Effects of Environmentally Relevant Concentrations of Antipsychotic Drugs (Sulpiride and Clozapine) on Serotonergic and Dopaminergic Neurotransmitter Systems in Octopus Brain Tissue
Abstract
:1. Introduction
2. Materials and Methods
2.1. Drugs
2.2. Experimental Animals and Disposal Methods
2.3. RNA Extraction and qPCR
2.4. Western Blot Analysis
2.5. Statistical Analysis
3. Results
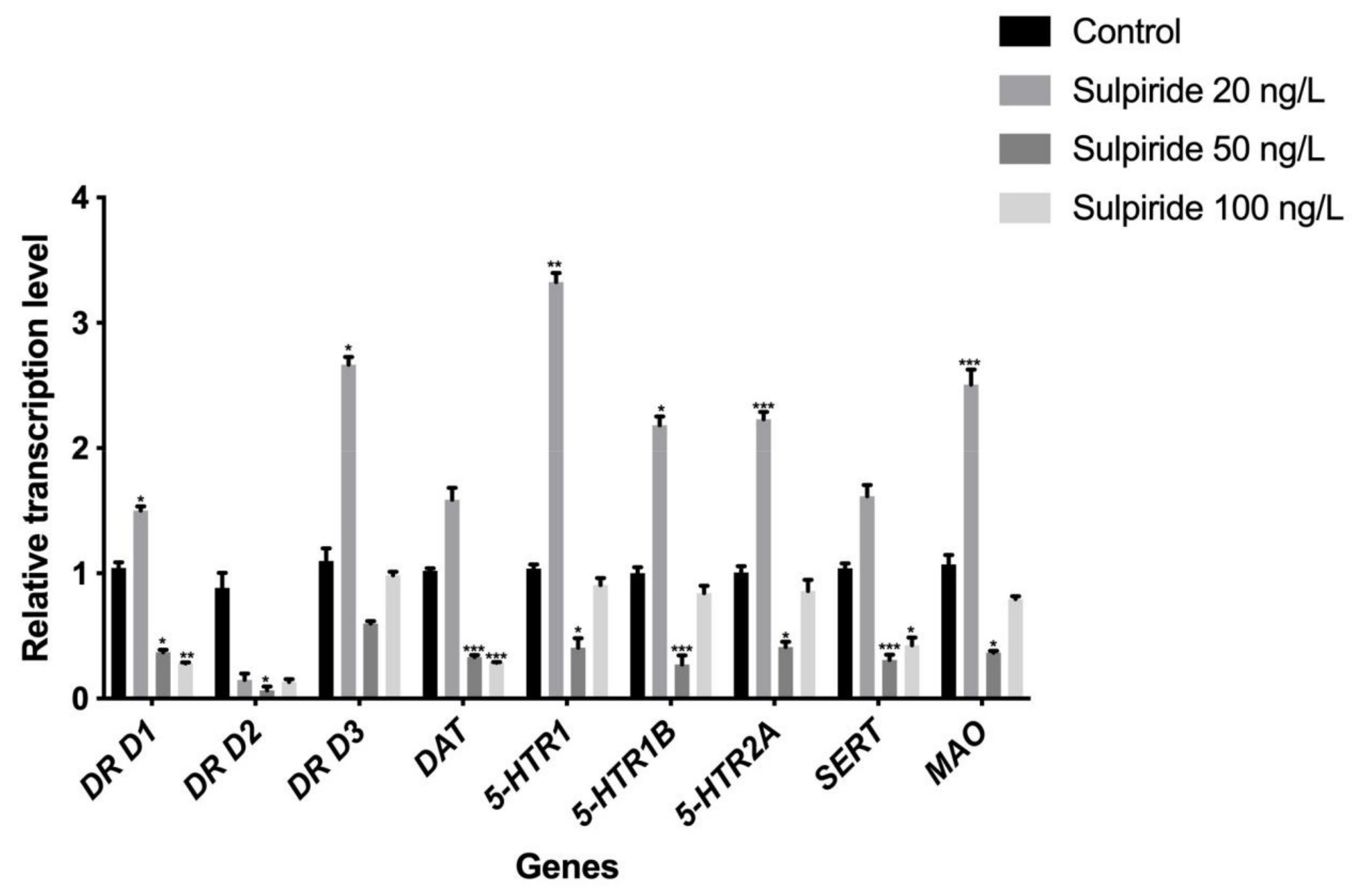
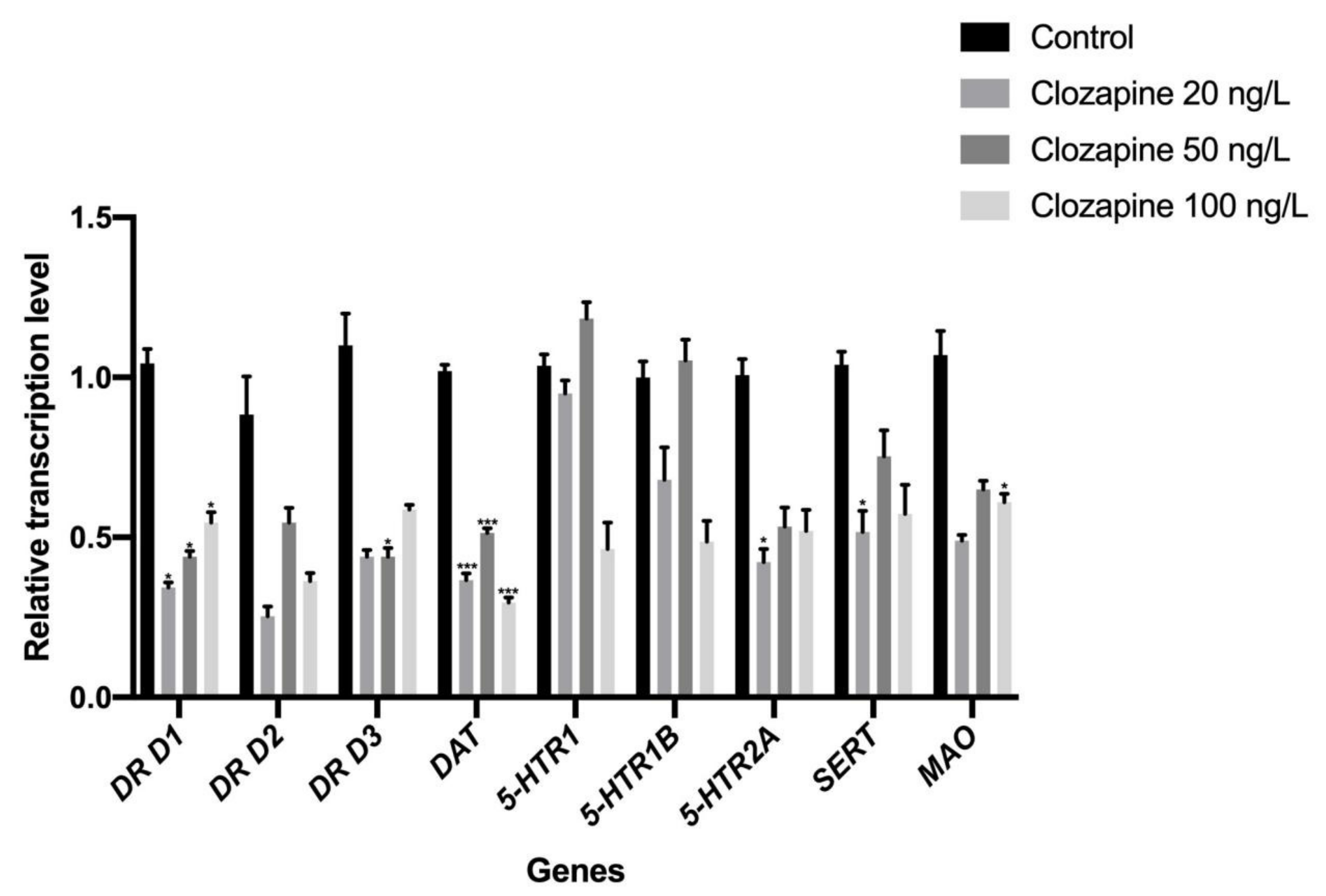
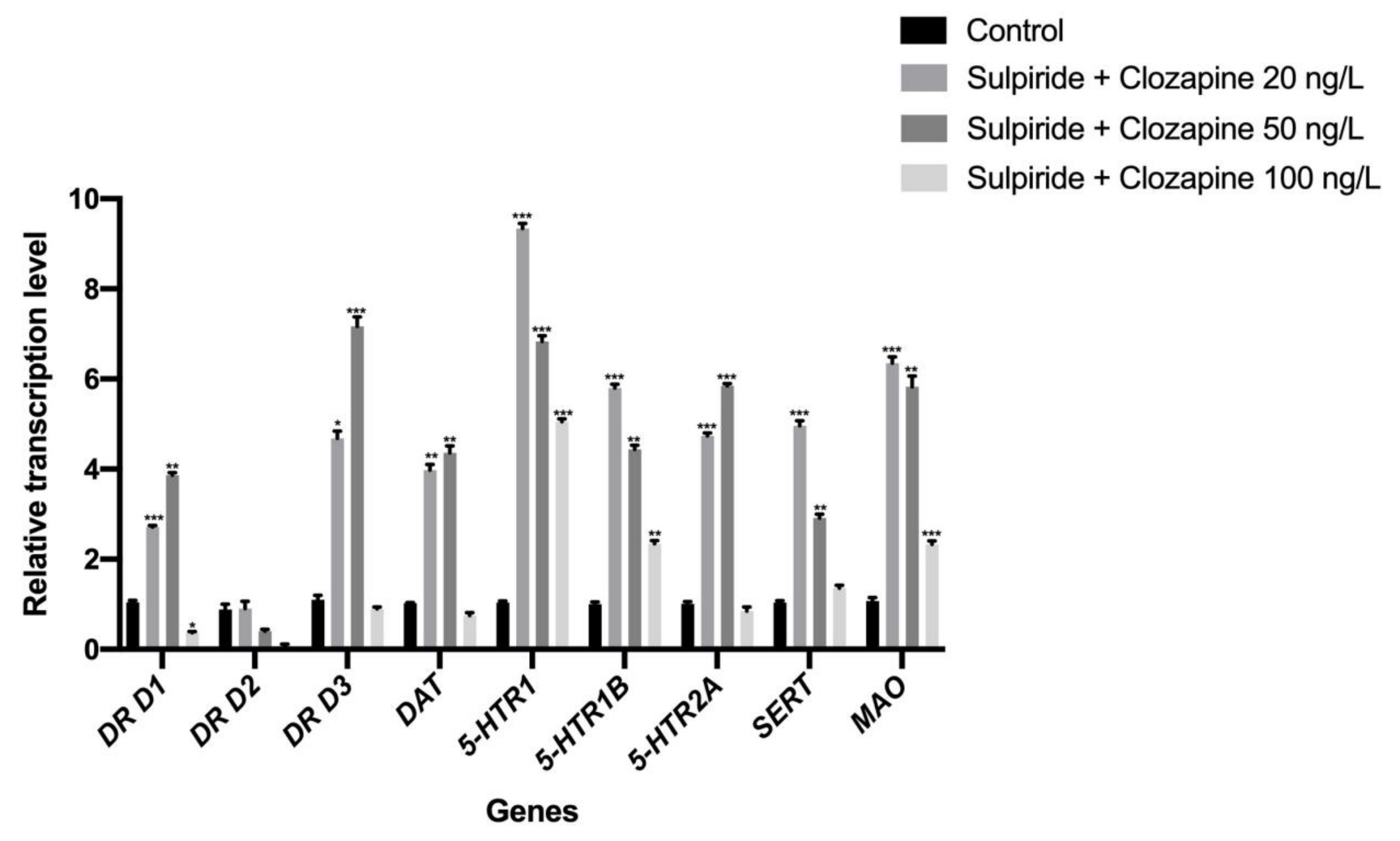
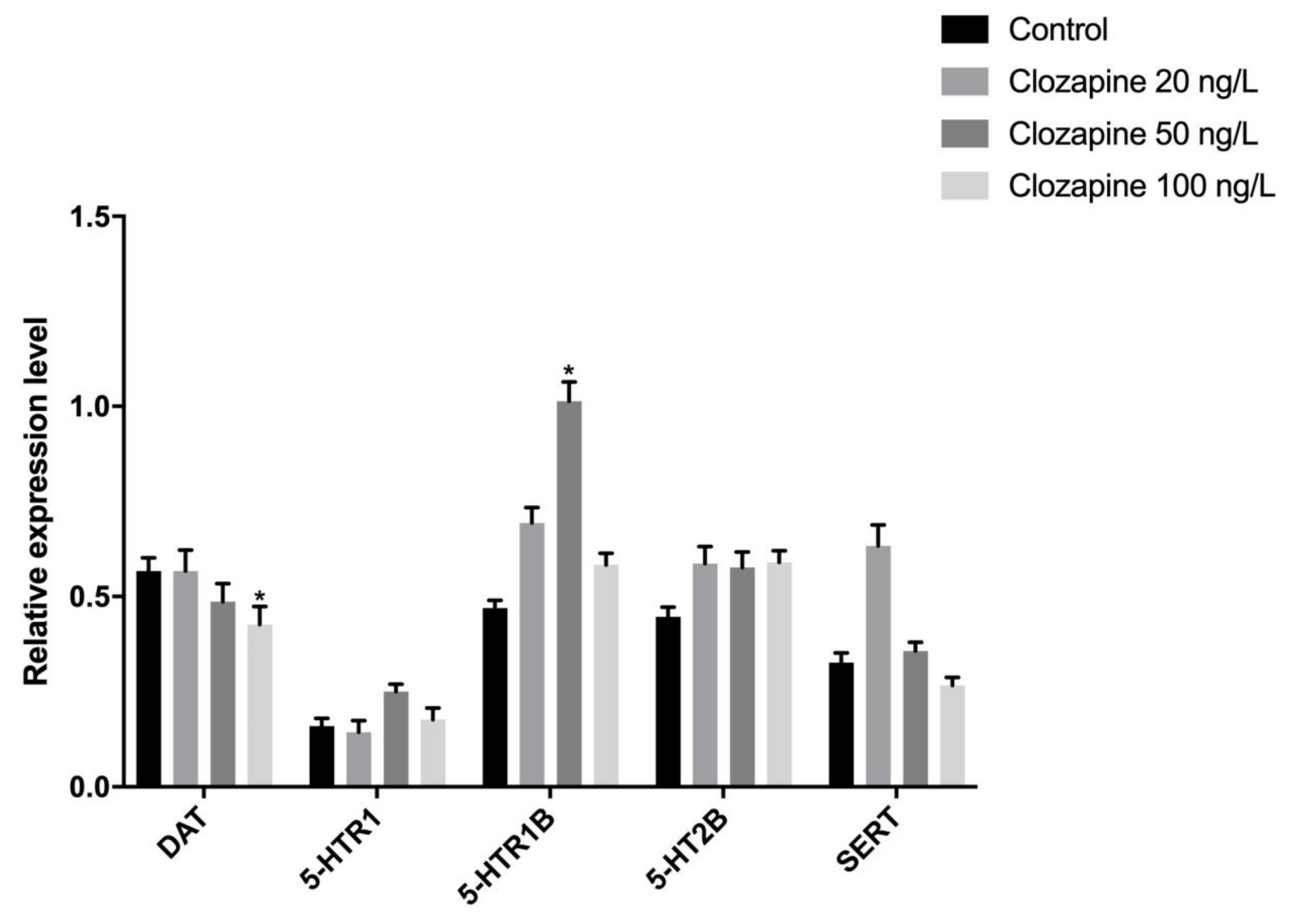
3.1. Effects of Sulpiride and Clozapine on the Transcription of Functional Molecules of DA and 5-HT Neurotransmitter Systems in Octopus Brain Tissue
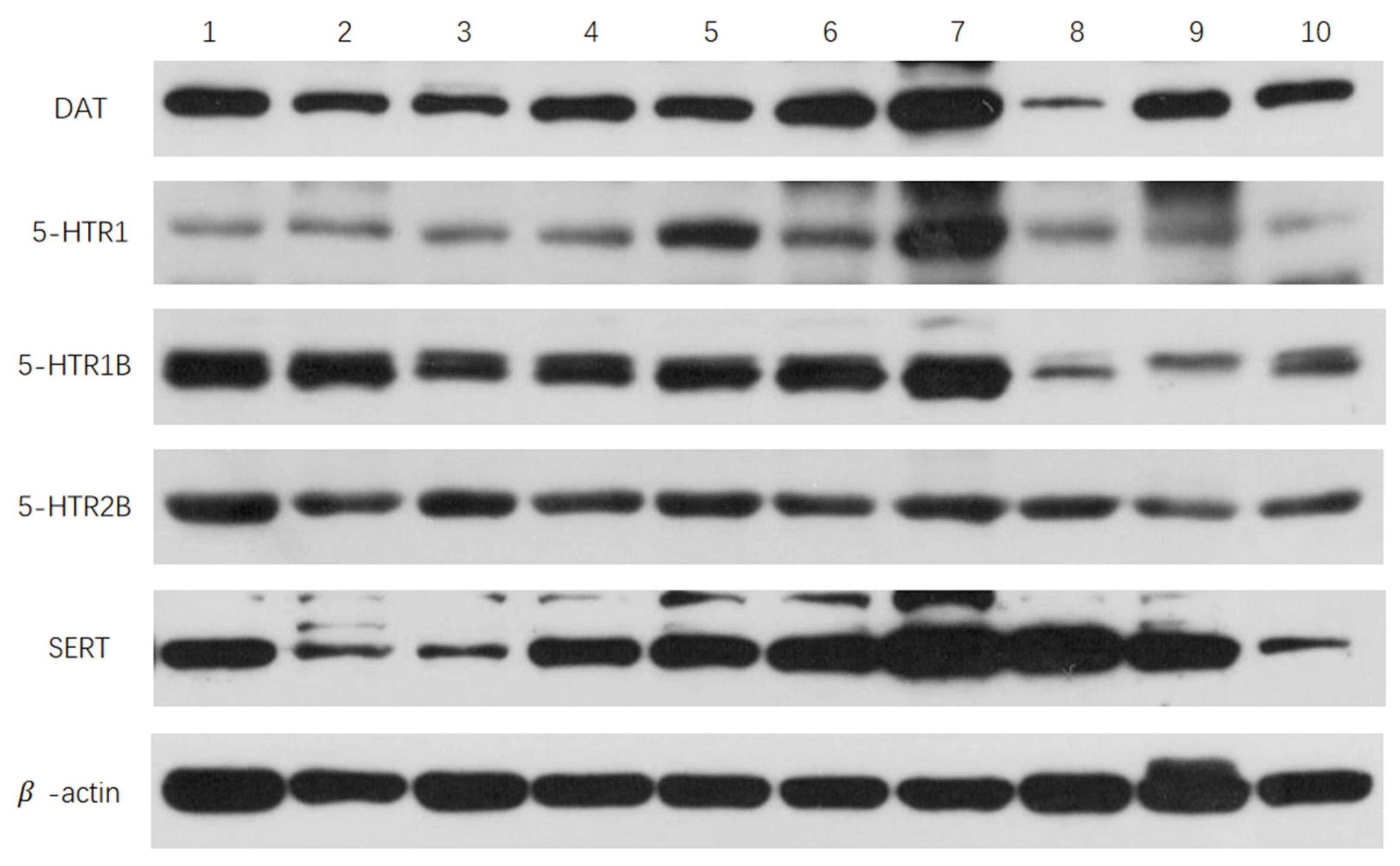
3.2. Effects of Sulpiride and Clozapine on Protein Expression Levels of Functional Molecules of DA and 5-HT Neurotransmitter System in Octopus Brain Tissue
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chaves, M.; Barbosa, S.C.; Malinowski, M.M.; Volpato, D.; Castro, I.B.; Franco, T.; Primel, E.G. Pharmaceuticals and personal care products in a Brazilian wetland of international importance: Occurrence and environmental risk assessment. Sci. Total. Environ. 2020, 734, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Freitas, L.; Radis-Baptista, G. Pharmaceutical Pollution and Disposal of Expired, Unused, and Unwanted Medicines in the Brazilian Context. J. Xenobiot. 2021, 11, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Pompei, C.; Campos, L.C.; Da, S.B.; Fogo, J.C.; Vieira, E.M. Occurrence of PPCPs in a Brazilian water reservoir and their removal efficiency by ecological filtration. Chemosphere 2019, 226, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Roveri, V.; Guimaraes, L.L.; Toma, W.; Correia, A.T. Occurrence and ecological risk assessment of pharmaceuticals and cocaine in the urban drainage channels of Santos beaches (Sao Paulo, Brazil): A neglected, but sensitive issue. Environ. Sci. Pollut. Res. Int. 2021, 28, 65595–65609. [Google Scholar] [CrossRef]
- Santos, L.H.; Araujo, A.N.; Fachini, A.; Pena, A.; Delerue-Matos, C.; Montenegro, M.C. Ecotoxicological aspects related to the presence of pharmaceuticals in the aquatic environment. J. Hazard. Mater. 2010, 175, 45–95. [Google Scholar] [CrossRef]
- Volk, D.W.; Jenkins, A.K.; Lidstone, P.A.; Lewis, D.A. Rosenberg’s Molecular and Genetic Basis of Neurological and Psychiatric Disease, 6th ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 653–662. [Google Scholar]
- Seeman, P. Dopamine D2 receptors as treatment targets in schizophrenia. Clin. Schizophr. Relat. Psychoses 2010, 4, 56–73. [Google Scholar] [CrossRef]
- Arranz, M.J.; Aitchison, K.J.; Kerwin, R.; Collier, D.A. Analyses of clozapine response and the 5-HT2 gene. Schizophr. Res. 1995, 15, 35. [Google Scholar] [CrossRef]
- Gros, M.; Petrovic, M.; Barcelo, D. Wastewater treatment plants as a pathway for aquatic contamination by pharmaceuticals in the ebro river basin (northeast Spain). Environ. Toxicol. Chem. 2007, 26, 1553–1562. [Google Scholar] [CrossRef]
- Lei, H.J.; Yang, B.; Ye, P.; Yang, Y.Y.; Zhao, J.L.; Liu, Y.S.; Xie, L.; Ying, G.G. Occurrence, fate and mass loading of benzodiazepines and their transformation products in eleven wastewater treatment plants in Guangdong province, China. Sci. Total. Environ. 2021, 755, 142648. [Google Scholar] [CrossRef]
- Bollmann, A.F.; Seitz, W.; Prasse, C.; Lucke, T.; Schulz, W.; Ternes, T. Occurrence and fate of amisulpride, sulpiride, and lamotrigine in municipal wastewater treatment plants with biological treatment and ozonation. J. Hazard. Mater. 2016, 320, 204–215. [Google Scholar] [CrossRef]
- Yuan, S.; Jiang, X.; Xia, X.; Zhang, H.; Zheng, S. Detection, occurrence and fate of 22 psychiatric pharmaceuticals in psychiatric hospital and municipal wastewater treatment plants in Beijing, China. Chemosphere 2013, 90, 2520–2525. [Google Scholar] [CrossRef]
- González, Á.F.; Pierce, G.J. Advances in the study of cephalopod fisheries and ecosystems. Fish. Res. 2021, 242, 105975. [Google Scholar] [CrossRef]
- Wells, M. Octopus: Physiology and Behaviour of an Advanced Invertebrate; Chapman and Hall: London, UK, 1978. [Google Scholar]
- Young, J. The Anatomy of the Nervous System of Octopus Vulgaris; Clarendon Press: Oxford, UK, 1971. [Google Scholar]
- Shomrat, T.; Zarrella, I.; Fiorito, G.; Hochner, B. The octopus vertical lobe modulates short-term learning rate and uses LTP to acquire long-term memory. Curr. Biol. 2008, 18, 337–342. [Google Scholar] [CrossRef]
- Di Poi, C.; Darmaillacq, A.S.; Dickel, L.; Boulouard, M.; Bellanger, C. Effects of perinatal exposure to waterborne fluoxetine on memory processing in the cuttlefish Sepia officinalis. Aquat. Toxicol. 2013, 132–133, 84–91. [Google Scholar] [CrossRef]
- Messenger, J.B. Review Neurotransmitters of cephalopods. Invertebr. Neurosci. 1996, 2, 95–114. [Google Scholar] [CrossRef]
- Juorio, A.V. Catecholamines and 5-hydroxytryptamine in nervous tissue of cephalopods. J. Physiol. 1971, 216, 213–226. [Google Scholar] [CrossRef]
- Juorio, A.V.; Molinoff, P.B. The normal occurrence of octopamine in neural tissue of the Octopus and other cephalopods. J. Neurochem. 1974, 22, 271–280. [Google Scholar] [CrossRef]
- Lin, R.; Liang, J.; Luo, M. The Raphe Dopamine System: Roles in Salience Encoding, Memory Expression, and Addiction. Trends Neurosci. 2021, 44, 366–377. [Google Scholar] [CrossRef]
- Ott, T.; Nieder, A. Dopamine and Cognitive Control in Prefrontal Cortex. Trends Cogn. Sci. 2019, 23, 213–234. [Google Scholar] [CrossRef]
- Andrews, P.L.R.; Messenger, J.B.; Tansey, E.M. The chromatic and motor effects of neurotransmitter injection in intact and brain-lesioned Octopus. J. Mar. Biol. Assoc. UK 1983, 63, 355–370. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, L.; Yan, Y.; Zheng, Y.; Ge, W.; Li, M.; Wang, W.; Song, X.; Song, L. D1 dopamine receptor is involved in shell formation in larvae of Pacific oyster Crassostrea gigas. Dev. Comp. Immunol. 2018, 84, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Hochner, B.; Shomrat, T.; Fiorito, G. The octopus: A model for a comparative analysis of the evolution of learning and memory mechanisms. Biol. Bull. 2006, 210, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Shomrat, T.; Feinstein, N.; Klein, M.; Hochner, B. Serotonin is a facilitatory neuromodulator of synaptic transmission and "reinforces" long-term potentiation induction in the vertical lobe of Octopus vulgaris. Neuroscience 2010, 169, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Courtiol, E.; Menezes, E.C.; Teixeira, C.M. Serotonergic regulation of the dopaminergic system: Implications for reward-related functions. Neurosci. Biobehav. Rev. 2021, 128, 282–293. [Google Scholar] [CrossRef] [PubMed]
- Herculano, A.M.; Maximino, C. Serotonergic modulation of zebrafish behavior: Towards a paradox. Prog. Neuropsychopharmacol. Biol. Psychiatry 2014, 55, 50–66. [Google Scholar] [CrossRef] [PubMed]
- Nowicki, M.; Tran, S.; Muraleetharan, A.; Markovic, S.; Gerlai, R. Serotonin antagonists induce anxiolytic and anxiogenic-like behavior in zebrafish in a receptor-subtype dependent manner. Pharm. Biochem. Behav. 2014, 126, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Rosen, S.C.; Weiss, K.R.; Goldstein, R.S.; Kupfermann, I. The role of a modulatory neuron in feeding and satiation in Aplysia: Effects of lesioning of the serotonergic metacerebral cells. J. Neurosci. 1989, 9, 1562–1578. [Google Scholar] [CrossRef]
- Mackey, S.; Carew, T.J. Locomotion in Aplysia: Triggering by serotonin and modulation by bag cell extract. J. Neurosci. 1983, 3, 1469–1477. [Google Scholar] [CrossRef]
- Glanzman, D.L.; Mackey, S.L.; Hawkins, R.D.; Dyke, A.M.; Lloyd, P.E.; Kandel, E.R. Depletion of serotonin in the nervous system of Aplysia reduces the behavioral enhancement of gill withdrawal as well as the heterosynaptic facilitation produced by tail shock. J. Neurosci. 1989, 9, 4200–4213. [Google Scholar] [CrossRef]
- Wang, Q.; He, M. Molecular characterization and analysis of a putative 5-HT receptor involved in reproduction process of the pearl oyster Pinctada fucata. Gen. Comp. Endocrinol. 2014, 204, 71–79. [Google Scholar] [CrossRef]
- Liu, Y.; Ji, M.; Wang, R.; Zhai, H.; Yu, S.; Liu, B.; Zhao, Y. Spatial distribution and comprehensive evaluation of emerging organic pollutants in effluents from wastewater treatment plants in northern cities of China. Desalin. Water Treat. 2019, 156, 20–31. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, X.H.; Huang, Y.; Wang, H. Comprehensive evaluation of pharmaceuticals and personal care products (PPCPs) in typical highly urbanized regions across China. Environ. Pollut. 2015, 204, 223–232. [Google Scholar] [CrossRef]
- He, Y.; Chen, W.; Zheng, X.; Wang, X.; Huang, X. Fate and removal of typical pharmaceuticals and personal care products by three different treatment processes. Sci. Total. Environ. 2013, 447, 248–254. [Google Scholar] [CrossRef]
- Di Paola, D.; Capparucci, F.; Abbate, J.M.; Cordaro, M.; Crupi, R.; Siracusa, R.; D′Amico, R.; Fusco, R.; Genovese, T.; Impellizzeri, D.; et al. Environmental Risk Assessment of Oxaliplatin Exposure on Early Life Stages of Zebrafish (Danio rerio). Toxics 2022, 10, 81. [Google Scholar] [CrossRef]
- Andrews, P.L.R.; Darmaillacq, A.; Dennison, N.; Gleadall, I.G.; Hawkins, P.; Messenger, J.B.; Osorio, D.; Smith, V.J.; Smith, J.A. The identification and management of pain, suffering and distress in cephalopods, including anaesthesia, analgesia and humane killing. J. Exp. Mar. Biol. Ecol. 2013, 447, 46–64. [Google Scholar] [CrossRef]
- Messenger, J.B.; Nixon, M.; Ryan, K.P. Magnesium chloride as an anaesthetic for cephalopods. Comp. Biochem. Physiol. C Comp. Pharm. Toxicol. 1985, 82, 203–205. [Google Scholar] [CrossRef]
- Di Paola, D.; Capparucci, F.; Lanteri, G.; Crupi, R.; Marino, Y.; Franco, G.A.; Cuzzocrea, S.; Spano, N.; Gugliandolo, E.; Peritore, A.F. Environmental Toxicity Assessment of Sodium Fluoride and Platinum-Derived Drugs Co-Exposure on Aquatic Organisms. Toxics 2022, 10, 272. [Google Scholar] [CrossRef]
- Di Paola, D.; Natale, S.; Gugliandolo, E.; Cordaro, M.; Crupi, R.; Siracusa, R.; D Amico, R.; Fusco, R.; Impellizzeri, D.; Cuzzocrea, S.; et al. Assessment of 2-Pentadecyl-2-oxazoline Role on Lipopolysaccharide-Induced Inflammation on Early Stage Development of Zebrafish (Danio rerio). Life 2022, 12, 128. [Google Scholar] [CrossRef]
- Aydin, S.; Beduk, F.; Ulvi, A.; Aydin, M.E. Simple and effective removal of psychiatric pharmaceuticals from wastewater treatment plant effluents by magnetite red mud nanoparticles. Sci. Total. Environ. 2021, 784, 147174. [Google Scholar] [CrossRef]
- Lo, Y.F.; Yang, F.C.; Huang, J.S.; Lin, Y.S.; Liang, C.S. Disentangling the complex bidirectional associations between COVID-19 and psychiatric disorder. Lancet Psychiat. 2021, 8, 179. [Google Scholar] [CrossRef]
- Putri, C.; Arisa, J.; Hananto, J.E.; Hariyanto, T.I.; Kurniawan, A. Psychiatric sequelae in COVID-19 survivors: A narrative review. World J. Psychiatry 2021, 11, 821–829. [Google Scholar] [CrossRef]
- Taquet, M.; Geddes, J.R.; Husain, M.; Luciano, S.; Harrison, P.J. 6-month neurological and psychiatric outcomes in 236 379 survivors of COVID-19: A retrospective cohort study using electronic health records. Lancet Psychiat. 2021, 8, 416–427. [Google Scholar] [CrossRef]
- Guruge, K.S.; Goswami, P.; Tanoue, R.; Nomiyama, K.; Wijesekara, R.; Dharmaratne, T.S. First nationwide investigation and environmental risk assessment of 72 pharmaceuticals and personal care products from Sri Lankan surface waterways. Sci. Total. Environ. 2019, 690, 683–695. [Google Scholar] [CrossRef]
- Singh, K.P.; Rai, P.; Singh, A.K.; Verma, P.; Gupta, S. Occurrence of pharmaceuticals in urban wastewater of north Indian cities and risk assessment. Environ. Monit. Assess. 2014, 186, 6663–6682. [Google Scholar] [CrossRef]
- Reis-Santos, P.; Pais, M.; Duarte, B.; Cacador, I.; Freitas, A.; Vila, P.A.; Barbosa, J.; Leston, S.; Rosa, J.; Ramos, F.; et al. Screening of human and veterinary pharmaceuticals in estuarine waters: A baseline assessment for the Tejo estuary. Mar. Pollut. Bull. 2018, 135, 1079–1084. [Google Scholar] [CrossRef]
- Seeman, P.; Van Tol, H.H. Dopamine receptor pharmacology. Curr. Opin. Neurol. Neurosurg. 1993, 6, 602–608. [Google Scholar] [CrossRef]
- Meltzer, H.Y. Relevance of dopamine autoreceptors for psychiatry: Preclinical and clinical studies. Schizophr. Bull. 1980, 6, 456–475. [Google Scholar] [CrossRef]
- Boothman, L.J.; Allers, K.A.; Rasmussen, K.; Sharp, T. Evidence that central 5-HT2A and 5-HT2B/C receptors regulate 5-HT cell firing in the dorsal raphe nucleus of the anaesthetised rat. Br. J. Pharm. 2003, 139, 998–1004. [Google Scholar] [CrossRef]
- Sharp, T.; Barnes, N.M. Central 5-HT receptors and their function; present and future. Neuropharmacology 2020, 177, 108155. [Google Scholar] [CrossRef]
- De Deurwaerdere, P.; Di Giovanni, G. Serotonergic modulation of the activity of mesencephalic dopaminergic systems: Therapeutic implications. Prog. Neurobiol. 2017, 151, 175–236. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Colborn, T.; Hayes, T.B.; Heindel, J.J.; Jacobs, D.J.; Lee, D.H.; Shioda, T.; Soto, A.M.; Vom, S.F.; Welshons, W.V.; et al. Hormones and endocrine-disrupting chemicals: Low-dose effects and nonmonotonic dose responses. Endocr. Rev. 2012, 33, 378–455. [Google Scholar] [CrossRef] [PubMed]
- Brodin, T.; Fick, J.; Jonsson, M.; Klaminder, J. Dilute concentrations of a psychiatric drug alter behavior of fish from natural populations. Science 2013, 339, 814–815. [Google Scholar] [CrossRef] [PubMed]
- David, A.; Lange, A.; Tyler, C.R.; Hill, E.M. Concentrating mixtures of neuroactive pharmaceuticals and altered neurotransmitter levels in the brain of fish exposed to a wastewater effluent. Sci. Total. Environ. 2018, 621, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Huerta, B.; Margiotta-Casaluci, L.; Rodriguez-Mozaz, S.; Scholze, M.; Winter, M.J.; Barcelo, D.; Sumpter, J.P. Anti-anxiety drugs and fish behavior: Establishing the link between internal concentrations of oxazepam and behavioral effects. Environ. Toxicol. Chem. 2016, 35, 2782–2790. [Google Scholar] [CrossRef]
- Valenti, T.W.; Gould, G.G.; Berninger, J.P.; Connors, K.A.; Keele, N.B.; Prosser, K.N.; Brooks, B.W. Human therapeutic plasma levels of the selective serotonin reuptake inhibitor (SSRI) sertraline decrease serotonin reuptake transporter binding and shelter-seeking behavior in adult male fathead minnows. Environ. Sci. Technol. 2012, 46, 2427–2435. [Google Scholar] [CrossRef]
- Margiotta-Casaluci, L.; Owen, S.F.; Cumming, R.I.; de Polo, A.; Winter, M.J.; Panter, G.H.; Rand-Weaver, M.; Sumpter, J.P. Quantitative cross-species extrapolation between humans and fish: The case of the anti-depressant fluoxetine. PLoS ONE 2014, 9, e110467. [Google Scholar] [CrossRef]
- Schultz, W. Multiple functions of dopamine neurons. F1000 Biol. Rep. 2010, 2, 2–4. [Google Scholar] [CrossRef]
- Wood, S.J.; Toth, M. Molecular pathways of anxiety revealed by knockout mice. Mol. Neurobiol 2001, 23, 101–119. [Google Scholar] [CrossRef]
- Zifa, E.; Fillion, G. 5-Hydroxytryptamine receptors. Pharm. Rev. 1992, 44, 401–458. [Google Scholar]


| Genes | Primer Type | Sequences |
|---|---|---|
| DRD1 | Forward | CGTCGTCGTCGTTGTCGTCATC |
| Reverse | AGGCAGGCAAGCTGTTGATGTG | |
| DRD2 | Forward | GAAGGTGCGTAGAGATGCGAAGAC |
| Reverse | CCCTCCAAACTCACTCCACTCATTC | |
| DRD3 | Forward | TGTAGGAGAGTCACCACGAAGGC |
| Reverse | ACTACCAGGGAATCGGTTGTCATTG | |
| DAT | Forward | TGATTGGTTGGTGCGTTGCTCTC |
| Reverse | GCTTGCTTGTGGTTAGTTGGATGTG | |
| 5-HTR1 | Forward | CGGAAGGCTGCCAGAGTGATTG |
| Reverse | TCTAAAGAGGCACGAGCGAAATTGG | |
| 5-HTR1B | Forward | CGGAAGGCTGCCAGAGTGATTG |
| Reverse | TCCGATCAAGGCAACCATGAAGAAG | |
| 5-HTR2A | Forward | CATCTCCTGCGAGTCCAAGACATTC |
| Reverse | TTAGTAGCCTTCCGTTCCGTTTGC | |
| SERT | Forward | AGTCTGGATAACAGCCACCGTACC |
| Reverse | TCCCATCACTGGAACCAGGAAGAG | |
| MAO | Forward | GCAACCCAATCGCCTTCAAATCAC |
| Reverse | CTGTCTTCCGAGTGTCCATGTTCC | |
| β-actin | Forward | CCCAGGTATTGCTGACCGTATGC |
| Reverse | GAAGGTGGACAGAGAAGCCAAGATG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, X.; Xu, Q.; Guo, Y.; Zhang, B. Effects of Environmentally Relevant Concentrations of Antipsychotic Drugs (Sulpiride and Clozapine) on Serotonergic and Dopaminergic Neurotransmitter Systems in Octopus Brain Tissue. Water 2022, 14, 2608. https://doi.org/10.3390/w14172608
Peng X, Xu Q, Guo Y, Zhang B. Effects of Environmentally Relevant Concentrations of Antipsychotic Drugs (Sulpiride and Clozapine) on Serotonergic and Dopaminergic Neurotransmitter Systems in Octopus Brain Tissue. Water. 2022; 14(17):2608. https://doi.org/10.3390/w14172608
Chicago/Turabian StylePeng, Xijian, Qiuxia Xu, Yuanming Guo, and Bo Zhang. 2022. "Effects of Environmentally Relevant Concentrations of Antipsychotic Drugs (Sulpiride and Clozapine) on Serotonergic and Dopaminergic Neurotransmitter Systems in Octopus Brain Tissue" Water 14, no. 17: 2608. https://doi.org/10.3390/w14172608
APA StylePeng, X., Xu, Q., Guo, Y., & Zhang, B. (2022). Effects of Environmentally Relevant Concentrations of Antipsychotic Drugs (Sulpiride and Clozapine) on Serotonergic and Dopaminergic Neurotransmitter Systems in Octopus Brain Tissue. Water, 14(17), 2608. https://doi.org/10.3390/w14172608





