Phytoremediation Potential of Selected Ornamental Woody Species to Heavy Metal Accumulation in Response to Long-Term Irrigation with Treated Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Soil and Water Characteristics
2.3. Plant Material
2.4. Sampling and Analysis of Plant Tissues, Soil, and Water
2.4.1. Plant Sampling
2.4.2. Irrigation Water and Soil Sampling
2.5. Chemical Analysis of Irrigation Water, Soil, and Plant Tissues
2.6. Bioconcentration and Translocation Factors, and Comprehensive Bioconcentration Index Analysis
2.7. Data Analysis
3. Results and Discussion
3.1. Chemical Analysis of Irrigation Water and Soil
3.2. Effects of Irrigation with TWW on HM Content in Different Tissues of Woody Trees
3.3. Effects of Irrigation with TWW on HM Bioconcentration and Translocation Factors in Different Parts of Woody Trees
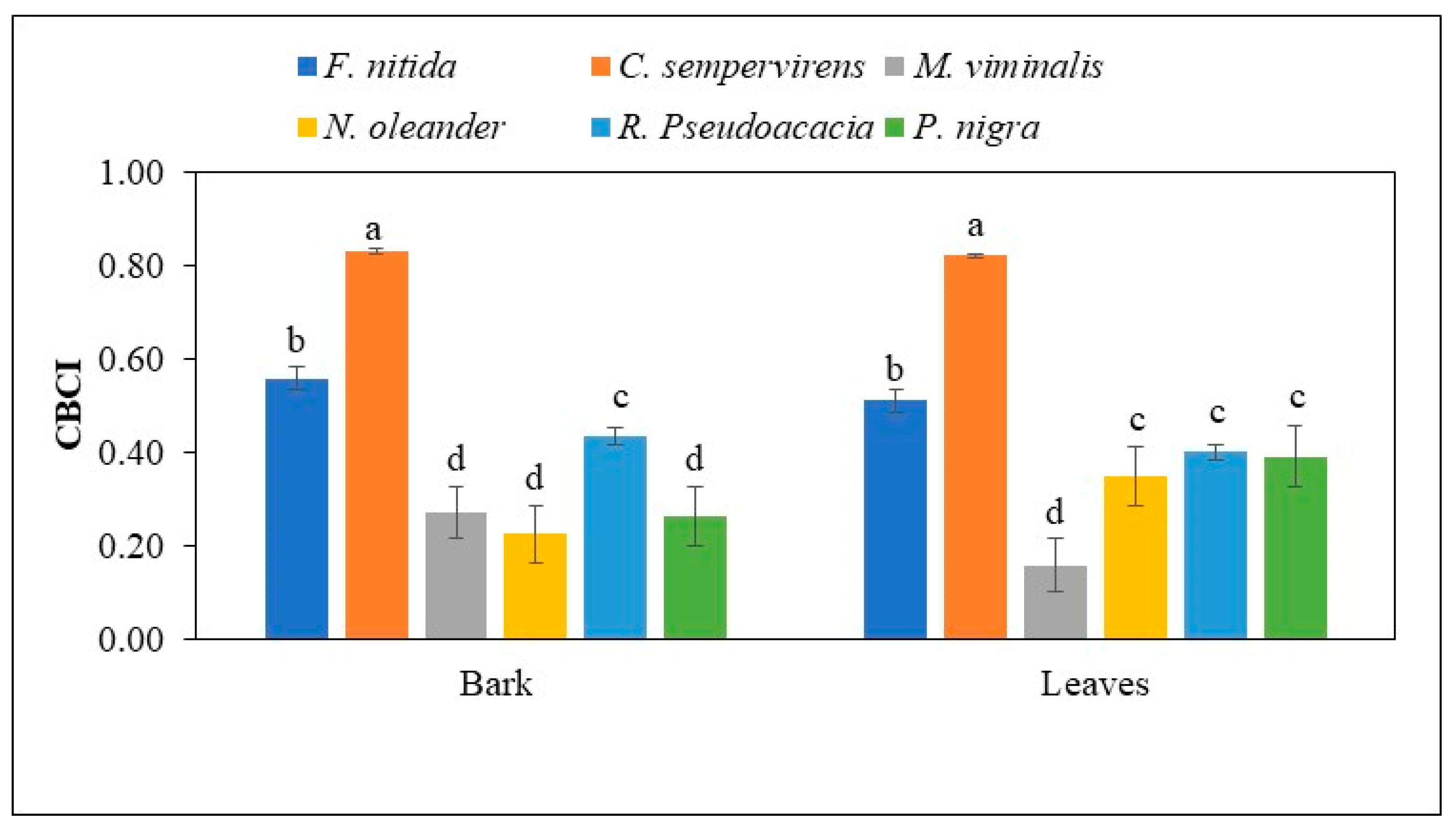
3.4. Comprehensive Metal Accumulation Index for Tree Species
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Le Houérou, H.N. The arid bioclimates in the Mediterranean isoclimatic zone. Ecol. Mediterr. 1982, 8, 103–114. [Google Scholar] [CrossRef]
- FAO. AQUASTAT Core Database. Food and Agriculture Organization of the United Nations. Database. 2020. Available online: https://www.fao.org/aquastat/es/databases/maindatabase/ (accessed on 1 January 2022).
- United Nations Development Programme. Sustainable Development Goals; UNDP Jordan: Amman, Jordan, 2019. [Google Scholar]
- Jackson, R.B.; Carpenter, S.R.; Dahm, C.N.; McKnight, D.M.; Naiman, R.J.; Postel, S.L.; Running, S.W. Water in a changing world. Ecol. Appl. 2001, 11, 1027–1045. [Google Scholar] [CrossRef]
- Yadav, R.K.; Minhas, P.S.; Dagar, J.C. Potential of wastewater disposal through tree plantations. In Agroforestry for the Management of Waterlogged Saline Soils and Poor-Quality Waters, 1st ed.; Dagar, J.C., Minhas, P., Eds.; Springer: New Delhi, India, 2016; pp. 163–179. [Google Scholar] [CrossRef]
- Khresat, S.E.; Al-Bakri, J.; Al-Tahhan, R. Impacts of land use/cover change on soil properties in the Mediterranean region of northwestern Jordan. Land Degrad. Dev. 2008, 19, 397–407. [Google Scholar] [CrossRef]
- Alrababah, M.A.; Bani-Hani, M.G.; Alhamad, M.N.; Bataineh, M.M. Boosting seedling survival and growth under semi-arid Mediterranean conditions: Selecting appropriate species under rainfed and wastewater irrigation. J. Arid Environ. 2008, 72, 1606–1612. [Google Scholar] [CrossRef]
- Al-Assaf, A.A. Applying contingent valuation to measure the economic value of forest services: A case study in Northern Jordan. Int. J. Sustain. Dev. World Ecol. 2015, 22, 242–250. [Google Scholar] [CrossRef]
- Singh, G.; Bhati, M. Growth, biomass production, and nutrient composition of eucalyptus seedlings irrigated with municipal effluent in loamy sand soil of Indian desert. J. Plant Nutr. 2003, 26, 2469–2488. [Google Scholar] [CrossRef]
- Alahabadi, A.; Ehrampoush, M.H.; Miri, M.; Aval, H.E.; Yousefzadeh, S.; Ghaffari, H.R.; Hosseini-Bandegharaei, A. A comparative study on capability of different tree species in accumulating heavy metals from soil and ambient air. Chemosphere 2017, 172, 459–467. [Google Scholar] [CrossRef]
- Justin, M.Z.; Pajk, N.; Zupanc, V.; Zupančič, M. Phytoremediation of landfill leachate and compost wastewater by irrigation of Populus and Salix biomass and growth response. Waste Manag. 2010, 30, 1032–1042. [Google Scholar] [CrossRef]
- Belaida, N.; Neel, C.; Lenain, J.F.; Buzier, R.; Kallel, M.; Ayoub, T.; Ayadi, A.; Bauduc, M. Assessment of metal accumulation in calcareous soil and forage crops subjected to long-term irrigation using treated wastewater: Case of El Hajeb-Sfax, Tunisia. Agric. Ecosyst. Environ. 2012, 158, 83–93. [Google Scholar] [CrossRef]
- Pedrero, F.; Kalavrouziotis, I.; Alarcón, J.J.; Koukoulakis, P.; Asano, T. Use of treated municipal wastewater in irrigated agriculture—Review of some practices in Spain and Greece. Agric. Water Manag. 2010, 97, 1233–1241. [Google Scholar] [CrossRef]
- Mishra, S.; Bharagava, R.N.; More, N.; Yadav, A.; Zainith, S.; Mani, S.; Chowdhary, P. Heavy metal contamination: An alarming threat to environment and human health. In Environmental Biotechnology: For Sustainable Future; Sobti, R., Arora, N., Kothari, R., Eds.; Springer: Singapore, 2019; pp. 103–125. [Google Scholar] [CrossRef]
- Oyewo, O.A.; Adeniyi, A.; Bopape, M.F.; Onyango, M.S. Heavy metal mobility in surface water and soil, climate change, and soil interactions. In Climate Change and Soil Interactions, 1st ed.; Prasad, M.N.V., Pietrzykowski, M., Eds.; Elsevier: Oxford, UK, 2020; pp. 51–88. [Google Scholar] [CrossRef]
- Nkwunonwo, U.C.; Odika, P.O.; Onyia, N.I. A review of the health implications of heavy metals in food chain in Nigeria. Sci. World J. 2020, 2020, 6594109. [Google Scholar] [CrossRef] [PubMed]
- Ibekwe, A.M.; Gonzalez-Rubio, A.; Suarez, D.L. Impact of treated wastewater for irrigation on soil microbial communities. Sci. Total Environ. 2018, 622, 1603–1610. [Google Scholar] [CrossRef] [PubMed]
- Othman, Y.A.; Al-Assaf, A.; Tadros, M.J.; Albalawneh, A. Heavy Metals and Microbes Accumulation in Soil and Food Crops Irrigated with Wastewater and the Potential Human Health Risk: A Metadata Analysis. Water 2021, 13, 3405. [Google Scholar] [CrossRef]
- Adriano, D.C. Bioavailability of trace metals. In Trace Elements in Terrestrial Environments; Springer: New York, NY, USA, 2001; pp. 61–89. [Google Scholar] [CrossRef]
- Liu, Y.; Du, Q.; Wang, Q.; Yu, H.; Liu, J.; Tian, Y.; Lei, J. Causal inference between bioavailability of heavy metals and environmental factors in a large-scale region. Environ. Pollut. 2017, 226, 370–378. [Google Scholar] [CrossRef]
- Jadia, C.D.; Fulekar, M.H. Phytoremediation of heavy metals: Recent techniques. Afr. J. Biotechnol. 2009, 8, 921–928. [Google Scholar]
- Irshad, M.; Ahmad, S.; Pervez, A.; Inoue, M. Phytoaccumulation of heavy metals in natural plants thriving on wastewater effluent at Hattar industrial estate, Pakistan. Int. J. Phytoremediat. 2014, 17, 154–158. [Google Scholar] [CrossRef]
- Vamerali, T.; Bandiera, M.; Coletto, L.; Zanetti, F.; Dickinson, N.M.; Mosca, G. Phytoremediation trials on metal-and arsenic-contaminated pyrite wastes (Torviscosa, Italy). Environ. Pollut. 2009, 157, 887–894. [Google Scholar] [CrossRef]
- Dickinson, N.M.; Punshon, T.; Hodkinson, R.B.; Lepp, N.W. Metal tolerance and accumulation in willows. In Proceedings of the Willow Vegetation Filters for Municipal Wastewater and Sludges, Swedish University of Agricultural Sciences, Uppsala, Sweden, 5–10 June 1994; pp. 121–127. [Google Scholar]
- Chaney, R.L.; Malik, M.; Li, Y.M.; Brown, S.L.; Brewer, E.P.; Angle, J.S.; Baker, A.J. Phytoremediation of soil metals. Curr. Opin. Biotechnol. 1997, 8, 279–284. [Google Scholar] [CrossRef]
- Salt, D.E.; Smith, R.D.; Raskin, I. Phytoremediation. Annu. Rev. Plant Biol. 1998, 49, 643–668. [Google Scholar] [CrossRef]
- Chandra, R.; Kumar, V.; Singh, K. Hyperaccumulator versus nonhyperaccumulator plants for environmental waste management. In Phytoremediation of Environmental Pollutants, 1st ed.; Chandra, R., Dubey, N.K., Kumar, V., Eds.; CRC Press: Boca Raton, FL, USA, 2018; pp. 43–80. [Google Scholar]
- Zhao, X.; Liu, J.; Xia, X.; Chu, J.; Wei, Y.; Shi, S.; Chang, E.; Yin, W.; Jiang, Z. The evaluation of heavy metal accumulation and application of a comprehensive bio-concentration index for woody species on contaminated sites in Hunan, China. Environ. Sci. Pollut. Res. 2014, 21, 5076–5085. [Google Scholar] [CrossRef]
- Ayoub, S.; Al-Shdiefat, S.; Rawashdeh, H.; Bashabsheh, I. Utilization of reclaimed wastewater for olive irrigation: Effect on soil properties, tree growth, yield and oil content. Agric. Water Manag. 2016, 176, 163–169. [Google Scholar] [CrossRef]
- Mcheik, M.; Toufaily, J.; Haj Hassan, B.; Hamieh, T.; Abi Saab, M.T.; Rouphael, Y.; Ferracin, E.; da Shio, B.; Bashabshah, I.; Al Hadidi, L. Reuse of treated municipal wastewater in irrigation: A case study from Lebanon and Jordan. Water Environ. J. 2017, 31, 552–558. [Google Scholar] [CrossRef]
- JISM. Water—Reclaimed Grey Water (JS 1776:2013); Jordan Standards and Metrology Organization: Amman, Jordan, 2013; pp. 1–12. [Google Scholar]
- WHO. Guidelines for the Safe Use of Wastewater, Excreta and Greywater; World Health Organization: Geneva, Switzerland, 2006; Volume 2. [Google Scholar]
- Al-Habahbeh, K.A.; Al-Nawaiseh, M.B.; Al-Sayaydeh, R.S.; Al-Hawadi, J.S.; Albdaiwi, R.N.; Al-Debei, H.S.; Ayad, J.Y. Long-term irrigation with treated municipal wastewater from the wadi-musa region: Soil heavy metal accumulation, uptake and partitioning in olive trees. Horticulturae 2021, 7, 152. [Google Scholar] [CrossRef]
- Duncan, H.J.; Flowers, T.H.; Pulford, I.D.; Wilson, W.D. Analytical procedures for the analysis of heavy metals in contaminated soils. In Proceedings of the Fifth International FZK/TNO Conference on Contaminated Soil, Maastricht, The Netherlands, 30 October–3 November 1995; Springer: Dordrecht, The Netherlands; pp. 731–732. [Google Scholar] [CrossRef]
- Estefan, G.; Sommer, R.; Ryan, J. Methods of Soil, Plant, and Water Analysis: A Manual for the West Asia and North Africa Region, 3rd ed.; ICARDA: Beirut, Lebanon, 2013; pp. 20–199. Available online: https://hdl.handle.net/20.500.11766/7512 (accessed on 1 January 2022).
- Pedrero, F.; Alarcón, J.J. Effects of treated wastewater irrigation on lemon trees. Desalination 2009, 246, 631–639. [Google Scholar] [CrossRef]
- Chapman, H.D. Cation-exchange capacity. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties, 1st ed.; Norman, A.G., Ed.; American Society of Agronomy: Washington, DC, USA, 1965; Volume 9, pp. 891–901. [Google Scholar] [CrossRef]
- Amundson, R.G.; Trask, J.; Pendall, E. A rapid method of soil carbonate analysis using gas chromatography. Soil Sci. Soc. Am. J. 1988, 52, 880–883. [Google Scholar] [CrossRef]
- Atafar, Z.; Mesdaghinia, A.; Nouri, J.; Homaee, M.; Yunesian, M.; Ahmadimoghaddam, M.; Mahvi, A.H. Effect of fertilizer application on soil heavy metal concentration. Environ. Monit. Assess. 2010, 160, 83–89. [Google Scholar] [CrossRef]
- Mellem, J.J. Bioaccumulation of Cr, Hg, As, Pb, Cu and Ni with the ability for hyperaccumulation by Amaranthus dubius. Afr. J. Agric. Res. 2012, 7, 591–596. [Google Scholar] [CrossRef] [Green Version]
- Al-Khashman, O.A. Study of water quality of springs in Petra region, Jordan: A three-year follow-up. Water Resour. Manag. 2007, 21, 1145–1163. [Google Scholar] [CrossRef]
- Bedbabis, S.; Ben Rouina, B.; Boukhris, M.; Ferrara, G. Effect of irrigation with treated wastewater on soil chemical properties and infiltration rate. J. Environ. Manag. 2014, 133, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Bourazanis, G.; Roussos, P.A.; Argyrokastritis, I.; Kosmas, C.; Kerkides, P. Evaluation of the use of treated municipal wastewater on the yield, oil quality, free fatty acids’ profile and nutrient levels in olive trees cv Koroneiki, in Greece. Agric. Water Manag. 2016, 163, 1–8. [Google Scholar] [CrossRef]
- Batarseh, M.I.; Rawajfeh, A.; Ioannis, K.K.; Prodromos, K.H. Treated Municipal Wastewater Irrigation Impact on Olive Trees (Olea Europaea L.) at Al-Tafilah, Jordan. Water Air Soil Pollut. 2010, 217, 185–196. [Google Scholar] [CrossRef]
- Ziadat, A.H.; Jiries, A.; Alojail, I. Accumulation of Heavy Metals on Soil Irrigated with Treated Wastewater at Al al-Bayt University-Jordan. In Proceedings of the 2019 Advances in Science and Engineering Technology International Conferences (ASET), Dubai, United Arab Emirates, 26 March–10 April 2019. [Google Scholar] [CrossRef]
- Manasreh, W.; Alzaydien, A.S. Analysis of Treated Wastewater Produced from Al-Lajoun Wastewater Treatment Plant, Jordan. E-J. Chem. 2009, 6 (Suppl. S1), S287–S303. [Google Scholar] [CrossRef]
- Ye, Y.; Ngo, H.H.; Guo, W.; Chang, S.W.; Nguyen, D.D.; Zhang, X.; Zhang, J.; Liang, S. Nutrient recovery from wastewater: From technology to economy. Bioresour. Technol. Rep. 2020, 11, 100425. [Google Scholar] [CrossRef]
- Bolto, B.; Dixon, D.; Eldridge, R. Ion exchange for the removal of natural organic matter. React. Funct. Polym. 2004, 60, 171–182. [Google Scholar] [CrossRef]
- Gama-Rodrigues, A.C. Soil organic matter, nutrient cycling and biological dinitrogen-fixation in agroforestry systems. Agrofor. Syst. 2011, 81, 191–193. [Google Scholar] [CrossRef]
- Brümmer, G.W. Heavy metal species, mobility and availability in soils. In The Importance of Chemical “Speciation” in Environmental Processes; Springer: Berlin/Heidelberg, Germany, 1986; pp. 169–192. [Google Scholar] [CrossRef]
- Kim, I.S.; Kang, K.H.; Johnson-Green, P.; Lee, E.J. Investigation of heavy metal accumulation in Polygonum thunbergii for phytoextraction. Environ. Pollut. 2003, 126, 235–243. [Google Scholar] [CrossRef]
- Capozzi, F.; Sorrentino, M.C.; Caporale, A.G.; Fiorentino, N.; Giordano, S.; Spagnuolo, V. Exploring the phytoremediation potential of Cynara cardunculus: A trial on an industrial soil highly contaminated by heavy metals. Environ. Sci. Pollut. Res. 2020, 27, 9075–9084. [Google Scholar] [CrossRef]
- Mishra, S.R.; Chandra, R.; Kaila, J. Kinetics and isotherm studies for the adsorption of metal ions onto two soil types. Environ. Technol. Innov. 2017, 7, 87–101. [Google Scholar] [CrossRef]
- Muthusaravanan, S.; Sivarajasekar, N.; Vivek, J.S.; Paramasivan, T.; Naushad, M.; Prakashmaran, J.; Gayathri, V.; Al-Duaij, O.K. Phytoremediation of heavy metals: Mechanisms, methods and enhancements. Environ. Chem. Lett. 2018, 16, 1339–1359. [Google Scholar] [CrossRef]
- Yaghoubian, Y.; Siadat, S.A.; Moradi Telavat, M.R.; Pirdashti, H.; Yaghoubian, I. Bio-removal of cadmium from aqueous solutions by filamentous fungi: Trichoderma spp. and Piriformospora indica. Environ. Sci. Pollut. Res. 2019, 26, 7863–7872. [Google Scholar] [CrossRef]
- Rajkumar, M.; Sandhya, S.; Prasad, M.N.; Freitas, H. Perspectives of plant-associated microbes in heavy metal phytoremediation. Biotechnol. Adv. 2012, 30, 1562–1574. [Google Scholar] [CrossRef]
- Mahler, R.L. Nutrients Plants Require for Growth; CIS; College of Agricultural and Life Science, Iniversity of Idaho: Moscow, ID, USA, 2004; p. 1124. [Google Scholar]
- Dogan, Y.; Unver, M.C.; Ugulu, I.; Calis, M.; Durkan, N. Heavy metal accumulation in the bark and leaves of Juglans regia planted in Artvin City, Turkey. Biotechnol. Biotechnol. Equip. 2014, 28, 643–649. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E.; Møller, I.M.; Murphy, A. Plant Physiology and Development, 4th ed.; Sinauer Associates Incorporated: Sunderland, UK, 2006; pp. 67–86. [Google Scholar]
- Long, X.X.; Yang, X.E.; Ni, W.Z.; Ye, Z.Q.; He, Z.L.; Calvert, D.V.; Stoffella, J.P. Assessing zinc thresholds for phytotoxicity and potential dietary toxicity in selected vegetable crops. Commun. Soil Sci. Plant Anal. 2011, 34, 1421–1434. [Google Scholar] [CrossRef]
- Yang, X.E.; Long, X.X.; Ni, W.Z.; Ye, Z.Q.; He, Z.L.; Stoffella, P.J.; Calvert, D.V. Assessing copper thresholds for phytotoxicity and potential dietary toxicity in selected vegetable crops. J. Environ. Sci. Health B 2002, 37, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Alloway, B.J. Heavy metals and metalloids as micronutrients for plants and animals. In Heavy Metals in Soils; Springer: Dordrecht, The Netherlands, 2013; pp. 195–209. [Google Scholar] [CrossRef]
- Arif, N.; Yadav, V.; Singh, S.; Singh, S.; Ahmad, P.; Mishra, R.K.; Sharma, S.; Tripathi, D.K.; Dubey, N.K.; Chauhan, D.K. Influence of high and low levels of plant-beneficial heavy metal ions on plant growth and development. Front. Environ. Sci. 2016, 4, 69. [Google Scholar] [CrossRef]
- Pinho, S.; Ladeiro, B. Phytotoxicity by Lead as Heavy Metal Focus on Oxidative Stress. J. Bot. 2012, 2012, 369572. [Google Scholar] [CrossRef]
- Anderson, L.L. Assessment of Thelypteris palustris, Asparagus sprengeri, and Lolium perenne for Their Potential Use in the Phytoremediation of Arsenic-Contaminated Soils. Ph.D. Thesis, Louisiana State University, Baton Rouge, LA, USA, 2007. Available online: https://digitalcommons.lsu.edu/gradschool_dissertations/2824/ (accessed on 1 January 2022).
- Liu, H.; Wang, H.; Ma, Y.; Wang, H.; Shi, Y. Role of transpiration and metabolism in translocation and accumulation of cadmium in tobacco plants (Nicotiana tabacum L.). Chemosphere 2016, 144, 1960–1965. [Google Scholar] [CrossRef]
- Albdaiwi, R.N.; Al-Hawadi, J.S.; Al-Rawashdeh, Z.B.; Al-Habahbeh, K.A.; Ayad, J.Y.; Al-Sayaydeh, R.S. Effect of Treated Wastewater Irrigation on the Accumulation and Transfer of Heavy Metals in Lemon Trees Cultivated in Arid Environment. Horticulturae 2022, 8, 514. [Google Scholar] [CrossRef]
- Yoon, J.; Cao, X.; Zhou, Q.; Ma, L.Q. Accumulation of Pb, Cu, and Zn in native plants growing on a contaminated Florida site. Sci. Total Environ. 2006, 368, 456–464. [Google Scholar] [CrossRef]
- Peuke, A.D.; Rennenberg, H. Phytoremediation-Molecular biology, requirements for application, environmental protection, public attention and feasibility. EMBO Rep. 2005, 6, 497–501. [Google Scholar] [CrossRef]
- Saba, G.; Parizanganeh, A.H.; Zamani, A.; Saba, J. Phytoremediation of heavy metals contaminated environments: Screening for native accumulator plants in Zanjan-Iran. Int. J. Environ. Res. 2015, 9, 309–316. [Google Scholar]
- Kaya, G.; Yaman, M. Trace metal concentrations in cupressaceae leaves as biomonitors of environmental pollution. Trace Elem. Electrolytes 2008, 25, 156–164. [Google Scholar] [CrossRef]
- Farahat, E.; Linderholm, H.W. The effect of long-term wastewater irrigation on accumulation and transfer of heavy metals in Cupressus sempervirens leaves and adjacent soils. Sci. Total Environ. 2015, 512–513, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yeo, C.K.; Tan, H.T.W. Ficus stranglers and Melastoma malabathricum: Potential tropical woody plants for phytoremediation of metals in wetlands. Nat. Singap. 2011, 4, 213–226. [Google Scholar]
- Parihar, J.K.; Parihar, P.K.; Pakade, Y.B.; Katnoria, J.K. Bioaccumulation potential of indigenous plants for heavy metal phytoremediation in rural areas of Shaheed Bhagat Singh Nagar, Punjab (India). Environ. Sci. Pollut. Res. 2021, 28, 2426–2442. [Google Scholar] [CrossRef] [PubMed]
- Aksoy, A.; Öztürk, M.A. Nerium oleander L. as a biomonitor of lead and other heavy metal pollution in Mediterranean environments. Sci. Total Environ. 1997, 205, 145–150. [Google Scholar] [CrossRef]
- Hussain, R.O.; Am, H. Phytoextraction of lead from polluted soil by oleander (Nerium Oleander). J. Univ. Duhok 2016, 19, 654–662. [Google Scholar]
- Ibrahim, N.; El Afandi, G. Phytoremediation uptake model of heavy metals (Pb, Cd and Zn) in soil using Nerium oleander. Heliyon 2020, 6, e04445. [Google Scholar] [CrossRef]
- Vural, A. Assessment of Heavy Metal Accumulation in the Roadside Soil and Plants of Robinia pseudoacacia, in Gumushane, Northeastern Turkey. Ekoloji Dergisi. 2013, 22, 1–10. [Google Scholar] [CrossRef]
- Bhargava, A.; Carmona, F.F.; Bhargava, M.; Srivastava, S. Approaches for enhanced phytoextraction of heavy metals. J. Environ. Manag. 2012, 105, 103–120. [Google Scholar] [CrossRef]
- Chen, H.; Peng, Y.; Zhang, Y.; Corlett, R.T. Winter cropping in Ficus tinctoria: An alternative strategy. Sci. Rep. 2015, 5, 16496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pajević, S.; Borišev, M.; Nikolić, N.; Arsenov, D.D.; Orlović, S.; Župunski, M. Phytoextraction of heavy metals by fast-growing trees: A review. Phytoremediation 2016, 3, 29–64. [Google Scholar] [CrossRef]
- Rockwood, D.L.; Naidu, C.V.; Carter, D.R.; Rahmani, M.; Spriggs, T.A.; Lin, C.; Alker, G.R.; Isebrands, J.G.; Segrest, S.A. Short rotation woody crops and phytoremediation: Opportunities for agroforestry? In New Vistas in Agroforestry, 1st ed.; Nair, P.K.R., Rao, M.R., Buck, L.E., Eds.; Springer: Dordrecht, The Netherlands, 2004; Volume 61–62, pp. 51–63. [Google Scholar] [CrossRef]
- Unterbrunner, R.; Puschenreiter, M.; Sommer, P.; Wieshammer, G.; Tlustoš, P.; Zupan, M.; Wenzel, W.W. Heavy metal accumulation in trees growing on contaminated sites in Central Europe. Environ. Pollut. 2007, 148, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Emara, M.M.; Farag, R.S.; Mubarak, M.F.; Ali, S.K. Synthesis of core–shell activated carbon/CaO composite from Ficus Nitida leaves, as an efficient adsorbent for removal of methylene blue. Nanotechnol. Environ. Eng. 2020, 5, 24. [Google Scholar] [CrossRef]
- Giovanelli, A.; De Carlo, A. Micropropagation of Mediterranean cypress (Cupressus sempervirens L.). In Protocols for Micropropagation of Woody Trees and Fruits, 1st ed.; Jain, S.M., Häggman, H., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 93–105. [Google Scholar] [CrossRef]
- Hameed, R.L.; Adil, A.M. Effect of Wounding, Auxins and Cinnamon Extract on the rooting and vegetative growth characteristics of bottle brush plant (Melaleuca viminalis L.) cuttings. Sci. J. Flowers Ornam. Plants 2019, 6, 105–111. [Google Scholar] [CrossRef]
- Sádlo, J.; Vítková, M.; Pergl, J.; Pyšek, P. Towards site-specific management of invasive alien trees based on the assessment of their impacts: The case of Robinia pseudoacacia. NeoBiota 2017, 35, 1. [Google Scholar] [CrossRef] [Green Version]
- Vanden Broeck, A. EUFORGEN Technical Guidelines for Genetic Conservation and Use for Black Poplar (Populus nigra); Bioversity International: Rome, Italy, 2003. [Google Scholar]
| Soil Analysis | Unit | Measurement |
|---|---|---|
| Sand (20–200 µm) | % weight | 77.45 ± 1.45 |
| Silt (2–20 µm) | % weight | 12.98 ± 0.54 |
| Clay (<20 µm) | % weight | 9.58 ± 1.16 |
| EC | dS m−1 | 1.90 ± 0.16 |
| pH | pH unit | 7.93 ± 0.51 |
| OM | g Kg−1 | 12.10 ± 1.28 |
| CaCO3 | g Kg−1 | 15.79 ± 1.09 |
| CEC | (mEq/100 g) | 6.26 ± 0.70 |
| Parameter | TWW | JISM 1 | WHO 2 |
|---|---|---|---|
| pH | 7.61 ± 0.25 | 6.0–9.0 | 6.5–8.0 |
| EC (dS m−1) | 1.85 ± 0.10 | 1.0–3.0 | 0.7–3.0 |
| BOD (mg L−1) | 10.48 ± 1.10 | 60 | 300 |
| COD (mg L−1) | 28.93 ± 1.08 | 120 | 500 |
| TDS (mg L−1) | 829.48 ± 33.42 | <2000 | 450–2000 |
| SAR (ratio) | 8.38 ± 0.86 | 9 | <13 |
| Total Coliforms (MPN/100 mL) | 1.85 ± 0.06 | <10 | <9 |
| Element | TWW | JISM 1 | WHO 2 | Element | Soil |
|---|---|---|---|---|---|
| N (mg L−1) | 14.69 ± 2.36 | 50 | 5–50 | N total (g Kg−1) | 14.2 |
| NO3− (mg L−1) | 42.32 ± 2.19 | 45 | 50 | K (mg Kg−1) | 684.2 |
| PO4−3 (mg L−1) | 16.16 ± 1.93 | 30 | 30 | P (mg Kg−1) | 94.8 |
| K (mg L−1) | 36.50 ± 3.11 | 80 | 80 | Na (mg Kg−1) | 374.1 |
| Mg (mg L−1) | 17.89 ± 1.98 | 60 | 60 | Cl (mg Kg−1) | 784.1 |
| Ca (mg L−1) | 90.04 ± 5.09 | 400 | 230 | Zn (mg Kg−1) | 1.26 |
| Na (mg L−1) | 93.03 ± 2.27 | 230 | 69–207 | Fe (mg Kg−1) | 1.55 |
| Cl (mg L−1) | 134.48 ± 8.16 | 400 | 140–350 | Mn (mg Kg−1) | 7.14 |
| Zn (mg L−1) | 0.81 ± 0.06 | 2 | <2.0 | Cu (mg Kg−1) | 0.30 |
| Fe (mg L−1) | 4.05 ± 0.34 | 5 | 0.1–1.5 | Cr (mg Kg−1) | 0.19 |
| Mn (mg L−1) | 0.02 ± 0.01 | - | 0.2 | Ni (mg Kg−1) | 0.08 |
| Cu (mg L−1) | 0.01 ± 0.01 | - | 0.2 | Cd (mg Kg−1) | 0.03 |
| Cr (mg L−1) | 0.72 ± 0.18 | - | 0.02 | Pb (mg Kg−1) | 2.44 |
| Ni (mg L−1) | 1.99 ± 0.20 | - | 0.2 | ||
| Cd (mg L−1) | 1.38 ± 0.10 | 0.01 | <0.01 | ||
| Pb (mg L−1) | 7.00 ± 1.69 | 5 | <5.0 |
| Part/Species | Zn | Fe | Mn | Cu | Cr | Ni | Cd | Pb |
|---|---|---|---|---|---|---|---|---|
| Roots | ||||||||
| F. nitida | 54.05 a * | 419.38 b | 27.25 ab | 41.25 a | 4.47 a | 4.95 a | 0.16 b | 24.94 cd |
| C. sempervirens | 25.58 b | 120.53 d | 15.59 ab | 7.13 d | 4.08 a | 4.74 ab | 0.18 b | 18.05 de |
| M. viminalis | 15.87 cd | 375.38 b | 11.11 ab | 34.86 b | 4.08 a | 4.37 ab | 0.71 a | 45.64 a |
| N. oleander | 13.46 cd | 360.98 b | 31.90 a | 11.54 d | 3.82 a | 3.38 bc | 0.55 a | 33.80 bc |
| R. pseudoacacia | 19.39 c | 625.53 a | 26.38 ab | 17.92 c | 4.56 a | 3.41 bc | 0.42 a | 36.96 ab |
| P. nigra | 11.09 d | 232.00 c | 8.99 b | 10.60 d | 1.82 b | 2.06 c | 0.36 a | 14.60 e |
| Bark | ||||||||
| F. nitida | 20.65 b | 215.31 a | 14.16 c | 13.55 b | 2.37 b | 3.94 a | 1.81 ab | 10.70 d |
| C. sempervirens | 13.64 c | 227.95 a | 19.45 bc | 20.68 a | 4.08 a | 4.14 a | 1.84 a | 18.79 b |
| M. viminalis | 9.95 d | 110.11 b | 22.50 b | 2.50 d | 0.83 d | 4.14 a | 1.71 ab | 26.43 a |
| N. oleander | 11.14 cd | 191.45 a | 15.30 c | 6.11 c | 2.24 bc | 2.24 b | 0.35 c | 13.45 c |
| R. pseudoacacia | 45.31 a | 119.19 b | 68.89 a | 5.49 c | 1.46 c | 2.35 b | 1.05 b | 15.75 bc |
| P. nigra | 8.64 d | 91.03 c | 7.03 d | 4.08 cd | 2.23 bc | 2.10 b | 0.46 bc | 3.19 e |
| Leaves | ||||||||
| F. nitida | 13.69 a | 277.93 ab | 57.63 b | 17.68 ab | 2.04 a | 4.14 ab | 0.77 a | 11.91 b |
| C. sempervirens | 16.15 a | 326.70 a | 101.86 a | 19.55 ab | 1.73 ab | 4.22 ab | 0.69 a | 22.13 a |
| M. viminalis | 10.83 b | 188.50 c | 38.22 bc | 7.54 c | 1.28 b | 3.73 b | 0.73 a | 19.68 a |
| N. oleander | 17.18 a | 228.06 bc | 48.50 b | 11.38 bc | 2.26 ab | 5.56 a | 0.26 b | 17.93 a |
| R. pseudoacacia | 17.50 a | 252.88 b | 142.19 a | 22.01 a | 1.31 b | 4.30 ab | 0.79 a | 21.66 a |
| P. nigra | 9.55 b | 180.28 c | 29.83 c | 5.56 c | 0.61 c | 4.06 ab | 0.40 b | 6.06 c |
| Part/Species | Zn | Fe | Mn | Cu | Cr | Ni | Cd | Pb |
|---|---|---|---|---|---|---|---|---|
| Roots | ||||||||
| F. nitida | 54.87 a * | 283.84 ab | 3.40 bc | 97.66 a | 38.02 a | 84.57 a | 5.64 bc | 12.72 ab |
| C. sempervirens | 28.04 b | 85.94 c | 3.63 bc | 31.05 cd | 29.23 a | 94.70 a | 2.14 c | 9.87 b |
| M. viminalis | 9.52 c | 291.67 a | 0.83 d | 84.80 ab | 17.78 b | 29.28 b | 35.63 a | 13.73 ab |
| N. oleander | 7.48 c | 279.88 ab | 5.68 a | 31.06 cd | 17.47 b | 52.94 ab | 10.05 bc | 10.87 b |
| R. pseudoacacia | 10.99 c | 346.55 a | 2.87 c | 53.68 bcd | 20.06 ab | 23.28 b | 33.40 a | 10.33 b |
| P. nigra | 26.94 b | 114.37 bc | 3.95 bc | 27.41 d | 9.21 c | 44.92 ab | 24.17 ab | 17.85 a |
| Bark | ||||||||
| F. nitida | 20.96 a | 145.73 ab | 1.77 cd | 42.34 b | 20.19 a | 67.35 a | 65.82 ab | 5.46 b |
| C. sempervirens | 14.95 a | 162.53 a | 4.53 ab | 90.09 a | 29.21 a | 82.75 a | 61.25 ab | 10.27 a |
| M. viminalis | 5.97 b | 85.56 bc | 1.68 d | 7.61 d | 3.60 d | 27.76 ab | 85.63 a | 7.95 a |
| N. oleander | 6.19 b | 148.44 ab | 2.73 b | 16.44 bcd | 10.24 b | 35.10 ab | 6.41 c | 4.32 b |
| R. pseudoacacia | 25.69 a | 66.03 bc | 7.49 a | 16.44 bcd | 6.44 c | 16.04 b | 84.00 a | 4.40 b |
| P. nigra | 20.99 a | 44.88 c | 3.09 bc | 10.54 cd | 11.28 b | 45.90 ab | 30.83 b | 3.90 b |
| Leaves | ||||||||
| F. nitida | 13.90 ab | 188.10 a | 7.20 c | 55.23 ab | 17.34 a | 70.73 a | 27.91 b | 6.08 b |
| C. sempervirens | 17.70 ab | 232.94 a | 23.74 a | 85.19 a | 12.42 a | 84.35 a | 22.92 b | 12.10 a |
| M. viminalis | 6.50 c | 146.46 ab | 2.85 d | 22.96 b | 5.56 b | 24.96 b | 36.25 b | 5.92 b |
| N. oleander | 9.56 bc | 176.83 ab | 8.64 c | 30.62 b | 10.33 ab | 87.25 a | 4.68 c | 5.76 b |
| R. pseudoacacia | 9.92 bc | 140.10 ab | 15.46 b | 65.96 ab | 5.78 b | 29.35 b | 63.00 a | 6.05 b |
| P. nigra | 23.21 a | 88.87 b | 13.10 b | 14.36 c | 3.08 c | 88.74 a | 26.50 b | 7.41 b |
| Part/Species | Zn | Fe | Mn | Cu | Cr | Ni | Cd | Pb |
|---|---|---|---|---|---|---|---|---|
| Bark | ||||||||
| F. nitida | 0.38 d * | 0.52 b | 0.52 b | 0.34 b | 0.53 b | 0.87 a | 12.49 b | 0.46 b |
| C. sempervirens | 0.54 cd | 1.92 a | 1.31 ab | 3.00 a | 1.00 a | 0.90 a | 27.63 a | 1.06 a |
| M. viminalis | 0.63 bc | 0.30 bc | 2.12 ab | 0.07 b | 0.21 b | 1.00 a | 2.80 c | 0.62 b |
| N. oleander | 0.83 b | 0.54 b | 0.48 b | 0.53 b | 0.60 b | 0.66 a | 0.64 c | 0.41 bc |
| R. pseudoacacia | 2.38 a | 0.19 c | 2.75 a | 0.32 b | 0.34 b | 0.69 a | 2.76 c | 0.43 c |
| P. nigra | 0.80 b | 0.40 bc | 0.83 ab | 0.38 b | 1.21 a | 1.03 a | 1.30 c | 0.22 bc |
| Leaves | ||||||||
| F. nitida | 0.26 d | 0.69 bc | 2.12 b | 0.44 d | 0.46 a | 0.91 c | 5.43 ab | 0.52 b |
| C. sempervirens | 0.64 c | 2.76 a | 6.73 a | 2.72 a | 0.43 a | 0.91 c | 10.40 a | 1.24 a |
| M. viminalis | 0.70 c | 0.51 bc | 3.37 b | 0.22 d | 0.32 a | 0.88 c | 1.28 b | 0.46 b |
| N. oleander | 1.28 a | 0.64 bc | 1.53 b | 1.00 bc | 0.63 a | 1.65 ab | 0.48 b | 0.53 b |
| R. pseudoacacia | 0.95 b | 0.41 c | 5.56 a | 1.24 b | 0.31 a | 1.27 bc | 1.95 b | 0.59 b |
| P. nigra | 0.85 bc | 0.78 b | 3.27 b | 0.51 cd | 0.32 a | 1.96 a | 1.37 b | 0.40 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Sayaydeh, R.S.; Al-Hawadi, J.S.; Al-Habahbeh, K.A.; Al-Nawaiseh, M.B.; Albdaiwi, R.N.; Ayad, J.Y. Phytoremediation Potential of Selected Ornamental Woody Species to Heavy Metal Accumulation in Response to Long-Term Irrigation with Treated Wastewater. Water 2022, 14, 2086. https://doi.org/10.3390/w14132086
Al-Sayaydeh RS, Al-Hawadi JS, Al-Habahbeh KA, Al-Nawaiseh MB, Albdaiwi RN, Ayad JY. Phytoremediation Potential of Selected Ornamental Woody Species to Heavy Metal Accumulation in Response to Long-Term Irrigation with Treated Wastewater. Water. 2022; 14(13):2086. https://doi.org/10.3390/w14132086
Chicago/Turabian StyleAl-Sayaydeh, Rabea S., Jehad S. Al-Hawadi, Khaled A. Al-Habahbeh, Mohamed B. Al-Nawaiseh, Randa N. Albdaiwi, and Jamal Y. Ayad. 2022. "Phytoremediation Potential of Selected Ornamental Woody Species to Heavy Metal Accumulation in Response to Long-Term Irrigation with Treated Wastewater" Water 14, no. 13: 2086. https://doi.org/10.3390/w14132086
APA StyleAl-Sayaydeh, R. S., Al-Hawadi, J. S., Al-Habahbeh, K. A., Al-Nawaiseh, M. B., Albdaiwi, R. N., & Ayad, J. Y. (2022). Phytoremediation Potential of Selected Ornamental Woody Species to Heavy Metal Accumulation in Response to Long-Term Irrigation with Treated Wastewater. Water, 14(13), 2086. https://doi.org/10.3390/w14132086






