Application of Internal Carbon Source from Sewage Sludge: A Vital Measure to Improve Nitrogen Removal Efficiency of Low C/N Wastewater
Abstract
:1. Introduction
2. Requirement of Carbon Source in Biological Nitrogen Removal Process
3. Conventional External Carbon Source
4. Internal Carbon Resources
4.1. Techniques Applied for VFAs Production from Sludge
4.1.1. Techniques Applied for VFAs Production from Primary Sludge
4.1.2. Techniques Applied for VFAs Production from Waste Activated Sludge
4.2. Techniques Applied for VFAs Production from Sludge
5. Conclusions
- How to produce as much as VFA as possible from PS or WAS with short sludge retention time and low cost
- As nearly all research has been conducted in the laboratory with intermittent flow, some important points have not been considered in continuous flow. Such points include the optimal ratio of sludge for producing a carbon source to sludge for recycling back to the main reaction flow, and into which reaction tank or in which period should the fermented sludge be added
- How microorganisms in the reaction tank will be affected after return of the internal carbon source.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Jin, L.; Zhang, G.; Tian, H. Current state of sewage treatment in China. Water Res. 2014, 66, 85–98. [Google Scholar] [CrossRef]
- Gao, Y.; Zhou, F.; Ciais, P.; Miao, C.; Yang, T.; Jia, Y.; Zhou, X.; Klaus, B.-B.; Yang, T.; Yu, G. Human activities aggravate nitrogen-deposition pollution to inland water over China. Natl. Sci. Rev. 2020, 7, 430–440. [Google Scholar] [CrossRef] [Green Version]
- Smith, V.H.; Tilman, G.D.; Nekola, J.C. Eutrophication: Impacts of excess nutrient inputs on freshwater, marine, and terrestrial ecosystems. Environ. Pollut. 1999, 100, 179–196. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Aber, J.D.; Howarth, R.W.; Likens, G.E.; Matson, P.A.; Schindler, D.W.; Schlesinger, W.H.; Tilman, D. Human alteration of the global nitrogen cycle: Sources and consequences. Ecol. Appl. 1997, 7, 737–750. [Google Scholar] [CrossRef] [Green Version]
- Van Loosdrecht, M.C.M.; Brdjanovic, D. Anticipating the next century of wastewater treatment. Science 2014, 344, 1452–1453. [Google Scholar] [CrossRef]
- Maroušek, J.; Maroušková, A. Economic Considerations on Nutrient Utilization in Wastewater Management. Energies 2021, 14, 3468. [Google Scholar] [CrossRef]
- Wagner, M.; Loy, A. Bacterial community composition and function in sewage treatment systems. Curr. Opin. Biotechnol. 2002, 13, 218–227. [Google Scholar] [CrossRef]
- Schmidt, I.; Sliekers, O.; Schmid, M.; Bock, E.; Fuerst, J.; Kuenen, J.G.; Jetten, M.S.M.; Strous, M. New concepts of microbial treatment processes for the nitrogen removal in wastewater. FEMS Microbiol. Rev. 2003, 27, 481–492. [Google Scholar] [CrossRef] [Green Version]
- Park, J.; Yoo, Y. Biological nitrate removal in industrial wastewater treatment: Which electron donor we can choose. Appl. Microbiol. Biotechnol. 2009, 82, 415–429. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Liu, S.; He, S.; Tian, Z.; Zheng, M. Minimization of N2O Emission through Intermittent Aeration in a Sequencing Batch Reactor (SBR): Main Behavior and Mechanism. Water 2021, 13, 210. [Google Scholar] [CrossRef]
- Caniani, D.; Esposito, G.; Gori, R.; Mannina, G. Towards A New Decision Support System for Design, Management and Operation of Wastewater Treatment Plants for the Reduction of Greenhouse Gases Emission. Water 2015, 7, 5599–5616. [Google Scholar] [CrossRef] [Green Version]
- Yang, G.; Zhang, G.; Wang, H. Current state of sludge production, management, treatment and disposal in China. Water Res. 2015, 78, 60–73. [Google Scholar] [CrossRef]
- Xiao, K.; Abbt-Braun, G.; Horn, H. Changes in the characteristics of dissolved organic matter during sludge treatment: A critical review. Water Res. 2020, 187, 116441. [Google Scholar] [CrossRef] [PubMed]
- Gallego-Schmid, A.; Tarpani, R.R.Z. Life cycle assessment of wastewater treatment in developing countries: A review. Water Res. 2019, 153, 63–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orhon, D. Evolution of the activated sludge process: The first 50 years. J. Chem. Technol. Biotechnol. 2015, 90, 608–640. [Google Scholar] [CrossRef]
- Ahn, Y.-H. Sustainable nitrogen elimination biotechnologies: A review. Process. Biochem. 2006, 41, 1709–1721. [Google Scholar] [CrossRef]
- Surampalli, R.Y.; Tyagi, R.D.; Scheible, O.K.; Heidman, J.A. Nitrification, denitrification and phosphorus removal in sequential batch reactors. Bioresour. Technol. 1997, 61, 151–157. [Google Scholar] [CrossRef]
- Strous, M.; Van Gerven, E.; Zheng, P.; Kuenen, J.G.; Jetten, M.S.M. Ammonium removal from concentrated waste streams with the anaerobic ammonium oxidation (anammox) process in different reactor configurations. Water Res. 1997, 31, 1955–1962. [Google Scholar] [CrossRef] [Green Version]
- Wagner, M.; Loy, A.; Nogueira, R.; Purkhold, U.; Lee, N.; Daims, H. Microbial community composition and function in wastewater treatment plants. Antonie Van Leeuwenhoek 2002, 81, 665–680. [Google Scholar] [CrossRef]
- Liwarska-Bizukojć, E.; Chojnacki, J.; Klink, M.; Olejnik, D. Effect of the type of the external carbon source on denitrification kinetics of wastewater. Desalination Water Treat. 2018, 101, 143–150. [Google Scholar] [CrossRef]
- Barker, P.S.; Dold, P.L. Cod and Nitrogen Mass Balances in Activated-Sludge Systems. Water Res. 1995, 29, 633–643. [Google Scholar] [CrossRef]
- Zhang, L.; Huang, Y.; Li, S.; He, P.; Wang, D. Optimization of Nitrogen Removal in Solid Carbon Source SND for Treatment of Low-Carbon Municipal Wastewater with RSM Method. Water 2018, 10, 827. [Google Scholar] [CrossRef] [Green Version]
- Seixo, J.; Varela, M.H.; Coutinho, J.A.P.; Coelho, M.A.Z. Influence of C/N ratio on autotrophic biomass development in a sequencing batch reactor. Biochem. Eng. J. 2004, 21, 131–139. [Google Scholar] [CrossRef]
- Roy, D.; Hassan, K.; Boopathy, R. Effect of carbon to nitrogen (C:N) ratio on nitrogen removal from shrimp production waste water using sequencing batch reactor. J. Ind. Microbiol. Biotechnol. 2010, 37, 1105–1110. [Google Scholar] [CrossRef]
- Gu, Y.; Wei, Y.; Xiang, Q.; Zhao, K.; Yu, X.; Zhang, X.; Li, C.; Chen, Q.; Xiao, H.; Zhang, X. C:N ratio shaped both taxonomic and functional structure of microbial communities in livestock and poultry breeding wastewater treatment reactor. Sci. Total Environ. 2019, 651, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Andreadakis, A.D. Nitrification-denitrification and activated sludge settlement. Environ. Technol. 1993, 14, 615–627. [Google Scholar] [CrossRef]
- Carrera, J.; Vicent, T.; Lafuente, J. Effect of influent COD/N ratio on biological nitrogen removal (BNR) from high-strength ammonium industrial wastewater. Process. Biochem. 2004, 39, 2035–2041. [Google Scholar] [CrossRef]
- Xia, S.; Li, J.; Wang, R. Nitrogen removal performance and microbial community structure dynamics response to carbon nitrogen ratio in a compact suspended carrier biofilm reactor. Ecol. Eng. 2008, 32, 256–262. [Google Scholar] [CrossRef]
- Fu, Z.; Yang, F.; Zhou, F.; Xue, Y. Control of COD/N ratio for nutrient removal in a modified membrane bioreactor (MBR) treating high strength wastewater. Bioresour. Technol. 2009, 100, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhang, J.; Jia, W.; Xie, H.; Gu, R.R.; Li, C.; Gao, B. Impact of COD/N ratio on nitrous oxide emission from microcosm wetlands and their performance in removing nitrogen from wastewater. Bioresour. Technol. 2009, 100, 2910–2917. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, M.; Bernat, K.; Cydzik-Kwiatkowska, A.; Sobolewska, J.; Wojnowska-Baryla, I. Nitrogen removal from wastewater and bacterial diversity in activated sludge at different COD/N ratios and dissolved oxygen concentrations. J. Environ. Sci. 2012, 24, 990–998. [Google Scholar] [CrossRef]
- Mohammed, R.N.; Abu-Alhail, S.; Wu, L.X. Intercross real-time control strategy in a novel phased isolation tank step feed process for treating low C/N real wastewater under ambient temperature. Korean J. Chem. Eng. 2014, 31, 1798–1809. [Google Scholar] [CrossRef]
- Wang, X.; Wang, S.; Xue, T.; Li, B.; Dai, X.; Peng, Y. Treating low carbon/nitrogen (C/N) wastewater in simultaneous nitrification-endogenous denitrification and phosphorous removal (SNDPR) systems by strengthening anaerobic intracellular carbon storage. Water Res. 2015, 77, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.P.; Nacher, C.P.I.; Merkey, B.; Zhou, Q.; Xia, S.Q.; Yang, D.H.; Sun, J.H.; Smets, B.F. Effective Biological Nitrogen Removal Treatment Processes for Domestic Wastewaters with Low C/N Ratios: A Review. Environ. Eng. Sci. 2010, 27, 111–126. [Google Scholar] [CrossRef] [Green Version]
- Manoharan, R.; Liptak, S.; Parkinson, P.; Mavinic, D.; Randall, C.W. Denitrific ation of a high ammonia leachate using an external carbon source. Environ. Technol. Lett. 1989, 10, 707–716. [Google Scholar] [CrossRef]
- Abu-Ghararah, Z.H. Biological denitrification of high nitrate water: Influence of type of carbon source and nitrate loading. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 1996, 31, 1651–1668. [Google Scholar] [CrossRef]
- Lee, M.W.; Park, J.M. Biological nitrogen removal from coke plant wastewater with external carbon addition. Water Environ. Res. 1998, 70, 1090–1095. [Google Scholar] [CrossRef]
- Mielcarek, A.; Rodziewicz, J.; Janczukowicz, W.; Dabrowska, D.; Ciesielski, S.; Thornton, A.; Struk-Sokolowska, J. Citric acid application for denitrification process support in biofilm reactor. Chemosphere 2017, 171, 512–519. [Google Scholar] [CrossRef]
- Mielcarek, A.; Rodziewicz, J.; Janczukowicz, W.; Struk-Sokolowska, J. The impact of biodegradable carbon sources on nutrients removal in post-denitrification biofilm reactors. Sci. Total Environ. 2020, 720, 137377. [Google Scholar] [CrossRef] [PubMed]
- Kłodowska, I.; Rodziewicz, J.; Janczukowicz, W.; Cydzik-Kwiatkowska, A.; Rusanowska, P. Influence of Carbon Source on the Efficiency of Nitrogen Removal and Denitrifying Bacteria in Biofilm from Bioelectrochemical SBBRs. Water 2018, 10, 393. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Zheng, Z.; Sun, F.; Miao, M.; Cui, M.-H.; Liu, H.; Zhang, H.; Zhang, C.; Hu, Z.; Liu, H. Valorization of citric acid production wastewater as alternative carbon source for biological nutrients removal: A pilot-scale case study. J. Clean. Prod. 2020, 258. [Google Scholar] [CrossRef]
- Quan, Z.X.; Jin, Y.S.; Yin, C.R.; Lee, J.J.; Lee, S.T. Hydrolyzed molasses as an external carbon source in biological nitrogen removal. Bioresour. Technol. 2005, 96, 1690–1695. [Google Scholar] [CrossRef] [PubMed]
- Dold, P.; Takács, I.; Mokhayeri, Y.; Nichols, A.; Hinojosa, J.; Riffat, R.; Bott, C.; Bailey, W.; Murthy, S. Denitrification with Carbon Addition—Kinetic Considerations. Water Environ. Res. 2008, 80, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Güven, D. Effects of Different Carbon Sources on Denitrification Efficiency Associated with Culture Adaptation and C/N Ratio. CLEAN Soil Air Water 2009, 37, 565–573. [Google Scholar] [CrossRef]
- Fernandez-Nava, Y.; Maranon, E.; Soons, J.; Castrillon, L. Denitrification of high nitrate concentration wastewater using alternative carbon sources. J. Hazard. Mater. 2010, 173, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Seon, J.; Byun, I.; Cho, S.; Park, T.; Lee, T. Comparison of nitrogen removal and microbial distribution in wastewater treatment process under different electron donor conditions. Bioresour. Technol. 2010, 101, 2988–2995. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Moon, T.; Park, S.; Choi, M.; Kim, C. Evaluation of industrial organic waste as an alternative external carbon source for denitrification in the biological nutrient removal process. Korean J. Chem. Eng. 2013, 30, 1911–1917. [Google Scholar] [CrossRef]
- Zhu, S.; Zheng, M.; Li, C.; Gui, M.; Chen, Q.; Ni, J. Special role of corn flour as an ideal carbon source for aerobic denitrification with minimized nitrous oxide emission. Bioresour. Technol. 2015, 186, 44–51. [Google Scholar] [CrossRef]
- Janczukowicz, W.; Rodziewicz, J.; Thornton, A.; Czaplicka, K. Effect of fermented wastewaters from butter production on phosphates removal in a sequencing batch reactor. Bioresour. Technol. 2012, 120, 34–39. [Google Scholar] [CrossRef]
- Mielcarek, A.; Rodziewicz, J.; Janczukowicz, W.; Dulski, T.; Ciesielski, S.; Thornton, A. Denitrification aided by waste beer in anaerobic sequencing batch biofilm reactor (AnSBBR). Ecol. Eng. 2016, 95, 384–389. [Google Scholar] [CrossRef]
- Guo, Y.; Guo, L.; Sun, M.; Zhao, Y.; Gao, M.; She, Z. Effects of hydraulic retention time (HRT) on denitrification using waste activated sludge thermal hydrolysis liquid and acidogenic liquid as carbon sources. Bioresour. Technol. 2017, 224, 147–156. [Google Scholar] [CrossRef]
- Guo, L.; Guo, Y.; Sun, M.; Gao, M.; Zhao, Y.; She, Z. Enhancing denitrification with waste sludge carbon source: The substrate metabolism process and mechanisms. Environ. Sci. Pollut. Res. Int. 2018, 25, 13079–13092. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, H.; Wang, Z.; Fang, Y.; Liu, Y.; Zeng, T.; Liu, Z.; Liu, M. Hydrocyclone breakage of activated sludge to exploit internal carbon sources and simultaneously enhance microbial activity. Process. Saf. Environ. Prot. 2018, 117, 651–659. [Google Scholar] [CrossRef]
- Liu, S.; Wang, Q.; Guan, Q.; Ning, P.; Luo, E. Rapid release of internal carbon source from excess sludge with synergistic treatment via thermophilic microaerobic digestion and microcurrent. Chem. Eng. J. 2019, 374, 637–647. [Google Scholar] [CrossRef]
- Gali, A.; Dosta, J.; Mata-Alvarez, J. Use of hydrolyzed primary sludge as internal carbon source for denitrification in a SBR treating reject water via nitrite. Ind. Eng. Chem. Res. 2006, 45, 7661–7666. [Google Scholar] [CrossRef]
- Aesoy, A.; Odegaard, H.; Bach, K.; Pujol, R.; Hamon, M. Denitrification in a packed bed biofilm reactor (BIOFOR)—Experiments with different carbon sources. Water Res. 1998, 32, 1463–1470. [Google Scholar] [CrossRef]
- Moser-Engeler, R.; Udert, K.M.; Wild, D.; Siegrist, H. Products from primary sludge fermentation and their suitability for nutrient removal. Water Sci. Technol. 1998, 38, 265–273. [Google Scholar] [CrossRef]
- Soares, A.; Kampas, P.; Maillard, S.; Wood, E.; Brigg, J.; Tillotson, M.; Parsons, S.A.; Cartmell, E. Comparison between disintegrated and fermented sewage sludge for production of a carbon source suitable for biological nutrient removal. J. Hazard. Mater. 2010, 175, 733–739. [Google Scholar] [CrossRef] [Green Version]
- Kampas, P.; Parsons, S.A.; Pearce, P.; Ledoux, S.; Vale, P.; Cartmell, E.; Soares, A. An internal carbon source for improving biological nutrient removal. Bioresour. Technol. 2009, 100, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Nam, Y.K.; Park, C.; Lee, M. Carbon source recovery from waste activated sludge by alkaline hydrolysis and gamma-ray irradiation for biological denitrification. Bioresour. Technol. 2009, 100, 5694–5699. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.-S.; Parameswaran, P.; Alder, J.M.; Rittmann, B.E. Feasibility of Focused-Pulsed Treated Waste Activated Sludge as a Supplemental Electron Donor for Denitrification. Water Environ. Res. 2010, 82, 2316–2324. [Google Scholar] [CrossRef]
- Chen, Y.; Jiang, S.; Yuan, H.; Zhou, Q.; Gu, G. Hydrolysis and acidification of waste activated sludge at different pHs. Water Res. 2007, 41, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.; Chen, Y. Recovery of nitrogen and phosphorus from alkaline fermentation liquid of waste activated sludge and application of the fermentation liquid to promote biological municipal wastewater treatment. Water Res. 2009, 43, 2969–2976. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, K.; Su, Y.; Zheng, X.; Wang, Q. Continuous bioproduction of short-chain fatty acids from sludge enhanced by the combined use of surfactant and alkaline pH. Bioresour. Technol. 2013, 140, 97–102. [Google Scholar] [CrossRef]
- Su, G.; Huo, M.; Yuan, Z.; Wang, S.; Peng, Y. Hydrolysis, acidification and dewaterability of waste activated sludge under alkaline conditions: Combined effects of NaOH and Ca(OH)2. Bioresour. Technol. 2013, 136, 237–243. [Google Scholar] [CrossRef]
- Vlyssides, A. Thermal-alkaline solubilization of waste activated sludge as a pre-treatment stage for anaerobic digestion. Bioresour. Technol. 2004, 91, 201–206. [Google Scholar] [CrossRef]
- Pang, L.; Ni, J.; Tang, X.; Chen, Q. Short-cut waste activated sludge fermentation and application of fermentation liquid to improve heterotrophic aerobic nitrogen removal by Agrobacterium sp. LAD9. Chem. Eng. J. 2015, 259, 911–917. [Google Scholar] [CrossRef]
- Zheng, X.; Su, Y.; Li, X.; Xiao, N.; Wang, D.; Chen, Y. Pyrosequencing reveals the key microorganisms involved in sludge alkaline fermentation for efficient short-chain fatty acids production. Environ. Sci. Technol. 2013, 47, 4262–4268. [Google Scholar] [CrossRef] [PubMed]
- Arnaiz, C.; Gutierrez, J.C.; Lebrato, J. Biomass stabilization in the anaerobic digestion of wastewater sludges. Bioresour. Technol. 2006, 97, 1179–1184. [Google Scholar] [CrossRef] [PubMed]
- Cokgor, E.U.; Oktay, S.; Tas, D.O.; Zengin, G.E.; Orhon, D. Influence of pH and temperature on soluble substrate generation with primary sludge fermentation. Bioresour. Technol. 2009, 100, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Ahn, Y.H.; Speece, R.E. Elutriated acid fermentation of municipal primary sludge. Water Res. 2006, 40, 2210–2220. [Google Scholar] [CrossRef]
- Bouzas, A.; Ribes, J.; Ferrer, J.; Seco, A. Fermentation and elutriation of primary sludge: Effect of SRT on process performance. Water Res. 2007, 41, 747–756. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.; Gavala, H.N.; Skiadas, I.V.; Mladenovska, Z.; Ahring, B.K. Improving anaerobic sewage sludge digestion by implementation of a hyper-thermophilic prehydrolysis step. J. Environ. Manag. 2008, 88, 881–889. [Google Scholar] [CrossRef]
- Ge, H.; Jensen, P.D.; Batstone, D.J. Pre-treatment mechanisms during thermophilic-mesophilic temperature phased anaerobic digestion of primary sludge. Water Res. 2010, 44, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, S.; Wang, S.; Wu, C.; Chen, Y.; Wang, Y.; Peng, Y. Enhanced biological nutrient removal in a simultaneous fermentation, denitrification and phosphate removal reactor using primary sludge as internal carbon source. Chemosphere 2013, 91, 635–640. [Google Scholar] [CrossRef]
- Eastman, J.A.; Ferguson, J.F. Solubilization of Particulate Organic-Carbon During The Acid Phase Of Anaerobic-Digestion. J. Water Pollut. Control. Fed. 1981, 53, 352–366. [Google Scholar]
- Canziani, R.; Pollice, A.; Ragazzi, M. Design considerations on primary sludge hydrolysis under psychrophilic conditions. Environ. Technol. 1996, 17, 747–754. [Google Scholar] [CrossRef]
- Zubrowska-Sudol, M.; Walczak, J. Enhancing combined biological nitrogen and phosphorus removal from wastewater by applying mechanically disintegrated excess sludge. Water Res. 2015, 76, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Chen, Y. Simultaneous Nitrogen and Phosphorus Recovery from Sludge-Fermentation Liquid Mixture and Application of the Fermentation Liquid to Enhance Municipal Wastewater Biological Nutrient Removal. Environ. Sci. Technol. 2009, 43, 6164–6170. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.; Chen, Y. Using Sludge Fermentation Liquid to Improve Wastewater Short-Cut Nitrification-Denitrification and Denitrifying Phosphorus Removal via Nitrite. Environ. Sci. Technol. 2010, 44, 8957–8963. [Google Scholar] [CrossRef]
- Li, X.; Chen, H.; Hu, L.; Yu, L.; Chen, Y.; Gu, G. Pilot-scale waste activated sludge alkaline fermentation, fermentation liquid separation, and application of fermentation liquid to improve biological nutrient removal. Environ. Sci. Technol. 2011, 45, 1834–1839. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wan, C.; Lee, D.J.; Du, M.; Pan, X.; Wan, F. Continuous volatile fatty acid production from waste activated sludge hydrolyzed at pH 12. Bioresour. Technol. 2014, 168, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Feng, L.; Chen, Y.; Li, X.; Chen, H.; Xiao, N.; Wang, D. Stimulating short-chain fatty acids production from waste activated sludge by nano zero-valent iron. J. Biotechnol. 2014, 187, 98–105. [Google Scholar] [CrossRef]
- Liu, K.; Chen, Y.; Xiao, N.; Zheng, X.; Li, M. Effect of humic acids with different characteristics on fermentative short-chain Fatty acids production from waste activated sludge. Environ. Sci. Technol. 2015, 49, 4929–4936. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Feng, L.; Chen, Y.; Sun, H.; Shen, Q.; Li, X.; Chen, H. Alkyl polyglucose enhancing propionic acid enriched short-chain fatty acids production during anaerobic treatment of waste activated sludge and mechanisms. Water Res. 2015, 73, 332–341. [Google Scholar] [CrossRef]
- Wang, B.; Wang, S.; Li, B.; Peng, C.; Peng, Y. Integrating waste activated sludge (WAS) acidification with denitrification by adding nitrite (NO2−). Biomass Bioenergy 2014, 67, 460–465. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, D.; Li, X.; Yang, Q.; Chen, H.; Zhong, Y.; Zeng, G. Free nitrous acid serving as a pretreatment method for alkaline fermentation to enhance short-chain fatty acid production from waste activated sludge. Water Res. 2015, 78, 111–120. [Google Scholar] [CrossRef] [Green Version]
- Yuan, H.Y.; Chen, Y.G.; Zhang, H.X.; Jiang, S.; Zhou, Q.; Gu, G.W. Improved bioproduction of short-chain fatty acids (SCFAs) from excess sludge under alkaline conditions. Environ. Sci. Technol. 2006, 40, 2025–2029. [Google Scholar] [CrossRef]
- Law, Y.; Ye, L.; Wang, Q.; Hu, S.; Pijuan, M.; Yuan, Z. Producing free nitrous acid—A green and renewable biocidal agent—From anaerobic digester liquor. Chem. Eng. J. 2015, 259, 62–69. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Ye, L.; Jiang, G.; Hu, S.; Yuan, Z. Side-stream sludge treatment using free nitrous acid selectively eliminates nitrite oxidizing bacteria and achieves the nitrite pathway. Water Res. 2014, 55, 245–255. [Google Scholar] [CrossRef]
- Wang, Q.; Ye, L.; Jiang, G.; Yuan, Z. A free nitrous acid (FNA)-based technology for reducing sludge production. Water Res. 2013, 47, 3663–3672. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Pijuan, M.; Fux, C.; Yuan, Z.; Keller, J. The role of nitrite/free nitrous acid on N2O production during denitrification. In Proceedings of the 6th IWA World Water Congress, Vienna, Austria, 7–12 September 2008. [Google Scholar]
- Du, F.; Freguia, S.; Yuan, Z.; Keller, J.; Pikaar, I. Enhancing toxic metal removal from acidified sludge with nitrite addition. Environ. Sci. Technol. 2015, 49, 6257–6263. [Google Scholar] [CrossRef] [PubMed]
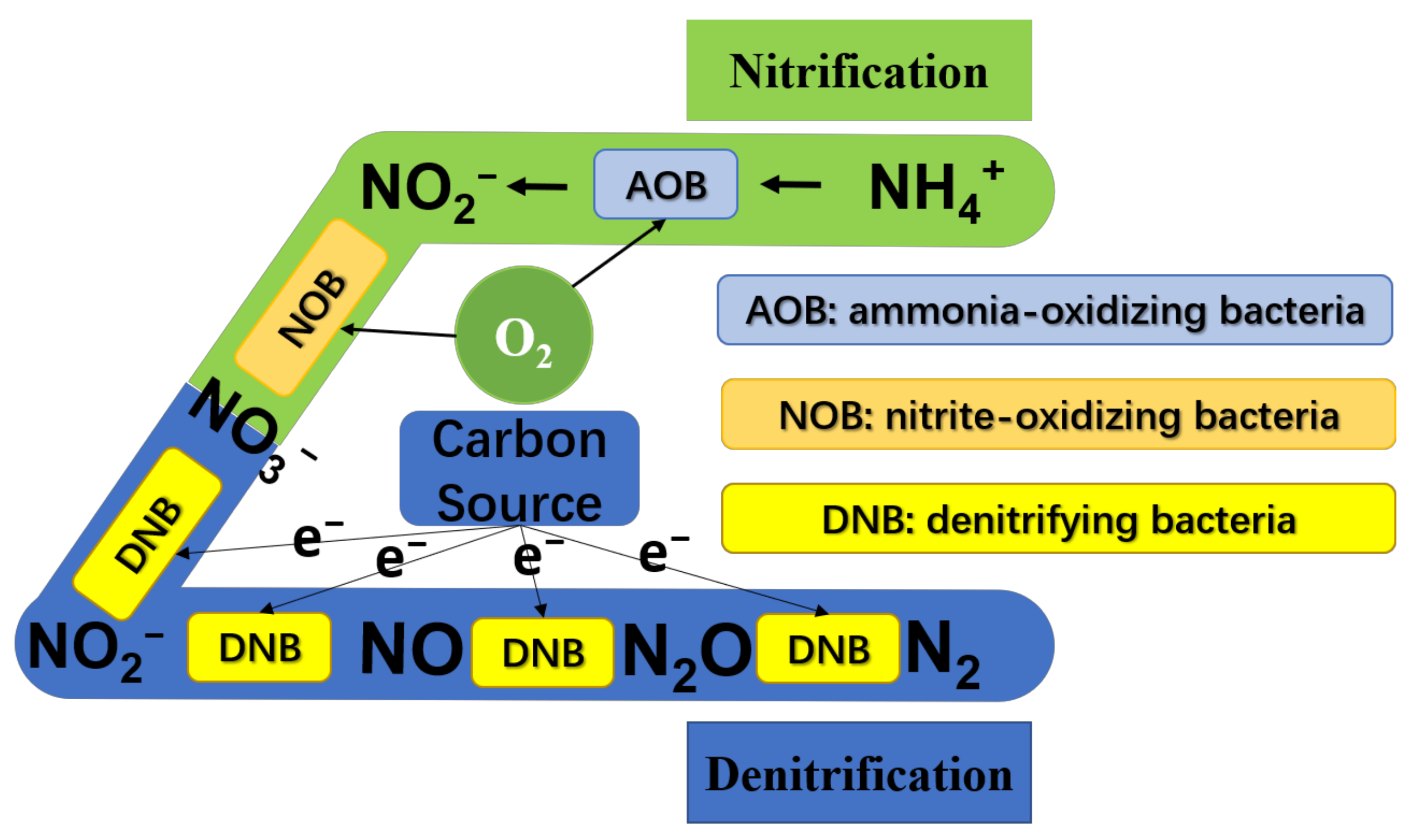
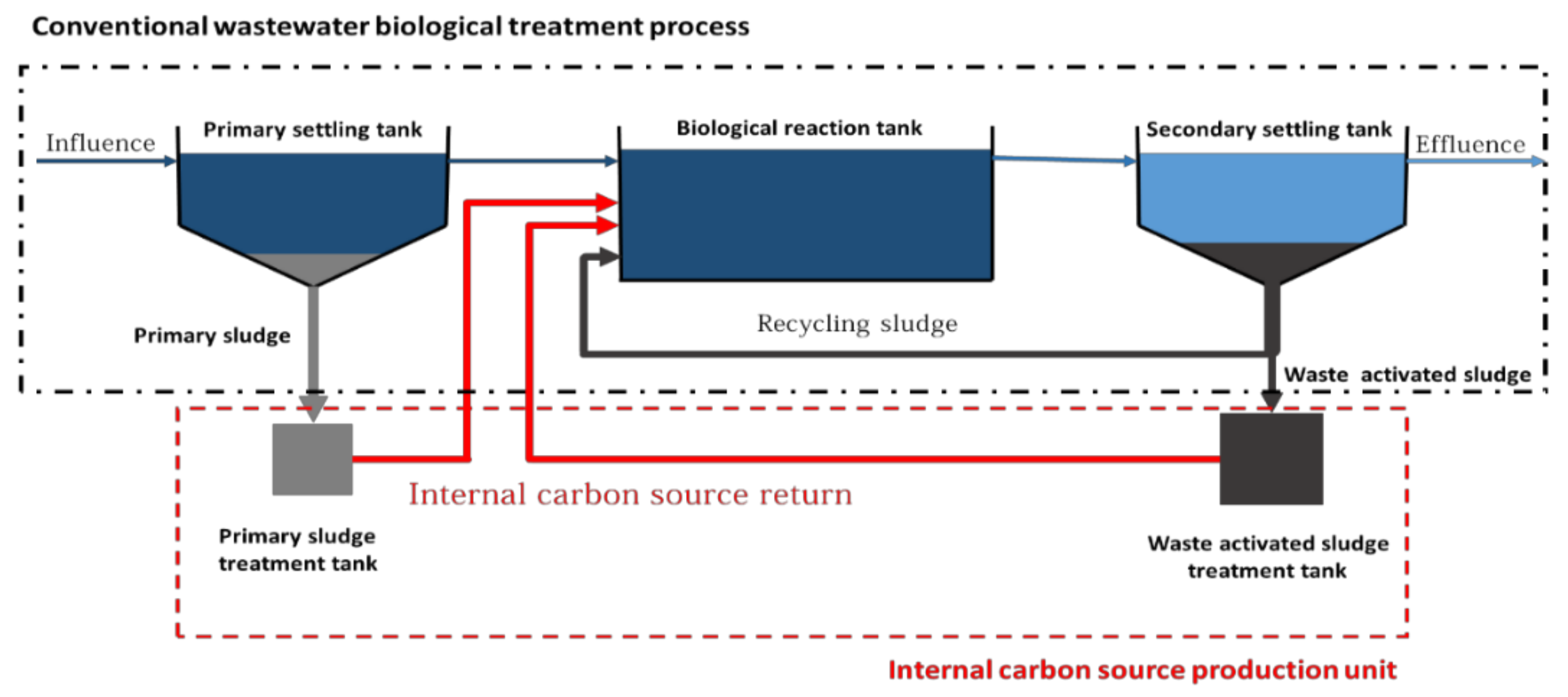
| Process | Wastewater Type | Operational Condition | Optimal C/N | Nitrogen Removal Efficiency (%) | References |
|---|---|---|---|---|---|
| Anoxic-aeration reactor | Sewage | Anoxic-aeration | 5 | 42.3 | [26] |
| Modified Ludzack-Ettinger process | Industrial w/w | Anoxic-aerobic-aerobic | 7.1 | - | [27] |
| SBR | Synthetic | Anoxic-aerobic | 11.0 | 99.6 | [23] |
| SCBR a | Synthetic | Aeration | 5 | 78.4 | [28] |
| Modified MBR | Synthetic | Anoxic-aerobic | 9.3 | 90.6 | [29] |
| Microcosm wetlands | Synthetic | Not given | 5 | 63.83 | [30] |
| SBR | Shrimp w/w | Aerobic-anaerobic-aerobic | 10 | 99 | [24] |
| SBR | Synthetic | Aeration, DO = 0.5 mg/L. | 6.8 | 50.3 | [31] |
| SBBR b | Livestock and poultry breeding wastewater | Aerobic-anaerobic-aerobic-anaerobic | 18.9 | ~96 | [25] |
| PITSF c system | Sewage | Intercross real-time control system | 4.3 | 83.2 | [32] |
| SNDPR-SBR d | Sewage | Anaerobic-aerobic | 3.5 | 77.7 | [33] |
| Sludge Treatment Technique | Carbon Source Production | References |
|---|---|---|
| Thermal-alkaline (pH = 9, T = 55 °C) | 0.18 g VFA a/g VSSCOD | [71] |
| Fermentation–elutriation (SRT b = 6 days, RSFR c = 4.5 L/h) | 36 mg/L HAc d | [72] |
| Hyper-thermophilic (70 °C, HRT = 2 days) hydrolysis followed by thermophilic (55 °C, HRT = 13 days) anaerobic digestion | 76 mg/L VFAs | [73] |
| Fermentation (T = 24 °C without pH control) | 2950 mg/L VFAs | [70] |
| Thermophilic–mesophilic anaerobic digestion (50–70 °C) | 3300 mg/L VFAs | [74] |
| Fermentation (T = 30 °C without pH control) | 0.99 gCOD/gVSS−1 | [75] |
| Treatment Technique | Treatment Condition | Internal Carbon Resource Production | Nitrogen Removal Performance | References |
|---|---|---|---|---|
| Mechanical disintegration | Disintegrated with a deflakerfor for 2–15 min | 530 mg/L VFA a | Denitrification rate was increased by 6.5 mg NO3-N g−1/VSS h−1 compared with the control vessel (30.2 mg NO3-N g−1/VSS h−1) | [59] |
| Focused-pulsed treatment | Treatment intensity = 28 kWh/m3 | 397 mg/L SCOD b | Maximum denitrification rate (0.25 g NO3-N g−1/VSS d−1) was greater than foruntreated WAS 0.05 g NO3-N g−1/VSS d−1) and methanol (0.15 g NO3-N g−1/VSS d−1) | [61] |
| Mechanical disintegration | Multi-use rotor driven by a motor with a power of P = 2 kw, revolutions n = 2800 r/min | 218.13 mg/L VFA | Denitrification efficiency increased from 49.2% to 76.2% | [78] |
| Alkaline fermentation | T = 21 °C, pH = 10, t = 8 days | 4225 mg/L SCFA c | TN removal efficiency increased to 83% compared to the control group, which was 63% | [79] |
| Alkaline fermentation | T = 20–22 °C, pH = 10, t = 8 days | 4189 mg/L SCFA | TN removal efficiency increased to 83.2% with the optimal volume ratio of fermentation liquid to municipal sewage equal to 1/35, compared to the control group, which was 63.3% | [63] |
| Alkaline fermentation | T = 20–22 °C, pH = 10, t = 8 days | 4035 mg/L BOD | TN removal efficiencies of sludge fermentation liquid were much higher than with acetic acid (98.7% versus 79.2%). | [80] |
| Alkaline fermentation | pH = 10, t = 8 days | 2480 mg/L SCFA | TN removal efficiencies of sludge fermentation liquid were much higher than control group (74.7% versus 27.1%) | [81] |
| Alkaline fermentation | T = 35 °C, pH = 12, t = 8 h | 365 mg VFAs g−1VSS 0.66 SCOD mg/mg TCOD d | NO3-N removal efficiency of acid fermentation liquor showed slight differences with that of sodium propionate, and the former had more stable performance than the latter. | [82] |
| Anaerobic fermentation with nano zero-valent iron addition | T = 20 °C without pH control, t = 4 days, 5.0 g/L nZVI e | 1307.8 mg/L SCFA | Not given | [83] |
| Anaerobic fermentation with humic acids addition (SHHA and SAHA) | T = 35 °C, pH = 7.0, t = 9 days, 1.0 g/g TCOD | 2741 mg/L SCFA | Not given | [84] |
| Anaerobic fermentation with alkyl polyglucose addition | T = 25 °C without pH control, t = 4 days, 0.3 g APG/g TSS | 2988 mg/L SCFA | Not given | [85] |
| Anaerobic fermentation with surfactant (SDBS) addition | T = 21 °C, pH = 10, t = 12 days | 2056 mg /L SCFA | Not given | [64] |
| Fermentation with nitrite (NO2−) | T = 35 °C, pH = 5, t = 28 days, 0.1 g NO2-N L−1 per day | 4500 mg/L SCFA | Not given | [86] |
| Fermentation with free nitrous acid addition | T = 20 °C, pH = 10,t = 2 days, 1.54 mg FNA f/L | 370.1 mg/g SCFA | Not given | [87] |
| Thermal-alkaline pretreated anaerobic digestion | T = 90 °C, pH = 11, t = 10 h | 69000 mg/L SCOD | Not given | [66] |
| Thermal-alkaline pretreated anaerobic digestion | T = 60 °C, pH = 12, t = 12 h | 3078 mg/L SCOD, 1096 mg/L VFA | TN removal efficiency within 12h was 51.6% higher than control group, which was 35.6% | [67] |
| Alkaline fermentation and gamma-ray irradiation | T = 25 °C, pH = 10, gamma-ray irradiation = 20 kGy | 2980 mg/L | Max TN removal efficiency with solubilized sludge as carbon source was 51.1%, similar to that of methanol, which was 55.6% | [60] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Jiang, C.; Wang, X.; Xu, S.; Zhuang, X. Application of Internal Carbon Source from Sewage Sludge: A Vital Measure to Improve Nitrogen Removal Efficiency of Low C/N Wastewater. Water 2021, 13, 2338. https://doi.org/10.3390/w13172338
Wang H, Jiang C, Wang X, Xu S, Zhuang X. Application of Internal Carbon Source from Sewage Sludge: A Vital Measure to Improve Nitrogen Removal Efficiency of Low C/N Wastewater. Water. 2021; 13(17):2338. https://doi.org/10.3390/w13172338
Chicago/Turabian StyleWang, Huacai, Cancan Jiang, Xu Wang, Shengjun Xu, and Xuliang Zhuang. 2021. "Application of Internal Carbon Source from Sewage Sludge: A Vital Measure to Improve Nitrogen Removal Efficiency of Low C/N Wastewater" Water 13, no. 17: 2338. https://doi.org/10.3390/w13172338
APA StyleWang, H., Jiang, C., Wang, X., Xu, S., & Zhuang, X. (2021). Application of Internal Carbon Source from Sewage Sludge: A Vital Measure to Improve Nitrogen Removal Efficiency of Low C/N Wastewater. Water, 13(17), 2338. https://doi.org/10.3390/w13172338








