Resource Utilization of Acid Mine Drainage (AMD): A Review
Abstract
:1. Introduction
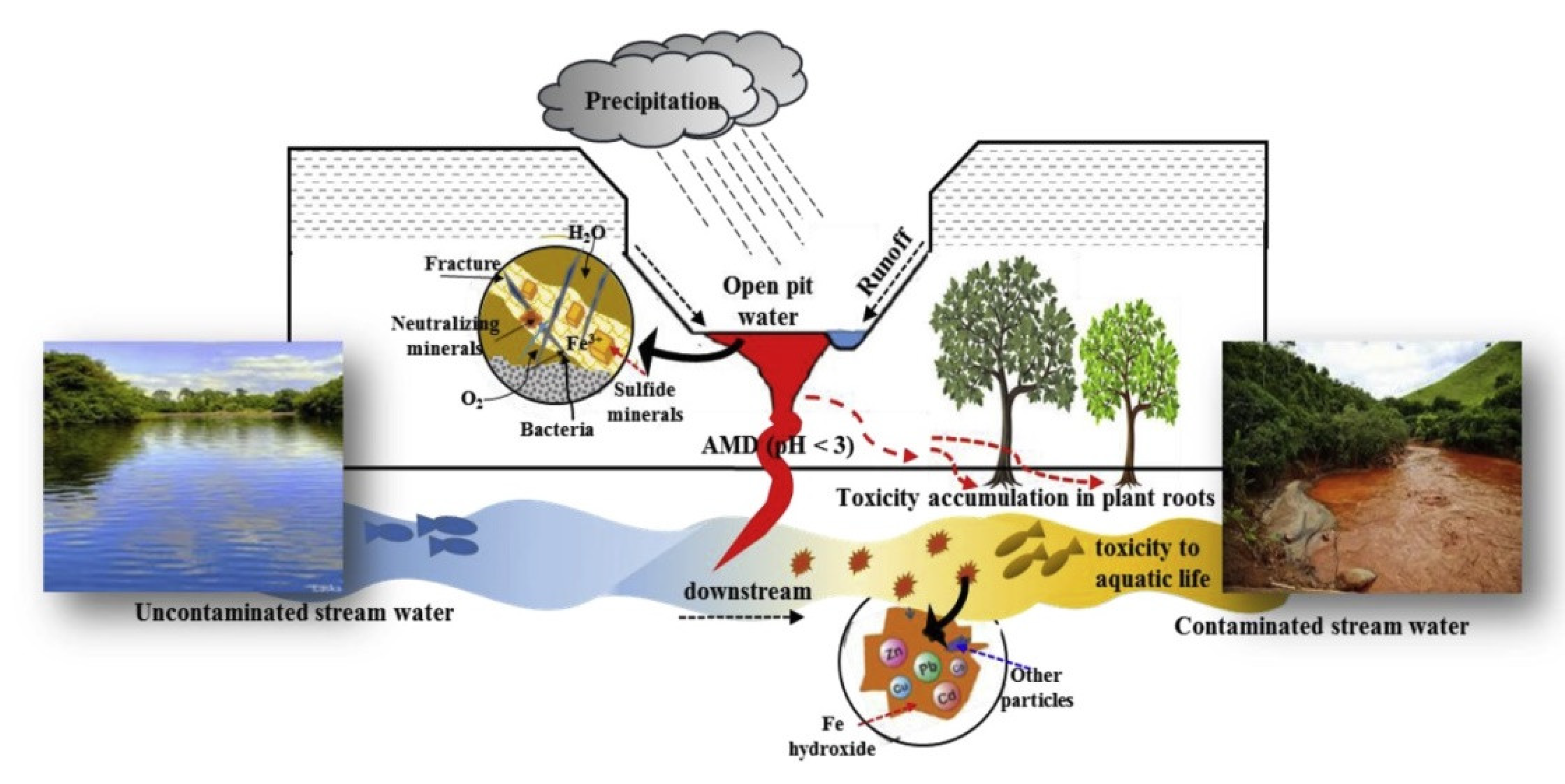
2. Formation and Hazard of Acid Mine Drainage
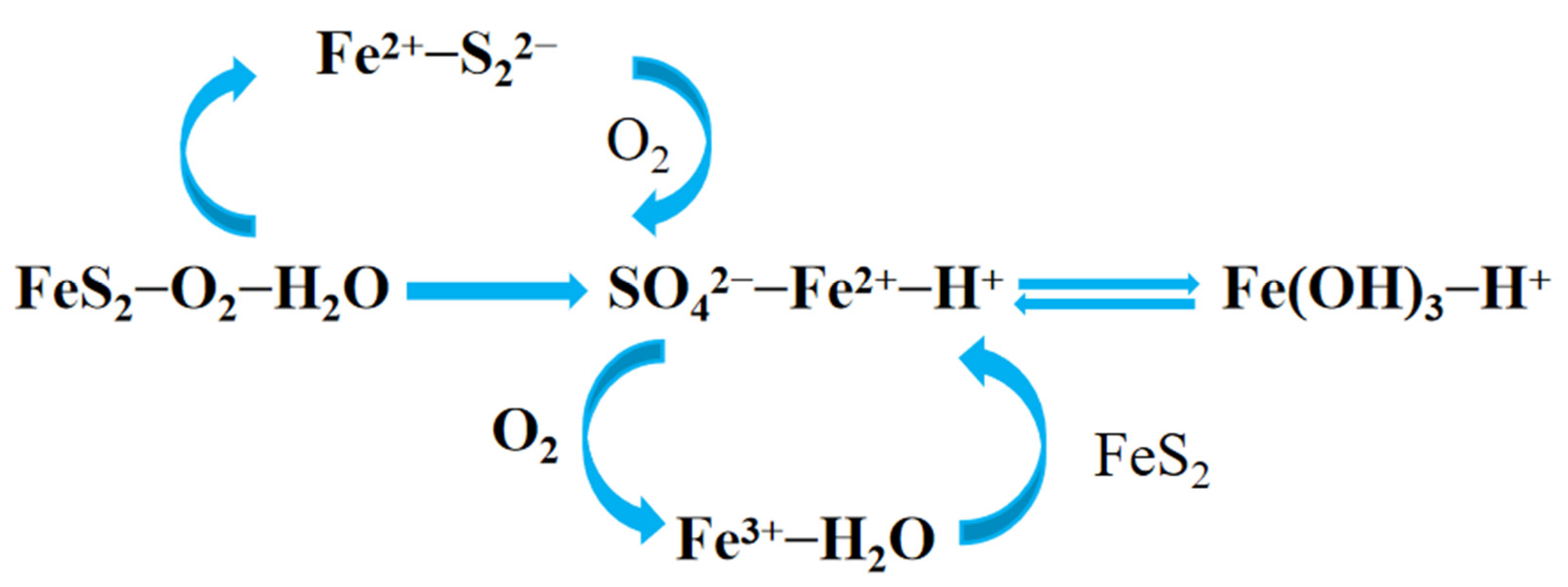
2.1. Formation of Acid Mine Drainage
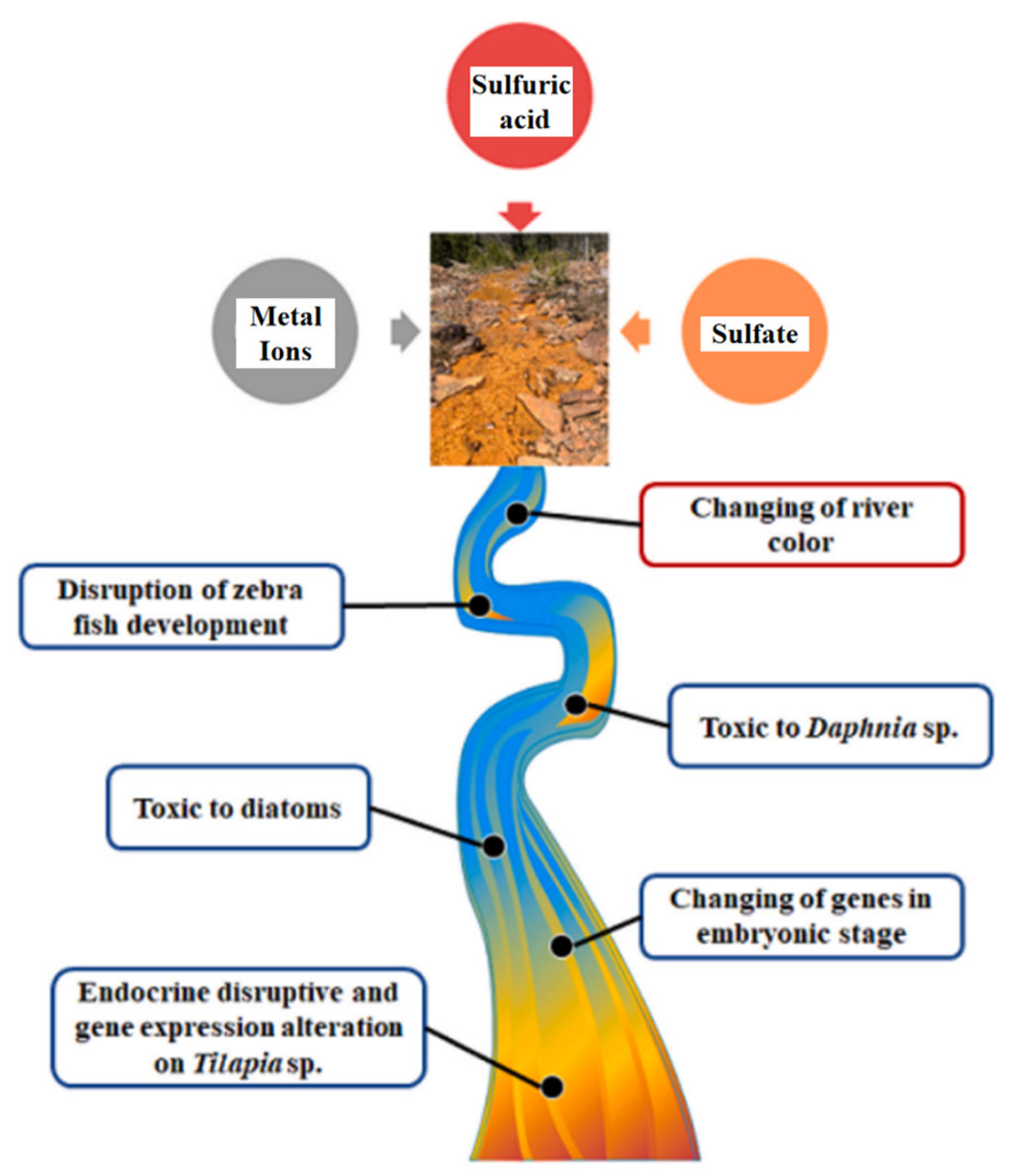
2.2. Hazards of Acid Mine Drainage
3. Resource Utilization of Acid Mine Drainage
3.1. Recovery of Water Resources from AMD
3.2. Recovery of Sulfuric Acid
3.3. Subsection Recovery of Valuable Metals and Synthetics
3.3.1. Recovery of Dissolved Metal Ions from AMD
3.3.2. Synthesis and Application of Valuable Minerals
4. Application of AMD in Fenton Oxidation Technology
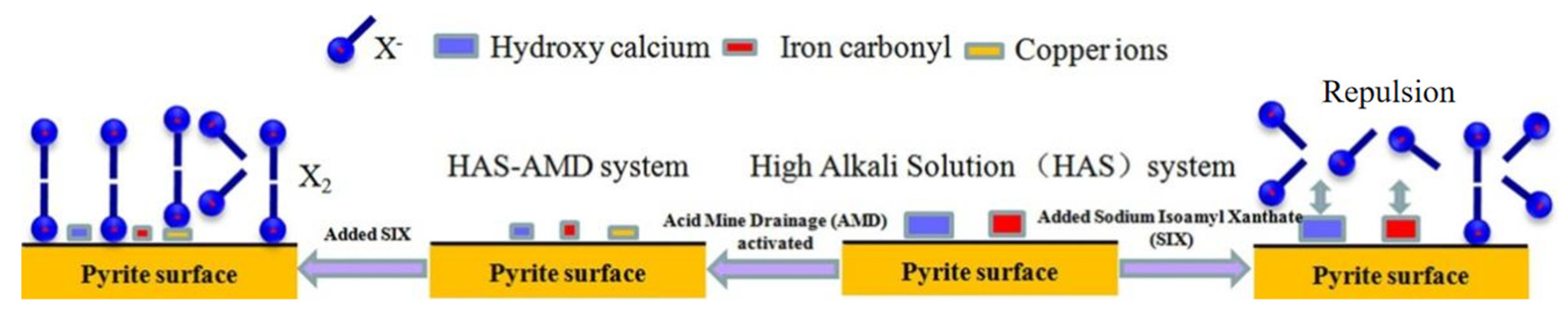
5. Application of AMD in Beneficiation
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Levio-Raiman, M.; Briceño, G.; Schalchli, H.; Bornhardt, C.; Diez, C.M. Alternative treatment for metal ion removal from acid mine drainage using an organic biomixture as a low cost adsorbent. Environ. Technol. Innov. 2021, 24, 101853. [Google Scholar] [CrossRef]
- Rambabu, K.; Banat, F.; Quan, M.P.; Ho, S.H.; Ren, N.Q.; Show, P.L. Biological remediation of acid mine drainage: Review of past trends and current outlook. Environ. Sci. Technol. 2020, 2, 100024. [Google Scholar] [CrossRef]
- Tang, H.; Luo, J.Z.; Zheng, L.W.; Liu, C.Y.; Li, H.H.; Wu, G.N.; Zeng, M.L.; Bai, X.S. Characteristics of pores in coals exposed to acid mine drainage. Energy. Rep. 2021, 7, 8772–8783. [Google Scholar] [CrossRef]
- Wang, Z.L.; Xu, Y.X.; Zhang, Z.X.; Zhang, Y.B. Review: Acid Mine Drainage (AMD) in Abandoned Coal Mines of Shanxi, China. Water 2020, 13, 8. [Google Scholar] [CrossRef]
- Yang, B.J.; Luo, W.; Hong, M.X.; Wang, J.; Liu, X.D.; Gan, M.; Qiu, G.Z. Inhibition of hematite on acid mine drainage caused by chalcopyrite biodissolution. Chin. J. Chem. Eng. 2022, 44, 94–104. [Google Scholar] [CrossRef]
- Carneiro Brandao Pereira, T.; Batista Dos Santos, K.; Lautert-Dutra, W.; de Souza Teodoro, L.; de Almeida, V.O.; Weiler, J.; Homrich Schneider, I.A.; Reis Bogo, M. Acid mine drainage (AMD) treatment by neutralization: Evaluation of physical-chemical performance and ecotoxicological effects on zebrafish (Danio rerio) development. Chemosphere 2020, 253, 126665. [Google Scholar] [CrossRef]
- Li, Q.; Ji, B.; Honaker, R.; Noble, A.; Zhang, W. Partitioning behavior and mechanisms of rare earth elements during precipitation in acid mine drainage. Colloids Surf. Physicochem. Eng. Asp. 2022, 641, 128563. [Google Scholar] [CrossRef]
- Núñez-Gómez, D.; Rodrigues, C.; Lapolli, F.R.; Lobo-Recio, M.Á. Adsorption of heavy metals from coal acid mine drainage by shrimp shell waste: Isotherm and continuous-flow studies. J. Environ. Chem. Eng. 2018, 7, 102787. [Google Scholar] [CrossRef]
- Alegbe, M.J.; Ayanda, O.S.; Ndungu, P.; Nechaev, A.; Petrik, L.F. Physicochemical Characteristics of Acid Mine Drainage, Simultaneous Remediation and use as Feedstock for Value Added Products. J. Environ. Chem. Eng. 2019, 7, 103097. [Google Scholar] [CrossRef]
- Fernández-Caliani, J.C.; Giráldez, M.I.; Barba-Brioso, C. Oral bioaccessibility and human health risk assessment of trace elements in agricultural soils impacted by acid mine drainage. Chemosphere 2019, 237, 124441. [Google Scholar] [CrossRef]
- Tomiyama, S.; Igarashi, T. The potential threat of mine drainage to groundwater resources. Curr. Opin. Environ. Sci. Health 2022, 27, 100347. [Google Scholar] [CrossRef]
- Aguiar, A.O.; Andrade, L.H.; Ricci, B.C.; Pires, W.L.; Miranda, G.A.; Amaral, M. Gold acid mine drainage treatment by membrane separation processes: An evaluation of the main operational conditions. Sep. Purif. Technol. 2016, 170, 360–369. [Google Scholar] [CrossRef]
- Kiiskila, J.D.; Sarkar, D.; Panja, S.; Sahi, S.V.; Datta, R. Remediation of acid mine drainage-impacted water by vetiver grass (Chrysopogon zizanioides): A multiscale long-term study. Ecol. Eng. 2019, 129, 97–108. [Google Scholar] [CrossRef]
- Magowo, W.E.; Sheridan, C.; Rumbold, K. Global Co-occurrence of Acid Mine Drainage and Organic Rich Industrial and Domestic Effluent: Biological sulfate reduction as a co-treatment-option. J. Water Process. 2020, 38, 101650. [Google Scholar] [CrossRef]
- Kefeni, K.K.; Msagati, T.A.M.; Mamba, B.B. Acid mine drainage: Prevention, treatment options, and resource recovery: A review. J. Clean. Prod. 2017, 151, 475–493. [Google Scholar] [CrossRef]
- Silva, D.; Weber, C.; Oliveira, C. Neutralization and uptake of pollutant cations from acid mine drainage (amd) using limestones and zeolites in a pilot-scale passive treatment system. Miner. Eng. 2021, 170, 107000. [Google Scholar] [CrossRef]
- Öztürk, Y.; Ekmekçi, Z. Removal of sulfate ions from process water by ion exchange resins. Miner. Eng. 2020, 159, 106613. [Google Scholar] [CrossRef]
- Geoffroy, N.; Demopoulos, G.P. The elimination of selenium(IV) from aqueous solution by precipitation with sodium sulfide. J. Hazard. Mater. 2011, 185, 148–154. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, Y.; Zhang, Z.; Li, H.; Wu, A.; Shi, H. Experimental research on various slags as a potential adsorbent for the removal of sulfate from acid mine drainage. J. Environ. Manag. 2020, 270, 110880. [Google Scholar] [CrossRef]
- Aguiar, A.; Andrade, L.; Grossi, L.; Pires, W.; Amaral, M. Acid mine drainage treatment by nanofiltration: A study of membrane fouling, chemical cleaning, and membrane ageing. Sep. Purif. Technol. 2018, 192, 185–195. [Google Scholar] [CrossRef]
- Foureaux, A.F.S.; Moreira, V.R.; Lebron, Y.A.R.; Santos, L.V.S.; Amaral, M.C.S. Direct contact membrane distillation as an alternative to the conventional methods for value-added compounds recovery from acidic effluents: A review. Sep. Purif. Technol. 2020, 236, 116251. [Google Scholar] [CrossRef]
- Chen, G.; Ye, Y.C.; Yao, N.; Hu, N.Y.; Zhang, J.; Huang, Y. A critical review of prevention, treatment, reuse, and resource recovery from acid mine drainage. J. Cleaner Prod. 2021, 329, 129666. [Google Scholar] [CrossRef]
- Nleya, Y.; Simate, G.S.; Ndlovu, S. Sustainability assessment of the recovery and utilisation of acid from acid mine drainage. J. Clean. Prod. 2016, 113, 17–27. [Google Scholar] [CrossRef]
- Crane, R.A.; Sapsford, D.J. Towards “Precision Mining” of wastewater: Selective recovery of Cu from acid mine drainage onto diatomite supported nanoscale zerovalent iron particles. Chemosphere 2018, 202, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Godoi, L.A.G.; Foresti, E.; Damianovic, M. Down-flow fixed-structured bed reactor: An innovative reactor configuration applied to acid mine drainage treatment and metal recovery. J. Environ. Manag. 2017, 197, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Naidu, G.; Ryu, S.; Thiruvenkatachari, R.; Choi, Y.; Jeong, S.; Vigneswaran, S. A critical review on remediation, reuse, and resource recovery from acid mine drainage. Environ. Pollut. 2019, 247, 1110–1124. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.P.; Ponou, J.; Matsuo, S.; Okaya, K.; Dodbiba, G.; Nazuka, T.; Fujita, T. Integrating sulfidization with neutralization treatment for selective recovery of copper and zinc over iron from acid mine drainage. Miner. Eng. 2013, 45, 100–107. [Google Scholar] [CrossRef]
- Flores, R.G.; Andersen, S.L.; Maia, L.K.; Jose, H.J.; Moreira Rde, F. Recovery of iron oxides from acid mine drainage and their application as adsorbent or catalyst. J. Environ. Manag. 2012, 111, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Kefeni, K.K.; Msagati, T.M.; Mamba, B.B. Synthesis and characterization of magnetic nanoparticles and study their removal capacity of metals from acid mine drainage. Chem. Eng. J. 2015, 276, 222–231. [Google Scholar] [CrossRef]
- Moreira, R.F.P.M.; Vandresen, S.; Luiz, D.B.; José, H.J.; Puma, G.L. Adsorption of arsenate, phosphate and humic acids onto acicular goethite nanoparticles recovered from acid mine drainage. J. Environ. Chem. Eng. 2017, 5, 652–659. [Google Scholar] [CrossRef]
- Ioffe, M.; Kundu, S.; Perez-Lapid, N.; Radian, A. Heterogeneous Fenton catalyst based on clay decorated with nano-sized amorphous iron oxides prevents oxidant scavenging through surface complexation. Chem. Eng. J. 2022, 433, 134609. [Google Scholar] [CrossRef]
- Baskar, A.V.; Bolan, N.; Hoang, S.A.; Sooriyakumar, P.; Kumar, M.; Singh, L.; Jasemizad, T.; Padhye, L.P.; Singh, G.; Vinu, A.; et al. Recovery, regeneration and sustainable management of spent adsorbents from wastewater treatment streams: A review. Sci. Total Environ. 2022, 822, 153555. [Google Scholar] [CrossRef]
- Rait, R.; Trumm, D.; Pope, J.; Craw, D.; Newman, N.; MacKenzie, H. Adsorption of arsenic by iron rich precipitates from two coal mine drainage sites on the West Coast of New Zealand. N. Z. J. Geol. Geophys. 2010, 53, 177–193. [Google Scholar] [CrossRef]
- Garrido-Ramírez, E.G.; Theng, B.K.G.; Mora, M.L. Clays and oxide minerals as catalysts and nanocatalysts in Fenton-like reactions—A review. Appl. Clay Sci. 2010, 47, 182–192. [Google Scholar] [CrossRef]
- Bai, S.J.; Bi, Y.X.; Li, J.; Yu, P.; Ding, Z.; Lv, C.; Wen, S.M. Innovative utilization of acid mine drainage (AMD): A promising activator for pyrite flotation once depressed in a high alkali solution (HAS)–Gearing towards a cleaner production concept of copper sulfide ore. Miner. Eng. 2021, 170, 106997. [Google Scholar] [CrossRef]
- Akcil, A.; Koldas, S. Acid Mine Drainage (AMD): Causes, treatment and case studies. J. Clean. Prod. 2006, 14, 1139–1145. [Google Scholar] [CrossRef]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef]
- Lin, S.Y.; Liu, R.Q.; Wu, M.R.; Hu, Y.H.; Sun, W.; Shi, Z.Z.; Han, H.S.; Li, W.H. Minimizing beneficiation wastewater through internal reuse of process water in flotation circuit. J. Clean. Prod. 2020, 245, 118898. [Google Scholar] [CrossRef]
- Acharya, B.S.; Kharel, G. Acid mine drainage from coal mining in the United States—An overview. J. Hydrol. 2020, 588, 125061. [Google Scholar] [CrossRef]
- Chen, Y.T.; Li, J.T.; Chen, L.X.; Hua, Z.S.; Huang, L.N.; Liu, J.; Xu, B.B.; Liao, B.; Shu, W.S. Biogeochemical processes governing natural pyrite oxidation and release of acid metalliferous drainage. Environ. Sci. Technol. 2014, 48, 5537–5545. [Google Scholar] [CrossRef]
- Ouyang, Y.; Liu, Y.; Zhu, R.; Ge, F.; Xu, T.; Luo, Z.; Liang, L. Pyrite oxidation inhibition by organosilane coatings for acid mine drainage control. Miner. Eng. 2015, 72, 57–64. [Google Scholar] [CrossRef]
- Park, I.; Tabelin, C.B.; Jeon, S.; Li, X.; Seno, K.; Ito, M.; Hiroyoshi, N. A review of recent strategies for acid mine drainage prevention and mine tailings recycling. Chemosphere 2019, 219, 588–606. [Google Scholar] [CrossRef]
- Tu, Z.; Wu, Q.; He, H.; Zhou, S.; Liu, J.; He, H.; Liu, C.; Dang, Z.; Reinfelder, J.R. Reduction of acid mine drainage by passivation of pyrite surfaces: A review. Sci. Total Environ. 2022, 832, 155116. [Google Scholar] [CrossRef]
- Chopard, A.; Benzaazoua, M.; Bouzahzah, H.; Plante, B.; Marion, P. A contribution to improve the calculation of the acid generating potential of mining wastes. Chemosphere 2017, 175, 97–107. [Google Scholar] [CrossRef]
- Khummalai, N.; Boonamnuayvitaya, V. Suppression of arsenopyrite surface oxidation by sol-gel coatings. J. Biosci. Bioeng. 2005, 99, 277–284. [Google Scholar] [CrossRef]
- Sahoo, P.K.; Kim, K.; Equeenuddin, S.M.; Powell, M.A. Current approaches for mitigating acid mine drainage. Rev. Environ. Contam. Toxicol. 2013, 226, 1–32. [Google Scholar] [CrossRef]
- Buzzi, D.C.; Viegas, L.S.; Rodrigues, M.A.S.; Bernardes, A.M.; Tenório, J.A.S. Water recovery from acid mine drainage by electrodialysis. Miner. Eng. 2013, 40, 82–89. [Google Scholar] [CrossRef]
- Simate, G.S.; Ndlovu, S. Acid mine drainage: Challenges and opportunities. J. Environ. Chem. Eng. 2014, 2, 1785–1803. [Google Scholar] [CrossRef]
- Thomas, G.; Sheridan, C.; Holm, P.E. A critical review of phytoremediation for acid mine drainage-impacted environments. Sci. Total Environ. 2022, 811, 152230. [Google Scholar] [CrossRef]
- Ighalo, J.O.; Kurniawan, S.B.; Iwuozor, K.O.; Aniagor, C.O.; Ajala, O.J.; Oba, S.N.; Iwuchukwu, F.U.; Ahmadi, S.; Igwegbe, C.A. A review of treatment technologies for the mitigation of the toxic environmental effects of acid mine drainage (AMD). Process. Saf. Environ. Prot. 2022, 157, 37–58. [Google Scholar] [CrossRef]
- Song, Y.W.; Guo, Z.H.; Wang, R.; Yang, L.L.; Cao, Y.X.; Wang, H.R. A novel approach for treating acid mine drainage by forming schwertmannite driven by a combination of biooxidation and electroreduction before lime neutralization. Water Res. 2022, 221, 118748. [Google Scholar] [CrossRef] [PubMed]
- Masindi, V. Recovery of drinking water and valuable minerals from acid mine drainage using an integration of magnesite, lime, soda ash, CO2 and reverse osmosis treatment processes. J. Environ. Chem. Eng. 2017, 5, 3136–3142. [Google Scholar] [CrossRef]
- España, J.S.; Esther, S.P.; Enrique, L.P. Iron terraces in acid mine drainage systems: A discussion about the organic and inorganic factors involved in their formation through observations from the Tintillo acidic river (Riotinto mine, Huelva, Spain). Geosphere 2007, 3, 133–151. [Google Scholar] [CrossRef] [Green Version]
- Brewster, E.T.; Freguia, S.; Edraki, M.; Berry, L.; Ledezma, P. Staged electrochemical treatment guided by modelling allows for targeted recovery of metals and rare earth elements from acid mine drainage. J. Environ. Manag. 2020, 275, 111266. [Google Scholar] [CrossRef]
- Balistrieri, L.S.; Seal, R.R.; Piatak, N.M.; Paul, B. Assessing the concentration, speciation, and toxicity of dissolved metals during mixing of acid-mine drainage and ambient river water downstream of the Elizabeth Copper Mine, Vermont, USA. Appl. Geochem. 2007, 22, 930–952. [Google Scholar] [CrossRef]
- Ambiado, K.; Bustos, C.; Schwarz, A.; Borquez, R. Membrane technology applied to acid mine drainage from copper mining. Water Sci. Technol. 2017, 75, 705–715. [Google Scholar] [CrossRef]
- Seo, E.Y.; Cheong, Y.W.; Yim, G.J.; Min, K.W.; Geroni, J.N. Recovery of Fe, Al and Mn in acid coal mine drainage by sequential selective precipitation with control of pH. CATENA 2017, 148, 11–16. [Google Scholar] [CrossRef]
- Brar, K.K.; Etteieb, S.; Magdouli, S.; Calugaru, L.; Brar, S.K. Novel approach for the management of acid mine drainage (AMD) for the recovery of heavy metals along with lipid production by Chlorella vulgaris. J. Environ. Manag. 2022, 308, 114507. [Google Scholar] [CrossRef]
- Santos, K.B.; Almeida, V.O.; Weiler, J.; Schneider, I.A.H. Removal of Pollutants from an AMD from a Coal Mine by Neutralization/Precipitation Followed by “In Vivo” Biosorption Step with the Microalgae Scenedesmus sp. Minerals 2020, 10, 711. [Google Scholar] [CrossRef]
- Abdullah, N.; Yusof, N.; Lau, W.J.; Jaafar, J.; Ismail, A.F. Recent trends of heavy metal removal from water/wastewater by membrane technologies. J. Ind. Eng. Chem. 2019, 76, 17–38. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Li, Z.; Wang, H.; He, X.; Wang, C. A novel method based on membrane distillation for treating acid mine drainage: Recovery of water and utilization of iron. Chemosphere 2021, 279, 130605. [Google Scholar] [CrossRef]
- Xie, H.Y.; Liu, Y.B.; Rao, B.; Wu, J.Z.; Gao, L.K.; Chen, L.Z.; Tian, X.S. Selective passivation behavior of galena surface by sulfuric acid and a novel flotation separation method for copper-lead sulfide ore without collector and inhibitor. Sep. Purif. Technol. 2021, 267, 118621. [Google Scholar] [CrossRef]
- Zhang, N.N.; Ejtemaei, M.; Nguyen, A.V.; Zhou, C.C. XPS analysis of the surface chemistry of sulfuric acid-treated kaolinite and diaspore minerals with flotation reagents. Miner. Eng. 2019, 136, 1–7. [Google Scholar] [CrossRef]
- Martí-Calatayud, M.C.; Buzzi, D.C.; García-Gabaldón, M.; Ortega, E.; Bernardes, A.M.; Tenório, J.A.S.; Pérez-Herranz, V. Sulfuric acid recovery from acid mine drainage by means of electrodialysis. Desalination 2014, 343, 120–127. [Google Scholar] [CrossRef] [Green Version]
- Cifuentes, L.; García, I.; Ortiz, R.; Casas, J.M. The use of electrohydrolysis for the recovery of sulphuric acid from copper-containing solutions. Sep. Purif. Technol. 2006, 50, 167–174. [Google Scholar] [CrossRef]
- Kesieme, U.K.; Milne, N.A.; Aral, H.; Cheng, C.Y.; Duke, M. Novel application of membrane distillation for acid and water recovery from mining waste waters. In Proceedings of the International Mine Water Association Symposium, Bunbury, Australia, 29 September–4 October 2012; McCullough, C.D., Lund, M.A., Wyse, L., Eds.; International Mine Water Association: Wendelstein, Germany, 2012. [Google Scholar]
- Menzel, K.; Barros, L.; García, A.; Ruby-Figueroa, R.; Estay, H. Metal sulfide precipitation coupled with membrane filtration process for recovering copper from acid mine drainage. Sep. Purif. Technol. 2021, 270, 118721. [Google Scholar] [CrossRef]
- Ryu, S.; Naidu, G.; Moon, H.; Vigneswaran, S. Selective copper recovery by membrane distillation and adsorption system from synthetic acid mine drainage. Chemosphere 2020, 260, 127528. [Google Scholar] [CrossRef]
- Sulonen, M.L.K.; Baeza, J.A.; Gabriel, D.; Guisasola, A. Optimisation of the operational parameters for a comprehensive bioelectrochemical treatment of acid mine drainage. J. Hazard. Mater. 2021, 409, 124944. [Google Scholar] [CrossRef]
- Hu, X.; Yang, H.; Tan, K.Y.; Hou, S.T.; Cai, J.Y.; Yuan, X.; Lan, Q.P.; Cao, J.R.; Yan, S.M. Treatment and recovery of iron from acid mine drainage: A pilot-scale study. J. Environ. Chem. Eng. 2022, 10, 106974. [Google Scholar] [CrossRef]
- Park, S.M.; Shin, S.Y.; Yang, J.S.; Ji, S.W.; Baek, K. Selective Recovery of Dissolved Metals from Mine Drainage Using Electrochemical Reactions. Electrochim. Acta 2015, 181, 248–254. [Google Scholar] [CrossRef]
- Macingova, E.; Luptakova, A. Recovery of metals from acid mine drainage. Chem. Eng. Trans. 2012, 28, 109–114. [Google Scholar] [CrossRef]
- Chen, T.; Yan, B.; Lei, C.; Xiao, X.M. Pollution control and metal resource recovery for acid mine drainage. Hydrometallurgy 2014, 147–148, 112–119. [Google Scholar] [CrossRef]
- Pino, L.; Beltran, E.; Schwarz, A.; Ruiz, M.C.; Borquez, R. Optimization of nanofiltration for treatment of acid mine drainage and copper recovery by solvent extraction. Hydrometallurgy 2020, 195, 105361. [Google Scholar] [CrossRef]
- Oh, C.; Han, Y.S.; Park, J.H.; Bok, S.; Cheong, Y.; Yim, G.; Ji, S. Field application of selective precipitation for recovering Cu and Zn in drainage discharged from an operating mine. Sci. Total Environ. 2016, 557–558, 212–220. [Google Scholar] [CrossRef]
- Le, V.G.; Vo, T.D.; Nguyen, B.S.; Vu, C.T.; Shih, Y.J.; Huang, Y.H. Recovery of iron(II) and aluminum(III) from acid mine drainage by sequential selective precipitation and fluidized bed homogeneous crystallization (FBHC). J. Taiwan. Inst. Chem. Eng. 2020, 115, 135–143. [Google Scholar] [CrossRef]
- Akinwekomi, V.; Maree, J.P.; Masindi, V.; Zvinowanda, C.; Osman, M.S.; Foteinis, S.; Mpenyana-Monyatsi, L.; Chatzisymeon, E. Beneficiation of acid mine drainage (AMD): A viable option for the synthesis of goethite, hematite, magnetite, and gypsum—Gearing towards a circular economy concept. Miner. Eng. 2020, 148, 106204. [Google Scholar] [CrossRef]
- Silva, R.d.A.; Castro, C.D.; Vigânico, E.M.; Petter, C.O.; Schneider, I.A.H. Selective precipitation/UV production of magnetite particles obtained from the iron recovered from acid mine drainage. Miner. Eng. 2012, 29, 22–27. [Google Scholar] [CrossRef]
- Silva, R.d.A.; Secco, M.P.; Lermen, R.T.; Schneider, I.A.H.; Hidalgo, G.E.N.; Sampaio, C.H. Optimizing the selective precipitation of iron to produce yellow pigment from acid mine drainage. Miner. Eng. 2019, 135, 111–117. [Google Scholar] [CrossRef]
- Akinwekomi, V.; Maree, J.P.; Zvinowanda, C.; Masindi, V. Synthesis of magnetite from iron-rich mine water using sodium carbonate. J. Environ. Chem. Eng. 2017, 5, 2699–2707. [Google Scholar] [CrossRef]
- Zhang, W.X. Nanoscale Iron Particles for Environmental Remediation: An Overview. J. Nanopart. Res. 2003, 5, 323–332. [Google Scholar] [CrossRef]
- Cheng, S.; Jang, J.H.; Dempsey, B.A.; Logan, B.E. Efficient recovery of nano-sized iron oxide particles from synthetic acid-mine drainage (AMD) water using fuel cell technologies. Water Res. 2011, 45, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Pignatello, J.J.; Oliveros, E.; MacKay, A. Advanced Oxidation Processes for Organic Contaminant Destruction Based on the Fenton Reaction and Related Chemistry. Crit. Rev. Environ. Sci. Technol. 2006, 36, 1–84. [Google Scholar] [CrossRef]
- Tufail, A.; Price, W.E.; Mohseni, M.; Pramanik, B.K.; Hai, F.I. A critical review of advanced oxidation processes for emerging trace organic contaminant degradation: Mechanisms, factors, degradation products, and effluent toxicity. J. Water Process. 2021, 40, 101778. [Google Scholar] [CrossRef]
- Chamarro, E.; Marco, A.; Esplugas, S. Use of Fenton reagent to improve organic chemical biodegradability. Water Res. 2001, 35, 1047–1051. [Google Scholar] [CrossRef]
- Lin, R.; Li, Y.; Yong, T.; Cao, W.; Wu, J.; Shen, Y. Synergistic effects of oxidation, coagulation and adsorption in the integrated fenton-based process for wastewater treatment: A review. J. Environ. Manag. 2022, 306, 114460. [Google Scholar] [CrossRef]
- Xiang, Y.; Liu, H.L.; Zhu, E.Y.; Yang, K.; Yuan, D.L.; Jiao, T.F.; Zhang, Q.R.; Tang, S.F. Application of inorganic materials as heterogeneous cocatalyst in Fenton/Fenton-like processes for wastewater treatment. Sep. Purif. Technol. 2022, 295, 121293. [Google Scholar] [CrossRef]
- Ji, Y.F.; Fan, Y.; Liu, K.; Kong, D.Y.; Lu, J.H. Thermo activated persulfate oxidation of antibiotic sulfamethoxazole and structurally related compounds. Water Res. 2015, 87, 1–9. [Google Scholar] [CrossRef]
- Moon, B.Y.; Park, Y.B.; Park, K.H. Fenton oxidation of Orange II by pre-reduction using nanoscale zero-valent iron. Desalination 2011, 268, 249–252. [Google Scholar] [CrossRef]
- Shi, X.G.; Ma, K.; Gu, Y.W.; Zhang, W.Q.; Sun, J. Accelerated degradation of sulfadiazine by wet mechanochemical synthesized nano-pyrite FeS2 based Fenton system: Performance, mechanism and applicability. Sep. Purif. Technol. 2022, 292, 121060. [Google Scholar] [CrossRef]
- Tavares, M.G.; Duarte, J.L.d.S.; Oliveira, L.M.T.M.; Fonseca, E.J.S.; Tonholo, J.; Ribeiro, A.S.; Zanta, C.L.P.S. Reusable iron magnetic catalyst for organic pollutant removal by Adsorption, Fenton and Photo Fenton process. J. Photochem. Photobiol. A Chem. 2022, 432, 114089. [Google Scholar] [CrossRef]
- Mahiroglu, A.; Tarlan-Yel, E.; Sevimli, M.F. Treatment of combined acid mine drainage (AMD)--flotation circuit effluents from copper mine via Fenton’s process. J. Hazard. Mater. 2009, 166, 782–787. [Google Scholar] [CrossRef]
- Sun, M.; Ru, X.R.; Zhai, L.F. In-situ fabrication of supported iron oxides from synthetic acid mine drainage: High catalytic activities and good stabilities towards electro-Fenton reaction. Appl. Catal. B 2015, 165, 103–110. [Google Scholar] [CrossRef]
- Sun, Y.M.; Zhai, L.F.; Duan, M.F.; Sun, M. In Situ Fabrication of Electro-Fenton Catalyst from Fe2+ in Acid Mine Drainage: Influence of Coexisting Metal Cations. ACS Sustain. Chem. Eng. 2018, 6, 14154–14161. [Google Scholar] [CrossRef]
- Huang, A.; Zhi, D.; Tang, H.; Jiang, L.; Luo, S.; Zhou, Y. Effect of Fe(2+), Mn(2+) catalysts on the performance of electro-Fenton degradation of antibiotic ciprofloxacin, and expanding the utilizing of acid mine drainage. Sci. Total Environ. 2020, 720, 137560. [Google Scholar] [CrossRef]
- Anawar, H.M. Sustainable rehabilitation of mining waste and acid mine drainage using geochemistry, mine type, mineralogy, texture, ore extraction and climate knowledge. J. Environ. Manag. 2015, 158, 111–121. [Google Scholar] [CrossRef]
- López, J.; Gibert, O.; Cortina, J.L. Integration of membrane technologies to enhance the sustainability in the treatment of metal-containing acidic liquid wastes. An overview. Sep. Purif. Rev. 2021, 265, 118485. [Google Scholar] [CrossRef]
- Zhou, H.B. Research and application of sulfur flotation with mine acidic sewage. Nonferrous Met. (Mineral. Processing Sect.) 1992, 12–15. (In Chinese) [Google Scholar]
- Bai, S.J.; Li, J.; Yuan, J.Q.; Bi, Y.X.; Ding, Z.; Dai, H.X.; Wen, S.M. An innovative option for the activation of chalcopyrite flotation once depressed in a high alkali solution with the addition of acid mine drainage. J. Cent. South. Univ. 2022, 29. in press. [Google Scholar]
- Gong, X.Y.; Ma, X.H.; Wu, X.L.; Peng, X. Research on the Application of Wastewater from Stope in Beneficiation of Pyrite. Hunan Nonferrous Met. 2016, 32, 7–8+19. (In Chinese) [Google Scholar]
- Duan, Z.H. Experimental study on application of mine acid wastewater in pyrite beneficiation system. Shandong Ind. Technol. 2017, 2, 54. (In Chinese) [Google Scholar] [CrossRef]
- Zhan, Y.H. Wastewater Utilization Practice in Dexing Copper Mine. Nonferrous Met. Eng. 2015, 5, 90–93. (In Chinese) [Google Scholar] [CrossRef]
| Country | Typical Mines | pH | Fe (mg/L) | Zn (mg/L) | Cu (mg/L) | Mn (mg/L) | Al (mg/L) | SO42− (mg/L) | References |
|---|---|---|---|---|---|---|---|---|---|
| China | Polymetallic Mine | 2.50 | 2490 | 500 | 2670 | 6590 | - | 24,530 | [51] |
| South Africa | Mpumalanga Coal Mine | 2 | 8000 | - | - | 75 | 300 | 30,000 | [52] |
| Spain | Riotinto Mine | 2.60–2.80 | 1824 | 557 | 184 | 329 | 2830 | 24,700 | [53] |
| Australia | Mount Morgan Gold Mine | 2.70 | 66 | 55 | 65 | 245 | 2317 | 29,547 | [54] |
| USA | Elizabeth Copper Mine | 3.30 | 123 | - | - | 2.60 | 13 | 1200 | [55] |
| Chile | Copper mine | 2.50 | 627.50 | - | 2298 | 224.50 | 1139 | 14,337 | [56] |
| Korea | Taejeong Coal Mine | 3.28 | 186 | - | - | 13 | 40 | 1950 | [57] |
| Canada | Gold Mine | 2.39 | 788 | 0.25 | 3.42 | 19.40 | 310 | 4520 | [58] |
| Brazil | Coal Mine | 2.33 | 611.38 | 62.65 | - | 37.98 | 269.37 | 7410.40 | [59] |
| Recovery Methods | Metal Ions Recovery | Description | References |
|---|---|---|---|
| Membrane Distillation and Adsorption System | Cu | The valuable copper resources were selectively recovered from the synthesized AMD solution by membrane distillation and adsorption systems. The pH of AMD was adjusted to the range of 5.0–5.2, and the KOH-treated AMD was continued to concentrate the solution by the direct contact membrane distillation (DCMD) process with the aim of achieving the selective adsorption of Cu for the multi-modified mesoporous silica SBA-15 material and the amount of Cu adsorption reached 24.53 mg/g. | [68] |
| Metal Sulfide Precipitation and Membrane Filtration Process | Cu | A novel process combining metal sulfide precipitation and membrane microfiltration was investigated for the recovery of copper from synthetic AMD. The recovery of copper was close to 100% and turbidity values in the treated solution were lower than 2 NTU for sulfide stoichiometric dosages of 120%. | [67] |
| Nanofiltration (NF) and Solvent Extraction (SX) | Cu | The feasibility of recovering copper from actual AMD was investigated by pilot-scale tests. Nanofiltration (NF) was applied to concentrate copper from AMD solutions, and then solvent extraction (SX) was performed to extract copper from the concentrated solution of NF. The results showed that the combination of NF and SX is promising for copper recovery and the copper recovery rate reached 97%. | [74] |
| Selective Precipitation (SP) | Cu and Zn | A field trial was conducted for the recovery of Cu and Zn from AMD produced at an operating mine using a selective precipitation (SP) pilot plant. Under the AMD condition of 1.4 L/min, Cu and Zn precipitates with a purity of 80% and a precipitation rate of 90% can be obtained. | [75] |
| Chemical Oxidation Technology (H2O2-NaOH Technology) | Fe | The chemical oxidation pilot process was used to treat AMD with high concentration of Fe. Under acidic conditions, H2O2 was able to rapidly oxidize Fe2+ to Fe3+. Followed by the addition of NaOH to adjust the pH to 3.8, Fe-contained precipitates were formed and the average Fe content in the recovered sludge was 26.85%. | [70] |
| Electrochemical Reactions | Fe, Al, Cu, Zn, and Ni | Electrochemical reactions are possible to oxidize Fe(II) to Fe(III) while producing neutralizing agents (containing a high concentration of hydroxide) for the selective recovery of dissolved metals (Fe, Al, Cu, Zn, and Ni) from AMD. | [71] |
| Sequential Selective Precipitation and Fluidized Bed Homogeneous Crystallization (FBHC) | Fe and Al | The Fe2+ and Al3+ were recovered from actual AMD using a combination of sequential selective precipitation and fluidized bed homogeneous crystallization (FBHC). Under the conditions of pH of about 9.25, [H2O2]/[Al(III)] molar ratio of 2.0 and upward flow rate (U) of 30.5 m/h. Ferric hydroxide (Fe(OH)3) and bayerite (α-Al(OH)3) pellets were prepared sequentially. | [76] |
| Sequential Selective Precipitation | Fe, Al, and Mn | The selective precipitation of dissolved iron, aluminum, and manganese in the AMD from the Samma-Taejeong coal mine by adding oxidants and neutralizers was investigated. In the case of oxidation (H2O2) and then neutralization of AMD, the sequence of metal removal was Fe>Al>Mn in order, and the recovery of dissolved Fe, Al, and Mn reached 99.2–99.3%, 70.4–82.2%, and 37.8–87.5%, respectively. | [57] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, J.; Ding, Z.; Bi, Y.; Li, J.; Wen, S.; Bai, S. Resource Utilization of Acid Mine Drainage (AMD): A Review. Water 2022, 14, 2385. https://doi.org/10.3390/w14152385
Yuan J, Ding Z, Bi Y, Li J, Wen S, Bai S. Resource Utilization of Acid Mine Drainage (AMD): A Review. Water. 2022; 14(15):2385. https://doi.org/10.3390/w14152385
Chicago/Turabian StyleYuan, Jiaqiao, Zhan Ding, Yunxiao Bi, Jie Li, Shuming Wen, and Shaojun Bai. 2022. "Resource Utilization of Acid Mine Drainage (AMD): A Review" Water 14, no. 15: 2385. https://doi.org/10.3390/w14152385







