Hydrocarbon Biodegradation in Utah’s Great Salt Lake
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Salinity of the North and South Arm of the Great Salt Lake
3.2. Gasoline Degradation
3.3. Crude Oil Degradation
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohammed, I.N.; Tarboton, D.G. An examination of the sensitivity of the Great Salt Lake to changes in inputs. Water Res. Res. 2012, 48, W11511. [Google Scholar] [CrossRef]
- Meuser, J.E.; Baxter, B.K.; Spear, J.R.; Peters, J.W.; Posewitz, M.C.; Boyd, E.S. Contrasting patterns of community assembly in the stratified water column of Great Salt Lake, Utah. Microb. Ecol. 2013, 66, 268–280. [Google Scholar] [CrossRef] [PubMed]
- Brock, T.D. Salinity and the ecology of Dunaliella from Great Salt Lake. J. Gen. Microbiol. 1975, 89, 285–292. [Google Scholar] [CrossRef]
- Post, F.J. The microbial ecology of the Great Salt Lake. Microb. Ecol. 1977, 3, 143–165. [Google Scholar] [CrossRef] [PubMed]
- Bortz, L.C. Heavy-Oil Deposit, Great Salt Lake, Utah: Section V. Exploration Histories. In SG 25: Exploration for Heavy Crude Oil and Natural Bitumen; AAPG Special issue; AAPG: Tulsa, OK, USA, 1987; pp. 555–563. [Google Scholar]
- Sinninghe Damsté, J.S.; De Leeuw, J.W.; Kock-van Dalen, A.C.; De Zeeuw, M.A.; De Lange, F.; Irene, W.; Rijpstra, C.; Schenck, P.A. The occurrence and identification of series of organic sulphur compounds in oils and sediment extracts. I. A study of Rozel Point Oil (USA). Geochim. Cosmochim. Acta 1987, 51, 2369–2391. [Google Scholar] [CrossRef]
- Abou Khalil, C.; Prince, V.L.; Prince, R.C.; Greer, C.W.; Lee, K.; Zhang, B.; Boufadel, M.C. Occurrence and biodegradation of hydrocarbons at high salinities. Sci. Total Environ. 2021, 762, 143165. [Google Scholar] [CrossRef] [PubMed]
- Ward, D.M.; Brock, T.D. Hydrocarbon biodegradation in hypersaline environments. Appl. Environ. Microbiol. 1978, 35, 353–359. [Google Scholar] [CrossRef]
- Sei, A.; Fathepure, B.Z. Biodegradation of BTEX at high salinity by an enrichment culture from hypersaline sediments of Rozel Point at Great Salt Lake. J. Appl. Microbiol. 2009, 107, 2001–2008. [Google Scholar] [CrossRef]
- Stephens, D.W.; Gillespie, D.M. Phytoplankton production in the Great Salt Lake, Utah, and a laboratory study of algal response to enrichment 1. Limnol. Oceanogr. 1978, 21, 74–87. [Google Scholar] [CrossRef]
- Bragg, J.R.; Prince, R.C.; Harner, E.J.; Atlas, R.M. Effectiveness of bioremediation for the Exxon Valdez oil spill. Nature 1994, 368, 413–418. [Google Scholar] [CrossRef]
- Prince, R.C.; Parkerton, T.F.; Lee, C. The primary aerobic biodegradation of gasoline hydrocarbons. Environ. Sci. Technol. 2007, 41, 3316–3321. [Google Scholar] [CrossRef] [PubMed]
- Uhler, R.M.; Healey, E.M.; McCarthy, K.J.; Uhler, A.D.; Stout, S.A. Molecular fingerprinting of gasoline by a modified EPA 8260 gas chromatography-mass spectrometry method. Int. J. Environ. Anal. Chem. 2003, 83, 1–20. [Google Scholar] [CrossRef]
- Prince, R.C.; Butler, J.D.; Redman, A.D. The rate of crude oil biodegradation in the sea. Environ. Sci. Technol. 2017, 51, 1278–1284. [Google Scholar] [CrossRef] [PubMed]
- Abou Khalil, C.; Fortin, N.; Prince, R.C.; Greer, C.W.; Lee, K.; Boufadel, M.C. Crude oil biodegradation in upper and supratidal seashores. J. Hazard. Mater. 2021, 416, 125919. [Google Scholar] [CrossRef] [PubMed]
- Tissot, B.P.; Welte, D.H. Petroleum Formation and Occurrence; Springer: Berlin/Heidelberg, Germany, 1984. [Google Scholar] [CrossRef]
- Prince, R.C.; McFarlin, K.M.; Butler, J.D.; Febbo, E.J.; Wang, F.C.; Nedwed, T.J. The primary biodegradation of dispersed crude oil in the sea. Chemosphere 2013, 90, 521–526. [Google Scholar] [CrossRef]
- Brakstad, O.G.; Nordtug, T.; Throne-Holst, M. Biodegradation of dispersed Macondo oil in seawater at low temperature and different oil droplet sizes. Mar. Pollut. Bull. 2015, 93, 144–152. [Google Scholar] [CrossRef]
- Wang, J.; Sandoval, K.; Ding, Y.; Stoeckel, D.; Minard-Smith, A.; Andersen, G.; Dubinsky, E.A.; Atlas, R.; Gardinali, P. Biodegradation of dispersed Macondo crude oil by indigenous Gulf of Mexico microbial communities. Sci. Total Environ. 2016, 557, 453–468. [Google Scholar] [CrossRef]
- Owens, E.H.; Taylor, E.; Marty, R.; Little, D.I. An inland oil spill response manual to minimize adverse environmental impacts. Proc. Int. Oil Spill Conf. 1993, 105–109. [Google Scholar] [CrossRef]
- Prince, R.C.; Garrett, R.M.; Bare, R.E.; Grossman, M.J.; Townsend, T.; Suflita, J.M.; Lee, K.; Owens, E.H.; Sergy, G.A.; Braddock, J.F.; et al. The roles of photooxidation and biodegradation in long-term weathering of crude and heavy fuel oils. Spill Sci. Technol. Bull. 2003, 8, 145–156. [Google Scholar] [CrossRef]
- Garrett, R.M.; Pickering, I.J.; Haith, C.E.; Prince, R.C. Photooxidation of crude oils. Environ. Sci. Technol. 1998, 32, 3719–3723. [Google Scholar] [CrossRef]
- Aeppli, C.; Mitchell, D.A.; Keyes, P.; Beirne, E.C.; McFarlin, K.M.; Roman-Hubers, A.T.; Rusyn, I.; Prince, R.C.; Zhao, L.; Parkerton, T.F.; et al. Oil irradiation experiments document changes in oil properties, molecular composition, and dispersant effectiveness associated with oil photo-oxidation. Environ. Sci. Technol. 2022, 56, 7789–7799. [Google Scholar] [CrossRef] [PubMed]
- Haws, E.S. Ecology of culturable organisms at Rozel Point, Great Salt Lake, Utah. Master’s Thesis, Brigham Young University, Provo, UT, USA, 2007. Available online: https://scholarsarchive.byu.edu/etd/857 (accessed on 21 July 2022).
- Almeida-Dalmet, S.; Baxter, B.K. Unexpected complexity at salinity saturation: Microbial diversity of the North Arm of Great Salt Lake. In Great Salt Lake Biology; Baxter, B., Butler, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; pp. 119–144. [Google Scholar] [CrossRef]
- Tazi, L.; Breakwell, D.P.; Harker, A.R.; Crandall, K.A. Life in extreme environments: Microbial diversity in Great Salt Lake, Utah. Extremophiles 2014, 18, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Corsellis, Y.Y.; Krasovec, M.M.; Sylvi, L.L.; Cuny, P.P.; Militon, C.C. Oil removal and effects of spilled oil on active microbial communities in close to salt-saturation brines. Extremophiles 2016, 20, 235–250. [Google Scholar] [CrossRef]
- Kebbouche-Gana, S.; Gana, M.L.; Khemili, S.; Fazouane-Naimi, F.; Bouanane, N.A.; Penninckx, M.; Hacene, H. Isolation and characterization of halophilic Archaea able to produce biosurfactants. J. Ind. Microbiol. Biotechnol. 2009, 36, 727–738. [Google Scholar] [CrossRef] [PubMed]
- Baxter, B.K.; Zalar, P. The extremophiles of Great Salt Lake: Complex microbiology in a dynamic hypersaline ecosystem. In Model Ecosystems in Extreme Environments; Elsevier Inc.: Amsterdam, The Netherlands, 2019; pp. 57–99. [Google Scholar] [CrossRef]
- Bayles, M.; Belasco, B.C.; Breda, A.J.; Cahill, C.B.; Da Silva, A.Z.; Regan, M.J., Jr.; Schlamp, N.K.; Slagle, M.P.; Baxter, B.K. The Haloarchaea of Great Salt Lake as models for potential extant life on Mars. In Extremophiles as Astrobiological Models; Seckbach, J., Stan-Lotter, H., Eds.; Wiley Online Library: Hoboken, NJ, USA, 2020; pp. 83–124. [Google Scholar] [CrossRef]
- Prince, R.C. Eukaryotic Hydrocarbon Degraders. InTaxonomy, Genomics and Ecophysiology of Hydrocarbon-Degrading Microbes. Handbook of Hydrocarbon and Lipid Microbiology; McGenity, T., Ed.; Springer: Cham, Switzerland, 2019; pp. 53–72. [Google Scholar] [CrossRef]
- Prince, R.C.; Walters, C.C. Biodegradation of oil hydrocarbons and its implications for source identification. In Standard Handbook Oil Spill Environmental Forensics; Stout, S.A., Wang, Z., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 869–916. [Google Scholar] [CrossRef]
- Stout, S.A.; Douglas, G.S.; Uhler, A.D. Assessing temporal and spatial variations of gasoline-impacted groundwater using relative mole fractions and PIANO fingerprinting. Environ. Forensics 2010, 11, 328–341. [Google Scholar] [CrossRef]
- Wang, Z.; Stout, S.A.; Fingas, M. Forensic fingerprinting of biomarkers for oil spill characterization and source identification. Environ. Forensics 2006, 7, 105–146. [Google Scholar] [CrossRef]
- National Research Council. In Situ Bioremediation: When Does It Work? National Academies Press: Washington, DC, USA, 1993; Available online: https://nap.nationalacademies.org/catalog/2131/in-situ-bioremediation-when-does-it-work (accessed on 21 July 2022).
- Domagalski, J.L.; Orem, W.H.; Eugster, H.P. Organic geochemistry and brine composition in Great Salt, Mono, and Walker Lakes. Geochim. Cosmochim. Acta 1989, 53, 2857–2872. [Google Scholar] [CrossRef]
- Geng, M.; Duan, Z. Prediction of oxygen solubility in pure water and brines up to high temperatures and pressures. Geochim. Cosmochim. Acta 2010, 74, 5631–5640. [Google Scholar] [CrossRef]
- Burant, A.; Lowry, G.V.; Karamalidis, A.K. Measurement of Setschenow constants for six hydrophobic compounds in simulated brines and use in predictive modeling for oil and gas systems. Chemosphere 2016, 144, 2247–2256. [Google Scholar] [CrossRef]
- Post, F.J. Microbiology of the Great Salt Lake North Arm. Hydrobiologia 1981, 81, 59–69. [Google Scholar] [CrossRef]
- Marcarelli, A.M.; Wurtsbaugh, W.A.; Griset, O. Salinity controls phytoplankton response to nutrient enrichment in the Great Salt Lake, Utah, USA. Can. J. Fish. Aquat. Sci. 2006, 63, 2236–2248. [Google Scholar] [CrossRef]
- ATTC. Microbial Media Formulations. 2019. Available online: https://www.atcc.org/en/Documents/Learning_Center/Microbial_Media_Formulations.aspx (accessed on 21 July 2022).
- Fitzpatrick, M.A.; Munawar, M.; Leach, J.H.; Haffner, G.D. Factors regulating primary production and phytoplankton dynamics in western Lake Erie. Fundam. Appl. Limnol. 2007, 11, 137–152. [Google Scholar] [CrossRef]
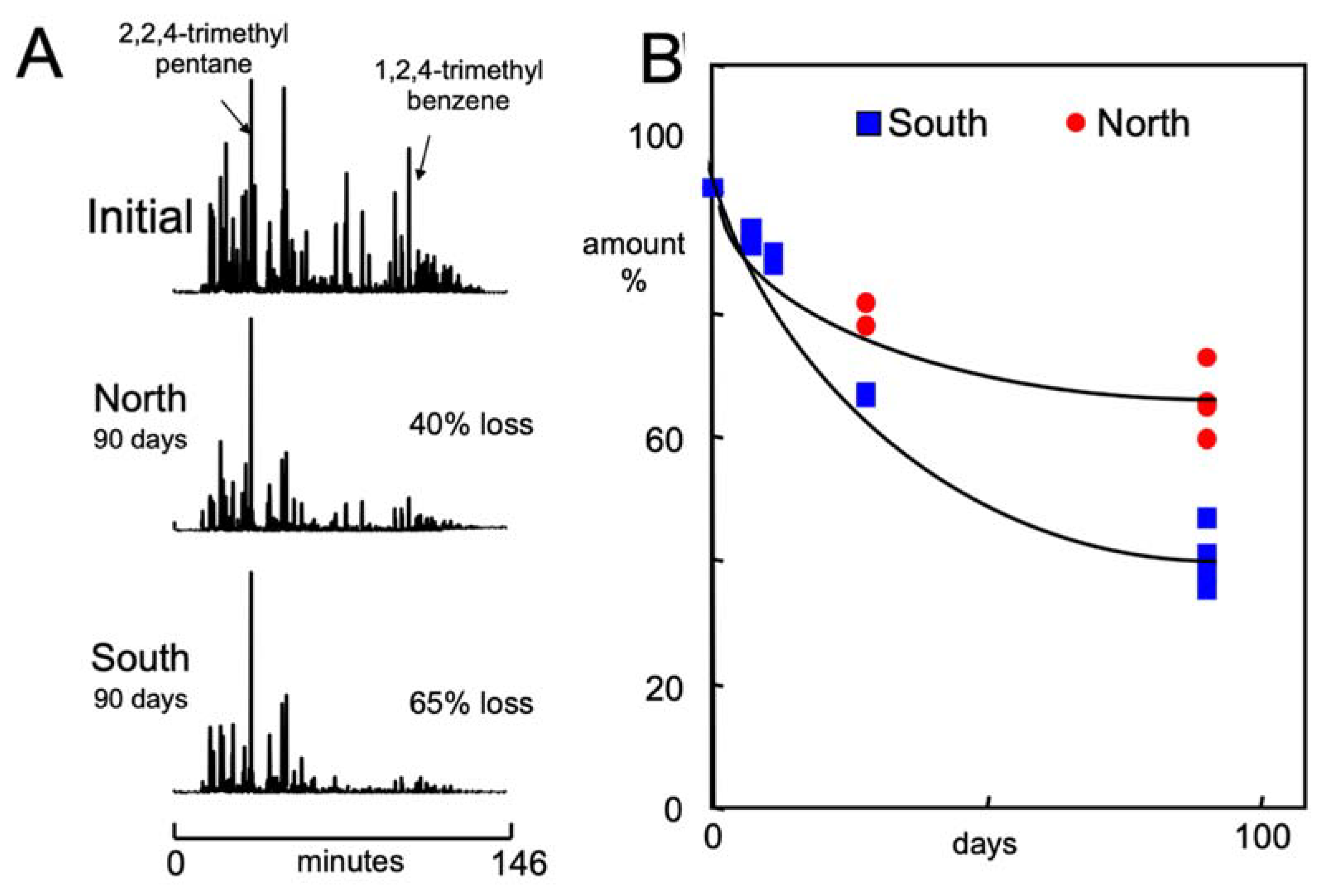

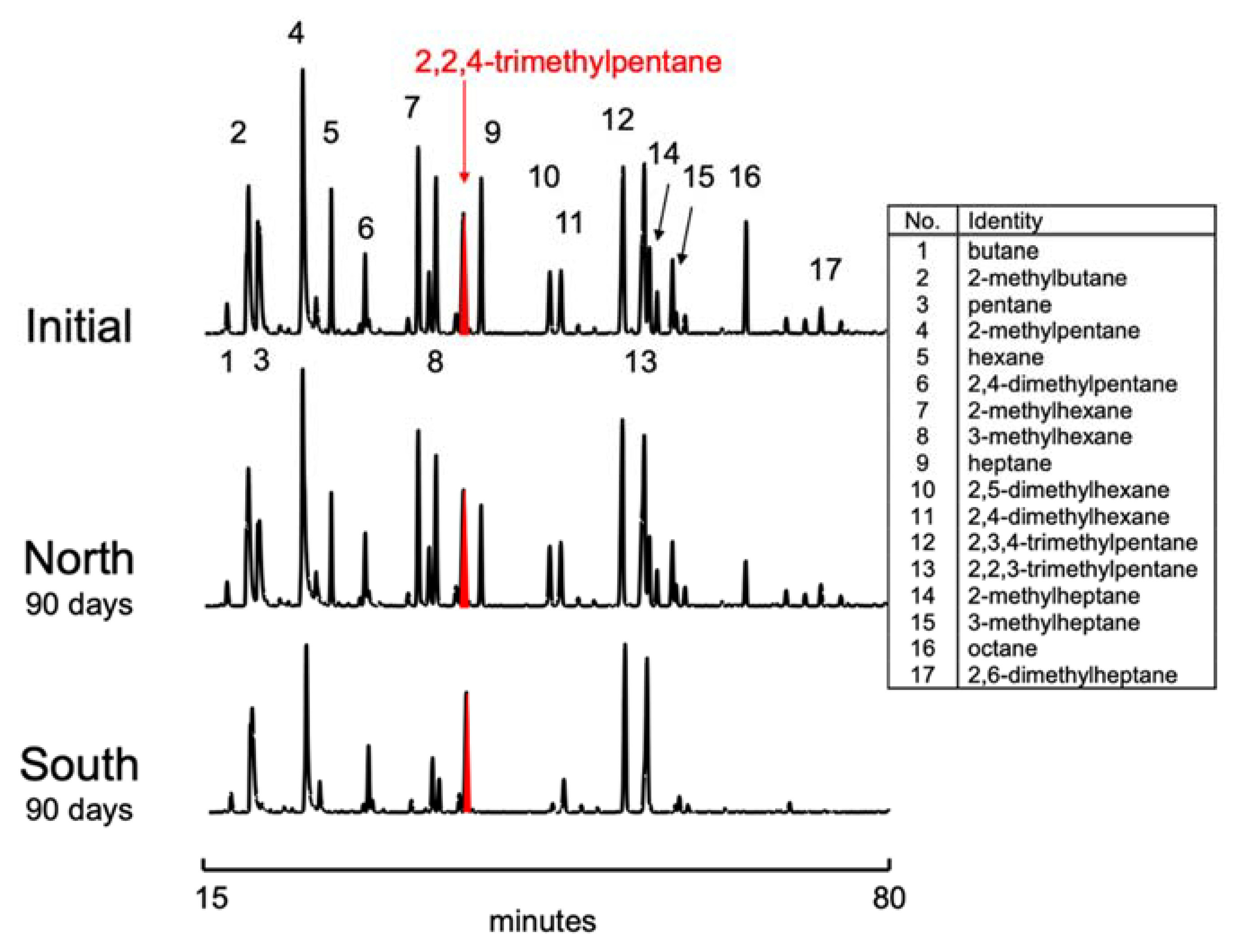
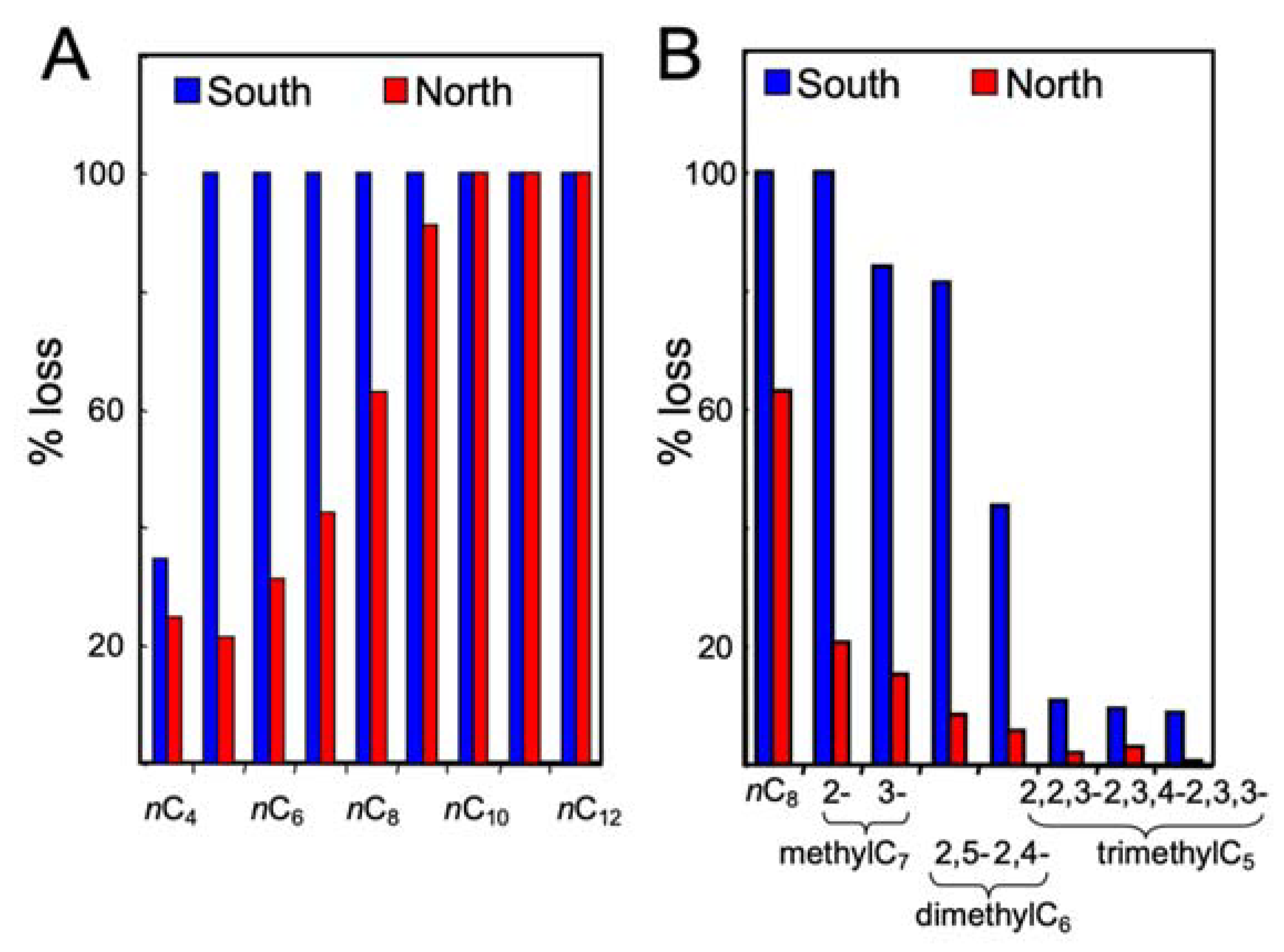
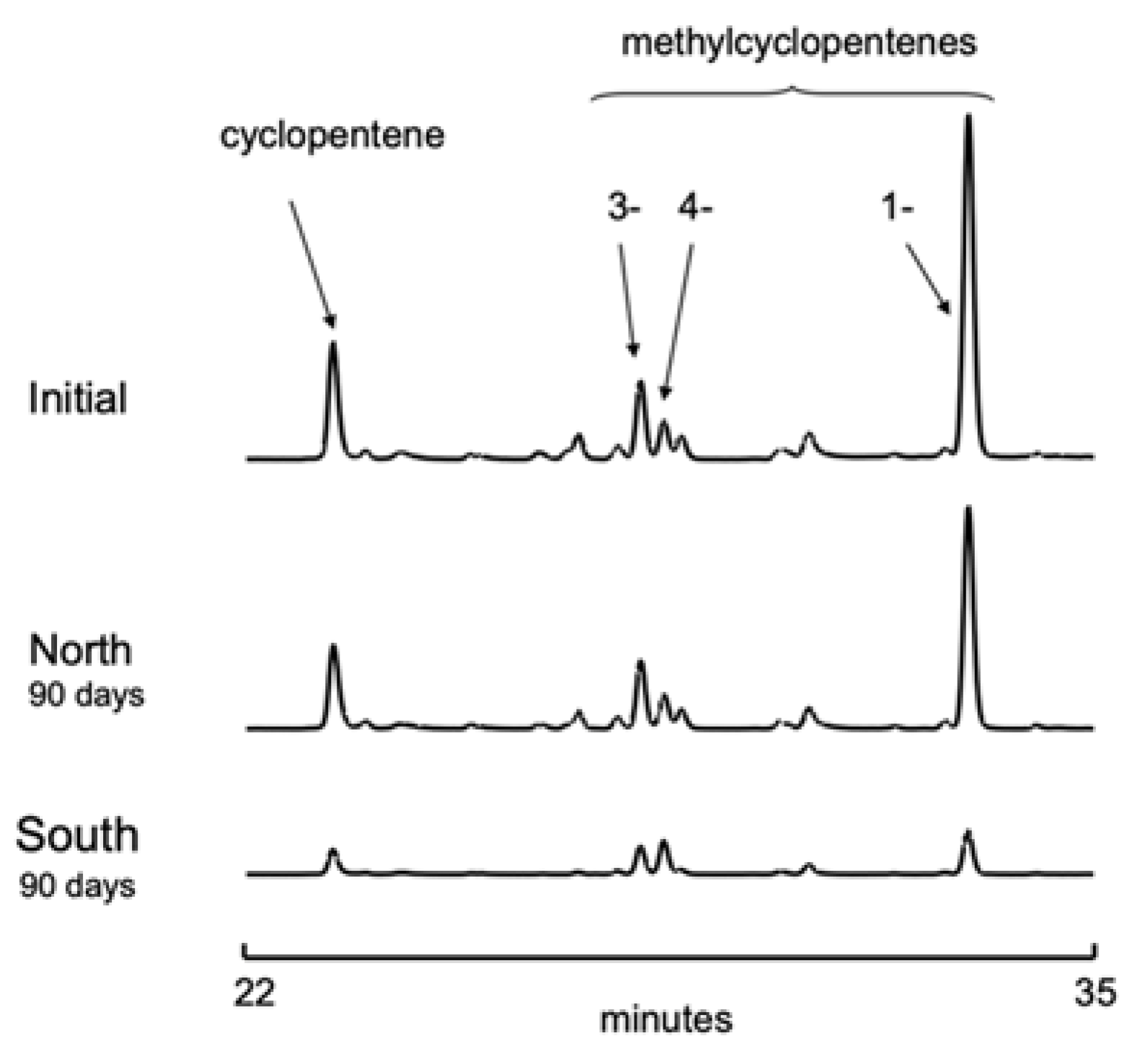

| % Consumed in 90 Days | % Consumed in 90 Days | ||||
|---|---|---|---|---|---|
| South Arm | North Arm | South Arm | North Arm | ||
| Aromatics | Linear and iso-Alkanes | ||||
| benzene | 100 | 60 | butane | 35 | 25 |
| toluene | 100 | 81 | pentane | 100 | 22 |
| ethylbenzene | 100 | 83 | hexane | 100 | 31 |
| p-xylene | 100 | 97 | heptane | 100 | 43 |
| m-xylene | 100 | 90 | octane | 100 | 63 |
| o-xylene | 100 | 74 | nonane | 100 | 91 |
| butyl benzene | 100 | 97 | decane | 100 | 100 |
| propylbenzene | 100 | 87 | undecane | 100 | 100 |
| isopropylbenzene | 100 | 51 | dodecane | 100 | 100 |
| 1-ethyl-2-methylbenzene | 68 | 63 | 2-methylbutane | 9 | 5 |
| 1-ethyl-3-methylbenzene | 100 | 89 | 2-methylpentane | 40 | 14 |
| 1-ethyl-4-methylbenzene | 100 | 97 | 3-methylpentane | 37 | 11 |
| 1,2,3-trimethylbenzene | 100 | 75 | 2-methylhexane | 95 | 15 |
| 1,2,4-trimethylbenzene | 100 | 91 | 3-methylhexane | 79 | 13 |
| 1,3,5-trimethylbenzene | 73 | 70 | 2-methylheptane | 100 | 21 |
| (1-methylpropyl) benzene | 87 | 47 | 3-methylheptane | 94 | 15 |
| (2-methylpropyl) benzene | 100 | 56 | 3-methyloctane | 100 | 37 |
| 1,2-diethylbenzene | 74 | 69 | 2-methylnonane | 100 | 84 |
| 1,4-diethylbenzene | 97 | 80 | 3-methylnonane | 100 | 74 |
| 1-methyl-2-propylbenzene | 70 | 69 | 4-methylnonane | 100 | 62 |
| 1-methyl-3-propylbenzene | 99 | 91 | 5-methylnonane | 99 | 34 |
| 1-methyl-4-propylbenzene | 100 | 97 | 2,2-dimethylpentane | 6 | 14 |
| 1-methyl-2(1-methylethyl) benzene | 48 | 44 | 2,3-dimethylpentane | 19 | 12 |
| 1-methyl-3(1-methylethyl) benzene | 77 | 66 | 2,3-dimethylpentane | 14 | 9 |
| 1-methyl-4(1-methylethyl) benzene | 100 | 77 | 3,3-dimethylpentane | 23 | 18 |
| 1-ethyl-3,5-dimethylbenzene | 68 | 67 | 2,4-dimethylpentane | 37 | 14 |
| 2-ethyl-1,3-dimethylbenzene | 62 | 61 | 2,4-dimethylhexane | 44 | 6 |
| 1,2-dimethyl-4-ethylbenzene | 89 | 85 | 2,5 dimethylhexane | 81 | 9 |
| 1,4-dimethyl-2-ethylbenzene | 98 | 79 | 2,5-dimethylheptane | 100 | 18 |
| 2,4-dimethyl-1-ethylbenzene | 97 | 86 | 2,6-dimethylheptane | 85 | 21 |
| 1,2,3,4-tetramethylbenzene | 74 | 79 | 3,5-dimethylheptane | 100 | 18 |
| 1,2,3,5-tetramethylbenzene | 75 | 77 | 2,5-dimethyloctane | 99 | 38 |
| 1,2,4,5-tetramethylbenzene | 81 | 83 | 2,6-dimethyloctane | 99 | 42 |
| naphthalene | 100 | 94 | 2,2,3-trimethylpentane | 11 | 2 |
| 1-methylnaphthalene | 97 | 77 | 2,3,3-trimethylpentane | 9 | 1 |
| 2-methylnaphthalene | 100 | 97 | 2,3,4-trimethylpentane | 10 | 3 |
| tetralin | 77 | 76 | Cyclic Alkanes | ||
| indan | 97 | 96 | methylcyclopentane | 20 | 4 |
| 1-methylindan | 93 | 92 | cis1,3-dimethylcyclopentane | 29 | 11 |
| 4-methylindan | 99 | 99 | trans1,3-dimethylcyclopentane | 9 | 4 |
| 5-methylindan | 91 | 96 | cyclohexane | 9 | 2 |
| 1,2,3,4-tetramethylbenzene | 74 | 79 | methylcyclohexane | 17 | 0 |
| 1,2,3,5-tetramethylbenzene | 75 | 77 | ethylcyclohexane | 76 | 9 |
| 1,2,4,5-tetramethylbenzene | 81 | 83 | 1,1-dimethylcyclohexane | 0 | 0 |
| naphthalene | 100 | 94 | cis1,2-dimethylcyclohexane | 11 | 2 |
| 1-methylnaphthalene | 97 | 77 | trans1,2-dimethylcyclohexane | 6 | 0 |
| 2-methylnaphthalene | 100 | 97 | cis1,3-dimethylcyclohexane | 7 | 0 |
| tetralin | 77 | 76 | trans1,3-dimethylcyclohexane | 11 | 0 |
| indan | 97 | 96 | propylcyclohexane | 100 | 34 |
| 1-methylindan | 93 | 92 | butylcyclohexane | 100 | 49 |
| 4-methylindan | 99 | 99 | Linear and iso-Alkenes | ||
| 5-methylindan | 91 | 96 | 2-methyl-1-butene | 10 | 12 |
| 2-methyl-2-butene | 40 | 26 | |||
| Cyclic Alkenes | cis2-pentene | 95 | 44 | ||
| cyclopentene | 52 | 22 | trans2-pentene | 84 | 33 |
| 1-methylcyclopentene | 71 | 34 | 3-methyl-2-pentene | 15 | 13 |
| 3-methylcyclopentene | 16 | 9 | cis2-hexene | 69 | 25 |
| 4-methylcyclopentene | 14 | 7 | trans2-hexene | 100 | 37 |
| 1-ethylcyclopentene | 81 | 12 | cis3-hexene | 90 | 41 |
| % Consumed in 66 Days | % Consumed in 66 Days | ||||
|---|---|---|---|---|---|
| South Arm | North Arm | South Arm | North Arm | ||
| Aromatics | Linear Alkanes | ||||
| naphthalene | 100 | 100 | tetradecane | 100 | 100 |
| C1-naphthalenes | 100 | 100 | pentadecane | 100 | 86 |
| C2-naphthalenes | 100 | 90 | hexadecane | 100 | 86 |
| C3-naphthalenes | 100 | 70 | heptadecane | 100 | 85 |
| C4-naphthalenes | 100 | 50 | octadecane | 100 | 75 |
| fluorene | 100 | 25 | nonadecane | 100 | 66 |
| 1-methylfluorene | 60 | 0 | icosane | 100 | 57 |
| 2/3-methylfluorene | 80 | 0 | henicosane | 100 | 5 |
| 4-methylfluorene | 80 | 0 | docosane | 99 | 5 |
| 9-methylfluorene | 80 | 0 | tricosane | 95 | 2 |
| C2-fluorenes | 40 | 0 | tetracosane | 90 | 1 |
| C3-fluorenes | 25 | 0 | pentacosane | 86 | 2 |
| phenanthrene | 30 | 10 | hexacosane | 83 | 0 |
| 1-methylphenanthrene | 15 | 7 | heptacosane | 81 | 0 |
| 2-methylphenanthrene | 15 | 7 | octacosane | 80 | 0 |
| 3-methylphenanthrene | 20 | 10 | nonacosane | 79 | 2 |
| 9-methylphenanthrene | 20 | 12 | triacontane | 79 | 1 |
| C2-phenanthrenes | 0 | 0 | hentriacontane | 78 | 0 |
| C3-phenanthrenes | 0 | 0 | dotriacontane | 76 | 3 |
| C4-phenanthrenes | 0 | 0 | tritriacontane | 66 | 0 |
| benz[a]anthracene | 20 | 10 | tetratriacontane | 34 | 1 |
| chrysene | 10 | 0 | |||
| 1-methylchrysene | 5 | 0 | iso-Alkanes | ||
| 2-methylchrysene | 5 | 0 | pristane | 99 | 67 |
| 3-methylchrysene | 0 | 0 | phytane | 95 | 63 |
| 4/6-methylchrysene | 5 | 0 | |||
| C2-chrysenes | 0 | 0 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prince, R.C.; Prince, V.L. Hydrocarbon Biodegradation in Utah’s Great Salt Lake. Water 2022, 14, 2661. https://doi.org/10.3390/w14172661
Prince RC, Prince VL. Hydrocarbon Biodegradation in Utah’s Great Salt Lake. Water. 2022; 14(17):2661. https://doi.org/10.3390/w14172661
Chicago/Turabian StylePrince, Roger C., and Victoria L. Prince. 2022. "Hydrocarbon Biodegradation in Utah’s Great Salt Lake" Water 14, no. 17: 2661. https://doi.org/10.3390/w14172661
APA StylePrince, R. C., & Prince, V. L. (2022). Hydrocarbon Biodegradation in Utah’s Great Salt Lake. Water, 14(17), 2661. https://doi.org/10.3390/w14172661








