Kanchan Arsenic Filters for Household Water Treatment: Unsuitable or Unsustainable?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy and Selection Criteria
2.2. Completeness of the Literature
3. The KanchanTM Arsenic Filter
4. Factors Affecting the Design and Operation of Fe0-Based Filters
4.1. General Aspects
4.2. Relevant Design Considerations
5. KAF Assessments
5.1. The Research Articles Co-Authored by Ngai
5.2. The Research Article Co-Authored by Chiew
5.3. The Research Article Co-Authored by Singh
5.4. The Research Article Co-Authored by Ogata
5.5. The Research Articles Co-Authored by Mueller
5.6. Critical Evaluation
6. KAF Design Mistakes and a Path to Improvement
- (i)
- The raw water quality: the prohibitive geochemical conditions have not yet been clearly defined as most of unsatisfactory results were even observed in conditions close to the “ideal” defined by Ngai et al. [30];
- (ii)
- The contact time: the current filtration rate (varying from 15 to 30 L h−1) is said to be too high and, hence, does not allow enough reaction time between contaminant scavengers (FeCPs) generated in situ and contaminants;
- (iii)
- The wet/dry cycle principle on which the filter operates: the intermittent immersion of the nails in the system causes a progressive decrease in their reactivity with time;
- (iv)
- The iron nails’ ineffectiveness in several cases to generate sufficient FeCPs for As and other contaminants’ collection: this was wrongly attributed solely to the limited contact time or nails’ reactivity loss resulting from their non-permanent immersion in the system;
- (v)
- O2 depletion in the Fe0 unit: this was proven to disturb the operation and functionality of the biosand unit;
- (vi)
- The long-term use of the filter without an established frequency for regular/periodic maintenance.
6.1. Specifications on the Main KAF Design Limitations
6.2. Design Amendments for Effective Improvement
6.3. Maintenance and Quality Control
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gadgil, A. Drinking water in developing countries. Annu. Rev. Energy Environ. 1998, 23, 253–286. [Google Scholar] [CrossRef] [Green Version]
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Marinas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Ali, I. Water Treatment by Adsorption Columns: Evaluation at Ground Level. Sep. Purif. Rev. 2014, 43, 175–205. [Google Scholar] [CrossRef]
- Hering, J.G.; Maag, S.; Schnoor, J.L. A call for synthesis of water research to achieve the sustainable development goals by 2030. Environ. Sci. Technol. 2016, 50, 6122–6123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogata, R.; Dangol, B.; Sakamoto, M. Sustainability assessment of long-term, widely used household Kanchan Arsenic Filters in Nepal. J. Environ. Sci. Health A 2020, 55, 517–527. [Google Scholar] [CrossRef]
- Mueller, B.; Dangol, B.; Ngai, T.K.K.; Hug, S.J. Kanchan arsenic filters in the lowlands of Nepal: Mode of operation, arsenic removal, and future improvements. Environ. Geochem. Health 2021, 43, 375–389. [Google Scholar] [CrossRef]
- Logsdon, G.S.; Kohne, R.; Abel, S.; LaBonde, S. Slow sand filtration for small water systems. J. Environ. Eng. Sci. 2002, 1, 339–348. [Google Scholar] [CrossRef]
- Haig, S.J.; Collins, G.; Davies, R.L.; Dorea, C.C.; Quince, C. Biological aspects of slow sand filtration: Past, present and future. Water Sci. Technol. Water Supply 2011, 11, 468–472. [Google Scholar] [CrossRef]
- Elliott, M.; Stauber, C.E.; DiGiano, F.A.; Aceituno, D.; Fabiszewski, A.; Sobsey, M.D. Investigation of E. coli and virus reductions using replicate, bench-scale biosand filter columns and two filter media. Int. J. Environ. Res. Public Health 2015, 12, 10276–10299. [Google Scholar] [CrossRef]
- Baumgartner, J.; Murcott, S.; Ezzati, M. Reconsidering ‘Appropriate Technology’: The Effects of Operating Conditions on the Bacterial Removal Performance of Two Household Drinking-water Filter Systems. Environ. Res. Lett. 2007, 2, 024003. [Google Scholar] [CrossRef]
- Johnson, S.M.; Peletz, R.L.; Murcott, S. Results from Household Ceramic Filter Evaluation in Northern Ghana. In Proceedings of the 33rd WEDC International Conference, Accra, Ghana, 7–11 April 2008; p. 463. [Google Scholar]
- Van Halem, D.; van der Lann, H.; Heijman, S.G.J.; van Dijk, J.C.; Amy, G.L. Assessing the Sustainability of the Silver-impregnated Ceramic Pot Filter for Low-cost Household Drinking Water Treatment. Phys. Chem. Earth 2009, 34, 36. [Google Scholar] [CrossRef]
- Smittakorn, S.; Jirawongboonrod, N.; Mongkolnchai-Arunya, S.; Durnford, D. Homemade bone charcoal adsorbent for defluoridation of groundwater in Thailand. J. Water Health 2010, 8, 826–836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Habuda-Stani, M.; Ravanèi, M.E.; Flanagan, A. A review on adsorption of fluoride from aqueous solution. Materials 2014, 7, 6317–6366. [Google Scholar] [CrossRef]
- Hassan, S.S.M.; Awwad, N.S.; Aboterika, A.H.A. Removal of mercury from wastewater using camel bone charcoal. J. Hazard. Mater. 2008, 154, 992–997. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, A.; Kumar, E.; Sillanpaa, M. Fluoride removal from water by adsorption: A review. Chem. Eng. J. 2011, 171, 811–840. [Google Scholar] [CrossRef]
- Harikumar, P.S.P.; Jaseela, C.; Megha, T. Defluoridation of water using biosorbents. Nat. Sci. 2012, 4, 245–251. [Google Scholar] [CrossRef] [Green Version]
- Biswas, K.; Saha, S.K.; Ghosh, U.C. Adsorption of fluoride from aqueous solution by a synthetic iron(III)-aluminium(III) mixed oxide. Ind. Eng. Chem. Res. 2007, 46, 5346–5356. [Google Scholar] [CrossRef]
- Tripathy, S.S.; Raichur, A.M. Abatement of fluoride from water using manganese dioxide coated activated alumina. J. Hazard. Mater. 2008, 153, 1043–1051. [Google Scholar] [CrossRef]
- Mulugeta, E.; Zewge, F.; Johnson, C.A.; Chandravanshi, B.S. Aluminium hydro(oxide)-based (AO) adsorbent for defluoridation of drinking water: Optimisation, performance comparison, and field testing. Water 2015, 41, 121–128. [Google Scholar] [CrossRef] [Green Version]
- Daschner, F.D. Holy Water, Tap Water, Mineral Water or Water Filters? J. Hosp. Infect. 1997, 35, 71. [Google Scholar] [CrossRef]
- Gulson, B.L.; Sheehan, A.; Giblin, A.M.; Chiaradia, M.; Conradt, B. The Efficiency of Removal of Lead and other Elements from Domestic Drinking Waters Using a Bench-top Water Filter System. Sci. Total Environ. 1997, 196, 205. [Google Scholar] [CrossRef]
- Levesque, S.; Rodriguez, M.J.; Serodes, J.; Beaulieu, C.; Proulx, F. Effects of Indoor Drinking Water Handling on Trihalomethanes and Haloacetic Acids. Water Res. 2006, 40, 2921. [Google Scholar] [CrossRef]
- Naseri, E.; Ndé-Tchoupé, A.I.; Mwakabona, H.T.; Nanseu-Njiki, C.P.; Noubactep, C.; Njau, K.N.; Wydra, K.D. Making Fe0-Based Filters a Universal Solution for Safe Drinking Water Provision. Sustainability 2017, 9, 1224. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Hu, R.; Ndé-Tchoupé, A.I.; Gwenzi, W.; Ruppert, H.; Noubactep, C. Designing the next generation of Fe0-based filters for decentralized safe drinking water treatment. Processes 2020, 8, 745. [Google Scholar] [CrossRef]
- Hussam, A.; Munir, A.K.M. A simple and effective arsenic filter based on composite iron matrix: Development and deployment studies for groundwater of Bangladesh. J. Environ. Sci. Health A 2007, 42, 1869–1878. [Google Scholar] [CrossRef]
- Bradley, I.; Straub, A.; Maraccini, P.; Markazi, S.; Nguyen, T.H. Iron oxide amended biosand filters for virus removal. Water Res. 2011, 45, 4501–4510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tepong-Tsindé, R.; Ndé-Tchoupé, A.I.; Noubactep, C.; Nassi, A.; Ruppert, H. Characterizing a Newly Designed Steel-Wool-Based Household Filter for safe Drinking Water Provision: Hydraulic Conductivity and Efficiency for Pathogen removal. Processes 2019, 7, 966. [Google Scholar] [CrossRef] [Green Version]
- Ngai, T.K.K.; Murcott, S.; Shrestha, R.R.; Dangol, B.; Maharjan, M. Development and dissemination of Kanchan™ Arsenic Filter in rural Nepal. Water Sci. Technol. Water Supply 2006, 6, 137–146. [Google Scholar] [CrossRef]
- Ngai, T.K.K.; Shrestha, R.R.; Dangol, B.; Maharjan, M.; Murcott, S.E. Design for sustainable development—Household drinking water filter for arsenic and pathogen treatment in Nepal. J. Environ. Sci. Health A 2007, 42, 1879–1888. [Google Scholar] [CrossRef] [PubMed]
- Mwakabona, H.T.; Ndé-Tchoupé, A.I.; Njau, K.N.; Noubactep, C.; Wydra, K.D. Metallic iron for safe drinking water provision: Considering a lost knowledge. Water Res. 2017, 117, 127–142. [Google Scholar] [CrossRef]
- Antia, D.D.J. Water treatment and desalination using the eco-materials n-Fe0 (ZVI), n-Fe3O4, n-FexOyHz[mH2O], and n-Fex[Cation] nOyHz[Anion]m[rH2O]. In Handbook of Nanomaterials and Nanocomposites for Energy and Environmental Applications; Kharissova, O.V., Torres-Martínez, L.M., Kharisov, B.I., Eds.; Springer Nature: Cham, Switzerland, 2020. [Google Scholar]
- Khan, A.H.; Rasul, S.B.; Munir, A.K.M.; Habibuddowla, M.; Alauddin, M.; Newaz, S.S.; Hussam, A. Appraisal of a simple arsenic removal method for ground water of Bangladesh. J. Environ. Sci. Health Part A 2000, 35, 1021–1041. [Google Scholar] [CrossRef]
- Banerji, T.; Kalawapudi, K.; Salana, S.; Vijay, R. Review of processes controlling arsenic retention and release in soils and sediments of Bengal basin and suitable iron based technologies for its removal. Groundw. Sustain. Dev. 2019, 8, 358–367. [Google Scholar] [CrossRef]
- Sharma, R.M. Research Study on Possible Contamination of Groundwater with Arsenic in Jhapa, Morang, and Sunsari Districts of Eastern Terai of Nepal; Report of WHO Project; DWSS Government of Nepal: Katmandu, Nepal, 1999. [Google Scholar]
- Siddiqui, S.I.; Naushad, M.; Chaudhry, S.A. Promising prospects of nanomaterials for arsenic water remediation: A comprehensive review. Process. Saf. Environ. Prot. 2019, 126, 60–97. [Google Scholar] [CrossRef]
- Choudhry, A.; Sharma, A.; Khan, T.A.; Saif Ali Chaudhry, S.A. Flax seeds based magnetic hybrid nanocomposite: An advance and sustainable material for water cleansing. J. Water Process. Eng. 2021, 42, 102150. [Google Scholar] [CrossRef]
- Sharma, A.; Mangla, D.; Shehnaz; Chaudhry, S.A. Recent advances in magnetic composites as adsorbents for wastewater remediation. J. Environ. Manag. 2022, 306, 114483. [Google Scholar] [CrossRef]
- Siddiqui, S.I.; Chaudhry, S.A. A review on graphene oxide and its composites preparation and their use for the removal of As3+ and As5+ from water under the effect of various parameters: Application of isotherm, kinetic and thermodynamics. Process. Saf. Environ. Prot. 2018, 119, 138–163. [Google Scholar] [CrossRef]
- Siddiqui, S.I.; Chaudhry, S.A. Iron oxide and its modified forms as an adsorbent for arsenic removal: A comprehensive recent advancement. Process. Saf. Environ. Prot. 2017, 111, 592–626. [Google Scholar] [CrossRef]
- Charlet, L.; Liger, E.; Gerasimo, P. Decontamination of TCE- and U-rich waters by granular iron: Role of sorbed Fe (II). J. Environ. Eng. 1998, 124, 25–30. [Google Scholar] [CrossRef]
- Ndé-Tchoupé, A.I.; Crane, R.A.; Mwakabona, H.T.; Noubactep, C.; Njau, K.N. Technologies for decentralized fluoride removal: Testing metallic iron based filters. Water 2015, 7, 6750–6774. [Google Scholar] [CrossRef] [Green Version]
- Banerji, T.; Chaudhari, S. A cost-effective technology for arsenic removal: Case study of zerovalent iron-based IIT Bombay arsenic filter in West Bengal. In Water and Sanitation in the New Millennium; Nath, K., Sharma, V., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 127–137. [Google Scholar] [CrossRef]
- Bretzler, A.; Nikiema, J.; Lalanne, F.; Hoffmann, L.; Biswakarma, J.; Siebenaller, L.; Demange, D.; Schirmer, M.; Hug, S.J. Arsenic removal with zero-valent iron filters in Burkina Faso: Field and laboratory insights. Sci. Total Environ. 2020, 737, 139466. [Google Scholar] [CrossRef]
- Chiew, H.; Sampson, M.L.; Huch, S.; Ken, S.; Bostick, B.C. Effect of groundwater iron and phosphate on the efficacy of arsenic removal by iron-amended biosand filters. Environ. Sci. Technol. 2009, 43, 6295–6300. [Google Scholar] [CrossRef]
- Singh, A.; Smith, L.S.; Shrestha, S.; Maden, N. Efficacy of arsenic filtration by Kanchan Arsenic Filter in Nepal. J. Water Health 2014, 12, 596–599. [Google Scholar] [CrossRef]
- Huang, Z.; Cao, V.; Nya, E.L.; Gwenzi, W.; Noubactep, C. Kanchan Arsenic Filters and the Future of Fe0-Based Filtration Systems for Single Household Drinking Water Supply. Processes 2021, 9, 58. [Google Scholar] [CrossRef]
- Noubactep, C.; Temgoua, E.; Rahman, M.A. Designing Iron-Amended Biosand Filters for Decentralized Safe Drinking Water Provision. Clean Soil Air Water 2012, 40, 798–807. [Google Scholar] [CrossRef] [Green Version]
- Wenk, C.B.; Kaegi, R.; Hug, S.J. Factors affecting arsenic and uranium removal with zero-valent iron: Laboratory tests with Kanchan-type iron nail filter columns with different groundwaters. Environ. Chem. 2014, 11, 547–557. [Google Scholar] [CrossRef] [Green Version]
- Timalsina, H.; Mainali, B.; Angove, M.J.; Komai, T.; Paudel, S.R. Potential modification of groundwater arsenic removal filter commonly used in Nepal: A review. Groundw. Sustain. Dev. 2021, 12, 100549. [Google Scholar] [CrossRef]
- Rahman, M.A.; Karmakar, S.; Salama, H.; Gactha-Bandjun, N.; Btatkeu-K, B.D.; Noubactep, C. Optimising the design of Fe0-based filtration systems for water treatment: The suitability of porous iron composites. J. Appl. Solut. Chem. Modeling 2013, 2, 165–177. [Google Scholar]
- Tepong-Tsindé, R. Designing and Piloting a household filter for the peri-urban population of Douala (Cameroon). Freib. Online Geosci. 2021, 61, 1–90. [Google Scholar]
- Caré, S.; Crane, R.; Calabro, P.S.; Ghauch, A.; Temgoua, E.; Noubactep, C. Modelling the permeability loss of metallic iron water filtration systems. Clean Soil Air Water 2013, 41, 275–282. [Google Scholar] [CrossRef] [Green Version]
- Domga, R.; Togue-Kamga, F.; Noubactep, C.; Tchatchueng, J.B. Discussing porosity loss of Fe0 packed water filters at ground level. Chem. Eng. J. 2015, 263, 127–134. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weststrate, J.; Dijkstra, G.; Eshuis, J.; Gianoli, A.; Rusca, M. The sustainable development goal on water and sanitation: Learning from the millennium development goals. Soc. Indic. Res. 2019, 143, 795–810. [Google Scholar] [CrossRef] [Green Version]
- Westerhoff, P.; James, J. Nitrate removal in zero-valent iron packed columns. Water Res. 2003, 37, 1818–1830. [Google Scholar] [CrossRef]
- Noubactep, C.; Schöner, A.; Waffo, P. Metallic Iron Filters for Universal Access to Save Drinking Water. Clean Soil Air Water 2009, 37, 930–937. [Google Scholar] [CrossRef] [Green Version]
- Hussam, A. Contending with a development disaster: SONO filters remove arsenic from well water in Bangladesh. Innovations 2009, 4, 89–102. [Google Scholar] [CrossRef]
- Bilardi, S.; Calabrò, P.S.; Caré, S.; Noubactep, C.; Moraci, N. Improving the sustainability of granular iron/pumice systems for water treatment. J. Environ. Manag. 2013, 121, 133–141. [Google Scholar] [CrossRef] [Green Version]
- Noubactep, C. Designing metallic iron packed-beds for water treatment: A critical review. Clean Soil Air Water 2016, 44, 411–421. [Google Scholar] [CrossRef]
- Leupin, O.X.; Hug, S.J.; Badruzzaman, A.B.M. Arsenic removal from Bangladesh tube well water with filter columns containing zerovalent iron filings and sand. Environ. Sci. Technol. 2005, 39, 8032–8037. [Google Scholar] [CrossRef]
- Noubactep, C.; Schöner, A. Fe0-based alloys for environmental remediation: Thinking outside the box. J. Hazard. Mater. 2009, 165, 1210–1214. [Google Scholar] [CrossRef]
- Su, C.; Puls, R.W. Arsenate and arsenite removal by zerovalent iron: Effects of phosphate, silicate, carbonate, borate, sulfate, chromate, molybdate and nitrate, relative to chloride. Environ. Sci. Technol. 2001, 35, 4562–4568. [Google Scholar] [CrossRef]
- Su, C.; Puls, R.W. Significance of iron (II, III) hydroxycarbonate green rust in arsenic remediation using zerovalent iron in laboratory column tests. Environ. Sci. Technol. 2004, 38, 5224–5231. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Puls, R.W. Arsenate and arsenite sorption on magnetite: Relations to groundwater arsenic treatment using zerovalent iron and natural attenuation. Water Air Soil Pollut. 2008, 193, 65–78. [Google Scholar] [CrossRef]
- Hu, R.; Ndé-Tchoupé, A.I.; Lufingo, M.; Nassi, A.; Noubactep, C.; Njau, K.N. The impact selected pre-treatment procedures on iron dissolution from metallic iron specimens used in water treatment. Sustainability 2019, 11, 671. [Google Scholar] [CrossRef] [Green Version]
- Hildebrant, B.; Ndé-Tchoupé, A.I.; Lufingo, M.; Licha, T.; Noubactep, C. Steel wool for water treatment: Intrinsic reactivity and defluoridation efficiency. Processes 2020, 8, 265. [Google Scholar] [CrossRef] [Green Version]
- Bischof, G. On putrescent organic matter in potable water I. Proc. R. Soc. Lond. 1877, 26, 179–184. [Google Scholar]
- Bischof, G. On putrescent organic matter in potable water II. Proc. R. Soc. Lond. 1878, 27, 258–261. [Google Scholar]
- Tucker, W.G. The purification of water by chemical treatment. Science 1892, 20, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Baker, M. Sketch of the history of water treatment. J. Am. Water Works Assoc. 1934, 26, 902–938. [Google Scholar] [CrossRef]
- Tepong-Tsindé, R.; Crane, R.; Noubactep, C.; Nassi, A.; Ruppert, H. Fundamentals for designing sustainable metallic iron based filters for water treatment. Water 2015, 7, 868–897. [Google Scholar] [CrossRef] [Green Version]
- Mueller, B. First step concerning improvement of arsenic removal by adapted Kanchan filters in the lowlands of Nepal. J. Chem. Appl. 2020, 2, 1–9. [Google Scholar] [CrossRef]
- Mueller, B. Results of the first improvement step regarding removal efficient of Kanchan Arsenic Filters in the lowlands of Nepal—A case study. Water 2021, 13, 1765. [Google Scholar] [CrossRef]
- Reardon, J.E. Anaerobic corrosion of granular iron: Measurement and interpretation of hydrogen evolution rates. Environ. Sci. Technol. 1995, 29, 2936–2945. [Google Scholar] [CrossRef] [PubMed]
- Noubactep, C.; Meinrath, G.; Dietrich, P.; Sauter, M.; Merkel, B. Testing the suitability of zerovalent iron materials for reactive Walls. Environ. Chem. 2005, 2, 71–76. [Google Scholar] [CrossRef]
- Lufingo, M.; Ndé-Tchoupé, A.I.; Hu, R.; Njau, K.N.; Noubactep, C. A novel and facile method to characterize the suitability of metallic iron for water treatment. Water 2019, 11, 2465. [Google Scholar] [CrossRef] [Green Version]
- Ndé-Tchoupé, A.I.; Hu, R.; Gwenzi, W.; Nassi, A.; Noubactep, C. Characterizing the Reactivity of Metallic Iron for Water Treatment: H2 Evolution in H2SO4 and Uranium Removal Efficiency. Water 2020, 12, 1523. [Google Scholar] [CrossRef]
- Cao, V.; Ndé-Tchoupé, A.I.; Hu, R.; Gwenzi, W.; Noubactep, C. The mechanisms of contaminants removal in Fe(0)/H2O systems: The burden of a poor literature review. Chemosphere 2021, 280, 130614. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-K.; Muralidharan, S.; Ha, T.-H.; Bae, J.-H.; Ha, Y.-C.; Lee, H.-G.; Scantlebury, J.D. Electrochemical studies on the alternating current corrosion of mild steelunder cathodic protection condition in marine environments. Electrochim. Acta 2006, 51, 5259–5267. [Google Scholar] [CrossRef]
- Noubactep, C. Characterizing the reactivity of metallic iron upon methylene blue discoloration in Fe0/MnO2/H2O systems. J. Hazard. Mater. 2009, 168, 1613–1616. [Google Scholar] [CrossRef] [Green Version]
- Btatkeu-K, B.D.; Miyajima, K.; Noubactep, C.; Caré, S. Testing the suitability of metallic iron for environmental remediation: Discoloration of methylene blue in column studies. Chem. Eng. J. 2013, 215–216, 959–968. [Google Scholar] [CrossRef] [Green Version]
- Konaku-Amoah, K.; Ndé-Tchoupé, A.I.; Hu, R.; Gwenzi, W.; Noubactep, C. Investigating the Fe0/H2O systems using the methylene blue method: Validity, applications and future directions. Chemosphere 2022, 291, 132913. [Google Scholar] [CrossRef]
- Ndé-Tchoupé, A.I. Design and Construction of Fe0-Based Filters for Households. Ph.D. Dissertation, University of Douala, Douala, Cameroon, 2019. (In French). [Google Scholar]
- Lee, K.P.; Arnot, T.C.; Mattia, D. A review of reverse osmosis membrane materials for desalination-development to date and future potential. J. Membr. Sci. 2011, 370, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Francesco, C. Modeling the effects of material chemistry on water flow enhancement in nanotube membranes. MRS Bull. 2017, 42, 289. [Google Scholar]
- Lilje, J.; Mosler, H.-J. Continuation of health behaviors: Psychosocial factors sustaining drinking water chlorination in a longitudinal study from Chad. Sustainability 2016, 8, 1149. [Google Scholar] [CrossRef] [Green Version]
- Kozicki, Z.A.; Baiyasi-Kozicki, S.J. The survival of mankind requires a Water Quality and Quantity Index (WQQI) and Water Applied Testing and Environmental Research (WATER) Centers. World Water Policy 2019, 5, 55–70. [Google Scholar] [CrossRef]
- Amrose, S.E.; Cherukumilli, K.; Wright, N.C. Chemical contamination of drinking water in resource-constrained settings: Global prevalence and piloted mitigation strategies. Annu. Rev. Environ. Resour. 2020, 45, 195–226. [Google Scholar] [CrossRef]
- Hubbart, J.A.; Gootman, K.S. A call to broaden investment in drinking water testing and community outreach programs. Challenges 2021, 12, 32. [Google Scholar] [CrossRef]
- Nya, E.L.; Feumba, R.; Fotsing-Kwetché, P.R.; Gwenzi, W.; Noubactep, C. A hybrid model for achieving universal safe drinking water in the medium-sized city of Bangangté (Cameroon). Water 2021, 13, 3177. [Google Scholar] [CrossRef]
- Noubactep, C. Metallic iron for environmental remediation: A review of reviews. Water Res. 2015, 85, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Casentini, B.; Falcione, F.T.; Amalfitano, S.; Fazi, S.; Rossetti, S. Arsenic removal by discontinuous ZVI two steps system for drinking water production at household scale. Water Res. 2016, 106, 135–145. [Google Scholar] [CrossRef]
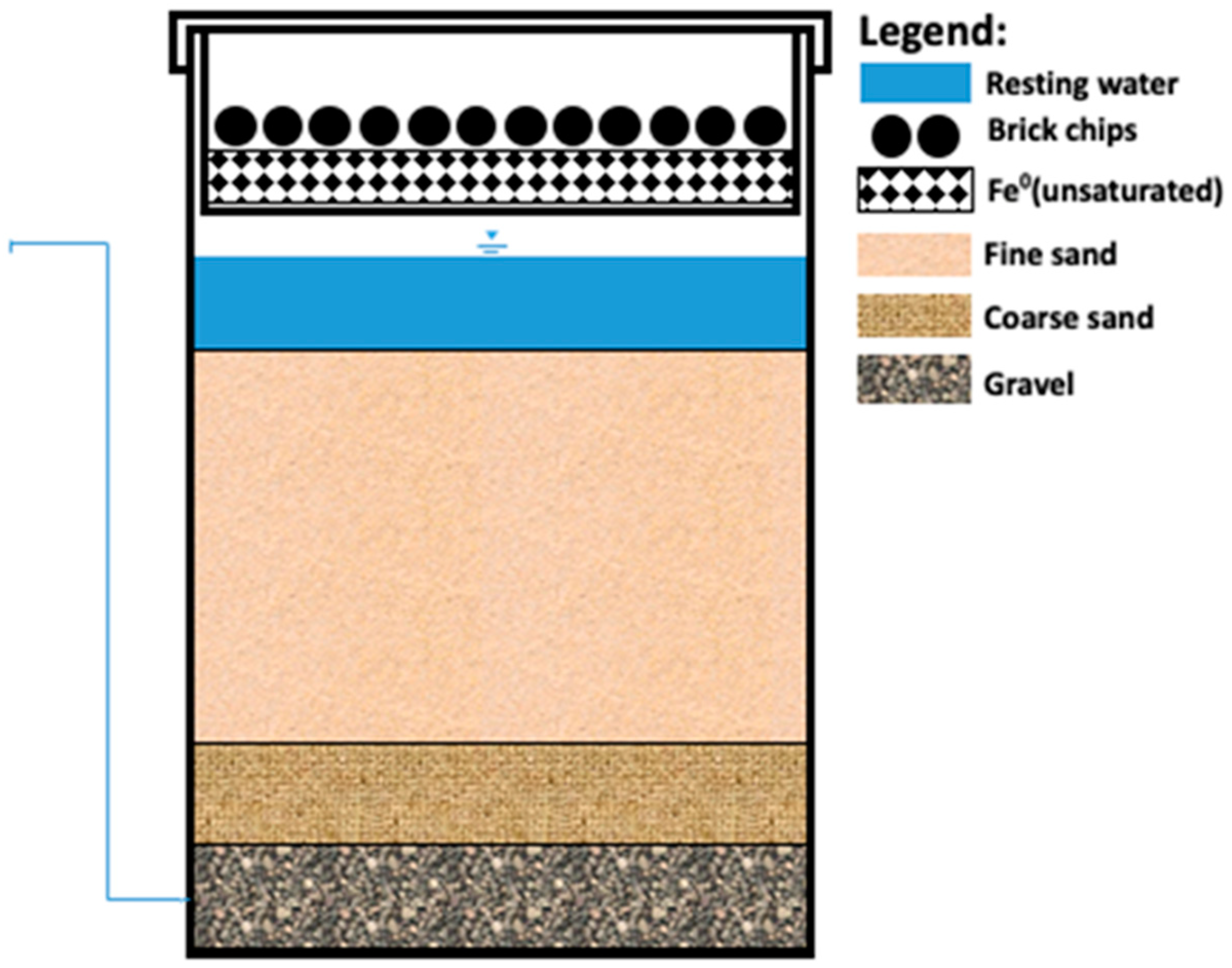
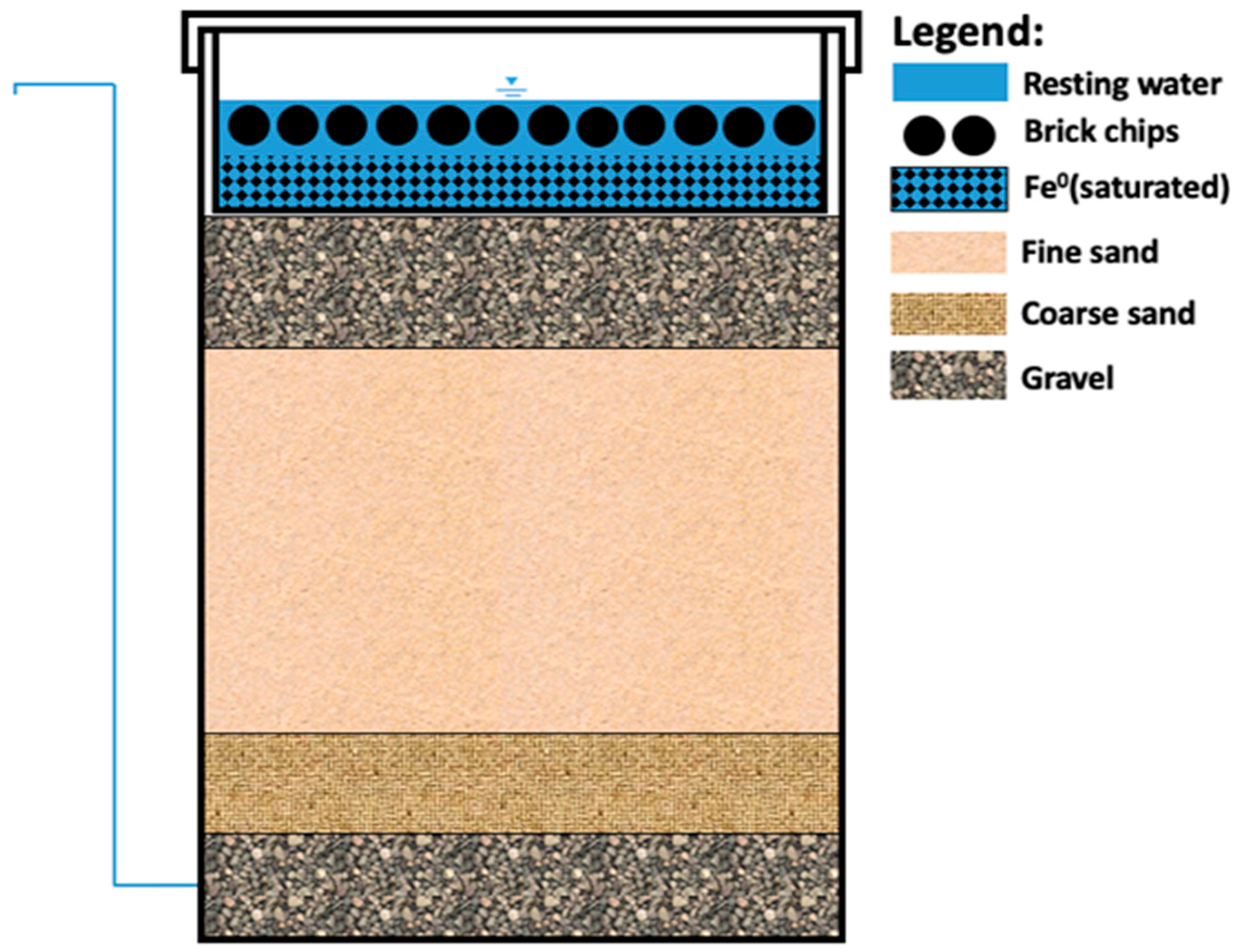
| Article | Number of Assessed KAFs | Duration | Performance Remarks |
|---|---|---|---|
| Ngai et al. [29] | 1034 | 3 months to 1 year | Good |
| Ngai et al. [30] | 1000 | 2 years | Good |
| Chiew et al. [45] | 3 | Almost 6 months | Poor |
| Singh et al. [46] | 62 | 6 months | Poor |
| Ogata et al. [5] | 2833 | 1 year | Poor |
| Mueller et al. [6] | 38 | 2 years | Poor |
| Article | Type | Covered Period | NT | Research Papers by KAF Developers | |
|---|---|---|---|---|---|
| Ngai et al. [29] | Ngai et al. [30] | ||||
| Chiew et al. [45] | Research | 1981–2008 | 33 | No | yes |
| Singh et al. [46] | Research | 2001–2012 | 11 | No | yes |
| Ogata et al. [5] | Research | 1995–2019 | 19 | No | yes |
| Mueller et al. [6] | Research | 1995–2020 | 51 | yes | yes |
| Tamalsina et al. [50] | Review | 1927–2020 | 83 | yes | yes |
| Huang et al. [47] | Review | 1890–2020 | 67 | yes | yes |
| N°. | Article | Some Key Observations | Comments |
|---|---|---|---|
| 1 | Ngai et al. [29] | The KAF performs satisfactorily, notably for the Terai region of Nepal type of groundwater. Cases of poor performance can only be due to improper filter construction and/or lack of maintenance. | This paper explains the development and describes the implementation process of the KAF in rural Nepal during the early days of its introduction. Question 1: Yes, Question 2: No |
| 2 | Ngai et al. [30] | The KAF is particularly suitable for natural waters that have the following characteristics: total arsenic ≤ 0.5 mg L−1, phosphate ≤ 2 mg L−1 and pH ≤ 8.0. | Research article on technical and social evaluation of the KAF at pilot scale in Nepal. Question 1: Yes, Question 2: No |
| 3 | Chiew et al. [45] | The KAF was found to be inefficient, particularly for As removal in Cambodia. The filter’s performance could be worse for MS2 coliphages (virus) removal than E. coli (bacteria). | KAF assessment at pilot scale in Cambodia. Question 1: No, Question 1: Yes |
| 4 | Singh et al. [46] | The KAF is inefficient and needs to be improved. | Pilot field assessment of KAF in the Nawalparasi District in Nepal. Question 1: No, Question 1: Yes |
| 5 | Ogata et al. [5] | They are currently more abandoned KAFs than those still in use in Nepal. Abandonments are mainly due to leaks, breakage and poor performances. | Mixture of literature review and field surveys on KAF in Nepal. Question 1: No, Question 1: Yes |
| 6 | Mueller et al. [6] | The KAF mostly performs well when influent water is initially highly Fe-rich. The filter needs design modifications (including increasing influent residence time and maintaining iron nails constantly immersed in the system) for performance improvement. | Mixture of literature review on KAF and an assessment study conducted in Nepal. Question 1: No, Question 1: Yes |
| 7 | Tamalsina et al. [50] | The KAF is particularly inefficient at treating As. Its performance could be improved by inserting an additional layer of human hair (which constituents have a high binding affinity for As(III) species) in the matrix of the filter’s biosand filter unit. | Reviews KAF and makes suggestions to sustainably improve its performance. Question 1: No, Question 1: Yes |
| Article | Influent Water Parameters | ||||||
|---|---|---|---|---|---|---|---|
| pH Value | [As] (μg L−1) | [Fe] (mg L−1) | [P] (mg L−1) | Hardness (mg/L) | Number of Tested KAFs | Location | |
| 1 | ˂8 | ˂500 | n.s. | ˂2 | n.s. | 1034 | Terai/Nepal |
| 2 | 7.18–7.26 | <10–1000 | 0.1–10 | 0.23 | n.s. | 1000 | Terai/Nepal |
| 3 | 7.5 ± 0.2 | 150–400 | ˂5 | >0.5 | n.s. | 3 | Cambodia |
| 4 | n.s. | 133.13 | n.s. | n.s. | n.s. | 62 | Newalparasi/Nepal |
| 5 | 6.0–7.5 | 0–1320 | 0–10.0 | 0–1 | 36–3687 | 2833 | Newalparasi/Nepal |
| 6 | 7.04–7.81 | 95.5–798.7 | 0.51–5.91 | 0.12 | n.s. | 38 | Terai/Nepal |
| KAF design specifications | |||||||
| KAF type | Filter’s height (cm) | Fe0 unit’s height (cm) | BSF’s height (cm) | Fe0 Type | Fe0 (kg) | Flow rate (L h−1) | |
| 1 | 1 (plastic round) | 49.6 * | 14.5 * | 28.1 * | Nails | 5 * | n.s. |
| 2 | 1 (plastic round) | 49.6 * | 14.5 * | 28.1 * | Nails | 5 * | 15–25 |
| 3 | 1 (concrete square) | 91 | 25 | 54 | Nails | 5 | n.s. (but contact time ~3 min) |
| 4 | n.s. | n.s. | n.s. | n.s. | Nails | n.s. | n.s. |
| 5 | 5 (all the types) | 49.6–91.0 | 14.5–25.0 | 28.1–56.0 | Nails | 5 | n.s. |
| 6 | n.s. | n.s. | n.s. | n.s. | Nails | 4.05–5.2 | n.s. |
| Effluent water parameters & investigation duration | |||||||
| pH | [Fe] (mg L−1) | [P] (mg L−1) | [As] (μg L−1) | Removed As (%) | Duration (year) | ||
| 1 | n.s. | n.s. | n.s. | n.s. | >95 | 0.25 to 1 | |
| 2 | 7.55–7.63 | <0.1–3 | 0.036 | ≤50 | 85–90 | 2 | |
| 3 | 7.9 ± 0.1 | <0.05 | n.s. | 74–226 | 39.4–74.9 | ~ 0.5 | |
| 4 | n.s. | n.s. | n.s. | 56.45 | ˂50 | 0.5 | |
| 5 | 6.0–7.5 | 0–5.0 | 0–2 | 0–590 | 0–100 | 1 | |
| 6 | n.s. | n.s. | n.s. | n.s. | 6.28–98.5 | 2 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ndé-Tchoupé, A.I.; Konadu-Amoah, B.; Gatcha-Bandjun, N.; Hu, R.; Gwenzi, W.; Noubactep, C. Kanchan Arsenic Filters for Household Water Treatment: Unsuitable or Unsustainable? Water 2022, 14, 2318. https://doi.org/10.3390/w14152318
Ndé-Tchoupé AI, Konadu-Amoah B, Gatcha-Bandjun N, Hu R, Gwenzi W, Noubactep C. Kanchan Arsenic Filters for Household Water Treatment: Unsuitable or Unsustainable? Water. 2022; 14(15):2318. https://doi.org/10.3390/w14152318
Chicago/Turabian StyleNdé-Tchoupé, Arnaud Igor, Bernard Konadu-Amoah, Nadège Gatcha-Bandjun, Rui Hu, Willis Gwenzi, and Chicgoua Noubactep. 2022. "Kanchan Arsenic Filters for Household Water Treatment: Unsuitable or Unsustainable?" Water 14, no. 15: 2318. https://doi.org/10.3390/w14152318
APA StyleNdé-Tchoupé, A. I., Konadu-Amoah, B., Gatcha-Bandjun, N., Hu, R., Gwenzi, W., & Noubactep, C. (2022). Kanchan Arsenic Filters for Household Water Treatment: Unsuitable or Unsustainable? Water, 14(15), 2318. https://doi.org/10.3390/w14152318









