1. Introduction
Environmental degradation caused by released industrial effluent has increased in South Africa and around the world [
1,
2]. Due to its extensive changes in high strength organics such as TOC, alkalinity, turbidity, acidity, COD, BOD, and volatile fatty acids, brewery wastewater is one of the major industrial effluents contributing to these impacts, [
2]. Various efforts have been made, and some are still being made, to identify new approaches for treating and reusing industrial wastewater [
3]. Due to the abundance of organic compounds such as sugars, soluble starches, ethanol, and organic acids, as well as particulate COD contents from barley and hops, brewery wastewater typically has high COD, BOD, and TSS [
3,
4]. The amount of TSS, BOD, COD, and the concentration of nitrogen and phosphorus in the form of orthophosphate characterize untreated brewery effluent [
3,
4]. Previous studies have shown that the brewery effluent temperature ranges from 18–40 °C with average to high operational temperatures [
4,
5].
Chemical coagulation is a critical component in most traditional wastewater treatment plants. It occurs in a physical purification system that comprises transportation operations, coagulant injection for chemical reactions, charge neutralization, and the formation of tiny flocs for agglomeration into bigger flocs. This improves the downstream processes’ ability to effectively remove recalcitrant pollutants [
6,
7,
8,
9]. Double layer compression, sweep flocculation, adsorption and charge neutralization, and adsorption and interparticle bridging are the four coagulation mechanisms or particle aggregation mechanisms that can occur. The colloids and the additional coagulant react to destabilize and neutralize the electric charges in the particles, while flocculation aids the agglomeration of flocs in the colloidal solution [
7]. The coagulant is responsible for the formation of small-scattered particles that eventually coalesce into bigger, more stable particle flocs [
7]. As a result, the flocs become heavier than the water, settling as sediments that can be removed. About 90% of the suspended stuff is removed because of this process [
6,
10,
11,
12].
1.1. Characteristics of Brewery Wastewater
Brewery wastewater has high COD content from organic matter such as sugars, soluble starch, ethanol, volatile fatty acids, and many more [
7,
10]. Studies conducted on brewery wastewater indicate that it usually has temperatures ranging from 25 °C to 38 °C, but occasionally reaches much higher temperatures, pH levels range between 2 and 12 and are influenced by the amount and type of chemicals used in cleaning and sanitizing which are normally caustic soda, phosphoric and nitric acid [
7,
10]. Brewery effluent can be characterized by the properties mentioned above as well as the quantity of TSS, BOD, COD, and the concentration of nitrogen and phosphorus [
4]. Due to the quantity of wastewater produced, it is very significant to pre-treat wastewater before it is discharged to sewage or surface water.
1.2. Brewery Wastewater Treatment Methods
1.2.1. Physical Technique
In this technique, physical forces are applied to remove contaminants. Floatation, granular—medium filtration, sedimentation, flow equalization, communication, and, lastly, screening are different methods used to physically remove solid matter only from brewery wastewater. These methods provide little success; most often they lead to incomplete contaminants removal [
1,
3,
9,
10,
11,
12,
13,
14,
15].
1.2.2. Chemical Technique
Chemical pre-treatment may involve pH adjustment or coagulation and flocculation [
6,
7,
8,
9,
10,
11,
12,
13]. The acidity or alkalinity of wastewater affects both wastewater treatment and the environment [
9]. Low pH indicates increasing acidity while a high pH indicates increasing alkalinity. The pH of wastewater needs to remain between 6 and 9 to protect organisms. Waste CO
2 may be used to neutralize caustic effluents from CIP systems and bottle washers [
6,
7,
8,
14]. Chlorination, disinfection, adsorption, and chemical precipitation are among other widely used chemical techniques. Coagulation is a physicochemical process commonly used for the removal of colloidal material or colour from water and wastewater. In water and wastewater treatment, coagulation implies the step where particles are destabilized by a coagulant, and this may include the formation of small aggregates by Brownian motion (perikinetic coagulation) [
6,
7,
8,
9,
10,
11,
12,
13].
1.2.3. Biological Technique
When we talk of biological methods or techniques, we are referring to low investment methods with high removal efficiency in both soluble COD and BOD, and we are referring to mature technological methods [
16]: anaerobic digestion, activated sludge, aerated lagoons, trickling filters, rotating biological contactors, biological nutrient removal, and pond stabilization to list a few [
1,
4]. However, biological treatment processes are particularly effective for wastewater treatment, they require a high energy input [
2,
17,
18,
19,
20,
21,
22]. The treatment of brewery wastewater can either be done by an aerobic or anaerobic biological method: where aerobic means the presence of air or oxygen and anaerobic means the absence of oxygen inside the reactor [
4,
5,
7,
16,
21].
1.3. Aim
This study aimed to test the efficiency of alum-chlorohydrate (1%) against polyamine (1%) chemical coagulants in a lab scale flocculator or jar test method for the removal of turbidity and particulate COD (inert or non-biodegradable COD) from brewery wastewater to achieve zero liquid effluent discharge. More specifically, we aimed to assess the impact of the combination of the two chemical coagulants aluminium chlorohydrate 1% and polyamine 1% to form PAC. The combination of these coagulants poses as a novelty for this current study. We aimed to enlighten the local wastewater treatment plants in South Africa of the incredible ability of PAC coagulant on the removal of inert or non-biodegradable brewery pollutants in a nonorganic state. Currently, the local South African wastewater treatment plants are more habituated to simple biological treatment of waste organic matter mostly in the form of suspended solids. The basic pH pre-treatment and roughing filters conventional methods coupled with anaerobic and partial aerobic sequencing is implemented. A modest amount of mostly biodegradable content is removed successfully, but the inert and non-biodegradable inorganics always remain untreated. This poses an unnecessary fine on local WWT plants during the dewatering phase as local municipal sewer dewatering standards are stringent. Therefore, this study focused precisely on bridging that gap of total COD removal considering that the polymeric flocculants have the ability to treat and remove inert solids and particulate COD, hence achieving total COD removal. This method is not precisely novel but adds a new combined dosage approach of these polymeric coagulants towards ZLED. More operational sequence and approach on this method has been succinctly presented in detail in the following sections.
2. Materials and Methods
Beyond this article, focus on a novel approach of removing high strength organic and biological pollutants synchronously to bioenergy production in a laboratory scale, sequential, double-chamber MFC, which will be integrated with an electrochemical coagulation process in the cathodic chamber for enhanced substrate removal, will be implemented. This bio-electrochemical method will be tested for scaling-up the production of electricity whilst treating industrial and brewery wastewater, specifically removing total phosphates, COD, TOC, TSS, and turbidity.
For these particulate inorganics or inert removals, the following specific chemical coagulants will be evaluated: polyamine (1%), aluminium chlorohydrate (1%), and the combination of the two, polyamine-aluminium chlorohydrate in adequate dosage ratios to ensure that the total chemical oxygen demand is entirely removed. The capacity of these viable chemical coagulants to extensively treat the undesirable turbidity and particulate COD that would be accumulated throughout the electrochemical processing in the MFC will be investigated and clearly studied in this article.
This section explains the planned, analytical emphasis, and experimental methods used in this study to achieve the effect of chemical coagulation in the jar test method in treating brewery wastewater for zero liquid effluent discharge. This study aimed to answer the above-mentioned problems and make recommendations for a cotreatment strategy that combines the MFC technique with a chemical coagulation method. In light of these study goals and objectives, a more fundamental and step-by-step strategy has been provided as a categorical technique to cover the influence of chemical coagulation of inerts removal. Factorial design in Design Expert version 7 accurately predicted the study’s experimental number of runs to be approximately 14 runs. All experiments were conducted at Mangosuthu University of Technology Research Laboratory over a period of 14 days. Given the varying dose ppm levels of the two coagulants and PAC, this aspect was confirmed using RSM motivated number of runs. Below is a precise and detailed jar test unit experimental set up, which covers all essential operational components of the bench top jar test procedure that was carried out as an evaluation study towards integrating it with the MFC unit to achieve ZLED.
2.1. Sample Harvesting and Storage and Characterisation
2.1.1. Sample Harvesting and Storage
Samples were collected using the composite sampling method at the South African Brewery (SAB), which is located in Durban Ispingo-Prospecton with respect to the APHA 2004 [
21] sampling standard method. The targeted sampling point for brewery wastewater was at the final treated effluent point before it is sent to the trade effluent discharge stream; samples were harvested using a 25 L poly-can. After sampling, runs were conducted within four hours, if not, the samples were preserved by storing them in the chemical engineering laboratory incubation unit at 4 °C, to minimize microbiological deviations in the sample.
2.1.2. Design of Experiments for Lab Scale Jar Test Method
Design Expert software version 7 was used to design the experiments. This aided in determining the exact number of runs to be performed in executing this jar test method, which was later combined and merged with the MFC unit in the main research study to evaluate and improve the removal of particulate COD while generating bioelectricity. The design expert analysis presented in
Table 1 is based on RSM and the 2-2 factorial approach for run projection.
2.2. Jar Tes
The jar test is a standard laboratory technique used to assess the optimum water or wastewater treatment operating conditions. This method allows changes in pH, coagulant or polymer dose variability, exchange of mixing speeds, or small-scale testing of different types of coagulants or polymers to predict the activity of a large-scale treatment plant. This method was instigated as a post-treatment stage after SAB anaerobic digestion and biodegradation of brewery wastewater. At this point, the emphasis was to investigate the feasibility of the poly/alum and PAC coagulants on the removal efficacy and its significance in achieving ZLED by complete removal of particulate and inert COD and turbidity contained in the BWW.
2.2.1. Jar Testing Apparatus: Lovibond Flocculator
The jar testing apparatus consists of six paddles, which stir the sample in the six 1000 mL beakers. One beaker is used as a control while the operating conditions are varied among the remaining five beakers. A stirring regulator on the top-left of the device allows for control of the mixing speed in all of the beakers. This unit is also equipped with the automatic time, which was used to time the running intervals in order to do away with human error.
Figure 1 depicts the typical process flow diagram of a real plant application and integration of a chemical coagulation process with the traditional anaerobic digester plant for the treatment of brewery wastewater. This schematic layout underpins the treatment stage that incorporates the chemical coagulation process to ensure total removal of particulate COD and other inert pollutants, such as turbidity, etc., for easy effluent dewatering.
2.2.2. Jar Test Operational Procedure
The following sequential procedure was developed and precisely carried out for the lab-scale jar test method in view of investigating the effect of the polyamine 1% and alum-chlorohydrate 1% chemical coagulants’ significance in inert constituents removal.
Measure turbidity, pH, and COD.
Using a 1000 mL graduated cylinder, add 1000 mL of raw effluent water to be flocculated to each of the jar test beakers.
Using a prepared stock solution (1.0% by weight), dose each beaker with increasing amounts of solution. One beaker was used as a control, while the other 5 beakers were adjusted at different doses.
After the coagulant dose, stir at approximately 160 rpm for 2 min. The rapid mix stage helps to disperse the coagulant throughout each beaker.
Reduce the stirring speed to 60 rpm and continue mixing for 15 min. This slower mixing speed helps promote floc formation by enhancing particle collisions, which leads to larger flocs.
Turn off the mixers and allow the beaker to settle for 30 min. Then, measure the final turbidity in each beaker.
The categorical dosing intervals that were varied and increased in fixed amounts from the least ppm concentration, according to the design of experiments that RSM suggested as set points, were implemented. This projected range was sufficient to test the effect of each chemical coagulant simultaneous to presenting its significance in particulate COD and inerts removal.
2.3. Analytical Methods and Data Analysis
Concentrations were analysed before and after treatment sequence as part of the performance monitoring step. COD quantities were analysed using high-range ampoules (HACH Chemical) with a spectrophotometer (HACH, DR5000); turbidity quantities were analysed using TL23 Benchtop HACH turbidity-meter which uses ratio nephelometric measurement technology to provide reliable measurement. The grab sampling method was also used to analyse the effluent characteristics, which are: BOD:COD, pH levels, and conductivity. All data were sampled and analysed in replicates to achieve the replicability analyses, hence eradicating the propagated human error during processing. More-over, further statistical analysis was implemented using the advanced excel to analyse the sample population standard deviation, project the sample median and also analyse the overall distribution of the sample in terms of the Skewness test. For future work in the MFC unit, full experimental runs with enough population size for the student
t-test method on unpaired data with unequal variances at 95% confidence interval will be implemented to gather the population size confidence interval levels. For the purpose of this research article, basic statistical analysis to determine the significance levels of the flocculants was done by linear regression on advance excel. Here, an empirical predictive model with a corresponding root-mean square factor (R
2) was ascertained to show the significance of the coagulant in terms of inorganics removal as the value of R
2 approaches 1. Clearly, the results demonstrated a high significance and criticalness of merging the chemical coagulants for successful WWT. The results section details the above statistical parameters by plots and data tables. Moreover, R-Studio statistical software was employed and presented in the following
Section 3. Its statistical findings on the ALUM, POLY, and PAC flocculants significance test is deliberated.
3. Results and Discussion
3.1. Industrial Wastewater Characterisation: Raw Brewery Wastewater
The industrial wastewater samples were collected for two purposes: first, to characterize the wastewater generated by the local industries in order to determine its organic pollutant strength post WW pre-treatment; then, to recommend a laboratory-scale flocculator unit for advanced inert and non-biodegradable content removal from these complex substrates.
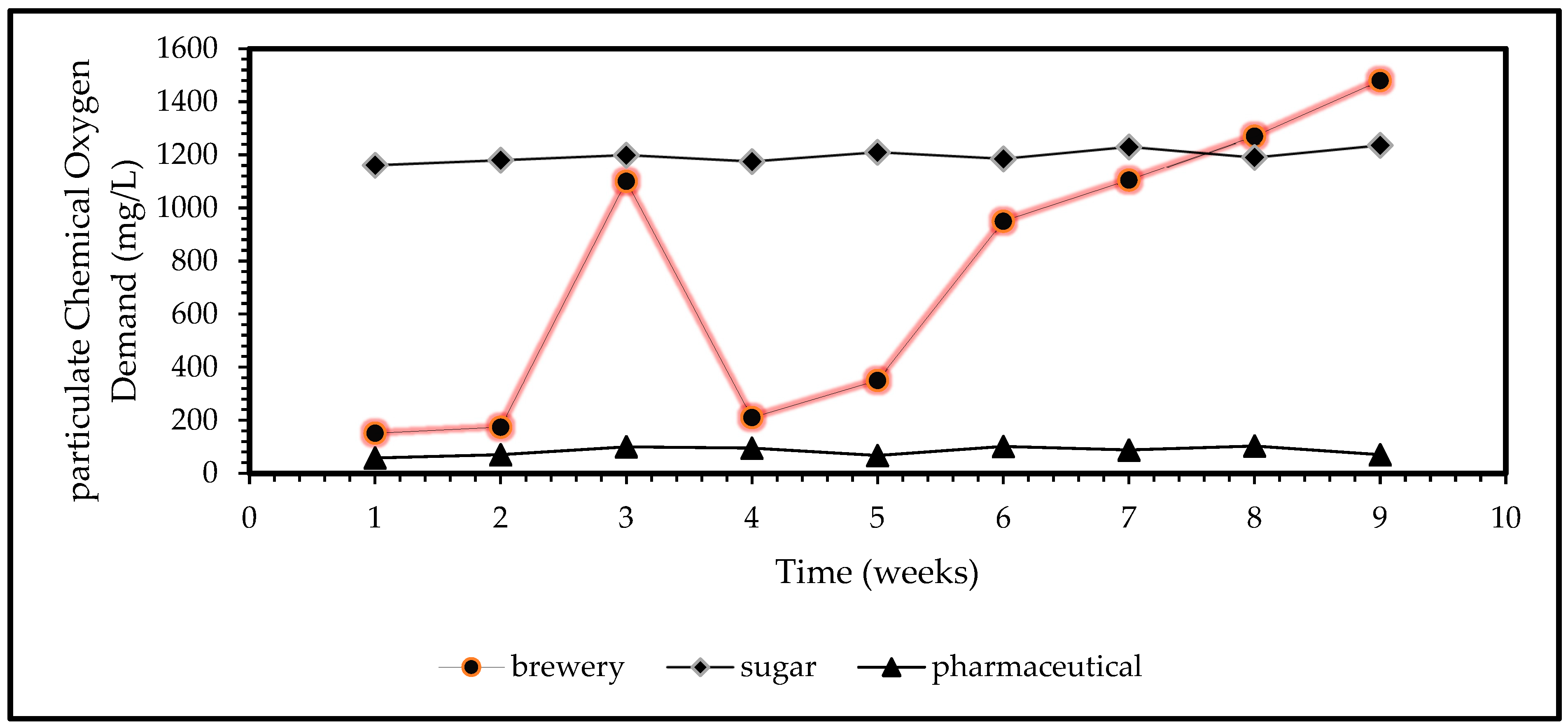
Figure 2 shows the characteristic organic contents of the three harvested complex substrates that were collected from local biorefinery, brewery, and pharmaceutical plants, respectively. This study focuses on the brewery wastewater for chemical coagulation treatment towards the removal of stagnant pollutants such as chemical or inert COD, turbidity, and TSS contained beyond the biological treatment stage. Based on the trends presented in
Figure 2, brewery wastewater shows an ever-changing nature and its complexity as an organic/inorganic substrate. The characterisation analysis presented a clear need for a further polishing stage of the brewery wastewater by chemical treatment method [
1,
9]. As seen in
Figure 2, the pollutant nature of brewery wastewater is highly unstable but clearly categorised as high range organic/inorganic pollutant strengthened wastewater due to highly fluctuating particulate COD and TSS concentrations. The need to treat and remove these inerts/inorganics is imperative to avoid nonconformance of local WWTP towards the local South African municipal sewer dewatering standards and high plant tariffs, thereof.
3.2. Effect of Flocculants Treatment Based on Characterised Brewery Wastewater
Trial runs were done over a period of 14 days in order to clearly investigate the removal of inert, inorganic, and organic pollutants from brewery wastewater by chemical coagulation/flocculation [
8,
22]. Flocculation, in principle, is distinguished onto three types of flocculation mechanisms: namely, charge neutralization, electrostatic patch, and polymer bridging. More precisely, this investigative comparison study focused on the effect of the latter class, i.e., the polymer bridging mechanism between polyamine (1%) feedstock flocculant and the aluminium chlorohydrate (1%) feedstock, and a combination of the two flocculants at variable dosages of polyamine flocculant. Due to the raw nature of brewery wastewater being characterized by high pollutants in terms of turbidity, COD, and BOD levels together with suspended solids, an initial dosage of 30 ppm was ideal with an increment of 10 ppm to 70 ppm. These variations were aimed at digressing the comparative study between polymeric polyamine (1%) linear flocculant against the effect of raw aluminium chlorohydrate (1%) flocculant on their flocculation efficiency and the capacity towards colour treatment on brewery wastewater. The combination of the two flocculants was aimed to be the optimal approach and was anticipated to render optimal removal of both biodegradable and non-biodegradable contents.
Figure 3, below, presents a sample of the treatment outcomes that were achieved after a typical experimental sequence in a jar test method. This pictorial result is evidence of the significance of chemical coagulation as an imperative post-treatment means by biological operation, in this case, from a typical SAB anaerobic digester vessel. In essence, this clearly shows the removal of inorganics, suspended solids, turbidity, and particulate COD with a good, anticipated effect on BOD, essentially. Even though the colour test was not successfully done for this manuscript, the visual observation of the effect of polyamine, alum-chlorohydrate, and combined poly/alum was clearly proven highly evident in
Figure 3. The before treatment condition is highly turbid and hazy, then in under 17 min of both rapid and slow mixing in the flocculator unit, all the suspended solids were rapidly coagulated into settleable flocs that were dense and quickly sedimented to the bottom of the beakers, leaving a clear effluent.
Results presented in
Figure 4,
Figure 5 and
Figure 6 show the mean-averages that were obtained with these two interchanging flocculants and their combined dosages of PAC. This implies that the replicability test was implemented when generating data of the operational parameters that were investigated: precisely, pCOD, BOD, turbidity, and TSS. Further statistical analysis was done on R-studio to determine the significance level of the compared flocculant treatment efficiencies. These findings presented the confidence interval level of the data and its credibility. The alpha values on the student
t-test also validated the significant difference on the compared flocculants poly/alum and poly alone with alum alone chemical coagulants.
3.2.1. Effect of Flocculants on Turbidity Removal from Brewery Wastewater
Figure 4 summarizes the turbidity removal profile using two types of coagulants at different dosages by bar graph. It can be observed from the graph that poly is more effective than alum, although they both yield quite notable results with alum reaching a percentage removal of 75.85%, whereas poly removed about 85.95% of turbidity. Poly-based coagulant was able to bring down turbidity to 10.70 NTU from 76.20 NTU of the influent while alum gave 18.40 NTU. As indicated in
Figure 4, the combined flocculant PAC had a balanced standard error from a statistical standpoint. This result corresponds to a 45-point standard deviation from a mean average of 101.
A positive skewness factor of 2 around the mean added to the statistical significance of this flocculant. The outliers were obviously dispersed positively around the mean value, as evidenced. Alum and poly mean values were 41 and 44, respectively. This is less than the variance seen in the combined flocculant. The skewness factor of 1 and 2 correspondingly revealed a clear and equitable distribution. Chemical coagulation’s efficiency has been shown to be statistically significant for the PAC (combined flocculants) when compared to poly and alum alone. However, it improved with the dosing configuration of the combined PAC coagulant. As mentioned by other authors, it is imperative to strike a balance of the flocculant concentrations to avoid the residual concentration of either alum or polyamine alone during this physicochemical treatment process [
8,
9,
11,
22].
3.2.2. Comparison of Flocculants Removal Efficiencies on Turbidity Removal
Figure 5 shows the effect of flocculants poly/alum and poly and alum alone on the removal of turbidity from brewery wastewater in a form of a best fit curve/plot. This level of relevance was predicted because chemical coagulation has the ability to coagulate sparse flocs and then agglomerate them into one heavy settleable floc that essentially settles off due to gravity [
12,
22]. A significant correlation factor of 0.9675 was obtained, indicating good removal efficiency concerning increasing flocculants dosage. At modest doses, the PAC coagulant was able to reach low particulate COD as compared to both poly and alum alone. This evidence demonstrates that PAC is more effective than the other flocculants in the treatment of turbidity. A significance correlation root mean-square factor of 0.9692, which attested to strong removal and treatment potential of PAC coagulant, was obtained. The combined PAC flocculant is preferable because of its high flocculation efficacy and as, by its nature of being a linear polymeric flocculant with a solid viscosity, it bears a good flocculation mechanism in bonding with suspended solids, hence quickly segregating them as flocs and thereby achieving good flocculation effect at low dosages, in addition to being an economically affordable flocculant from an engineering perspective [
22]. Chemical coagulation, as indicated in most of the literature, is an important step in water and wastewater treatment because it can break the chemical bonds of inerts and inorganics that cannot be handled by upstream techniques that solely rely on substrate biodegradation [
6,
7,
10,
11].
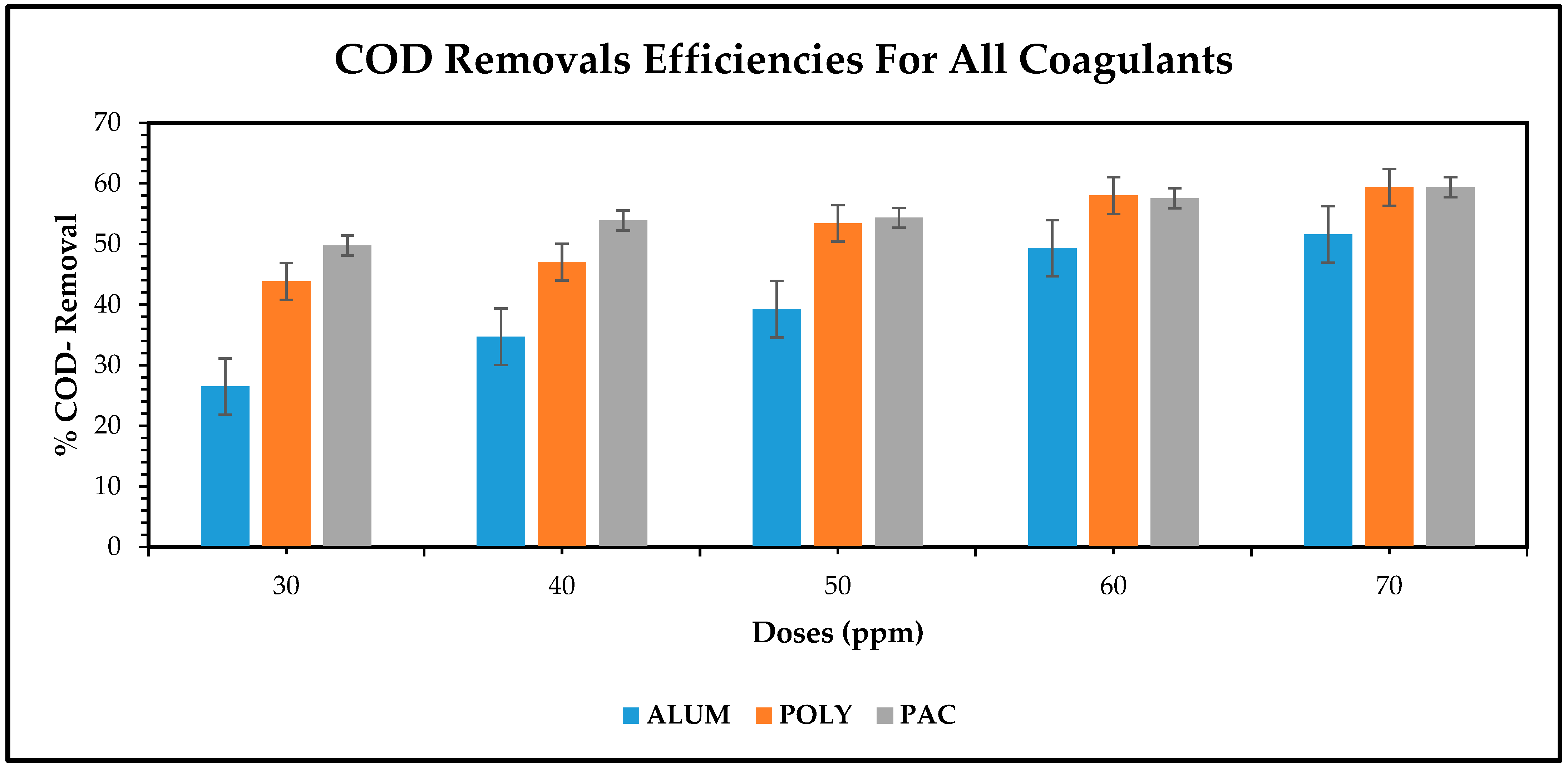
3.2.3. Effect of Flocculants on pCOD Removal from Brewery Wastewater
Figure 6 presents the COD removal profile at different dosage rates for both chemicals. It can be observed from the graph that poly is more effective than alum, although they both yield anticipated results. For both coagulants, previous studies have shown that they can yield COD removals of 55–65%. In this trial, alum was found to remove 51.59% and poly removed 59.36%. Overall, on COD removal, both coagulants were not far apart in terms of performance.
There is a clear outline in the removal efficiency of COD shown by
Figure 6. Scientifically, when applying a polymer-based flocculant, the chemical coagulation effect is seen to increase, this was clearly demonstrated by these effective coagulants on the pollutant removal from brewery wastewater. Moreover, varying the polyamine dosages tends to have a greater influence on the removal of suspended solids, sparingly breaking, and then agglomerating them to create settleable flocs. Polyamine literally has a stronger flocculating capacity, in principle, than alum-chlorohydrate coagulant and demonstrated a better statistical distribution analysis when skewness values were taken into account as presented in the previous section [
6,
7,
8,
9]. Polyamine can readily be recommended for industrial applications. The combination of poly and alum proved even more viable and had a high coagulation effectiveness on the removal of these unwanted inorganic and non-biodegradable pollutants in the form of particulate COD and turbidity. The subsequent graphical results accompanied by succinct trends clearly elucidate this phenomenon of effectively combined flocculants, polyamine/alum-chlorohydrate, to be commonly referred to as PAC.
3.2.4. Comparison of Flocculants Removal Efficiencies on pCOD
Figure 7 presents the PAC, poly and alum alone flocculants that were varied at standardized dosages. The pCOD was reduced quite significantly on the brewery wastewater, reducing to 65% COD removal efficiency. These results show the effect of the combined flocculants PAC as more effective in terms of treating the inert and inorganic pollutants from the brewery wastewater.
These results show a positive removal significance R
2 mean factor of 0.9649 for PAC and another strong correlation factor for both poly and alum with 0.9571 and 0.9728, respectively. When using PAC alone as a flocculant, a higher removal efficiency was observed, proving to be better than both poly and alum flocculants. From a scientific point of view, the PAC-based coagulant had a greater effect in terms of chemically treating the inorganic contents found in this brewery wastewater post the biological treatment phase. This insight can be confidently applied to all of the areas addressed above [
6,
8,
9,
22]. In simple terms, for an almost perfect ZLED, one can instigate the blended chemical coagulants of both polyamine and alum-chlorohydrate as an efficient inert removal coagulant with good effects in terms of particulate COD removal.
3.2.5. Particulate—Chemical Oxygen Demand Related to Biological Oxygen Demand Profile
The effects of poly, alum, and PAC on BOD and pCOD are summarized in
Figure 8. The brewery wastewater had an inflow of 219 mg COD/L and 131 mgBOD/L that was essentially removed to 106 mg/L and 64 mg/L for alum, 89 mg/L and 53 mg/L for poly, and same for PAC with 89 mg/L and 53 mg/L for pCOD and BOD, respectively. In general, all flocculants had evident removal effects on brewery wastewater as shown, with the same reduction on both BOD and pCOD at 70 ppm of the flocculant dosage but a different average removal effect, which set the two flocculants apart with PAC exuding a more effective treatment at mean averages of 101 mgCOD/L and 60 mgBOD/L whilst poly had 109 mgCOD/L and 59 mgBOD/L. The drop in the removal effect of both PAC and poly as we approached the 70 ppm dosage could be argued as a viable breakthrough or optimum operating flocculant dosing capacity for highly efficient flocculation of inert contents from BWW. Beyond this point, the issue of residual aluminium ion concentrations in the treated brewery wastewater can arise [
8,
9,
22]. Hence, it is imperative to note the optimum dosing concentration of these flocculants, which is, in this case, below 70 ppm.
The above trend also depicts the organic and biodegradable nature and content of brewery wastewater. As observed, the appreciable ratio of BOD:COD based on the above results was 0.6. This ratio has been widely reported in the literature as a sign of biodegradability and, hence, one can conclude by stating that brewery wastewater can be further treated by both biological and physicochemical means as we have envisaged the flocculation process to further treat the non-biodegradable contaminants.
3.2.6. Relation of Turbidity with Respect to Total Suspended Solids
Figure 9 shows the effect of the PAC, poly, and alum on the removal of turbidity and total suspended solids. The turbidity was reduced from, approximately, 76 NTUs to precisely 18 NTUs for alum alone treatment. The same was observed for poly-based alone treatment which reduced towards 10 NTUs at 70 ppm of the polymeric flocculant.
Although the colour test was not feasibly performed due to lab instrumentation constraints, the visual comparison of the before and after treatment samples, showed above in
Figure 3, presented a radical change in colour from a hazy, brownish turbid colour to crystal clear effluent. These results indicate that turbidity and TSS can be successfully removed by enhanced amounts of alum alone treatment from 30 ppm gradually to 70 ppm but being aware of the possibility of residual alum concentration once the dosage is overshot. The beneficial effect of using the alum/poly combined dosage is quite evidently established from the above presented findings. Although, as reported by previous authors Choi et al. [
22], the perfect dosage has to be reached as an optimal capacity of the flocculant proportional to its efficient coagulation of the suspended solids to perfectly aggregate them into settleable flocs without the residual formation of precipitation that inhibits the efficacy of this physicochemical treatment method.
3.2.7. Advanced Statistical Analysis on R-Studio
Advanced R-studio was undertaken to elucidate on the criticalness of the raw data harvested from the experimental runs. This analysis fortified the relevance, significance, median, mean, minimum, and maximum values from the raw data tables presented and discussed in the above Results subsections. The statistical insight shown was the complete balance and margin around the main operating variables such as particulate COD and turbidity that was observed when using these distinctive flocculants, alum, poly, and PAC. Poly proved more viable compared to alum; however, when these two coagulants were combined to form PAC, they even proved to have a more significant removal effect on inerts, and a high chemical coagulation efficacy as presented in
Table 2,
Table 3 and
Table 4. Here, all the performance monitoring factors such as particulate COD and turbidity removal efficiencies are comparatively steeper and a visible marginal difference is observed between these flocculants.
The following graphical trends present the overall statistical perception from R-studio when comparing these flocculants’ removal efficiencies over and above the versatility of combining the two base flocculants, poly and alum with the option of varying poly as it was proven above as more chemically significant towards solids removal, COD, and colour treatment in brewery wastewater. The combined chemical coagulants mentioned as PAC showed a more significant removal efficiency with substantial alpha values (
p) of 0.005 in the student
t-test paired analysis. The overall observation derived from this statistical exercise is that PAC is more convenient, significant, and highly effective for both particulate COD and turbidity removal. This aspect has been further presented graphically in the following
Figure 10 and
Figure 11, clearly elucidating a higher and stronger flocculation and chemical coagulation of PAC (combined coagulants) as compared to both poly and alum alone dosages.
3.3. Current Perspectives on the Use of Polymeric Polyamine (1%) and Aluminium Chlorohydrate (1%) Flocculants on Brewery Wastewater
The combination of polymeric polyamine (1%) linear feedstock as a flocculant with aluminium chlorohydrate (1%) for the treatment of raw brewery wastewater from a South African local brewery is purely a new knowledge contribution. The above mentioned feedstock flocculants have been reported by Choi et al. [
8,
9,
22] not for the treatment of brewery wastewater in its raw state but rather synthetic dye wastewater, and Nafriday et al. [
8,
9] argued the comparison of PAC as polyaluminium chloride and aluminium sulphate commonly known as alum.
From an engineering point of view, the above deliberated results have clearly demonstrated the high effective capabilities of polyamine and aluminium chlorohydrates as effective and significant flocculants for the post-treatment of brewery wastewater beyond the anaerobic plant digestion where only the biodegradable pollutants in the form of soluble COD and partially suspended solids have been reduced extensively. These flocculants have displayed appreciable differences between the influent and effluent concentrations, thereby exuding clear removal efficiencies for particulate COD, BOD, TSS, and turbidity that were reported in the above sections. We have observed the ability of the flocculants to adjust their effectiveness across unadjusted potential hydrogen (pH) levels of the brewery wastewater that was on average 7.7 with a temperature of 26 °C. These physical parameters did not inhibit the flocculation efficiency of these flocculants, and moreover, its flocculant bridging mechanism was exuded at times when there was slight deviation in pH but this still did not affect the flocculation efficiencies on the removal of these inorganic/organic pollutants: pCOD, BOD, TSS, and turbidity. We can confidently recommend these polymeric flocculants to local South African wastewater treatment plants due to their high ability to settle off suspended solids and the high average turbidity removal efficiencies of about 54%, 75%, and 76%, respectively, for alum, poly, and PAC. Moreover, the supply of these flocculant feedstocks is readily available in the USA and in local South African chemical laboratory and consumables suppliers. This makes the bulk procurement of these flocculants a possibility.
4. Conclusions and Recommendations
4.1. Conclusions
Previous studies indicate that the brewery effluent is high in organic matter, which is highly biodegradable. This is the type of wastewater that can be effectively treated by a biological treatment system. This current study has shown that chemical coagulation on brewery wastewater using a laboratory-scale flocculator have an impressive rate of removal of particulate COD, BOD, turbidity, and TSS when aluminium chlorohydrate (1%) coagulant and polyamine (1%) coagulant are combined into PAC combined doses. The findings above indicated that the removal of pCOD, colour, and turbidity can be optimised through enhanced doses of poly varied from 30 ppm to 70 ppm while maintaining 50% of alum-coagulant constant as a combined novel treatment technique. The beneficial effect of using a PAC combined coagulant was clearly depicted. However, we also observed that an overdose of these coagulants, either alum or poly, can produce higher residual aluminium ion concentrations in the water. This could negatively affect the sweeping effect of chemical coagulation on the brewery wastewater non-biodegradables removal. Recent studies have shown that a high aluminium ion concentration can cause fatal disease on consumption, hence it should be avoided at all times. Overall results of this article have indicated that chemical coagulation on brewery wastewater treatment is a feasible pragmatic approach, effective and worth conducting for ensuring complete effluent treatment and, therefore, avoiding recurrent local municipality sewer levies for dewatering nonconforming final effluent.
4.2. Recommendations
It is critical that the chemical industry and wastewater treatment plants shift towards a green energy paradigm. This means viewing industrial wastewater at large as a basic commodity resource rather than a waste stream. Industrial wastewater, by virtue of its pollutant content, harnesses some tATP energy in the form of chemical energy, which is imperative for bioenergy production. Hence, novel technologies, e.g., BET, precisely, the MFC technique, can be fully adopted to produce clean, renewable, and sustainable energy towards the rapid growing energy crisis, especially locally in South Africa.
Moreover, an integrated novel treatment approach that will combine the MFC technology with the chemical coagulation process for optimised bioenergy production and complete zero liquid effluent discharge is recommended for future study. This can be viably adopted by the local wastewater treatment plants and help save on utilities and effluent discharge financial obligations every time dewatering thresholds are not met with South African local municipalities.
Author Contributions
Conceptualization, K.P.S., J.K.B. and B.F.B.; methodology, K.P.S. and J.K.B.; validation, K.P.S.; formal analysis, K.P.S.; investigation, K.P.S.; resources, B.F.B.; data curation, K.P.S. and J.K.B.; writing—original draft preparation, K.P.S.; writing—review and editing, K.P.S. and J.K.B.; visualization, K.P.S.; supervision, B.F.B.; project administration, B.F.B.; funding acquisition, B.F.B. and K.P.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by MANGOSUTHU UNIVERSITY OF TECHNOLOGY (MUT), MUT-staff research grant and The APC was funded by MUT.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The department of research and innovation at MUT deserves our gratitude for financing this study work. More importantly, I am grateful to Prof Babatunde Femi Bakare, my supervisor, for his administrative and technical assistance with this article, and Joseph Kapukwu Bwapwa for his ongoing advice and expert review of my paper. Finally, I would like to thank Xolani Buthelezi, a Chemical Engineering B-tech student, for his help with lab runs and experimental setup.
Conflicts of Interest
The authors declare no conflict of interest.
List of Acronyms
| TOC | Total Organic Carbon |
| sCOD | Soluble Chemical Oxygen Demand |
| pCOD | Particulate Chemical Oxygen Demand |
| TCOD | Total Chemical Oxygen Demand |
| BOD5 | Five Days Biological Oxygen Demand |
| TSS | Total Suspended Solids |
| ZLED | Zero Liquid Effluent Discharge |
| CIP | Clean In Plates |
| pH | Potential of Hydrogen |
| MFC | Microbial Fuel Cell |
| RSM | Response Surface Methodology |
| SAB | South African Brewery |
| APHA | American Public Health Association |
| BWW | Brewery Wastewater |
| WWT | Wastewater Treatment |
| PAC | Polyamine—Aluminium—Chlorohydrate |
| Poly | Polyamine (1%) |
| Alum | Aluminium Chlorohydrate (1%) |
| ppm | Parts per million, (Solid liquid Mixture, also referred to mg/L) |
| NTU | Nephelometric Turbidity Units |
| rpm | Revolutions per minute |
| DOE | Design of Experiments |
| BET | Bioelectrochemical Technologies |
| tATP | Total Adenosine Triphosphate |
References
- Bakare, B.F.; Shabangu, K.; Chetty, M. Brewery wastewater treatment using laboratory scale aerobic sequencing batch reactor. South Afr. J. Chem. Eng. 2017, 24, 128–134. [Google Scholar] [CrossRef]
- Gul, H.; Raza, W.; Lee, J.; Azam, M.; Ashraf, M.; Kim, K.H. Progress in microbial fuel cell technology for wastewater treatment and energy harvesting. Chemosphere 2021, 281, 130828. [Google Scholar] [CrossRef] [PubMed]
- Kwaku Armah, E.; Chetty, M.; Adebisi Adedeji, J.; Kukwa, D.T.; Mutsvene, B.; Pearlman Shabangu, K.; Femi Bakare, B. Emerging Trends in Wastewater Treatment Technologies: The Current Perspective. In Promising Techniques for Wastewater Treatment and Water Quality Assessment; IntechOpen: London, UK, 2021. [Google Scholar]
- Budgen, J.; Le-Clech, P. Assessment of brewery wastewater treatment by an attached growth bioreactor. H2Open J. 2020, 3, 32–45. [Google Scholar] [CrossRef]
- Chen, H.; Chang, S.; Guo, Q.; Hong, Y.; Wu, P. Brewery wastewater treatment using an anaerobic membrane bioreactor. Biochem. Eng. J. 2016, 105, 321–331. [Google Scholar] [CrossRef]
- Swain, K.; Abbassi, B.; Kinsley, C. Combined Electrocoagulation and Chemical Coagulation in Treating Brewery Wastewater. Water 2020, 12, 726. [Google Scholar] [CrossRef]
- Pedersen, T.H.; Khumalo, S.M.; Bakare, B.F.; Rathilal, S.; Tetteh, E.K. Characterization of South African Brewery Wastewater: Oxidation-Reduction Potential Variation. Water 2022, 14, 1604. [Google Scholar] [CrossRef]
- Duan, J.; Wang, J.; Guo, T.; Gregory, J. Zeta potentials and sizes of aluminum salt precipitates-Effect of anions and organics and implications for coagulation mechanisms. J. Water Process Eng. 2014, 4, 224–232. [Google Scholar] [CrossRef]
- Aziz, N.; Effendy, N.; Basuki, K.T. Comparison of Poly Aluminium Chloride (PAC) and Aluminium Sulphate Coagulants Efficiency in Waste Water Treatment Plant. J. Inov. Tek. Kim. 2017, 2, 24–31. [Google Scholar] [CrossRef]
- Simate, G.S.; Cluett, J.; Iyuke, S.E.; Musapatika, E.T.; Ndlovu, S.; Walubita, L.F.; Alvarez, A.E. The treatment of brewery wastewater for reuse: State of the art. Desalination 2011, 273, 235–247. [Google Scholar] [CrossRef]
- Bwapwa, J.K. Treatment Efficiency of an Anaerobic Baffled Reactor Treating Low Biodegradable and Complex Particulate Wastewater (blackwater) in an ABR Membrane Bioreactor Unit(MBR-ABR). Int. J. Environ. Pollut. Remediat. 2012, 1, 51–58. [Google Scholar] [CrossRef]
- Mohammadi, A.; Khadir, A.; Tehrani, R.M.A. Optimization of nitrogen removal from an anaerobic digester effluent by electrocoagulation process. J. Environ. Chem. Eng. 2019, 7, 103195. [Google Scholar] [CrossRef]
- Dosta, J.; Rovira, J.; Galí, A.; Macé, S.; Mata-Álvarez, J. Integration of a Coagulation/Flocculation step in a biological sequencing batch reactor for COD and nitrogen removal of supernatant of anaerobically digested piggery wastewater. Bioresour. Technol. 2008, 99, 5722–5730. [Google Scholar] [CrossRef] [PubMed]
- Azwar, M.Y.; Hussain, M.A.; Abdul-Wahab, A.K. Development of biohydrogen production by photobiological, fermentation and electrochemical processes: A review. Renew. Sustain. Energy Rev. 2014, 31, 158–173. [Google Scholar] [CrossRef]
- Gudiukaite, R.; Nadda, A.K.; Gricajeva, A.; Shanmugam, S.; Nguyen, D.D.; Lam, S.S. Bioprocesses for the recovery of bioenergy and value-added products from wastewater: A review. J. Environ. Manag. 2021, 300, 113831. [Google Scholar] [CrossRef] [PubMed]
- Salahuddin, M.; Uddin, M.N.; Hwang, G.; Asmatulu, R. Superhydrophobic PAN nanofibers for gas diffusion layers of proton exchange membrane fuel cells for cathodic water management. Int. J. Hydrogen Energy 2018, 43, 11530–11538. [Google Scholar] [CrossRef]
- Ma, D.; Jiang, Z.H.; Lay, C.H.; Zhou, D. Electricity generation from swine wastewater in microbial fuel cell: Hydraulic reaction time effect. Int. J. Hydrogen Energy 2016, 41, 21820–21826. [Google Scholar] [CrossRef]
- Venkatramanan, V.; Shah, S.; Prasad, R. A Critical Review on Microbial Fuel Cells Technology: Perspectives on Wastewater Treatment. Open Biotechnol. J. 2021, 15, 131–141. [Google Scholar] [CrossRef]
- Liu, Y.C.; Hung, Y.H.; Hsu, C.C.; Ni, C.S.; Liu, T.Y.; Chang, J.K.; Chen, H.Y. Effects of surface functional groups of coal-tar-pitch-derived nanoporous carbon anodes on microbial fuel cell performance. Renew. Energy 2021, 171, 87–94. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Dairy Products, 17th ed.; American Public Health Association: Washington, DC, USA, 2004; Available online: https://scirp.org/reference/referencespapers.aspx?referenceid=1456317 (accessed on 19 July 2022).
- Choi, J.; Ahn, Y. Continuous electricity generation in stacked air cathode microbial fuel cell treating domestic wastewater. J. Environ. Manag. 2013, 130, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-H.; Shin, W.S.; Lee, S.-H.; Joo, D.-J.; Lee, J.-D.; Choi, S.J.; Park, L.S. Separation Science and Technology Application of Synthetic Polyamine Flocculants for Dye Wastewater Treatment. Sep. Sci. Technol. 2001, 36, 2945–2958. [Google Scholar] [CrossRef]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).