Metal Content and Enrichment in Bivalves within the Drainage Area of Seawater Used for a Desulfurization Process in Zhanjiang Bay, China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling Stations
2.2. Sample Collection, Preservation and Determination
2.2.1. Collection and Determination of Seawater Samples
2.2.2. Collection, Preservation and Determination of Biological Samples
2.3. Data Statistics and Analysis Methods
3. Results
3.1. Content and Distribution Characteristics of Dissolved Heavy Metals in Seawater
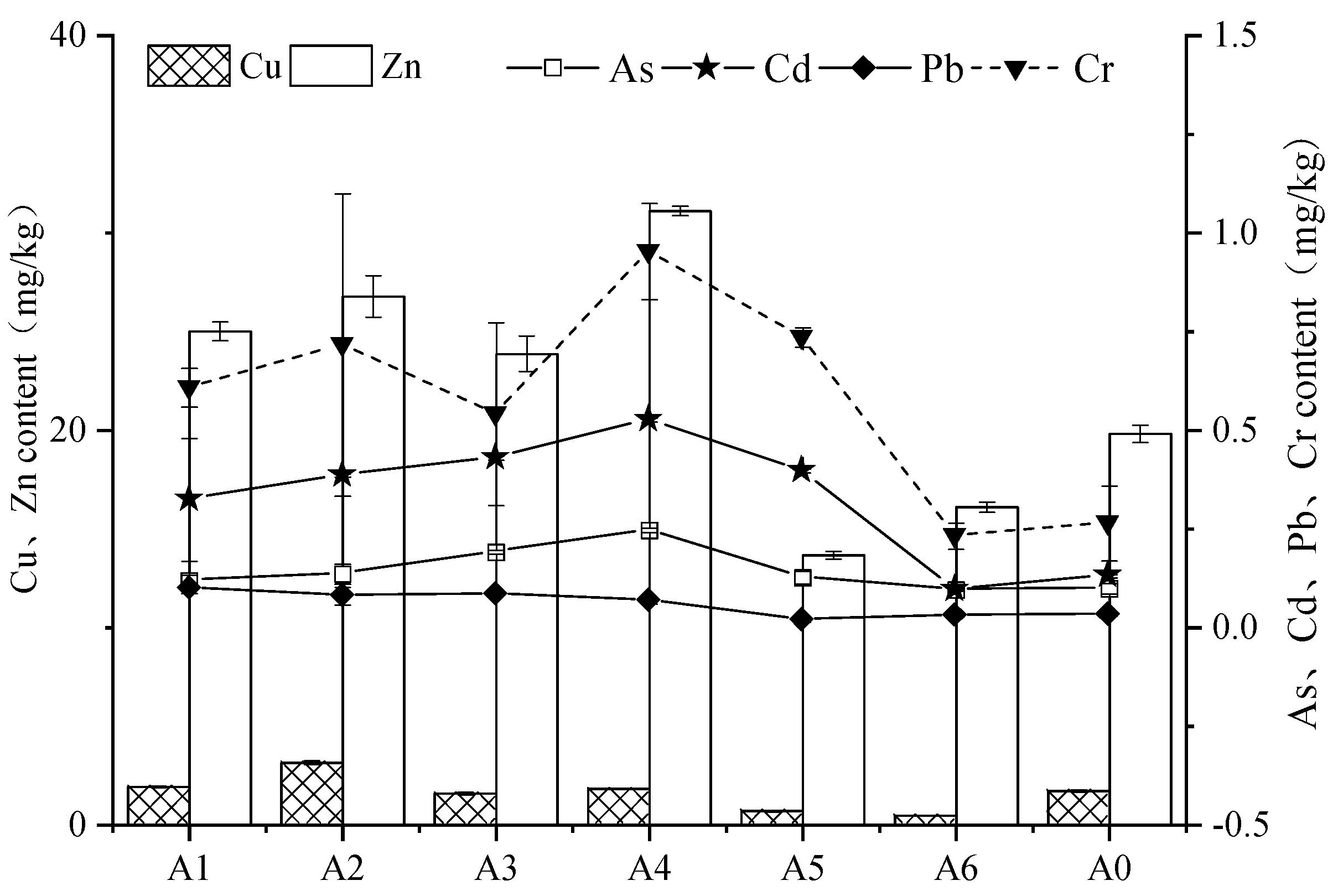
3.2. Content and Distribution Characteristics of Heavy Metals in Marine Fouling Organisms
3.3. Pollution Degree and Enrichment Behavior of Heavy Metals in Marine Organisms
4. Discussion
4.1. Effect of Seawater Desulfurization Drainage on Coastal Water Quality
4.2. Enrichment Level of Heavy Metals in Oyster and Barnacle Soft Tissue
4.3. Comparison of Heavy Metal Contents in Soft Tissues of Oysters and Barnacles in Other Estuaries and Sea Areas
5. Conclusions
- (1)
- When the seawater desulfurization system of Zhanjiang Bay coastal power plant was in normal operation, the heavy metals Cu, Zn, Cd, Pb and Cr in the seawater near the coastal power plant had obvious spatial distribution characteristics. In terms of regional distribution, the contents of Cu, Zn, Cd, Pb and Cr were higher near the drainage outlet and in the downstream waters. In the vertical direction, the concentrations of Cu, Zn, Cd, Pb, Cr and As in the surface layer were higher than those in the bottom layer within 100 m of the drainage outlet. The average concentrations of heavy metals Cu, Cr and As in the normal operation of seawater desulfurization process in Zhanjiang Bay were higher than those after the seawater desulfurization process stopped operating;
- (2)
- The content of heavy metals in the soft tissues of oysters and barnacles was Zn > Cu > Cr > Cd > Pb, which had obvious spatial distribution characteristics. In terms of regional distribution, the contents of heavy metals in the soft tissues of oysters and barnacles decreased with the increase in the distance from the outlet. In the vertical direction, the contents of heavy metals in the soft tissues of oysters and barnacles were in the order of surface layer > middle layer;
- (3)
- Single factor index (SFI) showed that the SFI of heavy metals Cu, Cr, As and Cd in the soft tissues of oysters and barnacles were in the normal range. Zn and Pb had potential pollution risks in surface water near the drainage outlet. The BCF of Zn in the soft tissues of oysters and barnacles was greater than 1000, and the BSAF was greater than 100%. The oysters and barnacles had high accumulation of Zn, and it decreased with the increase in the distance from the drainage outlet.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tokumura, M.; Baba, M.; Znad, H.T.; Kawase, Y.; Yongsiri, C.; Takeda, K. Neutralization of the acidified seawater effluent from the flue gas desulfurization process: Experimental investigation, dynamic modeling, and simulation. Ind. Eng. Chem. Res. 2006, 45, 6339–6348. [Google Scholar] [CrossRef]
- Pipitone, G.; Bolland, O. Modeling of Oxy-Combustion Flue Gas Desulfurization by Seawater Absorption. Environ. Prog. Sustain. 2009, 28, 20–29. [Google Scholar] [CrossRef]
- Wang, Z.L.; Zhang, Y.; Su, G.P.; Wei, S.Z. Properties of Flue Duct Evaporation Products by Desulfurization Waste Water in Coal-fired Power Plants. J. Power Eng. 2016, 36, 894–900. [Google Scholar]
- Ma, S.C.; Yu, W.J.; Jia, S.G.; Zhang, R.P.; Chai, J.; Hua, J.Z. Spray Evaporation Characteristics of Desulfurization Waste Water with Zero Discharge in Coal-fired Power Plant. J. Power Eng. 2018, 38, 291–297. [Google Scholar]
- Szatyowicz, E.; Krasowska, M. Assessment of heavy metals leaching from fly ashes as an indicator of their agricultural use. Desalination Water Treat. 2020, 199, 288–296. [Google Scholar] [CrossRef]
- Chen, Z.; You, C.; Wang, H.; Xie, N. A novel technical route based on wet flue gas desulfurization process for flue gas dehumidification, water and heat recovery. Appl. Therm. Eng. 2020, 171, 115102. [Google Scholar] [CrossRef]
- Ma, S.C.; Chai, J.; Chen, G.D.; Yu, W.J.; Zhu, S.J. Research on desulfurization wastewater evaporation: Present and future perspectives. Renew. Sustain. Energy Rev. 2016, 58, 1143–1151. [Google Scholar]
- Barrero, F.V.; Ollero, P.; Perales, A.L.V.; Gomez-Barea, A. Catalytic Seawater Flue Gas Desulfurization Model. Environ. Sci. Technol. 2009, 43, 9393–9399. [Google Scholar] [CrossRef]
- Huang, Y.H.; Peddi, P.K.; Tang, C.; Zeng, H.; Teng, X.J.S.; Technology, P. Hybrid zero-valent iron process for removing heavy metals and nitrate from flue-gas-desulfurization wastewater. Sep. Purif. Technol. 2013, 118, 690–698. [Google Scholar] [CrossRef]
- Rubright, S.M.; Pearce, L.L.; Peterson, J. Environmental toxicology ofhydrogen sulfide. Nitric Oxide 2017, 71, 1–13. [Google Scholar] [CrossRef]
- Luo, H.T.; Wang, Q.; Nie, X.P.; Ren, H.; Shen, Z.; Xie, X.F.; Yang, Y.F. Heavy Metal Contamination in the Cultivated Oyster Crassostrea rivularis and Associated Health Risks from a Typical Mariculture Zone in the South China Sea. Bull. Environ. Contam. Toxicol. 2018, 101, 33–41. [Google Scholar] [CrossRef]
- Jiang, Y.X.; Chao, S.H.; Liu, J.W.; Yang, Y.; Chen, Y.J.; Zhang, A.C.; Cao, H.B. Source apportionment and health risk assessment of heavy metals in soil for a township in Jiangsu Province, China. Chemosphere 2017, 168, 1658–1668. [Google Scholar] [CrossRef] [PubMed]
- Sanz-Medel, A. Trace element analytical speciation in biological systems: Importance, challenges and trends. Spectrochim. Acta Part B At. Spectrosc. 1998, 53, 197–211. [Google Scholar] [CrossRef]
- Rainbow, P.S. The significance of trace metal concentrations in marine invertebrates: A need for laboratory investigation of accumulation strategies. Mar. Pollut. Bull. 1990, 21, 321–324. [Google Scholar] [CrossRef]
- Sunda, W.G. Trace metal interactions with marine phytoplankton. Biol. Oceanogr. 1989, 6, 411–442. [Google Scholar]
- Baeyens, W.; Parmentier, K.; Goeyens, L.; Ducastel, G.; Gieter, M.D.; Leermakers, M. The biogeochemical behavior of Cd, Cu, Pb and Zn in the Scheldt estuary: Results of the 1995 surveys. Hydrobiologia 1998, 366, 45–62. [Google Scholar] [CrossRef]
- Phillips, D.J.E.P. The use of biological indicator organisms to monitor trace metal pollution in marine and estuarine environments—A review. Environ. Pollut. 1977, 13, 281–317. [Google Scholar] [CrossRef]
- Zhang, W.B.; Jin, M.; Zhou, Y. Discussion on Heavy Metal Pollution Indexes in Chinese Marine Shellfish Standards. Mar. Sci. 2004, 28, 72–74. (In Chinese) [Google Scholar] [CrossRef]
- Wang, J.F.; Zhang, X.C.; Shan, B.T. Effects of Enviromental Factors on the Accumulation of Dissolved Metals in Bivalves. J. Ocean. Univ. Qingdao 2005, 35, 382–386. [Google Scholar] [CrossRef]
- Rainbow, P.S.; Wang, W.X. Trace metals in barnacles: The significance of trophic transfer. Sci. China Ser. C.-Life Sci. 2005, 48, 110–117. [Google Scholar] [CrossRef]
- Li, L.; Wang, S.J.; Shen, X.Q.; Jiang, M. Ecological risk assessment of heavy metal pollution in the water of China’s coastal shellfish culture areas. Environ. Sci. Pollut. Res. 2020, 27, 18392–18402. [Google Scholar] [CrossRef] [PubMed]
- Yap, C.K.; Ismail, A.; Tan, S.G. Effects of Total Soft Tissue and Shell Thickness on the Accumulation of Heavy Metals (Cd, Cu, Pb, and Zn) in the Green-Lipped Mussel Perna viridis. Russ. J. Mar. Biol. 2003, 29, 323–327. [Google Scholar] [CrossRef]
- Reid, D.J.; Bone, E.K. The rise and fall of oyster cultivation in the highly urbanized Georges River estuary, Sydney, Australia: A review of lessons learned. Reg. Stud. Mar. Sci. 2020, 35, 101246. [Google Scholar] [CrossRef]
- Reinfelder, J.R.; Wang, W.X.; Luoma, S.N.; Fisher, N.S. Assimilation efficiencies and turnover rates of trace elements in marine bivalves: A comparison of oysters, clams and mussels. Mar. Biol. 1997, 129, 443–452. [Google Scholar] [CrossRef]
- Zhang, P.; Ruan, H.M.; Dai, P.D.; Zhao, L.R.; Zhang, J.B. Spatiotemporal river flux and composition of nutrients affecting adjacent coastal water quality in Hainan Island, China. J. Hydrol. 2020, 591, 125293. [Google Scholar] [CrossRef]
- Zhang, P.; Wei, S.S.; Zhang, J.B.; Ou, Z.; Yang, Y.Q.; Wang, M.Y. Occurrence, Composition, and Relationships in Marine Plastic Debris on the First Long Beach Adjacent to the Land-Based Source, South China Sea. J. Mar. Sci. Eng. 2020, 8, 666. [Google Scholar] [CrossRef]
- Zhang, P.; Peng, C.H.; Zhang, J.B.; Zou, Z.B.; Zhao, H.J.W. Spatiotemporal Urea Distribution, Sources, and Indication of DON Bioavailability in Zhanjiang Bay, China. Water 2020, 12, 633. [Google Scholar] [CrossRef]
- State Oceanic Administration. The Specification for Marine Monitoring—Part 3: Sample Collection, Storage and Transportation (GB 17378.3-2007); Beijing Standards Press of China: Beijing, China, 2007; pp. 9–11. (In Chinese) [Google Scholar]
- State Oceanic Administration. The Specification for Marine Monitoring—Part 6: Organism Analysis (GB 17378.6-2007); Beijing Standards Press of China: Beijing, China, 2007; pp. 1–5. (In Chinese) [Google Scholar]
- Ying, H.; Liang, C.; Zhang, X.; Zhang, D.; Zhang, X.; Yu, Y.; Fu, J.J.E. Trace elements in fish from Taihu Lake, China: Levels, associated risks, and trophic transfer. Ecotoxicol. Environ. Saf. 2013, 90, 89–97. [Google Scholar] [CrossRef]
- State Oceanic Administration. Marine Biological Quality (GB18421-2001); Beijing Standards Press of China: Beijing, China, 2001; pp. 1–2. (In Chinese) [Google Scholar]
- Rahayu, D.R.; Anggoro, S.; Soeprobowati, T.R.; Science, E. Metal Concentrations and Bio-Concentration Factor (BCF) in Surface Water and Economical Fish Species from Wadaslintang Multipurpose Dam, Wonosobo, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 2020, 593, 012024. [Google Scholar] [CrossRef]
- Rashed, M.N. Monitoring of environmental heavy metals in fish from Nasser Lake. Environ. Int. 2002, 27, 27–33. [Google Scholar] [CrossRef]
- Zhang, J.B.A.; Zhou, F.X.; Chen, C.L.; Sun, X.L.; Shi, Y.Z.; Zhao, H.; Chen, F.J. Spatial distribution and correlation characteristics of heavy metals in the seawater, suspended particulate matter and sediments in Zhanjiang Bay, China. PLoS ONE 2018, 13, e0201414. [Google Scholar] [CrossRef] [PubMed]
- National Environmental Protection Agency and National Oceanic Administration. Seawater Quality Standard (GB 3097-1997); Press of China: Beijing, China, 1997; pp. 2–3. (In Chinese) [Google Scholar]
- State Oceanic Administration. Technical Specification of Marine Biological Quality Monitoring (HY/T 078-2005); Beijing Standards Press of China: Beijing, China, 2005; pp. 4–5. (In Chinese) [Google Scholar]
- Qu, B.X.; Song, J.M.; Yuan, H.M.; Li, X.G.; Li, N.; Duan, L.Q. Intensive anthropogenic activities had affected Daya Bay in South China Sea since the 1980s: Evidence from heavy metal contaminations. Mar. Pollut. Bull. 2018, 135, 318–331. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Huo, S.L.; Xi, B.D.; Zhang, J.T.; Wu, F.C. Heavy metal contamination in sediments from typical lakes in the five geographic regions of China: Distribution, bioavailability, and risk. Ecol. Eng. 2015, 81, 243–255. [Google Scholar] [CrossRef]
- Li, R.; Tang, C.Y.; Li, X.; Jiang, T.; Shi, Y.P.; Cao, Y.J. Reconstructing the historical pollution levels and ecological risks over the past sixty years in sediments of the Beijiang River, South China. Sci. Total Environ. 2019, 649, 448–460. [Google Scholar] [CrossRef]
- Fang, H.W.; Huang, L.; Wang, J.Y.; He, G.J.; Reible, D. Environmental assessment of heavy metal transport and transformation in the Hangzhou Bay, China. J. Hazard. Mater. 2016, 302, 447–457. [Google Scholar] [CrossRef]
- Varol, M. Assessment of heavy metal contamination in sediments of the Tigris River (Turkey) using pollution indices and multivariate statistical techniques. J. Hazard. Mater. 2011, 195, 355–364. [Google Scholar] [CrossRef]
- Yi, Y.J.; Tang, C.H.; Yi, T.; Yang, Z.F.; Zhang, S.H. Health risk assessment of heavy metals in fish and accumulation patterns in food web in the upper Yangtze River, China. Ecotoxicol. Environ. Saf. 2017, 145, 295–302. [Google Scholar] [CrossRef]
- Naser, H.A. Assessment and management of heavy metal pollution in the marine environment of the Arabian Gulf: A review. Mar. Pollut. Bull. 2013, 72, 6–13. [Google Scholar] [CrossRef]
- Romic, M.; Romic, D. Heavy metals distribution in agricultural topsoils in urban area. Environ. Geol. 2003, 43, 795–805. [Google Scholar] [CrossRef]
- Tang, W.Z.; Shan, B.Q.; Zhang, H.; Mao, Z.P. Heavy metal sources and associated risk in response to agricultural intensification in the estuarine sediments of Chaohu Lake Valley, East China. J. Hazard. Mater. 2010, 176, 945–951. [Google Scholar] [CrossRef]
- Sheppard, C.; Al-Husiani, M.; Al-Jamali, F.; Al-Yamani, F.; Zainal, K.J.M.P.B. The Persian/Arabian Gulf: A young sea in decline. Mar. Pollut. Bull. 2010, 60, 13–38. [Google Scholar] [CrossRef] [PubMed]
- Sundaray, S.K.; Nayak, B.B.; Kanungo, T.K.; Bhatta, D. Dynamics and quantification of dissolved heavy metals in the Mahanadi river estuarine system, India. Environ. Monit. Assess. 2012, 184, 1157–1179. [Google Scholar] [CrossRef] [PubMed]
- Mo, H.; Wu, L.G. Heavy Metals Migration and Monitoring Analysis for Seawater Desulphurization Process in Coal-fired Power Plant. Environ. Sci. Technol. 2013, 36, 144–148. [Google Scholar]
- Zhang, X.C.; Somg, X.H.; Nie, X.H. The study on marine environmental impact of seawater flue gas desulfurization in the coastal fire power plant. Mar. Sci. 2008, 32, 94–96. (In Chinese) [Google Scholar]
- Zhang, C.P.; Zheng, Z.Y.; Yao, S.H.; Jia, H.L.; Xian, X.H.; Wang, L. Ecological risk of heavy metals in sediment around techeng island special marine reserves in zhanjiang bay. J. Ocean Univ. China 2020, 19, 561–568. [Google Scholar] [CrossRef]
- Peng, S.T.; Wu, C.; Dai, M.X.; Zhan, S.F.; Zhou, R.; Bai, Z.P. Distribution of dissolved heavy metals in seawater of the Bohai Bay. In Proceedings of the 6th International Symposium of Asia Institute of Urban Environment: Energy Conservation and Carbon Off in Asia City, Changchun, China, 20–23 September 2009; pp. 693–696. [Google Scholar]
- Cui, Y.; Chen, B.J.; Song, Y.L. Heavy metals content in sea water and marine organisms at jiaozhou bay. Chin. J. Appl. Ecol. 1997, 8, 650–654. [Google Scholar]
- Wang, D.L.; Lin, W.F.; Yang, X.Q.; Zhai, W.D.; Dai, M.H.; Chen, C.T. Ocurrences of dissolved trace metals (Cu, Cd, and Mn) in the Pearl River Estuary (China), a large river-groundwater-estuary system. Cont. Shelf Res. 2012, 50–51, 54–63. [Google Scholar] [CrossRef]
- Wang, Z.L.; Liu, C.Q. Distribution and partition behavior of heavy metals between dissolved and acid-soluble fractions along a salinity gradient in the Changjiang Estuary, eastern China. Chem. Geol. 2003, 202, 383–396. [Google Scholar] [CrossRef]
- Che, Y.; He, Q.; Lin, W.Q. The distributions of particulate heavy metals and its indication to the transfer of sediments in the Changjiang Estuary and Hangzhou Bay, China. Mar. Pollut. Bull. 2003, 46, 123–131. [Google Scholar] [CrossRef]
- Afarin, M.; Hamzeh, M.A.; Negarestan, H. Sedimentological and Geomorphological Classification of Chabahar Coastal Area (Chabahar-Gawater). J. Persian Gulf 2015, 6, 51–64. [Google Scholar]
- Bervoets, L.; Voets, J.; Covaci, A.; Chu, S.G.; Qadah, D.; Smolders, R.; Schepens, P.; Blust, R. Use of transplanted zebra mussels (Dreissena polymorpha) to assess the bioavailability of microcontaminants in Flemish surface waters. Environ. Sci. Technol. 2005, 39, 1492–1505. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.H.; Liu, H.; Zhou, H.L.; Ma, W.D.; Han, Q.; Diao, X.P.; Xue, Q.Z. Concentration distribution and potential health risk of heavy metals in Mactra veneriformis from Bohai Bay, China. Mar. Pollut. Bull. 2015, 97, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Xie, W.Z. Study on oyster as a monitoring organism of heavy metal Cu pollution in sea. Mar. Environ. Sci. 1998, 17, 17–23. (In Chinese) [Google Scholar] [CrossRef]
- Zhu, G.W.; Chi, Q.Q.; Qin, B.Q.; Wang, W.M. Heavy-metal contents in suspended solids of Meiliang Bay, Taihu Lake and its environmental significances. J. Environ. Sci. 2005, 17, 672–675. [Google Scholar] [CrossRef]
- Giusti, L.; Williamson, A.C.; Mistry, A. Biologically available trace metals in Mytilus edulis from the coast of Northeast England. Environ. Int. 1999, 25, 969–981. [Google Scholar] [CrossRef]
- Liang, L.N.; He, B.; Jiang, G.B.; Chen, D.Y.; Yao, Z.W. Evaluation of mollusks as biomonitors to investigate heavy metal contaminations along the Chinese Bohai Sea. Sci. Total Environ. 2004, 324, 105–113. [Google Scholar] [CrossRef]
- Chan, C.Y.; Wang, W.X. Seasonal and spatial variations of biomarker responses of rock oysters in a coastal environment influenced by large estuary input. Environ. Pollut. 2018, 242, 1253–1265. [Google Scholar] [CrossRef]
- Liu, J.W.; Zhang, J.B.; Lu, S.L.; Zhang, D.; Tong, Z.C.; Yan, Y.; Hu, B.Q. Interannual variation, ecological risk and human health risk of heavy metals in oyster-cultured sediments in the Maowei Estuary, China, from 2011 to 2018. Mar. Pollut. Bull. 2020, 154, 111039. [Google Scholar] [CrossRef]
- Aslam, S.; Chan, M.W.H.; Siddiqui, G.; Boczkaj, G.; Kazmi, S.J.H.; Kazmi, M.R. A comprehensive assessment of environmental pollution by means of heavy metal analysis for oysters’ reefs at Hab River Delta, Balochistan, Pakistan. Mar. Pollut. Bull. 2020, 153, 110970. [Google Scholar] [CrossRef]
- Anil, A.C.; Wagh, A.B. Accumulation of copper and zinc by Balanus amphitrite in a tropical estuary. Mar. Pollut. Bull. 1988, 19, 177–180. [Google Scholar] [CrossRef]
- Barber, S.; Trefry, J.H. Balanus eburneus: A sensitive indicator of copper and zinc pollution in the coastal zone. Bull. Environ. Contam. Toxicol. 1981, 27, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Paez-Osuna, F.; Bojorquez-Leyva, H. Regional variations of heavy metal concentrations in tissues of barnacles from the subtropical Pacific Coast of Mexico. Environ. Int. 1999, 25, 647–654. [Google Scholar] [CrossRef]
- Chen, L.G.; Lam, J.C.W.; Zhang, X.H.; Pan, K.; Guo, C.; Lam, P.K.S.; Wang, W.X.; Liu, H.B.; Qian, P.Y. Relationship between metal and polybrominated diphenyl ether (PBDE) body burden and health risks in the barnacle Balanus amphitrite. Mar. Pollut. Bull. 2015, 100, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Reis, P.A.; Salgado, M.A.; Vasconcelos, V. Barnacle species as biomonitors of metal contamination in the northwest coast of Portugal: Ecological quality classification approach. Hum. Ecol. Risk Assess. 2017, 23, 1219–1233. [Google Scholar] [CrossRef]

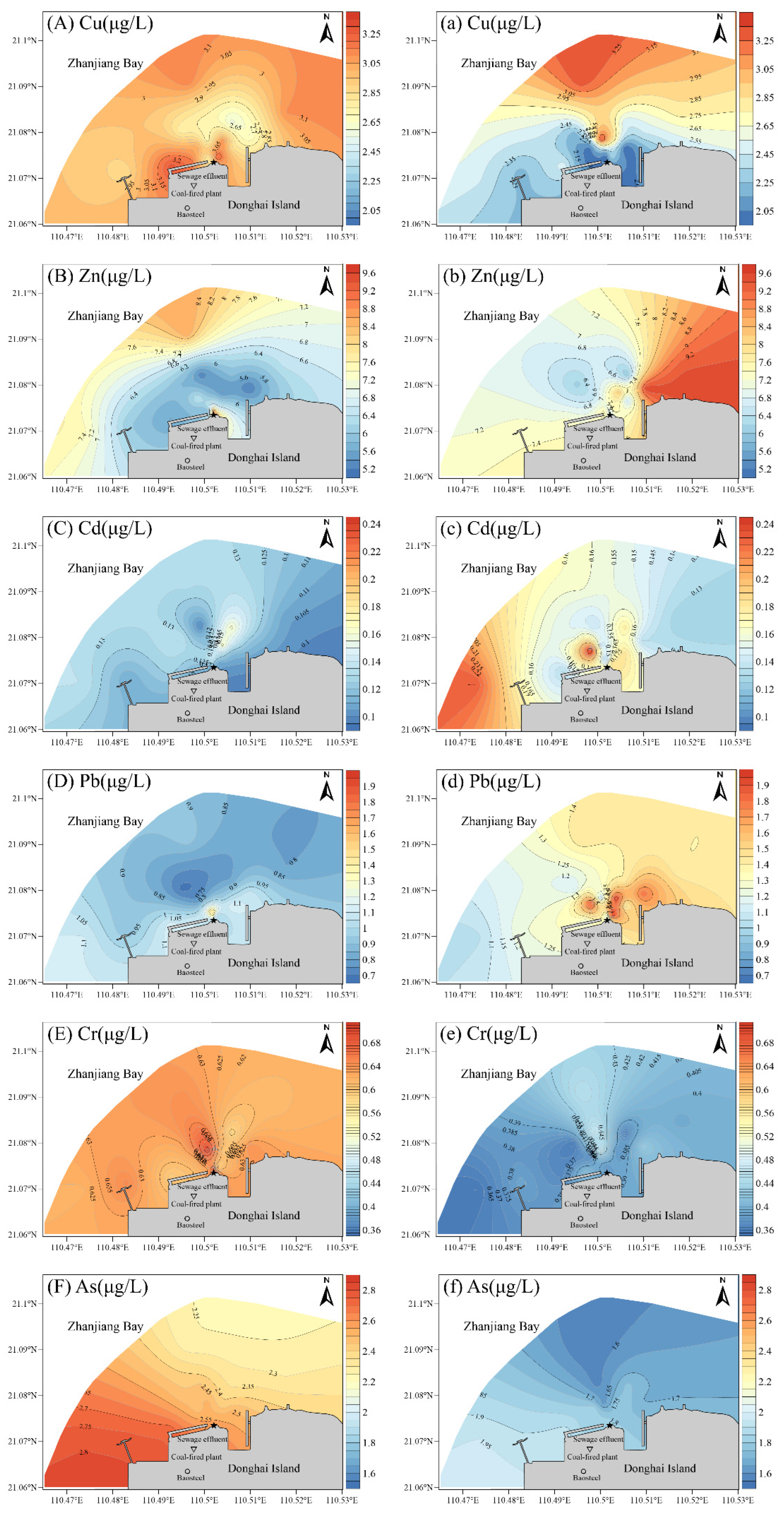



| Sampling Station | A1 | A2 | A3 | A4 | A5 | A6 | A0 |
|---|---|---|---|---|---|---|---|
| Distance | ~50 | ~100 | ~500 | ~1000 | ~800 | ~900 | ~2000 |
| Sampling Time | Layer | Cu | Zn | Cd | Pb | Cr | As |
|---|---|---|---|---|---|---|---|
| March 2015—January 2016 (Normal operation of desulfurization process) | Surface layer | 2.94 ± 0.21 | 6.42 ± 1.23 | 0.13 ± 0.02 | 1.00 ± 0.22 | 0.63 ± 0.04 | 2.51 ± 0.14 |
| Bottom layer | 2.95 ± 0.22 | 5.97 ± 1.23 | 0.12 ± 0.02 | 0.99 ± 0.23 | 0.68 ± 0.06 | 2.44 ± 0.18 | |
| February 2016—June 2016 (The desulfurization process was stopped) | Surface layer | 2.43 ± 0.42 | 7.26 ± 0.96 | 0.16 ± 0.03 | 1.42 ± 0.31 | 0.39 ± 0.03 | 1.77 ± 0.11 |
| Bottom layer | 2.57 ± 0.48 | 7.71 ± 1.83 | 0.17 ± 0.03 | 1.42 ± 0.31 | 0.41 ± 0.04 | 1.76 ± 0.09 |
| Fouling Organism | Layer | Eigenvalue | Cu | Zn | As | Cd | Pb | Cr |
|---|---|---|---|---|---|---|---|---|
| Oyster | Surface layer | Min | 3.41 | 21.88 | 0.08 | 0.06 | 0.03 | 0.08 |
| Max | 8.37 | 42.08 | 0.20 | 0.16 | 0.12 | 0.35 | ||
| M ± SD | 4.48 ± 1.61 | 33.1 ± 6.79 | 0.11 ± 0.04 | 0.1 ± 0.03 | 0.05 ± 0.03 | 0.15 ± 0.09 | ||
| Middle layer | Min | 2.31 | 27.57 | 0.07 | 0.06 | 0.03 | 0.06 | |
| Max | 9.18 | 38.76 | 0.15 | 0.13 | 0.06 | 0.34 | ||
| M ± SD | 5.06 ± 2.04 | 33.63 ± 3.85 | 0.11 ± 0.03 | 0.09 ± 0.02 | 0.04 ± 0.01 | 0.16 ± 0.09 | ||
| Barnacle | Surface layer | Min | 0.52 | 8.01 | 0.07 | 0.13 | 0.02 | 0.14 |
| Max | 3.54 | 28.49 | 0.23 | 0.84 | 0.08 | 2.00 | ||
| M ± SD | 1.61 ± 1.09 | 18.87 ± 6.70 | 0.15 ± 0.05 | 0.42 ± 0.26 | 0.04 ± 0.02 | 1.02 ± 0.63 | ||
| Middle layer | Min | 0.47 | 13.67 | 0.10 | 0.09 | 0.02 | 0.23 | |
| Max | 3.16 | 31.11 | 0.25 | 0.53 | 0.10 | 0.95 | ||
| M ± SD | 1.63 ± 0.82 | 22.34 ± 5.69 | 0.14 ± 0.05 | 0.33 ± 0.15 | 0.06 ± 0.03 | 0.58 ± 0.24 |
| Layers | Stations | Pi | |||||
|---|---|---|---|---|---|---|---|
| Cu | Zn | As | Cr | Cd | Pb | ||
| Surface layer | A1 | 0.335 | 0.842 | 0.039 | 0.174 | 0.079 | 0.059 |
| A2 | 0.141 | 0.627 | 0.024 | 0.042 | 0.056 | 0.016 | |
| A3 | 0.153 | 0.701 | 0.021 | 0.093 | 0.055 | 0.036 | |
| A4 | 0.175 | 0.626 | 0.019 | 0.053 | 0.043 | 0.025 | |
| A5 | 0.166 | 0.438 | 0.015 | 0.052 | 0.031 | 0.019 | |
| A6 | 0.149 | 0.841 | 0.017 | 0.059 | 0.041 | 0.022 | |
| A0 | 0.136 | 0.560 | 0.018 | 0.053 | 0.028 | 0.016 | |
| Middle layer | A1 | 0.367 | 0.775 | 0.030 | 0.171 | 0.065 | 0.027 |
| A2 | 0.210 | 0.725 | 0.016 | 0.068 | 0.050 | 0.015 | |
| A3 | 0.221 | 0.694 | 0.028 | 0.108 | 0.047 | 0.017 | |
| A4 | 0.218 | 0.675 | 0.023 | 0.054 | 0.042 | 0.028 | |
| A5 | 0.185 | 0.551 | 0.020 | 0.079 | 0.031 | 0.018 | |
| A6 | 0.093 | 0.720 | 0.015 | 0.041 | 0.029 | 0.025 | |
| A0 | 0.123 | 0.569 | 0.019 | 0.031 | 0.035 | 0.025 | |
| Layers | Stations | Pi | |||||
|---|---|---|---|---|---|---|---|
| Cu | Zn | As | Cr | Cd | Pb | ||
| Surface layer | A1 | 0.142 | 0.570 | 0.046 | 0.360 | 0.039 | 1.001 |
| A2 | 0.114 | 0.532 | 0.036 | 0.286 | 0.016 | 0.570 | |
| A3 | 0.071 | 0.279 | 0.027 | 0.421 | 0.012 | 0.913 | |
| A4 | 0.021 | 0.160 | 0.014 | 0.101 | 0.014 | 0.277 | |
| A5 | 0.036 | 0.307 | 0.035 | 0.149 | 0.009 | 0.305 | |
| A6 | 0.043 | 0.364 | 0.033 | 0.099 | 0.024 | 0.441 | |
| A0 | 0.024 | 0.430 | 0.022 | 0.067 | 0.021 | 0.070 | |
| Middle layer | A1 | 0.078 | 0.500 | 0.024 | 0.304 | 0.162 | 0.501 |
| A2 | 0.126 | 0.535 | 0.027 | 0.358 | 0.193 | 0.285 | |
| A3 | 0.064 | 0.477 | 0.038 | 0.270 | 0.215 | 0.456 | |
| A4 | 0.073 | 0.622 | 0.049 | 0.477 | 0.263 | 0.138 | |
| A5 | 0.028 | 0.273 | 0.025 | 0.368 | 0.198 | 0.152 | |
| A6 | 0.019 | 0.322 | 0.019 | 0.116 | 0.047 | 0.221 | |
| A0 | 0.069 | 0.396 | 0.020 | 0.132 | 0.065 | 0.035 | |
| Fouling Organism | Types of Heavy Metals | Study Area | Content (μg/g) | References |
|---|---|---|---|---|
| Oyster | Cu | Coastal water, Hong Kong | 211–825 | [63] |
| Maowei Estuary, China | 17.15 ± 8.61 | [64] | ||
| Hab River Delta, Balochistan, Pakistan | 0.791 ± 0.023 | [65] | ||
| Hab River Delta, Balochistan, Pakistan | 0.731 ± 0.045 | [65] | ||
| Zhanjing Bay, China | 14.46–39.91 | This study | ||
| Zn | Coastal water, Hong Kong | 1049–7351 | [63] | |
| Hab River Delta, Balochistan, Pakistan | 774.4 ± 9.723 | [65] | ||
| Hab River Delta, Balochistan, Pakistan | 737.8 ± 12.2 | [65] | ||
| Zhanjing Bay, China | 149.3–262.72 | This study | ||
| Cd | Coastal water, Hong Kong | 0.43–5.88 | [63] | |
| Maowei Estuary, China | 0.16 ± 0.14 | [64] | ||
| Hab River Delta, Balochistan, Pakistan | 0.027 ± 0.004 | [65] | ||
| Hab River Delta, Balochistan, Pakistan | 0.019 ± 0.003 | [65] | ||
| Zhanjing Bay, China | 0.35–0.68 | This study | ||
| Pb | Maowei Estuary, China | 17.58 ± 10.82 | [64] | |
| Hab River Delta, Balochistan, Pakistan | 1.322 ± 0.148 | [65] | ||
| Hab River Delta, Balochistan, Pakistan | 1.070 ± 0.092 | [65] | ||
| Zhanjing Bay, China | 0.15–0.51 | This study | ||
| Cr | Hab River Delta, Balochistan, Pakistan | 0.471 ± 0.039 | [65] | |
| Hab River Delta, Balochistan, Pakistan | 0.381 ± 0.030 | [65] | ||
| Zhanjing Bay, China | 0.40–1.51 | This study | ||
| As | Maowei Estuary, China | 10.27 ± 5.24 | [64] | |
| Hab River Delta, Balochistan, Pakistan | 2.059 ± 0.107 | [65] | ||
| Hab River Delta, Balochistan, Pakistan | 1.696 ± 0.031 | [65] | ||
| Zhanjing Bay, China | 0.42–0.85 | This study | ||
| Barnacle | Cu | West coast of India | 805 | [66] |
| Indian River Lagoon (Florida Coast) | 1937 | [66] | ||
| Mazatlan piers, Mexico | 11–64 | [67] | ||
| Topolobampo piers, Mexico | 10083 | [68] | ||
| Hebe Haven piers in Hong Kong | 915.1 ± 104.5 | [69] | ||
| northwest coast of Portugal | 0.93–5.70 | [70] | ||
| northwest coast of Portugal | 0.76–6.09 | [70] | ||
| Zhanjing Bay, China | 2.59–15.02 | This study | ||
| Zn | West coast of India | 3618 | [68] | |
| Mazatlan piers, Mexico | 29 | [68] | ||
| Guaymas Harbour, Mexico | 21.8 | [68] | ||
| Hebe Haven piers in Hong Kong | 18,440.4 ± 3089.4 | [69] | ||
| Tso Wo Hang piers in Hong Kong | 12,929.4 ± 1372.8 | [69] | ||
| Sai Kung piers in Hong Kong | 10,855.2 ± 913.7 | [69] | ||
| northwest coast of Portugal | 119–782 | [70] | ||
| northwest coast of Portugal | 413–976 | [70] | ||
| Zhanjing Bay, China | 40.07–155.54 | This study | ||
| Cd | Puerto Vallarta, Mexico | 63.1 | [68] | |
| Mazatlan Harbours, Mexico | 9.2–50.2 | [68] | ||
| northwest coast of Portugal | 0.39–1.98 | [70] | ||
| northwest coast of Portugal | 0.35–3.75 | [70] | ||
| Zhanjing Bay, China | 0.67–3.51 | This study | ||
| Pb | Puerto Vallarta, Mexico | 29.2 | [68] | |
| Topolobampo, Mexico | 25 | [68] | ||
| Guaymas Harbour, Mexico | 15.9 | [68] | ||
| Zhanjing Bay, China | 0.09–0.40 | This study | ||
| Cr | Coast of Mexico | <3.8 | [68] | |
| northwest coast of Portugal | 0.25–1.79 | [70] | ||
| northwest coast of Portugal | 0.45–3.13 | [70] | ||
| Zhanjing Bay, China | 0.70–8.01 | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Peng, D.; Zhang, P.; Rong, Y.; Hu, L.; Zhao, L.; Chen, C. Metal Content and Enrichment in Bivalves within the Drainage Area of Seawater Used for a Desulfurization Process in Zhanjiang Bay, China. Water 2022, 14, 2532. https://doi.org/10.3390/w14162532
Zhang J, Peng D, Zhang P, Rong Y, Hu L, Zhao L, Chen C. Metal Content and Enrichment in Bivalves within the Drainage Area of Seawater Used for a Desulfurization Process in Zhanjiang Bay, China. Water. 2022; 14(16):2532. https://doi.org/10.3390/w14162532
Chicago/Turabian StyleZhang, Jibiao, Demeng Peng, Peng Zhang, Yumei Rong, Lifang Hu, Lirong Zhao, and Chunliang Chen. 2022. "Metal Content and Enrichment in Bivalves within the Drainage Area of Seawater Used for a Desulfurization Process in Zhanjiang Bay, China" Water 14, no. 16: 2532. https://doi.org/10.3390/w14162532
APA StyleZhang, J., Peng, D., Zhang, P., Rong, Y., Hu, L., Zhao, L., & Chen, C. (2022). Metal Content and Enrichment in Bivalves within the Drainage Area of Seawater Used for a Desulfurization Process in Zhanjiang Bay, China. Water, 14(16), 2532. https://doi.org/10.3390/w14162532






