Synergistic Effect of Ferrate with Various Water Processing Techniques—A Review
Abstract
:1. Introduction
2. Water Treatment Performance of Synergistic Applications of Ferrate
| Synergistic Application of Ferrate | Chemicals | Sample condition | Removal% | Reference | |
|---|---|---|---|---|---|
| Ferrate + Persulfate | 5 mM ferrate + 2.5 mM peroxymonosulfate | Atrazine | 46.5 µM | 81.5 | [16] |
| 90 µM ferrate and 90 µM peroxymonosulfate by following the addition of the 90 µM Hydroxylamine | Ciprofloxacin | 30 µM | 91.5 | [17] | |
| 0.03 mM Ferrate + 0.03 mM peroxysulfate | Fluoroquinolones | 0.03 mM | 60 (improved from 10 by only ferrate) | [18] | |
| Photo + 0.5 mM Fe6+ + 0.5 mM peroxyonosulfate | sulfamethoxazole | 0.1 mM | 80 | [19] | |
| Ferrate + sulphite/bisulphite | 0.25 mM Sulphite + 0.05 mM ferrate + photo-activitation | Ciprofloaxin | 0.005 mM | >95 | [20] |
| 100 μM ferrate + 400 sulphite | DEET N, N-diethyk-3-toluamide | 10 μM | 78 | [21] | |
| 100 μM Fe(VI) + 100 μM sulphite/bisulphite | Flumequine | 20 μM | >40 | [10] | |
| 0.25 mM Sulphite + 0.05 mM ferrate 0 | Tert-butanol (TBA) | 10 mM | >40 | [20] | |
| 100 μM Fe(VI) + 100 μM sulphite/bisulphite | Trimethoprim | 2 μM | >90 | [10] | |
| 0.25 mM Sulphite + 0.05 mM ferrate | Sulfamethoxazole | 0.005 mM | >60 | [20] | |
| Ferrate + thiosulphate | [Thiosulphate]:[Fe(VI)] = 1:8 | chloramphenicol | 31 μM | 69 | [22] |
| Ferrate + adsorbent | 3 mg L−1 ferrate + 0.05 g MgO nanoparticles | Blue-203 dye | 25 mg L−1 | >97 | [23] |
| 0.5 mM ferrate and 4 g. L−1 silicate gel | Caffeine | 0.085–0.089 mM | >95 | [24] | |
| [Fe(VI)]: [Fe(III)] = (3:7) | Chemical weapon agent (Soman) | 0.38 mg.mL−1 | 99 | [25] | |
| 6.6 mg mL−1 ferrate + 16.6 mg Graphene oxide | Diclofenac | 2.5 mg L−1 | 99 | [15] | |
| Simultaneous 0.42 mM Ferrate + 0.12 mM PACl | Humic acids such as NOM | N/A | 37 | [26] | |
| 12 mM ferrate + 625 mg L−1 graphene oxide | RHB dye | 0.02 mg L−1 | 99 | [15] | |
| Simultaneous application of ferrate and montmorillonite [Fe (VI)]:[R] = 10:1, [montmorillonite] = 100 mg | Sulfadiazine | 0.02 mM | 73.70 | [27] | |
| Ferrate + UV | UV-Fe (VI) 8.96 mg L−1 ferrate + 40 mg L−1 TiO2 light intensity = 0.40 mW cm−2, pH 9 | Dimethyl phthalate | 11.6 mg L−1 | 68 | [28] |
| 0.1 g L−1 ferrate + UV fluence rate ¼ 0.198 mW cm−2 | phenolic compounds | 0.15 mM | 73 | [29] | |
| Ferrate + Ozonation | Ferrate, 0.1 mg L−1 + O3, 1.2 mg L−1 Ferrate 2.0 mg L−1 + O3, < 2.5 mg L−1 | (DMS) N,N-dimethylsulfamide Bromination | 10 µg L−1 Bromide concentration in raw water, 500 µg.L−1 | Completed removal and no NDMA formation 88–100 | [30] [30,31] |
| 0.15 mM ferrate + 10 mg L−1 ozone | DOC UV254 protein polysaccharide | 7.5 mg L−1 0.158 mg L−1 4.3 mg L−1 5.82 mg L−1 | 70.5 80.5 25.5 65.5 | [32] | |
| Simultaneous addition of 0.51 µmol L−1 ferrate and 0.51 µmol L−1 ozone | tetrabromobisphenol-A | 1.84 µM | 85.5 | [33] | |
| Ferrate + Other Chemical Additives | 0.1–0.5 mL of 0.5 M HCl in 161.9–569.2 µM Fe(VI) | Acesulfame potassium | 49.0–81.4 µM | 95 (from 64 by only ferrate) | [11] |
| 0.1–0.5 mL of 0.5 M HCl in 161.9–569.2 µM Fe(VI) | Atenolol | 41.6– 56.9 µM | 30 (from 0 by only ferrate) | [11] | |
| 0.1–0.5 mL of 0.5 M HCl in 88.5–634.5 µM Fe(VI). | Caffeine | DW 78.7–95.3 µM | 60 (from 12 by only ferrate) | [11] | |
| 10 mM ammonia + 0.6 mM ferrate | Flumequine | 0.03 mM | 90 | [9] | |
| Ferrate + UF | 2.0 mg L−1 ferrate + UF | DOC | Approx. 16 mg L−1 | 41 | [34] |
| Ferrate + ultrasound Ultrasound | 0.05 mM Fe6+ + Ultrasound (Ultrasonic frequency: 800 KHz) | Sulfadiazine Sulfamerazine Sulfamethoxazole | 0.02 mM | >95 | [35] |
| Ferrate + Chlorination | 0.25 mM ferrate + 5.0 mg L−1 chlorinated 0.25 mM ferrate + 5.0 mg L−1 chloraminated | halo-DBPs | N/A | 80.5 72.4 | [36] |
2.1. Mitigation of Membrane Fouling by Ferrate Pre-treatment
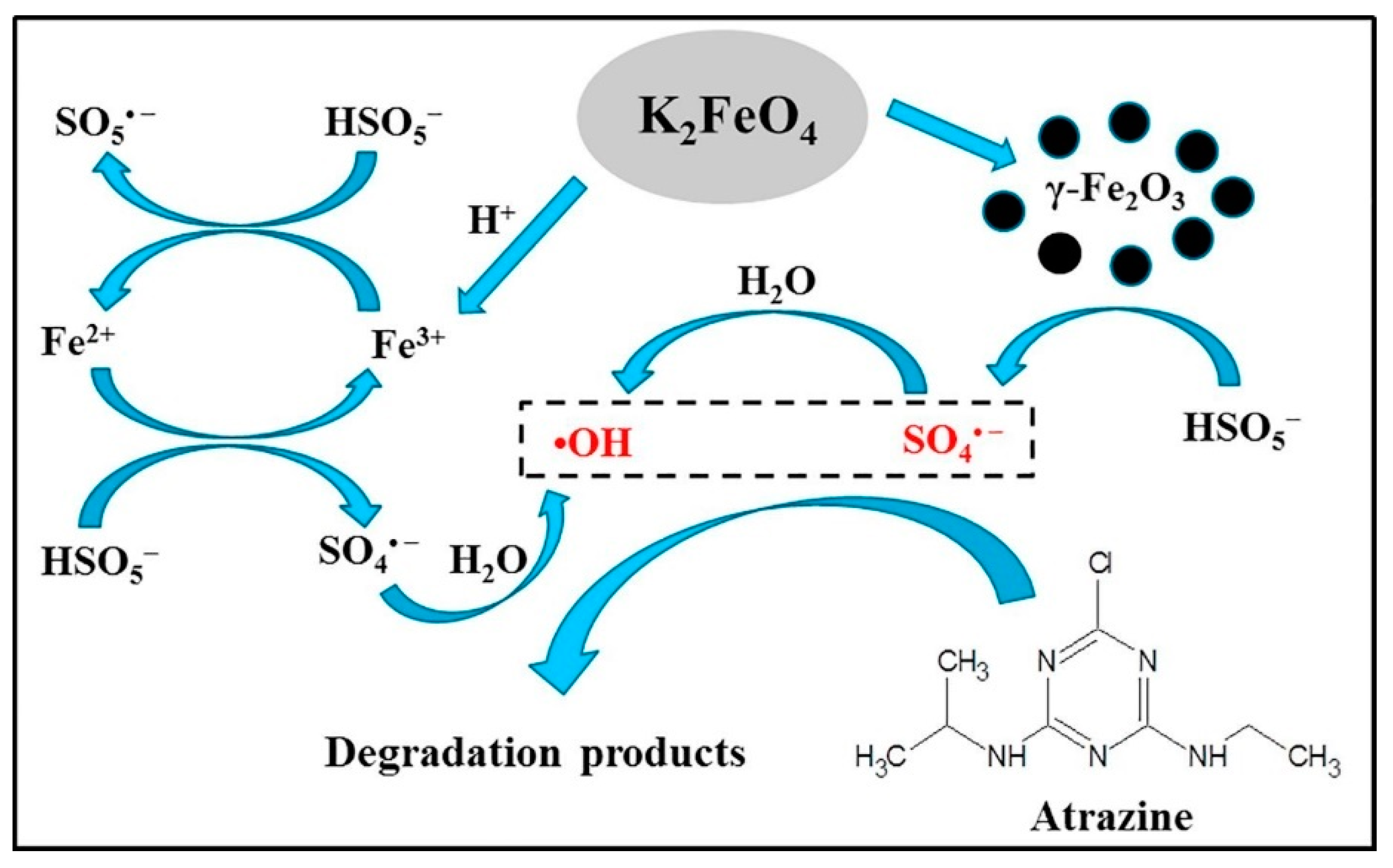
2.2. Ferrate Coupled with Persulfate (SO52−, S2O82−)
2.3. Sulphite (SO32−) + Ferrate
2.4. Thiosulphite (S2O32−) with Ferrate
2.5. Ferrate Coupled with Ultraviolet Light (UV) Radiation
2.6. Ferrate Coupled with Ozonation
2.7. Ferrate Coupled with Other Additives
2.7.1. Ferrate + Ammonia
2.7.2. Ferrate + Acids
2.7.3. Ferrate + Quinone
3. Conclusions and Further Works
3.1. Conclusions
- (1)
- Ferrate significantly improves membrane processing efficiency and alleviation membrane fouling; macromolecular natural organic matter can be oxidised into smaller organic particles by the ferrate. The properties and structure of flocs and DOC foulants were changed by ferrate oxidation so that the resultant flocs were more easily settled and difficult to deposit onto the membrane.
- (2)
- The high treatment performance by ferrate/sulphur compounds has mainly been attributed to the oxidative species formed during the interaction between ferrate and sulphur compounds such as Fe(V), Fe(IV), and free radicals such as SO4−, SO5−, SO3−, and OH∙−.
- (3)
- The high performance of ferrate coupled with other additives such as acids, quinone, and ammonia is because these chemical additives activate the ferrate’s decomposition, which increases the available electrons equivalence per unit of ferrate and modifies the properties and structure of the resultant ferric flocs, which can improve the efficiency of the adsorption and sedimentation.
3.2. Challenges and Future Work
- (1)
- The membrane-fouling mechanism varies after ferrate pre-oxidation. When the dosage of ferrate is increased to a certain threshold level, the DOC substances can be further degraded to smaller molecular weight substances such as protein and humic-like substances, which can penetrate to the membrane pore, and the accumulation of these substances can cause internal fouling by protein and humic-like substances. The optimisation of the logistics of the synergistic application of ferrate/membrane separation is required in future work.
- (2)
- On the ferric particles resulting from the sulphite/ferrate system, the crystalline structure of the non-activated particles is easier to prepare but has a low adsorption of pollutants. In contrast, the amorphous structure of the activated particles is more effective in adsorbing the pollutants but has a low precipitation rate. Therefore, future works need to study balanced treatment goals: the high adsorption of pollutants and/or quick precipitation via stochiometric of sulphite and ferrate ratios.
- (3)
- In the ferrate/ozonation system, the catalytic interaction between ferrate and ozone can be weakened by the sequence of ozonation, so ferrate pre-oxidation is the ideal way to interact with the ozone and enhance the synergistic effect sufficiently.
- (4)
- Adding an appropriate amount of quinone could activate the decomposition of the ferrate to produce the reactive ferrate species (Fe(V) and Fe(IV)), which enhanced the ability to oxidise the electron-rich moieties. However, the excessive addition of quinone could weaken the flocculation performance and retard the transformation of iron species. Thus, the logistics of quinone application need to be optimised.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Glassmeyer, S.T.; Furlong, E.T.; Kolpin, D.W.; Batt, A.L.; Benson, R.; Boone, J.S.; Conerly, O.; Donohue, M.J.; King, D.N.; Kostich, M.S. Nationwide reconnaissance of contaminants of emerging concern in source and treated drinking waters of the United States. Sci. Total Environ. 2017, 581, 909–922. [Google Scholar] [CrossRef] [PubMed]
- Lapworth, D.; Baran, N.; Stuart, M.; Ward, R. Emerging organic contaminants in groundwater: A review of sources, fate and occurrence. Environ. Pollut. 2012, 163, 287–303. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, A.A.; Parveen, T.; Umar, K.; Mohamad Ibrahim, M.N. Role of nanomaterials in the treatment of wastewater: A review. Water 2020, 12, 495. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ahmad, H.; Parveen, T.; Ahmad, A.; Oves, M.; Ismail, I.M.; Qari, H.A.; Umar, K.; Mohamad Ibrahim, M.N. Recent advances in metal decorated nanomaterials and their various biological applications: A review. Front. Chem. 2020, 8, 341. [Google Scholar] [CrossRef]
- Jiang, J.Q. Advances in the development and application of ferrate (VI) for water and wastewater treatment. J. Chem. Technol. Biotechnol. 2014, 89, 165–177. [Google Scholar] [CrossRef]
- Sharma, V.K.; Zboril, R.; Varma, R.S. Ferrates: Greener oxidants with multimodal action in water treatment technologies. Acc. Chem. Res. 2015, 48, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Shu, J.; Sharma, V.K.; Liu, C.; Xu, X.; Nesnas, N.; Wang, H. Unveiling the mechanism of imidacloprid removal by ferrate (VI): Kinetics, role of oxidation and adsorption, reaction pathway and toxicity assessment. Sci. Total Environ. 2022, 805, 150383. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Jiang, J.-Q.; Petri, M. Preliminarily comparative performance of removing bisphenol-S by ferrate oxidation and ozonation. npj Clean Water 2021, 4, 1. [Google Scholar] [CrossRef]
- Feng, M.; Cizmas, L.; Wang, Z.; Sharma, V.K. Activation of ferrate (VI) by ammonia in oxidation of flumequine: Kinetics, transformation products, and antibacterial activity assessment. Chem. Eng. J. 2017, 323, 584–591. [Google Scholar] [CrossRef]
- Feng, M.; Sharma, V.K. Enhanced oxidation of antibiotics by ferrate (VI)-sulfur (IV) system: Elucidating multi-oxidant mechanism. Chem. Eng. J. 2018, 341, 137–145. [Google Scholar] [CrossRef]
- Manoli, K.; Nakhla, G.; Ray, A.K.; Sharma, V.K. Enhanced oxidative transformation of organic contaminants by activation of ferrate (VI): Possible involvement of FeV/FeIV species. Chem. Eng. J. 2017, 307, 513–517. [Google Scholar] [CrossRef]
- Chen, Z.; Yang, B.; Wen, Q.; Tang, Y. Application of potassium ferrate combined with poly-aluminum chloride for mitigating ultrafiltration (UF) membrane fouling in secondary effluent: Comparison of oxidant dosing strategies. Chemosphere 2021, 274, 129862. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Liu, J.; Li, C.; Lin, Q.; Zhang, T.; Zhang, K.; Sharma, V.K. Ferrate (VI) pre-treatment and subsequent chlorination of blue-green algae: Quantification of disinfection byproducts. Environ. Int. 2019, 133, 105195. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhao, Z.; Liu, J.; Peng, W.; Peng, X.; Han, Y.; Xiao, P. Removal of Microcystis aeruginosa and control of algal organic matters by potassium ferrate (VI) pre-oxidation enhanced Fe (II) coagulation. Korean J. Chem. Eng. 2019, 36, 1587–1594. [Google Scholar] [CrossRef]
- Mura, S.; Jiang, Y.; Vassalini, I.; Gianoncelli, A.; Alessandri, I.; Granozzi, G.; Calvillo, L.; Senes, N.; Enzo, S.; Innocenzi, P. Graphene oxide/iron oxide nanocomposites for water remediation. ACS. Appl. Nano Mater. 2018, 1, 6724–6732. [Google Scholar] [CrossRef]
- Wu, S.; Li, H.; Li, X.; He, H.; Yang, C. Performances and mechanisms of efficient degradation of atrazine using peroxymonosulfate and ferrate as oxidants. Chem. Eng. J. 2018, 353, 533–541. [Google Scholar] [CrossRef]
- Li, C.; Lin, H.; Armutlulu, A.; Xie, R.; Zhang, Y.; Meng, X. Hydroxylamine-assisted catalytic degradation of ciprofloxacin in ferrate/persulfate system. Chem. Eng. J. 2019, 360, 612–620. [Google Scholar] [CrossRef]
- Feng, M.; Cizmas, L.; Wang, Z.; Sharma, V.K. Synergistic effect of aqueous removal of fluoroquinolones by a combined use of peroxymonosulfate and ferrate (VI). Chemosphere 2017, 177, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Chu, W.; Xu, K.; Xia, X.; Gong, H.; Tan, Y.; Pu, S. Efficient degradation, mineralisation and toxicity reduction of sulfamethoxazole under photo-activation of peroxymonosulfate by ferrate (VI). Chem. Eng. J. 2020, 389, 124084. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, L.; Shi, Z.; Gao, Y. Rapid removal of organic pollutants by activation sulfite with ferrate. Chemosphere 2017, 186, 576–579. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Pang, S.; Jiang, J.; Ma, J.; Huang, Z.; Zhang, J.; Liu, Y.; Xu, C.; Liu, Q.; Yuan, Y. The combination of ferrate (VI) and sulfite as a novel advanced oxidation process for enhanced degradation of organic contaminants. Chem. Eng. J. 2018, 333, 11–19. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, X.; Zhang, C.; Lu, S.; Xi, Y.; Huang, Y.; Xue, Z.; Yang, T. Combining ferrate (VI) with thiosulfate to oxidise chloramphenicol: Influencing factors and degradation mechanism. J. Environ. Chem. Eng. 2021, 9, 104625. [Google Scholar] [CrossRef]
- Eskandari, Z.; Talaiekhozani, A.; Talaie, M.R.; Banisharif, F. Enhancing ferrate (VI) oxidation process to remove blue 203 from wastewater utilising MgO nanoparticles. J. Environ. Manag. 2019, 231, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Manoli, K.; Nakhla, G.; Feng, M.; Sharma, V.K.; Ray, A.K. Silica gel-enhanced oxidation of caffeine by ferrate (VI). Chem. Eng. J. 2017, 330, 987–994. [Google Scholar] [CrossRef]
- Zboril, R.; Andrle, M.; Oplustil, F.; Machala, L.; Tucek, J.; Filip, J.; Marusak, Z.; Sharma, V.K. Treatment of chemical warfare agents by zero-valent iron nanoparticles and ferrate (VI)/(III) composite. J. Hazard. Mater. 2012, 211, 126–130. [Google Scholar] [CrossRef]
- Amano, M.; Lohwacharin, J.; Dubechot, A.; Takizawa, S. Performance of integrated ferrate–polyaluminum chloride coagulation as a treatment technology for removing freshwater humic substances. J. Environ. Manag. 2018, 212, 323–331. [Google Scholar] [CrossRef]
- Wang, H.; Wang, S.; Jiang, J.-Q.; Shu, J. Removal of sulfadiazine by ferrate (VI) oxidation and montmorillonite adsorption—synergistic effect and degradation pathways. J. Environ. Chem. Eng. 2019, 7, 103225. [Google Scholar] [CrossRef]
- Yuan, B.-l.; Li, X.-z.; Graham, N. Reaction pathways of dimethyl phthalate degradation in TiO2–UV–O2 and TiO2–UV–Fe (VI) systems. Chemosphere 2008, 72, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Liu, H.; Lin, Y.; Yang, C.; Lou, W.; Sun, J.; Du, C.; Zhang, D.; Nie, L.; Yin, K. Insights into mechanisms of UV/ferrate oxidation for degradation of phenolic pollutants: Role of superoxide radicals. Chemosphere 2020, 244, 125490. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.-Q.; Durai, H.B.P.; Petri, M.; Grummt, T.; Winzenbacher, R. Drinking water treatment by ferrate(VI) and toxicity assessment of the treated water. Desalination Water Treat 2016, 57, 26369–26375. [Google Scholar] [CrossRef]
- Han, Q.; Wang, H.; Dong, W.; Liu, T.; Yin, Y. Formation and inhibition of bromate during ferrate (VI)–Ozone oxidation process. Sep. Purif. Technol. 2013, 118, 653–658. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Z.; Chen, Q.; Zhang, X. Synergistic effect of ferrate (VI)-ozone integrated pre-treatment on the improvement of water quality and fouling alleviation of ceramic UF membrane in reclaimed water treatment. J. Membr. Sci. 2018, 567, 216–227. [Google Scholar] [CrossRef]
- Han, Q.; Dong, W.; Wang, H.; Ma, H.; Gu, Y.; Tian, Y. Degradation of tetrabromobisphenol A by a ferrate (vi)–ozone combination process: Advantages, optimisation, and mechanistic analysis. RSC Adv. 2019, 9, 41783–41793. [Google Scholar] [CrossRef] [PubMed]
- He, H.-Y.; Qiu, W.; Liu, Y.-L.; Xu, S.-Y.; Ma, J.; Wang, L. Ferrate Preoxidation Alleviating Membrane Fouling through the Formation of a Hydrophilic Prefiltration Layer. ACS ES T Eng. 2021, 1, 1576–1586. [Google Scholar] [CrossRef]
- Zhang, K.; Luo, Z.; Zhang, T.; Gao, N.; Ma, Y. Degradation effect of sulfa antibiotics by potassium ferrate combined with ultrasound (Fe (VI)-US). BioMed Res. Int. 2015, 2015, 169215. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lujan, H.; Dhungana, B.; Hockaday, W.C.; Sayes, C.M.; Cobb, G.P.; Sharma, V.K. Ferrate (VI) pre-treatment before disinfection: An effective approach to controlling unsaturated and aromatic halo-disinfection byproducts in chlorinated and chloraminated drinking waters. Environ. Int. 2020, 138, 105641. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.; Liu, B.; Xie, W.; Wang, P.; He, Q.; Bao, J.; Zhang, Y.; Zhang, Z.; Li, J.; Ma, J. Synergistic mechanism of combined ferrate and ultrafiltration process for shale gas wastewater treatment. J. Membr. Sci. 2022, 641, 119921. [Google Scholar] [CrossRef]
- Yu, W.; Yang, Y.; Graham, N. Evaluation of ferrate as a coagulant aid/oxidant pre-treatment for mitigating submerged ultrafiltration membrane fouling in drinking water treatment. J. Chem. Eng. 2016, 298, 234–242. [Google Scholar] [CrossRef]
- Wang, J.; Feng, K.; Lou, Y.; Lu, B.; Liu, B.; Xie, G.; Ren, N.; Xing, D. The synergistic effect of potassium ferrate and peroxymonosulfate application on biogas production and shaping microbial community during anaerobic co-digestion of a cow manure-cotton straw mixture. Bioresour. Technol. 2021, 333, 125166. [Google Scholar] [CrossRef]
- Sun, X.; Feng, M.; Dong, S.; Qi, Y.; Sun, L.; Nesnas, N.; Sharma, V.K. Removal of sulfachloropyridazine by ferrate (VI): Kinetics, reaction pathways, biodegradation, and toxicity evaluation J. Chem. Eng. 2019, 372, 742–751. [Google Scholar] [CrossRef]
- Bzdyra, B.M.; Spellman Jr, C.D.; Andreu, I.; Goodwill, J.E. Sulfite activation changes character of ferrate resultant particles. J. Chem. Eng. 2020, 393, 124771. [Google Scholar] [CrossRef]
- Wang, S.; Hu, Y.; Wang, J. Strategy of combining radiation with ferrate oxidation for enhancing the degradation and mineralisation of carbamazepine. Sci. Total Environ. 2019, 687, 1028–1033. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xiong, Y.; Wang, Z.; Chen, Y.; Chen, G.; Liu, Z. UV/ferrate (VI) oxidation of profenofos: Efficiency and mechanism. Desalination Water Treat 2015, 55, 506–513. [Google Scholar] [CrossRef]
- Matin, A.R.; Yousefzadeh, S.; Ahmadi, E.; Mahvi, A.; Alimohammadi, M.; Aslani, H.; Nabizadeh, R. A comparative study of the disinfection efficacy of H2O2/ferrate and UV/H2O2/ferrate processes on inactivation of Bacillus subtilis spores by response surface methodology for modeling and optimisation. Food Chem. Toxicol. 2018, 116, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Wang, J.; Zhang, H.; Jia, H.; Zhang, Y.; Gao, F. New insight into quinones triggered ferrate in-situ synthesised polynuclear Fe-hydroxyl complex for enhancing interfacial adsorption in highly efficient removal of natural organic matter. Sci. Total Environ. 2021, 770, 144844. [Google Scholar] [CrossRef] [PubMed]
| 4FeO42− + 20H+ → 4Fe3+ + 10OH− + 3O2 |
| 4FeO42− + 10H2O → 4Fe3+ + 20OH− + 3O2 |
| Fe3+ + HSO5− → Fe2+ + SO5− |
| Fe2+ + HSO3− → Fe3+ + SO4− + O− |
| ≡ Fe3+-OH + HSO5− → ≡ Fe3+-SO5− + H2O |
| ≡ Fe3+-SO5− + H2O → ≡ Fe3+-OH + SO5− + H+ |
| ≡ Fe3+-OH + HSO5− → ≡ Fe2+-SO5− + H2O |
| ≡ Fe2+-SO5− + H2O → ≡ Fe3+-OH + SO4− + H+ |
| SO4−+ H2O → SO42− + ∙OH + H+ |
| 4FeO42− + 20H+ → 4Fe3+ + 10OH− + 3O2 |
| 4FeO42− + 10H2O → 4Fe3+ + 20O− + 3O2 |
| NH2OH + S2O82− → NH2O + H+ +SO42− + SO4− |
| S2O8− + Fe3+ → Fe2+ + ∙S2O8− S2O8− + Fe2+ → Fe3+ + SO42 −+ SO4− SO4− − + H2O → H+ + SO42 − + ∙OH FeO42− + 3NH2OH + 5H+ → Fe3+ + 3NH2O + 4H2O NH2OH + Fe3+ → NH2O + Fe2+ + H+ |
| Fe6+ + SO32− → Fe5+ + SO − |
| SO3−+ O2 → SO5− |
| SO5− + HSO3 −/SO32− → SO4−+ SO42− + (H+) |
| SO4− + OH−→ SO42− + OH ∙ |
| Fe6+ + CAP → Fe2+/Fe3+ + CAP (oxidized) |
| Fe6+ + S2O32− → Fe5+/Fe4+ + SO4− |
| Fe6+ + S2O32− → Fe2+/Fe3+ + SO42− |
| Fe5+/Fe4+ + CAP → Fe2+/Fe3+ + CAP (oxidized) |
| Fe5+/Fe4+ + S2O32−→ Fe2+/Fe3+ + SO42− |
| SO3−/ ∙OH + CAP → SO42− + CAP (oxidized) |
| TiO2 + hv → hvvb+ + ecb− |
| HFeO4− + ecb− → HFeO42− |
| HFeO4− + ecb− → HFeO43− |
| HFeO43− + 3H2O + ec− → Fe(OH)3 + 4OH− |
| OH−produced from the direct decomposition of ozone | O3 + OH− → O− + HO2− → OH − |
| OH∙−produced from ferrate decomposition and interaction of ferrate species | Fe(VI) → Fe(V) → Fe(III) + H2O2 |
| Fe(VI) → Fe(IV) → Fe(II) | |
| Fe(II) + H2O2 → OH ∙ − + Fe(III) | |
| OH∙−produced from the interaction of surface hydroxyl on resultant ferrate particles (γ–Fe2O3 and γ–FeOOH) | O3 + > OH − → O2− + HO2− → OH − |
| Br− + O3 → BrO ∙ − + O2 |
| Br− + HO∙ → Br ∙+ OH − |
| OBr− + HO ∙ → BrO ∙+ OH − |
| 2FeO42− + 3H2O → 2Fe(OH)3 + 5 [O] |
| [O] + H2O → 2OH− |
| 2OH ∙→ H2O2 |
| 2HBrO + H2O2 → 2Br − + 2H2O + O2 |
| BrO3− + 6Fe2+ + 6H+ → Br − + 6Fe3+ + 3H2O |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, S.; Jiang, J.-Q. Synergistic Effect of Ferrate with Various Water Processing Techniques—A Review. Water 2022, 14, 2497. https://doi.org/10.3390/w14162497
Zhang S, Jiang J-Q. Synergistic Effect of Ferrate with Various Water Processing Techniques—A Review. Water. 2022; 14(16):2497. https://doi.org/10.3390/w14162497
Chicago/Turabian StyleZhang, Shaoqing, and Jia-Qian Jiang. 2022. "Synergistic Effect of Ferrate with Various Water Processing Techniques—A Review" Water 14, no. 16: 2497. https://doi.org/10.3390/w14162497
APA StyleZhang, S., & Jiang, J.-Q. (2022). Synergistic Effect of Ferrate with Various Water Processing Techniques—A Review. Water, 14(16), 2497. https://doi.org/10.3390/w14162497







