Dynamics of the Spatial Chlorophyll-A Distribution at the Polar Front in the Marginal Ice Zone of the Barents Sea during Spring
Abstract
:1. Introduction
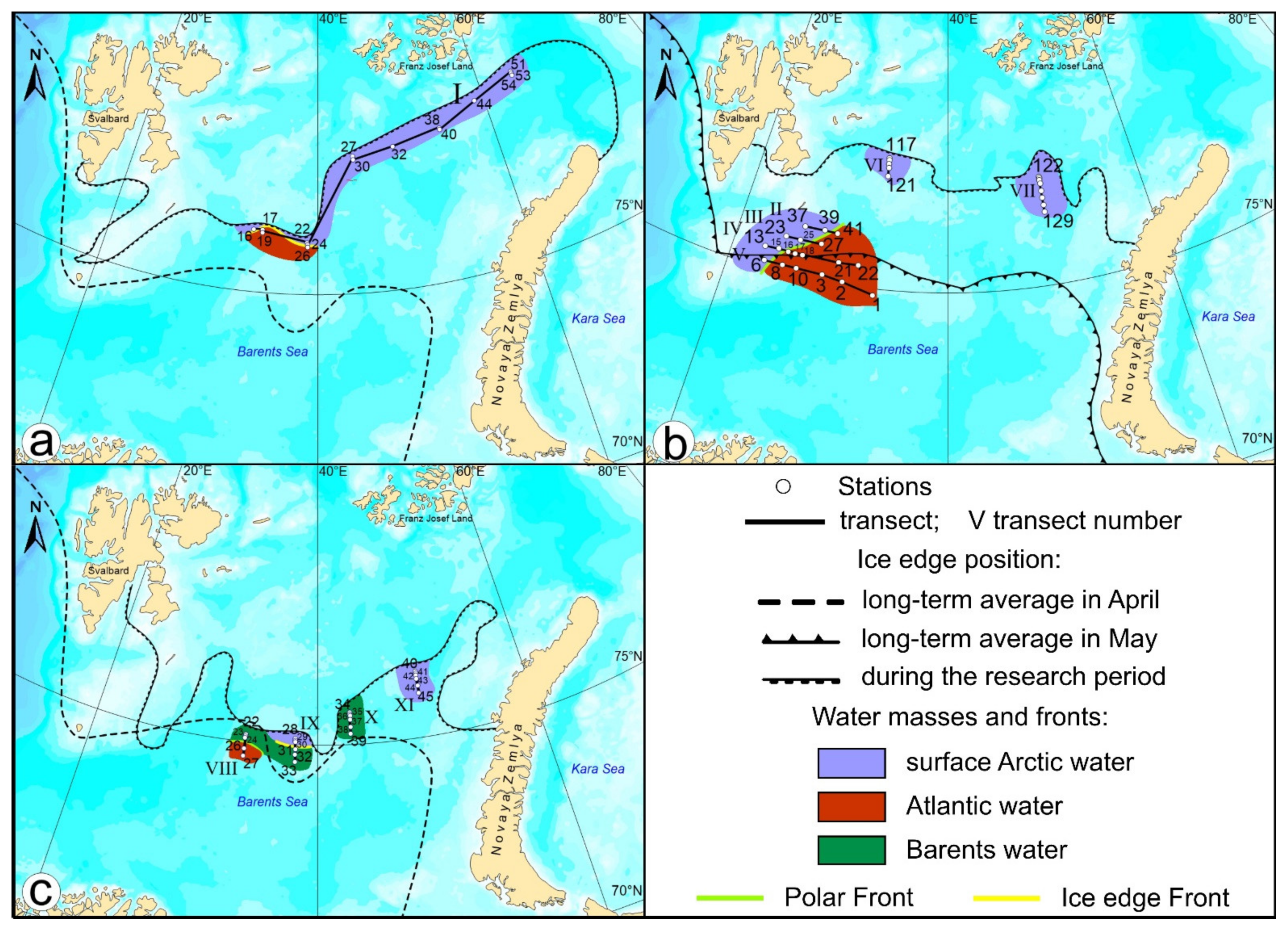
2. Materials and Methods
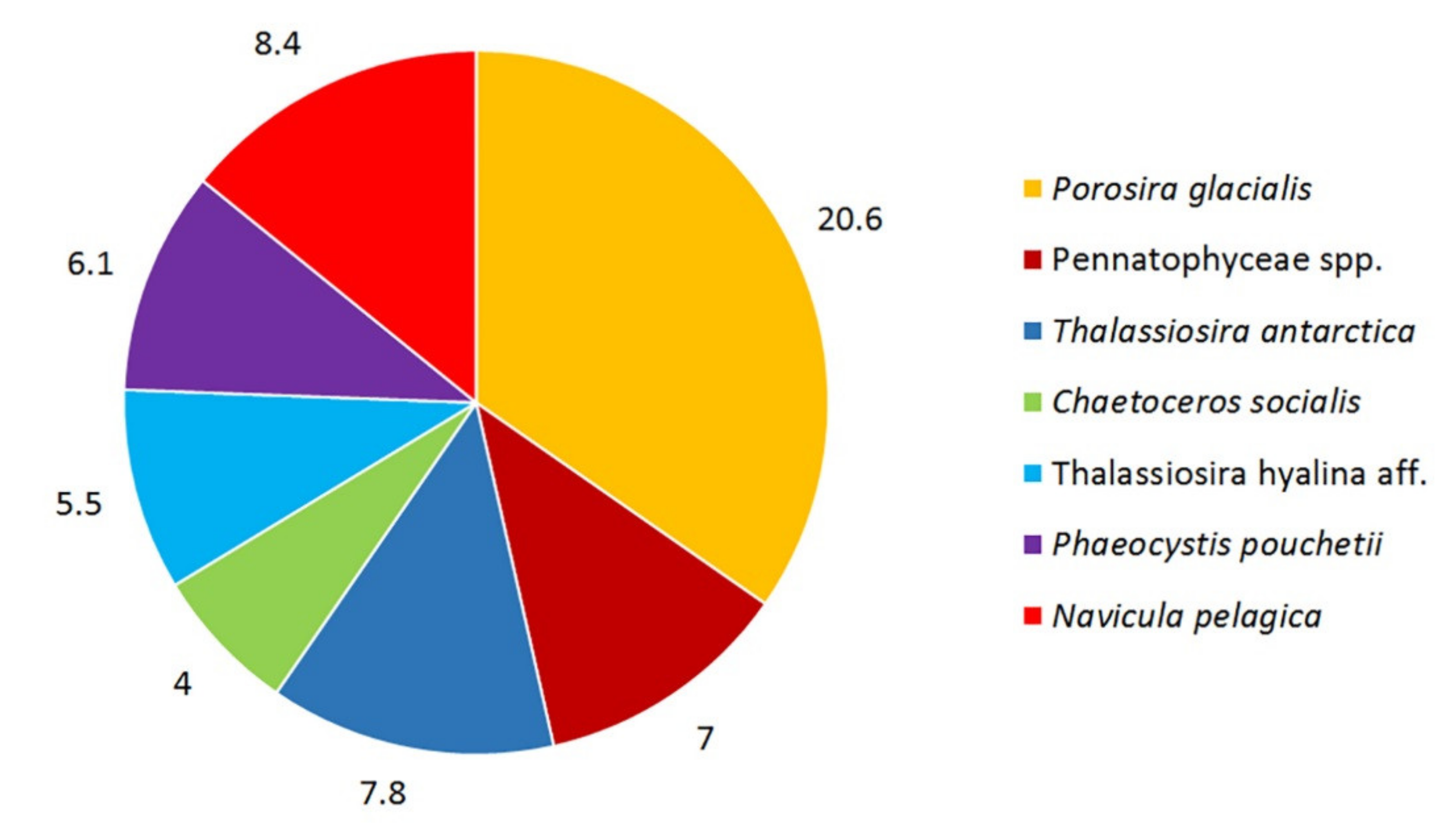
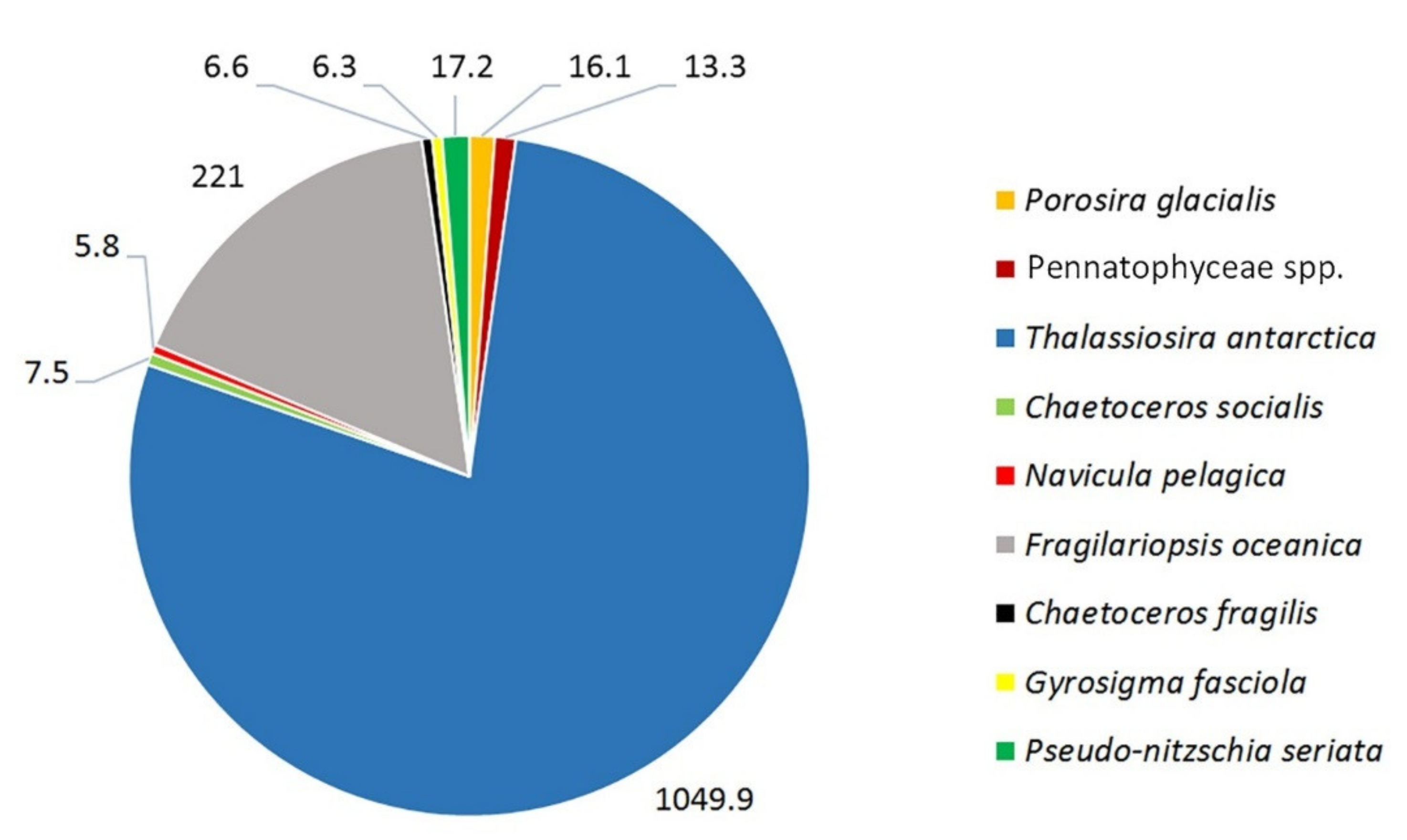
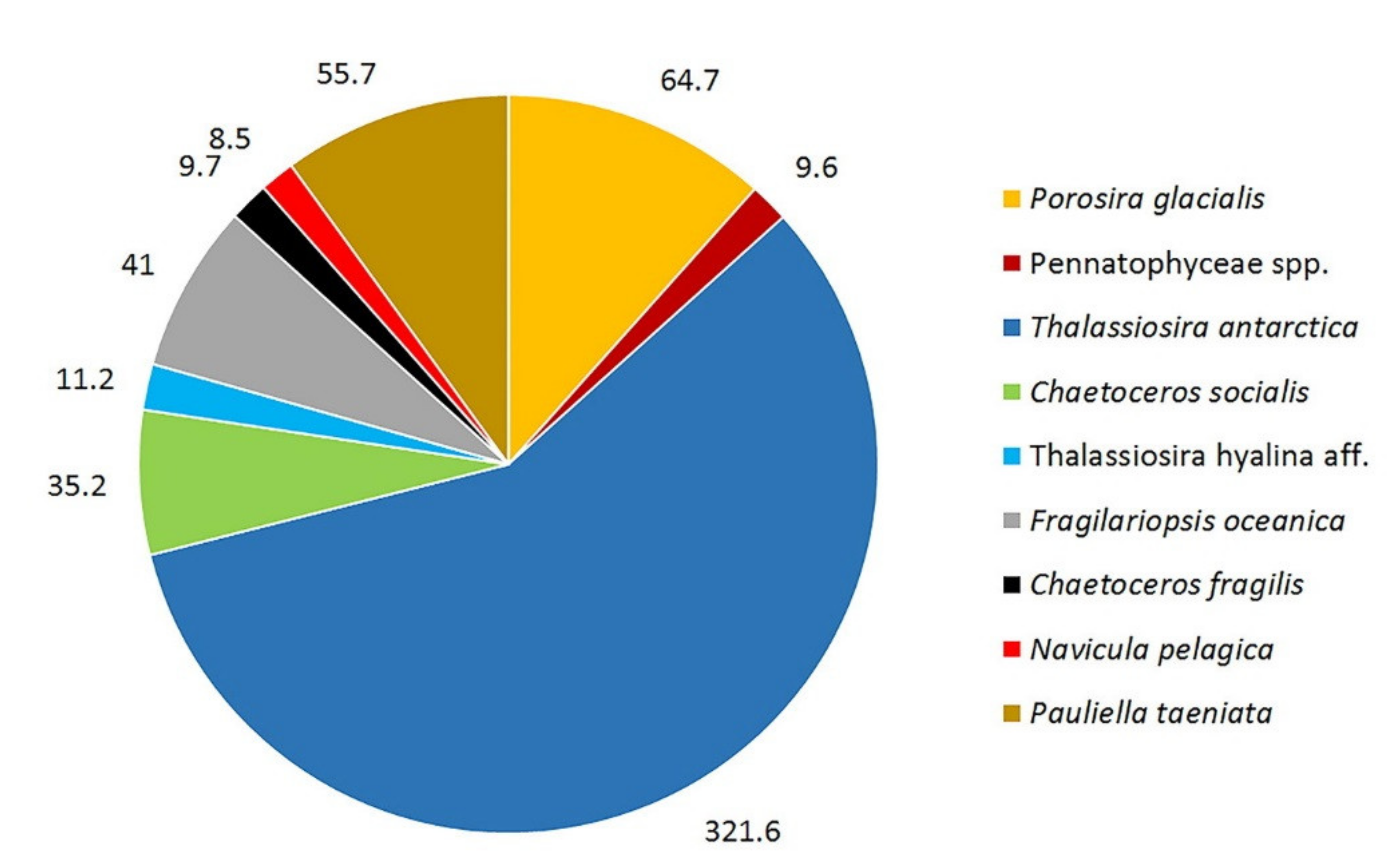
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
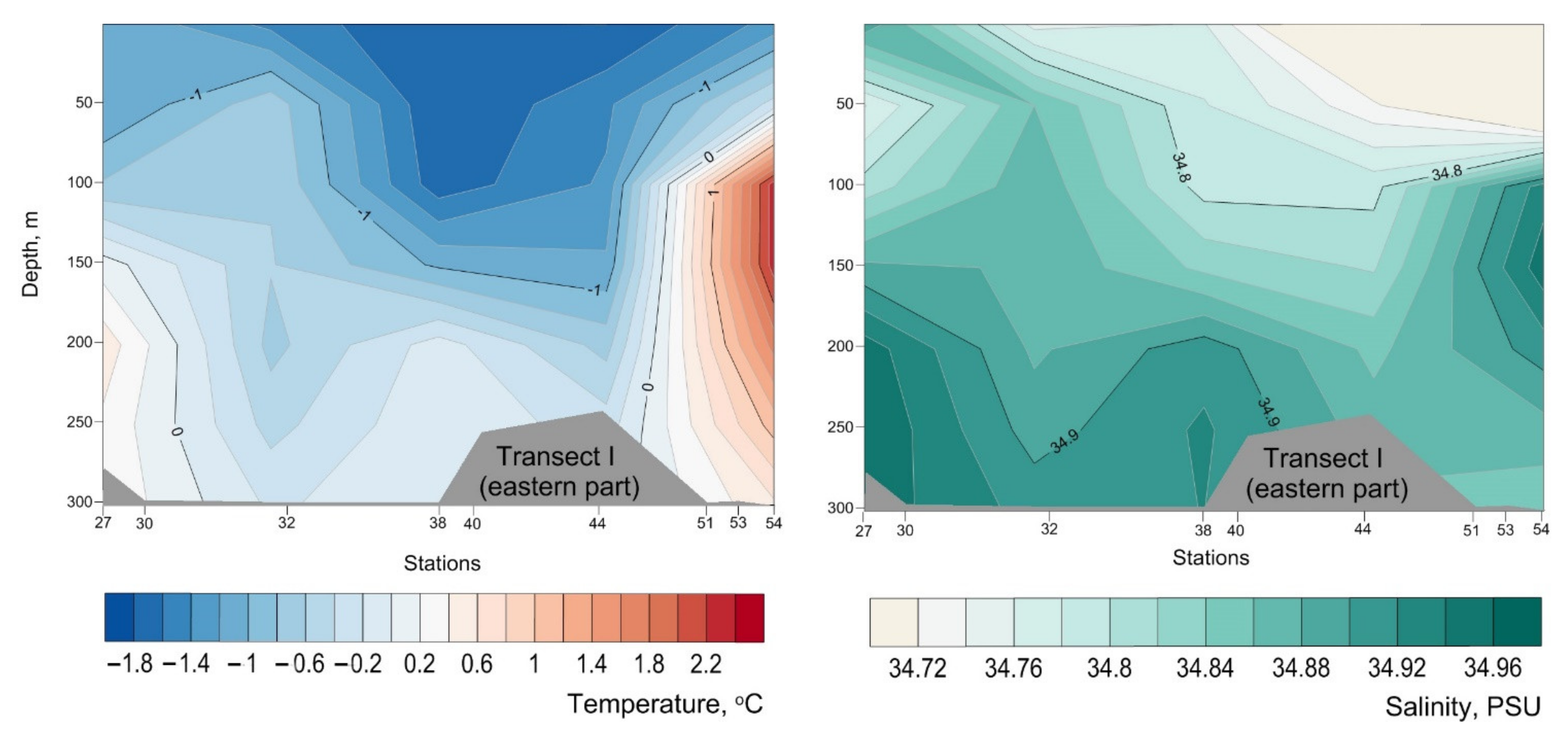
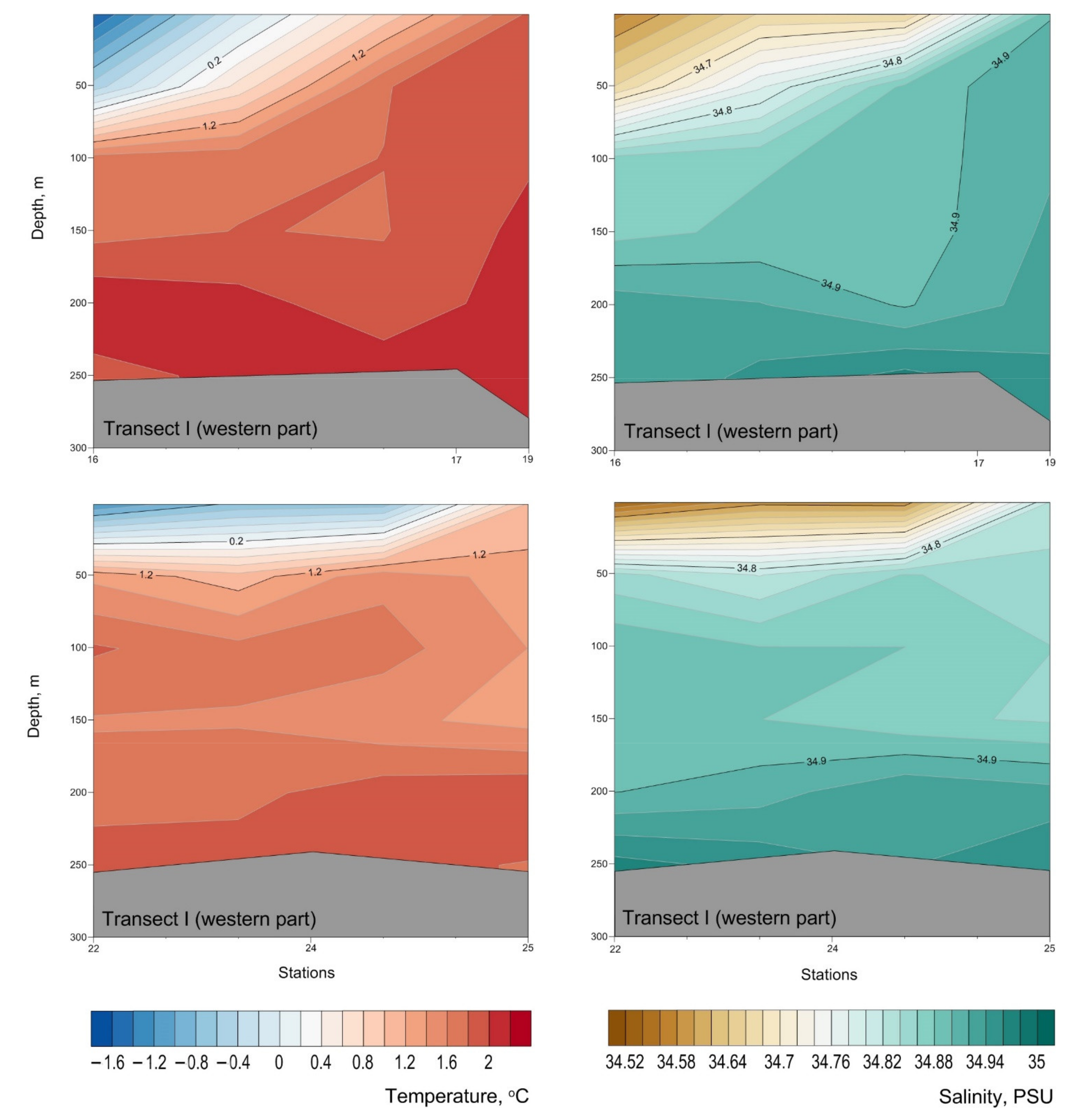
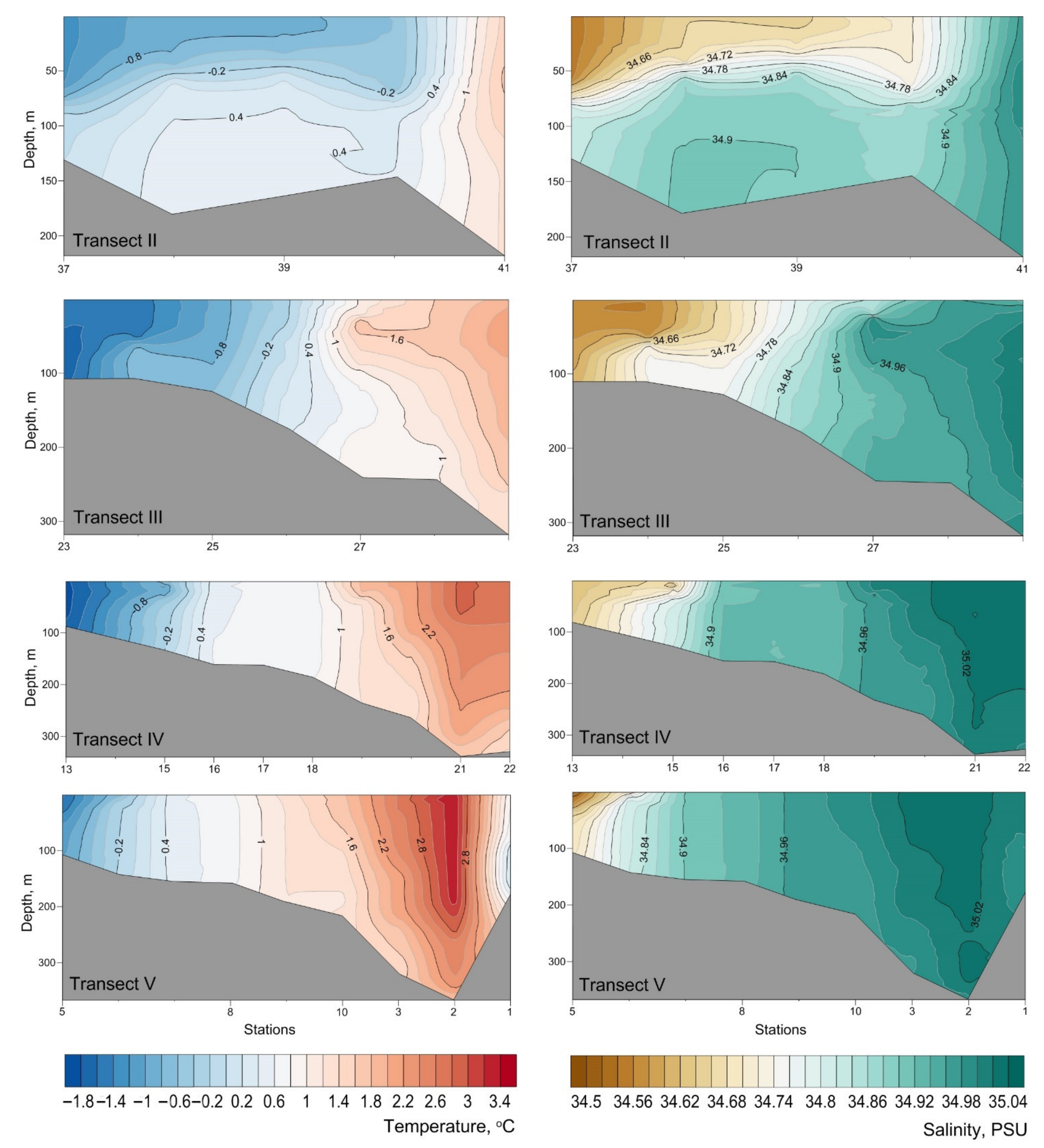
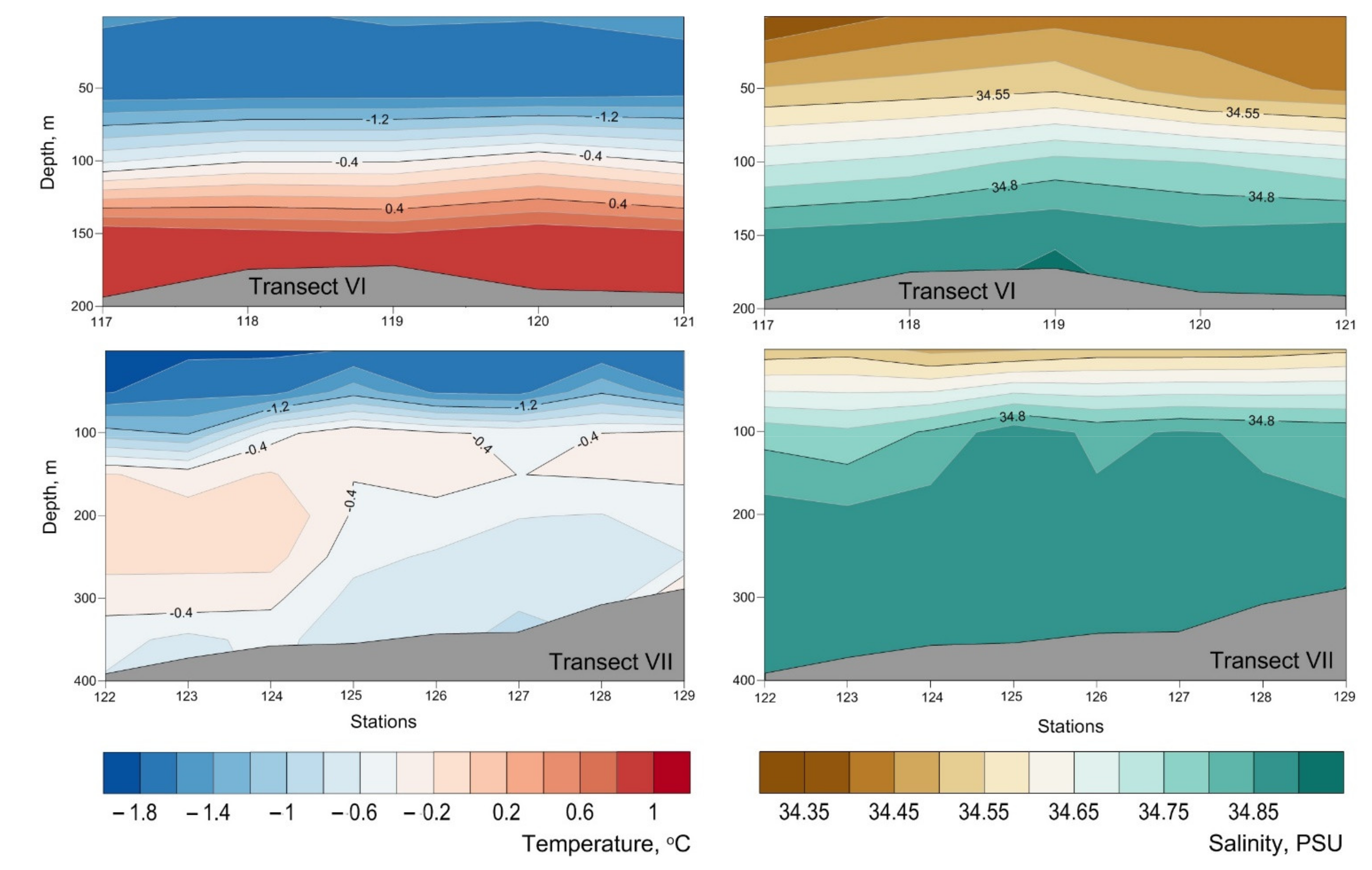
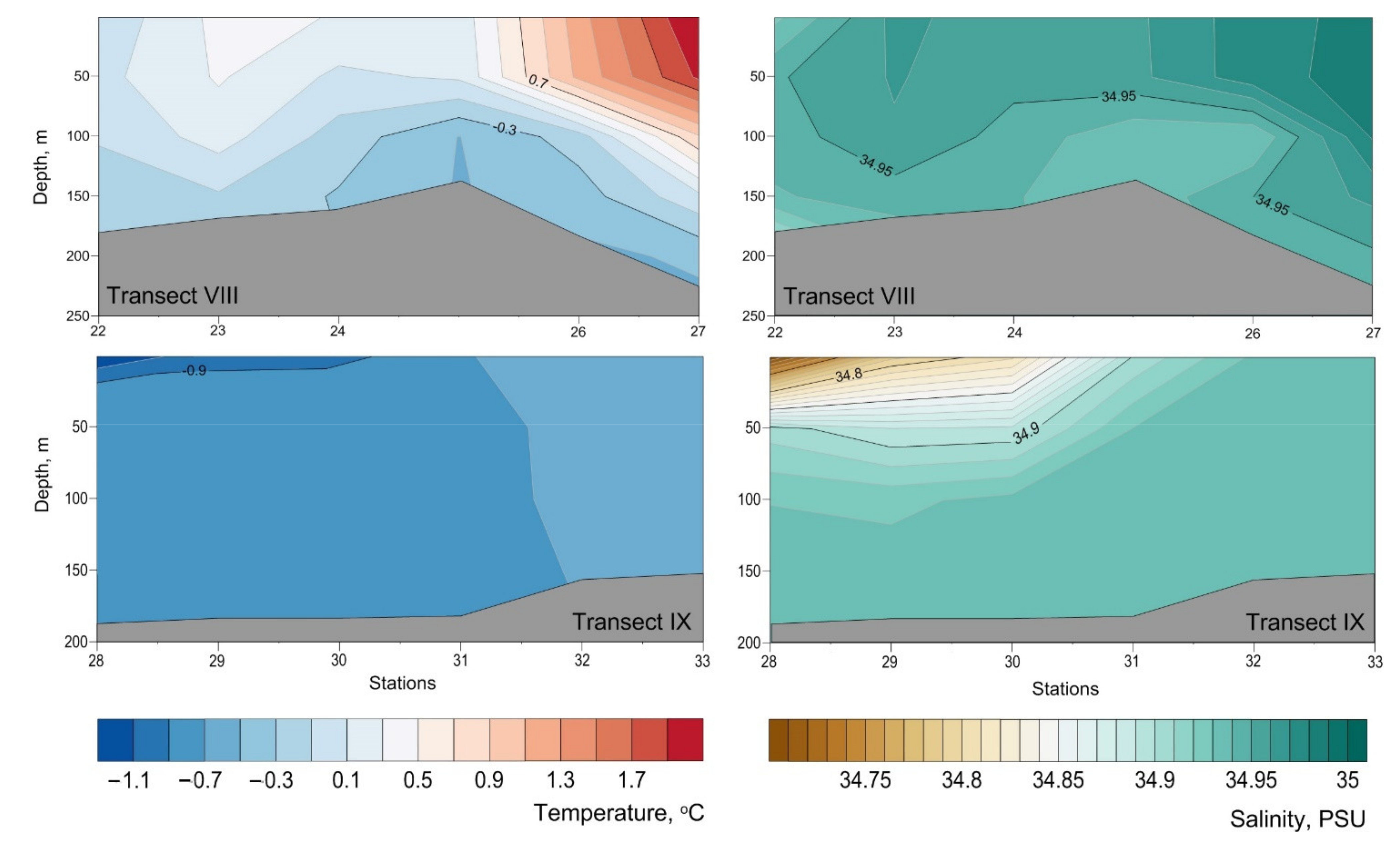
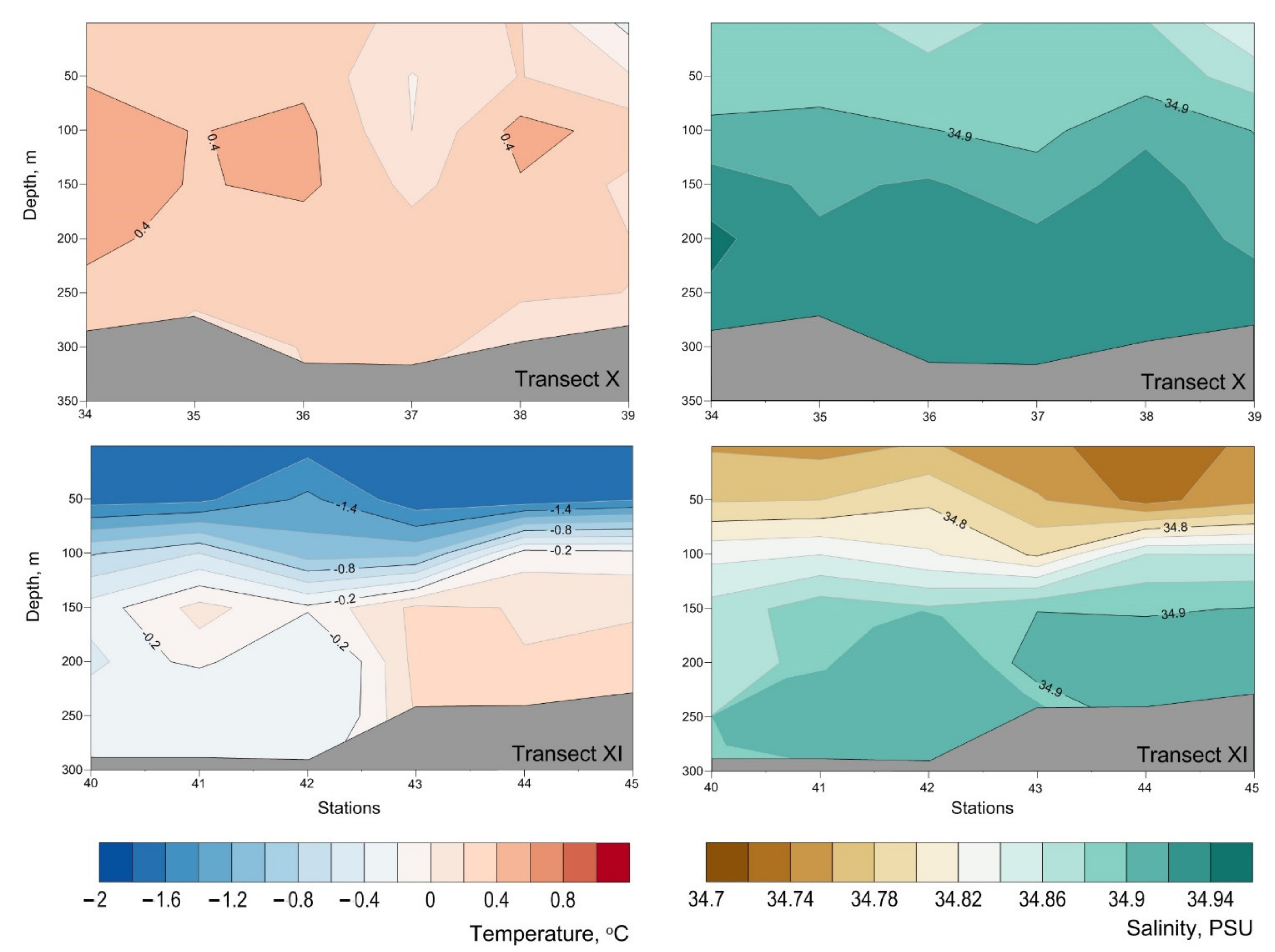
Appendix B
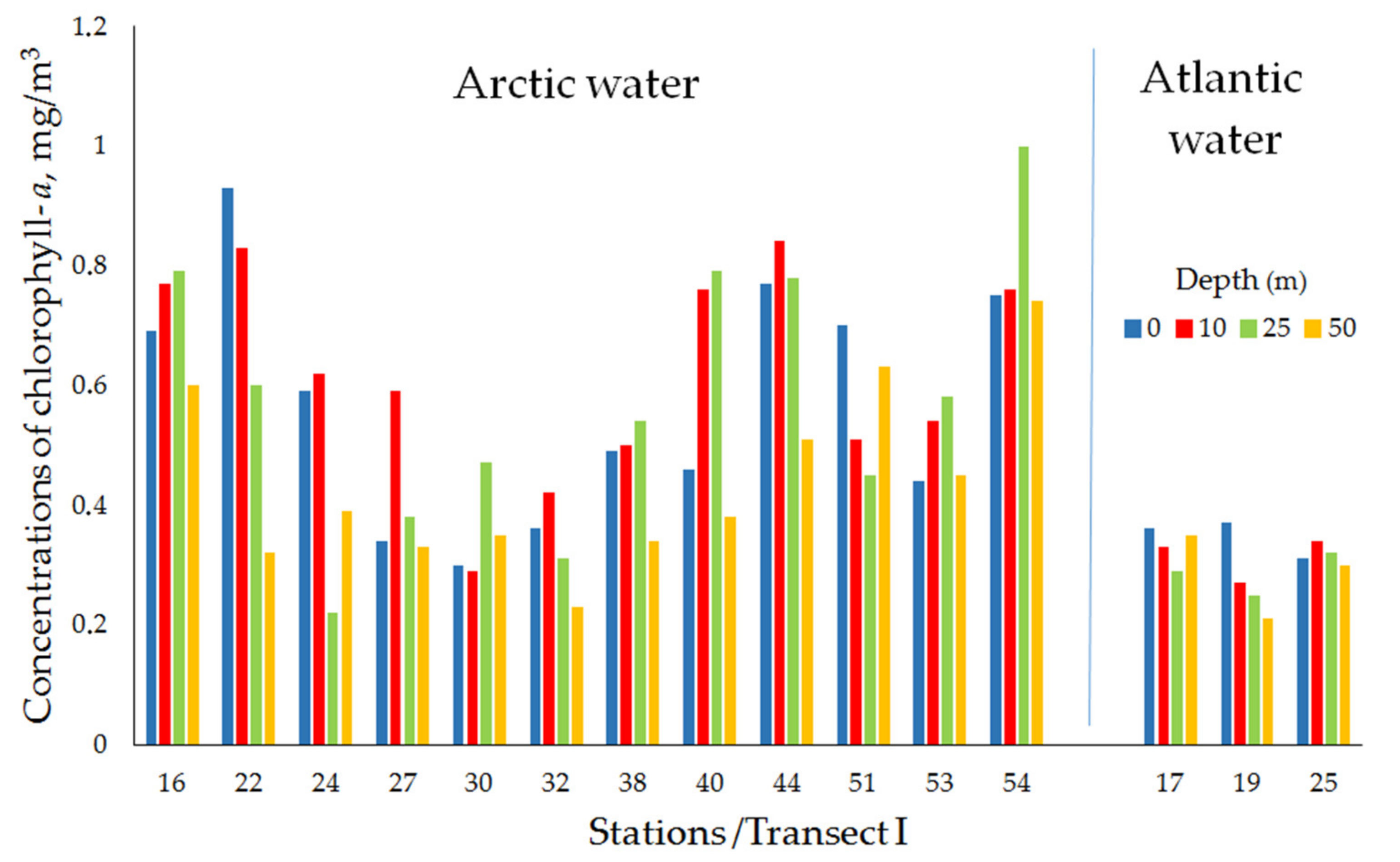
| Water Masses | St. | Date | Coordinates | Total Depth (m) | Chl-a (mg/m3) at Depth (m) | |||
|---|---|---|---|---|---|---|---|---|
| 0 | 10 | 25 | 50 | |||||
| Transect I (Ice-edge/Near-Edge Frontal Zone) | ||||||||
| Arctic water | 16 | 12 April 2016 | 76°26′ N, 33°42′ E | 254 | 0.69 | 0.77 | 0.79 | 0.60 |
| 22 | 14 April 2016 | 76°12′ N, 38°57′ E | 252 | 0.93 | 0.83 | 0.60 | 0.32 | |
| 24 | 14 April 2016 | 76°10′ N, 39°00′ E | 244 | 0.59 | 0.62 | 0.22 | 0.39 | |
| 27 | 16 April 2016 | 78°13′ N, 43°56′ E | 281 | 0.34 | 0.59 | 0.38 | 0.33 | |
| 30 | 16 April 2016 | 78°05′ N, 43°53′ E | 311 | 0.30 | 0.29 | 0.47 | 0.35 | |
| 32 | 17 April 2016 | 78°09′ N, 49°04′ E | 300 | 0.36 | 0.42 | 0.31 | 0.23 | |
| 38 | 20 April 2016 | 78°32′ N, 54°04′ E | 300 | 0.49 | 0.50 | 0.54 | 0.34 | |
| 40 | 20 April 2016 | 78°26′ N, 54°04′ E | 259 | 0.46 | 0.76 | 0.79 | 0.38 | |
| 44 | 21 April 2016 | 78°29′ N, 54°04′ E | 260 | 0.77 | 0.84 | 0.78 | 0.51 | |
| 51 | 25 April 2016 | 79°15′ N, 64°11′ E | 300 | 0.70 | 0.51 | 0.45 | 0.63 | |
| 53 | 26 April 2016 | 79°12′ N, 64°09′ E | 302 | 0.44 | 0.54 | 0.58 | 0.45 | |
| 54 | 26 April 2016 | 79°10′ N, 64°11′ E | 313 | 0.75 | 0.76 | 1.00 | 0.74 | |
| mean ± std | 0.57 ± 0.20 | 0.62 ± 0.17 | 0.57 ± 0.23 | 0.44 ± 0.15 | ||||
| Atlantic water | 17 | 13 April 2016 | 76°27′ N, 34°33′ E | 247 | 0.36 | 0.33 | 0.29 | 0.35 |
| 19 | 13 April 2016 | 76°24′ N, 34°34′ E | 283 | 0.37 | 0.27 | 0.25 | 0.21 | |
| 25 | 14 April 2016 | 75°45′ N, 39°00′ E | 256 | 0.31 | 0.34 | 0.32 | 0.30 | |
| mean ± std | 0.34 ± 0.03 | 0.31 ± 0.04 | 0.29 ± 0.04 | 0.28 ± 0.07 | ||||
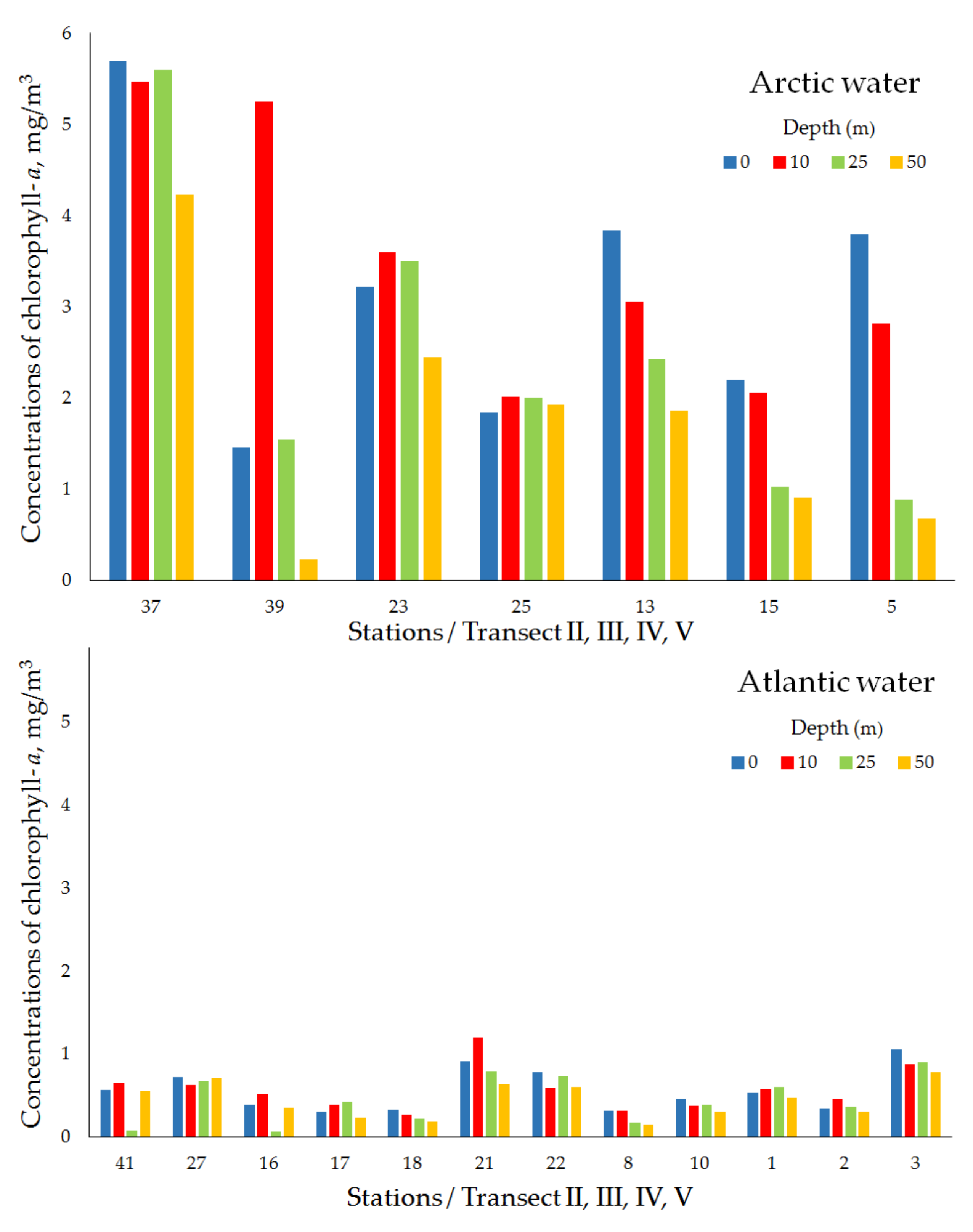
| Transect II (Polar Frontal Zone) | ||||||||
| Arctic water | 37 | 27 April 2018 | 76°10′ N, 26°06′ E | 138 | 5.69 | 5.47 | 5.60 | 4.23 |
| 39 | 27 April 2018 | 76°10′ N, 27°33′ E | 160 | 1.46 | 5.25 | 1.55 | 0.23 | |
| mean ± std | 3.57 ± 3.00 | 5.36 ± 0.15 | 3.57 ± 2.86 | 2.28 ± 2.76 | ||||
| Atlantic water | 41 | 26 April 2018 | 76°10′ N,28°58′ E | 230 | 0.56 | 0.65 | 0.07 | 0.55 |
| Transect III (Polar Frontal Zone) | ||||||||
| Arctic water | 23 | 26 April 2018 | 75°50′ N, 24°36′ E | 106 | 3.22 | 3.60 | 3.50 | 2.45 |
| 25 | 26 April 2018 | 75°50′ N, 25°60′ E | 118 | 1.84 | 2.01 | 2.00 | 1.93 | |
| mean ± std | 2.53 ± 0.97 | 2.80 ± 1.13 | 2.75 ± 1.06 | 2.19 ± 0.36 | ||||
| Atlantic water | 27 | 25 April 2018 | 75°50′ N, 27°21′ E | 242 | 0.72 | 0.62 | 0.67 | 0.71 |
| Transect IV (Polar Frontal Zone) | ||||||||
| Arctic water | 13 | 27 April 2018 | 75°30′ N, 22°54′ E | 73 | 3.84 | 3.05 | 2.42 | 1.86 |
| 15 | 22 April 2018 | 75°30′ N, 24°29′ E | 113 | 2.20 | 2.06 | 1.02 | 0.90 | |
| mean ± std | 2.52 ± 0.45 | 2.56 ± 0.70 | 1.72 ± 0.99 | 1.38 ± 0.68 | ||||
| Atlantic water | 16 | 22 April 2018 | 75°30′ N, 25°10′ E | 159 | 0.39 | 0.51 | 0.06 | 0.35 |
| 17 | 22 April 2018 | 75°30′ N, 25°50′ E | 154 | 0.30 | 0.39 | 0.42 | 0.23 | |
| 18 | 22 April 2018 | 75°30′ N, 26°31′ E | 173 | 0.33 | 0.27 | 0.22 | 0.18 | |
| 21 | 23 April 2018 | 75°30′ N, 29°55′ E | 364 | 0.91 | 1.20 | 0.79 | 0.64 | |
| 22 | 23 April 2018 | 75°30′ N, 31°42′ E | 348 | 0.78 | 0.59 | 0.73 | 0.60 | |
| mean ± std | 0.54 ± 0.28 | 0.59 ± 0.36 | 0.44 ± 0.32 | 0.40 ± 0.21 | ||||
| Transect V (Polar Frontal Zone) | ||||||||
| Arctic water | 5 | 21 April 2018 | 75°10′ N, 23°00′ E | 107 | 3.79 | 2.82 | 0.88 | 0.68 |
| Atlantic water | 8 | 21 April 2018 | 75°10′ N, 24°56′ E | 155 | 0.31 | 0.31 | 0.17 | 0.15 |
| 10 | 21 April 2018 | 75°10′ N, 26°12′ E | 207 | 0.46 | 0.37 | 0.38 | 0.30 | |
| 1 | 20 April 2018 | 74°51′ N, 33°19′ E | 172 | 0.53 | 0.58 | 0.60 | 0.47 | |
| 2 | 20 April 2018 | 75°04′ N, 30°28′ E | 386 | 0.34 | 0.46 | 0.36 | 0.30 | |
| 3 | 21 April 2018 | 75°09′ N, 28°35′ E | 336 | 1.05 | 0.87 | 0.90 | 0.78 | |
| mean ± std | 0.54 ± 0.30 | 0.52 ± 0.22 | 0.48 ± 0.28 | 0.40 ± 0.24 | ||||
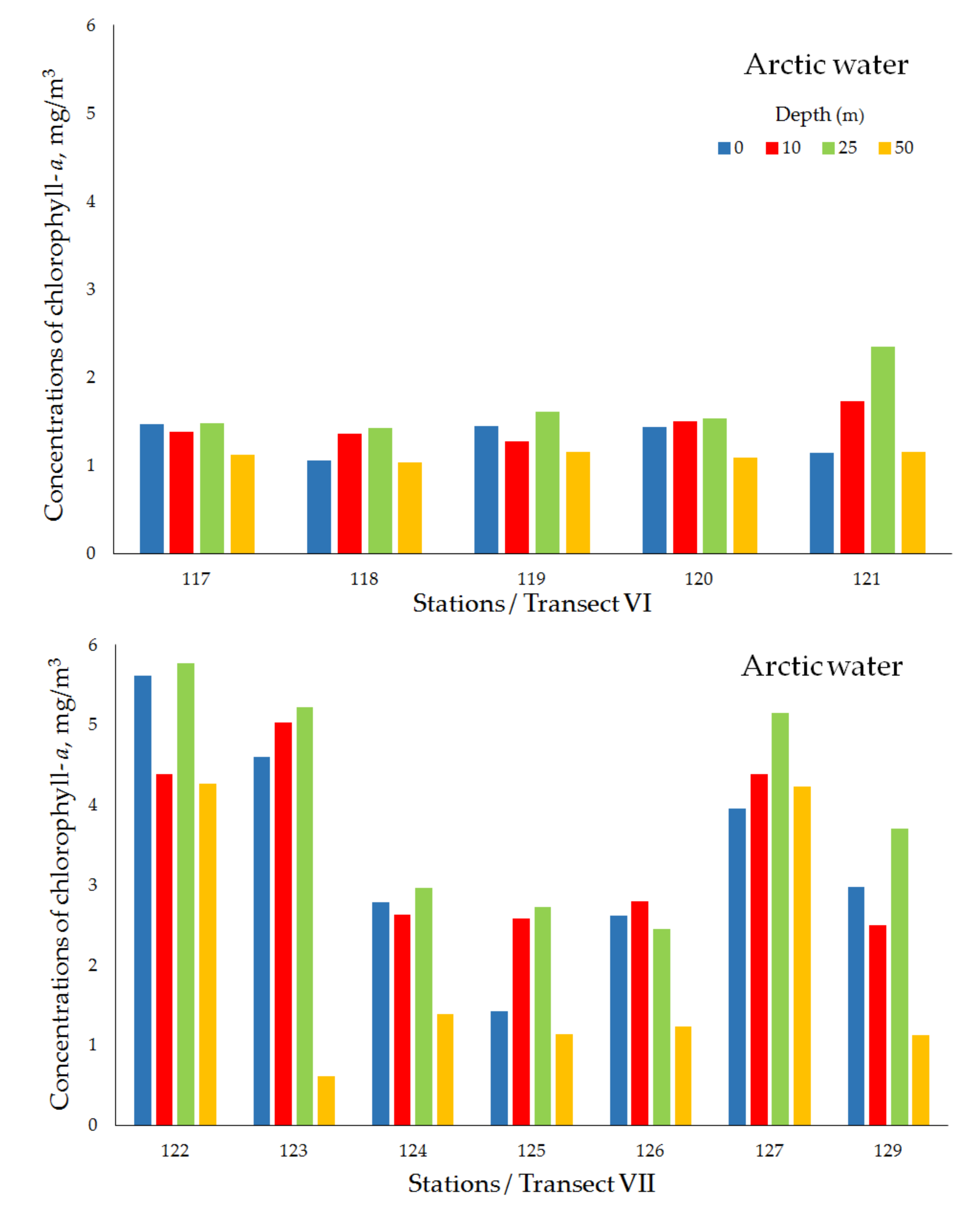
| Transect VI (Ice edge) | ||||||||
| Arctic water | 117 | 19 May 2018 | 78°02′ N, 33°30′ E | 195 | 1.47 | 1.38 | 1.48 | 1.12 |
| 118 | 19 May 2018 | 77°59′ N, 33°29′ E | 176 | 1.06 | 1.36 | 1.43 | 1.04 | |
| 119 | 20 May 2018 | 77°57′ N, 33°30′ E | 175 | 1.45 | 1.27 | 1.61 | 1.15 | |
| 120 | 20 May 2018 | 77°52′ N, 33°30′ E | 186 | 1.44 | 1.50 | 1.54 | 1.09 | |
| 121 | 20 May 2018 | 77°41′ N, 33°28′ E | 189 | 1.14 | 1.73 | 2.35 | 1.16 | |
| mean ± std | 1.31 ± 0.20 | 1.45 ± 0.18 | 1.68 ± 0.38 | 1.11 ± 0.05 | ||||
| Transect VII (Ice edge) | ||||||||
| Arctic water | 122 | 21 May 2018 | 77°31′ N, 49°40′ E | 398 | 5.61 | 4.38 | 5.77 | 4.27 |
| 123 | 22 May 2018 | 77°28′ N, 49°40′ E | 377 | 4.60 | 5.03 | 5.22 | 0.61 | |
| 124 | 22 May 2018 | 77°26′ N, 49°39′ E | 363 | 2.78 | 2.63 | 2.96 | 1.39 | |
| 125 | 22 May 2018 | 77°20′ N, 49°41′ E | 361 | 1.42 | 2.58 | 2.73 | 1.14 | |
| 126 | 22 May 2018 | 77°10′ N, 49°40′ E | 338 | 2.62 | 2.80 | 2.45 | 1.23 | |
| 127 | 23 May 2018 | 77°00′ N, 49°40′ E | 307 | 3.96 | 4.38 | 5.15 | 4.23 | |
| 129 | 23 May 2018 | 76°40′ N, 49°39′ E | 288 | 2.97 | 2.50 | 3.70 | 1.12 | |
| mean ± std | 3.42 ± 1.40 | 3.47 ± 1.08 | 4.00 ± 1.36 | 2.00 ± 1.56 | ||||
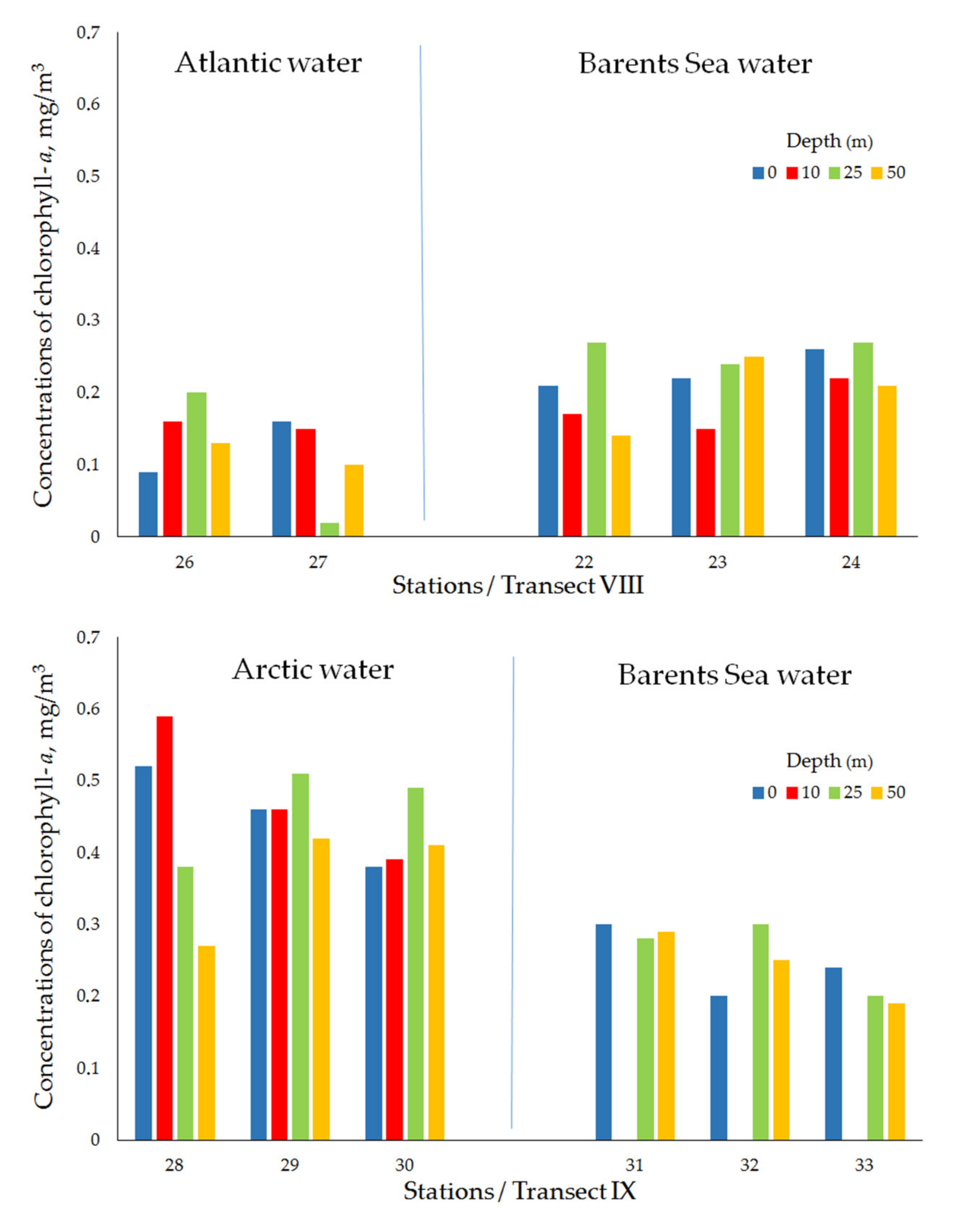
| Transect VIII (Ice edge/Polar Frontal Zone) | ||||||||
| Atlantic water | 26 | 10 April 2019 | 74°51′ N, 33°30′ E | 184 | 0.09 | 0.16 | 0.20 | 0.13 |
| 27 | 10 April 2019 | 74°40′ N, 33°29′ E | 227 | 0.16 | 0.15 | 0.02 | 0.10 | |
| mean ± std | 0.13 ± 0.05 | 0.15 ± 0.01 | 0.11 ± 0.13 | 0.12 ± 0.03 | ||||
| Barents Sea water | 22 | 9April 2019 | 75°00′ N, 33°30′ E | 180 | 0.21 | 0.17 | 0.27 | 0.14 |
| 23 | 9 April 2019 | 75°08′ N,33°30′ E | 169 | 0.22 | 0.15 | 0.24 | 0.25 | |
| 24 | 10 April 2019 | 75°06′ N, 33°29′ E | 160 | 0.26 | 0.22 | 0.27 | 0.21 | |
| mean ± std | 0.23 ± 0.03 | 0.18 ± 0.03 | 0.26 ± 0.02 | 0.20 ± 0.06 | ||||
| Transect IX (Ice edge/Near-Edge Frontal Zone) | ||||||||
| Arctic water | 28 | 11 April 2019 | 75°06′ N, 37°59′ E | 187 | 0.52 | 0.59 | 0.38 | 0.27 |
| 29 | 11 April 2019 | 75°03′ N, 38°00′ E | 182 | 0.46 | 0.46 | 0.51 | 0.42 | |
| 30 | 11 April 2019 | 75°02′ N, 37°60′ E | 182 | 0.38 | 0.39 | 0.49 | 0.41 | |
| mean ± std | 0.45 ± 0.07 | 0.48 ± 0.10 | 0.46 ± 0.07 | 0.37 ± 0.08 | ||||
| Barents Sea water | 31 | 11 April 2019 | 74°56′ N, 38°02′ E | 180 | 0.30 | no data | 0.28 | 0.29 |
| 32 | 12 April 2019 | 74°46′ N, 37°60′ E | 155 | 0.20 | no data | 0.30 | 0.25 | |
| 33 | 11 April 2019 | 74°36′ N, 37°60′ E | 148 | 0.24 | no data | 0.20 | 0.19 | |
| mean ± std | 0.25 ± 0.05 | no data | 0.26 ± 0.05 | 0.24 ± 0.05 | ||||
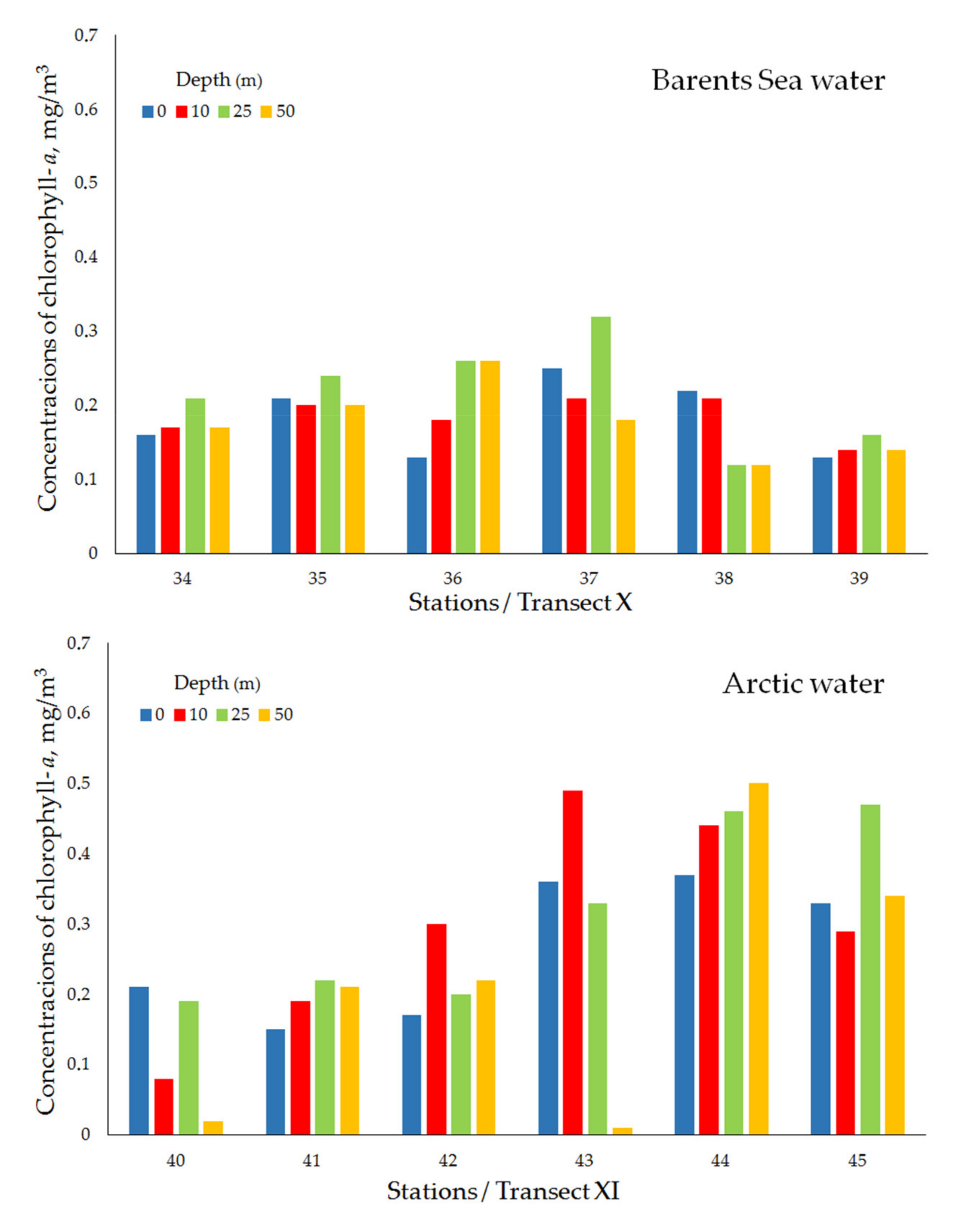
| Transect X (Ice edge) | ||||||||
| Barents Sea water | 34 | 13 April 2019 | 75°45′ N, 43°00′ E | 285 | 0.16 | 0.17 | 0.21 | 0.17 |
| 35 | 13 April 2019 | 75°43′ N, 42°59′ E | 271 | 0.21 | 0.20 | 0.24 | 0.20 | |
| 36 | 13 April 2019 | 75°40′ N, 42°60′ E | 313 | 0.13 | 0.18 | 0.26 | 0.26 | |
| 37 | 13 April 2019 | 75°35′ N, 43°01′ E | 318 | 0.25 | 0.21 | 0.32 | 0.18 | |
| 38 | 13 April 2019 | 75°25′ N, 43°01′ E | 294 | 0.22 | 0.21 | 0.12 | 0.12 | |
| 39 | 14 April 2019 | 75°15′ N, 42°60′ E | 280 | 0.13 | 0.14 | 0.16 | 0.14 | |
| mean ± std | 0.18 ± 0.05 | 0.18 ± 0.03 | 0.22 ± 0.07 | 0.18 ± 0.05 | ||||
| Transect XI (Ice edge) | ||||||||
| Arctic water | 40 | 14 April 2019 | 76°32′ N, 49°40′ E | 290 | 0.21 | 0.08 | 0.19 | 0.02 |
| 41 | 15 April 2019 | 76°29′ N, 49°41′ E | 290 | 0.15 | 0.19 | 0.22 | 0.21 | |
| 42 | 15 April 2019 | 76°27′ N, 49°41′ E | 292 | 0.17 | 0.30 | 0.20 | 0.22 | |
| 43 | 15 April 2019 | 76°22′ N, 49°40′ E | 246 | 0.36 | 0.49 | 0.33 | 0.01 | |
| 44 | 15 April 2019 | 76°12′ N, 49°40′ E | 245 | 0.37 | 0.44 | 0.46 | 0.50 | |
| 45 | 15 April 2019 | 76°02′ N, 49°41′ E | 235 | 0.33 | 0.29 | 0.47 | 0.34 | |
| mean ± std | 0.27 ± 0.10 | 0.30 ± 0.15 | 0.31 ± 0.13 | 0.21 ± 0.19 | ||||
Appendix C
| Depth (m) | ||||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 25 | 50 | 100 | |||||
| min | max | min | max | min | max | min | max | |
| P-РО4 (µM) | ||||||||
| 0.14 | 0.55 | 0.21 | 0.50 | 0.36 | 0.56 | 0.49 | 0.68 | |
| mean ± std | 0.37 ± 0.12 | 0.33 ± 0.08 | 0.46 ± 0.13 | 0.60 ± 0.05 | ||||
| N-NO3 (µM) | ||||||||
| 1.84 | 15.53 | 0.53 | 13.81 | 0.94 | 15.08 | 8.24 | 18.98 | |
| mean ± std | 8.99 ± 3.24 | 7.14 ± 4.21 | 7.09 ± 3.46 | 13.59 ± 2.54 | ||||
| Depth (m) | ||||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 25 | 50 | 100 | |||||
| min | max | min | max | min | max | min | max | |
| P-PO4 (µM) | ||||||||
| 0.43 | 0.85 | 0.31 | 0.59 | 0.43 | 0.70 | 0.41 | 0.81 | |
| mean ± std | 0.58 ± 0.05 | 0.46 ± 0.04 | 0.60 ± 0.04 | 0.58 ± 0.04 | ||||
| N-NO3 (µM) | ||||||||
| 4.63 | 12.01 | 2.18 | 10.26 | 1.53 | 10.98 | 4.83 | 11.81 | |
| mean ± std | 9.83 ± 0.95 | 7.24 ± 1.25 | 8.20 ± 1.39 | 8.79 ± 1.03 | ||||
References
- Lien, V.S.; Trofimov, A.G. Formation of Barents Sea Branch Water in the north-eastern Barents Sea. Polar Res. 2013, 32, 18905. [Google Scholar] [CrossRef] [Green Version]
- Rudels, B.; Anderson, L.G.; Jones, E.P. Formation and evolution of the surface mixed layer and halocline of the Arctic Ocean. J. Geophys. Res. 1996, 101, 8807–8821. [Google Scholar] [CrossRef]
- Averkiev, A.S. Transformation of Atlantic water masses in the Barents, Kara and Laptev Sea by observation data in september 2014 year. Int. Res. J. 2017, 7, 37–41. [Google Scholar] [CrossRef]
- Kazmin, A.S. Structure and Variability of Oceanic Frontal Zones: Analysis of Global Satellite Information. Ph.D. Dissertation, Science, IO RAS, Moscow, Russia, 2014; p. 43. [Google Scholar]
- Ivshin, V.; Trofimov, A.; Titov, O. Barents Sea thermal frontal zones in 1960–2017: Variability, weakening, shifting. Ices J. Mar. Sci. 2019, 76, 13–19. [Google Scholar] [CrossRef]
- Kostianoy, A.G.; Nihoul, J.C.J.; Rodionov, V.B. Physical Oceanography of Frontal Zones in the Subarctic Seas. Elsevier Oceanogr. Ser. 2004, 71, 5–8. [Google Scholar] [CrossRef]
- Sergeeva, V.M.; Mosharov, S.A.; Nedospasov, A.A.; Polukhin, A.A. Phytoplankton community structure in the polar front of the Eastern Barents Sea at the end of the growth season. Oceanology 2018, 58, 700–709. [Google Scholar] [CrossRef]
- Monaco, A.; Prouzet, P. Biodiversity of Phytoplankton: Responses to Environmental Changes in Coastal Zones. In Marine Ecosystems; Wiley&Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 25–80. [Google Scholar] [CrossRef]
- Benedetti, F.; Vogt, M.; Elizondo, U.H.; Righetti, D.; Zimmermann, N.E.; Gruber, N. Major restructuring of marine plankton assemblages under global warming. Nat. Commun. 2021, 12, 5226. [Google Scholar] [CrossRef]
- Perrette, M.; Yool, A.; Quartly, G.; Popova, E. Near-ubiquity of ice-edge blooms in the Arctic. Biogeosciences 2011, 8, 515–524. [Google Scholar] [CrossRef] [Green Version]
- Dong, K.; Kvile, Ø.K.; Stenseth, N.C.; Stige, L.C. Associations among temperature, sea ice and phytoplankton bloom dynamics in the Barents Sea. Mar. Ecol. Prog. Ser. 2020, 635, 25–36. [Google Scholar] [CrossRef]
- Engelsen, O.; Hegseth, E.; Hop, H.; Hansen, E.; Falk-Petersen, S. Spatial variability of chlorophyll-a in the marginal ice zone of the Barents Sea, with relations to sea ice and oceanographic conditions. J. Mar. Syst. 2002, 35, 79–97. [Google Scholar] [CrossRef]
- Hodal, H.; Kristiansen, S. The importance of small-celled phytoplankton in spring bloomsat the marginal ice zone in the northern Barents Sea. Deep Sea Res. 2008, 55, 2176–2185. [Google Scholar] [CrossRef] [Green Version]
- Reigstad, M.; Wassmann, P.; Riser, C.; Øygarden, S.; Rey, F. Variations in hydrography, nutrients and chlorophyll a in the marginal ice-zone and the central Barents Sea. J. Mar. Syst. 2002, 38, 9–29. [Google Scholar] [CrossRef]
- Wassmann, P.; Ratkova, T.; Andreassen, I.; Vernet, M.; Pedersen, G.; Rey, F. Spring bloom development in the marginal ice zone and the central Barents Sea. Mar. Ecol. 1999, 20, 321–346. [Google Scholar] [CrossRef] [Green Version]
- Sturluson, M.; Nielsen, T.; Wassmann, P. Bacterial abundance, biomass and production duringspring blooms in the northern Barents Sea. Deep Sea Res. 2008, 55, 2186–2198. [Google Scholar] [CrossRef]
- Degerlund, M.; Eilertsen, H.C. Main Species Characteristics of Phytoplankton Spring Blooms in NE Atlantic and Arctic Waters (68–80° N). Estuaries Coasts 2010, 33, 242–269. [Google Scholar] [CrossRef]
- Wang, Y.; Xiang, P.; Kang, J.; Ye, Y.Y.; Lin, G.M.; Yang, Q.L.; Lin, M. Microphytoplankton community structure in the western Arctic Ocean: Surface layer variability of geographic and temporal considerations in summer. Hydrobiologia 2018, 811, 295–312. [Google Scholar] [CrossRef]
- Johnsen, G.; Norli, M.; Moline, M.; Robbins, I.; Quillfeldt, S.; Søresen, K.; Cottier, F.; Berge, J. The advective origin of an under-ice spring bloom in the Arctic Ocean using multiple observational platforms. Polar Biol. 2018, 41, 1197–1216. [Google Scholar] [CrossRef] [Green Version]
- Makarevich, P.; Vodopianova, V.; Bulavina, A.; Kalinka, O. Influence of the Barents Sea Frontal zones on chlorophyll concentration in Spring. KnE Life Sci. 2020, 5, 536–546. [Google Scholar] [CrossRef] [Green Version]
- Mamaev, O.I. Thermohaline Analysis of the World Ocean Waters; Hydrometeoizdat: Leningrad, Russia, 1987. [Google Scholar]
- Ozhigin, V.; Ivshin, V.; Trofimov, A.; Karsakov, A.L.; Antsiferov, M. The Barents Sea Water: Structure, Circulation, Variability; PINRO Press: Murmansk, Russia, 2016. [Google Scholar]
- Murphy, J.; Riley, J.P. Modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Strickland, J.D.H.; Parsons, T.R. A practical handbook of seawater analysis. In Fisheries Research Board of Canada, 2nd ed.; Watson, J., Reinhart, J.M., Cook, D.J., Eds.; Alger Press Ltd.: Ottawa, ON, Canada, 1972; p. 311. [Google Scholar]
- Water. Spectrophotometric Determination of Chlorophyll-a (State Standard: 17.1.4.02–90); Standards: Moscow, Russia, 2001. [Google Scholar]
- Aminot, A.; Rey, F. Standard Procedure for the Determination of Chlorophyll a by Spectroscopic Methods; International Council for the Exploration of the Sea: Copenhagen, Denmark, 2000. [Google Scholar]
- Dodson, A.; Thomas, W. Concentrating plankton in a gentle fashion. Limnol. Oceanogr. 1964, 9, 455–456. [Google Scholar] [CrossRef]
- Sun, J.; Liu, D. Geometric models for calculating cell biovolume and surface area for phytoplankton. J. Plankton Res. 2003, 25, 1331–1346. [Google Scholar] [CrossRef] [Green Version]
- Mahotin, M.; Ivanov, V. Distribution of Atlantic water masses in the Barents Sea according to observations and numerical modeling. Proc. Hydrometeorol. Res. Cent. Russ. Fed. 2016, 361, 169–191. [Google Scholar]
- Zimin, A.; Konik, A.; Atadzhanova, O. Quantitative estimations of the variability of characteristics of the temperature of the sea (sst) surface in the front of the frontal zone of the Barents Sea. Sch. Notes RSHU 2018, 51, 99–108. [Google Scholar]
- Barton, B.; Lenn, Y.; Lique, C. Observed Atlantification of the Barents Sea Causes the Polar Front to Limit the Expansion of Winter Sea Ice. J. Phys. Oceanogr. 2018, 48, 1849–1866. [Google Scholar] [CrossRef]
- Plankton of the Sea of the Western Arctic; KSC RAS Press: Apatity, Russia, 1997.
- Zhichkin, A.P. Peculiarities of interannual and seasonal variations of the Barents Sea ice coverage anomalies. Russ. Meteorol. Hydrol. 2015, 40, 319–326. [Google Scholar] [CrossRef]
- Dalpadado, P.; Arrigo, K.; Hjøllo, S.; Rey, F.; Ingvaldsen, R.B.; Sperfeld, E.; Dijken, G.L.; Stige, L.C.; Olsen, A.; Ottersen, G. Productivity in the Barents Sea—Response to Recent Climate Variability. PLoS ONE 2014, 9, e95273. [Google Scholar] [CrossRef] [Green Version]
- Ryabchenko, V.; Gorchakov, V.; Dvornikov, A.; Pugalova, S. Estimating the effect of ice on the primary production of phytoplankton in the Barents Sea (based on three-dimensional modeling). Fundam. Prikl. Gidrofiz. 2016, 9, 41–51. [Google Scholar]
- Lien, V.; Schlichtholz, P.; Skagseth, O.; Vikebo, F. Wind-Driven Atlantic Water Flow as a Direct Mode for Reduced Barents Sea Ice Cover. J. Clim. 2017, 30, 803–812. [Google Scholar] [CrossRef]
- Onarheim, I.; Arthun, M. Toward an ice-free Barents Sea. Geophys. Res. Lett. 2017, 44, 8387–8395. [Google Scholar] [CrossRef]
- Kuznetsov, L.L.; Shoshina, E.V. Phytocenoses of the Barents Sea (Physiological and Structural Characteristics); SSC RAS Publ.: Rostov-on-Don, Russia, 2015. [Google Scholar]
- Raymont, J.E.G. Plankton and Productivity in the Oceans; Pergamon Press Ltd.: Oxford, UK, 1963; p. 660. Available online: https://archive.org/details/planktonproducti0000raym/page/n7/mode/2up (accessed on 26 November 2021).
- IPCC. IPCC Special Report on the Ocean and Cryosphere in a Changing Climate. 2019. Available online: https://www.ipcc.ch/srocc/ (accessed on 26 November 2021).
- Dalpadado, P.; Ingvaldsen, R.; Hassel, A. Zooplankton biomass variation in relation to climatic conditions in the Barents Sea. Polar Biol. 2003, 26, 233–241. [Google Scholar] [CrossRef]
- Wassmann, P.; Reigstad, M.; Haug, T.; Rudels, B.; Carroll, M.L.; Hop, H.; Gabrielsen, G.W.; Falk-Petersen, S.; Denisenko, S.G.; Arashkevich, E.; et al. Food webs and carbon flux in the Barents Sea. Prog. Oceanogr. 2006, 71, 232–287. [Google Scholar] [CrossRef]
- Dvoretsky, V.G. Zooplankton production in the Barents Sea in Summer. Izvestya TINRO 2012, 168, 169–183. [Google Scholar]
- Eiane, K.; Tande, K.S. Meso and macrozooplankton. In Ecosystem Barents Sea; Sakshaug, E., Johnsen, G., Kovacs, K.M., Eds.; Tapir Academic Press: Trondheim, Norway, 2009; pp. 209–234. [Google Scholar]
- Rey, F.; Loeng, H. The influence of ice and hydrographic conditions on the development of phytoplankton in the Barents Sea. In Marine Biology of Polar Regions and Effect of Stress on Marine Organisms; Gray, J.S., Christiansen, M.E., Eds.; Wiley&Sons Ltd.: Chichester, Great Britain, 1985; pp. 49–63. Available online: https://archive.org/details/marinebiologyofp0000euro/page/n7/mode/2up (accessed on 26 November 2021).
- Signorini, S.R.; McClain, C.R. Environmental factors controlling the Barents Sea spring-summer phytoplankton blooms. Geophys. Res. 2009, 36, 1–5. [Google Scholar] [CrossRef]
- Makarevich, P.R.; Druzhkova, E.I.; Larionov, V.V. Primary producers of the Barents Sea. In Diversity of Ecosystems; Mahamane, A., Ed.; InTech Open: Rijeka, Croatia, 2012; pp. 367–393. Available online: http://www.intechopen.com/books/diversity-of-ecosystems/primary-producers-of-the-barents-sea (accessed on 26 November 2021).
- Palmisano, A.C.; Garrison, D.L. Microorganisms in Antarctic sea ice. In Antarctic Microbiology; Friedman, E.I., Ed.; Wiley-Liss: New York, NY, USA, 1993; pp. 167–218. [Google Scholar]
- Poulin, M.; Daugbjerg, N.; Gradinger, R.; Ilyash, L.; Ratkova, T.; Quillfeldt, S. The pan-Arctic biodiversity of marine pelagic and sea-ice unicellular eukaryotes: A first-attempt assessment. Mar. Biodivers. 2011, 41, 13–28. [Google Scholar] [CrossRef]
- Fujiwara, A.; Hirawake, T.; Suzuki, K.; Imai, I.; Saitoh, S.I. Timing of sea ice retreat can alter phytoplankton community structure in the western Arctic Ocean. Biogeosciences 2014, 11, 1705–1716. [Google Scholar] [CrossRef] [Green Version]
- Park, J.; Kug, J.; Bader, J. Amplified Arctic warming by phytoplankton under greenhouse warming. Proc. Natl. Acad. Sci. USA 2015, 112, 5921–5926. [Google Scholar] [CrossRef] [Green Version]
- Ardyna, M.; Arrigo, K. Phytoplankton dynamics in a changing Arctic Ocean. Nat. Clim. Change 2020, 10, 892–903. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Makarevich, P.R.; Vodopianova, V.V.; Bulavina, A.S. Dynamics of the Spatial Chlorophyll-A Distribution at the Polar Front in the Marginal Ice Zone of the Barents Sea during Spring. Water 2022, 14, 101. https://doi.org/10.3390/w14010101
Makarevich PR, Vodopianova VV, Bulavina AS. Dynamics of the Spatial Chlorophyll-A Distribution at the Polar Front in the Marginal Ice Zone of the Barents Sea during Spring. Water. 2022; 14(1):101. https://doi.org/10.3390/w14010101
Chicago/Turabian StyleMakarevich, Pavel R., Veronika V. Vodopianova, and Aleksandra S. Bulavina. 2022. "Dynamics of the Spatial Chlorophyll-A Distribution at the Polar Front in the Marginal Ice Zone of the Barents Sea during Spring" Water 14, no. 1: 101. https://doi.org/10.3390/w14010101
APA StyleMakarevich, P. R., Vodopianova, V. V., & Bulavina, A. S. (2022). Dynamics of the Spatial Chlorophyll-A Distribution at the Polar Front in the Marginal Ice Zone of the Barents Sea during Spring. Water, 14(1), 101. https://doi.org/10.3390/w14010101








