Using Springs as Sentinels of Climate Change in Nature Parks North and South of the Alps: A Critical Evaluation of Methodological Aspects and Recommendations for Long-Term Monitoring
Abstract
:1. Introduction
1.1. An Interdisciplinary View on the Spring System
- A surprising taxonomic diversity, with the discovery of numerous animal species that are rare or new to science: Individual study sites can differ greatly regarding species diversity and fauna spectrum. Only after examining a large number of ecologically different springs can the contribution of these habitats to the biodiversity of a geographic region (landscape) be estimated (compare [7]).
- A high stability in the organismic colonisation: During annual repeat investigations in three selected spring complexes (1994 to 2005), no changes in the relative abundance patterns of the representatives of essential animal groups (snails and mussels, crustaceans, mites and caddisflies) could be determined.
- A deficit in terms of available data on settlement history: While changes in the distribution of flora or various animal groups (birds, beetles and butterflies) that have traditionally been observed intensively in large-scale landscape structures allow conclusions to be drawn about ecological developments over the past 200 years, there is a complete lack of corresponding information for spring biocoenoses.
1.2. Spring Types
- −
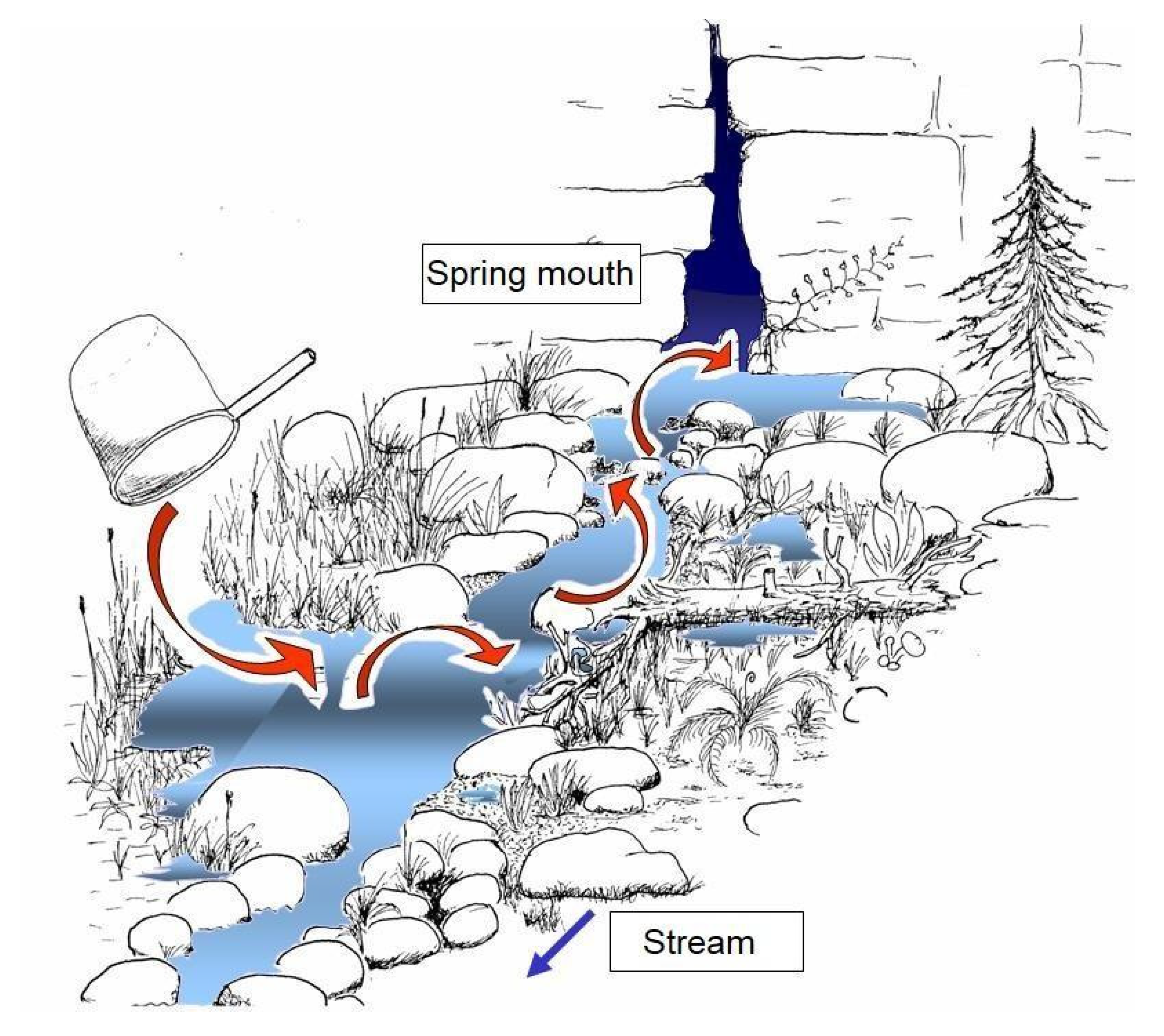
- Rheocrenes (Figure 1A): Water emerges at well-defined, often point-like spots and forms a clearly recognizable spring stream after a short distance. Compared to helocrenes (see below), they show a reduced species diversity due to a lower substratum differentiation but often show a pronounced biocenotic longitudinal gradient. In the immediate spring mouth area, populations of stygobiontic species can occur. In the following, often moss-rich actual source area, communities of crenobiontic and crenophilous species develop. Finally, in the subsequent transitional area spring/spring-fed rivulet also representatives of the typical brook fauna, e.g., filtering or larger predatory species, thrive. In springs of this type, it is expected that climate-related changes in living conditions are most likely to be reflected in modifications of the settlement structure. In addition to shifts in the boundaries in the longitudinal zonation and the immigration and emigration of stygobiontic/rhithrobiontic species and larger-scale zoogeographic migration movements could also be documented well here. A particular type of rheocrenes is characterised by the spring’s mouth shifting in elevation, dependent on the level of the groundwater reservoir. Such springs are of interest from a hydrographic point of view, since tendencies in groundwater capacities can be observed here. However, the instability of the emergence point severely limits possibilities for biological observations.
- −
- The structural diversity is particularly pronounced in helocrenes (Figure 1B). They occur in flat terrains or, with steeper gradients, as hillside spring mires and are characterised by diffuse, low discharge, with the formation of extensive shallow water areas. In springs of this type, biocoenoses of particular zoogeographical and biological interest have been discovered in recent decades, e.g., [11]. In addition to species that are strictly bound to springs (crenobionts), there are also several taxa with a preference for wet areas developing between water and land. Climate-related changes in the living conditions in such springs are difficult to document, because helocrenic springs can react to changes in temperature, discharge or chemistry by means of shifts within their complex structure.
- −
- Limnocrenes (Figure 1C) are characterised by water damming up at the spring’s mouth. The external factors affecting the groundwater discharge can be negligible if the dammed area is small. Still, they can become relevant with the increasing size of the pool (temperature stratification, formation of stagnant bank areas, etc.). Springs with relatively small pools at the spring mouth can be considered a special case of rheocrenes. As in rheocrenes, climate-related changes are likely to result in the emigration and immigration of species.
1.3. Weather and Climate Impacts on Spring Systems
- (1)
- Increases in water temperature and change in temperature amplitudes over the year;
- (2)
- Changed discharge behaviour in terms of seasonality and discharge volume;
- (3)
- Changed physical and chemical characteristics of the spring water;
- (4)
- Shifts in the distribution limits of species with narrow temperature tolerances to higher altitudes;
- (5)
- Spread of thermophilic species;
- (6)
- Immigration of species that were previously unable to gain a foothold due to other ecological limitations (e.g., duration of the growing season and food preferences);
- (7)
- Diffuse, randomly controlled immigration of species of different ecological valence due to the extended vegetation period.
2. Recommendations for the Long-Term Monitoring of Spring Habitats Targeting Climate-Change Effects
- −
- Site selection;
- −
- Mapping of the spring’s structure and the surrounding environment;
- −
- Recording of abiotic and biotic parameters.
2.1. Selection of the Springs to Be Studied
- −
- Hydrogeology: All critical hydrogeological units in the study area need to be considered. The porosity, location and dimension of the aquifer strongly influence the abiotic parameters of the spring water. Springs from deep aquifers show more stable thermal conditions than those from near-surface aquifers [27].
- −
- Geography: The essential characteristic landscape units are to be covered. In addition to the (hydro-)geological structuring, the diversity of surface forms and the substrate and vegetation conditions characterize the spring types. Due to their structure, specific landscape units must be disregarded if only spring types unsuitable for standardized monitoring (large limno- or helocrenes and linear rheocrenes) are available.
- −
- Elevation gradient: For a landscape unit, the selected locations must represent the entire vertical gradient over which springs are distributed in the area. The temperature gradient associated with the elevation gradient is crucial in climate observation.
- −
- Structural types: Flowing springs are best suited for standardized monitoring: Here, a low influence of external factors enables the most vital imprint by the aquifer. In addition, the reference points should be as diverse as possible (coarse substrate-high discharge and fine substrate-low discharge). In geographic areas without rheocrenes, helocrenes and limnocrenes can also be included in the monitoring by considering the following criteria.
- −
- Discharge: Springs with sporadically or seasonally interrupted discharge are unsuitable for long-term monitoring. Perennial discharge must be checked before selecting reference points (inspection after long dry phases or after long periods of frost when precipitation only occurs as ice or snow). Particularly in karst areas, the examination must be carried out several times, at least once several months after a drought, since discharge can be highly dynamic (see Figure 2) and can also be subject to latency with respect to meteorological conditions. As a rule, in low-altitude mountain ranges, early autumn and, in high-elevation mountains, winter are most suitable for verifying discharge stability. In questionable cases, a biological examination of a substrate sample is helpful: The presence of surface water microcrustaceans or species-rich colonization by mites or caddisflies indicate long-term bed stability; a poor fauna characterizes unstable sources, occasionally with the dominance of subterranean amphipods and turbellaria species, which retreat to groundwater in dry seasons. Observations show that, in contrast to running waters in Europe, no animal species are specially adapted to temporarily discharging springs. Resettlement occurs primarily through species that are not typical of the source (depending on the typology: river dwellers, species of temporary small bodies of water and groundwater species).
- −
- Temperature: Springs with stable temperatures over the year are particularly suitable for monitoring long-term changes in the groundwater system. However, locations with substantial daily and seasonal temperature variability also provide useful data, such as changes in amplitudes or shifts in maxima and minima.
- −
- Ecological integrity: Only near-natural (natural) springs are suitable for long-term, monitoring including biological aspects.
- −
- Limestone-Precipitating Springs (LPS): Due to their peculiar chemistry, “petrifying springs” (flora-fauna-habitat 7220) have relatively poor fauna [28]. Only a few animal species colonize the extreme habitat with lime coatings (Ger. “Sinter”). However, they deserve attention because climate change can affect carbonate precipitation. If LPS are characteristic of a landscape, at least one habitat of this type should be included in the long-term monitoring.
- −
- Logistics: For the practicability of a monitoring program, the accessibility of the locations is one of the selection criteria. To ensure regular data collection, the springs should be easily accessible, at least when there is no snow.
- −
- Data situation: If the suitability is otherwise equal, preference should be given to locations for which comparable results from earlier investigations are available. Such data from before the start of the monitoring extend the time series and increase the value of the study. If several similar sources are available, the site with the highest number of reference species should be preferred, or the availability of data from the catchment area (e.g., groundwater measuring points) should be decisive.
- −
- Comparability: Extreme habitats should not be selected as reference points, even if their characteristics possibly represent the particular trait of an area (e.g., rock-face springs, LPS, springs with a unique chemistry and thermal springs).
2.2. Overview on Monitoring Parameters
2.3. Mapping of the Spring Structure and of the Surrounding Environment
Mapping Method
2.4. Monitoring of Abiotic Parameters
2.4.1. Discharge
2.4.2. Water Temperature
2.4.3. pH
2.4.4. Conductivity
2.4.5. Dissolved Oxygen
2.4.6. Electrolytes and Main Algal Nutrients (N, P, S, Si)
2.5. Biological Monitoring
- Sampling of biological materials
- Sampling periodicity
2.5.1. Microbiology
Measuring Method
2.5.2. Botany
Phytobenthos Sampling
Field Work
Lab Work
2.5.3. Zoology
Reference Taxa
Zoobenthos Sampling
Field Work
Laboratory Work
2.5.4. Environmental DNA as a Tool to Monitor Spring Biota
2.6. Long-Term Data Storage and Evaluation
2.7. Framework Conditions for Monitoring at Springs
- −
- Data logger for continuous measurement of water temperature.
- −
- Measuring devices according to the defined standards for the in situ measurement of physical and chemical parameters.
- −
- Sample containers for physical, chemical and biological investigations.
- −
- Standard collection equipment for biological sampling.
- −
- Equipment for collection and preservation of eDNA samples (filter capsules, disposable syringes and isolation boxes with dry ice).
3. Findings from Previous Spring (Long-Term) Research in the Alpine Region: Relevance of Standardized Long-Term Spring Monitoring in the Context of Climate Change
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DIN 4049-3; Hydrologi: Begriffe zur Quantitativen Hydrologie. Beuth DIN: Berlin, Germany, 1994; Teil 3. 80p.
- Gerecke, R. Quellen: Leben auf der Schwelle zum Licht. In Wasser als Quelle des Lebens; Triebskorn, R., Wertheimer, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 169–187. [Google Scholar]
- Schröder, B.; Howein, H.; Gerecke, R. Quelltypen und Quellfauna. In Quellen im Nationalpark Berchtesgaden; Lebensgemeinschaften als Indikatoren des Klimawandels; Gerecke, R., Franz, H., Eds.; Nationalpark Berchtesgaden Forschungsbericht: Berchtesgaden, Germany, 2006; Volume 51, pp. 214–220. [Google Scholar]
- Gerecke, R.; Franz, H. (Eds.) Quellen im Nationalpark Berchtesgaden; Lebensgemeinschaften als Indikatoren des Klimawandels; Berchtesgadener Anzeiger: Berchtesgaden, Germany, 2006; Volume 51, p. 272. [Google Scholar]
- Taxböck, L.; Karger, D.N.; Kessler, M.; Spitale, D.; Cantonati, M. Diatom species richness in Swiss springs increases with habitat complexity and elevation. Water 2020, 12, 449. [Google Scholar] [CrossRef]
- Cantonati, M.; Stevens, L.E.; Segadelli, S.; Springer, A.E.; Goldscheider, N.; Celico, F.; Filippini, M.; Ogata, K.; Gargini, A. Ecohydrogeology: The interdisciplinary convergence needed to improve the study and stewardship of springs and other groundwater-dependent habitats, biota, and ecosystems. Ecol. Indic. 2020, 110, 105803. [Google Scholar] [CrossRef]
- Cantonati, M.; Segadelli, S.; Spitale, S.; Gabrieli, J.; Gerecke, R.; Angeli, N.; De Nardo, M.T.; Ogata, K.; Wehr, J.D. Geological and hydrochemical prerequisites of unexpectedly high biodiversity in spring ecosystems at the landscape level. Sci. Total Environ. 2020, 740, 140157. [Google Scholar] [CrossRef]
- Cantonati, M.; Bilous, O.; Spitale, D.; Angeli, N.; Segadelli, S.; Bernabè, D.; Lichtenwöhrer, K.; Gerecke, R.; Saber, A.A. Diatoms from the spring ecosystems selected for the long-term monitoring of climate-change effects in the Berchtesgaden National Park (Germany). Water 2022, 14, 381. [Google Scholar] [CrossRef]
- Steinmann, P. Praktikum der Süßwasserbiologie; Die Organismen des fließenden Wassers; Gebrüder Borntraeger (Sammlung naturwissenschaftlicher Praktika): Berlin, Germany, 1915; Teil I, Band VII; 184p. [Google Scholar]
- Thienemann, A. Hydrobiologische Untersuchungen an Quellen (I–IV). Arch. Hydrobiol. 1922, 14, 151–190. [Google Scholar]
- Gerecke, R.; Cantonati, M.; Spitale, D.; Stur, E.; Wiedenbrug, S. The challenges of long-term ecological research in springs in the northern and southern Alps: Indicator groups, habitat diversity, and medium term change. J. Limnol. 2011, 70, 168–187. [Google Scholar] [CrossRef]
- Stevens, L.E.; Schenk, E.R.; Springer, A.E. Springs ecosystem classification. Ecol. Appl. 2020, 31, 002218. [Google Scholar] [CrossRef]
- IPCC. Summary for Policymakers. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Masson-Delmotte, V., Zhai, P., Pirani, A., Connors, S.L., Péan, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M.I., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2021; pp. 3–32. [Google Scholar] [CrossRef]
- Kuhn, M.; Olefs, M. Elevation-dependent climate change in the European Alps. Oxf. Res. Encycl. Clim. Sci. 2020, 1–25. [Google Scholar] [CrossRef]
- Poschlod, B.; Ludwig, R. Internal variability and temperature scaling of future sub-daily rainfall return levels over Europe. Environ. Res. Lett. 2021, 16, 064097. [Google Scholar] [CrossRef]
- Poschlod, B.; Willkofer, F.; Ludwig, R. Impact of Climate Change on the Hydrological Regimes in Bavaria. Water 2020, 12, 1599. [Google Scholar] [CrossRef]
- Martin, P.; Gerecke, R.; Cantonati, M. Quellen. In Grundwassergeprägte Lebensräume; Eine Übersicht über Grundwasser, Quellen, das hyporheische Interstitial und Weitere Grundwassergeprägte Habitate. Limnologie Aktuell; Brendelberger, H., Martin, P., Brunke, M., Hahn, H.J., Eds.; Schweizerbart: Stuttgart, Germany, 2015; Volume 14, pp. 49–132. [Google Scholar]
- Hemmerle, H.; Bayer, P. Climate Change Yields Groundwater Warming in Bavaria, Germany. Front. Earth Sci. 2020, 8, 575894. [Google Scholar] [CrossRef]
- Jyväsjärvi, J.; Marttila, H.; Rossi, P.M.; Ala-Aho, P.; Olofsson, B.; Nisell, J.; Backman, B.; Ilmonen, J.; Virtanen, R.; Paasivirta, L.; et al. Climate-induced warming imposes a threat to north European spring ecosystems. Glob. Chang. Biol. Bioenergy 2015, 21, 4561–4569. [Google Scholar] [CrossRef] [PubMed]
- Kunstmann, H.; Strasser, U.; Warscher, M.; Laux, P.; Marke, T.; Lorenz, M. IPCC-AR5 Klimaszenarien (RCPs) zur Hydrologischen Klimaimpaktanalyse: Synthese Hochaufgelöster Regionalisierung, Multivariater Stochastischer Bias-Korrektur und Optimierter Hydrologischer Modell- und Prozessanalyse am Beispiel Nationalpark Berchtesgaden; BIAS II—Schlussbericht; Berchtesgadener Anzeiger: Berchtesgaden, Germany, 2019; 71p. [Google Scholar]
- Lichtenwöhrer, K.; Leonhardt, G.; Seifert, L.; Hotzy, R.; Schubert, E.; Gerecke, R.; Cantonati, M.; Blattner, L.; Lotz, A.; Poschlod, B. Erfassung von Klimawandelfolgen an Quellen in Bayern; Leitfaden für eine Langfristige Beobachtung von Quellen zur Erfassung von Klimawandelfolgen in Bayern; Nationalparkverwaltung Berchtesgaden, Ed.; Forschungsbericht: Berchtesgaden, Germany, 2022; Volume 57, 80p. [Google Scholar]
- Segadelli, S.; Grazzini, F.; Adorni, M.; De Nardo, M.T.; Fornasiero, A.; Chelli, A.; Cantonati, M. Predicting Extreme-Precipitation Effects on the Geomorphology of Small Mountain Catchments: Towards an Improved Understanding of the Consequences for Freshwater Biodiversity and Ecosystems. Water 2020, 12, 79. [Google Scholar] [CrossRef]
- Cantonati, M.; Gerecke, R.; Bertuzzi, E. Springs of the Alps—Sensitive ecosystems to environmental change: From biodiversity assessments to long-term studies. Hydrobiologia 2006, 562, 59–96. [Google Scholar] [CrossRef]
- Cantonati, M.; Ortler, K. Using spring biota of pristine mountain areas for long term monitoring—Hydrology. In Water Resources and Ecology in Headwaters, Proceedings of the Headwater’98 Conference, Merano/Meran, Italy, 20–23 April 1998; IAHS Publication: Wallingford, UK, 1998; Volume 248, pp. 379–385. [Google Scholar]
- Cantonati, M.; Bertuzzi, E.; Spitale, D. (Eds.) The spring habitat: Biota and sampling methods. In Museo Tridentino di Scienze Naturali 4; Museo Tridentino di Scienze Naturali: Trento, Italy, 2007; 350p. [Google Scholar]
- Van Rooij, P.; Pasmans, F.; Coen, Y.; Martel, A. Efficacy of chemical disinfectants for the containment of the salamander chytrid fungus Batrachochytrium salamandrivorans. PLoS ONE 2017, 12, e0186269. [Google Scholar] [CrossRef] [PubMed]
- Küry, D.; Lubini, V.; Stucki, P. Verletzlichkeit von Eintagsfliegen, Steinfliegen und Köcherfliegen alpiner Quellen gegenüber Klimaveränderungen. Jahrb. Des Ver. Zum Schutz Der Bergwelt 2018, 83, 199–218. [Google Scholar]
- Cantonati, M.; Segadelli, S.; Ogata, K.; Tran, H.; Sanders, D.; Gerecke, R.; Rott, E.; Filippini, M.; Gargini, A.; Celico, F. A global review on ambient Limestone-Precipitating Springs (LPS): Hydrogeological setting, ecology, and conservation. Sci. Total Environ. 2016, 568, 624–637. [Google Scholar] [CrossRef]
- LfU (Bayerisches Landesamt für Umwelt). Messeinrichtungen an Quellen; Nr. 2.1/10; Merkblatt: Augsburg, Germany, 2008; 33p. [Google Scholar]
- Hiscock, K.M.; Bense, V.F. Hydrogeology: Principles and Practice, 2nd ed.; Wiley-Blackwell: Chichester, UK, 2014; 519p. [Google Scholar]
- Gordon, N.D.; McMahon, T.A.; Finlayson, B.L.; Gippel, C.J.; Nathan, R.J. Stream Hydrology: An Introduction for Ecologists, 2nd ed.; Wiley-Blackwell: Chichester, UK, 2004; 448p. [Google Scholar]
- Hölting, B.; Coldewey, W.G. Hydrogeologie: Einführung in die Allgemeine und Angewandte Hydrogeologie; Spektrum: Heidelberg, Germany, 2009; 410p. [Google Scholar]
- Taylor, R.G.; Scanlon, B.; Döll, P.; Rodell, M.; Van Beek, R.; Wada, Y.; Longuevergne, L.; Leblanc, M.; Famiglietti, J.S.; Edmunds, M.; et al. Ground water and climate change. Nat. Clim. Chang. 2013, 3, 322–329. [Google Scholar] [CrossRef]
- Wilhelm, F. Physikalisch-Chemische Untersuchungen an Quellen in den Bayerischen Alpen und im Alpenvorland; Münchner Geographische Hefte; Michael Lassleben Kallmünz: Regensburg, Germany, 1956; Volume 10, 97p. [Google Scholar]
- Lahermo, P.; Valovirta, V.E.; Sarkioja, A. The geobotanical development of spring-fed mires in Finnish Lapland. Bull. Geol. Surv. Finl. 1977, 300, 1–44. [Google Scholar]
- Ebner, J.N.; Ritz, D.; Von Fumetti, S. Comparative proteomics of stenotopic caddisfly Crunoecia irrorata identifies acclimation strategies to warming. Mol. Ecol. 2019, 28, 4453–4469. [Google Scholar] [CrossRef]
- Brehm, J. Der pH-Wert von Buntsandsteinquellen und Bächen. II. Der Waldquell-pH-Wert nach der Höhenlage der Wasseraustritte und der Hangneigungsrichtung der Quelleneinzugsgebiete im Knüllgebirge. Beitr. Naturk. Osthessen 1985, 21, 53–66. [Google Scholar]
- Puhe, J.; Ulrich, B. Chemischer Zustand von Quellen im Kaufunger Wald. Arch. Hydrobiol. 1985, 102, 331–342. [Google Scholar] [CrossRef]
- Zollhofer, J.M. Regionale Quelltypologie für Jura und Mittelland der Schweiz. Crunoecia 1997, 5, 265–280. [Google Scholar]
- Brehm, J. Vierjährige hydrologische und chemische Untersuchungen an zwei benachbarten Buntsandstein-Waldquellen. Arch. Hydrobiol. 1978, 82, 49–65. [Google Scholar]
- Bakalowicz, M. Water geochemistry: Water Quality and Dynamics. In Groundwater Ecology; Gibert, J., Danielopol, D.L., Stanford, J., Eds.; Academic Press: London, UK, 1994; pp. 97–129. [Google Scholar]
- Hervant, F.; Mathieu, J.; Messana, G. Oxygen consumption and ventilation in declining oxygen tension and posthypoxic recovery in epigean and hypogean aquatic crustaceans. J. Crust. Biol. 1998, 18, 717–727. [Google Scholar] [CrossRef]
- Hervant, F.; Mathieu, J.; Culver, D.C. Comparative responses to severe hypoxia and subsequent recovery in closely related amphipod populations (Gammarus minus) from cave and surface habitats. Hydrobiologia 1999, 392, 197–204. [Google Scholar] [CrossRef]
- Maire, R.; Pomel, S. Karst Geomorphology and Environment. In Groundwater Ecology; Gibert, J., Danielopol, D.L., Stanford, J., Eds.; Academic Press: London, UK, 1994; pp. 130–157. [Google Scholar]
- Stanford, J.A.; Ward, J.V.; Ellis, B.K. Ecology of the Alluvial Aquifers of the Flathead River, Montana. In Groundwater Ecology; Gibert, J., Danielopol, D.L., Stanford, J., Eds.; Academic Press: London, UK, 1994; pp. 367–390. [Google Scholar]
- Pax, F. Die Tierwelt der mitteleuropäischen Schwefelquellen. Senckenbergiana 1948, 28, 139–152. [Google Scholar]
- Thies, H.; Nickus, U.; Tolotti, M.; Tessadri, R.; Krainer, K. Evidence of rock glacier melt impacts on water chemistry and diatoms in high mountain streams. Cold Reg. Sci. Technol. 2013, 96, 77–85. [Google Scholar] [CrossRef]
- Nadig, A. Hydrobiologische Untersuchungen in Quellen des Schweizerischen Nationalparks im Engadin (unter besonderer Berücksichtigung der Insektenfauna). Ergebn. wiss. Untersuch. schweiz. Nationalpark 1943, 1, 265–432. [Google Scholar]
- Karthe, D. Bedeutung des Klimawandels (insbesondere hydrometeorologischer Extremereignisse) für die Trinkwasserhygiene in Deutschland—Aktuelle Herausforderungen im Flussgebiets- und Hochwassermanagement—Prozesse, Methoden, Konzepte. Beiträge zum Tag der Hydrologie an der Universität Bonn. Forum Hydrol. Wasserbewirtsch. 2015, 35, 33–43. [Google Scholar]
- Cantonati, M. Springs—Groundwater-Borne Ecotones—A Typology, with an Overview on the Diversity of Photoautotrophs in Springs. In Encyclopedia of Inland Waters, 2nd ed.; Mehner, T., Tockner, K., Likens, G., Eds.; Elsevier Science: New York, NY, USA, 2022; Volume 3, pp. 488–509. [Google Scholar] [CrossRef]
- Cantonati, M.; Rott, E.; Pfister, P.; Bertuzzi, E. Benthic algae in springs of the Alps: Biodiversity and sampling methods. In Monografie del Museo Tridentino di Scienze Naturali 4; The Spring Habitat: Biota and Sampling Methods; Cantonati, M., Bertuzzi, E., Spitale, D., Eds.; Museo Tridentino di Scienze Naturali: Trento, Italy, 2007; pp. 77–112. [Google Scholar]
- Cantonati, M. Diatom communities of springs in the Southern Alps. Diatom Res. 1998, 13, 201–220. [Google Scholar] [CrossRef]
- Alles, E.; Nörpel-Schempp, M.; Lange-Bertalot, H. Zur Systematik und Ökologie charakteristischer Eunotia-Arten (Bacilariophyceae) in elektrolytarmen Bachoberläufen. Nova Hedwig. 1991, 53, 171–213. [Google Scholar]
- Lange-Bertalot, H.; Metzeltin, D. Indicators of oligotrophy. 800 taxa representative of three ecologically distinct lake types, carbonate buffered-oligodystrophic-weakly buffered soft waters. Iconogr. Diatomol. 1996, 2, 1–390. [Google Scholar]
- Andrén, C.; Jarlman, A. Benthic diatoms as indicators of acidity in streams. Fundam. Appl. Limnol. 2008, 173, 237–253. [Google Scholar] [CrossRef]
- Cantonati, M.; Lange-Bertalot, H. Diatom monitors of close-to-pristine, very-low alkalinity habitats: Three new Eunotia species from springs in Nature Parks of the south-eastern Alps. J. Limnol. 2011, 70, 209–221. [Google Scholar] [CrossRef]
- Cantonati, M.; Lange-Bertalot, H.; Scalfi, A.; Angeli, N. Cymbella tridentina sp. nov. (Bacillariophyta), A crenophilous diatom from carbonate springs of the Alps. J. N. Am. Benthol. 2010, 29, 775–788. [Google Scholar] [CrossRef]
- Cantonati, M.; Angeli, N.; Bertuzzi, E.; Spitale, D.; Lange-Bertalot, H. Diatoms in springs of the Alps: Spring types, environmental determinants, and substratum. Freshw. Sci. 2012, 31, 499–524. [Google Scholar] [CrossRef]
- Pfister, P.; Pipp, E. Leitfaden zur Erhebung der Biologischen Qualitätselemente; Teil 3A—Phytobenthos; Bundesministerium für Land- und Forstwirtschaft: Wien, Austria, 2013; 92p. [Google Scholar]
- Sambugar, B.; Martinez-Ansemil, E.; Giani, N. Oligochaeta from springs in southern Europe. Boll. Mus. Civ. Stor. Nat. Verona Bot. Zool. 2005, 29, 93–106. [Google Scholar]
- Blattner, L.; Ebner, J.N.; Zopfi, J.; von Fumetti, S. Targeted non-invasive bioindicator species detection in eDNA water samples to assess and monitor the integrity of vulnerable alpine freshwater environments. Ecol. Indic. 2021, 129, 107916. Available online: https://www.sciencedirect.com/science/article/pii/S1470160X21005811 (accessed on 9 September 2022). [CrossRef]
- Blattner, L.; Lucek, K.; Beck, N.; Berner, D.; von Fumetti, S. Intra-Alpine Islands: Population genomic inference reveals high degree of isolation between freshwater spring habitats. Divers. Distrib. 2022, 28, 291–305. [Google Scholar] [CrossRef]
- Gerecke, R.; Martin, P.; Gledhill, T. Water mites (Acari: Parasitengona: Hydrachnidia) as inhabitants of groundwater-influenced habitats—Considerations following an update of Limnofauna Europaea. Limnologica 2018, 69, 81–93. [Google Scholar] [CrossRef]
- Gerecke, R.; Martin, P.; Waltzberg, C.; Walisch, T. Verbreitungsatlas der Wassermilben des Großherzogtums Luxemburg. Ferrantia 2022, 85, 1–175. [Google Scholar]
- Hershkovitz, Y.; Dahm, V.; Lorenz, A.W.; Hering, D. A multi-trait approach for the identification and protection of European freshwater species that are potentially vulnerable to the impacts of climate change. Ecol. Indic. 2015, 50, 150–160. [Google Scholar] [CrossRef]
- Zollhöfer, J.M. Quellen—Die Unbekannten BIOTOPE im Schweizer Jura und Mittelland: Erfassen, Bewerten, Schützen. Bristol-Schriftenreihe Bristol-Stiftung, Ruth und Herbert Uhl-Forschungsstelle für Natur- und Umweltschutz; Flück-Wirth: Teufen, Switzerland, 1997; Band 6; 153p. [Google Scholar]
- Zollhöfer, J.M.; Brunke, M.A.; Gonser, T. A typology of springs in Switzerland by integrating habitat variables and fauna. Arch. Hydrobiol. 2000, 121, 349–376. [Google Scholar]
- Von Fumetti, S.; Nagel, P.; Baltes, B. Where a springhead becomes a springbrook—A regional zonation of springs. Fundam. Appl. Limnol. 2007, 169, 37–48. [Google Scholar] [CrossRef]
- Franz, H.; Gerecke, R.; Stur, E.; Wiedenbrug, S. Vorschläge für die langfristige Umweltbeobachtung, Ausblick. In Quellen im Nationalpark Berchtesgaden; Lebensgemeinschaften als Indikatoren des, Klimawandels; Gerecke, R., Franz, H., Eds.; Berchtesgadener Anzeiger: Berchtesgaden, Germany, 2006; Volume 51, pp. 255–263. [Google Scholar]
- Thomsen, P.F.; Willerslev, E. Environmental DNA—An emerging tool in conservation for monitoring past and present biodiversity. Biol. Conserv. 2015, 183, 4–18. [Google Scholar] [CrossRef]
- Senapati, D.; Bhattacharya, M.; Kar, A.; Chini, D.S.; Das, B.K.; Patra, B.C. Environmental DNA (eDNA): A Promising Biological Survey Tool for Aquatic Species Detection. Proc. Zool. Soc. 2019, 72, 211–228. [Google Scholar] [CrossRef]
- Elbrecht, V.; Steinke, D. Scaling up DNA metabarcoding for freshwater macrozoobenthos monitoring. Freshw. Biol. 2019, 64, 380–387. [Google Scholar] [CrossRef]
- Leese, F.; Sander, M.; Buchner, D.; Elbrecht, V.; Haase, P.; Zizka, V.M.A. Improved freshwater macroinvertebrate detection from environmental DNA through minimized nontarget amplification. Environ. DNA 2021, 3, 261–276. [Google Scholar] [CrossRef]
- Valentini, A.; Taberlet, P.; Miaud, C.; Civade, R.; Herder, J.; Thomsen, P.F.; Bellemain, E.; Besnard, A.; Coissac, E.; Boyer, F.; et al. Next-generation monitoring of aquatic biodiversity using environmental DNA metabarcoding. Mol. Ecol. 2016, 25, 929–942. [Google Scholar] [CrossRef]
- Tsuji, S.; Takahara, T.; Doi, H.; Shibata, N.; Yamanaka, H. The detection of aquatic macroorganisms using environmental DNA analysis—A review of methods for collection, extraction, and detection. Environ. DNA 2019, 1, 99–108. [Google Scholar] [CrossRef]
- Hernandez, C.; Bougas, B.; Perreault-Payette, A.; Simard, A.; Côté, G.; Bernatchez, L. 60 specific eDNA qPCR assays to detect invasive, threatened, and exploited freshwater vertebrates and invertebrates in Eastern Canada. Environ. DNA 2020, 2, 373–386. [Google Scholar] [CrossRef]
- Beng, K.C.; Corlett, R.T. Applications of environmental DNA (eDNA) in ecology and conservation: Opportunities, challenges and prospects. Biodiv. Conserv. 2020, 29, 2089–2121. [Google Scholar] [CrossRef]
- Fazio, M.; Celesti, A.; Puliafito, A.; Villari, M. Big Data Storage in the Cloud for Smart Environment Monitoring. Procedia Comput. Sci. 2015, 52, 500–506. [Google Scholar] [CrossRef]
- Horsburgh, J.S.; Reeder, S.L.; Jones, A.S.; Meline, J. Open source software for visualization and quality control of continuous hydrologic and water quality sensor data. Environ. Model. Softw. 2015, 70, 32–44. [Google Scholar] [CrossRef]
- Gerecke, R.; Franz, H.; Cantonati, M. Invertebrate diversity in springs of the National Park Berchtesgaden (Germany): Relevance for long-term monitoring. Int. Ver. Theor. Angew. Limnol. Verh. 2009, 30, 1229–1233. [Google Scholar] [CrossRef]
- Chang, Y.; Hartmann, A.; Liu, L.; Jiang, G.; Wu, J. Identifying More Realistic Model Structures by Electrical Conductivity Observations of the Karst Spring. Water Resour. Res. 2021, 57, e2020WR028587. [Google Scholar] [CrossRef]
- Einsiedl, F.; Maloszweski, P.; Stichler, W. Multiple isotope approache to the determination of the natural attenuation potential of a high-alpine karst system. J. Hydrol. 2009, 365, 113–121. [Google Scholar] [CrossRef]
- Kraller, G.; Strasser, U.; Franz, H. Effect of Alpine Karst on the hydrology of the basin “Berchtesgadener Ache”: A comprehensive summary of karst research in the Berchtesgaden Alps. J. Prot. Mt. Areas Res. Manag. 2011, 3, 19–28. [Google Scholar] [CrossRef]
- Garvelmann, J.; Warscher, M.; Leonhardt, G.; Franz, H.; Lotz, A.; Kunstmann, H. Quantification and characterization of the dynamics of spring and stream water systems in the Berchtesgaden Alps with a long-term stable isotope dataset. Environ. Earth Sci. 2017, 76, 766. [Google Scholar] [CrossRef]
- Kraller, G.; Warscher, M.; Vogl, S.; Marke, T.; Strasser, U.; Kunstmann, H. Water balance estimation in high Alpine terreain by combining distributed modeling and a neural network approach. Hydrol. Earth Syst. Sci. Discuss. 2012, 9, 1969–1990. [Google Scholar] [CrossRef]
- Warscher, M.; Kraller, G.; Franz, H.; Strasser, U.; Kunstmann, H. Performance of Complex Snow Cover Descriptions in a Distributed Hydrological Model System—A Case Study for the High Alpine Terrain of the Berchtesgaden Alps. Water Resour. Res. 2013, 49, 2619–2637. [Google Scholar] [CrossRef] [Green Version]
- Nationalpark Berchtesgaden. Quellmonitoring. Available online: http://www.nationalpark-berchtesgaden.bayern.de/forschung/monitoring/quellen/index.htm (accessed on 17 August 2022).
- Cantonati, M. (Ed.) Le Sorgenti del Parco Adamello-Brenta; Parco Adamello-Brenta: Trento, Italy, 1998; 177p, (In Italian with English and German Summaries). [Google Scholar]
- Merk, M.; Goeppert, N.; Goldscheider, N. Processes controlling spatial and temporal dynamics of spring water chemistry in the Black Forest National Park. Sci. Total Environ. 2020, 723, 137742. [Google Scholar] [CrossRef] [PubMed]
- Weigand, E.; Graf, W. Hydrobiologische Beweissicherung im Rahmen des LIFE-Projekts Nationalpark Kalkalpen. In Forschung im Nationalpark 2000; Bundesministerium für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft: Wien, Austria, 2000; pp. 36–37. [Google Scholar]
- Gerecke, R.; Haseke, H.; Klauber, J.; Maringer, A. (Eds.) Quellen; Schriften des Nationalparks Gesäuse: Weng im Gesäuse, Austria, 2012; Band 7; 391p. [Google Scholar]
- Kreiner, D.; Maringer, A.; Remschak, C.; Haseke, H.; Gerecke, R. Quellen—Forschung 2012–2017; Schriftenreihe des Nationalparks Gesäuse: Weng im Gesäuse, Austria, 2018; Volume 15, 193p. [Google Scholar]
- Remschak, C.; Haseke, H. Benthosuntersuchungen in Bächen, Quellen und Teichen im Rahmen des LIFE+ Projektes Ausseerland (Steirisches Salzkammergut); Mitteilungen des naturwissenschaftlichen Vereines für Steiermark: Graz, Austria, 2019; Volume 149, pp. 83–144. [Google Scholar]
- Von Fumetti, S.; Blattner, L. Faunistic assembalges of natural springs in different areas of the Swiss National Park: A small-scale comparison. Hydrobiologia 2017, 793, 175–184. [Google Scholar] [CrossRef]
- Lubini, V.; Stucki, P.; Vicentini, H.; Küry, D. Bewertung von Quell-Lebensräumen in der Schweiz. Entwurf für ein Strukturelles und Faunistisches Verfahren; Bericht im Auftrag des Bundesamtes für Umwelt BAFU: Zürich, Switzerland, 2014; pp. 1–46, (Update July 2016). [Google Scholar]
- Stevens, L.E.; Jenness, J.; Ledbetter, L.D. Springs and Springs-Dependent Taxa of the Colorado River Basin, Southwestern North America: Geography, Ecology and Human Impacts. Water 2020, 12, 1501. [Google Scholar] [CrossRef]
- Kurzweil, J.R.; Abdi, R.; Stevens, L.; Hogue, T.S. Utilization of ecological indicators to quantify distribution and conservation status of Mt. Tamalpais Springs, Marin County, California. Ecol. Indic. 2021, 125, 107544. [Google Scholar] [CrossRef]







| Parameter | Measurement Method | Monitoring Interval |
|---|---|---|
| Environment and structure mapping | Mapping sheet | every 5 years |
| Physical-chemical water properties | ||
| Discharge | Measurement container | 4(3) times per year (May, July, October) |
| Temperature (water and air) | Data logger | Hourly to four-hourly (loggers) |
| pH value | Multisensor device | 4(3) times per year |
| Electrical conductivity | Multisensor device | 4(3) times per year |
| Oxygen content (concentration and saturation) | Multisensor device | 4(3) times per year |
| Electrolytes/Nutrients | Ion chromatography | Once per year |
| Biological monitoring of aquatic organisms | ||
| Microbiology | Agar nutrient medium | Once per year |
| Botany—diatoms | Morphotaxonomy | every 5 years |
| Zoology—zoobenthos | Morphotaxonomy | every 5 years |
| Ecology | Zoogeography |
|---|---|
| Restricted nutritional range | Restricted or scattered distribution |
| Parasitic/symbiotic relations to other organisms | Population at border of distribution area |
| Sensitivity against increased nutrient contents | Changes in distribution during the past decades |
| Particular water chemistry requirements | Binding to particular vegetation ranges |
| Temperature sensitivity | Binding to particular elevation belts |
| Preference for a particular spring typology | Intraspecific variability |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cantonati, M.; Lichtenwöhrer, K.; Leonhardt, G.; Seifert, L.; Mustoni, A.; Hotzy, R.; Schubert, E.; Blattner, L.; Bilous, O.; Lotz, A.; et al. Using Springs as Sentinels of Climate Change in Nature Parks North and South of the Alps: A Critical Evaluation of Methodological Aspects and Recommendations for Long-Term Monitoring. Water 2022, 14, 2843. https://doi.org/10.3390/w14182843
Cantonati M, Lichtenwöhrer K, Leonhardt G, Seifert L, Mustoni A, Hotzy R, Schubert E, Blattner L, Bilous O, Lotz A, et al. Using Springs as Sentinels of Climate Change in Nature Parks North and South of the Alps: A Critical Evaluation of Methodological Aspects and Recommendations for Long-Term Monitoring. Water. 2022; 14(18):2843. https://doi.org/10.3390/w14182843
Chicago/Turabian StyleCantonati, Marco, Kurt Lichtenwöhrer, Gabi Leonhardt, Linda Seifert, Andrea Mustoni, Ralf Hotzy, Eva Schubert, Lucas Blattner, Olena Bilous, Annette Lotz, and et al. 2022. "Using Springs as Sentinels of Climate Change in Nature Parks North and South of the Alps: A Critical Evaluation of Methodological Aspects and Recommendations for Long-Term Monitoring" Water 14, no. 18: 2843. https://doi.org/10.3390/w14182843
APA StyleCantonati, M., Lichtenwöhrer, K., Leonhardt, G., Seifert, L., Mustoni, A., Hotzy, R., Schubert, E., Blattner, L., Bilous, O., Lotz, A., Poschlod, B., & Gerecke, R. (2022). Using Springs as Sentinels of Climate Change in Nature Parks North and South of the Alps: A Critical Evaluation of Methodological Aspects and Recommendations for Long-Term Monitoring. Water, 14(18), 2843. https://doi.org/10.3390/w14182843










