New Insights for Exploring the Risks of Bioaccumulation, Molecular Mechanisms, and Cellular Toxicities of AgNPs in Aquatic Ecosystem
Abstract
:1. Introduction
2. Discharge of AgNPs into the Aquatic Environment
Dissolution and Toxicity in Fresh and Marine Water
3. Uptake and Bioaccumulation of AgNPs
3.1. Bioaccumulation of AgNPs in Aquatic Organis
Mechanisms of Bioaccumulation
3.2. Ecological and Biological Toxic Effects of AgNPs
3.3. Toxicity of AgNPs on Algal Cells
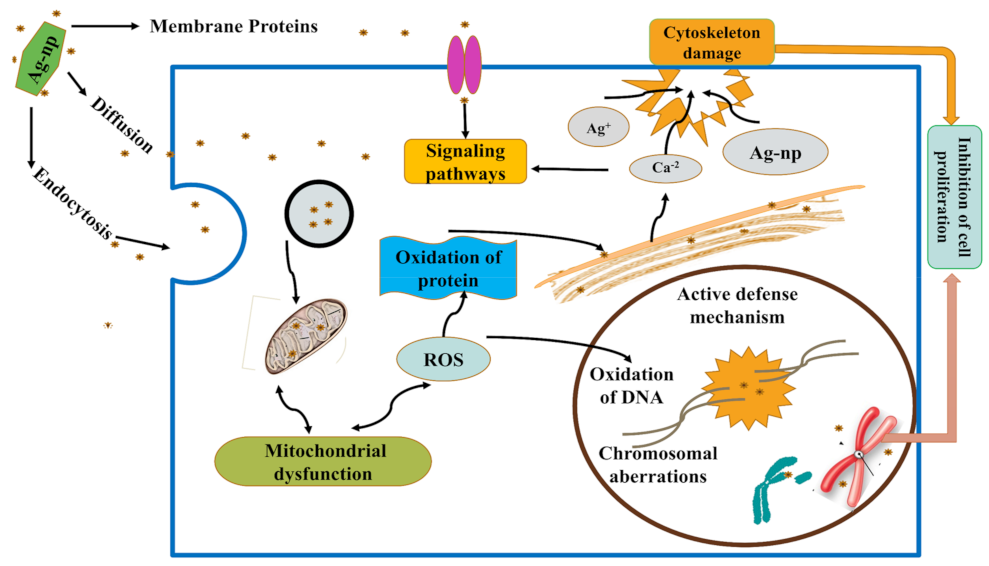
3.4. Different Toxicological Pathways of AgNPs
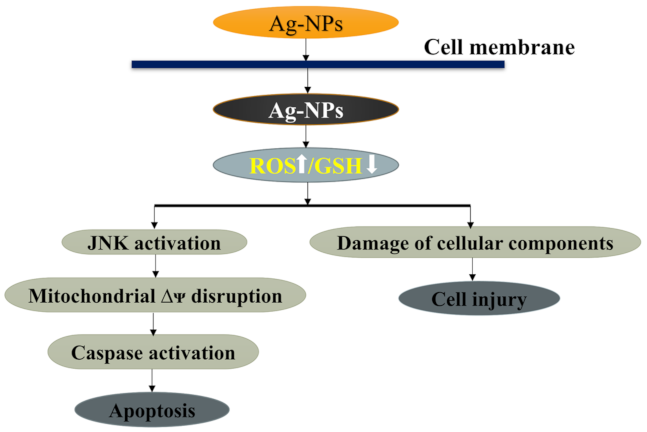
3.4.1. AgNPs Regulate Apoptotic Pathways
3.4.2. Mechanism of Apoptosis by AgNPs
3.4.3. Trojan-Horse Mechanism
4. Pharmacological Activities of AgNPs
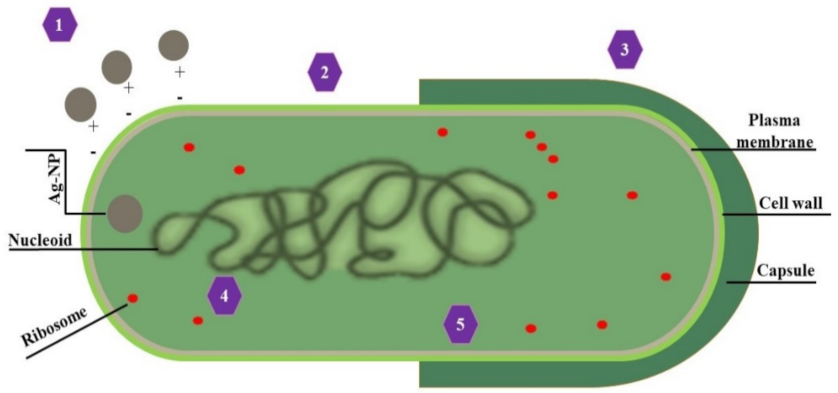
4.1. Antimicrobial Activities of AgNPs
4.2. Chronic and Acute Toxicity Effects of AgNPs
5. Different Methods for Silver Ions Detection
5.1. Biosensor
5.2. Chemical Sensor
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad, A.; Mohd-Setapar, S.H.; Chuong, C.S.; Khatoon, A.; Wani, W.A.; Kumar, R.; Rafatullah, M. Recent advances in new generation dye removal technologies: Novel search for approaches to reprocess wastewater. RSC Adv. 2015, 5, 30801–30818. [Google Scholar] [CrossRef]
- Vasistha, P.; Ganguly, R. Water quality assessment of natural lakes and its importance: An overview. Mater. Today Proc. 2020, 32, 544–552. [Google Scholar] [CrossRef]
- Elbehiry, F.; Alshaal, T.; Elhawat, N.; Elbasiouny, H. Environmental-Friendly and Cost-Effective Agricultural Wastes for Heavy Metals and Toxicants Removal from Wastewater. In The Handbook of Environmental Chemistry; Springer: Berlin/Heidelberg, Germany, 2021. [Google Scholar]
- Hullmann, A. The Economic Development of Nanotechnology—An Indicators Based Analysis; European Commission: Brussels, Belgium, 2006. [Google Scholar]
- Klaine, S.J.; Alvarez, P.J.J.; Batley, G.E.; Fernandes, T.F.; Handy, R.D.; Lyon, D.Y.; Mahendra, S.; McLaughlin, M.J.; Lead, J.R. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects. Environ. Toxicol. Chem. Int. J. 2008, 27, 1825–1851. [Google Scholar] [CrossRef]
- Fabrega, J.; Luoma, S.N.; Tyler, C.R.; Galloway, T.S.; Lead, J.R. Silver nanoparticles: Behaviour and effects in the aquatic environment. Environ. Int. 2011, 37, 517–531. [Google Scholar] [CrossRef] [PubMed]
- Xia, G.; Liu, T.; Wang, Z.; Hou, Y.; Dong, L.; Zhu, J.; Qi, J. The effect of silver nanoparticles on zebrafish embryonic development and toxicology. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1116–1121. [Google Scholar] [CrossRef]
- Vance, M.E.; Kuiken, T.; Vejerano, E.P.; McGinnis, S.P.; Hochella, M.F., Jr.; Rejeski, D.; Hull, M.S. Nanotechnology in the real world: Redeveloping the nanomaterial consumer products inventory. Beilstein J. Nanotechnol. 2015, 6, 1769–1780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fabrega, J.; Zhang, R.; Renshaw, J.C.; Liu, W.-T.; Lead, J.R. Impact of silver nanoparticles on natural marine biofilm bacteria. Chemosphere 2011, 85, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Qamar, S.U.R.; Ahmad, J.N. Nanoparticles: Mechanism of biosynthesis using plant extracts, bacteria, fungi, and their applications. J. Mol. Liq. 2021, 334, 116040. [Google Scholar] [CrossRef]
- Ottoni, C.A.; Simões, M.F.; Fernandes, S.; Dos Santos, J.G.; Da Silva, E.S.; de Souza, R.F.B.; Maiorano, A.E. Screening of filamentous fungi for antimicrobial silver nanoparticles synthesis. AMB Express 2017, 7, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Alexander, J.W. History of the medical use of silver. Surg. Infect. 2009, 10, 289–292. [Google Scholar] [CrossRef] [Green Version]
- Fabrega, J.; Fawcett, S.R.; Renshaw, J.C.; Lead, J.R. Silver nanoparticle impact on bacterial growth: Effect of pH, concentration, and organic matter. Environ. Sci. Technol. 2009, 43, 7285–7290. [Google Scholar] [CrossRef] [PubMed]
- McGillicuddy, E.; Murray, I.; Kavanagh, S.; Morrison, L.; Fogarty, A.; Cormican, M.; Dockery, P.; Prendergast, M.; Rowan, N.; Morris, D. Silver nanoparticles in the environment: Sources, detection and ecotoxicology. Sci. Total Environ. 2017, 575, 231–246. [Google Scholar] [CrossRef] [PubMed]
- Christen, V.; Capelle, M.; Fent, K. Silver nanoparticles induce endoplasmatic reticulum stress response in zebrafish. Toxicol. Appl. Pharmacol. 2013, 272, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.A.; McFerran, S.; Lazareva, A.; Suh, S. Global life cycle releases of engineered nanomaterials. J. Nanopart. Res. 2013, 15, 1–17. [Google Scholar] [CrossRef]
- Caballero-Guzman, A.; Sun, T.; Nowack, B. Flows of engineered nanomaterials through the recycling process in Switzerland. Waste Manag. 2015, 36, 33–43. [Google Scholar] [CrossRef]
- Ratte, H.T. Bioaccumulation and toxicity of silver compounds: A review. Environ. Toxicol. Chem. Int. J. 1999, 18, 89–108. [Google Scholar] [CrossRef]
- Bianchini, A.; Wood, C.M. Mechanism of acute silver toxicity in Daphnia magna. Environ. Toxicol. Chem. Int. J. 2003, 22, 1361–1367. [Google Scholar] [CrossRef]
- Kleiven, M.; Macken, A.; Oughton, D.H. Growth inhibition in Raphidocelis subcapita–Evidence of nanospecific toxicity of silver nanoparticles. Chemosphere 2019, 221, 785–792. [Google Scholar] [CrossRef]
- Katuli, K.K.; Massarsky, A.; Hadadi, A.; Pourmehran, Z. Silver nanoparticles inhibit the gill Na+/K+-ATPase and erythrocyte AChE activities and induce the stress response in adult zebrafish (Danio rerio). Ecotoxicol. Environ. Saf. 2014, 106, 173–180. [Google Scholar] [CrossRef]
- Brunetti, G.; Donner, E.; Laera, G.; Sekine, R.; Scheckel, K.G.; Khaksar, M.; Vasilev, K.; De Mastro, G.; Lombi, E. Fate of zinc and silver engineered nanoparticles in sewerage networks. Water Res. 2015, 77, 72–84. [Google Scholar] [CrossRef] [Green Version]
- Pulit-Prociak, J.; Banach, M. Silver nanoparticles–a material of the future…? Open Chem. 2016, 14, 76–91. [Google Scholar] [CrossRef]
- Luoma, S.N. Silver nanotechnologies and the environment. Proj. Emerg. Nanotechnol. Rep. 2008, 15, 12–13. [Google Scholar]
- Luoma, S.N.; Rainbow, P.S. Metal Contamination in Aquatic Environments: Science and Lateral Management; Cambridge University Press: Cambridge, UK, 2008; ISBN 0521860571. [Google Scholar]
- Levard, C.; Hotze, E.M.; Lowry, G.V.; Brown, G.E., Jr. Environmental transformations of silver nanoparticles: Impact on stability and toxicity. Environ. Sci. Technol. 2012, 46, 6900–6914. [Google Scholar] [CrossRef] [PubMed]
- Magesky, A.; Pelletier, É. Cytotoxicity and physiological effects of silver nanoparticles on marine invertebrates. Cell. Mol. Toxicol. Nanopart. 2018, 1048, 285–309. [Google Scholar]
- Cozzari, M.; Elia, A.C.; Pacini, N.; Smith, B.D.; Boyle, D.; Rainbow, P.S.; Khan, F.R. Bioaccumulation and oxidative stress responses measured in the estuarine ragworm (Nereis diversicolor) exposed to dissolved, nano-and bulk-sized silver. Environ. Pollut. 2015, 198, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Duong, C.N.; Cho, J.; Lee, J.; Kim, K.; Seo, Y.; Kim, P.; Choi, K.; Yoon, J. Toxicity of citrate-capped silver nanoparticles in common carp (Cyprinus carpio). J. Biomed. Biotechnol. 2012, 2012, 262670. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Ho, K.T.; Scheckel, K.G.; Wu, F.; Cantwell, M.G.; Katz, D.R.; Horowitz, D.B.; Boothman, W.S.; Burgess, R.M. Toxicity, bioaccumulation, and biotransformation of silver nanoparticles in marine organisms. Environ. Sci. Technol. 2014, 48, 13711–13717. [Google Scholar] [CrossRef]
- Ribeiro, F.; Gallego-Urrea, J.A.; Goodhead, R.M.; Van Gestel, C.A.M.; Moger, J.; Soares, A.M.V.M.; Loureiro, S. Uptake and elimination kinetics of silver nanoparticles and silver nitrate by Raphidocelis subcapitata: The influence of silver behaviour in solution. Nanotoxicology 2015, 9, 686–695. [Google Scholar] [CrossRef]
- Macken, A.; Byrne, H.J.; Thomas, K.V. Effects of salinity on the toxicity of ionic silver and Ag-PVP nanoparticles to Tisbe battagliai and Ceramium tenuicorne. Ecotoxicol. Environ. Saf. 2012, 86, 101–110. [Google Scholar] [CrossRef] [Green Version]
- Thio, B.J.R.; Montes, M.O.; Mahmoud, M.A.; Lee, D.-W.; Zhou, D.; Keller, A.A. Mobility of capped silver nanoparticles under environmentally relevant conditions. Environ. Sci. Technol. 2012, 46, 6985–6991. [Google Scholar] [CrossRef]
- Cleveland, D.; Long, S.E.; Pennington, P.L.; Cooper, E.; Fulton, M.H.; Scott, G.I.; Brewer, T.; Davis, J.; Petersen, E.J.; Wood, L. Pilot estuarine mesocosm study on the environmental fate of silver nanomaterials leached from consumer products. Sci. Total Environ. 2012, 421, 267–272. [Google Scholar] [CrossRef]
- Blaser, S.A.; Scheringer, M.; MacLeod, M.; Hungerbühler, K. Estimation of cumulative aquatic exposure and risk due to silver: Contribution of nano-functionalized plastics and textiles. Sci. Total Environ. 2008, 390, 396–409. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.-L. Effect of silver nanoparticles on tropical freshwater and marine microalgae. J. Chem. 2019, 2019, 9658386. [Google Scholar] [CrossRef]
- Mueller, N.C.; Nowack, B. Exposure modeling of engineered nanoparticles in the environment. Environ. Sci. Technol. 2008, 42, 4447–4453. [Google Scholar] [CrossRef] [PubMed]
- Luoma, S.N.; Rainbow, P.S. Why is metal bioaccumulation so variable? Biodynamics as a unifying concept. Environ. Sci. Technol. 2005, 39, 1921–1931. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.; Weissleder, R.; Bogdanov, A., Jr. Uptake of dextran-coated monocrystalline iron oxides in tumor cells and macrophages. J. Magn. Reson. Imaging 1997, 7, 1140–1145. [Google Scholar] [CrossRef]
- Zhao, C.-M.; Wang, W.-X. Biokinetic uptake and efflux of silver nanoparticles in Daphnia magna. Environ. Sci. Technol. 2010, 44, 7699–7704. [Google Scholar] [CrossRef]
- Sohn, E.K.; Johari, S.A.; Kim, T.G.; Kim, J.K.; Kim, E.; Lee, J.H.; Chung, Y.S.; Yu, I.J. Aquatic toxicity comparison of silver nanoparticles and silver nanowires. BioMed Res. Int. 2015, 2015, 893049. [Google Scholar] [CrossRef]
- Hartmann, S.; Beasley, A.; Mozhayeva, D.; Engelhard, C.; Witte, K. Defective defence in Daphnia daughters: Silver nanoparticles inhibit anti-predator defence in offspring but not in maternal Daphnia magna. Sci. Rep. 2020, 10, 8021. [Google Scholar] [CrossRef]
- Farkas, J.; Christian, P.; Urrea, J.A.G.; Roos, N.; Hassellöv, M.; Tollefsen, K.E.; Thomas, K.V. Effects of silver and gold nanoparticles on rainbow trout (Oncorhynchus mykiss) hepatocytes. Aquat. Toxicol. 2010, 96, 44–52. [Google Scholar] [CrossRef]
- Maurer-Jones, M.A.; Gunsolus, I.L.; Murphy, C.J.; Haynes, C.L. Toxicity of engineered nanoparticles in the environment. Anal. Chem. 2013, 85, 3036–3049. [Google Scholar] [CrossRef] [Green Version]
- Miao, A.-J.; Schwehr, K.A.; Xu, C.; Zhang, S.-J.; Luo, Z.; Quigg, A.; Santschi, P.H. The algal toxicity of silver engineered nanoparticles and detoxification by exopolymeric substances. Environ. Pollut. 2009, 157, 3034–3041. [Google Scholar] [CrossRef]
- Roh, J.; Sim, S.J.; Yi, J.; Park, K.; Chung, K.H.; Ryu, D.; Choi, J. Ecotoxicity of silver nanoparticles on the soil nematode Caenorhabditis elegans using functional ecotoxicogenomics. Environ. Sci. Technol. 2009, 43, 3933–3940. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.N.; Lord, C.A.; Yang, X.Y.; Turner, E.A.; Badireddy, A.R.; Marinakos, S.M.; Chilkoti, A.; Wiesner, M.R.; Auffan, M. Intracellular uptake and associated toxicity of silver nanoparticles in Caenorhabditis elegans. Aquat. Toxicol. 2010, 100, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Shoults-Wilson, W.A.; Reinsch, B.C.; Tsyusko, O.V.; Bertsch, P.M.; Lowry, G.V.; Unrine, J.M. Effect of silver nanoparticle surface coating on bioaccumulation and reproductive toxicity in earthworms (Eisenia fetida). Nanotoxicology 2011, 5, 432–444. [Google Scholar] [CrossRef] [PubMed]
- Heckmann, L.-H.; Hovgaard, M.B.; Sutherland, D.S.; Autrup, H.; Besenbacher, F.; Scott-Fordsmand, J.J. Limit-test toxicity screening of selected inorganic nanoparticles to the earthworm Eisenia fetida. Ecotoxicology 2011, 20, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.J.; Nallathamby, P.D.; Browning, L.M.; Osgood, C.J.; Xu, X.-H.N. In vivo imaging of transport and biocompatibility of single silver nanoparticles in early development of zebrafish embryos. ACS Nano 2007, 1, 133–143. [Google Scholar] [CrossRef] [Green Version]
- Nallathamby, P.D.; Lee, K.J.; Xu, X.-H.N. Design of stable and uniform single nanoparticle photonics for in vivo dynamics imaging of nanoenvironments of zebrafish embryonic fluids. ACS Nano 2008, 2, 1371–1380. [Google Scholar] [CrossRef] [Green Version]
- Yeo, M.-K.; Kang, M.-S. Effects of nanometer sized silver materials on biological toxicity during zebrafish embryogenesis. Bull. Korean Chem. Soc. 2008, 29, 1179–1184. [Google Scholar]
- Yeo, M.-K.; Pak, S.-W. Exposing zebrafish to silver nanoparticles during caudal fin regeneration disrupts caudal fin growth and p53 signaling. Mol. Cell. Toxicol. 2008, 4, 311–317. [Google Scholar]
- Griffitt, R.J.; Luo, J.; Gao, J.; Bonzongo, J.; Barber, D.S. Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. Environ. Toxicol. Chem. Int. J. 2008, 27, 1972–1978. [Google Scholar] [CrossRef]
- Bilberg, K.; Malte, H.; Wang, T.; Baatrup, E. Silver nanoparticles and silver nitrate cause respiratory stress in Eurasian perch (Perca fluviatilis). Aquat. Toxicol. 2010, 96, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Farkas, J.; Christian, P.; Gallego-Urrea, J.A.; Roos, N.; Hassellöv, M.; Tollefsen, K.E.; Thomas, K.V. Uptake and effects of manufactured silver nanoparticles in rainbow trout (Oncorhynchus mykiss) gill cells. Aquat. Toxicol. 2011, 101, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.J.; Kim, J.Y.; Yang, S.Y.; Lee, B.G.; Kim, S.D. Bioaccumulation of silver nanoparticles in Oryzias latipes through aqueous exposure. Korean Soc. Environ. Health Toxicol. 2012, 10, 410–411. [Google Scholar]
- Stampoulis, D.; Sinha, S.K.; White, J.C. Assay-dependent phytotoxicity of nanoparticles to plants. Environ. Sci. Technol. 2009, 43, 9473–9479. [Google Scholar] [CrossRef]
- Ma, X.; Geiser-Lee, J.; Deng, Y.; Kolmakov, A. Interactions between engineered nanoparticles (ENPs) and plants: Phytotoxicity, uptake and accumulation. Sci. Total Environ. 2010, 408, 3053–3061. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Cheng, Y.; Espinasse, B.; Colman, B.P.; Auffan, M.; Wiesner, M.; Rose, J.; Liu, J.; Bernhardt, E.S. More than the ions: The effects of silver nanoparticles on Lolium multiflorum. Environ. Sci. Technol. 2011, 45, 2360–2367. [Google Scholar] [CrossRef]
- Oberdörster, E. Manufactured nanomaterials (fullerenes, C60) induce oxidative stress in the brain of juvenile largemouth bass. Environ. Health Perspect. 2004, 112, 1058–1062. [Google Scholar] [CrossRef] [Green Version]
- Moore, M.N. Do nanoparticles present ecotoxicological risks for the health of the aquatic environment? Environ. Int. 2006, 32, 967–976. [Google Scholar] [CrossRef]
- Liu, J.; Hurt, R.H. Ion release kinetics and particle persistence in aqueous nano-silver colloids. Environ. Sci. Technol. 2010, 44, 2169–2175. [Google Scholar] [CrossRef]
- Brown, D.M.; Wilson, M.R.; MacNee, W.; Stone, V.; Donaldson, K. Size-dependent proinflammatory effects of ultrafine polystyrene particles: A role for surface area and oxidative stress in the enhanced activity of ultrafines. Toxicol. Appl. Pharmacol. 2001, 175, 191–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reidy, B.; Haase, A.; Luch, A.; Dawson, K.A.; Lynch, I. Mechanisms of silver nanoparticle release, transformation and toxicity: A critical review of current knowledge and recommendations for future studies and applications. Materials 2013, 6, 2295–2350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, V.K. Stability and toxicity of silver nanoparticles in aquatic environment: A review. Sustain. Nanotechnol. Environ. Adv. Achiev. 2013, 1124, 165–179. [Google Scholar]
- Zhang, L.; Wang, W.-X. Dominant role of silver ions in silver nanoparticle toxicity to a unicellular alga: Evidence from luminogen imaging. Environ. Sci. Technol. 2018, 53, 494–502. [Google Scholar] [CrossRef]
- Huang, Z.; Zeng, Z.; Chen, A.; Zeng, G.; Xiao, R.; Xu, P.; He, K.; Song, Z.; Hu, L.; Peng, M. Differential behaviors of silver nanoparticles and silver ions towards cysteine: Bioremediation and toxicity to Phanerochaete chrysosporium. Chemosphere 2018, 203, 199–208. [Google Scholar] [CrossRef]
- Abramenko, N.B.; Demidova, T.B.; Abkhalimov, E.V.; Ershov, B.G.; Krysanov, E.Y.; Kustov, L.M. Ecotoxicity of different-shaped silver nanoparticles: Case of zebrafish embryos. J. Hazard. Mater. 2018, 347, 89–94. [Google Scholar] [CrossRef]
- Kvitek, L.; Vanickova, M.; Panacek, A.; Soukupova, J.; Dittrich, M.; Valentova, E.; Prucek, R.; Bancirova, M.; Milde, D.; Zboril, R. Initial study on the toxicity of silver nanoparticles (NPs) against Paramecium caudatum. J. Phys. Chem. C 2009, 113, 4296–4300. [Google Scholar] [CrossRef]
- Braydich-Stolle, L.K.; Lucas, B.; Schrand, A.; Murdock, R.C.; Lee, T.; Schlager, J.J.; Hussain, S.M.; Hofmann, M.-C. Silver nanoparticles disrupt GDNF/Fyn kinase signaling in spermatogonial stem cells. Toxicol. Sci. 2010, 116, 577–589. [Google Scholar] [CrossRef] [Green Version]
- Mohammadinejad, R.; Moosavi, M.A.; Tavakol, S.; Vardar, D.Ö.; Hosseini, A.; Rahmati, M.; Dini, L.; Hussain, S.; Mandegary, A.; Klionsky, D.J. Necrotic, apoptotic and autophagic cell fates triggered by nanoparticles. Autophagy 2019, 15, 4–33. [Google Scholar] [CrossRef] [Green Version]
- Ahmadi, F.; Branch, S. Impact of different levels of silver nanoparticles (Ag-NPs) on performance, oxidative enzymes and blood parameters in broiler chicks. Pak. Vet. J. 2012, 32, 325–328. [Google Scholar]
- Haase, A.; Rott, S.; Mantion, A.; Graf, P.; Plendl, J.; Thünemann, A.F.; Meier, W.P.; Taubert, A.; Luch, A.; Reiser, G. Effects of silver nanoparticles on primary mixed neural cell cultures: Uptake, oxidative stress and acute calcium responses. Toxicol. Sci. 2012, 126, 457–468. [Google Scholar] [CrossRef]
- Perde-Schrepler, M.; Florea, A.; Brie, I.; Virag, P.; Fischer-Fodor, E.; Vâlcan, A.; Gurzău, E.; Lisencu, C.; Maniu, A. Size-dependent cytotoxicity and genotoxicity of silver nanoparticles in cochlear cells in vitro. J. Nanomater. 2019, 2019, 6090259. [Google Scholar] [CrossRef] [Green Version]
- Awasthi, K.K.; Awasthi, A.; Kumar, N.; Roy, P.; Awasthi, K.; John, P.J. Silver nanoparticle induced cytotoxicity, oxidative stress, and DNA damage in CHO cells. J. Nanopart. Res. 2013, 15, 1898. [Google Scholar] [CrossRef]
- Banerjee, V.; Das, K.P. Interaction of silver nanoparticles with proteins: A characteristic protein concentration dependent profile of SPR signal. Colloids Surfaces B Biointerfaces 2013, 111, 71–79. [Google Scholar] [CrossRef]
- Tripathi, D.K.; Tripathi, A.; Singh, S.; Singh, Y.; Vishwakarma, K.; Yadav, G.; Sharma, S.; Singh, V.K.; Mishra, R.K.; Upadhyay, R.G. Uptake, accumulation and toxicity of silver nanoparticle in autotrophic plants, and heterotrophic microbes: A concentric review. Front. Microbiol. 2017, 8, 7. [Google Scholar] [CrossRef]
- Oukarroum, A.; Bras, S.; Perreault, F.; Popovic, R. Inhibitory effects of silver nanoparticles in two green algae, Chlorella vulgaris and Dunaliella tertiolecta. Ecotoxicol. Environ. Saf. 2012, 78, 80–85. [Google Scholar] [CrossRef]
- Moreno-Garrido, I.; Pérez, S.; Blasco, J. Toxicity of silver and gold nanoparticles on marine microalgae. Mar. Environ. Res. 2015, 111, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Zhang, X.; Zhang, C.; Chen, Z. Molecular interactions between gold nanoparticles and model cell membranes. Phys. Chem. Chem. Phys. 2015, 17, 9873–9884. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Fang, J.; Cheng, H. Toxicity of silver nanoparticles to green algae M. aeruginosa and alleviation by organic matter. Environ. Monit. Assess. 2018, 190, 667. [Google Scholar] [CrossRef]
- Khoshnamvand, M.; Hao, Z.; Fadare, O.O.; Hanachi, P.; Chen, Y.; Liu, J. Toxicity of biosynthesized silver nanoparticles to aquatic organisms of different trophic levels. Chemosphere 2020, 258, 127346. [Google Scholar] [CrossRef]
- Ottoni, C.A.; Neto, M.C.L.; Léo, P.; Ortolan, B.D.; Barbieri, E.; De Souza, A.O. Environmental impact of biogenic silver nanoparticles in soil and aquatic organisms. Chemosphere 2020, 239, 124698. [Google Scholar] [CrossRef] [PubMed]
- Kruszewski, M.; Grzelak, A. Nanoparticle toxicity and reactive species: An overview. In Toxicology; Academic Press: Cambridge, MA, USA, 2021; pp. 11–21. [Google Scholar]
- Sun, J.; Wan, J.; Zhai, X.; Wang, J.; Liu, Z.; Tian, H.; Xin, L. Silver nanoparticles: Correlating particle size and ionic Ag release with cytotoxicity, genotoxicity, and inflammatory responses in human cell lines. Toxicol. Ind. Health 2021, 37, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liu, W.; Slaveykova, V.I. Effects of mixtures of engineered nanoparticles and metallic pollutants on aquatic organisms. Environments 2020, 7, 27. [Google Scholar] [CrossRef] [Green Version]
- Arora, S.; Jain, J.; Rajwade, J.M.; Paknikar, K.M. Interactions of silver nanoparticles with primary mouse fibroblasts and liver cells. Toxicol. Appl. Pharmacol. 2009, 236, 310–318. [Google Scholar] [CrossRef]
- Picó, Y.; Andreu, V. Analytical tools able to detect ENP/NM/MNs in both artificial and natural environmental water media. In Ecotoxicology of Nanoparticles in Aquatic Systems; CRC Press: Boca Raton, FL, USA, 2019; pp. 230–258. ISBN 1315158760. [Google Scholar]
- McShan, D.; Ray, P.C.; Yu, H. Molecular toxicity mechanism of nanosilver. J. Food Drug Anal. 2014, 22, 116–127. [Google Scholar] [CrossRef] [Green Version]
- Unrine, J.M.; Colman, B.P.; Bone, A.J.; Gondikas, A.P.; Matson, C.W. Biotic and abiotic interactions in aquatic microcosms determine fate and toxicity of Ag nanoparticles. Part 1. Aggregation and dissolution. Environ. Sci. Technol. 2012, 46, 6915–6924. [Google Scholar] [CrossRef]
- Azimzada, A.; Tufenkji, N.; Wilkinson, K.J. Transformations of silver nanoparticles in wastewater effluents: Links to Ag bioavailability. Environ. Sci. Nano 2017, 4, 1339–1349. [Google Scholar] [CrossRef] [Green Version]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on zinc oxide nanoparticles: Antibacterial activity and toxicity mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef] [Green Version]
- Sharifi, M.; Hosseinali, S.H.; Saboury, A.A.; Szegezdi, E.; Falahati, M. Involvement of planned cell death of necroptosis in cancer treatment by nanomaterials: Recent advances and future perspectives. J. Control. Release 2019, 299, 121–137. [Google Scholar] [CrossRef]
- Fu, P.P.; Xia, Q.; Hwang, H.-M.; Ray, P.C.; Yu, H. Mechanisms of nanotoxicity: Generation of reactive oxygen species. J. Food Drug Anal. 2014, 22, 64–75. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Zhou, Q. Silver nanoparticles cause oxidative damage and histological changes in medaka (Oryzias latipes) after 14 days of exposure. Environ. Toxicol. Chem. 2013, 32, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Asharani, P.V.; Hande, M.P.; Valiyaveettil, S. Anti-proliferative activity of silver nanoparticles. BMC Cell Biol. 2009, 10, 65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuchs, Y.; Steller, H. Programmed cell death in animal development and disease. Cell 2011, 147, 742–758. [Google Scholar] [CrossRef] [Green Version]
- Quevedo, A.C.; Lynch, I.; Valsami-Jones, E. Silver nanoparticle induced toxicity and cell death mechanisms in embryonic zebrafish cells. Nanoscale 2021, 13, 6142–6161. [Google Scholar] [CrossRef]
- Guicciardi, M.E.; Gores, G.J. Life and death by death receptors. FASEB J. 2009, 23, 1625–1637. [Google Scholar] [CrossRef] [Green Version]
- Ferdous, Z.; Nemmar, A. Health impact of silver nanoparticles: A review of the biodistribution and toxicity following various routes of exposure. Int. J. Mol. Sci. 2020, 21, 2375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, S.J.; Ryoo, I.; Lee, Y.J.; Kwak, M.-K. Role of the Nrf2-heme oxygenase-1 pathway in silver nanoparticle-mediated cytotoxicity. Toxicol. Appl. Pharmacol. 2012, 258, 89–98. [Google Scholar] [CrossRef]
- Li, Y.; Guo, M.; Lin, Z.; Zhao, M.; Xiao, M.; Wang, C.; Xu, T.; Chen, T.; Zhu, B. Polyethylenimine-functionalized silver nanoparticle-based co-delivery of paclitaxel to induce HepG2 cell apoptosis. Int. J. Nanomed. 2016, 11, 6693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akter, M.; Sikder, M.T.; Rahman, M.M.; Ullah, A.K.M.A.; Hossain, K.F.B.; Banik, S.; Hosokawa, T.; Saito, T.; Kurasaki, M. A systematic review on silver nanoparticles-induced cytotoxicity: Physicochemical properties and perspectives. J. Adv. Res. 2018, 9, 1–16. [Google Scholar] [CrossRef]
- Hsin, Y.-H.; Chen, C.-F.; Huang, S.; Shih, T.-S.; Lai, P.-S.; Chueh, P.J. The apoptotic effect of nanosilver is mediated by a ROS-and JNK-dependent mechanism involving the mitochondrial pathway in NIH3T3 cells. Toxicol. Lett. 2008, 179, 130–139. [Google Scholar] [CrossRef]
- Piao, M.J.; Kang, K.A.; Lee, I.K.; Kim, H.S.; Kim, S.; Choi, J.Y.; Choi, J.; Hyun, J.W. Silver nanoparticles induce oxidative cell damage in human liver cells through inhibition of reduced glutathione and induction of mitochondria-involved apoptosis. Toxicol. Lett. 2011, 201, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Quan, J.-H.; Gao, F.F.; Ismail, H.A.H.A.; Yuk, J.-M.; Cha, G.-H.; Chu, J.-Q.; Lee, Y.-H. Silver nanoparticle-induced apoptosis in ARPE-19 cells is inhibited by Toxoplasma gondii pre-infection through suppression of NOX4-dependent ROS generation. Int. J. Nanomedicine 2020, 15, 3695. [Google Scholar] [CrossRef] [PubMed]
- Limbach, L.K.; Wick, P.; Manser, P.; Grass, R.N.; Bruinink, A.; Stark, W.J. Exposure of engineered nanoparticles to human lung epithelial cells: Influence of chemical composition and catalytic activity on oxidative stress. Environ. Sci. Technol. 2007, 41, 4158–4163. [Google Scholar] [CrossRef]
- Baun, A.; Sørensen, S.N.; Rasmussen, R.F.; Hartmann, N.B.; Koch, C.B. Toxicity and bioaccumulation of xenobiotic organic compounds in the presence of aqueous suspensions of aggregates of nano-C60. Aquat. Toxicol. 2008, 86, 379–387. [Google Scholar] [CrossRef]
- Hsiao, I.-L.; Hsieh, Y.-K.; Wang, C.-F.; Chen, I.-C.; Huang, Y.-J. Trojan-horse mechanism in the cellular uptake of silver nanoparticles verified by direct intra-and extracellular silver speciation analysis. Environ. Sci. Technol. 2015, 49, 3813–3821. [Google Scholar] [CrossRef]
- Su, Y.; Yan, X.; Pu, Y.; Xiao, F.; Wang, D.; Yang, M. Risks of single-walled carbon nanotubes acting as contaminants-carriers: Potential release of phenanthrene in Japanese medaka (Oryzias latipes). Environ. Sci. Technol. 2013, 47, 4704–4710. [Google Scholar] [CrossRef]
- Zhang, W.; Xiao, B.; Fang, T. Chemical transformation of silver nanoparticles in aquatic environments: Mechanism, morphology and toxicity. Chemosphere 2018, 191, 324–334. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Jones, A.M.; Garg, S.; Pham, A.N.; Waite, T.D. Silver nanoparticle− reactive oxygen species interactions: Application of a charging− discharging model. J. Phys. Chem. C 2011, 115, 5461–5468. [Google Scholar] [CrossRef]
- Khan, F.R.; Misra, S.K.; Bury, N.R.; Smith, B.D.; Rainbow, P.S.; Luoma, S.N.; Valsami-Jones, E. Inhibition of potential uptake pathways for silver nanoparticles in the estuarine snail Peringia ulvae. Nanotoxicology 2015, 9, 493–501. [Google Scholar] [CrossRef]
- Navarro, E.; Piccapietra, F.; Wagner, B.; Marconi, F.; Kaegi, R.; Odzak, N.; Sigg, L.; Behra, R. Toxicity of silver nanoparticles to Chlamydomonas reinhardtii. Environ. Sci. Technol. 2008, 42, 8959–8964. [Google Scholar] [CrossRef]
- Khaydarov, R.R.; Khaydarov, R.A.; Gapurova, O. Remediation of contaminated groundwater using nano-carbon colloids. In Nanomaterials: Risks and Benefits; Springer: Berlin/Heidelberg, Germany, 2009; pp. 219–224. [Google Scholar]
- Choi, O.K.; Hu, Z.Q. Nitrification inhibition by silver nanoparticles. Water Sci. Technol. 2009, 59, 1699–1702. [Google Scholar] [CrossRef] [PubMed]
- Lok, C.-N.; Ho, C.-M.; Chen, R.; He, Q.-Y.; Yu, W.-Y.; Sun, H.; Tam, P.K.-H.; Chiu, J.-F.; Che, C.-M. Silver nanoparticles: Partial oxidation and antibacterial activities. JBIC J. Biol. Inorg. Chem. 2007, 12, 527–534. [Google Scholar] [CrossRef] [PubMed]
- De Matteis, V.; Malvindi, M.A.; Galeone, A.; Brunetti, V.; De Luca, E.; Kote, S.; Kshirsagar, P.; Sabella, S.; Bardi, G.; Pompa, P.P. Negligible particle-specific toxicity mechanism of silver nanoparticles: The role of Ag+ ion release in the cytosol. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 731–739. [Google Scholar] [CrossRef]
- Jiang, H.S.; Yin, L.; Ren, N.N.; Xian, L.; Zhao, S.; Li, W.; Gontero, B. The effect of chronic silver nanoparticles on aquatic system in microcosms. Environ. Pollut. 2017, 223, 395–402. [Google Scholar] [CrossRef]
- Rubino, F.M. Toxicity of glutathione-binding metals: A review of targets and mechanisms. Toxics 2015, 3, 20–62. [Google Scholar] [CrossRef] [Green Version]
- Navarro, D.A.; Kirby, J.K.; McLaughlin, M.J.; Waddington, L.; Kookana, R.S. Remobilisation of silver and silver sulphide nanoparticles in soils. Environ. Pollut. 2014, 193, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Quadros, M.E.; Marr, L.C. Environmental and human health risks of aerosolized silver nanoparticles. J. Air Waste Manag. Assoc. 2010, 60, 770–781. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moustafa, E.M.; Khalil, R.H.; Saad, T.T.; Amer, M.T.; Shukry, M.; Farrag, F.; Elsawy, A.A.; Lolo, E.E.; Sakran, M.I.; Hamouda, A.H. Silver nanoparticles as an antibacterial agent in Oreochromis niloticus and Sparus auratus fish. Aquac. Res. 2021, 52, 6218–6234. [Google Scholar] [CrossRef]
- Ghetas, H.A.; Abdel-Razek, N.; Shakweer, M.S.; Abotaleb, M.M.; Paray, B.A.; Ali, S.; Eldessouki, E.A.; Dawood, M.A.O.; Khalil, R.H. Antimicrobial activity of chemically and biologically synthesized silver nanoparticles against some fish pathogens. Saudi J. Biol. Sci. 2022, 29, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- Romo-Quiñonez, C.R.; Álvarez-Sánchez, A.R.; Álvarez-Ruiz, P.; Chávez-Sánchez, M.C.; Bogdanchikova, N.; Pestryakov, A.; Mejia-Ruiz, C.H. Evaluation of a new Argovit as an antiviral agent included in feed to protect the shrimp Litopenaeus vannamei against White Spot Syndrome Virus infection. PeerJ 2020, 8, e8446. [Google Scholar] [CrossRef] [Green Version]
- Ochoa-Meza, A.R.; Álvarez-Sánchez, A.R.; Romo-Quiñonez, C.R.; Barraza, A.; Magallón-Barajas, F.J.; Chávez-Sánchez, A.; García-Ramos, J.C.; Toledano-Magaña, Y.; Bogdanchikova, N.; Pestryakov, A. Silver nanoparticles enhance survival of white spot syndrome virus infected Penaeus vannamei shrimps by activation of its immunological system. Fish Shellfish Immunol. 2019, 84, 1083–1089. [Google Scholar] [CrossRef]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Agnihotri, S.; Mukherji, S.; Mukherji, S. Size-controlled silver nanoparticles synthesized over the range 5–100 nm using the same protocol and their antibacterial efficacy. Rsc Adv. 2014, 4, 3974–3983. [Google Scholar] [CrossRef] [Green Version]
- Mendis, E.; Rajapakse, N.; Byun, H.-G.; Kim, S.-K. Investigation of jumbo squid (Dosidicus gigas) skin gelatin peptides for their in vitro antioxidant effects. Life Sci. 2005, 77, 2166–2178. [Google Scholar] [CrossRef]
- Priyadarshini, S.; Gopinath, V.; Priyadharsshini, N.M.; MubarakAli, D.; Velusamy, P. Synthesis of anisotropic silver nanoparticles using novel strain, Bacillus flexus and its biomedical application. Colloids Surfaces B Biointerfaces 2013, 102, 232–237. [Google Scholar] [CrossRef]
- Dibrov, P.; Dzioba, J.; Gosink, K.K.; Häse, C.C. Chemiosmotic mechanism of antimicrobial activity of Ag+ in Vibrio cholerae. Antimicrob. Agents Chemother. 2002, 46, 2668–2670. [Google Scholar] [CrossRef] [Green Version]
- Hamouda, T.; Myc, A.; Donovan, B.; Shih, A.Y.; Reuter, J.D.; Baker, J.R. A novel surfactant nanoemulsion with a unique non-irritant topical antimicrobial activity against bacteria, enveloped viruses and fungi. Microbiol. Res. 2001, 156, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sondi, I.; Salopek-Sondi, B. Silver nanoparticles as antimicrobial agent: A case study on E. coli as a model for Gram-negative bacteria. J. Colloid Interface Sci. 2004, 275, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Qamar, S.U.R. Nanocomposites: Potential therapeutic agents for the diagnosis and treatment of infectious diseases and cancer. Colloid Interface Sci. Commun. 2021, 43, 100463. [Google Scholar] [CrossRef]
- Chaloupka, K.; Malam, Y.; Seifalian, A.M. Nanosilver as a new generation of nanoproduct in biomedical applications. Trends Biotechnol. 2010, 28, 580–588. [Google Scholar] [CrossRef]
- Kędziora, A.; Speruda, M.; Krzyżewska, E.; Rybka, J.; Łukowiak, A.; Bugla-Płoskońska, G. Similarities and differences between silver ions and silver in nanoforms as antibacterial agents. Int. J. Mol. Sci. 2018, 19, 444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rai, M.; Kon, K.; Ingle, A.; Duran, N.; Galdiero, S.; Galdiero, M. Broad-spectrum bioactivities of silver nanoparticles: The emerging trends and future prospects. Appl. Microbiol. Biotechnol. 2014, 98, 1951–1961. [Google Scholar] [CrossRef] [PubMed]
- Alshareef, A.; Laird, K.; Cross, R.B.M. Shape-dependent antibacterial activity of silver nanoparticles on Escherichia coli and Enterococcus faecium bacterium. Appl. Surf. Sci. 2017, 424, 310–315. [Google Scholar] [CrossRef]
- Abbas, Q.; Yousaf, B.; Ullah, H.; Ali, M.U.; Zia-ur-Rehman, M.; Rizwan, M.; Rinklebe, J. Biochar-induced immobilization and transformation of silver-nanoparticles affect growth, intracellular-radicles generation and nutrients assimilation by reducing oxidative stress in maize. J. Hazard. Mater. 2020, 390, 121976. [Google Scholar] [CrossRef]
- Jia, Z.; Zhou, W.; Yan, J.; Xiong, P.; Guo, H.; Cheng, Y.; Zheng, Y. Constructing multilayer silk protein/Nanosilver biofunctionalized hierarchically structured 3D printed Ti6Al4 V scaffold for repair of infective bone defects. ACS Biomater. Sci. Eng. 2018, 5, 244–261. [Google Scholar] [CrossRef] [PubMed]
- Marjaneh, R.M.; Rahmani, F.; Hassanian, S.M.; Rezaei, N.; Hashemzehi, M.; Bahrami, A.; Ariakia, F.; Fiuji, H.; Sahebkar, A.; Avan, A. Phytosomal curcumin inhibits tumor growth in colitis-associated colorectal cancer. J. Cell. Physiol. 2018, 233, 6785–6798. [Google Scholar] [CrossRef]
- Ribeiro, A.P.C.; Anbu, S.; Alegria, E.; Fernandes, A.R.; Baptista, P.V.; Mendes, R.; Matias, A.S.; Mendes, M.; da Silva, M.F.C.G.; Pombeiro, A.J.L. Evaluation of cell toxicity and DNA and protein binding of green synthesized silver nanoparticles. Biomed. Pharmacother. 2018, 101, 137–144. [Google Scholar] [CrossRef] [Green Version]
- Talebpour, Z.; Haghighi, F.; Taheri, M.; Hosseinzadeh, M.; Gharavi, S.; Habibi, F.; Aliahmadi, A.; Sadr, A.S.; Azad, J. Binding interaction of spherical silver nanoparticles and calf thymus DNA: Comprehensive multispectroscopic, molecular docking, and RAPD PCR studies. J. Mol. Liq. 2019, 289, 111185. [Google Scholar] [CrossRef]
- Cui, L.; Chen, P.; Chen, S.; Yuan, Z.; Yu, C.; Ren, B.; Zhang, K. In situ study of the antibacterial activity and mechanism of action of silver nanoparticles by surface-enhanced Raman spectroscopy. Anal. Chem. 2013, 85, 5436–5443. [Google Scholar] [CrossRef]
- Laban, G.; Nies, L.F.; Turco, R.F.; Bickham, J.W.; Sepúlveda, M.S. The effects of silver nanoparticles on fathead minnow (Pimephales promelas) embryos. Ecotoxicology 2010, 19, 185–195. [Google Scholar] [CrossRef]
- Choi, O.; Hu, Z. Size dependent and reactive oxygen species related nanosilver toxicity to nitrifying bacteria. Environ. Sci. Technol. 2008, 42, 4583–4588. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Hou, L.; Liu, M.; Newell, S.E.; Yin, G.; Yu, C.; Zhang, H.; Li, X.; Gao, D.; Gao, J. Effects of silver nanoparticles on nitrification and associated nitrous oxide production in aquatic environments. Sci. Adv. 2017, 3, e1603229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wood, C.M. Silver. In Fish Physiology; Elsevier: Amsterdam, The Netherlands, 2011; Volume 31, pp. 1–65. ISBN 1546-5098. [Google Scholar]
- Lekamge, S.; Ball, A.S.; Shukla, R.; Nugegoda, D. The toxicity of nanoparticles to organisms in freshwater. Rev. Environ. Contam. Toxicol. 2018, 248, 1–80. [Google Scholar]
- Kim, S.; Ryu, D. Silver nanoparticle-induced oxidative stress, genotoxicity and apoptosis in cultured cells and animal tissues. J. Appl. Toxicol. 2013, 33, 78–89. [Google Scholar] [CrossRef]
- Van Hoecke, K.; Quik, J.T.K.; Mankiewicz-Boczek, J.; De Schamphelaere, K.A.C.; Elsaesser, A.; Van der Meeren, P.; Barnes, C.; McKerr, G.; Howard, C.V.; Meent, D. Van De Fate and effects of CeO2 nanoparticles in aquatic ecotoxicity tests. Environ. Sci. Technol. 2009, 43, 4537–4546. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, W. Comparison of acute and chronic toxicity of silver nanoparticles and silver nitrate to Daphnia magna. Environ. Toxicol. Chem. 2011, 30, 885–892. [Google Scholar] [CrossRef] [PubMed]
- Cameron, P.; Gaiser, B.K.; Bhandari, B.; Bartley, P.M.; Katzer, F.; Bridle, H. Silver nanoparticles decrease the viability of Cryptosporidium parvum oocysts. Appl. Environ. Microbiol. 2016, 82, 431–437. [Google Scholar] [CrossRef] [Green Version]
- Juganson, K.; Mortimer, M.; Ivask, A.; Pucciarelli, S.; Miceli, C.; Orupold, K.; Kahru, A. Mechanisms of toxic action of silver nanoparticles in the protozoan Tetrahymena thermophila: From gene expression to phenotypic events. Environ. Pollut. 2017, 225, 481–489. [Google Scholar] [CrossRef]
- Greulich, C.; Braun, D.; Peetsch, A.; Diendorf, J.; Siebers, B.; Epple, M.; Köller, M. The toxic effect of silver ions and silver nanoparticles towards bacteria and human cells occurs in the same concentration range. RSC Adv. 2012, 2, 6981–6987. [Google Scholar] [CrossRef]
- Marambio-Jones, C.; Hoek, E. A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J. Nanopart. Res. 2010, 12, 1531–1551. [Google Scholar] [CrossRef]
- Ruparelia, J.P.; Chatterjee, A.K.; Duttagupta, S.P.; Mukherji, S. Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomater. 2008, 4, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramírez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuppusamy, P.; Ichwan, S.J.A.; Parine, N.R.; Yusoff, M.M.; Maniam, G.P.; Govindan, N. Intracellular biosynthesis of Au and Ag nanoparticles using ethanolic extract of Brassica oleracea L. and studies on their physicochemical and biological properties. J. Environ. Sci. 2015, 29, 151–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dietz, K.-J.; Herth, S. Plant nanotoxicology. Trends Plant Sci. 2011, 16, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Al-Huqail, A.A.; Hatata, M.M.; Al-Huqail, A.A.; Ibrahim, M.M. Preparation, characterization of silver phyto nanoparticles and their impact on growth potential of Lupinus termis L. seedlings. Saudi J. Biol. Sci. 2018, 25, 313–319. [Google Scholar] [CrossRef]
- Sun, J.; Wang, L.; Li, S.; Yin, L.; Huang, J.; Chen, C. Toxicity of silver nanoparticles to Arabidopsis: Inhibition of root gravitropism by interfering with auxin pathway. Environ. Toxicol. Chem. 2017, 36, 2773–2780. [Google Scholar] [CrossRef]
- Panda, K.K.; Achary, V.M.M.; Krishnaveni, R.; Padhi, B.K.; Sarangi, S.N.; Sahu, S.N.; Panda, B.B. In vitro biosynthesis and genotoxicity bioassay of silver nanoparticles using plants. Toxicol. In Vitr. 2011, 25, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Engelmann, P.; Bodó, K.; Najbauer, J.; Németh, P. Annelida: Oligochaetes (Segmented Worms): Earthworm immunity, quo vadis? Advances and new paradigms in the omics era. In Advances in Comparative Immunology; Springer: Berlin/Heidelberg, Germany, 2018; pp. 135–159. [Google Scholar]
- Wang, F.; Guan, W.; Xu, L.; Ding, Z.; Ma, H.; Ma, A.; Terry, N. Effects of nanoparticles on algae: Adsorption, distribution, ecotoxicity and fate. Appl. Sci. 2019, 9, 1534. [Google Scholar] [CrossRef] [Green Version]
- Kwak, J., II; Cui, R.; Nam, S.-H.; Kim, S.W.; Chae, Y.; An, Y.-J. Multispecies toxicity test for silver nanoparticles to derive hazardous concentration based on species sensitivity distribution for the protection of aquatic ecosystems. Nanotoxicology 2016, 10, 521–530. [Google Scholar] [CrossRef]
- Naguib, M.; Mahmoud, U.M.; Mekkawy, I.A.; Sayed, A.E.-D.H. Hepatotoxic effects of silver nanoparticles on Clarias gariepinus; Biochemical, histopathological, and histochemical studies. Toxicol. Rep. 2020, 7, 133–141. [Google Scholar] [CrossRef]
- Kakakhel, M.A.; Wu, F.; Sajjad, W.; Zhang, Q.; Khan, I.; Ullah, K.; Wang, W. Long-term exposure to high-concentration silver nanoparticles induced toxicity, fatality, bioaccumulation, and histological alteration in fish (Cyprinus carpio). Environ. Sci. Eur. 2021, 33, 1–11. [Google Scholar] [CrossRef]
- Hammond, S.A.; Carew, A.C.; Helbing, C.C. Evaluation of the effects of titanium dioxide nanoparticles on cultured Rana catesbeiana tailfin tissue. Front. Genet. 2013, 4, 251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hinther, A.; Vawda, S.; Skirrow, R.C.; Veldhoen, N.; Collins, P.; Cullen, J.T.; van Aggelen, G.; Helbing, C.C. Nanometals induce stress and alter thyroid hormone action in amphibia at or below North American water quality guidelines. Environ. Sci. Technol. 2010, 44, 8314–8321. [Google Scholar] [CrossRef] [PubMed]
- Jafari, S.A.; Cheraghi, S.; Mirbakhsh, M.; Mirza, R.; Maryamabadi, A. Employing response surface methodology for optimization of mercury bioremediation by Vibrio parahaemolyticus PG02 in coastal sediments of Bushehr, Iran. CLEAN–Soil Air Water 2015, 43, 118–126. [Google Scholar] [CrossRef]
- Ashokkumar, P.; Loashini, V.M.; Bhavya, V. Effect of pH, Temperature and biomass on biosorption of heavy metals by Sphaerotilus natans. Int. J. Microbiol. Mycol. 2017, 6, 32–38. [Google Scholar]
- Benazir, J.F.; Suganthi, R.; Rajvel, D.; Pooja, M.P.; Mathithumilan, B. Bioremediation of chromium in tannery effluent by microbial consortia. Afr. J. Biotechnol. 2010, 9, 3140–3143. [Google Scholar]
- Mariano, S.; Panzarini, E.; Inverno, M.D.; Voulvoulis, N.; Dini, L. Toxicity, bioaccumulation and biotransformation of glucose-capped silver nanoparticles in green microalgae Chlorella vulgaris. Nanomaterials 2020, 10, 1377. [Google Scholar] [CrossRef]
- Al-Garni, S.M.; Ghanem, K.M.; Ibrahim, A.S. Biosorption of mercury by capsulated and slime layerforming Gram-ve bacilli from an aqueous solution. Afr. J. Biotechnol. 2010, 9, 6413–6421. [Google Scholar]
- Igiri, B.E.; Okoduwa, S.I.R.; Idoko, G.O.; Akabuogu, E.P.; Adeyi, A.O.; Ejiogu, I.K. Toxicity and bioremediation of heavy metals contaminated ecosystem from tannery wastewater: A review. J. Toxicol. 2018, 2018, 2568038. [Google Scholar] [CrossRef]
- Durán, N.; Marcato, P.D.; Alves, O.L.; Da Silva, J.P.S.; De Souza, G.I.H.; Rodrigues, F.A.; Esposito, E. Ecosystem protection by effluent bioremediation: Silver nanoparticles impregnation in a textile fabrics process. J. Nanopart. Res. 2010, 12, 285–292. [Google Scholar] [CrossRef]
- Gao, Z.; Liu, G.G.; Ye, H.; Rauschendorfer, R.; Tang, D.; Xia, X. Facile colorimetric detection of silver ions with picomolar sensitivity. Anal. Chem. 2017, 89, 3622–3629. [Google Scholar] [CrossRef] [PubMed]
- Maruthupandi, M.; Vasimalai, N. Nanomolar detection of L-cysteine and Cu2+ ions based on Trehalose capped silver nanoparticles. Microchem. J. 2021, 161, 105782. [Google Scholar] [CrossRef]
- Hussain, F.; Hafeez, J.; Khalifa, A.S.; Naeem, M.; Ali, T.; Eed, E.M. In vitro and in vivo study of inhibitory potentials of α-glucosidase and acetylcholinesterase and biochemical profiling of M. charantia in alloxan-induced diabetic rat models. Am. J. Transl. Res. 2022, 14, 3824–3839. [Google Scholar]
- Safavi, A.; Ahmadi, R.; Mohammadpour, Z. Colorimetric sensing of silver ion based on anti aggregation of gold nanoparticles. Sens. Actuators B Chem. 2017, 242, 609–615. [Google Scholar] [CrossRef]
- Song, T.; Zhu, X.; Zhou, S.; Yang, G.; Gan, W.; Yuan, Q. DNA derived fluorescent bio-dots for sensitive detection of mercury and silver ions in aqueous solution. Appl. Surf. Sci. 2015, 347, 505–513. [Google Scholar] [CrossRef]
- Wang, H.; Xue, L.; Jiang, H. Ratiometric fluorescent sensor for silver ion and its resultant complex for iodide anion in aqueous solution. Org. Lett. 2011, 13, 3844–3847. [Google Scholar] [CrossRef]


| Nanoparticles | Size (nm) | Dissolution of Silver Ions from AgNPs over Time (ppb) | Ionic Membrane Potential (mV) | ||||
|---|---|---|---|---|---|---|---|
| Type of Nanoparticles | Size of Nanoparticles | Time (h) | Freshwater (%) | Marine Water (%) | Freshwater | Marine Water | References |
| AgNPs | 40 | 0–24 | 24.5 | 30–38 | −25.3 | 16.5 ± 2 | [27] |
| AgNPs | 20 | 0–48 | 1.8–4 | 35–95 | −17.5 | −6.5 ± 1 | [37] |
| AgNPs | 30 | 0–48 | 118 | 15–30 | −19.6 | −15.8 ± 2 | [30] |
| AgNPs | 50 | 0–48 | 0.3–1.4 | 14–23 | −16.2 | −5.6 ± 2 | [37] |
| AgNPs | 30 | 0–48 | 75 | 32.38 | −32.5 | −11.7 ± 1 | [24] |
| AgNPs | 42 | 0–72 | 45 | 20–38 | −17.8 | −15.8 ± 1 | [29] |
| AgNPs | 100 | 0–48 | <0.1 | 0.38–1.25 | −21.7 | −12.6 ± 2 | [37] |
| AgNPs | 20 | 0–96 | 200 | 35–45 | −23.4 | −19.4 ± 1 | [35] |
| AgNPs | 42 | 0–48 | 27 | 40–55 | −16.5 | −2.5 ± 2 | [6] |
| Aquatic Organisms Isolated Strains | NP Size (nm) | Concentration Range | Duration | Uptake Endpoints | Reference | |
|---|---|---|---|---|---|---|
| Diatom | Thalassiosira weissflogii | 10 | 10 mg/L | 48 h | Cellular distribution | [45] |
| Aquatic bacterium | Pseudomonas fluorescens | 30–50 | 0.002–2 mg/L | 24 h | Aggregation by nanoscale film formation | [9] |
| Eastern mud snails, Juvenile hard clams, Grass shrimp, Cordgrass, Biofilms | Vibrio harveyi | 20–80 | 1.62 mg/L | 60 d | Bioaccumulation and trophic transfer | [34] |
| Nematode | Caenorhabditis elegans | <100 | up to 0.5 mg/L | 24 h | Uptake/adsorption to body | [46] |
| 7–25 | 5–50 mg/L | 24 h | Uptake/transgenerational transfer to body | [47] | ||
| Earthworm | Eisenia fetida | 30–50 | 10–20 mg/L | 28 d | Bioaccumulation in a concentration-dependent manner | [48] |
| 10–50 | <0.1 mg/L | 48 h | Unpredictable | [48] | ||
| 29–39 | 1 mg/L | 28 d | Possible body distribution | [49] | ||
| Water flea | Daphnia magna | 40–50 | up to 5 mg/L | 8 h | Uptake and bioaccumulation | [40] |
| Zebrafish embryos | Danio rerio | 5–15 | 1.62 mg/L | 120 h | Uptake in embryos through chorion pore canals | [50] |
| 11.3 | 0.48 mg/L | 21 h | Adsorption to embryos | [51] | ||
| 20–30 | 1 × 10−8–2 × 10−8 mg/L | 24 h | Penetrated skin and blood tube as aggregated particles | [52] | ||
| 20–30 | 0–4 mg/L | 10 d | Bioaccumulation in muscle and intestine | [53] | ||
| 20–30 | 10 mg/L | 48 h | Possible body uptake | [54] | ||
| Eurasian perch | Perca fluviatilis | 30–40 | 0.000063–0.0003 mg/L | 25 h | Possible to adsorb into gill | [55] |
| Rainbow trout | Oncorhynchus mykiss | 5–15 | 10–20 mg/L | 48 h | Cellular compartmentalization, transport over epithelial layers | [56] |
| Japanese medaka | Oryzias latipes | 30–50 | 20 mg/L | 7 d | Bioaccumulation in liver and gill | [57] |
| Zucchini | Cucurbita pepo | 100 | 1000 mg/L | 12 d | Translocation through shoots | [58] |
| Thale cress | Arabidopsis thaliana | 20–80 | 10–20 mg/L | Uptake and accumulation of roots | [59] | |
| Common grass | Lolium multiflorum | 6–25 | 0–40 mg/L | 24 h | Uptake into roots and shoots | [60] |
| Groups | Role of AgNPs in Toxicity/Malfunctioning of Cells/Organ/Organisms | Example | Reference |
|---|---|---|---|
| Protozoa | Ag ions destroy the sporozoites by entering the oocyst and ultimately break the oocyst wall | Cryptosporidium parvum | [154] |
| The effects of protein-coated AgNPs (14.6 nm, Collargol) have shown in the viability, oxidative stress, and gene expression levels of ciliates species | Tetrahymena thermophila | [155] | |
| Monera | AgNPs are highly toxic to bacteria, often associated with ion release and induction of oxidative stress. AgNPs serve as an antibacterial against bacterial tension and thus avoids its horrendous impact | Bacteria | [156,157] |
| Inhibition of bacterial growth increased permeability due to the formation of “pits” | Escherichia coli | [134] | |
| The interaction of the bacterial cell with AgNPs causes Proton Motive Force dissipation leading to the death of the cell | Staphylococcus aureus | [158] | |
| Generation of ROS | Autotrophic nitrifying bacteria | [147] | |
| AgNPs caused toxicity in the membrane when they attached with less than ten nm-sized NPs | Salmonella typhi, Pseudomonas aeruginosa and Vibrio cholera | [159] | |
| Fungi | AgNPs show antifungal activity, which suppresses the growth of fungal cells | Aspergillus sp., Rhizoctonia solani, Sclerotinia sclerotiorum, S. minor | [160] |
| Plant | AgNPs changed/inhibited seed’s germination, the surface area of leaf, morphology, biomass, and growth potential | Spirodela polyrhiza | [161] |
| Metabolic disorders arise, foliar proline accumulation is caused by a decrease in the contents of sugar. Total protein and chlorophyll, elongation of shoots and roots become reduced | Lupinus termis | [162] | |
| Repressed down-regulated induction of auxin receptor-related genetics, gravitropism of root, and reduction in root tips accumulation of auxins | Arabidopsis thaliana | [163] | |
| DNA damages when cytotoxicity enhances at lethal concentration; LC50, i.e., up to 10 mg/L | Allium cepa | [164] | |
| Animals | Apoptosis occurs when AgNPs directly contact the intestinal epithelium. In specific, typhlosole wherein the apoptosis impaired chloragogenous cells have a role like that of the liver invertebrate species or tissue in molluscs and arthropods | Oligochaetes, vertebrates, molluscs, arthropoda | [165] |
| Acute toxicity/cause immobilization | Daphnia magna | [152] | |
| Algae | AgNPs increase toxicity and disrupts the Photosynthetic system, cell metabolism, and cell membrane. The percentage of overall NPs absorbed by algae cells was 21% and 31%, respectively, for both species | Chlorella vulgaris, Raphidocelis subcapitata | [166] |
| AgNPs cause inhibitory effects on algae species | Chlorella vulgaris Dunaliella tertiolecta | [79] | |
| Chronic toxicity/Growth inhibition | Euglena gracilis | [167] | |
| Chronic toxicity/Growth inhibition | Chlamydomonas reinhardtii | [167] | |
| Fishes | AgNPs induce changes in haematology parameters such as the mean corpuscular haemoglobin (MCH) and mean corpuscular volume (MCV) become decreased. In contrast, red blood cells (RBCs) and white blood cells (WBCs) become increased as the concentration (Conc.) of AgNPs increased | Rainbow trout (Oncorhynchus mykiss) | [154] |
| AgNPs decreased the concentration of albumin (Al), globulin (Gl), and total proteins (Tp). In contrast, the concentration of alkaline phosphatase (ALP), Aspartic aminotransferase (AST), Glucose (Glu), Alanine aminotransferase (ALT), and total lipids (Tl) increases. At tissue and cell levels, pyknotic nuclei, proliferation of hepatocytes, cytoplasmic vacuolation, hepatic necrosis, central vein wall rupture, infiltrations of inflammatory cells, melanoma-macrophages aggregation, and apoptotic cells occurs in the liver of AgNPs-exposed fish | Clarias gariepinus | [168] | |
| Acute toxicity/Abnormality | Oryzias latipes | [167] | |
| Recent findings revealed that AgNPs had influenced the fish behaviour at the highest concentration (0.09 mg/L). The bioaccumulation AgNPs was found high in the liver, intestine, gills, and muscles. Moreover, the results revealed that at the highest concentration (0.09 mg/L), the bioaccumulation of AgNPs led to histopathological alterations, including gill damage leading to necrosis | Cyprinus carpio | [169] | |
| Acute toxicity/Abnormality in different functions | Danio rerio | [167] | |
| Amphibians | The influence of AgNPs on stress and thyroid hormones is being studied with tadpole caudal fin cultures in vitro | Lithobates catesbeianus, Rana catesbeiana | [170,171] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramzan, U.; Majeed, W.; Hussain, A.A.; Qurashi, F.; Qamar, S.U.R.; Naeem, M.; Uddin, J.; Khan, A.; Al-Harrasi, A.; Razak, S.I.A.; et al. New Insights for Exploring the Risks of Bioaccumulation, Molecular Mechanisms, and Cellular Toxicities of AgNPs in Aquatic Ecosystem. Water 2022, 14, 2192. https://doi.org/10.3390/w14142192
Ramzan U, Majeed W, Hussain AA, Qurashi F, Qamar SUR, Naeem M, Uddin J, Khan A, Al-Harrasi A, Razak SIA, et al. New Insights for Exploring the Risks of Bioaccumulation, Molecular Mechanisms, and Cellular Toxicities of AgNPs in Aquatic Ecosystem. Water. 2022; 14(14):2192. https://doi.org/10.3390/w14142192
Chicago/Turabian StyleRamzan, Uzma, Waqar Majeed, Abdul Ahad Hussain, Fasiha Qurashi, Safi Ur Rehman Qamar, Muhammad Naeem, Jalal Uddin, Ajmal Khan, Ahmed Al-Harrasi, Saiful Izwan Abd Razak, and et al. 2022. "New Insights for Exploring the Risks of Bioaccumulation, Molecular Mechanisms, and Cellular Toxicities of AgNPs in Aquatic Ecosystem" Water 14, no. 14: 2192. https://doi.org/10.3390/w14142192
APA StyleRamzan, U., Majeed, W., Hussain, A. A., Qurashi, F., Qamar, S. U. R., Naeem, M., Uddin, J., Khan, A., Al-Harrasi, A., Razak, S. I. A., & Lee, T. Y. (2022). New Insights for Exploring the Risks of Bioaccumulation, Molecular Mechanisms, and Cellular Toxicities of AgNPs in Aquatic Ecosystem. Water, 14(14), 2192. https://doi.org/10.3390/w14142192












