Abstract
Emerging pollutants (EPs), also known as micropollutants, have been a major issue for the global population in recent years as a result of the potential threats they bring to the environment and human health. Pharmaceuticals and personal care products (PPCPs), antibiotics, and hormones that are used in great demand for health and cosmetic purposes have rapidly culminated in the emergence of environmental pollutants. EPs impact the environment in a variety of ways. EPs originate from animal or human sources, either directly discharged into waterbodies or slowly leached via soils. As a result, water quality will deteriorate, drinking water sources will be contaminated, and health issues will arise. Since drinking water treatment plants rely on water resources, the prevalence of this contamination in aquatic environments, particularly surface water, is a severe problem. The review looks into several related issues on EPs in water environment, including methods in removing EPs. Despite its benefits and downsides, the EPs treatment processes comprise several approaches such as physico-chemical, biological, and advanced oxidation processes. Nonetheless, one of the membrane-based filtration methods, ultrafiltration, is considered as one of the technologies that promises the best micropollutant removal in water. With interesting properties including a moderate operating manner and great selectivity, this treatment approach is more popular than conventional ones. This study presents a comprehensive summary of EP’s existence in the environment, its toxicological consequences on health, and potential removal and treatment strategies.
1. Introduction
EPs have become a significant concern for the global population in recent years, owing to the possible dangers posed to the environment and human health. EPs, also known as micropollutants, are produced by various sources, including synthetic and natural substances. A new class of chemical compounds known as new EPs have recently been discovered in surface water, food sources, municipal wastewater, groundwater, and even drinking water. Such pollutants, known as EPs, are chemical composites that are commonly found in the environment, particularly in soil and aquatic bodies. However, they have only recently been identified as significant water contaminants. Personal care products (PCPs), hormones, flame retardants, industrial additives, endocrine-disrupting chemicals (EDCs), pharmaceuticals, nanomaterials, and pesticides are examples of EPs that are widely used and indispensable in modern society [1,2,3,4]. Based on the NORMAN network, at least 700 substances classified into 20 classes were identified in the European aquatic environment [5]. US Geological Survey characterized EP as “any compound of engineered or normal root or any microorganism that is not usually observed in the surrounding, however it can possibly cause unfriendly environmental and additionally human wellbeing impacts”. These contaminants are usually found in trace concentration from few parts per trillion to parts per billion [6]. According to Dulio et al. [7], the term “EPs” are substances that can persist in the environment, bioaccumulate, and potentially life-threatening, such as causing abnormal growth, reduced fertility and reproductive health, neurodevelopmental delays, inhibiting wildlife species, degrading aquatic ecosystems, and possibly harming the human immune system [8]. It is important to highlight that the majority of emerging contaminants are not new or recently introduced pollutants into the environment. Most emerging contaminants, on the other hand, are well-established pollutants with a newly documented harmful effect or mode of action. Therefore, the term “emerging” applies to both the contaminant and the issues that have arisen. Hence, emerging contaminants are also known as “contaminants of emerging concern” or “chemicals of emerging concern”. In a broader sense, emerging pollutants can be classified according to the following criteria: (i) not necessarily a new compound, (ii) a compound that has long existed in the environment but whose presence has only recently been detected and whose significance is beginning to be recognized, and (iii) a long-known compound whose potential negative impact on humans and the environment has only recently been realized. Table 1 lists the EP groups and their major compounds.

Table 1.
Emerging pollutants groups and their major compounds [9,10,11] (Reprinted by permission).
Water resources quality has deteriorated due to contamination caused by urbanization, rapid population growth, agricultural activities, and industrial development [12]. Heavy metals, microbial pollutants, priority contaminants, and nutrients are the most commonly studied aspects of water quality. Nonetheless, recent research [11,13] revealed the presence of organic pollutants that have a significant impact on water parameters. The main issue with EPs is a lack of understanding about their long-term effects on aquatic life, the environment, and human health. The discovery of numerous new compounds in drinking, ground, and surface water has alarmed the public, mainly when human health-based guidelines are unavailable [14,15]. Numerous studies were carried out to determine the contaminants concentrations and sources in receiving water bodies [16,17]. Due to the various concentrations and the lack of systematic monitoring programs, information about their transformation products, metabolites, and drinking water treatment is still limited. Also, since most EPs are not subject to water and wastewater regulations, there is little information or data involved in water resources.
Nevertheless, policymakers have recently agreed that EPs must be addressed systematically and coherently, although many remain unregulated. The European Union (EU), for example, has established a complex set of regulatory frameworks for EPs for governing activities involving the commercialization, use, presence, and emissions of chemical pollutants in the environment. Additionally, through their relevant agencies, the United States (US) excelled in continuous monitoring practices and maximum limit regulations [18].
Sutherland and Ralph [19] presented an extensive review on the microalgal bioremediation potentials of emerging pollutants. They demonstrated that microalgae have ability to concentrate, filter, eliminate or biotransform a wide range of emerging pollutants. Gogoi et al. [20] investigated the fate and occurrence of these contaminants in wastewater treatment plants and in the environment. The writers indicated out that future research should concentrate on the improvement of risk-based screening framework and models. [8] carefully reviewed some treatment technologies such as biological, phase-change, advance oxidation process for removal of emerging pollutants from water. In addition, microplastics are classified as emerging pollutants, the interaction between pollutants and microplastics was evaluated by Abaroa-Pérez et al. [21]. Khan et al. [22] considered pharmaceuticals as emerging major source of pollution for the environment. They considered effluent discharge from hospitals and emphasized the treatment processes such as activate sludge process, sequencing batch reactor, membrane biological reactor, activated carbon treatment, carbon nanotubes treatment, upflow anaerobic sludge blank, UV/H2O2, Fenton, and ozone treatment. A recent study by Roy et al. [23] considered antibiotics as only form of emerging water pollutant and focused on the treatment technologies such as photocatalytic degradation and in combination with nanomaterials. Monitoring, challenges of emerging pollutants, implementing efficient and ecological methods for their removal are well described by Vasilachi et al. [11].
Although huge scientific studies are available on several aspects concerning emerging pollutant’s monitoring, analysis, we consider that it is urgently needed to review these research efforts in a holistic way from occurrence, distribution, categorize to treatment of such emerging pollutants. To our best knowledge, there is not yet a review paper that structures all the research efforts related to the occurrence, effects of emerging pollutants in water environment and different technological options. As a result, this review script aims to bring comprehensive literature studies till date regarding the occurrences of emerging pollutants in water sources, distribution, analytical methods, toxicological effects and treatment processes.
This review paper gave insight into EPs in the water environment, which recently received much attention. In this paper, peer-reviewed scientific literature on emerging pollutants or emerging contaminants were reviewed with special regards to their occurrence, detection techniques, analytical methods, fate in the environment, and toxicity assessment. The discussion in this research also involved the current state of various water treatment processes for EP removal. This review demonstrated that EPs might pose a significant risk to consumers. However, there is substantially limited information on the by-products’ formation and their toxicity. Therefore, more research is required to better understand the EPs that exist in the water environment and elucidate the entire degradation pathway. A total of 4000 documents (among research articles and review papers) from Scopus databases were appeared while using keywords occurrence, detection techniques, analytical methods, fate in the environment, and toxicity assessment. The current study is prepared by reviewing 238 scientific articles, with 185 review publications and the rest are research publications. These studies are from different countries across the world, published between the 2011 to 2021.
2. Sources of EPs and Their Occurrence in Water Resources
Both surface and groundwaters have been found to contain EPs. Due to dilution and natural attenuation processes, their concentrations in surface waters are frequently lower than those recorded immediately at the outflow of wastewater and sewage treatment plants. Surface water, on the other hand, has higher EPs concentrations than groundwater since it receives effluent directly from the WWTP and has a shorter residence time. But, if the aquifer is close to pollution sources, groundwater concentrations could rise. A few researchers have discovered that the occurrence of certain PCPs and pharmaceuticals in surface water varies considerably. The variability is highly probable due to the usage frequency and dosages in different regions, including the effectiveness of the WWTP system [24]. Numerous factors influence the transport, fate, and occurrence of EPs in the surroundings. Among them are the physico-chemical properties of the environment and water, as well as longitude and latitude. Furthermore, the source type influences the exposure degree and the substance’s properties [9]. Figure 1 depicts the sources and possible EPs routes released into the atmosphere and dissolving into various receptors (ground, surface, and drinking water).
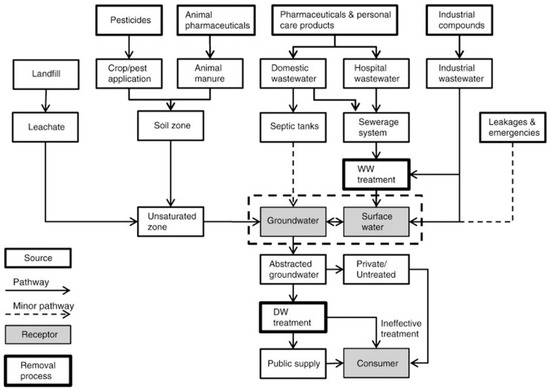
Figure 1.
Sources and potential routes of EPs in the water environment [25]. (Reproduced from [25] with Elsevier permission, license 5191080813331, from 16 November 2021).
2.1. Pharmaceuticals and Personal Care Products (PPCPs)
PCPs include various chemicals, including cosmetics and health care products (e.g., over-the-counter drugs, supplements, and prescription pharmaceuticals). After being released from leached farmland manure or sewage treatment plants, these pollutants end up in the community water systems and soil [26]. The pharmaceutical sector has a massive presence in WWTP operations [27]. Based on research made in the Ter River, Catalonia, Spain, the effect of effluent from WWTP is minimal compared to the river’s widespread presence of compounds. Likewise, research in groundwater on Cape Cod, Massachusetts, found that sewer systems from residential and industrial development, on top of other sources such as on-site wastewater treatment for nursing homes and health services, are a significant source on the incidence of EPs [28].
2.2. Antiseptics
On the other hand, triclosan is an antiseptic/antibacterial agent frequently found in household appliances, toothpaste, playthings, soaps, clothing, bedding, plastic, and fabric. Triclosan in tap water produces chloroform as one of the chlorinated by-products. Triclosan can break down in the environment to methyl triclosan or certain dioxins, particularly 2,4-dichlorophenol (2,4-DCP) and 2,8-dichlorodibenzo-p-dioxin (2,8-DCDD) [29]. The United States Geological Survey (USGS) of 95 different organic wastewater pollutants in the US streams revealed that at the highest concentrations, triclosan was one of the frequently detected compounds. Triclosan has also been discovered in numerous water bodies throughout the US, and researchers suspected that conventional treatment methods have not entirely removed it [30]. Additionally, the safety standards of triclosan in drinking water have yet to be established.
2.3. Hormones and Steroids
Among the various nonprescription and prescription or over-the-counter drugs are synthetic and natural steroid hormones such as gestagens, estrogens, and androgens [31]. These drugs are used for various purposes in humans and animals since they can modify their physiology and trigger essential regulatory functions in the body fluid. Ferguson et al. [32] discovered that in South-eastern Australia, the estrogens entering the estuary are most likely coming from a nearby WWTP. Its detection in freshwater indicates a secondary source, most likely from the agricultural area. Another study discovered that crop residues were the primary source of steroid estrogens, with the conjugated estrogens accounting for up to 22% of the total estrogen load from dairy farming. Furthermore, estrogens have been found in soil drainage water, streams draining stock grazing fields, runoff from dairy wastes application, on groundwater under unlined effluent holding ponds, and land [33].
2.4. Perfluoronated Compounds (PFCs)
PFCs are a type of compound that is applied in paints, food packaging, textiles, adhesives, polishes, waxes, electronics, and stain repellents, among other things. The most common are perfluorocarbon sulfonic acids (PFSAs) and perfluorinated carboxylic acids (PFCAs). In contrast, perfluoro octane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) are the most commonly known usage [34]. Due to releases from non-point sources, WWTPs, and industrial facilities, PFOA is detected in drinking and treated water sources [35]. It could also be present in drinking water wells via the contaminated groundwater plume passage.
Additionally, PFOA can enter groundwater through the air from nearby industrial sites before deposition onto soil and seepage into groundwater [36,37]. Pitter et al. [38] and Steenlan et al. [39] discovered that the release of PFOA in Ohio and West Virginia from an industrial facility could contaminate drinking water wells up to 20 miles away. This situation happened when PFOA-containing air pollution from an industrial source settled in the ground, accompanied by passage to groundwater and recharge the groundwater aquifer with contaminated surface water from the Ohio River.
2.5. Disinfection By-Products (DBPs)
On the other hand, the primary source of disinfection by-products (DBPs) came from the drinking water treatment plant (WTP) [40,41,42]. DBPs were determined in 15 WTPs in Beijing City from various water sources by Stalter et al. [40]. They discovered that halogenic acetic acids (HAAs) and trihalomethane (THMs) accounted for 38.1% and 42.6% of all DBPs, respectively, in all treated samples. DBPs in drinking water were found to vary with a water source, with surface water having the highest levels, mixed water sources, and groundwater having the lowest levels.
2.6. Pesticides
Pesticides can contaminate drinking water due to carelessness, such as back-siphoning, application in lawns and golf courses, and a sizeable accidental spill [43]. Pesticide metabolites have been highly prone to leaching in soil [44,45]. Reemtsma et al. [46] discovered pesticide metabolites were found in ground and surface water, with the diversity in the runoff samples attributed to multiple pesticide applications in minimal urban areas. Various metabolites in water matrices indicate that pesticides are widely used in city centers [46,47]. Belenguer et al. [48] discovered pesticides concentration and presence such as prochloraz, clofenvinphos, pyriproxyfen, imazalil, and dichlofenthion, in water are linked to intensive agricultural activities in the area. Table 2 shows a compilation of studies on EPs concentrations in various water bodies around the world.

Table 2.
Concentrations of EPs in water samples worldwide.
3. Toxicological Effects of EPs
EPs’ adverse effects have become a significant source of concern in society and the environment since they can cause cancer and endocrine disruption. EDCs are synthetic or natural chemicals that can block or mimic hormones and affect the living organisms’ endocrine systems [79]. This disruption could affect normal hormone levels, stimulating or inhibiting hormone production and metabolism [80,81]. EDCs are EP classes that include phthalates, polybrominated compounds, polychlorinated biphenyls (PCBs), steroid sex hormones, pesticides, pharmaceutical products, bisphenol A (BPA), alkylphenol ethoxylates, and alkylphenols. When a mother is exposed to EDCs such as BPA and phthalates, the sexual development of her offspring can be hampered [82,83,84]. Diethyltoluamide/insect repellents (DEET), ultraviolet (UV) screens, synthetic musk fragrances, and parabens are types of PCPs that may also act as EDC in water [81,85,86,87,88,89].
3.1. Hormones and Steroids
Several hormones, such as estrone (E1), 17-oestradiol (E2), 17-ethinylestradiol (EE2), estriol (E3), equilin, 17-oestradiol, norethindrone, equilenin, and mestranol, are listed as priority drinking water contaminants based on their health effects and environmental occurrence [90]. At low concentrations, 17-ethinylestradiol can cause estrogenic effects in fish. The impacts also include changes in sexual characteristics and sex ratios that cause decreasing egg fertilization in fish [90] and feminization in fish [91,92]. Androgen hormones also influence fish masculinization, whereas glucocorticoids impair reproduction and immune system development [93]. Endocrine disruptors, such as phytoestrogens, could also cause teratogenic, estrogenic, and other physiological problems in mammalian embryos and fish [94,95].
3.2. Antiseptics
Beauty and skincare products containing triclocarban and triclosan can cause the body’s hormonal or endocrine disruptions [96]. Furthermore, these ingredients are harmful to fish embryos [97], as well as algae, crustaceans, and fish [98,99]. Triclosan, as per [100], can disrupt the reproductive axis and thyroid hormone homeostasis. Triclosan also has been shown to harm phytoplankton accumulation in freshwaters at specific concentrations in the environment. Long-term exposure to parabens, even at low concentrations, might cause vitellogenin synthesis in fish [101].
3.3. Plasticizers
Since BPA is among the most commonly used industrial added chemicals, its adverse health effects have been extensively researched. Rochester et al. [102] and Weber et al. [103] conducted a comprehensive review of the impact of BPA on human health and wildlife. BPA has been linked to lower sperm quality, fertility, sex hormone concentrations, and self-reported sexual function in men. In women, exposure is linked to polycystic ovary syndrome, breast cancer, miscarriage, endometrial disorders, and premature births.
3.4. Flame Retardants
In breast milk, the levels of polybrominated diphenyl ethers (PBDEs) are linked to lower body mass and birth weights, as well as cryptorchidism (undescended testicles) in newborn boys [104,105]. Dorman et al. [106] discovered PBDE levels in humans and animals that cause neurotoxicity development in animal model experiments, implying that PBDEs may have a similar effect on humans. Due to their lipophilicity, PBDEs are known to bioaccumulate in fat tissue. They have also been linked to harmful animal health risks such as fetal malformations, hormone disruption, and decreased sperm count [29].
3.5. Disinfection By-Products (DBPs)
Congenital disabilities, early-term miscarriage, and bladder cancer have been associated with DBPs in drinking water. Meanwhile, DBPs in swimming pools can cause respiratory problems or asthma [107]. The cancer risk from inhalation, ingestion, and dermal contact exposure to THMs was evaluated by Gan et al. [108]. They discovered that bromodichloromethane (CHCl2Br) led to the most cancer risk from the ingestion pathway, while chloroform (CHCl3) contributed the most cancer risk from the inhalation pathway.
3.6. Pesticides
Pesticides include dichlorodiphenyltrichloroethane (DDT), chlordane, vinclozolin, endosulfan and dieldrin, aldrin, and atrazine. Those components can disrupt the endocrine system by causing hormonal imbalances [109]. Moreover, organochlorine accumulation was linked to a higher risk of several types of genotoxicity, human cancer, and mental and psychomotor development [110]. Nanomaterials have been shown in toxicological studies to be neurotoxic, cytotoxic, genotoxic, bactericidal, and ecotoxic. In rodents and humans, anatase (TiO2), zinc oxide (ZnO), and nano-sized silicon dioxide (SiO2) could cause pulmonary inflammation [111]. Moreover, once released into the environment, Argentum, a silver nanoparticle, could disrupt ecological equilibrium [112]. Table 3 shows the additional harmful effects of EPs on the environment and their exposure limits.

Table 3.
Toxicological effects of emerging pollutants.
4. Analytical Methods of EPs
In conventional ways targeting at emerging contaminants, advanced ultra-sensitive instrumental techniques such as electrospray ionisation-mass spectrometry (ESI(NI)-MS), Liquid chromatography -tandem mass spectrometry (LC-MS/MS), PTV-GC-EI-MS, gas chromatography-nitrogen-phosphorus detection (GC-NPD) etc. are commonly practised but not used for monitoring periodically. The use of such analytical instruments offer higher possibilities for detection of multiple emerging pollutants, and improve the detection limits even with very low concentration [125,126]. Several sample preparation techniques are available for various emerging contaminant’s extraction from water. However, SPE is one of methods which is well adapted for a wide range of analyte’s analysis, having dissimilar physical, chemical properties, and polarities. Moreover, SPE has a variety of available sorbents, their higher capacity compared to liquid–liquid extraction (LLE) [127]. Table 4 shows some analytical methods used so far for the extraction of EP samples.

Table 4.
Analytical methods used for quantitative analysis of emerging pollutants.
LC–MS/MS has several advantages; can be applied to a wide range of biological molecules and has fast scanning speeds. ESI(NI)-MS was used by several researcher since this analytical instrument can analyze large masses. Through PTV-GC-EI-MS, analytical sensitivity is greatly enhanced for analytes with low concentrations [128]. The efficiency of extraction and the sensitivity and selectivity enable detection at levels below the EU regulatory level of 0.1 μg/L−1 by GC-NPD and GC-MS/MS [131].
5. Treatment Methods for EPs Removal
The removal of EPs from the water and the potential formation of disinfection by-products determines the quality of drinking water supplies. EPs can be removed using various treatment methods, including biological, physico-chemical, and oxidation methods. Even so, most of the treatment methods have disadvantages, such as secondary pollution, high maintenance cost, and complicated procedures in the treatment [133]. Typical drinking WTPs (chlorination, filtration, and coagulation-flocculation) are less efficient at entirely removing EPs, such as PCPs, selected pharmaceuticals, and atrazine [97]. Research also discovered that metal salt coagulants (ferric sulfate and aluminium sulfate) were ineffective at removing compounds such as trimethoprim, sulfadimethoxine, and carbadox [134]. However, some studies demonstrated that treatment methods such as adsorption, nanofiltration (NF), and reverse osmosis (RO) on powdered activated carbon (PAC) and granular activated carbon (GAC) effectively removed the pollutants [81,87,88]. Recently, electrochemical oxidation (EO), along with other advanced oxidation processes (AOP), have been considered a promising technique for removing CEC from water and wastewater [135].
5.1. Biological Treatment
Since the early 1900s, microbial biomass used to degrade nutrients, contaminants, and organics in wastewater. Contrarily, the use of such biological treatment in drinking water is less common. Nonetheless, recent advancements are starting to expand the favorability, possibility, and applicability of biological treatment technologies of drinking water. These advancements include: (1) the high complexities and rising costs of managing water treatment for residuals, which is membrane concentrates; (2) the drive for a technique that effectively destroys contaminants rather than concentrating them (i.e., green technologies); (3) the introduction of new contaminants that are highly prone to biological degradation, such as perchlorate; (4) the emergence of membrane-based treatment systems that are highly vulnerable to biological fouling; and (5) regulations that limit DBPs formation [136,137,138].
Only polar contaminants discharged in the final effluent were removed during biological treatment [139]. It is well understood that the conventional activated sludge (CAS) is the most cost-effective method to degrade and eliminate contaminants. However, it does not eradicate micropollutants in sewage treatment [140,141]. In Europe, activated sludge with a 14-h hydraulic contact time could remove approximately 85% of estriol, 17β-estradiol, estrone, and mestranol [142]. Less than 10% of synthetic and natural estrogens were removed during the biodegradation process [143]. While some of the components were absorbed in the sludge, the majority were still soluble in the effluent. The use of CAS to treat pharmaceutical industry wastewater necessitated a lengthy hydraulic retention time [144,145]. Due to the limited operational requirements, the capital cost is cheaper when compared to advanced treatment. Even though CAS is less harmful to the environment compared to chlorination, it has some disadvantages, such as a tendency to produce higher amounts of sludge [146], high energy consumption, as well as possible formation of foam, color, and bulk sludge in secondary clarifiers [145].
Membrane bioreactors (MBRs) are another biological treatment efficient in eliminating bulk organics and could be used as hybrid systems or in conjunction with CAS. The primary advantages of MBRs over CAS are their capability to treat various wastewater compositions [147] and meet small footprint requirements [148]. MBR has been shown to achieve high biological oxygen demand (BOD) and chemical oxygen demand (COD) removal in pharmaceutical manufacturing facilities [149]. Research has found that membrane bioreactors could be used to remove estrogens from wastewater, moderate efficiency of 17β-estradiol was effectively removed up to 67% and estrone up to 91% [150]. Estrone-3- glucuronide, 17-estradiol-glucuronides, and estrone-3-sulfate, on the other hand, were not significantly removed.
Enzyme-based treatment processes are used in a recent biological treatment that has the potential to remove EPs. Under optimal conditions, [151] discovered that laccase treatment in the presence of the natural redox mediator syringaldehyde completely removed diclofenac cytotoxicity. Furthermore, syringaldehyde and laccase were successfully converted the triclosan dichlorination product into 2-phenoxy phenol as a non-toxic polymer [152]. Laccase-poly (lactic-co-glycolic acid) nanofiber in the presence of syringaldehyde can be a viable method for diclofenac detoxification and removal from aqueous sources [153]. Recent research has discovered that a combination of cross-linked enzyme aggregate (combi-CLEA) associated with enzymatic cascade reaction could remove pharmaceutically active compounds (PhACs) with a high removal rate of more than 80% [154]. Since it is environmentally friendly, enzymes application for treating EPs has future potential.
5.2. Physico-Chemical Treatment
Physico-chemical processes are typically used in conventional water treatment to eliminate pathogens, control taste and odour issues, and reduce turbidity. These processes may also have the added benefit of lowering a load of micropollutants in finished drinking water. However, the removal is frequently insufficient [155]. Activated carbon, coagulation-flocculation, and membrane filtration are all part of the physico-chemical process. Coagulation-flocculation is a standard physico-chemical process required for water treatment. Current research on one of the emerging DBPs (halo-benzoquinone) revealed that coagulation could not wholly eliminate the precursor components from drinking water sources [156].
Meanwhile, a different study found that reducing halogenic acetic acids (HAAs) and THMs precursors by poly aluminum chloride under improved coagulation could only achieve the highest removal rates of 59% and 51%, respectively [157]. The adsorption process with activated carbon has emerged as a viable option for treating purifying drinking water and industrial wastewater [158]. The most frequently used adsorbent for treating biological and chemical pollutants in raw drinking water and industrial wastewaters are PAC or GAC. The application is favourable since their porosity, surface area, and chemistry are highly developed [159,160].
The adsorption process is prevalent and influential in water treatment since it is simple to design, produces no undesirable by-products, and is insensitive to toxic substances [161]. Although activated carbons are preferred to remove various EPs, their use is sometimes limited due to their high cost. Even after it has been depleted, activated carbon can be regenerated for future use. However, carbon will be lost during the regeneration process, and the resulting product might have a lower adsorption capacity compared to the freshly prepared activated carbon [162]. Natural water spiked with 30 pharmaceuticals and a bench-scale WTP simulation model were used to test 80 different EDCs. Except for oxidation via ozonation and chlorination and PAC, Leusch et al. [87] discovered no significant compound removal. Following that, Abd El-Gawad el al. [88] and Bolong et al. [163] confirmed that GAC regeneration or PAC dose is required to get a high removal rate.
RO and NF have received much attention in recent years. Membrane filtration technologies are now widely used to treat wastewater reclamation and drinking water as they effectively remove most inorganic and organic compounds [164,165]. The use of NF and RO membranes is a promising technology for removing EPs [85,166]. Nonetheless, the RO membrane is far more effective than the NF at removing EPs, but RO requires more energy during the process, making it less desirable.
Numerous studies have discovered that using RO and NF membranes in water treatment plants effectively removes EDCs and PCPs, sometimes by up to 95% [167,168]. Also, a different study demonstrated that the NF application in aqueous solutions could effectively remove pesticides (simazine, atrazine, and diuron) as well as atrazine metabolite (DEA) [169]. For all solutes, the NF membrane outperforms particle coagulation-flocculation, dual media filtration, and sedimentation, with atrazine, has the highest retention rate, while diuron has the lowest [170]. The NF membrane was also used in another study to eliminate organic chlorine pesticides such as DDT from drinking water. The outcomes showed that DDT could be efficiently eliminated from water using an NF membrane, with up to a 95% removal rate [171].
Subsequently, Acero et al. [172] reported that regardless of the water matrix, the ultrafiltration (UF) membrane was the best for removing the selected herbicides. The NF and UF membranes proved to eliminate more than 90% BPA in drinking water with concentrations ranging from 60–600 µg/L. The mixture of humic acid and BPA hydrophobic adsorption mechanism was discovered to be effective for BPA retention [173,174]. Due to its size, it may be challenging to remove nanomaterials from water using conventional filtration without pre-coagulation [175]. However, it was determined that the UF membrane removed more than 99.6% of the SiO2 nanoparticles [176]. The use of membrane filtration as the final process could improve the removal efficiencies for nanomaterials. Nonetheless, the membrane filtration process has the disadvantage of fouling, which causes flux reduction and increases operational costs [177].
5.3. Oxidation Treatment
Oxidation, which uses chemical oxidants such as ozone (O3) and chlorine, is one of the fundamental techniques for removing EPs. The chemical reactions in water can be reactive, resulting in by-products. Therefore, before selecting this treatment, a careful selection of chemical oxidants is required. Because of its high oxidation capability, O3 has been widely used in water treatment for color removal, disinfection, the degradation of many organic contaminants, and taste and odor control for drinking water. O3 will react with organic pollutants directly or indirectly with molecular O3 and free radicals (hydroxyl radical OH) produced during O3 decomposition, respectively [178].
A study conducted by de Jesus Gaffney et al. [179] discovered that an O3 concentration of 2.5 mg/L could treat raw water at 20 different drinking WTPs across the States. In contrast, UV and chlorine concentrations of 40 mJ/cm2 and 2.5 mg/L, respectively, were less effective. Chemical oxidation with O3 is also efficiently treating various organic micropollutants in bench-, pilot- and full-scale drinking water and wastewater experiments [180,181,182,183,184,185].
Due to the enhanced generation of hydroxyl radicals and photon-initiated cleavage of carbon-halogen bonds, combining O3-based advanced oxidation processes (AOPs) such as O3/UV, photochemical, Fenton-type techniques, and O3/H2O2 is more efficient than ozonation alone. Hence, AOPs are favored to treat recalcitrant compounds [184]. Ionizing radiation was considered as an attractive option among the available AOPs to degrade various toxic organic pollutants, such as nitrophenols [186,187], chlorophenols [188,189], and antibiotics [190,191] in aqueous solution, which draws concern in many countries.
The benefits of ionizing radiation technology include: (1) good penetration range in the water matrix; (2) having no additional chemicals; (3) being insensitive to color and suspended particles; and (4) the recalcitrant compounds may be degraded in situ by reactive species formed during water radiolysis [192,193]. The high price of radioisotopes, as well as safety concerns, are significant factors limiting their application. Two types of irradiators are commonly used, namely gamma (ɣ) sources (137Cs or 60Co) and electron beam accelerators (EB) [194]. The dose rate of EB is high, and ɣ rays are extremely penetrating. Moreover, the cost of energy for ɣ irradiation is significantly higher [195]. As a result, EB is a viable candidate for practical application to alleviate public concerns about radioisotope safety measures [196].
UV photolysis (Suntest apparatus, Xe, and Hg lamps) has been successfully used to degrade organophosphorus pesticides, revealing mechanisms, diverse kinetics, and by-product formation. Although more toxic oxons were present in some cases [197], several pesticides were effectively degraded by low-pressure UV photolysis, such as pentachlorophenol, diuron, atrazine, clofenvinphos, and alachlor [198]. Nevertheless, pesticides in an aqueous solution could only be slightly degraded by a simple photolysis process. These lighting sources outperformed the photolysis process alone when combined with H2O2 or Fe (III) [199].
TiO2 is a semiconductor catalyst that has been extensively studied in heterogeneous photocatalytic processes, making it one of the advanced oxidation treatments’ options (AOT). Chemical stability, non-toxicity, and low cost are all advantages of TiO2. However, TiO2 has the disadvantage of being in powder form, necessitating a separation stage after treatment in order to employ it as a photocatalytic material in wastewater purification by photocatalytic treatment. According to the findings of the study by Borges et al. [200], TiO2 has strong photocatalytic activity for the removal of paracetamol from wastewater, with a removal rate of 99–100% after 4 h of irradiation.
5.4. Combine Treatment Processes
During water treatment at laboratory and pilot plant scales, numerous researchers have investigated some pharmaceutical drugs removal. Using a combination of treatment methods such as activated carbon adsorption, chlorine or O3 oxidation, RO, and filtration, 90% of the antibiotics were removed [61,201,202,203,204,205]. Nevertheless, integrating specific treatment processes such as coagulation-flocculation with iron salts or aluminum and UV disinfection treatments did not achieve satisfactory contaminant removal levels [134]. Certain pharmaceuticals (ibuprofen, clofibric acid, and diclofenac) cannot be removed using flocculation with activated carbon adsorption alone [206]. Advanced oxidation processes using hydrogen peroxide (H2O2) and O3 at 1.8 mg/L and 5 mg/L, respectively, achieved significant removals.
A study was conducted by Kovalova et al. [181] on a pilot plant scale in Germany to eliminate selected pharmaceuticals (diclofenac, clofibric acid, bezafibrate, and carbamazepine) from the source of drinking water. Either sand filtration in both anoxic and aerobic conditions or flocculation using iron (III) chloride (FeCl3) were ineffective in removing the desired pharmaceuticals. On the contrary, it was discovered that ozonation is very selective in removing these polar compounds. The presence of psychoactive stimulatory drugs in raw and finished drinking water from a Spanish drinking WTP was assessed by Watanabe et al. [207]. They discovered that amphetamine-type stimulants (except MDMA (ecstasy)) were wholly eliminated during pre-chlorination, flocculation, and sand filtration steps, resulting in concentrations lower than their limits of detection (LODs).
Moreover, combined treatment methods of ozonation activated carbon adsorption and coagulation-flocculation (with FeCl3) in removing EPs in pilot and drinking WTP was evaluated by Ternes et al. [180]. The EPs include bezafibrate, carbamazepine, diclofenac, and clofibric acid. The finding showed that the combined ozonation and activated carbon adsorption process successfully removed the pollutants.
A different study conducted by Luine et al. [208] on estrogen sorption and coagulation elimination performance processes using activated carbon was compared. Even in a hybrid system with NF membranes, they revealed that sorption by GAC and PAC was more effective compared to coagulation. A removal of 17α-ethinylestradiol (EE2) and 17β-estradiol (E2) in MBRs with and without PAC addition was investigated by Yang et al. [209]. The MBR alone removed EE2 and E2 at rates of 70.9% and 89.0%, respectively. With PAC, however, the EE2 and E2 rates of removal increased by 15.8% and 3.4%, respectively. The research also found that biodegradation was the most common method for removing EE2 and E2 in MBRs [209].
Subsequently, the oxidation of two pesticides (trifluralin and bromoxynil) in natural waters in a batch using O3 and O3 combined with H2O2 was investigated by Chelme-Ayala et al. [210]. The degradation levels for both pesticides were less than 50% based on the results. Nevertheless, the combined O3/H2O2 process increased the degradation level. It was also discovered that adding a photocatalyst of titanium dioxide (TiO2) to a coagulation-flocculation process increased the industrial chemical (1,4-dioxane) removal rate by two-fold in an hour. Moreover, at a UV dose of 0.35 WL−1, a continuous flow reactor with a residence time of 39 min removed more than 60% of the 1,4-dioxane [211]. Pesticides, over time, may pose a risk to human health via water and the atmosphere. Conventional portable water treatment methods, such as sedimentation, coagulation-flocculation, and dual media filtration, are less effective at removing pesticides residues [212]. By incorporating more advanced processes before pre-treatment, such as oxidation of O3 or H2O2, membrane filtration, or granular activated carbon filtration, advanced water treatment could enhance the efficacy of typical water treatment [212,213,214,215].
Table 5 depicts the available water treatment processes for EPs as well as their removal performance. The CW had a removal efficiency of 42%, the AS had 62%, the RBC had 63%, and the WSP had 82%. Except for the WSP system, all of these technologies demonstrated seasonal variability in removing emerging contaminants. The WSP is the only system that is safe in both seasons, whereas all methods could potentially reduce the aquatic risk, according to an ecotoxicological assessment study [216]. During UV/O3 treatment, a synergistic impact between O3 and UV was detected in the selected trace antibiotics degradation process [177].

Table 5.
Treatment processes for removing emerging pollutants.
UF, NF, and NH4Cl-induced activated carbon (NAC) have been used to remove pesticides such as phenyl-urea [171], diuron [217], and diazinon [218]. The UV/H2O2 technique was investigated to remove 6 iodinated trihalomethanes (6 ITHMs), EE2, paroxetine, venlafaxine, pindolol, bezafibrate sotalol, and metformin. The 6 ITHMs degradation rates in the UV/H2O2 system were significantly faster than in the UV system without H2O2 [219]. Dichloroacetic acid (DCAA) decomposition in water using a UV/H2O2/micro-aeration process was also analyzed [220]. DCAA was unremovable by H2O2 oxidation, UV radiation, or micro-aeration. Still, an integrated technique of UV/H2O2/micro-aeration was practical and could completely degrade DCAA.
6. Conclusions
Current knowledge of the risks of EPs to the environment and human health has been carried out. However, there is still a gap in our understanding of EPs’ long-term effects. EPs’ fate and adverse impacts on human health and aquatic life are restricted and sparse, necessitating a greater study and knowledge. Due to the rapid development of EPs in the environment, implementing cost-effective and sustainable detection, risk assessment, and removal programs for all of these elements is difficult.
In the meantime, EPs have become a challenge in sustainable water management, where climate change and population growth exacerbate water sources issues. The primary concern with these contaminants is their micro size, as they are not effectively eliminated by conventional water treatment. WWTPs are critical in separating contaminants before they are discharged into the river system. Some, though, are very persistent in the water and hard to remove or biodegrade quickly. Pollutant residues will spread in the atmosphere and contaminate drinking water sources.
Therefore, future research initiatives should emphasis the pollutants that have the greatest impact on human health and the aquatic environment, allowing for integrated research to alleviate pollution inputs while optimizing available resources.
Author Contributions
Conceptualization, M.R.S., S.S., and M.S.M.; writing—original draft preparation, M.S.M., N.Z.A., and T.H.N.; writing—review and editing, N.Z.A., T.H.N., M.R.S., and S.S.; supervision, M.R.S., M.M., S.S., and A.A. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Water Security and Sustainable Development Hub funded by the UK Research and Innovation’s Global Challenges Research Fund (GCRF) [grant number: ES/S008179/1] and Universiti Teknologi Malaysia (UTM), grant number R.J130000.7609.4C241.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
List of emerging pollutant abbreviations used throughout the manuscript:
| Abbreviation | Full form |
| 6 ITHM | 6 iodinated trihalomethanes |
| AOP | Advanced oxidation processes |
| AS | Aeration system |
| BOD | Biological oxygen demand |
| BPA | Bisphenol A |
| CAS | Conventional activated sludge |
| CHCl2Br | Bromodichloromethane |
| CHCL3 | Chloroform |
| COD | Chemical oxygen demand |
| CW | Constructed wetlands |
| DBP | Disinfection by-product |
| DCAA | Dichloroacetic acid |
| DDT | Dichlorodiphenyltrichloroethane |
| DEA | Atrazine metabolite |
| DEET | Diethyltoluamide |
| DES | Diethylstilbestrol |
| DWTP | Drinking water treatment plants |
| E1 | Estrone |
| E2 | 17β-estradiol |
| E3 | Estriol |
| EDC | Endocrine disrupting compound |
| EE2 | 17α-ethinylestradiol |
| EV | Estradiol valerat |
| GAC | Granular activated carbon |
| HAA | Halogenic acetic acid |
| H2O2 | Hydrogen peroxide |
| Hg | Mercury |
| LOD | Limits of detection |
| MBR | Membrane bioreactor |
| NAC | NH4Cl- induced activated carbon |
| NF | Nanofiltration |
| PAC | Powdered activated carbon |
| PBDE | Polybrominated diphenyl ethers |
| PCB | Polychlorinated biphenyl |
| PCP | Personal care product |
| PFC | Perfluorinated chemical |
| PFOA | Perfluorooctanoic acid |
| RBC | Rotating biological contactor |
| RO | Reverse osmosis |
References
- Su, C.; Cui, Y.; Liu, D.; Zhang, H.; Baninla, Y. Endocrine Disrupting Compounds, Pharmaceuticals and Personal Care Products in the Aquatic Environment of China: Which Chemicals Are the Prioritized Ones? Sci. Total Environ. 2020, 720, 137652. [Google Scholar] [CrossRef]
- Birch, G.F.; Drage, D.S.; Thompson, K.; Eaglesham, G.; Mueller, J.F. Emerging Contaminants (Pharmaceuticals, Personal Care Products, a Food Additive and Pesticides) in Waters of Sydney Estuary, Australia. Mar. Pollut. Bull. 2015, 97, 56–66. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Lai, W.W.-P.; Tung, H.-H.; Lin, A.Y.-C. Occurrence of Pharmaceuticals, Hormones, and Perfluorinated Compounds in Groundwater in Taiwan. Environ. Monit. Assess. 2015, 187, 256. [Google Scholar] [CrossRef]
- Lin, T.; Yu, S.; Chen, W. Occurrence, Removal and Risk Assessment of Pharmaceutical and Personal Care Products (PPCPs) in an Advanced Drinking Water Treatment Plant (ADWTP) around Taihu Lake in China. Chemosphere 2016, 152, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Peña-Guzmán, C.; Ulloa-Sánchez, S.; Mora, K.; Helena-Bustos, R.; Lopez-Barrera, E.; Alvarez, J.; Rodriguez-Pinzón, M. Emerging Pollutants in the Urban Water Cycle in Latin America: A Review of the Current Literature. J. Environ. Manag. 2019, 237, 408–423. [Google Scholar] [CrossRef] [PubMed]
- Sivaranjanee, R.; Kumar, P.S. A Review on Remedial Measures for Effective Separation of Emerging Contaminants from Wastewater. Environ. Technol. Innov. 2021, 23, 101741. [Google Scholar] [CrossRef]
- Dulio, V.; van Bavel, B.; Brorström-Lundén, E.; Harmsen, J.; Hollender, J.; Schlabach, M.; Slobodnik, J.; Thomas, K.; Koschorreck, J. Emerging Pollutants in the EU: 10 Years of NORMAN in Support of Environmental Policies and Regulations. Environ. Sci. Eur. 2018, 30, 1–13. [Google Scholar] [CrossRef]
- Rodriguez-Narvaez, O.M.; Peralta-Hernandez, J.M.; Goonetilleke, A.; Bandala, E.R. Treatment Technologies for Emerging Contaminants in Water: A Review. Chem. Eng. J. 2017, 323, 361–380. [Google Scholar] [CrossRef] [Green Version]
- Stefanakis, A.I.; Becker, J.A. A Review of Emerging Contaminants in Water: Classification, Sources, and Potential Risks. In Practice, Progress, and Proficiency in Sustainability; IGI Global: Hershey, PA, USA, 2015; pp. 55–80. [Google Scholar]
- Starling, M.C.V.M.; Amorim, C.C.; Leão, M.M.D. Occurrence, control and fate of contaminants of emerging concern inenvironmental compartments in Brazil. J. Hazard. Mater. 2019, 372, 17–36. [Google Scholar] [CrossRef] [PubMed]
- Vasilachi1, I.C.; Asiminicesei, D.M.; Fertu, D.I.; Gavrilescu, M. Occurrence and Fate of Emerging Pollutants in Water Environment and Options for Their Removal. Water 2021, 13, 181. [Google Scholar] [CrossRef]
- Patel, M.; Kumar, R.; Kishor, K.; Mlsna, T.; Pittman, C.U., Jr.; Mohan, D. Pharmaceuticals of Emerging Concern in Aquatic Systems: Chemistry, Occurrence, Effects, and Removal Methods. Chem. Rev. 2019, 119, 3510–3673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karpińska, J.; Kotowska, U. Removal of Organic Pollution in the Water Environment. Water 2019, 11, 2017. [Google Scholar] [CrossRef] [Green Version]
- Khatib, J.M.; Baydoun, S.; ElKordi, A.A. Water Pollution and Urbanisation Trends in Lebanon: Litani River Basin Case Study. In Urban Pollution; John Wiley & Sons, Ltd.: Chichester, UK, 2018; pp. 397–415. [Google Scholar]
- Baken, K.A.; Sjerps, R.M.A.; Schriks, M.; van Wezel, A.P. Toxicological Risk Assessment and Prioritization of Drinking Water Relevant Contaminants of Emerging Concern. Environ. Int. 2018, 118, 293–303. [Google Scholar] [CrossRef]
- Lim, F.; Ong, S.; Hu, J. Recent Advances in the Use of Chemical Markers for Tracing Wastewater Contamination in Aquatic Environment: A Review. Water 2017, 9, 143. [Google Scholar] [CrossRef]
- Huerta, B.; Rodriguez-Mozaz, S.; Nannou, C.; Nakis, L.; Ruhí, A.; Acuña, V.; Sabater, S.; Barcelo, D. Determination of a Broad Spectrum of Pharmaceuticals and Endocrine Disruptors in Biofilm from a Waste Water Treatment Plant-Impacted River. Sci. Total Environ. 2016, 540, 241–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vargas-Berrones, K.; Bernal-Jácome, L.; Díaz de León-Martínez, L.; Flores-Ramírez, R. Emerging Pollutants (EPs) in Latin América: A Critical Review of under-Studied EPs, Case of Study-Nonylphenol. Sci. Total Environ. 2020, 726, 138493. [Google Scholar] [CrossRef]
- Sutherland, D.L.; Ralph, P.J. Microalgal Bioremediation of Emerging Contaminants—Opportunities and Challenges. Water Res. 2019, 164, 114921. [Google Scholar] [CrossRef] [PubMed]
- Gogoi, A.; Mazumder, P.; Tyagi, V.K.; Tushara Chaminda, G.G.; An, A.K.; Kumar, M. Occurrence and Fate of Emerging Contaminants in Water Environment: A Review. Groundw. Sustain. Dev. 2018, 6, 169–180. [Google Scholar] [CrossRef]
- Abaroa-Pérez, B.; Sánchez-Almeida, G.; Hernández-Brito, J.J.; Vega-Moreno, D. In Situ Miniaturised Solid Phase Extraction (m-SPE) for Organic Pollutants in Seawater Samples. J. Anal. Methods Chem. 2018, 2018, 7437031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, N.A.; Khan, S.U.; Ahmed, S.; Farooqi, I.H.; Yousefi, M.; Mohammadi, A.A.; Changani, F. Recent Trends in Disposal and Treatment Technologies of Emerging-Pollutants—A Critical Review. Trends Analyt. Chem. 2020, 122, 115744. [Google Scholar] [CrossRef]
- Roy, N.; Alex, S.A.; Chandrasekaran, N.; Mukherjee, A.; Kannabiran, K. A Comprehensive Update on Antibiotics as an Emerging Water Pollutant and Their Removal Using Nano-Structured Photocatalysts. J. Environ. Chem. Eng. 2021, 9, 104796. [Google Scholar] [CrossRef]
- Munthe, J.; Brorström-Lundén, E.; Rahmberg, M.; Posthuma, L.; Altenburger, R.; Brack, W.; Bunke, D.; Engelen, G.; Gawlik, B.M.; van Gils, J.; et al. An Expanded Conceptual Framework for Solution-Focused Management of Chemical Pollution in European Waters. Environ. Sci. Eur. 2017, 29, 13. [Google Scholar] [CrossRef] [Green Version]
- Stuart, M.; Lapworth, D.; Crane, E.; Hart, A. Review of risk from potential emerging contaminants in UK groundwater. Sci. Total Environ. 2012, 416, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Parra-Saldivar, R.; Castillo-Zacarías, C.; Bilal, M.; Iqbal, H.M.N.; Barceló, D. Sources of Pharmaceuticals in Water. In The Handbook of Environmental Chemistry; Springer: Berlin/Heidelberg, Germany, 2020; pp. 33–47. [Google Scholar]
- Palli, L.; Spina, F.; Varese, G.C.; Vincenzi, M.; Aragno, M.; Arcangeli, G.; Mucci, N.; Santianni, D.; Caffaz, S.; Gori, R. Occurrence of Selected Pharmaceuticals in Wastewater Treatment Plants of Tuscany: An Effect-Based Approach to Evaluate the Potential Environmental Impact. Int. J. Hyg. Environ. Health 2019, 222, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Schaider, L.A.; Rodgers, K.M.; Rudel, R.A. Review of Organic Wastewater Compound Concentrations and Removal in Onsite Wastewater Treatment Systems. Environ. Sci. Technol. 2017, 51, 7304–7317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gwenzi, W.; Mangori, L.; Danha, C.; Chaukura, N.; Dunjana, N.; Sanganyado, E. Sources, Behaviour, and Environmental and Human Health Risks of High-Technology Rare Earth Elements as Emerging Contaminants. Sci. Total Environ. 2018, 636, 299–313. [Google Scholar] [CrossRef]
- Weatherly, L.M.; Gosse, J.A. Triclosan Exposure, Transformation, and Human Health Effects. J. Toxicol. Environ. Health B Crit. Rev. 2017, 20, 447–469. [Google Scholar] [CrossRef]
- Wöhler, L.; Niebaum, G.; Krol, M.; Hoekstra, A.Y. The Grey Water Footprint of Human and Veterinary Pharmaceuticals. Water Res. X 2020, 7, 100044. [Google Scholar] [CrossRef]
- Ferguson, E.M.; Allinson, M.; Allinson, G.; Swearer, S.E.; Hassell, K.L. Fluctuations in Natural and Synthetic Estrogen Concentrations in a Tidal Estuary in South-Eastern Australia. Water Res. 2013, 47, 1604–1615. [Google Scholar] [CrossRef]
- Tremblay, L.A.; Gadd, J.B.; Northcott, G.L. Steroid Estrogens and Estrogenic Activity Are Ubiquitous in Dairy Farm Watersheds Regardless of Effluent Management Practices. Agric. Ecosyst. Environ. 2018, 253, 48–54. [Google Scholar] [CrossRef]
- Bai, X.; Lutz, A.; Carroll, R.; Keteles, K.; Dahlin, K.; Murphy, M.; Nguyen, D. Occurrence, Distribution, and Seasonality of Emerging Contaminants in Urban Watersheds. Chemosphere 2018, 200, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Sznajder-Katarzyńska, K.; Surma, M.; Cieślik, I. A Review of Perfluoroalkyl Acids (PFAAs) in Terms of Sources, Applications, Human Exposure, Dietary Intake, Toxicity, Legal Regulation, and Methods of Determination. J. Chem. 2019, 2019, 2717528. [Google Scholar] [CrossRef] [Green Version]
- Xu, B.; Liu, S.; Zhou, J.L.; Zheng, C.; Weifeng, J.; Chen, B.; Zhang, T.; Qiu, W. PFAS and Their Substitutes in Groundwater: Occurrence, Transformation and Remediation. J. Hazard. Mater. 2021, 412, 125159. [Google Scholar] [CrossRef]
- Zhu, H.; Kannan, K. Distribution and Partitioning of Perfluoroalkyl Carboxylic Acids in Surface Soil, Plants, and Earthworms at a Contaminated Site. Sci. Total Environ. 2019, 647, 954–961. [Google Scholar] [CrossRef] [PubMed]
- Pitter, G.; Da Re, F.; Canova, C.; Barbieri, G.; Zare Jeddi, M.; Daprà, F.; Manea, F.; Zolin, R.; Bettega, A.M.; Stopazzolo, G.; et al. Serum Levels of Perfluoroalkyl Substances (PFAS) in Adolescents and Young Adults Exposed to Contaminated Drinking Water in the Veneto Region, Italy: A Cross-Sectional Study Based on a Health Surveillance Program. Environ. Health Perspect. 2020, 128, 27007. [Google Scholar] [CrossRef] [Green Version]
- Steenland, K.; Fletcher, T.; Stein, C.R.; Bartell, S.M.; Darrow, L.; Lopez-Espinosa, M.-J.; Barry Ryan, P.; Savitz, D.A. Review: Evolution of Evidence on PFOA and Health Following the Assessments of the C8 Science Panel. Environ. Int. 2020, 145, 106125. [Google Scholar] [CrossRef]
- Stalter, D.; O’Malley, E.; von Gunten, U.; Escher, B.I. Mixture Effects of Drinking Water Disinfection By-Products: Implications for Risk Assessment. Environ. Sci. (Camb.) 2020, 6, 2341–2351. [Google Scholar] [CrossRef]
- Chaves, R.S.; Guerreiro, C.S.; Cardoso, V.V.; Benoliel, M.J.; Santos, M.M. Toxicological Assessment of Seven Unregulated Drinking Water Disinfection By-Products (DBPs) Using the Zebrafish Embryo Bioassay. Sci. Total Environ. 2020, 742, 140522. [Google Scholar] [CrossRef]
- Mercier Shanks, C.; Sérodes, J.-B.; Rodriguez, M.J. Spatio-Temporal Variability of Non-Regulated Disinfection by-Products within a Drinking Water Distribution Network. Water Res. 2013, 47, 3231–3243. [Google Scholar] [CrossRef] [PubMed]
- Srivastav, A.L. Chemical Fertilizers and Pesticides: Role in Groundwater Contamination. In Agrochemicals Detection, Treatment and Remediation; Elsevier: Amsterdam, The Netherlands, 2020; pp. 143–159. [Google Scholar]
- Nowak, K.M.; Miltner, A.; Poll, C.; Kandeler, E.; Streck, T.; Pagel, H. Plant Litter Enhances Degradation of the Herbicide MCPA and Increases Formation of Biogenic Non-Extractable Residues in Soil. Environ. Int. 2020, 142, 105867. [Google Scholar] [CrossRef]
- Tasca, A.L.; Fletcher, A. State of the Art of the Environmental Behaviour and Removal Techniques of the Endocrine Disruptor 3,4-Dichloroaniline. J. Environ. Sci. Health A Tox. Hazard. Subst. Environ. Eng. 2018, 53, 260–270. [Google Scholar] [CrossRef]
- Reemtsma, T.; Alder, L.; Banasiak, U. Emerging Pesticide Metabolites in Groundwater and Surface Water as Determined by the Application of a Multimethod for 150 Pesticide Metabolites. Water Res. 2013, 47, 5535–5545. [Google Scholar] [CrossRef]
- Gallé, T.; Bayerle, M.; Pittois, D.; Huck, V. Allocating Biocide Sources and Flow Paths to Surface Waters Using Passive Samplers and Flood Wave Chemographs. Water Res. 2020, 173, 115533. [Google Scholar] [CrossRef]
- Belenguer, V.; Martinez-Capel, F.; Masiá, A.; Picó, Y. Patterns of Presence and Concentration of Pesticides in Fish and Waters of the Júcar River (Eastern Spain). J. Hazard. Mater. 2014, 265, 271–279. [Google Scholar] [CrossRef] [Green Version]
- K’oreje, K.O.; Vergeynst, L.; Ombaka, D.; De Wispelaere, P.; Okoth, M.; Van Langenhove, H.; Demeestere, K. Occurrence Patterns of Pharmaceutical Residues in Wastewater, Surface Water and Groundwater of Nairobi and Kisumu City, Kenya. Chemosphere 2016, 149, 238–244. [Google Scholar] [CrossRef]
- Simazaki, D.; Kubota, R.; Suzuki, T.; Akiba, M.; Nishimura, T.; Kunikane, S. Occurrence of Selected Pharmaceuticals at Drinking Water Purification Plants in Japan and Implications for Human Health. Water Res. 2015, 76, 187–200. [Google Scholar] [CrossRef]
- Sun, J.; Luo, Q.; Wang, D.; Wang, Z. Occurrences of Pharmaceuticals in Drinking Water Sources of Major River Watersheds, China. Ecotoxicol. Environ. Saf. 2015, 117, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Wang, B.; Ren, D.; Jin, W.; Liu, J.; Peng, J.; Pan, X. Occurrence, Removal and Bioaccumulation of Steroid Estrogens in Dianchi Lake Catchment, China. Environ. Int. 2013, 59, 262–273. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.; Lei, B.; Li, N.; Ma, M.; Wang, Z. Determination of Estrogens and Estrogenic Activities in Water from Three Rivers in Tianjin, China. J. Environ. Sci. (China) 2013, 25, 1164–1171. [Google Scholar] [CrossRef]
- Tan, R.; Liu, R.; Li, B.; Liu, X.; Li, Z. Typical Endocrine Disrupting Compounds in Rivers of Northeast China: Occurrence, Partitioning, and Risk Assessment. Arch. Environ. Contam. Toxicol. 2018, 75, 213–223. [Google Scholar] [CrossRef]
- Zhou, L.-J.; Li, J.; Zhang, Y.; Kong, L.; Jin, M.; Yang, X.; Wu, Q.L. Trends in the Occurrence and Risk Assessment of Antibiotics in Shallow Lakes in the Lower-Middle Reaches of the Yangtze River Basin, China. Ecotoxicol. Environ. Saf. 2019, 183, 109511. [Google Scholar] [CrossRef]
- Liu, Y.-H.; Zhang, S.-H.; Ji, G.-X.; Wu, S.-M.; Guo, R.-X.; Cheng, J.; Yan, Z.-Y.; Chen, J.-Q. Occurrence, Distribution and Risk Assessment of Suspected Endocrine-Disrupting Chemicals in Surface Water and Suspended Particulate Matter of Yangtze River (Nanjing Section). Ecotoxicol. Environ. Saf. 2017, 135, 90–97. [Google Scholar] [CrossRef]
- Chen, S.; Jiao, X.-C.; Gai, N.; Li, X.-J.; Wang, X.-C.; Lu, G.-H.; Piao, H.-T.; Rao, Z.; Yang, Y.-L. Perfluorinated Compounds in Soil, Surface Water, and Groundwater from Rural Areas in Eastern China. Environ. Pollut. 2016, 211, 124–131. [Google Scholar] [CrossRef] [PubMed]
- van der Aa, M.; Bijlsma, L.; Emke, E.; Dijkman, E.; van Nuijs, A.L.N.; van de Ven, B.; Hernández, F.; Versteegh, A.; de Voogt, P. Risk Assessment for Drugs of Abuse in the Dutch Watercycle. Water Res. 2013, 47, 1848–1857. [Google Scholar] [CrossRef] [PubMed]
- Jurado, A.; Vázquez-Suñé, E.; Pujades, E. Potential Uses of Pumped Urban Groundwater: A Case Study in Sant Adrià Del Besòs (Spain). Hydrogeol. J. 2017, 25, 1745–1758. [Google Scholar] [CrossRef]
- Szymonik, A.; Lach, J.; Malińska, K. Fate and Removal of Pharmaceuticals and Illegal Drugs Present in Drinking Water and Wastewater. Ecol. Chem. Eng. S 2017, 24, 65–85. [Google Scholar] [CrossRef] [Green Version]
- Dhodapkar, R.S.; Gandhi, K.N. Pharmaceuticals and Personal Care Products in Aquatic Environment: Chemicals of Emerging Concern? In Pharmaceuticals and Personal Care Products: Waste Management and Treatment Technology; Elsevier: Amsterdam, The Netherlands, 2019; pp. 63–85. [Google Scholar]
- Esteban, S.; Gorga, M.; Petrovic, M.; González-Alonso, S.; Barceló, D.; Valcárcel, Y. Analysis and Occurrence of Endocrine-Disrupting Compounds and Estrogenic Activity in the Surface Waters of Central Spain. Sci. Total Environ. 2014, 466–467, 939–951. [Google Scholar] [CrossRef]
- Madikizela, L.M.; Ncube, S.; Chimuka, L. Analysis, Occurrence and Removal of Pharmaceuticals in African Water Resources: A Current Status. J. Environ. Manag. 2020, 253, 109741. [Google Scholar] [CrossRef]
- Campo, J.; Masiá, A.; Picó, Y.; Farré, M.; Barceló, D. Distribution and Fate of Perfluoroalkyl Substances in Mediterranean Spanish Sewage Treatment Plants. Sci. Total Environ. 2014, 472, 912–922. [Google Scholar] [CrossRef]
- Lorenzo, M.; Campo, J.; Farré, M.; Pérez, F.; Picó, Y.; Barceló, D. Perfluoroalkyl Substances in the Ebro and Guadalquivir River Basins (Spain). Sci. Total Environ. 2016, 540, 191–199. [Google Scholar] [CrossRef]
- Kleywegt, S.; Pileggi, V.; Yang, P.; Hao, C.; Zhao, X.; Rocks, C.; Thach, S.; Cheung, P.; Whitehead, B. Pharmaceuticals, Hormones and Bisphenol A in Untreated Source and Finished Drinking Water in Ontario, Canada-–Occurrence and Treatment Efficiency. Sci. Total Environ. 2011, 409, 1481–1488. [Google Scholar] [CrossRef] [PubMed]
- Galindo-Miranda, J.M.; Guízar-González, C.; Becerril-Bravo, E.J.; Moeller-Chávez, G.; León-Becerril, E.; Vallejo-Rodríguez, R. Occurrence of Emerging Contaminants in Environmental Surface Waters and Their Analytical Methodology—A Review. Water Sci. Technol. Water Supply 2019, 19, 1871–1884. [Google Scholar] [CrossRef]
- Kibuye, F.A.; Gall, H.E.; Elkin, K.R.; Swistock, B.; Veith, T.L.; Watson, J.E.; Elliott, H.A. Occurrence, Concentrations, and Risks of Pharmaceutical Compounds in Private Wells in Central Pennsylvania. J. Environ. Qual. 2019, 48, 1057–1066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Padhye, L.P.; Yao, H.; Kung’u, F.T.; Huang, C.-H. Year-Long Evaluation on the Occurrence and Fate of Pharmaceuticals, Personal Care Products, and Endocrine Disrupting Chemicals in an Urban Drinking Water Treatment Plant. Water Res. 2014, 51, 266–276. [Google Scholar] [CrossRef]
- Backe, W.J. An Ultrasensitive (Parts-per-Quadrillion) and SPE-Free Method for the Quantitative Analysis of Estrogens in Surface Water. Environ. Sci. Technol. 2015, 49, 14311–14318. [Google Scholar] [CrossRef]
- Corrêa, J.M.M.; Sanson, A.L.; Machado, C.F.; Aquino, S.F.; Afonso, R.J.C.F. Occurrence of Contaminants of Emerging Concern in Surface Waters from Paraopeba River Basin in Brazil: Seasonal Changes and Risk Assessment. Environ. Sci. Pollut. Res. Int. 2021, 28, 30242–30254. [Google Scholar] [CrossRef]
- Lee, Y.-M.; Lee, J.-Y.; Kim, M.-K.; Yang, H.; Lee, J.-E.; Son, Y.; Kho, Y.; Choi, K.; Zoh, K.-D. Concentration and Distribution of Per- and Polyfluoroalkyl Substances (PFAS) in the Asan Lake Area of South Korea. J. Hazard. Mater. 2020, 381, 120909. [Google Scholar] [CrossRef]
- Yi, X.; Lin, C.; Ong, E.J.L.; Wang, M.; Zhou, Z. Occurrence and Distribution of Trace Levels of Antibiotics in Surface Waters and Soils Driven by Non-Point Source Pollution and Anthropogenic Pressure. Chemosphere 2019, 216, 213–223. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Das, P.; Shaw, A.; Mukherjee, A.; Civil, W.; Petersen, J.O.; Gooddy, D.C.; Wakefield, O.; Finlayson, A.; Krishan, G.; et al. Deep Urban Groundwater Vulnerability in India Revealed through the Use of Emerging Organic Contaminants and Residence Time Tracers. Environ. Pollut. 2018, 240, 938–949. [Google Scholar] [CrossRef] [Green Version]
- Sunantha, G.; Vasudevan, N. Assessment of Perfluorooctanoic Acid and Perfluorooctane Sulfonate in Surface Water—Tamil Nadu, India. Mar. Pollut. Bull. 2016, 109, 612–618. [Google Scholar] [CrossRef]
- Chakraborty, P.; Khuman, S.N.; Selvaraj, S.; Sampath, S.; Devi, N.L.; Bang, J.J.; Katsoyiannis, A. Polychlorinated Biphenyls and Organochlorine Pesticides in River Brahmaputra from the Outer Himalayan Range and River Hooghly Emptying into the Bay of Bengal: Occurrence, Sources and Ecotoxicological Risk Assessment. Environ. Pollut. 2016, 219, 998–1006. [Google Scholar] [CrossRef]
- Elfikrie, N.; Ho, Y.; Zaidon, S.; Juahir, H.; Tan, E. Occurrence of pesticides in surface water, pesticides removal efficiency in drinking water treatment plant and potential health risk to consumers in Tengi River Basin, Malaysia. Sci. Total Environ. 2020, 712, 136540. [Google Scholar] [CrossRef] [PubMed]
- Al-Odaini, N.; Zakaria, M.P.; Yaziz, M.I.; Surif, S. Detecting human pharmaceutical pollutants in Malaysian aquatic environment: A new challenge for water quality management. Res. Gate Febr. 2016. Available online: https://www.researchgate.net/profile/Najat-Al-Odaini/publication/279913351_Detecting_Human_Pharmaceutical_Pollutants_in_Malaysian_Aquatic_Environment_A_new_challenge_for_water_quality_management/links/559d947f08ae76bed0bb4270/Detecting-Human-Pharmaceutical-Pollutants-in-Malaysian-Aquatic-Environment-A-new-challenge-for-water-quality-management.pdf (accessed on 9 November 2021).
- Jiang, J.-Q.; Zhou, Z.; Sharma, V.K. Occurrence, Transportation, Monitoring and Treatment of Emerging Micro-Pollutants in Waste Water—A Review from Global Views. Microchem. J. 2013, 110, 292–300. [Google Scholar] [CrossRef]
- Lee, D.-H. Evidence of the Possible Harm of Endocrine-Disrupting Chemicals in Humans: Ongoing Debates and Key Issues. Endocrinol. Metab. (Seoul) 2018, 33, 44. [Google Scholar] [CrossRef]
- Haq, I.; Raj, A. Endocrine-Disrupting Pollutants in Industrial Wastewater and Their Degradation and Detoxification Approaches. In Emerging and Eco-Friendly Approaches for Waste Management; Springer: Singapore, 2019; pp. 121–142. [Google Scholar]
- Afshan, A.; Ali, M.N.; Bhat, F.A. Environmental Phthalate Exposure in Relation to Reproduction Outcomes and Health Endpoints. In Handbook of Research on Environmental and Human Health Impacts of Plastic Pollution; IGI Global: Hershey, PA, USA, 2020; pp. 340–369. [Google Scholar]
- Thambirajah, A.A.; Wade, M.G.; Verreault, J.; Buisine, N.; Alves, V.A.; Langlois, V.S.; Helbing, C.C. Disruption by Stealth—Interference of Endocrine Disrupting Chemicals on Hormonal Crosstalk with Thyroid Axis Function in Humans and Other Animals. Environ. Res. 2021, 203, 111906. [Google Scholar] [CrossRef]
- Nesan, D.; Sewell, L.C.; Kurrasch, D.M. Opening the Black Box of Endocrine Disruption of Brain Development: Lessons from the Characterization of Bisphenol A. Horm. Behav. 2018, 101, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Z.; Huang, Z.; Tang, X.; Xiong, C.; Tang, M.; Lu, Y. A Dually Charged Nanofiltration Membrane by PH-Responsive Polydopamine for Pharmaceuticals and Personal Care Products Removal. Sep. Purif. Technol. 2019, 211, 90–97. [Google Scholar] [CrossRef]
- Zhang, H.; West, D.; Shi, H.; Ma, Y.; Adams, C.; Eichholz, T. Simultaneous Determination of Selected Trace Contaminants in Drinking Water Using Solid-Phase Extraction-High Performance Liquid Chromatography-Tandem Mass Spectrometry. Water Air Soil Pollut. 2019, 230, 28. [Google Scholar] [CrossRef]
- Leusch, F.D.L.; Neale, P.A.; Busetti, F.; Card, M.; Humpage, A.; Orbell, J.D.; Ridgway, H.F.; Stewart, M.B.; van de Merwe, J.P.; Escher, B.I. Transformation of Endocrine Disrupting Chemicals, Pharmaceutical and Personal Care Products during Drinking Water Disinfection. Sci. Total Environ. 2019, 657, 1480–1490. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Gawad, S.A.; Abd ElAziz, H. Effective Removal of Chemical Oxygen Demand and Phosphates from Aqueous Medium Using Entrapped Activated Carbon in Alginate. MOJ Biol. Med. 2018, 3, 227–236. [Google Scholar] [CrossRef]
- Tran, N.H.; Reinhard, M.; Gin, K.Y.-H. Occurrence and Fate of Emerging Contaminants in Municipal Wastewater Treatment Plants from Different Geographical Regions—A Review. Water Res. 2018, 133, 182–207. [Google Scholar] [CrossRef]
- Richardson, S.D.; Kimura, S.Y. Water Analysis: Emerging Contaminants and Current Issues. Anal. Chem. 2020, 92, 473–505. [Google Scholar] [CrossRef]
- Flores, A.; Hill, E.M. Formation of Estrogenic Brominated Ethinylestradiol in Drinking Water: Implications for Aquatic Toxicity Testing. Chemosphere 2008, 73, 1115–1120. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Terasaki, M. Estrogen Agonistic/Antagonistic Activity of Brominated Parabens. Environ. Sci. Pollut. Res. Int. 2018, 25, 21257–21266. [Google Scholar] [CrossRef]
- Chen, Y.; Xie, H.; Junaid, M.; Xu, N.; Zhu, Y.; Tao, H.; Wong, M. Spatiotemporal Distribution, Source Apportionment and Risk Assessment of Typical Hormones and Phenolic Endocrine Disrupting Chemicals in Environmental and Biological Samples from the Mariculture Areas in the Pearl River Delta, China. Sci. Total Environ. 2021, 807, 150752. [Google Scholar] [CrossRef] [PubMed]
- Eustache, F.; Bennani Smires, B.; Moison, D.; Bergès, R.; Canivenc-Lavier, M.-C.; Vaiman, D.; Auger, J. Different Exposure Windows to Low Doses of Genistein and/or Vinclozolin Result in Contrasted Disorders of Testis Function and Gene Expression of Exposed Rats and Their Unexposed Progeny. Environ. Res. 2020, 190, 109975. [Google Scholar] [CrossRef] [PubMed]
- Maskey, E.; Crotty, H.; Wooten, T.; Khan, I.A. Disruption of Oocyte Maturation by Selected Environmental Chemicals in Zebrafish. Toxicol. Vitr. 2019, 54, 123–129. [Google Scholar] [CrossRef]
- Awfa, D.; Ateia, M.; Fujii, M.; Johnson, M.S.; Yoshimura, C. Photodegradation of Pharmaceuticals and Personal Care Products in Water Treatment Using Carbonaceous-TiO2 Composites: A Critical Review of Recent Literature. Water Res. 2018, 142, 26–45. [Google Scholar] [CrossRef] [PubMed]
- Falisse, E.; Voisin, A.-S.; Silvestre, F. Impacts of Triclosan Exposure on Zebrafish Early-Life Stage: Toxicity and Acclimation Mechanisms. Aquat. Toxicol. 2017, 189, 97–107. [Google Scholar] [CrossRef]
- Li, C.; Qu, R.; Chen, J.; Zhang, S.; Allam, A.A.; Ajarem, J.; Wang, Z. The PH-Dependent Toxicity of Triclosan to Five Aquatic Organisms (Daphnia Magna, Photobacterium Phosphoreum, Danio Rerio, Limnodrilus Hoffmeisteri, and Carassius Auratus). Environ. Sci. Pollut. Res. Int. 2018, 25, 9636–9646. [Google Scholar] [CrossRef]
- Battaglin, W.A.; Bradley, P.M.; Iwanowicz, L.; Journey, C.A.; Walsh, H.L.; Blazer, V.S. Pharmaceuticals, Hormones, Pesticides, and Other Bioactive Contaminants in Water, Sediment, and Tissue from Rocky Mountain National Park, 2012–2013. Sci. Total Environ. 2018, 643, 651–673. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Tan, Y.X.R.; Gong, Z.; Bae, S. The Toxic Effect of Triclosan and Methyl-Triclosan on Biological Pathways Revealed by Metabolomics and Gene Expression in Zebrafish Embryos. Ecotoxicol. Environ. Saf. 2020, 189, 110039. [Google Scholar] [CrossRef]
- Srain, H.S.; Beazley, K.F.; Walker, T.R. Pharmaceuticals and Personal Care Products and Their Sublethal and Lethal Effects in Aquatic Organisms. Environ. Rev. 2021, 29, 142–181. [Google Scholar] [CrossRef]
- Rochester, J.R. Bisphenol A and Human Health: A Review of the Literature. Reprod. Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef]
- Weber, D.N.; Hoffmann, R.G.; Hoke, E.S.; Tanguay, R.L. Bisphenol A Exposure during Early Development Induces Sex-Specific Changes in Adult Zebrafish Social Interactions. J. Toxicol. Environ. Health Part A 2015, 78, 50–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collet, B.; Desalegn, A.A.; Swart, K.; Naderman, M.; Iszatt, N.; Stigum, H.; Jensen, T.K.; Brouwer, A.; Eggesbø, M.; van der Burg, B. Anti-Androgenic Compounds in Breast Milk and Cryptorchidism among Norwegian Boys in the HUMIS Birth Cohort. Sci. Total Environ. 2021, 803, 149746. [Google Scholar] [CrossRef]
- Arendt, L.H.; Lindhard, M.S.; Henriksen, T.B.; Olsen, J.; Cnattingius, S.; Petersson, G.; Parner, E.T.; Ramlau-Hansen, C.H. Maternal Diabetes Mellitus and Genital Anomalies in Male Offspring: A Nationwide Cohort Study in 2 Nordic Countries. Epidemiology 2018, 29, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Dorman, D.C.; Chiu, W.; Hales, B.F.; Hauser, R.; Johnson, K.J.; Mantus, E.; Martel, S.; Robinson, K.A.; Rooney, A.A.; Rudel, R.; et al. Polybrominated Diphenyl Ether (PBDE) Neurotoxicity: A Systematic Review and Meta-Analysis of Animal Evidence. J. Toxicol. Environ. Health B Crit. Rev. 2018, 21, 269–289. [Google Scholar] [CrossRef]
- Richardson, S.D.; Postigo, C. Liquid Chromatography–Mass Spectrometry of Emerging Disinfection by-Products. In Advances in the Use of Liquid Chromatography Mass Spectrometry (LC-MS)—Instrumentation Developments and Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 267–295. [Google Scholar]
- Gan, W.; Guo, W.; Mo, J.; He, Y.; Liu, Y.; Liu, W.; Liang, Y.; Yang, X. The Occurrence of Disinfection By-Products in Municipal Drinking Water in China’s Pearl River Delta and a Multipathway Cancer Risk Assessment. Sci. Total Environ. 2013, 447, 108–115. [Google Scholar] [CrossRef]
- Mostafalou, S.; Abdollahi, M. Pesticides and Human Chronic Diseases: Evidences, Mechanisms, and Perspectives. Toxicol. Appl. Pharmacol. 2013, 268, 157–177. [Google Scholar] [CrossRef] [PubMed]
- Souza, R.C.; Portella, R.B.; Almeida, P.V.N.B.; Pinto, C.O.; Gubert, P.; Santos da Silva, J.D.; Nakamura, T.C.; do Rego, E.L. Human Milk Contamination by Nine Organochlorine Pesticide Residues (OCPs). J. Environ. Sci. Health B 2020, 55, 530–538. [Google Scholar] [CrossRef]
- Du, J.; Tang, J.; Xu, S.; Ge, J.; Dong, Y.; Li, H.; Jin, M. A Review on Silver Nanoparticles-Induced Ecotoxicity and the Underlying Toxicity Mechanisms. Regul. Toxicol. Pharmacol. 2018, 98, 231–239. [Google Scholar] [CrossRef]
- Gajewicz, A.; Rasulev, B.; Dinadayalane, T.C.; Urbaszek, P.; Puzyn, T.; Leszczynska, D.; Leszczynski, J. Advancing Risk Assessment of Engineered Nanomaterials: Application of Computational Approaches. Adv. Drug Deliv. Rev. 2012, 64, 1663–1693. [Google Scholar] [CrossRef]
- Ji, H.; Song, N.; Ren, J.; Li, W.; Xu, B.; Li, H.; Shen, G. Metabonomics Reveals Bisphenol A Affects Fatty Acid and Glucose Metabolism through Activation of LXR in the Liver of Male Mice. Sci. Total Environ. 2020, 703, 134681. [Google Scholar] [CrossRef] [PubMed]
- vom Saal, F.S.; Hughes, C. An Extensive New Literature Concerning Low-Dose Effects of Bisphenol A Shows the Need for a New Risk Assessment. Environ. Health Perspect. 2005, 113, 926–933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viñas, R.; Watson, C.S. Bisphenol S Disrupts Estradiol-Induced Nongenomic Signaling in a Rat Pituitary Cell Line: Effects on Cell Functions. Environ. Health Perspect. 2013, 121, 352–358. [Google Scholar] [CrossRef]
- Han, X.; Cui, Z.; Zhou, N.; Ma, M.; Li, L.; Li, Y.; Lin, H.; Ao, L.; Shu, W.; Liu, J.; et al. Urinary Phthalate Metabolites and Male Reproductive Function Parameters in Chongqing General Population, China. Int. J. Hyg. Environ. Health 2014, 217, 271–278. [Google Scholar] [CrossRef]
- Dhillon, G.S.; Kaur, S.; Pulicharla, R.; Brar, S.K.; Cledón, M.; Verma, M.; Surampalli, R.Y. Triclosan: Current Status, Occurrence, Environmental Risks and Bioaccumulation Potential. Int. J. Environ. Res. Public Health 2015, 12, 5657–5684. [Google Scholar] [CrossRef] [PubMed]
- Kunz, P.Y.; Kienle, C.; Carere, M.; Homazava, N.; Kase, R. In Vitro Bioassays to Screen for Endocrine Active Pharmaceuticals in Surface and Waste Waters. J. Pharm. Biomed. Anal. 2015, 106, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Gajewska, M.; Paini, A.; Sala Benito, J.V.; Burton, J.; Worth, A.; Urani, C.; Briesen, H.; Schramm, K.-W. In Vitro-to-in Vivo Correlation of the Skin Penetration, Liver Clearance and Hepatotoxicity of Caffeine. Food Chem. Toxicol. 2015, 75, 39–49. [Google Scholar] [CrossRef]
- Ventura, C.; Venturino, A.; Miret, N.; Randi, A.; Rivera, E.; Núñez, M.; Cocca, C. Chlorpyrifos Inhibits Cell Proliferation through ERK1/2 Phosphorylation in Breast Cancer Cell Lines. Chemosphere 2015, 120, 343–350. [Google Scholar] [CrossRef]
- Li, P.; Zhu, J.; Kong, Q.; Jiang, B.; Wan, X.; Yue, J.; Li, M.; Jiang, H.; Li, J.; Gao, Z. The Ethylene Bis-Dithiocarbamate Fungicide Mancozeb Activates Voltage-Gated KCNQ2 Potassium Channel. Toxicol. Lett. 2013, 219, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.C.; Miranda, L.F.C.; de Souza, A.O.; Dorta, D.J. BDE-154 Induces Mitochondrial Permeability Transition and Impairs Mitochondrial Bioenergetics. J. Toxicol. Environ. Health Part A 2014, 77, 24–36. [Google Scholar] [CrossRef]
- Wu, S.; Ji, G.; Liu, J.; Zhang, S.; Gong, Y.; Shi, L. TBBPA Induces Developmental Toxicity, Oxidative Stress, and Apoptosis in Embryos and Zebrafish Larvae (Danio Rerio): TBBPA INDUCES DEVELOPMENTAL TOXICITY, OXIDATIVE STRESS, AND APOPTOSIS. Environ. Toxicol. 2016, 31, 1241–1249. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Li, X.; Sun, M.; Wei, Z.; Wang, Y.; Gao, A.; Chen, D.; Zhao, X.; Feng, X. Exploring the Effects of Different Types of Surfactants on Zebrafish Embryos and Larvae. Sci. Rep. 2015, 5, 10107. [Google Scholar] [CrossRef] [Green Version]
- Rodil, R.; Quintana, J.B.; López-Mahía, P.; Muniategui-Lorenzo, S.; Prada-Rodríguez, D. Multi-Residue Analytical Method for the Determination of Emerging Pollutants in Water by Solid-Phase Extraction and Liquid Chromatography-Tandem Mass Spectrometry. J. Chromatogr. A 2009, 1216, 2958–2969. [Google Scholar] [CrossRef] [PubMed]
- La Farré, M.; Pérez, S.; Kantiani, L.; Barceló, D. Fate and Toxicity of Emerging Pollutants, Their Metabolites and Transformation Products in the Aquatic Environment. Trends Analyt. Chem. 2008, 27, 991–1007. [Google Scholar] [CrossRef]
- Lopez de Alda, M.J.; Díaz-Cruz, S.; Petrovic, M.; Barceló, D. Liquid Chromatography–(Tandem) Mass Spectrometry of Selected Emerging Pollutants (Steroid Sex Hormones, Drugs and Alkylphenolic Surfactants) in the Aquatic Environment. J. Chromatogr. A 2003, 1000, 503–526. [Google Scholar] [CrossRef] [Green Version]
- Norlock, F.M.; Jang, J.-K.; Zou, Q.; Schoonover, T.M.; Li, A. Large-Volume Injection PTV-GC-MS Analysis of Polycyclic Aromatic Hydrocarbons in Air and Sediment Samples. J. Air Waste Manag. Assoc. 2002, 52, 19–26. [Google Scholar] [CrossRef]
- Pitt, J.J. Principles and Applications of Liquid Chromatography-Mass Spectrometry in Clinical Biochemistry. Clin. Biochem. Rev. 2009, 30, 19–34. [Google Scholar]
- Quintana, J.B.; Rodil, R.; Reemtsma, T.; García-López, M.; Rodríguez, I. Organophosphorus Flame Retardants and Plasticizers in Water and Air II. Analytical Methodology. Trends Analyt. Chem. 2008, 27, 904–915. [Google Scholar] [CrossRef]
- Frenich, A.G.; Martínez Vidal, J.L.; Pablos Espada, M.C.; Gil García, M.D.; Arrebola, F.J. Comparison of Gas Chromatography with NPD, MS, and Tandem MS-MS in the Multiresidue Analysis of Pesticides in Environmental Waters. Chromatographia 2000, 52, 614–620. [Google Scholar] [CrossRef]
- Wille, K.; De Brabander, H.F.; Vanhaecke, L.; De Wulf, E.; Van Caeter, P.; Janssen, C.R. Coupled Chromatographic and Mass-Spectrometric Techniques for the Analysis of Emerging Pollutants in the Aquatic Environment. Trends Analyt. Chem. 2012, 35, 87–108. [Google Scholar] [CrossRef]
- Grassi, M.; Kaykioglu, G.; Belgiorno, V.; Lofrano, G. Removal of Emerging Contaminants from Water and Wastewater by Adsorption Process. In SpringerBriefs in Molecular Science; Springer: Dordrecht, The Netherlands, 2012; pp. 15–37. [Google Scholar]
- Adams, C.; Wang, Y.; Loftin, K.; Meyer, M. Removal of Antibiotics from Surface and Distilled Water in Conventional Water Treatment Processes. J. Environ. Eng. (N. Y.) 2002, 128, 253–260. [Google Scholar] [CrossRef]
- Martín de Vidales, M.J.; Rua, J.; Montero de Juan, J.L.; Fernández-Martínez, F.; Dos Santos-García, A.J. Degradation of Contaminants of Emerging Concern by Electrochemical Oxidation: Coupling of Ultraviolet and Ultrasound Radiations. Materials 2020, 13, 5551. [Google Scholar] [CrossRef]
- Prest, E.I.; Hammes, F.; van Loosdrecht, M.C.M.; Vrouwenvelder, J.S. Biological Stability of Drinking Water: Controlling Factors, Methods, and Challenges. Front. Microbiol. 2016, 7, 45. [Google Scholar] [CrossRef]
- Jensen, V.B.; Darby, J.L.; Seidel, C.; Gorman, C. Nitrate in Potable Water Supplies: Alternative Management Strategies. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2203–2286. [Google Scholar] [CrossRef]
- Rahman, I. Direct Biofiltration and Nutrient (Phosphorus) Enhancement for Polymeric Ultrafiltration Membrane Fouling Control. Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2013. [Google Scholar]
- Sgroi, M.; Roccaro, P.; Korshin, G.V.; Greco, V.; Sciuto, S.; Anumol, T.; Snyder, S.A.; Vagliasindi, F.G.A. Use of Fluorescence EEM to Monitor the Removal of Emerging Contaminants in Full Scale Wastewater Treatment Plants. J. Hazard. Mater. 2017, 323, 367–376. [Google Scholar] [CrossRef]
- Liu, Z.-H.; Lu, G.-N.; Yin, H.; Dang, Z.; Rittmann, B. Removal of Natural Estrogens and Their Conjugates in Municipal Wastewater Treatment Plants: A Critical Review. Environ. Sci. Technol. 2015, 49, 5288–5300. [Google Scholar] [CrossRef]
- Vilela, C.L.S.; Bassin, J.P.; Peixoto, R.S. Water Contamination by Endocrine Disruptors: Impacts, Microbiological Aspects and Trends for Environmental Protection. Environ. Pollut. 2018, 235, 546–559. [Google Scholar] [CrossRef]
- Praveena, S.M.; Lui, T.S.; Hamin, N.; Razak, S.Q.N.A.; Aris, A.Z. Occurrence of Selected Estrogenic Compounds and Estrogenic Activity in Surface Water and Sediment of Langat River (Malaysia). Environ. Monit. Assess. 2016, 188, 442. [Google Scholar] [CrossRef]
- Clouzot, L.; Choubert, J.-M.; Cloutier, F.; Goel, R.; Love, N.G.; Melcer, H.; Ort, C.; Patureau, D.; Plósz, B.G.; Pomiès, M.; et al. Perspectives on Modelling Micropollutants in Wastewater Treatment Plants. Water Sci. Technol. 2013, 68, 448–461. [Google Scholar] [CrossRef] [Green Version]
- Gadipelly, C.; Pérez-González, A.; Yadav, G.D.; Ortiz, I.; Ibáñez, R.; Rathod, V.K.; Marathe, K.V. Pharmaceutical Industry Wastewater: Review of the Technologies for Water Treatment and Reuse. Ind. Eng. Chem. Res. 2014, 53, 11571–11592. [Google Scholar] [CrossRef]
- Hussain, A.; Dubey, S.K. Specific Methanogenic Activity Test for Anaerobic Treatment of Phenolic Wastewater. Desalination Water Treat. 2014, 52, 7015–7025. [Google Scholar] [CrossRef]
- Sreekanth, D.; Sivaramakrishna, D.; Himabindu, V.; Anjaneyulu, Y. Thermophilic Treatment of Bulk Drug Pharmaceutical Industrial Wastewaters by Using Hybrid up Flow Anaerobic Sludge Blanket Reactor. Bioresour. Technol. 2009, 100, 2534–2539. [Google Scholar] [CrossRef]
- Chang, C.-Y.; Chang, J.-S.; Vigneswaran, S.; Kandasamy, J. Pharmaceutical Wastewater Treatment by Membrane Bioreactor Process—A Case Study in Southern Taiwan. Desalination 2008, 234, 393–401. [Google Scholar] [CrossRef]
- Mazzei, R.; Piacentini, E.; Gebreyohannes, A.Y.; Giorno, L. Membrane Bioreactors in Food, Pharmaceutical and Biofuel Applications: State of the Art, Progresses and Perspectives. Curr. Org. Chem. 2017, 21, 1671–1701. [Google Scholar] [CrossRef]
- Hamon, P.; Moulin, P.; Ercolei, L.; Marrot, B. Oncological Ward Wastewater Treatment by Membrane Bioreactor: Acclimation Feasibility and Pharmaceuticals Removal Performances. J. Water Proc.Eng. 2018, 21, 9–26. [Google Scholar] [CrossRef] [Green Version]
- Katibi, K.K.; Yunos, K.F.; Che Man, H.; Aris, A.Z.; Bin Mohd Nor, M.Z.; Binti Azis, R.S. Recent Advances in the Rejection of Endocrine-Disrupting Compounds from Water Using Membrane and Membrane Bioreactor Technologies: A Review. Polymers 2021, 13, 392. [Google Scholar] [CrossRef]
- Sathishkumar, P.; Mythili, A.; Hadibarata, T.; Jayakumar, R.; Kanthimathi, M.S.; Palvannan, T.; Ponraj, M.; Salim, M.R.; Mohd Yusoff, A.R. Laccase Mediated Diclofenac Transformation and Cytotoxicity Assessment on Mouse Fibroblast 3T3-L1 Preadipocytes. RSC Adv. 2014, 4, 11689. [Google Scholar] [CrossRef]
- Bokare, V.; Murugesan, K.; Kim, Y.-M.; Jeon, J.-R.; Kim, E.-J.; Chang, Y.S. Degradation of Triclosan by an Integrated Nano-Bio Redox Process. Bioresour. Technol. 2010, 101, 6354–6360. [Google Scholar] [CrossRef]
- Sathishkumar, P.; Chae, J.-C.; Unnithan, A.R.; Palvannan, T.; Kim, H.Y.; Lee, K.-J.; Cho, M.; Kamala-Kannan, S.; Oh, B.-T. Laccase-Poly(Lactic-Co-Glycolic Acid) (PLGA) Nanofiber: Highly Stable, Reusable, and Efficacious for the Transformation of Diclofenac. Enzym. Microb. Technol. 2012, 51, 113–118. [Google Scholar] [CrossRef]
- Touahar, I.E.; Haroune, L.; Ba, S.; Bellenger, J.-P.; Cabana, H. Characterization of Combined Cross-Linked Enzyme Aggregates from Laccase, Versatile Peroxidase and Glucose Oxidase, and Their Utilization for the Elimination of Pharmaceuticals. Sci. Total Environ. 2014, 481, 90–99. [Google Scholar] [CrossRef]
- Benner, J.; Helbling, D.E.; Kohler, H.-P.E.; Wittebol, J.; Kaiser, E.; Prasse, C.; Ternes, T.A.; Albers, C.N.; Aamand, J.; Horemans, B.; et al. Is Biological Treatment a Viable Alternative for Micropollutant Removal in Drinking Water Treatment Processes? Water Res. 2013, 47, 5955–5976. [Google Scholar] [CrossRef]
- Diemert, S.; Wang, W.; Andrews, R.C.; Li, X.-F. Removal of Halo-Benzoquinone (Emerging Disinfection by-Product) Precursor Material from Three Surface Waters Using Coagulation. Water Res. 2013, 47, 1773–1782. [Google Scholar] [CrossRef]
- Zhao, Y.; Xiao, F.; Wang, D.; Yan, M.; Bi, Z. Disinfection Byproduct Precursor Removal by Enhanced Coagulation and Their Distribution in Chemical Fractions. J. Environ. Sci. (China) 2013, 25, 2207–2213. [Google Scholar] [CrossRef]
- Jusoh, A.; Hartini, W.J.H.; Ali, N.; Endut, A. Study on the Removal of Pesticide in Agricultural Run off by Granular Activated Carbon. Bioresour. Technol. 2011, 102, 5312–5318. [Google Scholar] [CrossRef]
- Ova, D.; Ovez, B. 2,4-Dichlorophenoxyacetic Acid Removal from Aqueous Solutions via Adsorption in the Presence of Biological Contamination. J. Environ. Chem. Eng. 2013, 1, 813–821. [Google Scholar] [CrossRef]
- Nazifa, T.H.; Habba, N.; Aris, A.; Hadibarata, T. Adsorption of Procion Red MX-5B and Crystal Violet Dyes from Aqueous Solution onto Corncob Activated Carbon: Adsorption of Procion Red MX-5B and Crystal Violet Dyes. J. Chin. Chem. Soc. 2018, 65, 259–270. [Google Scholar] [CrossRef]
- Tong, D.S.; Zhou, C.H.C.; Lu, Y.; Yu, H.; Zhang, G.F.; Yu, W.H. Adsorption of Acid Red G Dye on Octadecyl Trimethylammonium Montmorillonite. Appl. Clay Sci. 2010, 50, 427–431. [Google Scholar] [CrossRef]
- Gupta, V.K.; Carrott, P.J.M.; Ribeiro Carrott, M.M.L.; Suhas. Low-Cost Adsorbents: Growing Approach to Wastewater Treatment—A Review. Crit. Rev. Environ. Sci. Technol. 2009, 39, 783–842. [Google Scholar] [CrossRef]
- Bolong, N.; Ismail, A.F.; Salim, M.R.; Matsuura, T. A Review of the Effects of Emerging Contaminants in Wastewater and Options for Their Removal. Desalination 2009, 239, 229–246. [Google Scholar] [CrossRef]
- Vatankhah, H.; Murray, C.C.; Brannum, J.W.; Vanneste, J.; Bellona, C. Effect of Pre-Ozonation on Nanofiltration Membrane Fouling during Water Reuse Applications. Sep. Purif. Technol. 2018, 205, 203–211. [Google Scholar] [CrossRef]
- Touati, K.; Gzara, L.; Mahfoudhi, S.; Bourezgui, S.; Hafiane, A.; Elfil, H. Treatment of Coastal Well Water Using Ultrafiltration-Nanofiltration-Reverse Osmosis to Produce Isotonic Solutions and Drinking Water: Fouling Behavior and Energy Efficiency. J. Clean. Prod. 2018, 200, 1053–1064. [Google Scholar] [CrossRef]
- Semião, A.J.C.; Foucher, M.; Schäfer, A.I. Removal of Adsorbing Estrogenic Micropollutants by Nanofiltration Membranes: Part B—Modeldevelopment. J. Memb. Sci. 2013, 431, 257–266. [Google Scholar] [CrossRef] [Green Version]
- Aziz, M.; Ojumu, T. Exclusion of Estrogenic and Androgenic Steroid Hormones from Municipal Membrane Bioreactor Wastewater Using UF/NF/RO Membranes for Water Reuse Application. Membranes 2020, 10, 37. [Google Scholar] [CrossRef] [Green Version]
- Amin, M.T.; Alazba, A.A.; Manzoor, U. A Review of Removal of Pollutants from Water/Wastewater Using Different Types of Nanomaterials. Adv. Mater. Sci. Eng. 2014, 2014, 825910. [Google Scholar] [CrossRef] [Green Version]
- Musbah, I.; Cicéron, D.; Saboni, A.; Alexandrova, S. Retention of Pesticides and Metabolites by Nanofiltration by Effects of Size and Dipole Moment. Desalination 2013, 313, 51–56. [Google Scholar] [CrossRef]
- Plakas, K.V.; Karabelas, A.J. Removal of Pesticides from Water by NF and RO Membranes—A Review. Desalination 2012, 287, 255–265. [Google Scholar] [CrossRef]
- Pang, W.; Gao, N.; Xia, S. Removal of DDT in Drinking Water Using Nanofiltration Process. Desalination 2010, 250, 553–556. [Google Scholar] [CrossRef]
- Acero, J.L.; Benitez, F.J.; Real, F.J.; García, C. Removal of Phenyl-Urea Herbicides in Natural Waters by UF Membranes: Permeate Flux, Analysis of Resistances and Rejection Coefficients. Sep. Purif. Technol. 2009, 65, 322–330. [Google Scholar] [CrossRef]
- Bing-zhi, D.; Lin, W.; Nai-yun, G. The Removal of Bisphenol A by Ultrafiltration. Desalination 2008, 221, 312–317. [Google Scholar] [CrossRef]
- Bing-zhi, D.; Hua-qiang, C.; Lin, W.; Sheng-ji, X.; Nai-yun, G. The Removal of Bisphenol A by Hollow Fiber Microfiltration Membrane. Desalination 2010, 250, 693–697. [Google Scholar] [CrossRef]
- Zhang, Y. Nanoparticle Separation in Cross-Flow Filtration by Introduction of Electrophoresis. Master’s Thesis, University of Delaware, Newark, DE, USA, 2014. [Google Scholar]
- Springer, F.; Laborie, S.; Guigui, C. Removal of SiO2 Nanoparticles from Industry Wastewaters and Subsurface Waters by Ultrafiltration: Investigation of Process Efficiency, Deposit Properties and Fouling Mechanism. Sep. Purif. Technol. 2013, 108, 6–14. [Google Scholar] [CrossRef]
- Liu, P.; Zhang, H.; Feng, Y.; Yang, F.; Zhang, J. Removal of Trace Antibiotics from Wastewater: A Systematic Study of Nanofiltration Combined with Ozone-Based Advanced Oxidation Processes. Chem. Eng. J. 2014, 240, 211–220. [Google Scholar] [CrossRef]
- Saravanan, A.; Senthil Kumar, P.; Jeevanantham, S.; Karishma, S.; Tajsabreen, B.; Yaashikaa, P.R.; Reshma, B. Effective Water/Wastewater Treatment Methodologies for Toxic Pollutants Removal: Processes and Applications towards Sustainable Development. Chemosphere 2021, 280, 130595. [Google Scholar] [CrossRef]
- de Jesus Gaffney, V.; Almeida, C.M.M.; Rodrigues, A.; Ferreira, E.; Benoliel, M.J.; Cardoso, V.V. Occurrence of Pharmaceuticals in a Water Supply System and Related Human Health Risk Assessment. Water Res. 2015, 72, 199–208. [Google Scholar] [CrossRef]
- Ternes, T.A.; Meisenheimer, M.; McDowell, D.; Sacher, F.; Brauch, H.-J.; Haist-Gulde, B.; Preuss, G.; Wilme, U.; Zulei-Seibert, N. Removal of Pharmaceuticals during Drinking Water Treatment. Environ. Sci. Technol. 2002, 36, 3855–3863. [Google Scholar] [CrossRef]
- Kovalova, L.; Siegrist, H.; von Gunten, U.; Eugster, J.; Hagenbuch, M.; Wittmer, A.; Moser, R.; McArdell, C.S. Elimination of Micropollutants during Post-Treatment of Hospital Wastewater with Powdered Activated Carbon, Ozone, and UV. Environ. Sci. Technol. 2013, 47, 7899–7908. [Google Scholar] [CrossRef] [Green Version]
- Kharel, S.; Stapf, M.; Miehe, U.; Ekblad, M.; Cimbritz, M.; Falås, P.; Nilsson, J.; Sehlén, R.; Bester, K. Ozone Dose Dependent Formation and Removal of Ozonation Products of Pharmaceuticals in Pilot and Full-Scale Municipal Wastewater Treatment Plants. Sci. Total Environ. 2020, 731, 139064. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, K.; Yu, J.; Li, H.; Yang, M.; He, X.; Lu, N.; Zhang, D.; Sun, Z.; Wu, X. Pesticides removal evaluation for O3/BAC with post filtration process in drinking water. Chin. J. Environ. Eng. 2016, 10, 2315–2320. [Google Scholar]
- Wang, J.; Zhuan, R. Degradation of Antibiotics by Advanced Oxidation Processes: An Overview. Sci. Total Environ. 2020, 701, 135023. [Google Scholar] [CrossRef]
- Chauveheid, E.; Scholdis, S. Removal of Pharmaceuticals by a Surface Water Treatment Plant. Water Sci. Technol. Water Supply 2019, 19, 1793–1801. [Google Scholar] [CrossRef]
- Yu, S.; Hu, J.; Wang, J. Gamma Radiation-Induced Degradation of p-Nitrophenol (PNP) in the Presence of Hydrogen Peroxide (H2O2) in Aqueous Solution. J. Hazard. Mater. 2010, 177, 1061–1067. [Google Scholar] [CrossRef]
- Shaoqing, Y.; Jun, H.; Jianlong, W. Radiation-Induced Catalytic Degradation of p-Nitrophenol (PNP) in the Presence of TiO2 Nanoparticles. Radiat. Phys. Chem. 2010, 79, 1039–1046. [Google Scholar] [CrossRef]
- Alkhuraiji, T.S.; Alkhuraiji, W.S. Detailed Study of Water Radiolysis-Based Degradation of Chloroorganic Pollutants in Aqueous Solutions. J. Hazard. Mater. 2019, 368, 569–577. [Google Scholar] [CrossRef]
- Xue, J.; Hu, J.; Wang, J.-L. Radiolysis of chlorophenols in aqueous solution by gamma ray. Huan Jing Ke Xue 2008, 29, 1919–1923. [Google Scholar]
- Liu, Y.; Wang, J. Degradation of Sulfamethazine by Gamma Irradiation in the Presence of Hydrogen Peroxide. J. Hazard. Mater. 2013, 250–251, 99–105. [Google Scholar] [CrossRef]
- Liu, Y.; Tourbin, M.; Lachaize, S.; Guiraud, P. Nanoparticles in Wastewaters: Hazards, Fate and Remediation. Powder Technol. 2014, 255, 149–156. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Wang, J. Application of Radiation Technology to Sewage Sludge Processing: A Review. J. Hazard. Mater. 2007, 143, 2–7. [Google Scholar] [CrossRef]
- Rauf, M.A.; Ashraf, S.S. Radiation Induced Degradation of Dyes—An Overview. J. Hazard. Mater. 2009, 166, 6–16. [Google Scholar] [CrossRef]
- Hossain, K.; Maruthi, Y.A.; Das, N.L.; Rawat, K.P.; Sarma, K.S.S. Irradiation of Wastewater with Electron Beam Is a Key to Sustainable Smart/Green Cities: A Review. Appl. Water Sci. 2018, 8, 6. [Google Scholar] [CrossRef] [Green Version]
- Lee, O.-M.; Kim, H.Y.; Park, W.; Kim, T.-H.; Yu, S. A Comparative Study of Disinfection Efficiency and Regrowth Control of Microorganism in Secondary Wastewater Effluent Using UV, Ozone, and Ionizing Irradiation Process. J. Hazard. Mater. 2015, 295, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chu, L. Irradiation Treatment of Pharmaceutical and Personal Care Products (PPCPs) in Water and Wastewater: An Overview. Radiat. Phys. Chem. 2016, 125, 56–64. [Google Scholar] [CrossRef]
- Li, W.; Zhao, Y.; Yan, X.; Duan, J.; Saint, C.P.; Beecham, S. Transformation Pathway and Toxicity Assessment of Malathion in Aqueous Solution during UV Photolysis and Photocatalysis. Chemosphere 2019, 234, 204–214. [Google Scholar] [CrossRef]
- Sanches, S.; Barreto Crespo, M.T.; Pereira, V.J. Drinking Water Treatment of Priority Pesticides Using Low Pressure UV Photolysis and Advanced Oxidation Processes. Water Res. 2010, 44, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Shawaqfeh, A.T.; Al Momani, F.A. Photocatalytic Treatment of Water Soluble Pesticide by Advanced Oxidation Technologies Using UV Light and Solar Energy. Sol. Energy 2010, 84, 1157–1165. [Google Scholar] [CrossRef]
- Borges, M.; García, D.; Hernández, T.; Ruiz-Morales, J.; Esparza, P. Supported Photocatalyst for Removal of Emerging Contaminants from Wastewater in a Continuous Packed-Bed Photoreactor Configuration. Catalysts 2015, 5, 77–87. [Google Scholar] [CrossRef] [Green Version]
- Gadupudi, C.K.; Rice, L.; Xiao, L.; Kantamaneni, K. Endocrine Disrupting Compounds Removal Methods from Wastewater in the United Kingdom: A Review. Science 2021, 3, 11. [Google Scholar] [CrossRef]
- Khan, S.J.; Wintgens, T.; Sherman, P.; Zaricky, J.; Schäfer, A.I. Removal of Hormones and Pharmaceuticals in the Advanced Water Recycling Demonstration Plant in Queensland, Australia. Water Sci. Technol. 2004, 50, 15–22. [Google Scholar] [CrossRef]
- Sahar, E.; David, I.; Gelman, Y.; Chikurel, H.; Aharoni, A.; Messalem, R.; Brenner, A. The Use of RO to Remove Emerging Micropollutants Following CAS/UF or MBR Treatment of Municipal Wastewater. Desalination 2011, 273, 142–147. [Google Scholar] [CrossRef]
- Dolar, D.; Gros, M.; Rodriguez-Mozaz, S.; Moreno, J.; Comas, J.; Rodriguez-Roda, I.; Barceló, D. Removal of Emerging Contaminants from Municipal Wastewater with an Integrated Membrane System, MBR-RO. J. Hazard. Mater. 2012, 239–240, 64–69. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Li, M.; Dong, J.; Sun, C.; Chen, G. Removal of Pharmaceutical and Personal Care Products (PPCPs) from Municipal Waste Water with Integrated Membrane Systems, MBR-RO/NF. Int. J. Environ. Res. Public Health 2018, 15, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, C.M.; Palaniandy, P.; Dahlan, I. Pharmaceutical Residues in Aquatic Environment and Water Remediation by TiO2 Heterogeneous Photocatalysis: A Review. Environ. Earth Sci. 2017, 76, 1–19. [Google Scholar] [CrossRef]
- Watanabe, K.; Batikian, C.M.; Pelley, D.; Carlson, B.; Pitt, J.; Gersberg, R.M. Occurrence of Stimulant Drugs of Abuse in a San Diego, CA, Stream and Their Consumption Rates in the Neighboring Community. Water Air Soil Pollut. 2020, 231, 1–11. [Google Scholar] [CrossRef]
- Luine, V.N. Estradiol and Cognitive Function: Past, Present and Future. Horm. Behav. 2014, 66, 602–618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, W.; Zhou, H.; Cicek, N. Removal Mechanisms of 17β-Estradiol and 17α-Ethinylestradiol in Membrane Bioreactors. Water Sci. Technol. 2012, 66, 1263–1269. [Google Scholar] [CrossRef]
- Chelme-Ayala, P.; El-Din, M.G.; Smith, D.W.; Adams, C.D. Oxidation Kinetics of Two Pesticides in Natural Waters by Ozonation and Ozone Combined with Hydrogen Peroxide. Water Res. 2011, 45, 2517–2526. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-C.; Choo, K.-H. Hybridization of TiO2 Photocatalysis with Coagulation and Flocculation for 1,4-Dioxane Removal in Drinking Water Treatment. Chem. Eng. J. 2013, 231, 227–235. [Google Scholar] [CrossRef]
- Guillon, A.; Videloup, C.; Leroux, C.; Bertin, H.; Philibert, M.; Baudin, I.; Bruchet, A.; Esperanza, M. Occurrence and Fate of 27 Triazines and Metabolites within French Drinking Water Treatment Plants. Water Sci. Technol. Water Supply 2019, 19, 463–471. [Google Scholar] [CrossRef]
- Wang, X.; Mao, Y.; Tang, S.; Yang, H.; Xie, Y.F. Disinfection Byproducts in Drinking Water and Regulatory Compliance: A Critical Review. Front. Environ. Sci. Eng. 2015, 9, 3–15. [Google Scholar] [CrossRef]
- Aldeguer Esquerdo, A.; Varo Galvañ, P.J.; Sentana Gadea, I.; Prats Rico, D. Activated Carbon and Ozone to Reduce Simazine in Water. Water 2020, 12, 2900. [Google Scholar] [CrossRef]
- Ormad, M.P.; Miguel, N.; Claver, A.; Matesanz, J.M.; Ovelleiro, J.L. Pesticides Removal in the Process of Drinking Water Production. Chemosphere 2008, 71, 97–106. [Google Scholar] [CrossRef]
- Matamoros, V.; Rodríguez, Y.; Albaigés, J. A Comparative Assessment of Intensive and Extensive Wastewater Treatment Technologies for Removing Emerging Contaminants in Small Communities. Water Res. 2016, 88, 777–785. [Google Scholar] [CrossRef]
- Wang, Y.; Shu, L.; Jegatheesan, V.; Gao, B. Removal and Adsorption of Diuron through Nanofiltration Membrane: The Effects of Ionic Environment and Operating Pressures. Sep. Purif. Technol. 2010, 74, 236–241. [Google Scholar] [CrossRef]
- Moussavi, G.; Hosseini, H.; Alahabadi, A. The Investigation of Diazinon Pesticide Removal from Contaminated Water by Adsorption onto NH4Cl-Induced Activated Carbon. Chem. Eng. J. 2013, 214, 172–179. [Google Scholar] [CrossRef]
- Xiao, Y.; Fan, R.; Zhang, L.; Yue, J.; Webster, R.D.; Lim, T.-T. Photodegradation of Iodinated Trihalomethanes in Aqueous Solution by UV 254 Irradiation. Water Res. 2014, 49, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Rana, D.B.; Yenkie, M.K.N.; Khaty, N.T. Grey Water Treatment by Combine Processes like Vertical Flow Constructed Wetland and H2O2/UV Photo-Assisted Oxidation Process to Obtain Water of Suitable Quality that can be Reused Directly. J. Chem. Chem. Sci. 2016, 6, 339–349. [Google Scholar]
- Barrios, J.A.; Becerril, E.; De León, C.; Barrera-Díaz, C.; Jiménez, B. Electrooxidation Treatment for Removal of Emerging Pollutants in Wastewater Sludge. Fuel 2015, 149, 26–33. [Google Scholar] [CrossRef]
- Miralles-Cuevas, S.; Oller, I.; Ruiz Aguirre, A.; Sánchez Pérez, J.A.; Malato Rodríguez, S. Removal of Pharmaceuticals at Microg L−1 by Combined Nanofiltration and Mild Solar Photo-Fenton. Chem. Eng. J. 2014, 239, 68–74. [Google Scholar] [CrossRef]
- Wilde, M.L.; Montipó, S.; Martins, A.F. Degradation of β-Blockers in Hospital Wastewater by Means of Ozonation and Fe2+/Ozonation. Water Res. 2014, 48, 280–295. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; He, C.; Liu, X.; Wu, J.; Hu, Y.; Zhang, Y. Removal of Perfluorinated Compounds by Membrane Bioreactor with Powdered Activated Carbon (PAC): Adsorption onto Sludge and PAC. Desalination 2014, 334, 23–28. [Google Scholar] [CrossRef]
- Illés, E.; Szabó, E.; Takács, E.; Wojnárovits, L.; Dombi, A.; Gajda-Schrantz, K. Ketoprofen Removal by O3 and O3/UV Processes: Kinetics, Transformation Products and Ecotoxicity. Sci. Total Environ. 2014, 472, 178–184. [Google Scholar] [CrossRef]
- Sarasidis, V.C.; Plakas, K.V.; Patsios, S.I.; Karabelas, A.J. Investigation of Diclofenac Degradation in a Continuous Photo-Catalytic Membrane Reactor. Influence of Operating Parameters. Chem. Eng. J. 2014, 239, 299–311. [Google Scholar] [CrossRef]
- Vergili, I. Application of Nanofiltration for the Removal of Carbamazepine, Diclofenac and Ibuprofen from Drinking Water Sources. J. Environ. Manag. 2013, 127, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Wols, B.A.; Hofman-Caris, C.H.M.; Harmsen, D.J.H.; Beerendonk, E.F. Degradation of 40 Selected Pharmaceuticals by UV/H2O2. Water Res. 2013, 47, 5876–5888. [Google Scholar] [CrossRef]
- Appleman, T.D.; Dickenson, E.R.V.; Bellona, C.; Higgins, C.P. Nanofiltration and Granular Activated Carbon Treatment of Perfluoroalkyl Acids. J. Hazard. Mater. 2013, 260, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Sanches, S.; Penetra, A.; Rodrigues, A.; Cardoso, V.V.; Ferreira, E.; Benoliel, M.J.; Barreto Crespo, M.T.; Crespo, J.G.; Pereira, V.J. Removal of Pesticides from Water Combining Low Pressure UV Photolysis with Nanofiltration. Sep. Purif. Technol. 2013, 115, 73–82. [Google Scholar] [CrossRef]
- Li, X.; Zhang, P.; Jin, L.; Shao, T.; Li, Z.; Cao, J. Efficient Photocatalytic Decomposition of Perfluorooctanoic Acid by Indium Oxide and Its Mechanism. Environ. Sci. Technol. 2012, 46, 5528–5534. [Google Scholar] [CrossRef]
- Yuan, H.; Zhang, Y.; Zhou, X. Degradation of Bezafibrate with UV/H2O2 in Surface Water and Wastewater Treatment Plant Effluent. Clean 2012, 40, 239–245. [Google Scholar] [CrossRef]
- Sanches, S.; Penetra, A.; Rodrigues, A.; Ferreira, E.; Cardoso, V.V.; Benoliel, M.J.; Barreto Crespo, M.T.; Pereira, V.J.; Crespo, J.G. Nanofiltration of Hormones and Pesticides in Different Real Drinking Water Sources. Sep. Purif. Technol. 2012, 94, 44–53. [Google Scholar] [CrossRef]
- Pereira, V.J.; Galinha, J.; Barreto Crespo, M.T.; Matos, C.T.; Crespo, J.G. Integration of Nanofiltration, UV Photolysis, and Advanced Oxidation Processes for the Removal of Hormones from Surface Water Sources. Sep. Purif. Technol. 2012, 95, 89–96. [Google Scholar] [CrossRef]
- Cesaro, A.; Belgiorno, V. Removal of Endocrine Disruptors from Urban Wastewater by Advanced Oxidation Processes (AOPs): A Review. Open Biotechnol. J. 2016, 10, 151–172. [Google Scholar] [CrossRef]
- Heo, J.; Flora, J.R.V.; Her, N.; Park, Y.-G.; Cho, J.; Son, A.; Yoon, Y. Removal of Bisphenol A and 17β-Estradiol in Single Walled Carbon Nanotubes–Ultrafiltration (SWNTs–UF) Membrane Systems. Sep. Purif. Technol. 2012, 90, 39–52. [Google Scholar] [CrossRef]
- Melo-Guimarães, A.; Torner-Morales, F.J.; Durán-Álvarez, J.C.; Jiménez-Cisneros, B.E. Removal and Fate of Emerging Contaminants Combining Biological, Flocculation and Membrane Treatments. Water Sci. Technol. 2013, 67, 877–885. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).