Review of Advanced Oxidation Processes Based on Peracetic Acid for Organic Pollutants
Abstract
:1. Introduction
2. Preparation and Characteristics of PAA
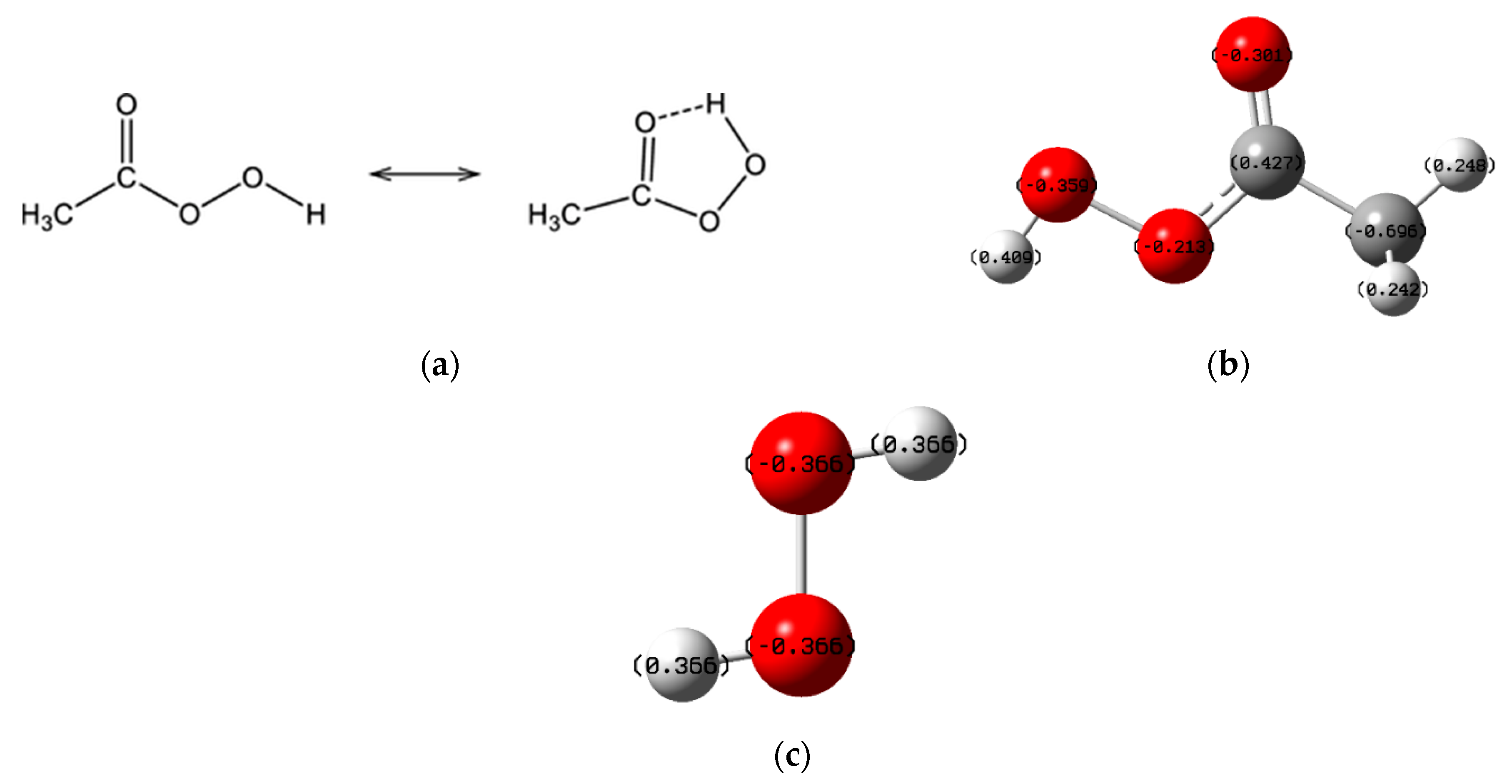
2.1. Characteristics of PAA
2.2. Synthesis and Detection of PAA
3. Methods and Mechanisms of Activating PAA
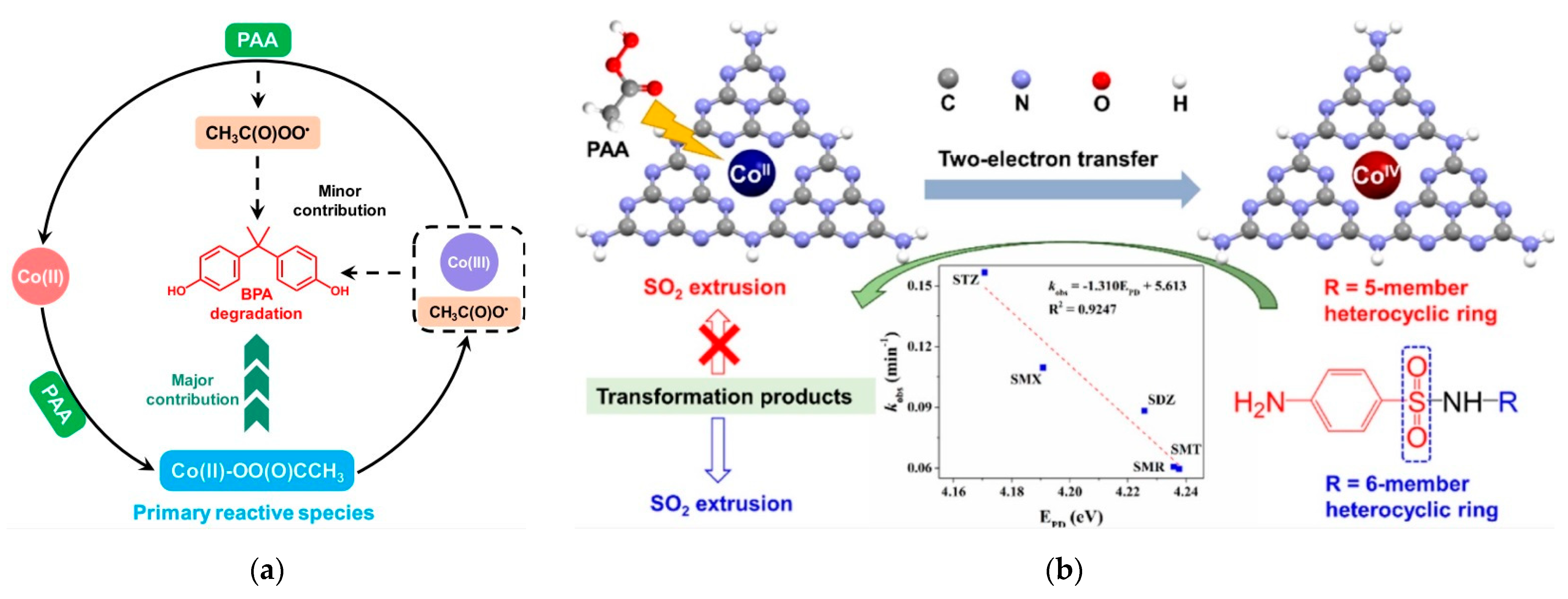
3.1. Free Radical Pathway
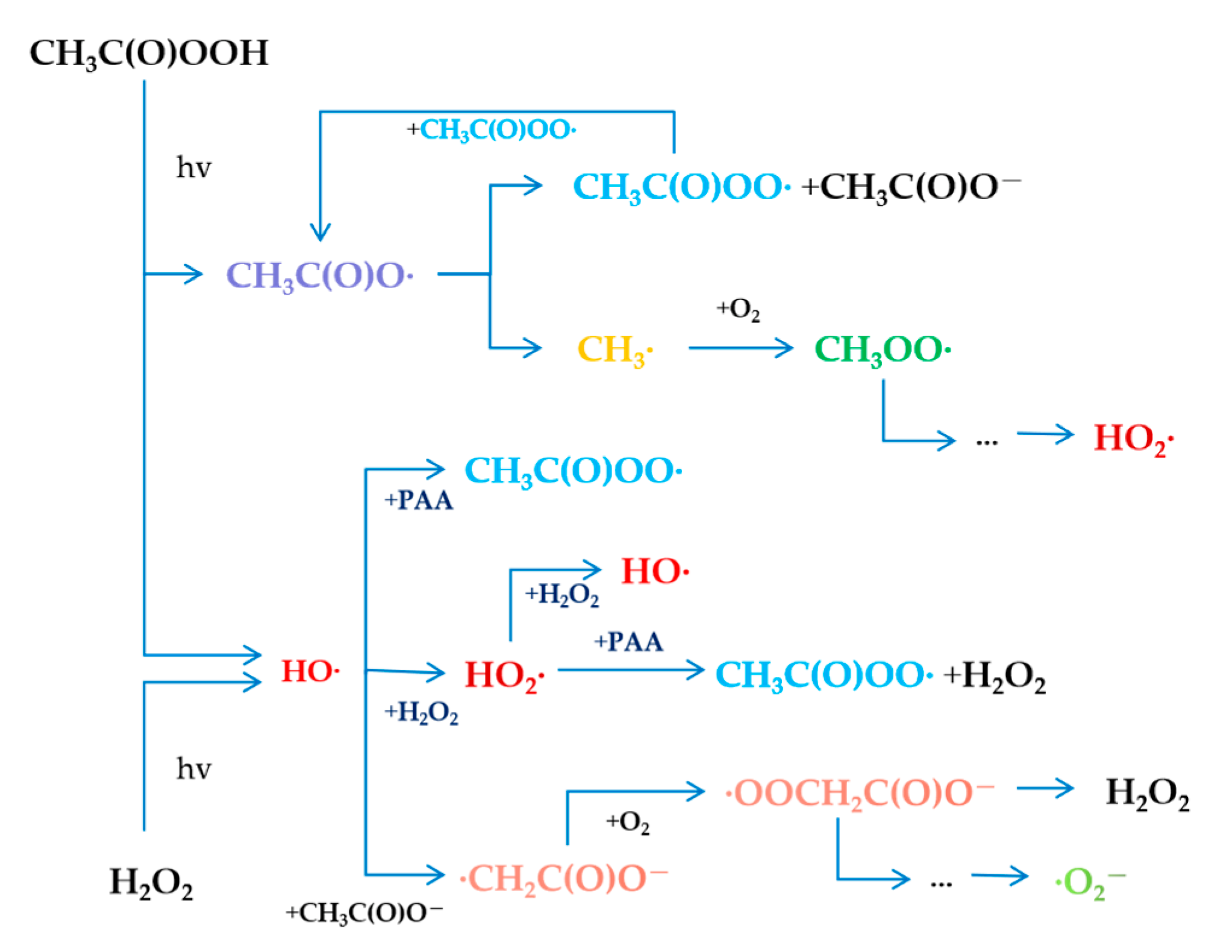
3.1.1. Energy Radiation
3.1.2. Transition Metal Catalysis
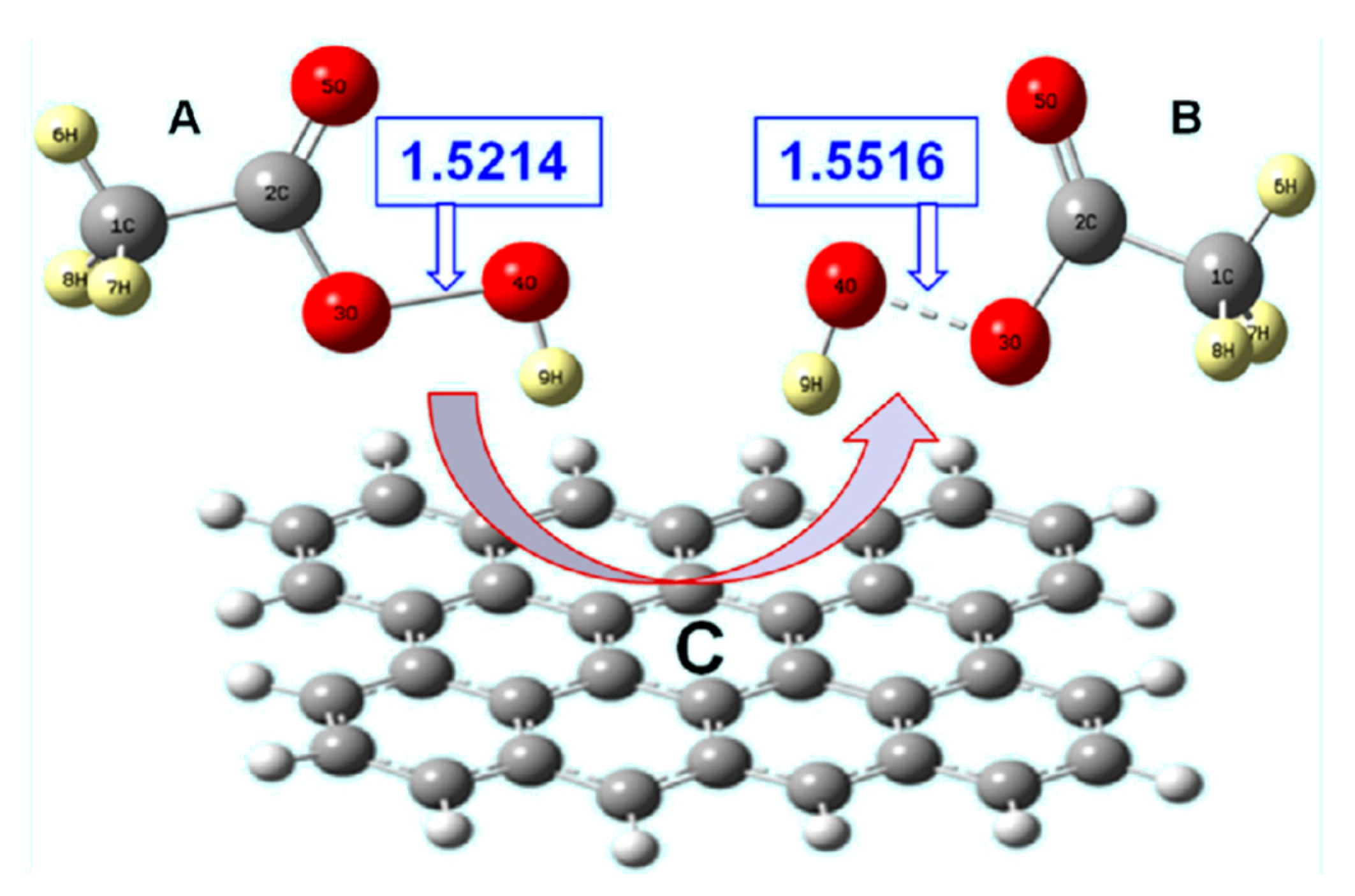
3.1.3. Activated Carbon Catalysis
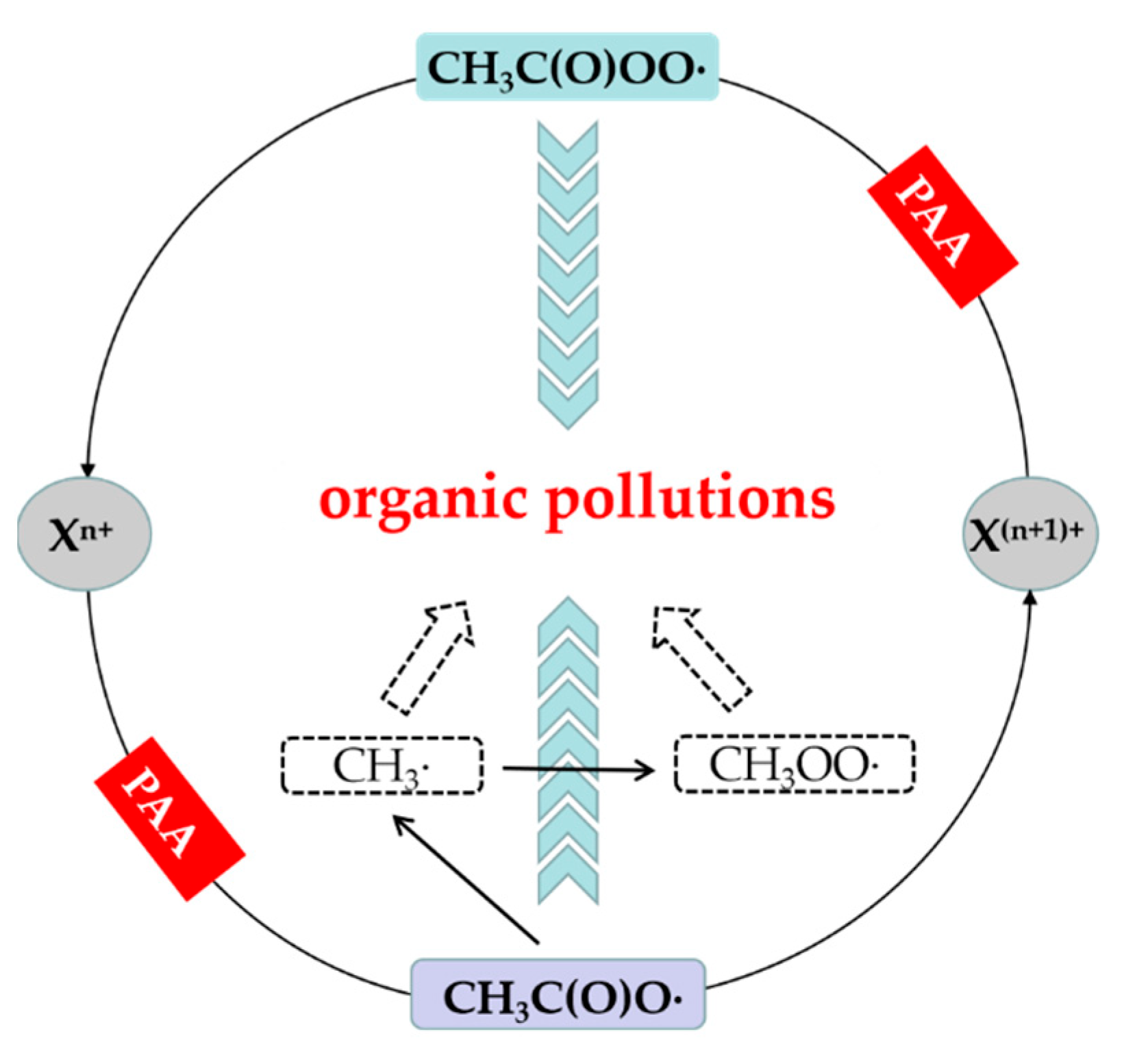
3.2. Non-Free Radical Pathway
4. Reactivity of PAA-Based AOPs
4.1. Oxidation of PAA Alone
4.2. PAA-Based AOPs
4.2.1. Homogeneous Catalysis
| Catalysts | CCatalyst (μM) | Contaminants | C0 (μM) | CPAA (μM) | pH | Time (min) | Max. Removal Efficiency (%) | References |
|---|---|---|---|---|---|---|---|---|
| Fe(VI) | 200 | SMX | 10 | 100 | 9.0 ± 0.1 | 1 | ~100 | [69] |
| Sulfadimethoxine | ~100 | |||||||
| Trimethoprim | >80 | |||||||
| Atenolol | >85 | |||||||
| Propranolol | >90 | |||||||
| Caffeine | 85 | |||||||
| Fe(II) | 100 | Methylene blue | 15 | 100 | 3.0~8.2 | 120 | ~90 | [54] |
| Naproxen | ~100 | |||||||
| Bisphenol-A | >80 | |||||||
| Fe(II) | 0.4 mM | Bisphenol-A | 60 mg·L−1 | 40 mg·L−1 | 3.5 | 10 | 100 | [72] |
| Fe(II) | 0.4 g·L−1 | polyacrylamide | 200 mg·L−1 | 10 mg·L−1 | 3 | 15 | >80 | [83] |
| Fe(II) | 5 mg·L−1 | Diclofenac | 1 | 100 | 7 | 1 | 80 | [84] |
| Cu(II) | 0.4 mM | Bisphenol-A | 60 mg·L−1 | 20 mg·L−1 | 3.5 | 60 | >95 | [72] |
| Cu(II)-HCO3−(CO32−) | 15 | Diclofenac | 1 | 60 | 9.3 | 20 | 85 | [80] |
| Co(II) | 15 | Bisphenol-A | 15 | 100 | 3.0~8.1 | 30 | 100 | [77] |
| Naproxen | 100 | |||||||
| SMX | 98.5 | |||||||
| Carbamazepine | 87.7 | |||||||
| Co(II) | 0.8 μM | SMX | 10 | 100 | 7 | 15 | ~90 | [56] |
| Co(II) | 0.01 mM | Acid orange 7 | 0.05 mM | 0.2 mM | 7 | 60 | 92 | [85] |
| Methylene blue | 52 | |||||||
| Congo red | 98 | |||||||
| Crystal Violet | 67 | |||||||
| Co(II) | 2.0 | Bisphenol-A | 8.0 | 100 | 7 | 20 | 93.6 | [68] |
| SMX | 99.3 | |||||||
| Phenol | 66.2 | |||||||
| Nitrobenzene | 18.8 | |||||||
| Mn(II) | 0.1~1 mg·L−1 | Orange II | 17.5 mg·L−1 | 38~3802 mg·L−1 | 9.5 | n.r. | n.r. | [86] |
| phosphate | 0.1 M | Diclofenac | 5 μM | 0.55 mM | 7.4 | 45 | ~100 | [42] |
| Cl− | 400 mM | Rhodamine B | 10 mg·L−1 | 2.0 mM | n.r. | 10 | ~100 | [82] |
4.2.2. Heterogeneous Catalysis
- Energy Catalysis
| Energy | Contaminants | C0 | CPAA | pH | Time (min) | Temperature (°C) | Max. Removal Efficiency (%) | Free Radicals | References |
|---|---|---|---|---|---|---|---|---|---|
| electrochemical oxidation | methylene blue | 10 mg·L−1 | 3.6 mmol·L−1 | 3 | 120 | 25 ± 2 | 93.99 | HO•>R−O• | [87] |
| Heat | SMX | 5 μM | 0.025–0.2 mM | 4–9 | 25 | 60 | ~100 | R−O• | [89] |
| microwave | thiophene sulfur | 0.10% | 0.50% | n.r. | 3 | n.r. | 12.07 | n.r. | [94] |
| solar radiation | Venlafaxine | 5 mg·L−1 | 28.6 mmol −1 | 2.3 | 60 | 40 | 98.5 | n.r. | [88] |
| UV254 | SMX | 5 mg·L−1 | 5~100 mg·L−1 | 7 ± 0.2 | 30 | n.r. | ~100 | R−O• | [36] |
| Fluoxetine | ~100 | R−O• | |||||||
| Carbamazepine | ~100 | R−O• | |||||||
| Naproxen | ~100 | R−O• | |||||||
| UV254 | methylene blue | 16 µM | 20 mg·L−1 | 7 | 14 | 20 ± 1 | ~80 | n.r. | [64] |
| UV254 | Bezafibrate | 1 µM | 1 mg·L−1 | 7.1 | 120 | n.r. | >90 | HO•>R−O• | [63] |
| Carbamazepine | 30 | >90 | |||||||
| Clofibric acid | 10 | >90 | |||||||
| Diclofenac | <5 | >90 | |||||||
| Ibuprofen | 30 | >90 | |||||||
| Naproxen | 10 | >95 | |||||||
| UV254 | methylene blue | 10 mg·L−1 | 50 mg·L−1 | 7.6 | 0–120 | n.r. | 85% | n.r. | [91] |
- 2.
- Catalyst Catalysis
5. The Factors That Influence the Removal of Organic Pollutants in PAA-Based Processes
5.1. PAA Dosage
5.2. Catalysts Dosage
5.3. pH
5.4. Temperature
5.5. Water Quality Components
5.5.1. Anions
5.5.2. Dissolved Organic Matter (DOM)
6. Conclusions and Future Prospects
Author Contributions
Funding
Conflicts of Interest
References
- Ling, M.; Lv, C.; Guo, X. Quantification method of water environmental value loss caused by water pollution based on emergy theory. Desalination Water Treat. 2018, 129, 299–303. [Google Scholar] [CrossRef] [Green Version]
- Liu, D.; Yuan, Y.; Wei, Y.; Zhang, H.; Si, Y.; Zhang, F. Removal of refractory organics and heavy metals in landfill leachate concentrate by peroxi-coagulation process. J. Environ. Sci. 2022, 116, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Ply, A.; Wjz, A.; Lhk, A.; Rfs, B.; Xjg, A.; Xi, Y.A.; Yan, C.A.; Wzl, A. Endow activated carbon with the ability to activate peroxymono-sulfate for the treatment of refractory organics. Green Chem. Eng. 2022. [Google Scholar] [CrossRef]
- Tawfik, A.; Alalm, M.G.; Awad, H.M.; Islam, M.; Qyyum, M.A.; Al-Muhtaseb, A.H.; Osman, A.I.; Lee, M. Solar photo-oxidation of recalcitrant industrial wastewater: A review. Environ. Chem. Lett. 2022, 20, 1839–1862. [Google Scholar] [CrossRef]
- Da Silva, F.C., Jr.; Felipe, M.B.M.C.; de Castro, D.E.F.; Araújo, S.C.D.S.; Sisenando, H.C.N.; de Medeiros, S.R.B. A look beyond the priority: A systematic review of the genotoxic, mutagenic, and carcinogenic endpoints of non-priority PAHs. Environ. Pollut. 2021, 278, 116838. [Google Scholar] [CrossRef]
- Sun, S.; Shen, J.; Li, D.; Li, B.; Sun, X.; Ma, L.; Qi, H. A new insight into the ARG association with antibiotics and non-antibiotic agents—Antibiotic resistance and toxicity. Environ. Pollut. 2021, 293, 118524. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Wang, X. Mechanism of removal of pharmaceuticals and personal care products by nanofiltration membranes. Desalination Water Treat. 2015, 53, 2816–2824. [Google Scholar] [CrossRef]
- Xu, R.; Fang, F.; Wang, L.; Luo, J.; Cao, J. Insight into the interaction between trimethoprim and soluble microbial products produced from biological wastewater treatment processes. J. Environ. Sci. 2022, 124, 130–138. [Google Scholar] [CrossRef]
- Lobato-Peralta, D.R.; Duque-Brito, E.; Ayala-Cortés, A.; Arias, D.; Longoria, A.; Cuentas-Gallegos, A.K.; Sebastian, P.; Okoye, P.U. Advances in activated carbon modification, surface heteroatom configuration, reactor strategies, and regeneration methods for enhanced wastewater treatment. J. Environ. Chem. Eng. 2021, 9, 105626. [Google Scholar] [CrossRef]
- Wang, T.; He, J.; Lu, J.; Zhou, Y.; Wang, Z.; Zhou, Y. Adsorptive removal of PPCPs from aqueous solution using carbon-based composites: A review. Chin. Chem. Lett. 2021, 33, 3585–3593. [Google Scholar] [CrossRef]
- Dong, F.; Li, C.; Ma, X.; Lin, Q.; He, G.; Chu, S. Degradation of estriol by chlorination in a pilot-scale water distribution system: Kinetics, pathway and DFT studies. Chem. Eng. J. 2020, 383, 123187. [Google Scholar] [CrossRef]
- Gan, W.; Ge, Y.; Zhu, H.; Huang, H.; Yang, X. ClO2 pre-oxidation changes the yields and formation pathways of chloroform and chloral hydrate from phenolic precursors during chlorination. Water Res. 2019, 148, 250–260. [Google Scholar] [CrossRef]
- Zheng, H.; Hou, Y.; Li, S.; Ma, J.; Nan, J.; Li, T. Recent advances in the application of metal organic frameworks using in advanced oxidation progresses for pollutants degradation. Chin. Chem. Lett. 2022. [Google Scholar] [CrossRef]
- Lin, Q.; Tan, X.; Almatrafi, E.; Yang, Y.; Wang, W.; Luo, H.; Qin, F.; Zhou, C.; Zeng, G.; Zhang, C. Effects of biochar-based materials on the bioavailability of soil organic pollutants and their biological impacts. Sci. Total Environ. 2022, 826, 153956. [Google Scholar] [CrossRef]
- Yang, L.; Zhou, Y.; Chen, L.; Chen, H.; Liu, W.; Zheng, W.; Andersen, M.E.; Zhang, Y.; Hu, Y.; Crabbe, M.J.C.; et al. Single enrichment systems possibly underestimate both exposures and biological effects of organic pollutants from drinking water. Chemosphere 2022, 292, 133496. [Google Scholar] [CrossRef]
- Zhang, T.; Dong, F.; Luo, F.; Li, C. Degradation of sulfonamides and formation of trihalomethanes by chlo-rination after pre-oxidation with Fe(VI). J. Environ. Sci. 2018, 73, 89–95. [Google Scholar] [CrossRef]
- Shi, Z.; Wang, D.; Gao, Z.; Ji, X.; Zhang, J.; Jin, C. Enhanced ferrate oxidation of organic pollutants in the presence of Cu(II) Ion. J. Hazard. Mater. 2022, 433, 128772. [Google Scholar] [CrossRef]
- Tian, S.-Q.; Wang, L.; Liu, Y.-L.; Ma, J. Degradation of organic pollutants by ferrate/biochar: Enhanced formation of strong intermediate oxidative iron species. Water Res. 2020, 183, 116054. [Google Scholar] [CrossRef]
- Rahmah, A.U.; Harimurti, S.; Murugesan, T. Experimental investigation on the effect of wastewater matrix on oxytetracycline mineralization using UV/H2O2 system. Int. J. Environ. Sci. Technol. 2017, 14, 1225–1233. [Google Scholar] [CrossRef]
- Chaves, F.P.; Gomes, G.; Della-Flora, A.; Dallegrave, A.; Sirtori, C.; Saggioro, E.M.; Bila, D.M. Comparative endocrine disrupting compound removal from real wastewater by UV/Cl and UV/H2O2: Effect of pH, estrogenic activity, transformation products and toxicity. Sci. Total Environ. 2020, 746, 141041. [Google Scholar] [CrossRef]
- Qu, C.; Liang, D.-W. Novel electrochemical advanced oxidation processes with H2O2 generation cathode for water treatment: A review. J. Environ. Chem. Eng. 2022, 10, 107896. [Google Scholar] [CrossRef]
- He, G.; Zhang, T.; Zheng, F.; Li, C.; Zhang, Q.; Dong, F.; Huang, Y. Reaction of fleroxacin with chlorine and chlorine dioxide in drinking water distribution systems: Kinetics, transformation mechanisms and toxicity evaluations. Chem. Eng. J. 2019, 374, 1191–1203. [Google Scholar] [CrossRef]
- Li, C.; Luo, F.; Duan, H.; Dong, F.; Chen, X.; Feng, M.; Zhang, Z.; Cizmas, L.; Sharma, V.K. Degradation of chloramphenicol by chlorine and chlorine dioxide in a pilot-scale water distribution system. Sep. Purif. Technol. 2019, 211, 564–570. [Google Scholar] [CrossRef]
- Su, R.; Huang, L.; Li, N.; Li, L.; Jin, B.; Zhou, W.; Gao, B.; Yue, Q.; Li, Q. Chlorine dioxide radicals triggered by chlorite under visible-light irradiation for enhanced degradation and detoxification of norfloxacin antibiotic: Radical mechanism and toxicity evaluation. Chem. Eng. J. 2021, 414, 128768. [Google Scholar] [CrossRef]
- Pearce, R.; Hogard, S.; Buehlmann, P.; Salazar-Benites, G.; Wilson, C.; Bott, C. Evaluation of preformed monochloramine for bromate control in ozonation for potable reuse. Water Res. 2022, 211, 118049. [Google Scholar] [CrossRef]
- Wang, Y.; Man, T.; Zhang, R.; Yan, X.; Wang, S.; Zhang, M.; Wang, P.; Ren, L.; Yu, J.; Li, C. Effects of organic matter, ammonia, bromide, and hydrogen peroxide on bromate formation during water ozonation. Chemosphere 2021, 285, 131352. [Google Scholar] [CrossRef]
- Zhang, K.; Zhou, X.; Zhang, T.; Yu, L.; Qian, Z.; Liao, W.; Li, C. Degradation of the earthy and musty odorant 2,4,6-tricholoroanisole by persulfate activated with iron of different valences. Environ. Sci. Pollut. Res. 2017, 25, 3435–3445. [Google Scholar] [CrossRef]
- Aziz, K.H.H. Application of different advanced oxidation processes for the removal of chloroacetic acids using a planar falling film reactor. Chemosphere 2019, 228, 377–383. [Google Scholar] [CrossRef]
- Nam, S.-N.; Choong, C.E.; Hoque, S.; Farouk, T.I.; Cho, J.; Jang, M.; Snyder, S.A.; Meadows, M.E.; Yoon, Y. Catalytic non-thermal plasma treatment of endocrine disrupting compounds, pharmaceuticals, and personal care products in aqueous solution: A review. Chemosphere 2022, 290, 133395. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, Q.; Ji, G.; Li, A. Degradation of antibiotic pollutants by persulfate activated with various carbon materials. Chem. Eng. J. 2021, 429, 132387. [Google Scholar] [CrossRef]
- Peng, W.; Dong, Y.; Fu, Y.; Wang, L.; Li, Q.; Liu, Y.; Fan, Q.; Wang, Z. Non-radical reactions in persulfate-based homogeneous degradation processes: A review. Chem. Eng. J. 2021, 421, 127818. [Google Scholar] [CrossRef]
- Ao, X.-W.; Eloranta, J.; Huang, C.-H.; Santoro, D.; Sun, W.-J.; Lu, Z.-D.; Li, C. Peracetic acid-based advanced oxidation processes for decontamination and disinfection of water: A review. Water Res. 2021, 188, 116479. [Google Scholar] [CrossRef] [PubMed]
- Collazo, C.; Charles, F.; Aguiló-Aguayo, I.; Marín-Sáez, J.; Lafarga, T.; Abadias, M.; Viñas, I. Decontamination of Listeria innocua from fresh-cut broccoli using UV-C applied in water or peroxyacetic acid, and dry-pulsed light. Innov. Food Sci. Emerg. Technol. 2019, 52, 438–449. [Google Scholar] [CrossRef] [Green Version]
- de Souza, J.B.; Valdez, F.Q.; Jeranoski, R.F.; Vidal, C.M.D.S.; Cavallini, G.S. Water and Wastewater Disinfection with Peracetic Acid and UV Radiation and Using Advanced Oxidative Process PAA/UV. Int. J. Photoenergy 2015, 2015, 860845. [Google Scholar] [CrossRef] [Green Version]
- Henao, L.D.; Turolla, A.; Antonelli, M. Disinfection by-products formation and ecotoxicological effects of effluents treated with peracetic acid: A review. Chemosphere 2018, 213, 25–40. [Google Scholar] [CrossRef]
- Hollman, J.; Dominic, J.A.; Achari, G. Degradation of pharmaceutical mixtures in aqueous solutions using UV/peracetic acid process: Kinetics, degradation pathways and comparison with UV/H2O2. Chemosphere 2020, 248, 125911. [Google Scholar] [CrossRef]
- Jahangiri-Rad, M.; Nadafi, K.; Mesdaghinia, A.; Nabizadeh, R.; Younesian, M.; Rafiee, M. Sequential study on reactive blue 29 dye removal from aqueous solution by peroxy acid and single wall carbon nanotubes: Experiment and theory. Iran. J. Environ. Health Sci. Eng. 2013, 10, 5. [Google Scholar] [CrossRef] [Green Version]
- Chhetri, R.K.; Baun, A.; Andersen, H.R. Acute toxicity and risk evaluation of the CSO disinfectants performic acid, peracetic acid, chlorine dioxide and their by-products hydrogen peroxide and chlorite. Sci. Total Environ. 2019, 677, 1–8. [Google Scholar] [CrossRef]
- Fraisse, A.; Temmam, S.; Deboosere, N.; Guillier, L.; Delobel, A.; Maris, P.; Vialette, M.; Morin, T.; Perelle, S. Comparison of chlorine and peroxyacetic-based disinfectant to inactivate Feline calicivirus, Murine norovirus and Hepatitis A virus on lettuce. Int. J. Food Microbiol. 2011, 151, 98–104. [Google Scholar] [CrossRef]
- Kiejza, D.; Kotowska, U.; Polińska, W.; Karpińska, J. Peracids—New oxidants in advanced oxidation processes: The use of peracetic acid, peroxymonosulfate, and persulfate salts in the removal of organic micropollutants of emerging concern—A review. Sci. Total Environ. 2021, 790, 148195. [Google Scholar] [CrossRef]
- Maurício, R.; Jorge, J.; Dias, R.; Noronha, J.P.; Amaral, L.; Daam, M.A.; Mano, A.P.; Diniz, M.S. The use of peracetic acid for estrogen removal from urban wastewaters: E2 as a case study. Environ. Monit. Assess. 2020, 192, 114. [Google Scholar] [CrossRef]
- Deng, J.; Wang, H.; Fu, Y.; Liu, Y. Phosphate-induced activation of peracetic acid for diclofenac degradation: Kinetics, influence factors and mechanism. Chemosphere 2022, 287, 132396. [Google Scholar] [CrossRef]
- Jian, L. Degradation of Antibiotics in Water by Advanced Oxidation Technologies Based on the Application of Peroxide. Master’s Thesis, Suzhou University of Science and Technology, Suzhou, China, 2019. [Google Scholar]
- Liu, B.; Guo, W.; Jia, W.; Wang, H.; Si, Q.; Zhao, Q.; Luo, H.; Jiang, J.; Ren, N. Novel Nonradical Oxidation of Sulfonamide Antibiotics with Co(II)-Doped g-C3N4-Activated Peracetic Acid: Role of High-Valent Cobalt–Oxo Species. Environ. Sci. Technol. 2021, 55, 12640–12651. [Google Scholar] [CrossRef]
- Shah, A.D.; Liu, Z.-Q.; Salhi, E.; Höfer, T.; von Gunten, U. Peracetic Acid Oxidation of Saline Waters in the Absence and Presence of H2O2: Secondary Oxidant and Disinfection Byproduct Formation. Environ. Sci. Technol. 2015, 49, 1698–1705. [Google Scholar] [CrossRef]
- Ghanbari, F.; Giannakis, S.; Lin, K.-Y.A.; Wu, J.; Madihi-Bidgoli, S. Acetaminophen degradation by a synergistic peracetic acid/UVC-LED/Fe(II) advanced oxidation process: Kinetic assessment, process feasibility and mechanistic considerations. Chemosphere 2021, 263, 128119. [Google Scholar] [CrossRef]
- Wang, J.; Xiong, B.; Miao, L.; Wang, S.; Xie, P.; Wang, Z.; Ma, J. Applying a novel advanced oxidation process of activated peracetic acid by CoFe2O4 to efficiently degrade sulfamethoxazole. Appl. Catal. B Environ. 2021, 280, 119422. [Google Scholar] [CrossRef]
- Zhou, F.; Lu, C.; Yao, Y.; Sun, L.; Gong, F.; Li, D.; Pei, K.; Lu, W.; Chen, W. Activated carbon fibers as an effective metal-free catalyst for peracetic acid activation: Implications for the removal of organic pollutants. Chem. Eng. J. 2015, 281, 953–960. [Google Scholar] [CrossRef]
- Da Silva, W.P.; Carlos, T.D.; Cavallini, G.S.; Pereira, D.H. Peracetic acid: Structural elucidation for applications in wastewater treatment. Water Res. 2020, 168, 115143. [Google Scholar] [CrossRef]
- Keller, B.; Wojcik, M.; Fletcher, T. A directly-dissociative stepwise reaction mechanism for gas-phase peroxyacetic acid. J. Photochem. Photobiol. A Chem. 2008, 195, 10–22. [Google Scholar] [CrossRef]
- Swern, D.; Silbert, L.S. Studies in the Structure of Organic Peroxides. Anal. Chem. 1963, 35, 880–885. [Google Scholar] [CrossRef]
- Rittenhouse, J.R.; Lobunez, W.; Swern, D.; Miller, J.G. The Electric Moments of Crganic Peroxides. II. Aliphatic Peracids1. J. Am. Chem. Soc. 1958, 80, 4850–4852. [Google Scholar] [CrossRef]
- Zhang, K.; Zhou, X.; Du, P.; Zhang, T.; Cai, M.; Sun, P.; Huang, C.-H. Oxidation of β-lactam antibiotics by peracetic acid: Reaction kinetics, product and pathway evaluation. Water Res. 2017, 123, 153–161. [Google Scholar] [CrossRef]
- Kim, J.; Zhang, T.; Liu, W.; Du, P.; Dobson, J.T.; Huang, C.-H. Advanced Oxidation Process with Peracetic Acid and Fe(II) for Contaminant Degradation. Environ. Sci. Technol. 2019, 53, 13312–13322. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, J.; Zhang, Y.; Yu, Z.; Ji, R.; Zhou, X. Activation of peracetic acid with cobalt anchored on 2D sandwich-like MXenes (Co@MXenes) for organic contaminant degradation: High efficiency and contribution of acetylperoxyl radicals. Appl. Catal. B Environ. 2021, 297, 120475. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, J.; Xiong, B.; Bai, F.; Wang, S.; Wan, Y.; Zhang, L.; Xie, P.; Wiesner, M.R. Application of Cobalt/Peracetic Acid to Degrade Sulfamethoxazole at Neutral Condition: Efficiency and Mechanisms. Environ. Sci. Technol. 2020, 54, 464–475. [Google Scholar] [CrossRef]
- Cao, C.; Zhang, S.; Zhang, F.; Zhang, K. Research Progress of Peracetic Acid (PAA): An Emerging Disinfectant in Drinking Water. China Water Wastewater 2018, 34, 36–40. [Google Scholar] [CrossRef]
- Ison, S.; Beattie, M. Disinfection, sterilization and preservation (5th ed). Aust. Infect. Control 2002, 7, 74. [Google Scholar] [CrossRef]
- Kitis, M. Disinfection of wastewater with peracetic acid: A review. Environ. Int. 2004, 30, 47–55. [Google Scholar] [CrossRef]
- Pohjanvesi, S.; Mustonen, E.L.; Pukkinen, A.; Lehtinen, R. Process for the Production of Percarboxylic Acid. U.S. Patent 177732[P], 4 April 2002. [Google Scholar]
- Palani, A.; Pandurangan, A. Single pot synthesis of peroxyacetic acid from acetic acid and hydrogen peroxide using various solid acid catalysts. Catal. Commun. 2006, 7, 875–878. [Google Scholar] [CrossRef]
- Saha, M.S.; Denggerile, A.; Nishiki, Y.; Furuta, T.; Ohsaka, T. Synthesis of peroxyacetic acid using in situ electrogenerated hydrogen peroxide on gas diffusion electrode. Electrochem. Commun. 2003, 5, 445–448. [Google Scholar] [CrossRef]
- Cai, M.; Sun, P.; Zhang, L.; Huang, C.-H. UV/Peracetic Acid for Degradation of Pharmaceuticals and Reactive Species Evaluation. Environ. Sci. Technol. 2017, 51, 14217–14224. [Google Scholar] [CrossRef]
- Chen, S.; Cai, M.; Liu, Y.; Zhang, L.; Feng, L. Effects of water matrices on the degradation of naproxen by reactive radicals in the UV/peracetic acid process. Water Res. 2019, 150, 153–161. [Google Scholar] [CrossRef]
- Davies, D.M.; Deary, M.E. Determination of peracids in the presence of a large excess of hydrogen peroxide using a rapid and convenient spectrophotometric method. Analyst 1988, 113, 1477–1479. [Google Scholar] [CrossRef]
- Tashkhourian, J.; Hormozi-Nezhad, M.R.; Khodaveisi, J.; Dashti, R. Localized surface plasmon resonance sensor for simultaneous kinetic determination of peroxyacetic acid and hydrogen peroxide. Anal. Chim. Acta 2013, 762, 87–93. [Google Scholar] [CrossRef]
- Zhang, T.; Huang, C.-H. Modeling the Kinetics of UV/Peracetic Acid Advanced Oxidation Process. Environ. Sci. Technol. 2020, 54, 7579–7590. [Google Scholar] [CrossRef]
- Zhao, Z.; Li, X.; Li, H.; Qian, J.; Pan, B. New Insights into the Activation of Peracetic Acid by Co(II): Role of Co(II)-Peracetic Acid Complex as the Dominant Intermediate Oxidant. ACS ES T Eng. 2021, 1, 1432–1440. [Google Scholar] [CrossRef]
- Manoli, K.; Li, R.; Kim, J.; Feng, M.; Huang, C.-H.; Sharma, V.K. Ferrate(VI)-peracetic acid oxidation process: Rapid degradation of pharmaceuticals in water. Chem. Eng. J. 2022, 429, 132384. [Google Scholar] [CrossRef]
- Carlos, T.D.; Bezerra, L.B.; Vieira, M.M.; Sarmento, R.A.; Pereira, D.H.; Cavallini, G.S. Fenton-type process using peracetic acid: Efficiency, reaction elucidations and ecotoxicity. J. Hazard. Mater. 2021, 403, 123949. [Google Scholar] [CrossRef]
- Chen, S. Degradation Mechanisms of Nap and Effects of Water Matrices on Free Radicals Conversion in UV/Peracetic Acid Process. Master’s Thesis, Beijing Forestry University, Beijing, China, 2019. [Google Scholar]
- Luukkonen, T.; Heyninck, T.; Rämö, J.; Lassi, U. Comparison of organic peracids in wastewater treatment: Disinfection, oxidation and corrosion. Water Res. 2015, 85, 275–285. [Google Scholar] [CrossRef]
- Maurício, R.; Semedo, F.; Dias, R.; Noronha, J.P.; Amaral, L.; Daam, M.; Mano, A.P.; Diniz, M. Efficacy assessment of peracetic acid in the removal of synthetic 17α-ethinyl estradiol contraceptive hormone in wastewater. J. Environ. Sci. 2020, 89, 1–8. [Google Scholar] [CrossRef]
- Wu, J.; Zheng, X.; Wang, Y.; Liu, H.; Wu, Y.; Jin, X.; Chen, P.; Lv, W.; Liu, G. Activation of peracetic acid via Co3O4 with double-layered hollow structures for the highly efficient removal of sulfonamides: Kinetics insights and assessment of practical applications. J. Hazard. Mater. 2022, 431, 128579. [Google Scholar] [CrossRef]
- Wu, W.; Tian, D.; Liu, T.; Chen, J.; Huang, T.; Zhou, X.; Zhang, Y. Degradation of organic compounds by peracetic acid activated with Co3O4: A novel advanced oxidation process and organic radical contribution. Chem. Eng. J. 2020, 394, 124938. [Google Scholar] [CrossRef]
- Hey, G.; Ledin, A.; Jansen, J.L.C.; Andersen, H.R. Removal of pharmaceuticals in biologically treated wastewater by chlorine dioxide or peracetic acid. Environ. Technol. 2012, 33, 1041–1047. [Google Scholar] [CrossRef]
- Kim, J.; Du, P.; Liu, W.; Luo, C.; Zhao, H.; Huang, C.-H. Cobalt/Peracetic Acid: Advanced Oxidation of Aromatic Organic Compounds by Acetylperoxyl Radicals. Environ. Sci. Technol. 2020, 54, 5268–5278. [Google Scholar] [CrossRef]
- Wang, H.; Deng, J.; Lu, X.; Wan, L.; Huang, J.; Liu, Y. Rapid and continuous degradation of diclofenac by Fe(II)-activated persulfate combined with bisulfite. Sep. Purif. Technol. 2021, 262, 118335. [Google Scholar] [CrossRef]
- Zhou, X.; Luo, H.; Sheng, B.; Chen, X.; Wang, Y.; Chen, Q.; Zhou, J. Cu2+/Cu+ cycle promoted PMS decomposition with the assistance of Mo for the degradation of organic pollutant. J. Hazard. Mater. 2021, 411, 125050. [Google Scholar] [CrossRef]
- Wang, Z.; Fu, Y.; Peng, Y.; Wang, S.; Liu, Y. HCO3–/CO32– enhanced degradation of diclofenac by Cu(II)-activated peracetic acid: Efficiency and mechanism. Sep. Purif. Technol. 2021, 277, 119434. [Google Scholar] [CrossRef]
- Zhang, L.; Fu, Y.; Wang, Z.; Zhou, G.; Zhou, R.; Liu, Y. Removal of diclofenac in water using peracetic acid activated by zero valent copper. Sep. Purif. Technol. 2021, 276, 119319. [Google Scholar] [CrossRef]
- Jingxiao, W.; Kean, Z.; Fei, C. Degradation performance and mechanism of Rhodamine B by chloride Ion activated peracetic acid. Res. Environ. Sci. 2021, 34, 2850–2858. [Google Scholar] [CrossRef]
- Haysar, A.; Jia, J.; Ren, D.; Liao, Y.; Wang, J.; Zhao, M. The removal of hpam with fenton-like reagents oxidation process. Spec. Petrochem. 2016, 33, 4. [Google Scholar] [CrossRef]
- Wang, Z.; Shi, H.; Wang, S.; Liu, Y.; Fu, Y. Degradation of diclofenac by Fe(II)-activated peracetic acid. Environ. Technol. 2021, 42, 4333–4341. [Google Scholar] [CrossRef] [PubMed]
- Tian, D.; Wu, W.; Shen, Z.; Huang, T.; Chen, J. Degradation of organic dyes with peracetic acid activated by Co(II). Acta Sci. Circumstantiae 2018, 38, 4023–4031. [Google Scholar] [CrossRef]
- Rothbart, S.; Ember, E.E.; van Eldik, R. Mechanistic studies on the oxidative degradation of Orange II by peracetic acid catalyzed by simple manganese(ii) salts. Tuning the lifetime of the catalyst. New J. Chem. 2012, 36, 732–748. [Google Scholar] [CrossRef]
- Yuan, D.; Yang, K.; Pan, S.; Xiang, Y.; Tang, S.; Huang, L.; Sun, M.; Zhang, X.; Jiao, T.; Zhang, Q.; et al. Peracetic acid enhanced electrochemical advanced oxidation for organic pollutant elimination. Sep. Purif. Technol. 2021, 276, 119317. [Google Scholar] [CrossRef]
- Bezerra, L.B.; Carlos, T.D.; das Neves, A.P.N.; Durães, W.A.; Sarmento, R.D.A.; Pereira, D.H.; Cavallini, G.S. Theoretical-experimental study of the advanced oxidative process using peracetic acid and solar radiation: Removal efficiency and thermodynamic elucidation of radical formation processes. J. Photochem. Photobiol. A Chem. 2022, 423, 113615. [Google Scholar] [CrossRef]
- Wang, J.; Wan, Y.; Ding, J.; Wang, Z.; Ma, J.; Xie, P.; Wiesner, M.R. Thermal Activation of Peracetic Acid in Aquatic Solution: The Mechanism and Application to Degrade Sulfamethoxazole. Environ. Sci. Technol. 2020, 54, 14635–14645. [Google Scholar] [CrossRef]
- Daswat, D.P.; Mukhopadhyay, M. Photochemical degradation of chlorophenol industry wastewater using peroxy acetic acid (PAA). Chem. Eng. J. 2012, 209, 1–6. [Google Scholar] [CrossRef]
- Rizzo, L.; Lofrano, G.; Gago, C.; Bredneva, T.; Iannece, P.; Pazos, M.; Krasnogorskaya, N.; Carotenuto, M. Antibiotic contaminated water treated by photo driven advanced oxidation processes: Ultraviolet/H2O2 vs ultraviolet/peracetic acid. J. Clean. Prod. 2018, 205, 67–75. [Google Scholar] [CrossRef]
- Li, Z.; Yong-sheng, F.; Yi-qing, L. Degradation of diclofenac in water by Cu2+ enhanced UV activation of peracetic acid. China Environ. Sci. 2020, 40, 5260–5269. [Google Scholar] [CrossRef]
- Yinhao, D.; Shaogui, Y.; Chengdu, Q.; Shiyin, L.; Xiaolong, D.; Huan, H.; Haiou, S. Activation of peracetic acid process for aquatic organic pollutants degradation: A review. Environ. Chem. Lett. 2021, 40, 497–508. [Google Scholar] [CrossRef]
- Tang, L.; Long, K.; Chen, S.; Gui, D.; He, C.; Li, J.; Tao, X. Removal of thiophene sulfur model compound for coal by microwave with peroxyacetic acid. Fuel 2020, 272, 117748. [Google Scholar] [CrossRef]
- Chin, J.F.; Heng, Z.W.; Teoh, H.C.; Chong, W.C.; Pang, Y.L. Recent development of magnetic biochar crosslinked chitosan on heavy metal removal from wastewater—Modification, application and mechanism. Chemosphere 2022, 291, 133035. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, S.; Sun, Y.; Du, Q.; Song, J.; Tsang, D.C. A novel electrochemical modification combined with one-step pyrolysis for preparation of sustainable thorn-like iron-based biochar composites. Bioresour. Technol. 2019, 274, 379–385. [Google Scholar] [CrossRef]
- Yang, J.; Zhao, Y.; Ma, S.; Zhu, B.; Zhang, J.; Zheng, C. Mercury Removal by Magnetic Biochar Derived from Simultaneous Activation and Magnetization of Sawdust. Environ. Sci. Technol. 2016, 50, 12040–12047. [Google Scholar] [CrossRef]
- Wang, S.; Wang, H.; Liu, Y.; Fu, Y. Effective degradation of sulfamethoxazole with Fe2+-zeolite/peracetic acid. Sep. Purif. Technol. 2020, 233, 115973. [Google Scholar] [CrossRef]
- Rokhina, E.V.; Makarova, K.; Lahtinen, M.; Golovina, E.A.; Van As, H.; Virkutyte, J. Ultrasound-assisted MnO2 catalyzed homolysis of peracetic acid for phenol degradation: The assessment of process chemistry and kinetics. Chem. Eng. J. 2013, 221, 476–486. [Google Scholar] [CrossRef]
- Rokhina, E.V.; Makarova, K.; Golovina, E.A.; Van As, H.; Virkutyte, J. Free Radical Reaction Pathway, Thermochemistry of Peracetic Acid Homolysis, and Its Application for Phenol Degradation: Spectroscopic Study and Quantum Chemistry Calculations. Environ. Sci. Technol. 2010, 44, 6815–6821. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Activation of persulfate (PS) and peroxymonosulfate (PMS) and application for the degradation of emerging contaminants. Chem. Eng. J. 2018, 334, 1502–1517. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Z.; Cheng, Y.; Cao, L.; Bai, F.; Yue, S.; Xie, P.; Ma, J. Molybdenum disulfide (MoS2): A novel activator of peracetic acid for the degradation of sulfonamide antibiotics. Water Res. 2021, 201, 117291. [Google Scholar] [CrossRef]
- Huang, T.; Chen, J.; Shen, Z.; Tian, D. A Method for Degrading Azo Dye Golden Orange G in Wastewater. CN108298668A, 20 July 2018. [Google Scholar]
- Zhang, L.; Liu, Y.; Fu, Y. Degradation kinetics and mechanism of diclofenac by UV/peracetic acid. RSC Adv. 2020, 10, 9907–9916. [Google Scholar] [CrossRef] [Green Version]
- Mukhopadhyay, M.; Daswat, D.P. Kinetic and mechanistic study of photochemical degradation of 4-chlorophenol using peroxy acetic acid (PAA). Desalination Water Treat. 2014, 52, 5704–5714. [Google Scholar] [CrossRef]
- Izadifard, M.; Achari, G.; Langford, C.H. Degradation of sulfolane using activated persulfate with UV and UV-Ozone. Water Res. 2017, 125, 325–331. [Google Scholar] [CrossRef] [PubMed]
| Items | Value | |
|---|---|---|
| pKa | 8.2 | |
| Eh0(V) | PAA | 1.0~1.96 |
| H2O2 | 1.78 | |
| Cl | 1.48 | |
| ClO2 | 1.28 | |
| FeVI | 0.9~1.9 | |
| O3 | 2.08 | |
| Molar mass (g/mol) | 76.05 | |
| Density (g/mol) | 1.0375 | |
| boiling point (°C) | 110 | |
| melting point (°C) | 0.2 | |
| Henry’s law constant (M/atm) | 4.68 × 102 | |
| log Kow (at pH 7) | −0.66 | |
| O-OH bond(kJ/mol) | PAA | 159 |
| H2O2 | 213 | |
| Contaminants | C0 (μg·L−1) | CPAA (mg·L−1) | pH | Time (min) | Max. Removal Efficiency (%) | Matrixes | References |
|---|---|---|---|---|---|---|---|
| 17 β-Estradiol | 50 | 15 | 7.9 | 10–20 | 100 | WW 1 | [41] |
| 17 α-ethinyl estradiol | 50 | 15 | 7.9 | 10–20 | 100 | WW | [73] |
| Acetaminophen | 20 mg·L−1 | 4 mM | 5 | 30 | ~15 | DI water 2 | [46] |
| Reactive Brilliant Red X-3B | 50 μM | 5 mM | 7 | 40 | <5 | DI water | [48] |
| sulfamethazine | 8 mg·L−1 | 0.4 mM | 6.3 | 60 | ~20 | DI water | [74] |
| 2,4-dichlorophenol | 20 μM | 0.26 mM | 7 | 20 | <5 | DI water | [55] |
| sulfamethazine | 10 μM | 100 μM | 7 ± 0.2 | 15 | <5 | DI water | [56] |
| Clofibric acid | 0.05 mM | 0.5 mM | 7 | 90 | <5 | DI water | [75] |
| Ibuprofen | 40 | 50 | 6.7 (WW 1 3), 7.0 (WW 2 4 and WW 3 5) | n.r. 6 | ~45 (WW 1), ~20 (WW 2), <5 (WW 3) | WW 1, WW 2, WW 3 | [76] |
| Naproxen | ~80 (WW 1), ~60 (WW 2), ~40 (WW 3) | ||||||
| Diclofenac | ~75 (WW 1), >95 (WW 2), ~20 (WW 3) | ||||||
| Mefenamic acid | >95 (WW 1), ~90 (WW 2), ~90 (WW 3) | ||||||
| Gemfibrozil | ~75 (WW 1), ~20 (WW 2), ~20 (WW 3) | ||||||
| Clofibric acid | ~60 (WW 1), ~45 (WW 2), <10 (WW 3) | ||||||
| Blue 29 dye | 30 mg ·L−1 | CH3C(O)OH/dye = 344/1 | 3–8 | 60–180 | 20.2~56.4 | DI water | [37] |
| Catalysts | CCatalyst (g·L−1) | Contaminants | C0 | CPAA (mM) | pH | Time (min) | Max. Removal Efficiency (%) | References |
|---|---|---|---|---|---|---|---|---|
| Ultrasound/MnO2 | 1 | Phenol | 98.8 mg·L−1 | 100 | 7 | 20 | 89 | [99] |
| MnO2 | 0.7 | Phenol | n.r. | 50 mg·L−1 | 9.5 | 120 | n.r. | [100] |
| Co3O4 | 0.2 | Orange G | n.r. | 1 | 7 | 90 | 100 | [75] |
| LaCoO3 | 0.1 | SMX | 8 mg·L−1 | 0.4 | n.r. | 60 | 100 | [74] |
| CoFe2O3 | 0.2 | SMX | n.r. | 0.2 | 7 | 30 | 74.7 | [47] |
| ACF | 2 | Reactive Brilliant Red X-3B | 30.8 mg·L−1 | 380 mg·L−1 | 7 | 40 | 92.5 | [48] |
| Co(II)/g-C3N4 | 0.1 | SMX | 10 μM | 0.2 | 4.24 | 30 | ~100 | [44] |
| Fe2+-zeolite | 0.8 | SMX | 5 μM | 400 μM | 7 | 50 | ~100 | [101] |
| Co@MXenes | 10 mg·L−1 | 2,4-dichloropheno | 20 μM | 0.26 | 7 | 20 | ~100 | [55] |
| MoS2 | 0.3 | SMX | 10 μM | 0.3 | 3 | 15 | 76.1 | [102] |
| Phenol-AC | 0.1–0.2 | Orange G | n.r. | 2.6 × 10−4~3.9 × 10−3 | 7 | n.r. | Effective degradation | [103] |
| N-rGO | 0.75 | SMX | 0.15 mM | 1 | 3 | 60 | 96 | [43] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, C.; Li, C.; Wang, Y.; Guo, J.; Barry, S.; Zhang, Y.; Marmier, N. Review of Advanced Oxidation Processes Based on Peracetic Acid for Organic Pollutants. Water 2022, 14, 2309. https://doi.org/10.3390/w14152309
Shi C, Li C, Wang Y, Guo J, Barry S, Zhang Y, Marmier N. Review of Advanced Oxidation Processes Based on Peracetic Acid for Organic Pollutants. Water. 2022; 14(15):2309. https://doi.org/10.3390/w14152309
Chicago/Turabian StyleShi, Changjie, Cong Li, Yong Wang, Jiaqi Guo, Sadou Barry, Yunshu Zhang, and Nicolas Marmier. 2022. "Review of Advanced Oxidation Processes Based on Peracetic Acid for Organic Pollutants" Water 14, no. 15: 2309. https://doi.org/10.3390/w14152309







