Hydrological Regime and Fish Predation Regulate the Zooplankton Community Size Structure in a Tropical Floodplain Lake
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Field Sampling
2.3. Data Analysis
3. Results
3.1. Hydrological Conditions
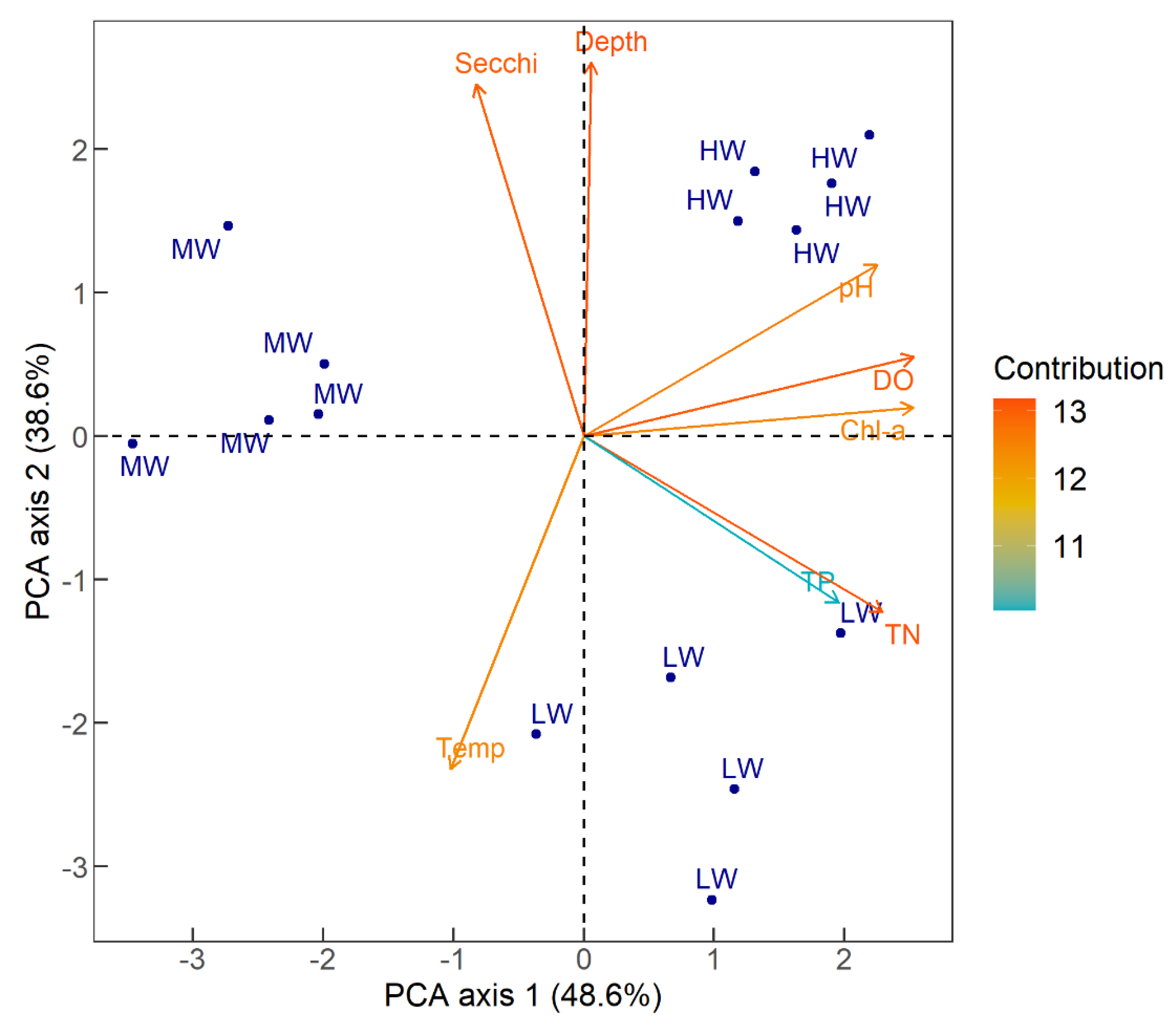
3.2. Physical and Chemical Parameters
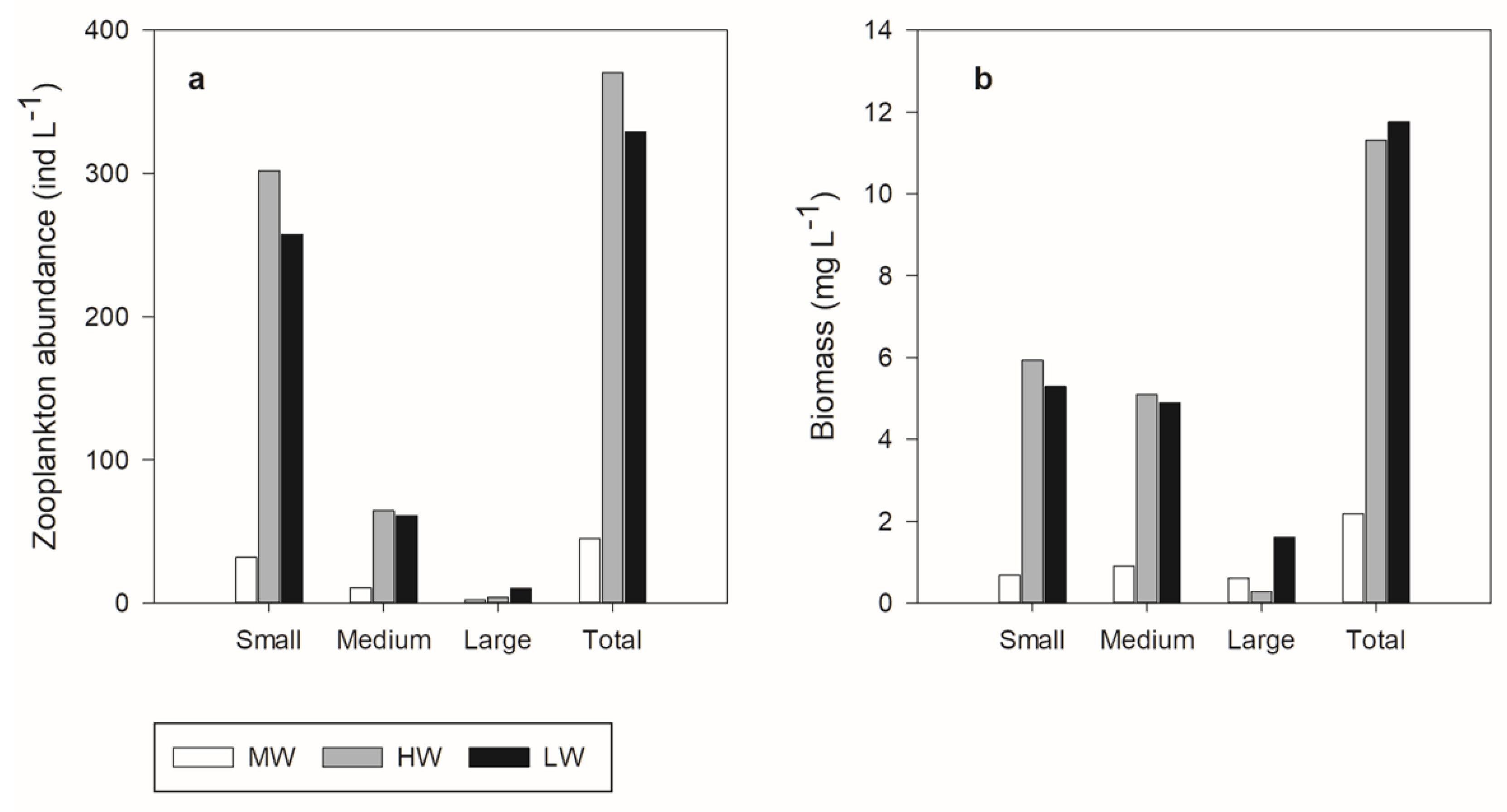
3.3. Zooplankton Community Size Structure
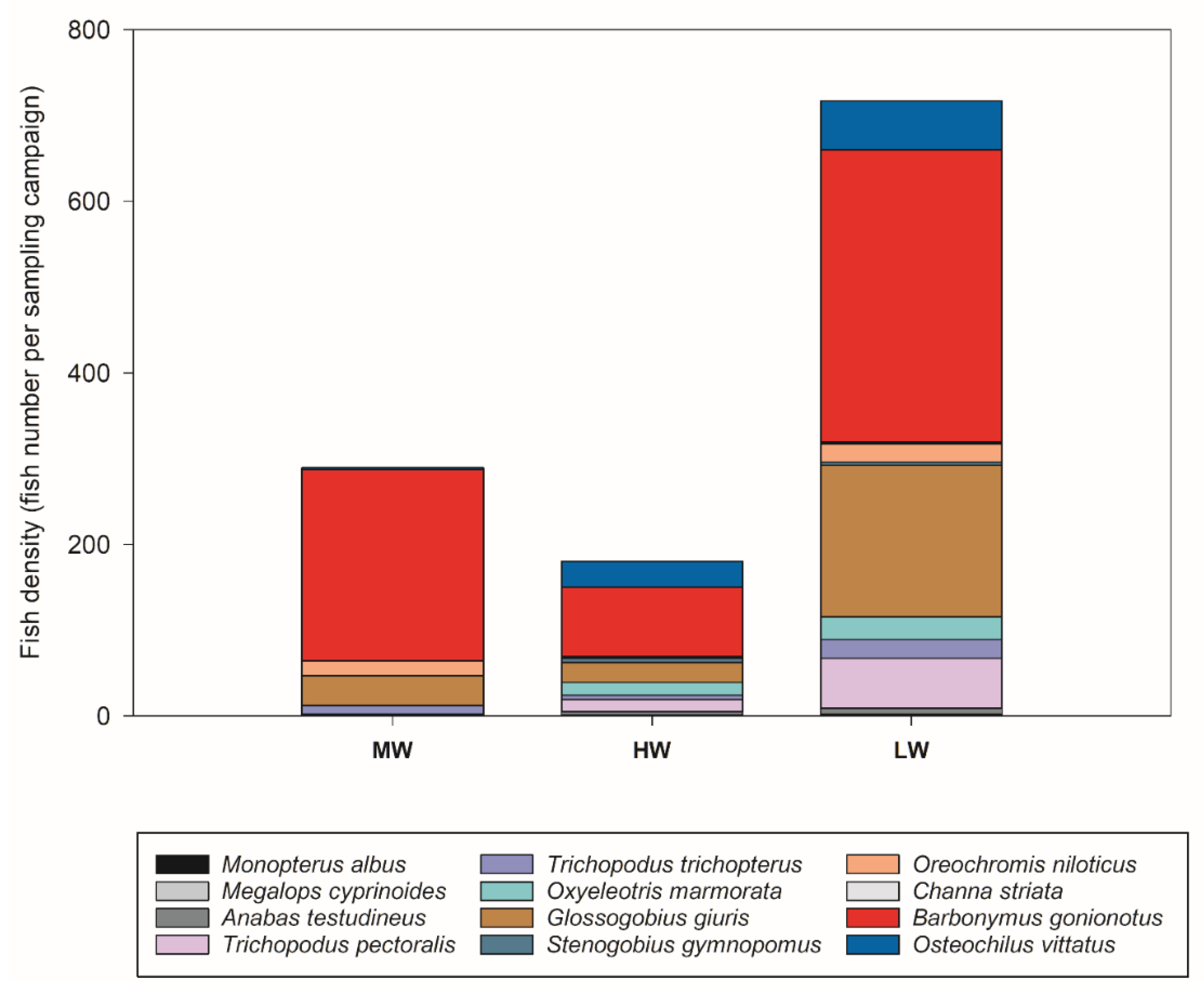
3.4. Fish Assemblages
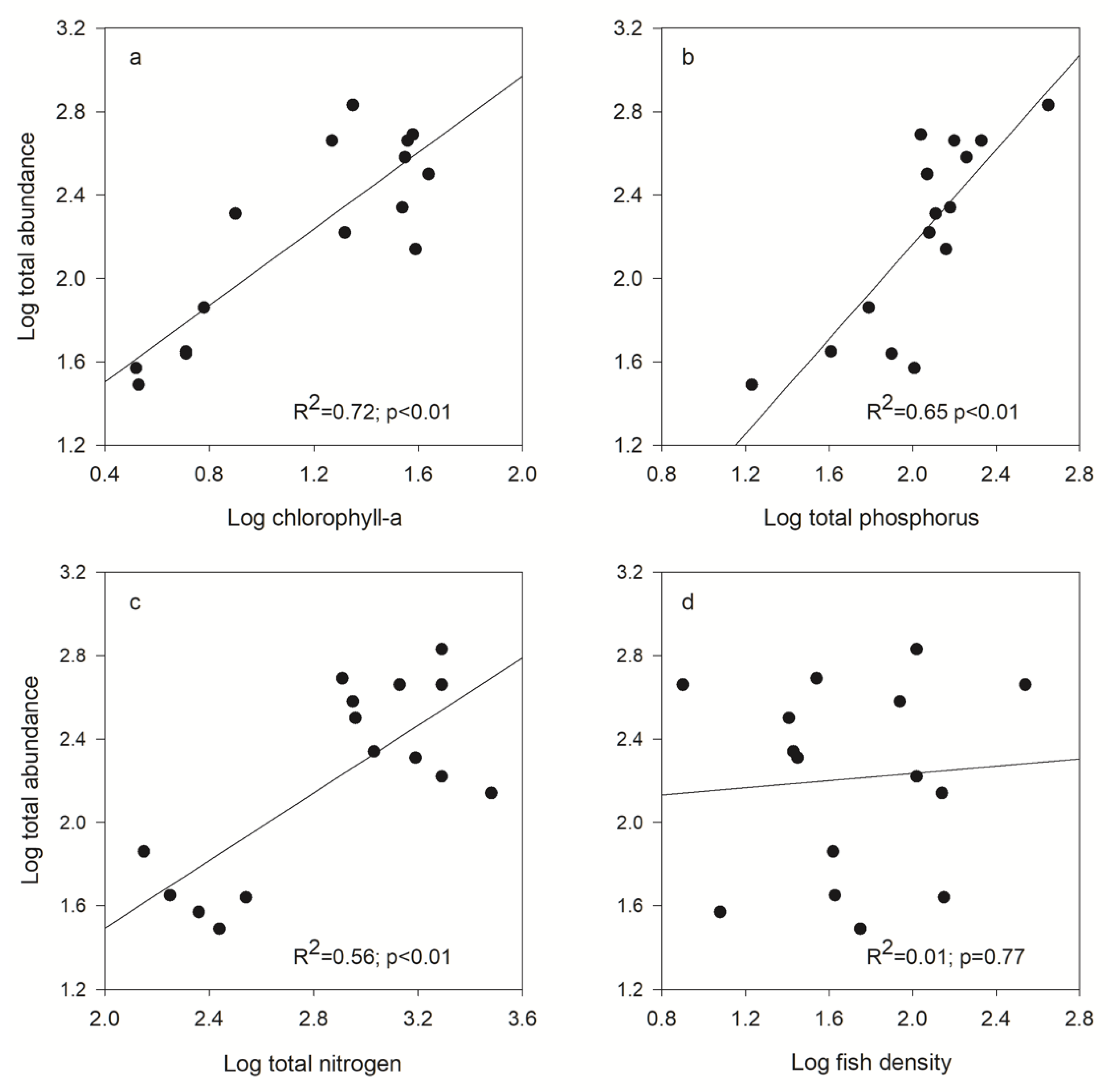
3.5. Correlation between Zooplankton Community, Fish Density, and Resource Availability
4. Discussion
4.1. Hydrological Regimes and Zooplankton Community
4.2. Environmental Variables Related to Temporal Variation in Zooplankton Community Size Structure
4.3. Effect of Fish Predation on Zooplankton Size Structure
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vörösmarty, C.J.; McIntyre, P.B.; Gessner, M.O.; Dudgeon, D.; Prusevich, A.; Green, P.; Glidden, S.; Bunn, S.E.; Sullivan, C.A.; Liermann, C.R.; et al. Global threats to human water security and river biodiversity. Nature 2010, 467, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Strayer, D.L.; Dudgeon, D. Freshwater biodiversity conservation: Recent progress and future challenges. J. N. Am. Benthol. Soc. 2010, 29, 344–358. [Google Scholar] [CrossRef]
- Junk, W.J.; Bayley, P.B.; Sparks, R.E. The flood pulse concept in River-Floodplain Systems. Can. Spec. Publ. Fish. Aquat. Sci. 1989, 106, 110–127. [Google Scholar]
- Golec-Fialek, C.; Lansac-Tôha, F.M.; Bonecker, C.C. Response of the zooplankton community to extreme hydrological variations in a temporary lake in a neotropical floodplain system. Limnologica 2021, 86, 125834. [Google Scholar] [CrossRef]
- Chaparro, G.; O’Farrell, I.; Hein, T. Multi-scale analysis of functional plankton diversity in floodplain wetlands: Effects of river regulation. Sci. Total Environ. 2019, 667, 338–347. [Google Scholar] [CrossRef]
- Tockner, K.; Pusch, M.; Borchardt, D.; Lorang, M.S. Multiple stressors in coupled river–floodplain ecosystems. Freshw. Biol. 2010, 55, 135–151. [Google Scholar] [CrossRef]
- Mayora, G.; Schneider, B.; Rossi, A. Turbidity and dissolved organic matter as significant predictors of spatio-temporal dynamics of phosphorus in a large river-floodplain system. River Res. Appl. 2018, 34, 629–639. [Google Scholar] [CrossRef]
- Dias, J.D.; Simões, N.R.; Meerhoff, M.; Lansac-Tôha, F.A.; Velho, L.F.M.; Bonecker, C.C. Hydrological dynamics drives zooplankton metacommunity structure in a Neotropical floodplain. Hydrobiologia 2016, 781, 109–125. [Google Scholar] [CrossRef]
- Schöll, K.; Kiss, A.; Dinka, M.; Berczik, Á. Flood-Pulse Effects on Zooplankton Assemblages in a River-Floodplain System (Gemenc Floodplain of the Danube, Hungary). Int. Rev. Hydrobiol. 2012, 97, 41–54. [Google Scholar] [CrossRef]
- Shiel, R.J.; Costelloe, J.F.; Reid, J.R.W.; Hudson, P.; Powling, J. Zooplankton diversity and assemblages in arid zone rivers of the Lake Eyre Basin, Australia. Mar. Freshw. Res. 2006, 57, 49–60. [Google Scholar] [CrossRef]
- Pinel-Alloul, B.; Patoine, A.; Marty, J. Multi-scale and multi-system perspectives of zooplankton structure and function in Canadian freshwaters. Can. J. Fish. Aquat. Sci. 2021, 78, 1543–1562. [Google Scholar] [CrossRef]
- Dodson, S.; Everhart, W.; Jandl, A.; Krauskopf, S. Effect of watershed land use and lake age on zooplankton species richness. Hydrobiologia 2007, 579, 393–399. [Google Scholar] [CrossRef]
- Jeppesen, E.; Lauridsen, T.; Mitchell, S.F.; Burns, C.W. Do planktivorous fish structure the zooplankton communities in New Zealand lakes? N. Zeal. J. Mar. Freshw. Res. 1997, 31, 163–173. [Google Scholar] [CrossRef]
- Molinari, B.; Stewart-Koster, B.; Adame, M.F.; Campbell, M.D.; McGregor, G.; Schulz, C.; Malthus, T.J.; Bunn, S. Relationships between algal primary productivity and environmental variables in tropical floodplain wetlands. Inland Waters 2021, 11, 180–190. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, C.; Zhao, J.; Xia, J.; Yu, Q.; Eamus, D. Zooplankton in highly regulated rivers: Changing with water environment. Ecol. Eng. 2013, 58, 323–334. [Google Scholar] [CrossRef]
- Mohan Dey, M.; Rab, M.A.; Paraguas, F.J.; Bhatta, R.; Ferdous Alam, M.; Koeshendrajana, S.; Ahmed, M. Status and economics of freshwater aquaculture in selected countries of Asia. Aquac. Econ. Manag. 2005, 9, 11–37. [Google Scholar] [CrossRef]
- Cao, L.; Wang, W.; Yang, Y.; Yang, C.; Yuan, Z.; Xiong, S.; Diana, J. Environmental impact of aquaculture and countermeasures to aquaculture pollution in China. Environ. Sci. Pollut. Res. Int. 2007, 14, 452–462. [Google Scholar]
- Jeppesen, E.; Nõges, P.; Davidson, T.; Haberman, J.; Nõges, T.; Blank, K.; Lauridsen, T.; Søndergaard, M.; Sayer, C.; Laugaste, R.; et al. Zooplankton as indicators in lakes: A scientific-based plea for including zooplankton in the ecological quality assessment of lakes according to the European Water Framework Directive (WFD). Hydrobiologia 2011, 676, 279–297. [Google Scholar] [CrossRef]
- Van Egeren, S.; Dodson, S.; Torke, B.; Maxted, J. The relative significance of environmental and anthropogenic factors affecting zooplankton community structure in Southeast Wisconsin Till Plain lakes. Hydrobiologia 2011, 668, 137–146. [Google Scholar] [CrossRef]
- Lampert, W. Zooplankton research: The contribution of limnology to general ecological paradigms. Aquat. Ecol. 1997, 31, 19–27. [Google Scholar] [CrossRef]
- Jeppesen, E.; Søndergaard, M.; Jensen, J.P.; Mortensen, E.; Sortkjær, O. Fish-induced changes in zooplankton grazing on phytoplankton and bacterioplankton: A long-term study in shallow hypertrophic Lake Søbygaard. J. Plankton Res. 1996, 18, 1605–1625. [Google Scholar] [CrossRef]
- Bertolo, A. Predation on Zooplankton. In Encyclopedia of Inland Waters, 2nd ed.; Mehner, T., Tockner, K., Eds.; Elsevier: Oxford, UK, 2022; pp. 196–206. [Google Scholar]
- Brooks, J.L.; Dodson, S.I. Predation, Body Size, and Composition of Plankton. Science 1965, 150, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Ghadouani, A.; Pinel-Alloul, B.; Prepas, E.E. Could increased cyanobacterial biomass following forest harvesting cause a reduction in zooplankton body size structure? Can. J. Fish. Aquat. Sci. 2006, 63, 2308–2317. [Google Scholar] [CrossRef]
- Finlay, B.J.; Esteban, G.F. Body size and biogegraphy. In Body Size: The Structure and Function of Aquatic Ecosystems; Hildrew, A.G., Raffaelli, D.G., Edmonds-Brow, R., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 167–185. [Google Scholar]
- Wu, B.; Dai, S.; Wen, X.; Qian, C.; Luo, F.; Xu, J.; Wang, X.; Li, Y.; Xi, Y. Chlorophyll-nutrient relationship changes with lake type, season and small-bodied zooplankton in a set of subtropical shallow lakes. Ecol. Indic. 2022, 135, 108571. [Google Scholar] [CrossRef]
- Dodson, S.I. Zooplankton Competition and Predation: An Experimental Test of the Size-Efficiency Hypothesis. Ecology 1974, 55, 605–613. [Google Scholar] [CrossRef]
- Ersoy, Z.; Jeppesen, E.; Sgarzi, S.; Arranz, I.; Cañedo-Argüelles, M.; Quintana, X.D.; Landkildehus, F.; Lauridsen, T.L.; Bartrons, M.; Brucet, S. Size-based interactions and trophic transfer efficiency are modified by fish predation and cyanobacteria blooms in Lake Mývatn, Iceland. Freshw. Biol. 2017, 62, 1942–1952. [Google Scholar]
- Bonecker, C.C.; Azevedo, F.d.; Simões, N.R. Zooplankton body-size structure and biomass in tropical floodplain lakes: Relationship with planktivorous fishes. Acta Limnol. Bras. 2011, 23, 217–228. [Google Scholar] [CrossRef]
- Zhang, J.; Xie, P.; Tao, M.; Guo, L.; Chen, J.; Li, L.; Zhang, X.; Zhang, L. The Impact of Fish Predation and Cyanobacteria on Zooplankton Size Structure in 96 Subtropical Lakes. PLoS ONE 2013, 8, e76378. [Google Scholar] [CrossRef]
- Liu, X.; Dur, G.; Ban, S.; Sakai, Y.; Ohmae, S.; Morita, T. Planktivorous fish predation masks anthropogenic disturbances on decadal trends in zooplankton biomass and body size structure in Lake Biwa, Japan. Limnol. Oceanogr. 2020, 65, 667–682. [Google Scholar] [CrossRef]
- Litchman, E.; Ohman, M.D.; Kiørboe, T. Trait-based approaches to zooplankton communities. J. Plankton Res. 2013, 35, 473–484. [Google Scholar] [CrossRef]
- Peters, R.H. The Ecological Implication of Body Size, 1st ed.; Cambridge University Press: Cambridge, UK, 1983. [Google Scholar]
- Finlay, K.; Beisner, B.E.; Patoine, A.; Pinel-Alloul, B. Regional ecosystem variability drives the relative importance of bottom-up and top-down factors for zooplankton size spectra. Can. J. Fish. Aquat. Sci. 2007, 64, 516–529. [Google Scholar] [CrossRef]
- Sato, K.; Matsuno, K.; Arima, D.; Abe, Y.; Yamaguchi, A. Spatial and temporal changes in zooplankton abundance, biovolume, and size spectra in the neighboring waters of Japan: Analyses using an optical plankton counter. Zool. Stud. 2015, 54, 18. [Google Scholar] [CrossRef] [PubMed]
- Braun, L.-M.; Brucet, S.; Mehner, T. Top-down and bottom-up effects on zooplankton size distribution in a deep stratified lake. Aquat. Ecol. 2021, 55, 527–543. [Google Scholar] [CrossRef]
- Fernando, C.H. Zooplankton, fish and fisheries in tropical freshwaters. Hydrobiologia 1994, 272, 105–123. [Google Scholar] [CrossRef]
- Toruan, R.L.; Setiawan, F. Hydrological Regimes and Zooplankton Ecology at Tempe Floodplains, Indonesia: Preliminary Study Before the Operation of the Downstream Barrage. In Trends in Asian Water Environmental Science and Technology; Kurisu, F., Ramanathan, A.L., Kazmi, A.A., Kumar, M., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 117–125. [Google Scholar]
- Sukmayanti. Evaluasi perikanan tangkap dan prospek pengembangannya di Danau Tempe. In Research Report; Faculty of Fishery, Universitas Cokroaminoto: Makassar, Indonesia, 2007. [Google Scholar]
- Ramadhan, A.; Triyanti, R.; Koeshendrajana, S. Karakteristik dan nilai ekonomi sumberdaya perikanan komplek Danau Tempe. J. Kebijak. Dan Ris. Soc. Ekon. Kelaut. Perikan. 2008, 3, 89–102. [Google Scholar]
- Jasalesmana, T.; Nomosatryo, S.; Harsono, E.; Dina, R.; Ali, F. Keterkaitan Parameter Fisika, Kimia dan Biologi di Danau Tempe, Sulawesi Selatan. In Proceedings of the Seminar Nasional Limnologi VII, Bogor, Indonesia, 16 September 2014; Subehi, L., Haryani, G.S., Henny, C., Wibowo, H., Eds.; Pusat Penelitian Limnologi LIPI: Bogor, Indonesia, 2014. [Google Scholar]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Public Health Association: Washington, DC, USA, 2013. [Google Scholar]
- Setiawan, F.; Wibowo, H. Karakteristik Danau Tempe Sebagai Danau Paparan Banjir. In Proceedings of the Pertemuan Ilmiah Masyarakat Limnologi, Cibinong, Indonesia, 3 December 2013; Chrismadha, T., Sulawesty, F., Fakhrudin, M., Nofdianto, Said, D.S., Eds.; Pusat Penelitian Limnologi: Cibinong, Indonesia, 2013. [Google Scholar]
- Catlin, A.K.; Collier, K.J.; Duggan, I.C. Zooplankton generation following inundation of floodplain soils: Effects of vegetation type and riverine connectivity. Mar. Freshw. Res. 2017, 68, 76–86. [Google Scholar] [CrossRef]
- Tockner, K.; Malard, F.; Ward, J.V. An extension of the flood pulse concept. Hydrol. Process. 2000, 14, 2861–2883. [Google Scholar] [CrossRef]
- Górski, K.; Collier, K.J.; Duggan, I.C.; Taylor, C.M.; Hamilton, D.P. Connectivity and complexity of floodplain habitats govern zooplankton dynamics in a large temperate river system. Freshw. Biol. 2013, 58, 1458–1470. [Google Scholar] [CrossRef]
- Ning, N.P.; Gawne, B.; Cook, R.; Nielsen, D. Zooplankton dynamics in response to the transition from drought to flooding in four Murray–Darling Basin rivers affected by differing levels of flow regulation. Hydrobiologia 2013, 702, 45–62. [Google Scholar] [CrossRef]
- Dodson, S.I.; Lillie, R.A.; Will-Wolf, S. Land Use, Water Chemistry, Aquatic Vegetation, and Zooplankton Community Structure of Shallow Lakes. Ecol. Appl. 2005, 15, 1191–1198. [Google Scholar] [CrossRef]
- Evans, L.E.; Hirst, A.G.; Kratina, P.; Beaugrand, G. Temperature-mediated changes in zooplankton body size: Large scale temporal and spatial analysis. Ecography 2020, 43, 581–590. [Google Scholar] [CrossRef]
- Pavluk, T.; Bij de Vaate, A. Trophic Index and Efficiency. In Encyclopedia of Ecology, 2nd ed.; Fath, B., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 495–502. [Google Scholar]
- Badosa, A.; Boix, D.; Brucet, S.; López-Flores, R.; Gascón, S.; Quintana, X.D. Zooplankton taxonomic and size diversity in Mediterranean coastal lagoons (NE Iberian Peninsula): Influence of hydrology, nutrient composition, food resource availability and predation. Estuar. Coast. Shelf Sci. 2007, 71, 335–346. [Google Scholar] [CrossRef]
- Napiórkowski, P.; Bąkowska, M.; Mrozińska, N.; Szymańska, M.; Kolarova, N.; Obolewski, K. The Effect of Hydrological Connectivity on the Zooplankton Structure in Floodplain Lakes of a Regulated Large River (the Lower Vistula, Poland). Water 2019, 11, 1924. [Google Scholar] [CrossRef]
- Li, H.; Gu, Y.; Cai, Q.; Dong, X.; Ye, L. Zooplankton Size Structure in Relation to Environmental Factors in the Xiangxi Bay of Three Gorges Reservoir, China. Front. Ecol. Evol. 2022, 10. [Google Scholar] [CrossRef]
- Carpenter, S.R.; Kitchell, J.F.; Hodgson, J.R. Cascading Trophic Interactions and Lake Productivity. BioScience 1985, 35, 634–639. [Google Scholar] [CrossRef]
- Schindler, D.W. Evolution of Phosphorus Limitation in Lakes. Science 1977, 195, 260–262. [Google Scholar] [CrossRef]
- Collins, S.F.; Wahl, D.H. Size-specific effects of bighead carp predation across the zooplankton size spectra. Freshw. Biol. 2018, 63, 700–708. [Google Scholar] [CrossRef]


| Zone | Description |
|---|---|
| Zone 1 (T1) |
|
| Zone 2 (T2) |
|
| Zone 3 (T3) |
|
| Zone 4 (T4) |
|
| Zone 5 (T5) |
|
| Characteristic | Moderate-Water | High-Water | Low-Water | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| pH | 7.6 | 0.26 | 8.76 | 0.41 | 8.09 | 0.44 |
| Electric conductivity (µS cm−1) | 158.4 | 0.55 | 153 | 8.33 | 196 | 4.95 |
| Dissolved oxygen (mg L−1) | 5.21 | 0.79 | 8.17 | 0.64 | 6.98 | 1.14 |
| Temperature (°C) | 29.9 | 0.92 | 28.13 | 0.36 | 30.54 | 0.81 |
| Chlorophyll-a (mg m−3) | 3.57 | 1.18 | 36.38 | 3.5 | 20.79 | 11.2 |
| Total phosphorus (µg L−1) | 59.20 | 32.93 | 141.80 | 30.77 | 208.80 | 134.63 |
| Total nitrogen (µg L−1) | 231.40 | 79.87 | 1006.50 | 219.07 | 2092.40 | 561.97 |
| Secchi depth (cm) | 69 | 17.08 | 69.6 | 5.55 | 17.2 | 2.9 |
| Depth (cm) | 211 | 4.97 | 313.80 | 16.54 | 111.14 | 8.53 |
| Factor | One-Way ANOVA, Fisher’s LSD (α = 0.05) | ||
|---|---|---|---|
| MW vs. HW | MW vs. LW | HW vs. LW | |
| Abiotic | |||
| Total nitrogen (TN) Total phosphorus (TP) Chlorophyll-a (chl-a) Secchi depth Mean depth | p < 0.01 NS p < 0.01 NS p < 0.01 | p <0.01 p = 0.01 p = 0.01 p < 0.01 p < 0.01 | p < 0.01 NS p <0.01 p < 0.01 p < 0.01 |
| Biotic | |||
| Zooplankton total abundance Zooplankton abundance small-sized Zooplankton abundance intermediate-sized Zooplankton abundance large sized Zooplankton total biomass Zooplankton biomass small-sized Zooplankton biomass medium-sized Zooplankton biomass large-sized Fish density | p < 0.01 p < 0.01 p < 0.01 p = 0.02 p = 0.02 p < 0.01 p = 0.02 NS p < 0.01 | p = 0.01 p = 0.01 p = 0.01 p = 0.01 p = 0.01 p = 0.01 p = 0.02 p = 0.04 p < 0.01 | NS NS NS NS p = 0.04 NS NS p = 0.01 p < 0.01 |
| Parameter | Hydrological Periods | ||
|---|---|---|---|
| Moderate-Water | High-Water | Low-Water | |
| Abundance (ind L−1) | |||
| Total | 44.74 (±15.82) | 370.34 (±107.17) | 328 (±233.505) |
| 300–500 µm (Small) | 31.84 (±8.26) | 301.82 (±80.99) | 257.39 (±188.45) |
| 501–750 µm (Medium) | 10.76 (±6.86)) | 64.33 (±30.75) | 61.14 (±39.28) |
| >750 µm (Large) | 2.14 (±1.16) | 4.19 (±1.87) | 10.17 (±7.05) |
| Biomass (mg L−1) | |||
| Total | 2.18 (±0.78) | 11.30 (±3.88) | 11.77 (±8.01) |
| 300–500 µm (Small) | 0.68 (±0.22) | 5.93 (±1.64) | 5.29 (±3.91) |
| 501–750 µm (Medium) | 0.90 (±0.58) | 5.10 (±2.48) | 4.88 (±3.08) |
| >750 µm (Large) | 0.63 (±0.54) | 0.27 (±0.18) | 1.59 (±1.06) |
| NBSS parameters | |||
| Slope (a) | −1.26 | −1.73 | −1.21 |
| Intercept (b) | 3.34 | 4.49 | 3.97 |
| Species | Fish Total Abundance | ||
|---|---|---|---|
| Moderate-Water (March) | High-Water (July) | Low-Water (September) | |
| Anabas testudineus Barbonymus gonionotus Channa striata Glossogobius giuris Megalops cyprinoides Monopterus albus Oreochromis niloticus Osteochilus vittatus Oxyeleotris marmorata Stenogobius gymnopomus Trichopodus pectoralis Trichopodus trichopterus | 0 (0%) 223 (77.2%) 0 (0%) 35 (12%) 0 (0.0%) 0 (0 %) 17 (6%) 2 (0.7%) 0 (0%) 0 (0) 2 (0.7%) 10 (3.5%) | 4 (2.2%) 81 (45%) 1 (0.6%) 23 (12.8%) 0 (0 %) 1(0.6%) 1 (0.6%) 30 (16.7%) 15 (8.3%) 5 (2.8%) 14 (7.8%) 5 (2.8%) | 7 (1%) 341 (47.6%) 2 (0.3%) 177 (24.7%) 1 (0.1%) 1 (0.1%) 21 (2.9%) 57 (7.9%) 26 (2.9%) 4 (0.6%) 2 (0.3%) 22 (3.1%) |
| Total abundance Total number of species | 256 6 | 180 11 | 715 12 |
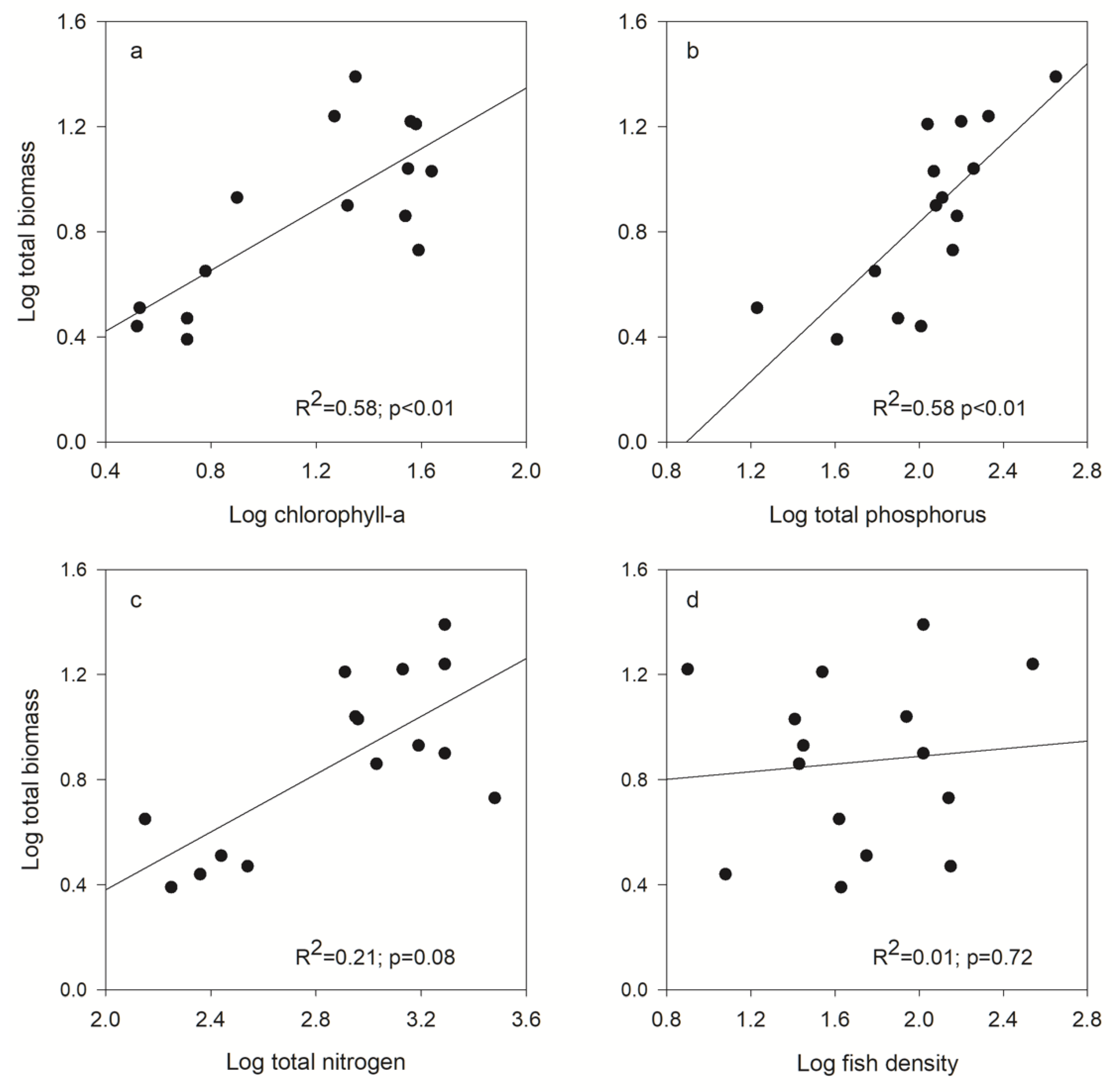
| Zooplankton Size Spectra | Chl-a | TP | TN | Fish Density |
|---|---|---|---|---|
| Biomass | ||||
| Total | R2 = 0.58; p < 0.01 | R2 = 0.58; p < 0.01 | R2 = 0.21; p = 0.08 | R2 = 0.01; p = 0.77 |
| Small | R2 = 0.67; p < 0.01 | R2 = 0.67; p < 0.01 | R2 = 0.53; p < 0.01 | R2 = 0.02; p = 0.61 |
| Intermediate | R2 = 0.54; p < 0.01 | R2 = 0.53; p < 0.01 | R2 = 0.44; p < 0.01 | R2 = 0.01; p = 0.76 |
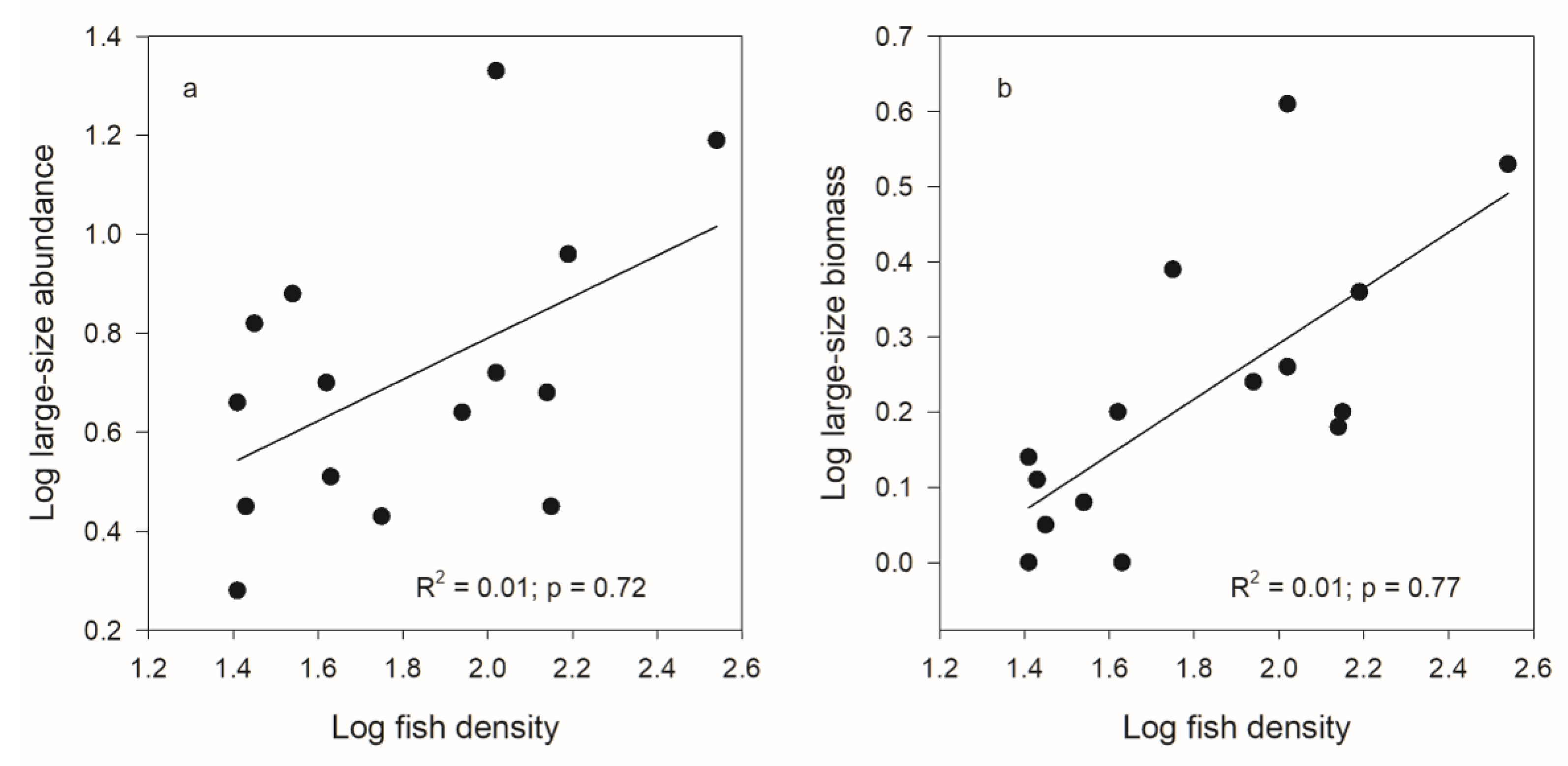
| Large | R2 = 0.01; p = 0.63 | R2 = 0.48; p < 0.01 | R2 = 0.15; p = 0.22 | R2 = 0.51; p < 0.01 |
| Abundance | ||||
| Total | R2 = 0.72; p < 0.01 | R2 = 0.65; p < 0.01 | R2 = 0.56; p < 0.01 | R2 = 0.01; p = 0.72 |
| Small | R2 = 0.74; p < 0.01 | R2 = 0.65; p < 0.01 | R2 = 0.59; p < 0.01 | R2 = 0.01; p = 0.73 |
| Intermediate | R2 = 0.59; p < 0.01 | R2 = 0.63; p < 0.01 | R2 = 0.46; p < 0.01 | R2 = 0.01; p = 0.75 |
| Large | R2 = 0.18; p < 0.12 | R2 = 0.42; p < 0.01 | R2 = 0.38; p = 0.02 | R2 = 0.25; p = 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toruan, R.L.; Dina, R.; Coggins, L.X.; Ghadouani, A. Hydrological Regime and Fish Predation Regulate the Zooplankton Community Size Structure in a Tropical Floodplain Lake. Water 2022, 14, 2518. https://doi.org/10.3390/w14162518
Toruan RL, Dina R, Coggins LX, Ghadouani A. Hydrological Regime and Fish Predation Regulate the Zooplankton Community Size Structure in a Tropical Floodplain Lake. Water. 2022; 14(16):2518. https://doi.org/10.3390/w14162518
Chicago/Turabian StyleToruan, Reliana Lumban, Rahmi Dina, Liah X. Coggins, and Anas Ghadouani. 2022. "Hydrological Regime and Fish Predation Regulate the Zooplankton Community Size Structure in a Tropical Floodplain Lake" Water 14, no. 16: 2518. https://doi.org/10.3390/w14162518






