4.1. Hydrochemical Characteristics of Thermal and Non-Thermal Groundwater
Generally, the low concentrations of hydrochemical parameters show that water-rock interaction is not intense [
31]. The considerable spatial inconsistency in EC and TDS concentrations proposed that groundwater chemistry was not homogeneous and was controlled by different processes [
32]. The isotopic and hydrochemical composition of groundwater during their ascent to the ground surface may have been altered because of the steam separation caused by adiabatic expansion and dilution by mixing with shallow groundwater [
12]. As described earlier in
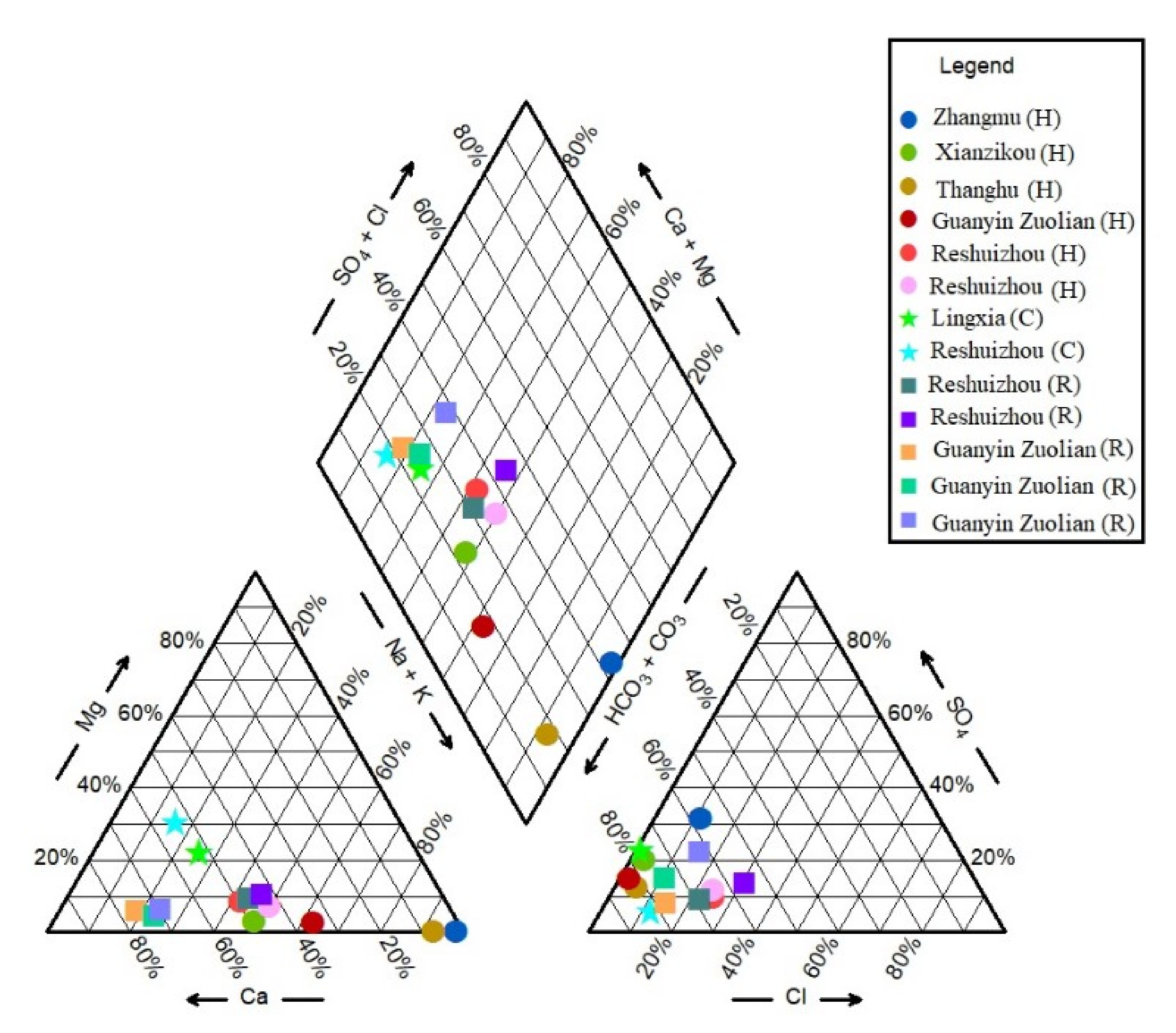
Figure 3, the dominant water type is HCO
3-Ca-Na. Due to the variability in hydrochemistry, however, the Na-HCO
3-SO
4 type can be seen exhibiting approximately 32% SO
4. RSZ04-1 belongs to the Na-Ca-HCO
3-Cl type. Coldwater sample RSZ02-1 belongs to the Ca-Mg-HCO
3 type containing Mg content up to 25%.
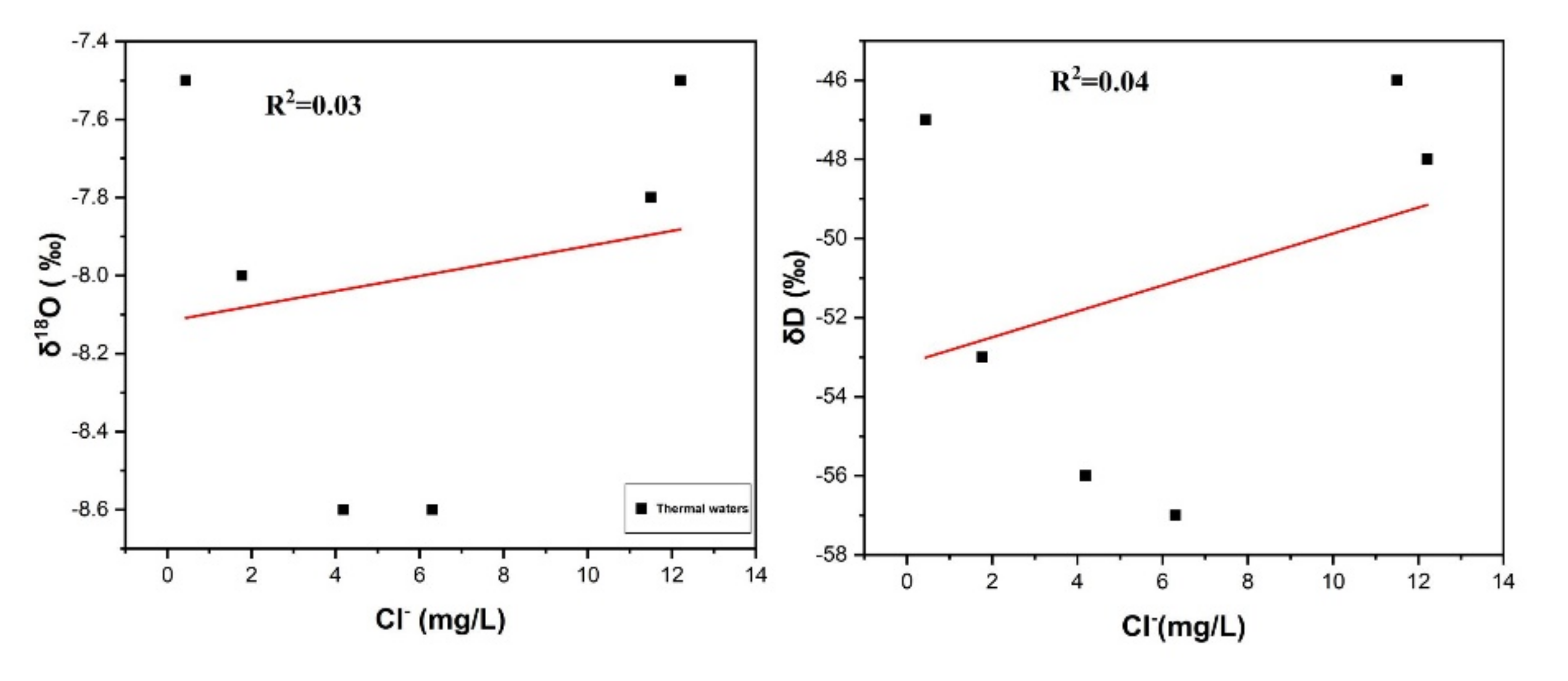
Cl
− is an important tool for assessing the mixing of geothermal water with cold groundwater in upflow zones of geothermal water systems. The correlation of δD and δ
18O with Cl
− is insignificant confirming the mixing of geothermal waters with shallow cold waters [
33,
34], as shown in
Figure 5. If the steam separation/evaporation process was dominant, then Cl
− enrichment would have been observed, but in the present study, low Cl
− contents minimized this possibility and supports the mixing process [
35,
36].
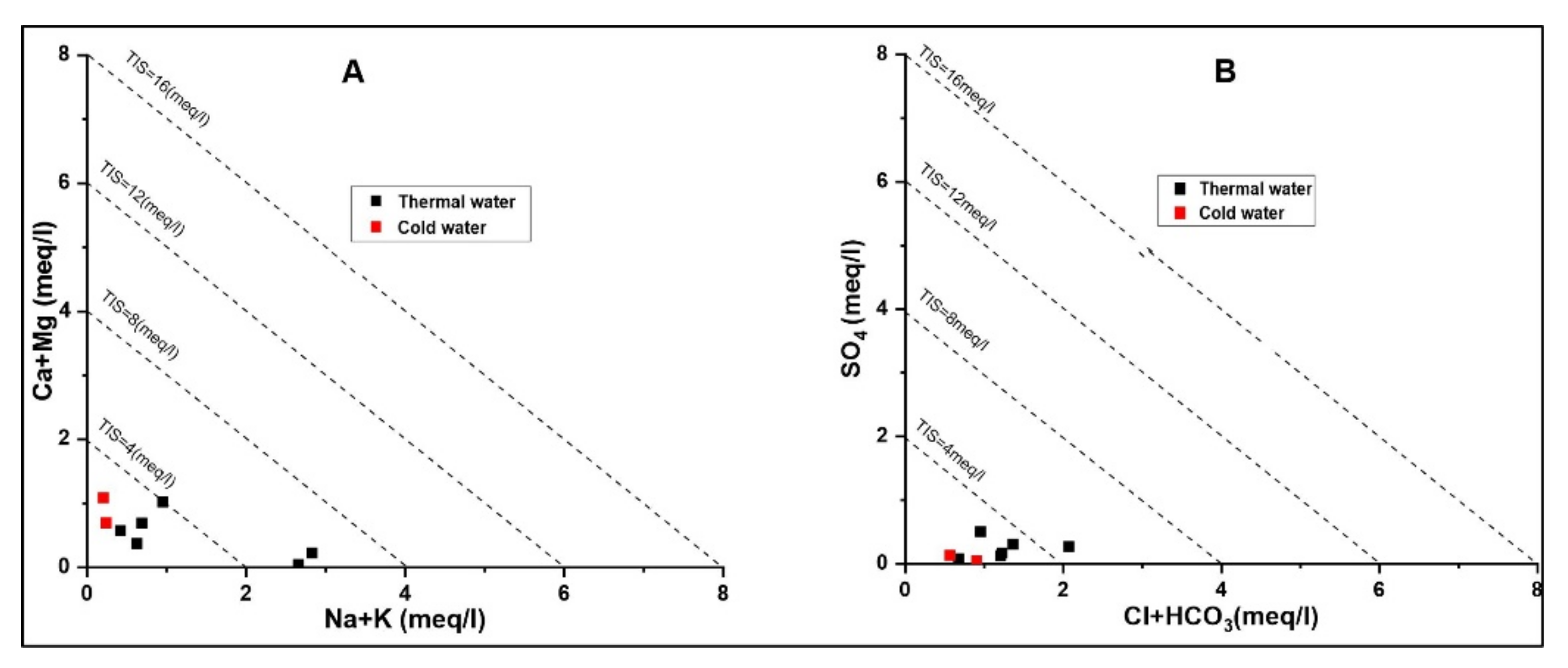
The TIS salinity plot is also an important index used for the classification of hydrochemistry where the (
Figure 6A) Ca + Mg vs. Na + K diagram shows the low salinity with minimum variations. Usually the high Ca and Mg concentrations are likely due to re-equilibration or mixing with Ca and Mg rich cold waters during infiltration of thermal waters [
37]. Furthermore, SO
4 vs. Cl + HCO
3 in the TIS salinity plot shows that the thermal and cold waters accounted for low salinity content (
Figure 6B).
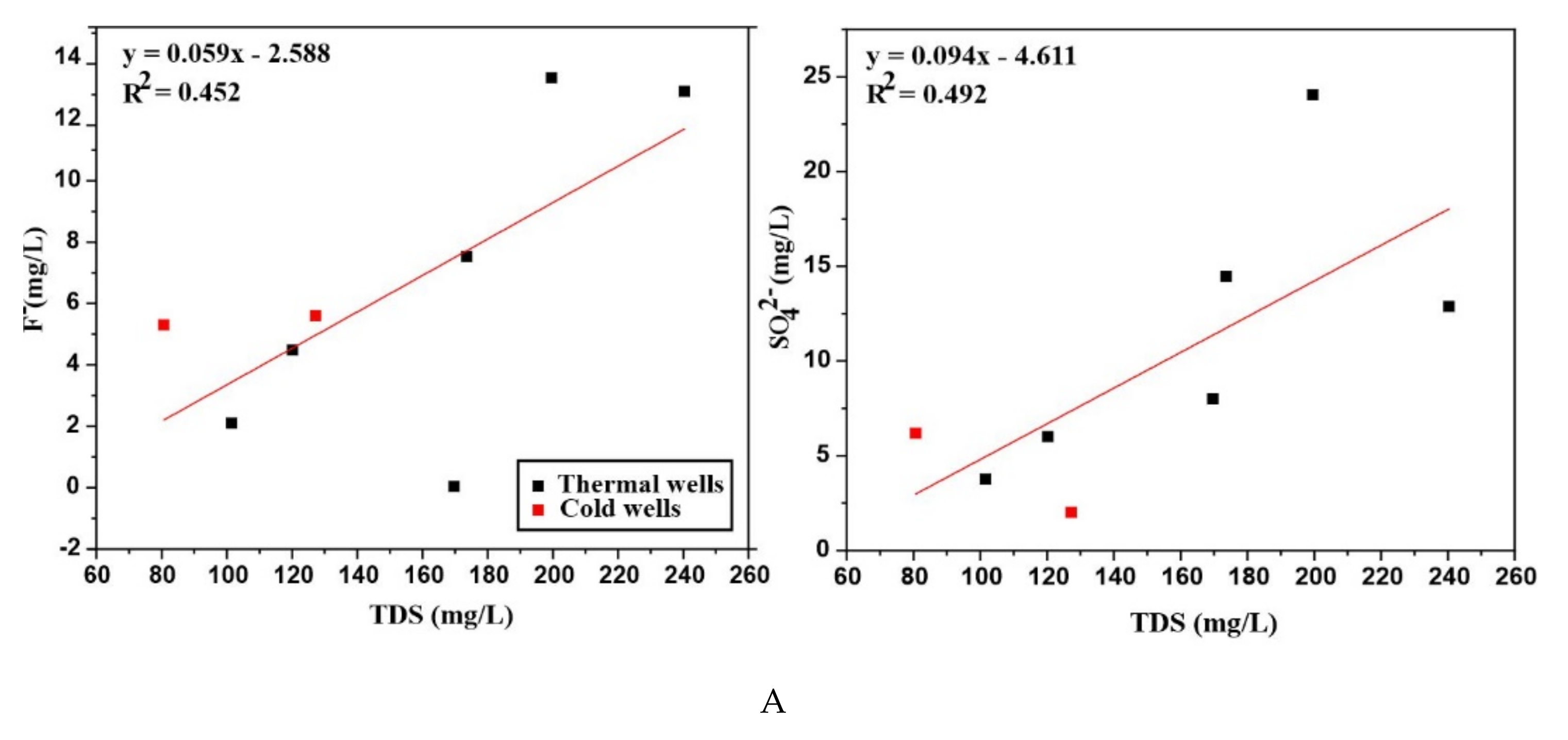
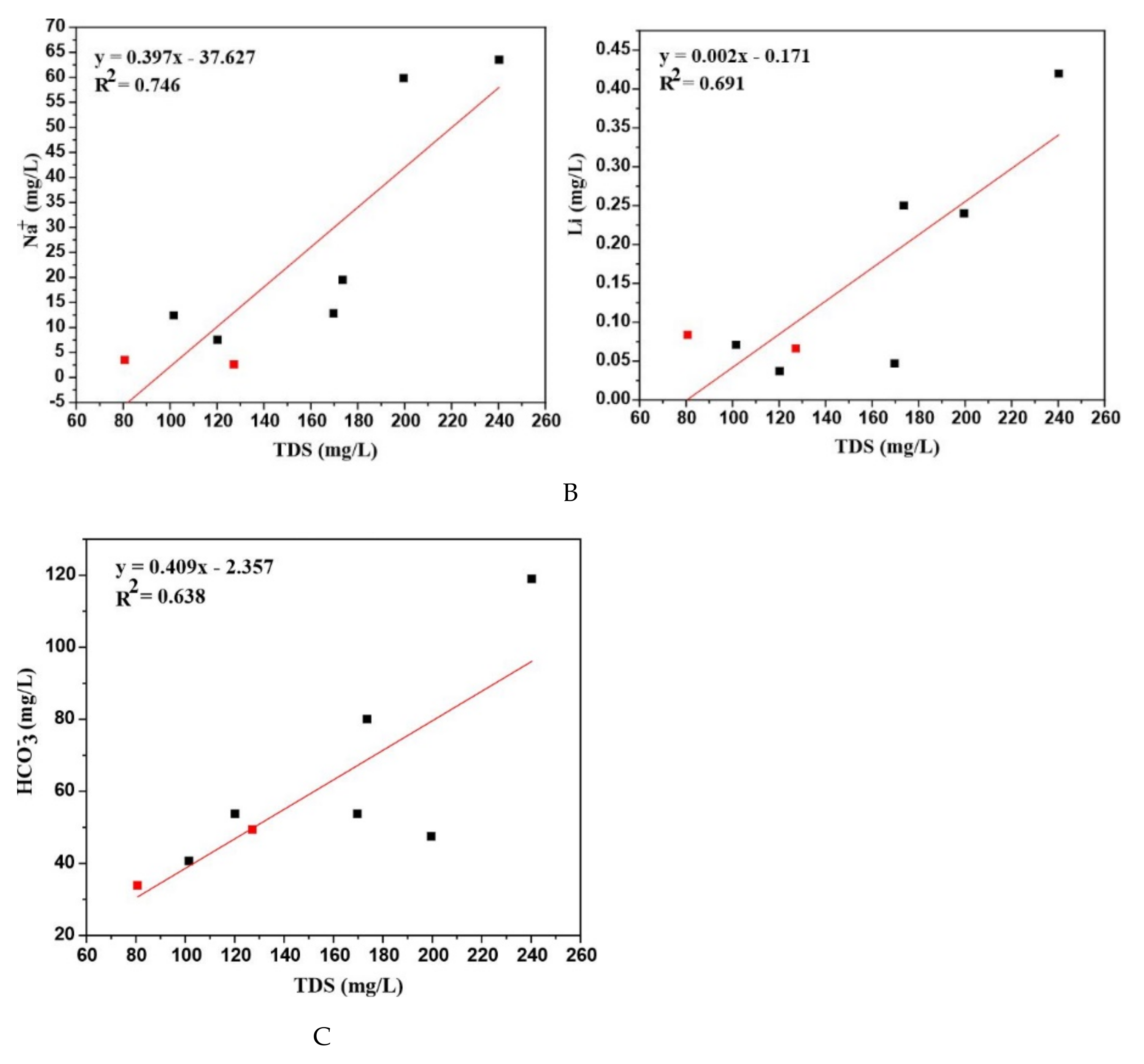
The correlation of various parameters is shown in (
Figure 7A–C). Na
+ exhibits a good linear correlation with R
2 = 0.746 when plotted against TDS. The little elevation in SO
42− might reflect the oxidation of hydrogen sulfide (H
2S) or other sulfide minerals involved in the formation of ore deposits and shows an agreeable correlation with TDS. Even though the continuing dissolution of gypsum and anhydrite as well as the interaction with evaporite deposits also leads to a possibility of elevated sulfate content [
38], simultaneously oxygen concentration should be increased by the shallow water table to promote the sulfide oxidation and hence the SO
42− concentration would be increased [
39]. All the water samples are dominated by HCO
3− ions and show higher pH > 6 rather than CO
3 ions that affect the pH of water. Because at low pH< 6 the carbonates occur mainly in the form of weak H
2CO
3 caused by the degassing of CO
2 [
34] and it can also reduce the pH of natural waters [
40]. The HCO
3− content of the cold water (RSZ02-1) and few thermal waters indicate the dilution by the HCO
3− content of shallow groundwater during upward flow. Nevertheless, the HCO
3− ion shows a good linear correlation with TDS.
The concentration of dissolved silica (SiO
2) measured for RSZ04-1 is the highest among all other thermal and non-thermal groundwater. But overall SiO
2 content is low, which is a sign of its dependence on host rock types because most probably its high content in the case of thermal groundwater could be reported in granitic rock types [
41]. The lowest concentrations of Li and Sr reported in the present study show the existence of different processes. Li shows a noticeable correlation. (
Figure 7B). One of the possibilities of low Sr concentration is that it might be controlled by the precipitation of calcite minerals [
42]. Volcanic rocks are possibly the main source of alkali metal (Li) in thermal groundwater, as it can be accumulated in different acidic rocks, particularly at the end of magma crystallization. The higher concentrations of Li and Sr are generally associated with deep circulating magmatic waters [
43,
44]. On the whole, fluoride concentration is very high and overreached the standards of World Health Organization (WHO) [
45]. In most of the groundwaters, the high fluoride content generally shows a function of pH > 6 and temperature > 50 °C [
5]. The granitic rock type in the study area satisfied the high content of fluoride due to the water-rock interaction between granite-bearing fluoride minerals and thermal waters. This is because fluoride is mainly derived from the weathering of silicate minerals and the existence of granitic rocks amalgamating with fluorite minerals defines its high concentration [
46]. Although there is another process by which atmospheric deposition results in dust particles in the form of soil but the significant source is the weathering of soil minerals [
47,
48].
Based on TDS values, it can be concluded that all the groundwater (thermal and non-thermal) belong to freshwater as (TDS < 1000 ppm) [
49,
50]. Normally, the higher values of TDS and EC for groundwater are probably the index of elevated ionic concentrations; this is because of intense weathering conditions resulting in the maximum contents of dissolved minerals. The mutual dependence of both the quantities is described by [
40]. TDS is a function of 0.6 times EC, as expressed below.
4.2. Isotopes of H and O and Origin of Thermal Groundwaters
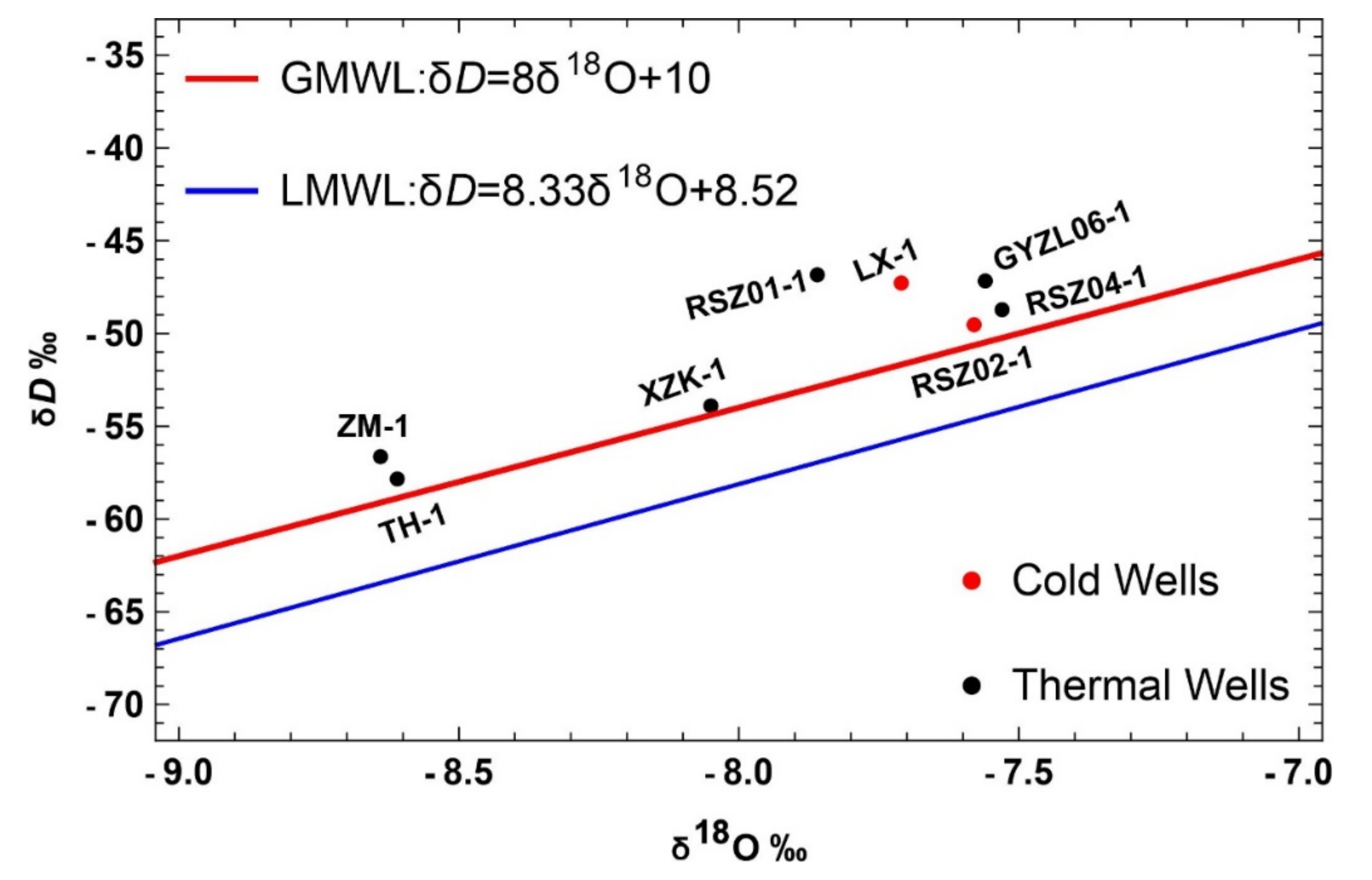
H and O isotopes for geothermal and cold water samples are plotted in
Figure 4. It is quite clear from the graph that no such deviation was observed from the two standard representative lines. The position of all the water samples close to the global meteoric water line (GMWL) [
51] and local meteoric water line (LMWL) of Jiangxi province [
52], with no deviation observed [
53], undoubtedly confirmed their meteoric type of origin. Another piece of evidence in support of the meteoric origin is that the concentration of environmental isotopes in the case of oceanic origin is around 0% (VSMOW), salinity ranges from 33,500 ppm to 37,600 ppm, and chloride is about 19,300 ppm [
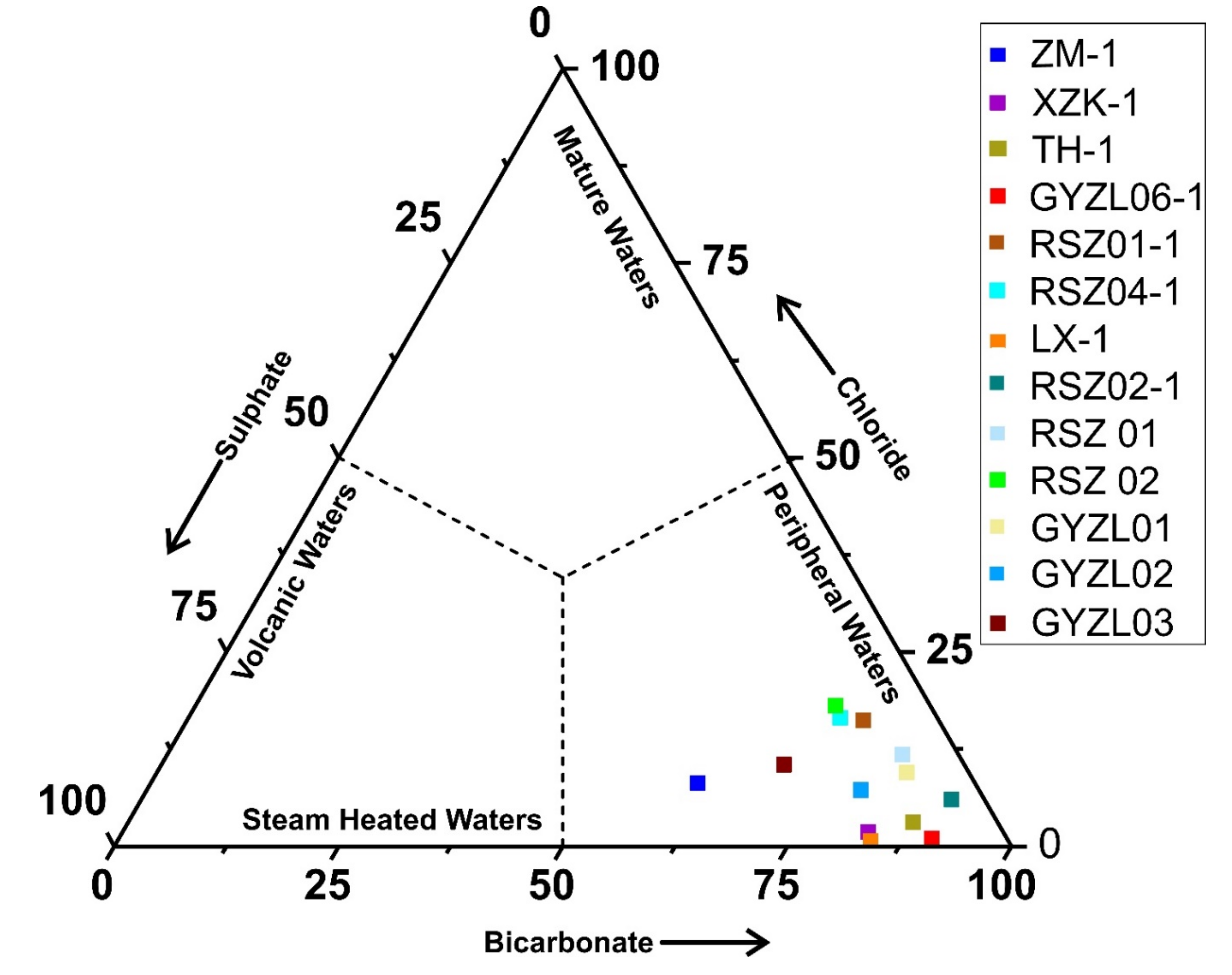
54]. The placement of all the water samples in a triangular plot shows only one dominant water type and that is HCO
3− with a low concentration of chlorine (
Figure 8). The analysis of the point position comparison among water samples confirms the interference of peripheral water characteristics and it increased up to a considerable level during the process of heat storage to the seepage of the upsurge of underground hot water and it is thus speculated that the mixing effect is enhanced.
There are no signs of volcanic or magmatic water origin or mixing due to the lack of notable δ
18O, δD content, which is +6 to +9‰ and −40 to −80‰ in the case of magmatic water, respectively [
12]. These facts confirm the meteoric origin of the Suichuan geothermal waters. Oxygen shift regarding geothermal waters is promoted by the duration of contact [
55]. In the present study, there was no significant phenomena observed, and the degree of closeness with no sample falling below the meteoric water lines excluded the probability of “oxygen-18 shift”.
4.3. Reservior Temperature
The Na-K-Mg diagram was used to assess the equilibrium condition between water and fluid-rock interaction, the degree of mixing, and the suitability of hydrothermal groundwater for geothermometry to measure reservoir temperature (
Figure 9) [
25]. Except two of the geothermal waters, the rest of all the water samples fall at the extreme right corner of the triangular plot showing that they are “immature waters”. The immaturity or disequilibrium of water samples showed the possible mixing with shallow cold water, rapid flow speed, and shallow depth of circulation [
56]. The mixing might have been triggered by the processes of hydrogen metasomatism, minor peripheral neutralization of CO
2, and also the involvement of potassium metasomatism as thermal water rises to the ground surface [
2]. ZM-1 and TH-1 are located in the partially equilibrated zone, showing that they did not achieve complete equilibrium due to the dilution process or perhaps because of the weak degree of water-rock interactions [
57]. These thermal water samples indicate “mature waters”, showing that they have not experienced significant mixing processes that could have resulted in notable alterations in chemical contents. The presence of carbonate rock types indicate the use of a Ca/Mg geothermometer because carbonate-evaporite rocks are the host of a distinct environment [
58]. The comparison of the results of the Ca/Mg geothermometer with the other geothermometers would provide a different approach to study the aqueous environments. However, in the present study, cations and silica geothermometers are applied to estimate the reservoir temperatures at which thermal groundwater last equilibrated with the surrounding environment. The application of chemical geothermometers accounted for a distinct range of temperatures. Different geothermometers calculated various temperature values (
Table 5), which are the signatures of different mineral phases. The range of reservoir temperature estimated by using the Na-K geothermometer was 700 °C to 8867 °C, which is extremely high. This thermometer is not suitable for the present study, as it depends on the Na/K ratios that rely on the dissolution balance of albite and k-feldspar minerals, usually in the case of high temperature geothermal systems while for low temperature reservoirs this ratio is dependent on the dissolution of host rock types [
59]. Few of the temperature calculations using chalcedony are lower than that of the measured temperatures. Therefore a chalcedony thermometer was not considered. Quartz and improved silica geothermometers show quite reasonable estimations, except that one of the samples yielded a lower temperature than the measured value. Because a quartz geothermometer using between the temperature range 150–225 °C might provide reliable results [
23] and improved SiO
2 show an error of about 2–3% when the temperature range is 20–210 °C [
24]. The silica content is low because of the mixing effect between thermal and cold waters, therefore its estimations are the minimum reservoir temperatures.
In the hydrothermal environment, the lithium content is likely scarce because it acts as a “soluble element”, and therefore the Na/Li ratio is barely altered during the upward flow of thermal fluids showing reasonable estimations [
26]. An Na-Li thermometer may provide a good range of deep temperatures, but in comparison with other thermometers, it can be concluded that this geothermometer yielded slightly higher reservoir temperatures. The temperature evaluation by Na-K-Ca-Mg falls between 94.80 °C and 120.35 °C, which are reasonable measurements probably showing no signatures of mixing activities. The temperature calculations carried out by the K-Mg geothermometer are within the scope of the present study because this geothermometer applies to medium-low geothermal fields, but the minimum involvement of clinochlore in chlorites might cause oddness in reservoir temperatures [
60]. K/Mg temperature estimations show that Na and Ca did not achieve water-rock equilibrium as well as revealing the fact that thermal water during the upward flow to the surface has not experienced any fluid-rock equilibrium reactions [
61].
The circulation depth influences the different salient features of hydrothermal systems. It can bring significant abnormalities in deep reservoir temperatures and the concentration of different solutes. It is one of the most reliable and important criteria that estimates the hydrothermal reservoir potential, the origin of thermal groundwater, and also the mechanism of the formation of hydrothermal water resources. The maximum circulation depth was estimated for RSZ04-1 locality (
Table 6).
4.4. Mineral Saturation Dynamics
Usually the decrease in the temperature of hydrothermal waters due to mixing with shallow waters or the decline in pressure possibly creates an appropriate environment for the precipitation of different minerals like carbonates, sulfate silicates, and others [
10]. The thermodynamic equilibrium saturation index is a standard that shows the possible saturation or precipitation attributes of an aqueous and terrestrial environment based on hydrochemical parameters [
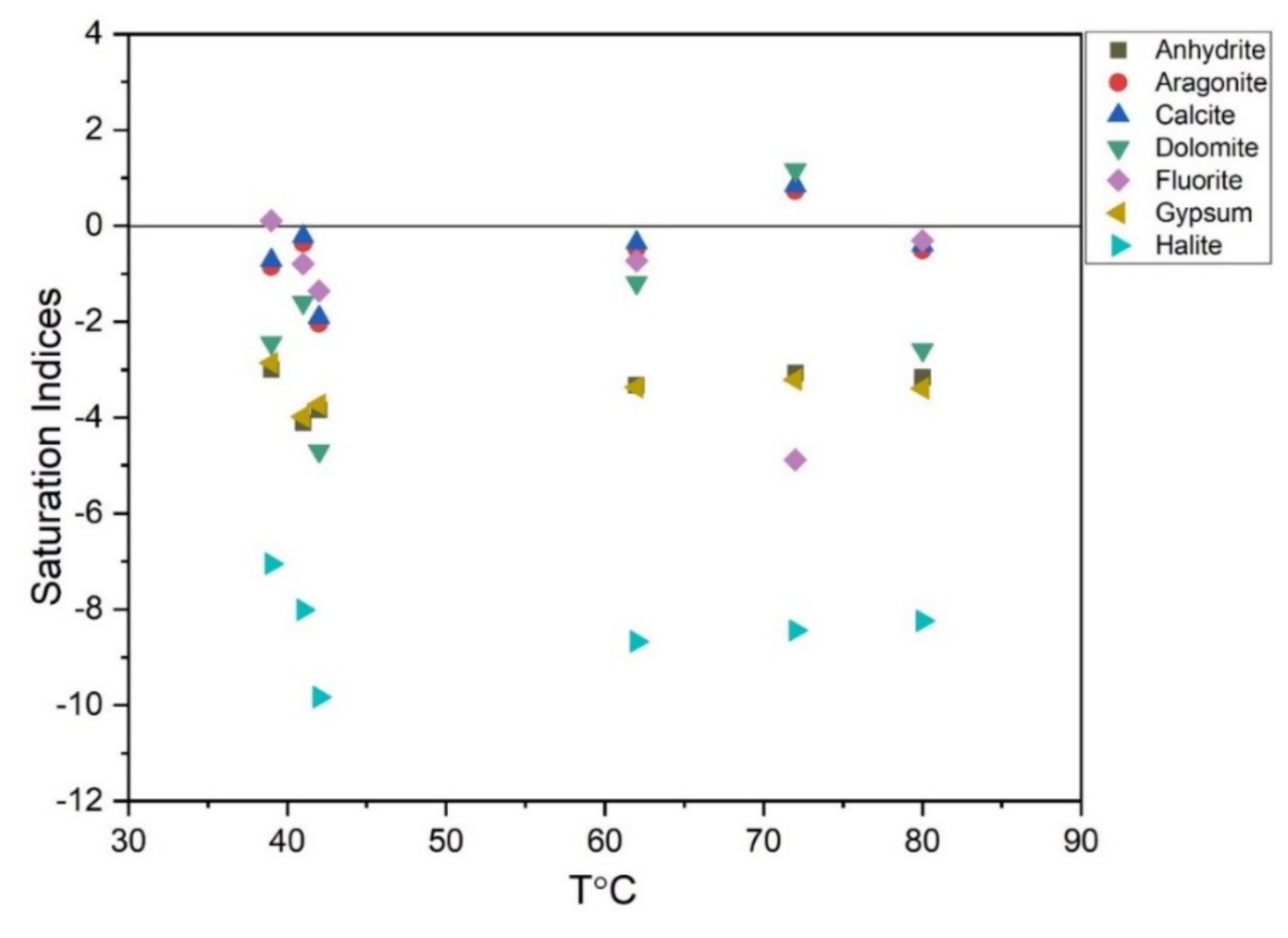
62]. Furthermore, it defines whether the water is corrosive or can cause scaling. The range that explains this phenomenon is −0.3 to +0.3. Water, having a saturation index of less than −0.3, will be corrosive and above +0.3 it will cause scaling that in turn creates a huge problem in the exploitation and utilization of thermal waters. The graphical representation in (
Figure 10) and (
Table 4) for thermal water saturation indices (Anhydrite, Aragonite, Calcite, Dolomite, Fluorite, Gypsum, and Halite) showed the unsaturation state of the respective water samples, as most of the minerals are distributed below the equilibrium line. The water sample XZK-1 might have corrosive characteristics concerning fluorite minerals, as it falls very close to the equilibrium standard line. This reveals the maximum chances of F
− to release from fluorite into the deep circulation of underground hydrothermal fluids [
7]. Similarly, notable variations have been observed in RSZ04-1, as it yields the characteristics of the supersaturation of aragonite, calcite, and dolomite minerals and an estimated SI of >+0.3, which can cause scaling problems. The present situation explains the dominance of certain environments that confirm the abundance of these carbonate minerals, as its concentration in natural waters is a function of dissolved carbon dioxide, temperature, pH, cations, and other dissolved salts.
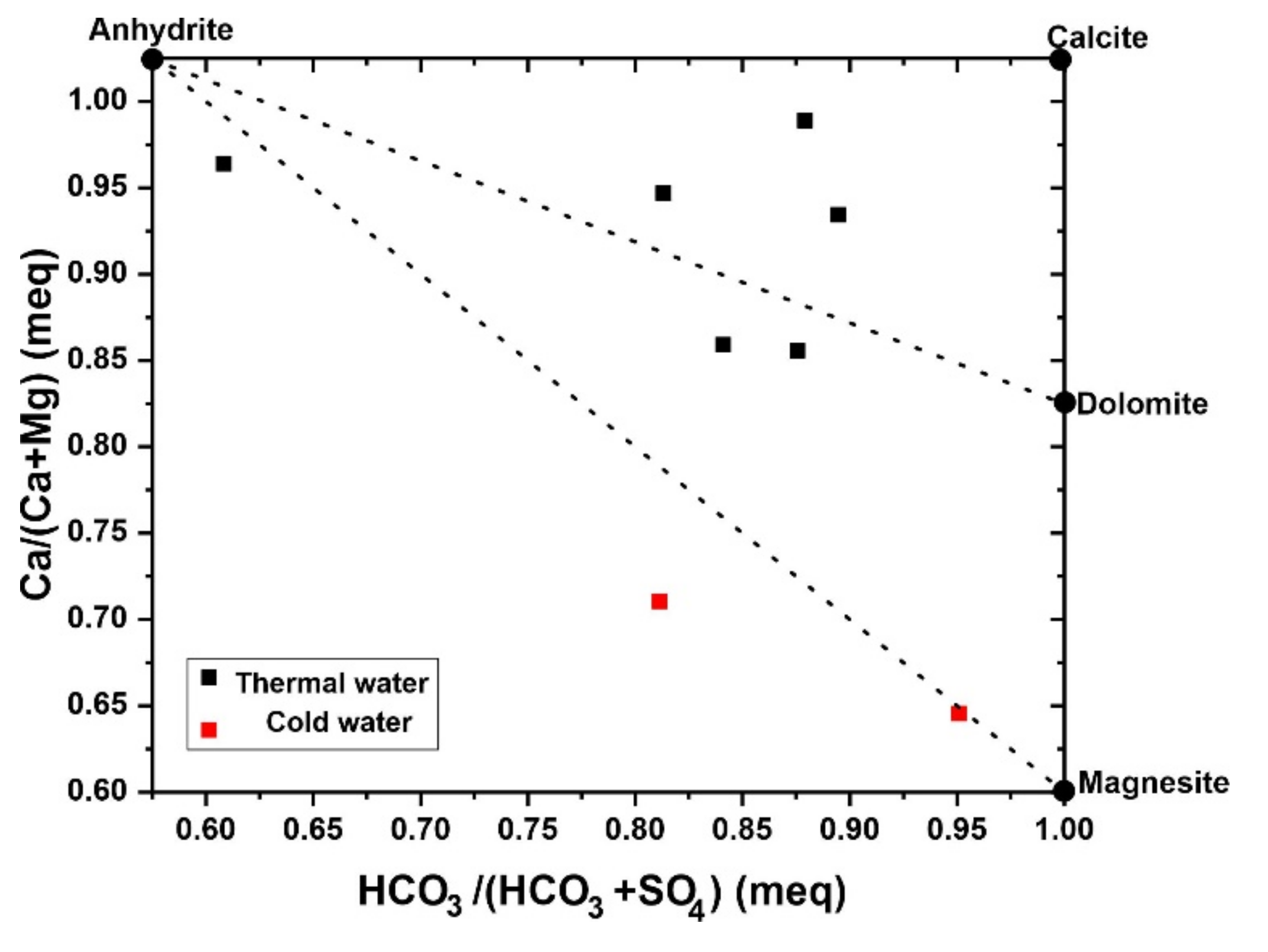
In (
Figure 11), most of the thermal waters are dispersed near dolomite and calcite tie lines, showing the dissolution process of carbonate rocks [
63], whereas thermal water located near the anhydrite corner indicates the dominance of evaporite minerals [
64].
Because the environment in which sediments rich in anhydrite and gypsum deposits exist, it is quite possible that aragonite and calcite would be precipitated [
66]. As these saturation indices, dominant carbonate (CaCO
3) forms, and the studies proved that they can reveal crucial and significant information concerning their environment including pH, dissolved Ca
2+, and other inorganic carbon (IC) as well [
67]. The infiltration and groundwater flow dissolve CaCO
3, and Ca-Mg(CO
3)
2 minerals and increase the Ca
2+ and Mg
2+ concentration in the ground water. (
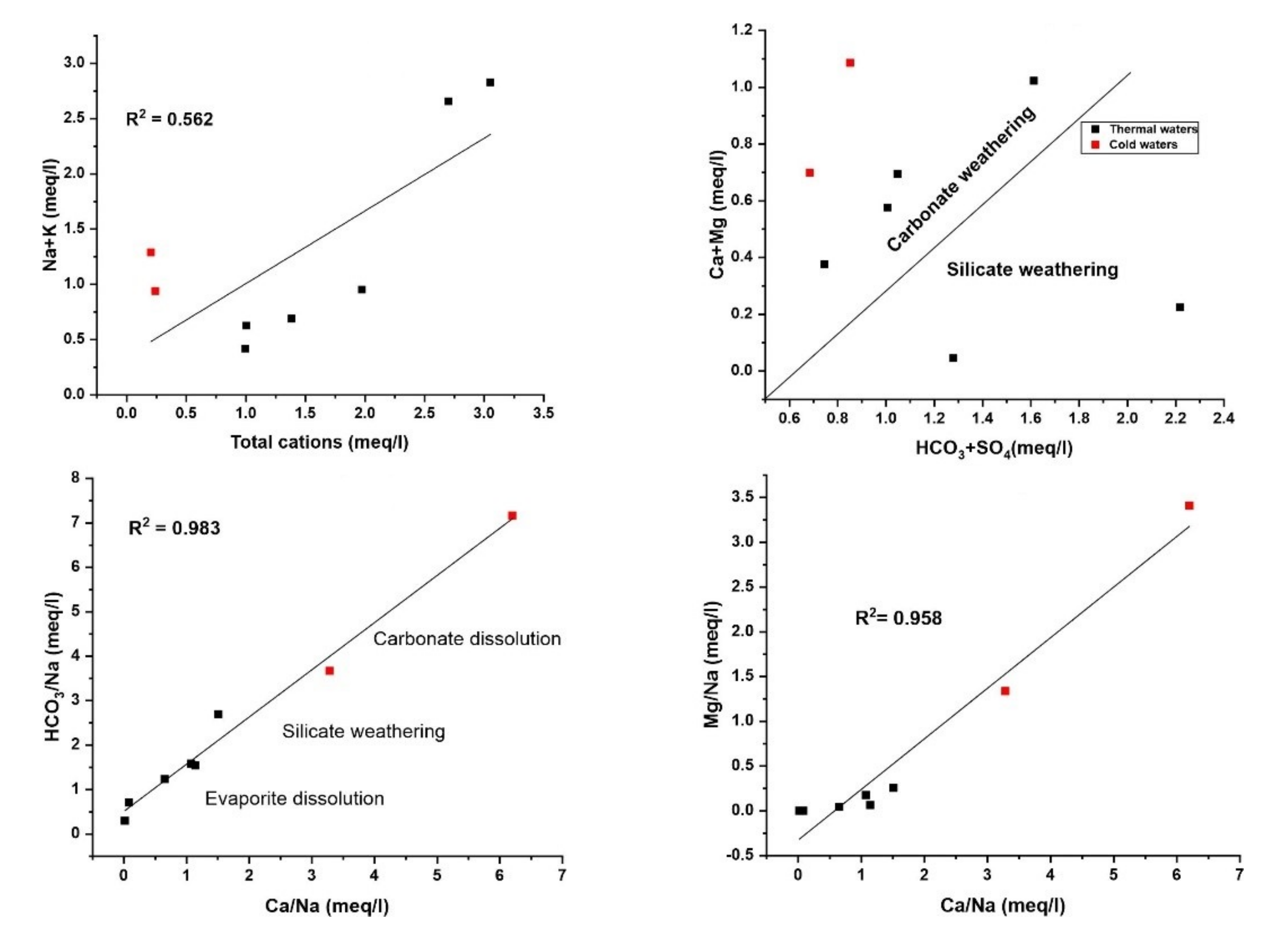
Figure 12) [
68] described that the water samples located above the equiline reveal the carbonate weathering, while those along the line show both carbonate and silicate weathering. The following equations illustrate the mechanism of Ca
2+ and Mg
2+ release in the water system:
The water samples fall below the equiline determined silicate weathering and evaporite dissolution. Thus, carbonate silicate weathering, and evaporite dissolution represent the prime source of Ca2+ and Mg2+ in the ground waters. The carbonate weathering resulting from the interaction of water and CO2 is an intensive process. Moreover, it can trigger Ca2+, Mg2+ and HCO3− in the surrounding water.
The correlation between the estimated water temperature and calculated saturation indices is insignificant. Due to weak fluid-rock activity, most of the thermal and cold ground waters are unsaturated. The estimation of SIs reveal the existence of fairly different hydrological conditions at quite considerable depth.
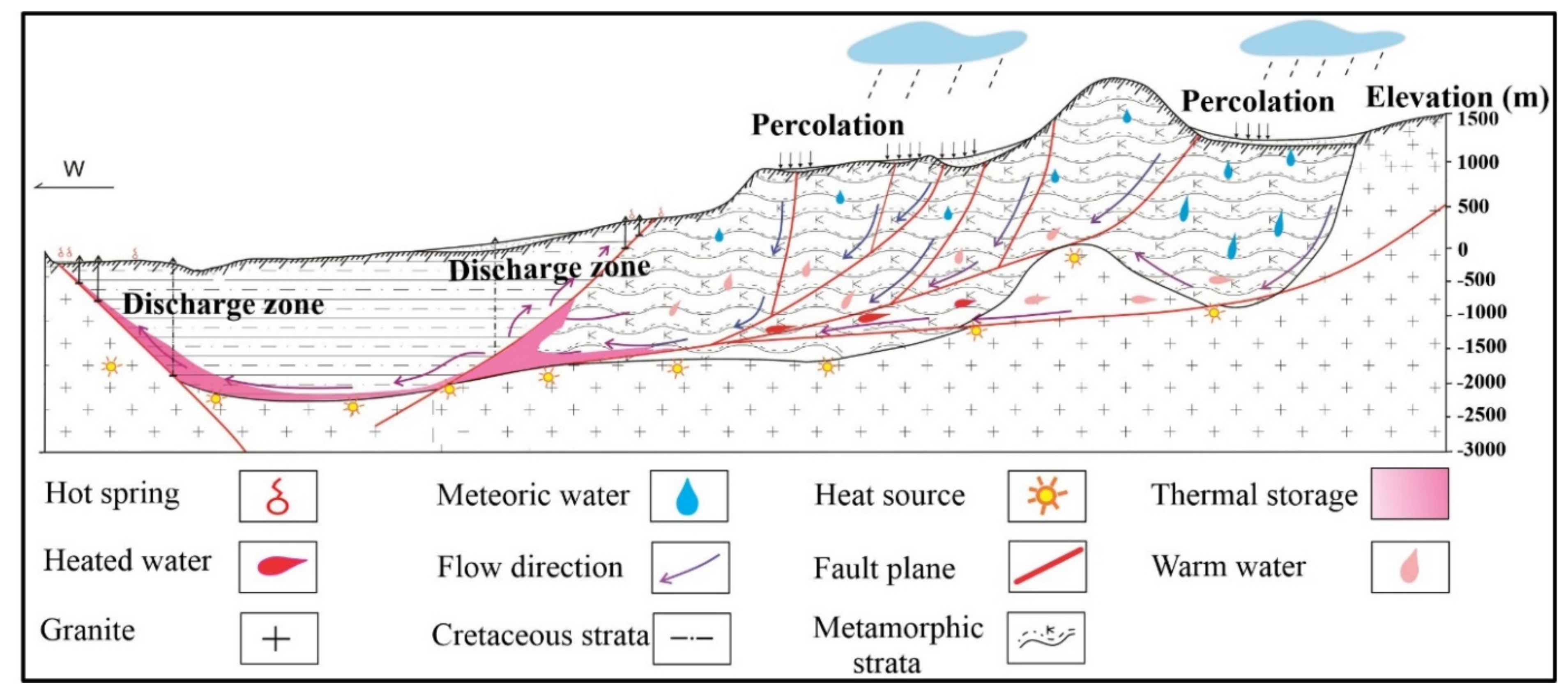
4.5. Circulation Mechanism and Genetic Model
The underground thermal fluid circulation of the Suichuan geothermal field (
Figure 13) is explained by combining the tidal response with hydrochemistry and isotopic characteristics. The tidal oscillations could be detectable in aquifers because of the existence of the solid earth tide [
69]. The tidal forces generate pressure affecting the orientation of fractures that develop as a result of fault slip [
70].
The most likely source of geothermal fluids is meteoric water that adopted a considerable deep circulation cycle as per the hydrochemical and stable isotopic data. The near dispersion of isotopic data along meteoric water lines confirms that no significant evaporation was observed before percolation [
71]. During infiltration, meteoric water came into contact with the surrounding rocks and dissolved various chemical elements, and gained its heat from surrounding rock heating and also from a deep heat source. The rise in temperature and pressure takes place with the continuous deep downward flow of meteoric water. The residence time of thermal fluids is minimal, satisfying the low chemical and isotopic concentrations with no “oxygen drift” because the fluid-rock interaction did not give rise to a notable change of δ
18O values. The deformed zones developed due to the tectonic stresses caused the change in fault aperture from upper widening to lower narrowing and lower widening to upper narrowing. This process created pressure differences in the surrounding rocks. When contraction takes place, the fractures showed low hydraulic conductance, while the extension causes the cracks to open and to provide a passage conducive to the infiltration of meteoric water [
72,
73].
The process continues until the groundwater encounters a barrier that hinders its downward migration and it starts flowing along the stratum interface or cross fissures. This cycle continues and, hence, groundwater attains the heat from surrounding rock heating, and the friction among faults and fractures is also a source of heat. However, the major heat source in the region is radioactive granite [
74,
75]. This is accelerated due to the increasing pressure and resulted in the fluid being discharged from the permeable fractures into the surrounding rocks [
76]. Therefore, thermal groundwater dissolving various chemical elements migrated upward from the areas such as tensional structures and zones of high permeability [
77,
78,
79,
80], oozing out as hot springs. Based on this mechanism and the hydrochemical results of thermal groundwater, the genetic model in Suichuan has been put forward.