Investigation of a Possible Relationship between Anthropogenic and Geogenic Water Contaminants and Birth Defects Occurrence in Rural Nebraska
Abstract
:1. Introduction
2. Materials and Methods
2.1. Case Definition, Study Population and Site, and Data Sources
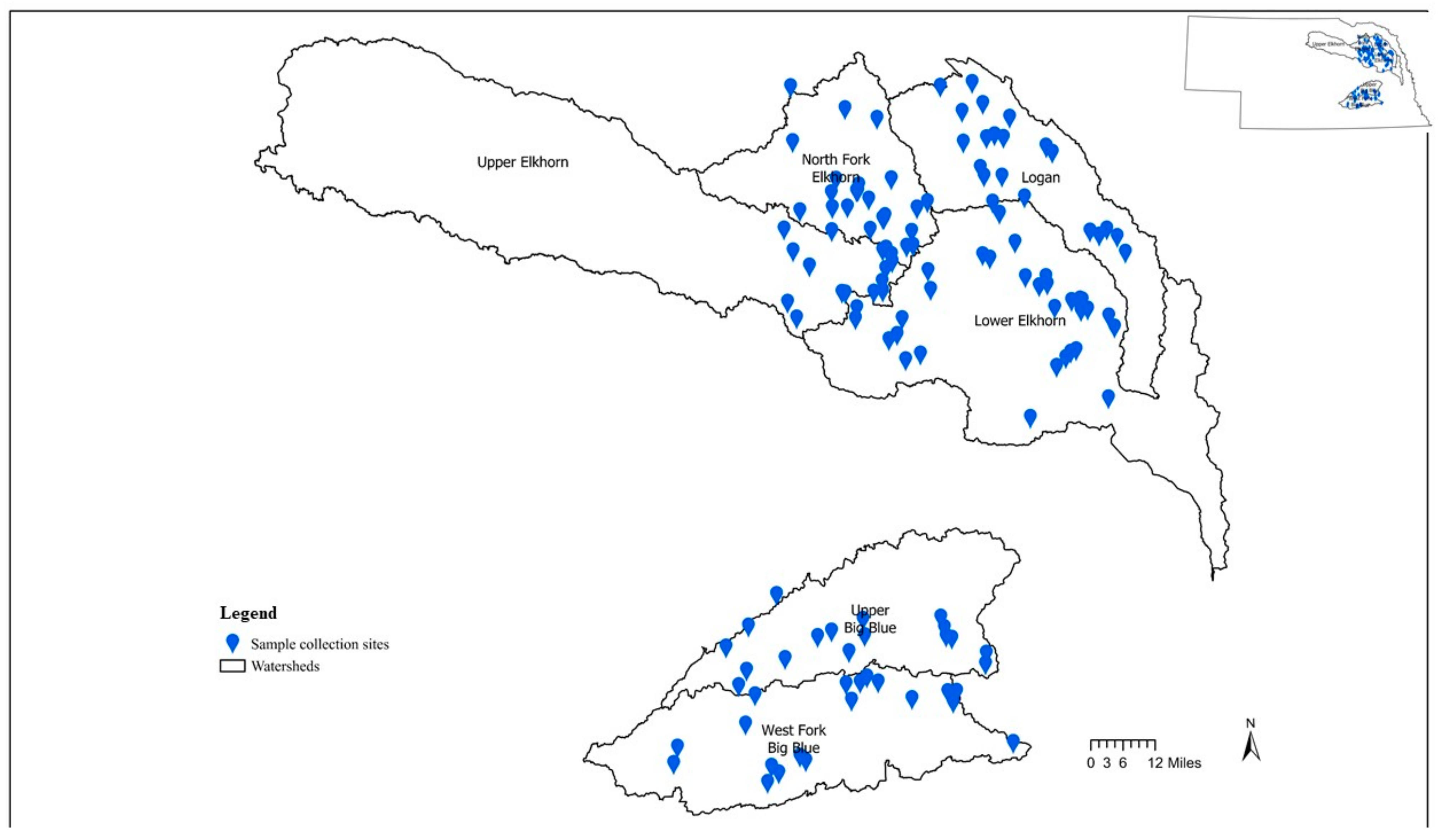
2.1.1. Study Sites
2.1.2. Birth Defect Data
2.1.3. Water Quality Data
2.1.4. Geographic Information System (GIS) Data
2.2. Data Analysis
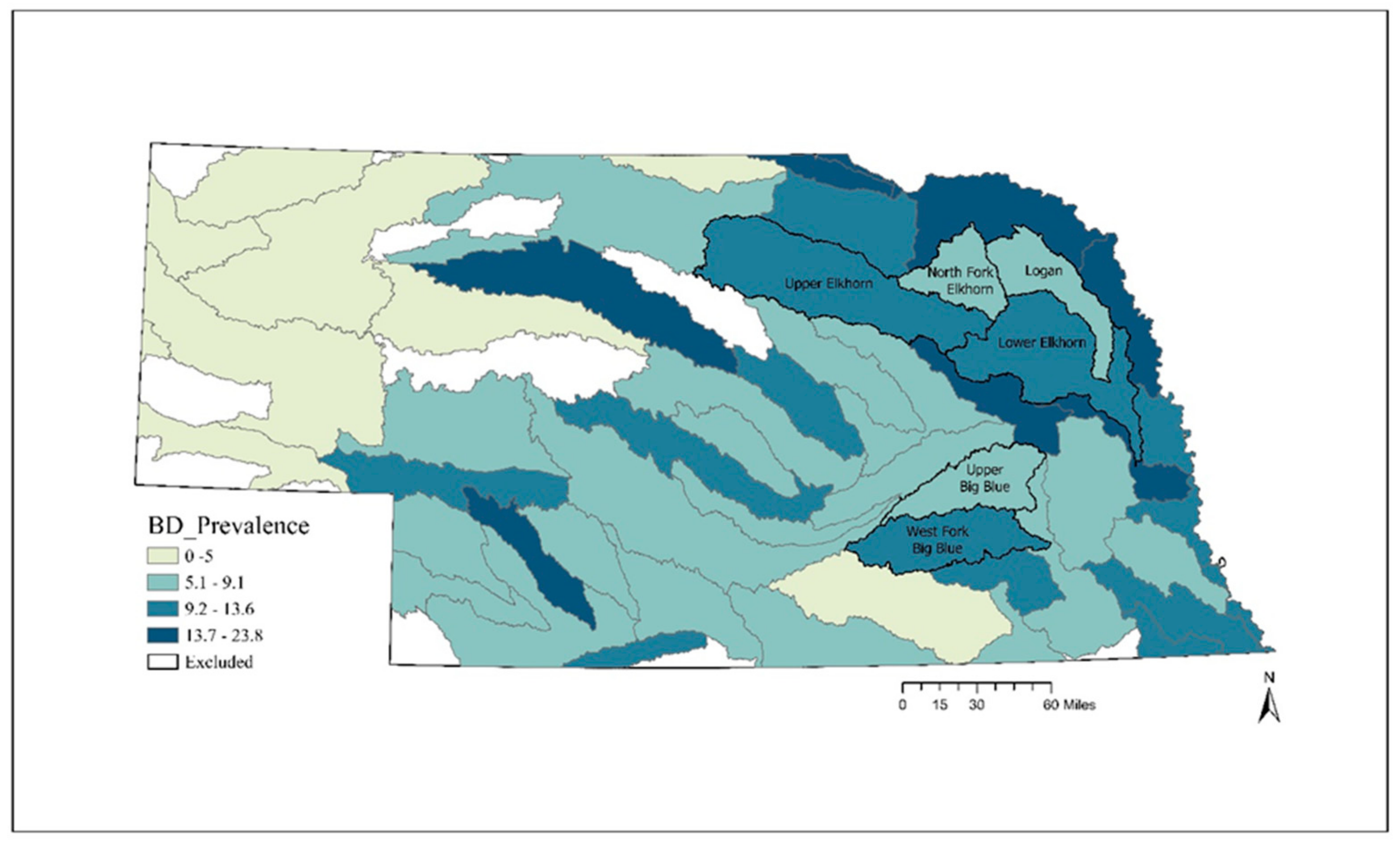
2.2.1. Birth Defect Prevalence
2.2.2. Analysis of the Association between Birth Defects and Water Quality Data
3. Results
3.1. Descriptive Statistics
3.1.1. Birth Defects
3.1.2. Groundwater Quality Data
3.1.3. Correlations between Variables
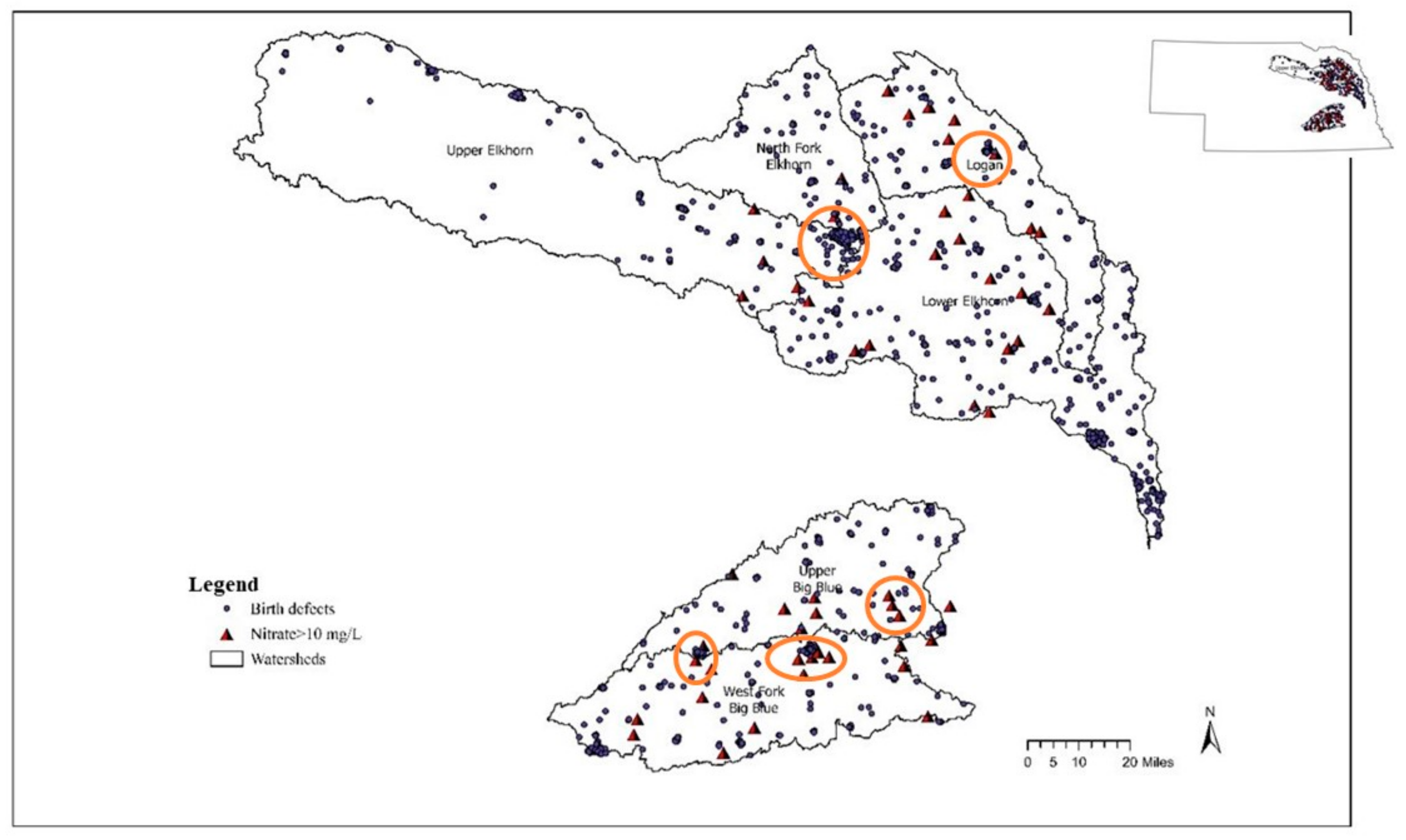
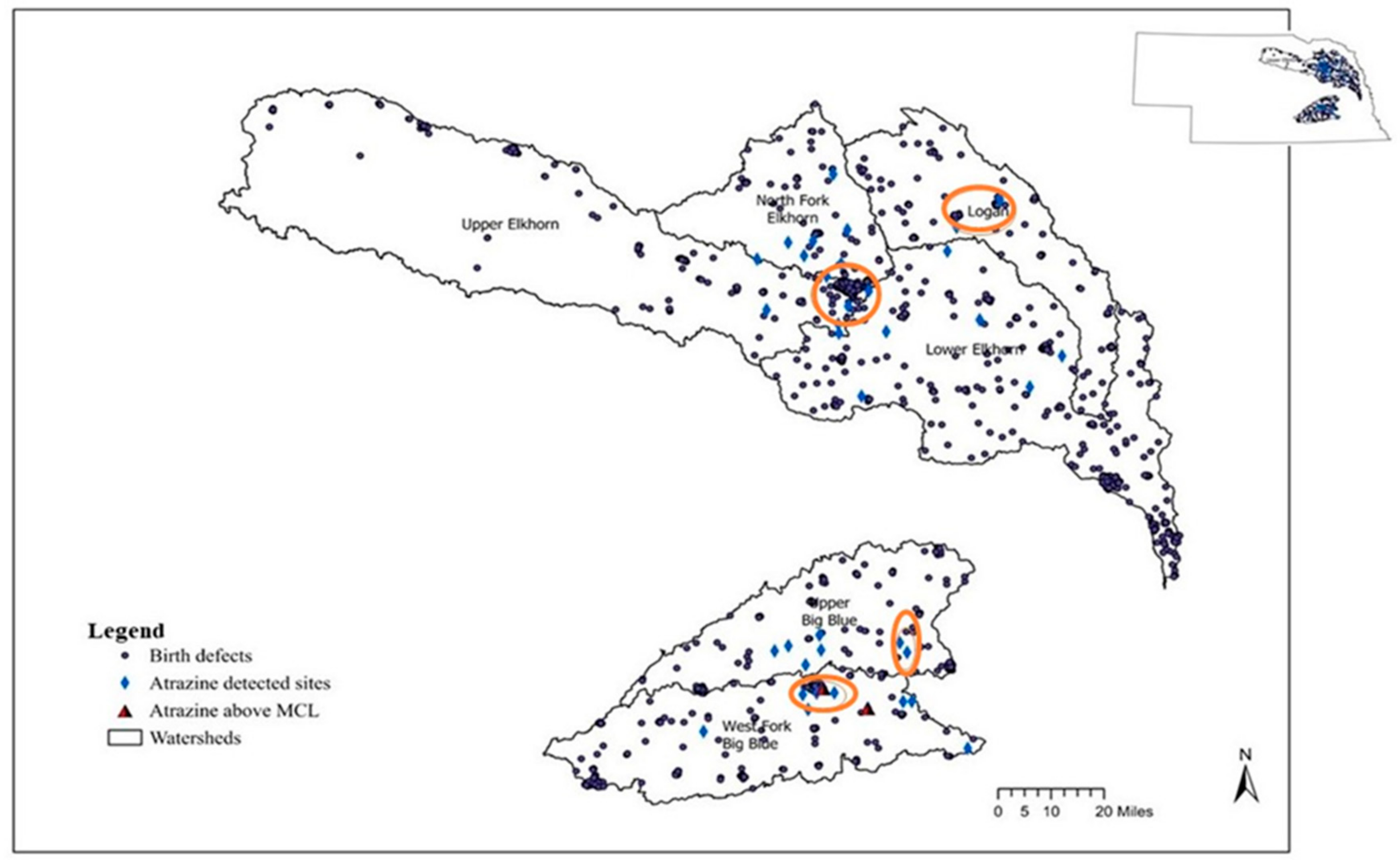
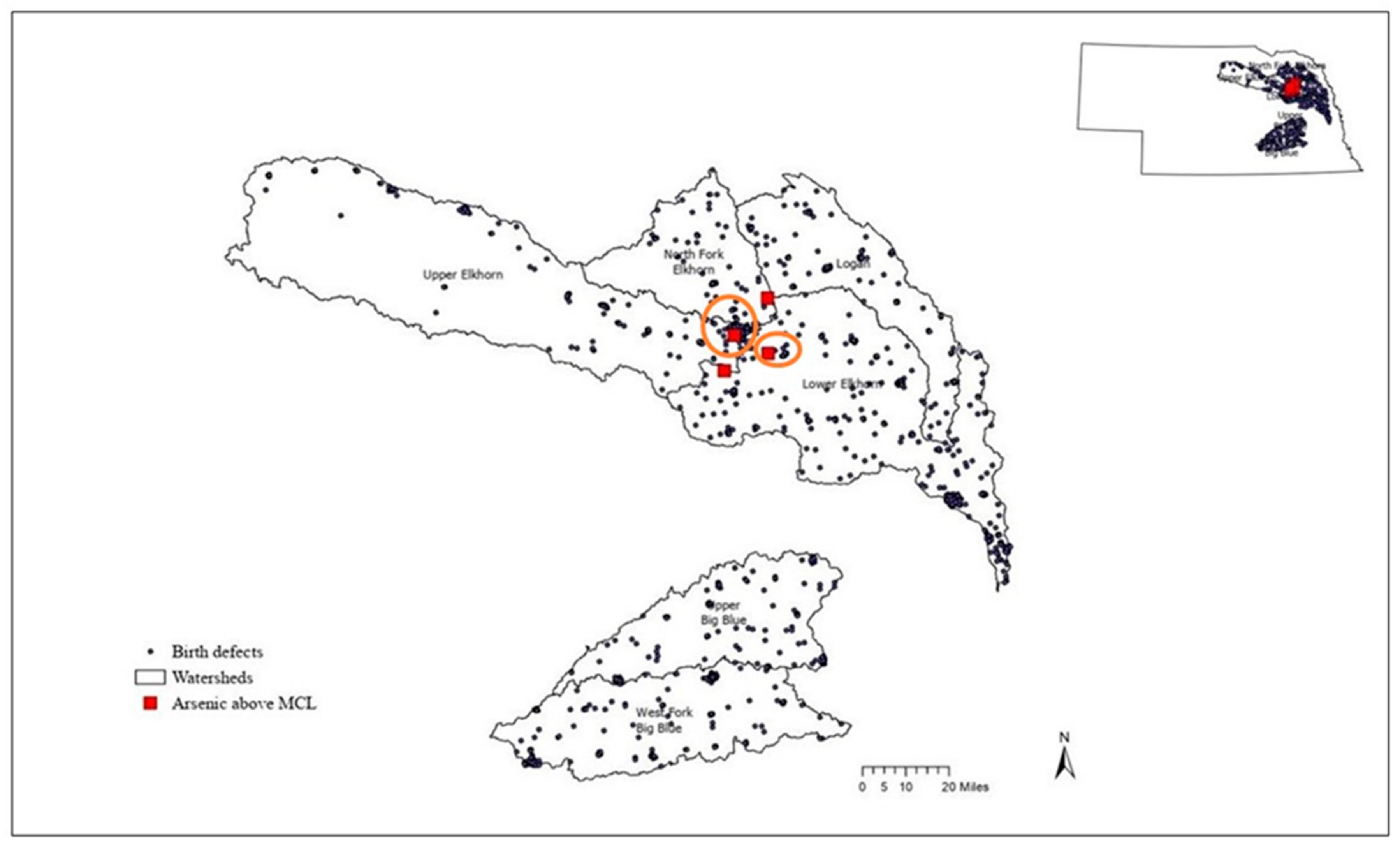
3.2. Geospatial Analysis
3.3. Inferential Statistics
3.3.1. Birth Defects and Nitrate
3.3.2. Birth Defects and Atrazine
3.3.3. Birth Defects and Uranium
3.3.4. Birth Defects and Arsenic
4. Discussion
Study Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Centers for Disease Control and Prevention. Update on Overall Prevalence of Major Birth Defects—Atlanta, Georgia, 1978–2005. MMWR Morb. Mortal. Wkly. Rep. 2008, 57, 1–5. [Google Scholar]
- World Health Organization. Birth Defects. 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/birth-defects (accessed on 13 May 2022).
- Nebraska Department of Health and Human Services. Nebraska 2015 Vital Statistics Report. 2015. Available online: https://dhhs.ne.gov/Vital%20Statistics%20Reports/Table%2028%202015.pdf#search=birth%20defects (accessed on 13 May 2022).
- Mathews, T.; MacDorman, M.E.; Thoma, M.E. Infant Mortality Statistics from the 2013 Period Linked Birth/Infant Death Data Set. 2015. Available online: https://stacks.cdc.gov/view/cdc/32752 (accessed on 13 May 2022).
- Russo, C.A.; Elixhauser, A. Hospitalizations for Birth Defects, 2004: Statistical Brief# 24. Agency for Healthcare Research and Quality. 2007. Available online: https://www.ncbi.nlm.nih.gov/books/NBK63495/?report=printable (accessed on 13 May 2022).
- Feldkamp, M.L.; Carey, J.C.; Byrne, J.L.; Krikov, S.; Botto, L.D. Etiology and clinical presentation of birth defects: Population based study. BMJ 2017, 357, j2249. [Google Scholar] [CrossRef] [Green Version]
- Centers for Disease Control and Prevention. What Are Birth Defects? 2021. Available online: https://www.cdc.gov/ncbddd/birthdefects/facts.html (accessed on 13 May 2022).
- Harris, B.S.; Bishop, K.C.; Kemeny, H.R.; Walker, J.S.; Rhee, E.; Kuller, J.A. Risk factors for birth defects. Obstet. Gynecol. Surv. 2017, 72, 123–135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agopian, A.; Lupo, P.J.; Canfield, M.A.; Langlois, P.H. Case–C ontrol Study of Maternal Residential Atrazine Exposure and Male Genital Malformations. Am. J. Med. Genet. Part A 2013, 161, 977–982. [Google Scholar] [CrossRef] [PubMed]
- Waller, S.A.; Paul, K.; Peterson, S.E.; Hitti, J.E. Agricultural-related chemical exposures, season of conception, and risk of gastroschisis in Washington State. Am. J. Obs. Gynecol. 2010, 202, e241–e246. [Google Scholar] [CrossRef] [PubMed]
- Holtby, C.E.; Guernsey, J.R.; Allen, A.C.; Vanleeuwen, J.A.; Allen, V.M.; Gordon, R.J. A population-based case-control study of drinking-water nitrate and congenital anomalies using Geographic Information Systems (GIS) to develop individual-level exposure estimates. Int. J. Environ. Res. Public Health 2014, 11, 1803–1823. [Google Scholar] [CrossRef] [Green Version]
- Brender, J.D.; Weyer, P.J. Agricultural Compounds in Water and Birth Defects. Curr. Environ. Health Rep. 2016, 3, 144–152. [Google Scholar] [CrossRef]
- Brender, J.D.; Weyer, P.J.; Romitti, P.A.; Mohanty, B.P.; Shinde, M.U.; Vuong, A.M.; Sharkey, J.R.; Dwivedi, D.; Horel, S.A.; Kantamneni, J.; et al. Prenatal nitrate intake from drinking water and selected birth defects in offspring of participants in the national birth defects prevention study. Environ. Health Perspect. 2013, 121, 1083–1089. [Google Scholar] [CrossRef] [Green Version]
- Blaisdell, J.; Turyk, M.E.; Almberg, K.S.; Jones, R.M.; Stayner, L.T. Prenatal exposure to nitrate in drinking water and the risk of congenital anomalies. Environ. Res. 2019, 176, 108553. [Google Scholar] [CrossRef]
- Ward, M.H.; Jones, R.R.; Brender, J.D.; de Kok, T.M.; Weyer, P.J.; Nolan, B.T.; Villanueva, C.M.; van Breda, S.G. Drinking Water Nitrate and Human Health: An Updated Review. Int. J. Environ. Res. Public Health 2018, 15, 1557. [Google Scholar] [CrossRef] [Green Version]
- Nebraska Department of Agriculture and USDA NASS, Nebraska Field Office. Nebraska Agriculture Fact Card. 2020. Available online: https://nda.nebraska.gov/facts.pdf (accessed on 13 May 2022).
- Nebraska Department of Agriculture. (ND); Nebraska Agriculture. Available online: https://nda.nebraska.gov/publications/ne_ag_facts_brochure.pdf (accessed on 14 May 2022).
- USDA ERS United States Department of Agriculture—Economic Research Service. Fertilizer Use and Price. 2019. Available online: https://www.ers.usda.gov/data-products/fertilizer-use-and-price/ (accessed on 14 May 2022).
- Wieben, C.M. Estimated Annual Agricultural Pesticide Use by Major Crop or Crop Group for States of the Conterminous United States, 1992−2017, Version 2.0, May 2020; data release. US Geol. Survey 2019, 10, P9HHG3CT. [Google Scholar] [CrossRef]
- Lockhart, K.M.; King, A.M.; Harter, T. Identifying sources of groundwater nitrate contamination in a large alluvial groundwater basin with highly diversified intensive agricultural production. J. Contam. Hydrol. 2013, 151, 140–154. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Drinking Water Standards and Regulations. 2020. Available online: https://www.cdc.gov/healthywater/drinking/public/regulations.html (accessed on 14 May 2022).
- United States Environmental Protection Agency (EPA). Ground Water and Drinking Water. In National Primary Drinking Water Regulations. 2020. Available online: https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations#Inorganic (accessed on 14 May 2022).
- Liesch, T.; Hinrichsen, S.; Goldscheider, N. Uranium in groundwater—Fertilizers versus geogenic sources. Sci. Total Environ. 2015, 536, 981–995. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.; Weber, K.A. Natural uranium contamination in major US aquifers linked to nitrate. Environ. Sci. Technol. Lett. 2015, 2, 215–220. [Google Scholar] [CrossRef] [Green Version]
- Westrop, J.P.; Snow, D.D.; Weber, K.A. Mobilization of Naturally Occurring Uranium in Groundwater Under Intensely Managed Farmland. In Food, Energy, and Water Nexus; Springer: Cham, Germany, 2022; pp. 215–231. [Google Scholar]
- Kingsbury, J.A. Groundwater Quality in Selected Stream Valley Aquifers, Western United States (No. 2021-3011). US Geological Survey. 2021. Available online: https://pubs.usgs.gov/fs/2021/3011/fs20213011.pdf (accessed on 14 May 2022).
- Gholkar, P. Spatial Distribution of Orofacial Cleft Birth Defects and Uranium-Radium Concentrations in Tap Water in Atascosa, Bee, Brooks, Calhoun, Duval, Goliad, Hidalgo, Jim Hogg, Jim Wells, Karnes, Kleberg, Live Oak, McMullen, Nueces, San Patricio, Refugio, Starr, Victoria, Webb, and Zavala Counties, Texas. The University of Texas School of Public Health. 2011. Available online: https://www.proquest.com/openview/64c46dc6681ac94b50dd85017697733d/1?pq-origsite=gscholar&cbl=18750 (accessed on 14 May 2022).
- Shrivastava, B.K. Elevated uranium and toxic elements concentration in groundwater in Punjab state of India: Extent of the problem and risk due to consumption of unsafe drinking water. Water Qual. Expo. Health 2015, 7, 407–421. [Google Scholar] [CrossRef]
- Yin, S.; Tian, T.; Wang, C.; Wang, D.; Pi, X.; Liu, M.; Jin, L.; Liu, J.; Wang, L.; Li, Z.; et al. Prenatal uranium exposure and risk for fetal neural tube defects: A case-control study in women living in a rural area of northern China. J. Hazard. Mater. 2022, 424, 127466. [Google Scholar] [CrossRef] [PubMed]
- Kwok, R.K.; Kaufmann, R.B.; Jakariya, M. Arsenic in drinking-water and reproductive health outcomes: A study of participants in the Bangladesh Integrated Nutrition Programme. J. Health Popul. Nutr. 2006, 24, 190–205. [Google Scholar]
- Mazumdar, M.; Valeri, L.; Rodrigues, E.G.; Hasan, O.S.I.; Hamid, R.; Paul, L.; Selhub, J.; Silva, F.; Mostofa, G.; Quamruzzaman, Q.; et al. Polymorphisms in maternal folate pathway genes interact with arsenic in drinking water to influence risk of myelomeningocele. Birth Defects Res. Part A Clin. Mol. Teratol. 2015, 103, 754–762. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rudnai, T.; Sándor, J.; Kádár, M.; Borsányi, M.; Béres, J.; Métneki, J.; Maráczi, G.; Rudnai, P. Arsenic in drinking water and congenital heart anomalies in Hungary. Int. J. Hyg. Environ. Health 2014, 217, 813–818. [Google Scholar] [CrossRef]
- Corley, B.; Bartelt-Hunt, S.; Rogan, E.; Coulter, D.; Sparks, J.; Baccaglini, L.; Howell, M.; Liaquat, S.; Commack, R.; Kolok, A.S. Using watershed boundaries to map adverse health outcomes: Examples from Nebraska, USA. Environ. Health Insights 2018, 12, 1178630217751906. [Google Scholar] [CrossRef]
- North Platte Natural Resource District. Protecting Lives. Protecting Property. Protecting the Future. 2022. Available online: https://www.npnrd.org/ (accessed on 14 May 2022).
- Environmental Systems Research Institute (ESRI). Geocode Addresses (Geocoding). 2021. Available online: https://pro.arcgis.com/en/pro-app/latest/tool-reference/geocoding/geocode-addresses.htm (accessed on 14 May 2022).
- MODERNWATER. Rapid EssayR Atrazine. 2013. Available online: https://www.modernwater.com/pdf/MW_Factsheet_Rapid-Assay_Atrazine.pdf (accessed on 14 May 2022).
- Nebraskamap.gov. HUC 8. 2020. Available online: https://www.nebraskamap.gov/datasets/nebraska::huc-8/about (accessed on 14 May 2022).
- National Birth Defect Prevention Network. NBDPN Guidelines for Conducting Birth Defects Surveillance. 2016. Available online: https://www.nbdpn.org/docs/Ch_8_Statistics6-04_2016DEC14.pdf (accessed on 14 May 2022).
- IBM Corp. IBM SPSS Statistics for Windows, Version 28.0. 2021. Available online: https://www.ibm.com/products/spss-statistics (accessed on 14 May 2022).
- Mai, C.T.; Isenburg, J.; Langlois, P.H.; Alverson, C.; Gilboa, S.M.; Rickard, R.; Canfield, M.A.; Anjohrin, S.B.; Lupo, P.J.; Jackson, D.R. Population-based birth defects data in the United States, 2008 to 2012: Presentation of state-specific data and descriptive brief on variability of prevalence. Birth Defects Res. Part A Clin. Mol. Teratol. 2015, 103, 972. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Centers for Disease Control and Prevention (CDC). Spontaneous abortions possibly related to ingestion of nitrate-contaminated well water—LaGrange County, Indiana, 1991–1994. MMWR Morb. Mortal. Wkly. Rep. 1996, 45, 569–572. [Google Scholar]
- Adeyemi, J.A.; da Cunha Martins-Junior, A.; Barbosa, F., Jr. Teratogenicity, genotoxicity and oxidative stress in zebrafish embryos (Danio rerio) co-exposed to arsenic and atrazine. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2015, 172, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, S.; Xin, Z.; Wang, Y.; Shou, K.; Gao, H.; Li, Y. The establishment of hypospadias rat model and embryoteratogenic test of Atrazine. Chin. J. Plast. Surg. 2007, 23, 340–343. [Google Scholar]
- Mattix, K.D.; Winchester, P.D.; Scherer, L.R. Incidence of abdominal wall defects is related to surface water atrazine and nitrate levels. J. Pediatr. Surg. 2007, 42, 947–949. [Google Scholar] [CrossRef]
- Agency for Toxic Substances and Disease Registry (ASTDR). What Are the Physiological Effects of Uranium Exposure? 2013. Available online: https://www.atsdr.cdc.gov/csem/uranium/physiological_effects.html (accessed on 14 May 2022).
- Bloom, M.S.; Fitzgerald, E.F.; Kim, K.; Neamtiu, I.; Gurzau, E.S. Spontaneous pregnancy loss in humans and exposure to arsenic in drinking water. Int. J. Hyg. Environ. Health 2010, 213, 401–413. [Google Scholar] [CrossRef]
- Kim, J.; Swartz, M.D.; Langlois, P.H.; Romitti, P.A.; Weyer, P.; Mitchell, L.E.; Luben, T.J.; Ramakrishnan, A.; Malik, S.; Lupo, P.J. Estimated maternal pesticide exposure from drinking water and heart defects in offspring. Int. J. Environ. Res. Public Health 2017, 14, 889. [Google Scholar] [CrossRef] [Green Version]
- Ouattara, B.S.; Puvvula, J.; Abadi, A.; Munde, S.; Kolok, A.S.; Bartelt-Hunt, S.; Bell, J.E.; Wichman, C.S.; Rogan, E. Geospatial Distribution of Age-Adjusted Incidence of the Three Major Types of Pediatric Cancers and Waterborne Agrichemicals in Nebraska. GeoHealth 2022, 6, e2021GH000419. [Google Scholar] [CrossRef]
| Watershed Name | Prevalence (per 100 Live Births) |
|---|---|
| Beaver | 9.96 |
| Big Nemaha | 10.16 |
| Big Papillion-Mosquito | 10.91 |
| Blackbird-Soldier | 18.67 |
| Cedar | 6.18 |
| Frenchman | 6.84 |
| Harlan County Reservoir | 5.59 |
| Horse | 4.86 |
| Keg-Weeping Water | 9.39 |
| Keya Paha | 3.45 |
| Lewis and Clark Lake | 20.47 |
| Little Nemaha | 8.62 |
| Logan | 7.71 |
| Loup | 5.24 |
| Lower Elkhorn | 10.19 |
| Lower Little Blue | 5.44 |
| Lower Lodgepole | 2.76 |
| Lower Middle Loup | 7.76 |
| Lower Niobrara | 13.61 |
| Lower North Loup | 9.52 |
| Lower North Platte | 7.68 |
| Lower Platte | 18.57 |
| Lower Platte-Shell | 15.32 |
| Lower South Platte | 9.31 |
| Medicine | 9.07 |
| Middle Big Blue | 8.07 |
| Middle Niobrara | 5.51 |
| Middle North Platte-Scotts Bluff | 4.57 |
| Middle Platte-Buffalo | 6.55 |
| Middle Platte-Prairie | 7.88 |
| Middle Republican | 6.68 |
| Mud | 5.31 |
| Niobrara Headwaters | 3.45 |
| North Fork Elkhorn | 6.87 |
| Ponca | 23.79 |
| Red Willow | 16.02 |
| Salt | 8.74 |
| South Fork Big Nemaha | 9.74 |
| South Loup | 10.11 |
| Stinking Water | 8.56 |
| Tarkio-Wolf | 13.53 |
| Turkey | 10.13 |
| Upper Big Blue | 7.32 |
| Upper Elkhorn | 12.37 |
| Upper Little Blue | 4.62 |
| Upper Middle Loup | 3.33 |
| Upper Niobrara | 4.61 |
| Upper North Loup | 19.50 |
| Upper Republican | 5.46 |
| Upper White | 3.97 |
| West Fork Big Blue | 13.15 |
| Wood | 7.34 |
| Nitrate (mg/L) MCL = 10 | Atrazine (µg/L) MCL = 3 | Uranium (µg/L) MCL = 30 | Arsenic (µg/L) MCL = 10 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Watershed | Samples (n) | Mean | Min | Max | Mean | Min | Max | Mean | Min | Max | Mean | Min | Max |
| Logan | 20 | 21.37 | 1.18 | 97.21 | 0.00 | 0.00 | 0.00 | 8.02 | 0.11 | 28.03 | 2.61 | 0.40 | 8.04 |
| Lower Elkhorn | 37 | 9.09 | 1.14 | 24.58 | 0.01 | 0.00 | 0.29 | 9.81 | 0.06 | 83.08 | 3.17 | 0.40 | 10.51 |
| North Fork Elkhorn | 21 | 8.38 | 0.56 | 39.67 | 0.04 | 0.00 | 0.76 | 3.92 | 0.00 | 8.32 | 4.73 | 0.47 | 14.08 |
| Upper Elkhorn | 17 | 6.94 | 1.36 | 16.47 | 0.09 | 0.00 | 0.63 | 5.99 | 0.04 | 34.24 | 4.84 | 0.11 | 19.84 |
| Upper Big Blue | 17 | 18.24 | 0.73 | 58.35 | 0.18 | 0.00 | 1.39 | 13.18 | 0.55 | 143.7 | 3.61 | 1.22 | 6.13 |
| West Fork Big Blue | 20 | 16.12 | 0.87 | 55.97 | 1.11 | 0.00 | 11.23 | 6.33 | 0.24 | 15.03 | 3.14 | 0.33 | 5.04 |
| Percent above MCL (%) | |||||
|---|---|---|---|---|---|
| Watershed | Samples (n) | Nitrate | Atrazine | Uranium | Arsenic |
| Logan | 20 | 50 (10/20) | 0.0 | 5 (1/20 | 0.0 |
| Lower Elkhorn | 37 | 41 (15/37) | 0.0 | 5 (2/37) | 3 (1/37) |
| North Fork Elkhorn | 21 | 19 (4/21) | 0.0 | 0.00 | 5 (1/21) |
| Upper Elkhorn | 17 | 24 (4/17) | 0.0 | 6 (1/17) | 12 (2/17) |
| Upper Big Blue | 17 | 65 (11/17) | 0.0 | 6 (1/17) | 0.0 |
| West Fork Big Blue | 20 | 65 (14/20) | 10 (2/20) | 0.0 | 0.0 |
| All | 132 | 43.9 (58/132) | 1.5 (2/132) | 3.8 (5/132) | 3.0 (4/132) |
| Nitrate | Atrazine | Uranium | Arsenic | Diabetes | Tobacco | Birth Defect Cases | |
|---|---|---|---|---|---|---|---|
| Nitrate | 1 | 0.02 | 0.03 | −0.18 | −0.23 | −0.26 | −0.24 |
| Atrazine | 0.02 | 1 | −0.01 | −0.04 | −0.09 | −0.03 | 0.10 |
| Uranium | 0.03 | −0.01 | 1 | −0.08 | 0.03 | 0.01 | −0.02 |
| Arsenic | −0.18 | −0.04 | −0.08 | 1 | −0.06 | −0.01 | −0.01 |
| Diabetes | −0.23 | −0.09 | 0.03 | −0.06 | 1 | 0.96 | 0.76 |
| Tobacco | −0.03 | −0.03 | 0.01 | −0.01 | 0.96 | 1 | 0.88 |
| Birth defect cases | −0.24 | 0.10 | −0.02 | −0.01 | 0.76 | 0.88 | 1 |
| Variables | Birth Defects | |
|---|---|---|
| Univariable Analysis IRRc (95% CI) a | Multivariable Analysis IRRa (95% CI) a | |
| Nitrate | ||
| Group 1 | Reference | Reference |
| Group 2 | 1.44 (1.40–1.50) | 0.86 (0.83–0.89) |
| Atrazine | ||
| Group 1 | Reference | Reference |
| Group 2 | 2.84 (2.75–2.93) | 1.62 (1.56–1.70) |
| Diabetes | 1.03 (1.03–1.04) | 1.03 (1.02–1.04) |
| Tobacco b | 1.01 (1.01–1.02) | N/A |
| Uranium | 1.03 (1.02–1.03) | 0.96 (0.95–0.97) |
| Arsenic | 0.85 (0.84–0.86) | 0.92 (0.89–0.94) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ouattara, B.S.; Zahid, M.; Rahman, F.I.; Weber, K.A.; Bartelt-Hunt, S.L.; Rogan, E.G. Investigation of a Possible Relationship between Anthropogenic and Geogenic Water Contaminants and Birth Defects Occurrence in Rural Nebraska. Water 2022, 14, 2289. https://doi.org/10.3390/w14152289
Ouattara BS, Zahid M, Rahman FI, Weber KA, Bartelt-Hunt SL, Rogan EG. Investigation of a Possible Relationship between Anthropogenic and Geogenic Water Contaminants and Birth Defects Occurrence in Rural Nebraska. Water. 2022; 14(15):2289. https://doi.org/10.3390/w14152289
Chicago/Turabian StyleOuattara, Balkissa S., Muhammad Zahid, Farzana I. Rahman, Karrie A. Weber, Shannon L. Bartelt-Hunt, and Eleanor G. Rogan. 2022. "Investigation of a Possible Relationship between Anthropogenic and Geogenic Water Contaminants and Birth Defects Occurrence in Rural Nebraska" Water 14, no. 15: 2289. https://doi.org/10.3390/w14152289







