The optimal conditions leading to the selective extraction of VOCs from contaminated water with the best total flux and selectivity were determined by carrying out the pervaporation technique in a static process. Various parameters influencing the pervaporation performance were investigated, such as the membrane thickness, stirring speed, and temperature. The optimal conditions obtained were then applied to examine in the continuous mode the extraction of these contaminants from a reservoir of a larger capacity to minimize depletion and approach the situation of actual contamination.
3.2.1. Pervaporation Using the Static Process
- (A)
Effect of the Membrane Thickness on the Extraction Performance
Regarding the diffusion of a solution through a dense and non-porous material, according to Fick’s model, the total flux was inversely proportional to the membrane thickness [
59]. Hasanoglu et al. [
60] examined the extraction of ethyl acetate from its esterification media using PDMS as a membrane with different thicknesses. The total flux obtained using PDMS-250 µm was higher than that obtained using PDMS-300 µm. Hyder et al. [
61] investigated the thickness of a selective membrane fabricated from poly (vinyl alcohol)/polysulfone cross-linked in the dehydration of alcohol. They reported that the total flux increased by 193% with a decrease in thickness from 52 to 4 μm. Similarly, Raisi et al. [
62] reported the effect of the membrane thickness on the permeation rate (flux) using two types of membrane, PDMS and poly (octylmethylsiloxane) (POMS), and a mixture of pomegranate aroma as the feed for separation. They concluded that the pervaporation flux was inversely proportional to the membrane thickness and explained this simply by a decrease in membrane resistance toward the transit of the molecules with decreasing thickness. By contrast, according to the same authors, the selectivity increased with increasing membrane thickness.
The present study examined effects of the SILICONE1200 membrane thickness on the extraction performance at a constant temperature and a stirring rate of 20 °C and 500 rpm, respectively.
Figure 4 and
Figure 5 illustrate the variations of the total flux and the selectivity, respectively. For all thicknesses, the total flux decreased continually with time and more significantly when the membrane thickness was 260 and 350 μm, respectively (
Figure 4). The thinner membranes had the highest total fluxes; 11.03 and 7.62 kg·m
−2·h
−1 were obtained after 1.5 h of separation using 260 and 350 μm membranes, respectively. This is expected in the separation of organic compounds using a membrane, agrees with Hasanoglu et al. [
60] and Hyder et al. [
62], and is mainly due to the residence time (transit duration) of molecules through the membrane. A thinner membrane has a shorter residence time, and vice versa, promoting the passage of a larger number of molecules. The continued decrease in total flux with time was due to the depletion of organic compounds in the feed.
At first sight, the total flux decreased with time and membrane thickness, achieving a minimum of 1.95 kg·m
−2·h
−1 and 11.03 kg·m
−2·h
−1 with the thickest (510 μm) and thinnest (260 μm) membranes, respectively, over the 1.5 h duration (
Figure 4). As the thickness of the membrane was increased, the total flux became increasingly stable with time. This is because the flux is relatively high when a thinner membrane is used, leading to rapid depletion of the organic mixture, in which the heavy phase in contact with the membrane contains mainly chloroform (
Scheme 3). Therefore, the flux also decreases rapidly with time of the separation process. By contrast, a thicker membrane results in a lower total flux and slower depletion of the feed in the organic components. This leads to pseudo-stability of the concentration of organic compounds in the feed at its minimum. The higher total flux observed immediately after starting the separation process was mainly due to the large amount of organic solvents absorbed by the membrane before the pervaporation process was run (−20 min).
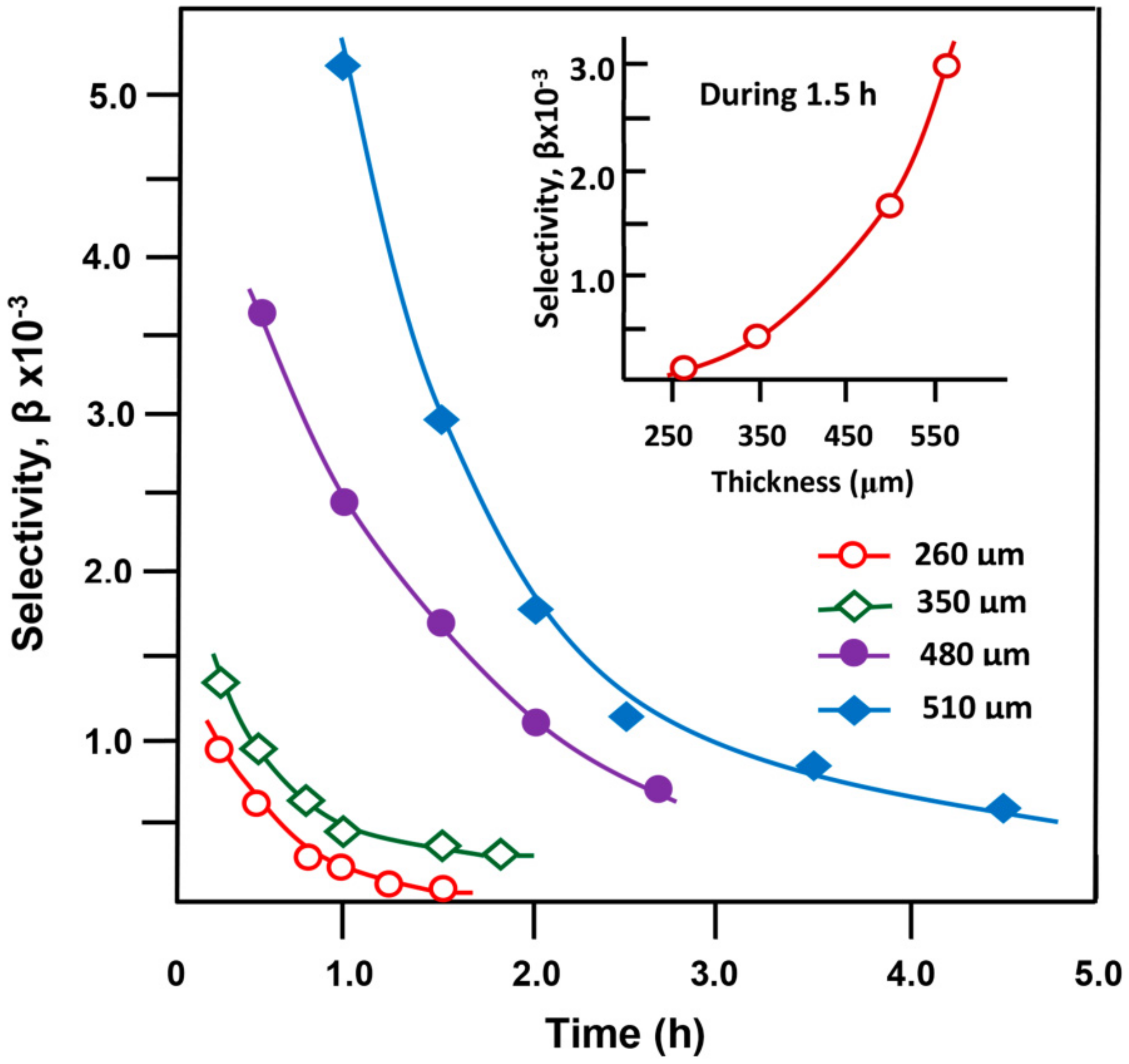
Figure 5 reveal for all membranes used a decrease in the selectivity versus time which is more marked when the thickness was 480 and 510 μm. The drop in selectivity is mainly due to the depletion of the organic compounds in the feed. This was confirmed by observation at the end of the separation process, because after 2 h there were practically no organic components were found in the feed when the membrane thickness was 260 μm and 6 h with the thickest membrane (510 μm). At the start of the separation, the heavy phase rich in chloroform (density = 1.48 g·cm
−3), which is immediately in contact with the membrane, favored by its relatively high affinity and relatively lower stirring rate (260 rpm), mainly passes into the permeate then comes thiophene (density = 1.05 g·cm
−3) and finally the rest of the organic compounds.
Regarding the variation of the selectivity with the membrane thickness (inset in
Figure 5), the curve profile indicates a significant increase in which a maximum selectivity of 2988 is reached for the 510-μm-thick membrane during a period of 1.5 h of the separation process. The increase of the selectivity with the increase of the membrane thickness agrees with comparable results reported in the literature [
62,
63,
64,
65].
With membrane thicknesses of 480 μm or more, the transit path of chloroform and other organic components through the membrane is long, this reduces the rate of exhaustion and therefore the separation period is slower. Hence, the selectivity of the membrane is governed only by molecules in the membrane and not by other factors, such as the rapid depletion of one component of the feed. In this case, the selectivity depends only on the membrane thickness, as it increased with increasing thickness.
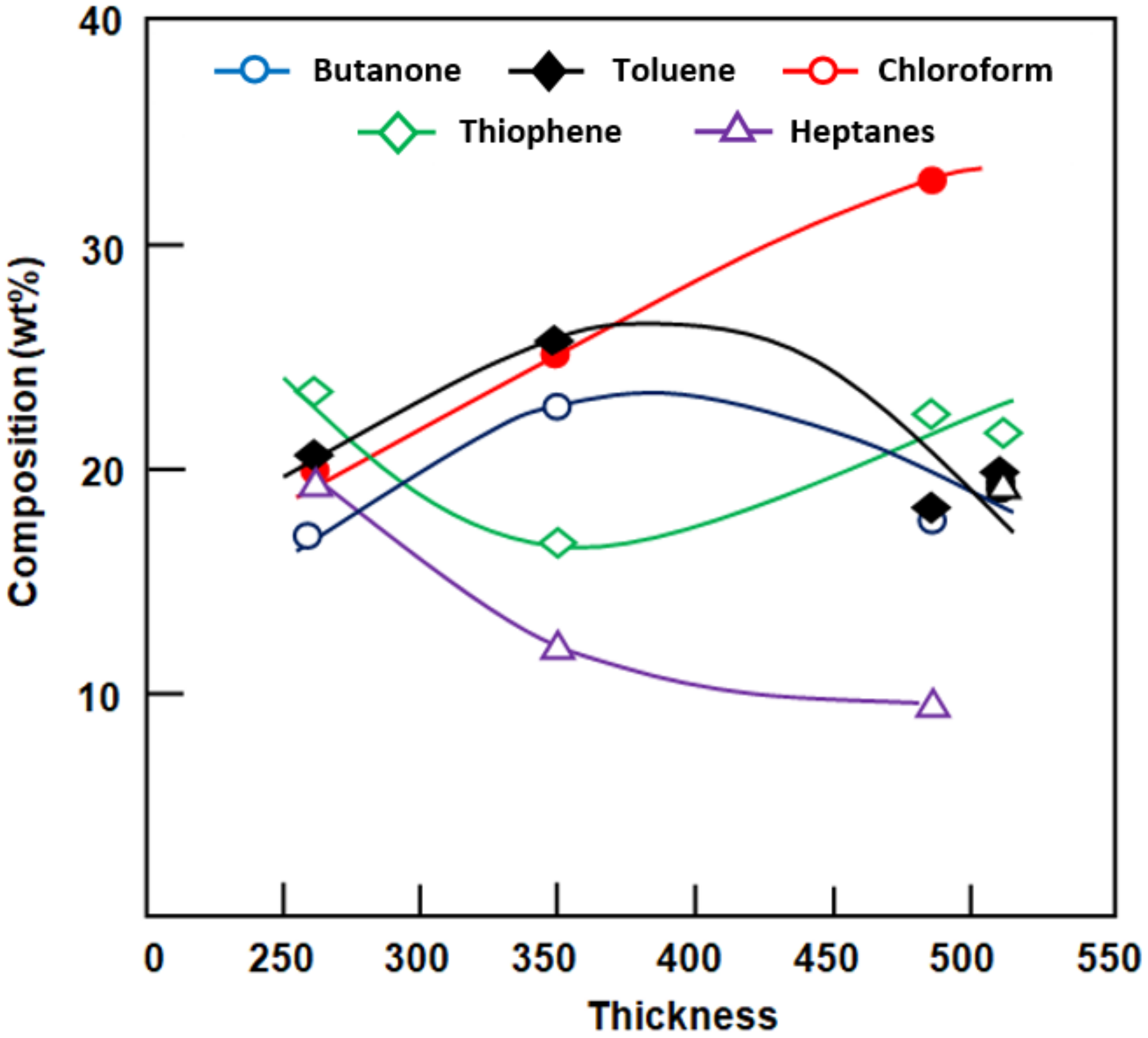
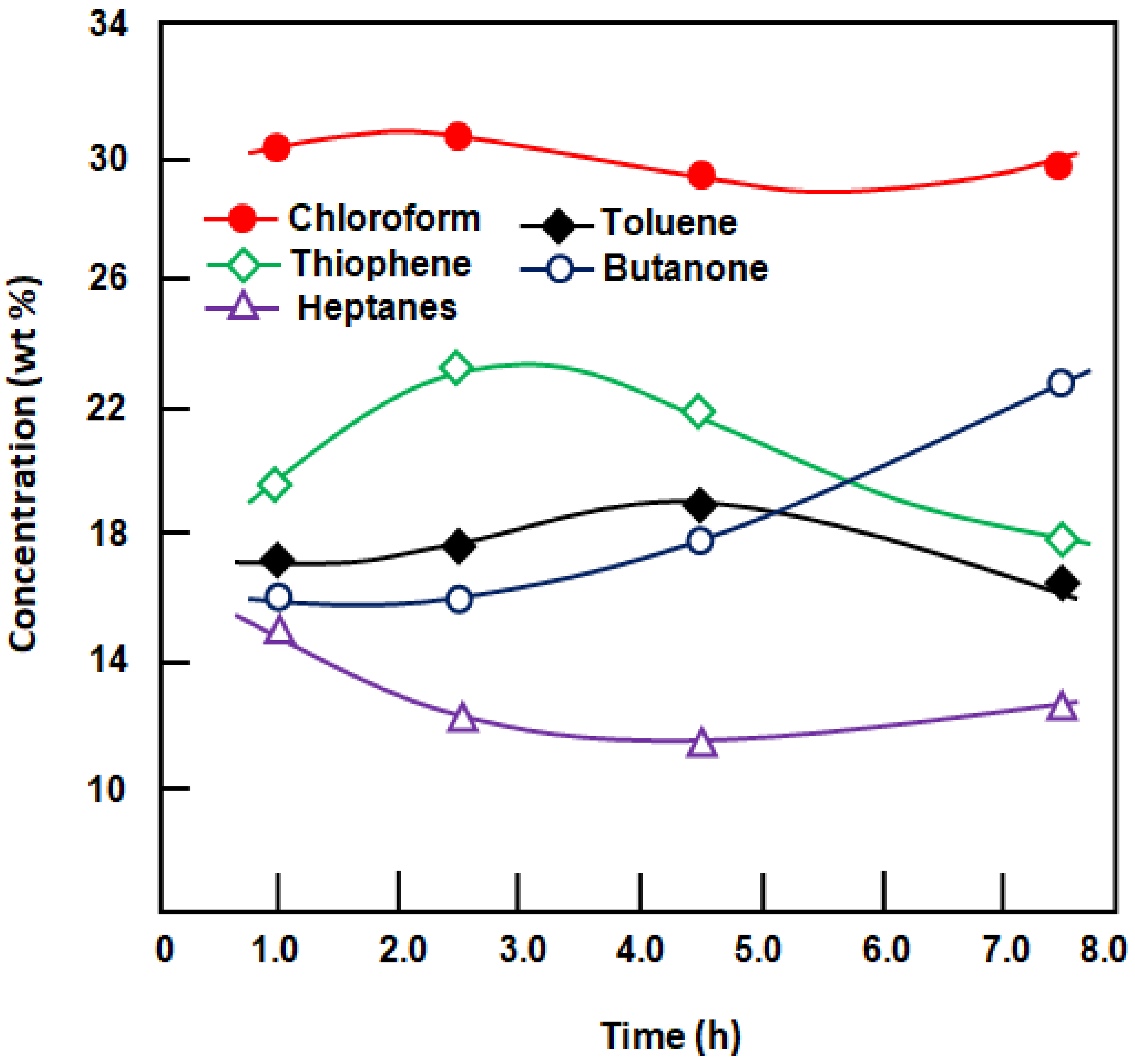
Figure 6 shows results of the quantitative analysis of the permeate using gas chromatography during the 1.5 h separation process using membranes of different thicknesses.
Over this period, the fraction of each component in the permeate did not follow a general rule when the membrane thickness was varied. For example, the toluene and butanone concentrations followed a similar trend, reaching the maximum when the membrane thickness was approximately 400 μm. By contrast, the concentration of chloroform increased linearly, reaching the highest concentration of 33 wt.%, thus confirming the high total flux and high selectivity of the organic mixture obtained. During this time, the concentration of heptanes decreased slowly, reaching the lowest value of 9.85 wt.% with the thickest membrane. A slight change in the concentration of the other components in the permeate was observed when the membrane thickness was the minimum and maximum.
- (B)
Effect of the Stirring Rate on the Extraction Performance
When the mixture to be separated contains three heterogeneous phases, the effect of stirring (mixing) should be considered. Therefore, it is essential to study the influence of the stirring rate on the pervaporation parameters. Accordingly, this study examined the effect of stirring at stirring rates of 250, 500, 750, and 1000 rpm with the temperature and membrane thickness maintained at 20 °C and 260 µm, respectively.
Figure 7 and
Figure 8 present the total flux and selectivity obtained, respectively. The total flux decreased with time and stabilized to a minimum of 0.30 kg·m
−2·h
−1 at 3.5 h when the stirring rate was greater than or equal to 750 rpm (
Figure 7). The best performance was obtained at a low stirring rate (250 and 500 rpm), in which the total flux reached 3.9–4.0 kg·m
−2·h
−1 during 1.5 h. This is because a stirring speed less than or equal to 500 rpm is insufficient to make a more homogeneous ternary mixture from a heavy organic phase rich in chloroform, an intermediate aqueous phase rich in water, and a light organic phase (heptanes/toluene/butanone). Under these conditions, the superior affinity of chloroform with respect to the membrane and its direct contact with the membrane promotes an increase in total flux. At a higher stirring rate, however, water in its majority and thiophene, which have practically the same density (1 and 1.05 g.mL
−1), in the middle phase at a slow stirring rate, come into contact with the membrane to produce a hydrophilic barrier that reduces the total flux significantly.
Figure 8 illustrates the effects of the stirring rate on the selectivity toward the organic mixture with respect to water. The selectivity as a function of time at different stirring rates diverged practically from the same region characterized by the selectivity varying between 450 and 995, depending on the stirring rate, reaching 4900, 2000, 200, and 200 after 3.5 h at stirring rates of 250 rpm, 500, 750, and 1000 rpm, respectively. At a stirring rate less than or equal to 500 rpm, the selectivity increased, with a significant increase observed at 250 rpm. By contrast, for stirring rates of 750 and 1000 rpm, the selectivity was minimal and followed a similar trend: a slow decrease with time to 100 to 3 h and relatively constant thereafter. These results can be explained in a similar manner to that of the total flux observed in the previous section. The selectivity increased at lower stirring rates because of the chloroform-rich phase that remained at the bottom of the feed and could be absorbed readily by the membrane.
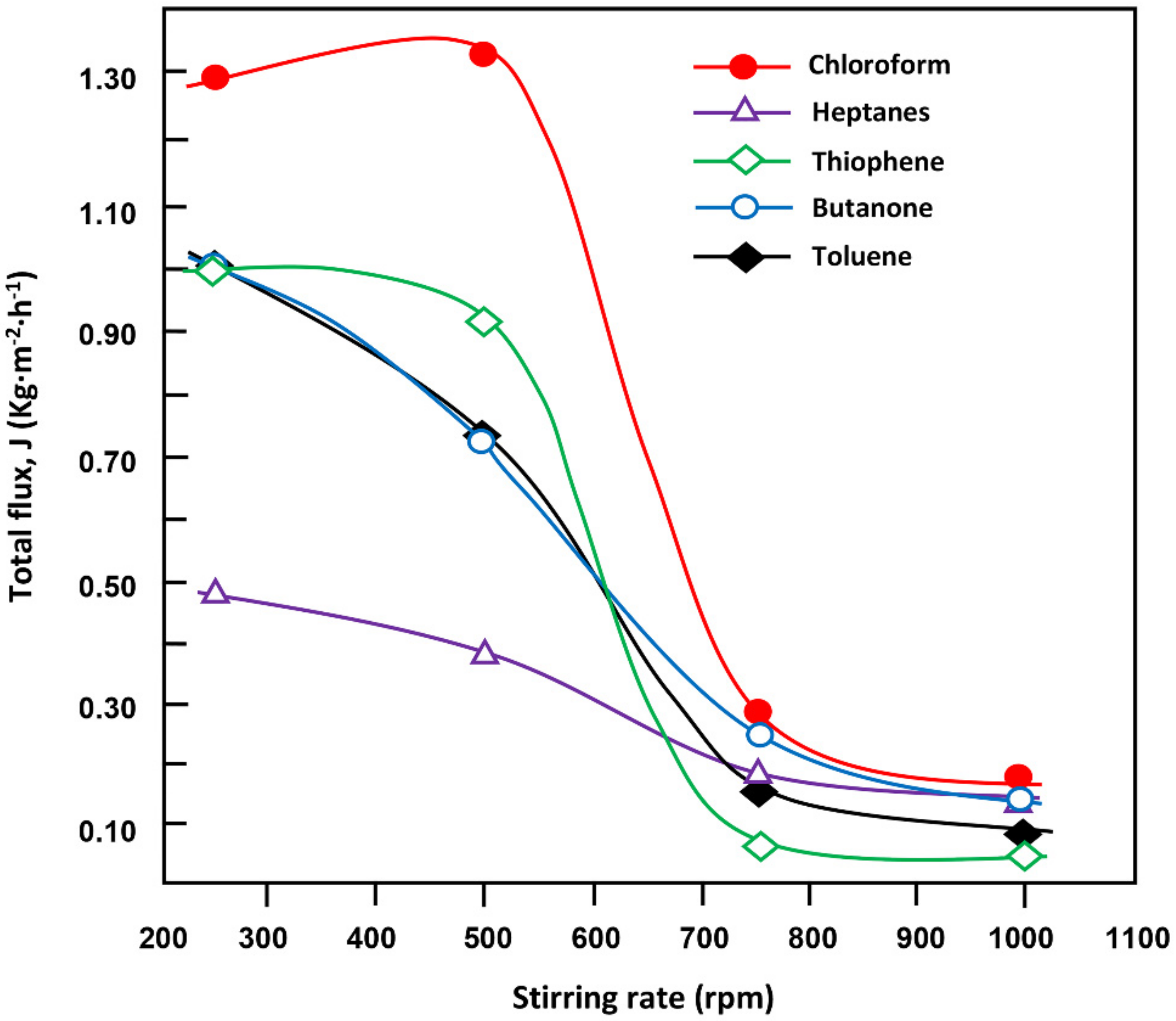
Figure 9 illustrates the variation of the cumulative flux of each organic component in the permeate versus the stirring rate during 1.5 h. All the curves have practically the same profiles describing sigmoids of degree of curvature varying according to the nature of the organic component when the agitation reached 500 rpm. Indeed, the cumulative flux of each organic component decreased irregularly with the stirring rate of the feed. This was expected because the presence of three distinct phases affects the separation process as previously showed in
Figure 1. For example, chloroform, which was at the bottom in direct contact with the membrane was the most enriched component in the permeate at any stirring rate, the cumulative flux of this component in the organic mixture reached a pseudo stability at its maximum of 1.29–1.31 kg·m
−2·h
−1 at lower stirring rate (≤500 rpm) then dramatically decreased to reach a minimum of 0.17 kg·m
−2·h
−1 at 1000 rpm. Thiophene (density, 1.05 g·cm
−3) in the middle, butanone (density, 0.80 g·mL
−1) and toluene (density, 0.867 g·mL
−1) being above in the feeds have substantially similar values of cumulative flux at minimum stirring rate then decrease together, diverging to finally achieve comparable pseudo-stabilities at stirring rate equal or greater than 750 rpm. Heptanes (density, 0.684 g·mL
−1) which is poorly soluble in water (solubility, 0.003 g·mL
−1), although it shows the best affinity with the PDMS membrane (
Δδ = 0.6 cal
0.5·cm
−1.5) shows the lowest flux values at any stirring rate. This indicates that the density of the compound to be separated played a primary role in separating these components from water notably at low stirring rates.
Increasing the stirring rate from 250 to 500 rpm did not appear to destroy this phase equilibrium. Under these conditions, most of the chloroform which is placed at the bottom of the feed coming into direct contact with the membrane and is absorbed immediately. A relatively slow decrease in toluene flux followed by stability was similar to that of butanone observed in the permeate when the stirring rate was increased. For these two components initially found mainly in the upper phase because of their low densities, the increased agitation at 750 and 1000 rpm was not enough to place the components in contact with the membrane. For thiophene, which has a similar density to that of water, a fraction of this component remains in the middle of the charge forming a pseudo-emulsion with water, and another fraction dissolves in chloroform because of the smaller difference in the solubility parameters between them [∆δ = 0.6 (cal·cm−3) 0.5]. Under these conditions, a stirring rate of 250 rpm was sufficient to increase the contact between the thiophene molecules and the membrane, causing a 7% by weight increase in thiophene concentration in the permeate compared to its initial concentration in the feed (20 wt.%). At a higher stirring rate, the concentration of thiophene in the permeate decreased significantly to a minimum of 7.8% by weight at 1000 rpm in favor of the concentration of heptanes, which increased symmetrically with increasing thiophene concentration, indicating a close link between these two components.
At a higher stirring rate, pseudo-homogenization of the five components occurs, leading to a large fraction of heptanes down the feed, which are absorbed immediately by the membrane because of their excellent affinity with the membrane [∆δ = 0.6 (cal·cm−3) 0.5]. However, a significant fraction of thiophene rises to the top of the feed.
- (C)
Effect of Temperature on the Total Flux and Selectivity
Effects of temperature on the membrane performance in separating organic compounds from water have been investigated [
37,
66,
67]. In general, the total flux increased with temperature, but the selectivity of the membrane decreased. For example, Byrne [
68] used PDMS membranes to extract ethyl acetate, methyl isobutylketone, methylethylketone, and aniline at temperatures ranging from 50 to 80 °C. They reported that the most successful separations, in terms of selectivity and flux, were obtained for methyl isobutyl ketone–water at 80 °C, in which 774 and 0.40 kg·m
−2·h
−1, respectively, were reached.
In this study, temperature had a significant effect on the pervaporation parameters for this ternary system. This is because an increase in temperature for an organic/water-immiscible system leads to an increase in the solubility of organic components in water, resulting in the enrichment of water in the organic component. This helps facilitate the transfer of a fraction of the organic components concentrated in the upper phase (toluene, heptanes, and butanone) to the lower phase rich in chloroform, which will then be absorbed easily by the membrane.
This section reports the selective separation of an organic mixture from water using the optimal conditions, which are a membrane thickness of 260 μm and a stirring rate of 250 rpm, by varying the temperature between 20 and 50 °C covering seasonal temperatures in KSA.
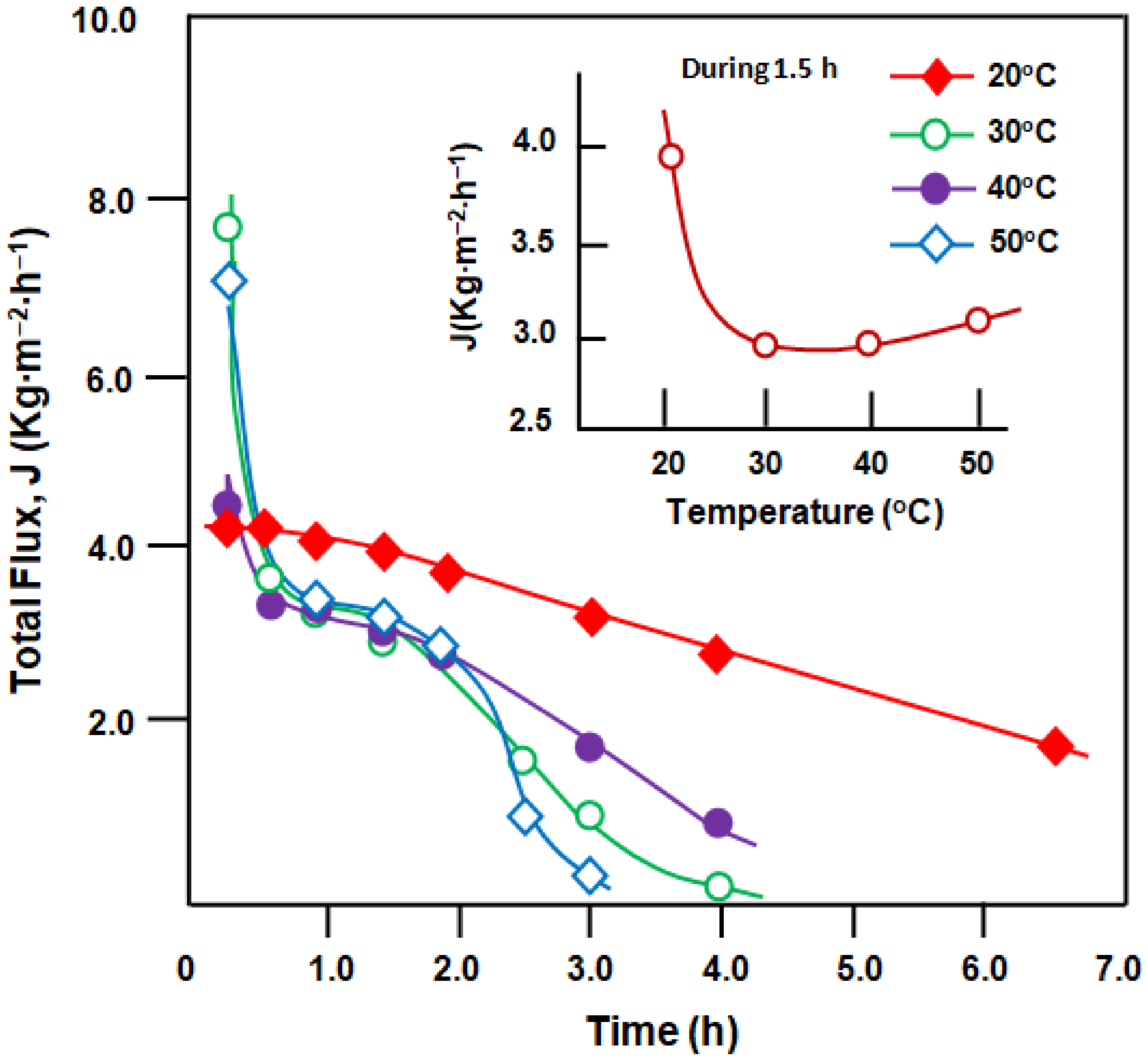
Figure 10 and
Figure 11 present the total flux and selectivity of the membrane with time of the separation presses at different temperatures, respectively. At all temperatures investigated, the total pervaporation flux decreased with time after pseudo-stabilization at 2.73–3.30 kg·m
−2·h
−1 between 0.5 and 2 h (
Figure 10), except for the processes carried out at 20 °C, in which the total flux decreased continually with time. The pseudo-stability of the total flux during this period was attributed to pseudo-stabilization of the concentration of organic compounds dissolved in water. Indeed, the concentration of organics in water was at a steady state due to the continuous dissolution of organic components from the organic phase at the top of the feed. After approximately 2 h of separation, the decrease in total flow was probably due to the exhaustion of organic substances in the feed, in which their concentration continued to decrease, leading to a decrease in the organic content in the permeate. During 1.5 h of the separation process, J decreased significantly with temperature, reaching a minimum of 2.96 kg·m
−2·h
−1 at 30 °C followed by a slight increase, reaching a total flux of 3.10 kg·m
−2·h
−1 at 50 °C (inset in
Figure 10). Overall, the best performance of the total flow was obtained at a temperature close to ambient temperature (20 °C).
The complexity of the system and its behavior during the separation process, i.e., the irregularity of the variations of the total flux with temperature, did not allow a determination of the activation energy through the flux according to the Arrhenius equation.
The selectivity of the membrane toward the organic mixture with regard to water increased gradually with time (
Figure 11), diverging from the same point characterized by a selectivity of −600 regardless of the temperature to reach a significant value of 7200 when the temperature was increased from 30 to 50 °C. During the first 1.5 h of the process, the selectivity increased from 1130 to 7810 when the temperature of the mixture was increased from 20 to 50 °C. This phenomenon was attributed to an increase in the solubility of the organic components with increasing temperature, particularly the slightly polar components. Although the selectivity decreased with increasing temperature, this remark was only valid in the case of miscible mixtures, which was not part of this case.
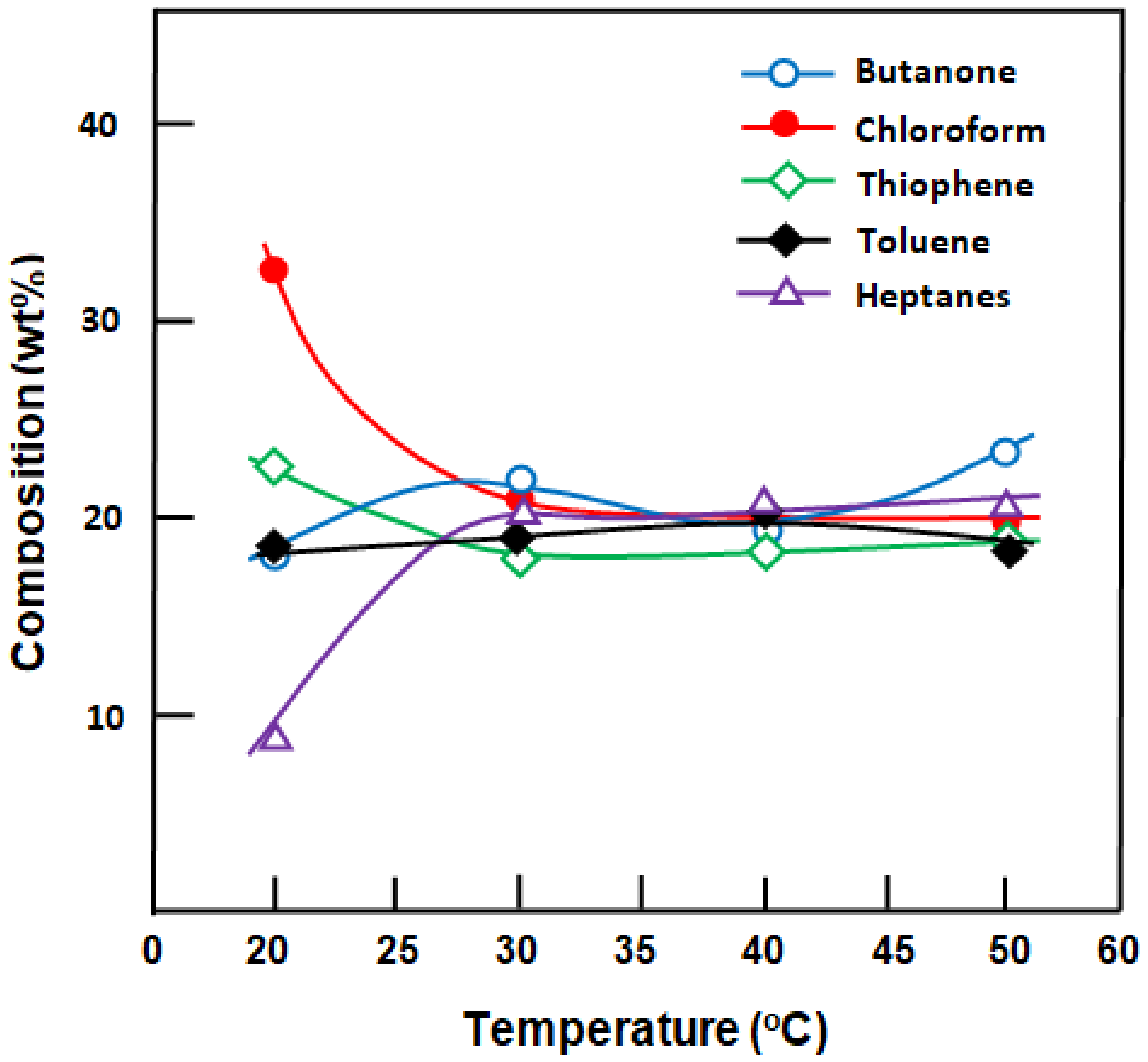
The composition of the organic mixture in the permeate during 1.5 h of separation was analyzed quantitatively (
Figure 12). The profiles of these curves clearly indicate the best enrichment of the organic mixture in chloroform when the separation process was carried out at 20 °C. Indeed, the concentration of this component increased initially from 20 wt.% in the feed to 32.5 wt.% in the permeate, resulting in a significant decrease in the mass percentage of heptanes from 20% to 9.5% by weight. In this case, a stirring rate of 250 rpm was insufficient to homogenize the dispersion of the organic mixture in the feed. Therefore, as reported in previous sections, most of the chloroform remained at the bottom of the feed in direct contact with the membrane. At higher temperatures, the composition of the mixture converged toward that at the start, from 30 to 50 °C, which oscillated between 19 and 24 wt.% depending on the nature of the component. The following two essential factors can explain the pseudo-stability of the composition of each organic component in the permeate extracted at higher temperatures: (i) an increase in free volume between the PDMS chains constituting the membrane and (ii) an increase in the solubility of the organic mixture in the aqueous phase when the temperature increases, thereby promoting the transfer of all organic molecules non-selectively.
- (D)
Organics/Water–SILICONE1200 Membrane Performance
Identification of the necessary conditions is crucial for achieving the best performance in the pervaporation parameters using the SILICONE1200 membrane to extract VOCs from wastewater. Indeed, the best performance of this membrane during the 1.5 h period was obtained when the membrane thickness, stirring rate of mixing, and temperature of the feed were 260 μm (higher total flux) (
Table 9), 500 rpm (higher selectivity) (
Table 10), and 50 °C (higher selectivity) (
Table 11), respectively.