Removal of Microplastics from Waters through Agglomeration-Fixation Using Organosilanes—Effects of Polymer Types, Water Composition and Temperature
Abstract
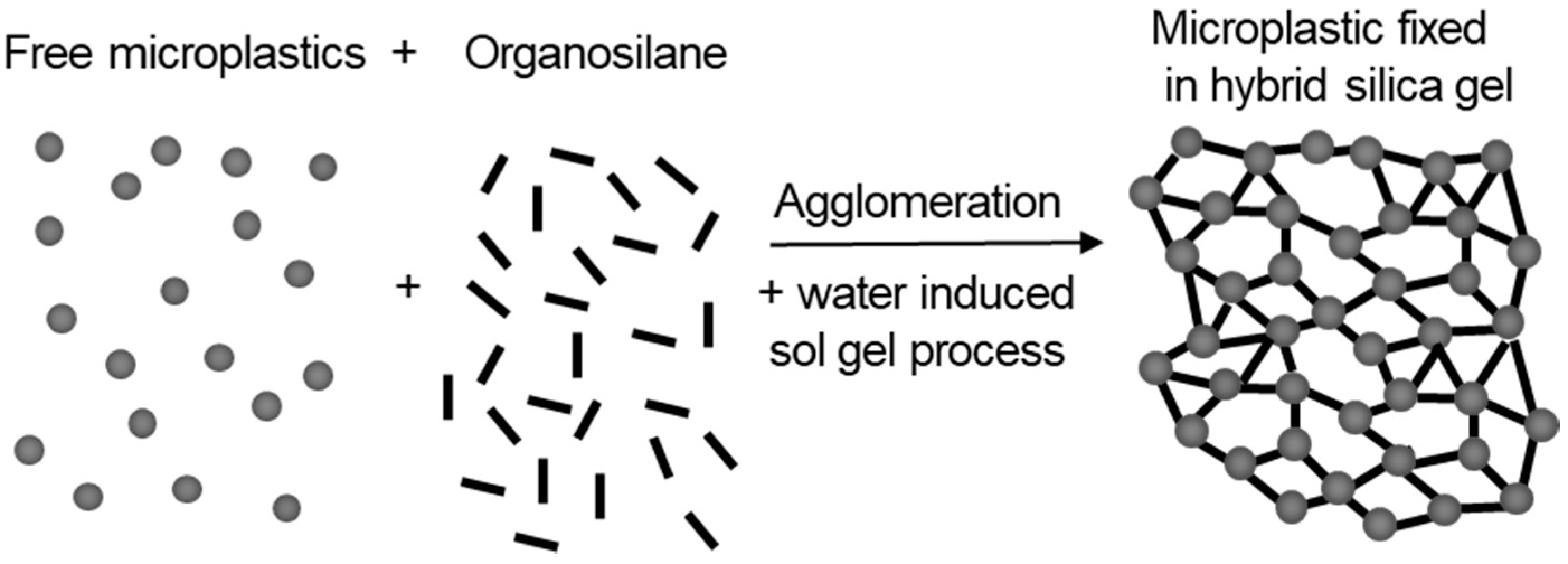
1. Introduction
2. Materials and Methods
2.1. Used Microplastics and Chemicals
2.2. Different Water Types and Temperatures
2.3. Determination of Removal Efficiency
2.4. Determination of the Organosilane Residues Dissolved in Water
2.5. Additional Analytics
3. Results and Discussion
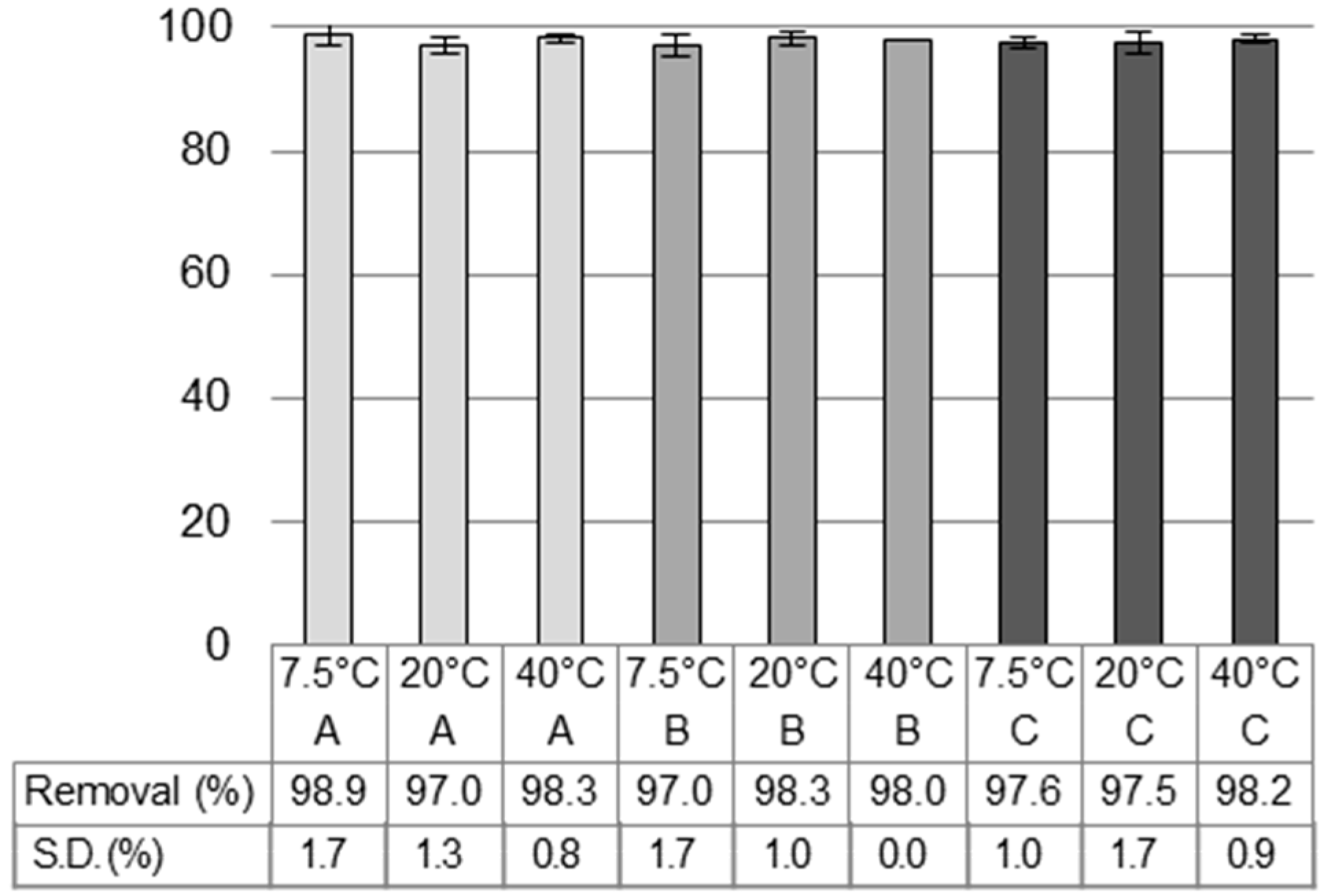
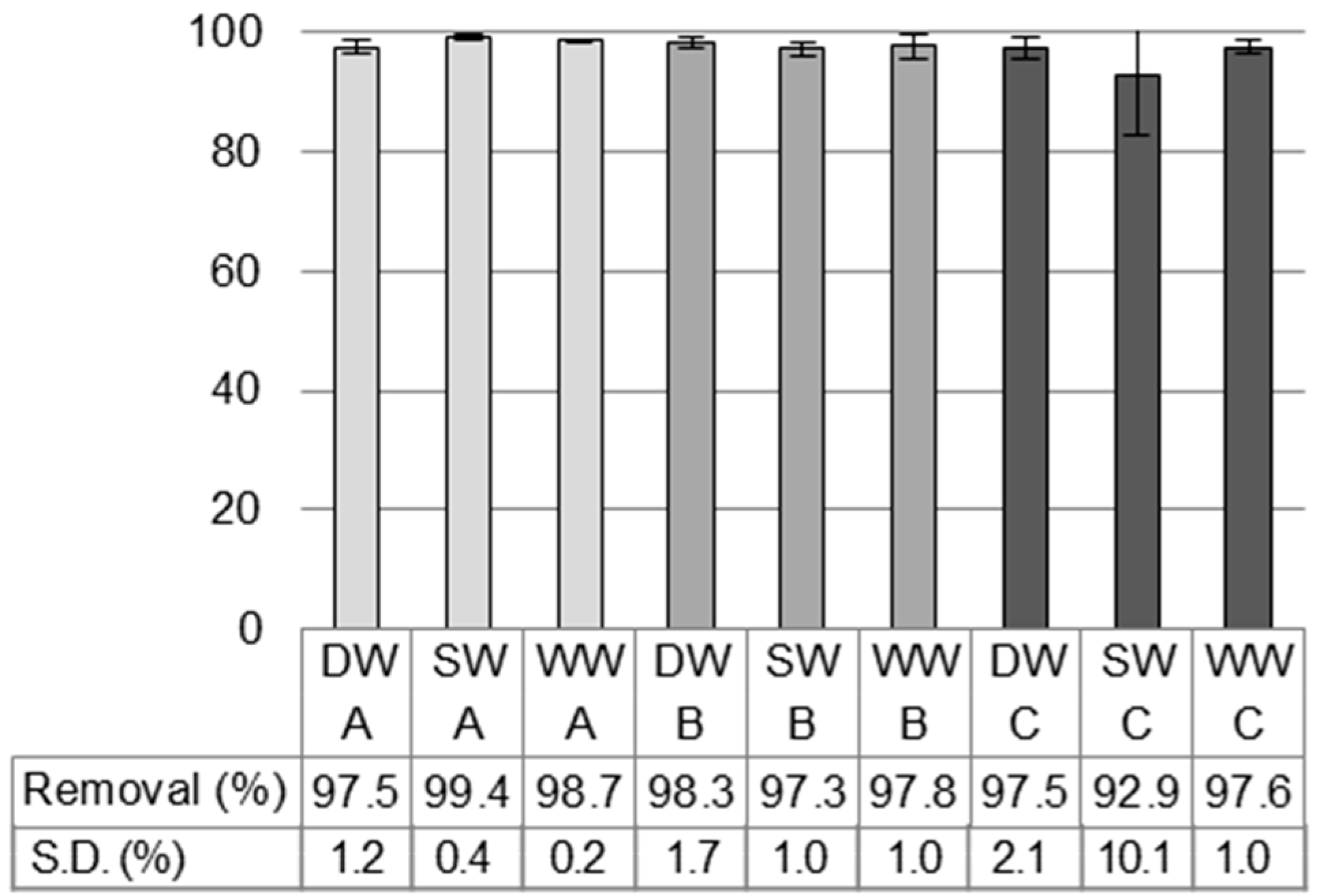
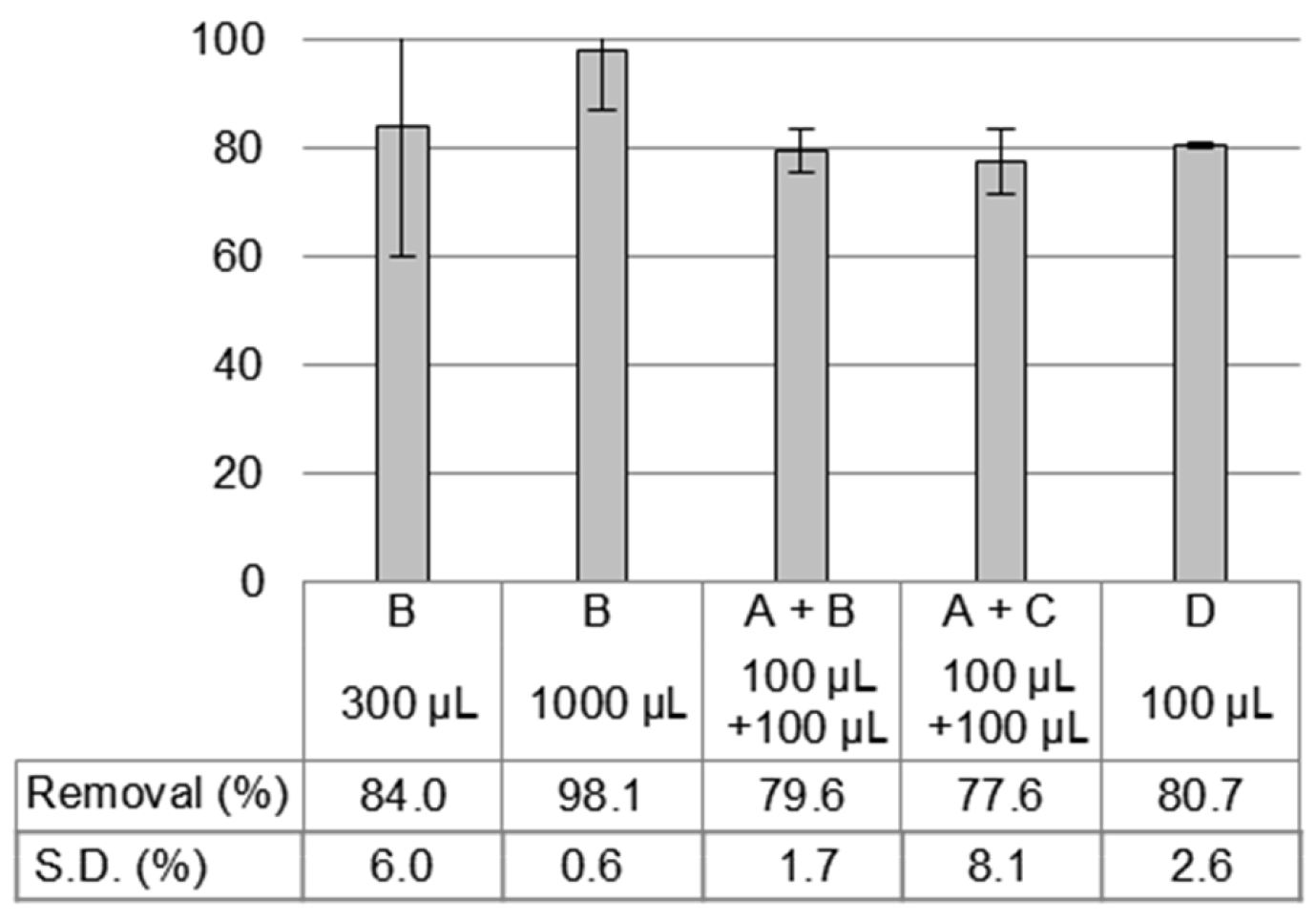
3.1. Different Water Types and Temperatures
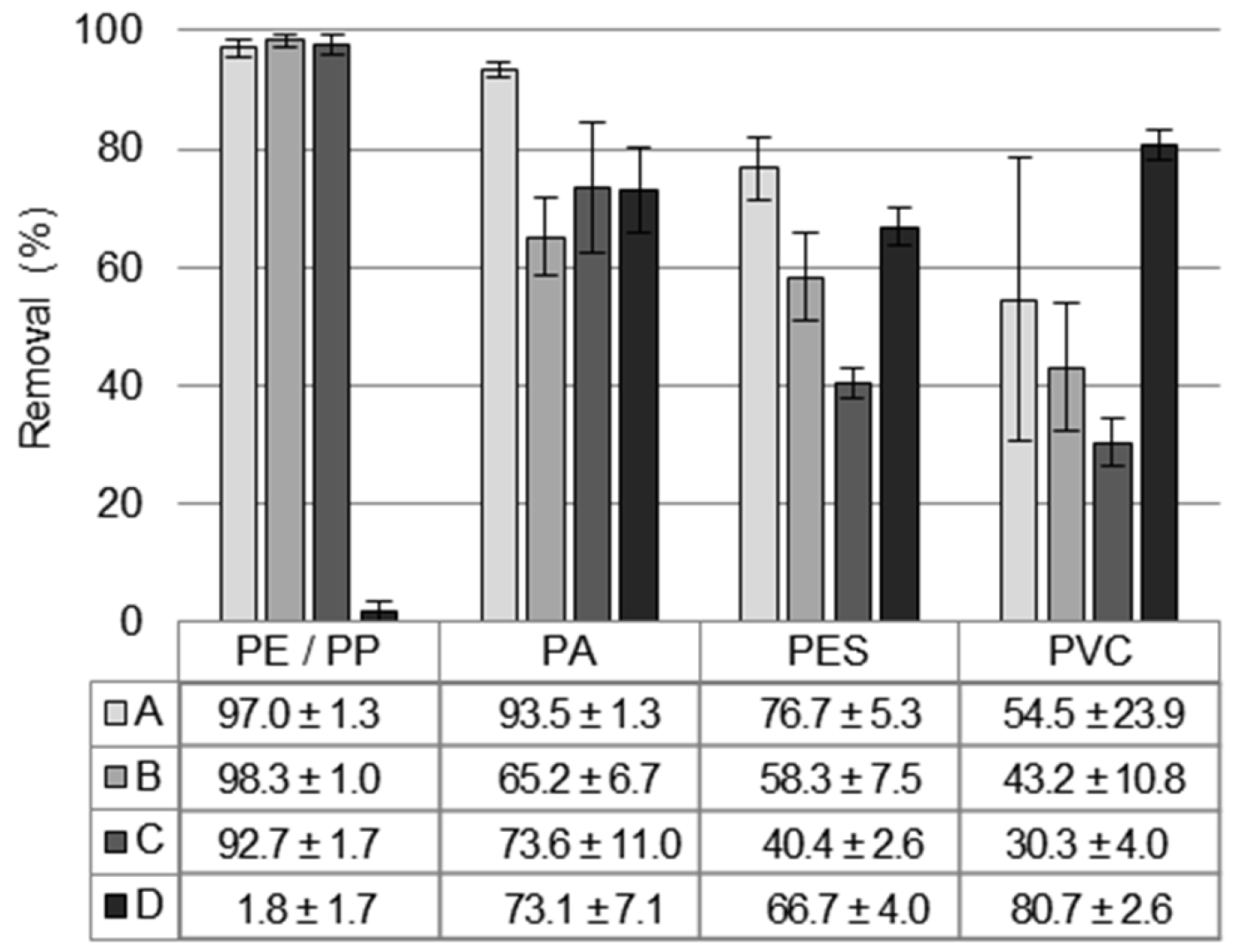
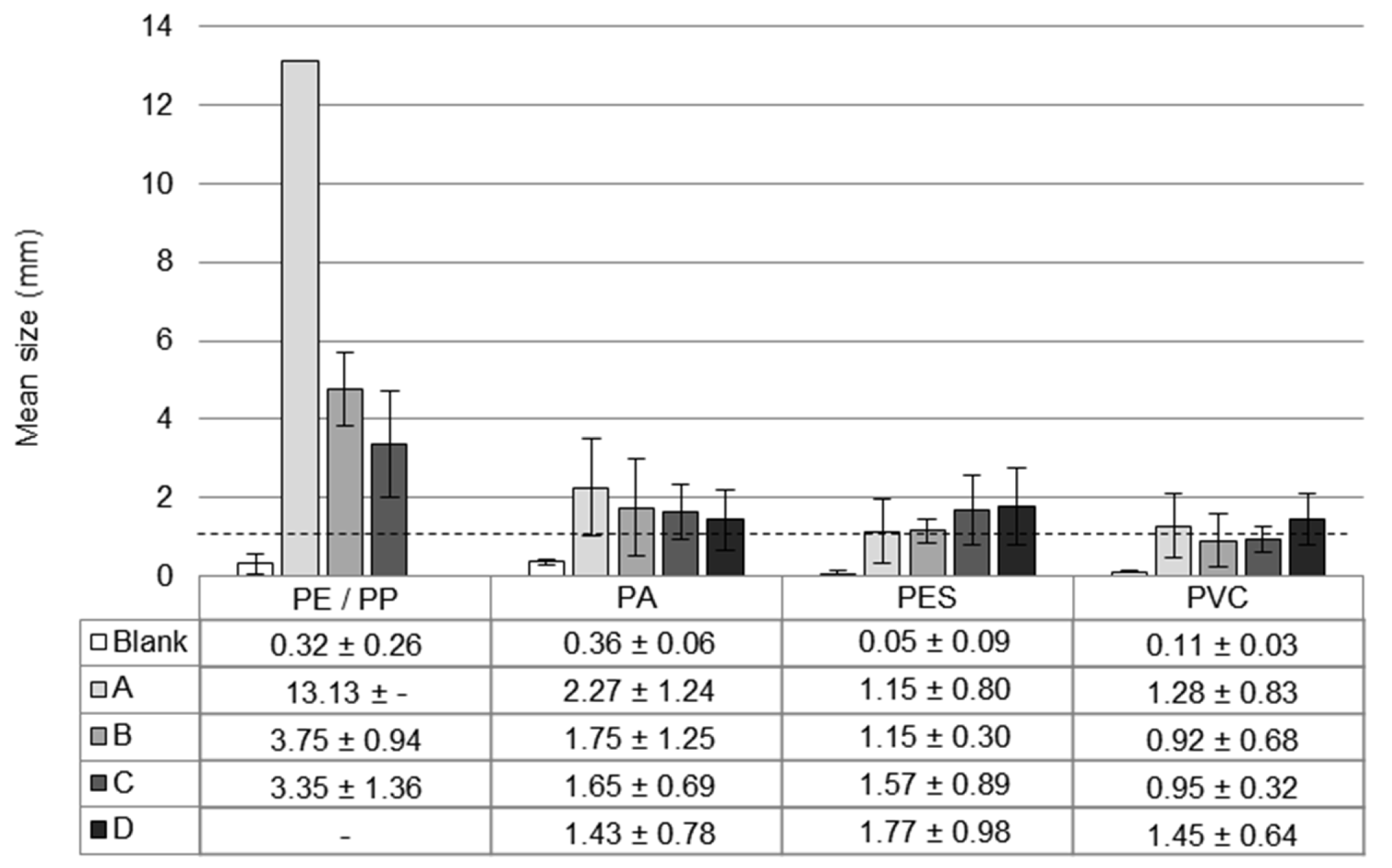
3.2. Comparison of Polymer Types
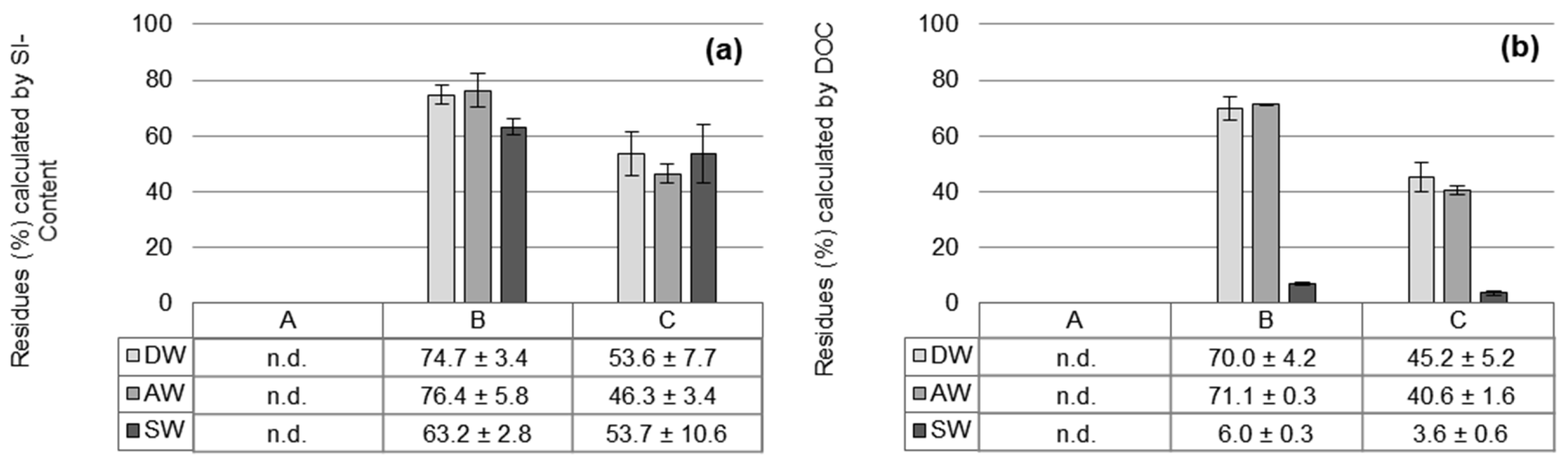
3.3. Organosilanes Residues Dissolved in Water
3.4. Additional Analytics
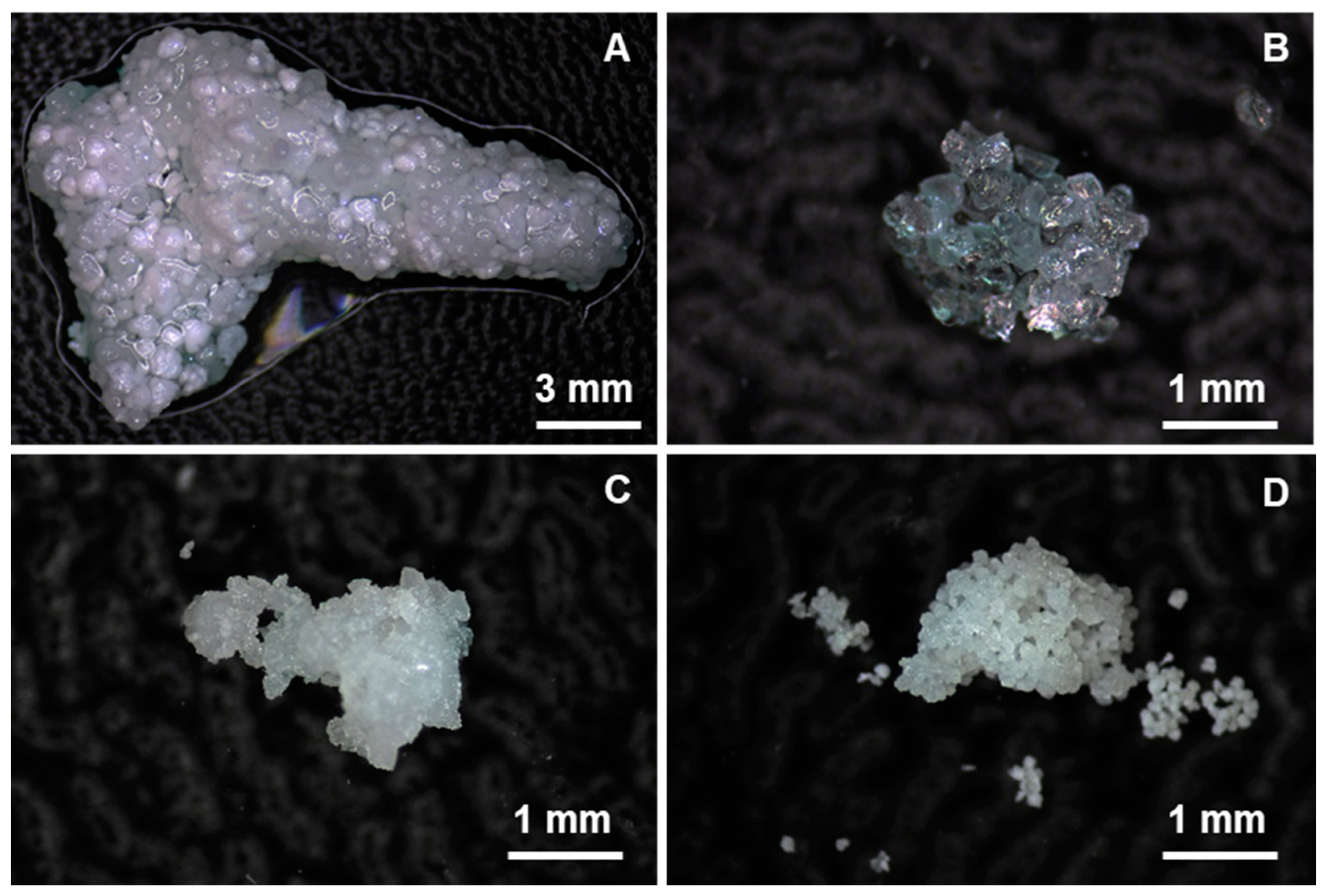
3.4.1. Microscope Images
3.4.2. IR-Spectra
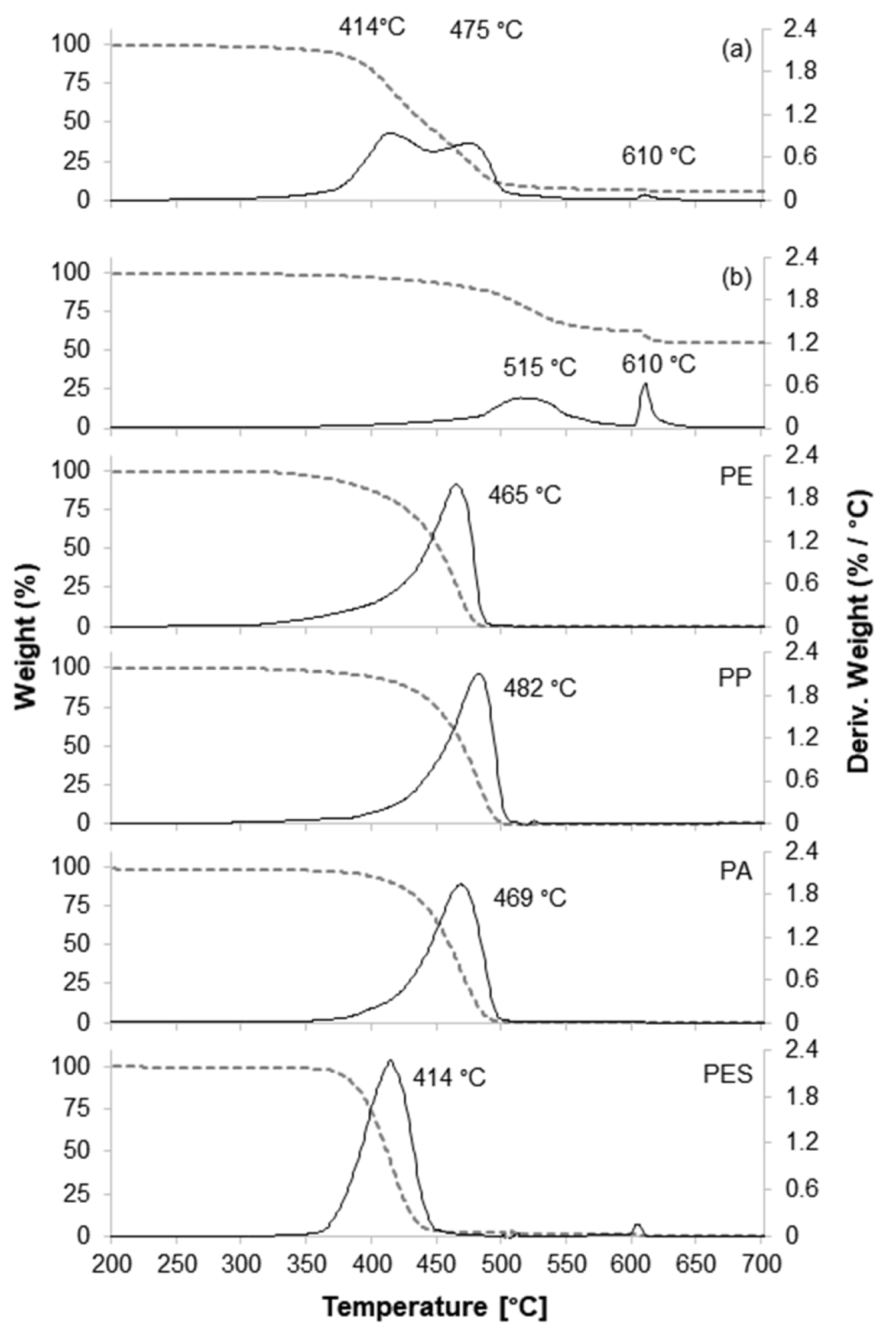
3.4.3. TGA
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barnes, D.K.A.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1985–1998. [Google Scholar] [CrossRef]
- Law, K.L.; Thompson, R.C. Microplastics in the seas. Science 2014, 345, 144–145. [Google Scholar] [CrossRef]
- Siegfried, M.; Koelmans, A.A.; Besseling, E.; Kroeze, C. Export of microplastics from land to sea. A modelling approach. Water Res. 2017, 127, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, J.P.; Santos, P.S.M.; Duarte, A.C.; Rocha-Santos, T. (Nano)plastics in the environment—Sources, fates and effects. Sci. Total Environ. 2016, 566–567, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Horton, A.A.; Walton, A.; Spurgeon, D.J.; Lahive, E.; Svendsen, C. Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ. 2017, 586, 127–141. [Google Scholar] [CrossRef]
- Gasperi, J.; Wright, S.L.; Dris, R.; Collard, F.; Mandin, C.; Guerrouache, M.; Langlois, V.; Kelly, F.J.; Tassin, B. Microplastics in air: Are we breathing it in? Curr. Opin. Environ. Sci. Health 2018, 1, 1–5. [Google Scholar] [CrossRef]
- Borrelle, S.B.; Ringma, J.; Law, K.L.; Monnahan, C.C.; Lebreton, L.; McGivern, A.; Murphy, E.; Jambeck, J.; Leonard, G.H.; Hilleary, M.A.; et al. Predicted growth in plastic waste exceeds efforts to mitigate plastic pollution. Science 2020, 369, 1515–1518. [Google Scholar] [CrossRef] [PubMed]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef]
- Wright, S.L.; Kelly, F.J. Plastic and Human Health: A Micro Issue? Environ. Sci. Technol. 2017, 51, 6634–6647. [Google Scholar] [CrossRef]
- Galloway, T.S.; Cole, M.; Lewis, C. Interactions of microplastic debris throughout the marine ecosystem. Nat. Ecol. Evol. 2017, 1, 116. [Google Scholar] [CrossRef]
- Schuhen, K.; Sturm, M.T. Microplastic Pollution and Reduction Strategies. In Handbook of Microplastics in the Environment; Rocha-Santos, T., Costa, M., Mouneyrac, C., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–33. ISBN 978-3-030-10618-8. [Google Scholar]
- Sun, J.; Dai, X.; Wang, Q.; van Loosdrecht, M.C.M.; Ni, B.-J. Microplastics in wastewater treatment plants: Detection, occurrence and removal. Water Res. 2019, 152, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Xue, W.; Hu, C.; Liu, H.; Qu, J.; Li, L. Characteristics of microplastic removal via coagulation and ultrafiltration during drinking water treatment. Chem. Eng. J. 2019, 359, 159–167. [Google Scholar] [CrossRef]
- Peixoto, D.; Pinheiro, C.; Amorim, J.; Oliva-Teles, L.; Guilhermino, L.; Vieira, M.N. Microplastic pollution in commercial salt for human consumption: A review. Estuar. Coast. Shelf Sci. 2019, 219, 161–168. [Google Scholar] [CrossRef]
- EFSA CONTAM Panel. Presence of microplastics and nanoplastics in food, with particular focus on seafood. Efsa J. 2016, 14, e04501. [Google Scholar] [CrossRef]
- Bui, X.-T.; Vo, T.-D.-H.; Nguyen, P.-T.; Nguyen, V.-T.; Dao, T.-S.; Nguyen, P.-D. Microplastics pollution in wastewater: Characteristics, occurrence and removal technologies. Environ. Technol. Innov. 2020, 19, 101013. [Google Scholar] [CrossRef]
- Pico, Y.; Alfarhan, A.; Barcelo, D. Nano- and microplastic analysis: Focus on their occurrence in freshwater ecosystems and remediation technologies. Trac. Trends Anal. Chem. 2019, 113, 409–425. [Google Scholar] [CrossRef]
- Poerio, T.; Piacentini, E.; Mazzei, R. Membrane Processes for Microplastic Removal. Molecules 2019, 24, 4148. [Google Scholar] [CrossRef]
- Talvitie, J.; Mikola, A.; Koistinen, A.; Setälä, O. Solutions to microplastic pollution—Removal of microplastics from wastewater effluent with advanced wastewater treatment technologies. Water Res. 2017, 123, 401–407. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Y.N.; Tian, L.; Ju, L.; Liu, Y. The removal efficiency and mechanism of microplastic enhancement by positive modification dissolved air flotation. Water Environ. Res. 2020. [Google Scholar] [CrossRef]
- Lapointe, M.; Farner, J.M.; Hernandez, L.M.; Tufenkji, N. Understanding and Improving Microplastic Removal during Water Treatment: Impact of Coagulation and Flocculation. Environ. Sci. Technol. 2020, 54, 8719–8727. [Google Scholar] [CrossRef]
- Harford, A.J.; Hogan, A.C.; Jones, D.R.; van Dam, R.A. Ecotoxicological assessment of a polyelectrolyte flocculant. Water Res. 2011, 45, 6393–6402. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.L.; Vidal, T.; Gonçalves, F.J.M.; Gabriel, R.G.; Costa, R.; Rasteiro, M.G. Is the aquatic toxicity of cationic polyelectrolytes predictable from selected physical properties? Chemosphere 2018, 202, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Herbort, A.F.; Schuhen, K. A concept for the removal of microplastics from the marine environment with innovative host-guest relationships. Environ. Sci. Pollut. Res. 2016, 11061–11065. [Google Scholar] [CrossRef] [PubMed]
- Herbort, A.F.; Sturm, M.T.; Schuhen, K. A new approach for the agglomeration and subsequent removal of polyethylene, polypropylene, and mixtures of both from freshwater systems—A case study. Environ. Sci. Pollut. Res. Int. 2018, 15226–15234. [Google Scholar] [CrossRef] [PubMed]
- Herbort, A.F.; Sturm, M.T.; Fiedler, S.; Abkai, G.; Schuhen, K. Alkoxy-silyl Induced Agglomeration: A New Approach for the Sustainable Removal of Microplastic from Aquatic Systems. J. Polym. Environ. 2018, 62, 4258–4270. [Google Scholar] [CrossRef]
- Sturm, M.T.; Herbort, A.F.; Horn, H.; Schuhen, K. Comparative study of the influence of linear and branched alkyltrichlorosilanes on the removal efficiency of polyethylene and polypropylene-based microplastic particles from water. Environ. Sci. Pollut. Res. 2020, 27, 10888–10898. [Google Scholar] [CrossRef]
- Schuhen, K.; Sturm, M.T.; Herbort, A.F. Technological Approaches for the Reduction of Microplastic Pollution in Seawater Desalination Plants and for Sea Salt Extraction. In Plastics in the Environment; Gomiero, A., Ed.; IntechOpen: London, UK, 2019; ISBN 978-1-83880-492-3. [Google Scholar]
- Brinker, C.J.; Scherer, G.W. Sol-gel Science: The physics and Chemistry of sol-gel Processing; Academic Press: Boston, MA, USA, 1990; ISBN 978-0-12-134970-7. [Google Scholar]
- Gurung, K.; Ncibi, M.C.; Sillanpää, M. Assessing membrane fouling and the performance of pilot-scale membrane bioreactor (MBR) to treat real municipal wastewater during winter season in Nordic regions. Sci. Total Environ. 2017, 579, 1289–1297. [Google Scholar] [CrossRef]
- Shore, J.L.; M’Coy, W.S.; Gunsch, C.K.; Deshusses, M.A. Application of a moving bed biofilm reactor for tertiary ammonia treatment in high temperature industrial wastewater. Bioresour. Technol. 2012, 112, 51–60. [Google Scholar] [CrossRef]
- Henze, M. Wastewater Treatment: Biological and Chemical Processes, 3rd ed.; Springer: New Delhi, India, 2010; ISBN 9783540422280. [Google Scholar]
- Akarsu, C.; Kumbur, H.; Gökdağ, K.; Kıdeyş, A.E.; Sanchez-Vidal, A. Microplastics composition and load from three wastewater treatment plants discharging into Mersin Bay, north eastern Mediterranean Sea. Mar. Pollut. Bull. 2020, 150, 110776. [Google Scholar] [CrossRef]
- Ben-David, E.A.; Habibi, M.; Haddad, E.; Hasanin, M.; Angel, D.L.; Booth, A.M.; Sabbah, I. Microplastic distributions in a domestic wastewater treatment plant: Removal efficiency, seasonal variation and influence of sampling technique. Sci. Total Environ. 2021, 752, 141880. [Google Scholar] [CrossRef]
- Ranade, V.V.; Bhandari, V.M.; Ahirrao, S. Industrial Wastewater Treatment, Recycling, and Reuse; Butterworth-Heinemann: Kidlington, Oxford, UK; Waltham, MA, USA, 2014; ISBN 9780444634030. [Google Scholar]
- Elderfield, H.; Holland, H.D. (Eds.) The Oceans and Marine Geochemistry, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2006; ISBN 9780080451015. [Google Scholar]
- Hammer, U.T. Saline Lake Ecosystems of the World; Junk: Dordrecht, The Netherlands, 1986; ISBN 9061935350. [Google Scholar]
- Rosner, D.; Markowitz, G. Persistent pollutants: A brief history of the discovery of the widespread toxicity of chlorinated hydrocarbons. Environ. Res. 2013, 120, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Hurkes, N.; Ehmann, H.M.A.; List, M.; Spirk, S.; Bussiek, M.; Belaj, F.; Pietschnig, R. Silanol-based surfactants: Synthetic access and properties of an innovative class of environmentally benign detergents. Chemistry 2014, 20, 9330–9335. [Google Scholar] [CrossRef] [PubMed]
- Baby, D.K. Rheology of hydrogels. In Rheology of Polymer Blends and Nanocomposites; Elsevier: Amsterdam, The Netherlands, 2020; pp. 193–204. ISBN 9780128169575. [Google Scholar]
- Al-Oweini, R.; El-Rassy, H. Synthesis and characterization by FTIR spectroscopy of silica aerogels prepared using several Si(OR)4 and R′′Si(OR′)3 precursors. J. Mol. Struct. 2009, 919, 140–145. [Google Scholar] [CrossRef]
- Noda, I.; Dowrey, A.E.; Haynes, J.L.; Marcott, C. Group Frequency Assignments for Major Infrared Bands Observed in Common Synthetic Polymers. In Physical Properties of Polymers Handbook; Mark, J.E., Ed.; Springer: New York, NY, USA, 2007; pp. 395–406. ISBN 978-0-387-31235-4. [Google Scholar]
- Jung, M.R.; Horgen, F.D.; Orski, S.V.; Rodriguez, C.V.; Beers, K.L.; Balazs, G.H.; Jones, T.T.; Work, T.M.; Brignac, K.C.; Royer, S.-J.; et al. Validation of ATR FT-IR to identify polymers of plastic marine debris, including those ingested by marine organisms. Mar. Pollut. Bull. 2018, 127, 704–716. [Google Scholar] [CrossRef] [PubMed]
| Polymer Type | Abbreviation | Mean Size [µm] | Supplier |
|---|---|---|---|
| Mixture of Polyethylene/Polypropylene (1:1) | PE/PP | 318 ± 258 | LyondellBasell, Basell Polyolefine GmbH, Frankfurt, Germany |
| Polyamide | PA | 357 ± 60 | EMS-Grilltech, Domat/Ems, Switzerland |
| Polyester | PES | 54 ± 87 | EMS-Grilltech, Domat/Ems, Switzerland |
| Polyvinylchloride | PVC | 110 ± 25 | Sigma-Adlrich, Taufkirchen, Germany |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sturm, M.T.; Horn, H.; Schuhen, K. Removal of Microplastics from Waters through Agglomeration-Fixation Using Organosilanes—Effects of Polymer Types, Water Composition and Temperature. Water 2021, 13, 675. https://doi.org/10.3390/w13050675
Sturm MT, Horn H, Schuhen K. Removal of Microplastics from Waters through Agglomeration-Fixation Using Organosilanes—Effects of Polymer Types, Water Composition and Temperature. Water. 2021; 13(5):675. https://doi.org/10.3390/w13050675
Chicago/Turabian StyleSturm, Michael Toni, Harald Horn, and Katrin Schuhen. 2021. "Removal of Microplastics from Waters through Agglomeration-Fixation Using Organosilanes—Effects of Polymer Types, Water Composition and Temperature" Water 13, no. 5: 675. https://doi.org/10.3390/w13050675
APA StyleSturm, M. T., Horn, H., & Schuhen, K. (2021). Removal of Microplastics from Waters through Agglomeration-Fixation Using Organosilanes—Effects of Polymer Types, Water Composition and Temperature. Water, 13(5), 675. https://doi.org/10.3390/w13050675







