Comparative Anaerobic Decolorization of Azo Dyes by Carbon-Based Membrane Bioreactor
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Ceramic Supported Carbon Membrane
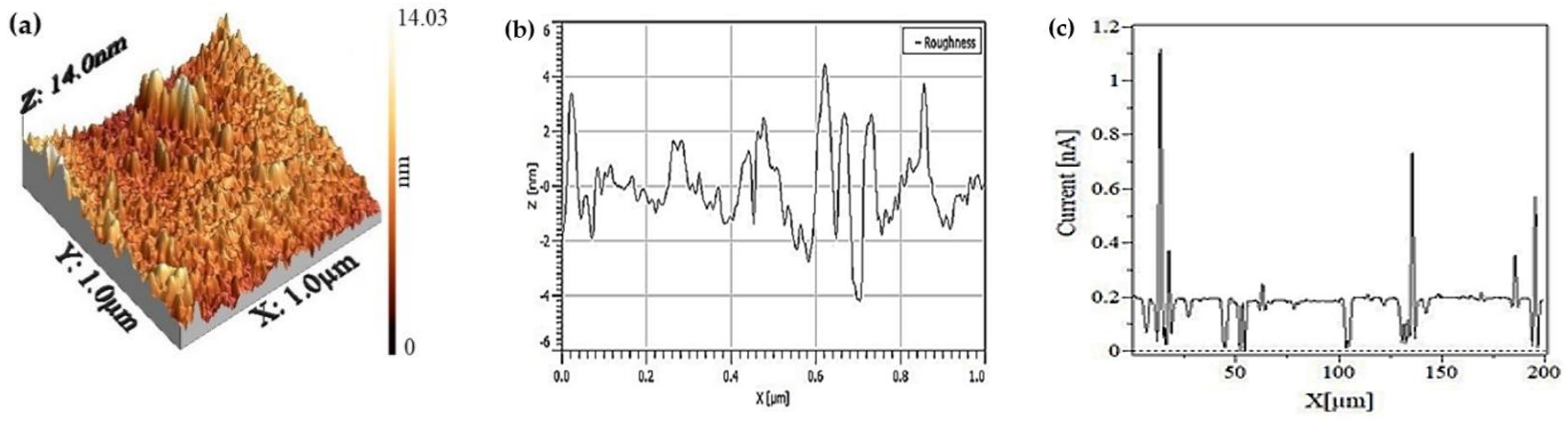
2.3. Membrane Characterization
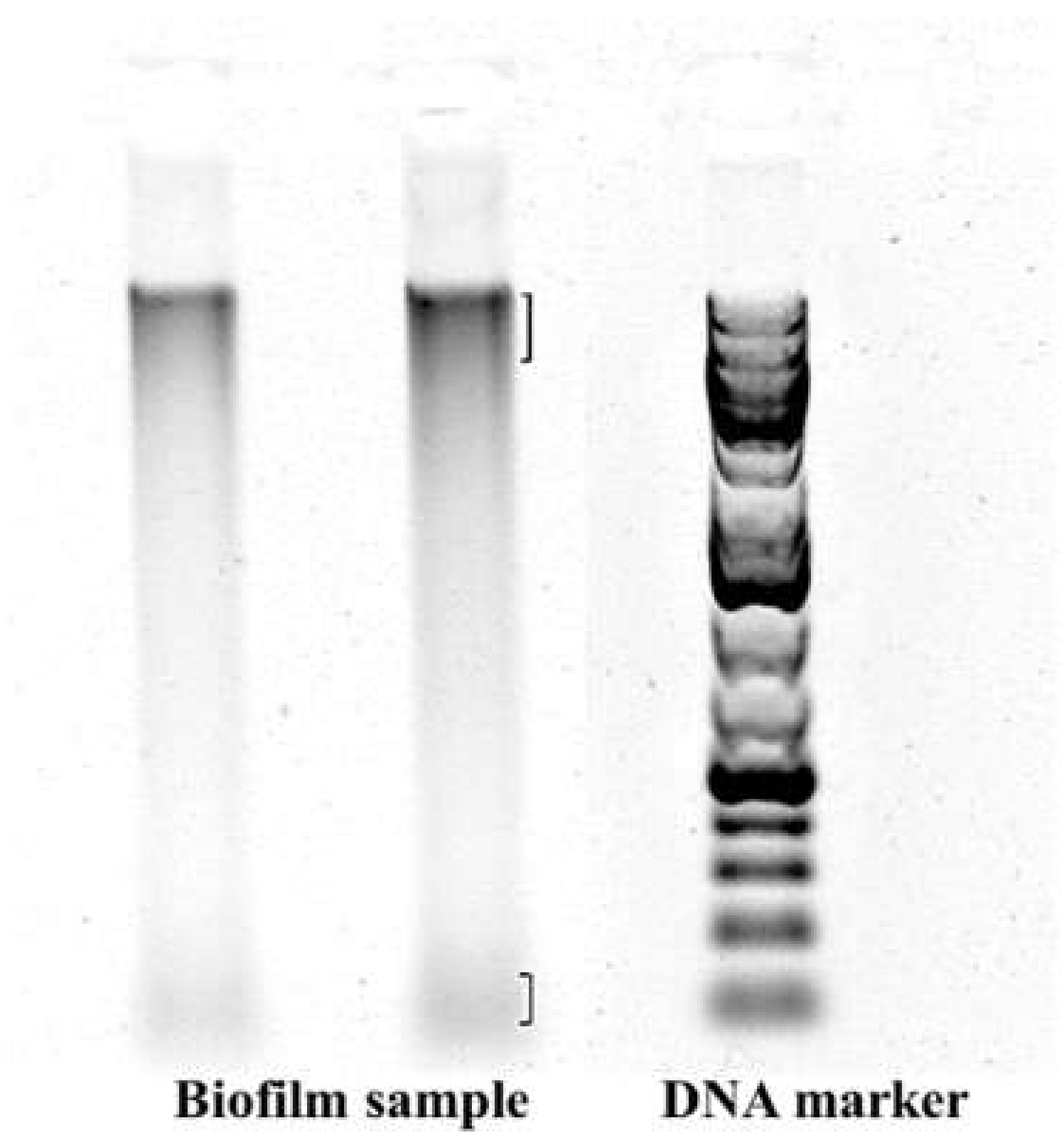
2.4. Microbial Analysis
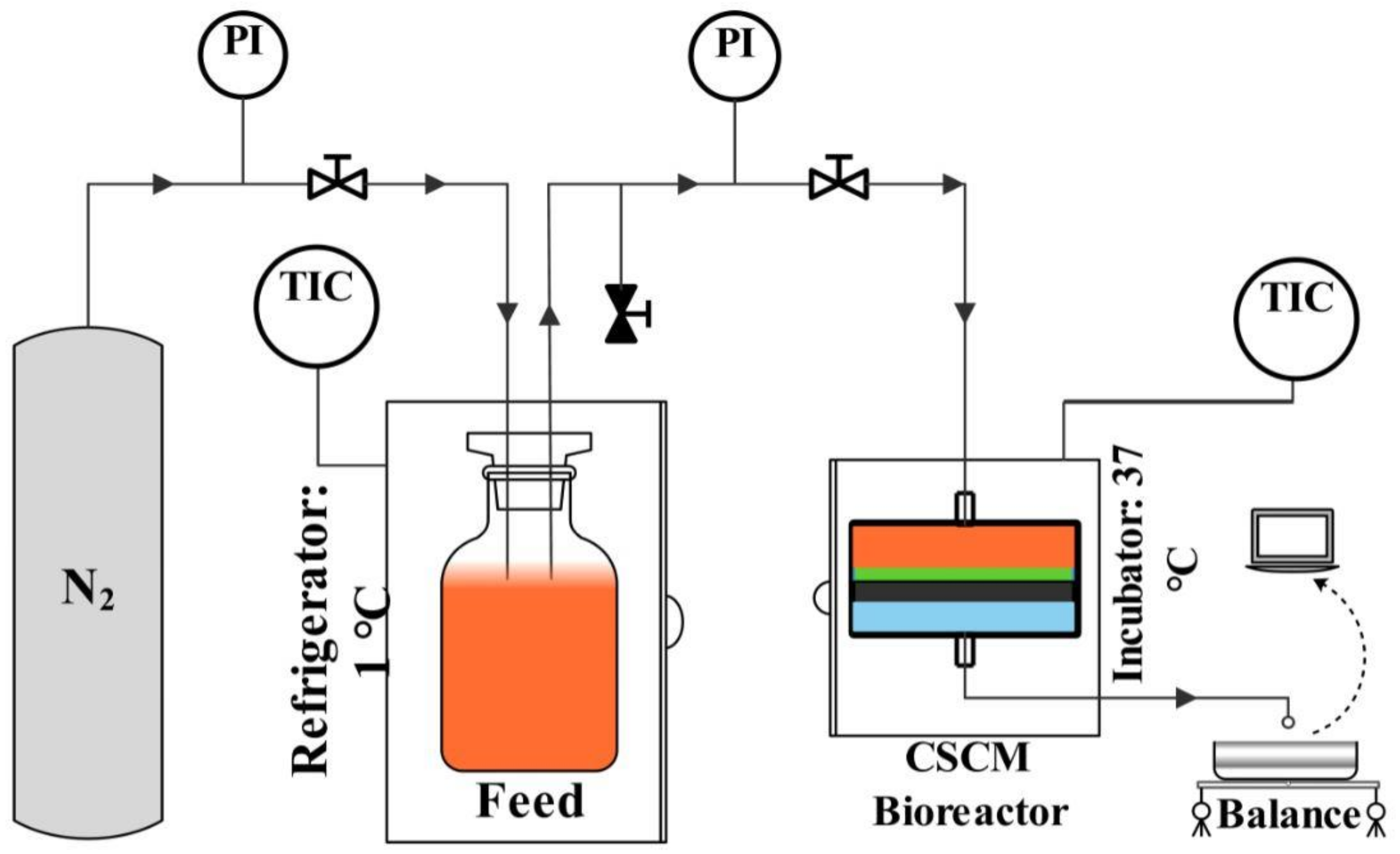
2.5. Experimental Set-Up for Anaerobic Biodegradation
2.6. Analytical Methods
3. Results and Discussions
3.1. Morphological Structure Analysis
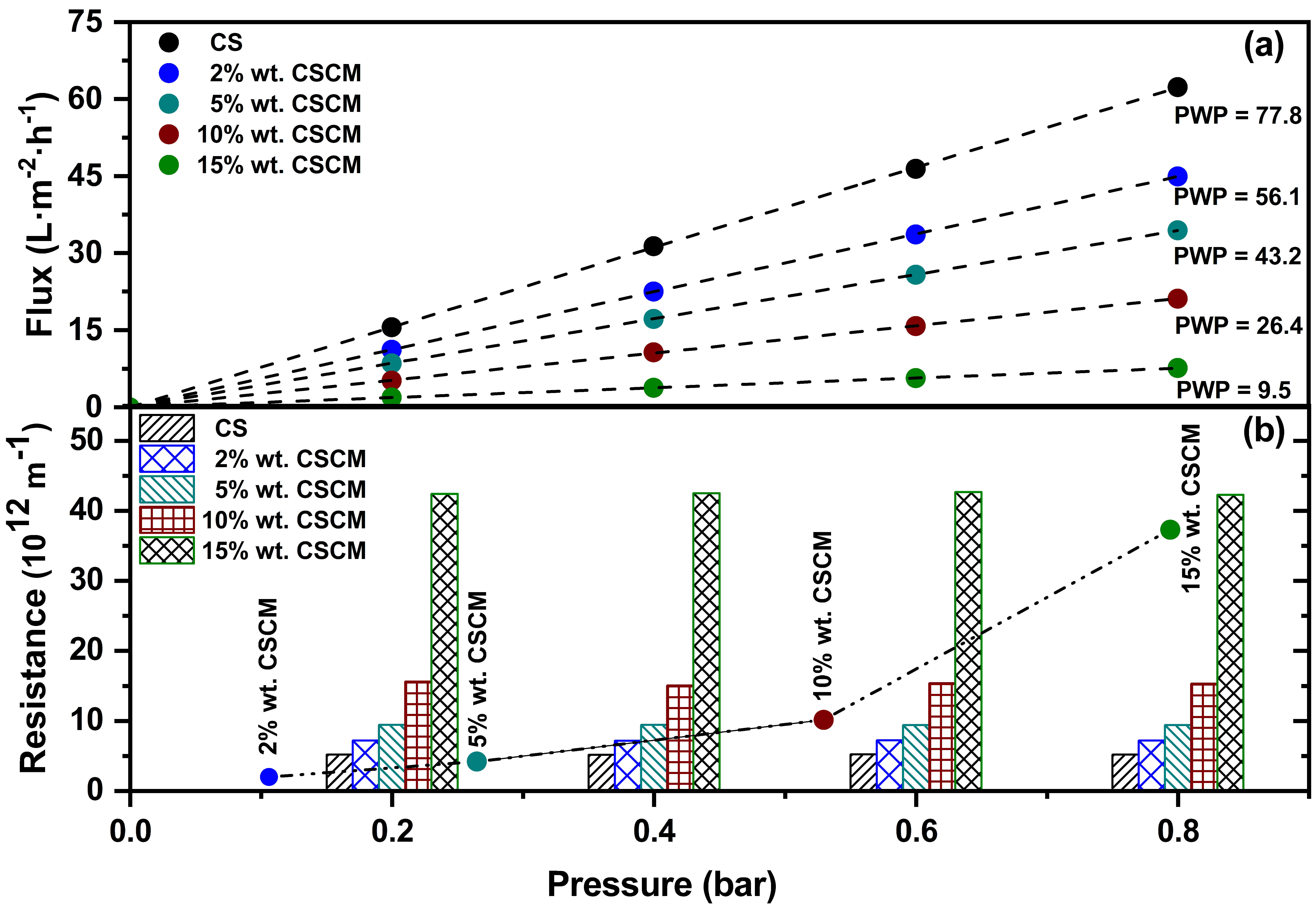
3.2. Impact of the Carbon Layer on Flux and Resistance
3.3. Role of the Carbon Layer on Anaerobic Biodegradation of Azo Dye
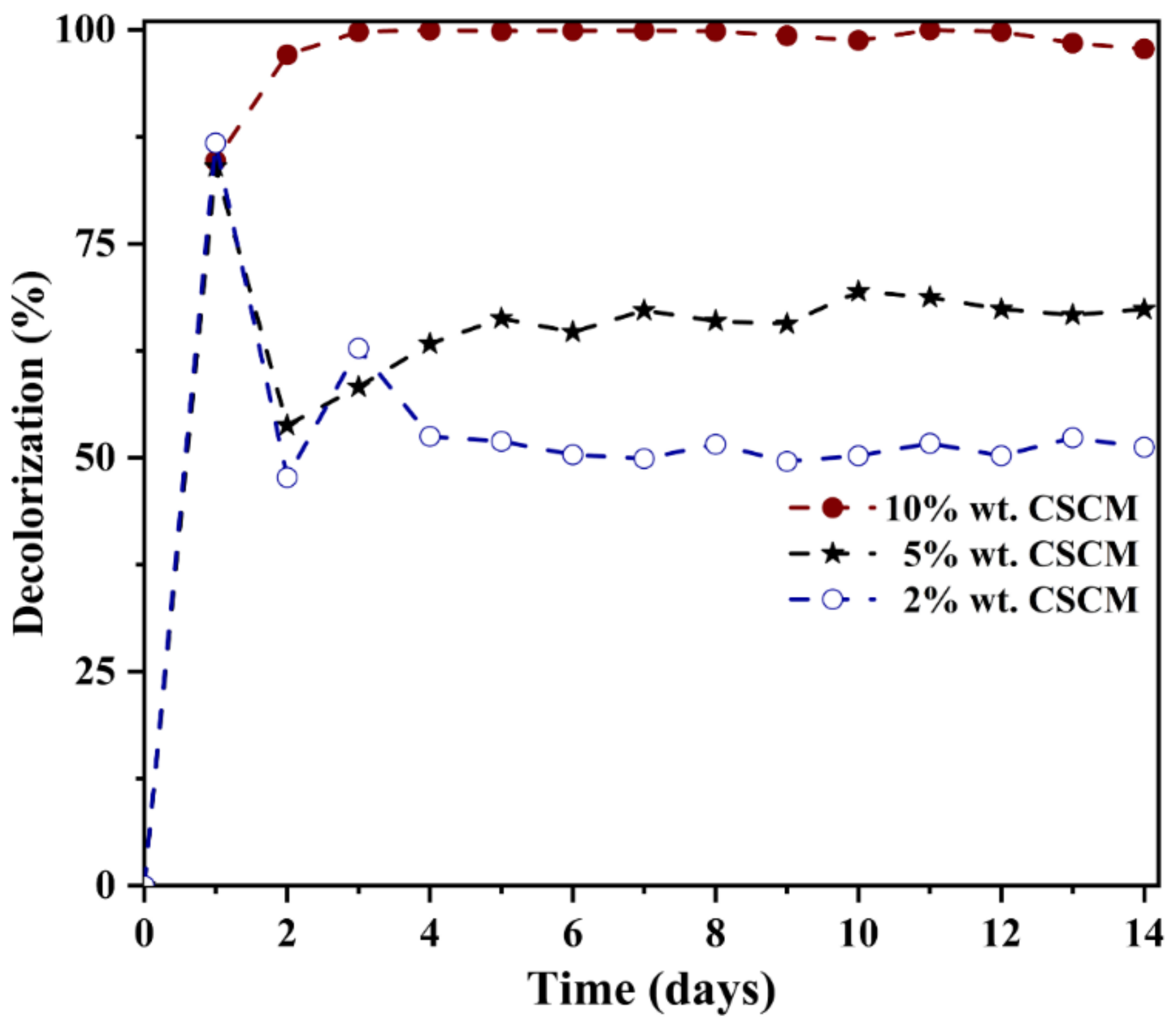
3.4. Effect of Precursor Concentration on Azo Dye Decolorization
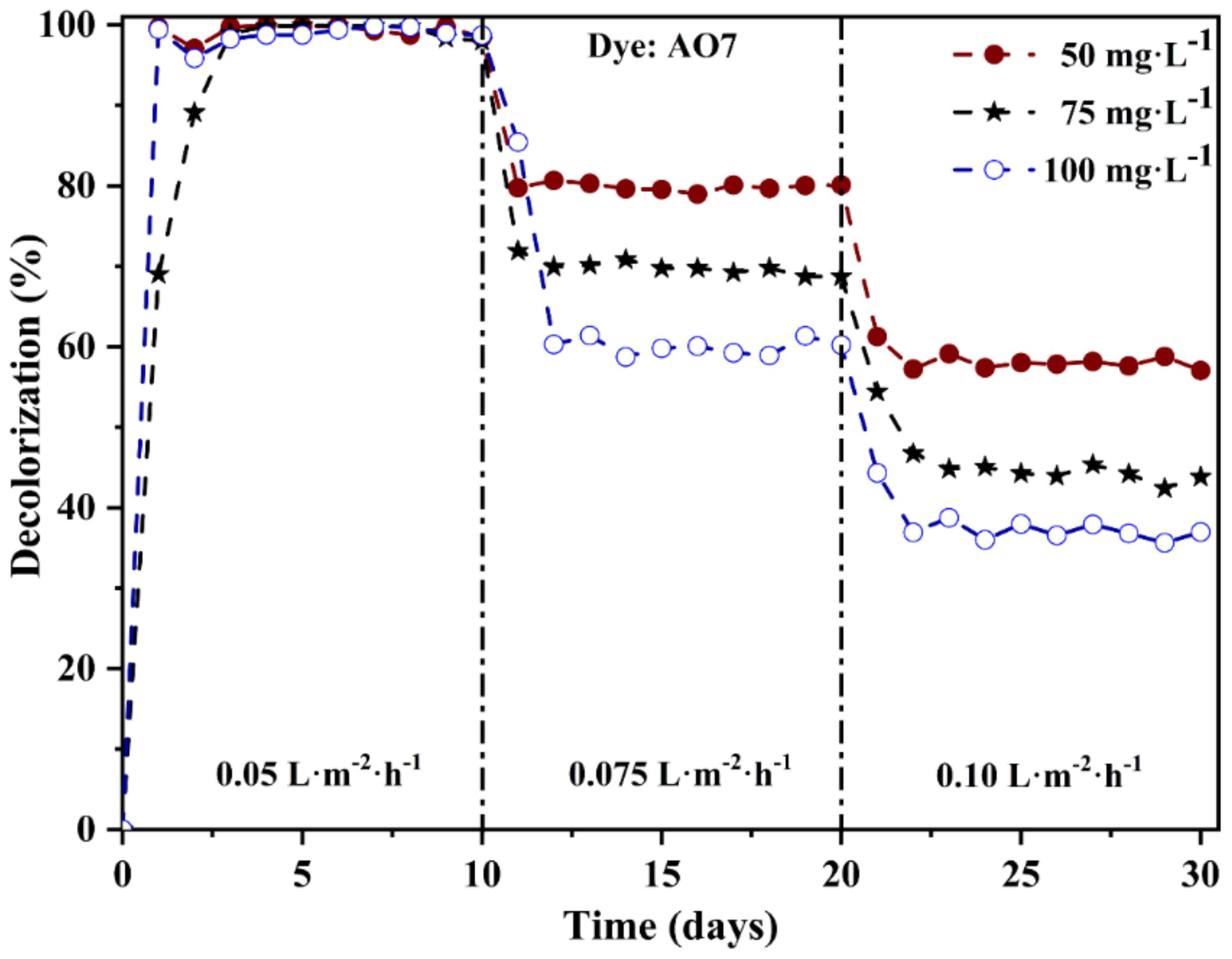
3.5. Effect of Flux and Feed Concentration on Azo Dye Decolorization
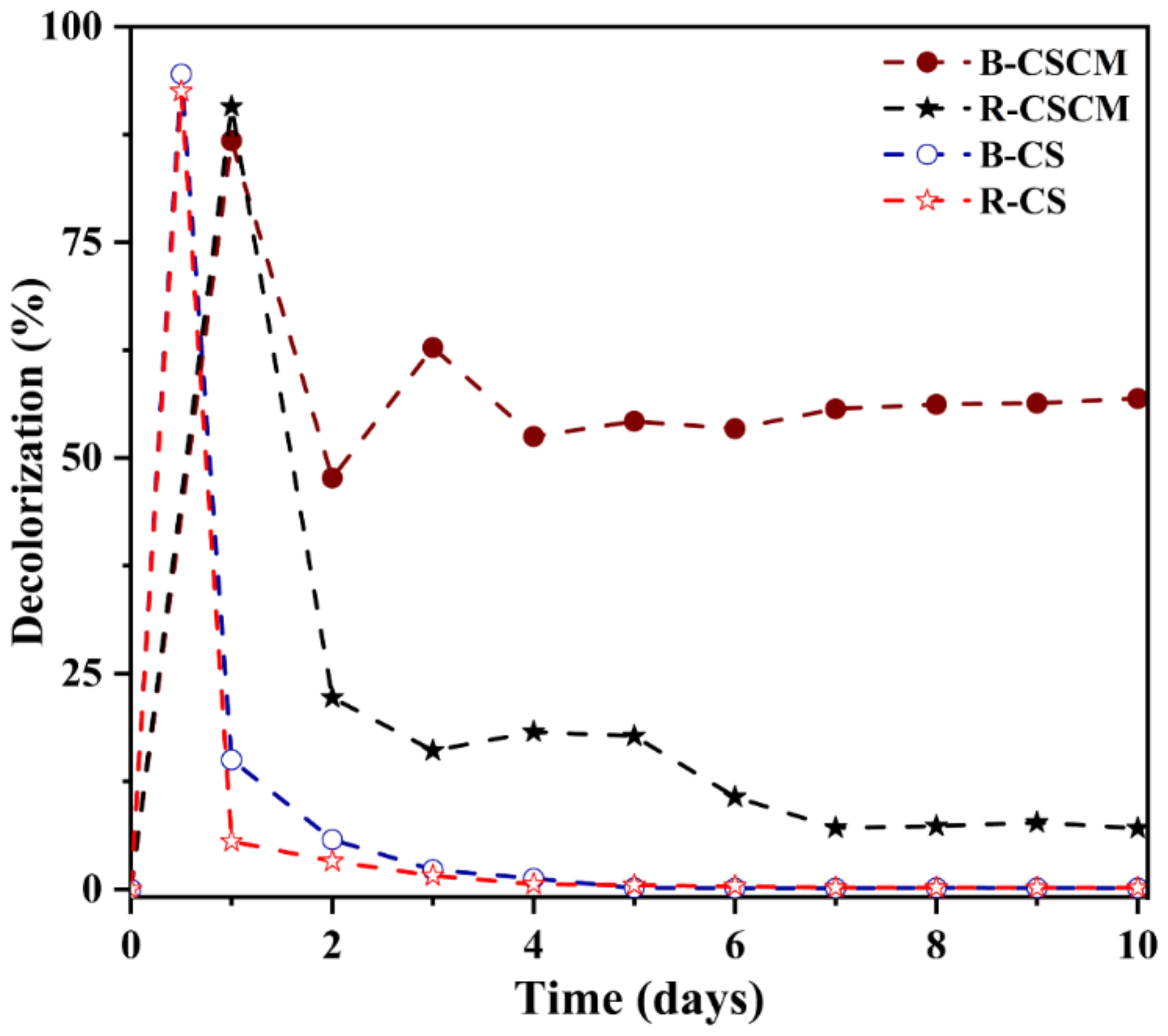
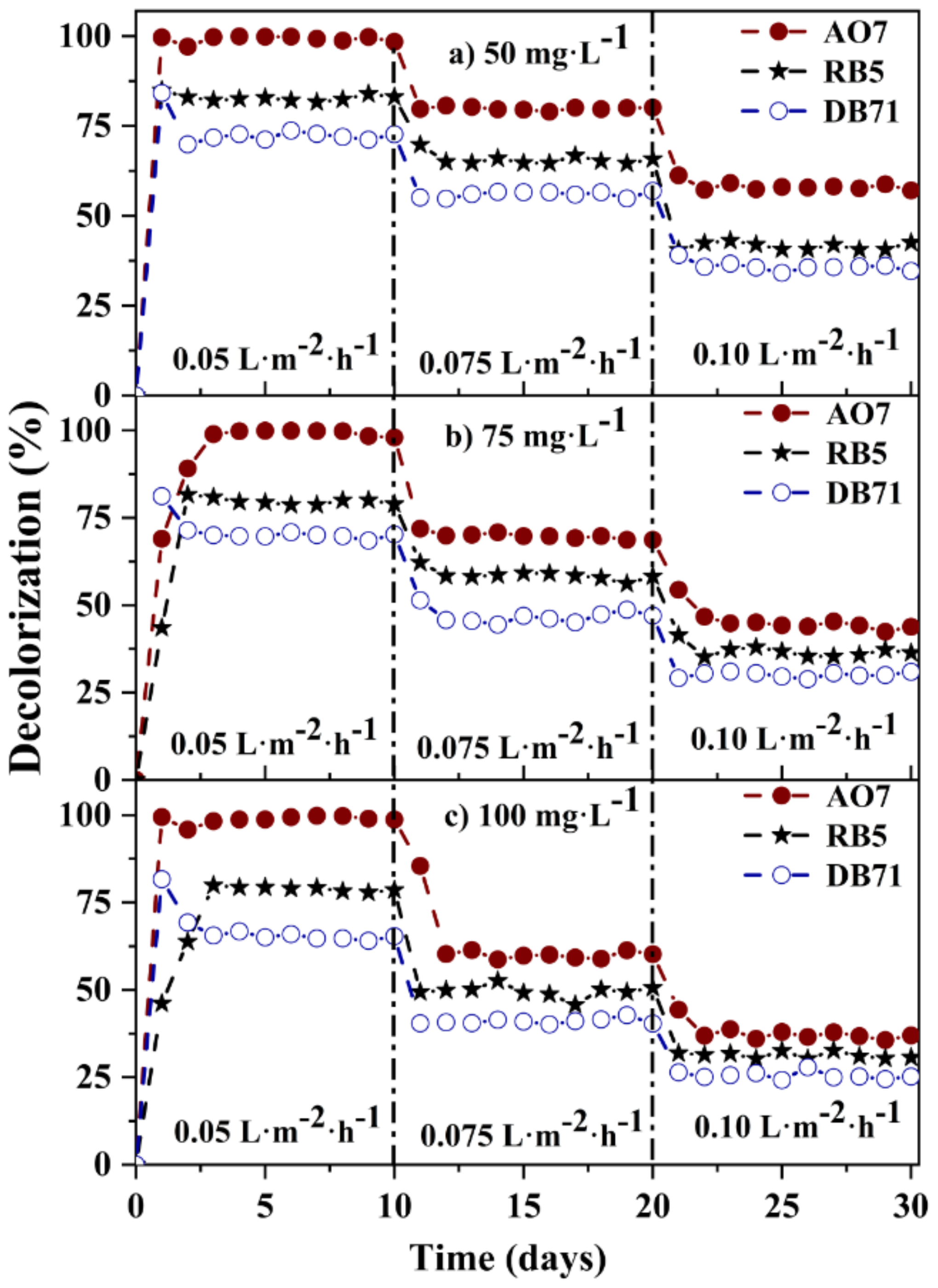
3.6. Comparative Decolorization of Azo Dyes
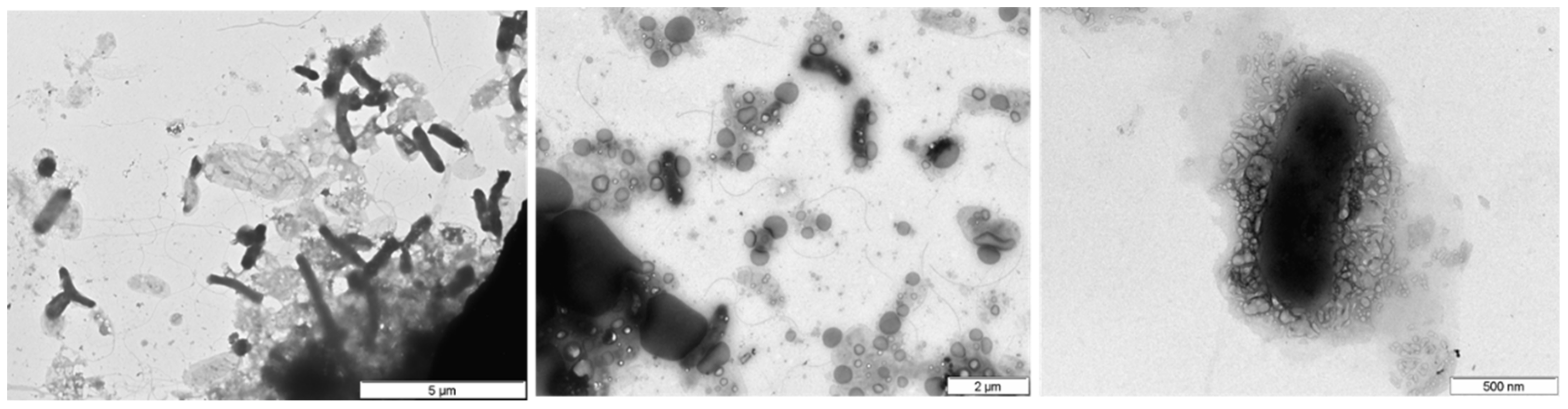
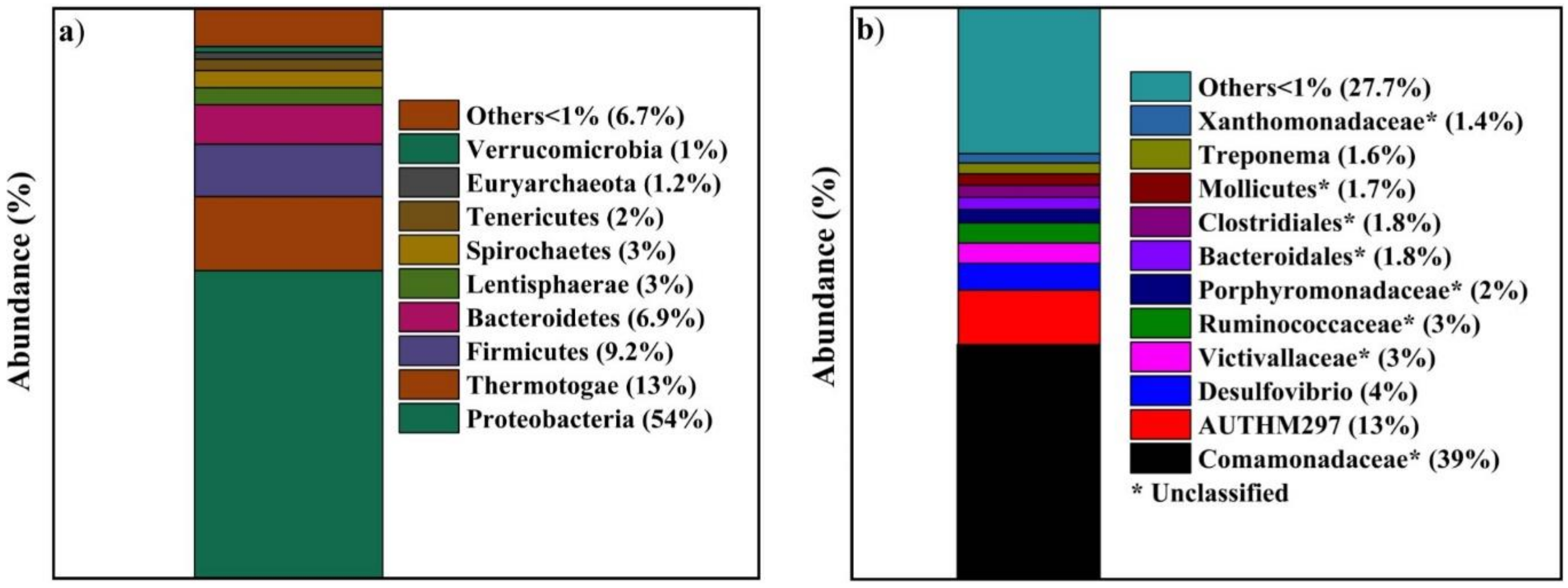
3.7. Microbial Community Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shanker, U.; Rani, M.; Jassal, V. Degradation of hazardous organic dyes in water by nanomaterials. Environ. Chem. Lett. 2017, 15, 623–642. [Google Scholar] [CrossRef]
- Puvaneswari, N.; Jayarama, M.; Gunasekaran, P. Toxicity assessment and microbial degradation of azo dyes. Indian J. Exp. Biol. 2006, 44, 618–626. [Google Scholar] [PubMed]
- Dos Santos, A.B.; Cervantes, F.J.; van Lier, J.B. Review paper on current technologies for decolourisation of textile wastewaters: Perspectives for anaerobic biotechnology. Bioresour. Technol. 2007, 98, 2369–2385. [Google Scholar] [CrossRef] [PubMed]
- Alves de Lima, R.O.; Bazo, A.P.; Salvadori, D.M.F.; Rech, C.M.; de Palma Oliveira, D.; de Aragão Umbuzeiro, G. Mutagenic and carcinogenic potential of a textile azo dye processing plant effluent that impacts a drinking water source. Mutat. Res. Genet. Toxicol. Environ. Mutagenesis 2007, 626, 53–60. [Google Scholar] [CrossRef]
- Lellis, B.; Fávaro-Polonio, C.Z.; Pamphile, J.A.; Polonio, J.C. Effects of textile dyes on health and the environment and bioremediation potential of living organisms. Biotechnol. Res. Innov. 2019, 3, 275–290. [Google Scholar] [CrossRef]
- Nawaz, M.S.; Gadelha, G.; Khan, S.J.; Hankins, N. Microbial toxicity effects of reverse transported draw solute in the forward osmosis membrane bioreactor (FO-MBR). J. Membr. Sci. 2013, 429, 323–329. [Google Scholar] [CrossRef]
- Poots, V.J.P.; McKay, G.; Healy, J.J. Removal of Basic Dye from Effluent Using Wood as an Adsorbent. J. Water Pollut. Control Fed. 1978, 50, 926–935. [Google Scholar]
- Rani, D.; Dahiya, R.P. COD and BOD removal from domestic wastewater generated in decentralised sectors. Bioresour. Technol. 2008, 99, 344–349. [Google Scholar] [CrossRef]
- Mezohegyi, G.; Kolodkin, A.; Castro, U.I.; Bengoa, C.; Stuber, F.; Font, J.; Fabregat, A.; Fortuny, A. Effective Anaerobic Decolorization of Azo Dye Acid Orange 7 in Continuous Upflow Packed-Bed Reactor Using Biological Activated Carbon System. Ind. Eng. Chem. Res. 2007, 46, 6788–6792. [Google Scholar] [CrossRef]
- Beydilli, M.I.; Pavlostathis, S.G.; Tincher, W.C. Biological Decolorization of the Azo Dye Reactive Red 2 under Various Oxidation-Reduction Conditions. Water Environ. Res. 2000, 72, 698–705. [Google Scholar] [CrossRef]
- Manu, B. Physico-chemical treatment of indigo dye wastewater. Coloration Technol. 2007, 123, 197–202. [Google Scholar] [CrossRef]
- Karcher, S.; Kornmüller, A.; Jekel, M. Anion exchange resins for removal of reactive dyes from textile wastewaters. Water Res. 2002, 36, 4717–4724. [Google Scholar] [CrossRef]
- Papić, S.; Koprivanac, N.; Božić, A.L. Removal of reactive dyes from wastewater using Fe(III) coagulant. Coloration Technol. 2000, 116, 352–358. [Google Scholar] [CrossRef]
- Uddin, M.J.; Islam, M.A.; Haque, S.A.; Hasan, S.; Amin, M.S.A.; Rahman, M.M. Preparation of nanostructured TiO2-based photocatalyst by controlling the calcining temperature and pH. Int. Nano Lett. 2012, 2, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Koyuncu, İ.; Afşar, H. Decomposition of dyes in the textile wastewater with ozone. J. Environ. Sci. Health Part A Environ. Sci. Eng. Toxicol. 1996, 31, 1035–1041. [Google Scholar] [CrossRef]
- Papić, S.; Koprivanac, N.; Božić, A.L.; Vujević, D.; Dragičević, S.K.; Kušić, H.; Peternel, I. Advanced Oxidation Processes in Azo Dye Wastewater Treatment. Water Environ. Res. 2006, 78, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Ennouri, R.; Lavecchia, R.; Zuorro, A.; Elaoud, S.C.; Petrucci, E. Degradation of chloramphenicol in water by oxidation on a boron-doped diamond electrode under UV irradiation. J. Water Process Eng. 2021, 41, 1–9. [Google Scholar] [CrossRef]
- Montanaro, D.; Lavecchia, R.; Petrucci, E.; Zuorro, A. UV-assisted electrochemical degradation of coumarin on boron-doped diamond electrodes. Chem. Eng. J. 2017, 323, 512–519. [Google Scholar] [CrossRef]
- Liakou, S.; Kornaros, M.; Lyberatos, G. Pretreatment of azo dyes using ozone. Water Sci. Technol. 1997, 36, 155–163. [Google Scholar] [CrossRef]
- Tzitzi, M.; Vayenas, D.V.; Lyberatos, G. Pretreatment of Textile Industry Wastewaters with Ozone. Water Sci. Technol. 1994, 29, 151–160. [Google Scholar] [CrossRef]
- Xu, Y.; Lebrun, R.E.; Gallo, P.-J.; Blond, P. Treatment of Textile Dye Plant Effluent by Nanofiltration Membrane. Sep. Sci. Technol. 1999, 34, 2501–2519. [Google Scholar] [CrossRef]
- Ghanbarlou, H.; Pedersen, N.L.; Simonsen, M.E.; Muff, J. Nitrogen-Doped Graphene Iron-Based Particle Electrode Outperforms Activated Carbon in Three-Dimensional Electrochemical Water Treatment Systems. Water 2020, 12, 3121. [Google Scholar] [CrossRef]
- Hansson, H.; Kaczala, F.; Marques, M.; Hogland, W. Photo-Fenton and Fenton Oxidation of Recalcitrant Industrial Wastewater Using Nanoscale Zero-Valent Iron. Int. J. Photoenergy 2012, 2012, 531076. [Google Scholar] [CrossRef] [Green Version]
- Khehra, M.S.; Saini, H.S.; Sharma, D.K.; Chadha, B.S.; Chimni, S.S. Decolorization of various azo dyes by bacterial consortium. Dye. Pigment. 2005, 67, 55–61. [Google Scholar] [CrossRef]
- Lau, Y.-Y.; Wong, Y.-S.; Teng, T.-T.; Morad, N.; Rafatullah, M.; Ong, S.-A. Coagulation-flocculation of azo dye Acid Orange 7 with green refined laterite soil. Chem. Eng. J. 2014, 246, 383–390. [Google Scholar] [CrossRef]
- Islam, M.A.; Amin, M.S.A.; Hoinkis, J. Optimal design of an activated sludge plant: Theoretical analysis. Appl. Water Sci. 2013, 3, 375–386. [Google Scholar] [CrossRef] [Green Version]
- Robinson, T.; McMullan, G.; Marchant, R.; Nigam, P. Remediation of dyes in textile effluent: A critical review on current treatment technologies with a proposed alternative. Bioresour. Technol. 2001, 77, 247–255. [Google Scholar] [CrossRef]
- Galán, J.; Rodríguez, A.; Gómez, J.M.; Allen, S.J.; Walker, G.M. Reactive dye adsorption onto a novel mesoporous carbon. Chem. Eng. J. 2013, 219, 62–68. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, L.; Zhang, G.; Wang, Z.; Xu, L.; Fan, Z. Influence of azo dye-TiO2 interactions on the filtration performance in a hybrid photocatalysis/ultrafiltration process. J. Colloid Interface Sci. 2013, 389, 273–283. [Google Scholar] [CrossRef]
- Wanyonyi, W.C.; Onyari, J.M.; Shiundu, P.M.; Mulaa, F.J. Effective biotransformation of Reactive Black 5 Dye Using Crude Protease from Bacillus Cereus Strain KM201428. Energy Procedia 2019, 157, 815–824. [Google Scholar] [CrossRef]
- Buckley, C.A. Membrane Technology for the Treatment of Dyehouse Effluents. Water Sci. Technol. 1992, 25, 203–209. [Google Scholar] [CrossRef]
- Żyłła, R.; Ledakowicz, S.; Boruta, T.; Olak-Kucharczyk, M.; Foszpańczyk, M.; Mrozińska, Z.; Balcerzak, J. Removal of Tetracycline Oxidation Products in the Nanofiltration Process. Water 2021, 13, 555. [Google Scholar] [CrossRef]
- Gupta, V.K.; Khamparia, S.; Tyagi, I.; Jaspal, D.; Malviya, A. Decolorization of Mixture of Dyes: A Critical Review. Glob. J. Environ. Sci. Manag. 2015, 1, 71–94. [Google Scholar]
- Mubashar, M.; Naveed, M.; Mustafa, A.; Ashraf, S.; Shehzad Baig, K.; Alamri, S.; Siddiqui, M.H.; Zabochnicka-Świątek, M.; Szota, M.; Kalaji, H.M. Experimental Investigation of Chlorella vulgaris and Enterobacter sp. MN17 for Decolorization and Removal of Heavy Metals from Textile Wastewater. Water 2020, 12, 3034. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, X.-Y.; Wang, S.; Tang, Q.-W.; Xie, T.; Lei, N.-Y.; Chen, Y.-L.; Qiao, W.-C.; Li, W.-W.; Lam, M.H.-W. Microbial community structure associated with treatment of azo dye in a start-up anaerobic sequenced batch reactor. J. Taiwan Inst. Chem. Eng. 2015, 54, 118–124. [Google Scholar] [CrossRef]
- Keck, A.; Klein, J.; Kudlich, M.; Stolz, A.; Knackmuss, H.J.; Mattes, R. Reduction of azo dyes by redox mediators originating in the naphthalenesulfonic acid degradation pathway of Sphingomonas sp. strain BN6. Appl. Environ. Microbiol. 1997, 63, 3684–3690. [Google Scholar] [CrossRef] [Green Version]
- Saratale, R.G.; Saratale, G.D.; Chang, J.S.; Govindwar, S.P. Bacterial decolorization and degradation of azo dyes: A review. J. Taiwan Inst. Chem. Eng. 2011, 42, 138–157. [Google Scholar] [CrossRef]
- Cervantes, F.; van der Zee, F.; Lettinga, G.; Field, J. Enhanced decolorisation of Acid Orange 7 in a continuous UASB reactor with quinones as redox mediators. Water Sci. Technol. A J. Int. Assoc. Water Pollut. Res. 2001, 44, 123–128. [Google Scholar] [CrossRef]
- Asif, M.B.; Ren, B.; Li, C.; Maqbool, T.; Zhang, X.; Zhang, Z. Powdered activated carbon—Membrane bioreactor (PAC-MBR): Impacts of high PAC concentration on micropollutant removal and microbial communities. Sci. Total Environ. 2020, 745, 1–9. [Google Scholar] [CrossRef]
- Mielgo, I.; Moreira, M.T.; Feijoo, G.; Lema, J.M. A packed-bed fungal bioreactor for the continuous decolourisation of azo-dyes (Orange II). J. Biotechnol. 2001, 89, 99–106. [Google Scholar] [CrossRef]
- Mezohegyi, G.; Bengoa, C.; Stuber, F.; Font, J.; Fabregat, A.; Fortuny, A. Novel bioreactor design for decolourisation of azo dye effluents. Chem. Eng. J. 2008, 143, 293–298. [Google Scholar] [CrossRef]
- Ong, S.-A.; Toorisaka, E.; Hirata, M.; Hano, T. Treatment of azo dye Orange II in aerobic and anaerobic-SBR systems. Process Biochem. 2005, 40, 2907–2914. [Google Scholar] [CrossRef]
- Brás, R.; Gomes, A.; Ferra, M.I.A.; Pinheiro, H.M.; Gonçalves, I.C. Monoazo and diazo dye decolourisation studies in a methanogenic UASB reactor. J. Biotechnol. 2005, 115, 57–66. [Google Scholar] [CrossRef] [Green Version]
- Spagni, A.; Casu, S.; Grilli, S. Decolourisation of textile wastewater in a submerged anaerobic membrane bioreactor. Bioresour. Technol. 2012, 117, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Waite, T.D.; Ong, P.E.A.; Schaefer, A. Assessment of Trace Estrogenic Contaminants Removal by Coagulant Addition, Powdered Activated Carbon Adsorption and Powdered Activated Carbon/Microfiltration Processes. J. Environ. Eng. 2004, 130, 736–742. [Google Scholar] [CrossRef] [Green Version]
- García-Martínez, Y.; Bengoa, C.; Stüber, F.; Fortuny, A.; Font, J.; Fabregat, A. Biodegradation of acid orange 7 in an anaerobic–aerobic sequential treatment system. Chem. Eng. Process. Process Intensif. 2015, 94, 99–104. [Google Scholar] [CrossRef]
- Sazali, N.; Salleh, W.N.W.; Nordin, N.A.H.M.; Ismail, A.F. Matrimid-based carbon tubular membrane: Effect of carbonization environment. J. Ind. Eng. Chem. 2015, 32, 167–171. [Google Scholar] [CrossRef]
- Fuertes, A.B.; Nevskaia, D.M.; Centeno, T.A. Carbon composite membranes from Matrimid® and Kapton® polyimides for gas separation. Microporous Mesoporous Mater. 1999, 33, 115–125. [Google Scholar] [CrossRef]
- Kiyono, M.; Williams, P.J.; Koros, W.J. Effect of pyrolysis atmosphere on separation performance of carbon molecular sieve membranes. J. Membr. Sci. 2010, 359, 2–10. [Google Scholar] [CrossRef]
- Horcas, I.; Fernández, R.; Gómez-Rodríguez, J.M.; Colchero, J.; Gómez-Herrero, J.; Baro, A.M. WSXM: A software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 2007, 78, 1–9. [Google Scholar] [CrossRef]
- NorgenBiotek. Soil DNA Isolation Kit. Available online: https://norgenbiotek.com/sites/default/files/resources/Soil-DNA-Isolation-Plus-Kit-Insert-PI64000-1.pdf (accessed on 25 February 2021).
- Lourenço, N.D.; Novais, J.M.; Pinheiro, H.M. Effect of some operational parameters on textile dye biodegradation in a sequential batch reactor. J. Biotechnol. 2001, 89, 163–174. [Google Scholar] [CrossRef]
- El Bouraie, M.; El Din, W.S. Biodegradation of Reactive Black 5 by Aeromonas hydrophila strain isolated from dye-contaminated textile wastewater. Sustain. Environ. Res. 2016, 26, 209–216. [Google Scholar] [CrossRef] [Green Version]
- Ismail, N.H.; Salleh, W.N.W.; Sazali, N.; Ismail, A.F. Effect of intermediate layer on gas separation performance of disk supported carbon membrane. Sep. Sci. Technol. 2017, 52, 2137–2149. [Google Scholar] [CrossRef]
- Barsema, J.N.; Klijnstra, S.D.; Balster, J.H.; van der Vegt, N.F.A.; Koops, G.H.; Wessling, M. Intermediate polymer to carbon gas separation membranes based on Matrimid PI. J. Membr. Sci. 2004, 238, 93–102. [Google Scholar] [CrossRef]
- Boussu, K.; Van der Bruggen, B.; Volodin, A.; Snauwaert, J.; Van Haesendonck, C.; Vandecasteele, C. Roughness and hydrophobicity studies of nanofiltration membranes using different modes of AFM. J. Colloid Interface Sci. 2005, 286, 632–638. [Google Scholar] [CrossRef]
- Shimp, R.J.; Pfaender, F.K. Effects of surface area and flow rate on marine bacterial growth in activated carbon columns. Appl. Environ. Microbiol. 1982, 44, 471–477. [Google Scholar] [CrossRef] [Green Version]
- Maximous, N.; Nakhla, G.; Wan, W.; Wong, K. Preparation, characterization and performance of Al2O3/PES membrane for wastewater filtration. J. Membr. Sci. 2009, 341, 67–75. [Google Scholar] [CrossRef]
- Abid, M.F.; Zablouk, M.A.; Abid-Alameer, A.M. Experimental study of dye removal from industrial wastewater by membrane technologies of reverse osmosis and nanofiltration. Iran. J. Environ. Health Sci. Eng. 2012, 9, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Méndez-Paz, D.; Omil, F.; Lema, J.M. Anaerobic treatment of azo dye Acid Orange 7 under batch conditions. Enzym. Microb. Technol. 2005, 36, 264–272. [Google Scholar] [CrossRef]
- Mezohegyi, G.; van der Zee, F.P.; Font, J.; Fortuny, A.; Fabregat, A. Towards advanced aqueous dye removal processes: A short review on the versatile role of activated carbon. J. Environ. Manag. 2012, 102, 148–164. [Google Scholar] [CrossRef] [PubMed]
- Manu, B.; Chaudhari, S. Anaerobic decolorisation of simulated textile wastewater containing azo dyes. Bioresour. Technol. 2002, 82, 225–231. [Google Scholar] [CrossRef]
- Brás, R.; Isabel, A.; Ferra, M.; Pinheiro, H.M.; Gonçalves, I.C. Batch tests for assessing decolourisation of azo dyes by methanogenic and mixed cultures. J. Biotechnol. 2001, 89, 155–162. [Google Scholar] [CrossRef]
- Evangelista-Barreto, N.S.; Albuquerque, C.D.; Vieira, R.H.S.F.; Campos-Takaki, G.M. Cometabolic Decolorization of the Reactive Azo Dye Orange II by Geobacillus stearothermophilus UCP 986. Text. Res. J. 2009, 79, 1266–1273. [Google Scholar] [CrossRef]
- Bragger, J.L.; Lloyd, A.W.; Soozandehfar, S.H.; Bloomfield, S.F.; Marriott, C.; Martin, G.P. Investigations into the azo reducing activity of a common colonic microorganism. Int. J. Pharm. 1997, 157, 61–71. [Google Scholar] [CrossRef]
- Zuorro, A.; Lavecchia, R.; Monaco, M.M.; Iervolino, G.; Vaiano, V. Photocatalytic Degradation of Azo Dye Reactive Violet 5 on Fe-Doped Titania Catalysts under Visible Light Irradiation. Catalysts 2019, 9, 645. [Google Scholar] [CrossRef] [Green Version]
- Tan, X.; Rodrigue, D. A Review on Porous Polymeric Membrane Preparation. Part II: Production Techniques with Polyethylene, Polydimethylsiloxane, Polypropylene, Polyimide, and Polytetrafluoroethylene. Polymers 2019, 11, 1310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahimi, M.; Aghel, B.; Sadeghi, M.; Ahmadi, M. Using Y-shaped microreactor for continuous decolorization of an Azo dye. Desalination Water Treat. 2014, 52, 5513–5519. [Google Scholar] [CrossRef]
- Meitiniarti, I.; Soetarto, E.S.; Sugiharto, E.; Timotius, K. Optimum concentration of glucose and Orange II for growth and decolorization of Orange II by Enterococcus faecalis ID6017 under static culture. Microb. Indones. 2008, 2, 73–78. [Google Scholar]
- Ding, J.; Zhang, Y.; Quan, X.; Chen, S. Anaerobic biodecolorization of AO7 by a newly isolated Fe(III)-reducing bacterium Sphingomonas strain DJ. J. Chem. Technol. Biotechnol. 2015, 90, 158–165. [Google Scholar] [CrossRef]
- Chen, B.-Y.; Chen, S.-Y.; Chang, J.-S. Immobilized cell fixed-bed bioreactor for wastewater decolorization. Process Biochem. 2005, 40, 3434–3440. [Google Scholar] [CrossRef]
- Popli, S.; Patel, U.D. Destruction of azo dyes by anaerobic–aerobic sequential biological treatment: A review. Int. J. Environ. Sci. Technol. 2015, 12, 405–420. [Google Scholar] [CrossRef] [Green Version]
- Mutambanengwe, C.C.Z.; Togo, C.A.; Whiteley, C.G. Decolorization and Degradation of Textile Dyes with Biosulfidogenic Hydrogenases. Biotechnol. Prog. 2007, 23, 1095–1100. [Google Scholar] [CrossRef]
- Kiriakidou, F.; Kondarides, D.I.; Verykios, X.E. The effect of operational parameters and TiO2-doping on the photocatalytic degradation of azo-dyes. Catal. Today 1999, 54, 119–130. [Google Scholar] [CrossRef]
- Chen, B.-Y. Understanding decolorization characteristics of reactive azo dyes by Pseudomonas luteola: Toxicity and kinetics. Process Biochem. 2002, 38, 437–446. [Google Scholar] [CrossRef]
- Khan, R.; Bhawana, P.; Fulekar, M.H. Microbial decolorization and degradation of synthetic dyes: A review. Rev. Environ. Sci. Bio Technol. 2013, 12, 75–97. [Google Scholar] [CrossRef]
- Solís, M.; Solís, A.; Pérez, H.I.; Manjarrez, N.; Flores, M. Microbial decolouration of azo dyes: A review. Process Biochem. 2012, 47, 1723–1748. [Google Scholar] [CrossRef]
- Chen, K.-C.; Wu, J.-Y.; Liou, D.-J.; Hwang, S.-C.J. Decolorization of the textile dyes by newly isolated bacterial strains. J. Biotechnol. 2003, 101, 57–68. [Google Scholar] [CrossRef]
- Franciscon, E.; Zille, A.; Fantinatti-Garboggini, F.; Silva, I.S.; Cavaco-Paulo, A.; Durrant, L.R. Microaerophilic–aerobic sequential decolourization/biodegradation of textile azo dyes by a facultative Klebsiella sp. strain VN-31. Process Biochem. 2009, 44, 446–452. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Segura, S.; Centellas, F.; Arias, C.; Garrido, J.A.; Rodríguez, R.M.; Cabot, P.L.; Brillas, E. Comparative decolorization of monoazo, diazo and triazo dyes by electro-Fenton process. Electrochim. Acta 2011, 58, 303–311. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, L.; Feng, L.; Chen, G.; Wang, Y.; Zhai, Z.; Zhang, Q. Exploration of the key functional strains from an azo dye degradation microbial community by DGGE and high-throughput sequencing technology. Environ. Sci. Pollut. Res. 2019, 26, 24658–24671. [Google Scholar] [CrossRef]
- Köchling, T.; Ferraz, A.D.N.; Florencio, L.; Kato, M.T.; Gavazza, S. 454-Pyrosequencing analysis of highly adapted azo dye-degrading microbial communities in a two-stage anaerobic–aerobic bioreactor treating textile effluent. Environ. Technol. 2017, 38, 687–693. [Google Scholar] [CrossRef]
- Cui, D.; Zhang, H.; He, R.; Zhao, M. The Comparative Study on the Rapid Decolorization of Azo, Anthraquinone and Triphenylmethane Dyes by Anaerobic Sludge. Int. J. Environ. Res. Public Health 2016, 13, 1053. [Google Scholar] [CrossRef] [PubMed]
- Balapure, K.H.; Jain, K.; Chattaraj, S.; Bhatt, N.S.; Madamwar, D. Co-metabolic degradation of diazo dye—Reactive blue 160 by enriched mixed cultures BDN. J. Hazard. Mater. 2014, 279, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Sun, Y.; Guo, D.; Wu, Z.; Jiang, D. Molecular diversity of bacterial community of dye wastewater in an anaerobic sequencing batch reactor. Afr. J. Microbiol. Res. 2012, 6, 6444–6453. [Google Scholar] [CrossRef]
- Yoo, E.; Libra, J.; Adrian, L. Mechanism of Dye Reduction of Azo Dyes in Anaerobic Mixed Culture. J. Environ. Eng. 2001, 127, 844–849. [Google Scholar] [CrossRef]
- Fernando, E.; Keshavarz, T.; Kyazze, G. Simultaneous co-metabolic decolourisation of azo dye mixtures and bio-electricity generation under thermophillic (50 °C) and saline conditions by an adapted anaerobic mixed culture in microbial fuel cells. Bioresour. Technol. 2013, 127, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Prigione, V.; Grosso, I.; Tigini, V.; Anastasi, A.; Varese, G.C. Fungal Waste-Biomasses as Potential Low-Cost Biosorbents for Decolorization of Textile Wastewaters. Water 2012, 4, 770–784. [Google Scholar] [CrossRef] [Green Version]

| Azo Dyes | Structure | Type | Solubility in Water |
|---|---|---|---|
| Acid Orange 7 (C16H11N2NaO4S) MW: 350.3 g/L λ max: 484 nm | 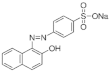 | Monoazo CI: 15510 | 116 g·L−1 (35 °C) |
| Reactive Black 5 (C26H21N5Na4O19S6) MW: 991.8 g/L λ max: 587 nm | 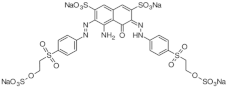 | Diazo CI: 20505 | 100 g·L−1 (30 °C) |
| Direct Blue 71 (C40H23N7Na4O13S4) MW: 1029.9 g/L λ max: 585 nm | 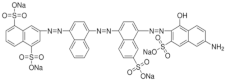 | Triazo CI: 34140 | 10 g·L−1 (60 °C) |
| C | O | Ti | Zr | |
|---|---|---|---|---|
| CS | n.d. | 44.0 | 52.2 | 3.8 |
| CSCM (2% wt. of Matrimid) | 9.8 | 25.9 | 2.6 | 61.7 |
| CSCM (5% wt. of Matrimid) | 34.5 | 19.1 | 1.2 | 45.2 |
| CSCM (10% wt. of Matrimid) | 65.6 | 3.1 | 0.6 | 30.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amin, M.S.A.; Stüber, F.; Giralt, J.; Fortuny, A.; Fabregat, A.; Font, J. Comparative Anaerobic Decolorization of Azo Dyes by Carbon-Based Membrane Bioreactor. Water 2021, 13, 1060. https://doi.org/10.3390/w13081060
Amin MSA, Stüber F, Giralt J, Fortuny A, Fabregat A, Font J. Comparative Anaerobic Decolorization of Azo Dyes by Carbon-Based Membrane Bioreactor. Water. 2021; 13(8):1060. https://doi.org/10.3390/w13081060
Chicago/Turabian StyleAmin, Mohammad Shaiful Alam, Frank Stüber, Jaume Giralt, Agustin Fortuny, Azael Fabregat, and José Font. 2021. "Comparative Anaerobic Decolorization of Azo Dyes by Carbon-Based Membrane Bioreactor" Water 13, no. 8: 1060. https://doi.org/10.3390/w13081060







