Recovery of Cr(III) from Tannery Effluents by Diafiltration Using Chitosan Modified Membranes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
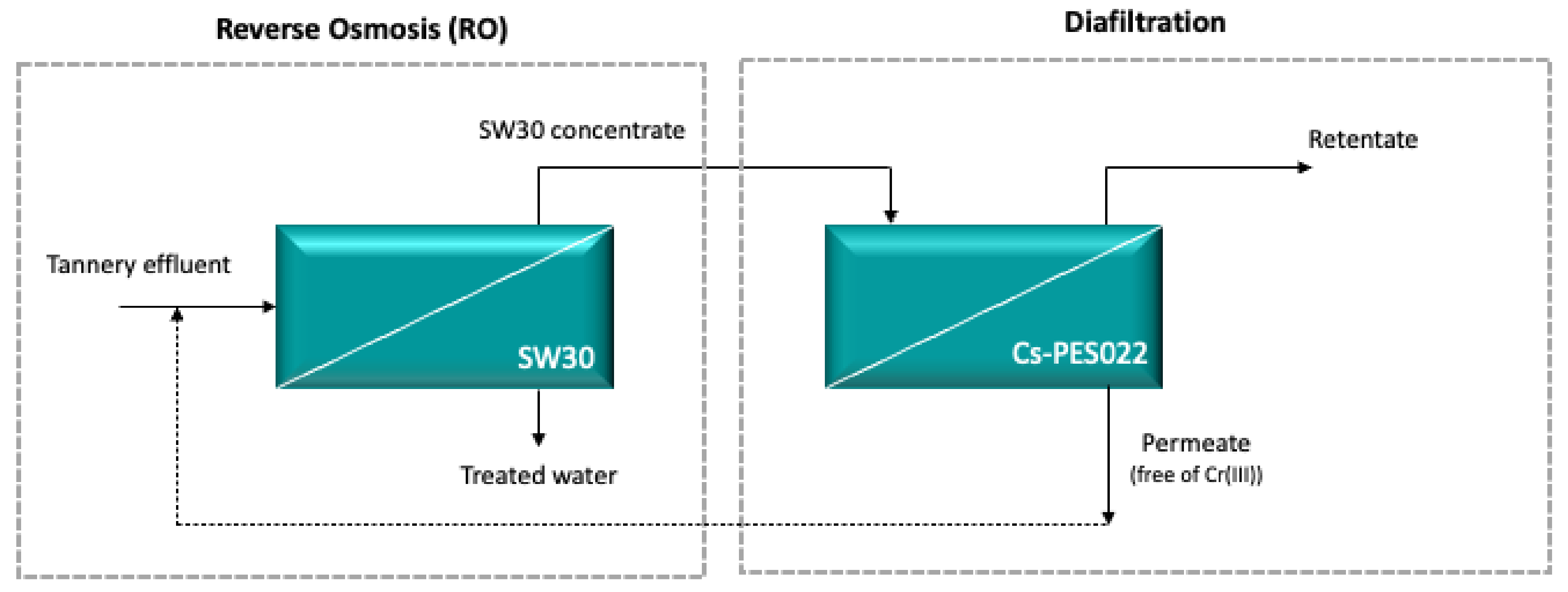
2.2.1. Concentrates of Real and Synthetic Tannery Effluents Obtained from Reverse Osmosis (RO)
2.2.2. Preparation of the Chitosan-Based Membranes (cs-PES MF022)
2.2.3. Diafiltration
2.2.4. Data Treatment
Membrane Permeability
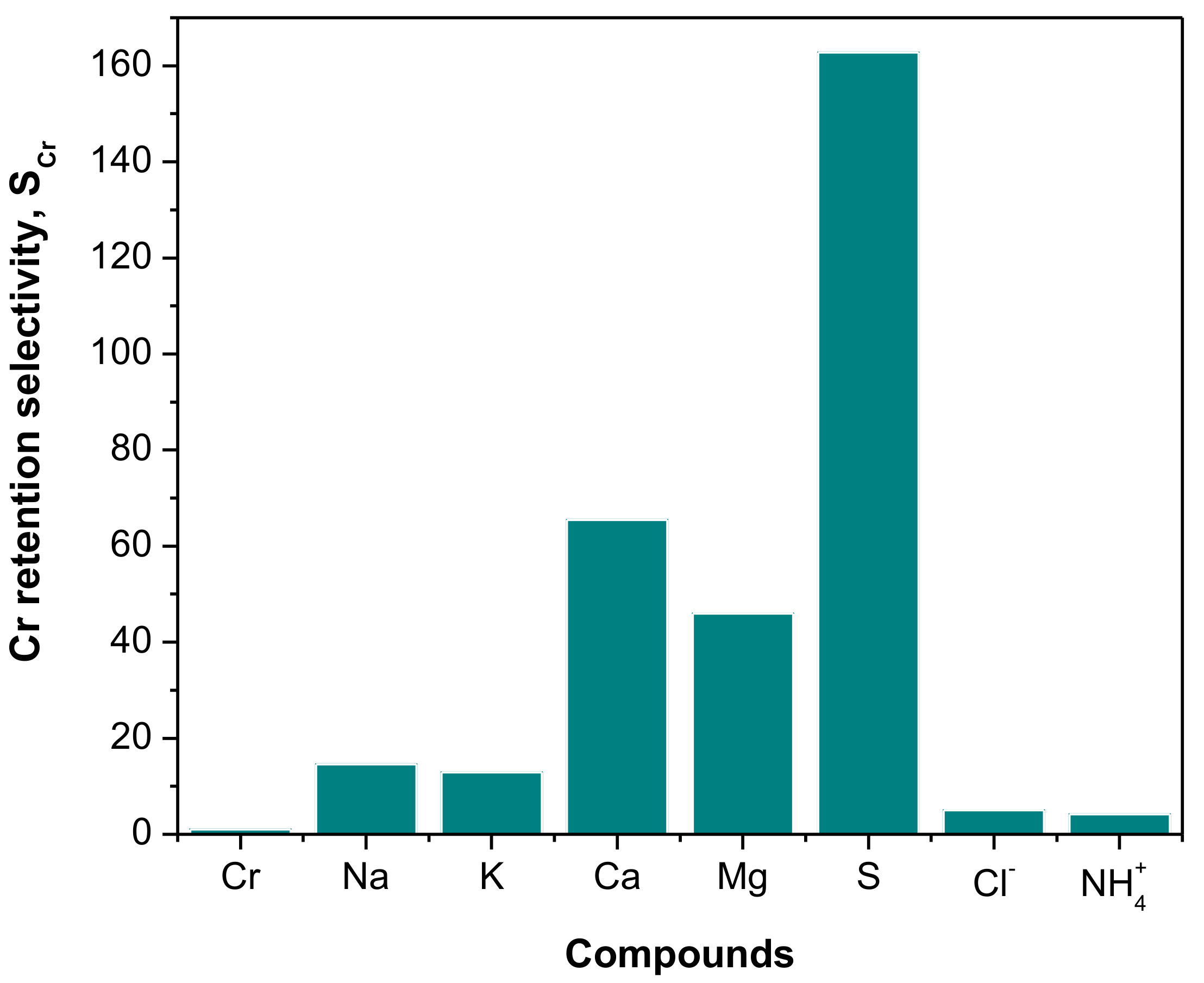
Determination of the Compounds Retained in the Membrane and Cr Retention Selectivity
2.2.5. pH Induced Desorption
2.2.6. Analytical Methods
Quantification of the Compounds
Characterization of Membrane Structure and Chemical Characterization
3. Results and Discussion
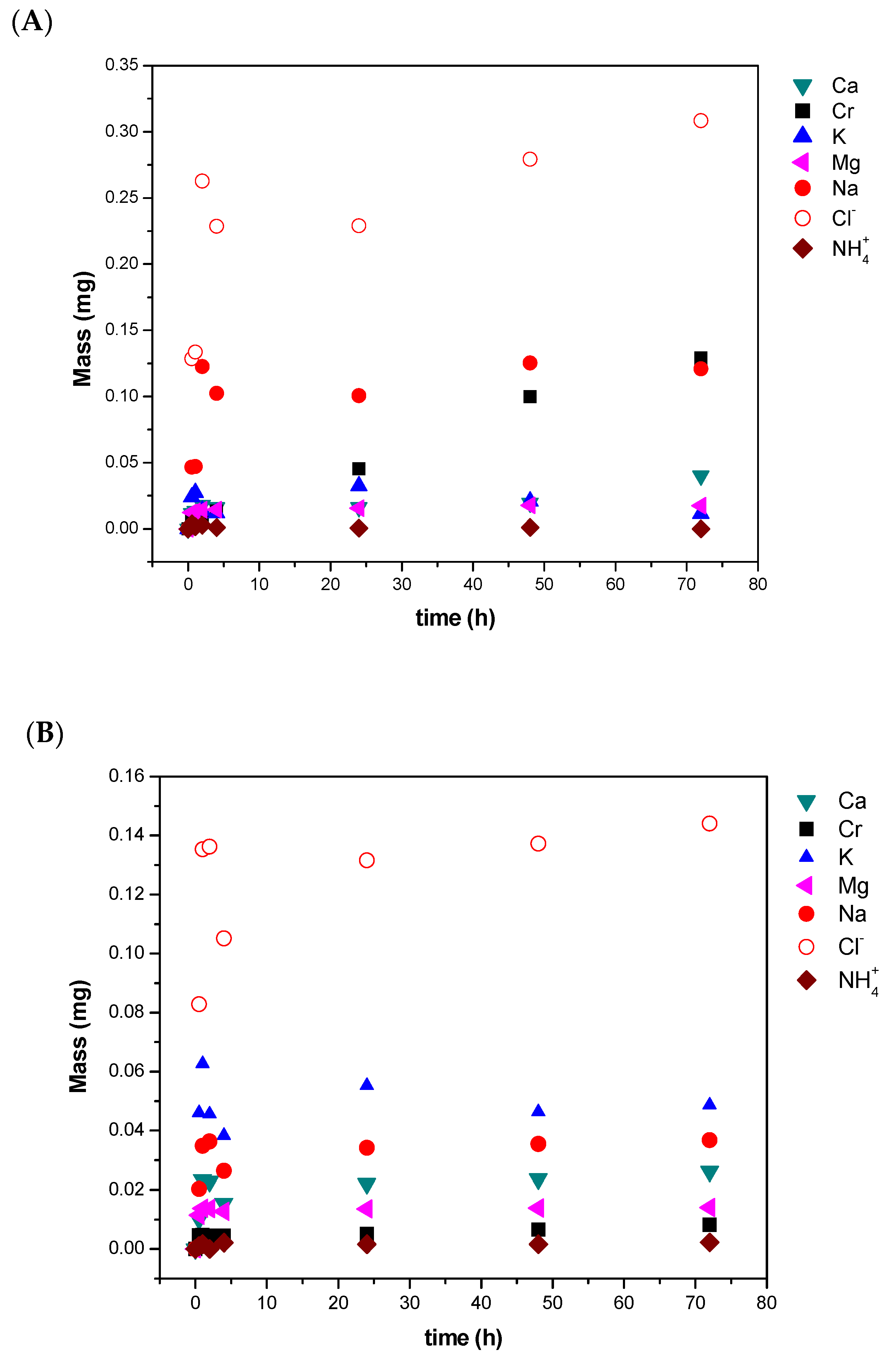
3.1. Diafiltration of the SW30 Concentrate Solution—Synthetic Tannery Effluent
3.2. Diafiltration of the SW30 Concentrate Solution—Real Tannery Effluent
3.3. pH Induced Desorption of Cr from the cs-PES MF022 Membrane
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saxena, G.; Chandra, R.; Bharagava, R.N. Environmental Pollution, Toxicity Profile and Treatment Approaches for Tannery Wastewater and Its Chemical Pollutants. Rev. Environ. Contam. Toxicol. 2016, 240, 31–69. [Google Scholar]
- Vignati, D.A.L.; Ferrari, B.J.D.; Roulier, J.-L.; Coquery, M.; Szalinska, E.; Bobrowski, A.; Czaplicka, A.; Kownacki, A.; Dominik, J. Chromium bioavailability in aquatic systems impacted by tannery wastewaters. Part 1: Understanding chromium accumulation by indigenous chironomids. Sci. Total. Environ. 2019, 653, 401–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Imran, Q.; Hanif, M.A.; Riaz, M.S.; Noureen, S.; Ansari, T.M.; Bhatti, H.N. Coagulation/Flocculation of Tannery Wastewater Using Immobilized Chemical Coagulants. J. Appl. Res. Technol. 2012, 10, 79–86. [Google Scholar]
- Genawi, N.M.; Ibrahim, M.H.; El-Naas, M.H.; Alshaik, A.E. Chromium Removal from Tannery Wastewater by Electrocoagulation: Optimization and Sludge Characterization. Water 2020, 12, 1374. [Google Scholar] [CrossRef]
- Ma, H.-R.; Li, H.; Wu, W.; Qiao, X.-R. Separation of Fe(III) and Cr(III) from tannery sludge bioleachate using organophosphorus acid extractants. Res. Chem. Intermed 2017, 43, 2333–2350. [Google Scholar] [CrossRef]
- Pietrelli, L.; Francolini, I.; Piozzi, A.; Sighicelli, M.; Silvestro, I.; Vocciante, M. Chromium(III) Removal from Wastewater by Chitosan Flakes. Appl. Sci. 2020, 10, 1925. [Google Scholar] [CrossRef] [Green Version]
- Angelucci, D.M.; Stazi, V.; Andrew, J.; Daugulis, A.J.; Tomei, M.C. Treatment of Synthetic Tannery Wastewater in a Continuous Two-Phase Partitioning Bioreactor: Biodegradation of the Organic Fraction and Chromium Separation. J. Clean. Prod. 2017, 152, 321. [Google Scholar] [CrossRef]
- Kavouras, P.; Pantazopoulou, E.; Varitis, S.; Vourlias, G.; Chrissafis, K.; Dimitrakopulos, G.P.; Mitrakas, M.; Zouboulis, A.I.; Karakostas, T.; Xenidis, A. Incineration of Tannery Sludge under Oxic and Anoxic Conditions: Study of Chromium Speciation. J. Hazard. Mater. 2015, 283, 672–679. [Google Scholar] [CrossRef]
- Kiliç, E.; Font, J.; Puig, R.; Çolak, S.; Çelik, D. Chromium Recovery from Tannery Sludge with Saponin and Oxidative Remediation. J. Hazard. Mater. 2011, 185, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Roca, J.A.; Galiana-Aleixandre, M.V.; Lora-García, J.; Bes-Piá, A. Purification of Tannery Effluents by Ultrafiltration in View of Permeate Reuse. Sep. Purif. Technol. 2010, 70, 296–301. [Google Scholar] [CrossRef]
- Religa, P.; Kowalik, A.; Gierycz, P. A new approach to chromium concentration from salt mixture solution using nanofiltration. Sep. Purif. Technol. 2011, 82, 114–120. [Google Scholar] [CrossRef]
- Zakmout, A.; Sadi, F.; Portugal, C.A.M.; Crespo, J.G.; Velizarov, S. Tannery Effluent Treatment by Nanofiltration, Reverse Osmosis and Chitosan Modified Membranes. Membranes 2020, 10, 378. [Google Scholar] [CrossRef]
- Vinduja, V.; Balasubramanian, N. Electrocoagulation-integrated hybrid membrane processes for the treatment of tannery wastewater. Environ. Sci. Pollut. Res. 2013, 20, 7441–7449. [Google Scholar]
- Deghles, A.; Kurt, U. Treatment of tannery wastewater by a hybrid electrocoagulation/ electrodialysis process. Chem. Eng. Process. 2016, 104, 43–50. [Google Scholar] [CrossRef]
- Dasgupta, J.; Mondal, D.; Chakraborty, S.; Sikder, J.; Curcio, S.; Arafat, H.A. Nanofiltration Based Water Reclamation from Tannery Effluent Following Coagulation Pretreatment. Ecotoxicol. Environ. Saf. 2015, 121, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Ashour, E.A.; Tony, M.A. Eco-friendly removal of hexavalent chromium from aqueous solution using natural clay mineral: Activation and modification effects. SN Appl. Sci. 2020, 2, 2042S. [Google Scholar]
- Mustapha, S.; Ndamitso, M.M.; Abdulkareem, A.S.; Tijani, J.O.; Mohammed, A.K.; Shuaib, D.T. Potential of using kaolin as a natural adsorbent for the removal of pollutants from tannery wastewater. Heliyon 2019, 5, e029234. [Google Scholar] [CrossRef]
- Tammaro, M.; Salluzzo, A.; Perfetto, R.; Lancia, A. A comparative evaluation of biological activated carbon and activated sludge processes for the treatment of tannery wastewater. J. Environ. Chem. Eng. 2014, 2, 1445–1455. [Google Scholar] [CrossRef]
- Ahmad, W.; Qaiser, S.; Ullah, R.; Jan, B.M.; Karakassides, M.A.; Salmas, C.E.; Ikram, G.K.R. Utilization of Tires Waste-Derived Magnetic-Activated Carbon for the Removal of Hexavalent Chromium from Wastewater. Materials 2021, 14, 34. [Google Scholar]
- Ihsanullah; Al-Khaldi, F.A.; Abu-Sharkh, B.; Abulkibash, A.M.; Qureshi, M.I.; Laoui, T.; Atieh, M.A. Effect of acid modification on adsorption of hexavalent chromium (Cr(VI)) from aqueous solution by activated carbon and carbon nanotubes. Desalination Water Treat. 2016, 57, 7232–7244. [Google Scholar] [CrossRef]
- Biswas, S.; Rashid, T.U.; Debnath, T.; Haque, P.; Rahman, M.M. Application of Chitosan-Clay Biocomposite Beads for Removal of Heavy Metal and Dye from Industrial Effluent. J. Compos. Sci. 2020, 4, 16. [Google Scholar] [CrossRef] [Green Version]
- Horzum, N.; Boyacı, E.; Eroglu, A.E.; Shahwan, T.; Demir, M.M. Sorption Efficiency of Chitosan Nanofibers toward Metal Ions at Low Concentrations. Biomacromolecules 2010, 11, 3301–3308. [Google Scholar] [CrossRef] [Green Version]
- Baroni, P.; Vieira, R.S.; Meneghetti, E.; da Silva, M.G.C.; Beppu, M.M. Evaluation of batch adsorption of chromium ions on natural and crosslinked chitosan membranes. J. Hazard. Mater. 2008, 152, 1155–1163. [Google Scholar] [CrossRef] [PubMed]
- Mirabedini, M.; Kassaee, M.Z.; Poorsadeghi, S. Novel Magnetic Chitosan Hydrogel Film, Cross-Linked with Glyoxal as an Efficient Adsorbent for Removal of Toxic Cr (VI) From Water. Arab. J. Sci. Eng. 2017, 42, 115–124. [Google Scholar] [CrossRef]
- Karaer, H.; Kaya, İ. Synthesis, characterization and using at the copper adsorption of chitosan/polyvinyl alcohol magnetic composite. J. Mol. Liq. 2017, 230, 152–162. [Google Scholar] [CrossRef]
- Salehi, E.; Madaeni, S.S.; Rajabi, L.; Derakhshan, A.A.; Daraei, S.; Vatanpour, V. Static and dynamic adsorption of copper ions on chitosan/polyvinyl alcohol thin adsorptive membranes: Combined effect of polyethylene glycol and aminated multi-walled carbon nanotubes. Chem. Eng. J. 2013, 215–216, 791–801. [Google Scholar] [CrossRef]
- Juang, R.-S.; Shiau, R.-C. Metal removal from aqueous solutions using chitosan-enhanced membrane filtration. J. Membr. Sci. 2000, 165, 159–167. [Google Scholar] [CrossRef]
- Li, L.; Li, Y.; Yang, C. Chemical filtration of Cr (VI) with electrospun chitosan nanofiber membranes. Carbohydr. Polym. 2016, 140, 299–307. [Google Scholar] [CrossRef]
- Desai, K.; Kit, K.; Li, J.; Davidson, P.M.; Zivanovic, S.; Meyer, H. Nanofibrous chitosan non-wovens for filtration applications. Polymer 2009, 50, 3661–3669. [Google Scholar] [CrossRef]
- Ghaee, A.; Shariaty-Niassar, M.; Barzin, J.; Matsuura, T.; Ismail, A.F. Preparation of chitosan/cellulose acetate composite nanofiltration membrane for wastewater treatment. Desalination Water Treat. 2015, 57, 14453–14460. [Google Scholar] [CrossRef]
- Căprărescu, S.; Zgârian, R.G.; Tihan, G.T.; Purcar, V.; Totu, E.E.; Modrogan, C.; Chiriac, A.L.; Nicolae, C.A. Biopolymeric Membrane Enriched with Chitosan and Silver for Metallic Ions Removal. Polymers 2020, 12, 1792. [Google Scholar] [CrossRef]
- Wang, L.; Yang, G.; Xing, W.; Xu, N. Mathematic model of the yield for diafiltration processes. Sep. Purif. Technol. 2008, 59, 206–213. [Google Scholar] [CrossRef]
- Veigas, B.; Portugal, C.A.M.; Valério, R.; Fortunato, E.; Crespo, J.G.; Baptista, P.V. Scalable approach for the production of functional DNA based gold nanoprobes. J. Membr. Sci. 2015, 492, 528–535. [Google Scholar] [CrossRef]
- Sabourian, P.; Tavakolian, M.; Yazdani, H.; Frounchi, M.; van de Ven Theo, G.M.; Maysinger, D.; Kakkar, A. Stimuli-responsive chitosan as an advantageous platform for efficient delivery of bioactive agents. J. Control. Release 2020, 317, 216–231. [Google Scholar] [CrossRef] [PubMed]
- Executive Decree, No. 06-141 of 20 Rabie El Aouel 1427 Corresponding to 19 April 2006, Official Journal of the Algerian Republic N 26. Available online: https://and.dz/site/wp-content/uploads/D%C3%A9cret-executif-n%C2%B0-06-141.pdf (accessed on 1 July 2021).
- Fikar, M.; Kovács, Z.; Czermak, P. Dynamic optimization of batch diafiltration processes. J. Membr. Sci. 2010, 355, 168–174. [Google Scholar] [CrossRef] [Green Version]
- Chemical Equilibrium Diagrams. Available online: https://sites.google.com/site/chemdiagr/ (accessed on 1 July 2021).
- Shao, J.; Zydney, A.L. Optimization of Ultrafiltration/Diafiltration Processes for Partially Bound Impurities. Biotechnol. Bioeng. 2004, 87, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Vinodhini, P.A.; Sudha, P.N. Removal of heavy metal chromium from tannery effluent using ultrafiltration membrane. Text. Cloth. Sustain. 2016, 2, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Xu, X.; Yue, C.; Song, L.; Lv, Y.; Liu, F.; Li, A. Insight into the efficient co-removal of Cr(VI) and Cr(III) by positively charged UiO-66-NH2 decorated ultrafiltration membrane. Chem. Eng. J. 2021, 404, 126546. [Google Scholar] [CrossRef]


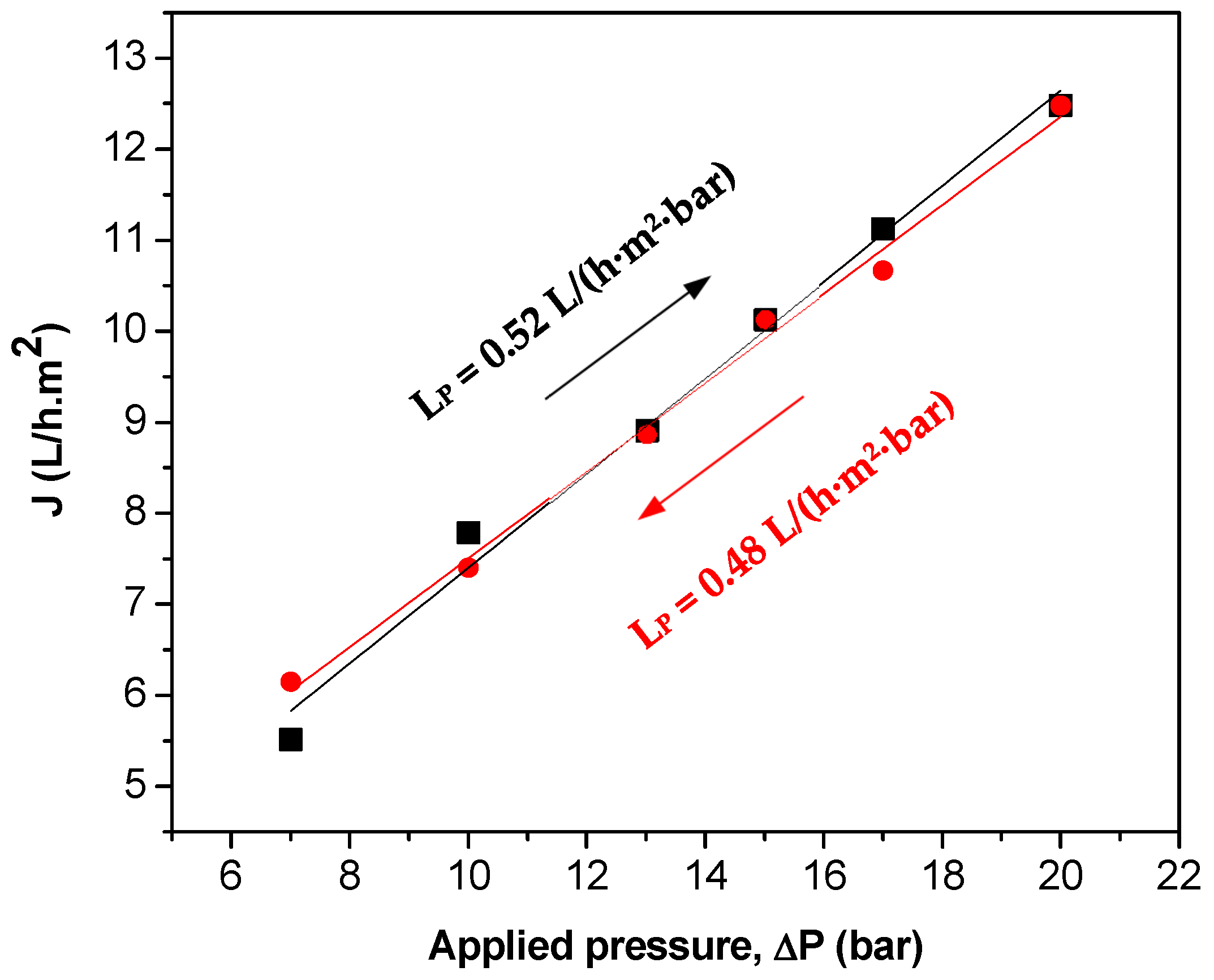
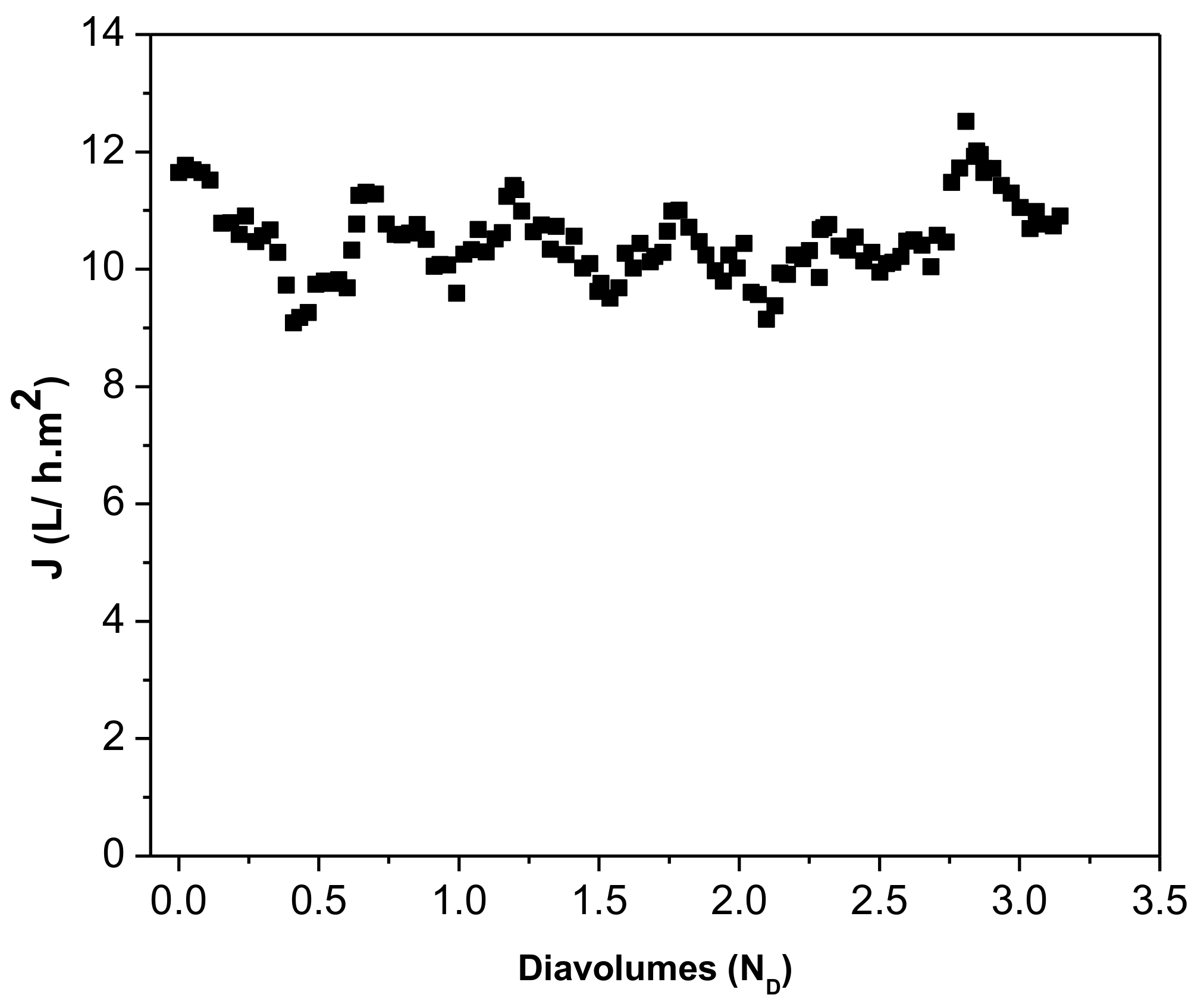
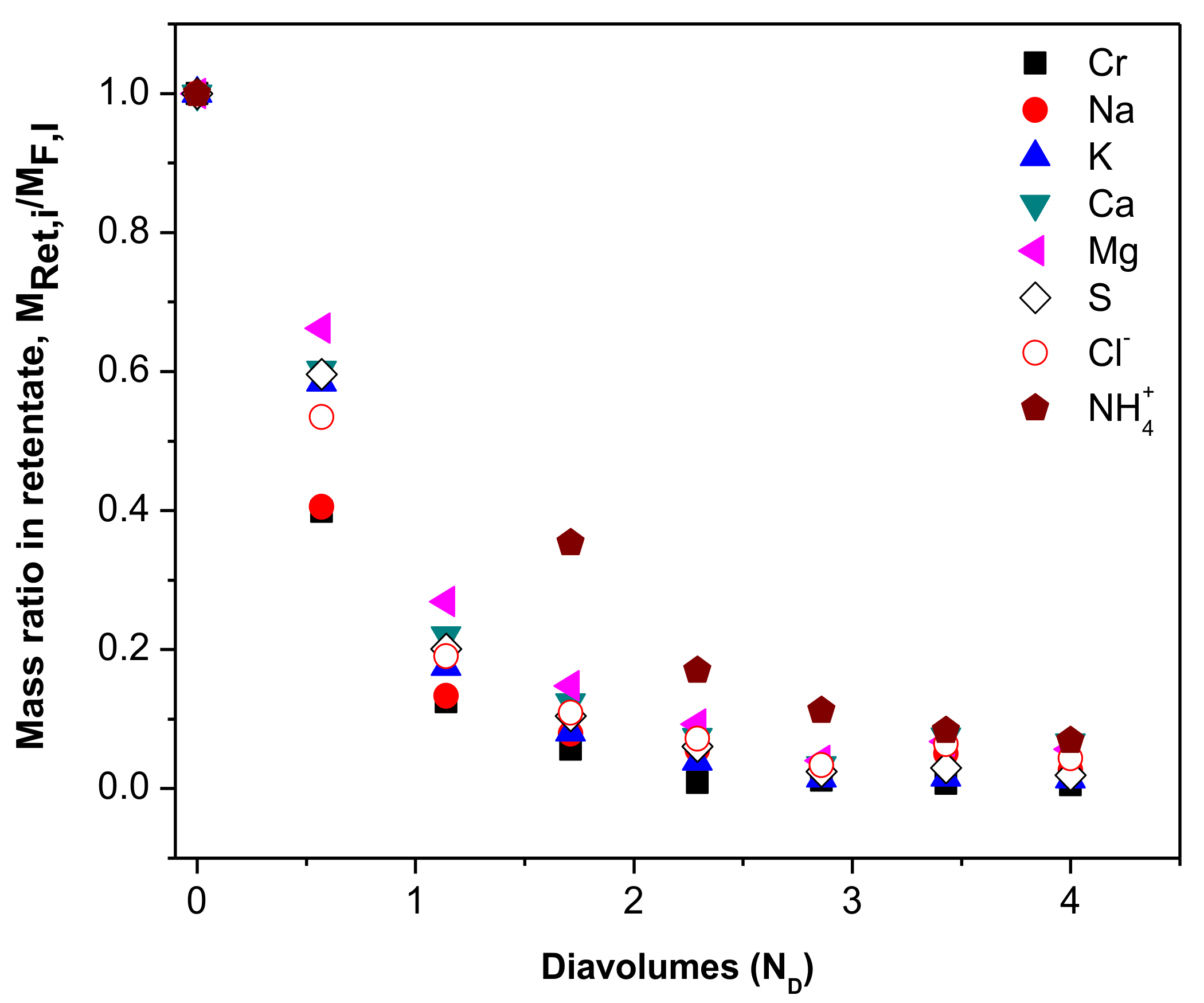

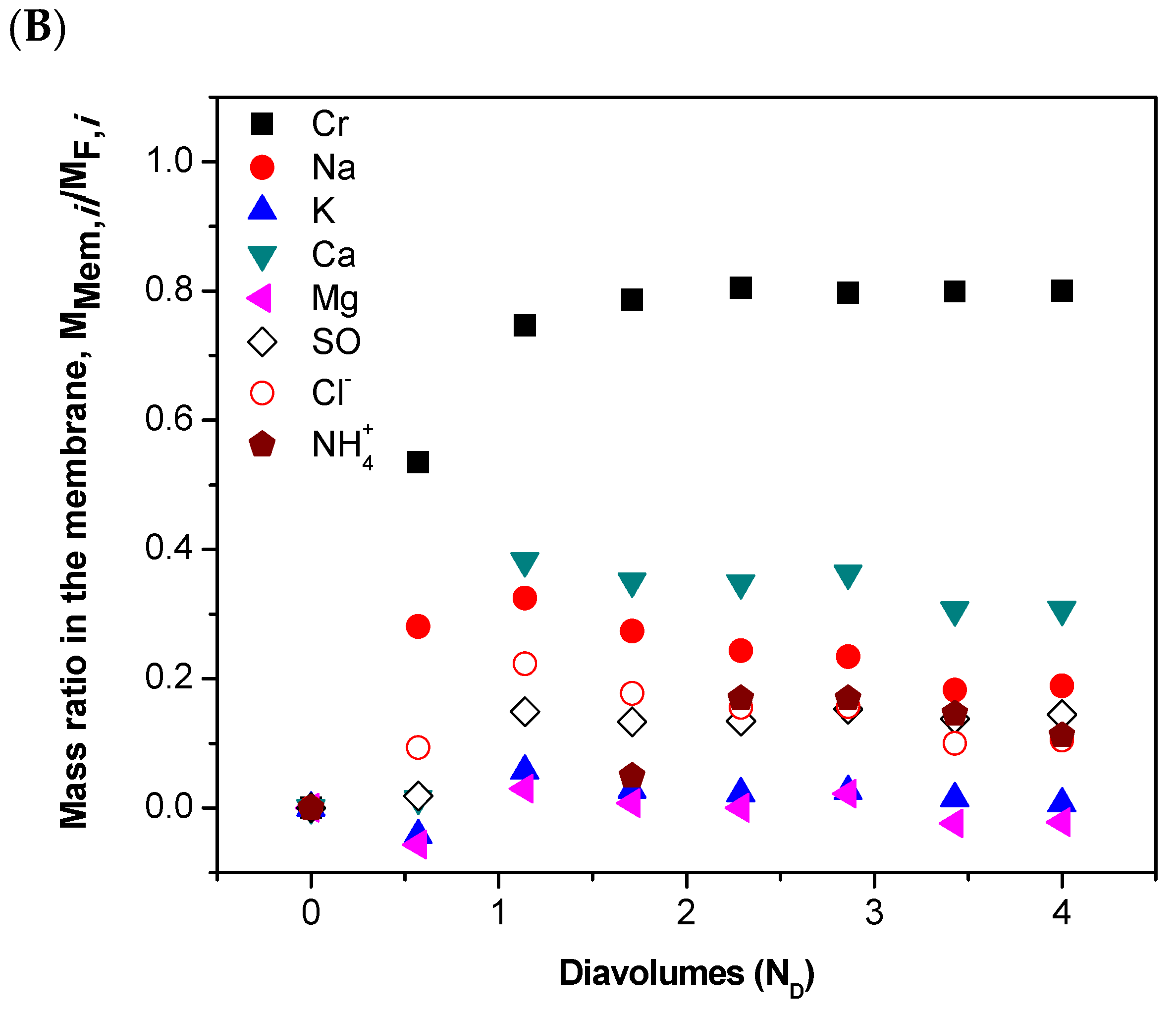
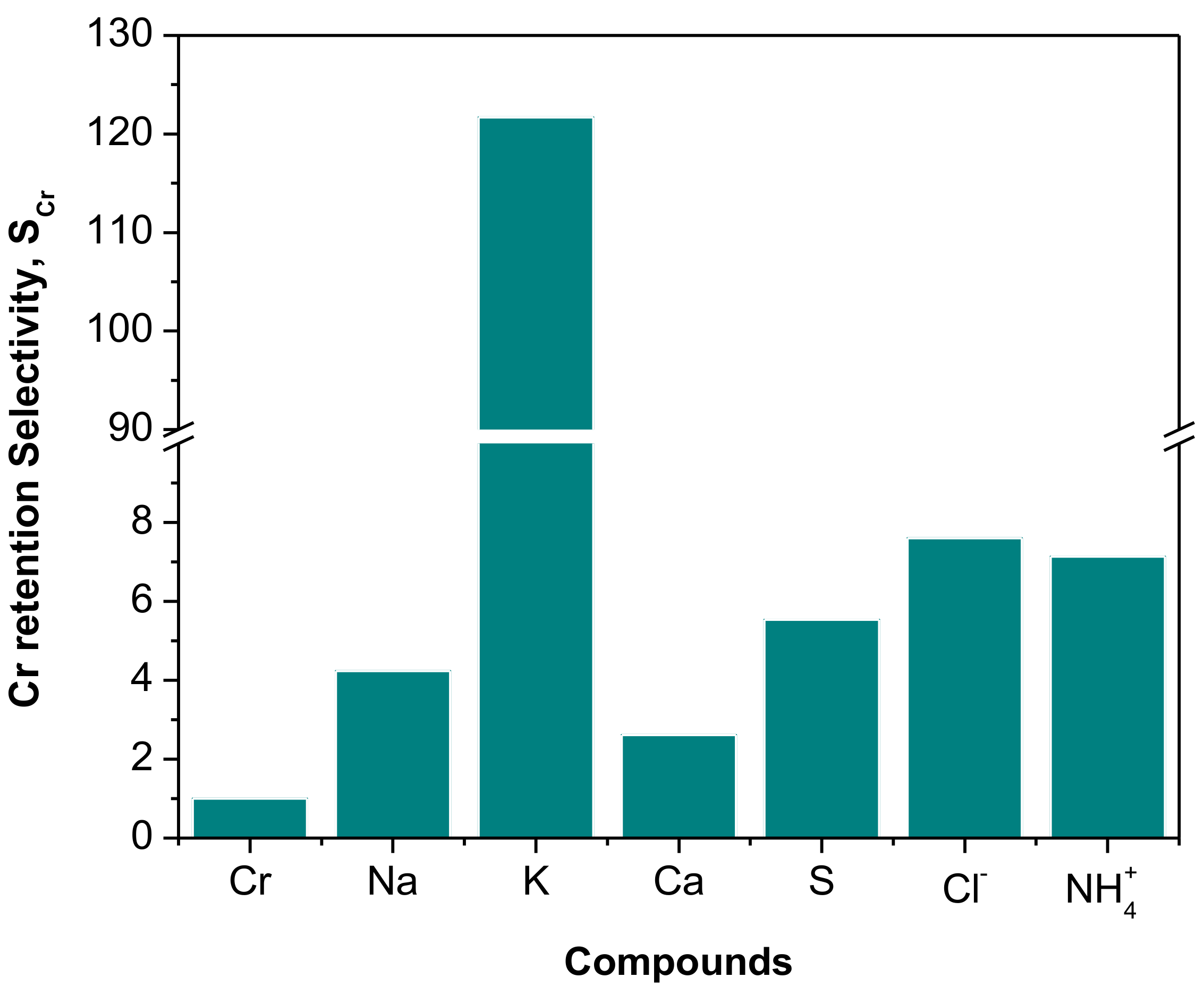
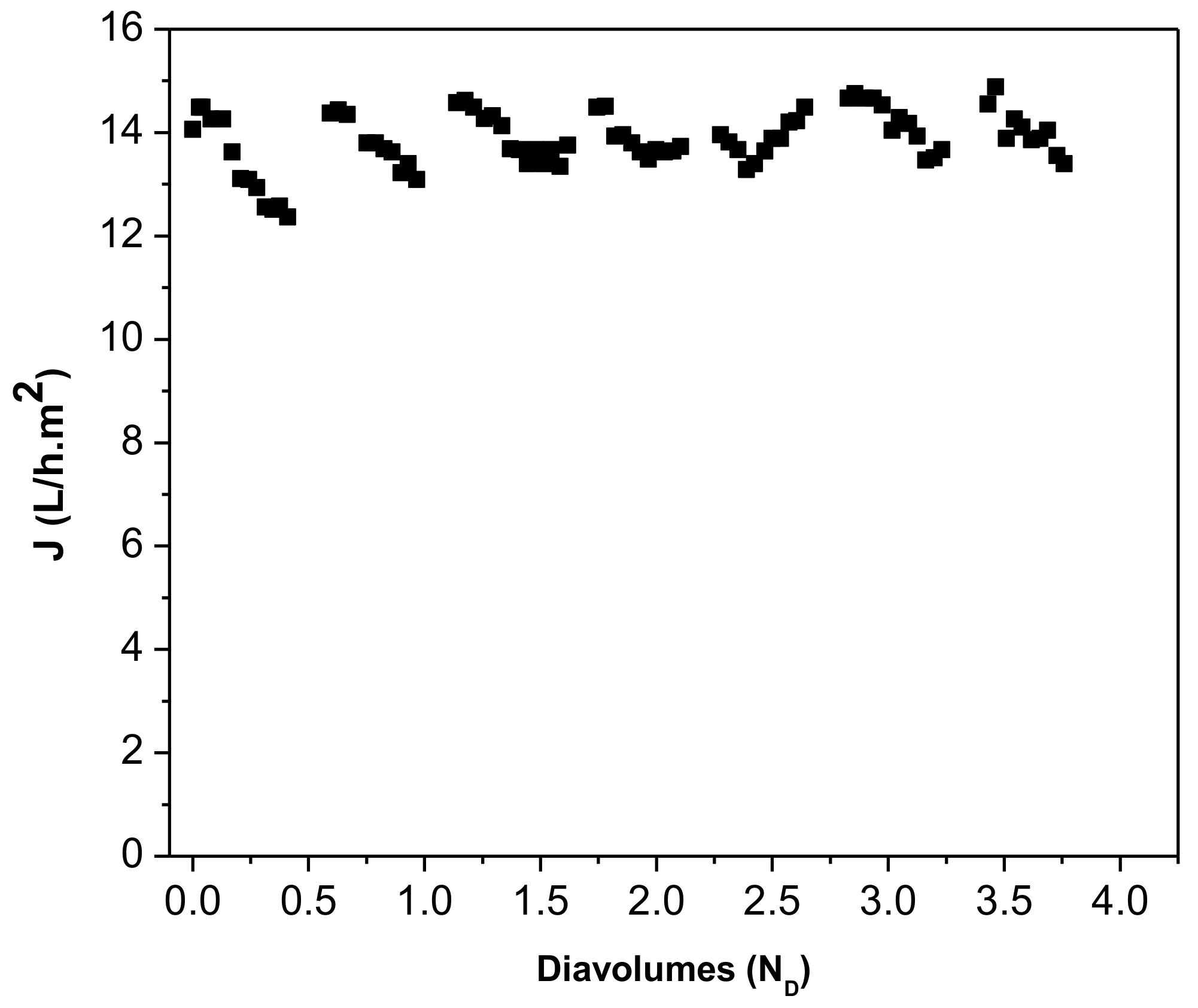
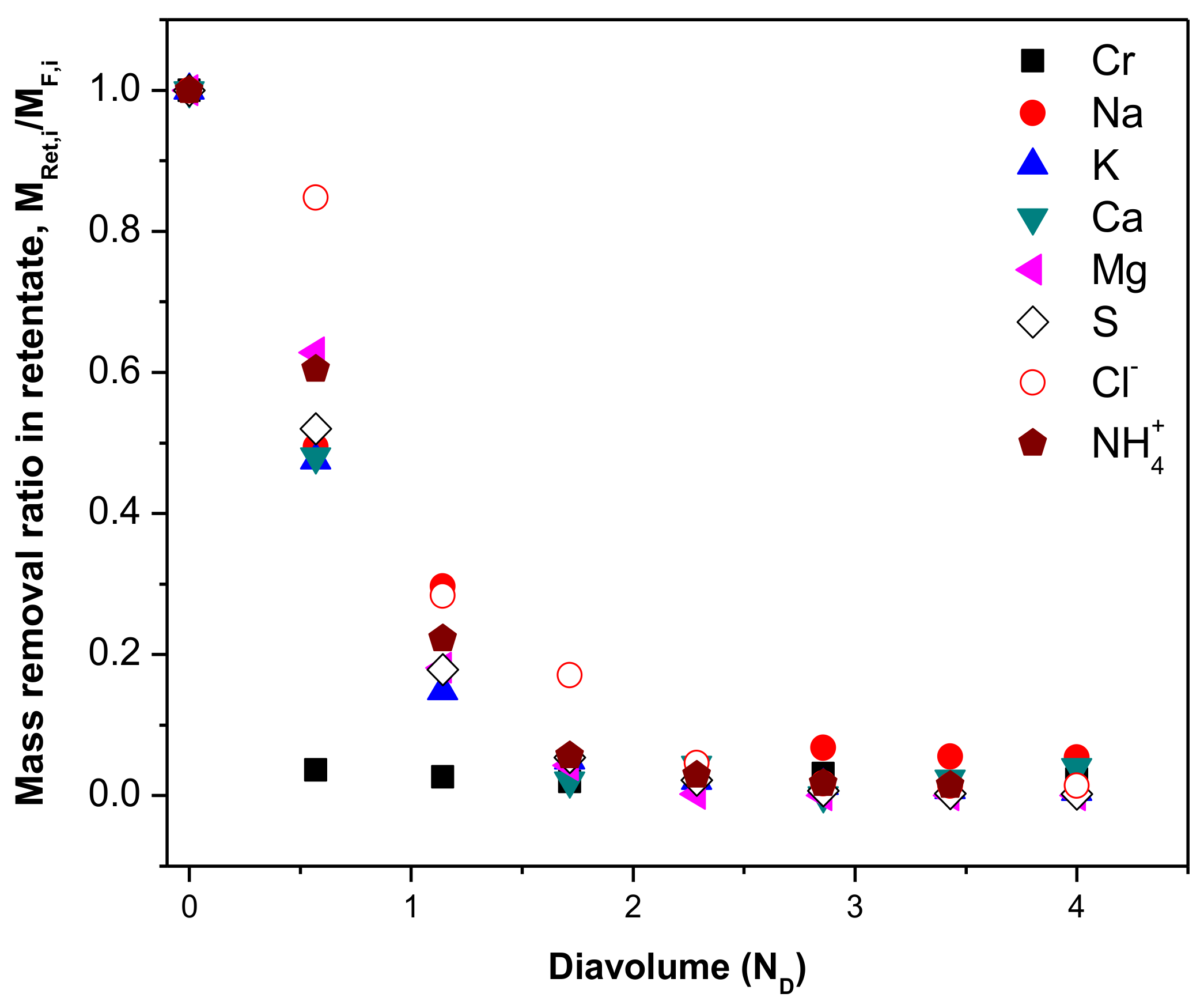

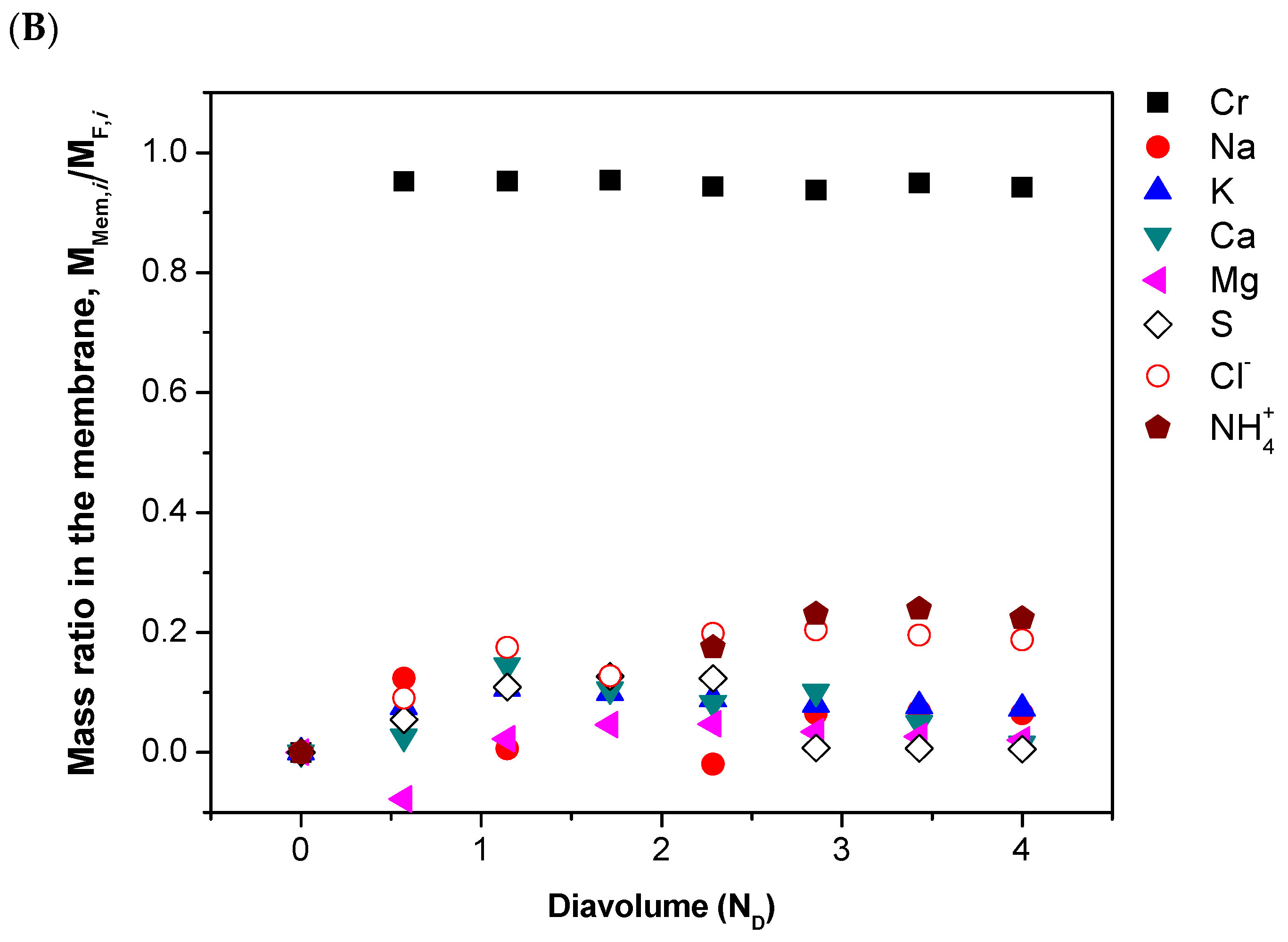
| Parameter 1 | Ca | Cr(III) | K | Mg | Na | S | Cl− | NH4+ |
|---|---|---|---|---|---|---|---|---|
| Unit | g/L | g/L | mg/L | g/L | g/L | g/L | g/L | g/L |
| Value | 0.64 | 0.18 | 0.76 | 0.33 | 2.18 | 0.88 | 5.01 | 0.15 |
| Paramete 1 | Ca | Cr(III) | K | Mg | Na | S | Cl− | NH4+ |
|---|---|---|---|---|---|---|---|---|
| Unit | g/L | g/L | g/L | g/L | g/L | g/L | g/L | g/L |
| Value | 0.14 | 0.12 | 2.77 | 2.80 | 1.17 | 2.09 | 28.67 | 0.30 |
| Compounds 1 | a | R2 |
|---|---|---|
| Cr | 1.69 ± 0.04 | 0.999 |
| Na | 1.60 ± 0.08 | 0.993 |
| K | 1.26 ± 0.10 | 0.984 |
| Ca | 1.11 ± 0.08 | 0.982 |
| Mg | 0.99 ± 0.07 | 0.982 |
| S | 1.18 ± 0.08 | 0.987 |
| Cl− | 1.24 ± 0.07 | 0.990 |
| NH4+ | 0.70 ± 0.03 | 0.993 |
| Compounds 1 | a | R2 |
|---|---|---|
| Cr | 5.85 ± 1.06 | 0.996 |
| Na | 1.13 ± 0.07 | 0.993 |
| K | 1.52 ± 0.07 | 0.995 |
| Ca | 1.45 ± 0.12 | 0.991 |
| Mg | 1.29 ± 0.15 | 0.968 |
| S | 1.41 ± 0.08 | 0.992 |
| Cl− | 0.93 ± 0.15 | 0.923 |
| NH4+ | 1.24± 0.11 | 0.980 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zakmout, A.; Sadi, F.; Velizarov, S.; Crespo, J.G.; Portugal, C.A.M. Recovery of Cr(III) from Tannery Effluents by Diafiltration Using Chitosan Modified Membranes. Water 2021, 13, 2598. https://doi.org/10.3390/w13182598
Zakmout A, Sadi F, Velizarov S, Crespo JG, Portugal CAM. Recovery of Cr(III) from Tannery Effluents by Diafiltration Using Chitosan Modified Membranes. Water. 2021; 13(18):2598. https://doi.org/10.3390/w13182598
Chicago/Turabian StyleZakmout, Asmaa, Fatma Sadi, Svetlozar Velizarov, João G. Crespo, and Carla A. M. Portugal. 2021. "Recovery of Cr(III) from Tannery Effluents by Diafiltration Using Chitosan Modified Membranes" Water 13, no. 18: 2598. https://doi.org/10.3390/w13182598
APA StyleZakmout, A., Sadi, F., Velizarov, S., Crespo, J. G., & Portugal, C. A. M. (2021). Recovery of Cr(III) from Tannery Effluents by Diafiltration Using Chitosan Modified Membranes. Water, 13(18), 2598. https://doi.org/10.3390/w13182598







