Engineering Microbial Consortia towards Bioremediation
Abstract
:1. Introduction
2. Microbial Cross-Feeding in Microbial Consortia
2.1. Transfer of Metabolites between Microbes
2.1.1. Costly Metabolites
2.1.2. Costless Metabolites
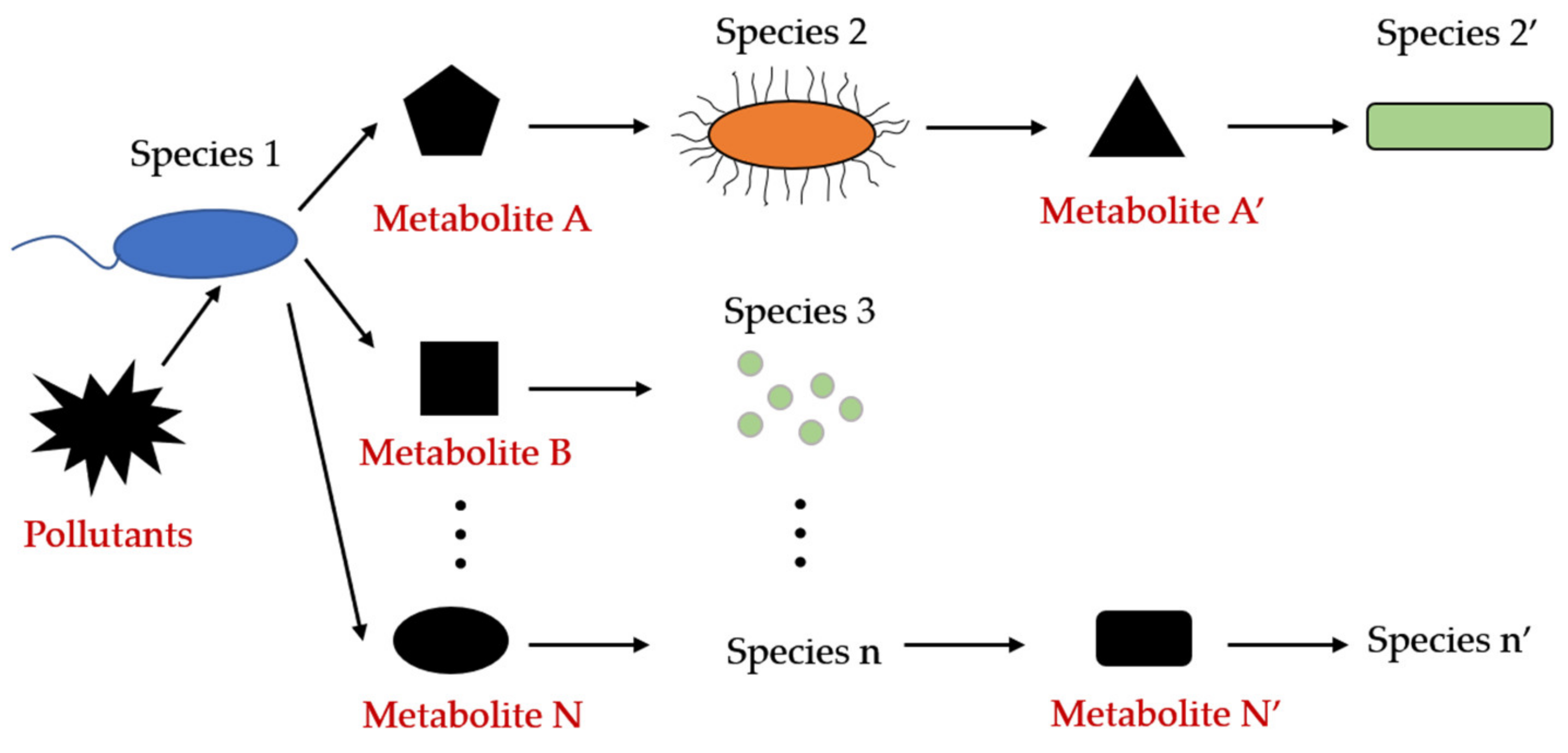
2.2. Mechanisms of Metabolites Transfer and Metabolic Interactions
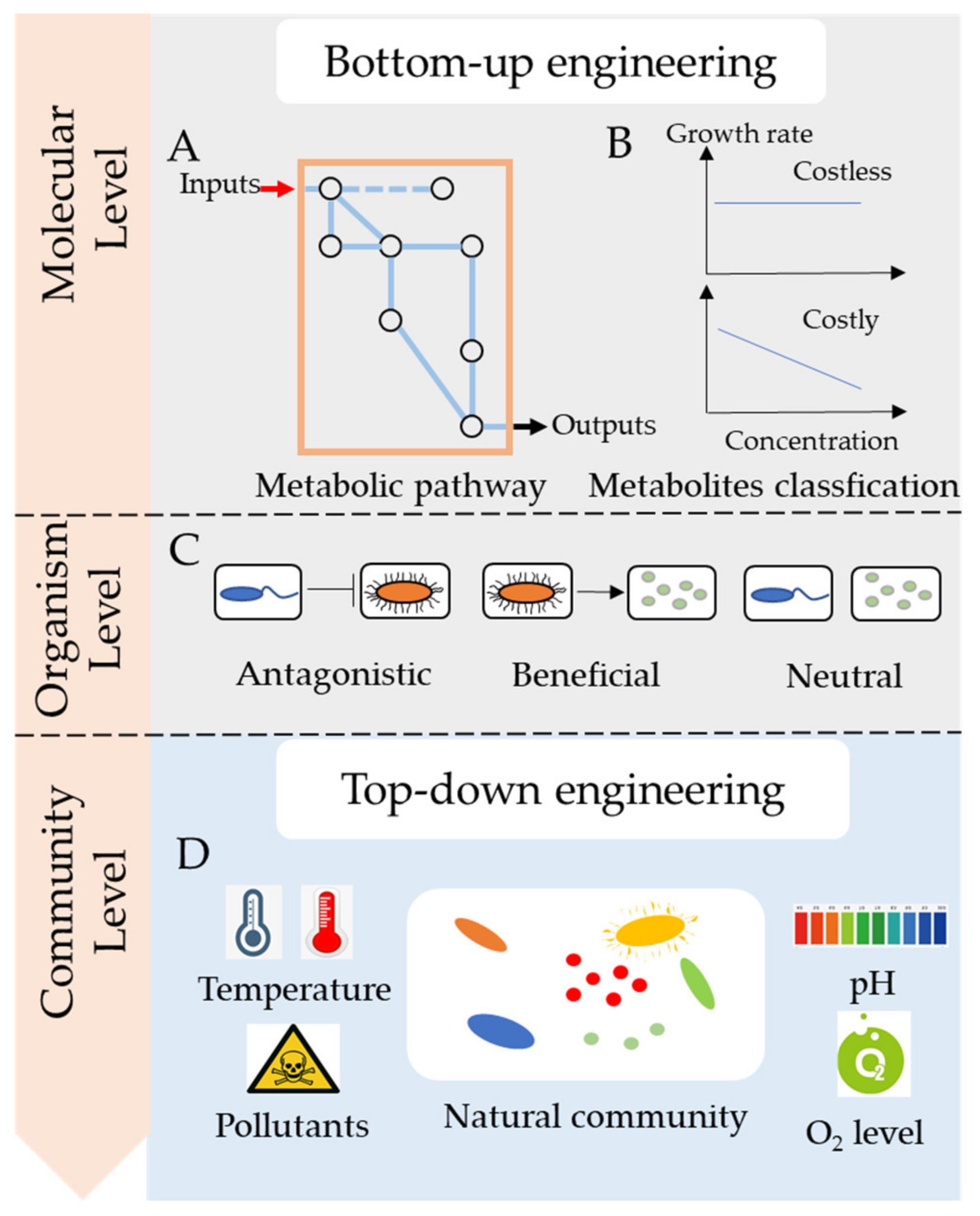
3. How to Engineer Microbial Consortia towards Bioremediation
3.1. Top-Down Engineering
Eliminating Stress Arising from Complex Pollutants
3.2. Bottom-Up Engineering
3.2.1. Making Division of Labor in Metabolic Pathways
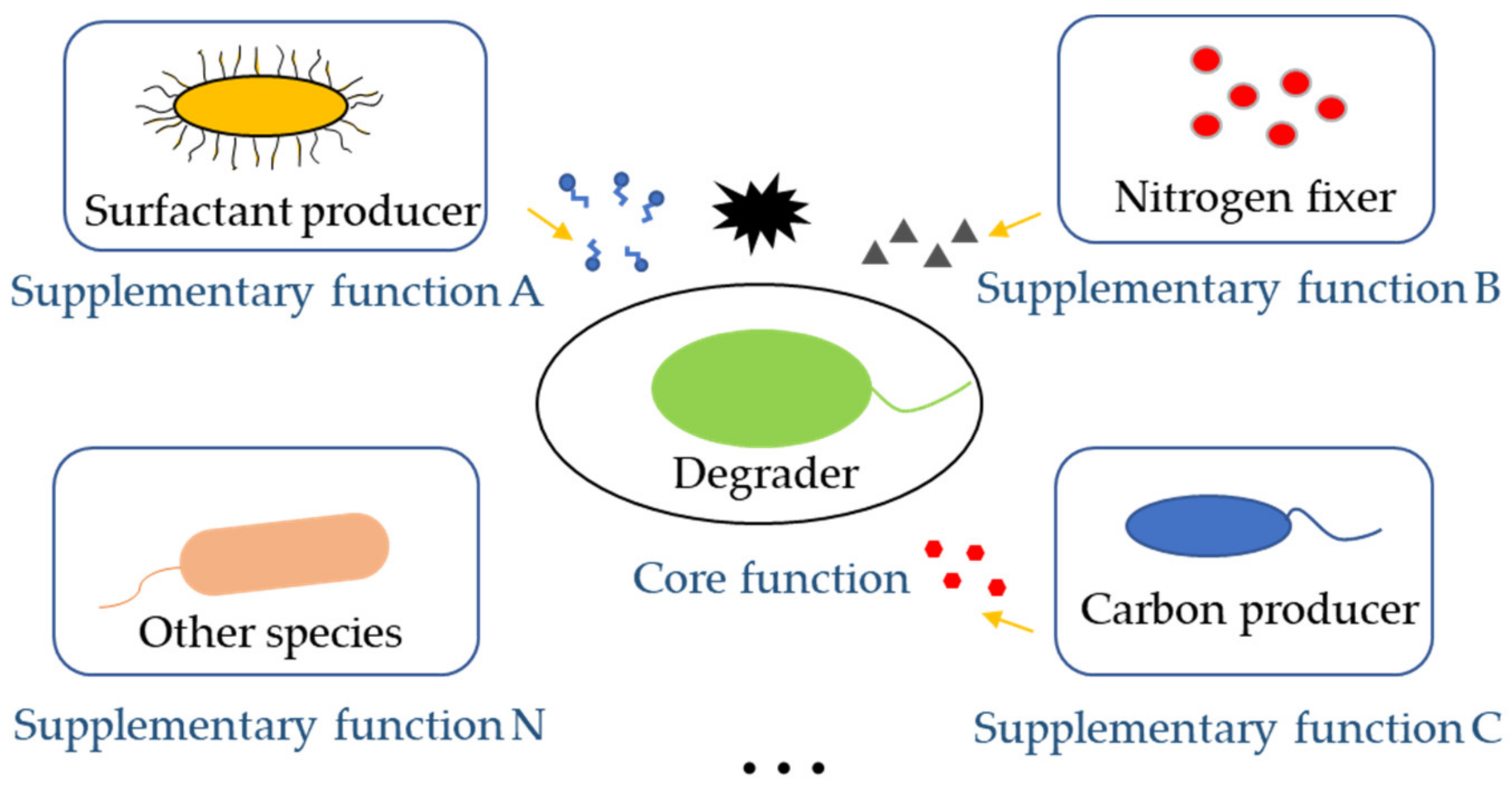
3.2.2. Keeping Multifunctionality of Microbial Communities
4. Engineering Microbial Consortia Promotes Bioremediation
4.1. Organic Pollutants
4.2. Heavy Metals
4.3. Complex Pollution
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Whitman, W.B.; Coleman, D.C.; Wiebe, W.J. Prokaryotes: The Unseen Majority. Proc. Natl. Acad. Sci. USA 1998, 95, 6578–6583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanaly, R.A.; Harayama, S. Biodegradation of High-Molecular-Weight Polycyclic Aromatic Hydrocarbons by Bacteria. J. Bacteriol. 2000, 182, 2059–2067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandrin, T.R.; Maier, R.M. Impact of Metals on the Biodegradation of Organic Pollutants. Environ. Health Perspect. 2003, 111, 1093–1101. [Google Scholar] [CrossRef]
- Brisson, V.L.; West, K.A.; Lee, P.K.H.; Tringe, S.G.; Brodie, E.L.; Alvarez-Cohen, L. Metagenomic Analysis of a Stable Trichloroethene-Degrading Microbial Community. ISME J. 2012, 6, 1702–1714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hansen, S.K.; Rainey, P.B.; Haagensen, J.A.J.; Molin, S. Evolution of Species Interactions in a Biofilm Community. Nature 2007, 445, 533–536. [Google Scholar] [CrossRef]
- Pacheco, A.R.; Moel, M.; Segrè, D. Costless Metabolic Secretions as Drivers of Interspecies Interactions in Microbial Ecosystems. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Seth, E.C.; Taga, M.E. Nutrient Cross-Feeding in the Microbial World. Front. Microbiol. 2014, 5, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Großkopf, T.; Soyer, O.S. Synthetic Microbial Communities. Curr. Opin. Microbiol. 2014, 18, 72–77. [Google Scholar] [CrossRef] [Green Version]
- Ponomarova, O.; Patil, K.R. Metabolic Interactions in Microbial Communities: Untangling the Gordian Knot. Curr. Opin. Microbiol. 2015, 27, 37–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giri, S.; Shitut, S.; Kost, C. Harnessing Ecological and Evolutionary Principles to Guide the Design of Microbial Production Consortia. Curr. Opin. Biotechnol. 2020, 62, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Hynes, W.F.; Chacón, J.; Segrè, D.; Marx, C.J.; Cady, N.C.; Harcombe, W.R. Bioprinting Microbial Communities to Examine Interspecies Interactions in Time and Space. Biomed. Phys. Eng. Express 2018, 4, 25–33. [Google Scholar] [CrossRef]
- Harcombe, W.R.; Chacón, J.M.; Adamowicz, E.M.; Chubiz, L.M.; Marx, C.J. Evolution of Bidirectional Costly Mutualism from Byproduct Consumption. Proc. Natl. Acad. Sci. USA 2018, 115, 12000–12004. [Google Scholar] [CrossRef] [Green Version]
- Harcombe, W. Novel Cooperation Experimentally Evolved between Species. Evolution 2010, 64, 2166–2172. [Google Scholar] [CrossRef] [PubMed]
- Van Tatenhove-Pel, R.J.; de Groot, D.H.; Bisseswar, A.S.; Teusink, B.; Bachmann, H. Population Dynamics of Microbial Cross-Feeding Are Determined by Co-Localization Probabilities and Cooperation-Independent Cheater Growth. ISME J. 2021, 15, 3050–3061. [Google Scholar] [CrossRef] [PubMed]
- Tarnita, C.E. The Ecology and Evolution of Social Behavior in Microbes. J. Exp. Biol. 2017, 220, 18–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Souza, G.; Shitut, S.; Preussger, D.; Yousif, G.; Waschina, S.; Kost, C. Ecology and Evolution of Metabolic Cross-Feeding Interactions in Bacteria. Nat. Prod. Rep. 2018, 35, 455–488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- West, S.A.; Murray, M.G.; Machado, C.A.; Griffin, A.S.; Herre, E.A. Testing Hamilton’s Rule with Competition between Relatives. Nature 2001, 409, 510–513. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Jacquiod, S.; Brejnrod, A.; Russel, J.; Burmølle, M.; Sørensen, S.J. Deciphering Links between Bacterial Interactions and Spatial Organization in Multispecies Biofilms. ISME J. 2019, 13, 3054–3066. [Google Scholar] [CrossRef] [PubMed]
- Mitri, S.; Clarke, E.; Foster, K.R. Resource Limitation Drives Spatial Organization in Microbial Groups. ISME J. 2016, 10, 1471–1482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammarlund, S.P.; Chacón, J.M.; Harcombe, W.R. A Shared Limiting Resource Leads to Competitive Exclusion in a Cross-Feeding System. Environ. Microbiol. 2019, 21, 759–771. [Google Scholar] [CrossRef] [PubMed]
- Stump, S.M.C.; Johnson, E.C.; Klausmeier, C.A. Local Interactions and Self-Organized Spatial Patterns Stabilize Microbial Cross-Feeding against Cheaters. J. R. Soc. Interface 2018, 15, 20170822. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCully, A.L.; LaSarre, B.; McKinlay, J.B. Growth-Independent Cross-Feeding Modifies Boundaries for Coexistence in a Bacterial Mutualism. Environ. Microbiol. 2017, 19, 3538–3550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zengler, K.; Zaramela, L.S. The Social Network of Microorganisms—How Auxotrophies Shape Complex Communities. Nat. Rev. Microbiol. 2018, 16, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Zuñiga, C.; Li, C.T.; Yu, G.; Al-Bassam, M.M.; Li, T.; Jiang, L.; Zaramela, L.S.; Guarnieri, M.; Betenbaugh, M.J.; Zengler, K. Environmental Stimuli Drive a Transition from Cooperation to Competition in Synthetic Phototrophic Communities. Nat. Microbiol. 2019, 4, 2184–2191. [Google Scholar] [CrossRef] [PubMed]
- Lawson, C.E.; Harcombe, W.R.; Hatzenpichler, R.; Lindemann, S.R.; Löffler, F.E.; O’Malley, M.A.; García Martín, H.; Pfleger, B.F.; Raskin, L.; Venturelli, O.S.; et al. Common Principles and Best Practices for Engineering Microbiomes. Nat. Rev. Microbiol. 2019, 17, 725–741. [Google Scholar] [CrossRef] [Green Version]
- Löffler, F.E.; Edwards, E.A. Harnessing Microbial Activities for Environmental Cleanup. Curr. Opin. Biotechnol. 2006, 17, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Olaniran, A.O.; Balgobind, A.; Pillay, B. Impacts of Heavy Metals on 1,2-Dichloroethane Biodegradation in Co-Contaminated Soil. J. Environ. Sci. 2009, 21, 661–666. [Google Scholar] [CrossRef]
- Qiu, R.; Zhang, D.; Diao, Z.; Huang, X.; He, C.; Morel, J.-L.; Xiong, Y. Visible Light Induced Photocatalytic Reduction of Cr(VI) over Polymer-Sensitized TiO2 and Its Synergism with Phenol Oxidation. Water Res. 2012, 46, 2299–2306. [Google Scholar] [CrossRef]
- Gauthier, P.T.; Norwood, W.P.; Prepas, E.E.; Pyle, G.G. Metal-Polycyclic Aromatic Hydrocarbon Mixture Toxicity in Hyalella Azteca. 2. Metal Accumulation and Oxidative Stress as Interactive Co-Toxic Mechanisms. Environ. Sci. Technol. 2015, 49, 11780–11788. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.; Lu, Y.; Zhou, Q.; Hong, J. Interaction of Polycyclic Aromatic Hydrocarbons and Heavy Metals on Soil Enzyme. Chemosphere 2005, 61, 1175–1182. [Google Scholar] [CrossRef]
- Bernstein, H.C.; Carlson, R.P. Microbial Consortia Engineering for Cellular Factories: In Vitro to in Silico Systems. Comput. Struct. Biotechnol. J. 2012, 3, e201210017. [Google Scholar] [CrossRef] [Green Version]
- Rosche, B.; Breuer, M.; Hauer, B.; Rogers, P.L. Biphasic Aqueous/Organic Biotransformation of Acetaldehyde and Benzaldehyde by Zymomonas Mobilis Pyruvate Decarboxylase. Biotechnol. Bioeng. 2004, 86, 788–794. [Google Scholar] [CrossRef] [PubMed]
- Gude, S.; Taga, M.E. Multi-Faceted Approaches to Discovering and Predicting Microbial Nutritional Interactions. Curr. Opin. Biotechnol. 2020, 62, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Embree, M.; Liu, J.K.; Al-Bassam, M.M.; Zengler, K. Networks of Energetic and Metabolic Interactions Define Dynamics in Microbial Communities. Proc. Natl. Acad. Sci. USA 2015, 112, 15450–15455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faust, K.; Raes, J. Microbial Interactions: From Networks to Models. Nat. Rev. Microbiol. 2012, 10, 538–550. [Google Scholar] [CrossRef] [PubMed]
- Kong, W.; Meldgin, D.R.; Collins, J.J.; Lu, T. Designing Microbial Consortia with Defined Social Interactions. Nat. Chem. Biol. 2018, 14, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Nishijima, W.; Soto, E.; Okada, M. Relationship between Growth Rate of Phenol Utilizing Bacteria and the Toxic Effect of Metabolic Intermediates of Trichloroethylene (TCE). Water Res. 1999, 33, 1085–1089. [Google Scholar] [CrossRef]
- Wanapaisan, P.; Laothamteep, N.; Vejarano, F.; Chakraborty, J.; Shintani, M.; Muangchinda, C.; Morita, T.; Suzuki-Minakuchi, C.; Inoue, K.; Nojiri, H.; et al. Synergistic Degradation of Pyrene by Five Culturable Bacteria in a Mangrove Sediment-Derived Bacterial Consortium. J. Hazard. Mater. 2018, 342, 561–570. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, B.; Jiang, Z.; Dong, X.; Hu, M.; Wang, Z. Metabolic Ability and Individual Characteristics of an Atrazine-Degrading Consortium DNC5. J. Hazard. Mater. 2012, 237–238, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Hempson, G.P.; Archibald, S.; Donaldson, J.E.; Lehmann, C.E.R. Alternate Grassy Ecosystem States Are Determined by Palatability–Flammability Trade-Offs. Trends Ecol. Evol. 2019, 34, 286–290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Isaac, P.; Martínez, F.L.; Bourguignon, N.; Sánchez, L.A.; Ferrero, M.A. Improved PAHs Removal Performance by a Defined Bacterial Consortium of Indigenous Pseudomonas and Actinobacteria FromPatagonia, Argentina. Int. Biodeterior. Biodegrad. 2015, 101, 23–31. [Google Scholar] [CrossRef]
- Yu, S.H.; Ke, L.; Wong, Y.S.; Tam, N.F.Y. Degradation of Polycyclic Aromatic Hydrocarbons by a Bacterial Consortium Enriched from Mangrove Sediments. Environ. Int. 2005, 31, 149–154. [Google Scholar] [CrossRef]
- Polti, M.A.; Aparicio, J.D.; Benimeli, C.S.; Amoroso, M.J. Simultaneous Bioremediation of Cr(VI) and Lindane in Soil by Actinobacteria. Int. Biodeterior. Biodegrad. 2014, 88, 48–55. [Google Scholar] [CrossRef]
- Roane, T.M.; Josephson, K.L.; Pepper, I.L. Dual-Bioaugmentation Strategy to Enhance Remediation of Cocontaminated Soil. Appl. Environ. Microbiol. 2001, 67, 3208–3215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- HuiJie, L.; CaiYun, Y.; Yun, T.; GuangHui, L.; TianLing, Z. Using Population Dynamics Analysis by DGGE to Design the Bacterial Consortium Isolated from Mangrove Sediments for Biodegradation of PAHs. Int. Biodeterior. Biodegrad. 2011, 65, 269–275. [Google Scholar] [CrossRef]
- Brune, K.D.; Bayer, T.S. Engineering Microbial Consortia to Enhance Biomining and Bioremediation. Front. Microbiol. 2012, 3, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Mosharaf, M.K.; Tanvir, M.Z.H.; Haque, M.M.; Haque, M.A.; Khan, M.A.A.; Molla, A.H.; Alam, M.Z.; Islam, M.S.; Talukder, M.R. Metal-Adapted Bacteria Isolated from Wastewaters Produce Biofilms by Expressing Proteinaceous Curli Fimbriae and Cellulose Nanofibers. Front. Microbiol. 2018, 9, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Smith, W.L. Hexavalent Chromium Reduction and Precipitation by Sulphate-Reducing Bacterial Biofilms. Environ. Geochem. Health 2001, 23, 297–300. [Google Scholar] [CrossRef]
- Dhal, B.; Thatoi, H.N.; Das, N.N.; Pandey, B.D. Chemical and Microbial Remediation of Hexavalent Chromium from Contaminated Soil and Mining/Metallurgical Solid Waste: A Review. J. Hazard. Mater. 2013, 250–251, 272–291. [Google Scholar] [CrossRef]
- Shen, H.; Wang, Y.T. Simultaneous Chromium Reduction and Phenol Degradation in a Coculture of Escherichia Coli ATCC 33456 and Pseudomonas Putida DMP-1. Appl. Environ. Microbiol. 1995, 61, 2754–2758. [Google Scholar] [CrossRef] [Green Version]
| Pollutants | Microorganism | Bioremediation Efficiency | References |
|---|---|---|---|
| Pyrene | (Mycobacterium spp. PO1 and PO2, Novosphin-gobium pentaromativorans PY1, Ochrobactrum sp. PW1, and Bacillus sp. FW1 | Three-fold higher degradation rate for pyrene than the individual degrader. | [38] |
| Atrazine | Arthrobacter sp. DNS10, Bacillus subtilis DNS4 and Variovorax sp. DNS12, Arthrobacter sp. DNS9 | Removed 100% of atrazine at initial concentration of 100 mg/L, faster than single species. | [39] |
| PAHs | Rhodococcus sp., Acinetobacter sp., and Pseudomonas sp. | 100% degradation of Fl and Phe in sediment-free liquid medium after 4 weeks of growth. | [42] |
| Cr(VI) | Streptomyces sp. A5, A11, M7, MC1 | Removed 86% of Cr(VI) at initial concentration of 50 mg/kg in soil. | [43] |
| Lindane | Removed 46% of lindane at initial concentration of 25 mg/kg in soil. | ||
| Cd | Bacillus sp. strain H9, Ralstonia eutropha JMP134 | Removed 42% of phenanthrene at initial concentration of 24 mg/L. | [44] |
| 2,4-D | Removed 100% of 2,4-D at initial concentration of 500 mg/L. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Wu, S.; Dong, Y.; Fan, H.; Bai, Z.; Zhuang, X. Engineering Microbial Consortia towards Bioremediation. Water 2021, 13, 2928. https://doi.org/10.3390/w13202928
Li X, Wu S, Dong Y, Fan H, Bai Z, Zhuang X. Engineering Microbial Consortia towards Bioremediation. Water. 2021; 13(20):2928. https://doi.org/10.3390/w13202928
Chicago/Turabian StyleLi, Xianglong, Shanghua Wu, Yuzhu Dong, Haonan Fan, Zhihui Bai, and Xuliang Zhuang. 2021. "Engineering Microbial Consortia towards Bioremediation" Water 13, no. 20: 2928. https://doi.org/10.3390/w13202928
APA StyleLi, X., Wu, S., Dong, Y., Fan, H., Bai, Z., & Zhuang, X. (2021). Engineering Microbial Consortia towards Bioremediation. Water, 13(20), 2928. https://doi.org/10.3390/w13202928









