Brown Trout Upstream Passage Performance for a Fishway with Water Drops between Pools beyond Fish Passage Design Recommendations
Abstract
:1. Introduction
2. Materials and Methods
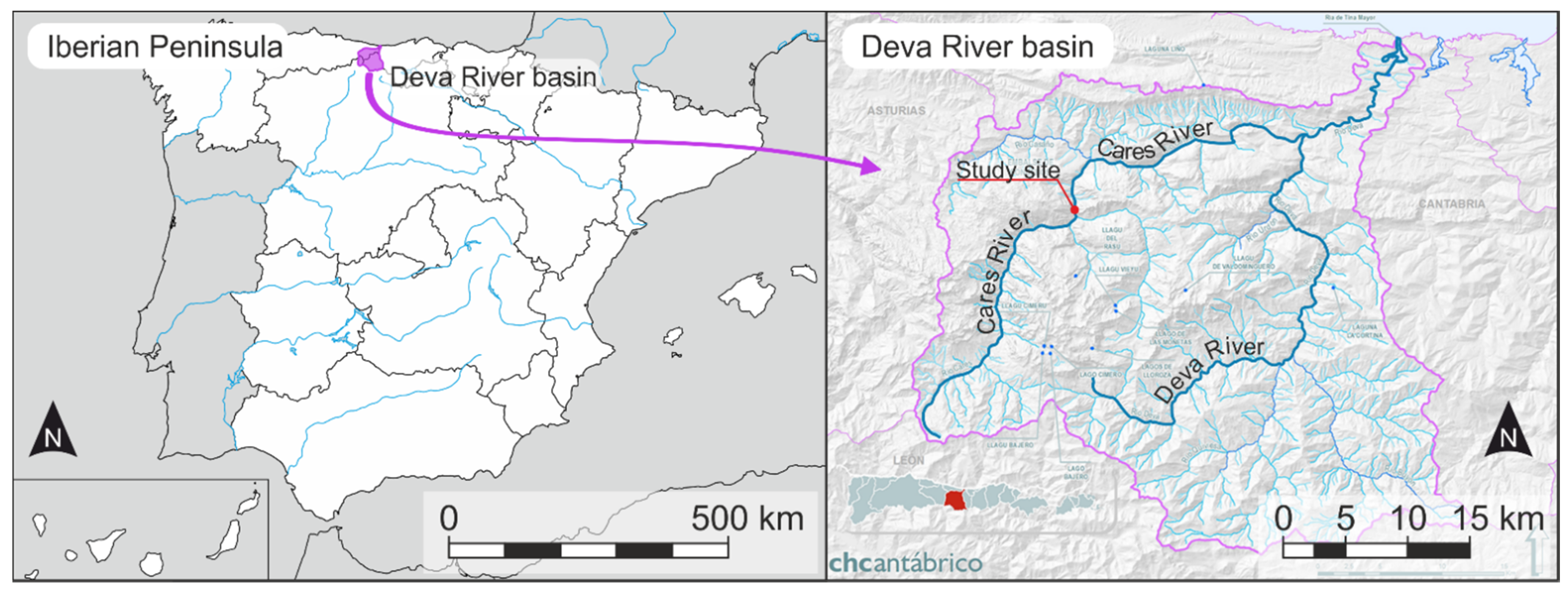
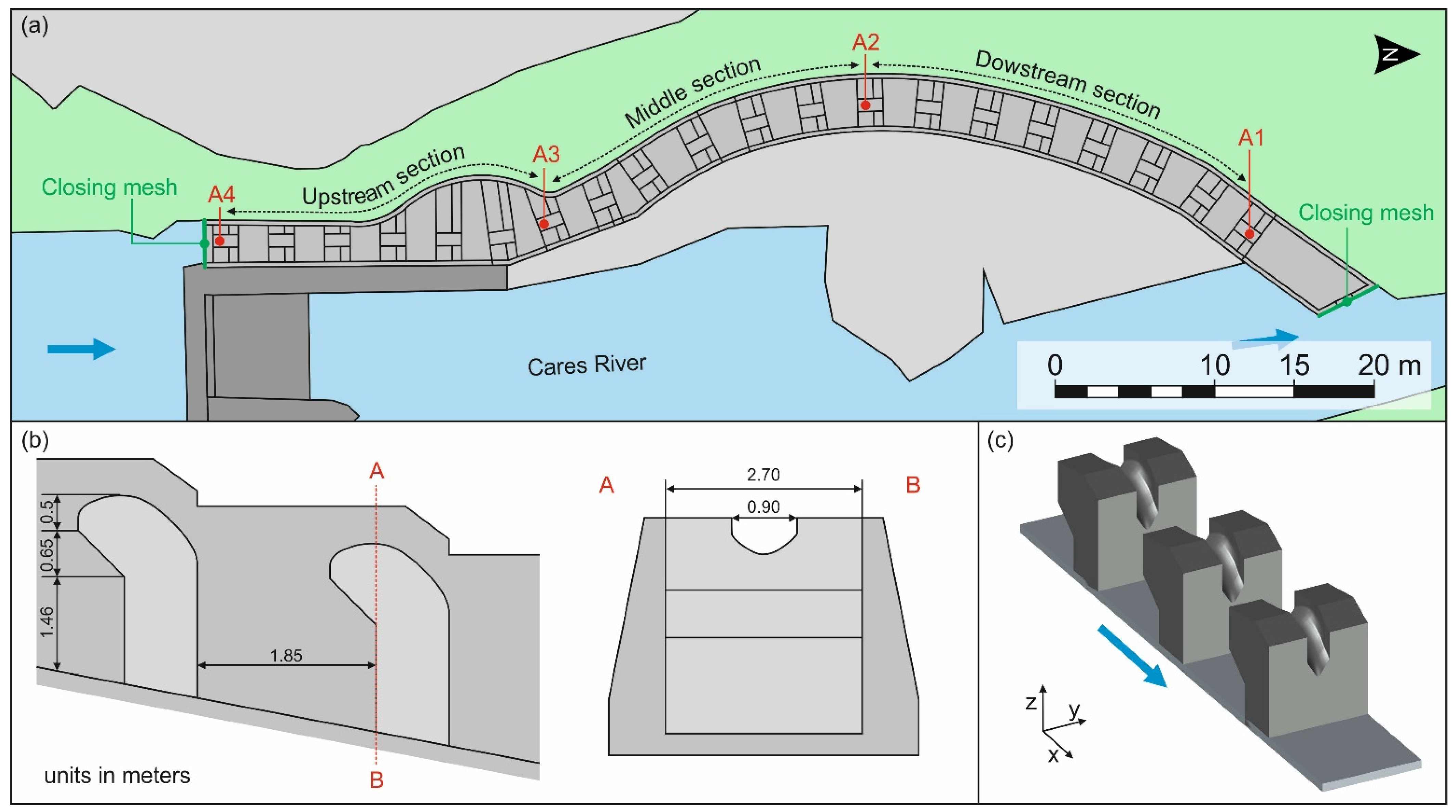
2.1. Study Site
2.2. The 3D Numerical Model
2.2.1. CFD Methods
2.2.2. Mesh, Boundary Conditions, and Time Sensitivity Analysis
2.2.3. CFD Data Treatment and Hydraulic Variables
2.3. Fish Response
2.3.1. Fish Collection and Tagging
2.3.2. Data Collection
- Fish with attempts were those with at least two records in A1, or one record in A1 and any other record in any of the subsequent antennas. The attempt percentage was defined as the total number of fish that made at least one attempt divided by the total number of fish.
- Successful events were assigned to those fish that reached A4. If not, they were considered failure events. The ascent success was defined as the total number of fish that reached A4 divided by the total number of fish that made at least one attempt.
- Height exceeded was a categorical variable based on the upstream antenna that the fish were able to overcome (A1 = 0.6 m/A2 = 3.8 m/A3 = 7.7 m/A4 = 11.8 m).
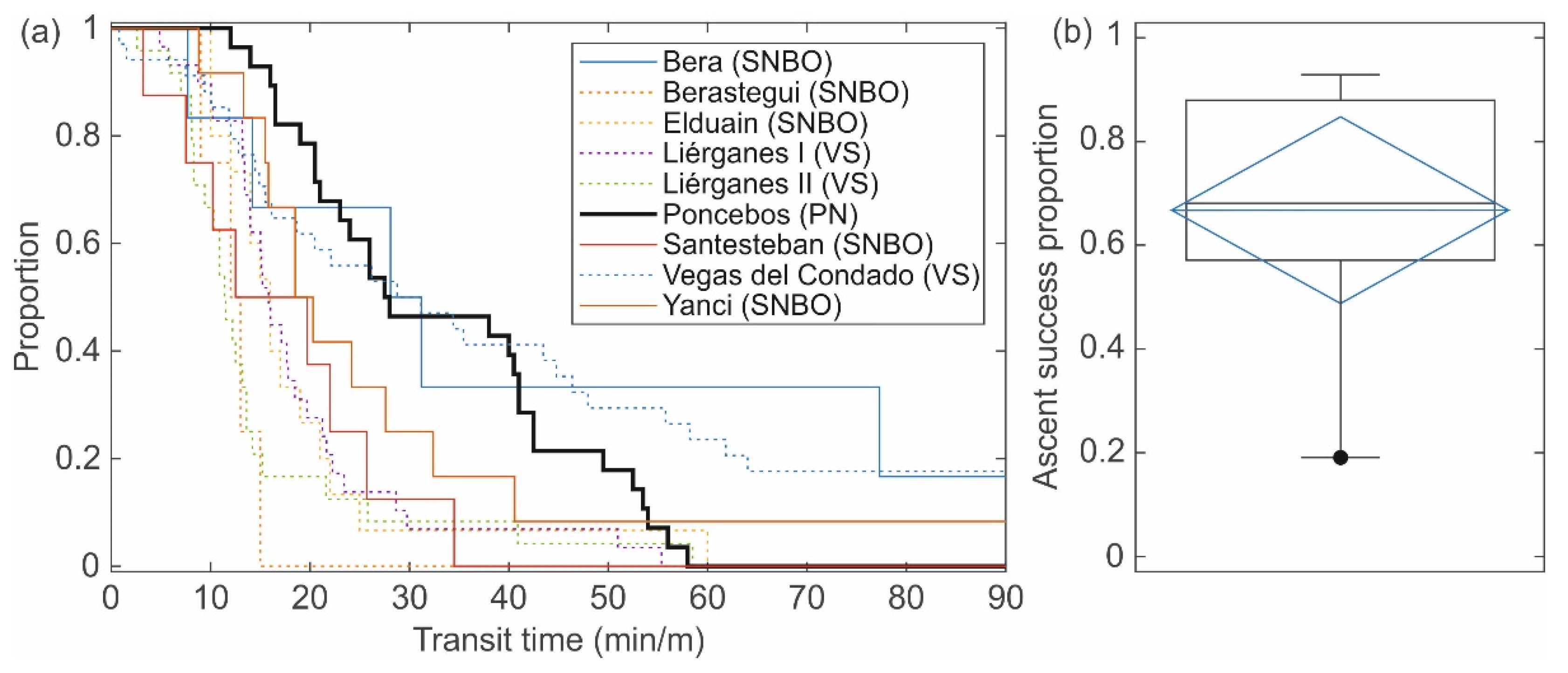
- The transit time was calculated as the time between the last detection in A1 and the first detection in A4 (only for fish with successful events). In addition, transit time was also calculated in each fishway section as the time between the downstream and the upstream antenna of the section (see Figure 2; downstream section: between A1 and A2, middle section: between A2 and A3, upstream section: between A3 and A4). To make possible the comparisons between other fishway assessments from the literature or between the different fishway sections, data of transit time were relativized by the water height difference between antennas, i.e., transit time per meter of ascended height [10].
- The time between the beginning of the trial and the first attempt was included as a sign of motivation. For those fish with at least one attempt, this time was considered as the lapse between the beginning of the trial and its first detection.
2.3.3. Data Analysis
3. Results
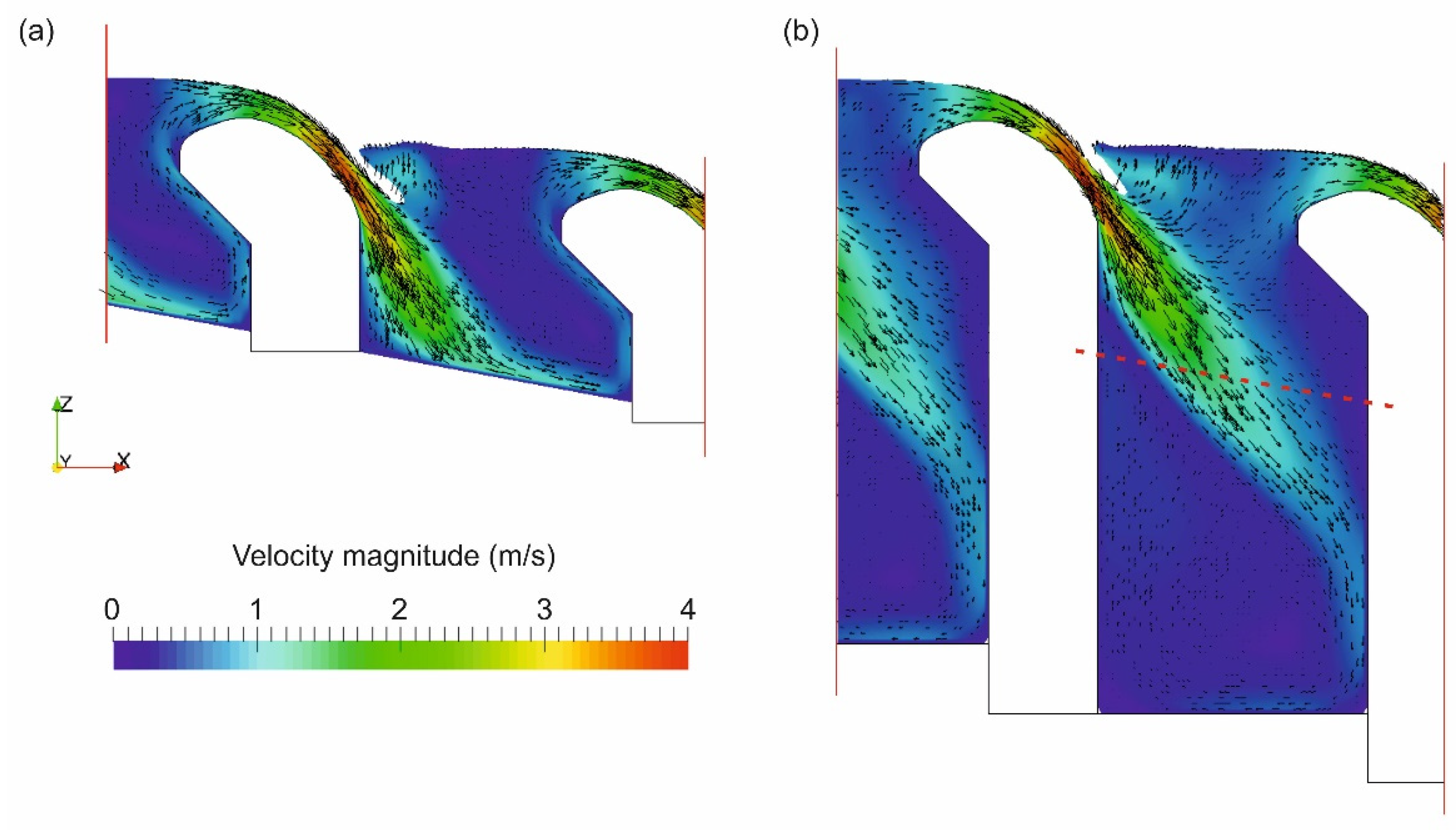
3.1. The 3D Numerical Models
3.2. Fish Response
4. Discussion
5. Summary and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nilsson, C.; Reidy, C.A.; Dynesius, M.; Revenga, C. Fragmentation and flow regulation of the world’s large river systems. Science 2005, 308, 405–408. [Google Scholar] [CrossRef] [PubMed]
- Reid, A.J.; Carlson, A.K.; Creed, I.F.; Eliason, E.J.; Gell, P.A.; Johnson, P.T.J.; Kidd, K.A.; MacCormack, T.J.; Olden, J.D.; Ormerod, S.J.; et al. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol. Rev. 2019, 94, 849–873. [Google Scholar] [CrossRef] [PubMed]
- European Commission Directive 2000/60/EC of the European Parliament and of the Council Establishing a Framework for the Community Action in the Field of Water Policy 2000. Available online: https://www.eea.europa.eu/policy-documents/directive-2000-60-ec-of (accessed on 3 March 2022).
- Habitats Directive Council Directive 92/43/EEC of 21 May 1992 on the conservation of natural habitats and wild fauna and flora. Off. J. Eur. Communities 1992, 206, 7–50. [CrossRef]
- Clay, C.H. Design of Fishways and Other Fish Facilities; CRC Press: Ottawa, ON, Canada, 1995; ISBN 1566701112. [Google Scholar]
- FAO/DVWK Fish Passes: Design, Dimensions, and Monitoring; FAO: Rome, Italy, 2002; ISBN 9251048940.
- Santos, J.M.; Silva, A.T.; Katopodis, C.; Pinheiro, P.; Pinheiro, A.; Bochechas, J.; Ferreira, M.T. Ecohydraulics of pool-type fishways: Getting past the barriers. Ecol. Eng. 2012, 48, 38–50. [Google Scholar] [CrossRef]
- Noonan, M.J.; Grant, J.W.A.; Jackson, C.D. A quantitative assessment of fish passage efficiency. Fish Fish. 2012, 13, 450–464. [Google Scholar] [CrossRef]
- Hershey, H. Updating the consensus on fishway efficiency: A meta-analysis. Fish Fish. 2021, 22, 735–748. [Google Scholar] [CrossRef]
- Bravo-Córdoba, F.J.; Valbuena-Castro, J.; García-Vega, A.; Fuentes-Pérez, J.F.; Ruiz-Legazpi, J.; Sanz-Ronda, F.J. Fish passage assessment in stepped fishways: Passage success and transit time as standardized metrics. Ecol. Eng. 2021, 162, 106172. [Google Scholar] [CrossRef]
- Larinier, M. Pool fishways, pre-barrages and natural bypass channels. Bull. Fr. Pêche Piscic. 2002, 364, 54–82. [Google Scholar] [CrossRef]
- Fuentes-Pérez, J.F.; Eckert, M.; Tuhtan, J.A.A.; Ferreira, M.T.; Kruusmaa, M.; Branco, P. Spatial preferences of Iberian barbel in a vertical slot fishway under variable hydrodynamic scenarios. Ecol. Eng. 2018, 125, 131–142. [Google Scholar] [CrossRef]
- Wu, S.; Rajaratnam, N.; Katopodis, C. Structure of flow in vertical slot fishway. J. Hydraul. Eng. 1999, 125, 351–360. [Google Scholar] [CrossRef]
- Ead, S.A.; Katopodis, C.; Sikora, G.J.; Rajaratnam, N. Flow regimes and structure in pool and weir fishways. J. Environ. Eng. Sci. 2004, 3, 379–390. [Google Scholar] [CrossRef]
- Rajaratnam, N.; Katopodis, C.; Mainali, A. Plunging and streaming flows in pool and weir fishways. J. Hydraul. Eng. 1988, 114, 939–944. [Google Scholar] [CrossRef]
- Fuentes-Pérez, J.F.; García-Vega, A.; Sanz-Ronda, F.J.; Martínez de Azagra Paredes, A. Villemonte’s approach: Validation of a general method for modeling uniform and non-uniform performance in stepped fishways. Knowl. Manag. Aquat. Ecosyst. 2017, 418, 11. [Google Scholar] [CrossRef]
- Rajaratnam, N.; Van der Vinne, G.; Katopodis, C. Hydraulics of vertical slot fishways. J. Hydraul. Eng. 1986, 112, 909–927. [Google Scholar] [CrossRef]
- Branco, P.; Santos, J.M.; Katopodis, C.; Pinheiro, A.; Ferreira, M.T. Pool-Type Fishways: Two Different Morpho-Ecological Cyprinid Species Facing Plunging and Streaming Flows. PLoS ONE 2013, 8, e65089. [Google Scholar] [CrossRef] [PubMed]
- Piper, A.T.; Wright, R.M.; Kemp, P.S. The influence of attraction flow on upstream passage of European eel (Anguilla anguilla) at intertidal barriers. Ecol. Eng. 2012, 44, 329–336. [Google Scholar] [CrossRef]
- Thompson, C. Effect of Flow on Performance and Behavior of Chinook Salmon in Fishways; No. 601.; US Department of the Interior, Bureau of Commercial Fisheries: Fairfax, VA, USA, 1970. [Google Scholar]
- Collins, C.; Elling, C. Fishway Research at the Fisheries-Engineering Research Laboratory; United States, Department of the Interior, Fish and Wildlife Service, Bureau of Commercial Fisheries: Fairfax, VA, USA, 1960. [Google Scholar]
- Fjeldstad, H.-P.; Alfredsen, K.; Forseth, T. Atlantic salmon fishways: The Norwegian experiences. Vann 2013, 2, 191–204. [Google Scholar]
- Kondratieff, M.C.; Myrick, C.a. How High Can Brook Trout Jump? A Laboratory Evaluation of Brook Trout Jumping Performance. Trans. Am. Fish. Soc. 2006, 135, 361–370. [Google Scholar] [CrossRef]
- Weaver, C.R. Fluence of Water Velocity upon Orientation and Perform-Ance of Adult Migrating Salmonids. Fish. Bull. Fish Wildl. Serv. 1964, 63, 97–121. [Google Scholar]
- Liu, M.; Rajaratnam, N.; Zhu, D.Z. Mean flow and turbulence structure in vertical slot fishways. J. Hydraul. Eng. 2006, 132, 765–777. [Google Scholar] [CrossRef]
- Enders, E.C.; Boisclair, D.; Roy, A.G. The effect of turbulence on the cost of swimming for juvenile Atlantic salmon (Salmo salar). Can. J. Fish. Aquat. Sci. 2003, 60, 1149–1160. [Google Scholar] [CrossRef]
- Enders, E.C.; Boisclair, D.; Roy, A.G. A model of total swimming costs in turbulent flow for juvenile Atlantic salmon (Salmo salar). Can. J. Fish. Aquat. Sci. 2005, 62, 1079–1089. [Google Scholar] [CrossRef] [Green Version]
- Guiny, E.; Ervine, D.A.; Armstrong, J.D. Hydraulic and biological aspects of fish passes for Atlantic salmon. J. Hydraul. Eng. 2005, 131, 542–553. [Google Scholar] [CrossRef]
- Aaserude, R.; Orsborn, J. New Concepts in Fish Ladder Design, Volume II of IV, Results of Laboratory and Field Research on New Concepts in Weir and Pool Fishways, 1982–1984 Final. 1985. Available online: https://digital.library.unt.edu/ark:/67531/metadc1100445/ (accessed on 26 February 2022).
- Flosi, G.; Downie, S.; Bird, M.; Coey, R.; Collins, B. California Salmonid Stream Habitat Restoration Manual. 2002. Available online: https://creeks.berkeley.edu/publications/california-salmonid-stream-habitat-restoration-manual-4th-ed-part-1 (accessed on 14 February 2022).
- Meixler, M.S.; Bain, M.B.; Todd Walter, M. Predicting barrier passage and habitat suitability for migratory fish species. Ecol. Modell. 2009, 220, 2782–2791. [Google Scholar] [CrossRef]
- Amaral, S.D.; Branco, P.; Katopodis, C.; Ferreira, M.T.; Pinheiro, A.N.; Santos, J.M. To swim or to jump? Passage behaviour of a potamodromous cyprinid over an experimental broad-crested weir. River Res. Appl. 2018, 34, 174–182. [Google Scholar] [CrossRef]
- Beamish, F.W.H. Swimming capacity. In Fish Physiology, Vol. VII. Locomotion; Hoar, W.H., Randall, D.J., Eds.; Academic Press: New York, NY, USA, 1978; pp. 101–187. [Google Scholar]
- Haro, A.; Castro-Santos, T.; Noreika, J.; Odeh, M. Swimming performance of upstream migrant fishes in open-channel flow: A new approach to predicting passage through velocity barriers. Can. J. Fish. Aquat. Sci. 2004, 61, 1590–1601. [Google Scholar] [CrossRef]
- Ruiz-Legazpi, J.; Sanz-Ronda, F.J.; Bravo-Córdoba, F.J.; Fuentes-Pérez, J.F.; Castro-Santos, T. Influencia de factores ambientales y biométricos en la capacidad de nado del barbo ibérico (Luciobarbus bocagei Steindachner, 1864), un ciprínido potamódromo endémico de la Península Ibérica. Limnetica 2018, 37, 251–265. [Google Scholar] [CrossRef]
- David, G.; Céline, C.; Morgane, B.; Franck, C. Ecological connectivity of the upper Rhône River: Upstream fish passage at two successive large hydroelectric dams for partially migratory species. Ecol. Eng. 2022, 178, 106545. [Google Scholar] [CrossRef]
- Brett, J.R. Energetic Responses of Salmon to Temperature. A Study of Some Thermal Relations in the Physiology and Freshwater Ecology of Sockeye Salmon (Oncorhynchus nerkd). Am. Zool. 1971, 11, 99–113. [Google Scholar] [CrossRef]
- Francis, F. Reports on Salmon Ladders; Horace Cox, 346, Strand, W.C.: London, UK, 1870. [Google Scholar]
- Doadrio, I.; Perea, S.; Garzón-Heydt, P.; González, J.L. Ictiofauna Continental Española: Bases Para Su Seguimiento; Ministerio de Medio Ambiente y Medio Rural y Marino: Madrid, Spain, 2011. [Google Scholar]
- CHC Río Cares. Las cuencas Cantábricas; 2016. Available online: https://www.chcantabrico.es/las-cuencas-cantabricas/marco-fisico/hidrologia/rios/cares (accessed on 3 April 2022).
- Huet, M. Biologie, profils en long et en travers des eaux courantes. Bull. Français Piscic. 1954, 175, 41–53. [Google Scholar] [CrossRef]
- Illies, J.; Botoseanu, L. Problèmes et méthodes de la classification et de la-zonation écologique des eaux courantes, considérées surtout-du point de vue faunistique. Mitt. Int. Ver. Fuer Theor. Unde Amgewandte Limnol. 1963, 12, 1–57. [Google Scholar] [CrossRef]
- Rosgen, D.L.; Silvey, H.L. Applied River Morphology; Wildland Hydrology: Pagosa Springs, CO, USA, 1996; Volume 1481. [Google Scholar]
- IGME. Estudio Hidrogeológico de la Cuenca Norte de España (Asturias); 1982. Available online: info.igme.es/SidPDF/018000/829/Volumen%204%20Informe%20t%C3%A9cnico%202/18829_0006.pdf (accessed on 27 March 2022).
- Allison, P.D. Survival Analysis using Sas: A Practical Guide; SAS Institute: Cary, NC, USA, 2010. [Google Scholar]
- O’Brien, R.M. A Caution Regarding Rules of Thumb for Variance Inflation Factors. Qual. Quant. 2007, 41, 673–690. [Google Scholar] [CrossRef]
- Kleinbaum, D.G.; Klein, M. Survival analysis: A Self-Learning Text, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2010; ISBN 1441966455. [Google Scholar]
- Akaike, H. A new look at the statistical model Identification. IEEE Trans. Automat. Contr. 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Towler, B.; Orvis, C.; Dow, D.; Haro, A. Fishway Inspection Guidelines; Univeristy of Massachusetts Amherst: Amherst, MA, USA, 2013; Volume 18. [Google Scholar]
- Baudoin, J.-M.; Burgun, V.; Chanseau, M.; Larinier, M.; Ovidio, M.; Sremski, W.; Steinbach, P.; Voegtle, B. Assessing the Passage of Obstacles by Fish. Concepts, Design and Application; Onema: Paris, France, 2015. [Google Scholar]
- Valbuena-Castro, J.; Fuentes-Pérez, J.F.; García-Vega, A.; Bravo-Córdoba, F.J.; Ruiz-Legazpi, J.; Martínez de Azagra-Paredes, A.; Sanz-Ronda, F.J. Coarse fishway assessment to prioritize retrofitting efforts: A case study in the Duero River basin. Ecol. Eng. 2020, 155, 105946. [Google Scholar] [CrossRef]
- Silva, A.T.; Lucas, M.C.; Castro-Santos, T.; Katopodis, C.; Baumgartner, L.J.; Thiem, J.D.; Aarestrup, K.; Pompeu, P.S.; O’Brien, G.C.; Braun, D.C. The future of fish passage science, engineering, and practice. Fish Fish. 2018, 19, 340–362. [Google Scholar] [CrossRef]
- Fuentes-Pérez, J.F.; Sanz-Ronda, F.J.; Martínez de Azagra-Paredes, A.; García-Vega, A. Non-uniform hydraulic behavior of pool-weir fishways: A tool to optimize its design and performance. Ecol. Eng. 2016, 86, 5–12. [Google Scholar] [CrossRef]
- Fuentes-Pérez, J.F.; Silva, A.T.; Tuhtan, J.A.; García-Vega, A.; Carbonell-Baeza, R.; Musall, M.; Kruusmaa, M. 3D modelling of non-uniform and turbulent flow in vertical slot fishways. Environ. Model. Softw. 2018, 99, 156–169. [Google Scholar] [CrossRef]
- DWA. Fischaufstiegsanlagen und Fischpassierbare Bauwerke. Gestaltung, Bemessung, Qualitätssicherung; Merkblatt DWA -M; DWA: Hennef, Germany, 2014; ISBN 9783942964913. [Google Scholar]
- Castro-Santos, T.; Sanz-Ronda, F.J.; Ruiz-Legazpi, J. Breaking the speed limit- comparative sprinting performance of brook trout (Salvelinus fontinalis) and brown trout (Salmo trutta). Can. J. Fish. Aquat. Sci. 2013, 70, 280–293. [Google Scholar] [CrossRef]
- Larinier, M.; Travade, F.; Porcher, J.P. Fishways: Biological basis, design criteria and monitoring. Bull. Français la Pêche la Piscic. 2002, 364, 208. [Google Scholar]
- Cooke, S.J.; Hinch, S.G. Improving the reliability of fishway attraction and passage efficiency estimates to inform fishway engineering, science, and practice. Ecol. Eng. 2013, 58, 123–132. [Google Scholar] [CrossRef]
- Ovidio, M.; Sonny, D.; Dierckx, A.; Watthez, Q.; Bourguignon, S.; de le Court, B.; Detrait, O.; Benitez, J.P. The use of behavioural metrics to evaluate fishway efficiency. River Res. Appl. 2017, 33, 1484–1493. [Google Scholar] [CrossRef]
- Bunt, C.M.; Castro-Santos, T.; Haro, A. Reinforcement and Validation of the Analyses and Conclusions Related to Fishway Evaluation Data from Bunt et al.: “Performance of Fish Passage Structures at Upstream Barriers to Migration”. River Res. Appl. 2016, 32, 2125–2137. [Google Scholar] [CrossRef]
- Elling, C.; Raymond, H. Fishway Capacity Experiment, 1956; US Department of Interior, Fish and Wildlife Service: Fairfax, VA, USA, 1959. [Google Scholar]
- USFWS (U.S. Fish and Wildlife Service). Fish Passage Engineering Design Criteria; USFWS: Hadley, MA, USA, 2017. [Google Scholar]
- Sánchez-González, J.R.; Morcillo, F.; Ruiz-Legazpi, J.; Sanz-Ronda, F.J. Fish morphology and passage through velocity barriers. Experience with northern straight-mouth nase (Pseudochondrostoma duriense Coelho, 1985) in an open channel flume. Hydrobiologia 2022, 849, 1351–1366. [Google Scholar] [CrossRef]
- Thurow, R.F. Life Histories of Potamodromous Fishes. In An Introduction to Fish Migration; Morais, P., Daverat, F., Eds.; CRC Press: Boca Raton, FL, USA, 2016; p. 29. [Google Scholar]
- Sanz-Ronda, F.J.; Bravo-Córdoba, F.J.; Sánchez-Pérez, A.; García-Vega, A.; Valbuena-Castro, J.; Fernandes-Celestino, L.; Torralva, M.; Oliva-Paterna, F.J. Passage Performance of Technical Pool-Type Fishways for Potamodromous Cyprinids: Novel Experiences in Semiarid Environments. Water 2019, 11, 2362. [Google Scholar] [CrossRef]
- Froese, R. Cube law, condition factor and weight–length relationships: History, meta-analysis and recommendations. J. Appl. Ichthyol. 2006, 22, 241–253. [Google Scholar] [CrossRef]
- Sanz-Ronda, F.J.; Fuentes-Pérez, J.F.; García-Vega, A.; Bravo-Córdoba, F.J. Fishways as downstream routes in small hydropower plants: Experiences with a potamodromous cyprinid. Water 2021, 13, 1041. [Google Scholar] [CrossRef]
- Fuentes-Pérez, J.F.; García-Vega, A.; Bravo-Córdoba, F.J.; Sanz-Ronda, F.J. A Step to Smart Fishways: An Autonomous Obstruction Detection System Using Hydraulic Modeling and Sensor Networks. Sensors 2021, 21, 6909. [Google Scholar] [CrossRef]
- Tamario, C.; Sunde, J.; Petersson, E.; Tibblin, P.; Forsman, A. Ecological and Evolutionary Consequences of Environmental Change and Management Actions for Migrating Fish. Front. Ecol. Evol. 2019, 7, 271. [Google Scholar] [CrossRef] [Green Version]

| Variables | Values |
|---|---|
| Number of pools | 20 |
| Total water height 1 | 13.33 m |
| Pool dimension 1 | |
| Length | 1.85 (1.50–2.00) m |
| Width | 2.70 (2.50–3.50) m |
| Depth | 1.80 (1.00–5.00) m |
| Width of the notch 1 | 0.60–0.90 m |
| Thickness of the notch 1 | 1.60 m |
| Maximum water velocity at the notches 2 | 3.40 (2.80–3.81) m/s |
| Drop between pools 1 | 0.65 (0.40–0.74) |
| Slope | 16.38% |
| Flow discharge 3 | 0.23 ± 0.08 m3/s |
| Volumetric power dissipation | 163 (57–390) W/m3 |
| Salmo trutta | N | Body Length (cm) | Weight (g) | K (g/cm3) | |||
|---|---|---|---|---|---|---|---|
| Mean ± SD | Range | Mean ± SD | Range | Mean ± SD | Range | ||
| Group 1 | 35 | 24.7 ± 4.0 | 17.5–40.5 | 193 ± 108 | 58–710 | 1.19 ± 0.11 | 0.84–1.46 |
| Group 2 | 34 | 24.6 ± 2.6 | 20.2–30.5 | 181 ± 60 | 94–356 | 1.18 ± 0.08 | 0.97–1.36 |
| p = 0.564 | p = 0.867 | p = 0.563 | |||||
| β ± SE | p-Value | HR | |
|---|---|---|---|
| Transit time (n = 11) | |||
| Body length (mm) | 0.038 ± 0.019 | 0.048 | 1.038 |
| Condition factor | 0.362 | ||
| Transit time by fishway section (n = 37) | |||
| Body length (mm) | 0.022 ± 0.005 | <0.001 | 1.022 |
| Condition factor | 0.080 | ||
| Fishway section= upstream section | Reference level | ||
| Fishway section = downstream section | −1.724 ± 0.493 | <0.001 | 0.178 |
| Fishway section = middle section | −1.282 ± 0.486 | <0.008 | 0.277 |
| Height exceeded (n = 69) | |||
| Body length (mm) | −0.021 ± 0.006 | 0.001 | 0.980 |
| Condition factor | 0.697 | ||
| Time between the beginning and the first attempt (n = 69) | |||
| Body length (mm) | 0.013 ± 0.004 | 0.002 | 1.013 |
| Condition factor | 0.263 | ||
| Downstream Section | Middle Section | Upstream Section | |
|---|---|---|---|
| Passage success (N success/N attempt) | 36% (21/59) | 71% (15/21) | 73% (11/15) |
| Median transit time (min–max) (min/m) | 53 (12–287) | 41 (16–207) | 21 (14–52) |
| Median VPD (min–max) (W/m3) | 243 (114–390) a | 89 (71–99) ab | 56 (51–62) b |
| p-value < 0.001 | |||
| Median water drop (min–max) (m) | 0.63 (0.39–0.74) | 0.65 (0.63–0.72) | 0.64 (0.63–0.66) |
| p-value = 0.816 | |||
| Median pool depth (min–max) (m) | 1.01 (0.85–2.5) a | 3.50 (3.00–4.00) ab | 5.00 (5.00–6.00) b |
| p-value < 0.001 | |||
| β | SE | |
|---|---|---|
| Transit time (n = 11) | ||
| Intercept | 4.999 | 0.620 |
| Body length (mm) | −0.006 | 0.002 |
| Shape | 0.198 | 0.048 |
| Transit time by fishway section (n = 37) | ||
| Intercept | 5.646 | 0.786 |
| Body length (mm) | −0.009 | 0.003 |
| Fishway section= upstream section | Reference level | |
| Fishway section = downstream | 0.804 | 0.296 |
| Fishway section = middle | 0.784 | 0.337 |
| Shape | 0.431 | 0.059 |
| Height exceeded (n = 69) | ||
| Intercept | −11.324 | 2.891 |
| Body length (mm) | 0.046 | 0.012 |
| Shape | 1.509 | 0.197 |
| Time between the beginning and the first attempt (n = 69) | ||
| Intercept | 9.883 | 1.508 |
| Body length (mm) | −0.019 | 0.006 |
| Shape | 0.991 | 0.107 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bravo-Córdoba, F.J.; Fuentes-Pérez, J.F.; García-Vega, A.; Peñas, F.J.; Barquín, J.; Sanz-Ronda, F.J. Brown Trout Upstream Passage Performance for a Fishway with Water Drops between Pools beyond Fish Passage Design Recommendations. Water 2022, 14, 2750. https://doi.org/10.3390/w14172750
Bravo-Córdoba FJ, Fuentes-Pérez JF, García-Vega A, Peñas FJ, Barquín J, Sanz-Ronda FJ. Brown Trout Upstream Passage Performance for a Fishway with Water Drops between Pools beyond Fish Passage Design Recommendations. Water. 2022; 14(17):2750. https://doi.org/10.3390/w14172750
Chicago/Turabian StyleBravo-Córdoba, Francisco Javier, Juan Francisco Fuentes-Pérez, Ana García-Vega, Francisco Jesús Peñas, José Barquín, and Francisco Javier Sanz-Ronda. 2022. "Brown Trout Upstream Passage Performance for a Fishway with Water Drops between Pools beyond Fish Passage Design Recommendations" Water 14, no. 17: 2750. https://doi.org/10.3390/w14172750










