Fish Ecology of the Alto Madre de Dios River Basin (Peru): Notes on Electrofishing Surveys, Elevation, Palm Swamp and Headwater Fishes
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Field Sampling and Analysis
2.3. Data Analysis
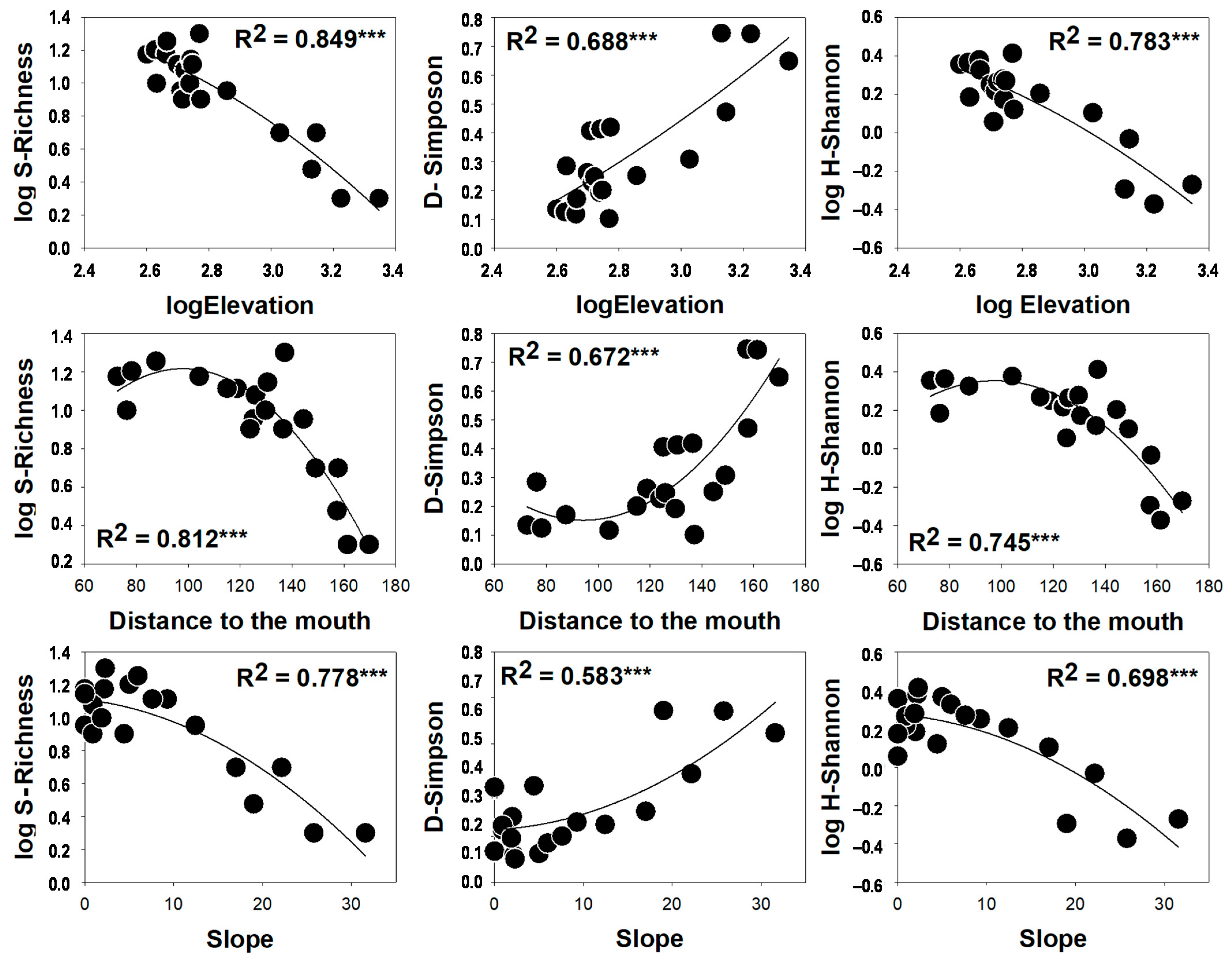
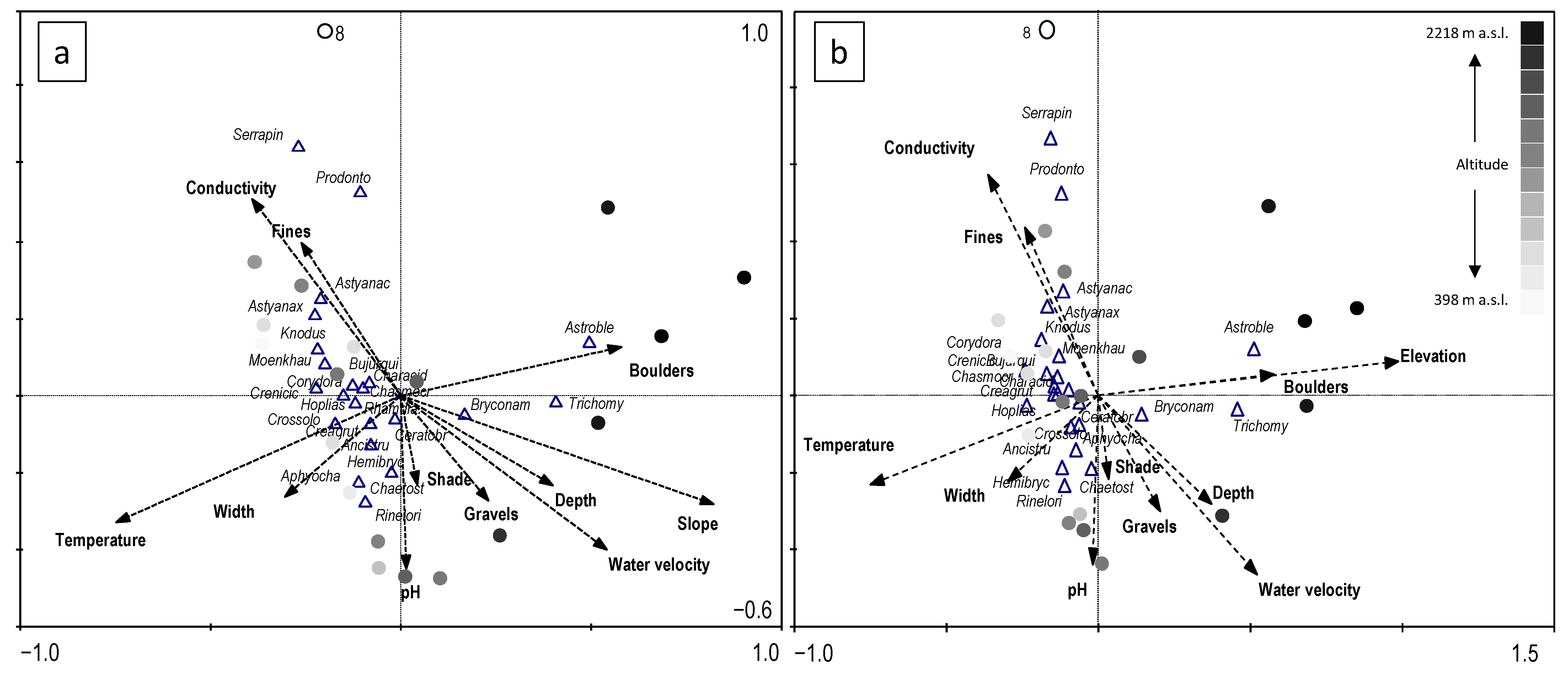
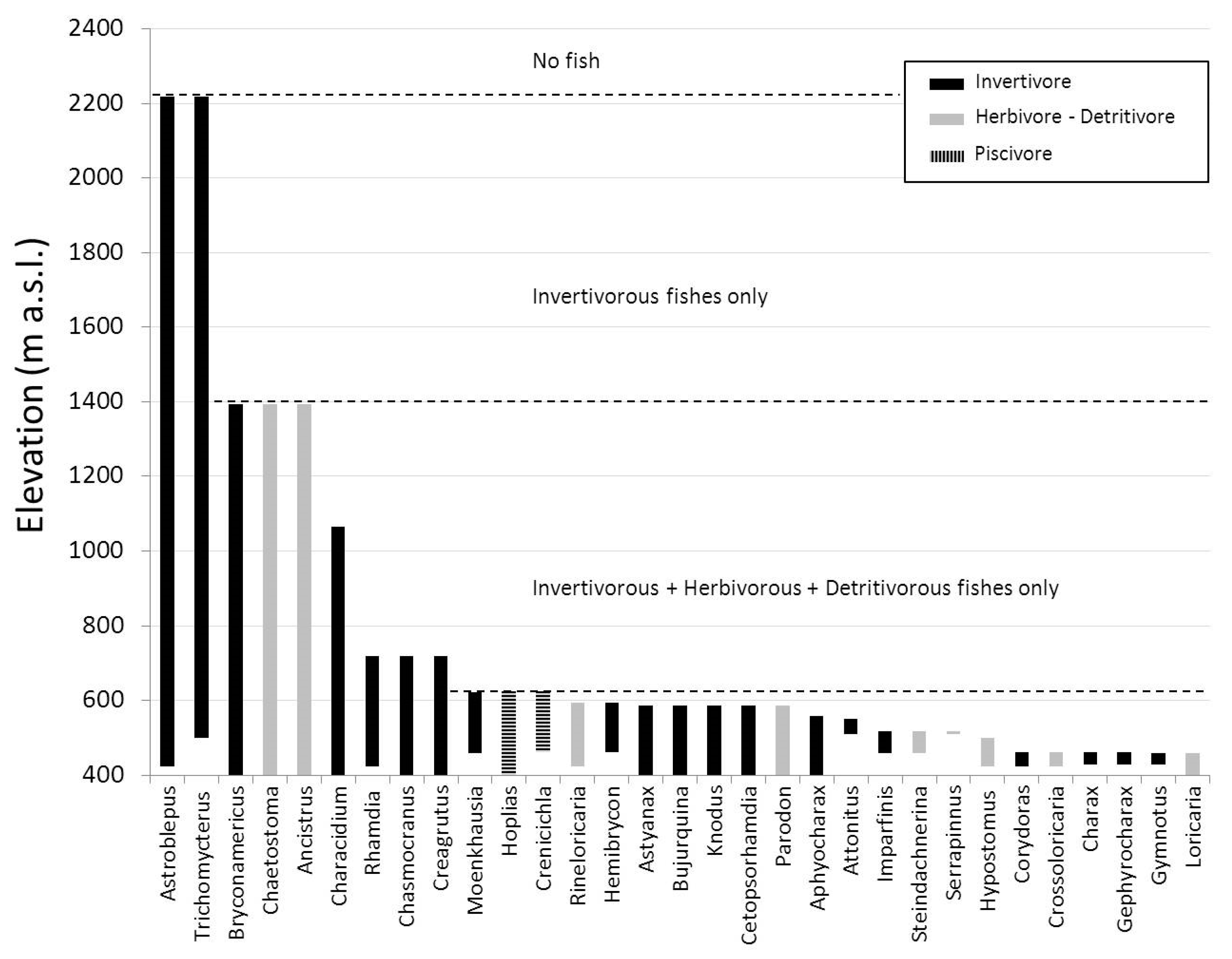
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gozlan, R.E.; Britton, J.R.; Cowx, I.G.; Copp, G.H. Current Knowledge on Non-Native Freshwater Fish Introductions. J. Fish Biol. 2010, 76, 751–786. [Google Scholar] [CrossRef]
- Ricciardi, A.; Rasmussen, J.B. Extinction Rates of North American Freshwater Fauna. Conserv. Biol. 1999, 13, 1220–1222. [Google Scholar] [CrossRef]
- Miqueleiz, I.; Böhm, M.; Ariño, A.H.; Miranda, R. Assessment Gaps and Biases in Knowledge of Conservation Status of Fishes. Aquat. Conserv. Mar. Freshw. Ecosyst. 2020, 30, 225–236. [Google Scholar] [CrossRef]
- Allan, J.D.; Flecker, A.S. Biodiversity Conservation in Running Waters. BioScience 1993, 43, 32. [Google Scholar] [CrossRef]
- Oberdorff, T.; Dias, M.S.; Jézéquel, C.; Albert, J.S.; Arantes, C.C.; Bigorne, R.; Carvajal-Vallejos, F.M.; de Wever, A.; Frederico, R.G.; Hidalgo, M.; et al. Unexpected Fish Diversity Gradients in the Amazon Basin. Sci. Adv. 2019, 5, eaav8681. [Google Scholar] [CrossRef] [PubMed]
- Lujan, N.K.; Roach, K.A.; Jacobsen, D.; Winemiller, K.O.; Vargas, V.M.; Ching, V.R.; Maestre, J.A. Aquatic Community Structure across an Andes-to-Amazon Fluvial Gradient. J. Biogeogr. 2013, 40, 1715–1728. [Google Scholar] [CrossRef]
- Bax, V.; Francesconi, W. Conservation Gaps and Priorities in the Tropical Andes Biodiversity Hotspot: Implications for the Expansion of Protected Areas. J. Environ. Manag. 2019, 232, 387–396. [Google Scholar] [CrossRef]
- Ortega, H.; Hidalgo, M.; Trevejo, G.; Correa, E.; Cortijo, A.M.; Meza, V.; Espino, J. Lista Anotada de Los Peces de Aguas Continentales Del Perú; Ministerio del Ambiente, Dirección General de Diversidad Biológica and Museo de Historia Natural, UNMSM: Lima, Peru, 2012; ISBN 9786124605321. [Google Scholar]
- Benejam, L.; Tobes, I.; Brucet, S.; Miranda, R. Size Spectra and Other Size-Related Variables of River Fish Communities: Systematic Changes along the Altitudinal Gradient on Pristine Andean Streams. Ecol. Indic. 2018, 90, 366–378. [Google Scholar] [CrossRef]
- Carvajal-Quintero, J.D.; Escobar, F.; Alvarado, F.; Villa-Navarro, F.A.; Jaramillo-Villa, Ú.; Maldonado-Ocampo, J.A. Variation in Freshwater Fish Assemblages along a Regional Elevation Gradient in the Northern Andes, Colombia. Ecol. Evol. 2015, 5, 2608–2620. [Google Scholar] [CrossRef]
- Pitman, N.C.A.; Salas, K.; Loyola Azáldegui, M.D.C.; Vigo, G.; Lutz, D.A. Historia e Impacto de La Literatura Científica Del Departamento de Madre de Dios, Perú. Rev. Peru. Biol. 2008, 15, 15–22. [Google Scholar] [CrossRef]
- Albert, J.S.; Tagliacollo, V.A.; Dagosta, F. Diversification of Neotropical Freshwater Fishes. Annu. Rev. Ecol. Evol. Syst. 2020, 51. [Google Scholar] [CrossRef]
- Townsend-Small, A.; McClain, M.E.; Hall, B.; Noguera, J.L.; Llerena, C.A.; Brandes, J.A. Suspended Sediments and Organic Matter in Mountain Headwaters of the Amazon River: Results from a 1-Year Time Series Study in the Central Peruvian Andes. Geochim. Cosmochim. Acta 2008, 72, 732–740. [Google Scholar] [CrossRef]
- Laraque, A.; Bernal, C.; Bourrel, L.; Darrozes, J.; Christophoul, F.; Armijos, E.; Fraizy, P.; Pombosa, R.; Guyot, J.L. Sediment Budget of the Napo River, Amazon Basin, Ecuador and Peru. Hydrol. Process. 2009, 23, 3509–3524. [Google Scholar] [CrossRef]
- Malhi, Y.; Roberts, J.T.; Betts, R.A.; Killeen, T.J.; Li, W.; Nobre, C.A. Climate Change, Deforestation, and the Fate of the Amazon. Science 2008, 319, 169–172. [Google Scholar] [CrossRef]
- Aldrich, S.; Walker, R.; Simmons, C.; Caldas, M.; Perz, S. Contentious Land Change in the Amazon’s Arc of Deforestation. Ann. Assoc. Am. Geogr. 2012, 102, 103–128. [Google Scholar] [CrossRef]
- Garcia, A.S.; Vívian, V.M.; Rizzo, R.; West, P.; Gerber, J.S.; Engstrom, P.M.; Maria, M.V. Assessing Land Use/Cover Dynamics and Exploring Drivers in the Amazon’s Arc of Deforestation through a Hierarchical, Multi-Scale and Multi-Temporal Classification Approach. Remote Sens. Appl. Soc. Environ. 2019, 15, 100233. [Google Scholar] [CrossRef]
- Brooks, T.M.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.B.; Rylands, A.B.; Konstant, W.R.; Flick, P.; Pilgrim, J.D.; Oldfield, S.; Magin, G.; et al. Habitat Loss and Extinction in the Hotspots of Biodiversity. Conserv. Biol. 2002, 16, 909–923. [Google Scholar] [CrossRef]
- Restrepo, J.D.; Kettner, A.J.; Syvitski, J.P.M. Recent Deforestation Causes Rapid Increase in River Sediment Load in the Colombian Andes. Anthropocene 2015, 10, 13–28. [Google Scholar] [CrossRef]
- Almeida, R.M.; Shi, Q.; Gomes-Selman, J.M.; Wu, X.; Xue, Y.; Angarita, H.; Barros, N.; Forsberg, B.R.; García-Villacorta, R.; Hamilton, S.K.; et al. Reducing Greenhouse Gas Emissions of Amazon Hydropower with Strategic Dam Planning. Nat. Commun. 2019, 10, 4281. [Google Scholar] [CrossRef]
- Anderson, E.P.; Jenkins, C.N.; Heilpern, S.; Maldonado-Ocampo, J.A.; Carvajal-Vallejos, F.M.; Encalada, A.C.; Rivadeneira, J.F.; Hidalgo, M.; Cañas, C.M.; Ortega, H.; et al. Fragmentation of Andes-to-Amazon Connectivity by Hydropower Dams. Sci. Adv. 2018, 4, eaao1642. [Google Scholar] [CrossRef]
- Yu, D.W.; Hendrickson, T.; Castillo, A. Ecotourism and Conservation in Amazonian Peru: Short-Term and Long-Term Challenges. Environ. Conserv. 1997, 24, 130–138. [Google Scholar] [CrossRef]
- Encalada, A.C.; Flecker, A.S.; Poff, N.L.; Suárez, E.; Herrera-R, G.A.; Ríos-Touma, B.; Jumani, S.; Larson, E.I.; Anderson, E.P. A Global Perspective on Tropical Montane Rivers. Science 2019, 365, 1124–1129. [Google Scholar] [CrossRef]
- Anderson, E.P.; Maldonado-Ocampo, J.A. A Regional Perspective on the Diversity and Conservation of Tropical Andean Fishes. Conserv. Biol. 2011, 25, 30–39. [Google Scholar] [CrossRef]
- Gentry, A. Changes in Plant Community Diversity and Floristic Composition on Environmental and Geographical Gradients. Ann. Mo. Bot. Gard. 1988, 75, 1–34. [Google Scholar] [CrossRef]
- McCoy, E.D. The Distribution of Insects along Elevational Gradients. Oikos 1990, 58, 313. [Google Scholar] [CrossRef]
- Fu, C.; Hua, X.; Li, J.; Chang, Z.; Pu, Z.; Chen, J. Elevational Patterns of Frog Species Richness and Endemic Richness in the Hengduan Mountains, China: Geometric Constraints, Area and Climate Effects. Ecography 2006, 29, 919–927. [Google Scholar] [CrossRef]
- Montaño-Centellas, F.A.; McCain, C.; Loiselle, B.A. Using Functional and Phylogenetic Diversity to Infer Avian Community Assembly along Elevational Gradients. Glob. Ecol. Biogeogr. 2020, 29, 232–245. [Google Scholar] [CrossRef]
- Lessmann, J.; Guayasamin, J.M.; Casner, K.L.; Flecker, A.S.; Funk, W.C.; Ghalambor, C.K.; Gill, B.A.; Jácome-Negrete, I.; Kondratieff, B.C.; Poff, L.N.; et al. Freshwater Vertebrate and Invertebrate Diversity Patterns in an Andean-Amazon Basin: Implications for Conservation Efforts. Neotrop. Biodivers. 2016, 2, 99–114. [Google Scholar] [CrossRef]
- Jaramillo-Villa, Ú.; Maldonado-Ocampo, J.A.; Escobar, F. Altitudinal Variation in Fish Assemblage Diversity in Streams of the Central Andes of Colombia. J. Fish Biol. 2010, 76, 2401–2417. [Google Scholar] [CrossRef]
- Salvador, S.; Clavero, M.; Leite Pitman, R. Large Mammal Species Richness and Habitat Use in an Upper Amazonian Forest Used for Ecotourism. Mamm. Biol. Z. Säugetierkunde 2011, 76, 115–123. [Google Scholar] [CrossRef]
- Thieme, M.L.; Lehner, B.; Abell, R.; Hamilton, S.K.; Kellndorfer, J.; Powell, G.; Riveros, J.C. Freshwater Conservation Planning in Data-Poor Areas: An Example from a Remote Amazonian Basin (Madre de Dios River, Peru and Bolivia). Biol. Conserv. 2007, 135, 484–501. [Google Scholar] [CrossRef]
- Fricke, R.; Eschmeyer, W.N.; van der Laan, R. Eschmeyer’s Catalog of Fishes: Genera, Species, References. Available online: http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp (accessed on 5 May 2020).
- Radinger, J.; Britton, J.R.; Carlson, S.M.; Magurran, A.E.; Alcaraz-Hernández, J.D.; Almodóvar, A.; Benejam, L.; Fernández-Delgado, C.; Nicola, G.G.; Oliva-Paterna, F.J.; et al. Effective Monitoring of Freshwater Fish. Fish Fish. 2019, faf.12373. [Google Scholar] [CrossRef]
- Godinho, F.N.; Ferreira, M.T.; Santos, J.M. Variation in Fish Community Composition along an Iberian River Basin from Low to High Discharge: Relative Contributions of Environmental and Temporal Variables. Ecol. Freshw. Fish 2000, 9, 22–29. [Google Scholar] [CrossRef]
- Clarke, K.R. Non-parametric Multivariate Analyses of Changes in Community Structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Bray, J.R.; Curtis, J.T. An Ordination of the Upland Forest Communities of Southern Wisconsin. Ecol. Monogr. 1957, 27, 325–349. [Google Scholar] [CrossRef]
- Lepš, J.; Šmilauer, P. Multivariate Analysis of Ecological Data Using CANOCO; Cambridge University Press: Cambridge, UK, 2003. [Google Scholar]
- Braak, C.J.F.F.; Verdonschot, P.F.M.M. Canonical Correspondence Analysis and Related Multivariate Methods in Aquatic Ecology. Aquat. Sci. 1995, 57, 255–289. [Google Scholar] [CrossRef]
- ter Braak, C.J.F.; Šmilauer, P. Canoco Reference Manual and CanoDraw for Windows User’s Guide. 2002, p. 500. Available online: https://library.wur.nl/WebQuery/wurpubs/reports/341885 (accessed on 19 May 2019).
- Blanchet, F.G.; Legendre, P.; Borcard, D. Forward selection of explanatory variables. Ecology 2008, 89, 2623–2632. [Google Scholar] [CrossRef]
- R Development Core Team R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 19 May 2019).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; Ohara, R.B.; Simpson, G.L.; Solymos, P.; et al. The Vegan Package. Community Ecology Package; Version 2.5-7. 2020. Available online: https://cran.r-project.org/web/packages/vegan/index.html (accessed on 19 September 2020).
- Lujan, N.K.; Winemiller, K.O.; Armbruster, J.W. Trophic Diversity in the Evolution and Community Assembly of Loricariid Catfishes. BMC Evol. Biol. 2012, 12. [Google Scholar] [CrossRef]
- Luz-Agostinho, K.D.G.; Agostinho, A.A.; Gomes, L.C.; Júlio, H.F. Influence of Flood Pulses on Diet Composition and Trophic Relationships among Piscivorous Fish in the Upper Paraná River Floodplain. Hydrobiologia 2008, 607, 187–198. [Google Scholar] [CrossRef]
- Hoeinghaus, D.J.; Winemiller, K.O.; Taphorn, D.C. Compositional Change in Fish Assemblages along the Andean Piedmont—Llanos Floodplain Gradient of the Río Portuguesa, Venezuela. Neotrop. Ichthyol. 2004, 2, 85–92. [Google Scholar] [CrossRef]
- Alexandre, C.V.; Esteves, K.E.; de Moura e Mello, M.A.M. Analysis of Fish Communities along a Rural–Urban Gradient in a Neotropical Stream (Piracicaba River Basin, São Paulo, Brazil). Hydrobiologia 2010, 641, 97–114. [Google Scholar] [CrossRef]
- Leunda, P.M.; Sistiaga, M.; Oscoz, J.; Miranda, R. Ichthyofauna of a Near-Natural Pyrenean River: Spatio-Temporal Variability and Reach-Scale Habitat. Environ. Eng. Manag. J. 2012, 11, 1111–1124. [Google Scholar] [CrossRef]
- Tobes, I.; Gaspar, S.; Peláez-Rodríguez, M.; Miranda, R. Spatial Distribution Patterns of Fish Assemblages Relative to Macroinvertebrates and Environmental Conditions in Andean Piedmont Streams of the Colombian Amazon. Inland Waters 2016, 6, 89–104. [Google Scholar] [CrossRef]
- Hamilton, S.K.; Kellndorfer, J.; Lehner, B.; Tobler, M. Remote Sensing of Floodplain Geomorphology as a Surrogate for Biodiversity in a Tropical River System (Madre de Dios, Peru). Geomorphology 2007, 89, 23–38. [Google Scholar] [CrossRef]
- Manzi, M.; Coomes, O.T. Managing Amazonian Palms for Community Use: A Case of Aguaje Palm (Mauritia Flexuosa) in Peru. For. Ecol. Manag. 2009, 257, 510–517. [Google Scholar] [CrossRef]
- Goulding, M.; Cañas, C.; Barthem, R.; Forsberg, B.; Ortega, H. Las Fuentes Del Amazonas: Ríos, Vida y Conservación de La Cuenca Del Madre de Dios; ACCA-ACA: Lima, Peru, 2003. [Google Scholar]
- Cotta, J.N. Contributions of Local Floodplain Resources to Livelihoods and Household Income in the Peruvian Amazon. For. Policy Econ. 2015, 59, 35–46. [Google Scholar] [CrossRef]
- van der Hoek, Y.; Solas, S.Á.; Peñuela, M.C. The Palm Mauritia Flexuosa, a Keystone Plant Resource on Multiple Fronts. Biodivers. Conserv. 2019, 28, 539–551. [Google Scholar] [CrossRef]
- Zhu, R.; Li, Q.; Wang, W.; Chu, L.; Yan, Y. Effects of Local, River-Network and Catchment Factors on Fish Assemblages in the Headwater Streams of the Xin’an Basin, China. J. Freshw. Ecol. 2017, 32, 300–313. [Google Scholar] [CrossRef]
- Trujillo-Jiménez, P.; López-López, E.; Díaz-Pardo, E.; Camargo, J.A. Patterns in the Distribution of Fish Assemblages in Río Amacuzac, Mexico: Influence of Abiotic Factors and Biotic Factors. Fish Biol. Fish. 2010, 20, 457–469. [Google Scholar] [CrossRef]
- Tejerina-Garro, F.L.; Maldonado, M.; Ibañez, C.; Pont, D.; Roset, N.; Oberdorff, T. Effects of Natural and Anthropogenic Environmental Changes on Riverine Fish Assemblages: A Framework for Ecological Assessment of Rivers. Braz. Arch. Biol. Technol. 2005, 48, 91–108. [Google Scholar] [CrossRef]
- Schaefer, S.A.; Arroyave, J. Rivers as Islands: Determinants of the Distribution of Andean Astroblepid Catfishes. J. Fish Biol. 2010, 77, 2373–2390. [Google Scholar] [CrossRef]
- Miranda, R.; Tobes, I.; Gaspar, S.; Peláez-Rodríguez, M. Patterns in the Distribution of Fish Assemblages and Their Association with Habitat Variables in the Suaza River on Its Way through the Cueva de Los Guácharos National Park, Colombia. Écoscience 2018, 25, 85–95. [Google Scholar] [CrossRef]
- Pouilly, M.; Barrera, S.; Rosales, C. Changes of Taxonomic and Trophic Structure of Fish Assemblages along an Environmental Gradient in the Upper Beni Watershed (Bolivia). J. Fish Biol. 2006, 68, 137–156. [Google Scholar] [CrossRef]
- Barthem, R.; Goulding, M.; Forsberg, B.; Cañas, C.; Ortega, H. Ecología Acuática Del Rio Madre de Dios: Bases Científicas Para La Conservación de Cabeceras Andino-Amazónicas; ACCA-ACA: Lima, Peru, 2003. [Google Scholar]
- Araujo Flores, J.M. Caracterización de La Biodiversidad Acuática En La Cuenca Andino-Amazónica de Madre de Dios—Perú; Universidad de Huelva: Huelva, Spain, 2016. [Google Scholar]
- Allard, L.; Grenouillet, G.; Khazraie, K.; Tudesque, L.; Vigouroux, R.; Brosse, S. Electrofishing Efficiency in Low Conductivity Neotropical Streams: Towards a Non-Destructive Fish Sampling Method. Fish. Manag. Ecol. 2014, 21, 234–243. [Google Scholar] [CrossRef]
- Mendonça, F.P.; Magnusson, W.E.; Zuanon, J. Relationships between Habitat Characteristics and Fish Assemblages in Small Streams of Central Amazonia. Copeia 2005, 2005, 751–764. [Google Scholar] [CrossRef]
- Zalewski, M. The Estimate of Fish Density and Biomass in Rivers on the Basis of Relationships between Specimen Size and Efficiency of Electrofishing. Fish. Res. 1985, 3, 147–155. [Google Scholar] [CrossRef]
- Meador, M.R.; McIntyre, J.P.; Pollock, K.H. Assessing the Efficacy of Single-Pass Backpack Electrofishing to Characterize Fish Community Structure. Trans. Am. Fish. Soc. 2003, 132, 39–46. [Google Scholar] [CrossRef]
- Swenson, J.J.; Carter, C.E.; Domec, J.-C.; Delgado, C.I. Gold Mining in the Peruvian Amazon: Global Prices, Deforestation, and Mercury Imports. PLoS ONE 2011, 6, e18875. [Google Scholar] [CrossRef]
- Finer, M.; Orta-Martínez, M. A Second Hydrocarbon Boom Threatens the Peruvian Amazon: Trends, Projections, and Policy Implications. Environ. Res. Lett. 2010, 5, 014012. [Google Scholar] [CrossRef]
- Tognelli, M.F.; Anderson, E.P.; Jiménez-Segura, L.F.; Chuctaya, J.; Chocano, L.; Maldonado-Ocampo, J.A.; Mesa-Salazar, L.; Mojica, J.I.; Carvajal-Vallejos, F.M.; Correa, V.; et al. Assessing Conservation Priorities of Endemic Freshwater Fishes in the Tropical Andes Region. Aquat. Conserv. Mar. Freshw. Ecosyst. 2019, 29, 1123–1132. [Google Scholar] [CrossRef]
- Giangrande, A. Biodiversity, Conservation, and the “Taxonomic Impediment”. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, 451–459. [Google Scholar] [CrossRef]
- Galvis, G.; Mojica, J.I.; Duque, S.R.; Castellanos, C.; Sánchez-Duarte, P.; Arce, M.; Gutiérrez, Á.; Jiménez, L.F.; Santos, M.; Vejarano, S.; et al. Peces Del Medio Amazonas, Región de Leticia; Conservación Internacional; Editorial Panamericana, Formas e Impresos: Bogotá, Colombia, 2006. [Google Scholar]
- Carvalho, T.P.; Flores, J.A.; Espino, J.; Trevejo, G.; Ortega, H.; Jerep, F.C.; Reis, R.E.; Albert, J.S. Fishes from the Las Piedras River, Madre de Dios Basin, Peruvian Amazon. Check List 2012, 8, 973. [Google Scholar] [CrossRef]
- Dubois, A. The Relationships between Taxonomy and Conservation Biology in the Century of Extinctions. Comptes Rendus Biol. 2003, 326, 9–21. [Google Scholar] [CrossRef]
- Jézéquel, C.; Tedesco, P.A.; Darwall, W.; Dias, M.S.; Frederico, R.G.; Hidalgo, M.; Hugueny, B.; Maldonado-Ocampo, J.; Martens, K.; Ortega, H.; et al. Freshwater Fish Diversity Hotspots for Conservation Priorities in the Amazon Basin. Conserv. Biol. 2020, 34, 956–965. [Google Scholar] [CrossRef]
- Tognelli, M.F.; Lasso, C.A.; Bota-Sierra, C.A.; Jiménez-Segura, L.F.; Cox, N.A. Estado de Conservación y Distribución de la Biodiversidad de Agua Dulce en los Andes Tropicales; UICN: Gland, Switzerland, 2016; ISBN 978-2-8317-1791-3. [Google Scholar]
- Donin, L.M.; Ferrer, J.; Carvalho, T.P. Taxonomical Study of Trichomycterus (Siluriformes: Trichomycteridae) from the Ribeira de Iguape River Basin Reveals a New Species Recorded in the Early 20th Century. J. Fish Biol. 2020, 96, 886–904. [Google Scholar] [CrossRef]
- Ochoa, L.E.; Melo, B.F.; García-Melo, J.E.; Maldonado-Ocampo, J.A.; Souza, C.S.; Albornoz-Garzón, J.G.; Conde-Saldaña, C.C.; Villa-Navarro, F.; Ortega-Lara, A.; Oliveira, C. Species Delimitation Reveals an Underestimated Diversity of Andean Catfishes of the Family Astroblepidae (Teleostei: Siluriformes). Neotrop. Ichthyol. 2020, 18, 1–19. [Google Scholar] [CrossRef]
- Schaefer, S.A.; Chakrabarty, P.; Geneva, A.J.; Sabaj Pérez, M.H. Nucleotide Sequence Data Confirm Diagnosis and Local Endemism of Variable Morphospecies of Andean Astroblepid Catfishes (Siluriformes: Astroblepidae). Zool. J. Linn. Soc. 2011, 162, 90–102. [Google Scholar] [CrossRef]
- Burgass, M.J.; Arlidge, W.N.S.; Addison, P.F.E. Overstating the Value of the IUCN Red List for Business Decision-Making. Conserv. Lett. 2018, 11, 1–8. [Google Scholar] [CrossRef]
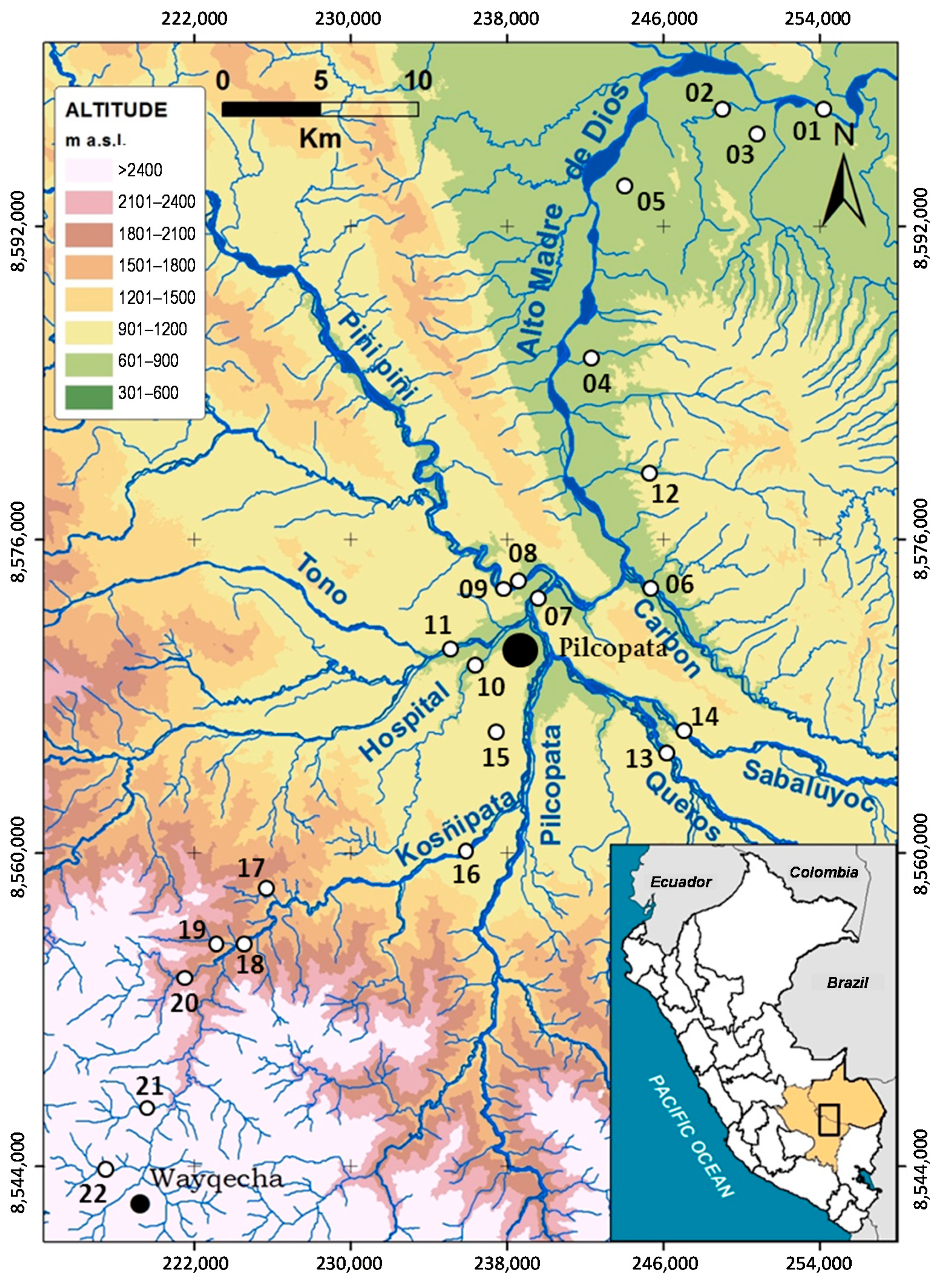


| Code | River Place | Date | Alt | pH | Ta | Tw | Cd | Wh | Dh | Vc | QBR | QHEI |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 01R | Aguas Calientes | 13 June 2012 | 398 | 7.95 | 24.3 | 21.7 | 105 | 18.2 | 25.1 | 0.51 | 100 | 72 |
| 02R | Shintuya | 12 June 2012 | 424 | 8.07 | 24.4 | 24.4 | 99 | 11.2 | 13.7 | 0.23 | 90 | 72 |
| 03A | Anaconda | 13 June 2012 | 428 | 7.37 | 26.4 | 21.4 | 119 | 3.45 | 16.8 | 0.05 | 95 | 61 |
| 04Q | Mascuitania | 14 June 2012 | 459 | 6.82 | 29.0 | 25.4 | 44 | 7.86 | 25.6 | 0.25 | 85 | 67 |
| 05Q | Puente a km 2,5 | 14 June 2012 | 462 | 8.03 | 26.1 | 22.4 | 169 | 5.54 | 18.7 | 0.15 | 100 | 74 |
| 06R | Carbón | 11 June 2012 | 499 | 8.78 | 26.3 | 25.3 | 102 | 14.4 | 34.5 | 0.41 | 100 | 85 |
| 07R | Pilcopata | 10 June 2012 | 511 | 6.94 | 24.8 | 25.0 | 119 | 15.7 | 15.6 | 0.04 | 100 | 74 |
| 08R | Piñi-Piñi | 7 June 2012 | 518 | 6.86 | 22.3 | 22.0 | 131 | 6.86 | 44.1 | 0.19 | 100 | 88 |
| 09Q | Villa Carmen | 7 June 2012 | 518 | 7.20 | 22.3 | 20.6 | 126 | 4.51 | 5.11 | 0.13 | 100 | 62 |
| 10R | Hospital | 8 June 2012 | 547 | 7.63 | 23.1 | 19.9 | 26 | 28.3 | 36.6 | 0.74 | 100 | 84 |
| 11R | Tono | 8 June 2012 | 551 | 7.70 | 19.1 | 18.5 | 50 | 6.94 | 22.1 | 0.30 | 100 | 68 |
| 12R | Salvación | 11 June 2012 | 558 | 7.26 | 24.0 | 23.2 | 33 | 4.42 | 30.1 | 0.39 | 100 | 66 |
| 13R | Queros | 9 June 2012 | 587 | 8.00 | 20.0 | 18.3 | 61 | 10.4 | 39.5 | 0.31 | 80 | 82 |
| 14R | Sabaloyacu | 9 June 2012 | 593 | 7.70 | 19.7 | 21.2 | 44 | 22.9 | 31.0 | 0.43 | 100 | 87 |
| 15A | Aguajal | 17 June 2012 | 622 | 5.77 | 23.9 | 22.0 | 26 | 2.49 | 57.4 | 0.04 | 50 | 63 |
| 16R | Asunción | 10 June 2012 | 720 | 7.40 | 26.4 | 19.3 | 65 | 5.16 | 19.9 | 0.32 | 100 | 78 |
| 17Q | Quitacalzon | 16 June 2012 | 1064 | 7.83 | 20.9 | 18.7 | 52 | 4.07 | 31.4 | 0.45 | 100 | 88 |
| 18R | Kosñipata | 16 June 2012 | 1347 | 7.53 | 22.2 | 16.0 | 89 | 6.42 | 24.4 | 0.15 | 95 | 70 |
| 19R | San Pedro | 16 June 2012 | 1394 | 7.81 | 20.1 | 18.5 | 43 | 7.74 | 49.4 | 0.48 | 95 | 88 |
| 20R | Unión | 17 June 2012 | 1673 | 7.72 | 20.2 | 15.3 | 64 | 8.41 | 32.0 | 0.72 | 75 | 66 |
| 21Q | Wayquecha | 18 June 2012 | 2218 | 7.40 | 17.3 | 11.4 | 51 | 3.99 | 30.2 | 0.83 | 100 | 84 |
| 22Q | Pacchayoc | 18 June 2012 | 2411 | 4.41 | 16.1 | 11.7 | 188 | 6.11 | 23.9 | 0.89 | 100 | 82 |
| Elevation-700 m | |||
|---|---|---|---|
| Taxa | % Contri. | U | A |
| Astroblepus | 18.36 | 1.03 | 9.00 |
| Trichomycterus | 10.76 | 0.78 | 5.78 |
| Knodus | 8.91 | 3.92 | - |
| Chaetostoma | 7.67 | 3.54 | 1.93 |
| Creagrutus | 6.35 | 3.09 | - |
| Bryconamericus | 6.01 | 1.84 | 2.64 |
| Ceratobranchia | 5.88 | 3.22 | - |
| Astyanax | 5.26 | 2.45 | - |
| Ancistrus | 4.57 | 2.48 | 0.28 |
| Bujurquina | 3.42 | 1.73 | - |
| Hemibrycon | 3.36 | 1.63 | - |
| Characidium | 3.01 | 1.33 | 0.28 |
| Aphyocharax | 2.67 | 1.52 | - |
| Rineloricaria | 2.36 | 1.05 | - |
| Hoplias | 1.96 | 0.87 | - |
| Richness | 24 | 6 | |
| (a) Slope | Marginal Effects | Intraset Correlations | Regression/Canonical Coefficients | ||
| Groups | λA | CCA1 | CCA2 | CCA1 | CCA2 |
| Slope | 0.44 | 0.786 | −0.261 | 0.755 | −0.114 |
| Water temperature | 0.38 | −0.715 | −0.304 | −0.418 | −0.433 |
| Water velocity | 0.25 | 0.517 | −0.370 | 0.047 | 0.002 |
| Boulders | 0.25 | 0.554 | 0.118 | 0.006 | 0.111 |
| Conductivity | 0.22 | −0.374 | 0.472 | 0.007 | 0.987 |
| Depth | 0.19 | 0.382 | −0.216 | −0.076 | 0.377 |
| Fines | 0.15 | −0.250 | 0.368 | −0.149 | 0.282 |
| Width | 0.14 | −0.291 | −0.243 | −0.132 | 0.064 |
| pH | 0.13 | 0.014 | −0.415 | −0.090 | −0.662 |
| Gravels | 0.07 | 0.220 | −0.252 | 0.018 | 0.236 |
| Shade | 0.07 | 0.041 | −0.214 | −0.266 | −0.289 |
| (b) Elevation | Marginal Effects | Intraset Correlations | Regression/Canonical Coefficients | ||
| Groups | λA | CCA1 | CCA2 | CCA1 | CCA2 |
| Elevation | 0.59 | 0.963 | 0.081 | 1.069 | 0.077 |
| Water temperature | 0.38 | −0.730 | −0.213 | 0.189 | −0.330 |
| Water velocity | 0.25 | 0.510 | −0.427 | 0.074 | −0.032 |
| Boulders | 0.25 | 0.563 | 0.051 | 0.091 | −0.010 |
| Conductivity | 0.22 | −0.354 | 0.526 | −0.068 | 1.064 |
| Depth | 0.19 | 0.364 | −0.259 | −0.080 | 0.402 |
| Fines | 0.15 | −0.236 | 0.400 | 0.045 | 0.287 |
| Width | 0.14 | −0.289 | −0.205 | −0.084 | 0.184 |
| pH | 0.13 | −0.018 | −0.403 | 0.164 | −0.787 |
| Gravels | 0.07 | 0.198 | −0.276 | −0.035 | 0.261 |
| Shade | 0.07 | 0.034 | −0.199 | −0.083 | −0.250 |
| (a) Slope | CCA Axis | 1 | 2 | 3 | 4 |
| Eingenvalues | 0.579 | 0.264 | 0.167 | 0.126 | |
| Species-environmental correlations | 0.956 | 0.924 | 0.884 | 0.950 | |
| Cumulative percentage variance | |||||
| of species data | 27.9 | 40.6 | 48.7 | 54.7 | |
| of species-environmental relation | 38.4 | 55.9 | 67.0 | 75.4 | |
| (b) Elevation | CCA Axis | 1 | 2 | 3 | 4 |
| Eingenvalues | 0.597 | 0.263 | 0.170 | 0.132 | |
| Species-environmental correlations | 0.976 | 0.918 | 0.903 | 0.931 | |
| Cumulative percentage variance | |||||
| of species data | 28.8 | 41.5 | 49.7 | 56.0 | |
| of species-environmental relation | 38.8 | 55.9 | 67.0 | 75.5 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tobes, I.; Ramos-Merchante, A.; Araujo-Flores, J.; Pino-del-Carpio, A.; Ortega, H.; Miranda, R. Fish Ecology of the Alto Madre de Dios River Basin (Peru): Notes on Electrofishing Surveys, Elevation, Palm Swamp and Headwater Fishes. Water 2021, 13, 1038. https://doi.org/10.3390/w13081038
Tobes I, Ramos-Merchante A, Araujo-Flores J, Pino-del-Carpio A, Ortega H, Miranda R. Fish Ecology of the Alto Madre de Dios River Basin (Peru): Notes on Electrofishing Surveys, Elevation, Palm Swamp and Headwater Fishes. Water. 2021; 13(8):1038. https://doi.org/10.3390/w13081038
Chicago/Turabian StyleTobes, Ibon, Adrián Ramos-Merchante, Julio Araujo-Flores, Andrea Pino-del-Carpio, Hernán Ortega, and Rafael Miranda. 2021. "Fish Ecology of the Alto Madre de Dios River Basin (Peru): Notes on Electrofishing Surveys, Elevation, Palm Swamp and Headwater Fishes" Water 13, no. 8: 1038. https://doi.org/10.3390/w13081038
APA StyleTobes, I., Ramos-Merchante, A., Araujo-Flores, J., Pino-del-Carpio, A., Ortega, H., & Miranda, R. (2021). Fish Ecology of the Alto Madre de Dios River Basin (Peru): Notes on Electrofishing Surveys, Elevation, Palm Swamp and Headwater Fishes. Water, 13(8), 1038. https://doi.org/10.3390/w13081038







