Natural and Anthropogenic Geochemical Tracers to Investigate Residence Times and Groundwater–Surface-Water Interactions in an Urban Alluvial Aquifer
Abstract
1. Introduction
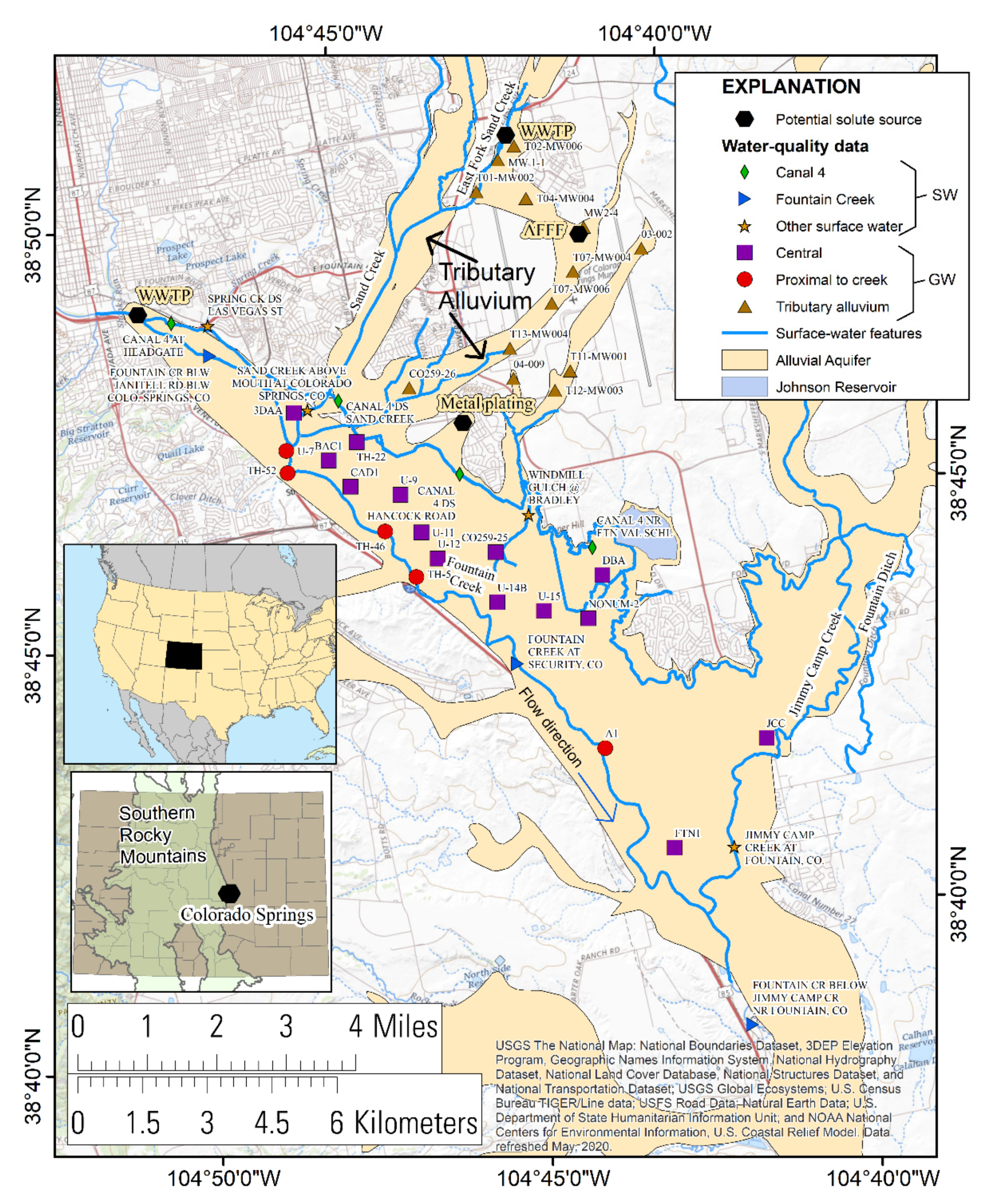
2. Materials and Methods
2.1. Sample Collection, Analysis, and Quality Control
2.2. Multivariate Evaluation
2.3. Noble Gases and Groundwater Residence-Time Tracers
2.3.1. Dissolved Gas Modeling
2.3.2. Apparent Ages and Lumped-Parameter Modeling
3. Results and Discussion
3.1. Major Ions and Trace Elements
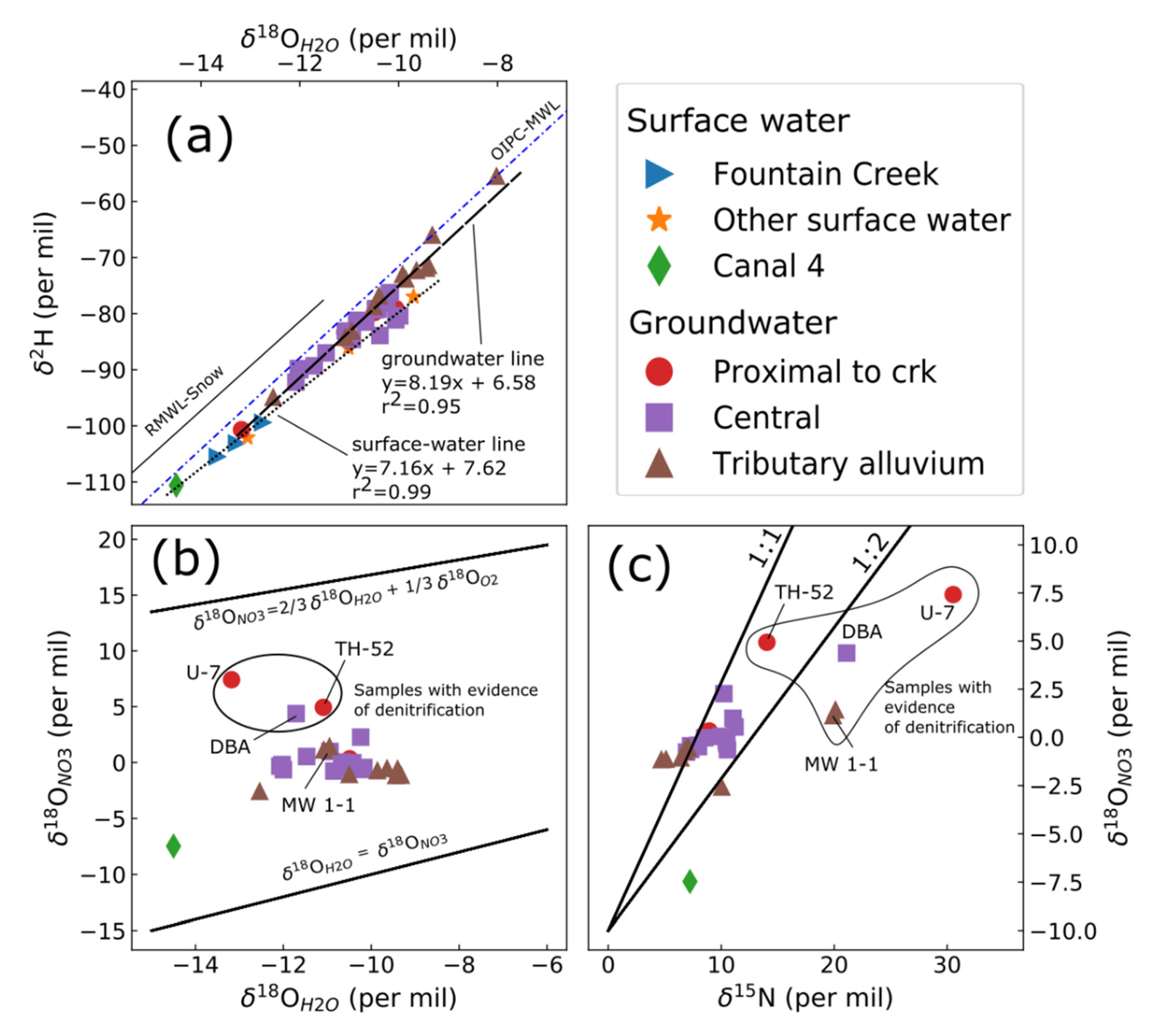
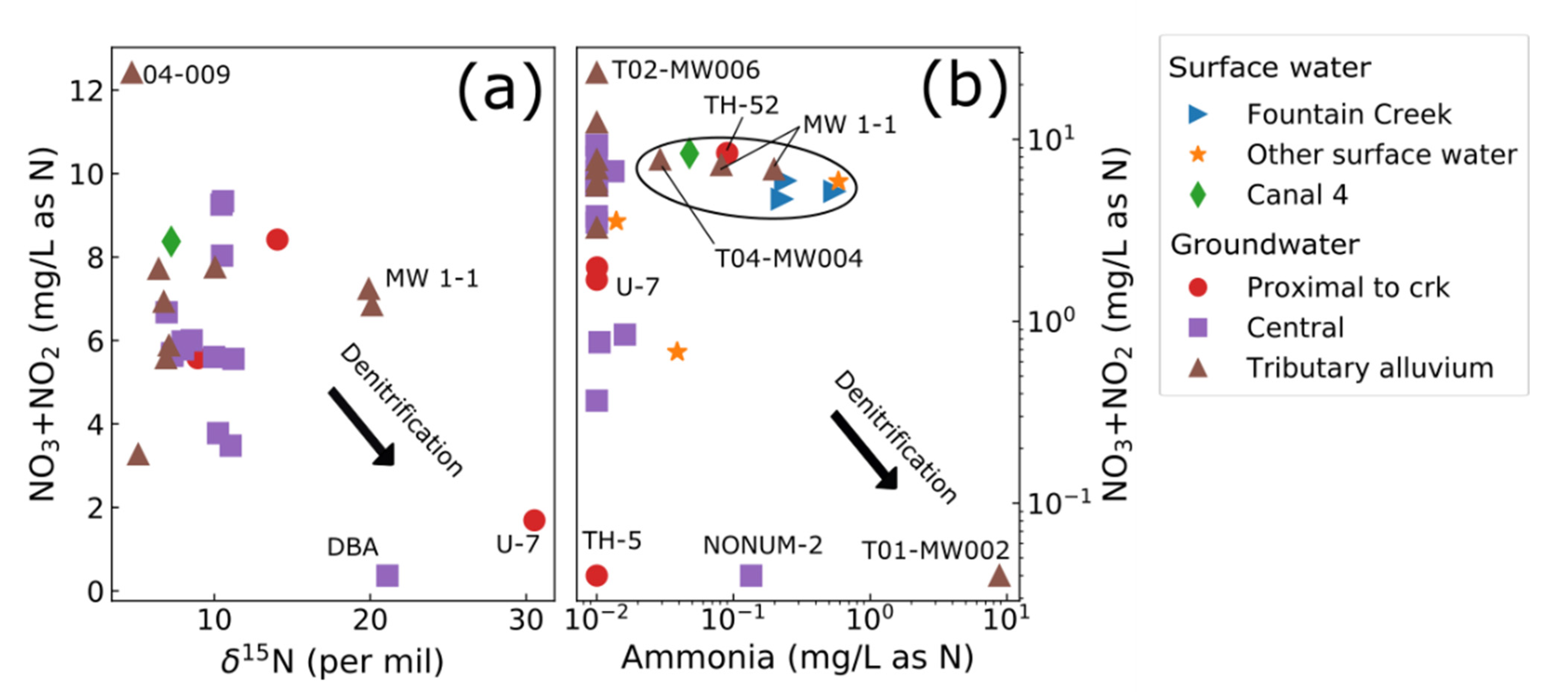
3.2. Stable Isotopes
3.3. Rare Earth Elements
3.4. Pharmaceutical Compounds and Wastewater-Indicator Compounds
3.5. Principal Component Analysis
3.6. Noble Gases and Groundwater Residence-Time Tracers
3.6.1. Noble-Gas Isotopes
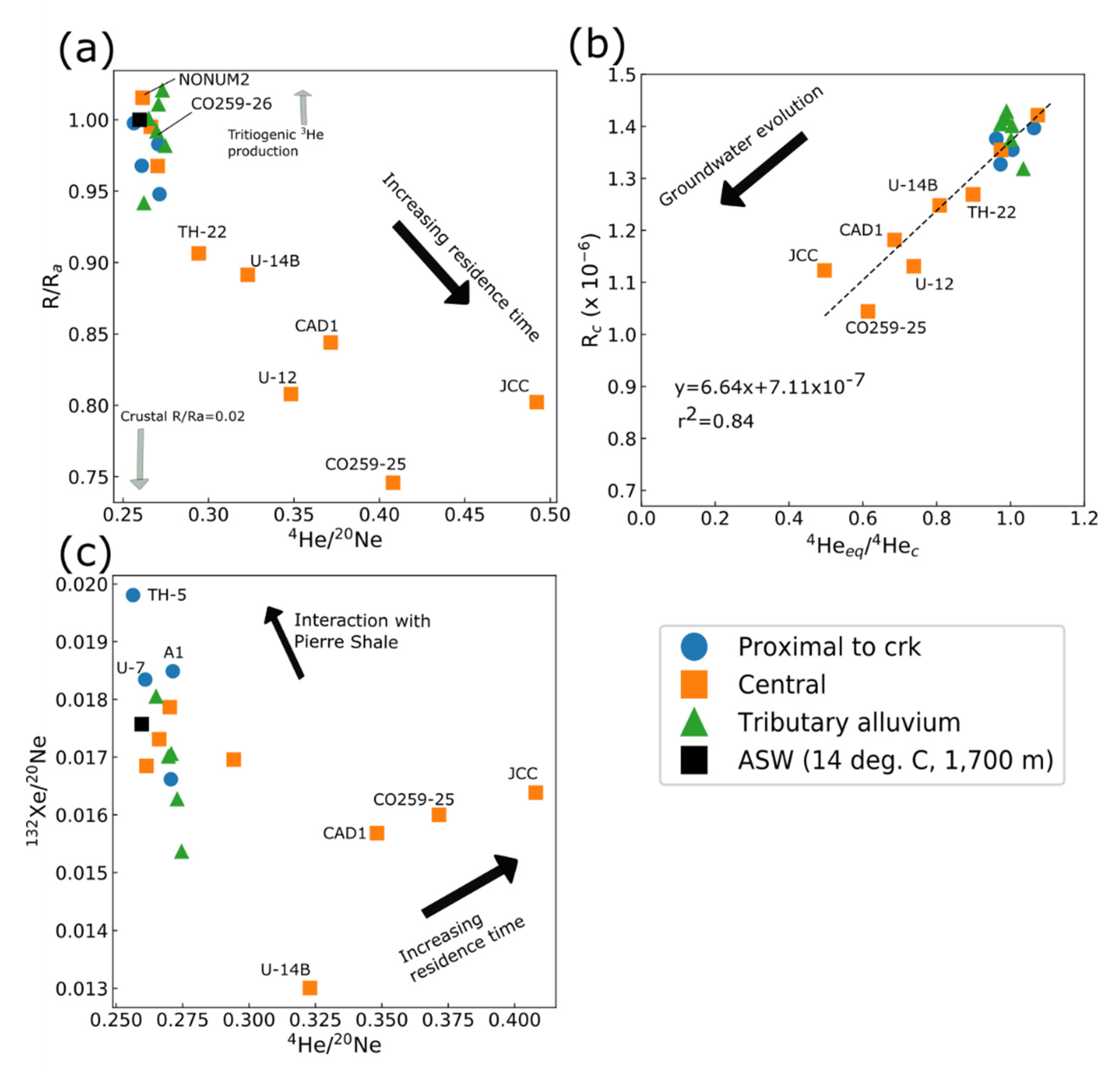
3.6.2. Apparent Ages and Presence of Atmospheric Tracer Gases
3.6.3. Lumped Parameter Models
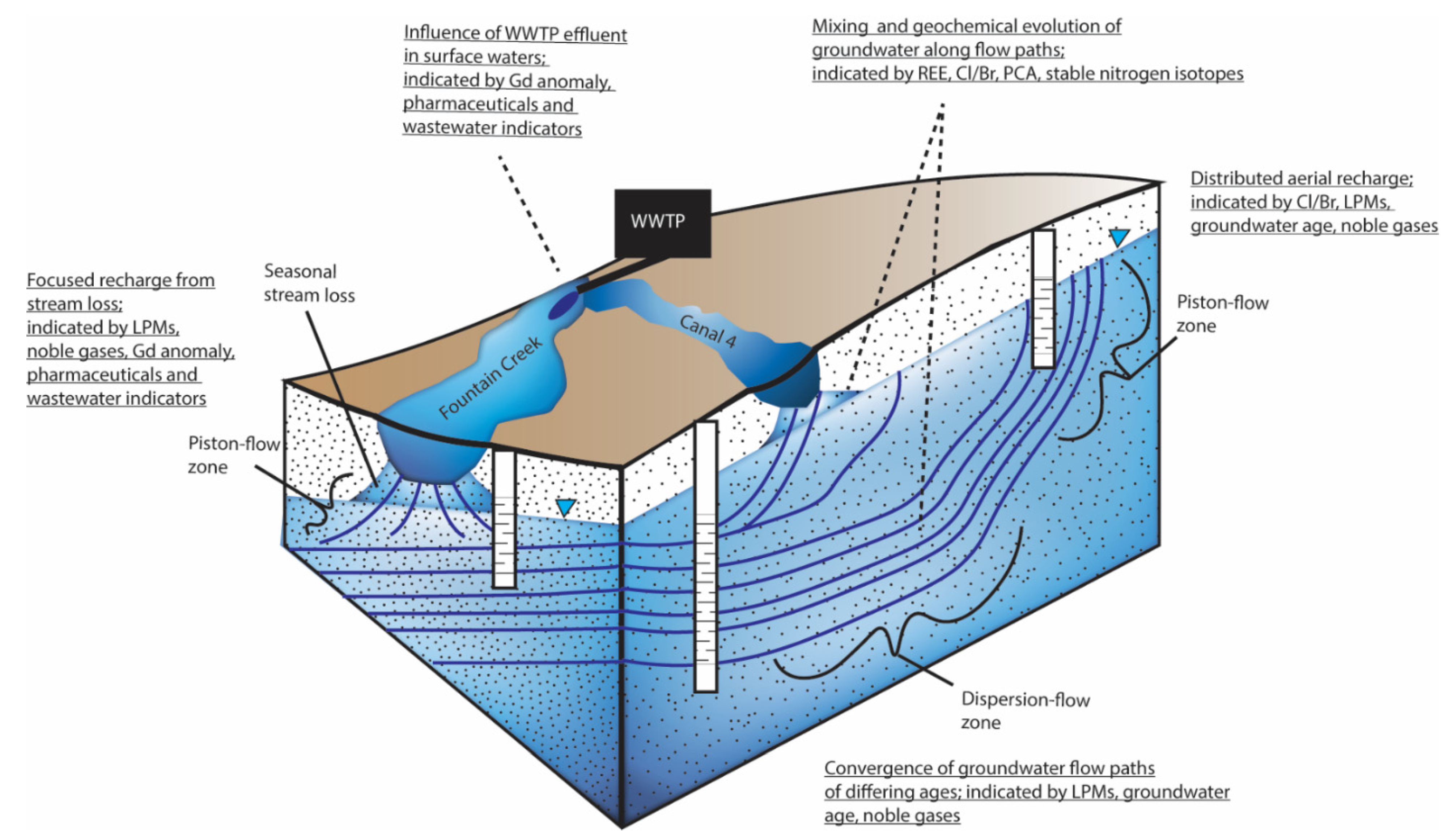
3.7. Conceptual Hydrologic Model
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Böhlke, J.K.; Verstraeten, I.M.; Kraemer, T.F. Effects of surface-water irrigation on sources, fluxes, and residence times of water, nitrate, and uranium in an alluvial aquifer. Appl. Geochem. 2007, 22, 152–174. [Google Scholar] [CrossRef]
- Bai, X.; Lutz, A.; Carroll, R.; Keteles, K.; Dahlin, K.; Murphy, M.; Nguyen, D. Occurrence, distribution, and seasonality of emerging contaminants in urban watersheds. Chemosphere 2018, 200, 133–142. [Google Scholar] [CrossRef]
- Focazio, M.J.; Kolpin, D.W.; Barnes, K.K.; Furlong, E.T.; Meyer, M.T.; Zaugg, S.D.; Barber, L.B.; Thurman, M.E. A national reconnaissance for pharmaceuticals and other organic wastewater contaminants in the United States—II) Untreated drinking water sources. Sci. Total. Environ. 2008, 402, 201–216. [Google Scholar] [CrossRef]
- Elliott, S.M.; Erickson, M.L.; Krall, A.L.; Adams, B.A. Concentrations of pharmaceuticals and other micropollutants in groundwater downgradient from large on-site wastewater discharges. PLoS ONE 2018, 13, e0206004. [Google Scholar] [CrossRef]
- Hu, X.C.; Andrews, D.Q.; Lindstrom, A.B.; Bruton, T.A.; Schaider, L.A.; Grandjean, P.; Lohmann, R.; Carignan, C.C.; Blum, A.; Balan, S.A.; et al. Detection of Poly- and Perfluoroalkyl Substances (PFASs) in U.S. Drinking Water Linked to Industrial Sites, Military Fire Training Areas, and Wastewater Treatment Plants. Environ. Sci. Technol. Lett. 2016, 3, 344–350. [Google Scholar] [CrossRef]
- Weber, A.K.; Barber, L.B.; Leblanc, D.R.; Sunderland, E.M.; Vecitis, C.D. Geochemical and Hydrologic Factors Controlling Subsurface Transport of Poly- and Perfluoroalkyl Substances, Cape Cod, Massachusetts. Environ. Sci. Technol. 2017, 51, 4269–4279. [Google Scholar] [CrossRef] [PubMed]
- Keefe, S.; Barber, L.; Hubbard, L.; Bradley, P.; Roth, D.; Kolpin, D. Behavior of major and trace elements in a transient surface water/groundwater system following removal of a long-term wastewater treatment facility source. Sci. Total. Environ. 2019, 668, 867–880. [Google Scholar] [CrossRef] [PubMed]
- Böhlke, J.K.; Denver, J.M. Combined Use of Groundwater Dating, Chemical, and Isotopic Analyses to Resolve the History and Fate of Nitrate Contamination in Two Agricultural Watersheds, Atlantic Coastal Plain, Maryland. Water Resour. Res. 1995, 31, 2319–2339. [Google Scholar] [CrossRef]
- Bern, C.R.; Walton-Day, K.; Naftz, D.L. Improved enrichment factor calculations through principal component analysis: Examples from soils near breccia pipe uranium mines, Arizona, USA. Environ. Pollut. 2019, 248, 90–100. [Google Scholar] [CrossRef]
- Zhang, X.; Lohmann, R.; Dassuncao, C.; Hu, X.C.; Weber, A.K.; Vecitis, C.D.; Sunderland, E.M. Source Attribution of Poly- and Perfluoroalkyl Substances (PFASs) in Surface Waters from Rhode Island and the New York Metropolitan Area. Environ. Sci. Technol. Lett. 2016, 3, 316–321. [Google Scholar] [CrossRef]
- Jurgens, B.C.; Bexfield, L.M.; Eberts, S.M. A Ternary Age-Mixing Model to Explain Contaminant Occurrence in a Deep Supply Well. Ground Water 2014, 52, 25–39. [Google Scholar] [CrossRef]
- Kulongoski, J.T.; Hilton, D.R. Applications of groundwater helium. In Handbook of Environmental Isotope Geochemistry; Baskaran, M., Ed.; Springer: Berlin, Germany, 2012; pp. 285–304. [Google Scholar] [CrossRef]
- McMahon, P.B.; Lindsey, B.D.; Conlon, M.D.; Hunt, A.G.; Belitz, K.; Jurgens, B.C.; Varela, B.A. Hydrocarbons in Upland Groundwater, Marcellus Shale Region, Northeastern Pennsylvania and Southern New York, U.S.A. Environ. Sci. Technol. 2019, 53, 8027–8035. [Google Scholar] [CrossRef]
- Moeck, C.; Radny, D.; Popp, A.; Brennwald, M.; Stoll, S.; Auckenthaler, A.; Berg, M.; Schirmer, M. Characterization of a managed aquifer recharge system using multiple tracers. Sci. Total. Environ. 2017, 609, 701–714. [Google Scholar] [CrossRef]
- Levy, Z.F.; Fram, M.S.; Faulkner, K.E.; Alpers, C.N.; Soltero, E.M.; Taylor, K.A. Effects of montane watershed development on vulnerability of domestic groundwater supply during drought. J. Hydrol. 2020, 583, 124567. [Google Scholar] [CrossRef]
- Bexfield, L.M.; Toccalino, P.L.; Belitz, K.; Foreman, W.T.; Furlong, E.T. Hormones and Pharmaceuticals in Groundwater Used As a Source of Drinking Water Across the United States. Environ. Sci. Technol. 2019, 53, 2950–2960. [Google Scholar] [CrossRef]
- Arnold, L.R.; Ortiz, R.F.; Brown, C.R.; Watts, K.R. Groundwater and Surface-Water Interaction, Water Quality, and Processes Affecting Loads of Dissolved Solids, Selenium, and Uranium in Fountain Creek, Pueblo County, Colorado, 2012–2014. In U.S. Geological Survey Scientific Investigation Report 2016–5134; U.S. Geological Survey: Reston, VA, USA, 2016; 78p. [Google Scholar] [CrossRef]
- Cain, D.; Edelmann, P. A reconnaissance water-quality appraisal of the Fountain Creek alluvial aquifer between Colorado Springs and Pueblo, Colorado, including trace elements and organic constituents. In U.S. Geological Survey Water-Resources Investigations Report 86-4085; U.S. Geological Survey: Reston, VA, USA, 1986; 45p. [Google Scholar]
- Chafin, D.T. Effects of land use on water quality of the Fountain Creek alluvial aquifer, east-central Colorado. In U.S. Geological Survey Water-Supply Paper 2381-D; U.S. Geological Survey: Reston, VA, USA, 1996; 99p. [Google Scholar]
- Edelmann, P.; Cain, D. Sources of water and nitrogen to the Widefield aquifer, southwestern El Paso County, Colorado. In U.S. Geological Survey Water-Resources Investigations Report 85-4162; U.S. Geological Survey: Reston, VA, USA, 1985; 81p. [Google Scholar]
- Lewis, M.E. Quality of water in the alluvial aquifer and tributary alluvium of the Fountain Creek valley, southwestern El Paso County, Colorado, 1991–1992. In U.S. Geological Survey Water-Resources Investigations Report 94-4118; U.S. Geological Survey: Reston, VA, USA, 1995; 39p. [Google Scholar]
- Mau, D.P.; Stogner, R.W.; Edelmann, P. Characterization of stormflows and wastewater treatment-plant effluent discharges on water quality, suspended sediment, and stream morphology for Fountain and Monument Creek watersheds, Colorado, 1981–2006. In U.S. Geological Survey Scientific Investigations Report 2007–5104; U.S. Geological Survey: Reston, VA, USA, 2007; 76p. [Google Scholar]
- U.S. Environmental Protection Agency. Lifetime Health Advisories and Health Effects Support Documents for Per-fluorooctanoic Acid and Perfluorooctane Sulfonate. Fed. Regist. 2016, 81, 33250–33251. [Google Scholar]
- U.S. Geological Survey. USGS water data for the Nation. In U.S. Geological Survey National Water Information System Database; U.S. Geological Survey: Reston, VA, USA, 2020. [Google Scholar] [CrossRef]
- AECOM. Peterson Air Force Base Conceptual Site Model: Report Prepared for US; Air Force Civil Engineering Center by AECOM: Denver, CO, USA, 2017; 24p. [Google Scholar]
- Topper, R.; Spray, K.L.; Bellis, W.H.; Hamilton, J.L.; Barkmann, P.E. Ground Water Atlas of Colorado: Colorado Geological Survey Special Publications-53. 2003. Available online: https://coloradogeologicalsurvey.org/water/colorado-groundwater-atlas/ (accessed on 1 January 2021).
- Aerostar. Final Preliminary Assessment Report for Perfluorinated Compounds at Peterson Air Force Base El Paso County, Colorado; Air Force Civil Engineering Center, Aerostar SES LLC.: Jacksonville, FL, USA, 2016; 145p. [Google Scholar]
- U.S. Environmental Protection Agency. Facility Registry Service—Geospatial Downloads. 2019. Available online: https://www.epa.gov/frs/geospatial-data-download-service (accessed on 1 April 2019).
- Kuhn, G.; Arnold, L.R. Application of a stream-aquifer model to Monument Creek for development of a method to estimate transit losses for reusable water, El Paso County, Colorado. In U.S. Geological Survey Scientific Investigations Report 2006–5184; U.S. Geological Survey: Reston, VA, USA, 2006; 111p. [Google Scholar]
- U.S. Geological Survey. Transit-Loss Accounting Program for Monument and Fountain Creeks, El Paso and Pueblo Counties, Colorado. 2019. Available online: https://www.usgs.gov/centers/co-water/science/transit-loss-accounting-program-monument-and-fountain-creeks-el-paso-and?qt-science_center_objects=0#qt-science_center_objects (accessed on 1 October 2019).
- Huntington, J.L.; Hegewisch, K.C.; Daudert, B.; Morton, C.G.; Abatzoglou, J.T.; McEvoy, D.J.; Erickson, T. Climate Engine: Cloud Computing and Visualization of Climate and Remote Sensing Data for Advanced Natural Resource Monitoring and Process Understanding. Bull. Am. Meteorol. Soc. 2017, 98, 2397–2410. [Google Scholar] [CrossRef]
- Jenkins, E.D. Ground water in Fountain and Jimmy Camp Valleys, El Paso County, Colorado. In U.S. Geological Survey Water-Supply Paper 1583; U.S. Geological Survey: Reston, VA, USA, 1964; 66p. [Google Scholar]
- U.S. Geological Survey; Variously Dated; National Field Manual for the Collection of Water-Quality Data. U.S. Geological Survey Techniques of Water-Resources Investigations, Book 9, Chaps A1–A10. Available online: http://pubs.water.usgs.gov/twri9A (accessed on 10 November 2019).
- Rounds, S.A. Alkalinity and acid neutralizing capacity (ver. 4.0, September 2012). In U.S. Geological Survey Techniques of Water-Resources Investigations; U.S. Geological Survey: Reston, VA, USA, 2012; Book 9, Chapter 6.6. [Google Scholar] [CrossRef]
- Aeschbach-Hertig, W.; Solomon, D.K. Noble gas thermometry in groundwater hydrology. In The Noble Gases as Geochemical Tracers; Burnard, P., Ed.; Springer: Berlin, Germany, 2013; pp. 81–122. [Google Scholar]
- Fishman, M.J.; Friedman, L.C. (Eds.) Methods for determination of inorganic substances in water and fluvial sediments. In U.S. Geological Survey Techniques of Water-Resources Investigations; U.S. Geological Survey: Reston, VA, USA, 1989; Book 5, Chapter A1; 545p. Available online: http://pubs.usgs.gov/twri/twri5-a1/html/pdf.html (accessed on 27 February 2014).
- Fishman, M.J. (Ed.) Methods of analysis by the U.S. Geological Survey National Water Quality Laborato-ry—Determination of inorganic and organic constituents in water and fluvial sediments. In U.S. Geological Survey OpenFile Report 93–125; U.S. Geological Survey: Reston, VA, USA, 1993; 217p. Available online: http://pubs.usgs.gov/of/1993/0125/report.pdf (accessed on 27 February 2014).
- Garbarino, J.R.; Struzeski, T.M. Methods of analysis by the U.S. Geological Survey National Water Quality Labor-atory—Determination of elements in whole-water digests using inductively coupled plasma-optical emission spectrometry and inductively coupled plasma-mass spectrometry. In U.S. Geological Survey Open-File Report 98–165; U.S. Geological Survey: Reston, VA, USA, 1998; 101p. Available online: http://nwql.usgs.gov/OFR-98-165.shtml (accessed on 27 February 2014).
- Furlong, E.T.; Noriega, M.C.; Kanagy, C.J.; Kanagy, L.K.; Coffey, L.J.; Burkhardt, M.R. Determination of human-use pharmaceuticals in filtered water by direct aqueous injection–high-performance liquid chromatography/tandem mass spec-trometry. In U.S. Geological Survey Techniques and Methods; U.S. Geological Survey: Reston, VA, USA, 2014; Book 5, Chapter B10; 49p. [Google Scholar]
- Coplen, T.B.; Qi, H.; Révész, K.; Casciotti, K.; Hannon, J.E. Determination of the δ15N and δ18O of Nitrate in Water; RSIL lab code 2900, chap. 17 of Stable Isotope-Ratio Methods, Methods of the Reston Stable Isotope Laboratory (Slightly revised from Version 1.0 Released in 2007). In U.S. Geological Survey Techniques and Methods; Révész, K., Coplen, T.B., Eds.; U.S. Geological Survey: Reston, VA, USA, 2012; 35p. Available online: https://pubs.usgs.gov/tm/2006/tm10c17/ (accessed on 1 January 2021).
- Kendall, C.; Doctor, D.H.; Young, M.B. Environmental isotope applications in hydrologic studies. In Treatise on Geochemistry, 2nd ed.; Holland, H.D., Turekian, K.K., Eds.; Elsevier Science: Amsterdam, The Netherlands, 2015; pp. 273–327. [Google Scholar]
- Révész, K.; Coplen, T.B. Determination of the δ(2H/1H) of water: RSIL lab code 1574. Chapter C1, Methods of the Reston Stable Isotope Laboratory. In U.S. Geological Survey Techniques and Methods 10–C1; Révész, K., Coplen, T.B., Eds.; U.S. Geological Survey: Reston, VA, USA, 2008; 27p. [Google Scholar]
- Révész, K.; Coplen, T.B. Determination of the δ(18O/16O) of water: RSIL lab code 489. Chapter C2, Methods of the Reston Stable Isotope Laboratory. In U.S. Geological Survey Techniques and Methods, 10–C2; Révész, K., Coplen, T.B., Eds.; U.S. Geological Survey: Reston, VA, USA, 2008; 28p. [Google Scholar]
- Hunt, A.G. Noble Gas Laboratory’s standard operating procedures for the measurement of dissolved gas in water samples. In U.S. Geological Survey Techniques and Methods; U.S. Geological Survey: Reston, VA, USA, 2015; Book 5, Chapter A11; 22p. [Google Scholar] [CrossRef]
- Busenberg, E.; Plummer, L.N. Dating young groundwater with sulfur hexafluoride: Natural and anthropogenic sources of sulfur hexafluoride. Water Resour. Res. 2000, 36, 3011–3030. [Google Scholar] [CrossRef]
- Mueller, D.K.; Schertz, T.L.; Martin, J.D.; Sandstrom, M.W. Design, analysis, and interpretation of field quali-ty-control data for water-sampling projects. In U.S. Geological Survey Techniques and Methods; U.S. Geological Survey: Reston, VA, USA, 2015; Book 4, Chapter C4; 54p. [Google Scholar] [CrossRef]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater, and Pollution, 2nd ed.; CRC Press: Amsterdam, The Netherlands, 2005; 649p. [Google Scholar]
- Lee, L.; Helsel, D. Baseline models of trace elements in major aquifers of the United States. Appl. Geochem. 2005, 20, 1560–1570. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: http://www.R-project.org/ (accessed on 5 June 2019).
- Reimann, R.C.; Filzmoser, P.; Garrett, R.G.; Dutter, R. Statistical Data Analysis Explained: Applied Environmental Statistics; John Wiley & Sons, Ltd.: Chichester, UK, 2008. [Google Scholar]
- Blondes, M.S.; Engle, M.A.; Geboy, N.J. A practical guide to the use of major elements, trace elements, and isotopes in compositional data analysis: Applications for deep formation brine geochemistry. In Compositional Data Analysis; Martín-Fernández, J.A., Thió-Henestrosa, S., Eds.; Springer: Cham, Switzerland, 2016; pp. 13–29. [Google Scholar]
- Filzmoser, P.; Hron, K.; Reimann, C. Principal component analysis for compositional data with outliers. Environmetrics 2009, 20, 621–632. [Google Scholar] [CrossRef]
- Jurgens, B.C.; Böhlke, J.K.; Eberts, S.M. TracerLPM (Version 1): An Excel® workbook for interpreting groundwater age distributions from environmental tracer data. In U.S. Geological Survey Techniques and Methods Report 4-F3; U.S. Geological Survey: Reston, VA, USA, 2012; 60p. [Google Scholar]
- Solomon, D.K.; Cook, P.G. 3H and 3He. In Environmental Tracers in Subsurface Hydrology; Cook, P.G., Herczeg, A.L., Eds.; Springer Science: New York, NY, USA, 2000; pp. 397–424. [Google Scholar]
- Chambers, L.; Gooddy, D.; Binley, A. Use and application of CFC-11, CFC-12, CFC-113 and SF6 as environmental tracers of groundwater residence time: A review. Geosci. Front. 2019, 10, 1643–1652. [Google Scholar] [CrossRef]
- Cartwright, I.; Cendón, D.; Currell, M.; Meredith, K. A review of radioactive isotopes and other residence time tracers in understanding groundwater recharge: Possibilities, challenges, and limitations. J. Hydrol. 2017, 555, 797–811. [Google Scholar] [CrossRef]
- Weiss, R.F. The solubility of nitrogen, oxygen and argon in water and seawater. Deep Sea Res. 1970, 17, 721–735. [Google Scholar] [CrossRef]
- Weiss, R. Carbon dioxide in water and seawater: The solubility of a non-ideal gas. Mar. Chem. 1974, 2, 203–215. [Google Scholar] [CrossRef]
- Benson, B.B.; Krause, D. The concentration and isotopic fractionation of gases dissolved in freshwater in equilibrium with the atmosphere. 1. Oxygen. Limnol. Oceanogr. 1980, 25, 662–671. [Google Scholar] [CrossRef]
- Benson, B.B.; Krause, D. The concentration and isotopic fractionation of oxygen dissolved in freshwater and seawater in equilibrium with the atmosphere1. Limnol. Oceanogr. 1984, 29, 620–632. [Google Scholar] [CrossRef]
- Peeters, F.; Beyerle, U.; Aeschbach-Hertig, W.; Holocher, J.; Brennwald, M.S.; Kipfer, R. Improving noble gas based paleoclimate reconstruction and groundwater dating using 20Ne/22Ne ratios. Geochim. Cosmochim. Acta 2003, 67, 587–600. [Google Scholar] [CrossRef]
- Manning, A.H.; Solomon, D. Using noble gases to investigate mountain-front recharge. J. Hydrol. 2003, 275, 194–207. [Google Scholar] [CrossRef]
- Manning, A.H. Mountain-block recharge, present and past, in the eastern Española Basin, New Mexico, USA. Hydrogeol. J. 2011, 19, 379–397. [Google Scholar] [CrossRef]
- Lindsey, B.D.; Jurgens, B.C.; Belitz, K. Tritium as an indicator of modern, mixed, and premodern groundwater age. Sci. Investig. Rep. 2019, 18. [Google Scholar] [CrossRef]
- McCallum, J.L.; Cook, P.G.; Simmons, C.T. Limitations of the Use of Environmental Tracers to Infer Groundwater Age. Ground Water 2015, 53, 56–70. [Google Scholar] [CrossRef]
- Cook, P.G.; Böhlke, J.K. Determining timescales for groundwater flow and transport. In Environmental Tracers in Subsurface Hydrology; Cook, P.G., Herczeg, A.L., Eds.; Springer Science: New York, NY, USA, 2000; pp. 1–30. [Google Scholar]
- Suckow, A. The age of groundwater—Definitions, models and why we do not need this term. Appl. Geochem. 2014, 50, 222–230. [Google Scholar] [CrossRef]
- Leray, S.; Engdahl, N.B.; Massoudieh, A.; Bresciani, E.; McCallum, J. Residence time distributions for hydrologic systems: Mechanistic foundations and steady-state analytical solutions. J. Hydrol. 2016, 543, 67–87. [Google Scholar] [CrossRef]
- Jasechko, S. Partitioning young and old groundwater with geochemical tracers. Chem. Geol. 2016, 427, 35–42. [Google Scholar] [CrossRef]
- Jurgens, B.C. Data for Tritium deposition in precipitation in the United States, 1953–2012. In U.S. Geological Survey Data Release; U.S. Geological Survey: Reston, VA, USA, 2018. [Google Scholar] [CrossRef]
- Jasechko, S.; Taylor, R.G. Intensive rainfall recharges tropical groundwaters. Environ. Res. Lett. 2015, 10, 124015. [Google Scholar] [CrossRef]
- Michel, R.L.; Jurgens, B.C.; Young, M.B. Tritium deposition in precipitation in the United States, 1953–2012. Sci. Investig. Rep. 2018, 11. [Google Scholar] [CrossRef]
- Piper, A.M. A graphic procedure in the geochemical interpretation of water-analyses. Trans. Am. Geophys. Union 1944, 25, 914–928. [Google Scholar] [CrossRef]
- Alcalá, F.J.; Custodio, E. Using the Cl/Br ratio as a tracer to identify the origin of salinity in aquifers in Spain and Portugal. J. Hydrol. 2008, 359, 189–207. [Google Scholar] [CrossRef]
- Davis, S.N.; Whittemore, D.O.; Fabryka-Martin, J. Uses of Chloride/Bromide Ratios in Studies of Potable Water. Ground Water 1998, 36, 338–350. [Google Scholar] [CrossRef]
- McArthur, J.; Sikdar, P.; Hoque, M.; Ghosal, U. Waste-water impacts on groundwater: Cl/Br ratios and implications for arsenic pollution of groundwater in the Bengal Basin and Red River Basin, Vietnam. Sci. Total. Environ. 2012, 437, 390–402. [Google Scholar] [CrossRef] [PubMed]
- Jurgens, B.C.; McMahon, P.B.; Chapelle, F.H.; Eberts, S.M. An Excel® workbook for identifying redox processes in ground water. In U.S. Geological Survey Open-File Report 2009–1004; U.S. Geological Survey: Reston, VA, USA, 2009; 8p. [Google Scholar]
- Anderson, L.; Berkelhammer, M.; Mast, M.A. Isotopes in North American Rocky Mountain Snowpack 1993–2014. Quat. Sci. Rev. 2016, 131, 262–273. [Google Scholar] [CrossRef]
- Bowen, G.J. The Online Isotopes in Precipitation Calculator, Version 3.1. 2020. Available online: http://www.waterisotopes.org (accessed on 2 July 2020).
- Solder, J.E.; Beisner, K.R. Critical evaluation of stable isotope mixing end-members for estimating groundwater recharge sources: Case study from the South Rim of the Grand Canyon, Arizona, USA. Hydrogeol. J. 2020, 28, 1–17. [Google Scholar] [CrossRef]
- Gammons, C.H.; Poulson, S.R.; Pellicori, D.A.; Reed, P.J.; Roesler, A.J.; Petrescu, E.M. The hydrogen and oxygen isotopic composition of precipitation, evaporated mine water, and river water in Montana, USA. J. Hydrol. 2006, 328, 319–330. [Google Scholar] [CrossRef]
- McMahon, P.B.; Böhlke, J.K. Regional Patterns in the Isotopic Composition of Natural and Anthropogenic Nitrate in Groundwater, High Plains, U.S.A. Environ. Sci. Technol. 2006, 40, 2965–2970. [Google Scholar] [CrossRef] [PubMed]
- Kendall, C.; Aravena. Nitrate isotopes in groundwater systems. In Environmental Tracers in Subsurface Hydrology; Cook, P.G., Herczeg, A.L., Eds.; Springer Science: New York, NY, USA, 2000; pp. 261–297. [Google Scholar]
- Leybourne, M.I.; Johannesson, K.H. Rare earth elements (REE) and yttrium in stream waters, stream sediments, and Fe–Mn oxyhydroxides: Fractionation, speciation, and controls over REE+Y patterns in the surface environment. Geochim. Cosmochim. Acta 2008, 72, 5962–5983. [Google Scholar] [CrossRef]
- Duvert, C.; Cendón, D.I.; Raiber, M.; Seidel, J.-L.; Cox, M.E. Seasonal and spatial variations in rare earth elements to identify inter-aquifer linkages and recharge processes in an Australian catchment. Chem. Geol. 2015, 396, 83–97. [Google Scholar] [CrossRef]
- Göb, S.; Loges, A.; Nolde, N.; Bau, M.; Jacob, D.E.; Markl, G. Major and trace element compositions (including REE) of mineral, thermal, mine and surface waters in SW Germany and implications for water–rock interaction. Appl. Geochem. 2013, 33, 127–152. [Google Scholar] [CrossRef]
- Rabiet, M.; Togola, A.; Brissaud, F.; Seidel, J.-L.; Budzinski, H.; Elbaz-Poulichet, F. Consequences of Treated Water Recycling as Regards Pharmaceuticals and Drugs in Surface and Ground Waters of a Medium-sized Mediterranean Catchment. Environ. Sci. Technol. 2006, 40, 5282–5288. [Google Scholar] [CrossRef]
- Barber, L.B.; Paschke, S.S.; Battaglin, W.A.; Douville, C.; Fitzgerald, K.C.; Keefe, S.H.; Roth, D.A.; Vajda, A.M. Effects of an Extreme Flood on Trace Elements in River Water—From Urban Stream to Major River Basin. Environ. Sci. Technol. 2017, 51, 10344–10356. [Google Scholar] [CrossRef]
- Verplanck, P.L.; Furlong, E.T.; Gray, J.L.; Phillips, P.J.; Wolf, R.E.; Esposito, K. Evaluating the Behavior of Gadolinium and Other Rare Earth Elements through Large Metropolitan Sewage Treatment Plants. Environ. Sci. Technol. 2010, 44, 3876–3882. [Google Scholar] [CrossRef]
- Noack, C.W.; Dzombak, D.A.; Karamalidis, A.K. Rare Earth Element Distributions and Trends in Natural Waters with a Focus on Groundwater. Environ. Sci. Technol. 2014, 48, 4317–4326. [Google Scholar] [CrossRef]
- Gromet, L.; Haskin, L.A.; Korotev, R.L.; Dymek, R.F. The “North American shale composite”: Its compilation, major and trace element characteristics. Geochim. Cosmochim. Acta 1984, 48, 2469–2482. [Google Scholar] [CrossRef]
- Stolpe, B.; Guo, L.; Shiller, A.M. Binding and transport of rare earth elements by organic and iron-rich nanocolloids in Alaskan rivers, as revealed by field-flow fractionation and ICP-MS. Geochim. Cosmochim. Acta 2013, 106, 446–462. [Google Scholar] [CrossRef]
- Barnes, K.K.; Kolpin, D.W.; Furlong, E.T.; Zaugg, S.D.; Meyer, M.T.; Barber, L.B. A national reconnaissance of pharmaceuticals and other organic wastewater contaminants in the United States—I) Groundwater. Sci. Total. Environ. 2008, 402, 192–200. [Google Scholar] [CrossRef]
- Schaider, L.A.; Rodgers, K.M.; Rudel, R.A. Review of Organic Wastewater Compound Concentrations and Removal in Onsite Wastewater Treatment Systems. Environ. Sci. Technol. 2017, 51, 7304–7317. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency. Reassessments of Two Exemptions from the Requirement of a Tolerance for Hexamethylenetetramine (HMTA; CAS Reg. No. 100-97-0). 2006. Available online: https://www.epa.gov/sites/production/files/2015-04/documents/hexamethyl.pdf (accessed on 19 March 2020).
- National Center for Biotechnology Information. PubChem Database, Methenamine, CID=4101. 2020. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Methenamine (accessed on 2 July 2020).
- Gardner, P.M.; Heilweil, V.M. A multiple-tracer approach to understanding regional groundwater flow in the Snake Valley area of the eastern Great Basin, USA. Appl. Geochem. 2014, 45, 33–49. [Google Scholar] [CrossRef]
- Kipfer, R.; Aeschbach-Hertig, W.; Peeters, F.; Stute, M. Noble Gases in Lakes and Ground Waters. Rev. Miner. Geochem. 2002, 47, 615–700. [Google Scholar] [CrossRef]
- Kulongoski, J.T.; Hilton, D.R.; Cresswell, R.G.; Hostetler, S.; Jacobson, G. Helium-4 characteristics of groundwaters from Central Australia: Comparative chronology with chlorine-36 and carbon-14 dating techniques. J. Hydrol. 2008, 348, 176–194. [Google Scholar] [CrossRef]
- Zinn, B.A.; Konikow, L.F. Potential effects of regional pumpage on groundwater age distribution. Water Resour. Res. 2007, 43, 06418. [Google Scholar] [CrossRef]
- Porcelli, D.; Ballentine, C.J.; Wieler, R. An Overview of Noble Gas Geochemistry and Cosmochemistry. Rev. Miner. Geochem. 2002, 47, 1–19. [Google Scholar] [CrossRef]
- Mahara, Y.; Ohta, T.; Morikawa, N.; Nakano, T.; Tokumasu, M.; Hukutani, S.; Tokunaga, T.; Igarashi, T. Effects of terrigenic He components on tritium–helium dating: A case study of shallow groundwater in the Saijo Basin. Appl. Geochem. 2014, 50, 142–149. [Google Scholar] [CrossRef]
- Visser, A.; Schaap, J.D.; Broers, H.P.; Bierkens, M.F. Degassing of 3H/3He, CFCs and SF6 by denitrification: Measurements and two-phase transport simulations. J. Contam. Hydrol. 2009, 103, 206–218. [Google Scholar] [CrossRef]
- Newman, C.P. Environmental-tracer modeling to support hydrogeochemical evaluation of the Fountain Creek alluvial aquifer, El Paso County, Colorado. In U.S. Geological Survey Data Release; U.S. Geological Survey: Reston, VA, USA, 2020. [Google Scholar] [CrossRef]
- Koh, D.-C.; Plummer, L.N.; Busenberg, E.; Kim, Y. Evidence for terrigenic SF6 in groundwater from basaltic aquifers, Jeju Island, Korea: Implications for groundwater dating. J. Hydrol. 2007, 339, 93–104. [Google Scholar] [CrossRef]
- Von Rohden, C.; Kreuzer, A.; Chen, Z.; Aeschbach-Hertig, W. Accumulation of natural SF6in the sedimentary aquifers of the North China Plain as a restriction on groundwater dating. Isot. Environ. Health Stud. 2010, 46, 279–290. [Google Scholar] [CrossRef]
- Santella, N.; Ho, D.T.; Schlosser, P.; Stute, M. Widespread elevated atmospheric SF6 mixing ratios in the Northeastern United States: Implications for groundwater dating. J. Hydrol. 2008, 349, 139–146. [Google Scholar] [CrossRef]
- Friedrich, R.; Vero, G.; von Rohden, C.; Lessmann, B.; Kipfer, R.; Aeschbach-Hertig, W. Factors controlling terrigenic SF6 in young groundwater of the Odenwald region (Germany). Appl. Geochem. 2013, 33, 318–329. [Google Scholar] [CrossRef]
- Cey, B.D.; Hudson, G.B.; Moran, J.E.; Scanlon, B.R. Evaluation of Noble Gas Recharge Temperatures in a Shallow Unconfined Aquifer. Ground Water 2009, 47, 646–659. [Google Scholar] [CrossRef] [PubMed]
- Manning, A.H.; Clark, J.F.; Diaz, S.H.; Rademacher, L.K.; Earman, S.; Plummer, L.N. Evolution of groundwater age in a mountain watershed over a period of thirteen years. J. Hydrol. 2012, 460-461, 13–28. [Google Scholar] [CrossRef]
- McMahon, P.B.; Plummer, L.N.; Böhlke, J.K.; Shapiro, S.D.; Hinkle, S.R. A comparison of recharge rates in aquifers of the United States based on groundwater-age data. Hydrogeol. J. 2011, 19, 779–800. [Google Scholar] [CrossRef]
- McCallum, J.L.; Dogramaci, S.; Bai, A.; Cook, P.G.; Engdahl, N.B.; Simmons, C.T.; Skrzypek, G.; Grierson, P.F. Assessing Temporal Changes in Groundwater Recharge Using Spatial Variations in Groundwater Ages. Water Resour. Res. 2020, 56. [Google Scholar] [CrossRef]
- Kaown, D.; Koh, D.-C.; Solomon, D.K.; Yoon, Y.-Y.; Yang, J.; Lee, K.-K. Delineation of recharge patterns and contaminant transport using 3H–3He in a shallow aquifer contaminated by chlorinated solvents in South Korea. Hydrogeol. J. 2014, 22, 1041–1054. [Google Scholar] [CrossRef]
- Colorado Department of Public Health and the Environment. Evaluation of Tetrachloroethylene Vapor Intrusion into Buildings Located above a Contaminated Aquifer, Schlage Lock Company, Security, El Paso County, Colo-Rado, Epa Facility ID: COD082657420. 2006; 33p. Available online: https://www.atsdr.cdc.gov/HAC/pha/SchlageLockCompany/SchlageLockCompanyHC113006.pdf (accessed on 25 March 2020).
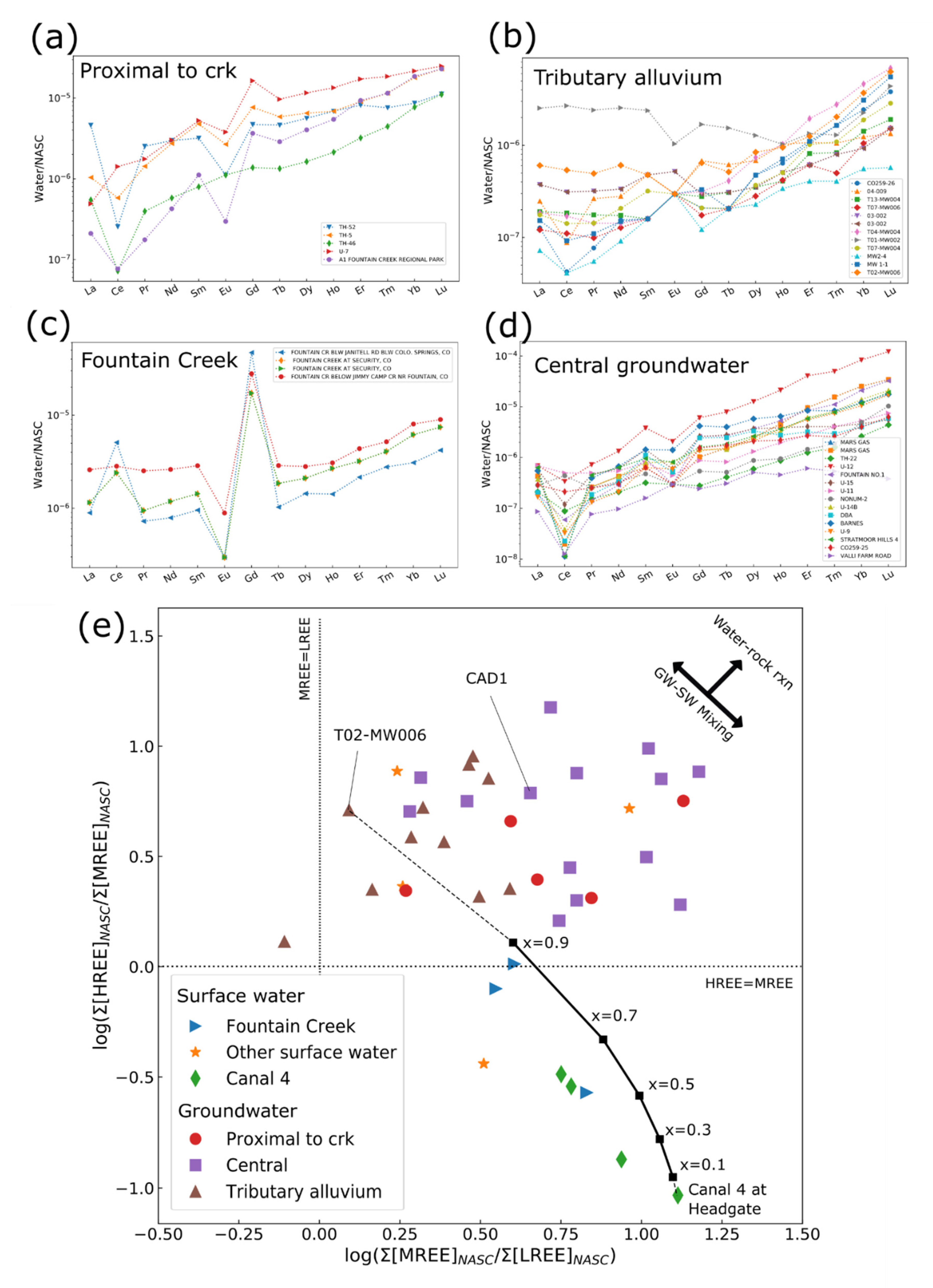
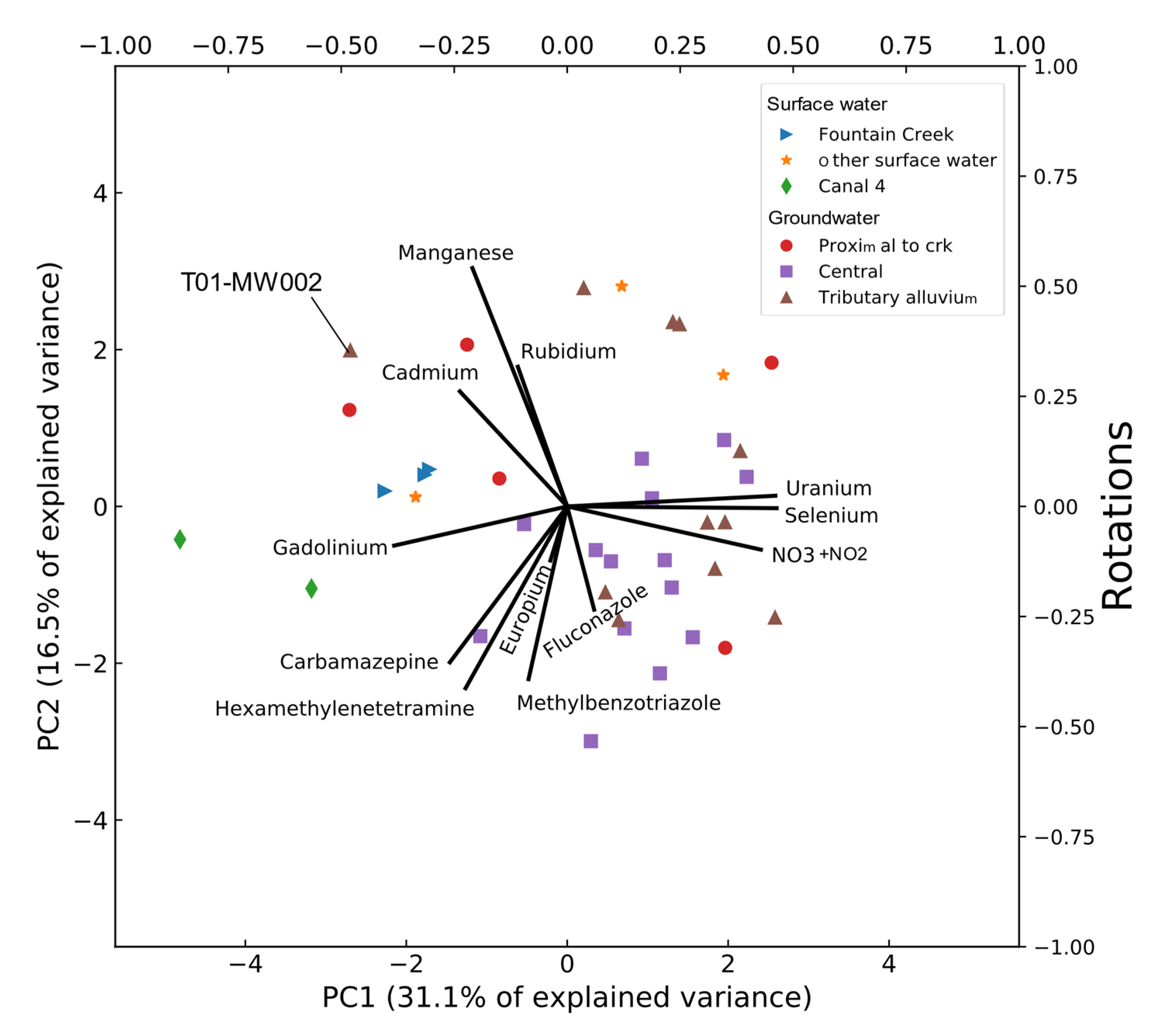
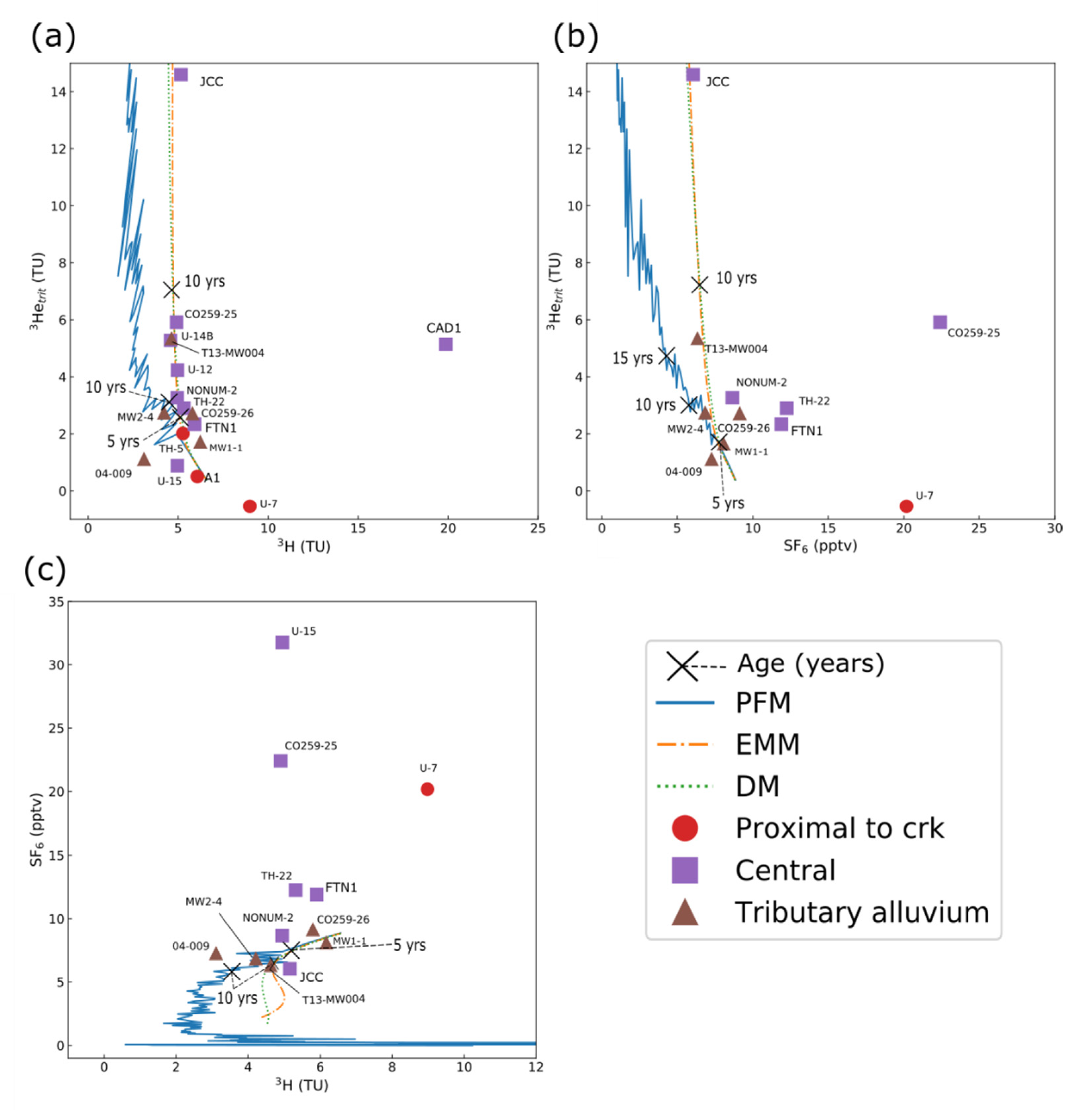
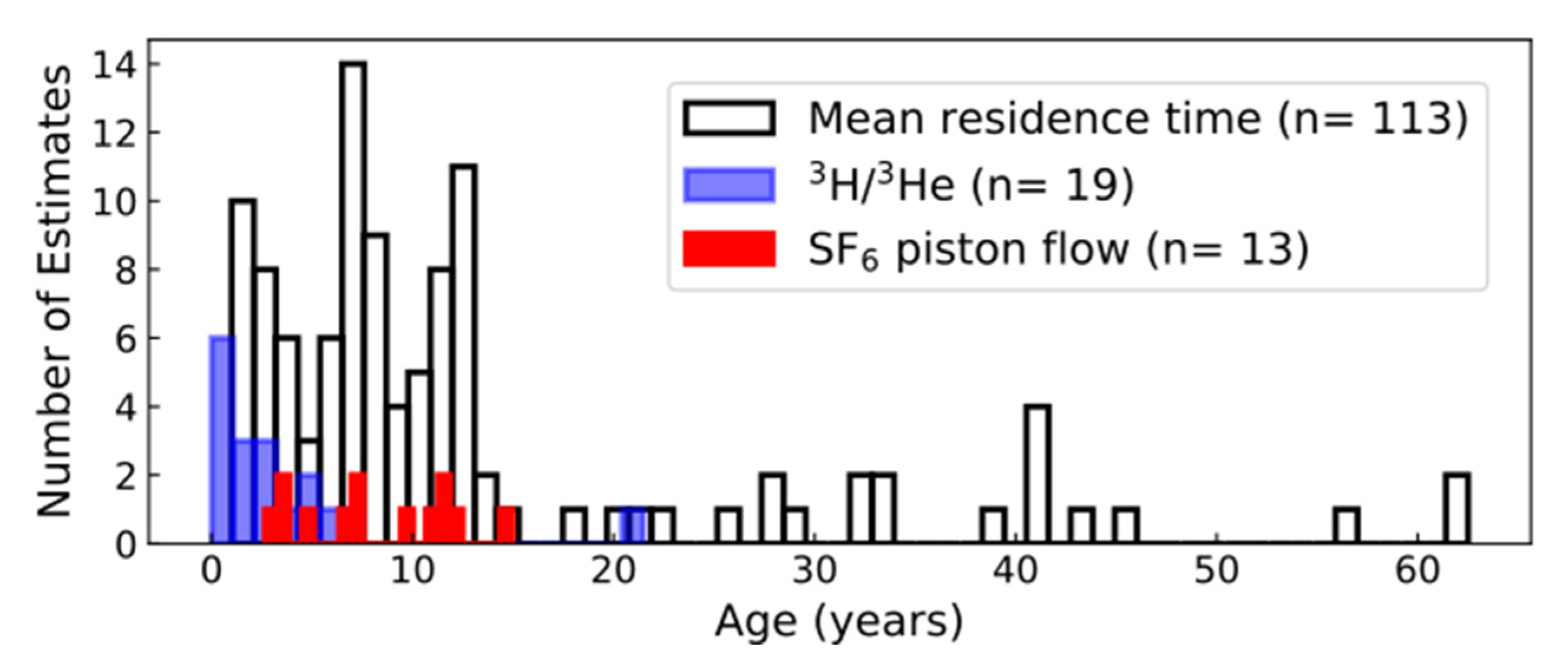
| Compound Name | Percent Detections in Groundwater Samples | Percent Detections in Surface-Water Samples | Occurrence in Groundwater Downgradient from Wastewater Effluent in Minnesota, USA 1 | Occurrence in Surface Water near Denver, Colorado, USA 2 |
|---|---|---|---|---|
| Methylbenzotriazole | 70% | 92% | X | |
| Carbamazepine | 57% | 83% | X | X |
| Sulfamethoxazole | 13% | 83% | X | X |
| Fluconazole | 40% | 83% | X | |
| Lidocaine | 20% | 83% | X | X |
| Hexamethylenetetramine 3 | 7% | 92% | ||
| Bupropion | 13% | 75% | X | X 4 |
| Gabapentin | 7% | 83% | X | |
| Tramadol | 10% | 75% | X | X |
| Meprobamate | 7% | 83% | X | X |
| Metformin | 3% | 33% | X | X |
| USGS Site ID | Site Alias | R/Ra | 3H (TU) | 3H-3He Age (Years) 1 | Fpost-1953 | SF6 Concentration (fmol/kg) | SF6 Concentration (pptv) 1 | SF6 Piston-Flow Age (Years) 2,3 |
|---|---|---|---|---|---|---|---|---|
| 384824104405101 | 03-002 | 1.011 | 6.79 | 3.3 | 0.8 | 2.56 | 8.03 | 5.4 |
| 384710104431201 | 04-009 | 0.942 | 3.11 | 0 | 0.36 | 2.7 | 7.26 | 7.9 |
| 384233104425801 | A1 | 0.948 | 6.07 | 0 | 0.72 | 35.4 | 110.4 | MF |
| 384718104463701 | 3DAA | -- | 8.95 | -- | 1.06 | 28.08 | 90.79 | MF |
| 384509104435901 | CO259-25 | 0.746 | 4.91 | 10.8 | 0.58 | 10.41 | 22.41 | MF |
| 384719104444701 | CO259-26 | 0.998 | 5.8 | 1.2 | 0.68 | 2.74 | 9.13 | 1.9 |
| 384437104422601 | DBA | -- | 5.8 | -- | 0.68 | -- | -- | -- |
| 384112104421301 | FTN1 | 0.995 | 5.91 | 1.1 | 0.7 | 3.82 | 11.9 | MF |
| 384639104461401 | BAC1 | -- | 8.49 | -- | 1.01 | 83.07 | 196.38 | MF |
| 384949104424501 | MW1-1 | 1.001 | 6.18 | 4.6 | 0.73 | 2.71 | 8.13 | 4.9 |
| 384848104413901 | MW2-4 | 1.021 | 4.21 | 4.9 | 0.49 | 2.28 | 6.84 | 9.9 |
| 384408104424701 | NONUM-2 | 1.015 | 4.95 | 2.7 | 0.58 | 4.68 | 8.65 | 2.9 |
| 384617104455901 | CAD1 | 0.844 | 19.9 | 3.2 | 2.38 | 369.71 | 714.58 | MF |
| 384929104431101 | T01-MW002 | -- | 8.33 | -- | 0.99 | 3.54 | 7.88 | 5.9 |
| 384956104422801 | T02-MW006 | -- | 4.58 | -- | 0.54 | -- | -- | -- |
| 384917104422701 | T04-MW004 | -- | 4.94 | -- | 0.58 | 2.31 | 5.58 | 14.4 |
| 384818104415701 | T07-MW004 | -- | 4.22 | -- | 0.49 | 3.48 | 7.65 | 6.9 |
| 384758104422301 | T07-MW006 | -- | 4.31 | -- | 0.5 | 2.97 | 6.81 | 9.9 |
| 384732104430901 | T13-MW004 | 0.982 | 4.62 | 3.2 | 0.54 | 2.57 | 6.33 | 11.4 |
| 384648104454501 | TH-22 | 0.895 | 5.32 | 1.1 | 0.63 | 3.69 | 12.25 | MF |
| 384540104453601 | TH-46 | -- | 4.79 | -- | 0.56 | 219.2 | 616.75 | MF |
| 384503104451601 | TH-5 | 0.998 | 5.27 | 0.6 | 0.62 | 25.28 | 62.35 | MF |
| 384636104465401 | TH-52 | 0.97 | 6.48 | 0.7 | 0.77 | 13.57 | 51.94 | MF |
| 384534104450302 | U-11 | -- | 4.52 | -- | 0.53 | 3.58 | 8.61 | 3.4 |
| 384513104445302 | U-12 | 0.808 | 4.96 | 5.6 | 0.58 | 33.84 | 72.45 | MF |
| 384433104440701 | U-14B | 0.891 | 4.57 | 6.9 | 0.54 | 98.94 | 248.12 | MF |
| 384420104432601 | U-15 | 0.968 | 4.95 | 0.6 | 0.58 | 8.93 | 31.76 | MF |
| 384652104465101 | U-7 | 0.968 | 8.98 | 0 | 1.07 | 7.78 | 20.17 | MF |
| 384604104451502 | U-9 | -- | 6.66 | -- | 0.79 | 3.54 | 8.46 | 3.9 |
| 384217104402901 | JCC | 0.802 | 5.16 | 21.5 | 0.61 | 2.66 | 6.05 | 12.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This is an open access article, free of all copyright, and may be freely reproduced, distributed, transmitted, modified, built upon, or otherwise used by anyone for any lawful purpose. The work is made available under the Creative Commons CC0 public domain dedication. (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Newman, C.P.; Paschke, S.S.; Keith, G. Natural and Anthropogenic Geochemical Tracers to Investigate Residence Times and Groundwater–Surface-Water Interactions in an Urban Alluvial Aquifer. Water 2021, 13, 871. https://doi.org/10.3390/w13060871
Newman CP, Paschke SS, Keith G. Natural and Anthropogenic Geochemical Tracers to Investigate Residence Times and Groundwater–Surface-Water Interactions in an Urban Alluvial Aquifer. Water. 2021; 13(6):871. https://doi.org/10.3390/w13060871
Chicago/Turabian StyleNewman, Connor P., Suzanne S. Paschke, and Gabrielle Keith. 2021. "Natural and Anthropogenic Geochemical Tracers to Investigate Residence Times and Groundwater–Surface-Water Interactions in an Urban Alluvial Aquifer" Water 13, no. 6: 871. https://doi.org/10.3390/w13060871
APA StyleNewman, C. P., Paschke, S. S., & Keith, G. (2021). Natural and Anthropogenic Geochemical Tracers to Investigate Residence Times and Groundwater–Surface-Water Interactions in an Urban Alluvial Aquifer. Water, 13(6), 871. https://doi.org/10.3390/w13060871






