Adsorption Mechanism of High-Concentration Ammonium by Chinese Natural Zeolite with Experimental Optimization and Theoretical Computation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization Techniques
2.3. Liquid-Phase Adsorption Experiments
2.4. Simulation of Zeolites’ Ammonium Capture and Bonding Strength
3. Results
3.1. Characterization of the Chinese Natural Zeolite
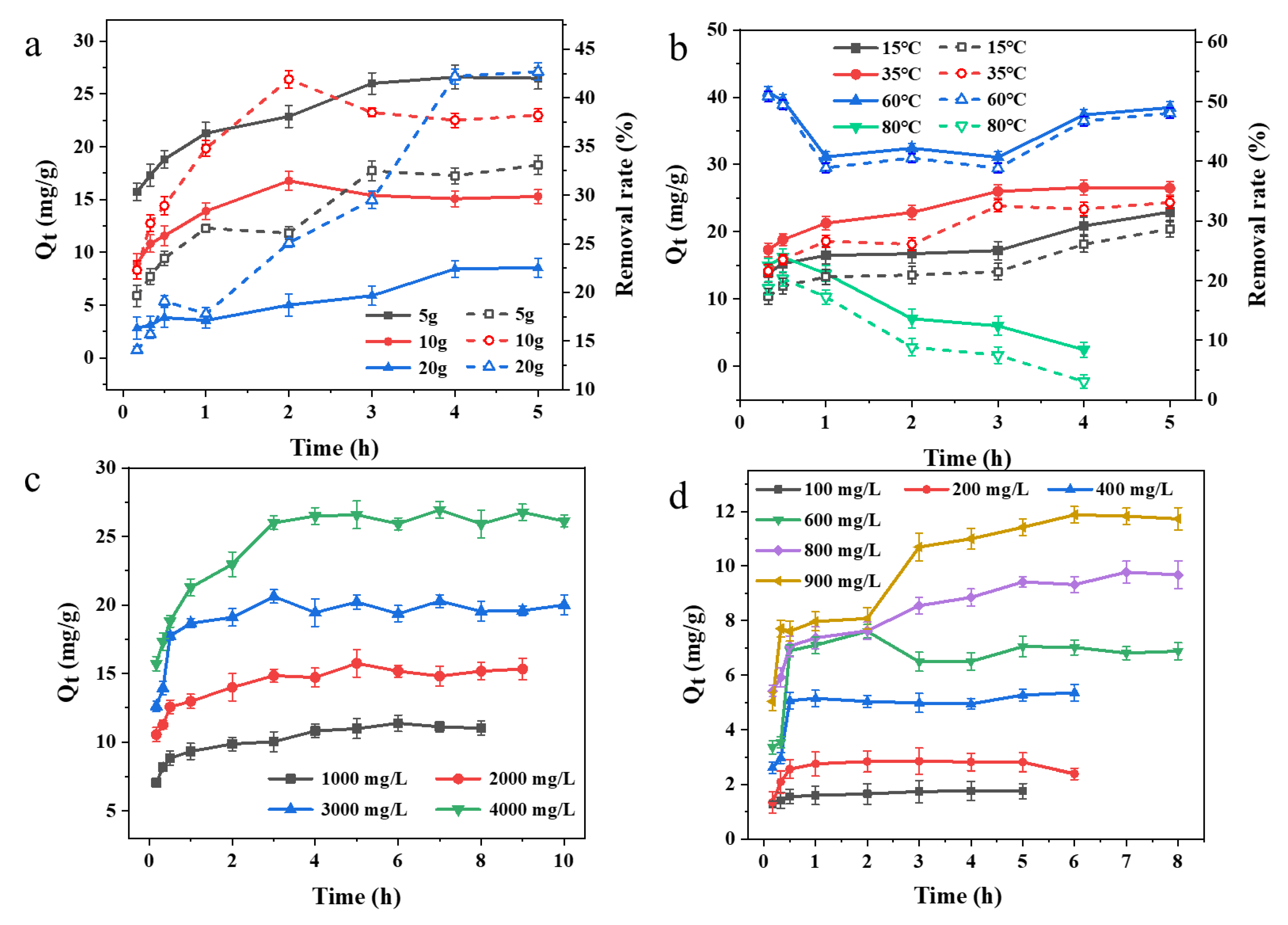
3.2. Adsorption Characteristics of the Chinese Natural Zeolite
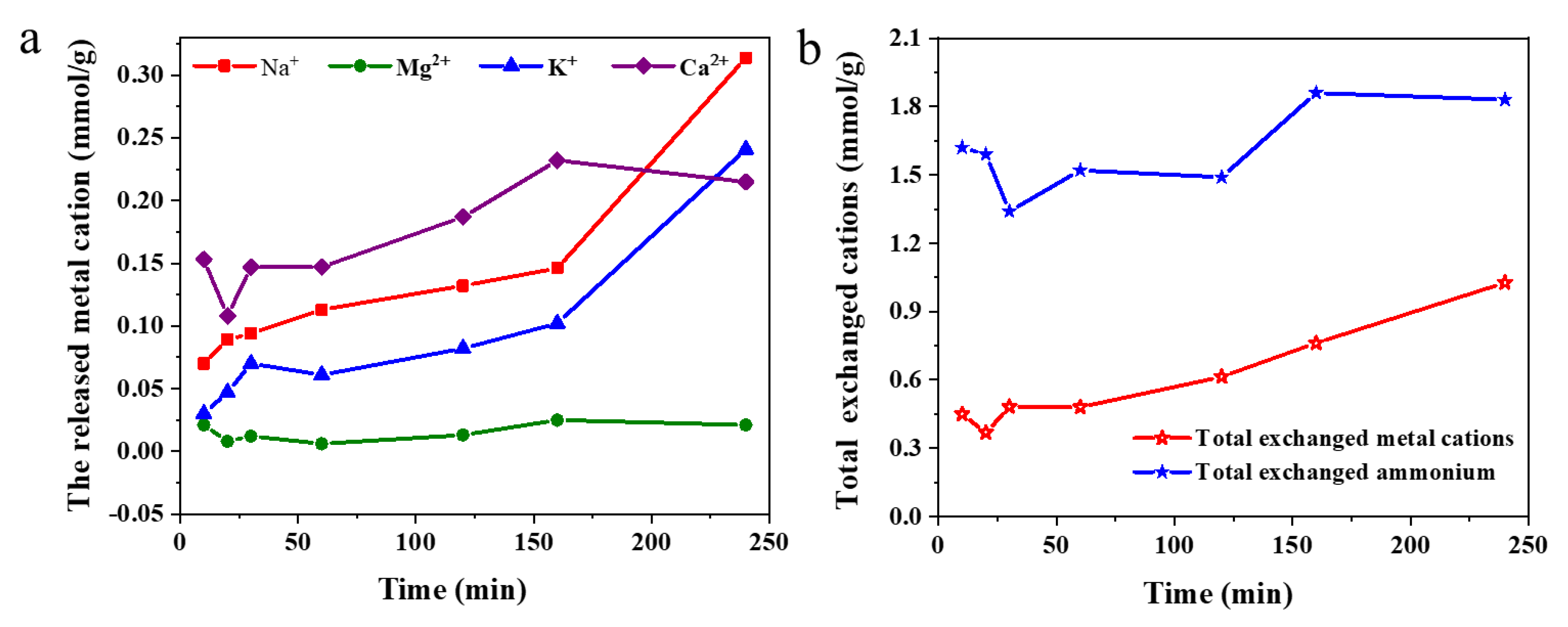
3.3. Total Ion Exchange Capacity and Selectivity for Ammonium
3.4. Adsorption Kinetics and Isotherm Properties
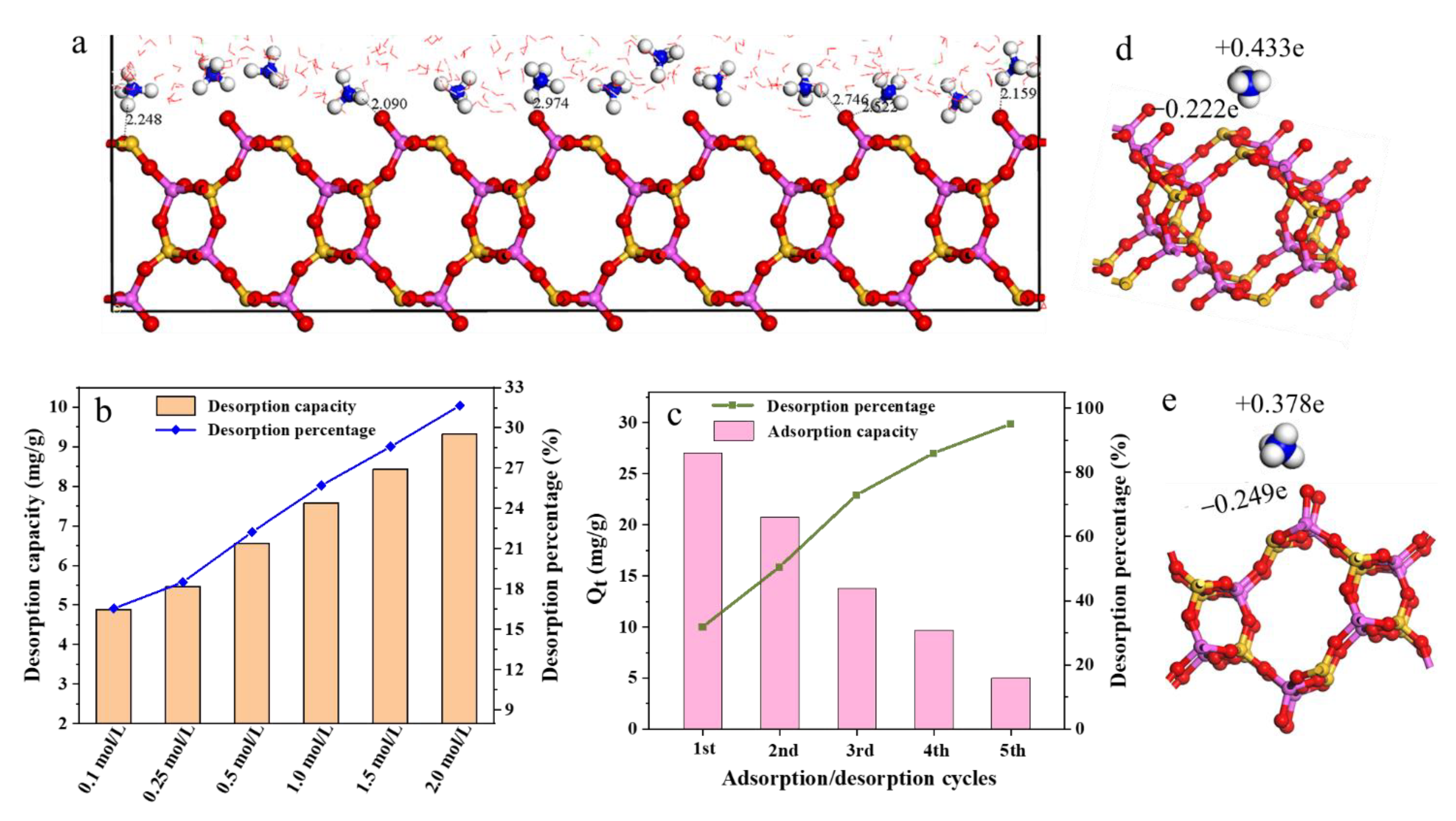
3.5. Simulation of Zeolite’s Ammonium Capture
3.6. Structural Changes of Natural Zeolite after Adsorption of Ultrahigh Concentration NH4+
3.7. Elucidation of Mechanism in Ammonium Adsorption Process
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ates, A. Effect of alkali-treatment on the characteristics of natural zeolites with different compositions. J. Colloid Interface Sci. 2018, 523, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Hudcová, B.; Osacký, M.; Vítková, M.; Mitzia, A.; Komárek, M. Investigation of zinc binding properties onto natural and synthetic zeolites: Implications for soil remediation. Microporous Mesoporous Mater. 2021, 317, 111022. [Google Scholar] [CrossRef]
- Wang, H.; Xu, J.; Sheng, L. Purification mechanism of sewage from constructed wetlands with zeolite substrates: A review. J. Clean. Prod. 2020, 258, 120760. [Google Scholar] [CrossRef]
- Valdés, H.; Alejandro, S.; Zaror, C.A. Natural zeolite reactivity towards ozone: The role of compensating cations. J. Hazard. Mater. 2012, 227–228, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Wan, C.; Lee, D.-J.; Lei, Z.; Liu, X. Ammonium assists orthophosphate removal from high-strength wastewaters by natural zeolite. Sep. Purif. Technol. 2014, 133, 351–356. [Google Scholar] [CrossRef] [Green Version]
- Valdés, H.; Riquelme, A.L.; Solar, V.A.; Azzolina-Jury, F.; Thibault-Starzyk, F. Removal of chlorinated volatile organic compounds onto natural and Cu-modified zeolite: The role of chemical surface characteristics in the adsorption mechanism. Sep. Purif. Technol. 2021, 258, 118080. [Google Scholar] [CrossRef]
- Valdés, H.; Solar, V.A.; Cabrera, E.H.; Veloso, A.F.; Zaror, C.A. Control of released volatile organic compounds from industrial facilities using natural and acid-treated mordenites: The role of acidic surface sites on the adsorption mechanism. Chem. Eng. J. 2014, 244, 117–127. [Google Scholar] [CrossRef]
- Guo, J. Adsorption characteristics and mechanisms of high-levels of ammonium from swine wastewater using natural and MgO modified zeolites. Desalination Water Treat. 2015, 57, 5452–5463. [Google Scholar] [CrossRef]
- Alshameri, A.; Yan, C.; Al-Ani, Y.; Dawood, A.S.; Ibrahim, A.; Zhou, C.; Wang, H. An investigation into the adsorption removal of ammonium by salt activated Chinese (Hulaodu) natural zeolite: Kinetics, isotherms, and thermodynamics. J. Taiwan Inst. Chem. Eng. 2014, 45, 554–564. [Google Scholar] [CrossRef]
- Tang, H.; Xu, X.; Wang, B.; Lv, C.; Shi, D. Removal of Ammonium from Swine Wastewater Using Synthesized Zeolite from Fly Ash. Sustainability 2020, 12, 3423. [Google Scholar] [CrossRef] [Green Version]
- Guaya, D.; Valderrama, C.; Farran, A.; Armijos, C.; Cortina, J.L. Simultaneous phosphate and ammonium removal from aqueous solution by a hydrated aluminum oxide modified natural zeolite. Chem. Eng. J. 2015, 271, 204–213. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Lee, Y.W.; Jahng, D. Ammonia stripping for enhanced biomethanization of piggery wastewater. J. Hazard. Mater. 2012, 199–200, 36–42. [Google Scholar] [CrossRef]
- Yuan, M.-H.; Chen, Y.-H.; Tsai, J.-Y.; Chang, C.-Y. Ammonia removal from ammonia-rich wastewater by air stripping using a rotating packed bed. Process Saf. Environ. Prot. 2016, 102, 777–785. [Google Scholar] [CrossRef]
- Wang, Y.; Pelkonen, M.; Kotro, M. Treatment of High Ammonium–Nitrogen Wastewater from Composting Facilities by Air Stripping and Catalytic Oxidation. Water Air Soil Pollut. 2009, 208, 259–273. [Google Scholar] [CrossRef]
- Huang, J.; Kankanamge, N.R.; Chow, C.; Welsh, D.T.; Li, T.; Teasdale, P.R. Removing ammonium from water and wastewater using cost-effective adsorbents: A review. J. Environ. Sci. 2018, 63, 174–197. [Google Scholar] [CrossRef]
- Abukhadra, M.R.; Mostafa, M. Effective decontamination of phosphate and ammonium utilizing novel muscovite/phillipsite composite; equilibrium investigation and realistic application. Sci. Total Environ. 2019, 667, 101–111. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, Z.; Liu, Y.; Feng, J.; Han, J.; Yan, W. Effective removal of ammonium nitrogen using titanate adsorbent: Capacity evaluation focusing on cation exchange. Sci. Total Environ. 2021, 771, 144800. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Zhu, Q.; Xing, Z. Adsorption of ammonia nitrogen in low temperature domestic wastewater by modification bentonite. J. Clean. Prod. 2019, 233, 720–730. [Google Scholar] [CrossRef]
- Lin, L.; Lei, Z.; Wang, L.; Liu, X.; Zhang, Y.; Wan, C.; Lee, D.-J.; Tay, J.H. Adsorption mechanisms of high-levels of ammonium onto natural and NaCl-modified zeolites. Sep. Purif. Technol. 2013, 103, 15–20. [Google Scholar] [CrossRef] [Green Version]
- Manto, M.; Xie, P.; Keller, M.A.; Liano, W.E.; Pu, T.; Wang, C. Recovery of ammonium from aqueous solutions using ZSM-5. Chemosphere 2018, 198, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Lo, B.T.; Qu, J.; Wilkinson, I.; Hughes, T.; Murray, C.A.; Tang, C.C.; Tsang, S.C.E. Probing atomic positions of adsorbed ammonia molecules in zeolite. Chem. Commun. 2016, 52, 3422–3425. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, E.; Kavanagh, O.N.; Chovan, D.; Madden, D.G.; Cronin, P.; Albadarin, A.B.; Walker, G.M.; Ryan, A. Highly selective trace ammonium removal from dairy wastewater streams by aluminosilicate materials. J. Ind. Eng. Chem. 2020, 86, 39–46. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berendsen, H.J.C.; Grigera, J.R.; Straatsma, T.P. The missing term in effective pair potentials. J. Phys. Chem. 1987, 91, 6269–6271. [Google Scholar] [CrossRef]
- Wiedemair, M.J.; Hofer, T.S. Towards a dissociative SPC-like water model-probing the impact of intramolecular Coulombic contributions. Phys. Chem. Chem. Phys. 2017, 19, 31910–31920. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Ren, P.; Fried, J.R. The COMPASS force field: Parameterization and validation for phosphazenes. Comput. Theor. Polym. Sci. 1998, 8, 229–246. [Google Scholar] [CrossRef]
- Sun, H. COMPASS: An ab initio force-field optimized for condensed-phase applications—overview with details on alkane and benzene compounds. J. Phys. Chem. 1998, 102, 7338–7364. [Google Scholar] [CrossRef]
- Sing, K.S.W. Reporting physisorption data for gas-solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Alshameri, A.; He, H.; Zhu, J.; Xi, Y.; Zhu, R.; Ma, L.; Tao, Q. Adsorption of ammonium by different natural clay minerals: Characterization, kinetics and adsorption isotherms. Appl. Clay Sci. 2018, 159, 83–93. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, B.; Zhang, X.; Wang, J.; Liu, J.; Chen, R. Preparation of highly ordered cubic NaA zeolite from halloysite mineral for adsorption of ammonium ions. J. Hazard. Mater. 2010, 178, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Peng, Y. Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- Malekian, R.; Abedi-Koupai, J.; Eslamian, S.S.; Mousavi, S.F.; Abbaspour, K.C.; Afyuni, M. Ion-exchange process for ammonium removal and release using natural Iranian zeolite. Appl. Clay Sci. 2011, 51, 323–329. [Google Scholar] [CrossRef]
- Huang, H.; Xiao, X.; Yan, B.; Yang, L. Ammonium removal from aqueous solutions by using natural Chinese (Chende) zeolite as adsorbent. J. Hazard. Mater. 2010, 175, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Alshameri, A.; Ibrahim, A.; Assabri, A.M.; Lei, X.; Wang, H.; Yan, C. The investigation into the ammonium removal performance of Yemeni natural zeolite: Modification, ion exchange mechanism, and thermodynamics. Powder Technol. 2014, 258, 20–31. [Google Scholar] [CrossRef]
- Inglezakis, V.J. The concept of "capacity" in zeolite ion-exchange systems. J. Colloid Interface Sci. 2005, 281, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Yamada, H.; Kokusen, H.; Tanaka, J.; Moriyoshi, Y.; Komatsu, Y. Ion Exchange Behavior of Natural Zeolites in Distilled Water, Hydrochloric Acid, and Ammonium Chloride Solution. Sep. Sci. Technol. 2007, 38, 1519–1532. [Google Scholar] [CrossRef]
- Kantiranis, N.; Sikalidis, K.; Godelitsas, A.; Squires, C.; Papastergios, G.; Filippidis, A. Extra-framework cation release from heulandite-type rich tuffs on exchange with NH4+. J. Environ. Manag. 2011, 92, 1569–1576. [Google Scholar] [CrossRef]
- Li, X.; Zhang, D.; Sheng, F.; Qing, H. Adsorption characteristics of Copper (II), Zinc (II) and Mercury (II) by four kinds of immobilized fungi residues. Ecotoxicol. Environ. Saf. 2018, 147, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Alasadi, A.M.; Khaili, F.I.; Awwad, A.M. Adsorption of Cu(II), Ni(II) and Zn(II) Ions by Nano Kaolinite Thermodynamics and Kinetics Studies. J. Environ. Manag. 2019, 246, 547–556. [Google Scholar] [CrossRef]
- Iqbal, M.; Abbas, M.; Nisar, J.; Nazir, A.; Qamar, A. Bioassays based on higher plants as excellent dosimeters for ecotoxicity monitoring: A review. Chem. Int. 2019, 5, 1–80. [Google Scholar] [CrossRef]
- Awwad, A.M.; Amer, M.W.; Al-aqarbeh, M.M. TiO2-Kaolinite Nanocomposite Prepared from the Jordanian Kaolin Clay Adsorption and Thermodynamics of Pb(II) and Cd(II) Ions in Aqueous Solution. Chem. Int. 2020, 6, 168–178. [Google Scholar] [CrossRef]
- Lima, E.C.; Hosseini-Bandegharaei, A.; Moreno-Piraján, J.C.; Anastopoulos, I. A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van't Hoof equation for calculation of thermodynamic parameters of adsorption. J. Mol. Liq. 2019, 273, 425–434. [Google Scholar] [CrossRef]
- Benhammou, A.; Yaacoubi, A.; Nibou, L.; Tanouti, B. Adsorption of metal ions onto Moroccan stevensite: Kinetic and isotherm studies. J. Colloid Interface Sci. 2005, 282, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Shaban, M.; Abukhadra, M.R.; Shahien, M.G.; Ibrahim, S.S. Novel bentonite/zeolite-NaP composite efficiently removes methylene blue and Congo red dyes. Environ. Chem. Lett. 2017, 16, 275–280. [Google Scholar] [CrossRef]
- Gilli, G.; Gilli, P. Towards an unified hydrogen-bond theory. J. Mol. Struct. 2000, 552, 1–15. [Google Scholar] [CrossRef]
- Rakhym, A.B.; Seilkhanova, G.A.; Mastai, Y. Physicochemical evaluation of the effect of natural zeolite modification with didodecyldimethylammonium bromide on the adsorption of Bisphenol-A and Propranolol Hydrochloride. Microporous Mesoporous Mater. 2021, 318, 111020. [Google Scholar] [CrossRef]
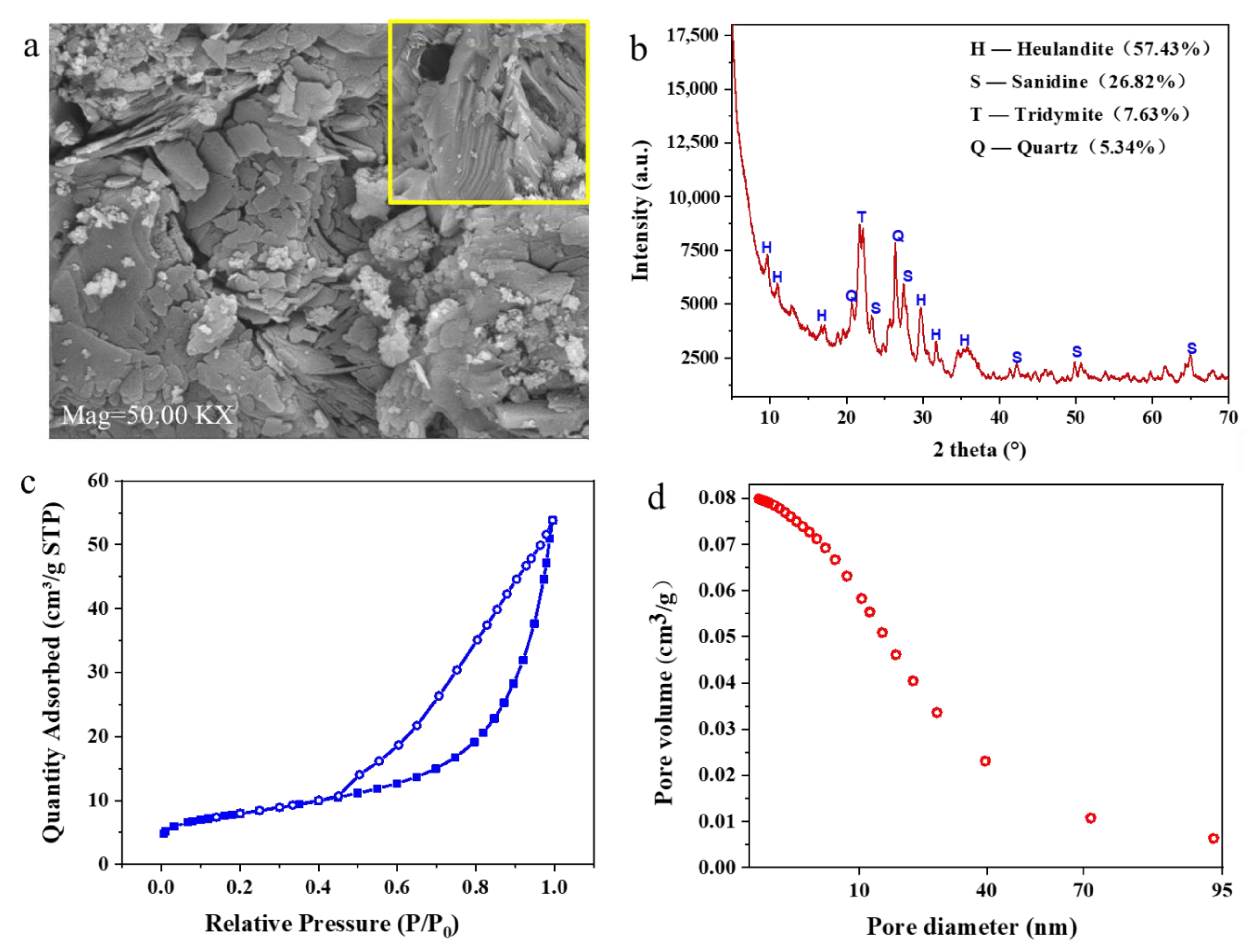

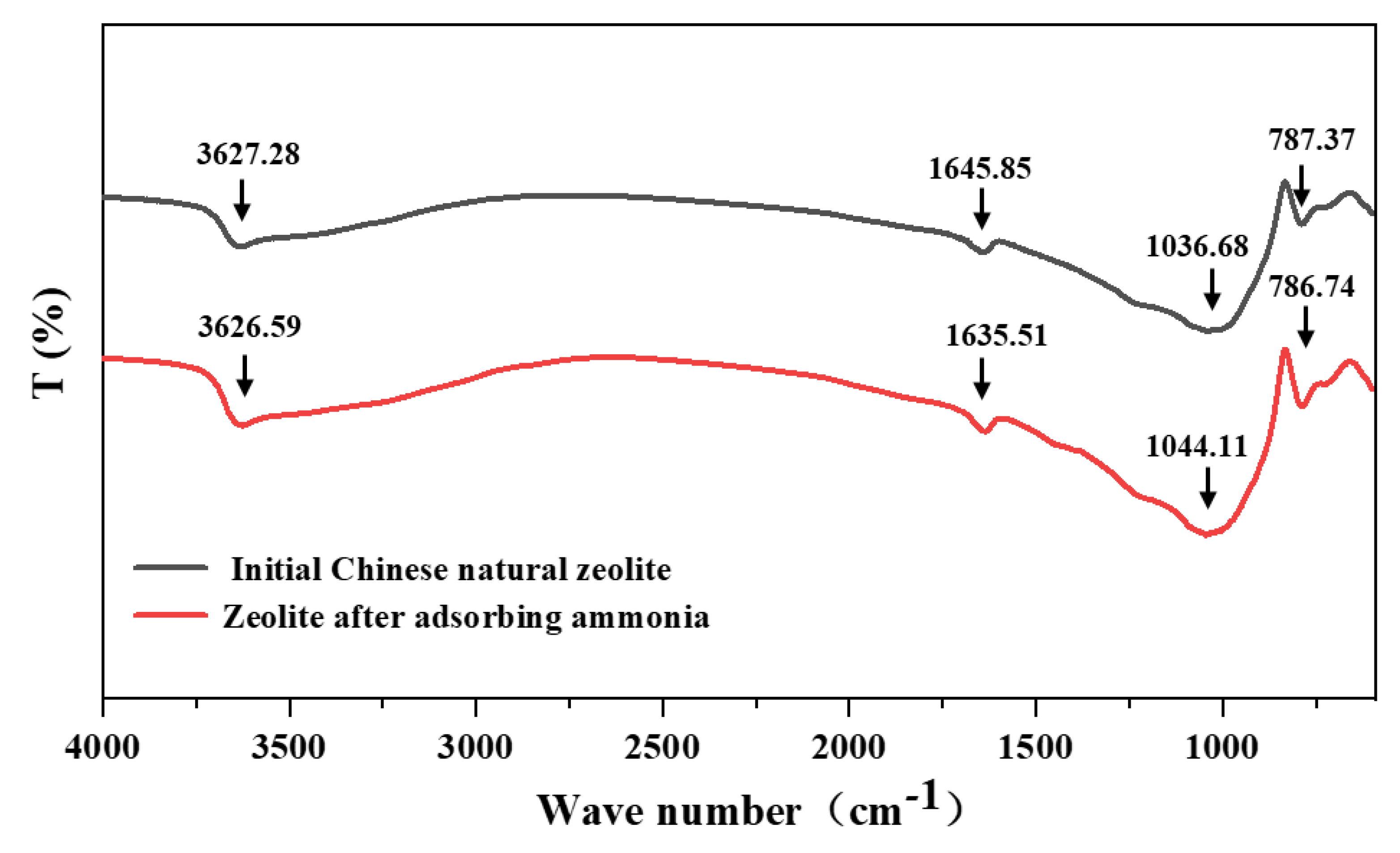
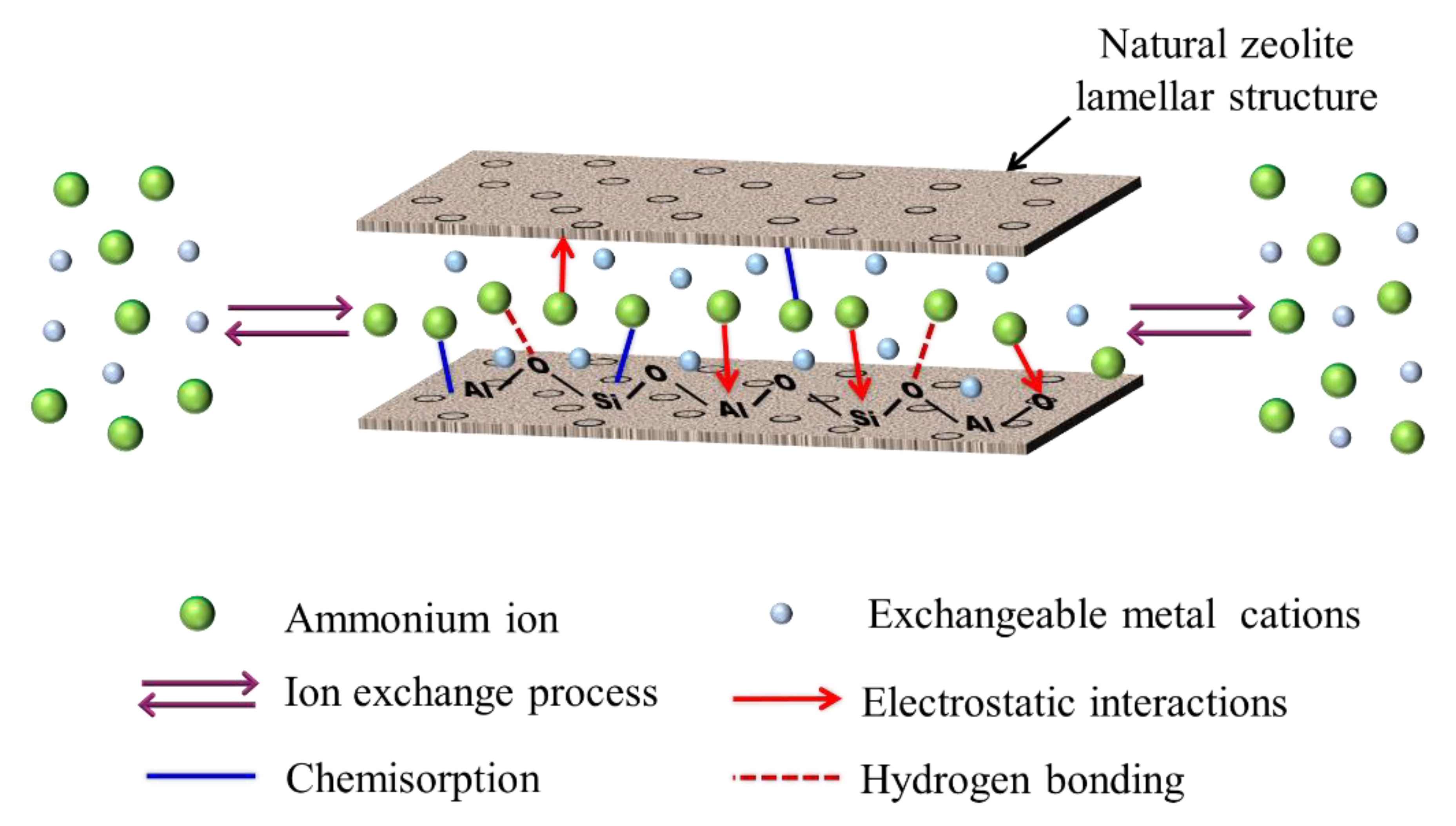
| Sample Name | Capacity (mg/g) | BET Surface Area (m2/g) | Mean Pore Diameter (nm) | Micropores Volume (cm3/g) | Mesopores Volume (cm3/g) | Total Volume (cm3/g) | Reference |
|---|---|---|---|---|---|---|---|
| Chinese natural zeolite | 26.94 | 28.15 | 11.87 | 0.00271 | 0.0559 | 0.0586 | this study |
| Natural zeolite | 14.3 | 14.33 | 16.19 | 0.044 | [19] | ||
| NaCl-modified zeolite | 17.3 | 60.83 | 29.24 | 0.065 | [19] |
| Chemical Elements | wt.% | Chemical Elements | wt.% |
|---|---|---|---|
| O | 47.12 | Na | 0.82 |
| Si | 26.87 | Ca | 0.77 |
| Al | 5.72 | Mg | 0.34 |
| K | 2.76 | others | 14.2 |
| Fe | 1.40 |
| Samples | C0 (mg/L) | Equilibrium Time (min) | Qexp,max 1 (mg/g) | QLan,max 2 (mg/g) | Reference |
|---|---|---|---|---|---|
| Natural zeolite | 1000~4000 | 180 | 26.94 | 27.06 | this study |
| Iranian zeolite | 90~3620 | 60 | 13.27 | 11.52 | [32] |
| Natural Chinese (Chende) zeolite | 50~300 | 180 | 9.41 | 9.41 | [33] |
| Na-Yemeni natural zeolite | 10~250 | 20 | 11.2 | [34] | |
| Modified bentonite | 0~350 | 60 | 5.85 | 5.8503 ± 0.08 | [18] |
| Langmuir | Freundlich | ||||
| qm | KL | R2 | KF | 1/n | R2 |
| 27.06 | 0.0015 | 0.935 | 0.872 | 0.415 | 0.969 |
| Pseudo First-Order | Pseudo Second-Order | ||||
| qe | k1 | R2 | qe | k2 | R2 |
| 25.56 | 0.059 | 0.754 | 26.65 | 0.016 | 0.927 |
| T (K) | Kc (×10−3) | ΔG0 (kJ·mol−1) | ΔH0 (kJ·mol−1) | ΔS0 (J·mol−1·K−1) |
|---|---|---|---|---|
| 288 | 0.58 | −15.52 | 21.36 | 128.05 |
| 298 | 0.95 | −16.80 | ||
| 308 | 1.23 | −18.21 | ||
| 323 | 1.62 | −19.85 | ||
| 338 | 2.42 | −21.89 |
| Adsorption Site | Bridged O Site | Unbonded O Site | Si Site | Al Site |
|---|---|---|---|---|
| Eads,cal (kJ/mol) | −86.77 | −136.77 | −40.90 | −61.32 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, P.; Zhang, A.; Liu, Y.; Liu, Z.; Liu, X.; Yang, L.; Yang, Z. Adsorption Mechanism of High-Concentration Ammonium by Chinese Natural Zeolite with Experimental Optimization and Theoretical Computation. Water 2022, 14, 2413. https://doi.org/10.3390/w14152413
Liu P, Zhang A, Liu Y, Liu Z, Liu X, Yang L, Yang Z. Adsorption Mechanism of High-Concentration Ammonium by Chinese Natural Zeolite with Experimental Optimization and Theoretical Computation. Water. 2022; 14(15):2413. https://doi.org/10.3390/w14152413
Chicago/Turabian StyleLiu, Pan, Aining Zhang, Yongjun Liu, Zhe Liu, Xingshe Liu, Lu Yang, and Zhuangzhuang Yang. 2022. "Adsorption Mechanism of High-Concentration Ammonium by Chinese Natural Zeolite with Experimental Optimization and Theoretical Computation" Water 14, no. 15: 2413. https://doi.org/10.3390/w14152413
APA StyleLiu, P., Zhang, A., Liu, Y., Liu, Z., Liu, X., Yang, L., & Yang, Z. (2022). Adsorption Mechanism of High-Concentration Ammonium by Chinese Natural Zeolite with Experimental Optimization and Theoretical Computation. Water, 14(15), 2413. https://doi.org/10.3390/w14152413






