Study on the Breeding and Characterization of High-Efficiency Oil-Degrading Bacteria by Mutagenesis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Isolation and Culture of Petroleum Hydrocarbon Degrading Bacteria
2.3. Mutagenesis Test of Petroleum Hydrocarbon Degrading Bacteria
2.4. Degradation Test of Pollutants by Mutant Strain MXM3U2
2.5. Analysis of Petroleum Hydrocarbons
2.5.1. Determination of the Degradation Proportion of Petroleum Hydrocarbons by UV Spectrophotometry
2.5.2. Determination of the Growth of Petroleum Hydrocarbon-Degrading Bacteria by UV Spectrophotometry
3. Results and Discussion
3.1. Screening and Characteristics of Petroleum Hydrocarbon Degrading Bacteria MX1 Strain
3.2. 16S rDNA Sequence Homology and Phylogenetic Analysis
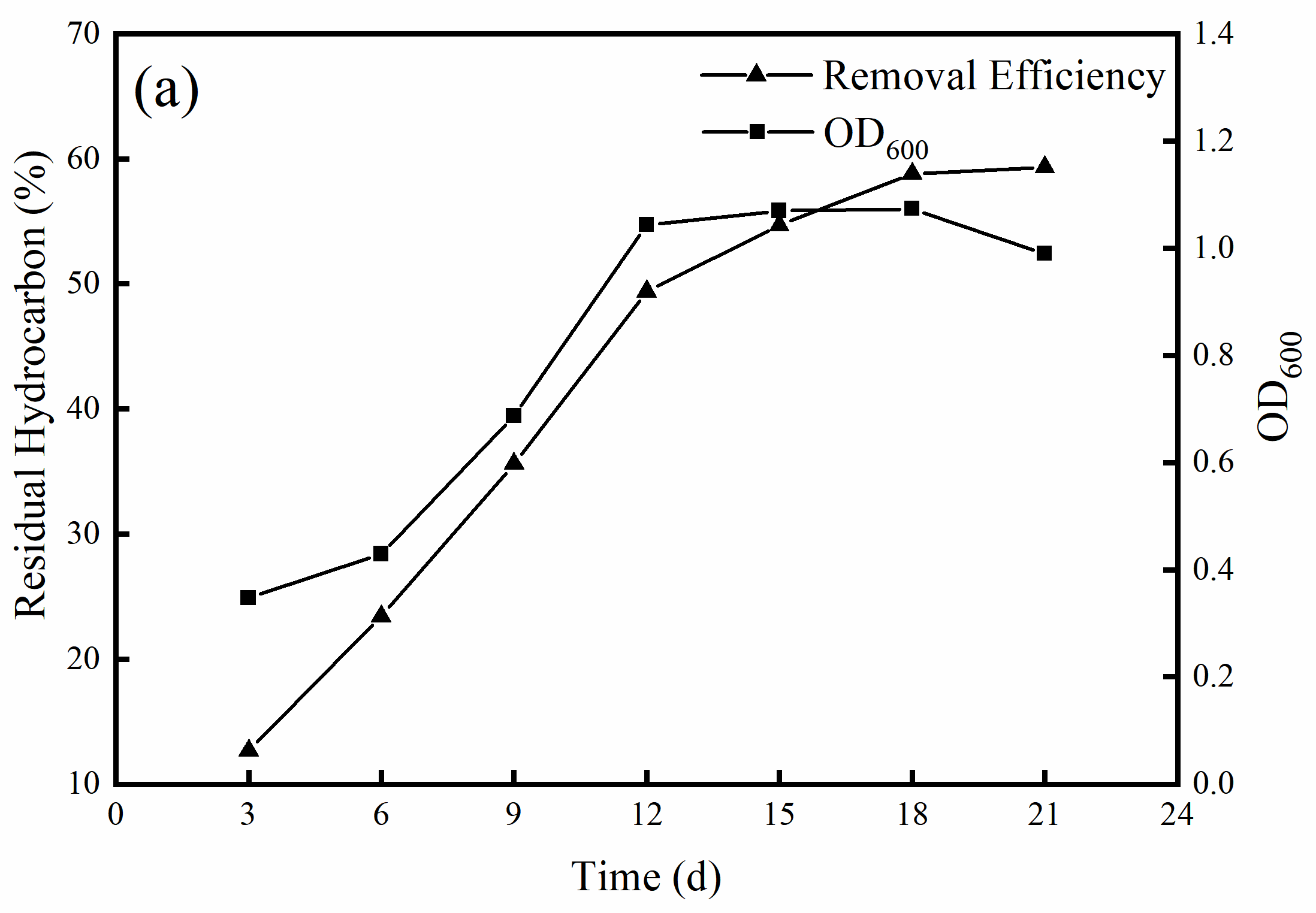
3.3. Determination of Optimal Growth and Degradation Conditions of the MX1 Strain
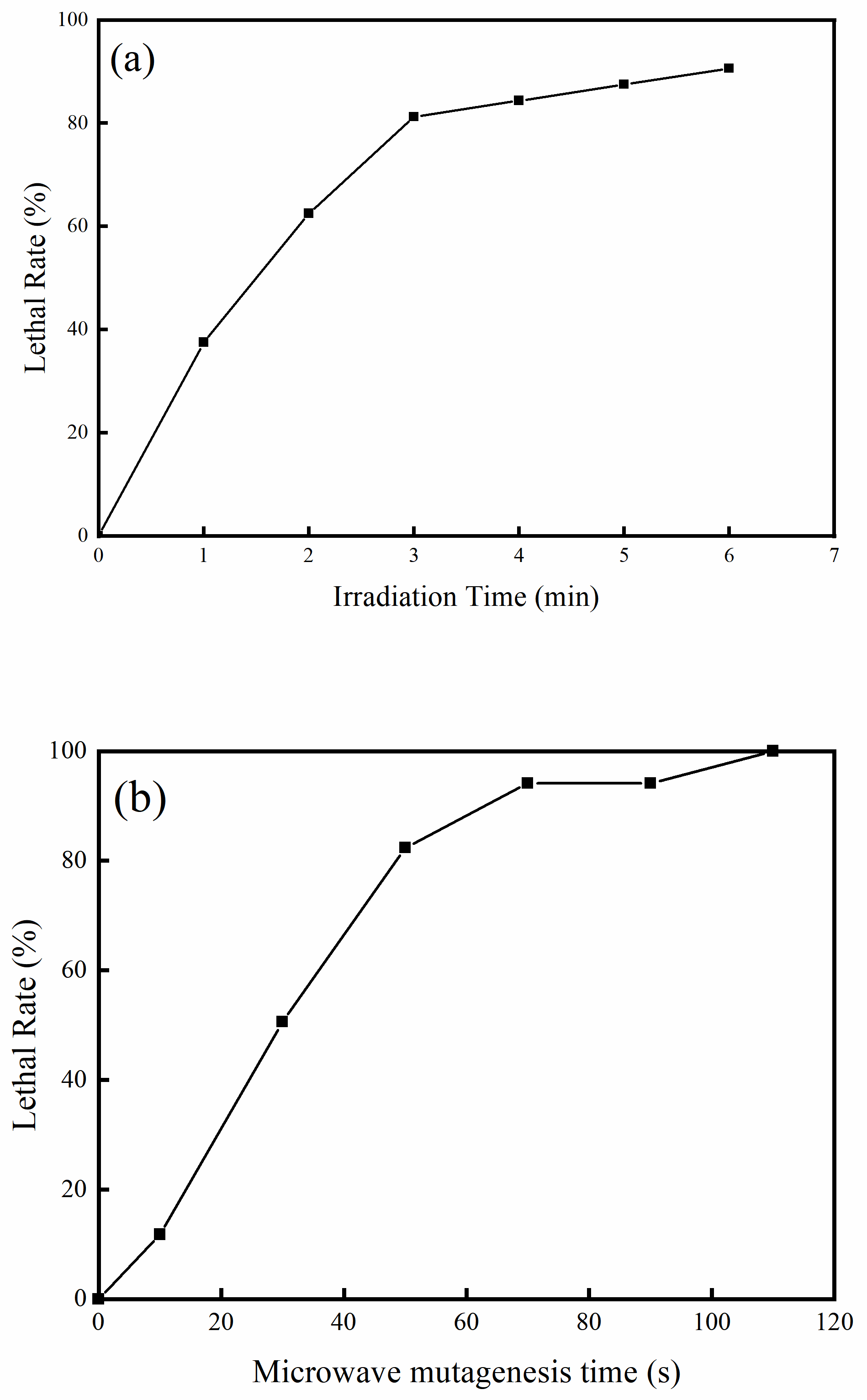
3.4. Microwave-UV Mutagenesis of Petroleum Hydrocarbon Degrading Bacteria
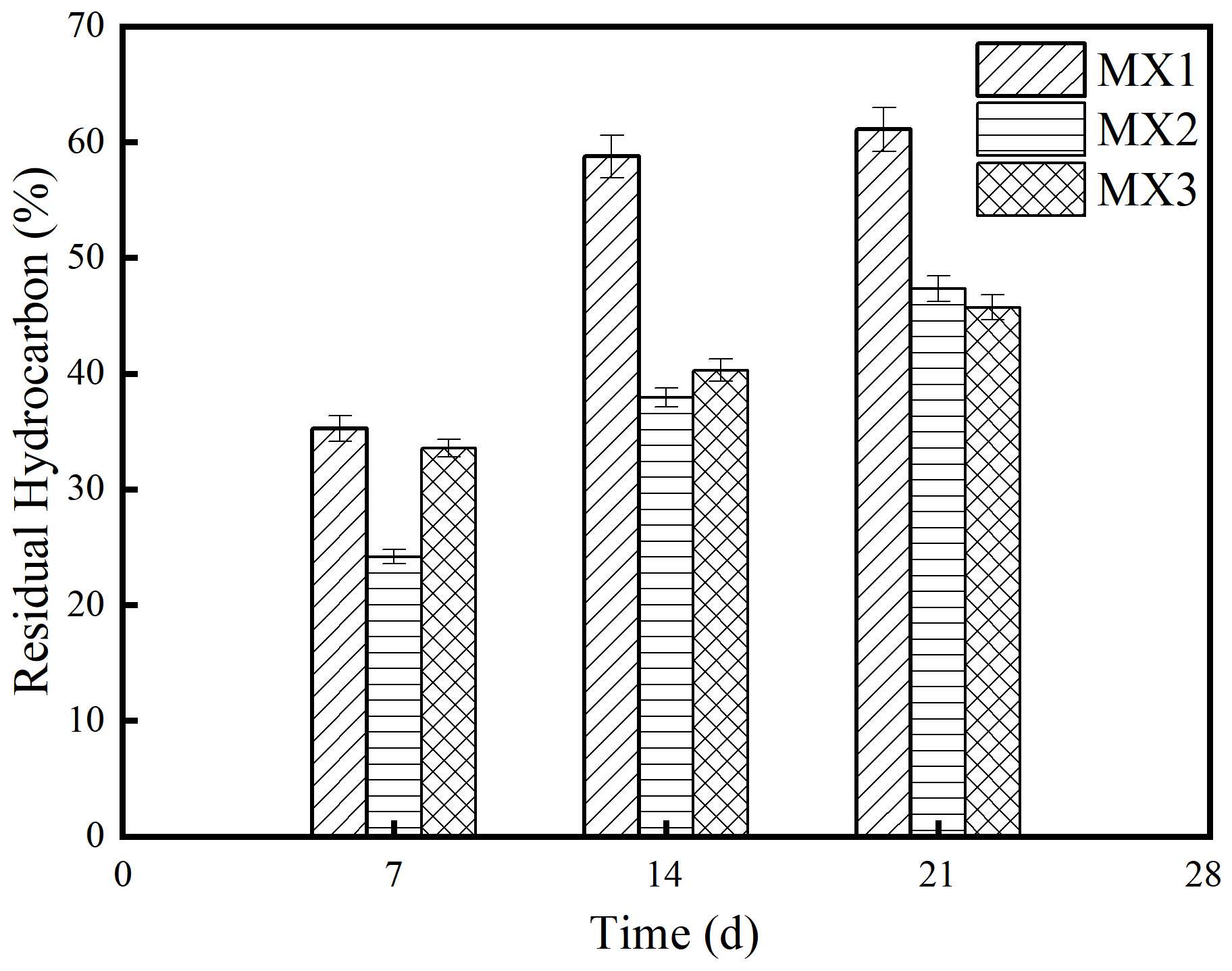
3.5. Screening of Mutant Strains by UV and Microwave
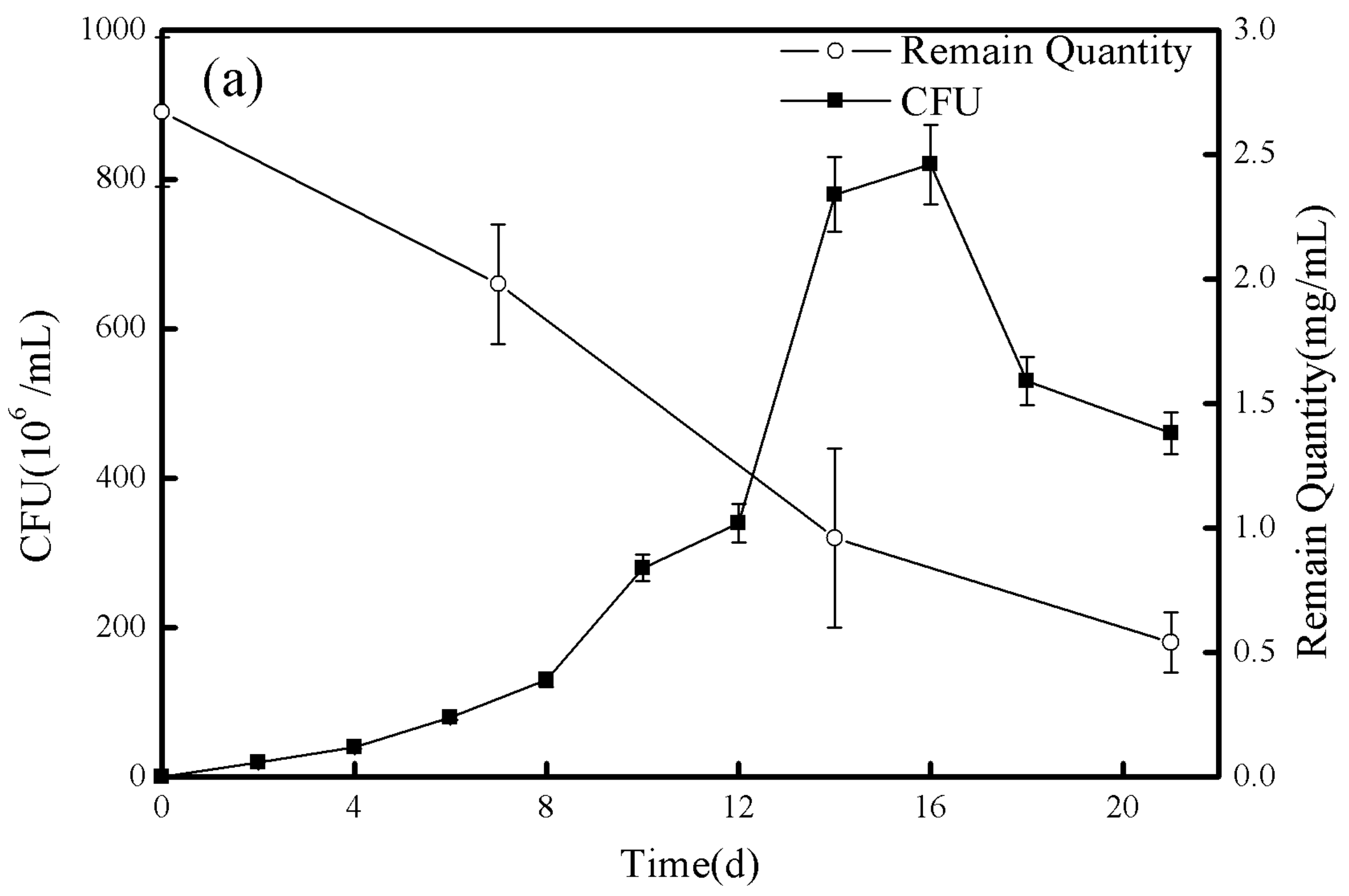
3.6. Degradation of Alkanes by Mutant Strain MXM3U2
3.7. Degradation Pathway of Pristane, Naphthalene, and Phenanthrene by Mutant Strains
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Romeiko, X.X. A comparative life cycle assessment of crop systems irrigated with the groundwater and reclaimed water in northern china. Sustainability 2019, 11, 2743. [Google Scholar] [CrossRef]
- Zulkefli, N.F.; Alias, N.H.; Jamaluddin, N.S.; Abdullah, N.; Abdul Manaf, S.F.; Othman, N.H.; Marpani, F.; Mat-Shayuti, M.S.; Kusworo, T.D. Recent mitigation strategies on membrane fouling for oily wastewater treatment. Membranes 2022, 12, 26. [Google Scholar] [CrossRef] [PubMed]
- Adetunji, A.I.; Olaniran, A.O. Treatment of industrial oily wastewater by advanced technologies: A review. Appl. Water Sci. 2021, 11, 1–19. [Google Scholar] [CrossRef]
- Ahmad, N.; Goh, P.; Abdul Karim, Z.; Ismail, A. Thin film composite membrane for oily waste water treatment: Recent advances and challenges. Membranes 2018, 8, 86. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Jing, J.; Li, J.; Zhu, D.; Zou, A.; Zheng, L.; Jin, Z. New exploration strategy in igneous petroliferous basins—Enlightenment from simulation experiments. Energ. Explor. Exploit. 2018, 36, 971–985. [Google Scholar] [CrossRef]
- Wen, Y.; Xu, L.; Xue, C.; Jiang, X.; Wei, Z. Assessing the impact of oil types and grades on tocopherol and tocotrienol contents in vegetable oils with chemometric methods. Molecules 2020, 25, 5076. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W. Dilemma of multisubject Co-Governance of global marine ecological environment and implementation path. Sustainability 2021, 13, 11145. [Google Scholar] [CrossRef]
- Mahamane, S.; Wan, N.; Porter, A.; Hancock, A.S.; Campbell, J.; Lyon, T.E.; Jordan, K.E. Natural categorization: Electrophysiological responses to viewing natural versus built environments. Front. Psychol. 2020, 11, 990. [Google Scholar] [CrossRef]
- Sembiring, K.C.; Saka, S. Renewable hydrocarbon fuels from plant oils for diesel and gasoline. J. Jpn. Petrol. Inst. 2019, 62, 157–172. [Google Scholar] [CrossRef]
- Smith, H.B.; Kim, H.; Walker, S.I. Scarcity of scale-free topology is universal across biochemical networks. Sci. Rep. 2021, 11, 6542. [Google Scholar] [CrossRef]
- Lesin, Y.; Gogolin, V.; Murko, E.; Markov, S.; Kretschmann, J. The choice of methods of quarry wastewater purifying. E3S Web Conf. 2018, 41, 1039. [Google Scholar] [CrossRef]
- Zhao, D.; Cheng, X.; Zhang, H.; Niu, Y.; Qi, Y.; Zhang, H. Evaluation of the ability of spectral indices of hydrocarbons and seawater for identifying oil slicks utilizing hyperspectral images. Remote Sens. 2018, 10, 421. [Google Scholar] [CrossRef]
- Haule, K.; Toczek, H.; Borzycka, K.; Darecki, M. Influence of dispersed oil on the remote sensing reflectance—Field experiment in the baltic sea. Sensors 2021, 21, 5733. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Wang, Z.; Xu, M. Progress in treatment of oily wastewater by inorganic porous ceramic membrane. MATEC Web Conf. 2017, 114, 2016. [Google Scholar] [CrossRef]
- Lee, B.; Schlautman, M. Effects of polymer molecular weight on adsorption and flocculation in aqueous kaolinite suspensions dosed with nonionic polyacrylamides. Water 2015, 7, 5896–5909. [Google Scholar] [CrossRef]
- Ghasemi, N.; Zare, F.; Hosano, H. A review of pulsed power systems for degrading water pollutants ranging from microorganisms to organic compounds. IEEE Access 2019, 7, 150863–150891. [Google Scholar] [CrossRef]
- Cao, L.; Liu, X.; Zhang, S.; Lyu, M. Comparison of regional urban water pollutants emission standards and determination of factors influencing their integration—A case study of the biopharmaceutical industry in the yangtze river delta urban agglomeration. Sustainability 2022, 14, 4741. [Google Scholar] [CrossRef]
- Capodaglio, A.G. Critical perspective on advanced treatment processes for water and wastewater: AOPs, ARPs, and AORPs. Appl. Sci. 2020, 10, 4549. [Google Scholar] [CrossRef]
- Valkov, A.; Raik, K.; Mualem-Sinai, Y.; Nakonechny, F.; Nisnevitch, M. Water disinfection by immobilized photosensitizers. Water 2019, 11, 26. [Google Scholar] [CrossRef]
- Poblete, I.B.S.; Araujo, O.D.Q.F.; de Medeiros, J.L. Sewage-Water treatment and Sewage-Sludge management with power production as bioenergy with carbon capture system: A review. Processes 2022, 10, 788. [Google Scholar] [CrossRef]
- Sayara, T.; Sánchez, A. Bioremediation of PAH-Contaminated soils: Process enhancement through composting/compost. Appl. Sci. 2020, 10, 3684. [Google Scholar] [CrossRef]
- Madhavan, J.; Theerthagiri, J.; Balaji, D.; Sunitha, S.; Choi, M.Y.; Ashokkumar, M. Hybrid advanced oxidation processes involving ultrasound: An overview. Molecules 2019, 24, 3341. [Google Scholar]
- Ko, B.H.; Hasa, B.; Shin, H.; Jeng, E.; Overa, S.; Chen, W.; Jiao, F. The impact of nitrogen oxides on electrochemical carbon dioxide reduction. Nat. Commun. 2020, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Alexander, M.; Alarwan, N.; Chandrasekaran, M.; Sundaram, A.; Milde, T.; Rasool, S. Experiments and modeling for investigation of oily sludge biodegradation in a wastewater pond environment. Appl. Sci. 2020, 10, 1659. [Google Scholar] [CrossRef]
- Xu, R.; Pei, Z.; Wang, W.; Zhang, M.; Zhang, L.; Zhang, J.; Wang, W.; Sun, L.; Zhang, Y. Assessment of biological toxicity and ecological safety for urban black-odor river remediation. Int. J. Environ. Res. Public Health 2020, 17, 1025. [Google Scholar] [CrossRef]
- Husti, A.; Cantor, M.; Stefan, R.; Miclean, M.; Roman, M.; Neacsu, I.; Contiu, I.; Magyari, K.; Baia, M. Assessing the indoor pollutants effect on ornamental plants leaves by FT-IR spectroscopy. Acta Physica Polonica A 2016, 129, 142–149. [Google Scholar] [CrossRef]
- Qin, C.; Li, Z.; Xie, P.; Hao, Q.; Tang, X.; Wu, Y.; Du, P. Temporal variation and reduction strategy of nutrient loads from an urban river catchment into a Eutrophic Lake, China. Water 2019, 11, 166. [Google Scholar] [CrossRef]
- Darabi, B.; Tabatabaei, T.; Amiri, F.; Jorfi, S. Integrated treatment of saline oily wastewater using Sono-Electrokinetic process, degradation mechanism, and toxicity assessment. Chem. Biochem. Eng. Q. 2020, 34, 181–191. [Google Scholar] [CrossRef]
- Yaacob, N.; Goh, P.S.; Ismail, A.F.; Mohd Nazri, N.A.; Ng, B.C.; Zainal Abidin, M.N.; Yogarathinam, L.T. ZrO2-TiO2 incorporated PVDF dual-layer hollow fiber membrane for oily wastewater treatment: Effect of air gap. Membranes 2020, 10, 124. [Google Scholar] [CrossRef]
- Akratos, C.S.; Tekerlekopoulou, A.G.; Vayenas, D.V. Agro-Industrial wastewater treatment with decentralized biological treatment methods. Water 2021, 13, 953. [Google Scholar] [CrossRef]
- Cárdenas-Talero, J.L.; Silva-Leal, J.A.; Pérez-Vidal, A.; Torres-Lozada, P. The influence of municipal wastewater treatment technologies on the biological stabilization of sewage sludge: A systematic review. Sustainability 2022, 14, 5910. [Google Scholar] [CrossRef]
- Dzhumagulova, N.; Svetkov, I.; Smetanin, V.; Dinh Dap, N. Fractal analysis of biological wastewater treatment efficiency. MATEC Web Conf. 2018, 251, 6005. [Google Scholar] [CrossRef]
- Marques, E.; Silva, G.; Dias, J.; Gross, E.; Costa, M.; Rezende, R. Cave drip water-related samples as a natural environment for aromatic hydrocarbon-degrading bacteria. Microorganisms 2019, 7, 33. [Google Scholar] [CrossRef]
- Guo, X.; Zhang, M.; Gao, Y.; Li, W.; Lu, D. Determining survival fractions of Saccharomyces cerevisiae in response to ionizing radiation in liquid culture. J. Radiat. Res. 2018, 59, 760–764. [Google Scholar] [CrossRef]
- Li, X.; Gao, J.; Du, H.; Jia, J.; Zhao, X.; Ling, T. Relationship between the void and sound absorption characteristics of epoxy porous asphalt mixture based on CT. Coatings 2022, 12, 328. [Google Scholar] [CrossRef]
- Lin, C.; Wan, J.; Lu, Y.; Zhang, H.; Chen, X.; Su, Y.; Zhu, W. Active bacterial communities of pig fecal microbiota transplantation suspension prepared and preserved under different conditions. AMB Express 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Borodina, I.; Zaitsev, B.; Teplykh, A.; Burygin, G.; Guliy, O. Sensor based on PZT ceramic resonator with lateral electric field for immunodetectionof bacteria in the conducting aquatic environment †. Sensors 2020, 20, 3003. [Google Scholar] [CrossRef]
- Gutman, I. Topological properties of altan-benzenoid hydrocarbons. J. Serb. Chem. Soc. 2014, 79, 1515–1521. [Google Scholar] [CrossRef]
- Hu, W.; Li, W.; Chen, J. Recent advances of microbial breeding via heavy-ion mutagenesis at IMP. Lett. Appl. Microbiol. 2017, 65, 274–280. [Google Scholar] [CrossRef]





| Bacteria | Straight Rod-Shaped, 0.6~1.0 μm × 1.2~3.0 μm in Size, Gram-Negative, with Pericytic Flagella. | ||
|---|---|---|---|
| colony | Round or nearly round, 1–4 mm in diameter, milky white. The surface is slightly moist and shiny, with a slightly raised center and a nearly crenellated edge. | ||
| exercise observation | + | Gelatin Liquefaction Test | + |
| Anaerobic growth | + | Citrate Utilization Test | + |
| aerobic growth | + | Fluorochrome | − |
| VP test | + | KCN | + |
| indole test | − | contact enzyme | − |
| oxidase | + | H2S production test | − |
| MR test | − | Sugar fermentation test | + |
| methyl red test | + | Urease | − |
| Serial Number | t/min | Degradation Products | m/e (%) |
|---|---|---|---|
| 1 | 11.2 | 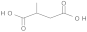 | 129 (18.12), 128 (13.43), 101 (12.13), 100 (11.57), 87 (8.19), 69 (12.92), 59 (100.00), 41 (21.08), 39 (10.87) 129 (29.35), 100 (16.58), 59 (100.00), 58 (60.96), 42 (12.87), 41 (21.93) |
| 2 | 22.6 |  | 169 (11.54), 141 (7.09), 129 (9.33), 113 (12.74), 99 (72.95), 88 (100.00), 71 (33.18), 59 (35.37), 43 (41.56) 169, 141, 129, 113, 99, 88, 71, 59, 43 standard from the library NIST |
| 3 | 25.7 |  | 238 (11.35), 226 (7.16), 212 (12.84), 197 (6.39), 185 (24.74), 167 (26.26), 155 (12.54), 125 (4.78), 113 (12.25), 85 (34.73), 71 (11.25), 69 (20.04), 58 (35.79), 43 (100.00) 238, 226, 212, 197, 185, 167, 155, 125, 113, 85, 71, 69, 59, 43 standard from the library NIST |
| 4 | 25.4 |  | 113 (12.43), 99 (10.77), 85 (28.27), 71 (77.91), 69 (11.25), 57 (100.00) 56 (20.62), 55 (20.65), 43 (65.33), 41 (30.34) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, P.; You, Z.; Chen, T.; Zhao, L.; Zhu, J.; Shi, W.; Meng, Q.; Sun, Y. Study on the Breeding and Characterization of High-Efficiency Oil-Degrading Bacteria by Mutagenesis. Water 2022, 14, 2544. https://doi.org/10.3390/w14162544
Zhang P, You Z, Chen T, Zhao L, Zhu J, Shi W, Meng Q, Sun Y. Study on the Breeding and Characterization of High-Efficiency Oil-Degrading Bacteria by Mutagenesis. Water. 2022; 14(16):2544. https://doi.org/10.3390/w14162544
Chicago/Turabian StyleZhang, Pan, Zhaoyang You, Tianfang Chen, Li Zhao, Jianguo Zhu, Weihong Shi, Qinwei Meng, and Yongjun Sun. 2022. "Study on the Breeding and Characterization of High-Efficiency Oil-Degrading Bacteria by Mutagenesis" Water 14, no. 16: 2544. https://doi.org/10.3390/w14162544






