1. Introduction
Irrigation with reclaimed water is a key method of alleviating agricultural water shortages (FAO, 2012) [
1]. Recycled water can save water resources and promote the circulation of nutrients, but pollutants in reclaimed water and soil have potential to cause health risks to humans, especially the endocrine disrupter chemicals (EDCs), which have attracted wide attention recently. EDCs can lead to endocrine disorders in aquatic organisms, affecting reproductive development and the immune system [
2,
3].
Reclaimed water from wastewater treatment plants (WWTPs) is regarded as one of the main sources of EDCs in the agricultural environment. In addition to reclaimed water or effluent, treated sewage sludge is also used all over the world in agriculture soil [
4,
5,
6,
7]. Municipal landfills also contain leachate with significant amounts of EDCs that could migrate into groundwater [
8,
9,
10,
11]. In recent years, the fate of EDCs in groundwater and reclaimed water has received much attention [
12,
13,
14,
15].
Nonylphenol (NP) and bisphenol A (BPA) are two typical EDCs with high detection frequencies and concentrations in soils irrigated with reclaimed water [
16,
17]. The concentrations of NP in reclaimed water and agricultural soils were 0.05–63 μg·L
−1 and 14.2–60.3 mg·kg
−1, respectively [
13,
18,
19,
20,
21]. The concentrations of BPA in reclaimed water and agricultural soil were ND-101.6 μg·L
−1 and ND-147 μg·kg
−1, respectively [
16,
22,
23,
24,
25]. The Danish Institute of Safety and Toxicology (DIST) derived a preliminary tolerable daily intake (TDI) value for NP of 5 μg·(day kg)
−1 body weight [
26]. USEPA estimated a reference dose for BPA of 50 μg·(day kg)
−1 body weight [
27].
NP is composed of varieties of isomers. The estrogenic activity and the environmental fate were heavily dependent on the isomer structures, such as the side-chain length, degree of branching, α-substituent type and steric index [
28,
29,
30]. The most toxic isomer among these isomers is 4-(3-ethyl-2-methylhexan-2-yl) phenol, which is noted as NP
7 [
19,
31] (
Supplementary Material S1) in this study. Adsorption, migration, and degradation are the main processes for organic substances in soil. Degradation is the main fate of NP and BPA in soil during the reclaimed water irrigation. NP in sewage irrigation soil completed the rapid degradation stage within 20 days, and the removal rates reached above 80% [
32], while adsorption is the first step for NP and BPA during the degradation process, which determines the rate of migration and transformation of NP and BPA into groundwater [
33]. Adsorption of NP and BPA on soils is affected by a variety of factors, including dissolved organic matter (DOM), temperature, pH and ionic content [
34,
35,
36,
37,
38], among which dissolved DOM plays a significant role [
38,
39,
40].
The Daxing irrigation district, in the southeastern part of Beijing, China, was one of the typical sewage irrigation districts for more than 40 years. Previous studies of this district focused on inorganic pollution or total NP isomers [
41,
42]. The study of individual isomers is quite few because of the difficulty in the separation of NP isomers, while the risks of the different isomers to the local ecological environment were different. In particular, the most toxic isomer, NP
7, has much estrogenic activity to the environment, so the study on adsorption and desorption of the NP isomer on field soil is imperative. The aim of this work is to investigate: (1) Comparison of the adsorption of NP, NP
7 and BPA on field soil irrigated with reclaimed water; (2) The effect of different environmental factors of pH, temperature and different polyvalent metal ions (Na
+, Ca
2+, As(III)) on the adsorption of NP, NP
7 and BPA; (3) The mechanism of sorption of NP and BPA on soil by Fourier Transform Infrared Spectrometer (FTIR) analysis.
2. Materials and Methods
2.1. Reagents and Materials
Soils (0~20 cm) were collected from Daxing reclaimed water irrigation district, China (39°36′ N, 116°21′ E). After the plants’ debris and residues were removed, the soils were freeze-dried at −20 °C and then 0.9 mm-sieved. The physicochemical properties of the soil are shown in
Table 1. The background concentrations of NP, NP
7 and BPA in collected soils were 37 μg·kg
−1, 2 μg·kg
−1 and below the method detection limit (MDL), respectively, and are reported in the former study [
17].
NP (0.25 g, 100%) and BPA (0.25 g, 99.8%) were purchased from Dr. Ehrenstorfer GmbH and dissolved in methanol with a concentration of 1000 mg·L−1 NaN3 (200 g, 99%). Methanol (HPLC grade) and dichloromethane (HPLC grade) were purchased from Honeywell (Morris Plains, NJ, USA). As(III) were obtained from Sigma-Aldrich (St. Louis, MO, USA). Anhydrous calcium chloride (CaCl2) (500 g, >96%), sodium chloride (NaCl) (500 g, >99.5%) and anhydrous sodium sulfate (Na2SO4) (500 g, >98%) were obtained from the Tianjin Fuchen chemical reagents factory.
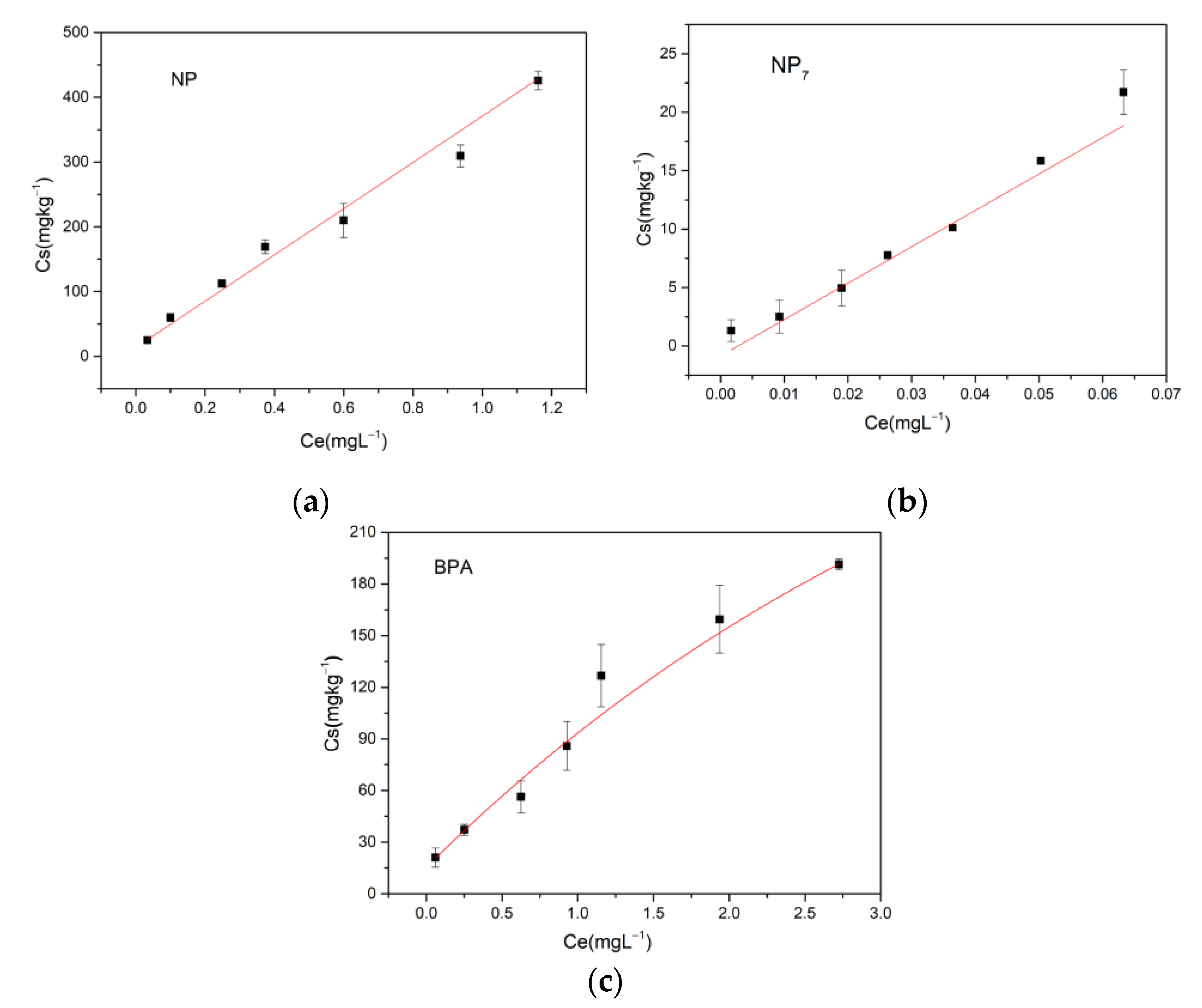
2.2. Isothermal Adsorption Experiment
The isothermal adsorption experiments were carried out under experimental conditions of pH7 at 25 °C. We added 0.2 g soil into a 40 mL brown vial, with 30 mL 0.01 M CaCl
2 solution as the background electrolyte (except for the polyvalent metal ions experiment). NaN
3 was added at a concentration of 200 mg L
−1 to sterilize the soil. NP (or BPA) solution was added at an initial concentration of 200, 500, 1000, 1500, 2000, 3000 and 4000 µg·L
−1. The brown bottles were placed in a thermostatic shaker at 25 °C. NP and BPA were allowed to adsorb on the soil for 24 h to reach equilibrium. After the samples were centrifuged for 5 min (3000 rpm), NP (or BPA) in the supernatant was extracted (
Supplementary Material S3) according to USEPA 3550C [
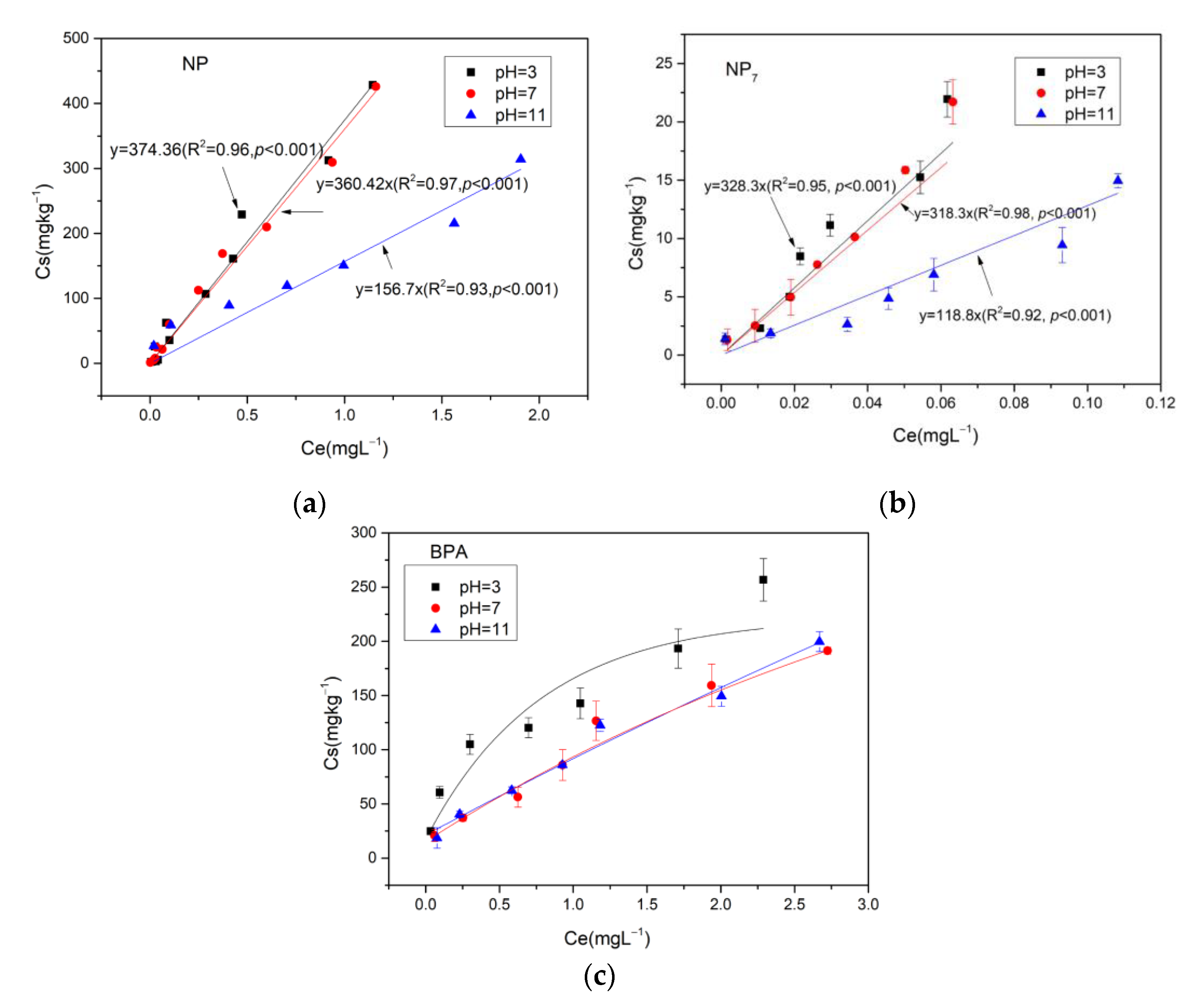
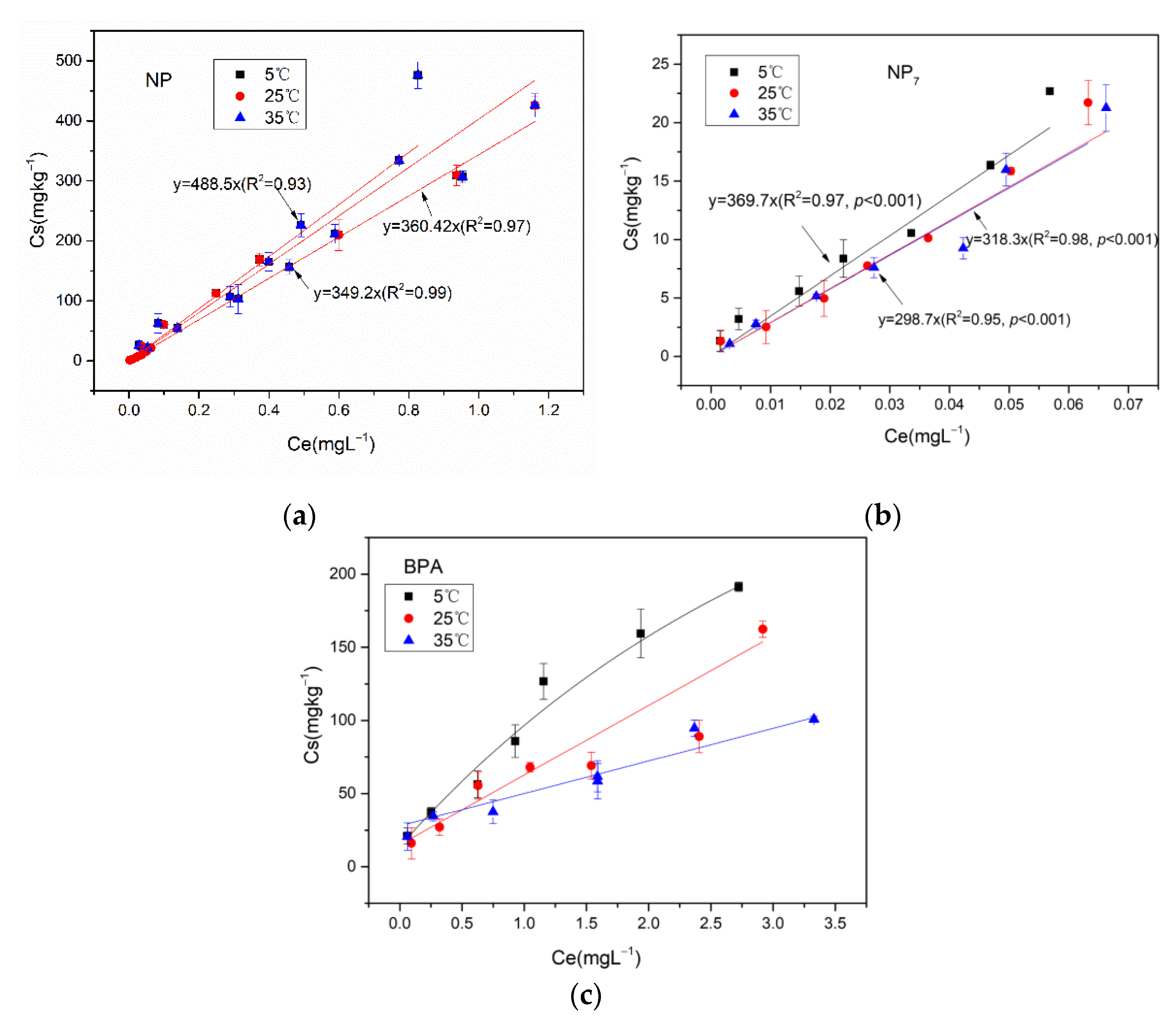
43], and determined by Gas Chromatograph Mass Spectrometer (GC-MS). The effects of different environmental factors on the adsorption were studied, including pH of the solution (pH 3.7, and 11), temperatures (5 °C, 25 °C and 35 °C) and polyvalent metal ions (Na
+, Ca
2+ and As(III)) with a concentration of 0.002 M. For the polyvalent metal ions experiment, the background electrolytes were Na
+, Ca
2+ and As(III) solution, respectively. The experiment was conducted in triplicate.
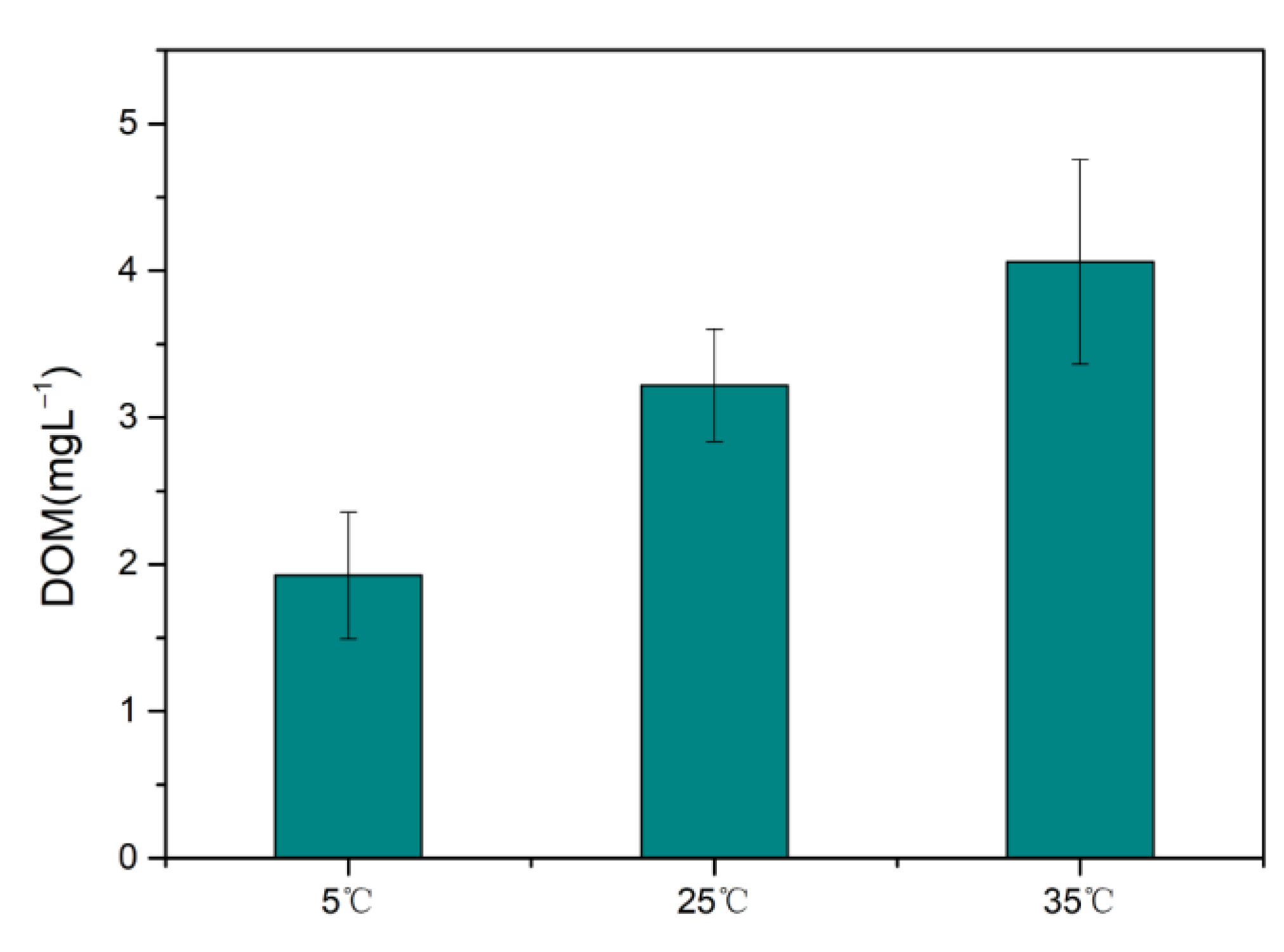
2.3. Dissolved Organic Matter (DOM)
In order to verify the effect of temperature on the dissolution of DOM, soil and water were mixed at a ratio of 1:150, shaking for 24 h at 5, 25 and 35 °C, respectively. Then the solution was centrifuged at 3000 rpm. DOM in the supernatant solution was measured with TOC instrument.
2.4. Data Analysis
The commonly used isothermal adsorption models are Linear adsorption model, Langmuir model and Freundlich model. The formulas are as follows:
2.4.1. Linear Adsorption Model
Kd: Adsorption equilibrium constant (L·kg−1).
Ce: Concentration of pollutants in the liquid phase at equilibrium (mg·L−1).
Cs: Concentration of pollutants in the solid phase at equilibrium (mg·kg−1).
2.4.2. Langmuir Adsorption Model
Qmax: Maximum sorption amount of pollutants on solids (mg·kg−1).
Ce: Concentration of pollutants in the liquid phase at equilibrium (mg·L−1).
Cs: Concentration of pollutants in the solid phase at equilibrium (mg·kg−1).
KL: Adsorption equilibrium constant (L mg−1).
2.4.3. Freundlich Adsorption Model
Kf: Adsorption equilibrium constant (mg1−1/n L1/n/kg).
Ce: Concentration of pollutants in the liquid phase at equilibrium (mg L−1).
Cs: Concentration of pollutants in the solid phase at equilibrium (mg·kg−1).
1/n: Adsorption force parameter (dimensionless).
2.4.4. Organic Carbon Adsorption Constants
The soil sorption constants were normalized to the organic carbon sorption constants (K
oc). Because the main adsorbent is organic carbon in soil, and K
oc is a characterization of the sorption capacity of organic carbon. The formula is:
Koc: Adsorption coefficient of pollutants on organic carbon (mg·kg−1).
Kd: Adsorption coefficient of pollutants on soil (mg·kg−1).
f: Organic carbon content in soil (%).
2.4.5. Determination of NP and BPA
NP and BPA were detected with GC-MS (Agilent, 6890 N/5975), with automatic sampler (7683B Series), capillary-column (60 m × 0.25 μm × 0.25 mm, DB-5MS, Agilent Technologies, Palo Alto, CA, USA). The parameters were as follows: the inlet and the detector temperatures were set at 250 °C and 280 °C, respectively. Helium was used as the carrier gas, and the flow rate was 1 mL min
−1. The injection mode was splitless, at a sample volume of 1 μL. The oven was heated as follows: Initial temperature was set at 40 °C and maintained for 1 min, heated to 150 °C at a rate of 8 °C min
−1 and maintained for 1 min, and then heated to 230 °C at a rate of 5 °C min
−1 and maintained for 2 min. The ion source was electron impact and the temperatures of the ion source and triple-quadrupole were 230 °C and 150 °C, respectively; the voltage was 70 eV. Data were acquired with both Scan and SIM mode, and the scan range was 50 to 300 amu. The MDLs for NP isomers were 0.86–2.65 μg·kg
−1, and the MDL of BPA was 2.0 μg·kg
−1. The method recovery for NP and BPA ranged from 70% to 110%, which met the standard of the U.S. EPA [
17].
2.4.6. FTIR Analysis
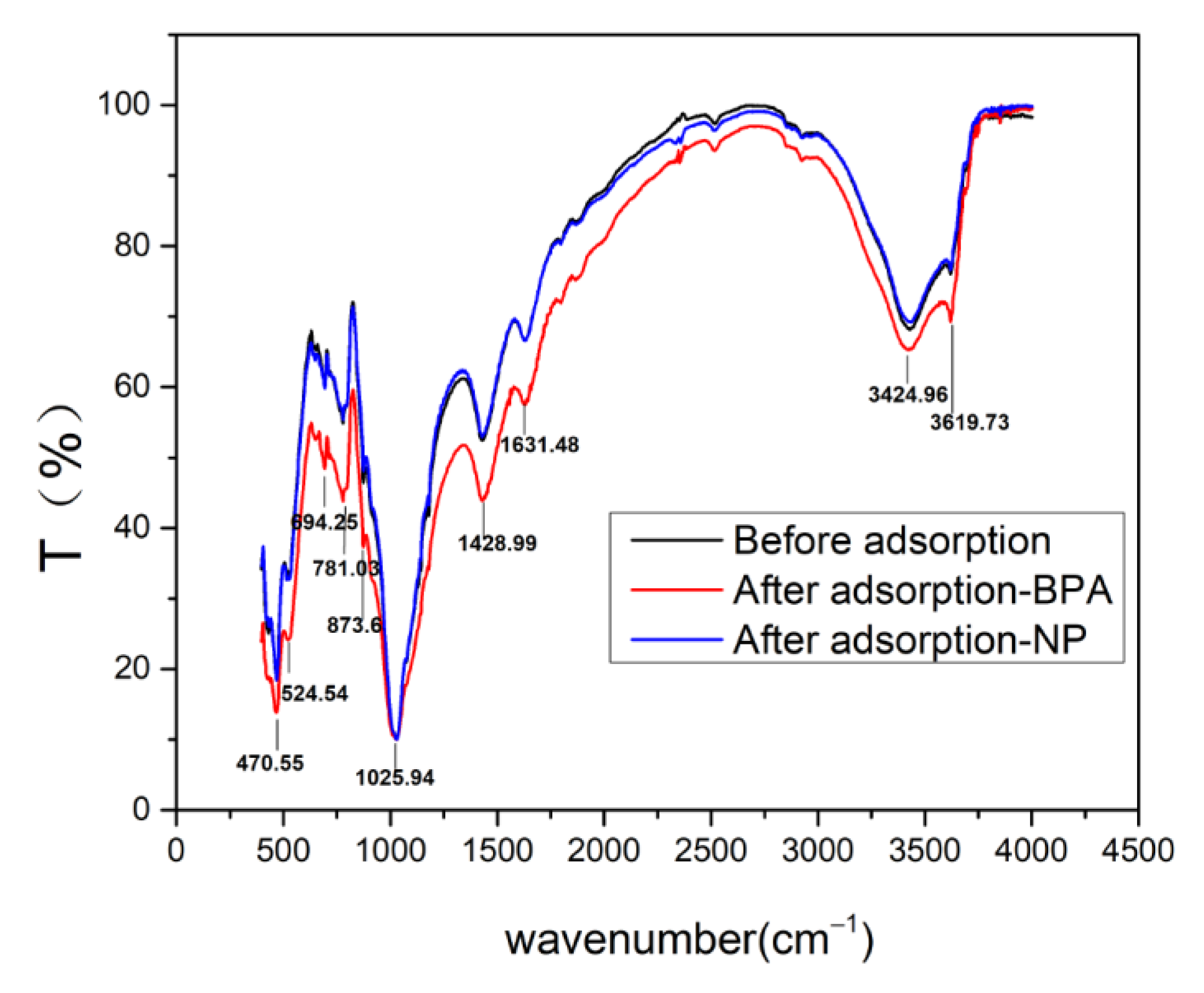
Soil before and after adsorption by NP and BPA were characterized by FTIR-650 with a deuterated triglycine sulfate (DTGS) detector. The instrument was under continuous dry air purge to eliminate atmospheric water vapor. Interferograms were averaged for 400–4000 scans at 4 cm−1 resolutions.
2.4.7. Statistical Analysis
IBM SPSS Statistics 22 software was used for the statistical analysis. The statistical differences were assessed using the one-way ANOVA test. The significance level of 0.05 and 0.001 was selected for all tests. In addition, different adsorption models and thermodynamic equations were used to fit the experimental data. The plotting and fitting of data were conducted using the Origin 9 software (OriginLab Corporation, Northampton, MA, USA).
4. Conclusions
This study, based on laboratory batch experiments, was successful in studying the thermodynamics of adsorption of NP and NP7 on reclaimed water-irrigated soils. The results indicated that the adsorptions of NP and NP7 on soil were in accordance with the Linear model, and the adsorption of BPA on soil was in accordance with the Freundlich model. The adsorptions of NP, NP7 and BPA on soils decreased with the increasing temperatures and pH. The adsorption capacity of NP, NP7 and BPA to soils was different with different cation valence: neutrally > divalent cations > mono- cations. The adsorption of NP7 on soil was quite different from the total NP, which was a further study of the previous study focusing on the total NP. Alkyl chains of NP and BPA seemed to form van der Waals interactions with the cavity of soil. But reclaimed water-irrigated soils are quite complex, and the mechanism of the differences between the total NP and individual isomer need further study. This study makes the risk assessment of NP isomers more targeted. In the future, researchers can assess the NP risks, not from the points of total NP but focusing on the isomer, which has higher estrogen activity. This study provided indispensable foundations for the human health risks of EDCs in the future.