Environmental Conditions along Tuna Larval Dispersion: Insights on the Spawning Habitat and Impact on Their Development Stages
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Sampling
2.2. Laboratory and Image Processing
2.3. Age Estimation
2.4. Backward Trajectories Calculation
2.5. Remote Sensing Dataset
2.6. Stages Classification
3. Results
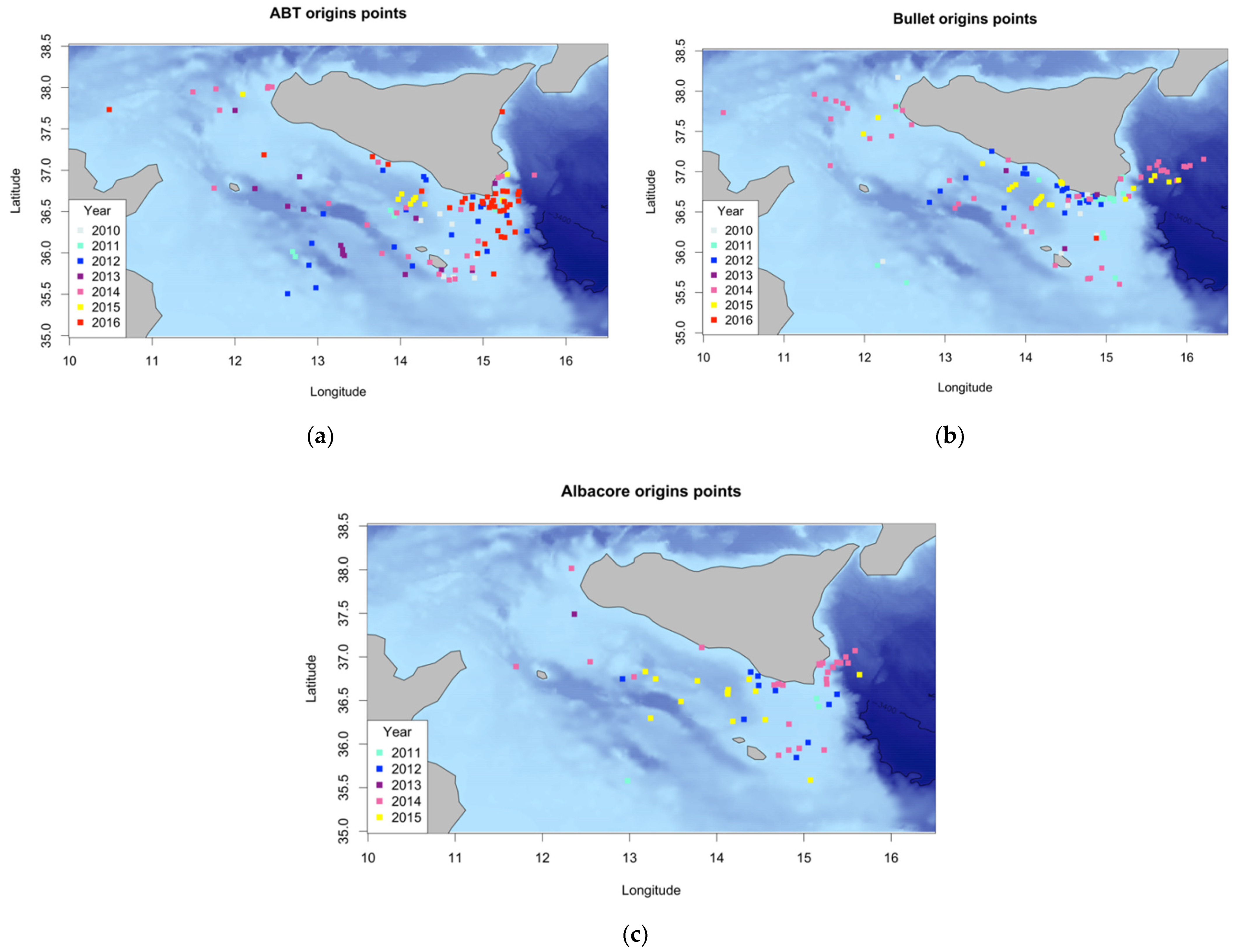
3.1. Backward Trajectory Calculation
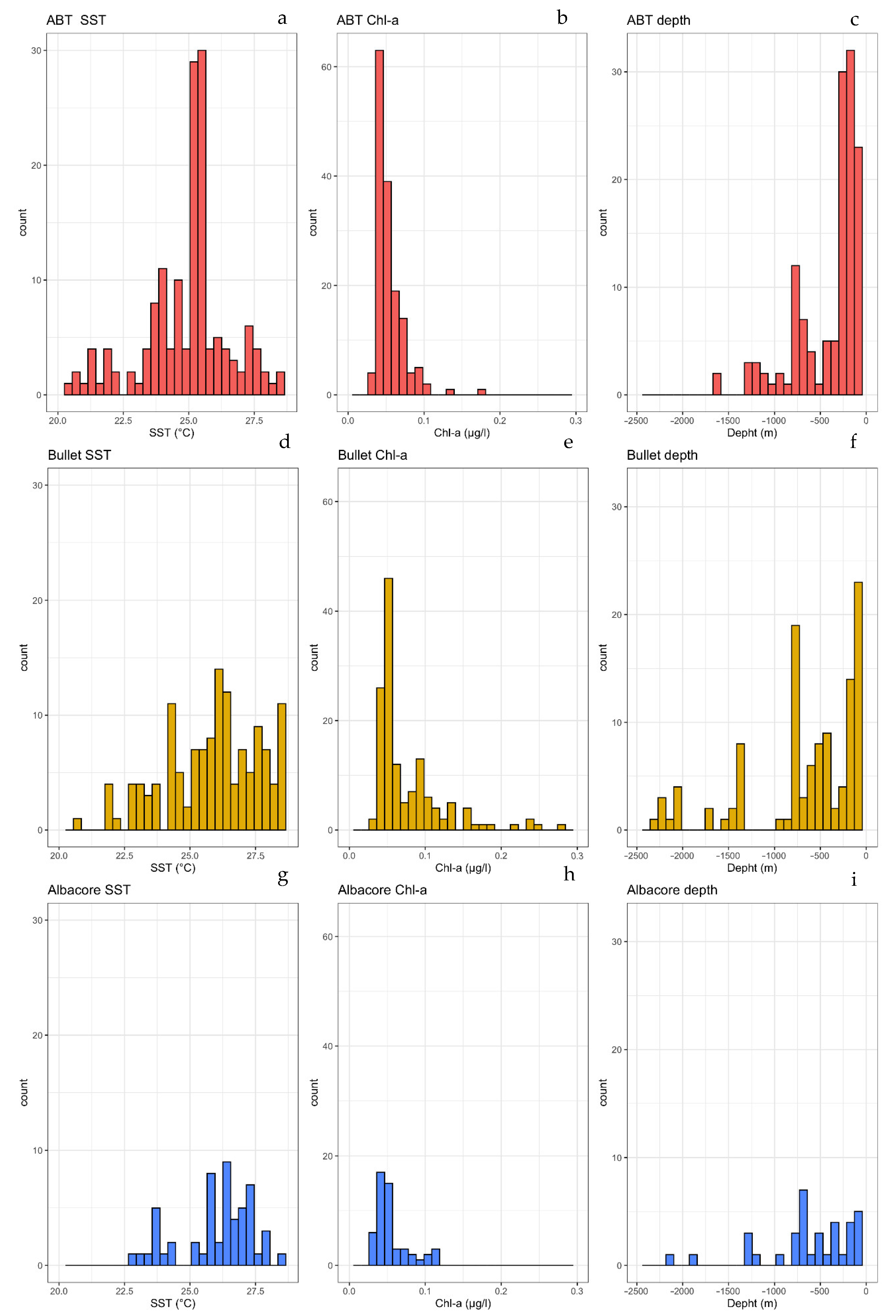
3.2. Characterization of Estimated Hatching Sites
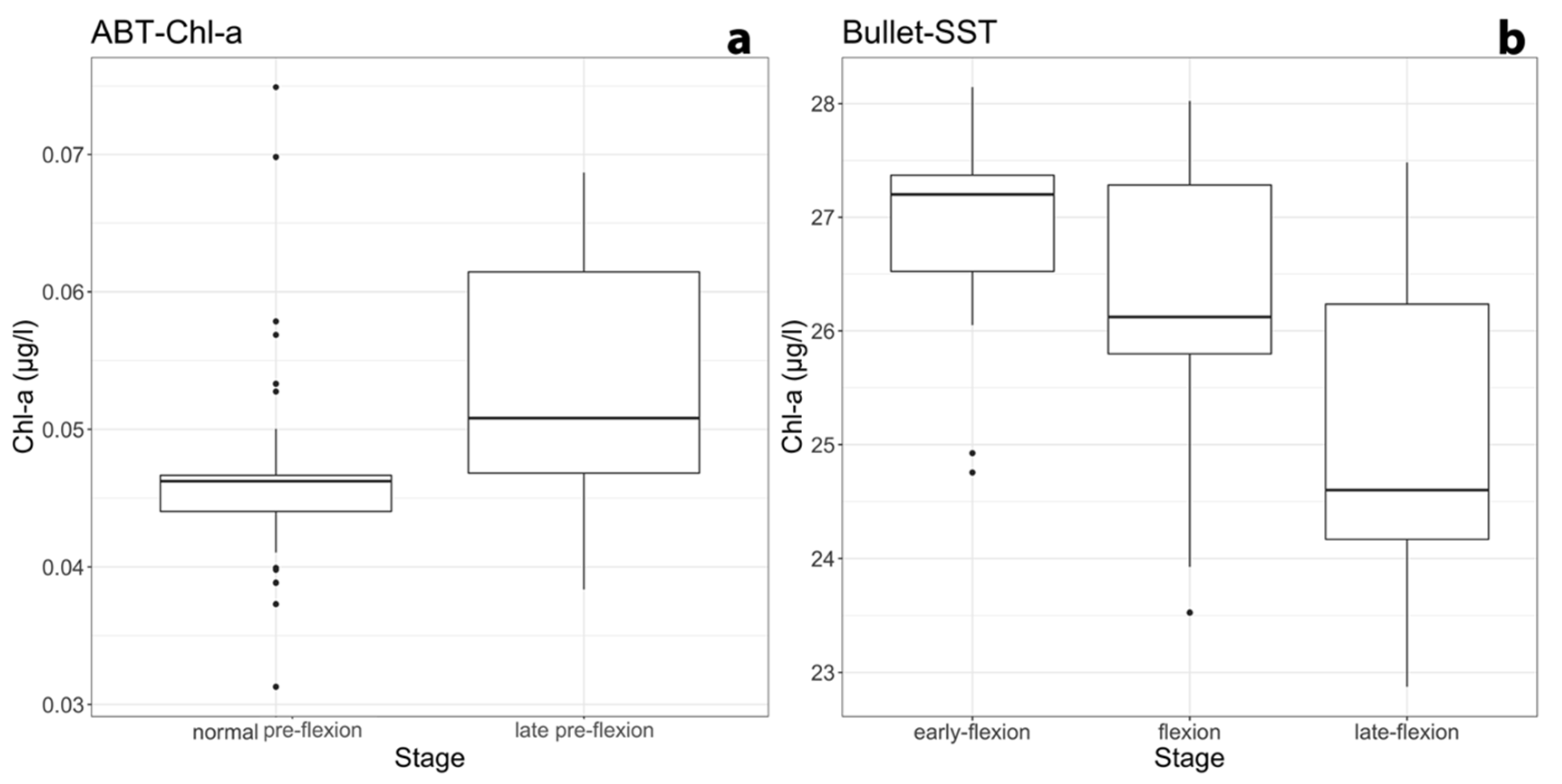
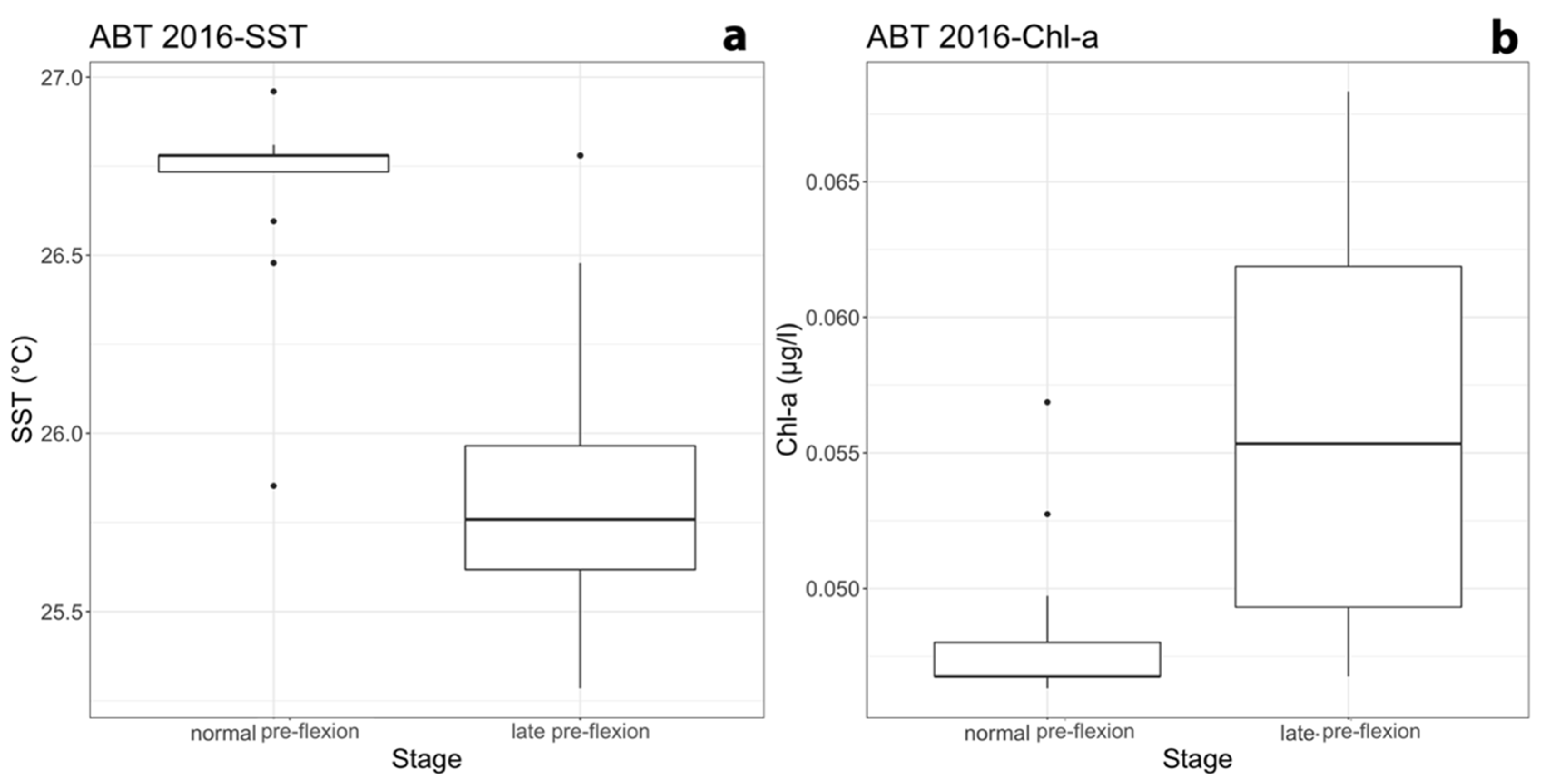
3.3. Relationships among Developmental Larval Traits and Environmental Forcings
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cuttitta, A.; Torri, M.; Zarrad, R.; Zgozi, S.; Jarboui, O.; Quinci, E.M.; Hamza, M.; Abdulfatah, E.; Haddoud, D.; El Turki, A.; et al. Linking surface hydrodynamics to planktonic ecosystem: The case study of the ichthyoplanktonic assemblages in the Central Mediterranean Sea. Hydrobiologia 2018, 821, 191–214. [Google Scholar] [CrossRef]
- Anderson, J.T. A review of size dependent survival during pre-recruit stage of fishes in relation to recruitment. J. Northw. Atl. Fish. Sci. 1988, 8, 55–66. [Google Scholar] [CrossRef]
- Houde, E.D. Recruitment variability. In Fish Reproductive Biology. Implications for Assessment and Management, 1st ed.; Jakobsen, T., Fogarty, M.J., Megrey, B.A., Moksness, E., Eds.; Wiley Blackwell UK: West Sussex, UK, 2009; pp. 91–171. [Google Scholar]
- Rodríguez, J.M.; Alemany, F.; García, A. A Guide to The Eggs and Larvae of 100 Common Western Mediterranean Sea Bony Fish. Species; FAO: Rome, Italy, 2017; pp. 1–256. [Google Scholar]
- Vasile, R.; Hartmann, K.; Hobday, A.J.; Oliver, E.; Tracey, S. Evaluation of hydrodynamic ocean models as a first step in larval dispersal modelling. Cont. Shelf Res. 2018, 152, 38–49. [Google Scholar] [CrossRef]
- Okubo, A. The Role of Diffusion and Related Physical Processes in Dispersal and Recruitment of Marine Populations and Estuarine Studies; American Geophysical Union: Washington, DC, USA, 1994; p. 20. [Google Scholar]
- García, A.; Cortés, D.; Quintanilla, J.; Rámirez, T.; Quintanilla, L.; Rodríguez, J.M.; Alemany, F. Climate-induced environmental conditions influencing interannual variability of Mediterranean bluefin (Thunnus thynnus) larval growth. Fish. Oceanogr. 2013, 22, 273–287. [Google Scholar] [CrossRef]
- Ishihara, T.; Watai, M.; Ohshimo, S.; Abe, O. Differences in larval growth of Pacific bluefin Tuna (Thunnus orientalis) between two spawning areas, and an evaluation of the growth-dependent mortality hypothesis. Environ. Biol. Fish. 2019, 102, 581–594. [Google Scholar] [CrossRef]
- Shiroza, A.; Malca, E.; Lamkin, J.T.; Gerard, T.; Landry, M.R.; Stukel, M.R.; Swalethorp, R. Active prey selection in developing larvae of Atlantic bluefin tuna (Thunnus thynnus) in spawning grounds of the Gulf of Mexico. J. Plankton Res. 2021, 1–19. [Google Scholar] [CrossRef]
- Kodama, T.; Hirai, J.; Tawa, A.; Ishihara, T.; Ohshimo, S. Feeding habits of the Pacific Bluefin tuna (Thunnus orientalis) larvae in two nursery grounds based on morphological and metagenomic analyses. Deep. Sea Res. Part II: Top. Stud. Oceanogr. 2020, 175, 104745. [Google Scholar] [CrossRef]
- Mariani, P.; Křivan, V.; MacKenzie, B.R.; Mullon, C. The migration game in habitat network: The case of Tuna. Theor. Ecol. 2016, 9, 219–232. [Google Scholar] [CrossRef]
- McMahon, K.W.; Hamady, L.L.; Thorrold, S.R. A review of ecogeochemistry approaches to estimating movements of marine animals. Limnol. Oceanogr. 2013, 58, 697–714. [Google Scholar] [CrossRef]
- Rooker, J.R.; Arrizabalaga, H.; Fraile, I.; Secor, D.H.; Dettman, D.L.; Abid, N.; Addis, P.; Deguara, S.; Karakulak, F.S.; Kimoto, A.; et al. Crossing the line: Migratory and homing behaviors of Atlantic bluefin Tuna. Mar. Ecol. Prog. Ser. 2014, 504, 265–276. [Google Scholar] [CrossRef]
- Bromage, N.R.; Porter, M.J.R.; Randall, C.F. The environmental regulation of maturation in farmed finfish with special reference to the role of photoperiod and melatonin. Aquaculture 2001, 197, 63–98. [Google Scholar] [CrossRef]
- Gabilondo, A.R.; Pérez, L.H.; Rodríguez, R.M. Hormonal and neuroendocrine control of reproductive function in teleost fish. Review in Bionatura. 2022, 6, 2122. [Google Scholar]
- Zanuy, S.; Carrillo, M.; Felip, A.; Rodríguez, L.; Blázquez, M.; Ramos, J.; Piferrer, F. Genetic, hormonal and environmental approaches for the control of reproduction in the European sea bass (Dicentrarchus labrax L.). Aquaculture 2001, 202, 187–203. [Google Scholar] [CrossRef]
- Migaud, H.; Davie, A.; Taylor, J.F. Current knowledge on the photoneuroendocrine regulation of reproduction in temperate fish species. J. Fish Biol. 2010, 76, 27–68. [Google Scholar] [CrossRef] [PubMed]
- Reglero, P.; Ciannelli, L.; Alvarez-Berastegui, D.; Balbín, R.; López-Jurado, J.L.; Alemany, F. Geographically and environmentally driven spawning distributions of Tuna species in the western Mediterranean Sea. Mar. Ecol. Prog. Ser. 2012, 463, 273–284. [Google Scholar] [CrossRef]
- Mather, F.J.; Mason, J.M.; Jones, A.C. Historical Document: Life History and Fisheries of Atlantic Bluefin Tuna; US Department of Commerce, NOAA, NMFS, Southeast Fisheries Science Center: Silver Spring, MD, USA, 1995; pp. 1–165.
- Osse, J.W.M.; Van den Boogaart, J.G.M.; Van Snik, G.M.J.; Van der Sluys, L. Priorities during early growth of fish larvae. Aquaculture 1997, 155, 249–258. [Google Scholar] [CrossRef]
- Cahu, C.; Infante, J.Z.; Takeuchi, T. Nutritional components affecting skeletal development in fish larvae. Aquaculture 2003, 227, 245–258. [Google Scholar] [CrossRef]
- Alvarez-Berastegui, D.; Tugores, M.P.; Martín, M.; Leyva, R.; Saber, S.; Macías, D.; Reglero, P. Assessing the spawning stock biomass of albacore (Thunnus alalunga) in the western mediterranean sea from a non-linear larval index (2001–2019). Collect. Vol. Sci. Pap. ICCAT 2021, 78, 276–293. [Google Scholar]
- Mariani, P.; MacKenzie, B.R.; Iudicone, D.; Bozec, A. Modelling retention and dispersion mechanisms of bluefin Tuna eggs and larvae in the northwest Mediterranean Sea. Prog. Oceanogr. 2010, 86, 45–58. [Google Scholar] [CrossRef]
- Burgess, S.C.; Bode, M.; Leis, J.M.; Mason, L.B. Individual variation in marine larval-fish swimming speed and the emergence of dispersal kernels. Oikos 2022, 2022, e08896. [Google Scholar] [CrossRef]
- Kitagawa, T.; Kimura, S. (Eds.) Biology and Ecology of Bluefin Tuna; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Wolanski, E.; Hamner, W.M. Topographically controlled fronts in the ocean and their biological influence. Science 1988, 241, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Tawa, A.; Kodama, T.; Sakuma, K.; Ishihara, T.; Ohshimo, S. Fine-scale horizontal distributions of multiple species of larval tuna off the Nansei Islands, Japan. Mar. Ecol. Prog. Ser. 2020, 636, 123–137. [Google Scholar] [CrossRef]
- Alemany, F.; Quintanilla, L.; Velez-Belchí, P.; García, A.; Cortés, D.; Rodríguez, J.M.; Fernández de Puelles, M.L.; González-Pola, C.; López-Jurado, J.L. Characterization of the spawning habitat of Atlantic Bluefin Tuna and related species in the Balearic Sea (western Mediterranean). Progr. Oceanogr. 2010, 86, 21–38. [Google Scholar] [CrossRef]
- Díaz-Barroso, L.; Hernández-Carrasco, I.; Orfila, A.; Reglero, P.; Balbín, R.; Hidalgo, M.; Tintoré, J.; Alemany, F.; Alvarez-Berastegui, D. Singularities of surface mixing activity in the Western Mediterranean influence bluefin tuna larval habitats. Mar. Ecol. Prog. Ser. 2022, 685, 69–84. [Google Scholar] [CrossRef]
- Russo, S.; Torri, M.; Patti, B.; Reglero, P.; Álvarez-Berastegui, D.; Cuttitta, A.; Sarà, G. Unveiling the Relationship Between Sea Surface Hydrographic Patterns and Tuna Larval Distribution in the Central Mediterranean Sea. Front. Mar. Sci. 2021, 8, 1116. [Google Scholar] [CrossRef]
- Koched, W.; Hattour, A.; Alemana, F.; Zarrad, R.; Garcia, A. Distribution of Tuna larvae in Tunisian east coasts and its environmental scenario. Cahiers de Biologie Marine 2012, 53, 505–515. [Google Scholar]
- Koched, W.; Ben Ismail, S.; Alemany, F.; Benmessaoud, R.; Hattour, A.; Garcia, A. Environmental conditions influencing the larval fish assemblage during summer in the Gulf of Gabes (Tunisia: South central Mediterranean). Mediterr. Mar. Sci. 2015, 16, 666–681. [Google Scholar] [CrossRef][Green Version]
- Zarrad, R.; Rodríguez, J.M.; Alemany, F.; Charef, A.; Jarboui, O.; Missaoui, H. Larval fish community composition and distribution of the central-southern mediterranean under summer and winter conditions. Acta Ichthyol. Piscat. 2020, 50, 313–324. [Google Scholar] [CrossRef]
- Robinson, A.R.; Sellschopp, J.; Warn-Varnas, A.; Leslie, W.G.; Lozano, C.J.; Haley, P.J., Jr.; Anderson, L.A.; Lermusiaux, P.F.J. The Atlantic Ionian stream. J. Mar. Syst. 1999, 20, 129–156. [Google Scholar] [CrossRef]
- Koched, W.; Hattour, A.; Alemany, F.; Garcia, A.; Said, K. Spatial distribution of Tuna larvae in the Gulf of Gabes (Eastern Mediterranean) in relation with environmental parameters. Mediterr. Mar. Sci. 2013, 14, 5–14. [Google Scholar] [CrossRef][Green Version]
- Patti, B.; Torri, M.; Cuttitta, A. General surface circulation controls the interannual fluctuations of anchovy stock biomass in the Central Mediterranean Sea. Sci. Rep. 2020, 10, 1554. [Google Scholar] [CrossRef] [PubMed]
- Cuttitta, A.; Patti, B.; Maggio, T.; Quinci, E.M.; Pappalardo, A.M.; Ferrito, V.; De Pinto, V.; Torri, M.; Falco, F.; Nicosia, A.; et al. Larval population structure of Engraulis encrasicolus in the Strait of Sicily as revealed by morphometric and genetic analysis. Fish. Oceanogr. 2015, 24, 135–149. [Google Scholar] [CrossRef]
- Torri, M.; Pappalardo, A.M.; Ferrito, V.; Giannì, S.; Armeri, G.M.; Patti, C.; Mangiaracina, F.; Biondo, G.; Di Natale, M.; Musco, M.; et al. Signals from the deep-sea: Genetic structure, morphometric analysis, and ecological implications of Cyclothone braueri (Pisces, Gonostomatidae) early life stages in the Central Mediterranean Sea. Mar. Environ. Res. 2021, 169, 105379. [Google Scholar] [CrossRef] [PubMed]
- Catalán, I.A.; Tejedor, A.; Alemany, F.; Reglero, P. Trophic ecology of Atlantic bluefin Tuna Thunnus thynnus larvae. J. Fish. Biol. 2011, 78, 1545–1560. [Google Scholar] [CrossRef]
- García, A.; Cortés, D.; Ramírez, T.; Fehri-Bedoui, R.; Alemany, F.; Rodríguez, J.M.; Carpena, Á.; Álvarez, J.P. First data on growth and nucleic acid and protein content of field-captured Mediterranean bluefin (Thunnus thynnus) and Albacore (Thunnus alalunga) Tuna larvae: A comparative study. Sci. Mar. 2006, 70, 67–78. [Google Scholar] [CrossRef]
- Laiz-Carrión, R.; Quintanilla, J.M.; Torres, A.P.; Alemany, F.; García, A. Hydrographic patterns conditioning variable trophic pathways and early life dynamics of Bullet Tuna Auxis rochei larvae in the Balearic Sea. Mar. Ecol. Prog. Ser. 2013, 475, 203–212. [Google Scholar] [CrossRef]
- NOAA (National Oceanic and Atmospheric Administration). General NOAA Oil Modelling Environment (GNOME) User’s Manual; NOAA: Seattle, WA, USA, 2002; pp. 1–94. [Google Scholar]
- Beegle-Krause, J.C.; O’Connor, C. GNOME Data Formats and Associated Example Data Files; NOAA Office of Response and Restoration, Emergency Response Division (formerly Hazardous Materials Response Division): Seattle, WA, USA, 2005; p. 49. [Google Scholar]
- Engie, K.T. Klinger. Modeling passive dispersal through a large estuarine system to evaluate marine reserve network connections. Estuaries Coasts 2007, 30, 201–213. [Google Scholar] [CrossRef]
- Palatella, L.; Bignami, F.; Falcini, F.; Lacorata, G.; Lanotte, A.S.; Santoleri, R. Lagrangian simulations and interannual variability of anchovy egg and larva dis- persal in the Sicily Channel. J. Geophys. Res. Ocean. 2014, 119, 1306–1323. [Google Scholar] [CrossRef]
- Beegle-Krause, C.J. General NOAA Oil Modeling Environment (GNOME): A New Spill Trajectory Model. In Proceedings of the IOSC 2001 Proceedings, Tampa, FL, USA, 26–29 March 2001; Volume 2. [Google Scholar]
- NASA; GSFC; NOAA. Cross-Calibrated Multi-Platform Ocean Surface Wind Vector L3.0 First-Look Analyses. Ver. 1. PO.DAAC, CA, USA. 2009. Available online: https://doi.org/10.5067/CCF30-01XXX (accessed on 30 September 2016).
- Buongiorno Nardelli, B.; Tronconi, C.; Pisano, A.; Santoleri, R. High and Ultra-High resolution processing of satellite Sea Surface Temperature data over Southern European Seas in the framework of MyOcean project. Rem. Sens. Env. 2013, 129, 1–16. [Google Scholar] [CrossRef]
- Lee, Z.P.; Carder, K.; Arnone, R. Deriving inherent optical properties from water color: A multi- band quasi-analytical algorithm for optically deep waters. Appl. Opt. 2002, 41, 5755–5772. [Google Scholar] [CrossRef]
- Berthon, J.F.; Zibordi, G. Bio-optical relationships for the northern Adriatic Sea. Int. J. Remote Sens. 2004, 25, 1527–1532. [Google Scholar] [CrossRef]
- Mélin, F.; Vantrepotte, V. How optically diverse is the coastal ocean? Remote Sens. Environ. 2015, 160, 235–251. [Google Scholar] [CrossRef]
- Volpe, G.; Buongiorno Nardelli, B.; Colella, S.; Pisano, A.; Santoleri, R. An Operational Interpolated Ocean Colour Product in the Mediterranean Sea. In New Frontiers in Operational Oceanography; Chassignet, E.P., Pascual, A., Tintorè, J., Verron, J., Eds.; GODAE Ocean View International School: Mallorca, Spain, 2018; pp. 227–244. [Google Scholar]
- Volpe, G.; Colella, S.; Brando, V.E.; Forneris, V.; Padula, F.L.; Cicco, A.D.; Sammartino, M.; Bracaglia, M.; Artuso, F.; Santoleri, R. Mediterranean ocean colour Level 3 operational multi-sensor processing. Ocean. Sci. 2019, 15, 127–146. [Google Scholar] [CrossRef]
- Pante, E.; Bouhet, S.B. marmap: A Package for Importing, Plotting and Analyzing Bathymetric and Topographic Data in R. PLoS ONE 2013, 8, e73051. [Google Scholar] [CrossRef] [PubMed]
- Blanco, E.; Reglero, P.; de Rojas, A.H.; Ortega, A.; de la Gándara, F.; Folkvord, A. The effect of nutritional condition by two nucleic acid derived indices on the growth to post-flexion of Atlantic bluefin Tuna and Atlantic bonito larvae. J. Exp. Mar. Biol. Ecol. 2019, 519, 151–182. [Google Scholar] [CrossRef]
- García-Lafuente, J.; García, S.; Mazzola, L.; Quintanilla, A.; Delgado, A.; Cuttitta, B. Hydrographic phenomena influencing early life stages of the Sicilian Channel anchovy. Fish. Oceanogr 2002, 11, 31–44. [Google Scholar] [CrossRef]
- Prants, S.V. Marine life at Lagrangian fronts. Prog. Oceanogr. 2022, 204, 102790. [Google Scholar] [CrossRef]
- Cuttitta, A.; Quinci, E.M.; Patti, B.; Bonomo, S.; Bonanno, A.; Musco, M.; Torri, M.; Placenti, F.; Basilone, G.; Genovese, S.; et al. Different key roles of mesoscale oceanographic structures and ocean bathymetry in shaping larval fish distribution pattern: A case study in Sicilian waters in summer 2009. J. Sea Res. 2016, 115, 6–17. [Google Scholar] [CrossRef]
- Ciannelli, L.; Bailey, K.; Olsen, E.M. Evolutionary and ecological constraints of fish spawning habitats. ICES J. Mar. Sci. 2015, 72, 285–296. [Google Scholar] [CrossRef]
- Torri, M.; Corrado, R.; Falcini, F.; Cuttitta, A.; Palatella, L.; Lacorata, G.; Patti, B.; Arculeo, M.; Mifsud, R.; Mazzola, S.; et al. Planktonic stages of small pelagic fishes (Sardinella aurita and Engraulis encrasicolus) in the central Mediterranean Sea: The key role of physical forcings and implications for fisheries management. Prog. Oceanogr. 2018, 162, 25–39. [Google Scholar] [CrossRef]
- Patti, B.; Zarrad, R.; Jarboui, O.; Cuttitta, A.; Basilone, G.; Aronica, S.; Placenti, F.; Tranchida, G.; Armeri, G.M.; Buffa, G.; et al. Anchovy (Engraulis encrasicolus) early life stages in the Central Mediterranean Sea: Connectivity issues emerging among adjacent sub-areas across the Strait of Sicily. Hydrobiologia 2018, 821, 25–40. [Google Scholar] [CrossRef]
- Reglero, P.; Ortega, A.; Balbín, R.; Abascal, F.J.; Medina, A.; Blanco, E.; de la Gándara, F.; Alvarez-Berastegui, D.; Hidalgo, M.; Rasmuson, L.; et al. Atlantic bluefin tuna spawn at suboptimal temperatures for their offspring. Proc. R. Soc. 2018, 285, 20171405. [Google Scholar] [CrossRef] [PubMed]
- Pepin, P.; Dower, J.F.; Davidson, F.J.M. A spatially explicit study of prey–predator interactions in larval fish: Assessing the influence of food and predator abundance on larval growth and survival. Fish. Oceanogr. 2003, 12, 19–33. [Google Scholar] [CrossRef]
- Petitgas, P.; Masse, J.; Bourriau, P.; Beillois, P.; Delmas, D.; Herbland, A.; Koueta, N.; Froidefond, J.M.; Santos, M. Hydro-plankton characteristics and their relationship with sardine and anchovy distributions on the French shelf of the Bay of Biscay. Sci. Mar. 2006, 70, 161–172. [Google Scholar] [CrossRef][Green Version]
- Cortés, D.; García, A.; Ramírez, T. First data on the nutritional condition of the Mediterranean bullet tuna larvae, Auxis rochei, from the Balearic archipelago. Rapp. Comm. Int. Mer. Médit. 2004, 37, 337. [Google Scholar]
- Laiz-Carrión, R.; Pérez-Torres, A.; Quintanilla, J.; García, A.; Alemany, F. Variable growth of bullet tuna larvae (Auxis rochei) related to hydrographic scenarios off the Balearic Sea. Rapp. Comm. Int. Mer. Medit. 2010, 39, 563. [Google Scholar]
- Bakun, A.; Broad, K. Environmental ‘loopholes’ and fish population dynamics: Comparative pattern recognition with focus on El Niño effects in the Pacific. Fish. Oceanogr. 2003, 12, 458–473. [Google Scholar] [CrossRef]
- Green, B.S.; McCormick, M.I. Influence of larval feeding history on the body condition of Amphiprion melanopus. J. Fish. Biol. 1999, 55, 1273–1289. [Google Scholar] [CrossRef]
- Green, B.S.; Fisher, R. Temperature influences swimming speed, growth and larval duration in coral reef fish larvae. J. Exp. Mar. Biol. Ecol. 2004, 299, 115–132. [Google Scholar] [CrossRef]
- Utne-Palm, A.; Stiansen, J. Effect of larval ontogeny, tur- bulence and light on prey attack rate and swimming activ- ity in herring larvae. J. Exp. Mar. Biol. Ecol. 2002, 268, 147–170. [Google Scholar] [CrossRef]
- Pepin, P.; Myers, R.A. Significance of egg and larval size to recruitment variability of temperate marine fish. Can. J. Fish. Aquat. Sci. 1991, 48, 1820–1828. [Google Scholar] [CrossRef]
- Chambers, R.C.; Leggett, W.C.; Brown, J.A. Egg size, female effects, and the correlation between early life history traits of capelin, Mallotus villosus: An appraisal at the individual level. Fish Bull. 1989, 87, 515–523. [Google Scholar]
- Green, B.S.; McCormick, M.I. Maternal and paternal effects determine size, growth and performance in larvae of a tropical reef fish. Mar. Ecol. Prog. Ser. 2005, 289, 263–272. [Google Scholar] [CrossRef]
- Rouyer, T.; Bernard, S.; Kerzérho, V.; Giordano, N.; Giordano, F.; Ellul, S.; Ellul, G.; Derridj, O.; Canet, R.; Deguara, S. Electronic tagging of Bluefin Tunas from the Maltese spawning ground suggests size-dependent migration dynamics. Environ. Biol. Fishes 2022. [Google Scholar] [CrossRef]
- Hjort, J. Fluctuations in the great fisheries of northern Europe. Rapp. Proces Verbaux La Comm. Int. Pour L’exploration Sci. La Mer 1914, 20, 1–228. [Google Scholar]
- Cowen, R.; Sponaugle, S. Larval dispersal and marine population connectivity. Annu. Rev. Mar. Sci. 2009, 1, 443–466. [Google Scholar] [CrossRef]
- Chavez, F.P.; Ryan, J.; Lluch-Cota, S.E.; Ñiquen, M. From anchovies to sardines and back: Multidecadal change in the Pacific Ocean. Science 2003, 299, 217–221. [Google Scholar] [CrossRef]
- Pörtner, H.O.; Knust, R. Climate change affects marine fishes through the oxygen limitation of thermal tolerance. Science 2007, 315, 95–97. [Google Scholar] [CrossRef]

| Macro-Category | Sub-Category | ABT | Bullet | Albacore |
|---|---|---|---|---|
| Pre-flexion | Normal | 98 | 63 | 21 |
| Late | 33 | 22 | 7 | |
| Flexion | Early | 10 | 16 | 10 |
| Normal | 18 | 31 | 20 | |
| Late | 10 | 16 | 10 | |
| Post-flexion | Early | 9 | 8 | 2 |
| Normal | 24 | 23 | 5 |
| Environmental Variable | ABT | Bullet | Albacore | |
|---|---|---|---|---|
| SST | Minimum value | 20.47 | 20.71 | 22.82 |
| Maximum value | 28.67 | 28.67 | 28.47 | |
| Median value | 25.16 | 25.98 | 26.32 | |
| Chl-a | Minimum value | 0.029 | 0.033 | 0.029 |
| Maximum value | 0.174 | 0.280 | 0.346 | |
| Median value | 0.048 | 0.055 | 0.049 | |
| Bottom Depth | Minimum value | −1607 | −2470 | −2484 |
| Maximum value | −15.7 | −9.00 | −11.28 | |
| Median value | −283 | −209 | −507 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Russo, S.; Torri, M.; Patti, B.; Musco, M.; Masullo, T.; Di Natale, M.V.; Sarà, G.; Cuttitta, A. Environmental Conditions along Tuna Larval Dispersion: Insights on the Spawning Habitat and Impact on Their Development Stages. Water 2022, 14, 1568. https://doi.org/10.3390/w14101568
Russo S, Torri M, Patti B, Musco M, Masullo T, Di Natale MV, Sarà G, Cuttitta A. Environmental Conditions along Tuna Larval Dispersion: Insights on the Spawning Habitat and Impact on Their Development Stages. Water. 2022; 14(10):1568. https://doi.org/10.3390/w14101568
Chicago/Turabian StyleRusso, Stefania, Marco Torri, Bernardo Patti, Marianna Musco, Tiziana Masullo, Marilena Vita Di Natale, Gianluca Sarà, and Angela Cuttitta. 2022. "Environmental Conditions along Tuna Larval Dispersion: Insights on the Spawning Habitat and Impact on Their Development Stages" Water 14, no. 10: 1568. https://doi.org/10.3390/w14101568
APA StyleRusso, S., Torri, M., Patti, B., Musco, M., Masullo, T., Di Natale, M. V., Sarà, G., & Cuttitta, A. (2022). Environmental Conditions along Tuna Larval Dispersion: Insights on the Spawning Habitat and Impact on Their Development Stages. Water, 14(10), 1568. https://doi.org/10.3390/w14101568









