A Multidisciplinary Approach to Posidonia oceanica Detritus Management (Port of Sperlonga, Italy): A Story of Turning a Problem into a Resource
Abstract
:1. Introduction
- Extraction of active ingredients for phytocosmetic, medicinal and pharmaceutical applications as described in the literature [23].
- Production of engineering materials (e.g., insulating products for the construction industry, especially for roofs and floors, due to their thermal insulating and fireproof properties).
- Production of biocomposites as natural products obtained by combining different raw materials to obtain an environmentally friendly final product with excellent performance.
- Determination of chemical pollutants (according to M.D. 173/2016 [9]) to evaluate the compatibility of the raw material as a substrate for the extraction of molecules of cosmetic and pharmaceutical interest for the production of compost and for underground experiments and reuse in the adjacent marine areas.
- Determination of macronutrient contents (i.e., TOC, TN, TP) to determine potential for composting/soil amendment production and/or their reuse in situ.
- Ecotoxicological impacts on marine species exposed to leaf biomass elutriates to assess environmental impacts associated with in-situ reuse (e.g., seashore burial or offshore dispersal above 3MN).
- Determination of their structural and mechanical properties of leaves waste.
2. Materials and Methods
2.1. The Study Area
2.2. Sampled Detritus
2.3. Physical-Chemical Analyses
2.4. Ecotoxicological Tests
2.5. Ultrastructural and Mechanical Analyses
2.6. Quality Assurance and Quality Control
3. Results
3.1. Macronutrients
3.2. Pollutants
3.3. Ecotoxicity
3.4. Mechanical Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Renzi, M.; Perra, G.; Guerranti, C.; Mariottini, M.; Baroni, D.; Volterrani, M.; Graziosi, M.; Specchiulli, A.; Focardi, S. Assessment of environmental pollutants in ten southern Italy port sediments. Toxicol. Ind. Health 2009, 25, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Ausili, A.; Mecozzi, M.; Gabellini, M.; Ciuffa, G.; Mellara, F. Phisico-chemical characteristics and multivarate analysis of contaminated port sediments. Water Sci. Technol. 1998, 37, 131–139. [Google Scholar] [CrossRef]
- Fortune, J. The Grainsize and Heavy Metal Content of Sediment in Darwin Port; Report No. 14/2006D; Aquatic Health Unit, Environmental Protection Agency: Washington, DC, USA, 2006; p. 68. [Google Scholar]
- Bortone, G.; Arevalo, E.; Deibel, I.; Detzner, H.D.; de Propris, L.; Elskens, F.; Giordano, A.; Hakstege, P.; Hamer, K.; Harmsen, J.; et al. Sediment and dredged material treatment. J. Soils Sedim. 2004, 4, 225–232. [Google Scholar] [CrossRef]
- Jensen, A.; Mogensen, B. Effects, Ecology and Economy. Environmental Aspects of Dredging—Guide No. 6. In Proceedings of the International Association of Dredging Companies (IADC) and Central Dredging Association (CEDA). 2000, p. 119. Available online: https://www.dhigroup.com/presences/nala/usa/news/2000/12/31/environmentalaspectsofdredging (accessed on 31 August 2022).
- Birch, G.F.; Taylor, S.E. Assessment of possible sediment toxicity of contaminated sediments in Port Jackson, Sydney, Australia. Hydrobiologia 2002, 472, 19–27. [Google Scholar] [CrossRef]
- Newell, R.C.; Seiderer, L.J.; Hitchcock, D.R. The impact of dredging works in coastal waters: A review of the sensitivity to disturbance and subsequent recovery of biological resources on the seabed. Ocean Mar. Biol. Ann. Rev. 1998, 36, 127–178. [Google Scholar]
- Erftemeijer, P.L.A.; Lewis, R.R. Environmental impacts of dredging on seagrasses: A review. Mar. Poll. Bull. 2006, 52, 1553–1572. [Google Scholar] [CrossRef]
- M.D. 173/2016 (Ministerial Decree 173/2016), Decreto Ministeriale 173 del 15 Luglio del 2016. Regolamento Recante Modalità e Criteri Tecnici per L’autorizzazione All’immersione in Mare dei Materiali di Escavo di Fondali Marini. In 16G00184—GU Serie Generale; 2016; Volume 208. Available online: https://www.isprambiente.gov.it/it/archivio/notizie-e-novita-normative/notizie-ispra/2016/09/regolamento-recante-modalita-e-criteri-tecnici-per-l2019autorizzazione-all2019immersione-in-mare-dei-materiali-di-escavo-di-fondali-marini (accessed on 31 August 2022). (In Italian)
- Renzi, M.; Tozzi, A.; Baroni, D.; Focardi, S. Factors affecting the distribution of trace elements in port sediments. Chem. Ecol. 2010, 27, 235–250. [Google Scholar] [CrossRef]
- Buia, M.C.; Gambi, M.C.; Zupo, V. Structure and functioning of Mediterranean seagrass ecosystems: An overview. Biol. Mar. Mediterr. 2000, 7, 167–190. [Google Scholar]
- Fornes, A.; Basterretxea, G.; Orfila, A.; Jordi, A.; Alvarez, A.; Tintore, J. Mapping Posidonia oceanica from IKONOS. ISPRS J. Photogramm. Remote Sens. 2006, 60, 315–322. [Google Scholar] [CrossRef]
- Decreto Ministeriale. 22 Gennaio 2009 da Parte del Ministero Delle Politiche Agricole Alimentari e Forestali. In Zone di Tutela Biologica: Nuove Determinazioni; Ministero delle Politiche Agricole Alimentari e Forestali: Rome, Italy, 2009; Volume 37, Available online: https://www.ambientediritto.it/Legislazione/INQUINAMENTO/2009/delmin_26gen2009.htm (accessed on 31 August 2022). (In Italian)
- Pergent, G.; Romero, J.; Pergent-Martini, C.; Mateo, M.A.; Boudouresque, C.F. Primary production, stocks and fluxes in the Mediterranean seagrass Posidonia oceanica. Mar. Ecol. Prog. Ser. 1994, 106, 139–146. [Google Scholar] [CrossRef]
- Bigongiari, N.; Cipriani, L.E.; Pranzini, E.; Renzi, M.; Vitale, G. Assessing shelf aggregate environmental compatibility and suitability for beach nourishment: A case study for Tuscany (Italy). Mar. Poll. Bull. 2010, 93, 183–193. [Google Scholar] [CrossRef]
- Bettaieb, F.; Khiari, R.; Hassan, M.L.; Belgacem, M.N.; Bras, J.; Dufresne, A.; Mhenni, M.F. Preparation and characterization of new cellulose nanocrystals from marine biomass Posidonia oceanica. Ind. Crop. Prod. 2015, 72, 175–182. [Google Scholar] [CrossRef]
- Khiari, R.; Belgacem, M.N. Potential for using multiscale Posidonia oceanica waste: Current status and prospects in material science. Woodhead Publishing Series in Composites Science and Engineering; In Lignocellulosic Fibre and Biomass-Based Composite Materials Processing, Properties and Applications; Woodhead Publishing: Sawston, UK, 2017; Volume 21, pp. 447–471. Available online: https://www.researchgate.net/publication/317507321_Potential_for_using_multiscale_Posidonia_oceanica_waste (accessed on 31 August 2022).
- Tarchoun, A.F.; Trache, D.; Klapötke, T.M. Microcrystalline cellulose from Posidonia oceanica brown algae: Extraction and characterization. Int. J. Biol. Macromol. 2019, 138, 837–845. [Google Scholar] [CrossRef]
- Barbieri, S.; Sambito, M.; Neduzha, L.; Severino, A. Pollutant emissions in Ports: A Comprehensive review. Infrastrucutres 2021, 6, 114. [Google Scholar] [CrossRef]
- European Union. Available online: https://european-union.europa.eu/select-language?destination=/node/1 (accessed on 10 August 2022).
- Kammoun, M.; Ayeb, H.; Bettaieb, T.; Richel, A. Chemical characterisation and technical assessment of agri-food residues, marine matrices, and wild grasses in the South Mediterranean area: A considerable inflow for biorefineries. Waste Manag. 2020, 118, 247–257. [Google Scholar] [CrossRef]
- Legislative Decree No. 75 (Circular 8838/2019). Gestione degli accumuli di Posidonia oceanica spiaggiati. m_amte.RIN.REGISTRO UFFICIALE.USCITA.Prot.0008838.20-05-2019. (In Italian)
- Jang, J.H.; So, B.R.; Yeo, H.J.; Kang, H.J.; Kim, M.J.; Lee, J.J.; Jung, S.K.; Jung, Y.H. Preparation of cellulose microfibril (CMF) from Gelidium amansii and feasibility of CMF as a cosmetic ingredient. Carbohydr. Polym. 2021, 257, 117569. [Google Scholar] [CrossRef]
- Comune di Fondi. Misure di Conservazione del SIC IT6000014 Fondali tra Terracina e Lago Lungo. Available online: https://www.comunedifondi.it/public/modulistica/4773095855_bozza%20misure%20di%20conservazione%20SIC%20IT6000014-1.pdf (accessed on 10 August 2022). (In Italian).
- Ardizzone, G.; Belluscio, A.; Maiorano, L. Long-term change in the structure of a Posidonia oceanica landscape and its reference for a monitoring plan. Mar. Ecol. 2006, 27, 299–309. [Google Scholar] [CrossRef]
- Guidelines 192/2020 ISPRA. 2020. La Spiaggia Ecologica: Gestione Sostenibile della Banquette di Posidonia oceanica Sugli Arenili del Lazio. Linee Guida 192/2020. Available online: https://www.isprambiente.gov.it/it/pubblicazioni/manuali-e-linee-guida/la-spiaggia-ecologica-gestione-sostenibile-della-banquette-di-posidonia-oceanica-sugli-arenili-del-lazio (accessed on 31 August 2022). (In Italian)
- Magara, G.; Prearo, M.; Vercelli, C.; Barbero, R.; Micera, M.; Botto, A.; Caimi, C.; Caldaroni, B.; Bertea, C.M.; Mannino, G.; et al. Modulation of Antioxidant Defense in Farmed Rainbow Trout (Oncorhynchus mykiss) Fed with a Diet Supplemented by the Waste Derived from the Supercritical Fluid Extraction of Basil (Ocimum basilicum). Antioxidants 2022, 11, 415. [Google Scholar] [CrossRef]
- Mari, M.; Macagnano, A.; Mazziotti, C.; Renzi, M.; Spiga, O.; Santucci, A.; Fardelli, A. Exploitation of Overrun Macroalgae as Model to Lead the Circular Economy Transition and the Bioeconomy Growth Monografia. In IIA, Istituto Sull’inquinamento atmosferico, Edizioni CNR (Italy); Consiglio Nazionale delle Ricerche: Rome, Italy, 2021; p. 39. ISBN 978-88-622-4021-5. (In Italian) [Google Scholar]
- Pepi, M.; Lampariello, L.R.; Altieri, R.; Esposito, A.; Perra, G.; Renzi, M.; Lobianco, A.; Feola, A.; Gasperini, S.; Focardi, S.E. Tannic acid degradation by bacterial strains Serratia spp. and Pantoea sp. isolated from olive mill waste mixtures. Int. Biodeterior. Biodegrad. 2010, 64, 73–80. [Google Scholar] [CrossRef]
- Vella, F.M.; Laratta, B.; La Cara, F.; Morana, A. Recovery of bioactive molecules from chestnut (Castanea sativa Mill.) by-products through extraction by different solvents. Nat. Prod. Res. 2018, 32, 1022–1032. [Google Scholar] [CrossRef]
- Gokce, G.; Haznedaroglu, M.Z. Evaluation of antidiabetic, antioxidant and vasoprotective effects of Posidonia oceanica extract. J. Ethnopharmacol. 2008, 115, 122–130. [Google Scholar] [CrossRef]
- Egadi Cosmesi Project. Available online: http://www.ampisoleegadi.it/?idPlugin=22859&idx=1632#testo_html_23476 (accessed on 31 August 2022).
- PHEE Company. Available online: https://phee.gr (accessed on 31 August 2022).
- Caript Foundation; The Research Institute of Terrestrial Ecosystems of the National Research Council (CNR-Iret); the Zelari farm in Pieve a Nievole (PT). Research Project Posidonia oceanica and Sediments for the Production of Substrates for Nurseries. Available online: https://www.cnr.it/it/news/allegato/1677 (accessed on 31 August 2022).
- Renzi, M.; Giovani, A.; Focardi, S.E. Biofuel production from the Orbetello lagoon macrophytes: Efficiency of lipid extraction using Accelerate Solvent Extraction technique. J. Environ. Prot. 2013, 4, 1224–1229. [Google Scholar] [CrossRef] [Green Version]
- Bastianoni, S.; Coppola, F.; Tiezzi, E.; Colacevich, A.; Borghini, F.; Focardi, S. Biofuel potential production from the Orbetello lagoonmacroalgae: A comparison with sunflower feedstock. Biomass Bioenergy 2008, 32, 619–628. [Google Scholar] [CrossRef]
- Lafabrie, C.; Pergent-Martini, C.; Pergent, G. Metal contamination of Posidonia oceanica meadows along the Corsican coastline (Mediterranean). Environ. Pollut. 2008, 151, 262–268. [Google Scholar] [CrossRef]
- Basset, A.; Pinna, M.; Renzi, M.A. Do Mining Activities Significantly Affect Feeding Behavior of Freshwater Benthic Macroinvertebrates? A Case Study in South Sardinia (Italy). Mine Water Environ. 2007, 36, 239–247. [Google Scholar] [CrossRef]
- Vengosh, A. Salinization and Saline Environments, 9.09. In Treatise on Geochemistry; Sherwood Lollar, B., Ed.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 9, p. 612. ISBN 0-08-043751-6. [Google Scholar]
- Hanson, B.; Evans, D.W.; Colby, D.R. Assessment of elemental contamination in estuarine and coastal environments based on geochemical and statistical modelling of sediments. Mar. Environ. Res. 1993, 36, 237–266. [Google Scholar] [CrossRef]
- Arizzi-Novelli, A.; Argese, E.; Tagliapietra, D.; Bettiol, C.; Volpi Ghirardini, A. Toxicity of tributyltin and triphenyltin to early life stages of Paracentrotus lividus (Echinodermata: Echinoidea). Environ. Toxicol. Chem. 2002, 21, 859–864. [Google Scholar] [CrossRef]
- Gopalakrishnan, S.; Thilagam, H.; Vivek Raja, P. Comparison of heavy metal toxicity in life stages (spermiotoxicity, egg toxicity, embryotoxicity and larval toxicity) of Hydroides elegans. Chemosphere 2008, 71, 515–528. [Google Scholar] [CrossRef]
- Calevro, F.; Campani, S.; Ragghianti, M.; Bucci, S.; Mancino, G. Tests of toxicity and teratogenicity in biphasic vertebrates treated with heavy metals (Cr3+, A13+, Cd2+). Chemosphere 1998, 37, 3011–3017. [Google Scholar] [CrossRef]




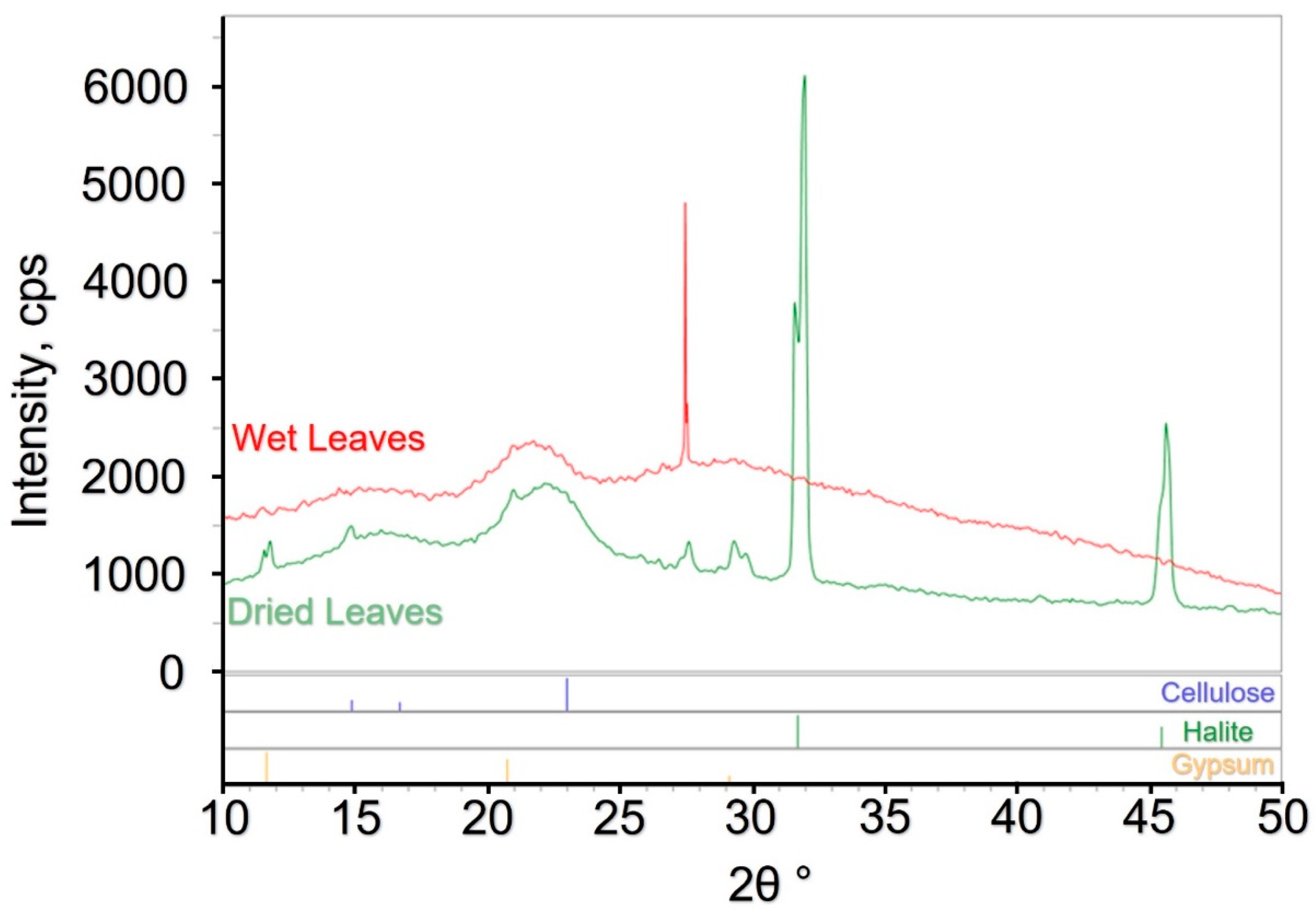
| Variable | Abbreviation | Method | Units | LOQ | |
|---|---|---|---|---|---|
| Nutrients | Total organic carbon | TOC | UNI EN 15936:2012 | % | 0.3 |
| Total nitrogen | TN | UNI EN 15407-2011 | % | 0.1 | |
| Total phosphorous | TP | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 2.5 | |
| Metals and metalloids | Aluminum | Al | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 1 |
| Arsenic | As | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 0.5 | |
| Cadmium | Cd | UNI EN 16174-2012 + UNI EN 16171-2016 | mg/kg | 0.03 | |
| Chromium | Cr | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 1 | |
| Chromium VI | Cr VI | CNR IRSA 16 Q 64 Vol 3 1986 | mg/kg | 0.1 | |
| Iron | Fe | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 1 | |
| Mercury | Hg | UNI EN 16174-2012 +UNI EN 16171-2016 | mg/kg | 0.03 | |
| Nickel | Ni | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 1 | |
| Lead | Pb | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 1 | |
| Copper | Cu | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 1 | |
| Vanadium | V | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 0.5 | |
| Zinc | Zn | EPA 3051 A 2007 + EPA 6010 D 2018 | mg/kg | 1 | |
| Hydrocarbons | Acenaphthylene | At | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 |
| Benzo(a)anthracene | BaA | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Fluoranthene | Fluo | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Naphthalene | Nap | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Anthracene | A | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Benzo(a)pyrene | BaP | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Benzo(b)fluoranthene | BbF | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Benzo(k)fluoranthene | BkF | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Benzo(g,h,i)perylene | BghiP | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Acenaphthene | Acn | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Fluorene | Flur | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Phenanthrene | Phe | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Pyrene | Py | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Dibenzo(a,h)anthracene | DahA | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Chrysene | Cr | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Indeno(1,2,3-c,d)pyrene | I(1,2,3)P | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 1 | |
| Hydrocarbons C > 12 | C > 12 | EPA 3550 C 2007 + EPA 8015 C 2007 | mg/kg | 5 | |
| POPs | Polychlorinated biphenyls | PCB | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 0.1 |
| Pesticides | - | EPA 3545 A 2007 + EPA 8270 E 2018 | μg/kg | 0.1 | |
| Organotin | BTs | ICRAM (2001)-App. 1 | μg/kg | 1 |
| Methods | Acute Toxicity (Type II) | Chronic Toxicity (Type II) | Chronic Toxicity (Type III) | |||
|---|---|---|---|---|---|---|
| Species | Paracentrotus lividus | Phaeodactylum tricornutum | Paracentrotus lividus | |||
| Method | EPA/600/R-95-136/s16 + ISPRA 11/17 | UNI EN ISO 10253:2017 | EPA/600/R-95-136/S15 + ISPRA 11/17 | |||
| Endpoint | Fertilization inhibition | Growth inhibition | Embryotoxicity | |||
| 20 min | 72 h | 72 h | ||||
| Unit | % | % | % | |||
| QA/QC | Average | SD/range | Average | SD/range | Average | SD/range |
| Negative control | 13.3 | 1.5 | 0.0 | 2.1 | 9.0 | 1.0 |
| Positive control | 40.06 | (35.98–44.59) | 23.15 | (20.63–25.99) | 33.08 | (28.93–37.83) |
| Fresh Leaves | Dried Leaves | |||
|---|---|---|---|---|
| w.w. | d.w. (Converted Values) | d.w. | ||
| TOC | % | 16.6 | 25.4 | 18.9 |
| TN | % | 0.22 | 0.34 | 0.59 |
| TP | mg/kg | 128 | 195 | 700 |
| Fresh Leaves | Dried Leaves | |||
|---|---|---|---|---|
| w.w. | d.w. (Converted Values) | d.w. | ||
| Al | mg/kg | 1362.4 | 2080 | 1810 |
| As | mg/kg | 4.86 | 7.42 | 9.02 |
| Cd | mg/kg | 0.14 | 0.22 | 0.19 |
| Cr | mg/kg | 7.2 | 11.0 | 9.1 |
| Fe | mg/kg | 2581 | 3940 | 5380 |
| Ni | mg/kg | 8.9 | 13.6 | 20.1 |
| Pb | mg/kg | 3.8 | 5.8 | 7.1 |
| Cu | mg/kg | 4.4 | 6.7 | 15.7 |
| V | mg/kg | 15.2 | 23.2 | 32.2 |
| Zn | mg/kg | 16.2 | 24.7 | 30.8 |
| C > 12 | mg/kg | 15.7 | 23.9 | 92.1 |
| BaA | μg/kg | 28.8 | 44 | <1 |
| Fluo | μg/kg | 34.1 | 52 | 11 |
| A | μg/kg | 3.1 | 4.8 | <1 |
| BaP | μg/kg | 34.7 | 53 | 5.4 |
| BbF | μg/kg | 41.9 | 64 | <1 |
| BkF | μg/kg | 17.0 | 26 | <1 |
| BghiP | μg/kg | 23.6 | 36 | 4 |
| Phe | μg/kg | 10.5 | 16 | 6.3 |
| Py | μg/kg | 28.2 | 43 | 8.6 |
| DahA | μg/kg | 4.2 | 6.4 | <1 |
| Cr | μg/kg | 32.8 | 50 | 4.8 |
| I(1,2,3)P | μg/kg | 22.3 | 34 | 2.9 |
| Total PAH | μg/kg | 281 | 429 | 43.3 |
| PCB 28 + PCB 31 | μg/kg | 0.10 | 0.19 | <0.1 |
| PCB 52 | μg/kg | 0.10 | 0.23 | <0.1 |
| PCB 101 | μg/kg | 0.40 | 0.57 | 0.29 |
| PCB 118 | μg/kg | 0.20 | 0.29 | <0.1 |
| PCB 138 | μg/kg | 0.60 | 0.84 | 0.48 |
| PCB 153 | μg/kg | 0.60 | 0.91 | 0.47 |
| PCB 180 | μg/kg | 0.30 | 0.48 | 0.24 |
| Total PCB | μg/kg | 2.30 | 3.50 | 1.48 |
| DDD o,p’ + p,p’ | μg/kg | 0.2 | 0.3 | <0.1 |
| DDE o,p’ + p,p’ | μg/kg | 0.3 | 0.4 | 0.5 |
| DDT o,p’ + p,p’ | μg/kg | 0.1 | 0.2 | 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renzi, M.; Guerranti, C.; Anselmi, S.; Provenza, F.; Leone, M.; La Rocca, G.; Cavallo, A. A Multidisciplinary Approach to Posidonia oceanica Detritus Management (Port of Sperlonga, Italy): A Story of Turning a Problem into a Resource. Water 2022, 14, 2856. https://doi.org/10.3390/w14182856
Renzi M, Guerranti C, Anselmi S, Provenza F, Leone M, La Rocca G, Cavallo A. A Multidisciplinary Approach to Posidonia oceanica Detritus Management (Port of Sperlonga, Italy): A Story of Turning a Problem into a Resource. Water. 2022; 14(18):2856. https://doi.org/10.3390/w14182856
Chicago/Turabian StyleRenzi, Monia, Cristiana Guerranti, Serena Anselmi, Francesca Provenza, Massimo Leone, Gabriele La Rocca, and Andrea Cavallo. 2022. "A Multidisciplinary Approach to Posidonia oceanica Detritus Management (Port of Sperlonga, Italy): A Story of Turning a Problem into a Resource" Water 14, no. 18: 2856. https://doi.org/10.3390/w14182856





