Essential Trace Elements and Arsenic in Thermal Springs, Afghanistan
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.1.1. Location and Climate Condition
2.1.2. Geological Setting
2.1.3. Hydrological Setting
2.2. Water Sampling and Field Parameter Measurements
2.3. Water Analysis Methods
2.4. Water Type Determination
3. Results
3.1. Physical Parameters of Thermal Springs
3.2. Hydrochemistry of Thermal Springs
3.2.1. Major Ions
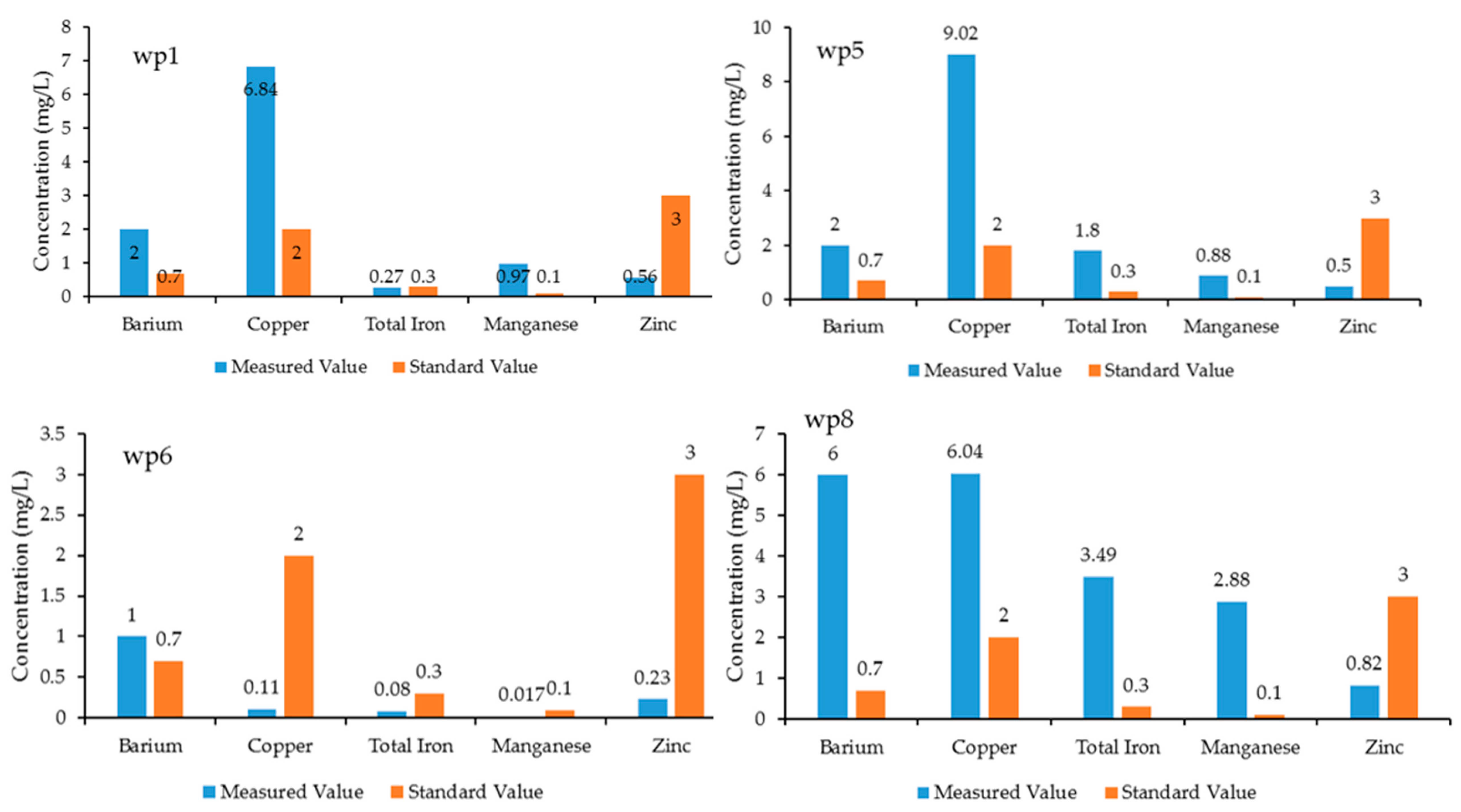
3.2.2. Trace Element Concentrations
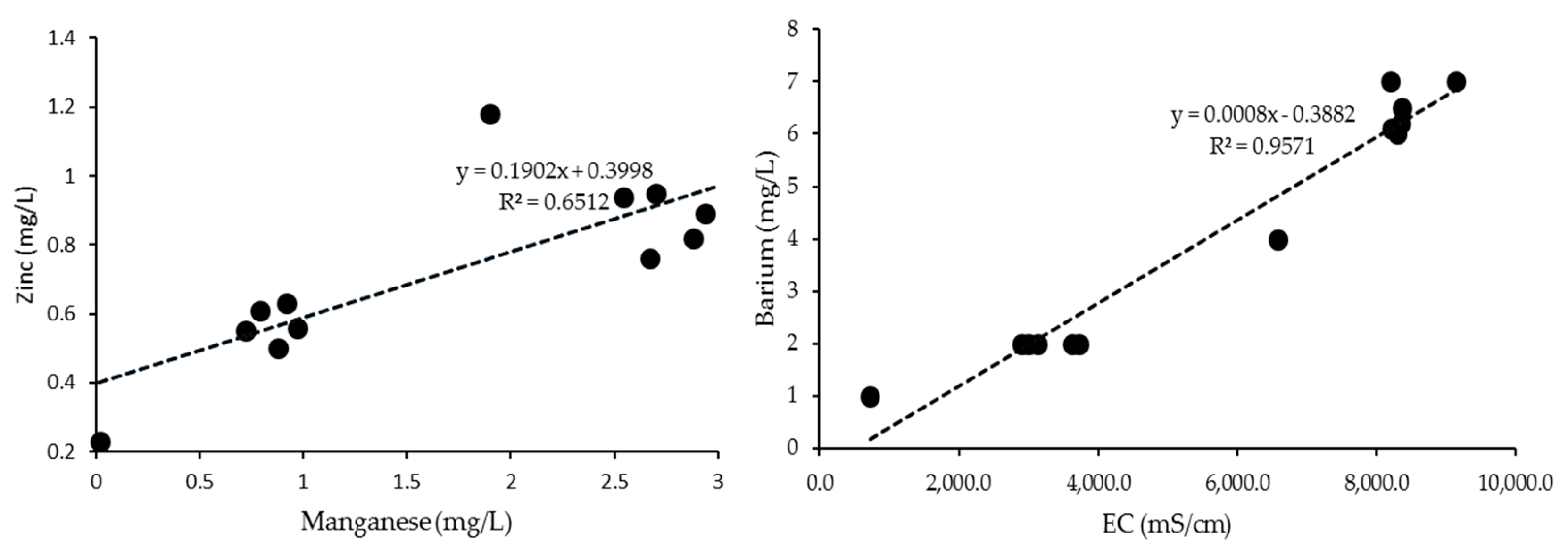
3.3. Correlations
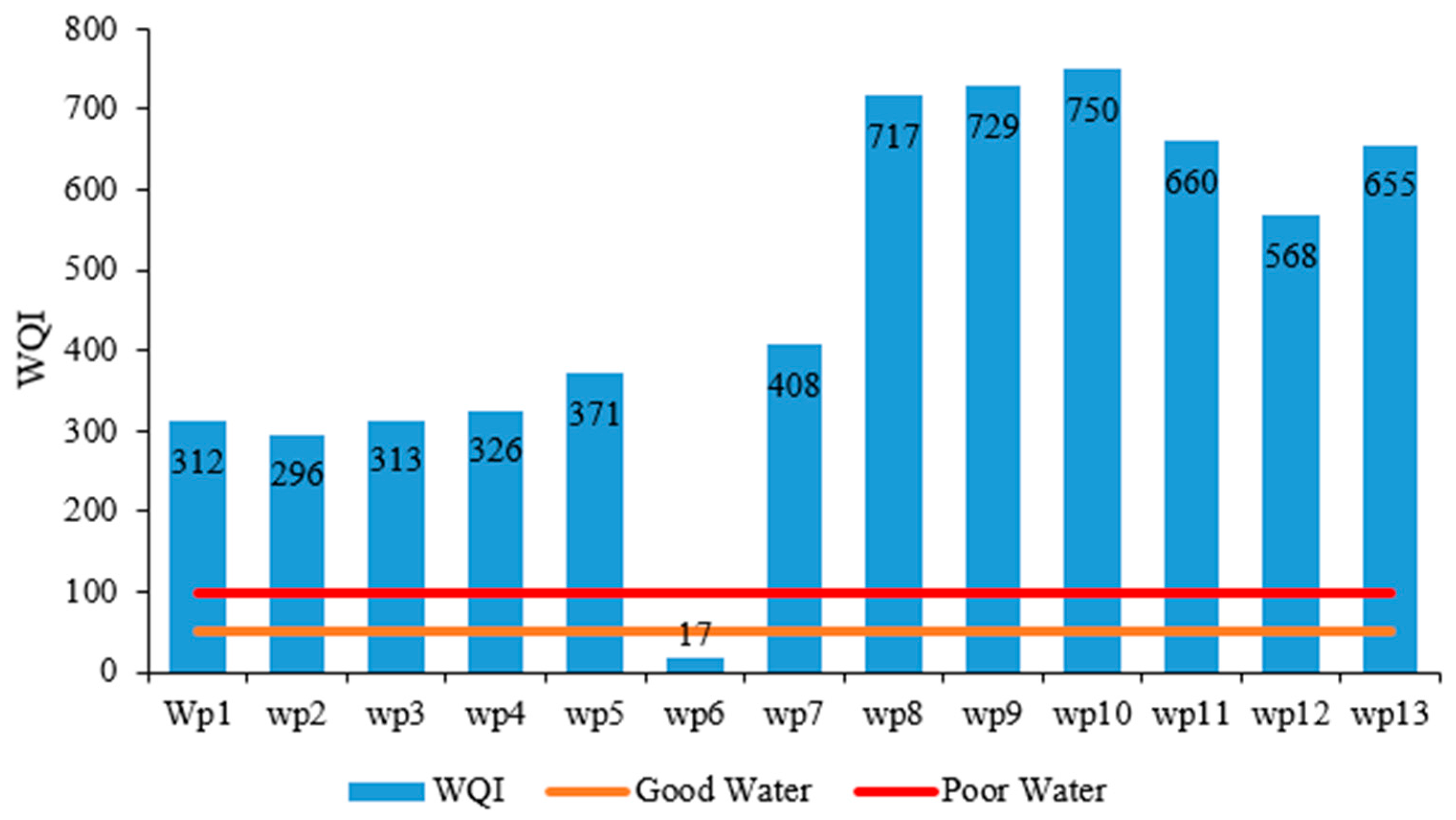
3.4. Water Type and Health Risk Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Mahala, S.C. Geology, Chemistry and Genesis of Thermal Springs of Odisha, India; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Dulanya, Z.; Morales-Simfors, N.; Sivertun, A. Comparative study of the silica and cation geothermometry of the Malawi hot springs: Potential alternative energy source. J. Afr. Earth Sci. 2010, 57, 321–327. [Google Scholar] [CrossRef]
- Wada, O. What are trace elements? Their deficiency and excess states. Jpn. Med. Assoc. 2003, 129, 607–612. [Google Scholar]
- Munoz, M.; Bhattacharya, P.; Stacek, O.; Ramos, O.; Aguirre, J.; Bundschuh, J.; Maity, J. Arsenic and other trace elements in thermal springs and in cold waters from drinking water wells on the Bolivian Altiplano. J. South Am. Earth Sci. 2015, 60, 10–20. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, Z.; Gao, S.; Günther, D.; Xu, J.; Gao, C.; Chen, H. In situ analysis of major and trace elements of anhydrous minerals by LA-ICP-MS with applying an internal standard. Chem. Geol. 2008, 257, 34–43. [Google Scholar] [CrossRef]
- Jones-Hughes, T.; Peters, J.; Whear, R.; Cooper, C.; Evans, H.; Depledge, M.; Pearson, M. Are interventions to reduce the impact of arsenic contamination of groundwater on human health in developinng countries affective? A systematic review. Environ. Evid. 2013, 2, 11. [Google Scholar] [CrossRef]
- Johannesson, K.; Tang, J. Conservative behavior of arsenic and other oxyanion-forming trace elements in an axic groundwater flow system. J. Hydrol. 2009, 378, 13–28. [Google Scholar] [CrossRef]
- Phan, K.; Phan, S.; Huoy, L.; Suy, B.; Wong, M.; Hashim, J.; Kim, K. Assessing mixed trace elements in groundwater and their health risk of residents living in the Mekong River basin of Cambodia. Environ. Pollut. 2013, 182, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Reimann, C.; Robert, G. Geochemical background- concept and reality. Sci. Total Environ. 2005, 350, 12–27. [Google Scholar] [CrossRef]
- Das, D.; Chatterjee, A.; Mandal, B.K.; Samanta, G.; Chakraborti, D. Arsenic in groundwater in six district of West Bengal, India: The biggest arsenic calamity in the world: Part2. Arsenic concentration I drinking water, hair, nails, urine, skin scale and liver tissue (biopsy) of the affected people. Analyst 1995, 120, 917–924. [Google Scholar] [CrossRef]
- Smith, A.H.; Lingas, E.O.; Rahman, M. Concentration of drinking water by arsenic in Bangladesh: A public health emergency. Bull. WHO 2000, 78, 1093–1101. [Google Scholar]
- Tan, J. The Atlas of Endemic Diseases and Their Environments in the People’s Republic of China; Science Press: Beijing, China, 1989. [Google Scholar]
- Combs, G.F.; Welch, R.M.; Duxbury, J.M.; Uphoff, N.T.; Nesheim, M.C. Food-Based Approaches to Preventing Micronutrient Malnutrition: An International Research Agenda; Cornell University: Ithaca, NY, USA, 1996. [Google Scholar]
- Vengosh, A.; Cahit, H.; Ismai, H.K. Geochemical constraints for the origen of thermal waters from western Turkey. Appl. Geochem. 2002, 18, 1117–1119. [Google Scholar] [CrossRef]
- Binda, G.; Andrea, P.; Alessandro, M.M.; Paula, J.N.; Michael, R.R. Towards the understanding of hydrogeochemical seismic responses in karst aquifers: A retrospective meta-analysis focused on the apennines (Italy). Minerals 2020, 10, 1058. [Google Scholar] [CrossRef]
- Tonani, F. Geochemical methods of exploration for geothermal energy. Geothermics 1970, 2, 492–515. [Google Scholar] [CrossRef]
- White, D.E. Geochemistry applied to the discovery, evaluation, and exploration of geothermal energy resources. Geothermics 1970, 2, 58–80. [Google Scholar]
- Fournier, R.O.; Truesdell, A.H. An empirical Na-K-Ca geothermometer for natural waters. Geochim. Cosmochim. Acta 1973, 37, 1255–1275. [Google Scholar] [CrossRef]
- Ellis, A.J.; Mahon, W.A.J. Chemistry and Geothermal Systems; Academic Press: New York, NY, USA, 1977. [Google Scholar]
- Fournier, R.O. A revised equation for Na-Kgeothermometer. Trans. Geotherm. Resour. Counc. 1979, 3, 221–224. [Google Scholar]
- Giggenbach, W.F.; Gonfiantini, R.; Jangi, B.L.; Truesdell, A.H. Isotopic and chemical composition of Parbati valley geothermal discharges, NW-Himalaya, India. Geothermics 1983, 12, 199–222. [Google Scholar] [CrossRef]
- Giggenbach, W.F. Geothermal solute equilibria. Derivation of Na–K–Mg–Ca geoindicators. Geochim. Cosmochim. Acta 1988, 52, 2749–2765. [Google Scholar] [CrossRef]
- King, M.; Sturtewagen, B. Making the Most of Afghanistan’s River Basins: Opportunities for Regional Cooperation; EastWest Institute: New York, NY, USA, 2010. [Google Scholar]
- Favre, R.; Kamal, G. Water Atlas of Afghanistan; FAO: Kabul, Afghanistan, 2004. [Google Scholar]
- Tünnermeier, T.; Houben, G. Hydrogeology of the Kabul Basin, Part 1&2; BGR: Hannover, Germany, 2005. [Google Scholar]
- Mack, T. Groundwater availability in the Kabul basin, Afghanistan. Groundw. South Asia 2018, 23–35. [Google Scholar] [CrossRef]
- Mack, T.; Akbari, M.; Ashoor, M.; Chornack, M.; Coplen, T.; Emerson, D.; Verstraeten, I. Conceptual Model of Water Resources in the Kabul Basin, Afghanistan; U.S.Geological Survey: Reston, VA, USA, 2010.
- Broshears, R.; Akbari, M.; Chornack, M.; Mueller, D.; Ruddy, B. Inventory of Groundwater Resources in the Kabul Basin, Afghanistan; U.S. Geological Survey: Reston, VA, USA, 2005.
- Saba, D.; Najaf, M.E.; Musazai, A.; Taraki, S. Geothermal Energy in Afghanistan: Prospects and Potential; Center on International Cooperation: New York, NY, USA, 2004. [Google Scholar]
- Abullah, S.; Chmyriov, V. Geology and Mineral Resources of Afghanistan; British Geological Survey: Amersham, UK, 2008. [Google Scholar]
- Kyriazis, D.; Zagana, E.; Stamatis, G.; Fillippidis, F.; Psomiadis, E. Assessment of groundwater pollution in relation to heavy metals of the alluvial aquifer of Tn hriasion Plan (NW Attica). Bull. Geol. Soc. Greece 2013, 47, 731–739. [Google Scholar] [CrossRef][Green Version]
- Azimi, M. Geography of Bamyan Province (Physical, Human, Economical); Alhoda: Kabul, Afghanistan, 2012. [Google Scholar]
- Aliyar, Q. Temperature change during three dicades in Central Bamyan. Acad. J. Bamyan Univ. 2019, 11, 1–9. [Google Scholar]
- Cook, D. Bamyan Province Climatology and Temperature Extremes in Afghanistan; Defense Technology Agency: Kabul, Afghanistan, 2011. [Google Scholar]
- USGS. Geological Map of Afghanistan; MicroImages: Lincoln, NE, USA, 2005.
- Jawadi, H.A.; Jay, S.; Daniel, D.S. A detailed assessment of groundwater quality in the Kabul Basin, Afghanistan, and suitability for future development. Water 2020, 12, 2890. [Google Scholar] [CrossRef]
- Batabyal, A.; Chakraborty, S. Hydrogeochemistry and water quality index in the assessment of groundwater quality for drinking use. Water Environ. Res. 2015, 607–619. [Google Scholar] [CrossRef] [PubMed]
- Ghoreyshinia, S.K.; Deshaee, A. Hydrogeochemistry, circulation path and arsenic distribution in Tahlab aquifer, East of Taftan Volcano, SE Iran. Appl. Geochem. 2020, 119. [Google Scholar] [CrossRef]
- Lafta, J. Analysis of water quality using chemical-physical- biological paramters of the kinds fo water used for drinking in the Baghdad Province- Al Adhamiya city. Chem. Mater. Res. 2015, 7, 1–4. [Google Scholar]
- Patil, P.; Sawant, D.; Deshmukh, R. Physico-chemical paramters for testing of water- A review. Int. J. Environ. Sci. 2012, 3, 1194–1207. [Google Scholar]
- Schwenzer, S.; Tommaseo, C.; Kersten, M.; Kirnbauer, T. Speciation and oxidation kinetics of arsenic in the thermal springs of Wiesbade spa, Germany. Fresenius’ J. Anal. Chem. 2001, 371, 927–933. [Google Scholar] [CrossRef]
- Kikawada, Y.; Fuji, H.; Ohno, Y.; Oi, T. Mobility of arsenic in hot springs water accompanying its penetration into the ground. J. Radioanal. Nucl. Chem. 2008, 278, 323–326. [Google Scholar] [CrossRef]
- Etim, E.; Odoh, R.; Itodo, A.; Umoh, S.; Lawal, U. Water quality index for the assessment of water quality from different sources in the Niger Delta region of Nigeria. Front. Sci. 2013, 3, 89–95. [Google Scholar] [CrossRef]
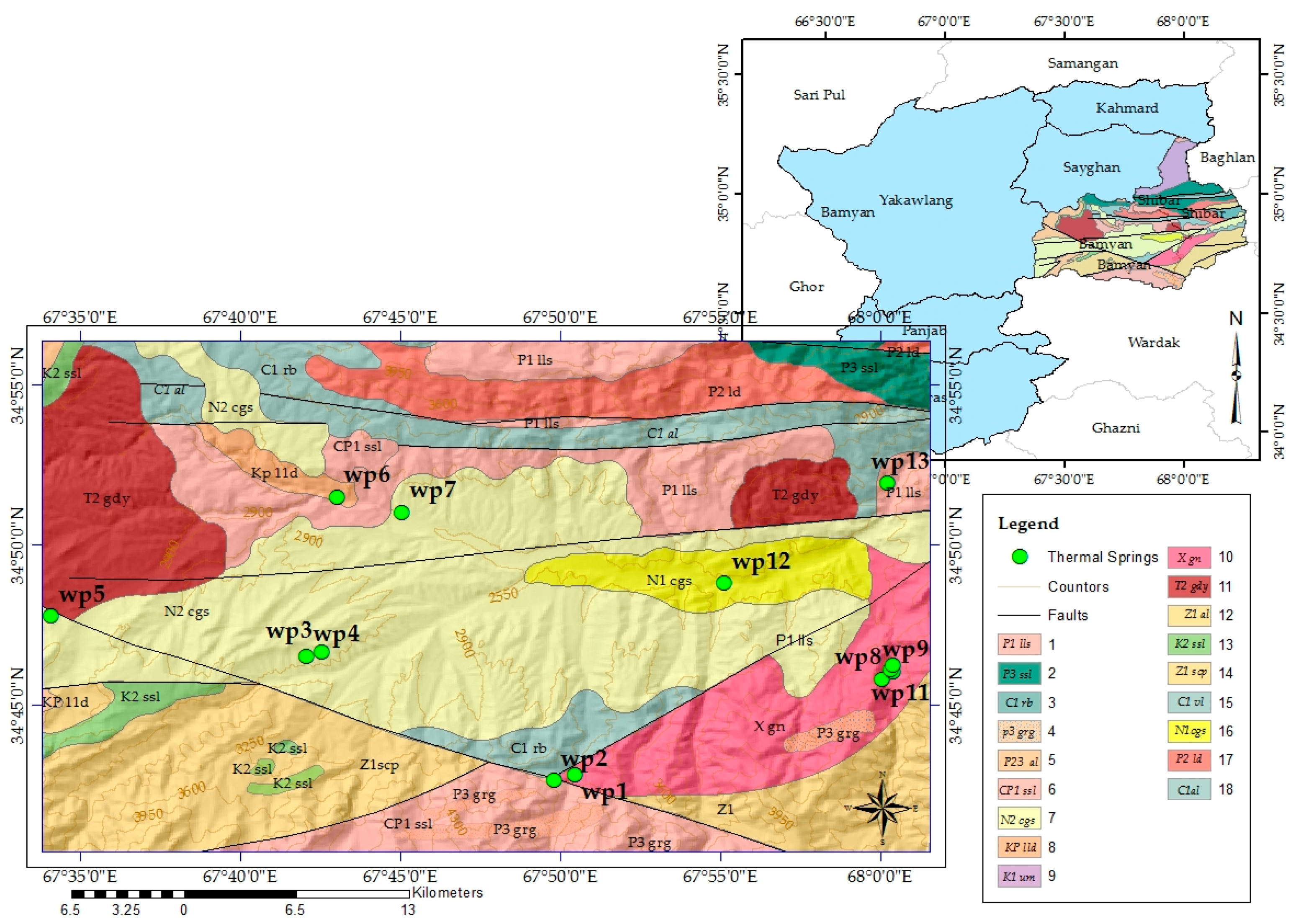
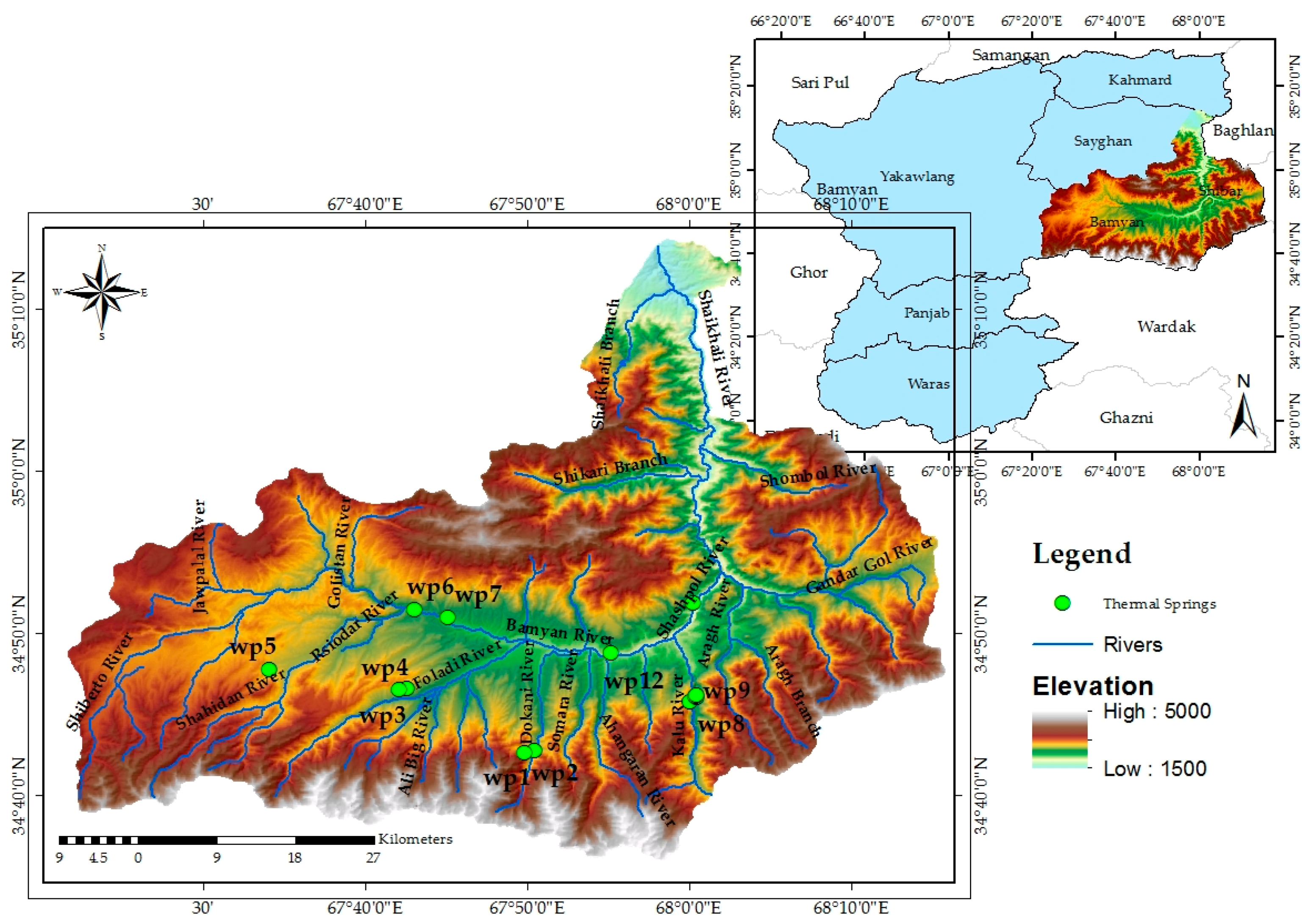

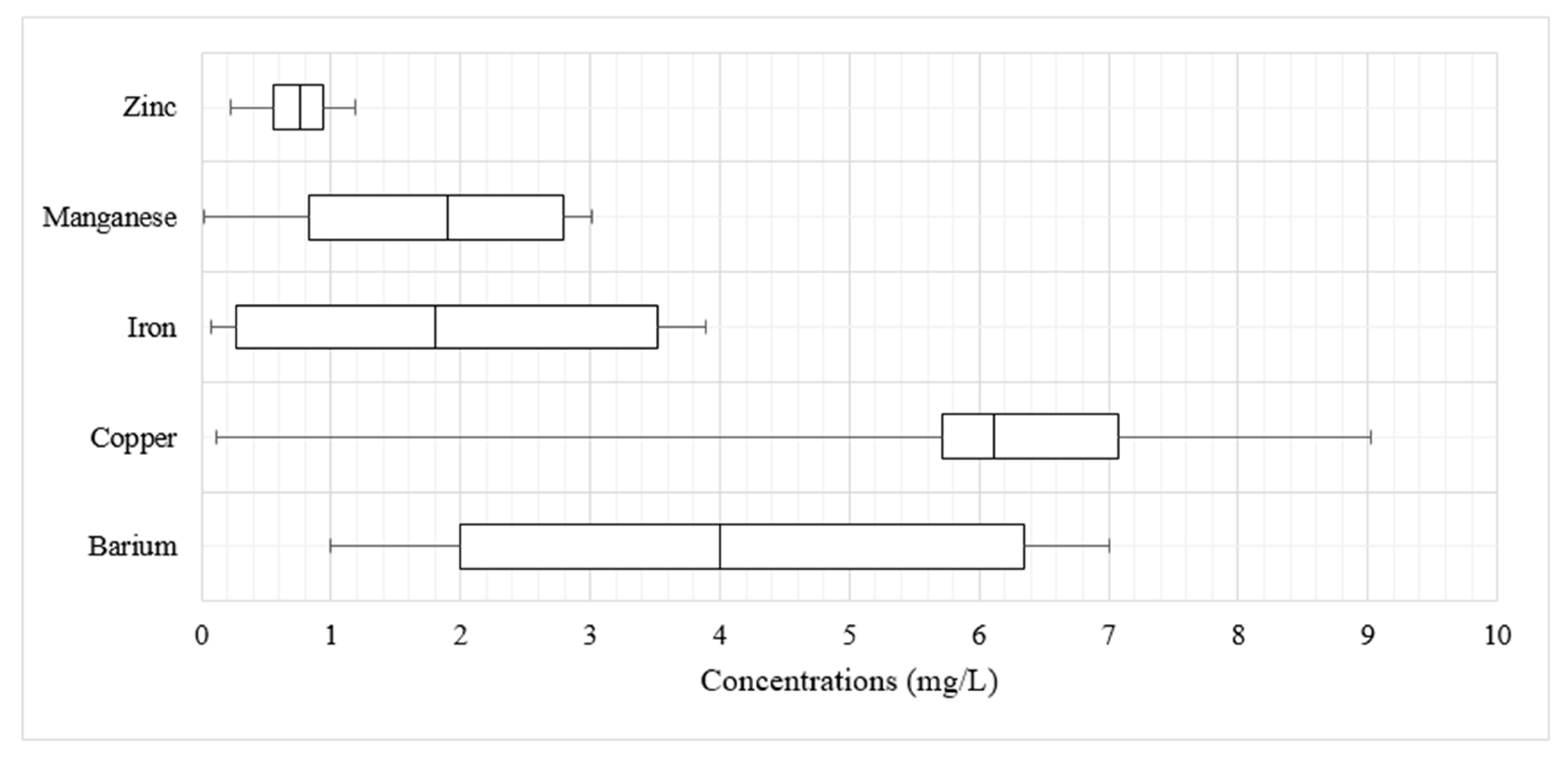
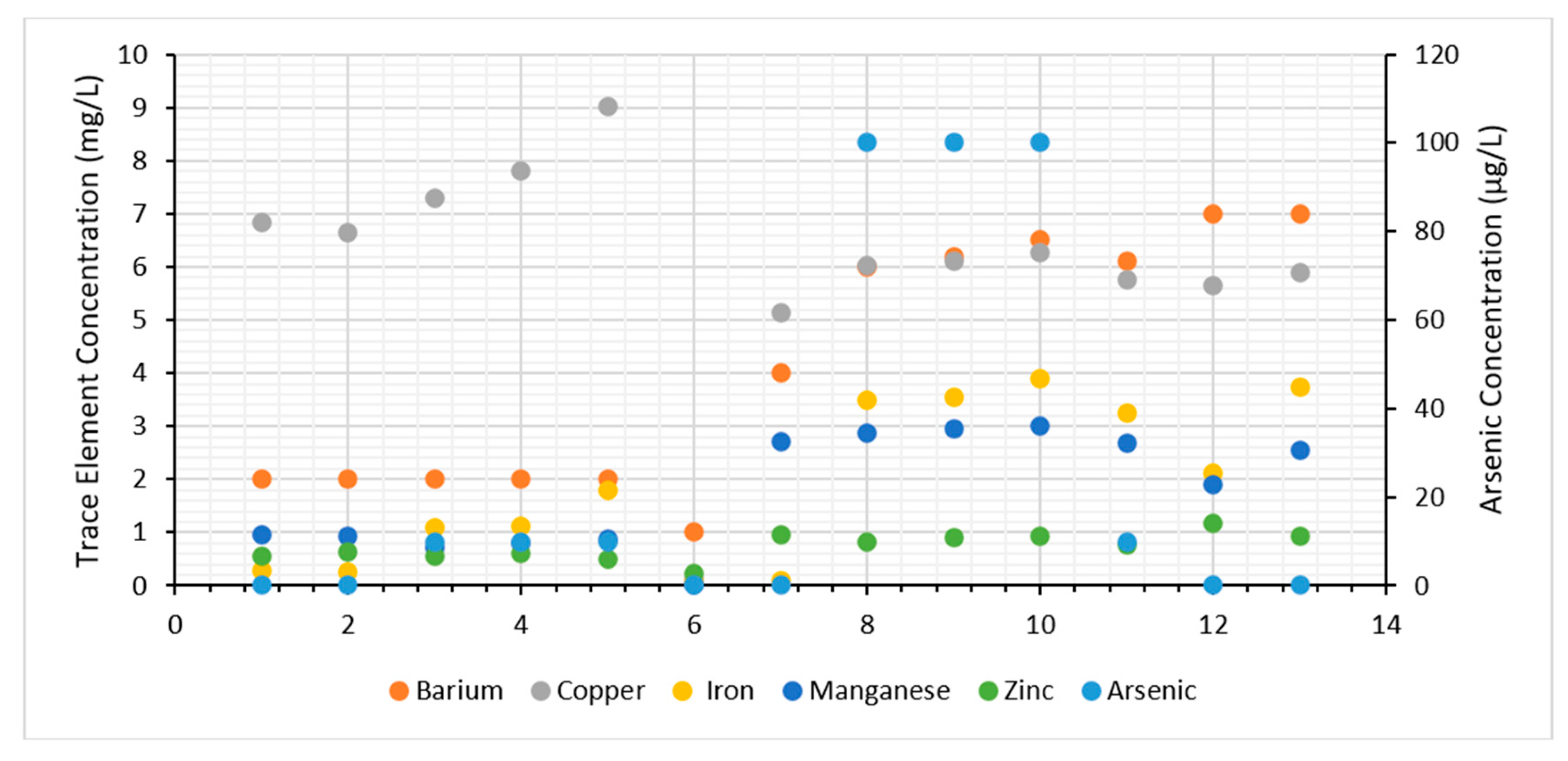
| Symbol | Location | Description of Location | Coordinates (WGS1984) | Elevation (m) | |
|---|---|---|---|---|---|
| Longitude | Latitude | ||||
| wp1 | Darra-e-Dokani1 | Mountainous region, located at the end of Dokani valley close to Baba Mountain ridge | 67.84 | 34.71 | 2939 |
| wp2 | Darra-e-Dokani 2 | Mountainous region, located at the end of Dokani valley close to Baba Mountain ridge | 67.83 | 34.71 | 2945 |
| wp3 | Syalayag Foladi1 | Located at the end and right side of Foladi valley, near Baba Mountain ridge, southwestern part of Bamyan City | 67.709 | 34.777 | 3530 |
| wp4 | Syalayag Foladi 2 | Located at the end and right side of Floadi valley, near Baba Mountain ridge, southwestern part of Bamyan City | 67.701 | 34.775 | 3537 |
| wp5 | Azdar Shahidan | Located in the Shahidan area, left side of Bamyan–Yakawlang main road with travertine and limestone sediments | 67.57 | 34.80 | 3068 |
| wp6 | Khoja Ali | Located near the southern entrance gate of Bamyan City, at contact of sedimentary and igneous rocks | 67.72 | 34.86 | 2635 |
| wp7 | Asidar Bamyan | Located at the end of Azhdar valley, south of Bamyan City with travertine and limestone | 67.75 | 34.85 | 2729 |
| wp8 | Paymory Kalo1 | Located at the right side of Kabul–Bamyan highway, middle point of Kalu valley, eastern part of Bamyan City | 67.05 | 34.79 | 2505 |
| wp9 | Paymory Kalo2 | Located at the lift side of Kabul–Bamyan highway, middle point of Kalu valley, eastern part of Bamyan City | 68.007 | 34.767 | 2504 |
| wp10 | Paymory Kalo 3 | Located at the lift side of Kabul–Bamyan highway, middle point of Kalu valley, eastern part of Bamyan City | 68.006 | 34.768 | 2502 |
| wp11 | Paymory Kalo 4 | Located at the lift side of Kabul–Bamyan highway, middle point of Kalu valley, eastern part of Bamyan City | 68.007 | 34.77 | 2502 |
| wp12 | Dahana Ahangaran | Located at the beginning of Ahangaran valley, northeastern part of Bamyan City | 67.92 | 34.81 | 2431 |
| wp13 | Shash Pul | Located at Shash Pol area, norteastern part of Bamyan City | 67.45 | 34.51 | 2361 |
| Physical Tests | ||||
|---|---|---|---|---|
| Tests | Unit | Methods | * MCL as per USEPA | * MCL as per WHO |
| pH | ( ) | HACH #8156 | 6.5–8.5 | 6.5–8.5 |
| Color | (CoPt) | HACH #8125 | 15 | 15 |
| Conductivity | (µS/cm) | HACH #8160 | - | - |
| Resistivity | (Ω.cm) | HACH-HQ Direct measurement | - | - |
| Salinity | (%) | HACH-HQ Direct measurement | - | - |
| Turbidity | (NTU) | HACH #8237 | 5 | 5 |
| TDS | (mg/L) | HACH-HQ Direct measurement | 500 | 1000 |
| Chemical Tests | ||||
| Arsenic | (µg/L) | EZ Arsenic Test Kit | 50 | 10 |
| Barium | (mg/L) | HACH #8014 | 2 | 0.7 |
| Chloride | (mg/L) | HACH #8206 | 250 | 250 |
| Cyanide | (mg/L) | HACH# 8027 | 0.2 | 0.1 |
| Copper | (mg/L) | HACH #8026 | 1.3 | 2 |
| Hardness | (mg/Las CaCO3) | HACH #8329 | 500 | 500 |
| Iron | (mg/L) | HACH #8008 | 0.3 | 0.3 |
| Manganese | (mg/L) | HACH #8149 | 0.05 | 0.1 |
| Nitrite | (mg/L) | HACH #8507 | 1 | 3 |
| Nitrate | (mg/L) | HACH #8039 | 10 | 10 |
| Phosphate | (mg/L) | HACH #8048 | - | - |
| Sulfate | (mg/L) | HACH #8051 | 250 | 400 |
| Zinc | (mg/L) | HACH #8009 | 5 | 3 |
| Study Points | Temperature | pH | Color | Conductivity | Resistivity | Turbidity | TDS |
|---|---|---|---|---|---|---|---|
| (°C) | (* SU) | (CoPt) | (µS/cm) | (Ω.cm) | (NTU) | (mg/L) | |
| wp1 | 25 | 6.68 | 27 | 3630 | 276 | 46 | 1876 |
| wp2 | 25 | 6.8 | 30 | 3720 | 260 | 40 | 1923 |
| wp3 | 25 | 6.7 | 43 | 3123 | 290 | 50 | 1526 |
| wp4 | 24 | 6.5 | 46 | 3000 | 305 | 52 | 1534 |
| wp5 | 24 | 6.43 | 51 | 2900 | 349 | 57 | 1471 |
| wp6 | 23 | 7.24 | 3 | 719 | 1392 | 0.21 | 351 |
| wp7 | 16 | 6.46 | 14 | 6590 | 151.7 | 0.44 | 3530 |
| wp8 | 32 | 6.41 | 120 | 8310 | 120.4 | 41.6 | 4490 |
| wp9 | 31 | 6.39 | 125 | 8354 | 121.1 | 42.5 | 4502 |
| wp10 | 31 | 6.44 | 129 | 8363 | 125 | 43.3 | 4512 |
| wp11 | 32 | 6.56 | 117 | 8213 | 118.9 | 40.5 | 4456 |
| wp12 | 20 | 6.39 | 133 | 9150 | 109.3 | 34 | 4960 |
| wp13 | 23 | 6.4 | 130 | 8205 | 134 | 38.6 | 4687 |
| Symbol | Chloride | Cyanide | Hardness (as CaCO3) | Nitrite | Nitrate | Phosphate | Sulfate | Arsenic (As) | Barium (Ba) | Copper (Cu) | Iron (Fe) | Manganese (Mn) | Zinc (Zn) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (mg/L) | (µg/L) | (mg/L) | |||||||||||
| Wp1 | 365 | 0.01 | 1260 | 0.013 | 2.5 | 0.35 | 488 | 0 | 2 | 6.84 | 0.27 | 0.97 | 0.56 |
| wp2 | 378 | 0.02 | 1287 | 0.02 | 3.1 | 0.65 | 502 | 0 | 2 | 6.65 | 0.25 | 0.92 | 0.63 |
| wp3 | 283 | 0.01 | 1011 | 0.02 | 2.7 | 0.15 | 310 | 10 | 2 | 7.3 | 1.1 | 0.72 | 0.55 |
| wp4 | 272 | 0.01 | 994 | 0.02 | 2.82 | 0.13 | 298 | 10 | 2 | 7.8 | 1.13 | 0.79 | 0.61 |
| wp5 | 262 | 0.009 | 982 | 0.02 | 3.1 | 0.19 | 340 | 10 | 2 | 9.02 | 1.8 | 0.88 | 0.5 |
| wp6 | 49 | 0.004 | 284 | 0.01 | 1.8 | 0.12 | 64 | 0 | 1 | 0.11 | 0.08 | 0.017 | 0.23 |
| wp7 | 590 | 0.006 | 2340 | 0.006 | 1.3 | 0.08 | 870 | 0 | 4 | 5.14 | 0.1 | 2.7 | 0.95 |
| wp8 | 1280 | 0.022 | 2615 | 1.11 | 17 | 0.48 | 1370 | 100 | 6 | 6.04 | 3.49 | 2.88 | 0.82 |
| wp9 | 1297 | 0.02 | 2722 | 1.13 | 19 | 0.52 | 1392 | 100 | 6.2 | 6.12 | 3.55 | 2.94 | 0.89 |
| wp10 | 1303 | 0.02 | 2781 | 1.15 | 21 | 0.61 | 1401 | 100 | 6.5 | 6.27 | 3.89 | 3.01 | 0.94 |
| wp11 | 1286 | 0.021 | 2804 | 1.12 | 19.4 | 0.55 | 1362 | 10 | 6.1 | 5.77 | 3.25 | 2.67 | 0.76 |
| wp12 | 1630 | 0.013 | 2490 | 0.79 | 9.8 | 0.23 | 1430 | 0 | 7 | 5.66 | 2.11 | 1.9 | 1.18 |
| wp13 | 1346 | 0.02 | 2549 | 0.97 | 12.6 | 0.34 | 1375 | 0 | 7 | 5.9 | 3.73 | 2.54 | 0.94 |
| USEPA | 250 | 0.2 | 500 | 1 | 10 | - | 250 | 10 | 0.7 | 1–2 | 0.3 | 0.1 | 3 |
| WHO | 250 | 0.1 | 500 | 3 | 10 | 0.70 | 400 | 10 | 0.7 | 1–2 | 0.3 | 0.1 | 3 |
| PH | EC | Turbidity | TDS | Arsenic | Barium | Copper | Iron | Manganese | Zinc | |
|---|---|---|---|---|---|---|---|---|---|---|
| PH | 1 | |||||||||
| EC | −0.73 | 1 | ||||||||
| Turbidity | −0.46 | 0.06 | 1 | |||||||
| TDS | −0.72 | 0.99 | 0.05 | 1 | ||||||
| Arsenic | −0.38 | 0.49 | 0.22 | 0.48 | 1 | |||||
| Barium | −0.68 | 0.97 | 0.06 | 0.98 | 0.49 | 1 | ||||
| Copper | −0.66 | 0.11 | 0.84 | 0.09 | 0.08 | 0.03 | 1 | |||
| Iron | −0.63 | 0.77 | 0.39 | 0.78 | 0.67 | 0.84 | 0.19 | 1 | ||
| Manganese | −0.71 | 0.93 | −0.01 | 0.93 | 0.61 | 0.89 | 0.10 | 0.76 | 1 | |
| Zinc | −0.78 | 0.92 | 0.03 | 0.91 | 0.31 | 0.87 | 0.21 | 0.56 | 0.81 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jawadi, H.A.; Malistani, H.A.; Moheghy, M.A.; Sagin, J. Essential Trace Elements and Arsenic in Thermal Springs, Afghanistan. Water 2021, 13, 134. https://doi.org/10.3390/w13020134
Jawadi HA, Malistani HA, Moheghy MA, Sagin J. Essential Trace Elements and Arsenic in Thermal Springs, Afghanistan. Water. 2021; 13(2):134. https://doi.org/10.3390/w13020134
Chicago/Turabian StyleJawadi, Hussain Ali, Hasan Ali Malistani, Mohammad Anvar Moheghy, and Jay Sagin. 2021. "Essential Trace Elements and Arsenic in Thermal Springs, Afghanistan" Water 13, no. 2: 134. https://doi.org/10.3390/w13020134
APA StyleJawadi, H. A., Malistani, H. A., Moheghy, M. A., & Sagin, J. (2021). Essential Trace Elements and Arsenic in Thermal Springs, Afghanistan. Water, 13(2), 134. https://doi.org/10.3390/w13020134







