Characteristics of Low-Oxygen Oxidation Ditch with Improved Nitrogen Removal
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Wastewater Characteristics
2.3. Analytical Methods and Calculations
2.4. Microbiological Analysis
2.5. Experimental Procedure
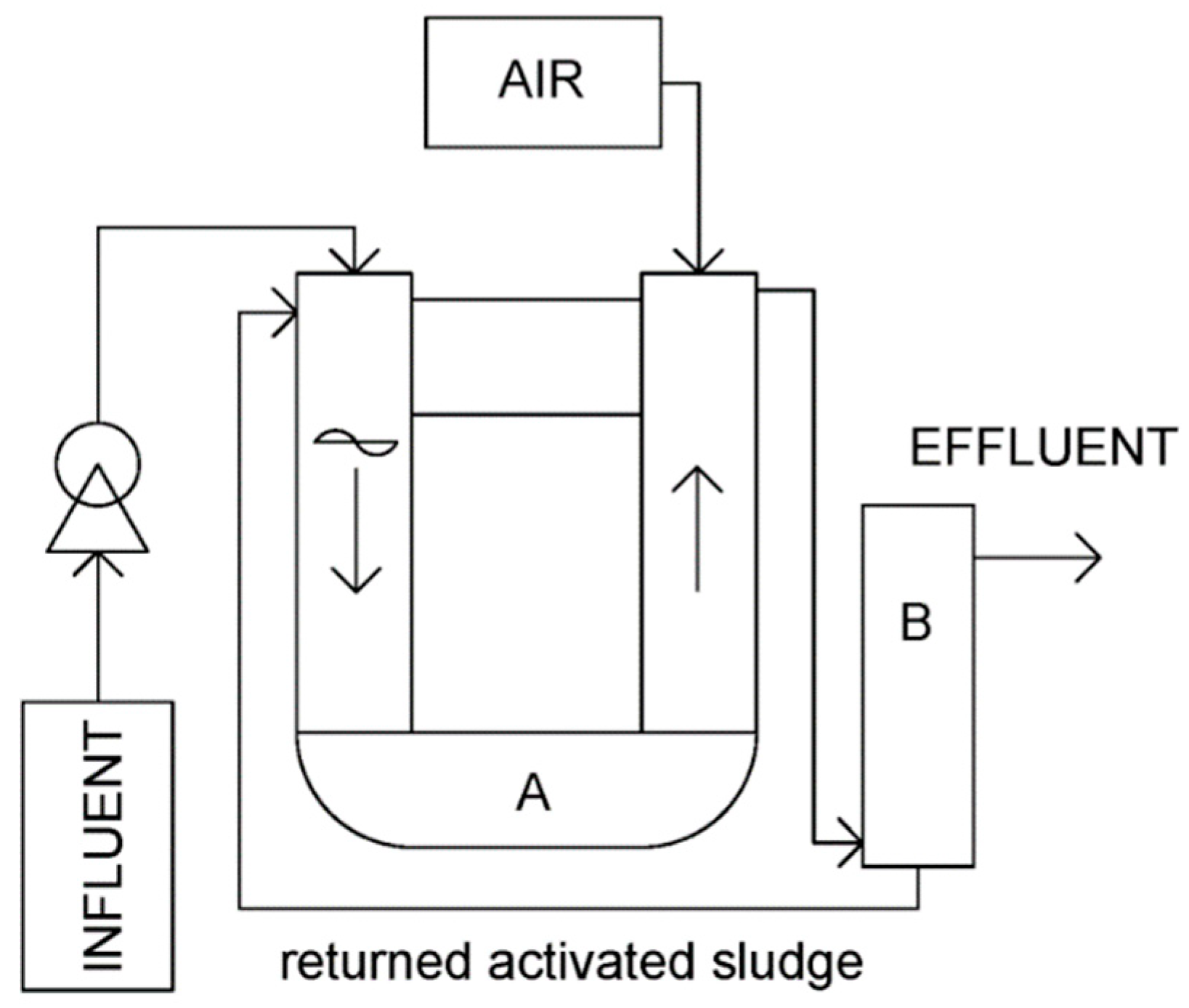
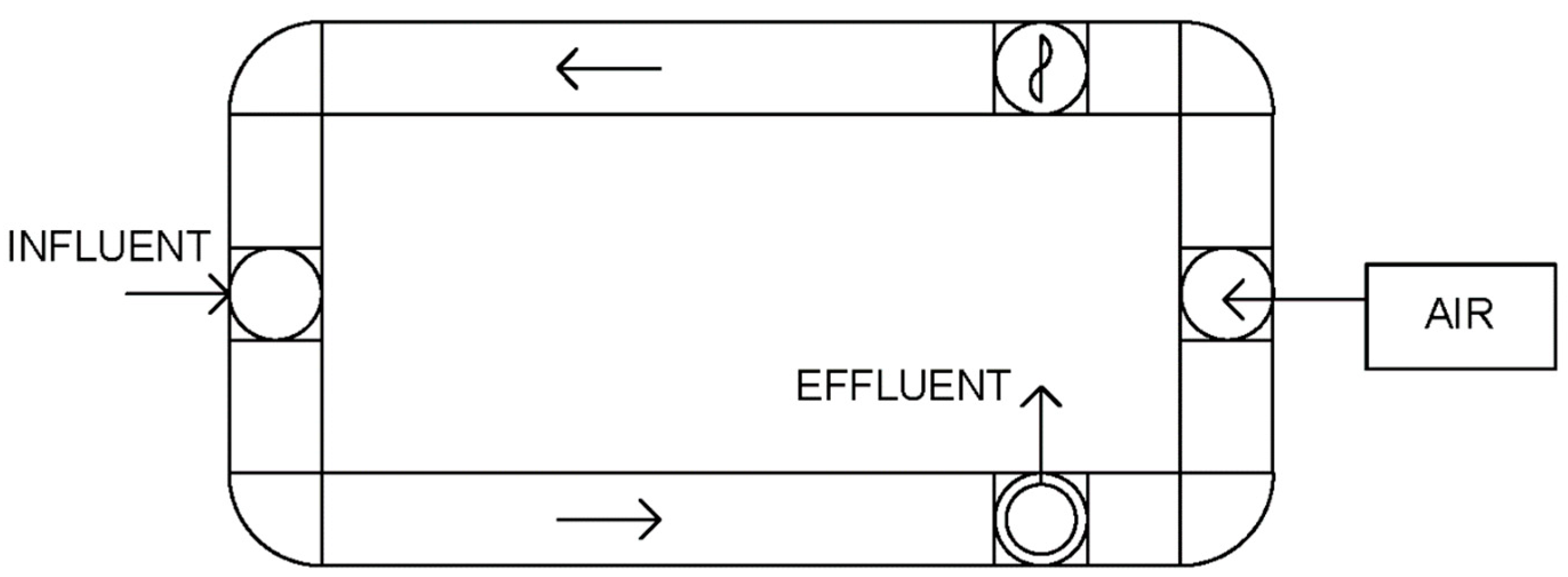
2.5.1. Experimental Set-Up at Stage 1
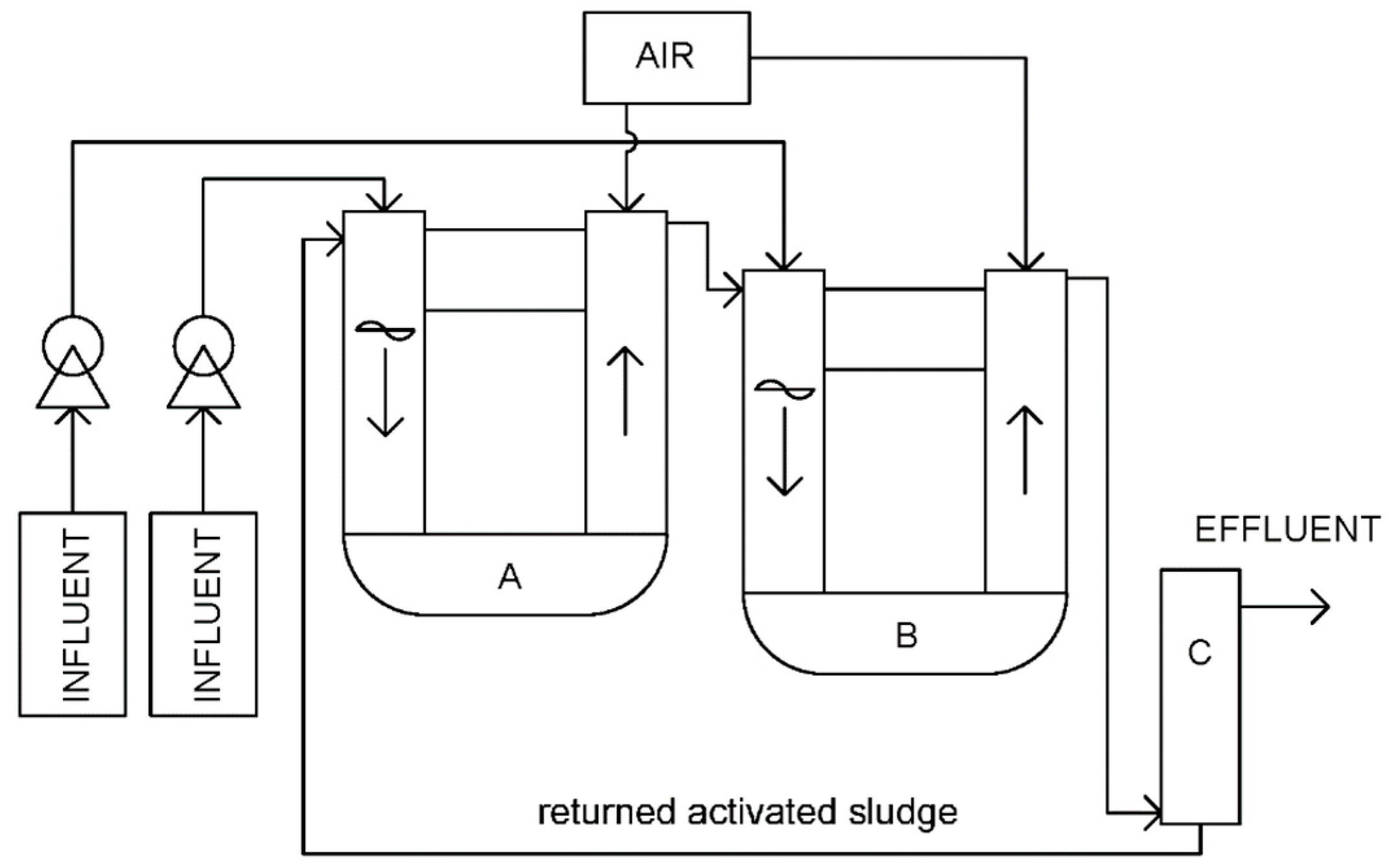
2.5.2. Experimental Set-Up at Stage 2
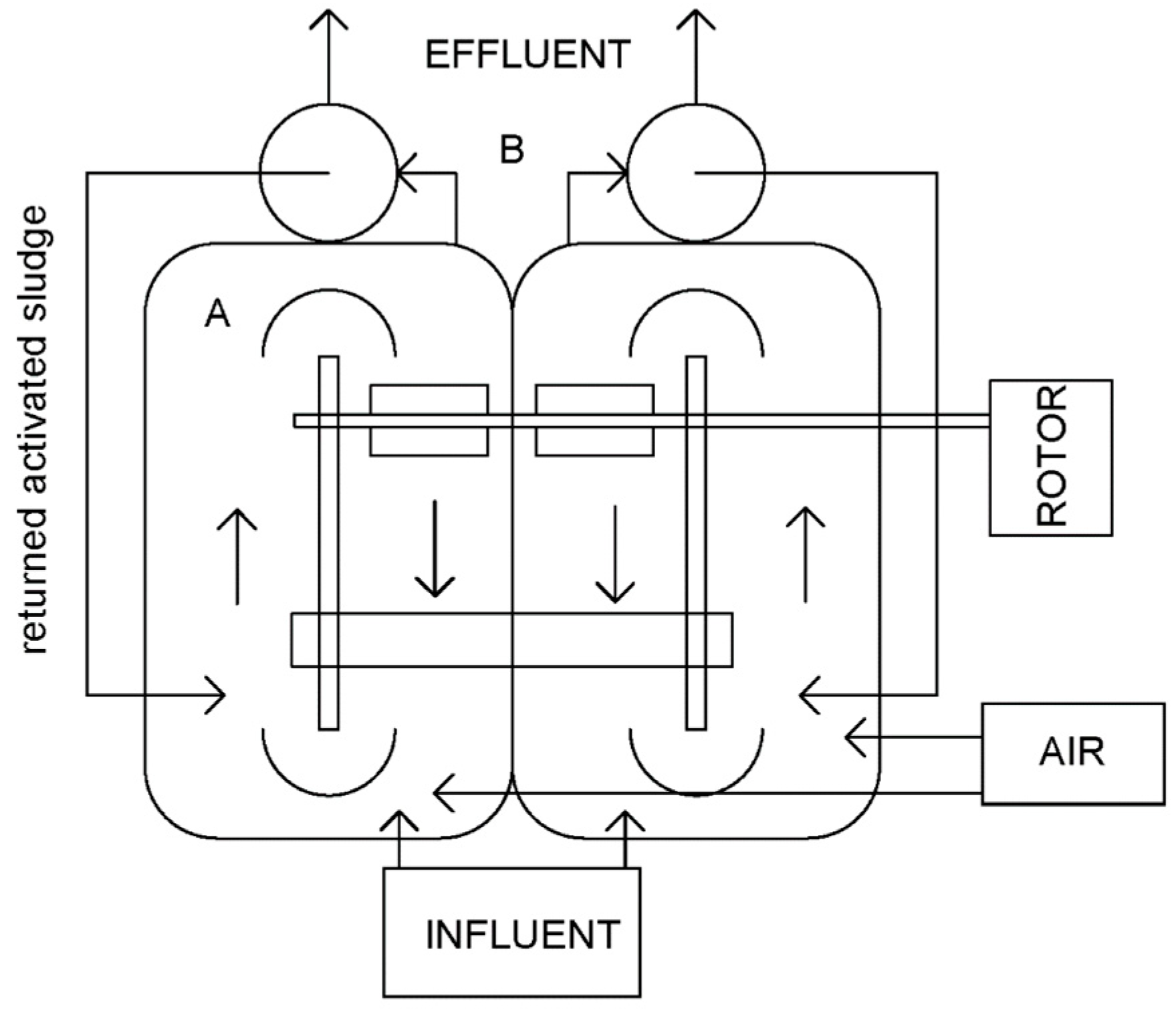
2.5.3. Experimental Set-Up at Stage 3
2.5.4. Operational Parameters of the Lab-Scale Reactors
3. Results and Discussion
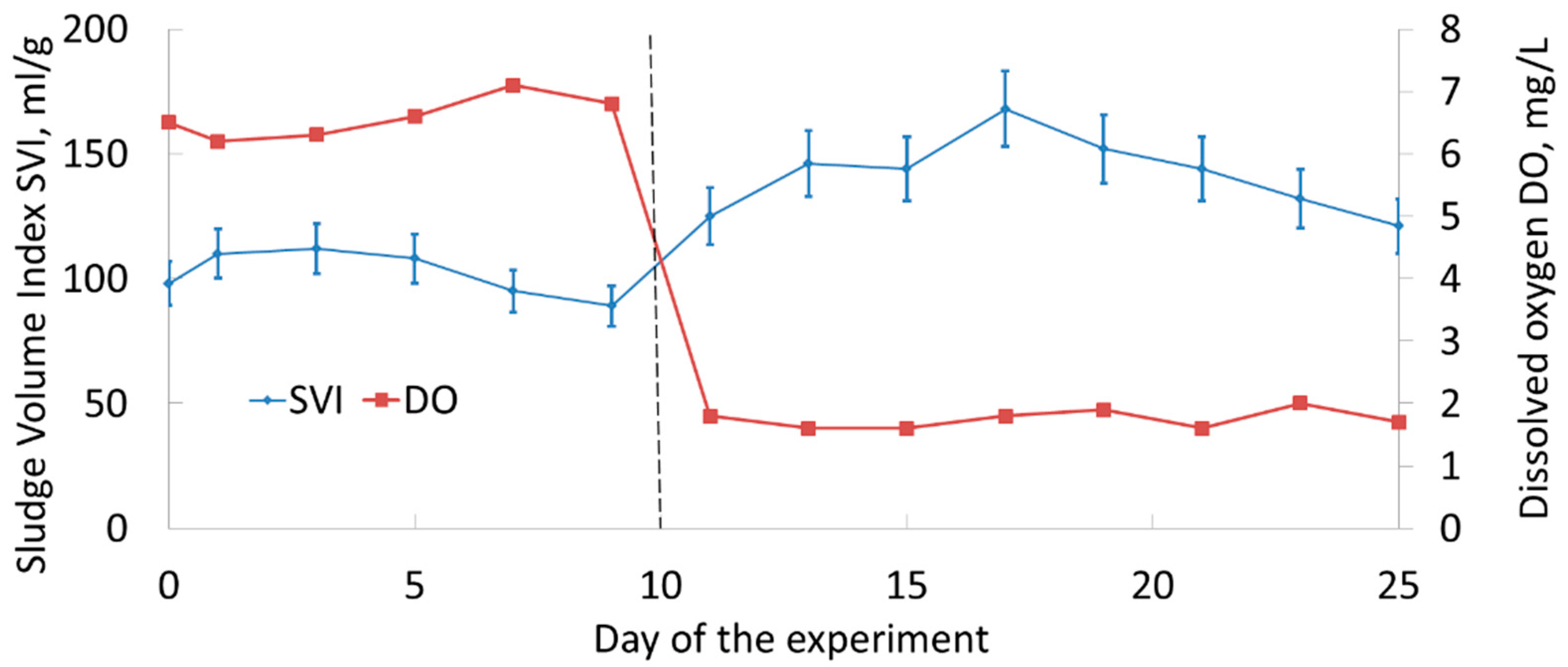
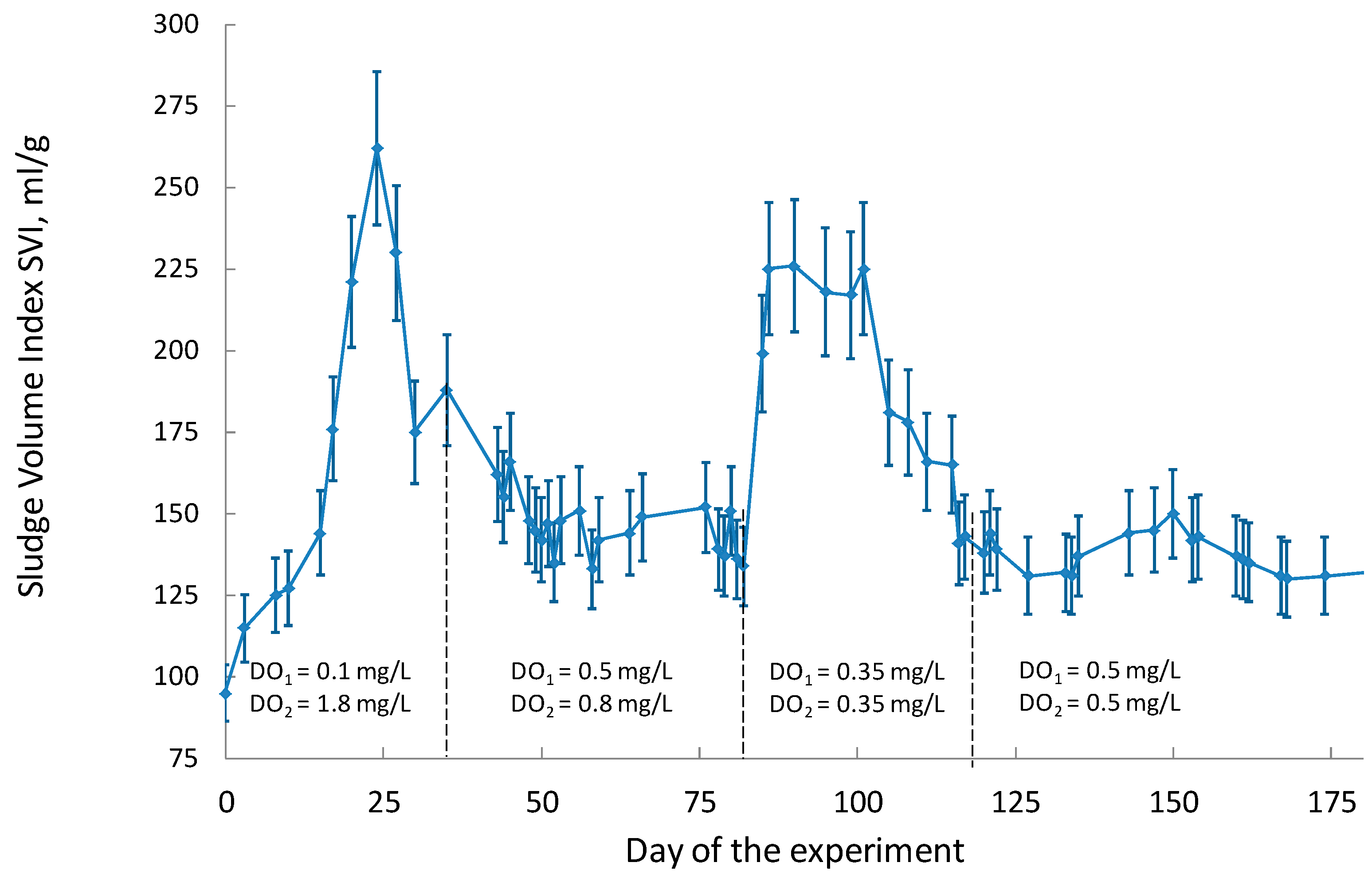
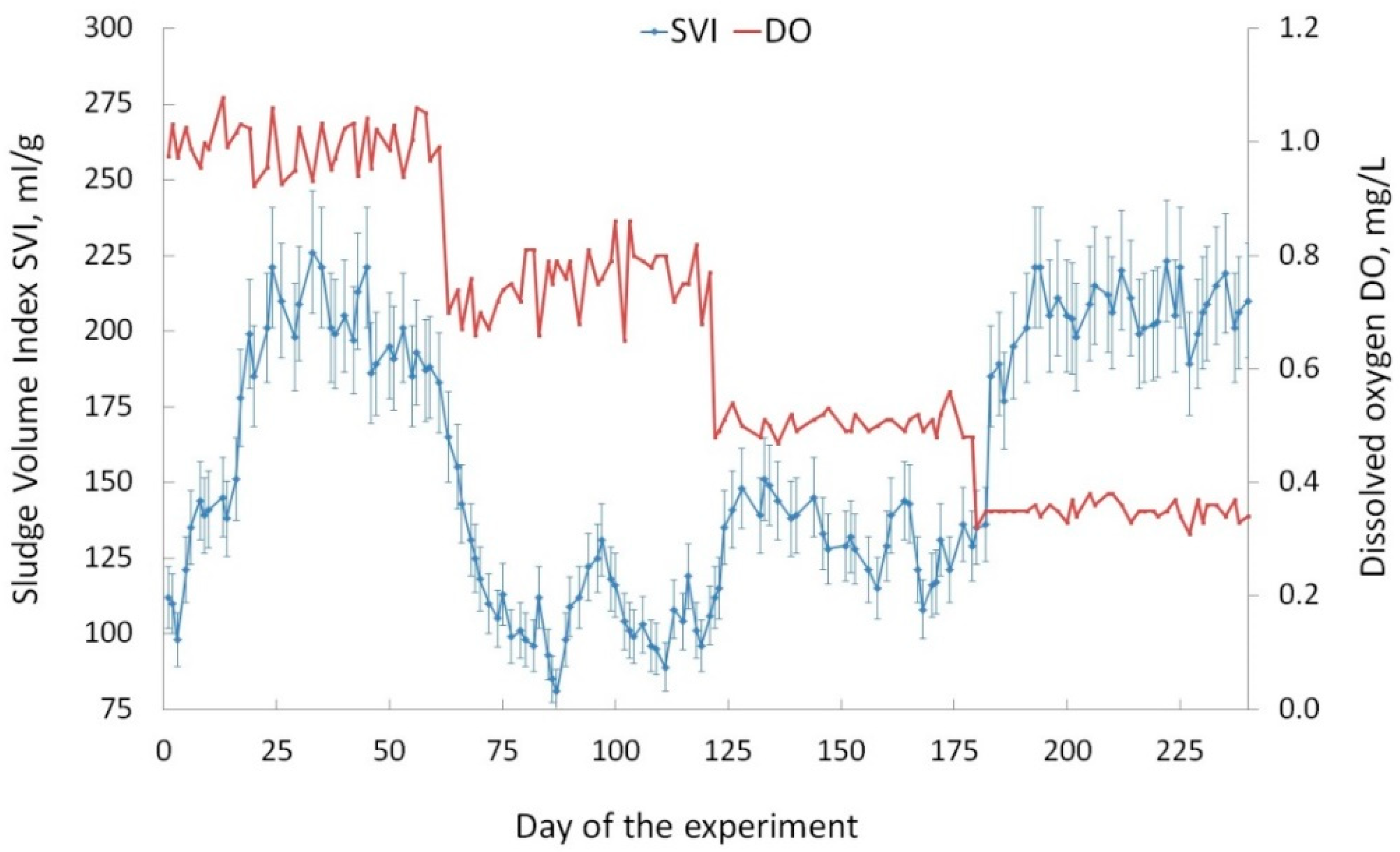
3.1. Activated Sludge: Charactreristics and Stability
3.2. PCR Testing
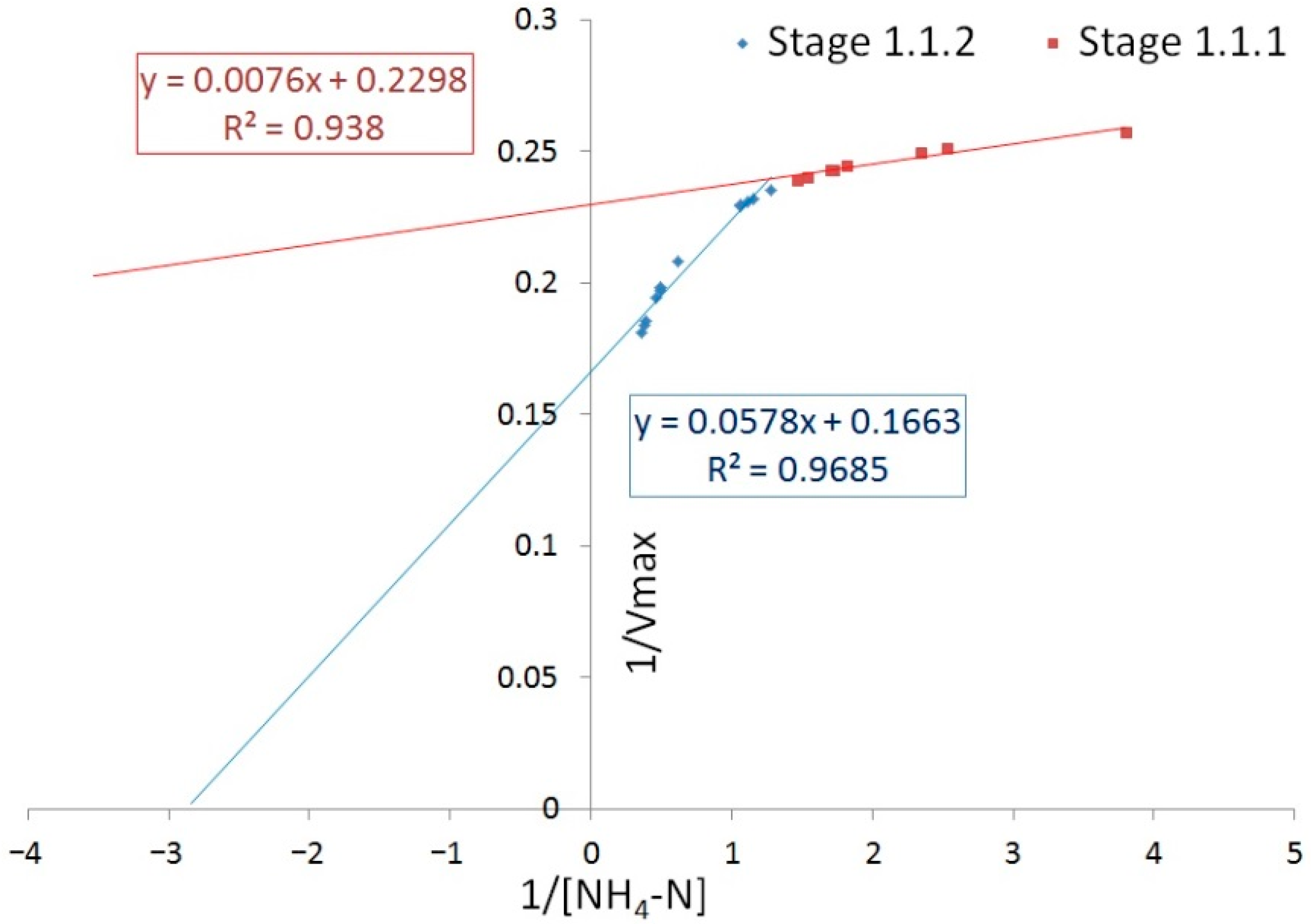
3.3. Reaction Rates
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pierre, K.C. Computational Fluid Dynamics (CFD) Analysis of the Hydraulic Performance and Bio-Kinetics in A Full-Scale Oxidation Ditch. Doctoral Dissertation, University of South Florida, Tampa, FL, USA, 9 July 2021. [Google Scholar]
- Uçan, R.; Şanlıer, H.; Özay, M.E. Investigation of Job Satisfaction and Occupational Safety Perceptions of Employees in Wastewater Treatment Plants: Study of Istanbul. Int. J. Occup. Saf. Health 2020, 10, 3–10. [Google Scholar] [CrossRef]
- Gandiglio, M.; Lanzini, A.; Soto, A.; Leone, P.; Santarelli, M. Enhancing the Energy Efficiency of Wastewater Treatment Plants through Co-digestion and Fuel Cell Systems. Front. Environ. Sci. 2017, 5, 70. [Google Scholar] [CrossRef] [Green Version]
- Luo, Y.; Yao, J.; Wang, X.; Zheng, M.; Guo, D.; Chen, Y. Efficient municipal wastewater treatment by oxidation ditch process at low temperature: Bacterial community structure in activated sludge. Sci. Total Environ. 2020, 703, 135031. [Google Scholar] [CrossRef]
- Li, X.; Zhang, J.; Zhang, X.; Li, J.; Liu, F.; Chen, Y. Start-up and nitrogen removal performance of CANON and SNAD processes in a pilot-scale oxidation ditch reactor. Process. Biochem. 2019, 84, 134–142. [Google Scholar] [CrossRef]
- Matko, T.; Chew, J.; Wenk, J.; Chang, J.; Hofman, J. Computational fluid dynamics simulation of two-phase flow and dissolved oxygen in a wastewater treatment oxidation ditch. Process. Saf. Environ. Prot. 2021, 145, 340–353. [Google Scholar] [CrossRef]
- Xu, D.; Li, J.; Ma, T. Rapid aerobic sludge granulation in an integrated oxidation ditch with two-zone clarifiers. Water Res. 2020, 175, 115704. [Google Scholar] [CrossRef]
- Wang, X.; Chen, T.; Jin, P.; Zhang, A.; Gao, C.; Qi, X.; Zhang, Y. Enhanced total nitrogen removal performance in a full scale Orbal oxidation ditch by a novel step aeration mode. Bioresour. Technol. 2019, 294, 122228. [Google Scholar] [CrossRef]
- Agbewornu, K.C.D.; Adyel, T.M.; Zhai, J. Optimizing nitrogen removal in a hybrid oxidation ditch. J. Environ. Chem. Eng. 2021, 9, 105443. [Google Scholar] [CrossRef]
- Xu, X.; Wei, W.; Liu, F.; Wei, W.; Liu, Z. Experimental study on aeration efficiency in a pilot-scale decelerated oxidation ditch equipped with fine bubble diffusers and impellers. Can. J. Chem. Eng. 2021, 99, 1410–1420. [Google Scholar] [CrossRef]
- Zhang, M.; Yao, J.; Wang, X.; Hong, Y.; Chen, Y. The microbial community in filamentous bulking sludge with the ultra-low sludge loading and long sludge retention time in oxidation ditch. Sci. Rep. 2019, 9, 13693. [Google Scholar] [CrossRef] [PubMed]
- Subtil, E.L.; Silva, M.V.; Lotto, B.A.; Moretto, M.R.D.; Mierzwa, J.C. Pilot-scale investigation on the feasibility of simultaneous nitrification and denitrification (SND) in a continuous flow single-stage membrane bioreactor. J. Water Process. Eng. 2019, 32, 100995. [Google Scholar] [CrossRef]
- Wang, J.; Rong, H.; Cao, Y.; Zhang, C. Factors affecting simultaneous nitrification and denitrification (SND) in a moving bed sequencing batch reactor (MBSBR) system as revealed by microbial community structures. Bioprocess Biosyst. Eng. 2020, 43, 1833–1846. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Jia, G.; Xu, J.; He, X.; Quan, X. Simultaneous nitrification and denitrification in continuous flow MBBR with novel surface-modified carriers. Environ. Technol. 2021, 42, 3607–3617. [Google Scholar] [CrossRef] [PubMed]
- Pochana, K.; Keller, J. Study of factors affecting simultaneous nitrification and denitrification (SND). Water Sci. Technol. 1999, 39, 61–68. [Google Scholar] [CrossRef]
- Zhao, H.W.; Mavinic, D.S.; Oldham, W.K.; Koch, F.A. Controlling factors for simultaneous nitrification and denitrification in a two-stage intermittent aeration process treating domestic sewage. Water Res. 1999, 33, 961–970. [Google Scholar] [CrossRef]
- Rittmann, B.E.; Langeland, W.E. Simultaneous denitrification with nitrification in single-channel oxidation ditches. J. Water Pollut. Control Fed. 1985, 57, 300–308. [Google Scholar]
- Sun, S.-P.; Pellicer-Nàcher, C.; Merkey, B.; Zhou, Q.; Xia, S.-Q.; Yang, D.-H.; Sun, J.-H.; Smets, B.F. Effective Biological Nitrogen Removal Treatment Processes for Domestic Wastewaters with Low C/N Ratios: A Review. Environ. Eng. Sci. 2010, 27, 111–126. [Google Scholar] [CrossRef] [Green Version]
- Liu, B.; Lin, H.; Yu, G.; Zhang, S.; Zhao, C. Fate of dissolved organic nitrogen during biological nutrient removal wastewater treatment processes. J. Environ. Biol. 2013, 34, 325. [Google Scholar]
- Hao, O.J.; Huang, J. Alternating aerobic-anoxic process for nitrogen removal: Process evaluation. Water Environ. Res. 1996, 68, 83–93. [Google Scholar] [CrossRef]
- Jayasvasti, M.; Ratanatamskul, C. Appropriate Lab Scale Oxidation Ditch Tank for Cafeteria Building Wastewater Treatment. Int. J. Adv. Agric. Environ. Eng. 2014, 1, 127–130. [Google Scholar]
- Zhang, P.; Qi, Z. Simultaneous nitrification and denitrification in activated sludge system under low oxygen concentration. Front. Environ. Sci. Eng. China 2007, 1, 49–52. [Google Scholar] [CrossRef]
- Zhu, G.-C.; Lu, Y.-Z.; Xu, L.-R. Effects of the carbon/nitrogen (C/N) ratio on a system coupling simultaneous nitrification and denitrification (SND) and denitrifying phosphorus removal (DPR). Environ. Technol. 2021, 42, 3048–3054. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wei, W.; Lv, B.; Yang, X. Research on optimal radius ratio of impellers in an oxidation ditch by using numerical simulation. Desalination Water Treat. 2013, 52, 2811–2816. [Google Scholar] [CrossRef]
- Pang, H.; Shi, H.; Shi, H. Flow characteristic and wastewater treatment performance of a pilot-scale airlift oxidation ditch. Front Environ. Sci. Eng 2009, 3, 470. [Google Scholar] [CrossRef]
- Mpongwana, N.; Ntwampe, S.K.O.; Omodanisi, E.I.; Chidi, B.S.; Razanamahandry, L.C.; Dlangamandla, C.; Mukandi, M.R. Bio-Kinetics of Simultaneous Nitrification and Aerobic Denitrification (SNaD) by a Cyanide- Degrading Bacterium Under Cyanide-Laden Conditions. Appl. Sci. 2020, 10, 4823. [Google Scholar] [CrossRef]
- Medhi, K.; Singhal, A.; Chauhan, D.; Thakur, I.S. Investigating the nitrification and denitrification kinetics under aerobic and anaerobic conditions by Paracoccus denitrificans ISTOD1. Bioresour. Technol. 2017, 242, 334–343. [Google Scholar] [CrossRef]
- Münch, E.V.; Lant, P.; Keller, J. Simultaneous nitrification and denitrification in bench-scale sequencing batch reactors. Water Res. 1996, 30, 277–284. [Google Scholar] [CrossRef]
- Henkel, J. Oxygen Transfer Phenomena in Activated Sludge. Doctoral Dissertation, TU Darmstadt, Darmstadt, Hessen, Germany, 2010. [Google Scholar]
- Hocaoglu, S.M.; Insel, G.; Cokgor, E.U.; Orhon, D. Effect of low dissolved oxygen on simultaneous nitrification and denitrification in a membrane bioreactor treating black water. Bioresour. Technol. 2011, 102, 4333–4340. [Google Scholar] [CrossRef] [PubMed]
- Mantziaras, I.D.; Katsiri, A. Reaction rate constants and mean population percentage for nitrifiers in an alternating oxidation ditch system. Bioprocess Biosyst. Eng. 2010, 34, 57–65. [Google Scholar] [CrossRef]
- Gogina, E.; Gulshin, I. Simultaneous Nitrification and Denitrification with Low Dissolved Oxygen Level and C/N ratio. Procedia Eng. 2016, 153, 189–194. [Google Scholar] [CrossRef] [Green Version]
- Gulshin, I. The Settling Behaviour of An Activated Sludge with Simultaneous Nitrification and Dentrification. In Web of Conferences; EDP Sciences: Les Ulis, France, 2017; Volume 106, p. 07002. [Google Scholar]
- Water Environment Federation; APH Association. Standard Methods for the Examination of Water and Wastewater; American Public Health Association (APHA): Washington, DC, USA, 2005. [Google Scholar]
- Van Loosdrecht, M.C.; Nielsen, P.H.; Lopez-Vazquez, C.M.; Brdjanovic, D. Experimental Methods in Wastewater Treatment; IWA Publishing: London, UK, 2016. [Google Scholar]
- Zhou, X.; Han, Y.; Guo, X. Identification and evaluation of SND in a full-scale multi-channel oxidation ditch system under different aeration modes. Chem. Eng. J. 2015, 259, 715–723. [Google Scholar] [CrossRef]
- Vesilind, P.A. Design of prototype thickeners from batch settling tests. Water Sew. Work. 1968, 115, 302–307. [Google Scholar]
- Fierer, N.; Jackson, J.A.; Vilgalys, R.; Jackson, R.B. Assessment of Soil Microbial Community Structure by Use of Taxon-Specific Quantitative PCR Assays. Appl. Environ. Microbiol. 2005, 71, 4117–4120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hallin, S.; Jones, C.; Schloter, M.; Philippot, L. Relationship between N-cycling communities and ecosystem functioning in a 50-year-old fertilization experiment. ISME J. 2009, 3, 597–605. [Google Scholar] [CrossRef]
- Michotey, V.; Méjean, V.; Bonin, P. Comparison of Methods for Quantification of Cytochrome cd 1 -Denitrifying Bacteria in Environmental Marine Samples. Appl. Environ. Microbiol. 2000, 66, 1564–1571. [Google Scholar] [CrossRef] [Green Version]
- Throbäck, I.N.; Enwall, K.; Jarvis, Å.; Hallin, S. Reassessing PCR primers targeting nirS, nirK and nosZ genes for community surveys of denitrifying bacteria with DGGE. FEMS Microbiol. Ecol. 2004, 49, 401–417. [Google Scholar] [CrossRef]
- Yu, Y.; Lee, C.; Hwang, S. Analysis of community structures in anaerobic processes using a quantitative real-time PCR method. Water Sci. Technol. 2005, 52, 85–91. [Google Scholar] [CrossRef]
- Park, S.-J.; Park, B.-J.; Rhee, S.-K. Comparative analysis of archaeal 16S rRNA and amoA genes to estimate the abundance and diversity of ammonia-oxidizing archaea in marine sediments. Extremophiles 2008, 12, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Wilén, B.-M.; Balmér, P. The effect of dissolved oxygen concentration on the structure, size and size distribution of activated sludge flocs. Water Res. 1999, 33, 391–400. [Google Scholar] [CrossRef]
- Casey, T.G.; Wentzel, M.C.; Ekama, G.A. Filamentous organism bulking in nutrient removal activated sludge systems. A historical overview of causes and control. Water S. Afr. 1995, 21, 231–238. [Google Scholar]
- Jenkins, D.; Richard, M.G.; Daigger, G.T. Manual on the Causes and Control of Activated Sludge Bulking, Foaming, and Other Solids Separation Problems; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Liu, Y.; Shi, H.; Xia, L.; Shi, H.; Shen, T.; Wang, Z.; Wang, G.; Wang, Y. Study of operational conditions of simultaneous nitrification and denitrification in a Carrousel oxidation ditch for domestic wastewater treatment. Bioresour. Technol. 2010, 101, 901–906. [Google Scholar] [CrossRef]
- Hao, X.; Doddema, H.J.; van Groenestijn, J.W. Conditions and mechanisms affecting simultaneous nitrification and denitrification in a Pasveer oxidation ditch. Bioresour. Technol. 1997, 59, 207–215. [Google Scholar] [CrossRef]
- Härtel, L.; Pöpel, H.J. A Dynamic Secondary Clarifier Model including Processes of Sludge Thickening. Water Sci. Technol. 1992, 25, 267–284. [Google Scholar] [CrossRef]
- Koopman, B.; Cadee, K. Prediction of thickening capacity using diluted sludge volume index. Water Res. 1983, 17, 1427–1431. [Google Scholar] [CrossRef]
- Pitman, A.R. Settling of Nutrient Removal Activated Sludges. Water Sci. Technol. 1985, 17, 493–504. [Google Scholar] [CrossRef]
- Uby, L. Handbook of Mixing for Wastewater and Similar Applications; Xylem Water Solutions AB: Sundbyberg, Sweden, 2012. [Google Scholar]
- Xu, D.; Liu, S.; Chen, Q.; Ni, J. Microbial community compositions in different functional zones of Carrousel oxidation ditch system for domestic wastewater treatment. AMB Express 2017, 7, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.-D.; Regan, J.; Noguera, D. Molecular analysis of ammonia-oxidizing bacterial populations in aerated-anoxic Orbal processes. Water Sci. Technol. 2002, 46, 273–280. [Google Scholar] [CrossRef] [Green Version]
- Meyer, R.L.; Zeng, R.J.; Giugliano, V.; Blackall, L.L. Challenges for simultaneous nitrification, denitrification, and phosphorus removal in microbial aggregates: Mass transfer limitation and nitrous oxide production. FEMS Microbiol. Ecol. 2005, 52, 329–338. [Google Scholar] [CrossRef] [Green Version]
- Wen, J.; Lechevallier, M.W.; Tao, W. Microbial community similarity and dissimilarity inside and across full-scale activated sludge processes for simultaneous nitrification and denitrification. Water Sci. Technol. 2020, 81, 333–344. [Google Scholar] [CrossRef]
- Xia, Z.; Wang, Q.; She, Z.; Gao, M.; Zhao, Y.; Guo, L.; Jin, C. Nitrogen removal pathway and dynamics of microbial community with the increase of salinity in simultaneous nitrification and denitrification process. Sci. Total Environ. 2019, 697, 134047. [Google Scholar] [CrossRef]
- Applegate, C.S.; Wilder, B.; DeShaw, J.R. Total nitrogen removal in a multichannel oxidation system. J. Water Pollut. Control Fed. 1980, 52, 568–577. [Google Scholar]
- Daigger, G.T.; Littleton, H.X. Characterization of Simultaneous Nutrient Removal in Staged, Closed-Loop Bioreactors. Water Environ. Res. 2000, 72, 330–339. [Google Scholar] [CrossRef]
- Gao, S.-Y.; Peng, Y.-Z.; Wang, S.-Y.; Yan, J. Novel strategy of nitrogen removal from domestic wastewater using pilot Orbal oxidation ditch. J. Environ. Sci. 2006, 18, 833–839. [Google Scholar] [CrossRef]
- WRC. Theory, Design and Operation of Nutrient Removal Activated Sludge Processes; Water Research Commission of South Africa: Pretoria, South Africa, 1984. [Google Scholar]
- Eckenfelder, W.W.; Argaman, Y.; Miller, E. Process selection criteria for the biological treatment of industrial wastewaters. Environ. Prog. 1989, 8, 40–45. [Google Scholar] [CrossRef]
- Shvetsov, V.; Stepanov, S.; Khar’kina, O. Comparison of the calculation results for aeration tanks using the NII VODGEO/SAMGTU and ASM2d models. Vodosnabzhenie Sanitarnaya Tekhnika 2021, 5, 18–29. [Google Scholar] [CrossRef]
| Parameter | Max. | Min. | Average |
|---|---|---|---|
| COD, mg/L | 215 | 52.8 | 145 |
| BOD5, mg/L | 185 | 35.3 | 138 |
| NH4-N, mg/L | 81.8 | 18.5 | 37 |
| PO4-P, mg/L | 13.5 | 2.8 | 7.2 |
| TSS, mg/L | 194.15 | 89.88 | 115.36 |
| pH | 8.7 | 7.3 | 7.7 |
| Parameter | Max. | Min. | Average |
|---|---|---|---|
| COD, mg/L | 325 | 64.8 | 152 |
| BOD5, mg/L | 211 | 56 | 143 |
| NH4-N, mg/L | 73.5 | 29.6 | 42.8 |
| PO4-P, mg/L | 32.6 | 5.3 | 8.9 |
| TSS, mg/L | 236.4 | 144.31 | 178.89 |
| pH | 8.3 | 7.6 | 7.9 |
| Target Group or Process | Target Gene | Primer | Primer Sequence (F, R) | Standard Source | Refs |
|---|---|---|---|---|---|
| Bacteria | 16S rRNA | Eub338 Eub518 | ACTCCTACGGGAGGCAGCAG ATTACCGCGGCTGCTGG | Escherichia coli | [38] |
| Archaea | 16S rRNA | 915f 1059r | AGGAA TTGGC GGGGG AGCAC GCCAT GCACC WCCTC T | strain FG-07 Halobacterium salinarum | [42] |
| Nitrification | bacterial amoA | amoA-1F amoA-2R | GGGGTTTCTACTGGTGGT CCCCTCKGSAAAGCCTTCTTC | Soil | [39] |
| archaeal amoA | Arch-amoAF Arch-amoAR | GCTCTAATTATGACAGTATAC AYCATGTTGAAYAATGGTAATGAC | - | [43] | |
| Denitrification | nirK | nirK876 nirK1040 | ATY GGC GGV CAY GGC GA GCC TCG ATC AGR TTR TGG TT | Sinorhizobium meliloti | [39] |
| nirS | cd3af R3cd | GTSAACGTSAAGGARACSGG GASTTCGGRTGSGTCTTGA | Pseudomonas sp. | [40] |
| Stage | Lab-Scale Reactor | Operational Modes |
|---|---|---|
| 1.1.1 | Lab-scale reactor operating in the mode close to the conditions of ideal mixing with discernible longitudinal mixing (synthetic wastewater). | DO—6–7 mg/L; horizontal flow velocity—0.02 m/s |
| 1.1.2 | DO—1.5–2.0 mg/L; horizontal flow velocity—0.2 m/s | |
| 1.2.1 | Lab-scale reactor operating in the mode close to the conditions of ideal mixing with discernible longitudinal mixing. Consists of two modules capable of operating in various oxygen modes under the conditions of intensive longitudinal mixing (synthetic wastewater). | DO—the first zone: 0.1 mg/L, the second zone: 1.8 mg/L, average concentration—0.95 mg/L; horizontal flow velocity—0.15 m/s |
| 1.2.2 | DO—the first zone: 0.5 mg/L, the second zone: 0.8 mg/L, average concentration—0.65 mg/L; horizontal flow velocity—0.15 m/s | |
| 1.2.3 | DO—the first zone: 0.35 mg/L, the second zone: 0.35 mg/L, average concentration—0.35 mg/L; horizontal flow velocity—0.2 m/s | |
| 1.2.4 | DO—the first zone: 0.5 mg/L, the second zone: 0.5 mg/L, average concentration—0.5 mg/L; horizontal flow velocity—0.2 m/s | |
| 2.1 | Laboratory reactor with mechanical aeration and a flow circulation system. There are no dedicated oxygen zones. Observation of the SND process (synthetic wastewater). | DO—average concentration—1.00 mg/l; horizontal flow velocity—0.2 m/s; BOD specific load—0.41 g/g/d; HRT—6.5 h |
| 2.2 | DO—average concentration—0.75 mg/l; horizontal flow velocity—0.2 m/s; BOD specific load—0.29 g/g/d; HRT—7 h | |
| 2.3 | DO—average concentration—0.50 mg/l; horizontal flow velocity—0.3 m/s; BOD specific load—0.23 g/g/d; HRT—10 h | |
| 2.4 | DO—average concentration—0.35 mg/l; horizontal flow velocity—0.3 m/s; BOD specific load—0.13 g/g/d; HRT—12 h | |
| 3.1 | Laboratory reactor with dedicated oxygen zones along the length of the channel. The dissolved oxygen concentration gradient is determined by the oxygen consumption along the channel. Mixing and aeration are combined. The unit has a built-in secondary clarifier (real wastewater). | DO—the first zone: 2.5 mg/L, the second zone: 1.3 mg/L, average concentration—1.9 mg/L; horizontal flow velocity—0.2 m/s; internal recirculation rate—250% |
| 3.2 | DO—the first zone: 1.9 mg/L, the second zone: 0.15 mg/L, average concentration—1.03 mg/L; horizontal flow velocity—0.25 m/s; internal recirculation rate—350% |
| Stage | Average Sizes of Floccules by the Equivalent Diameter, μm |
|---|---|
| 1.1 | 78 |
| 1.2.2 | 115 |
| 1.2.4 | 168 |
| 2.2 | 104 |
| 2.3 | 244 |
| 3.1 | 369 |
| 3.2 | 452 |
| Sample | Stage |
|---|---|
| 1 | 1.2.1 |
| 2 | 1.2.2 |
| 3 | 1.2.3 |
| 4 | 1.2.4 |
| 5 | 2.1 |
| 6 | 2.2 |
| 7 | 2.3 |
| 8 | 2.4 |
| 9 | 3 |
| 10 | Full-scale WWTP (control comparison) |
| Stage | BOD | NH4-N | NO2-N | NO3-N | ENSND | PO4-P | |||
|---|---|---|---|---|---|---|---|---|---|
| Efficiency, % | Min., mg/L | Efficiency, % | Min., mg/L | Min., mg/L | Min., mg/L | Efficiency, % | Efficiency, % | Min., mg/L | |
| 1.1.1 | 97.8 | 2.8 | 98.5 | 0.26 | 0.05 | 6.4 | 21.3 | - | - |
| 1.1.2 | 96.9 | 6.9 | 92.9 | 0.78 | 0.04 | 5.1 | 49.5 | - | - |
| 1.2.2 | 98.0 | 2.5 | 99.5 | 0.31 | 0.05 | 35.0 | 69.7 | 35.9 | 9.2 |
| 1.2.4 | 97.8 | 2.1 | 98.1 | 0.26 | 0.07 | 25.6 | 77.5 | 37.2 | 7.8 |
| 2.2 | 97.8 | 1.2 | 97.6 | 0.85 | 0.01 | 17.7 | 37.8 | 6.9 | 12.1 |
| 2.3 | 98.0 | 1.3 | 96.9 | 0.58 | 0.01 | 11.7 | 63.1 | 23.8 | 8.2 |
| 3.1 | 96.4 | 4.1 | 97.2 | 0.21 | 0.02 | 17.7 | 29.5 | 23.6 | 8.3 |
| 3.2 | 97.8 | 2.2 | 98.9 | 0.37 | 0.03 | 8.4 | 69.4 | 36.3 | 7.8 |
| Refs | Organic Loading Rate, g/g/d | DO, mg/L | HRT, h | ENSND, % | RNSND, mg/L/h |
|---|---|---|---|---|---|
| [17] | 0.04 | 0.1–0.5 | 20.5 | 75.7 | 1.19 |
| [17] | 0.11 | 0.1–0.5 | 17.2 | 97.6 | 1.42 |
| [48] | 0.02–0.21 | >1.0 | 60.0 | 45 | 1.58 |
| [58] | 0.03 | 0.1 | 33.1 | 76 | 0.56 |
| [59] | 0.05 | <0.1 | 35.9 | 90.5 | 0.41 |
| [56] | - | Near 0 | 30–43 | 97.9 | 0.39 |
| [60] | 0.06 | 0.1 | 16.5 | 72.1 | 3.63 |
| [36] | 0.26 | 0.2 | 10.5 | 74.9 | 5.91 |
| This study | 0.23 | Near 1.0 | 12.0 | 69.4 | 1.59 |
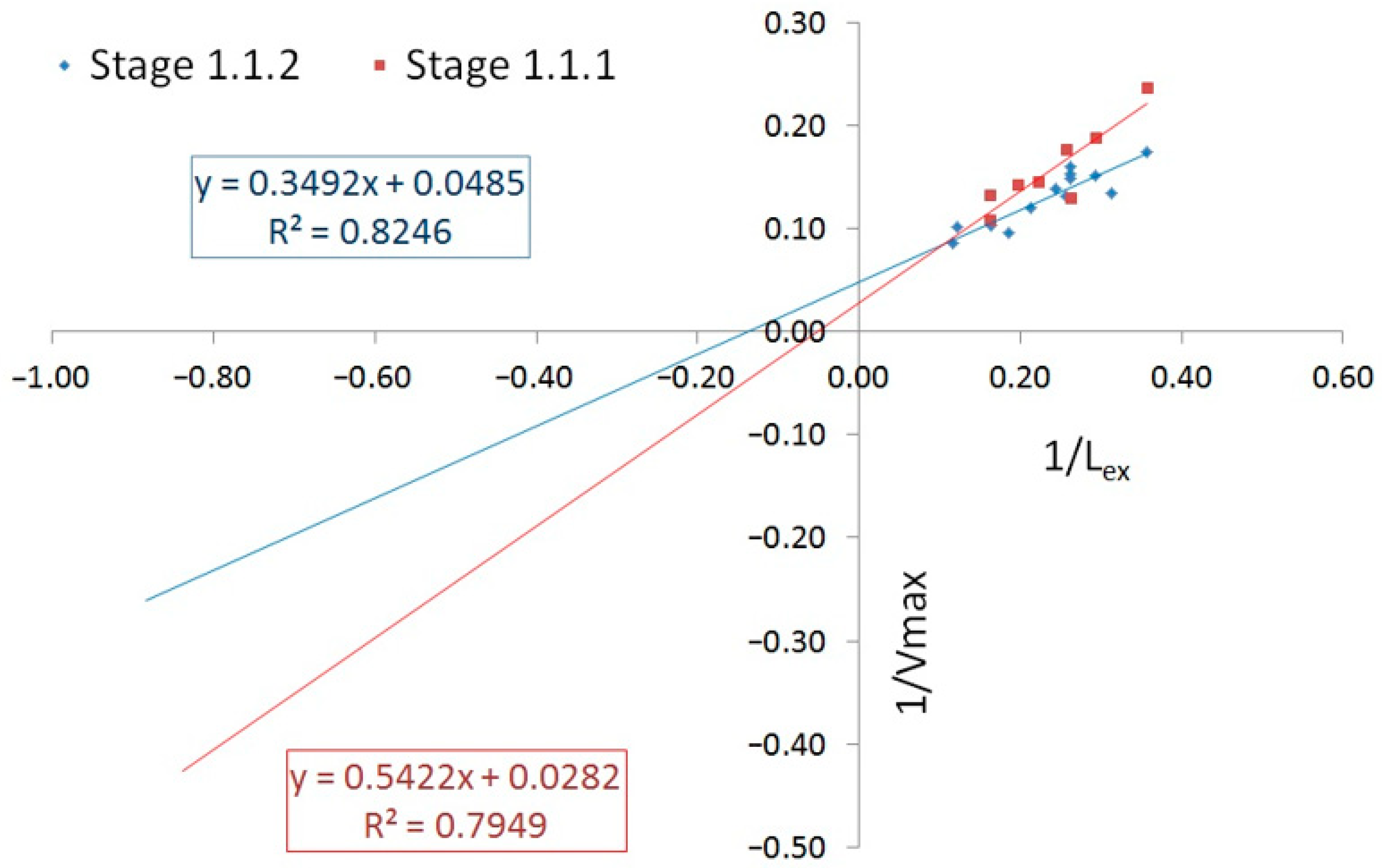
| Stage | Organic Matter Removal | Nitrification | Nitrification Rate, mg/L/h | Nitrate Formation Rate, mg/L/h | Specific Denitrification Rate, mg/g/h | ||
|---|---|---|---|---|---|---|---|
| Vmax | KM | Vmax | KM | ||||
| 1.1.1 | 35.46 | 19.23 | 4.35 | 0.03 | 8.15 | 7.91 | 6.15 |
| 1.1.2 | 20.61 | 7.2 | 6.03 | 0.35 | 6.51 | 6.66 | 8.56 |
| 1.2.2 | 35.71 | 3.64 | 5.03 | 0.08 | 9.15 | 9.11 | 8.51 |
| 1.2.4 | 30.67 | 2.53 | 4.58 | 0.04 | 8.62 | 8.73 | 9.28 |
| 2.2 | 33.44 | 2.87 | 5.63 | 0.23 | 8.99 | 8.76 | 11.12 |
| 2.3 | 14.35 | 0.61 | 4.76 | 0.08 | 8.15 | 7.91 | 6.15 |
| 3.1 | 33.90 | 8.98 | 4.22 | 0.02 | 7.53 | 7.62 | 8.98 |
| 3.2 | 27.17 | 6.29 | 4.44 | 0.04 | 9.15 | 9.11 | 8.51 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gogina, E.; Gulshin, I. Characteristics of Low-Oxygen Oxidation Ditch with Improved Nitrogen Removal. Water 2021, 13, 3603. https://doi.org/10.3390/w13243603
Gogina E, Gulshin I. Characteristics of Low-Oxygen Oxidation Ditch with Improved Nitrogen Removal. Water. 2021; 13(24):3603. https://doi.org/10.3390/w13243603
Chicago/Turabian StyleGogina, Elena, and Igor Gulshin. 2021. "Characteristics of Low-Oxygen Oxidation Ditch with Improved Nitrogen Removal" Water 13, no. 24: 3603. https://doi.org/10.3390/w13243603
APA StyleGogina, E., & Gulshin, I. (2021). Characteristics of Low-Oxygen Oxidation Ditch with Improved Nitrogen Removal. Water, 13(24), 3603. https://doi.org/10.3390/w13243603







