Heavy Metals and Nutrients Loads in Water, Soil, and Crops Irrigated with Effluent from WWTPs in Blantyre City, Malawi
Abstract
1. Introduction
2. Materials and Methods
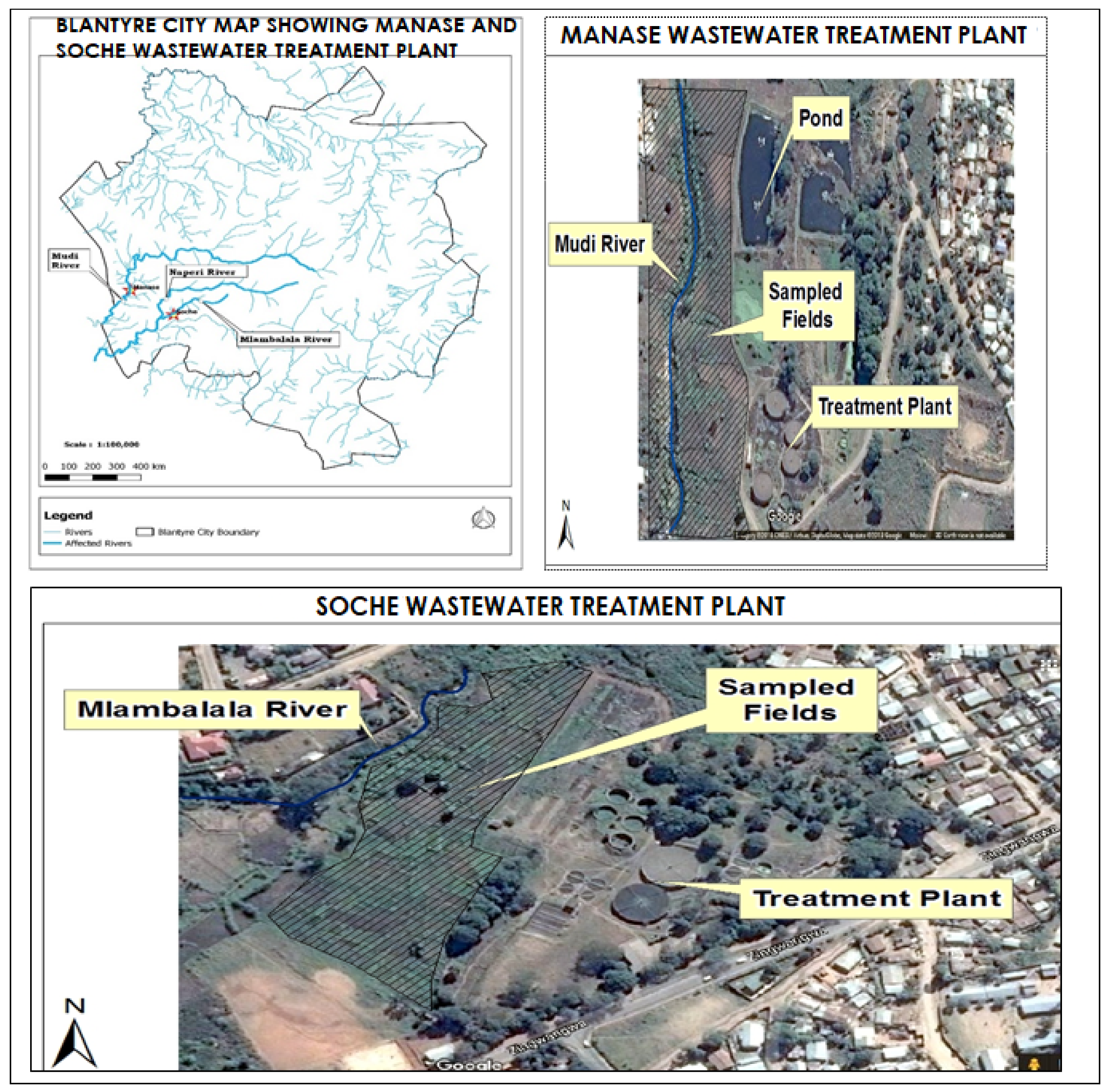
2.1. Description of Study Area
2.2. Collection of Crop Samples
2.3. Collection of Soil Samples
2.4. Collection of Water Samples
2.5. Chemical Analysis
Instruments and Other Analytical Issues
2.6. Analytical Methods
2.7. Soil Analysis
2.8. Plant Heavy Metal Analysis
3. Results and Discussion
3.1. pH, Temperature, Electrical Conductivity, and Total Dissolved Solids in Effluent and Soil Samples
3.2. Fecal Coliform
3.3. BOD
3.4. COD
3.5. Nitrate in Water Sample
Nitrate in Soil Samples
3.6. Phosphates
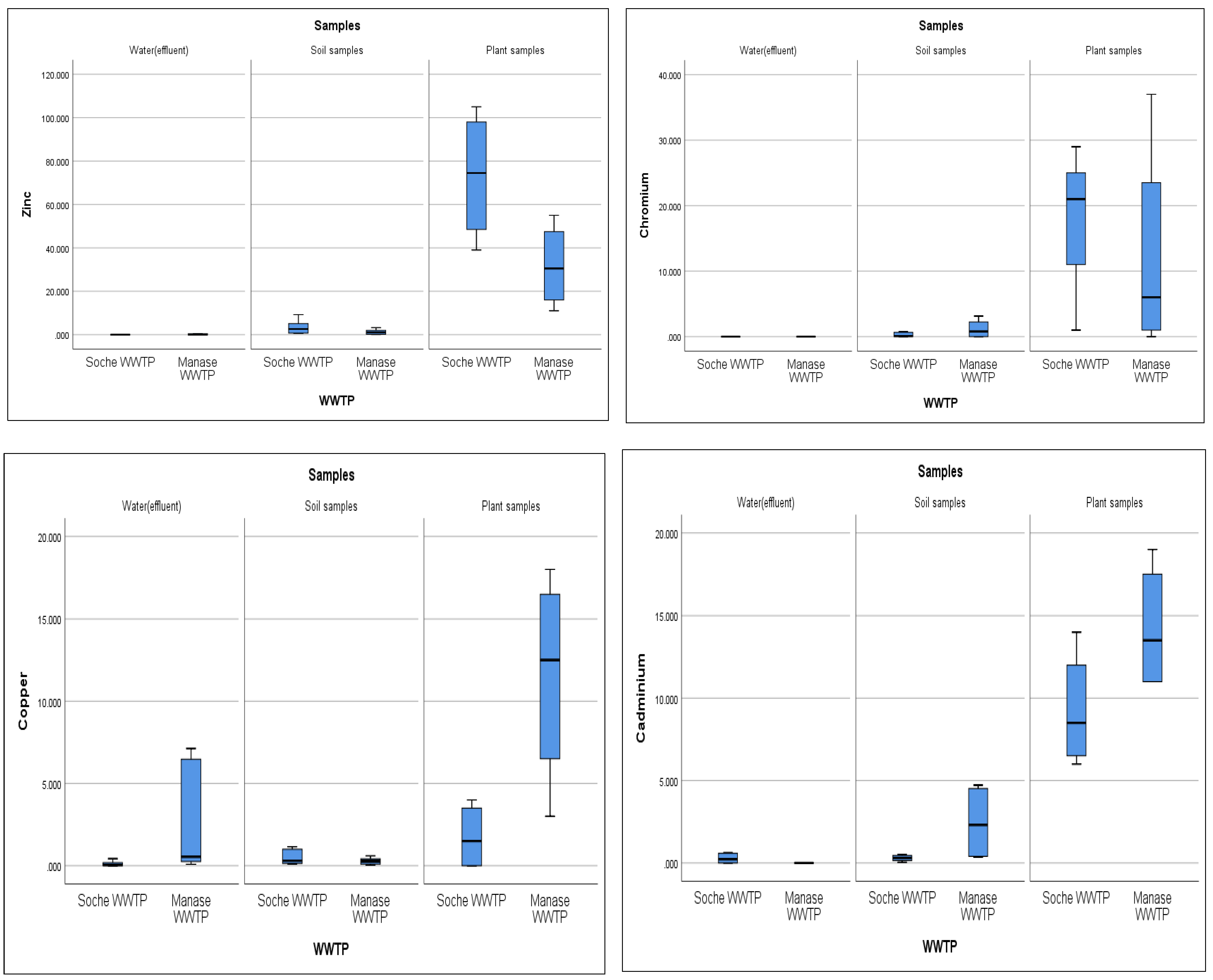
3.7. Heavy Metals
3.8. Health Risk Assessment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kalulu, K.; Thole, B.; Mkandawire, T.; Kululanga, G. Application of process intensification in the treatment of pit latrine sludge from informal settlements in Blantyre City, Malawi. Int. J. Environ. Res. Public Health 2020, 17, 3296. [Google Scholar] [CrossRef]
- Kayira, F.; Wanda, E.M.M. Evaluation of the performance of Mzuzu Central Hospital wastewater oxidation ponds and its effect on water quality in Lunyangwa River, Northern Malawi. Phys. Chem. Earth 2021, 123, 103015. [Google Scholar] [CrossRef]
- Vunain, E.; Mike, P.; Mpeketula, G.; Monjerezi, M.; Etale, A. Evaluation of coagulating e ffi ciency and water borne pathogens reduction capacity of Moringa oleifera seed powder for treatment of domestic wastewater from Zomba, Malawi. J. Environ. Chem. Eng. 2019, 7, 103118. [Google Scholar] [CrossRef]
- Ngoma, W.; Hoko, Z.; Misi, S.; Chidya, R.C.G. Assessment of efficiency of a decentralized wastewater treatment plant at Mzuzu University, Mzuzu, Malawi. Phys. Chem. Earth 2020, 118–119, 102903. [Google Scholar] [CrossRef]
- Abosede, A.; Solomon, O.; Peter, A.; Eromosele, H.; Emmanuel, A. Wastewater conservation and reuse in quality vegetable cultivation: Overview, challenges and future prospects. Food Control 2018, 98, 489–500. [Google Scholar] [CrossRef]
- Rizzo, L. Science of the Total Environment Best available technologies and treatment trains to address current challenges in urban wastewater reuse for irrigation of crops in EU countries. Sci. Total Environ. 2020, 710, 136312. [Google Scholar] [CrossRef]
- Truchado, P.; Garre, A.; Gil, M.I.; Simón-andreu, P.J.; Sánchez, G.; Allende, A. Science of the Total Environment Monitoring of human enteric virus and coliphages throughout water reuse system of wastewater treatment plants to irrigation endpoint of leafy greens. Sci. Total Environ. 2021, 782, 146837. [Google Scholar] [CrossRef] [PubMed]
- Pourret, O. It’s Time to Replace the Term ‘Heavy Metals’ with ‘Potentially Toxic Elements’ when Reporting Environmental Research. Int. J. Environ. Res. Public Health 2019, 16, 4446. [Google Scholar] [CrossRef]
- Letshwenyo, M.W.; Sima, T.V. Phosphorus removal from secondary wastewater effluent using copper smelter slag. Heliyon 2020, 6, e04134. [Google Scholar] [CrossRef]
- Alagha, O.; Manzar, M.S.; Zubair, M.; Anil, I. Comparative Adsorptive Removal of Phosphate and Nitrate from Wastewater Using Biochar-MgAl LDH Nanocomposites: Coexisting Anions Effect and Mechanistic Studies. Nanomaterials 2020, 10, 336. [Google Scholar] [CrossRef] [PubMed]
- Al-housni, M.; Hussein, A.H.; Yeboah, D.; Al Khaddar, R. Electrochemical removal of nitrate from wastewater electrochemical removal of nitrate from wastewater. IOP Conf. Ser. Mater. Sci. Eng. 2020, 888, 012037. [Google Scholar] [CrossRef]
- Taghipour, H.; Mosaferi, M. Heavy Metals in the vegetables collected from production sites. Health Promot. Perspect. 2013, 3, 185–193. [Google Scholar] [CrossRef]
- Duruibe, J.; Ogwuegbu, M.O.C.; Egwurugwu, J.N. Heavy metal pollution and human biotoxic effects. Int. J. Phys. Sci. 2007, 2, 112–118. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater, 16th ed.; American Public Health Association: New York, NY, USA, 1985. [Google Scholar]
- Ametepey, S.T.; Samuel, J.C.; Felix, J.A.; Abudu, B.D.; Zita, N.A. Health risk assessment and heavy metal contamination levels in vegetables from Tamale Metropolis, Ghana. Int. J. Food Contam. 2018, 5, 1–8. [Google Scholar] [CrossRef]
- Usepa, I.R.I.S. Integrated Risk Information System; Environmental Protection Agency Region I: Washington, DC, USA, 2011.
- WHO. Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2011; Volume 216, pp. 303–304. [Google Scholar]
- Malawi Bureau of Standards (MBS). Malawi Standard, Drinking Water—Specification; MS 214:2005, ICS 13.030.40 (First Revision); Malawi Standards Board: Blantyre, Malawi, 2005. [Google Scholar]
- Balkhair, K.S. Microbial contamination of vegetable crop and soil profile in arid regions under controlled application of domestic wastewater. Saudi J. Biol. Sci. 2016, 23, S83–S92. [Google Scholar] [CrossRef] [PubMed]
- Bauder, T.A.; Waskom, R.M.; Davis, J.G. Irrigation Water Quality Criteria; Fact Sheet No 0.506; Colorado State University, Cooperative Extension: Fort Collins, CO, USA, 2004. [Google Scholar]
- Kumar, M.; Avinash, P. A review of permissible limits of drinking water. Indian J. Occup. Environ. Med. 2012, 16, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, U.J.; Duncan, D.M.; Anne, P.; Guillermo, R.P.; Rebecca, S. Guidelines for the microbiological quality of treated wastewater used in agriculture: Recommendations for revising WHO guidelines. Bull. World Health Organ. 2000, 78, 1104–1116. [Google Scholar]
- Hong, H.; Qiu, J.; Yan, L. Environmental factors influencing the distribution of total and fecal coliform bacteria in six water storage reservoirs in the Pearl River Delta Region, China. J. Environ. Sci. 2010, 22, 663–668. [Google Scholar] [CrossRef]
- Ishii, S.; Michael, J.S. Escherichia coli in the environment: Implications for water quality and human health. Microbes Environ. 2008, 23, 101–108. [Google Scholar] [CrossRef]
- Mtethiwa, A.H.; Munyenyembe, A.; Jere, W.; Nyali, E. Efficiency of oxidation ponds in wastewater treatment. Int. J. Environ. Res. 2008, 2, 149–152. [Google Scholar]
- Ekhator, O.C.; Udowelle, N.A.; Igbiri, S.; Asomugha, R.N.; Igweze, Z.N.; Orisakwe, O.E. Safety evaluation of potential toxic metals exposure from street foods consumed in mid-west Nigeria. J. Environ. Public Health 2017, 2017, 8458057. [Google Scholar] [CrossRef]
- Grant, W.; Steele, G.; Isiorho, S.A. Spontaneous abortion possibly related to ingestion of nitrate-contaminated well water—LaGrange County, Indiana, 1991–1994. MMWR Morb. Mortal. Wkly. Rep. 1996, 45, 569–572. [Google Scholar]
- Masona, C.; Loveness, M.; Stenly, M.; Revai, M. Assessment of heavy metal accumulation in wastewater irrigated soil and uptake by maize plants (Zea mays L.) at Firle Farm in Harare. J. Sustain. Dev. 2011, 4, 132. [Google Scholar] [CrossRef][Green Version]
- Zhou, H.; Yang, W.; Zhou, X.; Liu, L.; Gu, J.; Wang, W.; Zou, J.; Tian, T.; Peng, P.; Liao, B. Accumulation of heavy metals in vegetable species planted in contaminated soils and the health risk assessment. Int. J. Environ. Res. Public Health 2016, 13, 289. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, F.R.; Ibrahim, N.H.; Ismail, S.N.S. Heavy metal (As, Cd, and Pb) concentration in selected leafy vegetables from Jengka, Malaysia, and potential health risks. SN Appl. Sci. 2020, 2, 1430. [Google Scholar] [CrossRef]
| In Effluent | ||||||||||
| Dry Season | Nitrate (mg/L) | Phosphate (mg/L) | Temp (°C) | pH | EC (µs/cm) | TDS | E. coli (CFU/g) | Total Coliforms (CFU/g) | COD | BOD |
| Manase WWTP | 39 ± 34 | 2 ± 0.2 | 17 | 6.9 ± 0.06 | 1224 ± 44 | 859 ± 28 | 185,800 ± 60,895 | 433,333 ± 194,415 | 195 ± 89 | 78 ± 45 |
| Soche WWTP | 37 ± 18 | 1.2 ± 0.5 | 22 | 7.66 ± 0.16 | 705 ± 2 | 479 ± 1 | 200,233 ± 82,323 | 647,000 ± 239,380 | 285 ± 86 | 69 ± 23 |
| Rainy season | ||||||||||
| Manase WWTP | 75 ± 56 | 1.4 ± 0.3 | 25 | 6.77 ± 0.18 | 450 ± 40 | 298 ± 28 | 111,466 ± 65,121 | 266,666 ± 30,550 | 137 ± 5 | 73 ± 30 |
| Soche WWTP | 445 ± 92 | 1.75 ± 0.12 | 26 | 6.98 ± 0.11 | 483 ± 23 | 320 ± 16 | 108,453 ± 40,896 | 408,666 ± 204,081 | 133 ± 0.6 | 73 ± 23 |
| MW (MS579:2013) | 50 | 0.15 | ||||||||
| EPA | 5 mg/L | |||||||||
| In Soil Samples | ||||||||||
| Dry Season | Nitrate (mg/L) | Phosphate (mg/L) | pH | EC (µs/cm) | TDS | E. coli (CFU/g) | Total Coliforms (CFU/g) | |||
| Manase WWTP | 187 ± 175 | 649 ± 97 | 5.4 ± 0.22 | 1813 ± 43 | 1289 ± 33 | 171,000 ± 101,424 | 723,000 ± 547,029 | |||
| Soche WWTP | 84 ± 45 | 688 ± 174 | 5.54 ± 0.5 | 2409 ± 158 | 1752 ± 124 | 77,077 ± 21,752 | 723,000 ± 547,029 | |||
| Rainy season | ||||||||||
| Manase WWTP | 1.89 ± 0.14 | 567 ± 98 | 4.85 ± 0.21 | 1793 ± 14 | 1272 ± 10 | 1,887,266 ± 854,892 | 15,280,000 ± 19,505,856 | |||
| Soche WWTP | 12 ± 7 | 582 ± 106 | 4.78 ± 0.32 | 1769 ± 58 | 1255 ± 44 | 526,543 ± 227,594 | 253,233 ± 11,551 | |||
| Heavy Metals in Effluent (Water) | ||||||||
| Rainy Season | Dry Season | |||||||
| Cu (mg/L) | Zn (mg/L) | Cr (mg/L) | Cd (mg/L) | Cu (mg/L) | Zn (mg/L) | Cr (mg/L) | Cd (mg/L) | |
| Manase | 0.298 ± 0.23 | 0.01 ± 0.009 | BDL | BDL | 6.94 ± 0.41 | 0.41 ± 0.23 | BDL | BDL |
| Soche | 0.236 ± 0.17 | 0.004 ± 0.004 | BDL | BDL | BDL | BDL | BDL | 0.55 ± 0.08 |
| MW (MS 579:201) | 2 | 5 | 0.05 | 0.05 | 2 | 5 | 0.05 | 0.05 |
| WHO (1996) | 0.017 | 0.2 | 0.05 | 0.05 | 0.017 | 0.2 | 0.05 | 0.05 |
| Heavy Metals in Soil | ||||||||
| Rainy Season | Dry Season | |||||||
| Cu (mg/kg) | Zn (mg/kg) | Cr (mg/kg) | Cd (mg/kg) | Cu (mg/kg) | Zn (mg/kg) | Cr (mg/kg) | Cd (mg/kg) | |
| Manase | 0.20 ± 0.2 | 0.159 ± 0.14 | 0.002 | 0.38 ± 0.05 | 0.48 ± 0.10 | 2.33 ± 0.75 | 2.32 ± 0.79 | 4.48 ± 0.26 |
| Soche | 0.114 ± 0.62 | 0.625 ± 0.47 | 0.0003 ± 0.0001 | 0.446 ± 11 | 0.87 ± 0.4 | 4.415 ± 5 | 0.062 ± 0.08 | 0.06 ± 0.07 |
| WHO (1996) | 50 | 36 | 100 | 0.8 | 50 | 36 | 100 | 0.8 |
| Heavy Metals in Plants | ||||||||
| Rainy Season | Dry Season | |||||||
| Cu (mg/L) | Zn (mg/L) | Cr (mg/L) | Cd (mg/L) | Cu (mg/L) | Zn (mg/L) | Cr (mg/L) | Cd (mg/L) | |
| Manase | 10 ± 6 | 25 ± 15 | 11 ± 11 | 12 ± 4 | 12 ± 7 | 32 ± 19 | 15 ± 15 | 14 ± 4 |
| Soche | 3 ± 1 | 39 ± 13 | 14 ± 5 | 4.7 ± 4.8 | 0.8 ± 0.3 | 14 ± 5 | 17 ± 11 | 7 ± 5 |
| WHO (1996) | 10 | 0.6 | 1.3 | 0.02 | 10 | 0.6 | 1.3 | 0.02 |
| Heavy Metal | ADD | THQ |
|---|---|---|
| Cu | 0.16 | 4 |
| Zn | 0.43 | 1.4 |
| Cr | 0.2 | 0.13 |
| Cd | 0.187 | 187 |
| Heavy Metal | ADD | THQ |
|---|---|---|
| Cu | 0.01 | 0.27 |
| Zn | 0.19 | 0.62 |
| Cr | 0.23 | 0.15 |
| Cd | 0.09 | 94 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malikula, R.S.; Kaonga, C.C.; Mapoma, H.W.T.; Thulu, F.G.D.; Chiipa, P. Heavy Metals and Nutrients Loads in Water, Soil, and Crops Irrigated with Effluent from WWTPs in Blantyre City, Malawi. Water 2022, 14, 121. https://doi.org/10.3390/w14010121
Malikula RS, Kaonga CC, Mapoma HWT, Thulu FGD, Chiipa P. Heavy Metals and Nutrients Loads in Water, Soil, and Crops Irrigated with Effluent from WWTPs in Blantyre City, Malawi. Water. 2022; 14(1):121. https://doi.org/10.3390/w14010121
Chicago/Turabian StyleMalikula, Raymond S., Chikumbusko C. Kaonga, Harold W. T. Mapoma, Fabiano G. D. Thulu, and Petra Chiipa. 2022. "Heavy Metals and Nutrients Loads in Water, Soil, and Crops Irrigated with Effluent from WWTPs in Blantyre City, Malawi" Water 14, no. 1: 121. https://doi.org/10.3390/w14010121
APA StyleMalikula, R. S., Kaonga, C. C., Mapoma, H. W. T., Thulu, F. G. D., & Chiipa, P. (2022). Heavy Metals and Nutrients Loads in Water, Soil, and Crops Irrigated with Effluent from WWTPs in Blantyre City, Malawi. Water, 14(1), 121. https://doi.org/10.3390/w14010121







