Carbamazepine Levels Related to the Demographic Indicators in Groundwater of Densely Populated Area
Abstract
:1. Introduction
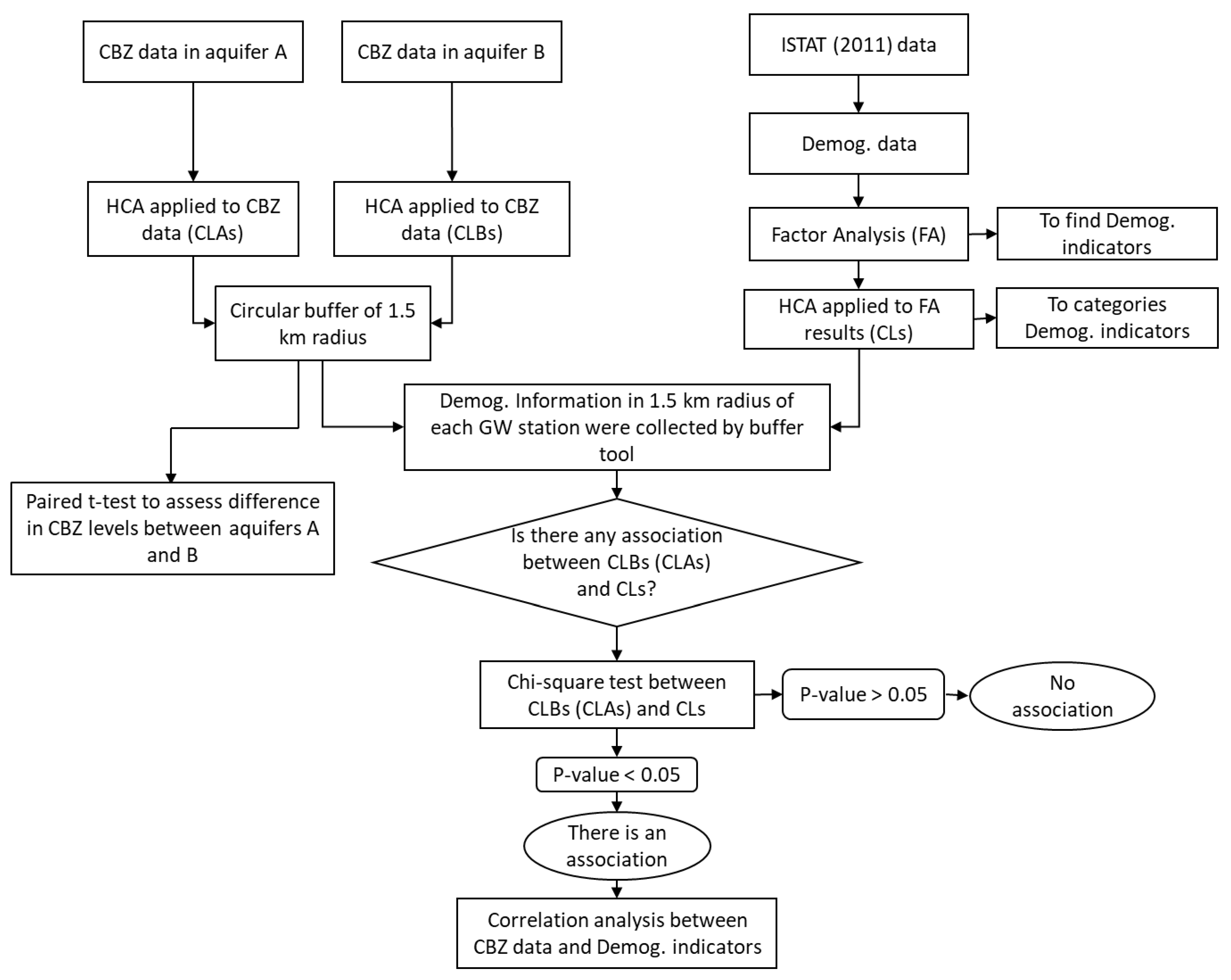
2. Materials and Methods
2.1. Study Area
Hydrogeological Study
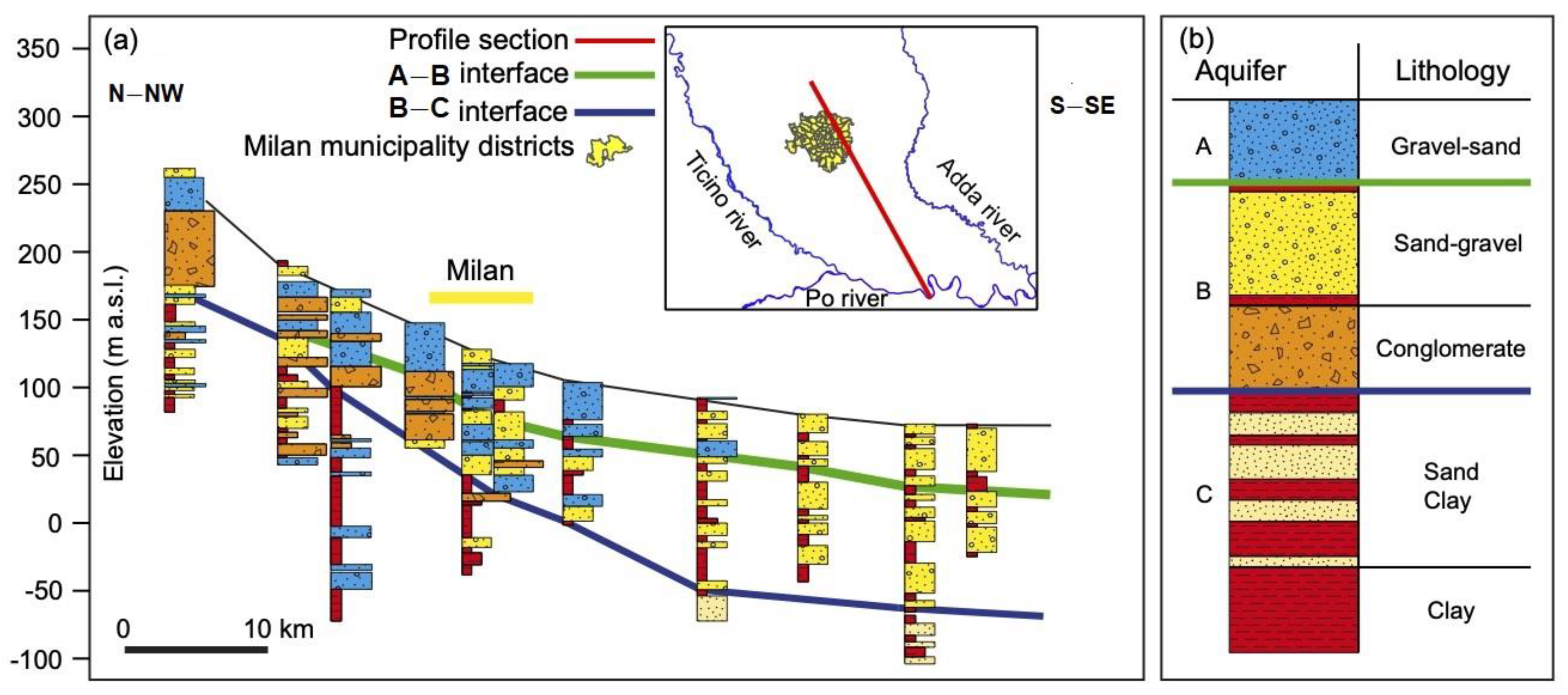
2.2. Input Data
2.2.1. Groundwater Samples
2.2.2. Analytical Method
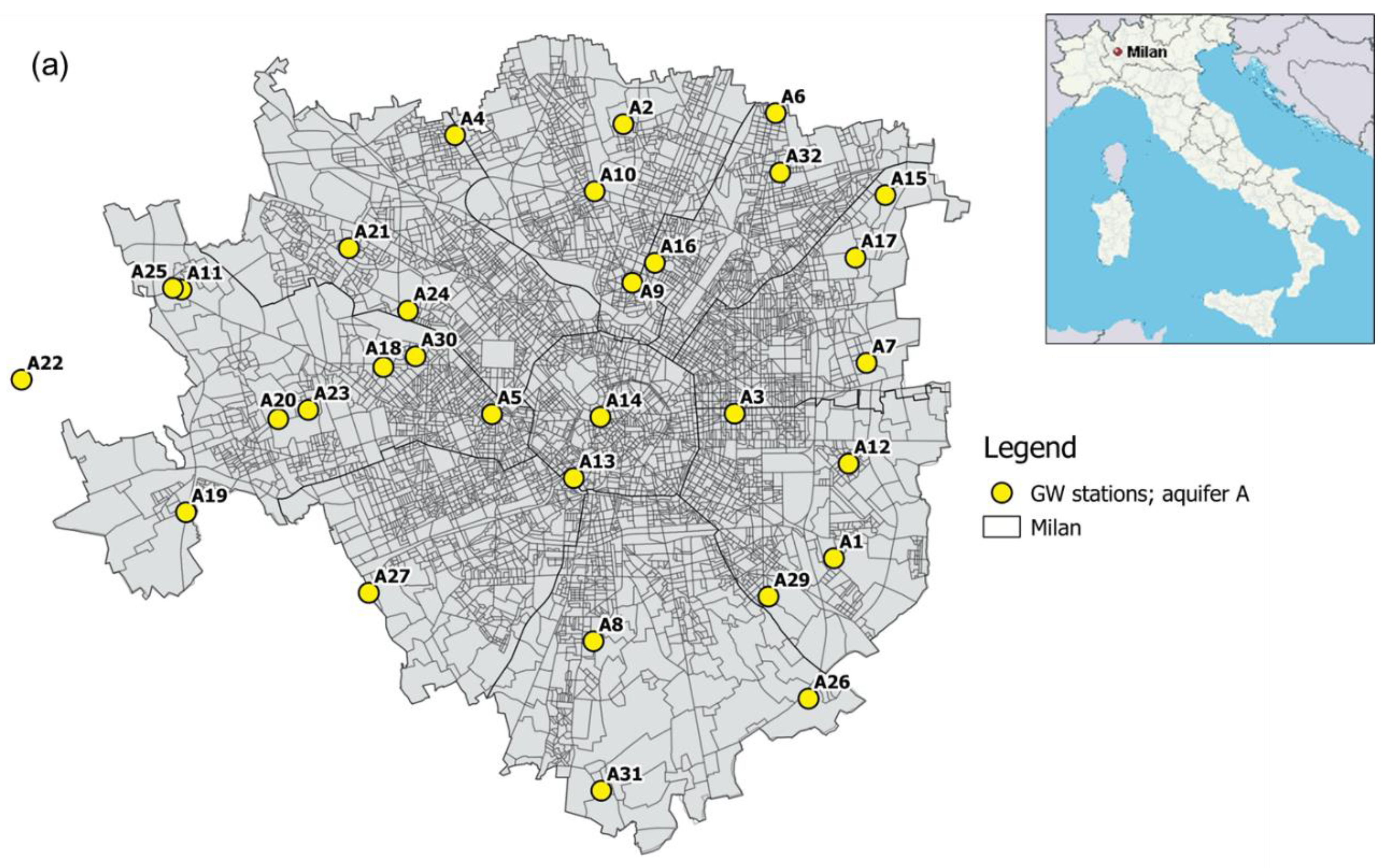
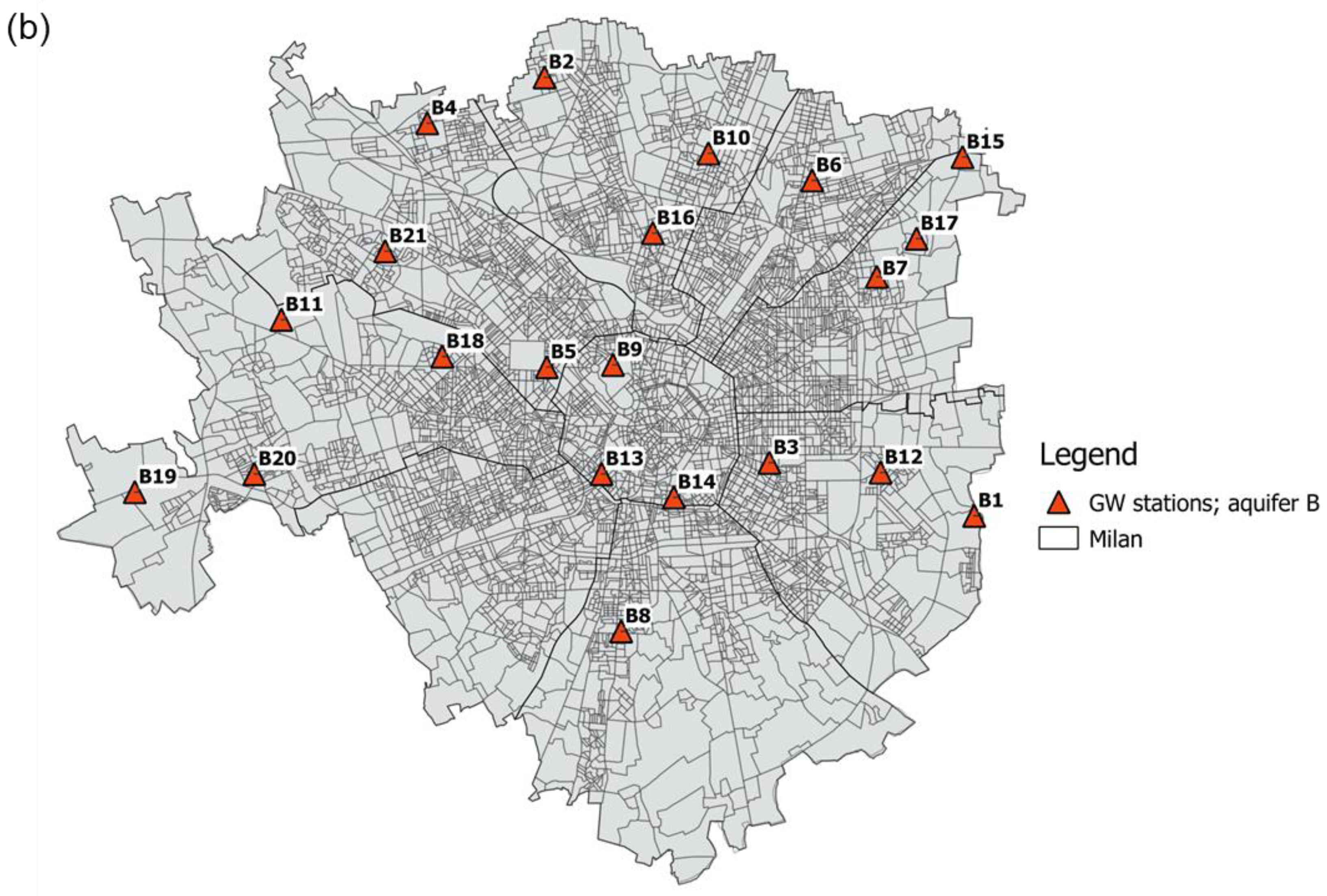
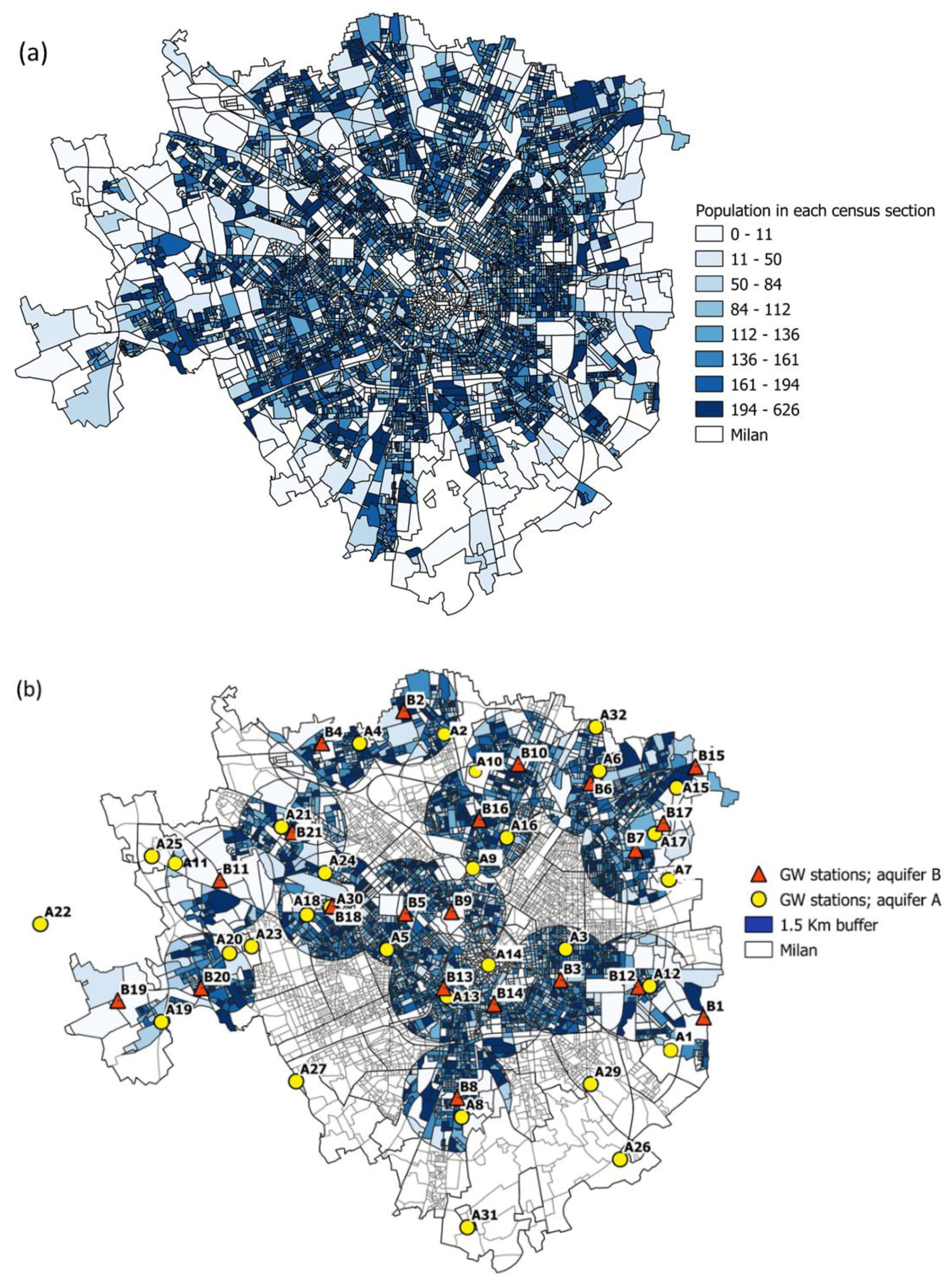
2.2.3. Demographic Variables
2.3. Statistical Analysis
3. Results
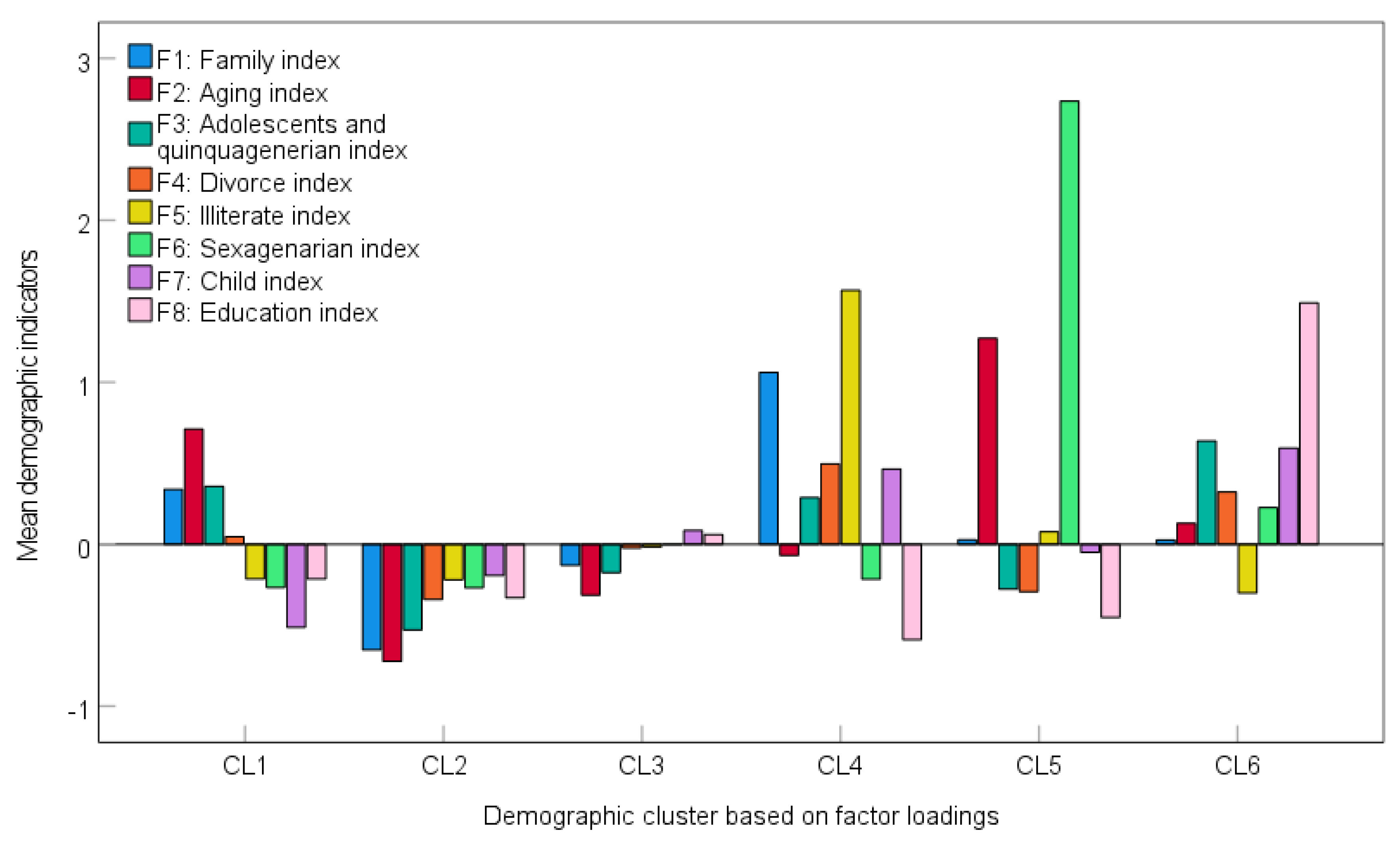
3.1. Factor Analysis Applied to the Demographic Variables
3.2. Hierarchical Cluster Analysis (HCA) Applied to Eight Factor Loadings
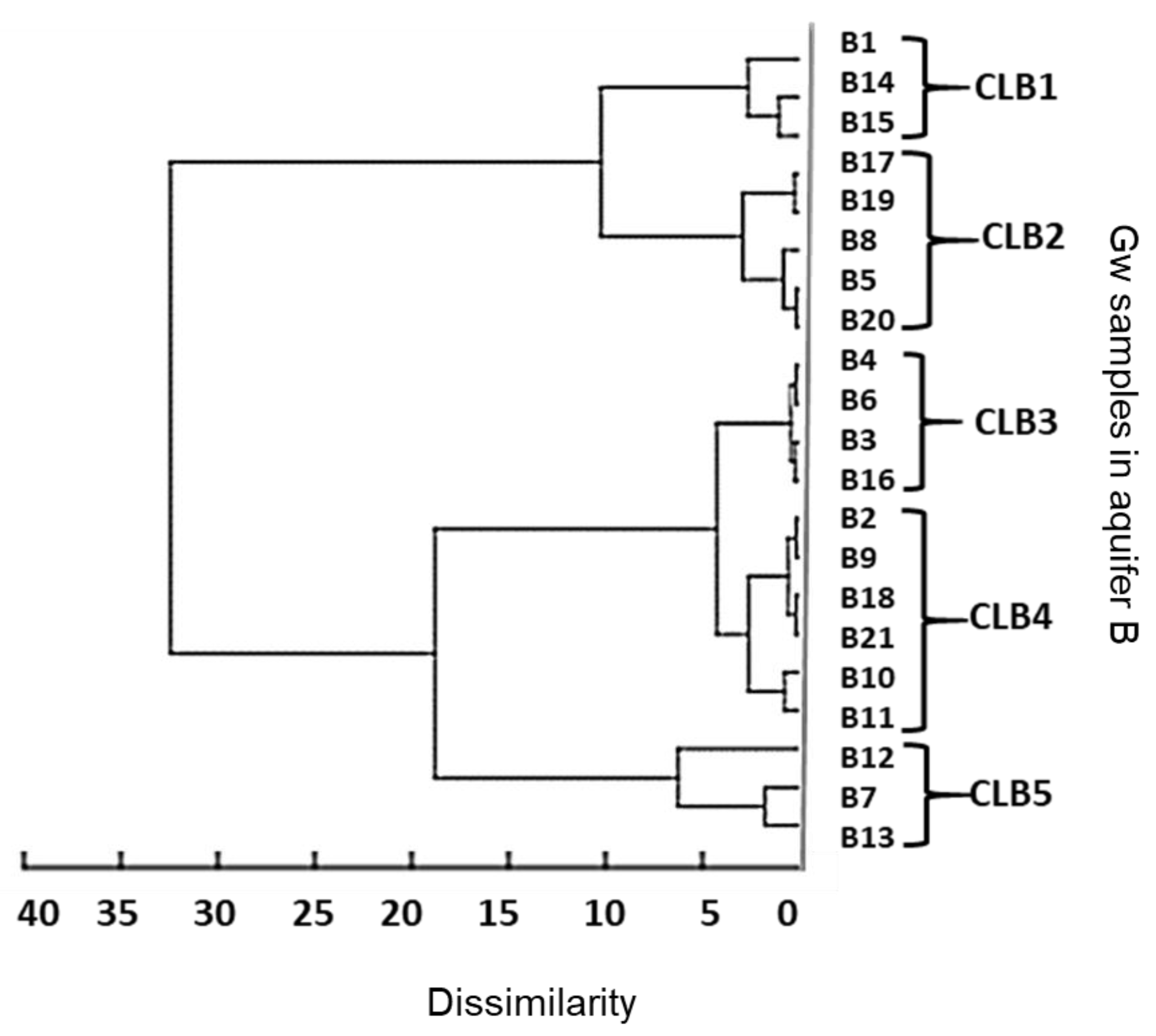
3.3. Statistical Analysis of CBZ Levels in Aquifer B
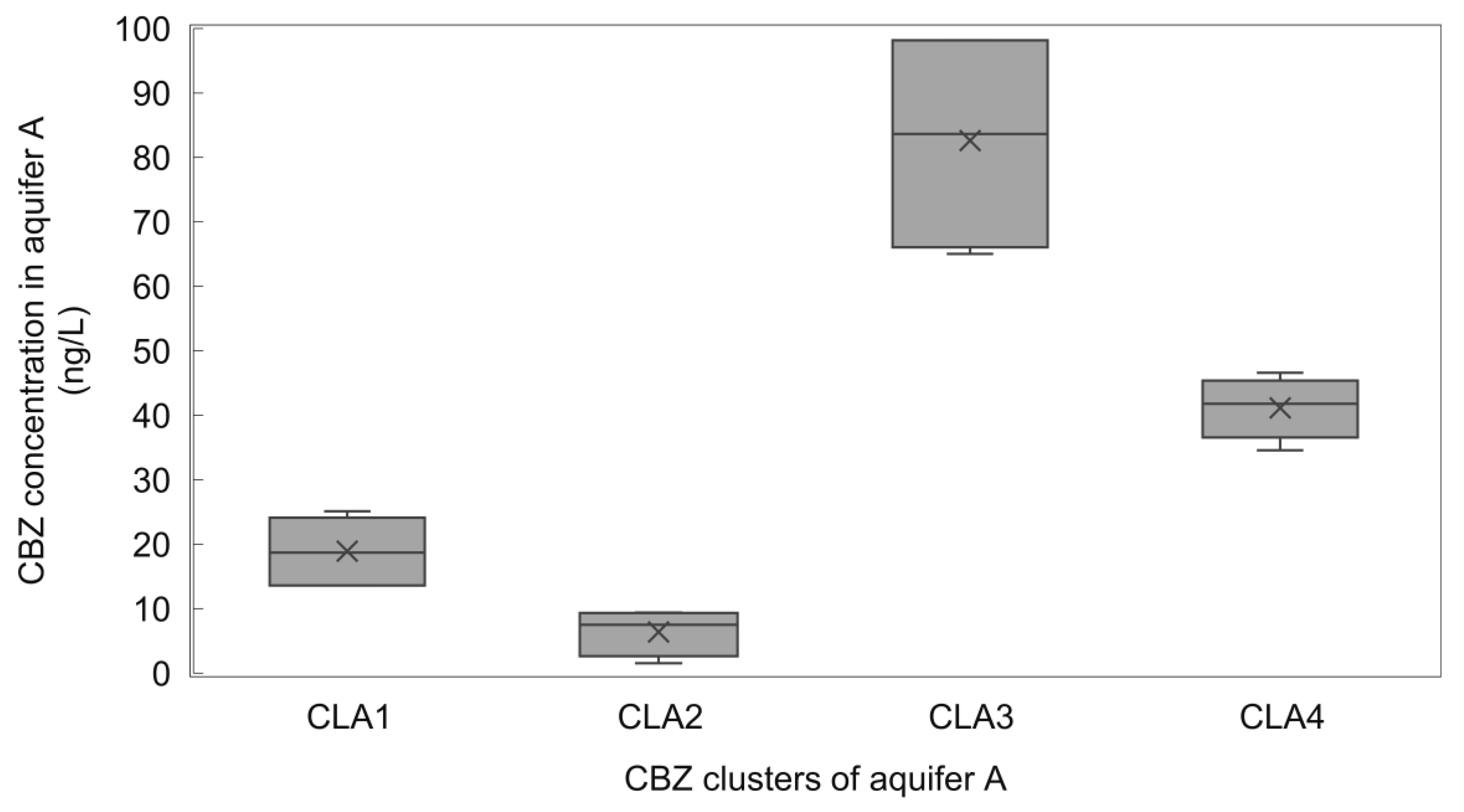
3.4. Statistical Analysis of CBZ Levels in Aquifer A
4. Discussion
5. Conclusions
- Demographic information can provide a better qualitative understanding of CBZ contamination in the groundwater of Milan city.
- CBZ concentrations in groundwater were higher in the area with a higher presence of the aging population over 70 years old.
- The consumption of CBZ in children less than 5 years old living with their parents may also significantly influence the amount of CBZ in groundwater.
- In addition to age classes, social characteristics (i.e., level of education, marital status) may also influence CBZ levels.
- CBZ levels present the same pattern in both the upper aquifer (A) and the lower aquifer (B) in the Milan urban area, supporting the hypothesis that CBZ may derive from sewage leakages and then leak to the lower aquifers.
- Given that the semi-confined aquifer (B) is used for drinking water purposes in the Milan area, the contamination of groundwater may have implications for human health, and supports the need for specific water treatments. A better understanding of the source and impact of CBZ may support more efficient management in terms of both better drug use and more cost-efficient treatment.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ebrahimzadeh, S.; Wols, B.; Azzellino, A.; Martijn, B.J.; van der Hoek, J.P. Quantification and modelling of organic micropollutant removal by reverse osmosis (RO) drinking water treatment. J. Water Process. Eng. 2021, 42, 102164. [Google Scholar] [CrossRef]
- Richardson, S.D.; Ternes, T.A. Water Analysis: Emerging Contaminants and Current Issues. Anal. Chem. 2018, 90, 398–428. [Google Scholar] [CrossRef]
- Glassmeyer, S.T.; Furlong, E.T.; Kolpin, D.W.; Batt, A.L.; Benson, R.; Boone, J.S.; Conerly, O.; Donohue, M.J.; King, D.N.; Kostich, M.S.; et al. Nationwide reconnaissance of contaminants of emerging concern in source and treated drinking waters of the United States. Sci. Total Environ. 2017, 581, 909–922. [Google Scholar] [CrossRef]
- Schapira, M.; Manor, O.; Golan, N.; Kalo, D.; Mordehay, V.; Kirshenbaum, N.; Goldsmith, R.; Chefetz, B.; Paltiel, O. Involuntary human exposure to carbamazepine: A cross-sectional study of correlates across the lifespan and dietary spectrum. Environ. Int. 2020, 143, 105951. [Google Scholar] [CrossRef] [PubMed]
- Heberer, T. Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: A review of recent research data. Toxicol. Lett. 2002, 131, 5–17. [Google Scholar] [CrossRef]
- Metcalfe, C.D.; Koening, B.G.; Bennie, D.T.; Servos, M.; Ternes, T.A.; Hirsch, R. Occurrence of neutral and acidic drugs in the effluents of Canadian sewage treatment plants. Environ. Toxicol. Chem. 2003, 22, 2872–2880. [Google Scholar] [CrossRef] [PubMed]
- Kolpin, D.W.; Skopec, M.; Meyer, M.; Furlong, E.; Zauggd, S.D. Urban contribution of pharmaceuticals and other organic wastewater contaminants to streams during differing flow conditions. Sci. Total Environ. 2004, 328, 119–130. [Google Scholar] [CrossRef]
- Castiglioni, S.; Davoli, E.; Riva, F.; Palmiotto, M.; Camporini, P.; Manenti, A.; Zuccato, E. Mass balance of emerging contaminants in the water cycle of a highly urbanized and industrialized area of Italy. Water Res. 2018, 131, 287–298. [Google Scholar] [CrossRef]
- Conn, K.; Barber, L.B.; Brown, G.K.; Siegrist, R.L. Occurrence and Fate of Organic Contaminants during Onsite Wastewater Treatment. Environ. Sci. Technol. 2006, 40, 7358–7366. [Google Scholar] [CrossRef]
- Carrara, C.; Ptacek, C.J.; Robertson, W.D.; Blowes, D.W.; Moncur, M.; Sverko, E.; Backus, S. Fate of Pharmaceutical and Trace Organic Compounds in Three Septic System Plumes, Ontario, Canada. Environ. Sci. Technol. 2008, 42, 2805–2811. [Google Scholar] [CrossRef]
- Lee, H.-J.; Kim, K.Y.; Hamm, S.-Y.; Kim, M.; Kim, H.K.; Oh, J.-E. Occurrence and distribution of pharmaceutical and personal care products, artificial sweeteners, and pesticides in groundwater from an agricultural area in Korea. Sci. Total Environ. 2019, 659, 168–176. [Google Scholar] [CrossRef]
- Sui, Q.; Cao, X.; Lu, S.; Zhao, W.; Qiu, Z.; Yu, G. Occurrence sources and fate of pharmaceutical and personal care products in the groundwater: A review. Emerg. Contam. 2015, 1, 14–24. [Google Scholar] [CrossRef] [Green Version]
- Kolar, B.; Finizio, A. Assessment of environmental risks to groundwater ecosystems related to use of veterinary medicinal products. Regul. Toxicol. Pharmacol. 2017, 88, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Ou, W.; Wang, C.; Wang, Z.; Huang, Q.; Jin, J.; Tan, J. Occurrence and ecological potential of pharmaceuticals and personal care products in groundwater and reservoirs in the vicinity ofmunicipal landfills in China. Sci. Total Environ. 2014, 490, 889–898. [Google Scholar] [CrossRef]
- Yao, L.; Wang, Y.; Tong, L.; Deng, Y.; Li, Y.; Gan, Y.; Guo, W.; Dong, C.; Duan, Y.; Zhao, K. Occurrence and risk assessment of antibiotics in surface water and groundwater from different depths of aquifers: A case study at Jianghan Plain, central China. Ecotoxicol. Environ. Saf. 2017, 135, 236–242. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Baran, N.; Stuart, M.E.; Ward, R.S. Emerging organic contaminants in groundwater: A review of sources fate and occurrence. Environ. Pollut. 2012, 163, 287–303. [Google Scholar] [CrossRef] [Green Version]
- Mohan Sharma, B.; Bečanová, J.; Scheringer, M.; Sharma, A.; Bharat, G.K.; Whitehead, P.G.; Klánová, J.; Nizzetto, L. Health and ecological risk assessment of emerging contaminants (pharmaceuticals, personal care products, and artificial sweeteners) in surface and groundwater (drinking water) in the Ganges River Basin, India. Sci. Total Environ. 2019, 646, 1459–1467. [Google Scholar] [CrossRef] [PubMed]
- Kibuye, F.A.; Gall, H.E.; Elkin, K.R.; Ayers, B.; Veith, T.L.; Miller, M.; Jacob, S.; Hayden, K.R.; Watson, J.E.; Elliott, H.A. Fate of pharmaceuticals in a spray-irrigation system: From wastewater to groundwater. Sci. Total Environ. 2019, 654, 197–208. [Google Scholar] [CrossRef]
- Edwards, Q.A.; Sultana, T.; Kulikov, S.M.; Garner-O’Neale, L.D.; Metcalfe, C.D. Micropollutants related to human activity in groundwater resources in Barbados, West Indies. Sci. Total Environ. 2019, 671, 76–82. [Google Scholar] [CrossRef]
- Stuart, M.; Lapworth, D.; Crane, E.; Hart, A. Review of risk from potential emerging contaminants in UK groundwater. Sci. Total Environ. 2012, 416, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Buerge, I.J.; Buser, H.R.; Kahle, M.; Müller, M.D.; Poiger, T. Ubiquitous Occurrence of the Artificial Sweetener Acesulfame in the Aquatic Environment: An Ideal Chemical Marker of Domestic Wastewater in Groundwater. Environ. Sci. Technol. 2009, 43, 4381–4385. [Google Scholar] [CrossRef] [PubMed]
- Clara, M.; Strenn, B.; Kreuzinger, N. Carbamazepine as a possible anthropogenic marker in the aquatic environment: Investigations on the behaviour of Carbamazepine in wastewater treatment and during groundwater infiltration. Water Res. 2004, 38, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Nakada, N.; Kiri, K.; Shinohara, H.; Harada, A.; Kuroda, K.; Takizawa, S.; Takada, H. Evaluation of Pharmaceuticals and Personal Care Products as Water-soluble Molecular Markers of Sewage. Environ. Sci. Technol. 2008, 42, 6347–6353. [Google Scholar] [CrossRef]
- James, C.A.; Miller-Schulze, J.P.; Ultican, S.; Gipe, A.D.; Baker, J.E. Evaluating contaminants of emerging concern as tracers of wastewater from septic systems. Water Res. 2016, 101, 241–251. [Google Scholar] [CrossRef]
- Kuroda, K.; Murakami, M.; Oguma, K.; Muramatsu, Y.; Takada, H.; Takizawa, S. Assessment of groundwater pollution in Tokyo using PPCPs as sewage markers. Environ. Sci. Technol. 2012, 46, 1455–1464. [Google Scholar] [CrossRef]
- Müller, B.; Scheytt, T.; Asbrand, M.; de Casas, A.M. Pharmaceuticals as indictors of sewage-influenced groundwater. Hydrogeol. J. 2012, 20, 1117–1129. [Google Scholar] [CrossRef]
- Robertson, W.D.; Van Stempvoort, D.R.; Roy, J.W.; Brown, S.J.; Spoelstra, J.; Schiff, S.L.; Rudolph, D.R.; Danielescu, S.; Graham, G. Use of an artificial sweetener to identify sources of groundwater nitrate contamination. Groundwater 2016, 54, 579–587. [Google Scholar] [CrossRef]
- Robertson, W.D.; Van Stempvoort, D.R.; Solomon, D.K.; Homewood, J.; Brown, S.J.; Spoelstra, J.; Schiff, S.L. Persistence of artificial sweeteners in a 15-year-old septic system plume. J. Hydrol. 2013, 477, 43–54. [Google Scholar] [CrossRef]
- Wolf, L.; Zwiener, C.; Zemann, M. Tracking artificial sweeteners and pharmaceuticals introduced into urban groundwater by leaking sewer networks. Sci. Total Environ. 2012, 430, 8–19. [Google Scholar] [CrossRef]
- Yang, Y.-Y.; Toor, G.S.; Wilson, P.C.; Williams, C.F. Micropollutants in groundwater from septic systems: Transformations, transport mechanisms, and human health risk assessment. Water Res. 2017, 123, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Krall, A.L.; Elliott, S.M.; Erickson, M.L.; Adams, B.A. Detecting sulfamethoxazole and carbamazepine in groundwater: Is ELISA a reliable screening tool? Environ. Pollut. 2018, 234, 420–428. [Google Scholar] [CrossRef]
- Garcia-Fresca, B. Urban-enhanced groundwater recharge: Review and case study of Austin, Texas, USA. In Urban Groundwater, Meeting the Challenge; Ken, H., Ed.; IAH Selected Papers on Hydrogeology; CRC Press: Los Angeles, CA, USA, 2007. [Google Scholar]
- Vatovec, C.; Phillips, P.; Van Wagoner, E.; Scott, T.M.; Furlong, E. Investigating dynamic sources of pharmaceuticals: Demographic and seasonal use are more important than down-the-drain disposal in wastewater effluent in a University City setting. Sci. Total Environ. 2016, 572, 906–914. [Google Scholar] [CrossRef] [PubMed]
- ter Laak, T.L.; Kooij, P.J.F.; Tolkamp, H.; Hofman, J. Different compositions of pharmaceuticals in Dutch and Belgian rivers explained by consumption patterns and treatment efficiency. Environ. Sci. Pollut. Res. 2014, 21, 12843–12855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khorasani, H.; Kerachian, R.; Aghayi, M.M.; Zahraie, B.; Zhu, Z. Assessment of the Impacts of Sewerage Network on Groundwater Quantity and Nitrate Contamination: Case Study of Tehran. In World Environmental and Water Resources Congress; American Society of Civil Engineers: Reston, VA, USA, 2020; pp. 53–66. [Google Scholar]
- Daughton, C.G. Pharmaceuticals and the Environment (PiE): Evolution and impact of the published literature revealed by bibliometric analysis. Sci. Total Environ. 2016, 562, 391–426. [Google Scholar] [CrossRef] [Green Version]
- Petrie, B.; Youdan, J.; Barden, R.; Kasprzyk-Hordern, B. New Framework to Diagnose the Direct Disposal of Prescribed Drugs in Wastewater—A Case Study of the Antidepressant Fluoxetine. Environ. Sci. Technol. 2016, 50, 3781–3789. [Google Scholar] [CrossRef] [Green Version]
- Bogunović, M.; Ivančev-Tumbas, I.; Česen, M.; Sekulić, T.D.; Prodanović, J.; Tubić, A.; Heath, D.; Heath, E. Removal of selected emerging micropollutants from wastewater treatment plant effluent by advanced non-oxidative treatment—A lab-scale case study from Serbia. Sci. Total Environ. 2021, 765, 142764. [Google Scholar] [CrossRef]
- Edefell, E.; Falås, P.; Kharel, S.; Hagman, M.; Christensson, M.; Cimbritz, M.; Bester, K. MBBRs as post-treatment to ozonation: Degradation of transformation products and ozone-resistant micropollutants. Sci. Total Environ. 2021, 754, 142103. [Google Scholar] [CrossRef] [PubMed]
- Brunner, A.M.; Bertelkamp, C.; Dingemans, M.M.; Kolkman, A.; Wols, B.; Harmsen, D.; Siegers, W.; Martijn, B.J.; Oorthuizen, W.A.; ter Laak, T.L. Integration of target analyses, non-target screening and effect-based monitoring to assess OMP related water quality changes in drinking water treatment. Sci. Total Environ. 2020, 705, 135779. [Google Scholar] [CrossRef]
- Venkatesan, A.K.; Halden, R.U. Wastewater treatment plants as chemical observatories to forecast ecological and human health risks of manmade chemicals. Sci. Rep. 2014, 4, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giacchetta, G.; Marchetti, B. Medical waste management: A case study in a small size hospital of central Italy. Strat. Outsourc. Int. J. 2013, 6, 65–84. [Google Scholar] [CrossRef]
- Verlicchi, P.; Galletti, A.; Petrovic, M.; Barceló, D. Hospital effluents as a source of emerging pollutants: An overview of micropollutants and sustainable treatment options. J. Hydrol. 2010, 389, 416–428. [Google Scholar] [CrossRef]
- Riva, F.; Castiglioni, S.; Fattore, E.; Manenti, A.; Davoli, E.; Zuccato, E. Monitoring emerging contaminants in the drinking water of Milan and assessment of the human risk. Int. J. Hyg. Environ. Health 2018, 221, 451–457. [Google Scholar] [CrossRef]
- Simazaki, D.; Kubota, R.; Suzuki, T.; Akiba, M.; Nishimura, T.; Kunikane, S. Occurrence of selected pharmaceuticals at drinking water purification plants in Japan and implications for human health. Water Res. 2015, 76, 187–200. [Google Scholar] [CrossRef]
- Castiglioni, S.; Bagnati, R.; Calamari, D.; Fanelli, R.; Zuccato, E. A multiresidue analytical method using solid-phase extraction and high-pressure liquid chromatography tandem mass spectrometry to measure pharmaceuticals of different therapeutic classes in urban wastewaters. J. Chromatogr. A 2005, 1092, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Palmiotto, M.; Castiglioni, S.; Zuccato, E.; Manenti, A.; Riva, F.; Davoli, E. Personal care products in surface, ground and wastewater of a complex aquifer system, a potential planning tool for contemporary urban settings. J. Environ. Manag. 2018, 214, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Spina, E.; Perugi, G. Antiepileptic drugs: Indications other than epilepsy. Epileptic Disorders. 2004, 6, 57–75. [Google Scholar] [PubMed]
- Iorio, M.L.; Moretti, U.; Colcera, S.; Magro, L.; Meneghelli, I.; Motola, D.; Rivolta, A.L.; Salvo, F.; Velo, G.P. Use and safety profile of antiepileptic drugs in Italy. Eur. J. Clin. Pharmacol. 2007, 63, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Putignano, D.; Clavenna, A.; Campi, R.; Bortolotti, A.; Fortino, I.; Merlino, L.G.; Vignoli, A.; Canevini, M.P.; Bonati, M. Antiepileptic drug use in Italian children over a decade. Eur. J. Clin. Pharmacol. 2017, 73, 241–248. [Google Scholar] [CrossRef]
- Scheytt, T.; Mersmann, P.; Lindstädt, R.; Heberer, T. Determination of sorption coefficients of pharmaceutically active substances carbamazepine, diclofenac, and ibuprofen, in sandy sediments. Chemosphere 2005, 60, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Dvory, N.Z.; Livshitz, Y.; Kuznetsov, M.; Adar, E.; Gasser, G.; Pankratov, I.; Lev, O.; Yakirevich, A. Caffeine vs. carbamazepine as indicators of wastewater pollution in a karst aquifer. Hydrol. Earth Syst. Sci. 2018, 22, 6371–6381. [Google Scholar] [CrossRef] [Green Version]
- Fenz, R.; Blaschke, A.; Clara, M.; Kroiss, H.; Mascher, D.; Zessner, M. Quantification of sewer exfiltration using the anti-epileptic drug carbamazepine as marker species for wastewater. Water Sci. Technol. 2005, 52, 209–217. [Google Scholar] [CrossRef]
- Gasser, G.; Rona, M.; Voloshenko, A.; Shelkov, R.; Tal, N.; Pankratov, I.; Elhanany, S.; Lev, O. Quantitative Evaluation of Tracers for Quantification of Wastewater Contamination of Potable Water Sources. Environ. Sci. Technol. 2010, 44, 3919–3925. [Google Scholar] [CrossRef]
- Arduini, C.; Dadomo, A.; Martinelli, G.; Porto, G.; Zelioli, A.; Carraro, E.; Gangemi, M. Nitrates in groundwater (Northern Italy): Isotopic prospection in high vulnerability area. In Proceedings of the International Symposium “Consoil 2008”, Milan, Italy, 3–6 June 2008; pp. F205–F215. [Google Scholar]
- Phillips, P.J.; Schubert, C.; Argue, D.; Fisher, I.; Furlong, E.T.; Foreman, W.; Gray, J.; Chalmers, A. Concentrations of hormones, pharmaceuticals and other micropollutants in groundwater affected by septic systems in New England New York. Sci. Total Environ. 2015, 512, 43–54. [Google Scholar] [CrossRef]
- Sacchi, E.; Acutis, M.; Bartoli, M.; Brenna, S.; Delconte, D.A.; Laini, A.; Pennisi, M. Origin and fate of nitrates in groundwater from the central Po plain: Insights from isotopic investigations. Appl. Geochem. 2013, 34, 164–180. [Google Scholar] [CrossRef]
- Demographic Statistics ISTAT. Annual Survey on Movement and Calculation of the Resident Population. ISTAT Roma. Available online: www.demo.istat.it (accessed on 30 July 2021).
- Shahvi, S.; Orsi, E.; Canziani, R.; Larcan, E.; Becciu, G. Study on industrial macropollutants discharges in Milan sewer system. Manag. Environ. Qual. 2016, 27, 194–209. [Google Scholar] [CrossRef]
- Colombo, L.; Gattinoni, P.; Scesi, L. Influence of underground structures and infrastructures on the groundwater level in the urban area of Milan, Italy. Int. J. Sustain. Dev. Plan. 2017, 12, 176–184. [Google Scholar] [CrossRef]
- Gattinoni, P.; Scesi, L. The groundwater rise in the urban area of Milan (Italy) and its interactions with underground structures and infrastructures. Tunn. Undergr. Space Technol. 2017, 62, 103–114. [Google Scholar] [CrossRef]
- De Luca, D.A.; Destefanis, E.; Forno, M.G.; Lasagna, M.; Masciocco, L. The genesis and the hydrogeological features of the Turin Po Plain fontanili, typical lowland springs in Northern Italy. Bull. Eng. Geol. Environ. 2014, 73, 409–427. [Google Scholar] [CrossRef]
- De Caro, M.; Crosta, G.B.; Frattini, P. Hydrogeochemical characterization and Natural Background Levels in urbanized areas: Milan Metropolitan area (Northern Italy). J. Hydrol. 2017, 547, 455–473. [Google Scholar] [CrossRef]
- Lombardia, R. Eni Division Agip (2002) Geologia Degli Acquiferi Padani della Regione Lombardia, a Cura di Cipriano Carcano e Andrea Piccin. S. EL. CA. (Firenze). Available online: https://www.arpalombardia.it (accessed on 30 July 2021). (In Italian).
- Kaufman, L. Finding Groups in Data: An Introduction to Cluster Analysis; Wiley: New York, NY, USA, 1990. [Google Scholar]
- Jolliffe, I.T. Principal Component Analysis; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar]
- Afifi, A.; Clark, V. Computer-Aided Multivariate Analysis; Van Nostrand Reinhold Company: New York, NY, USA, 1990. [Google Scholar]
- Menció, A.; Mas-Pla, J. Assessment by multivariate analysis of ground water–surface water interactions in urbanized Mediterranean streams. J. Hydrol. 2008, 362, 355–366. [Google Scholar] [CrossRef]
- Kohberger, R.; Willet, P. Similarity and Clustering in Chemical Information Systems. Technometrics 1990, 32, 359. [Google Scholar] [CrossRef]
- Azzellino, A.; Colombo, L.; Lombi, S.; Marchesi, V.; Piana, A.; Andrea, M.; Alberti, L. Groundwater diffuse pollution in functional urban areas: The need to define anthropogenic diffuse pollution background levels. Sci. Total Environ. 2019, 656, 1207–1222. [Google Scholar] [CrossRef]
- OECD. Pharmaceutical Residues in Freshwater; OECD: Paris, France, 2019; Available online: https://www.oecd.org (accessed on 30 July 2021).
- Hauser, W.A.; Annegers, J.F.; Kurland, L.T. Prevalence of Epilepsy in Rochester, Minnesota: 1940–1980. Epilepsia 1991, 32, 429–445. [Google Scholar] [CrossRef]
- Savica, R.; Beghi, E.; Mazzaglia, G.; Innocenti, F.; Brignoli, O.; Cricelli, C.; Caputi, A.P.; Musolino, R.; Spina, E.; Trifirò, G. Prescribing patterns of antiepileptic drugs in Italy: A nationwide population-based study in the years 2000–2005. Eur. J. Neurol. 2007, 14, 1317–1321. [Google Scholar] [CrossRef]
- Rowan, A.J.; Ramsay, R.E.; Collins, J.F.; Pryor, F.; Boardman, K.D.; Uthman, B.M.; Spitz, M.; Frederick, T.; Towne, A.; Carter, G.S.; et al. New onset geriatric epilepsy: A randomized study of gabapentin, lamotrigine, and carbamazepine. Neurology 2005, 64, 1868–1873. [Google Scholar] [CrossRef]
- Punyawudho, B.; Ramsay, E.R.; Brundage, R.C.; Macias, F.M.; Collins, J.F.; Birnbaum, A.K. Population Pharmacokinetics of Carbamazepine in Elderly Patients. Ther. Drug Monit. 2012, 34, 176–181. [Google Scholar] [CrossRef] [Green Version]
- Djordjevic, N.; Jankovic, S.M.; Milovanovic, J.R. Pharmacokinetics and Pharmacogenetics of Carbamazepine in Children. Eur. J. Drug Metab. Pharmacokinet. 2017, 42, 729–744. [Google Scholar] [CrossRef]
- Djordjevic, N.; Milovanovic, D.D.; Radovanović, M.; Radosavljević, I.; Obradović, S.; Jakovljevic, M.; Milovanovic, D.; Milovanovic, J.R.; Jankovic, S. CYP1A2 genotype affects carbamazepine pharmacokinetics in children with epilepsy. Eur. J. Clin. Pharmacol. 2016, 72, 439–445. [Google Scholar] [CrossRef]
- Tolou-Ghamari, Z.; Zare, M.; Habibabadi, J.M.; Najafi, M.R. A quick review of carbamazepine pharmacokinetics in epilepsy from 1953 to 2012. J. Res. Med. Sci. 2013, 18, S81–S85. [Google Scholar]
- Italian Medicines Agency. AIFA 2018. Available online: http://www.aifa.gov.it/ (accessed on 30 July 2021).
- Sullivan, G.; Davis, S. Is carbamazepine a potential drug of abuse? J. Psychopharmacol. 1997, 11, 93–94. [Google Scholar] [CrossRef]
- Hosseini, S.H.; Ahmadi, A. Abuse potential of carbamazepine for euphorigenic effects. Drug Res. 2015, 65, 223–224. [Google Scholar] [CrossRef]
- Xu, W.; Chen, Y.-L.; Zhao, Y.; Wang, L.-J.; Li, J.-J.; Liu, C.-F. A Clinical Study of Toxication Caused by Carbamazepine Abuse in Adolescents. BioMed Res. Int. 2018, 2018, 1–6. [Google Scholar] [CrossRef]
| Well Num. | GW Monitoring Station in Aquifer B | CBZ Concentration in Aquifer B (ng/L) | GW Monitoring Station in Aquifer A | CBZ Concentration in Aquifer A (ng/L) |
|---|---|---|---|---|
| 1 | B1 | 3.9 | A1 | 3.00 |
| 2 | B2 | 14.45 | A2 | 23.8 |
| 3 | B3 | 16.19 | A3 | 13.6 |
| 4 | B4 | 16.34 | A4 | 46.6 |
| 5 | B5 | 10.39 | A5 | 25.1 |
| 6 | B6 | 16.47 | A6 | 16.14 |
| 7 | B7 | 25.34 | A7 | 65.03 |
| 8 | B8 | 9.41 | A8 | 9.4 |
| 9 | B9 | 14.58 | A9 | 41.8 |
| 10 | B10 | 12.13 | A10 | 34.57 |
| 11 | B11 | 13.1 | A11 | 13.6 |
| 12 | B12 | 31.57 | A12 | 69.15 |
| 13 | B13 | 22.86 | A13 | 21.23 |
| 14 | B14 | 1.61 | A14 | 9.31 |
| 15 | B15 | 0.2 | A15 | 44.15 |
| 16 | B16 | 15.99 | A16 | 38.57 |
| 17 | B17 | 7.33 | A17 | 98.13 |
| 18 | B18 | 14.94 | A18 | 9.1 |
| 19 | B19 | 7.56 | A19 | 5.9 |
| 20 | B20 | 10.29 | A20 | 1.53 |
| 21 | B21 | 15.08 | A21 | 98.16 |
| Varifactors | Initial Eigenvalues | Extraction Sums of Squared Loadings | Rotation Sums of Squared Loadings | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | % of Variance | Cumulative % | Total | % of Variance | Cumulative % | Total | % of Variance | Cumulative % | |
| F1 | 51.8 | 68.170 | 68.170 | 51.8 | 68.170 | 68.17 | 22.12 | 29.11 | 29.110 |
| F2 | 4.72 | 6.210 | 74.380 | 4.72 | 6.210 | 74.38 | 14.7 | 19.45 | 48.561 |
| F3 | 3.76 | 4.952 | 79.332 | 3.76 | 4.952 | 79.33 | 11.4 | 15.00 | 63.569 |
| F4 | 2.05 | 2.704 | 82.035 | 2.05 | 2.704 | 82.03 | 5.32 | 7.01 | 70.579 |
| F5 | 1.51 | 1.992 | 84.027 | 1.51 | 1.992 | 84.03 | 4.91 | 6.47 | 77.048 |
| F6 | 1.23 | 1.629 | 85.656 | 1.23 | 1.629 | 85.65 | 3.87 | 5.09 | 82.144 |
| F7 | 1.02 | 1.350 | 87.006 | 1.02 | 1.350 | 87.006 | 2.73 | 3.60 | 85.748 |
| F8 | 1.00 | 1.321 | 88.327 | 1.00 | 1.321 | 88.33 | 1.96 | 2.6 | 88.327 |
| Pearson Correlation | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | CBZ |
|---|---|---|---|---|---|---|---|---|---|
| F1: Family index | 1 | 0.24 | −0.16 | 0.27 | 0.61 | −0.27 | −0.48 | −0.08 | 0.38 * |
| F2: Aging index | 0.24 | 1 | 0.82 | −0.40 | 0.22 | 0.31 | 0.67 | −0.54 | 0.27 * |
| F3: Adolescents and quinquagenarian index | −0.16 | 0.82 | 1 | −0.49 | 0.30 | 0.76 | 0.94 | −0.78 | −0.14 |
| F4: Divorce index | 0.27 | −0.40 | −0.49 | 1 | 0.12 | −0.45 | −0.44 | −0.04 | 0.61 * |
| F5: Illiterate index | 0.61 | 0.22 | 0.30 | 0.12 | 1 | 0.54 | 0.02 | −0.70 | −0.18 |
| F6: Sexagenarian index | −0.27 | 0.31 | 0.76 | −0.45 | 0.54 | 1 | 0.70 | −0.77 | −0.64 * |
| F7: Child index | −0.48 | 0.67 | 0.94 | −0.44 | 0.02 | 0.70 | 1 | −0.68 | −0.13 |
| F8: Education index | −0.08 | −0.54 | −0.78 | −0.04 | −0.70 | −0.77 | −0.68 | 1 | 0.05 |
| CBZ | 0.38 | 0.27 | −0.14 | 0.61 | −0.18 | −0.64 | −0.13 | 0.05 | 1 |
| Pearson Correlation | F1 | F2 | F3 | F4 | F5 | F6 | F7 | F8 | CBZ |
|---|---|---|---|---|---|---|---|---|---|
| F1: Family index | 1 | 0.63 | −0.02 | −0.85 | −0.06 | −0.57 | −0.38 | −0.50 | 0.87 * |
| F2: Aging index | 0.63 | 1 | −0.78 | −0.94 | 0.01 | −0.98 | −0.36 | −0.74 | 0.75 * |
| F3: Adolescents and quinquagenarian index | −0.02 | −0.78 | 1 | 0.53 | −0.23 | 0.83 | 0.00 | 0.67 | −0.32 |
| F4: Divorce index | −0.85 | −0.94 | 0.53 | 1 | 0.06 | 0.90 | 0.44 | 0.69 | 0.87 * |
| F5: Illiterate index | −0.06 | 0.01 | −0.23 | 0.06 | 1 | −0.20 | 0.92 | −0.67 | 0.27 |
| F6: Sexagenarian index | −0.57 | −0.98 | 0.83 | 0.90 | −0.20 | 1 | 0.17 | 0.85 | −0.76 * |
| F7: Child index | −0.38 | −0.36 | 0.00 | 0.44 | 0.92 | 0.17 | 1 | −0.34 | −0.09 |
| F8: Education index | −0.50 | −0.74 | 0.67 | 0.69 | −0.67 | 0.85 | −0.34 | 1 | −0.78 * |
| CBZ | 0.87 | 0.75 | −0.32 | 0.87 | 0.27 | −0.76 | −0.09 | −0.78 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ebrahimzadeh, S.; Castiglioni, S.; Riva, F.; Zuccato, E.; Azzellino, A. Carbamazepine Levels Related to the Demographic Indicators in Groundwater of Densely Populated Area. Water 2021, 13, 2539. https://doi.org/10.3390/w13182539
Ebrahimzadeh S, Castiglioni S, Riva F, Zuccato E, Azzellino A. Carbamazepine Levels Related to the Demographic Indicators in Groundwater of Densely Populated Area. Water. 2021; 13(18):2539. https://doi.org/10.3390/w13182539
Chicago/Turabian StyleEbrahimzadeh, Salma, Sara Castiglioni, Francesco Riva, Ettore Zuccato, and Arianna Azzellino. 2021. "Carbamazepine Levels Related to the Demographic Indicators in Groundwater of Densely Populated Area" Water 13, no. 18: 2539. https://doi.org/10.3390/w13182539







