Chromium Removal in the Presence of NOM during Fe(II) Reductive Precipitation for Drinking Water Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Examined Waters
2.2. Experimental Set-up and Procedures
2.2.1. General Jar Test Procedure for Reductive Precipitation of Chromium
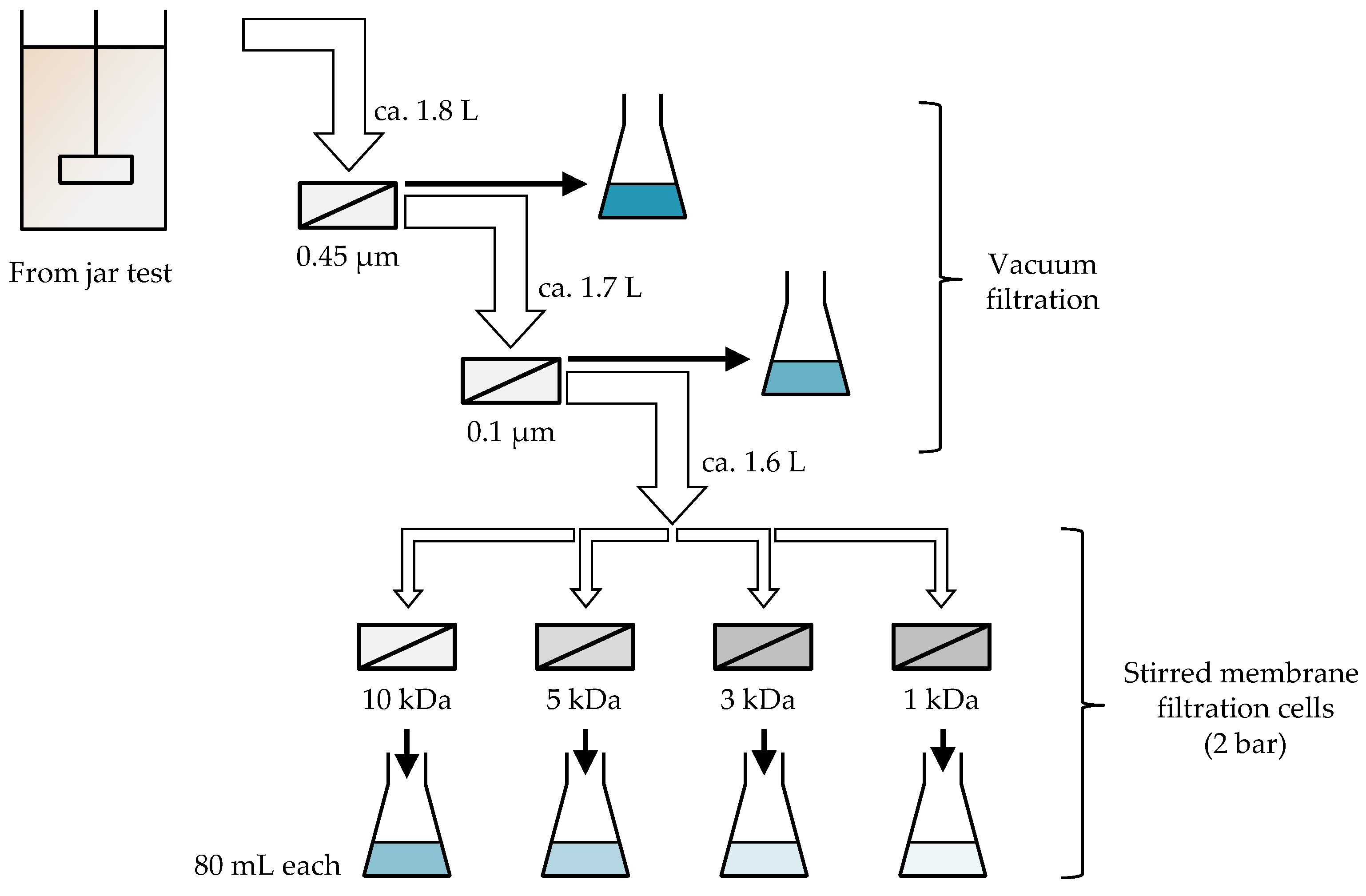
2.2.2. NOM Fractionation Tests
2.2.3. Jar Tests with Pre-Formed FeOH3 Sludge
2.3. Analytical Methods
3. Results and Discussion
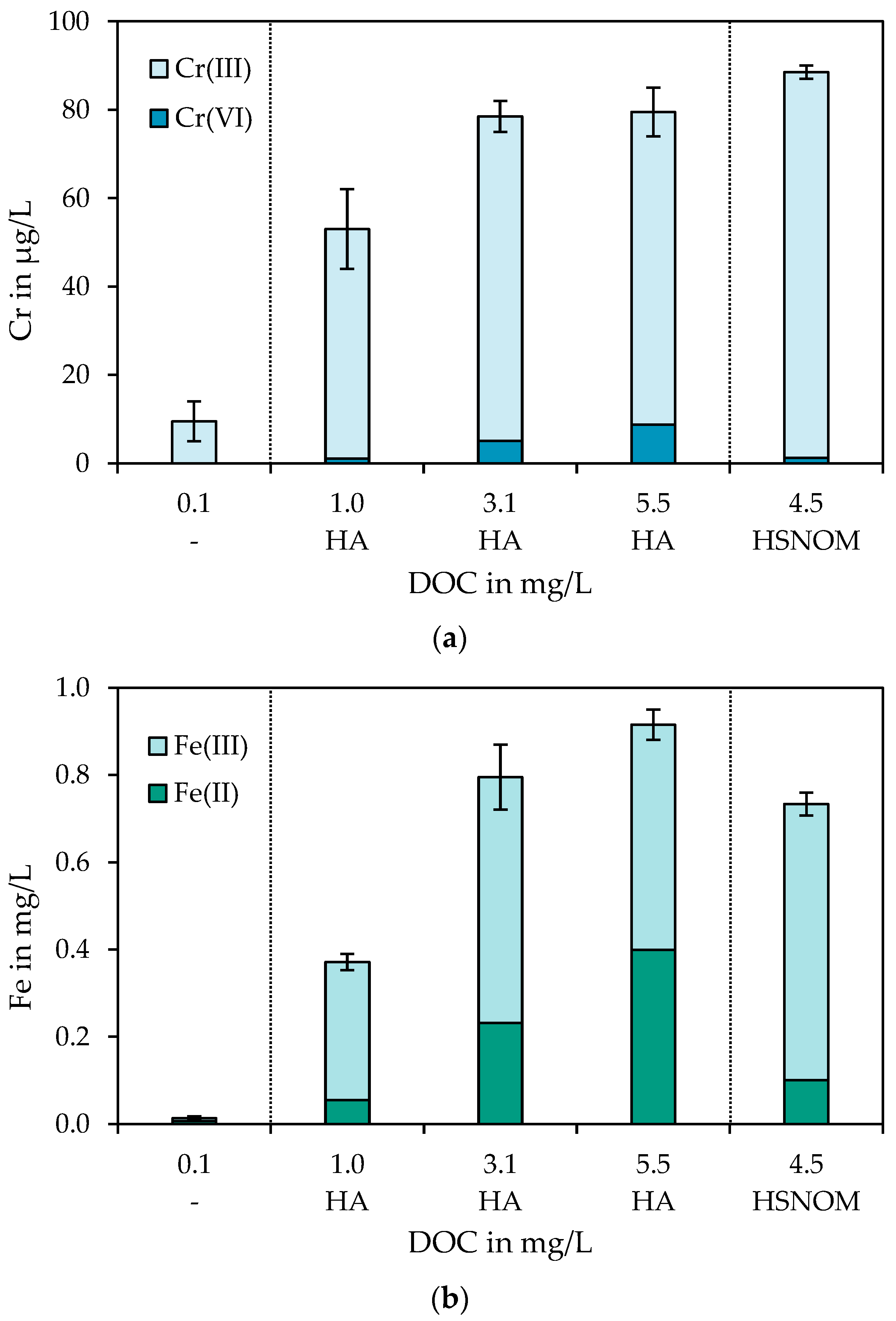
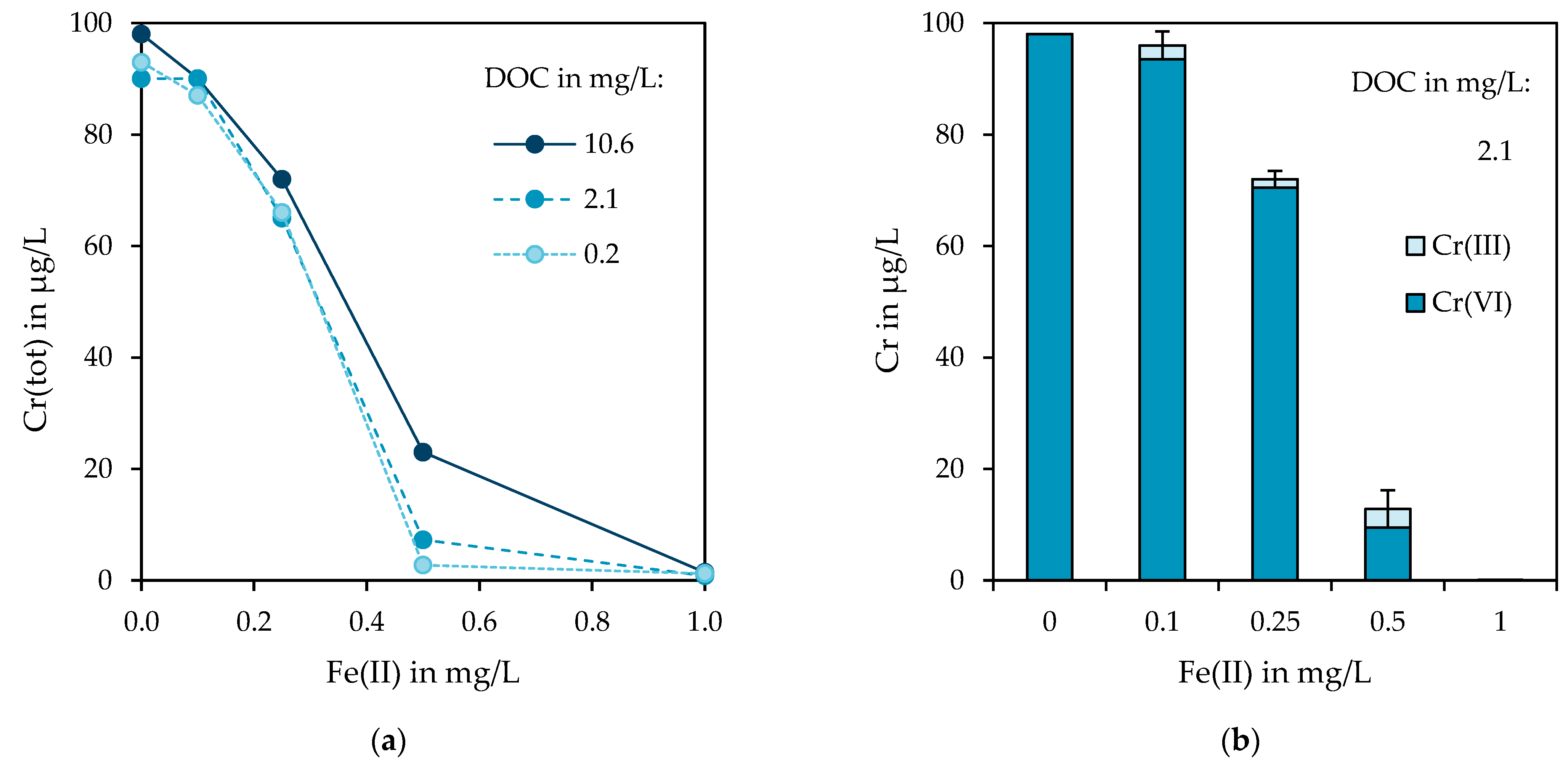
3.1. Impairment of Chromium Removal by NOM
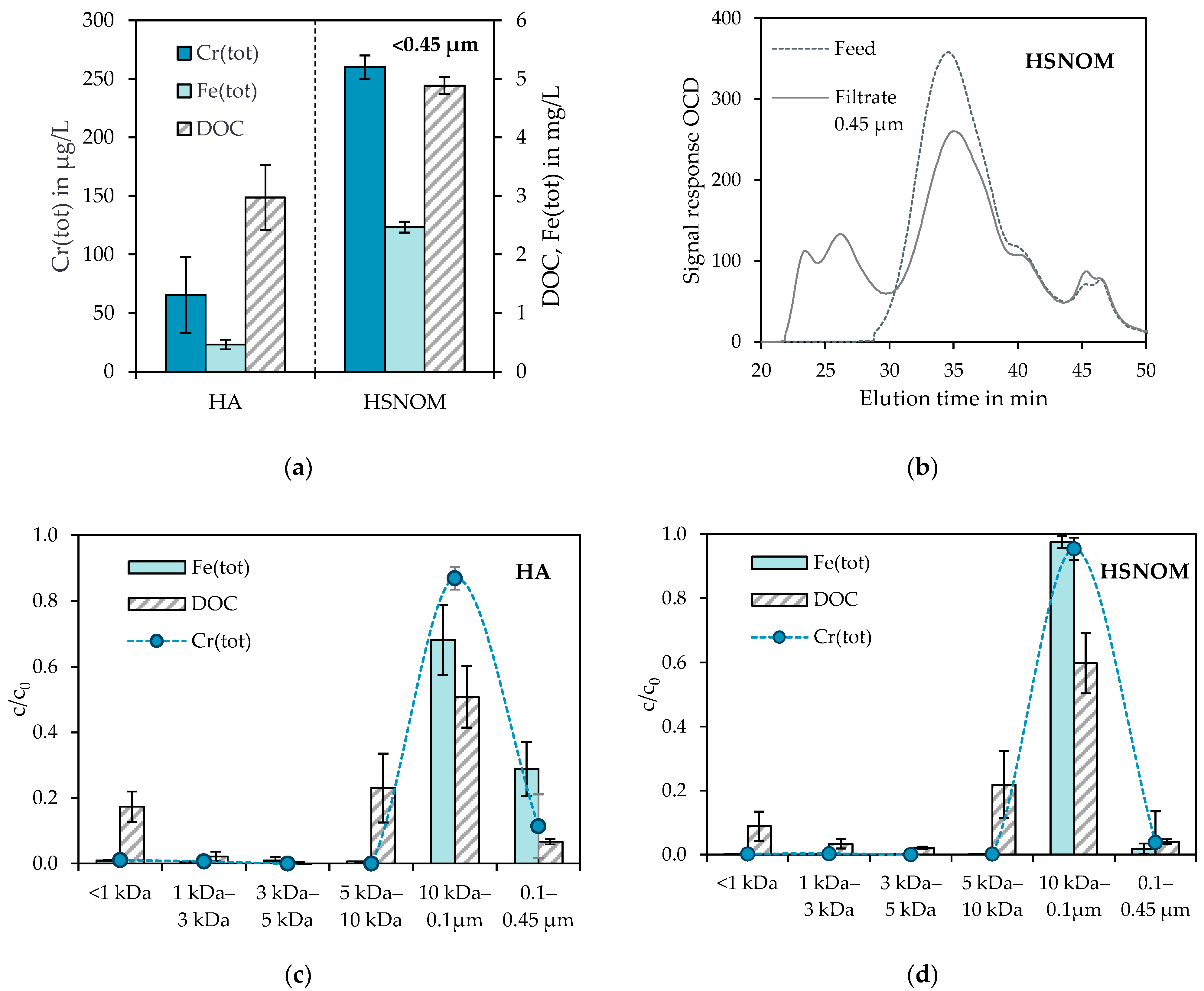
3.2. Fate of Residual Chromium, Iron and Organic Matter
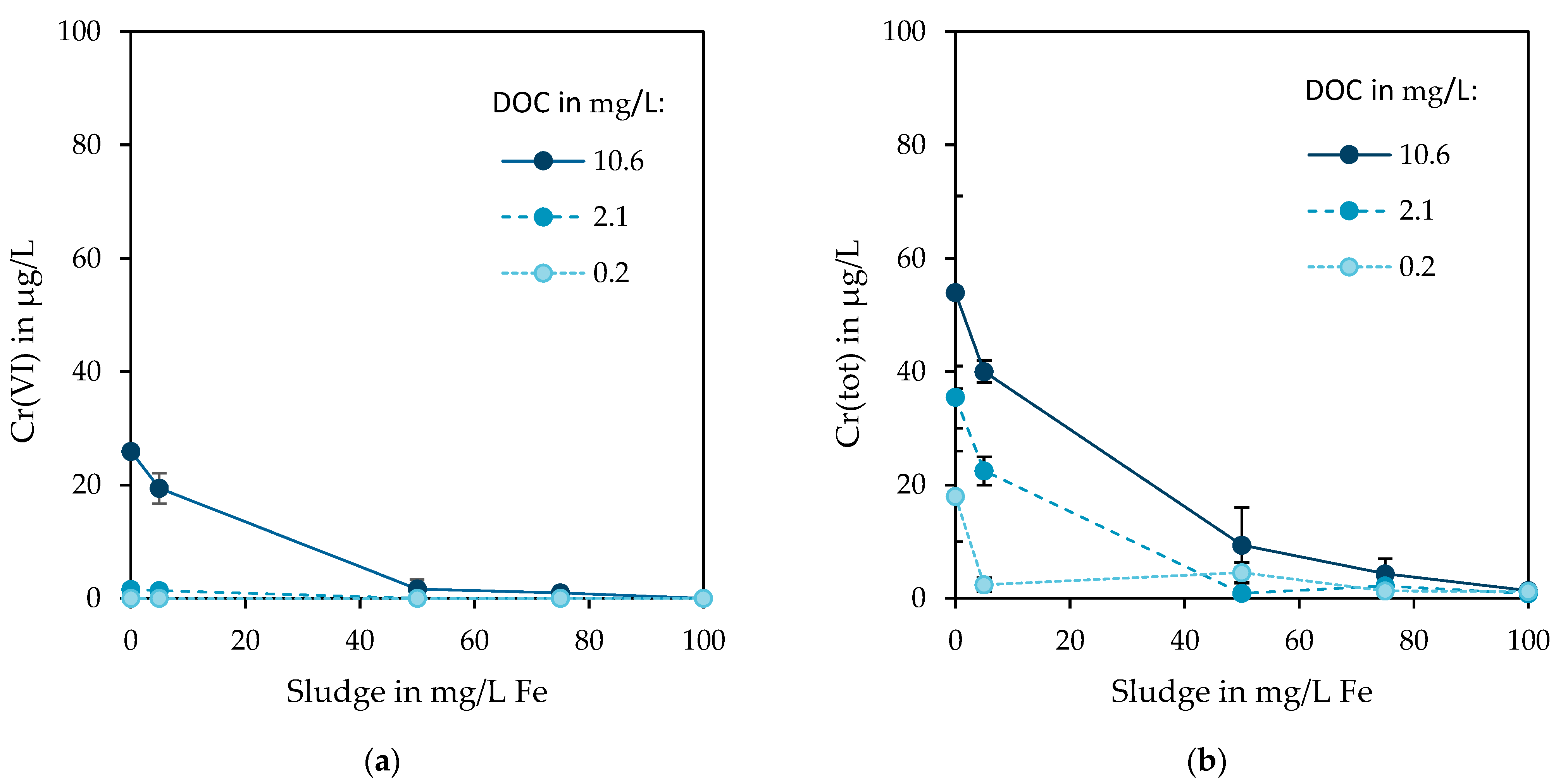
3.3. Removal of Residual Chromium in Presence of Precipitated Fe(OH)3
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kazakis, N.; Kantiranis, N.; Voudouris, K.S.; Mitrakas, M.; Kaprara, E.; Pavlou, A. Geogenic Cr oxidation on the surface of mafic minerals and the hydrogeological conditions influencing hexavalent chromium concentrations in groundwater. Sci. Total Environ. 2015, 514, 224–238. [Google Scholar] [CrossRef] [PubMed]
- Tumolo, M.; Ancona, V.; de Paola, D.; Losacco, D.; Campanale, C.; Massarelli, C.; Uricchio, V.F. Chromium Pollution in European Water, Sources, Health Risk, and Remediation Strategies: An Overview. Int. J. Environ. Res. Public Health 2020, 17, 5438. [Google Scholar] [CrossRef] [PubMed]
- Kaprara, E.; Kazakis, N.; Simeonidis, K.; Coles, S.; Zouboulis, A.I.; Samaras, P.; Mitrakas, M. Occurrence of Cr(VI) in drinking water of Greece and relation to the geological background. Hexavalent Chromium Geoenviron. Occur. Remediat. 2015, 281, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Rai, D.; Eary, L.E.; Zachara, J.M. Environmental chemistry of chromium. Sci. Total Environ. 1989, 86, 15–23. [Google Scholar] [CrossRef]
- McLean, J.E.; McNeill, L.S.; Edwards, M.; Parks, J.L. Hexavalent Chromium Review: Part 1—Health Effects, Regulations, and Analysis. J. Am. Water Work. Assoc. 2012, 104, 348–357. [Google Scholar] [CrossRef]
- Rai, D.; Sass, B.M.; Moore, D.A. Chromium(III) hydrolysis constants and solubility of chromium(III) hydroxide. Inorg. Chem. 1987, 26, 345–349. [Google Scholar] [CrossRef]
- Sun, H.; Brocato, J.; Costa, M. Oral Chromium Exposure and Toxicity. Curr. Environ. Health Rep. 2015, 2, 295–303. [Google Scholar] [CrossRef]
- Saha, R.; Nandi, R.; Saha, B. Sources and Toxicity of Hexavalent Chromium. J. Coord. Chem. 2011, 64, 1782–1806. [Google Scholar] [CrossRef]
- Zhitkovich, A. Chromium in Drinking Water: Sources, Metabolism, and Cancer Risks. Chem. Res. Toxicol. 2011, 24, 1617–1629. [Google Scholar] [CrossRef]
- Linos, A.; Petralias, A.; Christophi, C.A.; Christoforidou, E.; Kouroutou, P.; Stoltidis, M.; Veloudaki, A.; Tzala, E.; Makris, K.C.; Karagas, M.R. Oral ingestion of hexavalent chromium through drinking water and cancer mortality in an industrial area of Greece—An ecological study. Environ. Health 2011, 10, 50. [Google Scholar] [CrossRef]
- WHO. Chromium in Drinking-Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2020; Available online: https://www.who.int/publications/m/item/background-documents-for-development-of-who-guidelines-for-drinking-water-quality-and-guidelines-for-safe-recreational-water-environments (accessed on 16 April 2022).
- Council of the European Union. Directive (EU) 2020/2184 of the European Parliament and of the Council of 16 December 2020 on the Quality of Water Intended for Human Consumption: 2020/2184/EU; Council of the European Union: Brussels, Belgium, 2020. [Google Scholar]
- California Water Boards. Chromium-6 Drinking Water MCL—Announcements. Available online: https://www.waterboards.ca.gov/drinking_water/certlic/drinkingwater/Chromium6.html (accessed on 12 July 2022).
- Das Eidgenössische Departement des Innern (EDI). Verordnung des Eidgenössischen Departments des Innern (EDI) über Trinkwasser sowie Wasser in Öffentlich Zugänglichen Bädern und Duschanlagen: TBDV, 2017; Das Eidgenössische Departement des Innern (EDI): Bern, Switzerland, 2017. [Google Scholar]
- Sharma, S.K.; Petrusevski, B.; Amy, G. Chromium removal from water: A review. J. Water Supply: Res. Technol. Aqua 2008, 57, 541–553. [Google Scholar] [CrossRef]
- Brandhuber, P.; Frey, M.; McGuire, M.J.; Chao, P.; Seidel, C.; Amy, G.; Yoon, J.; McNeill, L.S.; Banerjee, K. Low-Level Hexavalent chromium Treatment Options: Bench-Scale Evaluation; Awwa Research Foundation: Denver, CO, USA, 2004; ISBN 978-1843399247. [Google Scholar]
- McGuire, M.J.; Blute, N.K.; Seidel, C.; Qin, G.; Fong, L. Pilot-scale studies of Hexavalent Chromium Removal from drinking water. J. Am. Water Work. Assoc. 2006, 98, 134–143. [Google Scholar] [CrossRef]
- Sass, B.M.; Rai, D. Solubility of amorphous chromium(III)-iron(III) hydroxide solid solutions. Inorg. Chem. 1987, 26, 2228–2232. [Google Scholar] [CrossRef]
- Mitrakas, M.G.; Pantazatou, A.S.; Tzimou-Tsitouridou, R.; Sikalidis, C.A. Influence of pH and temperature on Cr(VI) removal from a natural water using Fe(II): A pilot and full scale case study. Desalination Water Treat. 2011, 33, 77–85. [Google Scholar] [CrossRef]
- Qin, G.; McGuire, M.J.; Blute, N.K.; Seidel, C.; Fong, L. Hexavalent Chromium Removal by Reduction with Ferrous Sulfate, Coagulation, and Filtration: A Pilot-Scale Study. Environ. Sci. Technol. 2005, 39, 6321–6327. [Google Scholar] [CrossRef]
- Hering, J.G.; Lee, G. Removal of chromium(VI) from drinking water by redox-assisted coagulation with iron(II). J. Water Supply Res. Technol. Aqua 2003, 52, 319–332. [Google Scholar]
- Gröhlich, A.; Langer, M.; Mitrakas, M.; Zouboulis, A.; Katsoyiannis, I.; Ernst, M. Effect of Organic Matter on Cr(VI) Removal from Groundwaters by Fe(II) Reductive Precipitation for Groundwater Treatment. Water 2017, 9, 389. [Google Scholar] [CrossRef]
- Aoki, T.; Munemori, M. Recovery of chromium(VI) from wastewaters with iron(III) hydroxide—I: Adsorption mechanism of chromium(VI) on iron(III) hydroxide. Groundw. Arsen. Genes. Sustain. Remediat. 1982, 16, 793–796. [Google Scholar] [CrossRef]
- Fendorf, S.E.; Li, G. Kinetics of Chromate Reduction by Ferrous Iron. Environ. Sci. Technol. 1996, 30, 1614–1617. [Google Scholar] [CrossRef]
- Kamjunke, N.; Hertkorn, N.; Harir, M.; Schmitt-Kopplin, P.; Griebler, C.; Brauns, M.; von Tümpling, W.; Weitere, M.; Herzsprung, P. Molecular change of dissolved organic matter and patterns of bacterial activity in a stream along a land-use gradient. Water Res. 2019, 164, 114919. [Google Scholar] [CrossRef]
- Thurman, E.M. Organic Geochemistry of Natural Waters; Springer Science and Business Media: Dordrecht, The Netherlands, 1985. [Google Scholar]
- Frimmel, F.H. Characterization of natural organic matter as major constituents in aquatic systems. J. Contam. Hydrol. 1998, 35, 201–216. [Google Scholar] [CrossRef]
- Aiken, G.R.; Hsu-Kim, H.; Ryan, J.N. Influence of Dissolved Organic Matter on the Environmental Fate of Metals, Nanoparticles, and Colloids. Environ. Sci. Technol. 2011, 45, 3196–3201. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.; Troyer, L.D.; Liao, P.; Catalano, J.G.; Li, W.; Giammar, D.E. Effect of Humic Acid on the Removal of Chromium(VI) and the Production of Solids in Iron Electrocoagulation. Environ. Sci. Technol. 2017, 51, 6308–6318. [Google Scholar] [CrossRef] [PubMed]
- Kerndorff, H.; Schnitzer, M. Sorption of metals on humic acid. Geochim. Cosmochim. Acta 1980, 44, 1701–1708. [Google Scholar] [CrossRef]
- Lipczynska-Kochany, E. Effect of climate change on humic substances and associated impacts on the quality of surface water and groundwater: A review. Sci. Total Environ. 2018, 640–641, 1548–1565. [Google Scholar] [CrossRef] [PubMed]
- McDonough, L.K.; Santos, I.R.; Andersen, M.S.; O’Carroll, D.M.; Rutlidge, H.; Meredith, K.; Oudone, P.; Bridgeman, J.; Gooddy, D.C.; Sorensen, J.P.R.; et al. Changes in global groundwater organic carbon driven by climate change and urbanization. Nat. Commun. 2020, 11, 1279. [Google Scholar] [CrossRef]
- Agrawal, S.G.; Fimmen, R.L.; Chin, Y.-P. Reduction of Cr(VI) to Cr(III) by Fe(II) in the presence of fulvic acids and in lacustrine pore water. Chem. Geol. 2009, 262, 328–335. [Google Scholar] [CrossRef]
- Hori, M.; Shozugawa, K.; Matsuo, M. Reduction process of Cr(VI) by Fe(II) and humic acid analyzed using high time resolution XAFS analysis. J. Hazard. Mater. 2015, 285, 140–147. [Google Scholar] [CrossRef]
- Buerge, I.J.; Hug, S.J. Influence of Organic Ligands on Chromium(VI) Reduction by Iron(II). Environ. Sci. Technol. 1998, 32, 2092–2099. [Google Scholar] [CrossRef]
- Mak, M.S.; Lo, I.M. Influences of redox transformation, metal complexation and aggregation of fulvic acid and humic acid on Cr(VI) and As(V) removal by zero-valent iron. Chemosphere 2011, 84, 234–240. [Google Scholar] [CrossRef]
- Wittbrodt, P.R.; Palmer, C.D. Effect of temperature, ionic strength, background electrolytes, and Fe(III) on the reduction of hexavalent chromium by soil humic substances. Environ. Sci. Technol. 1996, 30, 2470–2477. [Google Scholar] [CrossRef]
- Daugherty, E.E.; Gilbert, B.; Nico, P.S.; Borch, T. Complexation and Redox Buffering of Iron(II) by Dissolved Organic Matter. Environ. Sci. Technol. 2017, 51, 11096–11104. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Tsang, D.C.W.; Lo, I.M.C. Chromium(VI) Reduction Kinetics by Zero-Valent Iron in Moderately Hard Water with Humic Acid: Iron Dissolution and Humic Acid Adsorption. Environ. Sci. Technol. 2008, 42, 2092–2098. [Google Scholar] [CrossRef]
- Stylianou, S.; Simeonidis, K.; Mitrakas, M.; Zouboulis, A.; Ernst, M.; Katsoyiannis, I.A. Reductive precipitation and removal of Cr(VI) from groundwaters by pipe flocculation-microfiltration. Environ. Sci. Pollut. Res. 2017, 25, 12256–12262. [Google Scholar] [CrossRef] [PubMed]
- Benecke, J. Gypsum Scaling during Reverse Osmosis Desalination—Characterization and Effects of Natural Organic Matter. Ph.D. Dissertation, Technische Universität Hamburg, Hamburg, Germany, 2018. [Google Scholar]
- Frimmel, F.H.; Abbt-Braun, G.; Heumann, K.G.; Hock, B.; Lüdemann, H.-D.; Spiteller, M. (Eds.) Refractory Organic Substances in the Environment; Wiley-VCH: Weinheim, Germany, 2002. [Google Scholar]
- Amy, G.; Chen, H.; Drizo, A.; von Gunten, U.; Brandhuber, P.; Hund, R.; Chowdhury, Z.; Kommineni, S.; Sinha, S.; Jekel, M.; et al. Adsorbent Treatment Technologies for Arsenic Removal; Awwa Research Foundation: Denver, CO, USA, 2005. [Google Scholar]
- DVGW. Arbeitsblatt W 218, Flockung in der Wasseraufbereitung—Flockungstestverfahren: DVGW W 218; DVGW Deutscher Verein des Gas- und Wasserfaches e.V.: Bonn, Germany, 1998. [Google Scholar]
- Sacher, F.; Thoma, A. Vorkommen von Chromat in Roh- und Trinkwässern in Deutschland. DVGW Energ. Wasser-Prax. 2013, 64, 52–56. [Google Scholar]
- Sharp, E.L.; Jarvis, P.; Parsons, S.A.; Jefferson, B. Impact of fractional character on the coagulation of NOM. Colloids Surf. A Physicochem. Eng. Asp. 2006, 286, 104–111. [Google Scholar] [CrossRef]
- Huber, S.A.; Balz, A.; Abert, M.; Pronk, W. Characterisation of aquatic humic and non-humic matter with size-exclusion chromatography—organic carbon detection—organic nitrogen detection (LC-OCD-OND). Groundw. Arsen. Genes. Sustain. Remediat. 2011, 45, 879–885. [Google Scholar] [CrossRef]
- Huang, B.; Qi, C.; Yang, Z.; Guo, Q.; Chen, W.; Zeng, G.; Lei, C. Pd/Fe3O4 nanocatalysts for highly effective and simultaneous removal of humic acids and Cr(VI) by electro-Fenton with H2O2 in situ electro-generated on the catalyst surface. J. Catal. 2017, 352, 337–350. [Google Scholar] [CrossRef]
- Rahmani, A.; Hossieni, E.; Poormohammadi, A. Removal of Chromium (VI) from Aqueous Solution Using Electro-Fenton Process. Environ. Processes 2015, 2, 419–428. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Langer, M.; Jamal, M.U.; Conklin, A.; Ernst, M. Chromium Removal in the Presence of NOM during Fe(II) Reductive Precipitation for Drinking Water Treatment. Water 2022, 14, 2903. https://doi.org/10.3390/w14182903
Langer M, Jamal MU, Conklin A, Ernst M. Chromium Removal in the Presence of NOM during Fe(II) Reductive Precipitation for Drinking Water Treatment. Water. 2022; 14(18):2903. https://doi.org/10.3390/w14182903
Chicago/Turabian StyleLanger, Margarethe, Mohammad Umair Jamal, Amanda Conklin, and Mathias Ernst. 2022. "Chromium Removal in the Presence of NOM during Fe(II) Reductive Precipitation for Drinking Water Treatment" Water 14, no. 18: 2903. https://doi.org/10.3390/w14182903
APA StyleLanger, M., Jamal, M. U., Conklin, A., & Ernst, M. (2022). Chromium Removal in the Presence of NOM during Fe(II) Reductive Precipitation for Drinking Water Treatment. Water, 14(18), 2903. https://doi.org/10.3390/w14182903







