The Sorbents Based on Acrylic Fiber Impregnated by Iron Hydroxide (III): Production Methods, Properties, Application in Oceanographic Research
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
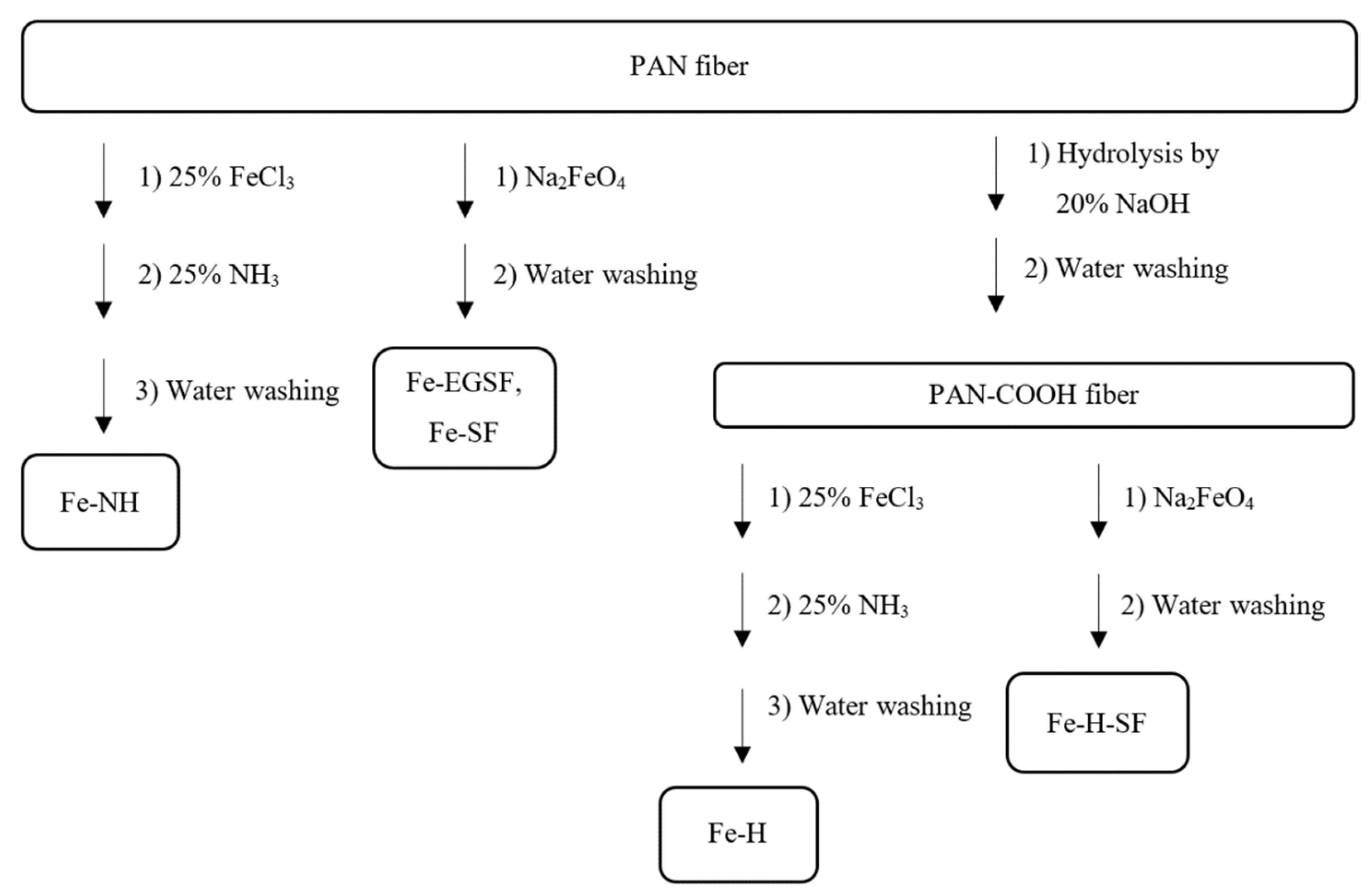
2.2. Preparation of PAN-Fe(OH)3 by Various Methods
2.2.1. Preparation of Fe-NH
2.2.2. Preparation of Fe-EGSF Using Na2FeO4 Generated Electrochemically
2.2.3. Preparation of Fe-SF Using Prepared Na2FeO4
2.2.4. Preparation of Fe-H
2.2.5. Preparation of Fe-H-SF
2.3. Study of the Sorbent by Structural Methods
2.4. Study of the Fe(OH)3 Washout from the Sorbent
2.5. Determining the Sorption Parameters of Phosphorus and Beryllium under Static Conditions
2.6. Sorption of Phosphorus and Beryllium under Dynamic Conditions
2.7. Recovery of the Radionuclides from Large-Volume Samples
2.8. Measurement of Gamma Radionuclides
3. Results and Discussion
3.1. Obtaining Sorbents
3.1.1. Preparation of Fe-NH Sorbent
3.1.2. Preparation of Fe-EGSF Sorbent Using Electrochemically Generated Na2FeO4
3.1.3. Preparation of Fe-SF Sorbent Using Ready-Made Na2FeO4
3.1.4. Preparation of Fe-H Sorbent
3.1.5. Preparation of Fe-H-SF Sorbent
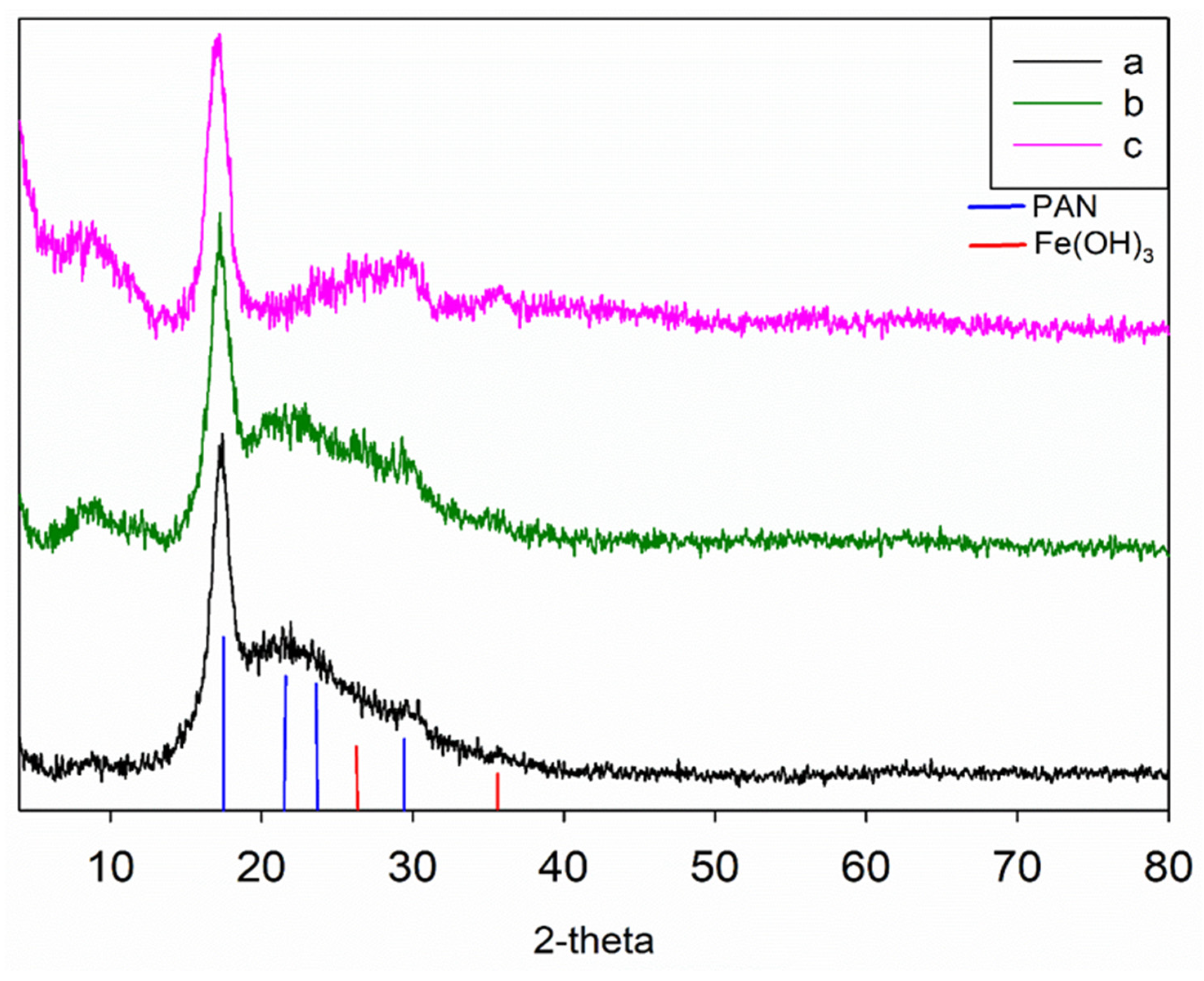
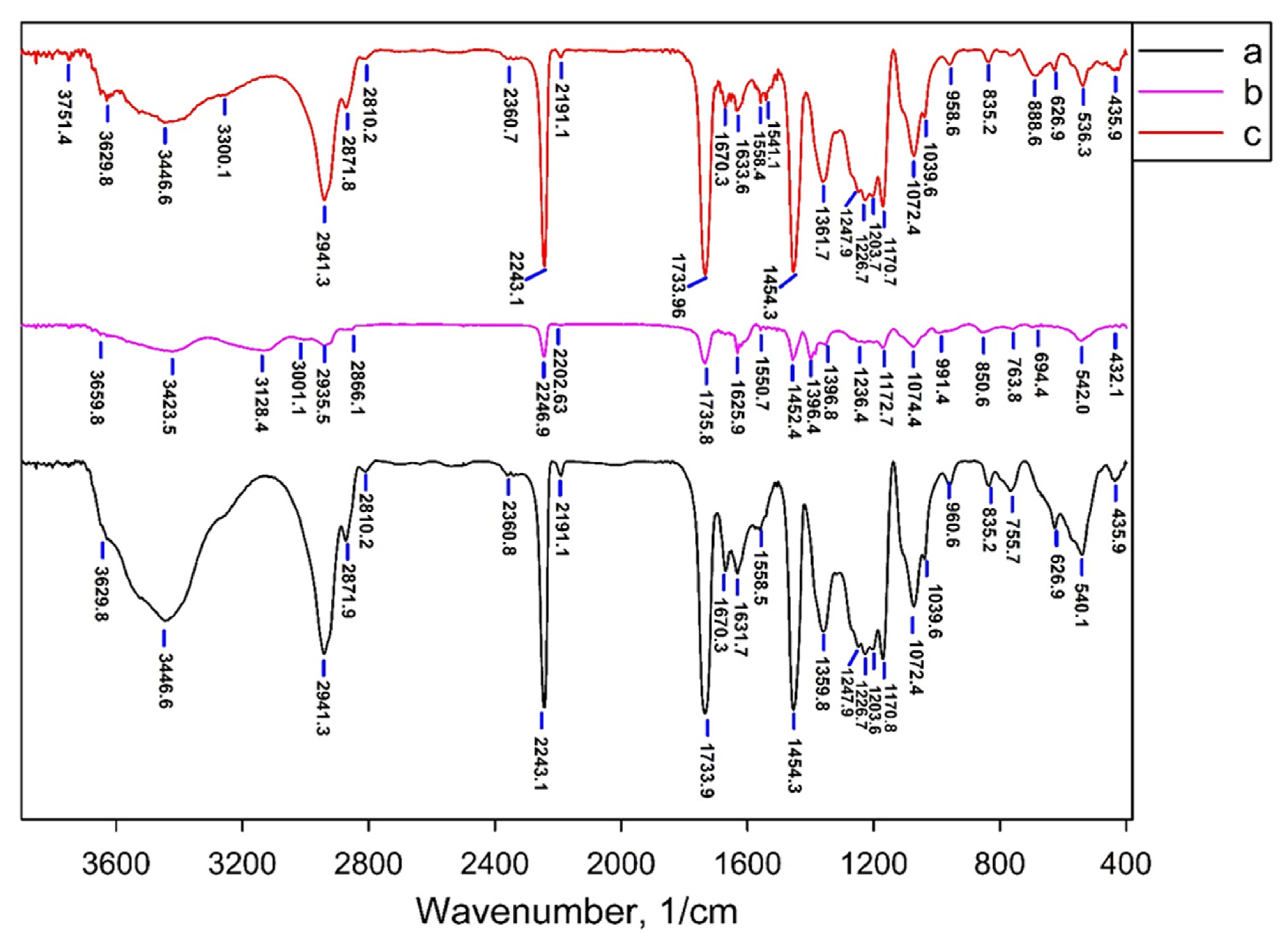
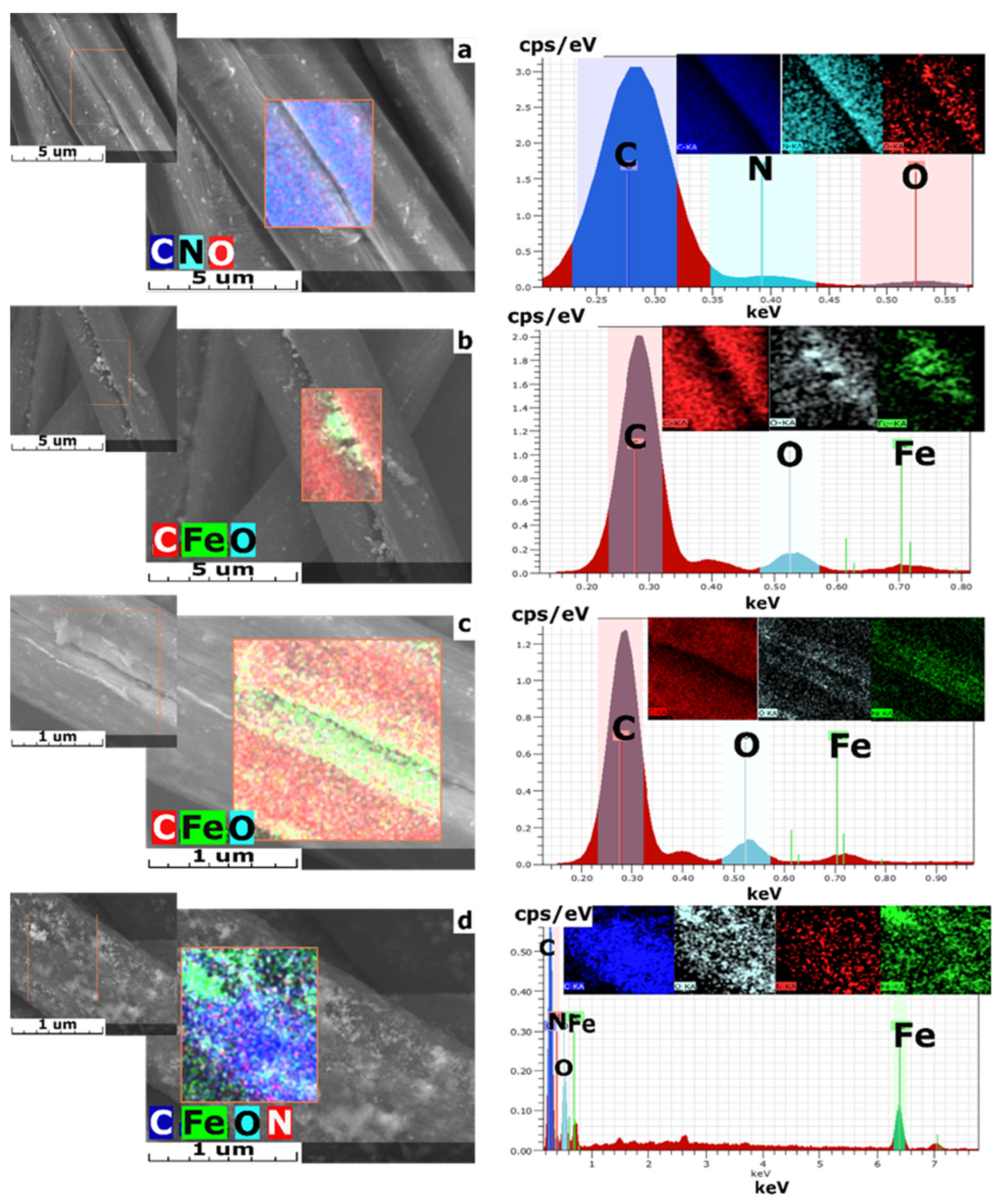
3.2. The Structure of the Obtained Sorbents
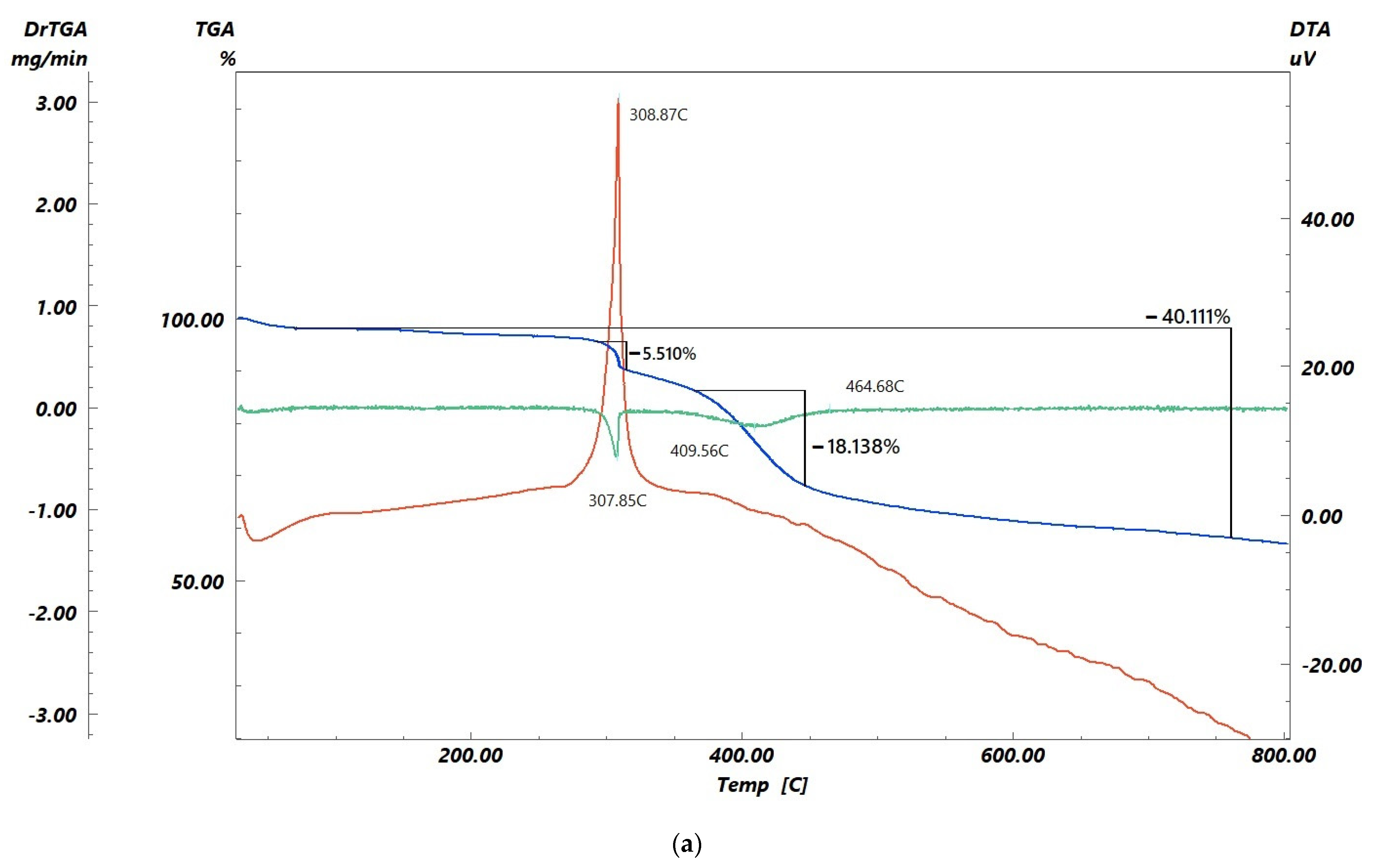
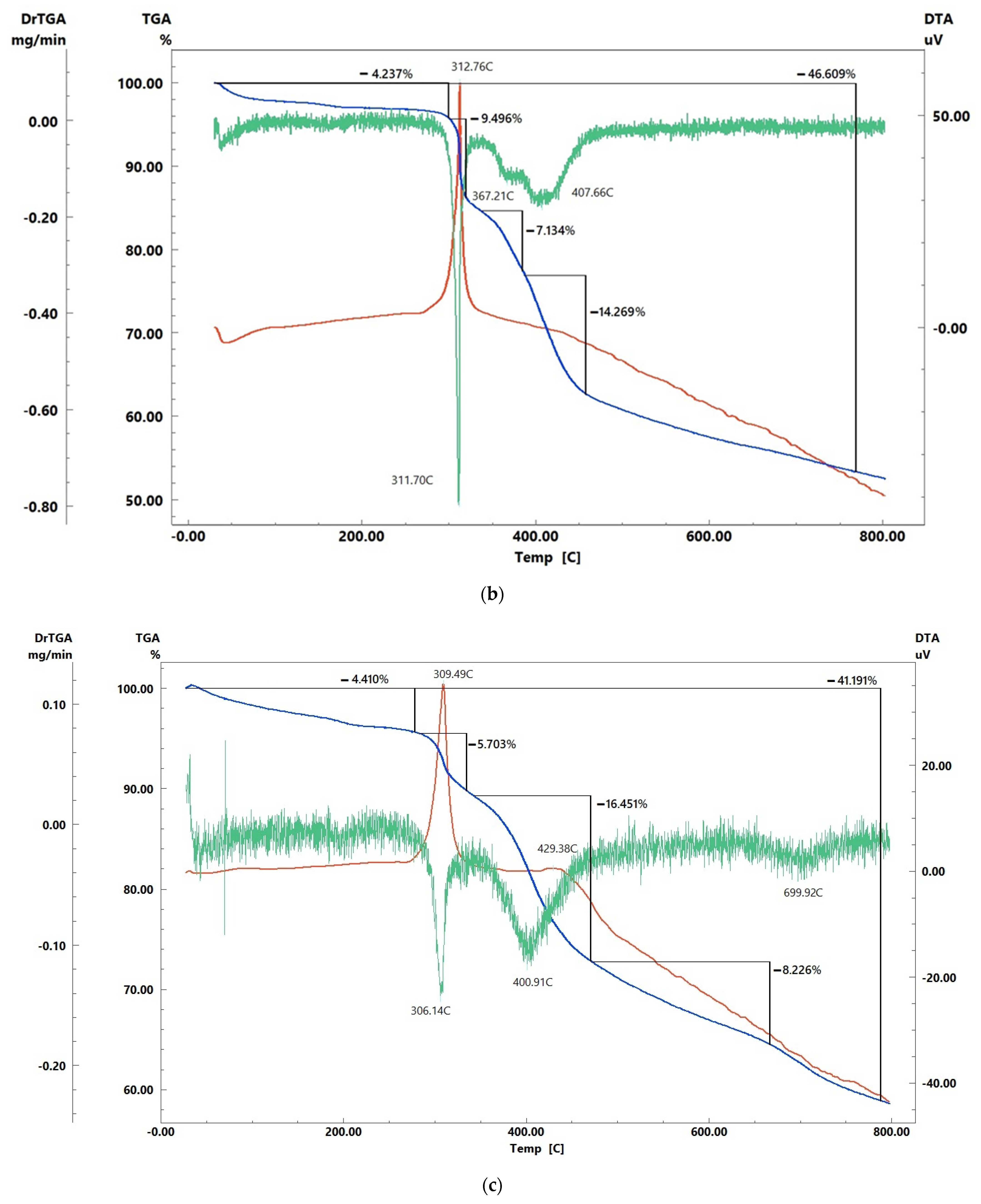
3.3. Fe(OH)3 Washout from the Sorbent
3.4. Determining the Parameters of Sorption of Phosphorus and Beryllium under Static Conditions
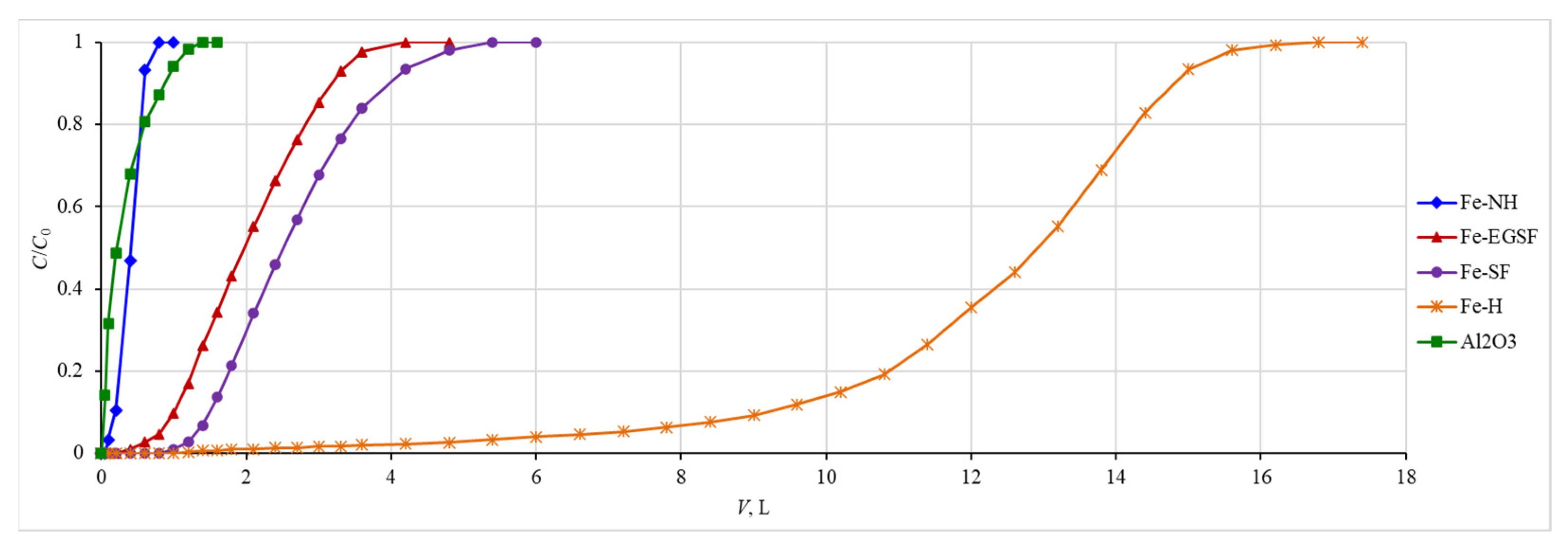
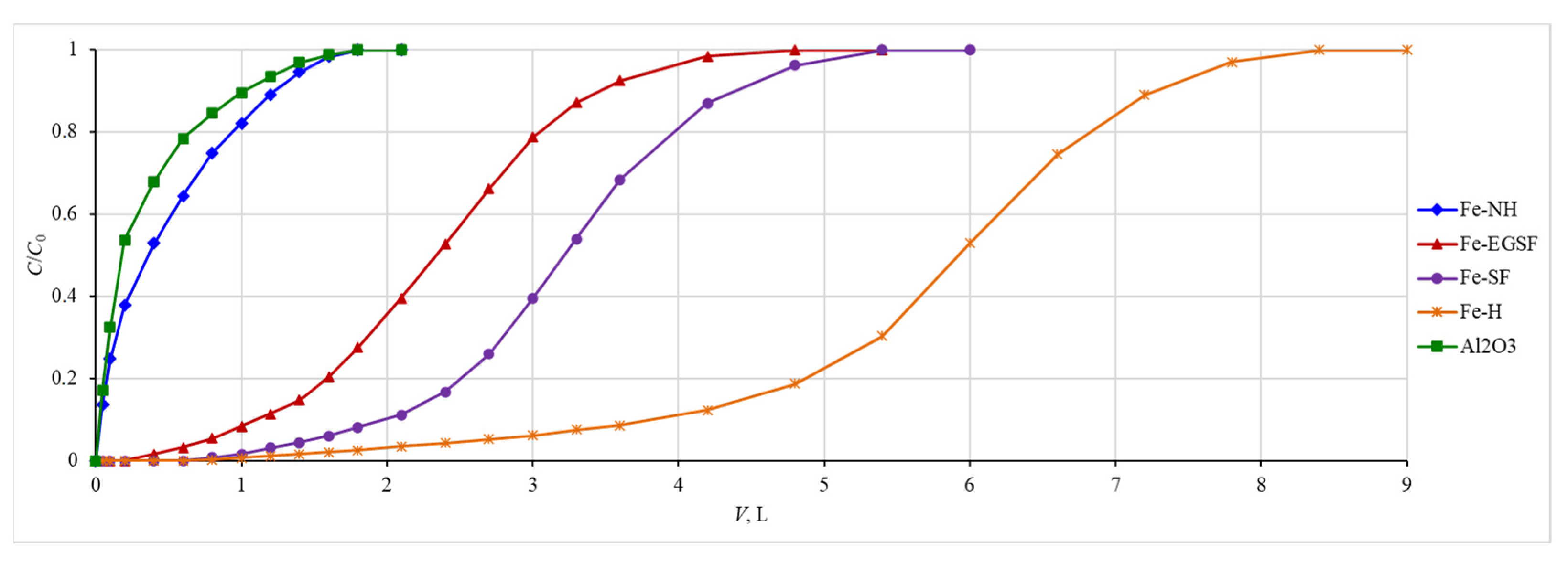
3.5. Sorption Curves for Stable Isotopes of Phosphorus and Beryllium under Laboratory Conditions
3.6. Sorption of Radionuclides from Large-Volume Samples
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Krishnaswami, S.; Lal, D.; Somayajulu, B.L.K.; Dixon, F.S.; Stonecipher, S.A.; Craig, H. Silicon, radium, thorium, and lead in seawater: In-situ extraction by synthetic fibre. Earth Planet. Sci. Lett. 1972, 16, 84–90. [Google Scholar] [CrossRef]
- Somayajulu, B.L.K.; Rengarajan, R.; Lal, D.; Weiss, R.F.; Craig, H. GEOSECS Atlantic 32Si profiles. Earth Planet. Sci. Lett. 1987, 85, 329–342. [Google Scholar] [CrossRef] [Green Version]
- Lal, D.; Chung, Y.; Platt, T.; Lee, T. Twin cosmogenic radiotracer studies of phosphorus recycling and chemical fluxes in the upper ocean. Limnol. Oceanogr. 1988, 33 Pt 2, 1559–1567. [Google Scholar] [CrossRef]
- Lal, D.; Lee, T. Cosmogenic 32P and 33P used as tracers to study phosphorus recycling in the upper ocean. Nature 1988, 333, 752–754. [Google Scholar] [CrossRef]
- Lee, T.; Lal, D. Techniques for extraction of dissolved inorganic and organic phosphorus from large volumes of sea water. Anal. Chim. Acta 1992, 260, 113–121. [Google Scholar] [CrossRef]
- Lee, T.; Barg, E.; Lal, D. Studies of vertical mixing in the Southern California Bight with cosmogenic radionuclides 32P and 7Be. Limnol. Oceanogr. 1991, 36, 1044–1052. [Google Scholar] [CrossRef]
- Haskell, W.Z.; Kadko, D.; Hammond, D.E.; Knapp, A.N.; Prokopenko, M.G.; Berelson, W.M.; Capone, D.G. Upwelling velocity and eddy diffusivity from 7Be measurements used to compare vertical nutrient flux to export POC flux in the Eastern Tropical South Pacific. Mar. Chem. 2015, 168, 140–150. [Google Scholar] [CrossRef]
- Kremenchutskii, D.A.; Batrakov, G.F.; Dovhyi, I.I.; Sapozhnikov, Y.A. The role of suspended matter in transformation of beryllium-7 (7Be) field in the Black Sea surface layer. Mar. Syst. 2021, 217, 10351. [Google Scholar] [CrossRef]
- Nakanishi, T.; Kusakabe, M.; Aono, T.; Yamada, M. Simultaneous measurements of cosmogenic radionuclides 32P, 33P and 7Be in dissolved and particulate forms in the upper ocean. J. Radioanal. Nucl. Chem. 2009, 279, 769–776. [Google Scholar] [CrossRef]
- Benitez-Nelson, C.R.; Buesseler, K.O. Measurement of Cosmogenic 32P and 33P Activities in Rainwater and Seawater. Anal. Chem. 1998, 70, 64–72. [Google Scholar] [CrossRef]
- Clevenger, S.J.; Benitez-Nelson, C.R.; Drysdale, J.; Pike, S.; Puigcorbé, V.; Buesseler, K.O. Review of the analysis of 234Th in small volume (2–4 L) seawater samples: Improvements and recommendations. J. Radioanal. Nucl. Chem. 2021, 329, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Roca-Martí, M.; Puigcorbé, V.; Castrillejo, M.; Casacuberta, N.; Garcia-Orellana, J.; Cochran, J.K.; Masqué, P. Quantifying 210Po/210Pb Disequilibrium in Seawater: A Comparison of Two Precipitation Methods with Differing Results. Front. Mar. Sci. 2021, 8, 70. [Google Scholar] [CrossRef]
- Silker, W.B.; Robertson, D.E.; Rieck, H.G., Jr.; Perkins, R.W.; Prospero, J.M. Beryllium-7 in Ocean Water. Science 1968, 161, 879–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bezhin, N.A.; Dovhyi, I.I.; Tananaev, I.G. Sorption methods in marine radiochemistry. Russ. Chem. Rev. 2021, 90, 1544–1565. [Google Scholar] [CrossRef]
- Colley, S.; Thomson, J. Particulate/solution analysis of 226Ra, 230Th and 210Pb in sea water sampled by in-situ large volume filtration and sorption by manganese oxyhydroxide. Sci. Total Environ. 1994, 155, 273–283. [Google Scholar] [CrossRef]
- Hartman, M.C.; Buesseler, K.O. Adsorbers for In-Situ Collection and At-Sea Gamma Analysis of Dissolved Thorium-234 in Seawater; Technical Report; Woods Hole Oceano-Graphic Institution: Falmouth, MA, USA, 1994. [Google Scholar]
- LLC “Ural Process Engineering Company (UPEC)”. Trade Mark RU 377834. 2009. Available online: https://www1.fips.ru/fips_servl/fips_servlet?DB=RUTM&DocNumber=377834 (accessed on 1 May 2022). (In Russian).
- CMER. Extraction of 7Be from Seawater Using Fe-Fibers. Available online: https://cmer.whoi.edu/recipe/extraction-of-7be-from-seawater-using-fe-fibers/ (accessed on 28 May 2022). (In Russian).
- Harvey, D. Modern Analytical Chemistry; McGraw-Hill Higher Education: New York, NY, USA, 2000. [Google Scholar]
- Bezhin, N.A.; Dovhyi, I.I.; Milyutin, V.V.; Kaptakov, V.O.; Kozlitin, E.A.; Egorin, A.M.; Tokar’, E.A.; Tananaev, I.G. Study of sorbents for analysis of radiocesium in seawater samples by one-column method. J. Radioanal. Nucl. Chem. 2021, 327, 1095–1103. [Google Scholar] [CrossRef]
- Karamanev, D.G.; Nikolov, L.N.; Mamatarkova, V. Rapid simultaneous quantitative determination of ferric and ferrous ions in drainage waters and similar solutions. Miner. Eng. 2002, 15, 341–346. [Google Scholar] [CrossRef]
- Remez, V.P.; Zelenin, V.I.; Smirnov, A.L.; Raspopin, S.P.; Matern, A.I.; Morzherin, Y.Y. Cellulose-inorganic sorbents in radiochemical analysis I. Promising sorbents for radiochemical analysis. Sorpt. Chromatogr. Process. 2009, 9, 627–632. (In Russian) [Google Scholar]
- Xia, X.; Dong, F.; Nie, X.; Pan, N.; Liu, C.; Ding, C.; Wang, J.; Cheng, W.; He, H.; Sun, S.; et al. Efficient adsorption of U(VI) using in low-level radioactive wastewater containing organic matter by amino groups modified polyacrylonitrile fibers. J. Radioanal. Nucl. Chem. 2022, 331, 921–936. [Google Scholar] [CrossRef]
- Bondar, Y.; Olkhovyk, Y.; Kuzenko, S. Nanocomposite adsorbent based on polyacrylonitrile fibers for rapid and selective removal of Cs radionuclides. J. Radioanal. Nucl. Chem. 2021, 330, 1221–1231. [Google Scholar] [CrossRef]
- Moore, W.S.; Reid, D.F. Extraction of radium from natural waters using manganese-impregnated acrylic fibers. J. Geophys. Res. 1973, 78, 8880–8886. [Google Scholar] [CrossRef]
- Karki, H.P.; Kafle, L.; Ojha, D.P.; Song, J.H.; Kim, H.J. Cellulose/polyacrylonitrile electrospun composite fiber for effective separation of the surfactant-free oil-inwater mixture under a versatile condition. Sep. Purif. Technol. 2019, 210, 913–919. [Google Scholar] [CrossRef]
- Waser, N.A.; Fleer, A.P.; Hammar, T.R.; Buesseler, K.O.; Bacon, M.P. Determination of natural 32P and 33P in rainwater, marine particles and plankton by low-level beta counting. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrom. Detect. Assoc. Equip. 1994, 338, 560–567. [Google Scholar] [CrossRef]
- Samodurov, A.S.; Chukharev, A.M.; Kazakov, D.A. Basic Regularities of Vertical Turbulent Exchange in the Mixed and Stratified Layers of the Black Sea. Phys. Oceanogr. 2021, 28, 405–422. [Google Scholar] [CrossRef]
- Orekhova, N.A. Nutrients Dynamics in the Surface Waters of the Black Sea. Phys. Oceanogr. 2021, 28, 660–676. [Google Scholar] [CrossRef]




| Sorbent | Recoverable Radionuclide | Sorbent Volume in a Column, L | Passed Volume, L | Flow Rate, mL/min | Sorption Efficiency, % | Reference |
|---|---|---|---|---|---|---|
| Acrylate fiber impregnated with Fe(OH)3 (University of California, Berkeley, CA, USA) | 32P, 33P | 1 | 10,000 | 1600 | 90.0 | [3] |
| 3300 | 88.0 | |||||
| 8300 | 90.0 | |||||
| 16,500 | 85.0 | |||||
| Fe(OH)3-form Dowex 20–50-mesh (University of California, Berkeley, CA, USA) | 2500 | 30.0 | ||||
| 4100 | 28.0 | |||||
| 7500 | 20.0 | |||||
| 14,100 | 3.00 | |||||
| 28,300 | 2.00 | |||||
| Fe(OH)3-form Dowex 50–100-mesh (University of California, Berkeley, CA, USA) | 800 | 50.0 | ||||
| 1600 | 35.0 | |||||
| 3300 | 30.0 | |||||
| 5800 | 28.0 | |||||
| 9100 | 28.0 | |||||
| LV-FiCS based on cartridges impregnated with Fe(OH)3 (National Institute of Radiological Sciences, Chiba, Japan) | 7Be | – 1 | 5100 | 29,400 | 85.0 ± 4.6 | [9] |
| 5500 | 22,800 | 86.4 ± 3.9 | ||||
| 4400 | 18,300 | 91.0 ± 3.8 | ||||
| 5000 | 20,900 | 90.6 ± 3.2 | ||||
| 5100 | 21,200 | 87.2 ± 5.5 | ||||
| 5500 | 22,800 | 84.2 ± 3.5 | ||||
| 4700 | 19,500 | 87.3 ± 3.9 | ||||
| 32P | – 1 | 5100 | 29,400 | 65.9 ± 1.2 | ||
| 5500 | 22,800 | 66.5 ± 0.5 | ||||
| 4400 | 18,300 | 77.9 ± 1.0 | ||||
| 5000 | 20,900 | 76.5 ± 0.8 | ||||
| 5100 | 21,200 | 72.8 ± 0.9 | ||||
| 5500 | 22,800 | 66.7 ± 0.6 | ||||
| 4700 | 19,500 | 78.9 ± 0.8 | ||||
| Cartridges impregnated with Fe(OH)3 (Woods Hole Oceanographic Institution, Massachusetts, MA, USA) | 32P, 33P | 1.4 | 1000 | 4000–6000 | 100 | [10] |
| 1500 | 99.0 | |||||
| 3300 | 90.0 | |||||
| 3900 | 82.0 | |||||
| 4000 | 70.0 | |||||
| Acrylate fiber impregnated with Fe(OH)3 (Woods Hole Oceanographic Institution, Massachusetts, MA, USA) | 250 | 97.0 | ||||
| 400 | 95.0 | |||||
| 550 | 92.0 | |||||
| 650 | 89.0 | |||||
| 750 | 86.0 | |||||
| 1000 | 81.0 | |||||
| 1200 | 77.0 | |||||
| Cartridges impregnated with manganese oxyhydroxide (Institute of Oceanographic Sciences, Southampton, UK) | 210Pb | – 1 | 950–2000 | – 1 | 96.5 ± 2.5 | [15] |
| 234Th | 91.0 ± 6.0 | |||||
| Hytrex II cartridge impregnated with MnO2 (Osmonics Inc., Minnesota, MS, USA) | 234Th | – 1 | 800 | 8000 | 86.0 | [16] |
| 10,000 | 39.0 | |||||
| 15,000 | 40.0 | |||||
| Beta Pure cartridge impregnated with MnO2 (CUNO Inc., Connecticut, CT, USA) | 8000 | 70.0 |
| Element | PAN | Fe-NH | Fe-SF | Fe-H |
|---|---|---|---|---|
| C, % | 69.04 | 61.53 | 65.66 | 42.50 |
| O, % | - | 22.52 | 20.63 | 26.28 |
| N, % | 27.96 | 8.51 | 5.82 | 12.40 |
| Fe, % | - | 7.44 | 7.89 | 18.82 |
| Element | Sorbent | Kd, mL/g | R, % | qe, mg/g |
|---|---|---|---|---|
| P | Fe-NH | 1100 | 91.5 | 0.0094 |
| Fe-EGSF | 1700 | 94.4 | 0.0085 | |
| Fe-SF | 4100 | 97.6 | 0.0088 | |
| Fe-H | 61,500 | 99.8 | 0.0095 | |
| Al2O3 | 546 | 84.5 | 0.0087 | |
| Be | Fe-NH | 516 | 83.8 | 0.0218 |
| Fe-EGSF | 653 | 86.7 | 0.0217 | |
| Fe-SF | 1140 | 91.9 | 0.0238 | |
| Fe-H | 735 | 88.0 | 0.0228 | |
| Al2O3 | 825 | 89.2 | 0.0232 |
| Element | Sorbent | DEC, mg/g | TDEC, mg/g |
|---|---|---|---|
| P | Fe-NH | 0.0034 | 0.0174 |
| Fe-EGSF | 0.0121 | 0.0754 | |
| Fe-SF | 0.0301 | 0.0924 | |
| Fe-H | 0.0375 | 0.3940 | |
| Al2O3 | 0.0017 | 0.0162 | |
| Be | Fe-NH | 0.0043 | 0.0545 |
| Fe-EGSF | 0.0345 | 0.2395 | |
| Fe-SF | 0.0690 | 0.3112 | |
| Fe-H | 0.0676 | 0.6563 | |
| Al2O3 | 0.0035 | 0.0435 |
| Radionuclide | Fe-SF | Fe-H |
|---|---|---|
| Sorption Efficiency, % | Sorption Efficiency, % | |
| 7Be | 45.2 | 92.6 |
| 210Pb | 50.3 | 91.5 |
| 234Th | 100 | 84.1 |
| 32,33P | 56.6 | 92.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bezhin, N.A.; Frolova, M.A.; Dovhyi, I.I.; Kozlovskaia, O.N.; Slizchenko, E.V.; Shibetskaia, I.G.; Khlystov, V.A.; Tokar’, E.A.; Tananaev, I.G. The Sorbents Based on Acrylic Fiber Impregnated by Iron Hydroxide (III): Production Methods, Properties, Application in Oceanographic Research. Water 2022, 14, 2303. https://doi.org/10.3390/w14152303
Bezhin NA, Frolova MA, Dovhyi II, Kozlovskaia ON, Slizchenko EV, Shibetskaia IG, Khlystov VA, Tokar’ EA, Tananaev IG. The Sorbents Based on Acrylic Fiber Impregnated by Iron Hydroxide (III): Production Methods, Properties, Application in Oceanographic Research. Water. 2022; 14(15):2303. https://doi.org/10.3390/w14152303
Chicago/Turabian StyleBezhin, Nikolay A., Mariya A. Frolova, Illarion I. Dovhyi, Ol’ga N. Kozlovskaia, Evgenii V. Slizchenko, Iuliia G. Shibetskaia, Vasiliy A. Khlystov, Eduard A. Tokar’, and Ivan G. Tananaev. 2022. "The Sorbents Based on Acrylic Fiber Impregnated by Iron Hydroxide (III): Production Methods, Properties, Application in Oceanographic Research" Water 14, no. 15: 2303. https://doi.org/10.3390/w14152303
APA StyleBezhin, N. A., Frolova, M. A., Dovhyi, I. I., Kozlovskaia, O. N., Slizchenko, E. V., Shibetskaia, I. G., Khlystov, V. A., Tokar’, E. A., & Tananaev, I. G. (2022). The Sorbents Based on Acrylic Fiber Impregnated by Iron Hydroxide (III): Production Methods, Properties, Application in Oceanographic Research. Water, 14(15), 2303. https://doi.org/10.3390/w14152303








