Simultaneous Removal of Nitrate and Tetracycline by an Up-Flow Immobilized Biofilter
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
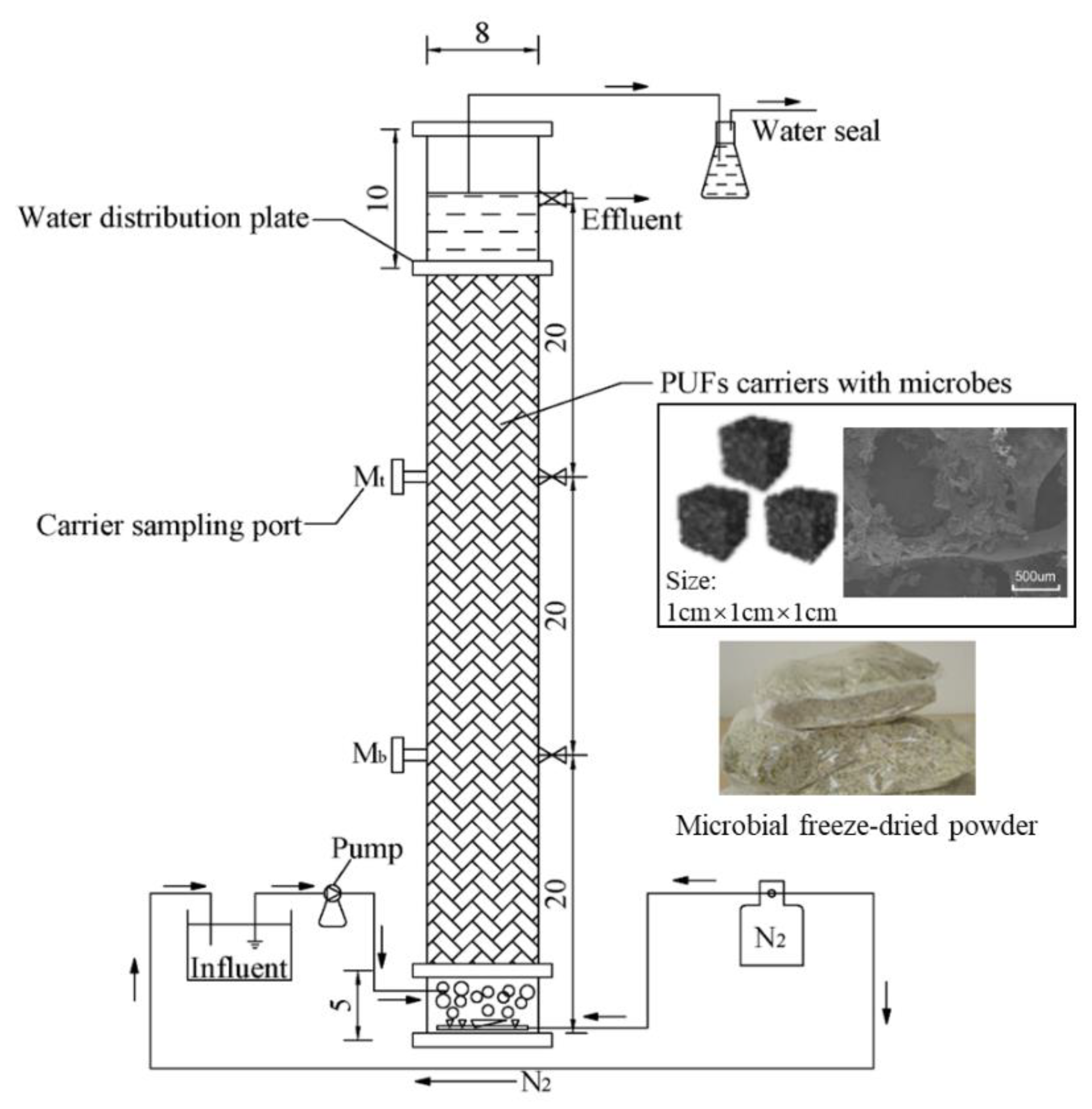
2.2. Experimental Apparatus
2.3. Experiment Procedure
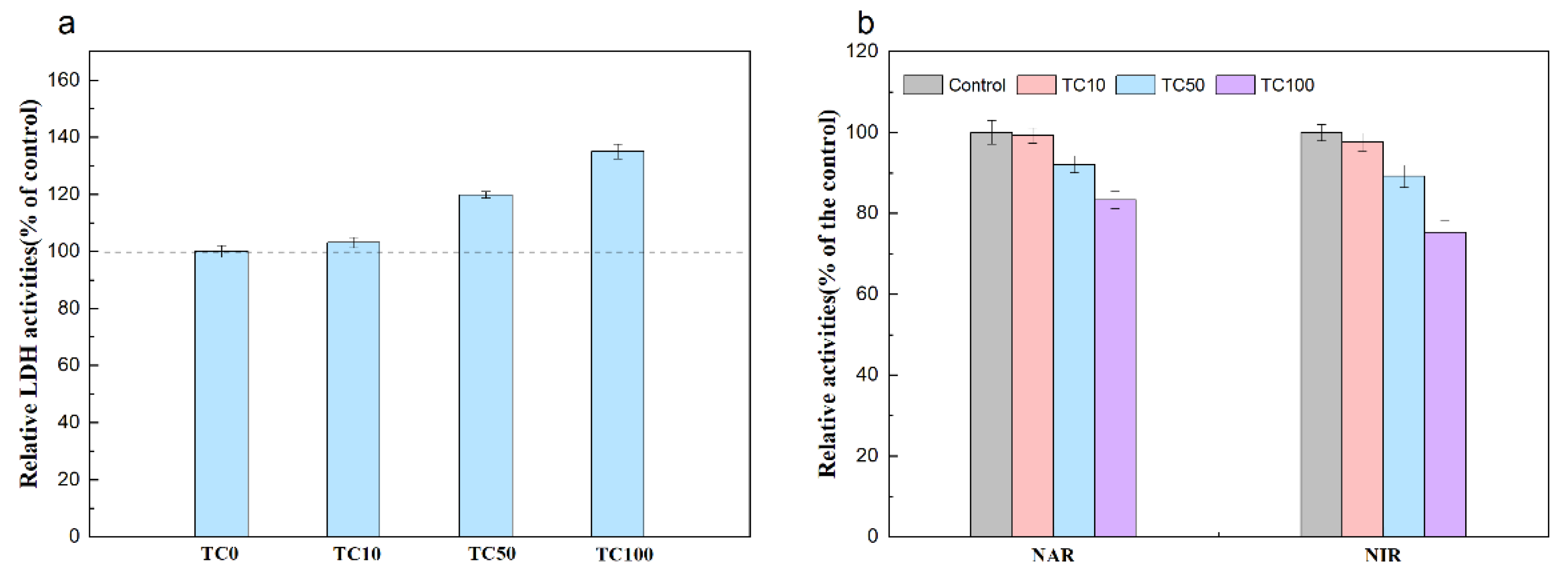
2.4. Enzyme Activities Assays
2.5. Analytical Methods
3. Results and Discussion
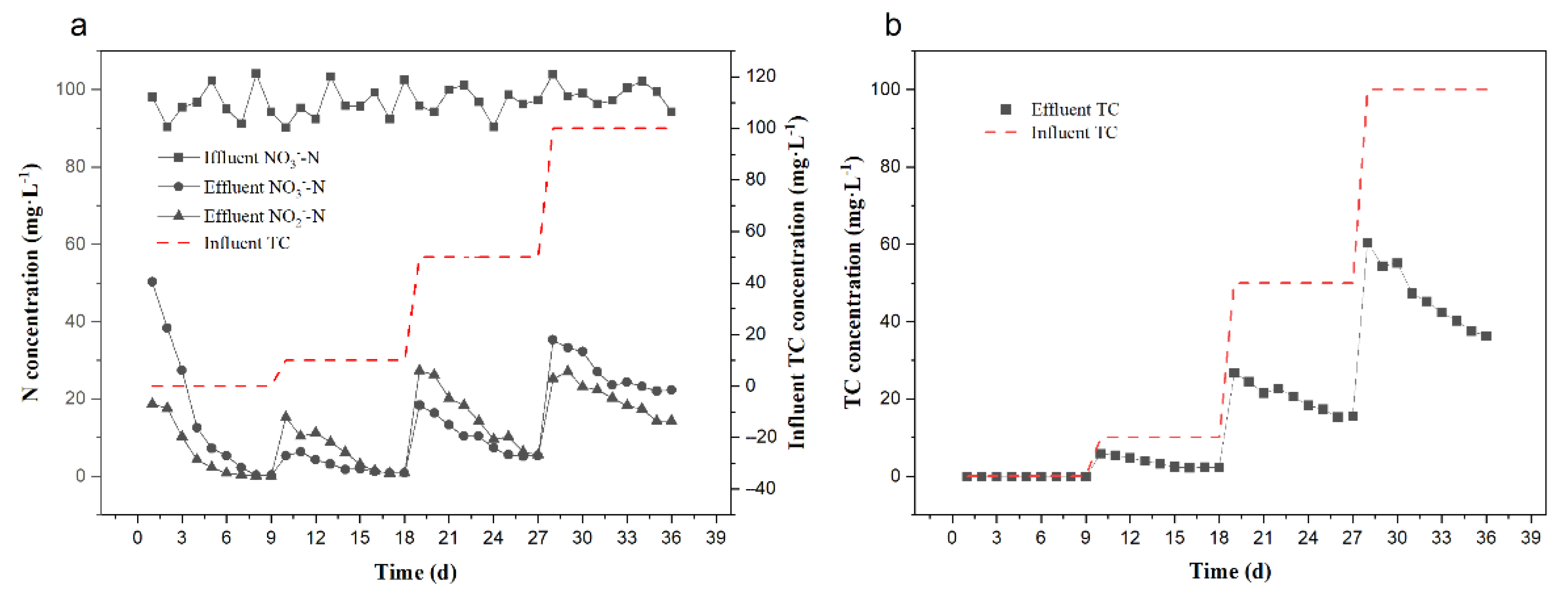
3.1. Denitrification Performance under Different TC Concentrations
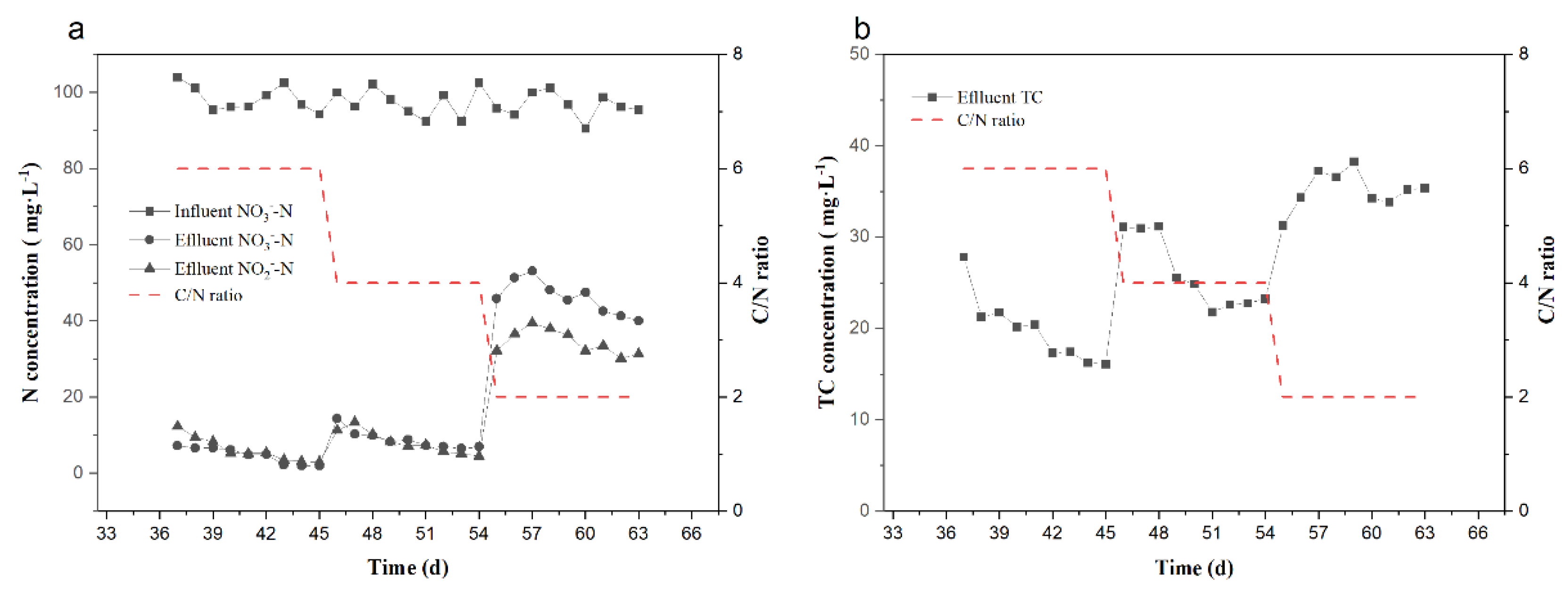
3.2. Effect of C/N Ratio on Denitrification and TC Removal
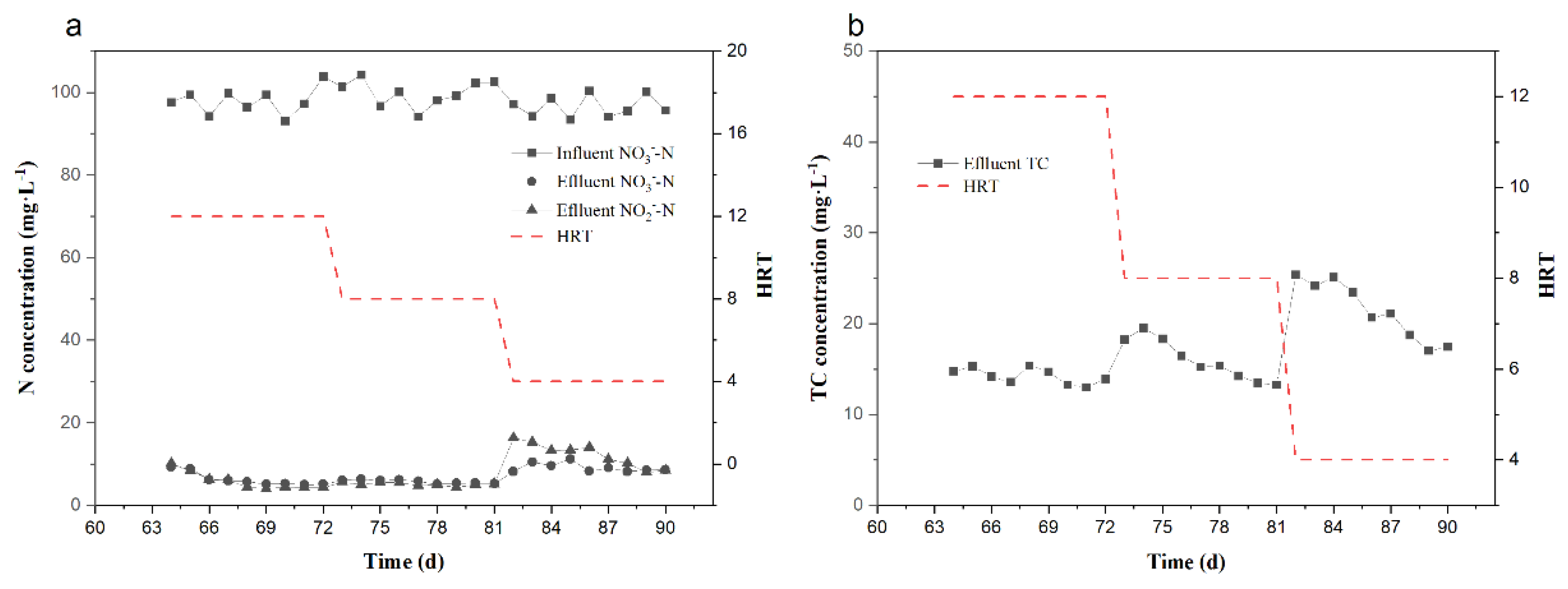
3.3. Effect of HRT on Denitrification and TC Removal
3.4. Inhibition Mechanism of TC on Denitrification
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. Fisheries and Aquaculture Statistics. Global aquaculture production 1950–2020. In FAO Fisheries and Aquaculture Department. Available online: www.fao.org/fishery/en/statistics/collections (accessed on 16 July 2022).
- Wang, J.; Beusen, A.H.W.; Liu, X.; Bouwman, A.F. Aquaculture production is a large, spatially concentrated source of nutrients in chinese freshwater and coastal seas. Environ. Sci. Technol. 2020, 54, 1464–1474. [Google Scholar] [CrossRef]
- Zhang, Y.; Bleeker, A.; Liu, J. Nutrient discharge from China’s aquaculture industry and associated environmental impacts. Environ. Res. Lett. 2015, 10, 45002. [Google Scholar] [CrossRef]
- Yang, X.; Song, X.; Hallerman, E.; Huang, Z. Microbial community structure and nitrogen removal responses of an aerobic denitrification biofilm system exposed to tetracycline. Aquaculture 2020, 529, 735665. [Google Scholar] [CrossRef]
- Brüssow, H. Growth promotion and gut microbiota: Insights from antibiotic use. Environ. Microbiol. 2015, 7, 2216–2227. [Google Scholar] [CrossRef]
- Shao, Y.; Wang, Y.; Yuan, Y.; Xie, Y. A systematic review on antibiotics misuse in livestock and aquaculture and regulation implications in China. Sci. Total Environ. 2021, 798, 149205. [Google Scholar] [CrossRef]
- Chen, J.; Sun, R.; Pan, C.; Sun, Y.; Mai, B.; Li, Q.X. Antibiotics and food safety in aquaculture. J. Agric. Food Chem. 2020, 68, 11908–11919. [Google Scholar] [CrossRef]
- Leal, J.F.; Henriques, I.S.; Correia, A.; Santos, E.; Esteves, V.I. Antibacterial activity of oxytetracycline photoproducts in marine aquaculture’s water. Environ. Pollut. 2017, 220, 644–649. [Google Scholar] [CrossRef]
- Zeng, X.; Sun, X.; Wang, Y. Photocatalytic degradation of flumequine by N-doped TiO2 catalysts under simulated sunlight. Environ. Eng. Res. 2021, 26, 200524. [Google Scholar] [CrossRef]
- Gorito, A.M.; Ribeiro, A.R.L.; Rodrigues, P.; Pereira, M.F.R.; Guimarães, L.; Almeida, C.M.R.; Silva, A.M.T. Antibiotics removal from aquaculture effluents by ozonation: Chemical and toxicity descriptors. Water Res. 2022, 218, 118497. [Google Scholar] [CrossRef]
- Prieto-Rodríguez, L.; Oller, I.; Klamerth, N.; Agüera, A.; Rodríguez, E.M.; Malato, S. Application of solar AOPs and ozonation for elimination of micropollutants in municipal wastewater treatment plant effluents. Water Res. 2013, 47, 1521–1528. [Google Scholar] [CrossRef]
- Semedo, M.; Song, B.; Sparrer, T.; Phillips, R.L. Antibiotic effects on microbial communities responsible for denitrification and N2O production in grassland soils. Front. Microbiol. 2018, 9, 2121. [Google Scholar] [CrossRef] [Green Version]
- Chen, A.; Chen, Y.; Ding, C.; Liang, H.; Yang, B. Effects of tetracycline on simultaneous biological wastewater nitrogen and phosphorus removal. Rsc. Adv. 2015, 5, 59326–59334. [Google Scholar] [CrossRef]
- Shao, S.; Hu, Y.; Cheng, J.; Chen, Y. Effects of carbon source, nitrogen source, and natural algal powder-derived carbon source on biodegradation of tetracycline (TEC). Bioresour. Technol. 2019, 288, 121567. [Google Scholar] [CrossRef]
- Bai, Y.; Su, J.; Ali, A.; Wen, Q.; Chang, Q.; Gao, Z.; Wang, Y. Efficient removal of nitrate, manganese, and tetracycline in a novel loofah immobilized bioreactor: Performance, microbial diversity, and functional genes. Bioresour. Technol. 2022, 344, 126228. [Google Scholar] [CrossRef]
- Taskan, B.; Hanay, O.; Taskan, E.; Erdem, M.; Hasar, H. Hydrogen-based membrane biofilm reactor for tetracycline removal: Biodegradation, transformation products, and microbial community. Environ. Sci. Pollut. Res. Int. 2016, 23, 21703–21711. [Google Scholar] [CrossRef]
- Li, W.; Shi, C.; Yu, Y.; Ruan, Y.; Kong, D.; Lv, X.; Xu, P.; Awasthi, M.K.; Dong, M. Interrelationships between tetracyclines and nitrogen cycling processes mediated by microorganisms: A review. Bioresour. Technol. 2021, 319, 124036. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, G.; Song, K.; Wang, Z.; Zhao, Q.; Li, S.; Ye, Z. Biological treatment of 2,4,6-trinitrotoluene (TNT) red water by immobilized anaerobic–aerobic microbial filters. Chem. Eng. J. 2015, 259, 876–884. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Ye, Z.F.; Zhang, M.H. Bioremediation of 2,4-dinitrotoluene (2,4-DNT) in immobilized micro-organism biological filter. J. Appl. Microbiol. 2011, 110, 1476–1484. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, Y.; Ye, Z.; Borthwick, A.G.L.; Ni, J. Oil field wastewater treatment in Biological Aerated Filter by immobilized microorganisms. Process Biochem. 2006, 41, 1475–1483. [Google Scholar] [CrossRef]
- Gan, Y.; Ye, Z.; Zhao, Q.; Li, L.; Lu, X. Spatial denitrification performance and microbial community composition in an up-flow immobilized biofilter for nitrate micro-polluted water treatment. J. Clean Prod. 2020, 258, 120913. [Google Scholar] [CrossRef]
- Xu, W.; Yang, Z.; Tang, H.; Wang, C.; Ye, Z. Response of immobilized denitrifying bacterial consortium to tetracycline exposure. Ecotox. Environ. Saf. 2022, 239, 113652. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Zhang, Q.; Wu, L.; Cao, J.; Feng, Q.; Fang, F.; Chen, Y. Inhibition of 1, 4-dioxane on the denitrification process by altering the viability and metabolic activity of Paracoccus denitrificans. Environ. Sci. Pollut. Res. Int. 2018, 25, 27274–27282. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, Y.; Wang, L.; Zhu, M.; Zhu, X.; Qian, C.; Li, W. Responses of biofilm microorganisms from moving bed biofilm reactor to antibiotics exposure: Protective role of extracellular polymeric substances. Bioresour. Technol. 2018, 254, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Su, Y.; Chen, Y.; Wan, R.; Liu, K.; Li, M.; Yin, D. Zinc oxide nanoparticles cause inhibition of microbial denitrification by affecting transcriptional regulation and enzyme activity. Environ. Sci. Technol. 2014, 48, 13800–13807. [Google Scholar] [CrossRef]
- Chen, Q.; Wu, W.; Zhang, Z.; Xu, J.; Jin, R. Inhibitory effects of sulfamethoxazole on denitrifying granule properties: Short- and long-term tests. Bioresour. Technol. 2017, 233, 391–398. [Google Scholar] [CrossRef]
- Shu, Y.; Liang, D. Effect of tetracycline on nitrogen removal in Moving Bed Biofilm Reactor (MBBR) System. PLoS ONE 2022, 17, 261306. [Google Scholar] [CrossRef]
- Scaria, J.; Anupama, K.V.; Nidheesh, P.V. Tetracyclines in the environment: An overview on the occurrence, fate, toxicity, detection, removal methods, and sludge management. Sci. Total Environ. 2021, 771, 145291. [Google Scholar] [CrossRef]
- Cheng, D.; Ngo, H.H.; Guo, W.; Chang, S.W.; Nguyen, D.D.; Liu, Y.; Shan, X.; Nghiem, L.D.; Nguyen, L.N. Removal process of antibiotics during anaerobic treatment of swine wastewater. Bioresour. Technol. 2020, 300, 122707. [Google Scholar] [CrossRef]
- Fu, X.; Hou, R.; Yang, P.; Qian, S.; Feng, Z.; Chen, Z.; Wang, F.; Yuan, R.; Chen, H.; Zhou, B. Application of external carbon source in heterotrophic denitrification of domestic sewage: A review. Sci. Total Environ. 2022, 817, 153061. [Google Scholar] [CrossRef]
- Krishna Mohan, T.V.; Nancharaiah, Y.V.; Venugopalan, V.P.; Satya Sai, P.M. Effect of C/N ratio on denitrification of high-strength nitrate wastewater in anoxic granular sludge sequencing batch reactors. Ecol. Eng. 2016, 91, 441–448. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Microbial degradation of sulfamethoxazole in the environment. Appl. Microbiol. Biot. 2018, 102, 3573–3582. [Google Scholar] [CrossRef] [PubMed]
- Niu, W.; Guo, J.; Lian, J.; Ngo, H.H.; Li, H.; Song, Y.; Li, H.; Yin, P. Effect of fluctuating hydraulic retention time (HRT) on denitrification in the UASB reactors. Biochem. Eng. J. 2018, 132, 29–37. [Google Scholar] [CrossRef]
- He, Y.; Wang, Y.; Song, X. High-effective denitrification of low C/N wastewater by combined constructed wetland and biofilm-electrode reactor (CW–BER). Bioresour. Technol. 2016, 203, 245–251. [Google Scholar] [CrossRef]
- Su, Y.; Zheng, X.; Chen, Y.; Li, M.; Liu, K. Alteration of intracellular protein expressions as a key mechanism of the deterioration of bacterial denitrification caused by copper oxide nanoparticles. Sci. Rep. UK 2015, 5, 15824. [Google Scholar] [CrossRef] [Green Version]
- Deng, Z.; Wang, Z.; Zhang, P.; Xia, P.; Ma, K.; Zhang, D.; Wang, L.; Yang, Y.; Wang, Y.; Chen, S.; et al. Effects of divalent copper on microbial community, enzymatic activity and functional genes associated with nitrification and denitrification at tetracycline stress. Enzyme Microb. Technol. 2019, 126, 62–68. [Google Scholar] [CrossRef] [PubMed]
| Periods | 1 | 2 | 3 |
|---|---|---|---|
| Operating Time (d) | 1–36 | 37–63 | 64–90 |
| NO3−-N (mg·L−1) | 100 | 100 | 100 |
| TC (mg·L−1) | 0, 10, 50, 100 | 50 | 50 |
| C/N ratio | 5 | 6, 4, 2 | 5 |
| HRT (h) | 12 | 12 | 12, 8, 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, W.; Luo, M.; Lu, X.; Ye, Z.; Jeong, T. Simultaneous Removal of Nitrate and Tetracycline by an Up-Flow Immobilized Biofilter. Water 2022, 14, 2595. https://doi.org/10.3390/w14172595
Xu W, Luo M, Lu X, Ye Z, Jeong T. Simultaneous Removal of Nitrate and Tetracycline by an Up-Flow Immobilized Biofilter. Water. 2022; 14(17):2595. https://doi.org/10.3390/w14172595
Chicago/Turabian StyleXu, Wenjie, Minghan Luo, Xinyue Lu, Zhengfang Ye, and Taeseop Jeong. 2022. "Simultaneous Removal of Nitrate and Tetracycline by an Up-Flow Immobilized Biofilter" Water 14, no. 17: 2595. https://doi.org/10.3390/w14172595








