Spatial Variability of Groundwater Quality for Freshwater Production in a Semi-Arid Area
Abstract
:1. Introduction
2. Materials and Methods
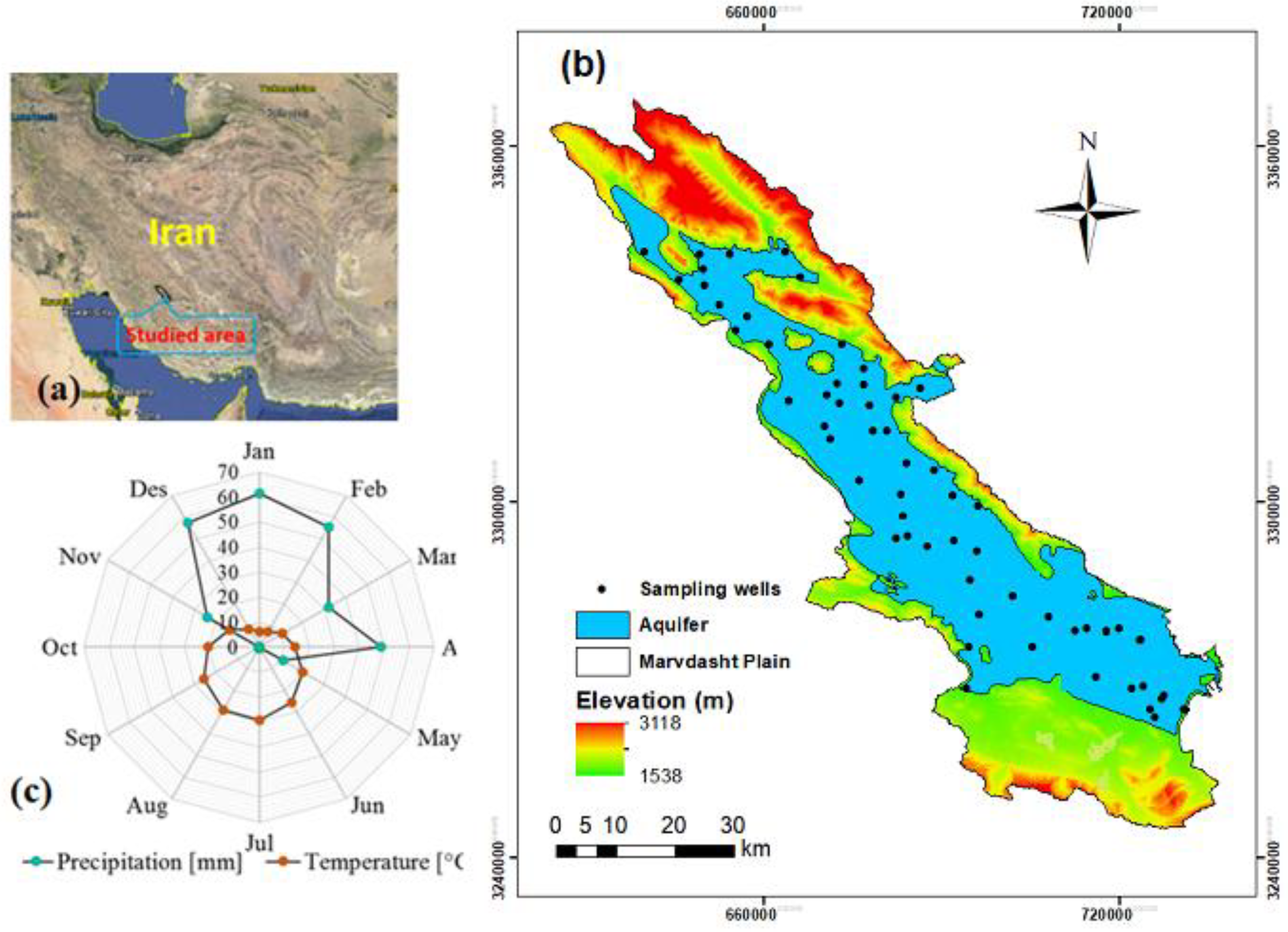
2.1. Study Area
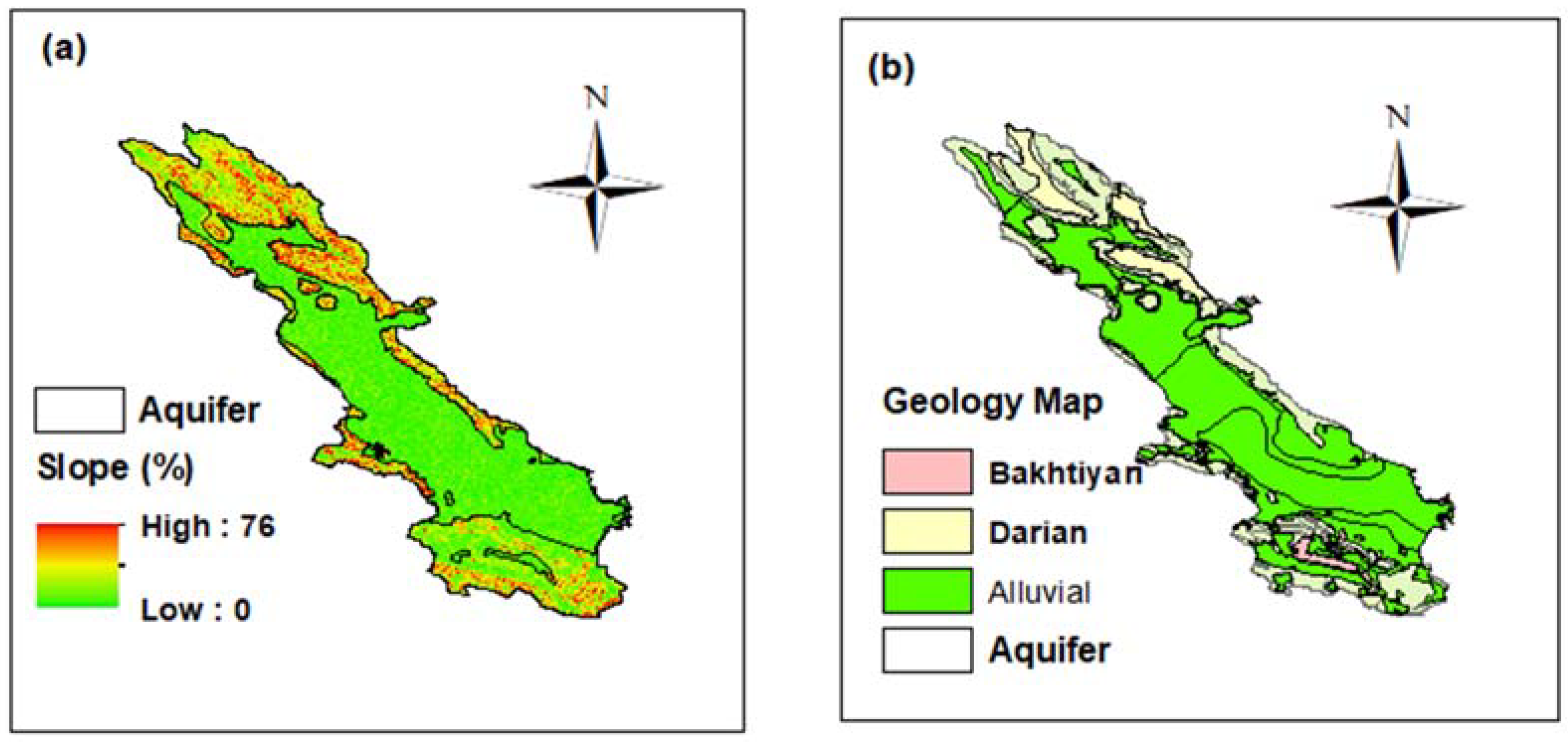
2.2. Marvdasht Aquifer
2.3. Geology of the Study Site
2.4. Sampling and Analysis
2.5. Drinking Groundwater Index (DGWQI)
2.6. DGWQI Sensitivity Analysis
2.7. Geostatistical Analysis
2.8. Performance Evaluation
3. Results and Discussion
3.1. Descriptive of Groundwater Quality Indicator
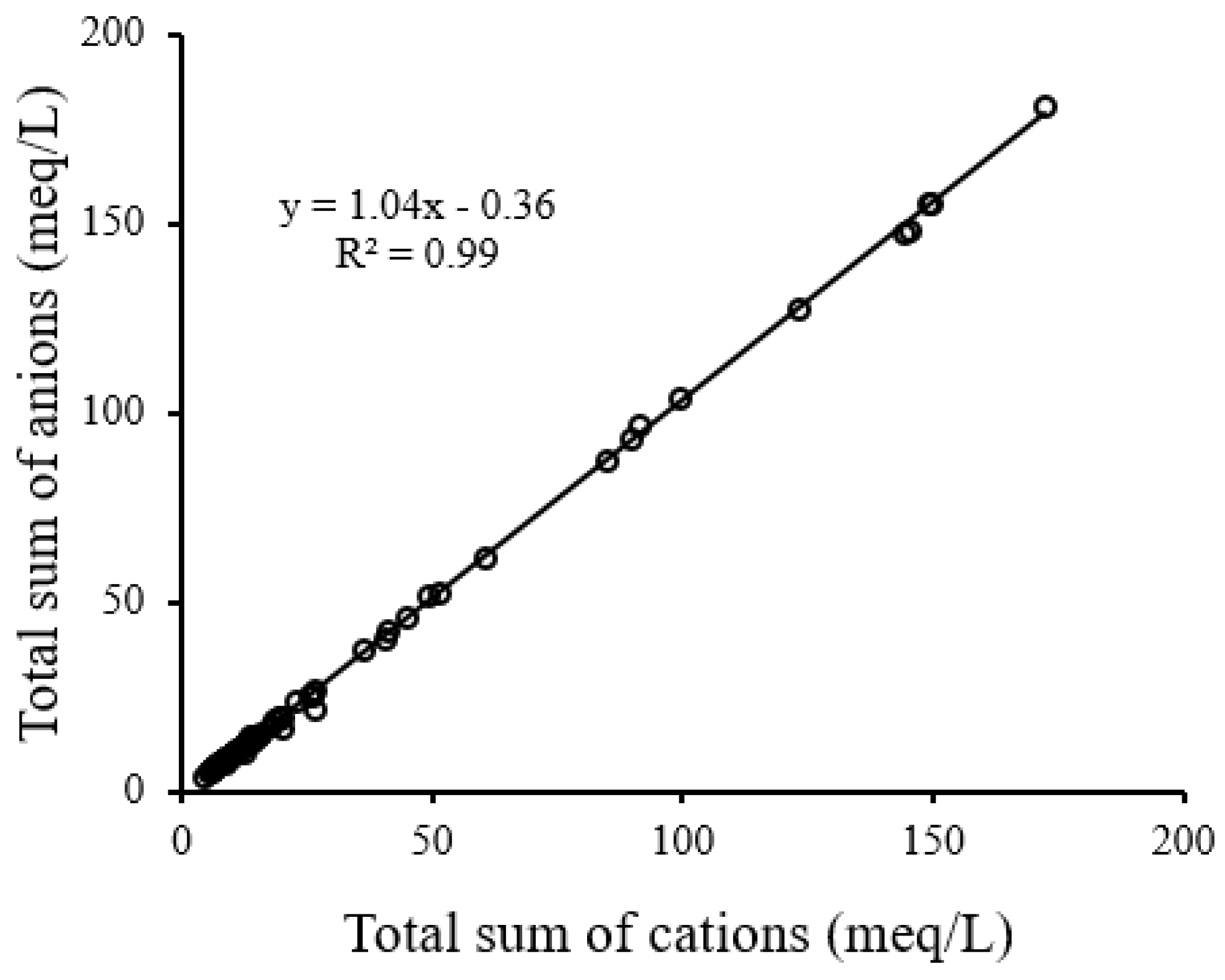
3.2. Hydro-Geochemical Assessment
3.3. Calculation of Drinking Groundwater Quality Index (DGWQI)
3.4. Sensitivity Analysis
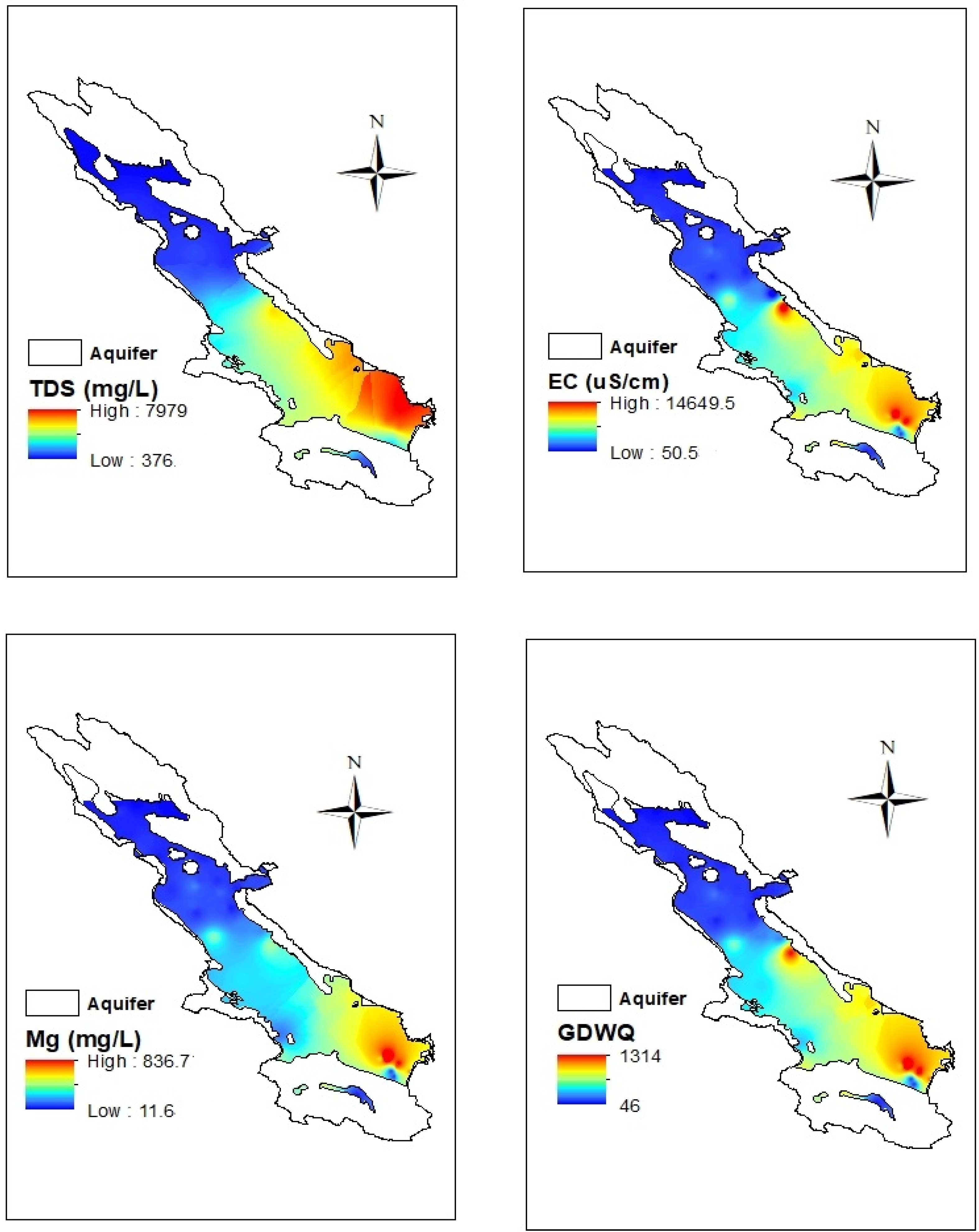
3.5. Geostatistical Analysis of DGWQI
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Honarbakhsh, A.; Azma, A.; Nikseresht, N.; Mousazadeh, M.; Eftekhari, M.; Ostovari, Y. Hydro-chemical assessment and GIS-mapping of groundwater quality indicators in semi-arid regions. J. Water Supply Res. Technol.-Aqua 2019, 68, 509–522. [Google Scholar] [CrossRef]
- Honarbakhsh, A.; Tahmoures, M.; Tashayo, B.; Mousazadeh, M.; Ingram, B.; Ostovari, Y. GIS-based assessment of groundwater quality for drinking purpose in northern part of Fars province, Marvdasht. J. Water Supply Res. Technol.-Aqua 2019, 68, 187–196. [Google Scholar] [CrossRef]
- Ostovari, Y.; Asgari, K.; Beigi-Harchegani, H. A fuzzy logic approach for assessment and mapping of groundwater irrigation quality: A case study of Marvdasht aquifer, Iran. Arch. Agron. Soil Sci. 2015, 61, 711–723. [Google Scholar] [CrossRef]
- Ostovari, Y.; Beigi, H.; Heshmati, S. A GIS-based assessment of drinking quality of Lordegan groundwater using a GWQ index. Iran. Remote Sens. GIS 2016, 7, 107–120. (In Persian) [Google Scholar]
- Babiker, I.; Mohamed, A.; Hiyama, T. Assessing groundwater quality using GIS. Water Resour. Manag. 2007, 21, 699–715. [Google Scholar] [CrossRef]
- Bawoke, G.; Anteneh, Z.L. Spatial assessment and appraisal of groundwater suitability for drinking consumption in Andasa watershed using water quality index (WQI) and GIS techniques: Blue Nile Basin, Northwestern Ethiopia. Cogent Eng. 2020, 7, 1748950. [Google Scholar] [CrossRef]
- Sandara-Kumar, K.; Kumar Pitta, S.; Ratnakanth, B.; Hanumantha, R. Assessment and mapping of groundwater quality using geographical information systems. Int. J. Eng. Sci. Technol. 2010, 2, 6035–6046. [Google Scholar]
- Zhang, K.; Wang, Q.; Chao, L.; Ye, J.; Li, Z.; Yu, Z.; Tao, Y.; Ju, Q. Ground observation-based analysis of soil moisture spatiotemporal variability across a humid to semi-humid transitional zone in China. J. Hydrol. 2019, 574, 903–914. [Google Scholar] [CrossRef]
- Olusola, F.O. Groundwater quality evaluation for drinking, domestic and irrigation uses in parts of ode irele local government area of Ondo state, Nigeria. Water. Conserv. Manag. 2020, 4, 32–41. [Google Scholar] [CrossRef]
- Heshmati, S.S. Mapping of some ground water quality index in Shahrekord Plain for various used. Master’s Thesis, Shahrekord University, Shahrekord, Iran, 2011; p. 130. (In Persian). [Google Scholar]
- Jafari, K.; Asghari, F.B.; Hoseinzade, E.; Heidari, Z.; Radfard, M.; Faraji, H. Groundwater quality assessment for drinking and agriculture purposes in Abhar city, Iran. Data Brief 2019, 19, 1033–1039. [Google Scholar] [CrossRef]
- Mehrjerdi, R.; Zareian, M.; Mahmodi, S.; Heidari, A. Spatial distribution of groundwater quality with geostatistics (Case study: Yazd-Ardakan plain). World. Appl. Sci. J. 2008, 4, 9–17. [Google Scholar]
- Wang, P.; Wang, L.; Leung, H.; Zhang, G. Super-Resolution Mapping Based on Spatial-Spectral Correlation for Spectral Imagery. IEEE. Trans. Geosci. Remote Sens. 2021, 59, 2256–2268. [Google Scholar] [CrossRef]
- Yang, S.; Wan, X.; Wei, K.; Ma, W.; Wang, Z. Silicon recovery from diamond wire saw silicon powder waste with hydrochloric acid pretreatment: An investigation of Al dissolution behavior. Waste Manag. 2021, 120, 820–827. [Google Scholar] [CrossRef] [PubMed]
- Boyd, C.E. Water Quality, an Introduction; Springer International Publishing: Cham, Switzerland, 2000; 323p. [Google Scholar]
- Xiong, Z.; Tang, Z.; Chen, X.; Zhang, X.; Zhang, K.; Ye, C. Research on Image Retrieval Algorithm Based on Combination of Color and Shape Features. J. Signal Process. Syst. 2019, 93, 139–146. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; Recommendations; World Health Organization (WHO): Geneva, Switzerland, 2011; Volume 1, p. 564. [Google Scholar]
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; Incorporating the 1st Addendum; Health Organization (WHO): Geneva, Switzerland, 2017; Volume 24, 631p. [Google Scholar]
- Chiles, J.P.; Delfiner, P. Geostatistics. In Modeling Spatial Uncertainty; John Wiley and Sons: New York, NY, USA, 1999; 695p. [Google Scholar]
- Belkhiri, L.; Tiri, A.; Mouni, L. Spatial distribution of the groundwater quality using kriging and Co-kriging interpolations. Groundw. Sustain. Dev. 2020, 11, 100473. [Google Scholar] [CrossRef]
- Julínek, T.; Duchan, D.; Říha, J. Mapping of uplift hazard due to rising groundwater level during floods. J. Flood Risk Manag. 2020, 34, 162–174. [Google Scholar] [CrossRef]
- Venkatramanan, S.; Chung, S.Y.; Kim, T.H.; Prasanna, M.V.; Hamm, S.Y. 2014 Assessment and Distribution of Metals Contamination in Groundwater: A Case Study of Busan City, Korea. Water Qual. Expo Health 2015, 7, 219–225. [Google Scholar] [CrossRef]
- Venkatramanan, S.; Viswanathan, P.M.; Chung, S.Y. GIS and Geostatistical Techniques for Groundwater Science, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2019; 389p. [Google Scholar]
- Petrula, L.; Říha, J.; Julínek, T. Evaluation of groundwater contamination in former military airport area. Gradevinar 2018, 70, 337–343. [Google Scholar]
- Mohebbi, M.R.; Reza Saeedi, R.; Montazeri, A.; Labbafi, S.; Oktaie, S.; Abtahi, M.; Mohagheghian, A. Assessment of water quality in groundwater resources of Iran using a modified drinking water quality index (DWQI). Ecol. Indic. 2013, 30, 28–34. [Google Scholar] [CrossRef]
- Soleimani, H.; Nasri, O.; Ojaghi, B.; Pasalarib, H.; Hosseini, M.; Hashemzadeh, B.; Kavosi, A.; Masoumi, S.; Radfard, M.; Adibzadeh, A.; et al. Data on drinking water quality using water quality index (WQI) and assessment of groundwater quality for irrigation purposes in Qorveh & Dehgolan, Kurdistan, Iran. Data Brief 2018, 20, 375–386. [Google Scholar]
- Bidhuri, S.; Khan, M.M.A. Assessment of ground water quality of central and southeast districts of NCT of Delhi. J. Geol. Soc. India 2020, 95, 95–103. [Google Scholar] [CrossRef]
- USDA. Keys to Soil Taxonomy, 11th ed.; USDA National Resources Conservation Service: Washington, DC, USA, 2010. [Google Scholar]
- Sangab-Zgros, C. Report of the Fars Regional Water Authority; Detailed Project on the Study of Groundwater of Marvdasht-Kharame Plain; Shiraz Water Organization: Shiraz, Iran, 2009; 225p. [Google Scholar]
- APHA. Standard Methods for Examination of Water and Wastewater, 20th ed.; American Public Health Association Inc.: Washington, DC, USA, 1998. [Google Scholar]
- Boyd, C.E. Water Quality, an Introduction, 3rd ed.; Springer International Publishing: Cham, Switzerland, 2016; 323p. [Google Scholar]
- Ramakrishnaiah, C.R.; Sadadhiv, C.; Rangnna, G. Assessment of water quality index for the groundwater in Tumkur Taluk, Karnataka State, India. J. Chem. 2009, 6, 523–530. [Google Scholar] [CrossRef] [Green Version]
- Lodwick, W.A.; Monson, W.; Svoboda, L. Attribute error and sensitivity analysis ofmap operations in geographical information systems: Suitability analysis. Int. J. Geogr. Inform. Syst. 1990, 4, 413–428. [Google Scholar]
- Machiwal, D.; Madan, K.J.; Bimal, C.M. GIS-based assessment and characterization of groundwater quality in a hard-rock hilly terrain of Western India. Environ. Monit. Assess. 2011, 174, 645–663. [Google Scholar] [CrossRef]
- ESRI Inc. ArcGisTM. Version 10.0. 2010. Available online: www.esri.com (accessed on 3 October 2021).
- Matheron, G. Principles of geostatistics. Econ. Geol. 1963, 58, 1246–1266. [Google Scholar] [CrossRef]
- Webster, R.; Oliver, M.A. Geostatistics for Environmental Scientist; John Wiley and Sons: Hoboken, NJ, USA, 2001. [Google Scholar]
- Zekai, S. Spatial Modeling Principles in Earth Sciences; Springer: Berlin/Heidelberg, Germany, 2009; 358p. [Google Scholar]
- Mirzaee, S.; Ghorbani-Dashtaki, S.; Mohammadi, J.; Asadi, H.; Asadzadeh, F. Spatial variability of soil organic matter using remote sensing data. Catena 2016, 145, 118–127. [Google Scholar] [CrossRef]
- Tashayo, B.; Honarbakhsh, A.; Akbari, M.; Ostovari, Y. Digital mapping of Philip model parameters for prediction of water infiltration at the watershed scale in a semi-arid region of Iran. Geoderma Reg. 2020, 22, e00301. [Google Scholar] [CrossRef]
- Ostovari, Y.; Ghorbani, S.; Bahrami, H. Soil loss prediction by an integrated system using RUSLE. GIS and remote sensing in semi-arid region. Geoderma Reg. 2017, 11, 28–36. [Google Scholar] [CrossRef]
- Ostovari, Y.; Ghorbani, S.; Bahrami, H. Modification of the USLE K factor for soil erodibility assessment on calcareous soils in Iran. Geomorphology 2016, 273, 385–395. [Google Scholar] [CrossRef]
- Ostovari, Y.; Moosavi, A.A.; Pourghasemi, H.R. Soil loss tolerance in calcareous soils of a semiarid region: Evaluation, prediction, and influential parameters. Land Degrad. Dev. 2020, 31, 2156–2167. [Google Scholar] [CrossRef]
- Kalantari, N.; Rahimi, M.H.; Akbari, A. Hydrochemical study of the intermediate water plain using statistical models, hydro-chemical charts and fuzzy logic. Geol. Q. Iran. 2009, 9, 15–25. (In Persian) [Google Scholar]
- Ishaku, J.M. Assessment of groundwater quality index for Jimeta-Yola area, Northeastern Nigeria. J. Geol. Min. Res. 2011, 3, 219–231. [Google Scholar]
- Rafferty, R. Scaling in Geothermal Heat Pump Systems; Geo-Heat Center Oregon Institute of Technology, Campus Drive: Klamath Falls, OR, USA, 2000; pp. 11–15. [Google Scholar]
- Sanches, F. Mapping groundwater quality variables using PCA and geostatistics: A case study of Bajo Andarax, southeastern Spain. Hydrol. Sci.-J. 2001, 46, 227–242. [Google Scholar]
- Lin, X.; Li, Z.; Liang, B.; Zhai, H.; Cai, W.; Nan, J.; Tao, Y.; Wang, A. Accelerated microbial reductive dechlorination of 2,4,6-trichlorophenol by weak electrical stimulation. Water Res. 2019, 162, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Tao, L.; Yang, H.; Iglauer, S.; Wang, X.; Askari, R.; Sun, H. Stress sensitivity of fractured and vuggy Carbonate: An X-Ray computed tomography analysis. J. Geophys. Res. Solid Earth 2020, 125, e2019JB018759. [Google Scholar] [CrossRef]
- Chao, L.; Zhang, K.; Wang, J.; Feng, J.; Zhang, M. A Comprehensive evaluation of five evapotranspiration datasets based on ground and GRACE satellite observations: Implications for improvement of evapotranspirationr Algorithm. Remote Sens. 2021, 13, 2414. [Google Scholar] [CrossRef]


| Parameter | Unit | Mean | Min | Max | SD | CV | WHO (2017) |
|---|---|---|---|---|---|---|---|
| pH | 7.53 | 6.9 | 8.0 | 0.2 | 2.8 | 7.5–8.5 | |
| EC | μS/cm | 4801.2 | 50.5 | 14,649.0 | 2635.1 | 127.1 | 1500 |
| TDS | mg/L | 2400.7 | 347.2 | 7979.2 | 2823.0 | 117.1 | 1000 |
| TH | mg/L | 1153.2 | 199.6 | 5480.4 | 1364.5 | 121.1 | 500 |
| HCO3− | mg/L | 309.1 | 100.0 | 552.1 | 89.6 | 29.4 | 300 |
| SO42− | mg/L | 301.5 | 13.2 | 1501.6 | 377.3 | 125.2 | 200 |
| Cl− | mg/L | 941.6 | 13.5 | 5117.7 | 1418.1 | 150.5 | 200 |
| Ca2+ | mg/L | 217.1 | 52.0 | 838.6 | 235.1 | 108.2 | 75 |
| Mg2+ | mg/L | 148.1 | 11.6 | 836.5 | 187.4 | 128.2 | 30 |
| K | mg/L | 7.4 | 0.5 | 32.0 | 0.8 | 51 | 12 |
| Na+ | mg/L | 398.6 | 6.0 | 2200.4 | 548.8 | 137.7 | 200 |
| Parameter | Unit | WHO (2011) | Weight of Parameters | Relative Weight (GWi) | Quality Ratio (qi) | QW |
|---|---|---|---|---|---|---|
| pH | 6.5–8.5 | 2 | 0.060 | 103 | 6.1 | |
| EC | μS/cm | 500 | 3 | 0.090 | 686 | 62 |
| TDS | mg/L | 600 | 5 | 0.121 | 480 | 57 |
| TH | mg/L | 500 | 4 | 0.121 | 130 | 16 |
| TA | mg/L | 500 | 3 | 0.090 | 123 | 11 |
| SO42- | mg/L | 250 | 3 | 0.090 | 119 | 10.5 |
| Cl− | mg/L | 250 | 4 | 0.121 | 376 | 49 |
| Na+ | mg/L | 200 | 3 | 0.090 | 200 | 18 |
| K+ | mg/L | 12 | 2 | 0.060 | 61 | 3.71 |
| Ca2+ | mg/L | 100 | 2 | 0.060 | 217 | 19 |
| Mg2+ | mg/L | 150 | 2 | 0.060 | 97 | 58 |
| Sum | - | - | - | 1 | - | - |
| Parameter | pH | EC | TDS | TH | TA | SO42− | Cl− | Ca2+ | Mg2+ | K+ | Na+ |
|---|---|---|---|---|---|---|---|---|---|---|---|
| EC | −0.67 * | ||||||||||
| TDS | −0.68 * | 0.99 * | |||||||||
| TH | −0.53 * | 0.96 * | 0.97 * | ||||||||
| TA | 0.24 | −0.14 | −0.21 | −0.25 | |||||||
| SO42− | −0.53 * | 0.74 * | 0.87 * | 0.90 * | −0.21 | ||||||
| Cl− | −0.70 * | 0.98 * | 0.99 * | 0.96 * | −0.23 | 0.81 * | |||||
| Ca2+ | −0.69 * | 0.84 * | 0.96 * | 0.97 * | −0.34 | 0.87 * | 0.94 * | ||||
| Mg2+ | −0.55 * | 0.74 * | 0.83 * | 0.98 * | −0.16 | 0.88 * | 0.84 * | 0.93 * | |||
| K+ | −0.55 * | 0.98 * | 0.99 * | 0.96 * | −0.20 | 0.85 * | 0.99 * | 0.95 * | 0.93 * | ||
| Na+ | −0.66 * | 0.84 * | 0.97 * | 0.89 * | −0.16 | 0.79 * | 0.87 * | 0.85 * | 0.87 * | 0.77 * | |
| DGWQI | −0.68 * | 0.99 * | 0.99 * | 0.98 * | −0.19 | 0.87 * | 0.99 * | 0.95 * | 0.97 * | 0.99 * | 0.96 * |
| Removed Parameter | Mean | Min | Max | CV |
|---|---|---|---|---|
| EC | 20.68 | 0.30 | 28.88 | 3.33 |
| TDS | 19.04 | 14.35 | 28.41 | 2.05 |
| TH | 8.5 | 5.6 | 14.5 | 3.5 |
| TA | 9.65 | 7.5 | 21.02 | 6.51 |
| SO42− | 3.28 | 1.11 | 8.42 | 1.35 |
| Cl− | 9.54 | 1.26 | 21.98 | 6.55 |
| Ca2+ | 7.79 | 3.84 | 15.80 | 2.90 |
| Mg2+ | 18.98 | 9.33 | 28.16 | 4.27 |
| K+ | 1.14 | 0.38 | 2.92 | 0.47 |
| Na+ | 4.50 | 0.56 | 9.93 | 2.39 |
| Parameter | Model | Range (m) | Sill (C0 + C) (mg/L)2 | Nugget (C0) (mg/L)2 | Spatial Dependency | |
|---|---|---|---|---|---|---|
| TDS | spherical | 56,347 | 7,900,000 | 2,140,000 | 0.27 | Relatively strong |
| EC | spherical | 65,530 | 170,000,000 | 5,100,000 | 0.30 | Relatively strong |
| Mg2+ | spherical | 22,410 | 20,206 | 4550 | 0.44 | Relatively strong |
| DGWQI | spherical | 49,210 | 130,000 | 19,852 | 0.39 | Relatively strong |
| Parameters | Interpolation Method | R2 | RMSE | ME | CV |
|---|---|---|---|---|---|
| TDS | Ordinary kriging | 0.75 | 111.2 | 11.1 | 21.45 |
| EC | Ordinary kriging | 0.72 | 132.8 | 15.2 | 17.15 |
| Mg2+ | Ordinary kriging | 0.68 | 20.3 | 12.3 | 35.32 |
| DGWQI | Ordinary kriging | 0.61 | 22.3 | 9.5 | 24.80 |
| DGWQI Class | Water Quality Class | Area (%) | Area (km2) |
|---|---|---|---|
| 0–100 | Appropriate | 31 | 615.7 |
| 100–200 | Poor | 14 | 278.04 |
| 200–300 | Very poor | 5 | 99.3 |
| >300 | Unsuitable for drinking | 50 | 993.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Q.; Zhou, B.; Dong, F.; Liu, Z.; Ostovari, Y. Spatial Variability of Groundwater Quality for Freshwater Production in a Semi-Arid Area. Water 2021, 13, 3024. https://doi.org/10.3390/w13213024
Zhang Q, Zhou B, Dong F, Liu Z, Ostovari Y. Spatial Variability of Groundwater Quality for Freshwater Production in a Semi-Arid Area. Water. 2021; 13(21):3024. https://doi.org/10.3390/w13213024
Chicago/Turabian StyleZhang, Qi, Baohua Zhou, Fang Dong, Zhanhong Liu, and Yaser Ostovari. 2021. "Spatial Variability of Groundwater Quality for Freshwater Production in a Semi-Arid Area" Water 13, no. 21: 3024. https://doi.org/10.3390/w13213024
APA StyleZhang, Q., Zhou, B., Dong, F., Liu, Z., & Ostovari, Y. (2021). Spatial Variability of Groundwater Quality for Freshwater Production in a Semi-Arid Area. Water, 13(21), 3024. https://doi.org/10.3390/w13213024





