Abstract
Stream and terrestrial ecosystems are intimately connected by riparian zones that support high biodiversity but are also vulnerable to human impacts. Landscape disturbances, overgrazing, and diffuse pollution of agrochemicals threaten riparian biodiversity and the delivery of ecosystem services in agricultural landscapes. We assessed how terrestrial invertebrate communities respond to changes in riparian vegetation in Romanian agricultural catchments, with a focus on the role of forested riparian buffers. Riparian invertebrates were sampled in 10 paired sites, with each pair consisting of an unbuffered upstream reach and a downstream reach buffered with woody riparian vegetation. Our results revealed distinct invertebrate community structures in the two site types. Out of 33 invertebrate families, 13 were unique to either forested (6) or unbuffered (7) sites. Thomisidae, Clubionidae, Tetragnathidae, Curculionidae, Culicidae, and Cicadidae were associated with forested buffers, while Lycosidae, Chrysomelidae, Staphylinidae, Coccinellidae, Tettigoniidae, Formicidae, and Eutichuridae were more abundant in unbuffered sites. Despite statistically equivocal results, invertebrate diversity was generally higher in forested riparian buffers. Local riparian attributes significantly influenced patterns in invertebrate community composition. Our findings highlight the importance of local woody riparian buffers in maintaining terrestrial invertebrate diversity and their potential contribution as a multifunctional management tool in agricultural landscapes.
1. Introduction
Riparian zones are areas of transition between stream and terrestrial ecosystems that provide heterogeneous microhabitats [1] for both terrestrial and aquatic populations, thus supporting biodiverse communities [2]. These organisms help connect aquatic and terrestrial systems: Aquatic consumers derive energy from allochthonous inputs of leaf litter and prey originating in the adjacent riparian zones, whereas aquatic organisms are consumed by riparian predators, thereby facilitating the reciprocal transfer of aquatic subsidies to terrestrial food webs [3]. Emergent aquatic insects and riparian invertebrate consumers, including spiders and beetles, play a key role in linking stream and terrestrial trophic networks [4] and contribute to biodiversity at local and catchment scales.
However, riparian zones are often highly degraded in agricultural catchments. Land-use change, deforestation (including riparian trees), overgrazing, inputs of pesticides, and nutrients from agricultural sources all threaten riparian biodiversity and ecosystem services at different spatial scales. Thus, protecting and enhancing riparian zones are often seen as the first steps towards reversing the impacts of agricultural land use [5]. Riparian buffers, typically comprising uncultivated, vegetated areas alongside streams and rivers, can play an essential role in preserving the integrity of stream–riparian networks in modified landscapes [6].
Riparian buffers help increase streambank stability and shading, thus reducing temperatures and inputs of diffuse pollutants (e.g., sediment, nutrients, and pathogens) to receiving stream ecosystems [7,8,9]. Riparian vegetation can provide habitat for plants and animals in highly fragmented agricultural landscapes [6,9]. The positive influence of riparian buffers on aquatic macroinvertebrate community structure has been documented in a number of studies [6,9,10,11,12,13]. Consequently, water quality, biodiversity, and other ecosystem services provided by streams in human-impacted landscapes are highly dependent on riparian buffer properties [9].
The enhanced biodiversity and community dynamics provided by riparian buffers may also be valuable to agricultural productivity. Many invertebrates that act as agents of biocontrol by feeding on plant or animal crop pests (e.g., carabid and staphylinid beetles) or that help pollinate crops (e.g., certain dipterans) use riparian buffers to complete their life-cycles [9,14,15,16]. These attributes add to the co-benefits of riparian buffers and point to their use as multifunctional management tools in agricultural landscapes [17].
Despite the recognition of riparian buffers as essential features to preserve ecosystem integrity in agricultural catchments, there remain gaps in our knowledge. Integrated ecological studies in recent decades have tested the effects of buffer properties, such as width, longitudinal continuity, age, species, and canopy cover, mainly on the stream community composition, diversity, and functionality [9,11,12]. However, studies addressing riparian invertebrate communities are relatively rare and have focused only on certain groups, such as beetles or spiders [2,4,10,18,19,20]. In this context, our case study aims to highlight how forested riparian buffers structure riparian invertebrate communities in a basin impacted by agriculture. We present a case study in the Argeș River Basin (Romania), with predominantly agricultural land use, which following the fall of communism, has been characterized by highly fragmented land ownership and little coordination of riparian zone management.
We investigated terrestrial invertebrate community composition and diversity in riparian zones with and without a riparian buffer of woody vegetation. We also assessed the relative and shared effects of reach (local) and catchment-scale environmental variables in structuring invertebrate community composition. We expected that the presence of woody vegetation in riparian zones would influence the diversity and composition of riparian invertebrate communities. Specifically, we hypothesized that woody riparian buffers have a positive influence on terrestrial invertebrate diversity at the local scale by providing habitat heterogeneity and refuge from disturbance. We also hypothesized that patches of woody riparian vegetation influence beta-diversity, thus helping to mitigate the effects of agricultural land-uses at the catchment scale. Providing evidence for the positive effects of woody riparian buffers would demonstrate their efficacy as a tool for biodiversity conservation and improving landscape integrity in a human-impacted landscape.
2. Materials and Methods
2.1. Study Area and Site Selection
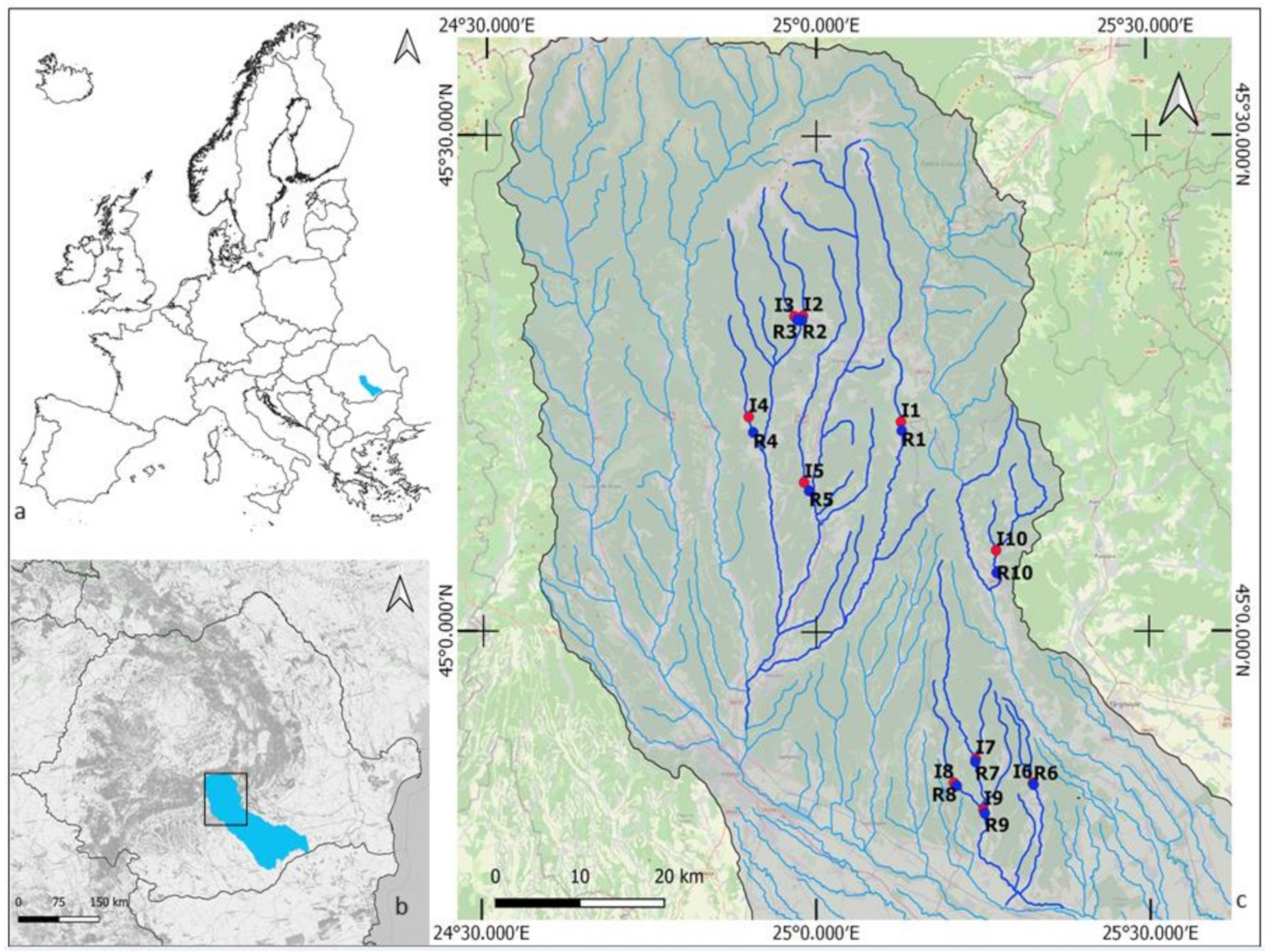
Our field sampling took place between May and June 2018 in the Middle Argeș River Basin, located in south-eastern Romania (Figure 1, Tables S1 and S2). The Argeș River is a major tributary of the River Danube, and its catchment is representative of a sylvo-steppe landscape. Agriculture is the dominant land use, representing 56% of the total catchment area of 125 km2. Other human pressures include aquaculture, forestry, hydropower, industry, and urban development [21]. Human impacts on the region’s stream–riparian networks have serious consequences, including increased flooding, water pollution, and the spread of invasive species (i.e., Fallopia japonica). Our case study was part of the CROSSLINK project, and the study design was based on the CROSSLINK paired sites approach (see [6]). We choose 10 paired stream–riparian sites in a landscape modified by agriculture, with the no woody riparian buffer site (UBF) located upstream of the forested vegetation buffer site (FBF) (see [6], Figure 1). All streams are 1st to 3rd order [22], mostly 2–6 m wide, with the streambed dominated by gravels and cobbles. Woody riparian buffers were a minimum of 50 m in length, moving upstream from the downstream end of the sampling reach, with a width of 2–3 times the wetted stream width and consist mainly of large trees (Diameter at Breast Height (DBH) > 5 cm) on both banks. The vegetation of the UBF sites (hereafter named unbuffered sites) was dominated by grass and herbs, with few shrubs and small trees (DBH < 5 cm). Information on the geographical location is listed in Table S1 (Supplementary Materials).
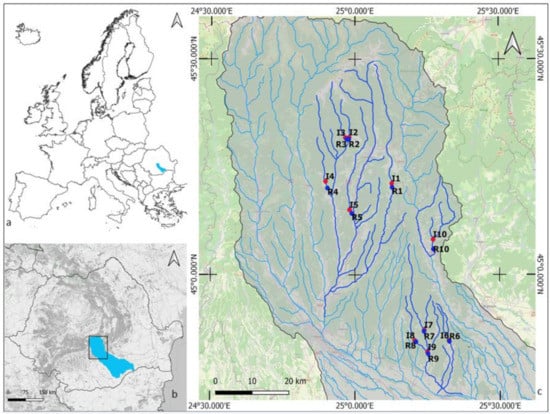
Figure 1.
The study site locations in the Argeș basin, Romania. Forested buffer sites (R1–R10) are marked in blue, and the unforested buffer sites (I1–I10) in red.
2.2. Sampling Design and Methods
We established six riparian sampling plots per site, three on each bank of the stream, attempting to cover the full heterogeneity of the study sites. The plots did not overlap and were in close proximity to each other. The plot size was 10 m in length (parallel to the stream) and 5 m in width (perpendicular to the stream), resulting in a sampling reach length of 30 m at each site (for details, see [6,10]).
2.2.1. Environmental Variables
We wanted to describe environmental factors contributing to the variation in riparian invertebrate community composition. Specifically, we wanted to reveal the role of forested riparian buffers in structuring riparian invertebrate communities. To do this, we used three categories of explanatory variables (Table 1) representing key groups of influence factors. The first category involved catchment attributes at broader spatial scales quantified from Geographical Information System (GIS) data: catchment area and elevation, Corine land cover and land use data, and the distance to the next 100 m forested buffer upstream of the site pair. The second category involved riparian habitat characteristics recorded from the quantitative field survey of the six 50 m2 rectangular plots at each sampling site. The percentage area of different vegetation/habitat categories was estimated as a vertical projection on the ground. Canopy cover at the zenith was measured in each plot’s center using the smartphone app “CanopyApp” (University of New Hampshire, Durham, NH, USA), and width and length of riparian forest patches were quantified based on the GIS data. All values were averaged to provide a single value for a site. The third and final category involved riparian integrity scores assigned for 13 qualitative attributes (Table 1). They were assessed over a 50 m study reach on each bank (30 m along of the three sampling plots and 20 m upstream) and graded from poor (1) to excellent (5), following the protocol described by Harding et al. [23] and adapted to European conditions (see Table S1, [6]). Subsequently, at each study site, bank scores were averaged to provide a single value for each attribute.

Table 1.
Explanatory variables used to explain the patterns observed in the structure of the riparian invertebrate community and assess the role of the riparian forested buffers.
2.2.2. Riparian Invertebrates
Riparian invertebrates were sampled during the transition between spring and summer (May–June) in European temperate zone regions (Table S2). The timing of sampling was intended to occur after leaf-out was complete for all tree and shrub species and adults of most invertebrate species were present and active in the field. Following Burdon et al. [6], we used a standardized semi-quantitative sampling strategy to allow for between-site comparisons. The methods used involved two approaches. The first approach used visual searching (i.e., looking for spider webs, turning over stones and wood, and riffling through leaf litter) and then collection by hand (i.e., guiding the invertebrates into labeled sample tubes). The second approach involved sweep netting in each sampling plot (see [6]). There was an exception in one buffered site (R6) where only one bank (three plots) was sampled due to the steepness and inaccessibility of the other bank. This site was considered an outlier and was excluded from the analysis. Plots were sampled methodically along transects parallel to the stream channel to allocate effort evenly. The sampled area and sampling duration (a maximum of 15 min) were recorded for each plot. Buffered and unbuffered sites in a pair were sampled on the same day, under similar weather conditions to ensure the comparability of results (Table S2). To reduce the sampling biases that may be introduced by different field workers (e.g., sampling effort and selection) [24], two trained researchers sampled all sites.
In the field, adult individuals were separated into broad taxonomic groups (i.e., spiders, beetles, dipterans), placed in separate tubes, labeled, and transported to the laboratory in a cooler bag. In the laboratory, all collected individuals were identified to the family level for most taxa and genus level for Carabidae, Staphylinidae, and Araneae, under a stereomicroscope using specific identification keys [25,26,27,28,29]. Araneae and beetles (Carabidae and Staphylinidae) are considered reliable indicators in monitoring programs [30] and are also frequently studied [31,32,33,34]. They were grouped and analyzed together as the consumer (predator) trophic group.
2.3. Data Analysis
2.3.1. Environmental Variables
To visualize the differences between the site types induced by the explanatory variables selected at different scales, we used the non-Metric Multidimensional Scaling (nMDS), with Euclidean distances, based on standardized values of the environmental variables, in accordance with [35]:
where STD is the standardized variable, x = the values of each explanatory variable, xmin and xmax = minimum and maximum values of each explanatory variable across the sites. We then used PERMANOVA (PERmutational Multivariate ANalysis Of VAriance) to assess differences in the environmental variables between sites. All these analyses were performed with PAST 4.03, the Palaeontological Association software [36].
2.3.2. Patterns in Invertebrate Community Structure
To estimate the relative abundance of invertebrates and to allow for the comparability between sites, we used a “Catch Per Unit Effort” (CPUE) approach [6]:
Based on these estimates, we investigated the patterns in invertebrate community composition (taxa and consumer abundance, %) and diversity (richness, Shannon diversity—H′ index and Pielou’s evenness index—J′) in buffered and unbuffered sites. The dissimilarity between community composition in buffered and unbuffered sites and the identity of taxa and consumer whose abundance explained up to 70–80% from the observed dissimilarity between sites were identified with SIMPER analysis (Bray–Curtis similarity measure, [37]). All these analyses were performed with PAST 4.03, the Palaeontological Association software [36]. To identify the significant differences between the two categories of sites, we used one-way ANOVA, with sqrt (for abundance) or ln (x) or ln (x + 1) (for diversity) transformed data (lmer function in the R package lme4). One-way ANOVA was also used to compare the patterns in consumer diversity. To account for spatial variation among pairs, stream identity was fitted as a blocking factor.
2.3.3. Influence of Selected Environmental Variables on Invertebrate Community Structure
The percentage abundance and consumer structure of riparian invertebrates and the explanatory data obtained from different sources (Table 1) were combined for statistical analysis, including redundancy analyses (RDA) and variance partitioning in R, version 3.6.3 (2020-02-29). To normalize data and reduce homescedascity, the percentage data were logit transformed, whereas the metric data (e.g., distance to the next 100 m riparian forested buffer block, upstream the site, riparian buffer length and width) were log or log+1 transformed using R packages car and vegan. All data were standardized by the decostand function in R (package vegan). Decostand is a widely used R function enabling standardization methods for community ecology. We applied Hellinger transformation for the invertebrate data [38] that does not give high weights to the rare species.
To detect spatial structures in communities (e.g., spatial autocorrelated data), we explicitly incorporated the spatial predictors into the statistical model. Principal Coordinates of Neighborhoods Matrix (PCNM) in R (vegan, dist) was used to decompose the spatial location of sites into PCNM functions used to express distances in the rectangular for ecologically relevant spatial scales. The descriptors of spatial relationships (PCNM base functions) were obtained by principal coordinate analysis of a truncated matrix of Euclidean (geographic) distances among the sampling sites [39]. PCNM base variables were used as spatial descriptors in the multivariate variation partitioning [40,41]. Variation partitioning (varpart, vegan package in R) was used to reveal the relative and shared effect of significant variables in shaping the riparian invertebrate communities’ structure. We used Venn diagrams to visualize the variation partitioning results. To reduce the predictor list and select significant variables for RDA and variation partitioning models, we fitted variables for each group individually (catchment-wide properties, riparian habitat characteristics, and riparian integrity condition scores) and fit global models for estimation of corresponding adjusted R2. The forward selection method in RDA, with appropriate stopping criteria (the adjusted coefficient of multiple determination, R2 adj, permutation test with 999 randomizations, and an alpha significance level of 0.05) was used to assess which variables are essential (forward.sel function in the packfor package in R). The forward selection method prevents overestimation of the explained variance [39].
To test for association between explanatory variables (paired samples), function cor.test in stats v 3.6.2 in R was used. The significance of different components was tested using ANOVA. Highly collinear predictors (e.g., p < 0.05, r > 0.65) were removed from the analysis to avoid over-parametrizing models. Function vif.cca, defined as generic in R package vegan (vif), gave the variance inflation factors for each constraint or contrast in factor constraints. Variance inflation factors are useful diagnostic tools for detecting multicollinearity. Predictor variables with VIF scores > 10 were excluded. For data visualization, the R packages gplots, ggplot2, and grid were used.
2.3.4. The Effect of Forested Riparian Buffers on Riparian Invertebrate Community Structure
To reveal the association of the riparian invertebrate structure with local riparian attributes, we specified a partial redundancy analysis model (pRDA) that conditioned out confounding influences of spatial location and characteristics at the catchment level (i.e., land-use). For this, we combined the explanatory variables from the two categories related to riparian properties: the riparian habitat characteristics and the scores from the attributes used in the Riparian Condition Index. As described above, auto-correlated variables (e.g., p < 0.05, r > 0.65) were not considered in the analysis.
Additionally, we compared the effect size (log response ratios ± 95% CI) by calculating the change in diversity indices between downstream sites buffered with forested riparian vegetation and the unbuffered upstream sites (i.e., without woody riparian vegetation).
3. Results
3.1. Environmental Differences between Site Types
There was little variation in the catchment-scale environmental variables between site types. In both site types, forest was the dominant upstream land use, followed by orchards and vineyards, natural grassland, and pasture (Table S1). Regarding local riparian habitat properties, forested buffer sites had a higher percentage cover and richness of trees (DBH > 5 cm), an abundance of dead wood, and shading (canopy cover; Figure S1). The percentage composition of habitat attributes was also different in forested buffer sites. The %cover of unmanaged grass, trees and shrubs, and plant litter was higher in forested buffer sites, while unbuffered sites recorded greater %cover of bare ground and managed grasses (Figure S2). Considering the riparian integrity conditions scores, we found the scores for adjacent ground and vegetation cover, buffer ground and vegetation cover, buffer width, and intactness were all higher in forested buffer sites. The shading attribute was also higher in forested buffer sites. In contrast, the scores for soil denitrification potential and livestock access were slightly higher in unbuffered sites (Figure S3).
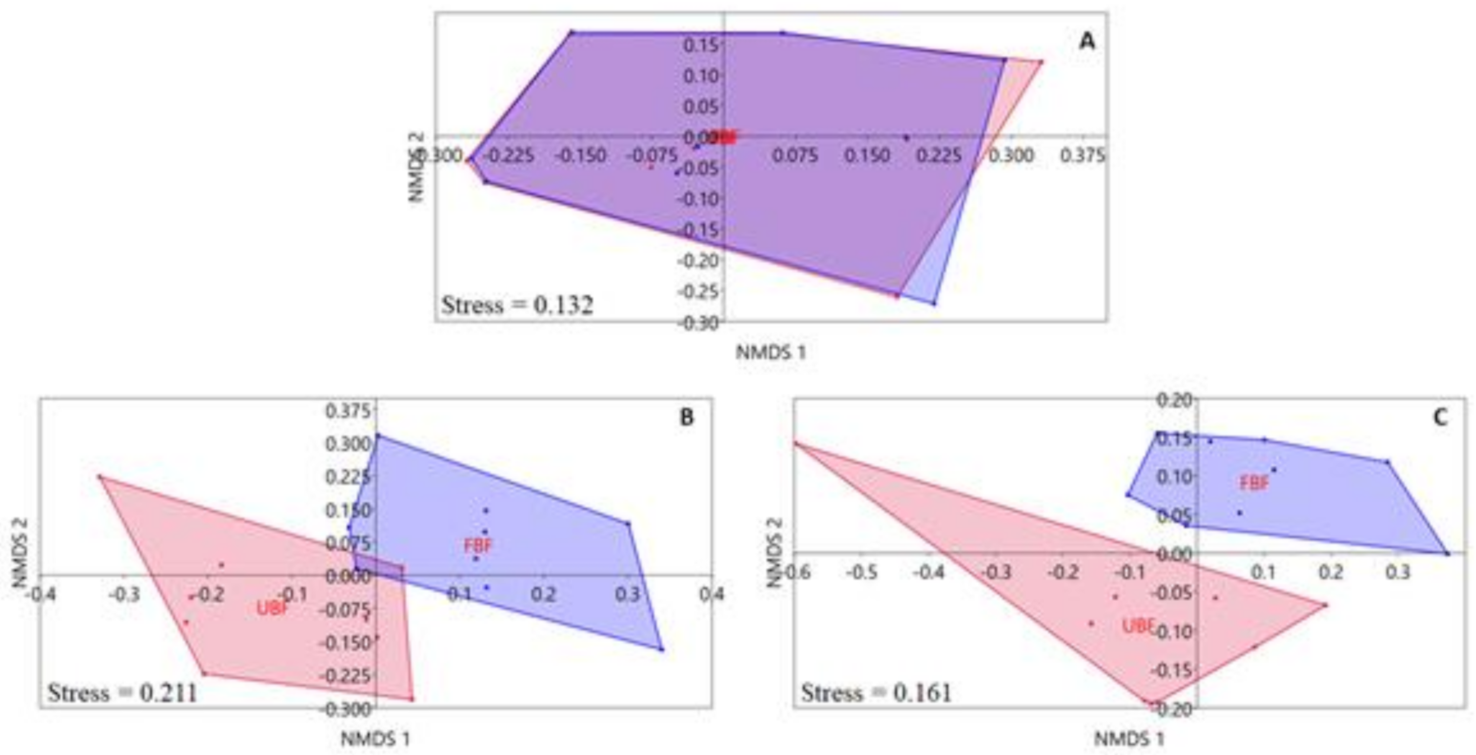
The unconstrained ordination analyses of selected environmental variables showed differences in our site types that were highly dependent on the spatial scale (Figure 2). The variables recorded at the catchment scale showed little dissimilarity among site types (Figure 2A), and the difference was statistically insignificant (PERMANOVA, F = 0.017, p = 0.991). In contrast, the variables recorded at the local spatial scale showed clear differences among the two site types (Figure 2B,C). The differences between the two groups of sites were statistically significant for both riparian habitat characteristics (PERMANOVA, F= 7.296, p = 0.0003) and riparian integrity condition scores (PERMANOVA, F = 5.349, p = 0.0005).
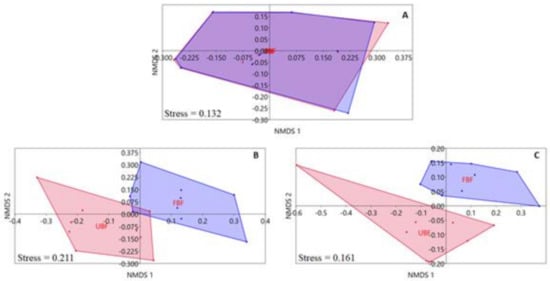
Figure 2.
The unconstrained (nMDS) ordination of sites based on values of environmental variables from the catchment (A) and the local scales: riparian habitat characteristics (B), riparian integrity condition scores (C). Unbuffered sites (UBF) = red, forested buffered sites (FBF) = blue.
3.2. Patterns in Invertebrate Community Structure
We sampled 1384 individuals (735 in unbuffered and 649 in forested buffer sites) belonging to 39 taxa in total. The 39 taxa were identified as 33 families and 6 orders (Figure S4). Adult Plecoptera were excluded from analyses due to their low number (two individuals) and restricted presence (in only one site). Seven taxa were present only in unbuffered sites: Gnaphosidae and Agelenidae (Araneae); Melyridae, Dermestidae, Oedemeridae, and Cleridae (Coleoptera), and specimens from the Neuroptera. Six taxa were only present in buffered sites: Opiliones (Arachnida); Blattidae (Blattodea); Anthicidae and Heteroceridae (Coleoptera); Panorpidae (Mecoptera); and Julidae (Diplopoda: Julida). The Lycosidae (Araneae) was the only family present in all samples from both site types. Chrysomelidae (Coleoptera) and Tettigoniidae (Orthoptera) were identified in all samples from unbuffered sites. In contrast, Thomisidae, Clubionidae, and Tetragnathidae (Araneae); Cicadidae (Hemiptera); and other hemipterans (hereafter referred to as Hemiptera) were identified in all samples from buffered sites. The most abundant taxa were Lycosidae (~22%), Chrysomelidae (11%), and Hemiptera (~11%) in unbuffered sites, and Culicidae (Diptera; 12%), Hemiptera (~10%), Thomisidae and Clubionidae (~8%) in buffered sites. Spiders (Araneae) were the most abundant taxa in the trophic group of secondary consumers (beetles and spiders) (Figure S5), with Lycosidae: Alopecosa sp. (19%) and Trochosa sp. (16%) dominating in unbuffered sites and Clubiona sp. (22%) (Clubionidae) and Tetragnatha sp. (17%) (Tetragnathidae), in buffered sites.
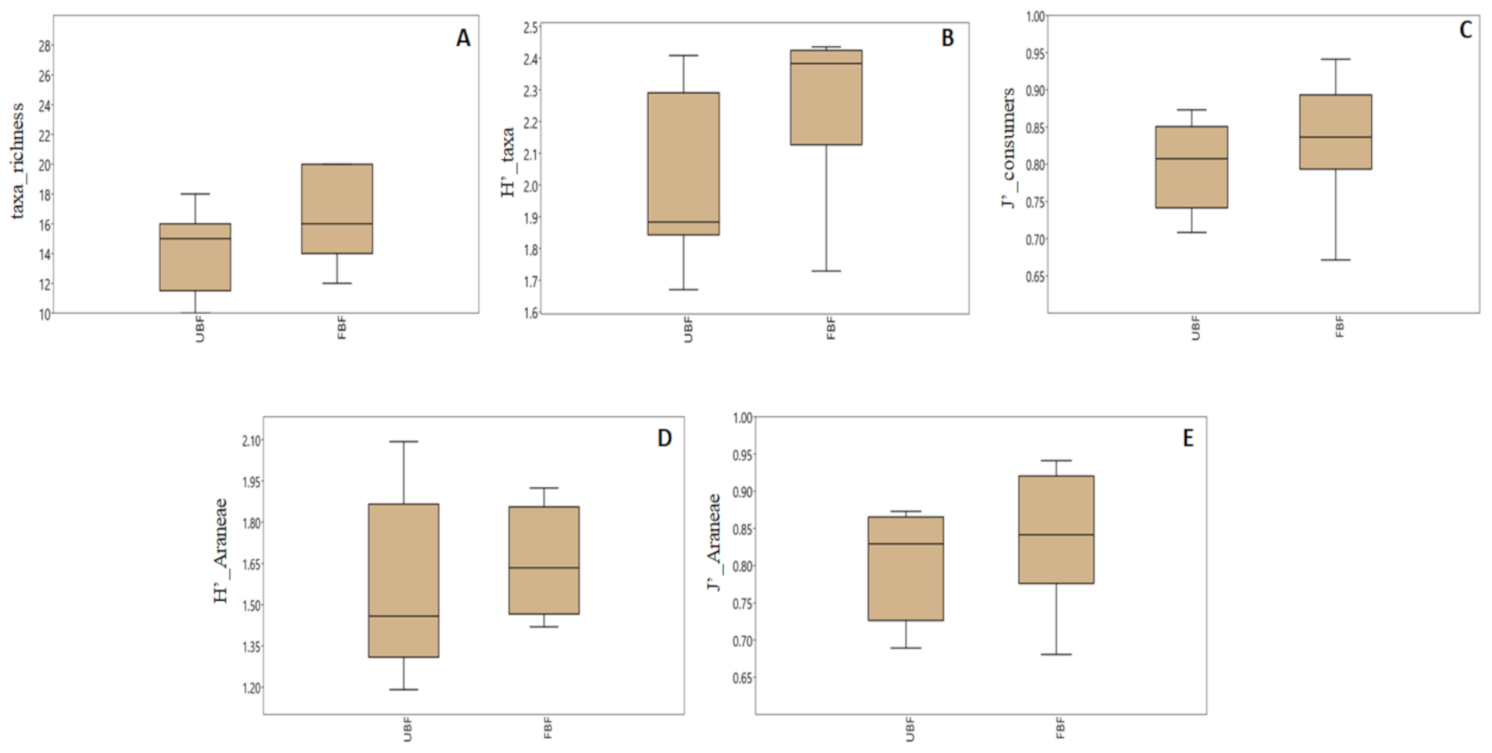
Our diversity metrics were generally higher in the buffered sites (Figure 3, Table S3). However, most of the differences between the two site types were statistically insignificant at α = 0.05, with the exception of invertebrate taxa richness and Araneae Pielou’s evenness (ANOVA, Table 2). The differences in invertebrate and Araneae Shannon diversity, as well as Pielou’s evenness of secondary consumers, were significant at α = 0.1 (Table 2).
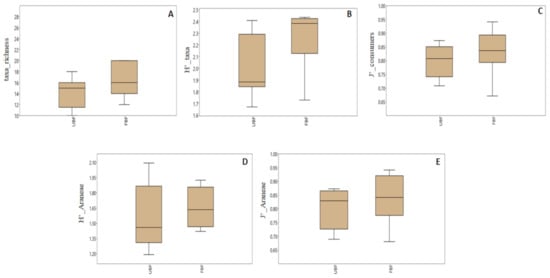
Figure 3.
The median value of diversity indices of riparian invertebrate community in unbuffered (UBF) and buffered sites (FBF). Only the indices showing significant (* p < 0.05) and marginally significant (p < 0.1) differences between the site types (see Table 2) are shown. The box = 25–75 percent quartiles, the whiskers = minimal and maximal values. A = taxa richness, B = taxa Shannon diversity, C = consumer Pielou’s evenness, D = Araneae Shannon diversity, E = Araneae Pielou’s evenness.

Table 2.
Differences in diversity metrics values between unbuffered and forested buffered sites (One-way ANOVA). H′ = Shannon diversity index; J’ = Pielou’s evenness. Only statistically significant indices are shown.
The highest genera richness (9) and Shannon diversity (H′ = 1.9) of ground beetles (Carabidae and Staphylinidae) were recorded in buffered sites. In contrast, the highest values of genera richness and Shannon diversity of spiders (Araneae) were found in the open unbuffered sites (11 and 2.09, respectively). However, no significant differences were identified between site types (ANOVA, p > 0.1).
3.3. Effects of Selected Environmental Variables on Riparian Invertebrate Community Structure
In total, our forward selection procedure (RDA) showed that nine environmental variables significantly influenced riparian invertebrate community composition in the Argeș basin (Table 3). Of these, only the PCNM1 and PCNM2 descriptors of spatial relationships, along with buffer intactness and pasture, were used for pRDA and variation partitioning analyses. Buffer intactness scores were significantly correlated with shading (p < 0.001), %trees with DBH > 5 cm (p < 0.001), and no. of dead wood elements (p = 0.088); %bare ground was correlated with PCNM (p = 0.023) and no. of dead wood elements (p = 0.096); and PCNM1 with buffer length (p < 0.001). Along the first two RDA axes, spatial location independently accounted for 23% of community variation, land-use at the catchment level explained 10%, and the riparian integrity condition score was able to explain 20% of the total variation. All effects were significant at α = 0.05.

Table 3.
Variables with a significant independent contribution in shaping the taxonomic structure of the riparian invertebrate community in the Argeș basin (RDA).
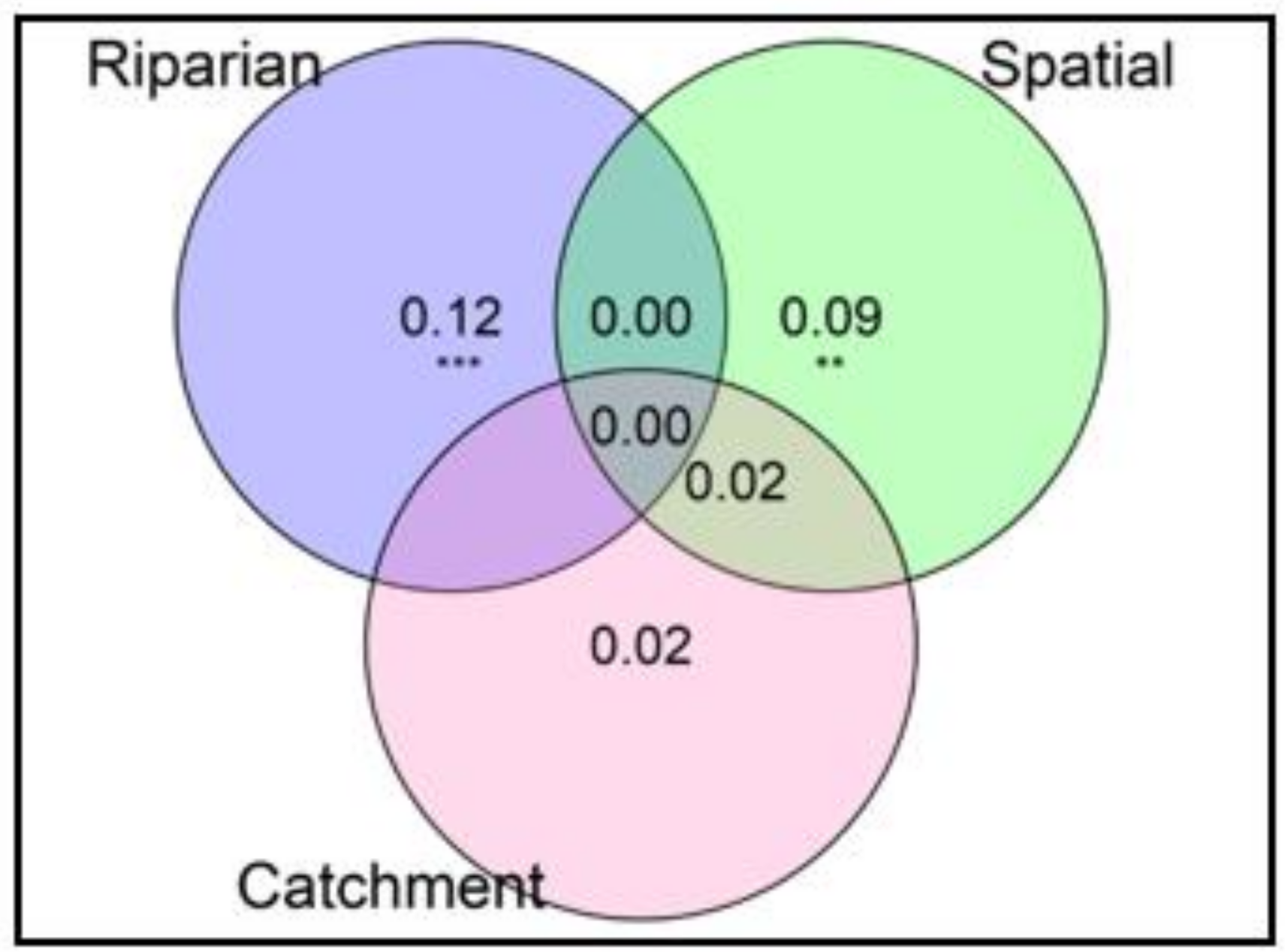
Variation partitioning revealed that spatial structuring (i.e., spatial relationships at the landscape scale) of terrestrial invertebrate communities was highly significant (F2,13 = 1.92, p = 0.003) and explained 9% of the observed variation in community composition. At the local scale, buffer intactness (Riparian Condition Index scores) showed a significant independent effect (F1,13 = 3.28, p = 0.001) and explained 12% of the total variation in community structure. At the catchment scale, upstream land-use (% of pasture) was not significant in shaping the structure of the invertebrate community (F1,13 = 1.41, p = 0.125) (Figure 4). Spatial location and catchment land-use combined explained only 2% of the total variation, indicating a weak autocorrelation of catchment-wide land use impacts. The unexplained (residual) variation was 75%.
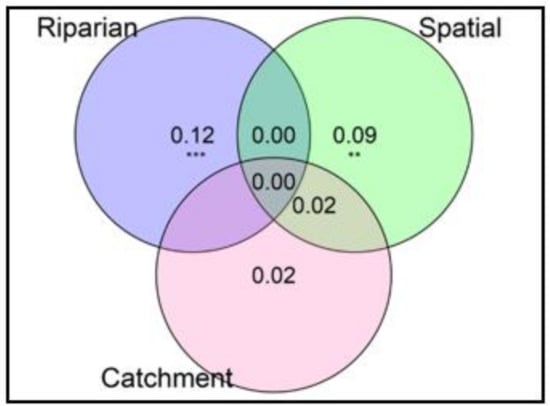
Figure 4.
Variation partitioning results showing the portions of variation in the taxonomic structure of riparian invertebrate community explained by groups of predictors at nine paired sites in the Argeș basin. Riparian = Buffer intactness scores, Spatial = PCNM1 and PCNM2 axes, Catchment variables = % of Pasture. The unexplained (residual) variation was 75%; Values < 0 are not shown. ** p < 0.01, *** p < 0.001.
3.4. The Effect of Forested Riparian Buffers on Riparian Invertebrate Community Structure
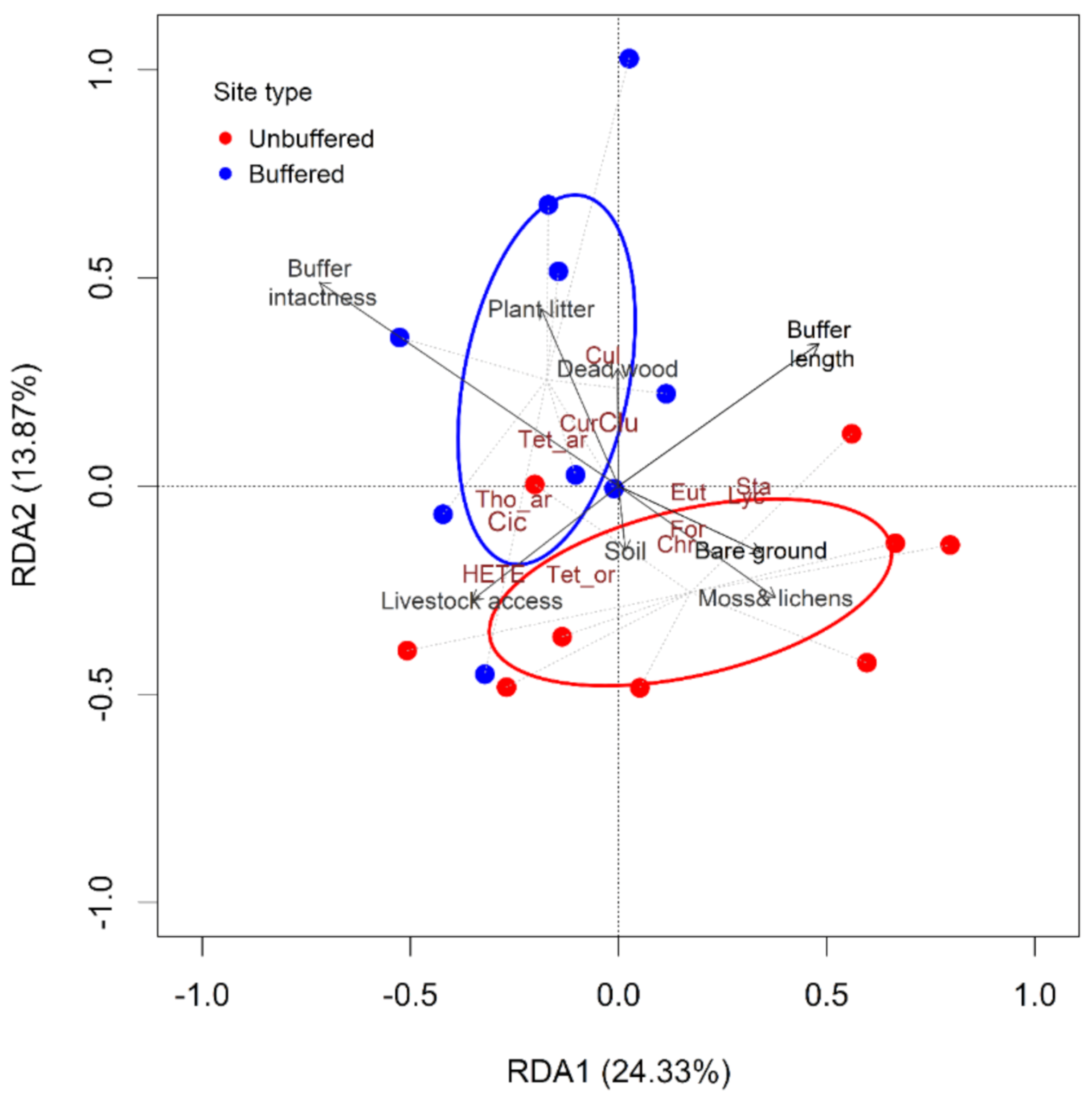
The partial redundancy analysis model (pRDA) conditioned out confounding influences of spatial location and characteristics at the catchment scale helped to reveal the association of terrestrial invertebrate structure with riparian properties (i.e., habitat and riparian condition attributes) (Figure 5). The first two pRDA axes explained 38% of the variation among sites. The taxa scores indicated clear differences in taxa abundances between unbuffered and buffered sites (Figure 5, Table S4). The taxa with the highest scores along the first two pRDA axes were Thomisidae, Tetragnathidae, Culicidae, Clubionidae, Curculionidae, Cicadidae, which are associated with forested buffered sites, and Hemiptera (other than Cicadidae), Tettigoniidae, Formicidae (Hymenoptera), Staphylinidae, Chrysomelidae, Lycosidae, and Eutichuridae (Araneae), which were associated with unbuffered sites (Figure 5).
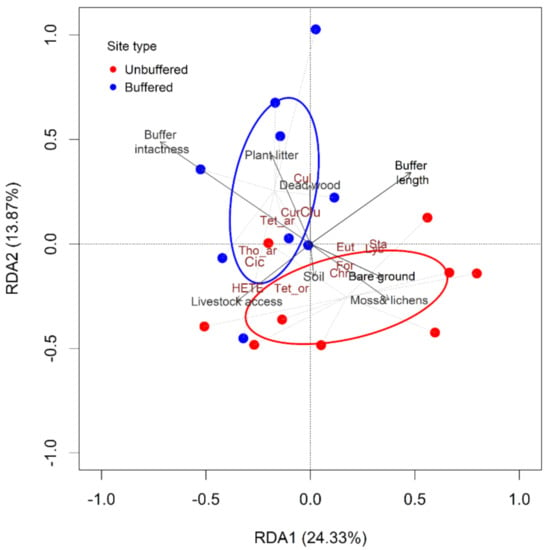
Figure 5.
Redundancy analysis (pRDA) of riparian invertebrate communities at nine paired sites in the Argeș basin, Romania, showing the influence of riparian attributes after conditioning out confounding Spatial location and Catchment level variables (i.e., land use). Riparian attributes used in the pRDA are shown in black (auto-correlated variables were not included in the analysis; Pearson’s correlation, p < 0.05). Buffer intactness, Buffer length, %Bare-ground, %Small trees and shrubs, %Moss and lichens, %Plant litter, %Herbs, Dead wood volume, and Soil drainage were included. The species with the highest scores along the first two pRDA axes are shown in dark brown: Araneae: Thomisidae = Tho_ar, Tetragnathidae = Tet_ar, Clubionidae = Clu, Culicidae = Cul, Coleoptera: Curculionidae = Cur, Hemiptera: Cicadidae = Cic are associated with buffered sites (blue ellipse and blue dots), whereas other Hemiptera = HETE, Orthoptera: Tettigoniidae = Tet_or, Hymenoptera: Formicidae = For, Coleoptera: Staphylinidae = Sta, Chrysomelidae = Chr, Araneae: Lycosidae = Lyc, Eutichuridae = Eut, are associated with unbuffered sites (red ellipse and red dots).
The biplot values for riparian attributes associated with forest buffered sites were positive along the RDA2 axis and either positive (buffer length 0.51) or negative (buffer intactness −0.72; % plant litter −0.18) along the RDA1. In contrast, the biplot values along the RDA2 axis were negative for %bare ground and %moss and lichens associated mostly with sites without forested riparian vegetation.
The pRDA results are consistent with results from our SIMPER (Similarity percentage) analysis, which revealed an overall average dissimilarity between buffered and unbuffered sites of 65%. Up to 80% of these differences were due to the abundance of 14 out of the 39 identified taxa. Seven of them have higher abundances in buffered sites: Thomisidae, Clubionidae, Tetragnathidae (Araneae); Curculionidae (Coleoptera); Culicidae (Diptera); Cicadidae (Hemiptera), and Gastropoda, while Lycosidae (Araneae), Chrysomelidae, Staphylinidae, Coccinellidae (Coleoptera), Tettigoniidae (Orthoptera), Formicidae (Hymenoptera), and Hemiptera (other than Cicadidae) were more abundant in unbuffered sites (Figure S4). One-way ANOVA proved statistically significant differences between the abundance of Tetragnathidae (F1,16= 12.47, p = 0.0075), Culicidae (F1,16 = 7.06, p = 0.0171), and Lycosidae (F1,16 = 6.36, p = 0.0222), and statistically marginally significant differences of Clubionidae (F1,16 = 4.22, p = 0.0565).
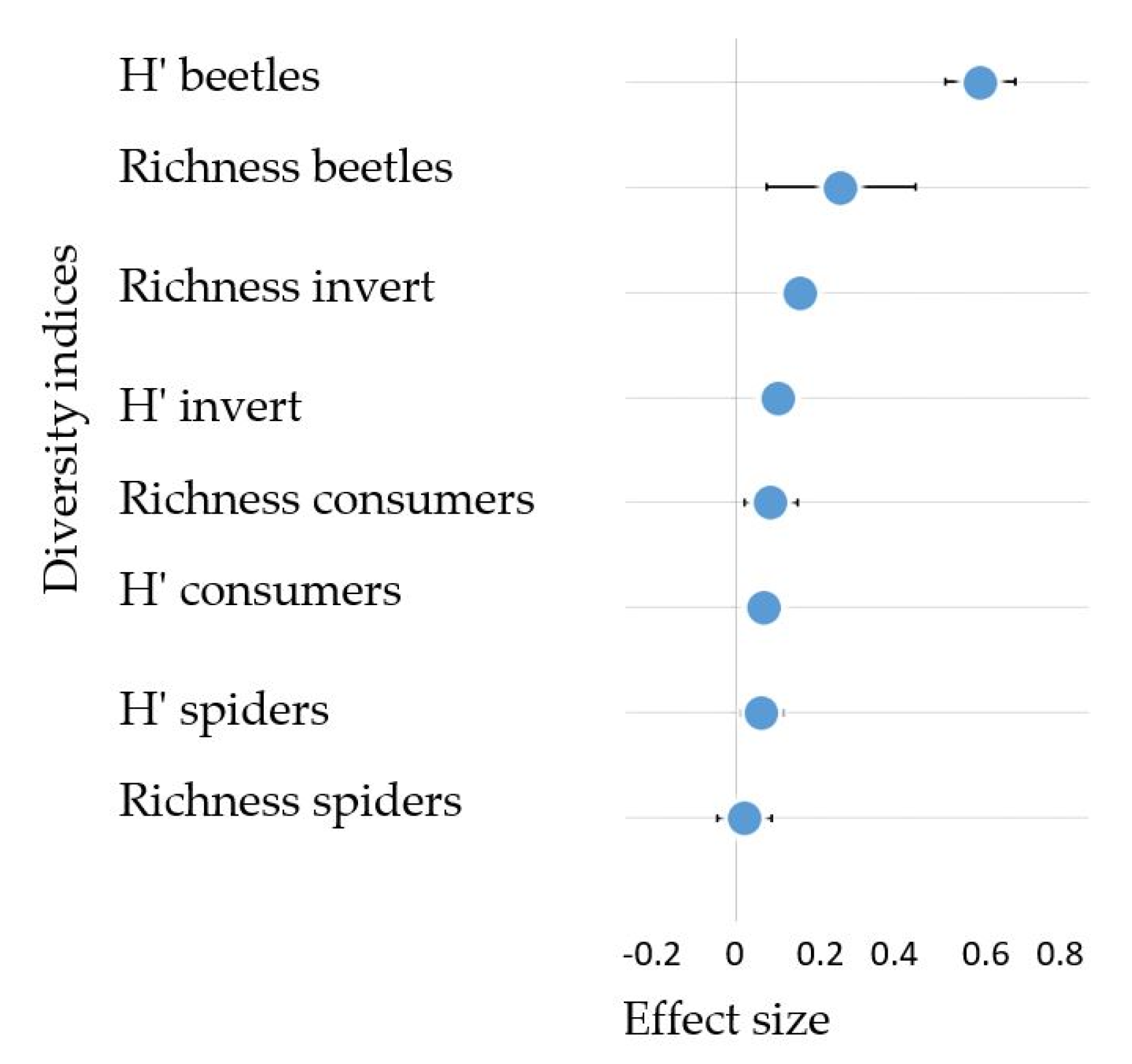
We also tested the mean effect size of the changes in riparian invertebrate community conferred by improved riparian conditions (i.e., forested buffers). None of the diversity indices showed negative effects with the presence of forest buffers. In particular, forest buffers had a strong positive effect on beetle diversity (Shannon Index) and a weak positive effect on beetle richness and richness and diversity of total invertebrate community (Figure 6).
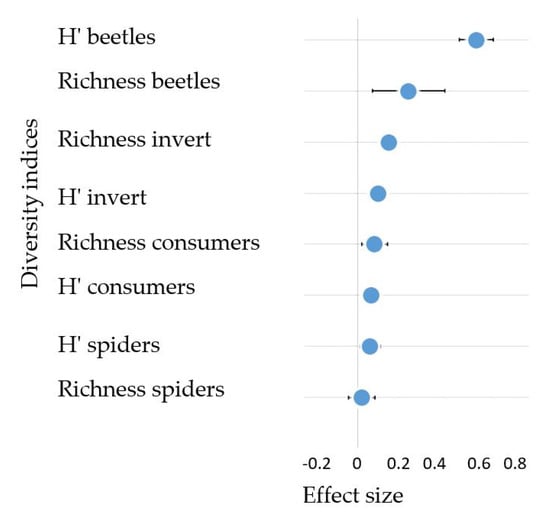
Figure 6.
Mean effect size (log response ratios ± 95% CI) showing the change between forested buffered sites situated downstream and upstream sites without forested riparian buffers for diversity indices that characterize the riparian invertebrate community (Richness = taxa richness, H′ = Shannon diversity index, Consumers = Araneae, Carabidae and Staphylinidae, beetles = Carabidae and Staphylinidae, spiders = Araneae, invert = all 39 identified invertebrate taxa).
4. Discussion
4.1. Patterns in Invertebrate Community Structure
Our results revealed that the forested riparian buffers supported a distinct invertebrate assemblage with a tendency for higher overall invertebrate diversity. These findings suggest that forest buffers play a key role in supporting biodiversity in agricultural landscapes. Out of 33 invertebrate families, 13 were unique either in forested (6) or unbuffered (7) sites. The ground-hunting spiders Lycosidae (Araneae), were the only family present at all sites. Overall, Tetragnathidae (Araneae) and Culicidae (Diptera) were prominent in buffered sites, whereas Lycosidae were more common in unbuffered sites. Regarding the Araneae, the percentage abundance of common riparian spiders was influenced by riparian vegetation structure. Tetragnathidae, Thomisidae, and Clubionidae were prominent in forested buffered sites, while the Lycosidae favored open, unbuffered sites. We found greater proportions of web-building spiders (e.g., Tetragnathidae) in the forest buffer sites. Our results are similar to those of Ramberg et al. [4], who found that the web-building family, Linyphiidae, was more abundant in buffered sites, while the Lycosidae was abundant in unbuffered sites [4]. Higher abundances of web-building spiders in forested riparian zones have been associated with vegetation structure (e.g., the presence of trees and dead wood structures) that aids the construction of webs [42,43]. Moreover, the Tetragnathidae are a web-building family that typically rely on aquatic prey from streams, meaning that they may respond numerically to changes in abundances of emergent stream insects [44,45]. Thus, lower abundances of web-building spiders in unbuffered streams could also reflect reduced abundances of emergent insects [10,42,45].
The Lycosidae are a family of free-living ground spiders that have greater mobility and feed on both aquatic and terrestrial prey [42]. Invertebrate species from the abundant families in our unbuffered sites have been cited as prey for lycosids [46]. These spiders may be less constrained by the vegetation structure, and Burdon and Harding [45] highlighted that open spaces (e.g., stony banks, gravel bars) may be beneficial to ground-dwelling spiders just as the close proximity of riparian vegetation may assist web-building spiders in headwater streams [45]. In contrast, the Thomisidae and Clubionidae are families of free-living ground spiders that contain ambush-hunting species [47]. The Clubionidae may use leaf litter and woody debris as cover [48] to capture prey, meaning they favor the ground habitat conditions provided by overhead forest cover. The Thomisidae are typically reliant on vegetation to ambush flying insects, so the presence of complex vegetation structure and a reduction in disturbance (e.g., grazing) may explain their greater abundance in the forest buffer sites.
We found some interesting patterns in invertebrate groups other than the Araneae. The preference of Culicidae for shaded areas [49] could help explain the prevalence of these dipterans in forest buffer sites, although water-filled tree-holes may also provide crucial habitat for the aquatic phase of their life cycles [50]. The association of hemipteran Cicadidae [51] and coleopteran Curculionidae [52] species with tree presence are well known and can reflect dead wood consumption [52], plant litter [53], or egg-laying substrate [52]. In contrast, we found that other hemipteran and coleopteran (e.g., Staphylinidae) taxa were associated with unbuffered sites. Unmanaged riparian zones with a mixture of grassland and bush habitat were reported to be the best reservoirs for beetles (Carabidae and Staphylinidae), where they reach high densities and abundances and dispersion to agrosystems [54,55,56]. Similar to our results, increased rove beetle diversity (Staphylinidae) has been reported in tropical and temperate regions that have undergone a land-use change from forest to pasture and cropping [57,58,59,60].
A major challenge in biomonitoring is resolving diversity at the species level since this requires a high level of expertise and may be affected by the presence of cryptic and undescribed species [61]. Although our study provides a useful overview of invertebrate diversity inhabiting riparian zones in a Romanian agricultural landscape, we likely missed substantial diversity at the species level. Most taxa were identified to the family level except Carabidae, Staphylinidae, and Araneae that were identified to the genus level. However, other studies have concluded that higher taxonomic identifications (e.g., family-level, genus-level) are sufficient to represent biodiversity patterns [62,63].
4.2. The Effects of Environmental Variables and the Role of Forested Riparian Buffers
The effects of anthropogenic pressures can be scale-dependent [64,65]. Patterns observed in local assemblages are not determined solely by local mechanisms but also result from processes operating at larger spatial scales [66]. We aimed to understand the independent and shared effects of influence factors at different spatial scales better and to identify those most significant in shaping the biodiversity patterns in riparian zones with contrasting vegetation structure. This knowledge would help optimize the ecological benefits and ecosystem services provided by woody riparian buffers by highlighting their contribution to local diversity in a Romanian agricultural landscape.
Our results demonstrated the strong influence of local-scale environmental factors (including both riparian habitat characteristics and scores from riparian condition attributes) in structuring riparian invertebrate communities. This contrasted with the relatively small contribution of land-use effects at the catchment scale. Riparian predictors independently explained 12% of the variation in terrestrial invertebrate community structure among sites, with a residual (unexplained) variation of 75%. Our partial redundancy analysis (pRDA) indicated that the buffer intactness attribute from the Riparian Condition Index (i.e., for rapid habitat assessment) together with other local riparian properties, including woody riparian buffer length, %plant litter, and dead wood volume (i.e., quantitative assessment attributes), were potentially strong indicators of the microhabitats used by taxa that were more abundant in forested buffered sites. The influence of local riparian conditions on terrestrial invertebrate assemblages supports results from other studies [4,10]. Forio et al. [10] found that local riparian conditions influenced spiders and carabids in contrast to stream macroinvertebrates, which were more influenced by properties of the riparian zone over broader spatial scales. Rodrigues et al. [67] showed a positive effect of canopy cover on invertebrate abundance and richness, as well as the influence of vegetation structure on species composition. Cole et al. [9] found that wider vegetated riparian buffers support greater densities of predatory hemipterans and coleopterans (e.g., Carabidae) that overwinter as adults. However, Stockan et al. [55] suggested that the distributions of carabid beetles in riparian zones within agricultural landscapes are shaped by the three factors at different spatial scales: the site type of the riparian zone, the local environmental conditions, and the catchment within which these stream–riparian networks are located. This suggests that processes acting at different spatial scales contribute to local biodiversity in riparian zones, with the presence of forested riparian buffers providing niche requirements for a wide range of invertebrate taxa.
Zermeño-Hernández et al. [68] found that the structure and diversity of riparian vegetation were strongly influenced by the riparian condition. In our study, the lower abundances of web-building spiders (e.g., Tetragnathidae) at unbuffered sites in the Argeș basin are likely the result of reduced habitat for web construction. The reduced vegetation structure (e.g., bare ground) in unbuffered sites could be the result of livestock overgrazing and disturbing habitat [69]. Other studies have made similar observations. Freiberg et al. [70] mentioned Tetragnathidae species “exclusively” in ungrazed areas, in contrast with Lycosidae species that were abundant in grazed areas.
The intensity of human and animal activity in our unbuffered sites may degrade riparian habitat with negative consequences for other ecosystem services provided by riparian zones. For instance, livestock presence may lead to soil compaction that favors anaerobic bacteria activity with increased denitrification rates and N2O emissions [71,72,73]. The lack of woody riparian vegetation (e.g., trees) and the increased soil nitrogen from livestock, crop fertilizers, and waste disposal may further increase denitrification rates [74,75]. These ecosystem “disservices” may also reflect changes to the terrestrial invertebrate communities in our study. For instance, species of staphylinid coleopteran Paederus favor open habitats with higher temperatures [76,77], and Philonthus spp. have been associated with the presence of animal dung [77], potentially helping to explain increased abundances of Staphylinidae at our unbuffered sites.
In our study, we found strong local effects of riparian vegetation structure on terrestrial invertebrate communities, while the influence of catchment land uses was weak. This may be a feature of the catchments used in our case study, and more generally, the strength of local riparian effects may be context-dependent. For instance, other studies have found that local effects of the riparian condition are weaker than catchment-scale environmental variables when anthropogenic impacts are high [6,78]. This observation may be more relevant to aquatic communities because those authors found evidence that riverine invertebrate assemblages are more degraded by upstream catchment-scale impacts, such as urbanisation [6,78] and land use and water chemistry degradation [6] than by local riparian impacts (e.g., deforestation and vegetation structure). Future research should consider what role human impacts at different spatial scales play in shaping invertebrate assemblages and ecosystem functioning since land use intensity and habitat fragmentation linked to biodiversity loss are particularly relevant for terrestrial invertebrate communities [79].
4.3. Management Implications
The maintenance of a habitat mosaic in agricultural landscapes is an important tool to support biodiversity conservation from local to catchment scales [4,80,81,82]. Due to their invertebrate faunas [83,84,85,86,87], riparian zones are extremely valuable for invertebrate conservation [88,89]. Our study and others have shown that woody riparian buffers are important habitat patches by providing food for terrestrial invertebrates (e.g., Cicadidae), sites for web construction (e.g., Tetragnathidae) and ambush-hunting (e.g., Thomisidae and Clubionidae), life-stage microhabitats and shelter (e.g., Culicidae), and sites for overwintering and laying eggs (e.g., Carabidae) [9,43,49,90,91]. Woody riparian buffers may provide essential habitat diversity (via plant diversity) that helps support diverse assemblages of riparian invertebrates (e.g., consumers) [81]. Some of these invertebrates are natural enemies of crop pests [9,92,93] or potential pollinators [94,95], so their presence and abundance could be beneficial for adjacent agricultural systems.
In our study, the diversity and richness of coleopterans in the riparian zone responded to changes in vegetation structure. Woody riparian buffers supported beetle assemblages with greater taxonomic richness and diversity. Other studies have shown that carabid assemblages are useful ecological indicators for riparian zones [86,96,97] and river management [98,99]. As predatory invertebrates feeding on prey items of both aquatic and terrestrial origin, carabid beetles are key indicators of aquatic–terrestrial linkages [4,100] that respond to vegetation structure [101,102], habitat heterogeneity, and distribution [103,104]. Being active early in the season, carabids may provide an important first line of defense against spring-active crop pests, such as aphids [105]. In open riparian zones, predatory rove beetles (Staphylinidae) exhibited a high degree of tolerance to changes in environmental conditions derived from human activities, enabling them to prevail in disturbed areas [106]. Studies suggest a high dispersal ability for certain species of carabids and staphylinids (e.g., Aleochara bilineata with distances of 1.4 to 5 km [107]) through human-modified landscapes [108,109]. Such dispersal abilities could enable these predators to persist in more disturbed unbuffered riparian zones and adjacent agricultural crops, thus meaning they may provide valuable ecosystem services of pest biocontrol.
Other positive interactions may be fostered by woody riparian buffers. A wide variety of dipterans contribute to plant pollination, and despite their disservices for human and animal health as potential disease vectors, there are studies that link species of mosquitos (Culicidae) and pollination. Although an earlier study by Foster [110] talked about the reduced role that Culicidae play in pollination, more recent studies have documented the role mosquitos and other dipterans contribute as pollinators [94,95]. Species of the Aedes genus (inclusive A. aegypti) and even the common mosquito (Culex pipiens) were mentioned as being pollinators for some species of plants [94,95]. Insect species, such as dipterans, that favor forested riparian buffers may provide valuable ecosystem services of plant pollination, adding to the co-benefits these multifunctional habitat features generate in agricultural catchments.
In contrast, higher abundances of herbivorous species from the orthopteran subfamily Phaneropterinae (Tettigoniidae) and the coleopteran family Chrysomelidae in our unbuffered sites could pose threats to agricultural crops as pests [111,112,113,114]. It is not clear if these taxa represent “spillover” from adjacent crops or are potential source populations that could move into adjacent agricultural systems, thus threatening crop production.
Managing these myriad interactions requires effective biomonitoring focused on measuring abundances of indicator fauna and habitat properties. Spider and ground beetle communities respond structurally to environmental change, indicating that they can provide valuable information for ecological impact assessment [101]. Both Tetragnathidae and Lycosidae are regarded as adequate biological indicators of habitat modification due to their potential to provide early warnings of environmental change [115,116,117]. These predatory invertebrates (e.g., tetragnathid and lycosid spiders, as well as carabid and staphylinid beetles) may be important sentinels of change because of their importance in controlling crop pest populations [92,118]. For example, Maisonneuve and Rioux [82] found that the proportion of pest species decreased with the complexity of riparian vegetation structure, while insectivorous species increased in abundance in woody riparian buffers. Accordingly, the presence and abundance of predatory invertebrates in our paired sites are highly relevant for the management of agricultural systems. The vegetation structure of the riparian zone can support different predator assemblages, thus suggesting the importance of both forested and grassy riparian vegetation in agricultural landscapes.
However, managing stream–riparian networks needs to embrace a multifunctional perspective that accounts for positive co-benefits as well as ecosystem disservices. For example, patches of riparian forest in catchments can help protect stream health (e.g., stabilizing streambanks and shading the channel, helping to regulate water quality and stream temperatures, providing plant litter and woody detritus inputs for shredding invertebrates) [6,10,68]. Forested riparian buffers can also provide refuge (microclimatic conditions, overwintering, or aestivation sites) and food resources for natural enemies of crop pests [119]. These habitat features may also serve as dispersal corridors between other non-crop habitats, as well as population sources of predatory invertebrates that disperse into crop fields [54,120]. These multiple co-benefits can help increase stream and terrestrial biodiversity [4,9,10], and future research should focus on the location and size of woody riparian buffers to optimize management goals that balance societal and conservation needs [6].
5. Conclusions
Our results showed that the composition of terrestrial invertebrate communities differed between sites with forested and unforested riparian zones. The forested riparian buffers supported higher invertebrate diversity and distinctive communities overall. Spider and beetle families that are natural enemies of crop pests were found at both riparian site types, with their taxonomic identity influenced by the vegetation structure of the riparian zone. The compositional differences in invertebrate communities (i.e., β-diversity) at buffered riparian sites could be important for maintaining biodiversity and ecological resilience at the catchment scale, thus indicating positive influences on ecosystem functioning. Local riparian properties were the most influential factors driving differences in community structure. This response underpins an important message for managers because while interventions at the catchment scale are often not logistically and economically feasible, local-scale changes in riparian management practice are within reach of most landowners and river authorities. Hence, the multiple co-benefits (shading, habitat diversity, plant litter and woody detritus inputs in terrestrial and stream systems, biological control of crops pest populations) that come from restoring and maintaining vegetated riparian buffers may safeguard biodiversity across spatial scales in both aquatic and terrestrial ecosystems.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4441/13/2/188/s1, Table S1. GIS data for catchments and riparian properties: up-stream catchment area (Area), Corine Land Cover (level 2, %), forested buffer length and width, and the distance to the next forested buffer upstream (m), Table S2: Sampling time of riparian invertebrates, weather and soil conditions in sampling sites, Table S3: Diversity metrics values (average) in unbuffered and buffered sites, Table S4: Riparian invertebrate taxa scores from the partial redundancy analysis model (pRDA) conditioning out confounding influences of spatial location and catchment land-uses. Invertebrate taxa are sorted from lowest to highest scores on RDA1, which explained 24.33% of the variation among sites, Figure S1: Average values of riparian habitat characteristic ± SE in unbuffered (UBF) and buffered (FBF) sites, Figure S2: Habitat type percentages in UBF and FBF sites, Figure S3: Average scores of riparian integrity conditions ± SE in unbuffered (UBF) and buffered (FBF) sites, Figure S4. The relative abundance (%) of riparian invertebrate taxa in the unbuffered (UBF) and buffered (FBF) sites, Figure S5. The relative abundance (%) of taxa in consumer trophic group (beetles and spiders) ± SE in the unbuffered (UBF) and buffered (FBF) sites.
Author Contributions
Conceptualization, C.P. and G.R.; methodology, C.P., M.O.-P., V.D., C.C., F.J.B., M.A.E.F., B.K., N.F., P.G., B.G.M. and G.R.; validation, G.R., C.P., B.G.M. and F.J.B.; formal analysis, C.P. and G.R.; investigation, C.P., M.O.-P., V.D. and G.R.; resources C.P. and G.R.; data curation, C.P., G.R., M.O.-P., V.D. and C.C.; writing—original draft preparation, C.P. and G.R.; writing—review and editing, C.P., M.O.-P., V.D., C.C., F.J.B., M.A.E.F., B.K., N.F., P.G., B.G.M. and G.R.; visualization, C.P. and G.R.; supervision, G.R., B.G.M. and F.J.B.; project administration, G.R., B.G.M., N.F. and P.G.; funding acquisition, G.R., B.G.M., N.F. and P.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was conducted as part of the CROSSLINK project funded through the 2015–2016 BiodivERsA COFUND call for research proposals. National funders: the Swedish Research Council for Sustainable Development (FORMAS, projects 2016-01945) and the Swedish Environmental Protection Agency; The Research Council of Norway (NFR, project 264499); The Research Foundation of Flanders (FWO, project G0H6516N), Belgium; and the Romanian National Authority for Scientific Research and Innovation (CCCDI–UEFISCDI, project BiodivERsA3-2015-49-CROSSLINK, within PNCDI III.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in Supplementary Materials.
Acknowledgments
We thank Georgeta Niculae, Aglaia Parvu, Marius Bujor, and Liliana Gheorghe for the help provided with the fieldwork and initial database development. Students who volunteered for riparian assessment are kindly acknowledged: Cezara Tudose, Mădălina Ivan, Raluca Zoican, Cristiana Bobeș, Miruna Mirică, Cristian Murgu.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Rykken, J.J.; Moldenke, A.R.; Olson, D.H. Headwater riparian forest-floor invertebrate communities associated with alternative forest management practices. Ecol. Appl. 2007, 17, 1168–1183. [Google Scholar] [CrossRef]
- Ramey, T.L.; Richardson, J.S. Terrestrial Invertebrates in the Riparian Zone: Mechanisms Underlying Their Unique Diversity. Bioscience 2017, 67, 808–819. [Google Scholar] [CrossRef]
- Raitif, J.; Plantegenest, M.; Roussel, J.M. From stream to land: Ecosystem services provided by stream insects to agriculture. Agric. Ecosyst. Environ. 2019, 270–271, 32–40. [Google Scholar] [CrossRef]
- Ramberg, E.; Burdon, F.J.; Sargac, J.; Kupilas, B.; Rîsnoveanu, G.; Lau, D.C.P.; Johnson, R.K.; McKie, B.G. The Structure of Riparian Vegetation in Agricultural Aquatic-Terrestrial Linkages. Water 2020, 12, 2855. [Google Scholar] [CrossRef]
- Burdon, F.J.; McIntosh, A.R.; Harding, J.S. Habitat loss drives threshold response of benthic invertebrate communities to deposited sediment in agricultural streams. Ecol. Appl. 2013, 23, 1036–1047. [Google Scholar] [CrossRef]
- Burdon, F.J.; Ramberg, E.; Sargac, J.; Forio, M.A.E.; de Saeyer, N.; Mutinova, P.T.; Moe, T.F.; Pavelescu, M.O.; Dinu, V.; Cazacu, C.; et al. Assessing the benefits of forested riparian zones: A qualitative index of riparian integrity is positively associated with ecological status in European streams. Water 2020, 12, 1178. [Google Scholar] [CrossRef]
- Hefting, M.M.; Clement, J.C.; Bienkowski, P.; Dowrick, D.; Guenat, C.; Butturini, A.; Topa, S.; Pinay, G.; Verhoeven, J.T.A. The role of vegetation and litter in the nitrogen dynamics of riparian buffer zones in Europe. Ecol. Eng. 2005, 24, 465–482. [Google Scholar] [CrossRef]
- Buckley, C.; Hynes, S.; Mechan, S. Supply of an ecosystem service-Farmers’ willingness to adopt riparian buffer zones in agricultural catchments. Environ. Sci. Policy 2012, 24, 101–109. [Google Scholar] [CrossRef]
- Cole, L.J.; Stockan, J.; Helliwell, R. Managing riparian buffer strips to optimise ecosystem services: A review. Agric. Ecosyst. Environ. 2020, 296, 106891. [Google Scholar] [CrossRef]
- Forio, M.A.E.; De Troyer, N.; Lock, K.; Witing, F.; Baert, L.; De Saeyer, N.; Rîșnoveanu, G.; Popescu, C.; Burdon, F.J.; Kupilas, B.; et al. Small Patches of Riparian Woody Vegetation Enhance Biodiversity of Invertebrates. Water 2020, 12, 3070. [Google Scholar] [CrossRef]
- Iñiguez-Armijos, C.; Hampel, H.; Breuer, L. Land-use effects on structural and functional composition of benthic and leaf-associated macroinvertebrates in four Andean streams. Aquat. Ecol. 2018, 52, 77–92. [Google Scholar] [CrossRef]
- Fierro, P.; Bertrán, C.; Tapia, J.; Hauenstein, E.; Peña-Cortés, F.; Vergara, C.; Cerna, C.; Vargas-Chacoff, L. Effects of local land-use on riparian vegetation, water quality, and the functional organization of macroinvertebrate assemblages. Sci. Total Environ. 2017, 609, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Silva-Araújo, M.; Silva-Junior, E.F.; Neres-Lima, V.; Feijó-Lima, R.; Tromboni, F.; Lourenço-Amorim, C.; Thomas, S.A.; Moulton, T.P.; Zandonà, E. Effects of riparian deforestation on benthic invertebrate community and leaf processing in Atlantic forest streams. Perspect. Ecol. Conserv. 2020, 18, 277–282. [Google Scholar] [CrossRef]
- Ssymank, A.; Kearns, C.A.; Pape, T.; Thompson, F.C.; Ssymank, A.; Kearns, C.A.; Pape, T.; Thompson, F.C. Plant diversity and agricultural production Pollinating Flies (Diptera): A major contribution to plant diversity and agricultural production. Biodiversity 2011, 86–89. [Google Scholar]
- Qodri, A.; Raffiudin, R.; Noerdjito, W.A. Diversity and Abundance of Carabidae and Staphylinidae (Insecta: Coleoptera) in Four Montane Habitat Types on Mt. Bawakaraeng, South Sulawesi. HAYATI J. Biosci. 2016, 23, 22–28. [Google Scholar] [CrossRef][Green Version]
- Bartholomée, O.; Aullo, A.; Becquet, J.; Vannier, C.; Lavorel, S. Pollinator presence in orchards depends on landscape-scale habitats more than in-field flower resources. Agric. Ecosyst. Environ. 2020, 293, 106806. [Google Scholar] [CrossRef]
- Stutter, M.I.; Chardon, W.J.; Kronvang, B. Riparian Buffer Strips as a Multifunctional Management Tool in Agricultural Landscapes: Introduction. J. Environ. Qual. 2012, 41, 297–303. [Google Scholar] [CrossRef]
- Burdon, F. Agriculture and Mining Contamination Contribute to a Productivity Gradient Driving Cross-Ecosystem Associations between Stream Insects and Riparian Arachnids. In Contaminants and Ecological Subsidies; Kraus, J.M., Walters, D.M., Mills, M.A., Eds.; Springer: Cham, Switzerland, 2020; pp. 61–90. [Google Scholar] [CrossRef]
- Williamson, J.; Slade, E.M.; Luke, S.H.; Swinfield, T.; Chung, A.Y.C.; Coomes, D.A.; Heroin, H.; Jucker, T.; Lewis, O.T.; Vairappan, C.S.; et al. Riparian buffers act as microclimatic refugia in oil palm landscapes. J. Appl. Ecol. 2020, 1–12. [Google Scholar] [CrossRef]
- Nsor, C.A.; Oppong, S.K.; Danquah, E.; Ochem, M.; Antobre, O.O. Spatiotemporal dynamics of terrestrial invertebrate assemblages in the riparian zone of the Wewe river, Ashanti region, Ghana. Open Life Sci. 2020, 15, 331–345. [Google Scholar] [CrossRef]
- Domnişoru, A. Long Term Effects of Climate Change on Europe’s Water Resources Case Study: Arges, Romania; Kiwa N.V.: Nieuwegein, The Netherlands, 2006; pp. 40–72. [Google Scholar]
- Strahler, A.N. Quantitative analysis of watershed geomorphology. EOS Trans. Am. Geophys. Union 1957, 38, 913–920. [Google Scholar] [CrossRef]
- Harding, J.; Clapcott, J.; Quinn, J.; Hayes, J.; Joy, M.; Storey, R.; Greig, H.; Hay, J.; James, T.; Beech, M.; et al. Stream Habitat Assessment Protocols for wadable rivers and streams of New Zealand; School of Biological Sciences, University of Canterbury: Christchurch, New Zealand, 2009. [Google Scholar]
- Anderson, J.T.; Davis, C.A. Wetland Techniques Volume 2: Organisms; Wetland Techniques; Springer: Drodrecht, The Netherlands, 2013; Volume 2, pp. 1–332. [Google Scholar]
- Makarov, K. A key to the genera of the Ground-beetle larvae (Coleoptera, Carabidae) of the Paleartic region. Boll. Mus. Reg. Sci. Nat. Torino 1994, 12, 221–254. [Google Scholar]
- Klimaszewski, J.; Watt, J.C. Coleoptera: Family-group review and keys to identification. Fauna N. Zeal. 1997, 37, 1–199. [Google Scholar]
- Key to Adult Insects. Available online: https://www.amentsoc.org/insects/what-bug-is-this/adult-key.html (accessed on 30 June 2020).
- Insect Identification Key. Available online: http://www.knowyourinsects.org/ (accessed on 30 June 2020).
- Insect Identification Key. Available online: https://www.insectidentification.org/insect-key.asp (accessed on 30 June 2020).
- Canedoli, C.; Ferrè, C.; Abu El Khair, D.; Comolli, R.; Liga, C.; Mazzucchelli, F.; Proietto, A.; Rota, N.; Colombo, G.; Bassano, B.; et al. Evaluation of ecosystem services in a protected mountain area: Soil organic carbon stock and biodiversity in alpine forests and grasslands. Ecosyst. Serv. 2020, 44, 101135. [Google Scholar] [CrossRef]
- Michaels, K.F. Using staphylinid and tenebrionid beetles as indicators of sustainable landscape management in Australia: A review. Aust. J. Exp. Agric. 2007, 47, 435–449. [Google Scholar] [CrossRef]
- Gerlach, J.; Samways, M.; Pryke, J. Terrestrial invertebrates as bioindicators: An overview of available taxonomic groups. J. Insect Conserv. 2013, 17, 831–850. [Google Scholar] [CrossRef]
- Johan Kotze, D.; Brandmayr, P.; Casale, A.; Dauffy-Richard, E.; Dekoninck, W.; Koivula, M.J.; Lövei, G.L.; Mossakowski, D.; Noordijk, J.; Paarmann, W.; et al. Forty years of carabid beetle research in Europe—From taxonomy, biology, ecology and population studies to bioindication, habitat assessment and conservation. Zookeys 2011, 100, 55–148. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Nielsen, A.B.; Hedblom, M. Reviewing the strength of evidence of biodiversity indicators for forest ecosystems in Europe. Ecol. Indic. 2015, 57, 420–434. [Google Scholar] [CrossRef]
- Soliveres, S.; Van Der Plas, F.; Manning, P.; Prati, D.; Gossner, M.M.; Renner, S.C.; Alt, F.; Arndt, H.; Baumgartner, V.; Binkenstein, J.; et al. Biodiversity at multiple trophic levels is needed for ecosystem multifunctionality. Nature 2016, 536, 456–459. [Google Scholar] [CrossRef]
- Hammer, O.; Harper, D.; Ryan, P. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Legendre, P.; Gallagher, E.D. Ecologically meaningful transformations for ordination of species data. Oecologia 2001, 129, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Blanchet, F.G.; Legendre, P.; Borcard, D. Forward selection of explanatory variables. Ecology 2008, 89, 2623–2632. [Google Scholar] [CrossRef] [PubMed]
- Borcard, D.; Legendre, P. All-scale spatial analysis of ecological data by means of principal coordinates of neighbour matrices. Ecol. Model. 2002, 153, 51–68. [Google Scholar] [CrossRef]
- Borcard, D.; Legendre, P. Environmental control and spatial structure in ecological communities: An example using oribatid mites (Acari, Oribatei). Environ. Ecol. Stat. 1994, 1, 37–61. [Google Scholar] [CrossRef]
- Krell, B.; Röder, N.; Link, M.; Gergs, R.; Entling, M.H.; Schäfer, R.B. Aquatic prey subsidies to riparian spiders in a stream with different land use types. Limnologica 2015, 51, 1–7. [Google Scholar] [CrossRef]
- Tourinho, A.L.; Benchimol, M.; Porto, W.; Peres, C.A.; Storck-Tonon, D. Marked compositional changes in harvestmen assemblages in Amazonian forest islands induced by a mega dam. Insect Conserv. Divers. 2020, 13, 432–444. [Google Scholar] [CrossRef]
- Marczak, L.B.; Richardson, J.S. Spiders and subsidies: Results from the riparian zone of a coastal temperate rainforest. J. Anim. Ecol. 2007, 76, 687–694. [Google Scholar] [CrossRef]
- Burdon, F.J.; Harding, J.S. The linkage between riparian predators and aquatic insects across a stream-resource spectrum. Freshw. Biol. 2008, 53, 330–346. [Google Scholar] [CrossRef]
- Leslie Hayes, J.; Lockley, T.C. Prey and nocturnal activity of Wolf spiders (Araneae: Lycosidae) in cotton fields in the Delta Region of Mississippi. Environ. Entomol. 1990, 19, 1512–1518. [Google Scholar] [CrossRef][Green Version]
- Hunt, J.L.; Paterson, H.; Close, P.; Pettit, N.E. Riparian condition influences spider community structure and the contribution of aquatic carbon subsidies to terrestrial habitats. Sci. Total Environ. 2020, 746, 141109. [Google Scholar] [CrossRef]
- Jennings, D.T. Crab spiders (Araneae: Thomisidae) preying on scarab beetles (Coleoptera: Scarabaeidae). Coleopterists’ Bull. 1974, 41–43. [Google Scholar]
- Török, E.; Kolcsár, L.P.; Popescu, O.; Keresztes, L. Faunistic survey on Culicidae (Diptera) and their arboviruses in the area of a metropolis Cluj-Napoca, Romania. North West. J. Zool. 2019, 15, 24–29. [Google Scholar]
- Bradshaw, M.E.; Holzapfel, C.M. Resource limitation, habitat segregation, and species interactions of british tree-hole mosquitoes in nature. Oecologia 1992, 90, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Zamanian, H.; Mehdipour, M.; Ghaemi, N. The study and analysis of the mating behavior and sound production of male cicada Psalmocharias alhageos (Kol.) (Homoptera:Cicadidae) to make disruption in mating. Pakistan J. Biol. Sci. PJBS 2008, 11, 2062–2072. [Google Scholar] [CrossRef]
- Anderson, R.S. CURCULIONIDAE Latreille, 1802. In American Beetles; Arnett, R.H., Jr., Thomas, M.C., Skelley, P.E., Frank, J.H., Eds.; CRC Press: Boca Raton, FL, USA, 2002; pp. 722–815. [Google Scholar]
- Olivier, R.; Aranda, R.; Godoi, M.; Vilalva, A. Habitat structure in the composition of leaf-litter insects in mosaic environment. Acta Biológica Parana. 2015, 44, 71–87. [Google Scholar] [CrossRef]
- Riis, T.; Kelly-Quinn, M.; Aguiar, F.C.; Manolaki, P.; Bruno, D.; Bejarano, M.D.; Clerici, N.; Fernandes, M.R.; Franco, J.C.; Pettit, N.; et al. Global overview of ecosystem services provided by riparian vegetation. Bioscience 2020, 70, 501–514. [Google Scholar] [CrossRef]
- Stockan, J.A.; Baird, J.; Langan, S.J.; Young, M.R.; Iason, G.R. Effects of riparian buffer strips on ground beetles (Coleoptera, Carabidae) within an agricultural landscape. Insect Conserv. Divers. 2014, 7, 172–184. [Google Scholar] [CrossRef]
- Bennewicz, J.; Barczak, T. Ground beetles (Carabidae) of field margin habitats. Biologia 2020, 75, 1631–1641. [Google Scholar] [CrossRef]
- Caballero, U.; León-Cortés, J.L.; Morón-Ríos, A. Response of rove beetles (Staphylinidae) to various habitat types and change in Southern Mexico. J. Insect Conserv. 2009, 13, 67–75. [Google Scholar] [CrossRef]
- Vásquez-Vélez, L.M.; Bermúdez, C.; Chacón, P.; Lozano-Zambrano, F.H. Analysis of the richness of Staphylinidae (Coleoptera) on different scales of a sub-Andean rural landscape in Colombia. Biodivers. Conserv. 2010, 19, 1917–1931. [Google Scholar] [CrossRef]
- Hoffmann, H.; Michalik, P.; Görn, S.; Fischer, K. Effects of fen management and habitat parameters on staphylinid beetle (Coleoptera: Staphylinidae) assemblages in north-eastern Germany. J. Insect Conserv. 2016, 20, 129–139. [Google Scholar] [CrossRef]
- Klimaszewski, J.; Brunke, A.J.; Work, T.T.; Venier, L. Rove Beetles (Coleoptera, Staphylinidae) as Bioindicators of Change in Boreal Forests and Their Biological Control Services in Agroecosystems: Canadian Case Studies BT—Biology of Rove Beetles (Staphylinidae): Life History, Evolution, Ecology and Distri; Betz, O., Irmler, U., Klimaszewski, J., Eds.; Springer: Cham, Switzerland, 2018; pp. 161–181. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Penton, E.H.; Burns, J.M.; Janzen, D.H.; Hallwachs, W. Ten species in one: DNA barcoding reveals cryptic species in the neotropical skipper butterfly Astraptes fulgerator. Proc. Natl. Acad. Sci. USA 2004, 101, 14812–14817. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, S.S.; Ortega, J.C.G.; dos Santos Ribas, L.G.; Lopes, V.G.; Bini, L.M. Higher taxa are sufficient to represent biodiversity patterns. Ecol. Indic. 2020, 111, 105994. [Google Scholar] [CrossRef]
- Timms, L.L.; Bowden, J.J.; Summerville, K.S.; Buddle, C.M. Does species-level resolution matter? Taxonomic sufficiency in terrestrial arthropod biodiversity studies. Insect Conserv. Divers. 2013, 6, 453–462. [Google Scholar] [CrossRef]
- Allan, J.D.; Erickson, D.L.; Fay, J. The influence of catchment land use on stream integrity across multiple spatial scales. Freshw. Biol. 1997, 37, 149–161. [Google Scholar] [CrossRef]
- Feld, C.K.; Fernandes, M.R.; Ferreira, M.T.; Hering, D.; Ormerod, S.J.; Venohr, M.; Gutiérrez-Cánovas, C. Evaluating riparian solutions to multiple stressor problems in river ecosystems—A conceptual study. Water Res. 2018, 139, 381–394. [Google Scholar] [CrossRef]
- Brosse, S.; Arbuckle, C.J.; Townsend, C.R. Habitat scale and biodiversity: Influence of catchment, stream reach and bedform scales on local invertebrate diversity. Biodivers. Conserv. 2003, 12, 2057–2075. [Google Scholar] [CrossRef]
- Rodrigues, E.N.L.; Mendonça, M.D.S.; Costa-Schmidt, L.E. Spider diversity responds strongly to edge effects but weakly to vegetation structure in riparian forests of Southern Brazil. Arthropod Plant Interact. 2014, 8, 123–133. [Google Scholar] [CrossRef]
- Zermeño-Hernández, I.; Benítez-Malvido, J.; Suazo-Ortuño, I.; Méndez-Toribio, M. Impact of adjacent land use on the ecological condition of riparian habitats: The relation between condition and vegetation properties. Appl. Veg. Sci. 2020, 23, 610–621. [Google Scholar] [CrossRef]
- Sigsgaard, L. Early season natural biological control of insect pests in rice by spiders—And some factors in the management of the cropping system that may affect this control. Eur. Arachnol. 2000, 2000, 57–64. [Google Scholar]
- Freiberg, J.A.; de Sales Dambros, C.; Rodrigues, E.N.L.; Teixeira, R.A.; Vieira, Â.D.H.N.; de Almeida, H.S.; de Faccio Carvalho, P.C.; Jacques, R.J.S. Increased grazing intensity in pastures reduces the abundance and richness of ground spiders in an integrated crop-livestock system. Agron. Sustain. Dev. 2020, 40. [Google Scholar] [CrossRef]
- Bakken, L.R.; Børresen, T.; Njøs, A. Effect of soil compaction by tractor traffic on soil structure, denitrification, and yield of wheat (Triticum aestivum L.). J. Soil Sci. 1987, 38, 541–552. [Google Scholar] [CrossRef]
- Bhandral, R.; Saggar, S.; Bolan, N.; Hedley, M.J. Nitrous oxide fluxes in soil as influenced by compaction. Proc. N. Zeal. Grassl. Assoc. 2003, 265–271. [Google Scholar] [CrossRef]
- Thomas, S.M.; Fraser, P.M.; Hu, W.; Clough, T.J.; Van Der Klei, G.; Wilson, S.; Tregurtha, R.; Baird, D. Tillage, compaction and wetting effects on NO3, N2O and N2 losses. Soil Res. 2019, 57, 670–688. [Google Scholar] [CrossRef]
- Sgouridis, F.; Ullah, S. Denitrification potential of organic, forest and grassland soils in the Ribble-Wyre and Conwy River catchments, UK. Environ. Sci. Process. Impacts 2014, 16. [Google Scholar] [CrossRef]
- Johnson, A.D. Denitrification and Organic Carbon in a Series of Riparian Buffers in the Bear Creek National Demonstration Watershed; Iowa State University: Ames, IA, USA, 2003; p. 78. [Google Scholar]
- Rana, H.; Khan, M.F.; Fahim, M.; Tariq, S.A. Abundance and Comparative Population Fluctuation of Rove Beetles (Paederus Littoralis and Paederus Fuscipes) in Sindh, Pakistan. Int. J. Biol. Biotechnol. 2013, 10, 271–274. [Google Scholar]
- Irmler, U.; Lipkow, E. Effect of environmental conditions on distribution patterns of rove beetles. In Biology of Rove Beetles (Staphylinidae); Betz, O., Irmler, U., Klimaszewski, J., Eds.; Springer: Cham, Switzerland, 2018; pp. 117–144. ISBN 9783319702551. [Google Scholar]
- Walsh, C.J.; Waller, K.A.; Gehling, J.; Mac Nally, R. Riverine invertebrate assemblages are degraded more by catchment urbanisation than by riparian deforestation. Freshw. Biol. 2007, 52, 574–587. [Google Scholar] [CrossRef]
- Didham, R.K.; Barker, G.M.; Bartlam, S.; Deakin, E.L.; Denmead, L.H.; Fisk, L.M.; Peters, J.M.R.; Tylianakis, J.M.; Wright, H.R.; Schipper, L.A. Agricultural intensification exacerbates spillover effects on soil biogeochemistry in adjacent forest remnants. PLoS ONE 2015, 10, 1–32. [Google Scholar] [CrossRef]
- Batáry, P.; Báldi, A.; Samu, F.; Szuts, T.; Erdos, S. Are spiders reacting to local or landscape scale effects in Hungarian pastures? Biol. Conserv. 2008, 141, 2062–2070. [Google Scholar] [CrossRef]
- Dassou, A.G.; Dépigny, S.; Canard, E.; Vinatier, F.; Carval, D.; Tixier, P. Contrasting effects of plant diversity across arthropod trophic groups in plantain-based agroecosystems. Basic Appl. Ecol. 2016, 17, 11–20. [Google Scholar] [CrossRef]
- Maisonneuve, C.; Rioux, S. Importance of riparian habitats for small mammal and herpetofaunal communities in agricultural landscapes of southern Québec. Agric. Ecosyst. Environ. 2001, 83, 165–175. [Google Scholar] [CrossRef]
- Eyre, M.D.; Lott, D.A.; Garside, A. Assessing the potential for environmental monitoring using ground beetles (Coleoptera: Carabidae) with riverside and Scottish data. Ann. Zool. Fennici 1996, 33, 157–163. [Google Scholar]
- Eyre, M.D.; Luff, M.L.; Lott, D.A. The importance of exposed riverine sediments for phytophagous beetles (Coleoptera) in Scotland and Northern England. Aquat. Conserv. Mar. Freshw. Ecosyst. 2002, 12, 553–566. [Google Scholar] [CrossRef]
- Andersen, J.; Hanssen, O. Riparian Beetles, a Unique, but Vulnerable Element in the Fauna of Fennoscandia. Biodivers. Conserv. 2005, 14, 3497–3524. [Google Scholar] [CrossRef]
- Lambeets, K.; Vandegehuchte, M.L.; Maelfait, J.P.; Bonte, D. Integrating environmental conditions and functional life-history traits for riparian arthropod conservation planning. Biol. Conserv. 2009, 142, 625–637. [Google Scholar] [CrossRef]
- Haddaway, N.R.; Brown, C.; Eales, J.; Eggers, S.; Josefsson, J.; Kronvang, B.; Randall, N.P.; Uusi-Kämppä, J. The multifunctional roles of vegetated strips around and within agricultural fields. Environ. Evid. 2018, 7, 1–43. [Google Scholar] [CrossRef]
- Bonn, A.; Hagen, K.; Wohlgemuth-Von Reiche, D. The significance of flood regimes for Carabid beetle and spider communities in riparian habitats—A comparison of three major rivers in Germany. River Res. Appl. 2002, 18, 43–64. [Google Scholar] [CrossRef]
- Cole, L.J.; Brocklehurst, S.; Robertson, D.; Harrison, W.; McCracken, D.I. Riparian buffer strips: Their role in the conservation of insect pollinators in intensive grassland systems. Agric. Ecosyst. Environ. 2015, 211, 207–220. [Google Scholar] [CrossRef]
- Halaj, J.; Cady, A.B.; Uetz, G.W. Modular habitat refugia enhance generalist predators and lower plant damage in soybeans. Environ. Entomol. 2000, 29, 383–393. [Google Scholar] [CrossRef]
- Aldebron, C.; Jones, M.S.; Snyder, W.E.; Blubaugh, C.K. Soil organic matter links organic farming to enhanced predator evenness. Biol. Control 2020, 146, 104278. [Google Scholar] [CrossRef]
- Baba, Y.G.; Tanaka, K. Factors affecting abundance and species composition of generalist predators (Tetragnatha spiders) in agricultural ditches adjacent to rice paddy fields. Biol. Control 2016, 103, 147–153. [Google Scholar] [CrossRef]
- Nyffeler, M.; Breene, R.G. Spiders associated with selected European hay meadows, and the effects of habitat disturbance, with the predation ecology of the crab spiders, Xysticus spp. (Araneae, Thomisidae). J. Appl. Entomol. 1990, 110, 149–159. [Google Scholar] [CrossRef]
- Lahondère, C.; Vinauger, C.; Okubo, R.P.; Wolff, G.H.; Chan, J.K.; Akbari, O.S.; Riffell, J.A. The olfactory basis of orchid pollination by mosquitoes. Proc. Natl. Acad. Sci. USA 2020, 117, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Peach, D.A.H.; Carroll, C.; Meraj, S.; Gomes, S.; Galloway, E.; Balcita, A.; Coatsworth, H.; Young, N.; Uriel, Y.; Gries, R.; et al. Nectar-dwelling microbes of common tansy are attractive to its mosquito pollinator, Culex pipiens L. bioRxiv 2020, 1–9. [Google Scholar] [CrossRef]
- van Looy, K.; Vanacker, S.; Jochems, H.; De Blust, G.; Dufrêne, M. Ground beetle habitat templets and riverbank integrity. River Res. Appl. 2005, 21, 1133–1146. [Google Scholar] [CrossRef]
- Januschke, K.; Verdonschot, R.C.M. Effects of river restoration on riparian ground beetles (Coleoptera: Carabidae) in Europe. Hydrobiologia 2016, 769, 93–104. [Google Scholar] [CrossRef]
- Šustek, Z. Impact of water management on a carabid community (Insecta, Coleoptera) in a central European floodplain forest. Quad. Staz. Ecol. Civ. Mus. St. Nat. Ferrara 1994, 6, 293–313. [Google Scholar]
- Skalsk, T.; Kedzior, R.; Radecki-Pawlik, A. Riverine ground beetles as indicators of inundation frequency of mountain stream: A case study of the ochotnica stream, southern Poland. Balt. J. Coleopterol. 2012, 12, 117–126. [Google Scholar]
- Jähnig, S.C.; Brunzel, S.; Gacek, S.; Lorenz, A.W.; Hering, D. Effects of re-braiding measures on hydromorphology, floodplain vegetation, ground beetles and benthic invertebrates in mountain rivers. J. Appl. Ecol. 2009, 46, 406–416. [Google Scholar] [CrossRef]
- Greenwood, M.T.; Bickerton, M.A.; Petts, G.E. Patial distribution of spiders on the floodplain of the river trent, UK: The role of hydrological setting. Regul. Rivers Res. Manag. 1995, 10, 303–313. [Google Scholar] [CrossRef]
- Ng, K.; Barton, P.S.; Blanchard, W.; Evans, M.J.; Lindenmayer, D.B.; Macfadyen, S.; McIntyre, S.; Driscoll, D.A. Disentangling the effects of farmland use, habitat edges, and vegetation structure on ground beetle morphological traits. Oecologia 2018, 188, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Eyre, M.D.; Luff, M.L. The use of ground beetles (Coleoptera: Carabidae) in conservation assessments of exposed riverine sediment habitats in Scotland and northern England. J. Insect Conserv. 2002, 6, 25–38. [Google Scholar] [CrossRef]
- Latty, E.F.; Werner, S.M.; Mladenoff, D.J.; Raffa, K.F.; Sickley, T.A. Response of ground beetle (Carabidae) assemblages to logging history in northern hardwood-hemlock forests. For. Ecol. Manag. 2006, 222, 335–347. [Google Scholar] [CrossRef]
- Cole, L.J.; Brocklehurst, S.; Elston, D.A.; McCracken, D.I. Riparian field margins: Can they enhance the functional structure of ground beetle (Coleoptera: Carabidae) assemblages in intensively managed grassland landscapes? J. Appl. Ecol. 2012, 49, 1384–1395. [Google Scholar] [CrossRef]
- Madra, A.; Konwerski, S.; Matuszewski, S. Necrophilous Staphylininae (Coleoptera: Staphylinidae) as indicators of season of death and corpse relocation. Forensic Sci. Int. 2014, 242, 32–37. [Google Scholar] [CrossRef]
- Tomlin, A.D.; McLeod, D.G.R.; Moore, L.V.; Whistlecraft, J.W.; Miller, J.J.; Tolman, J.H. Dispersal of Aleochara bilineata [Col.: Staphylinidae] following inundative releases in urban gardens. Entomophaga 1992, 37, 55–63. [Google Scholar] [CrossRef]
- Parker, J. Staphylinids. Curr. Biol. 2017, 27, R49–R51. [Google Scholar] [CrossRef]
- Hoffmann, H.; Kleeberg, A.; Görn, S.; Fischer, K. Riverine fen restoration provides secondary habitat for endangered and stenotopic rove beetles (Coleoptera: Staphylinidae). Insect Conserv. Divers. 2018, 11, 194–203. [Google Scholar] [CrossRef]
- Foster, W.A. Mosquito sugar feeding and reproductive energetics. Annu. Rev. Entomol. 1995, 40, 443–474. [Google Scholar] [CrossRef]
- Baniţã, E.; Cantoreanu, M.J.A. Contribution to the study of Cicada (Homoptera-Aucheenarrhyncha) populations from winter wheat crops in Oltenia Plain. Rom. Agric. Res. 1996, 5–6, 87–93. [Google Scholar]
- Bidau, C.J. Patterns in Orthoptera biodiversity. I. Adaptations in ecological and evolutionary contexts. J. Insect Biodivers. 2014, 2, 1. [Google Scholar] [CrossRef]
- Faiz, A.U.H.; Hassan, M.U.; Bagaturov, M.F.; Mahmood, M.; Faiz, L.Z.; Tariq, G. Insect Diversity and Association with Plants: A Case Study in Rural Areas of Dhirkot, Azad Kashmir Pakistan. J. Bioresour. Manag. 2020, 7, 21–32. [Google Scholar] [CrossRef]
- Steinger, T.; Klötzli, F.; Ramseier, H. Experimental Assessment of the Economic Injury Level of the Cereal Leaf Beetle (Coleoptera: Chrysomelidae) in Winter Wheat. J. Econ. Entomol. 2020, 113, 1823–1830. [Google Scholar] [CrossRef] [PubMed]
- Pearce, J.L.; Venier, L.A. The use of ground beetles (Coleoptera: Carabidae) and spiders (Araneae) as bioindicators of sustainable forest management: A review. Ecol. Indic. 2006, 6, 780–793. [Google Scholar] [CrossRef]
- Buchholz, S. Ground spider assemblages as indicators for habitat structure in inland sand ecosystems. Biodivers. Conserv. 2010, 19, 2565–2595. [Google Scholar] [CrossRef]
- Cobbold, S.M.; Supp, S.R. Patch shape alters spider community structure: Links between microhabitat choice and sensitivity to increased edge habitat. J. Insect Conserv. 2012, 16, 581–589. [Google Scholar] [CrossRef]
- Imbert, C.; Papaïx, J.; Husson, L.; Warlop, F.; Lavigne, C. Pests, but not predators, increase in mixed fruit tree–vegetable plots compared to control vegetable plots in a Mediterranean climate. Agrofor. Syst. 2020, 94, 627–638. [Google Scholar] [CrossRef]
- Jonsson, M.; Stenroth, K. True autochthony and allochthony in aquatic—Terrestrial resource fluxes along a landuse gradient. Freshw. Sci. 2016, 35, 882–894. [Google Scholar] [CrossRef]
- Luke, S.H.; Slade, E.M.; Gray, C.L.; Annammala, K.V.; Drewer, J.; Williamson, J.; Agama, A.L.; Ationg, M.; Mitchell, S.L.; Vairappan, C.S.; et al. Riparian buffers in tropical agriculture: Scientific support, effectiveness and directions for policy. J. Appl. Ecol. 2019, 56, 85–92. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).