Application of Two Indigenous Strains of Microalgal Chlorella sorokiniana in Cassava Biogas Effluent Focusing on Growth Rate, Removal Kinetics, and Harvestability
Abstract
:1. Introduction
2. Materials and Methods
2.1. Wastewater Source
2.2. Microalgal Isolation and Screening
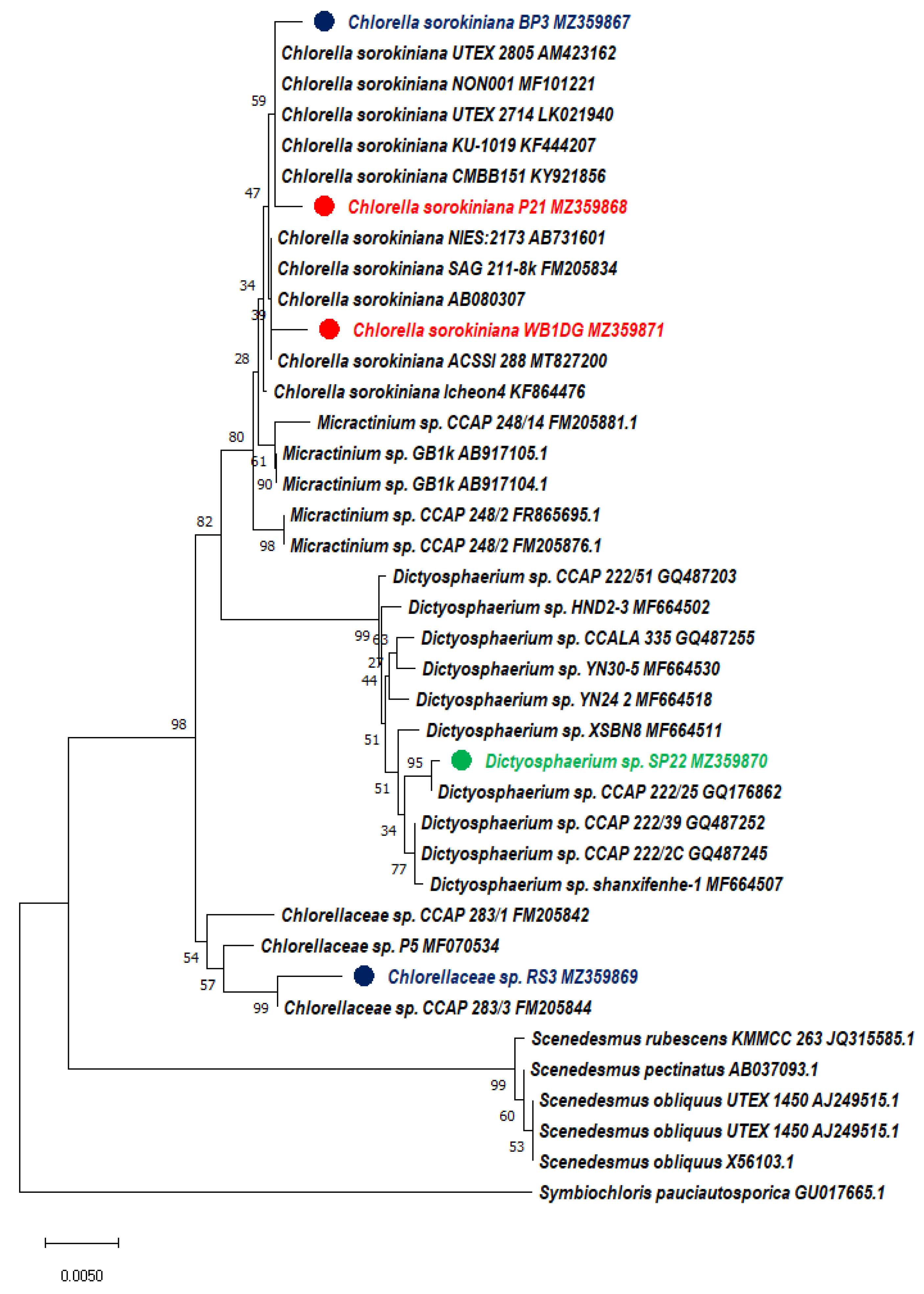
2.3. Morphological and Molecular Identification
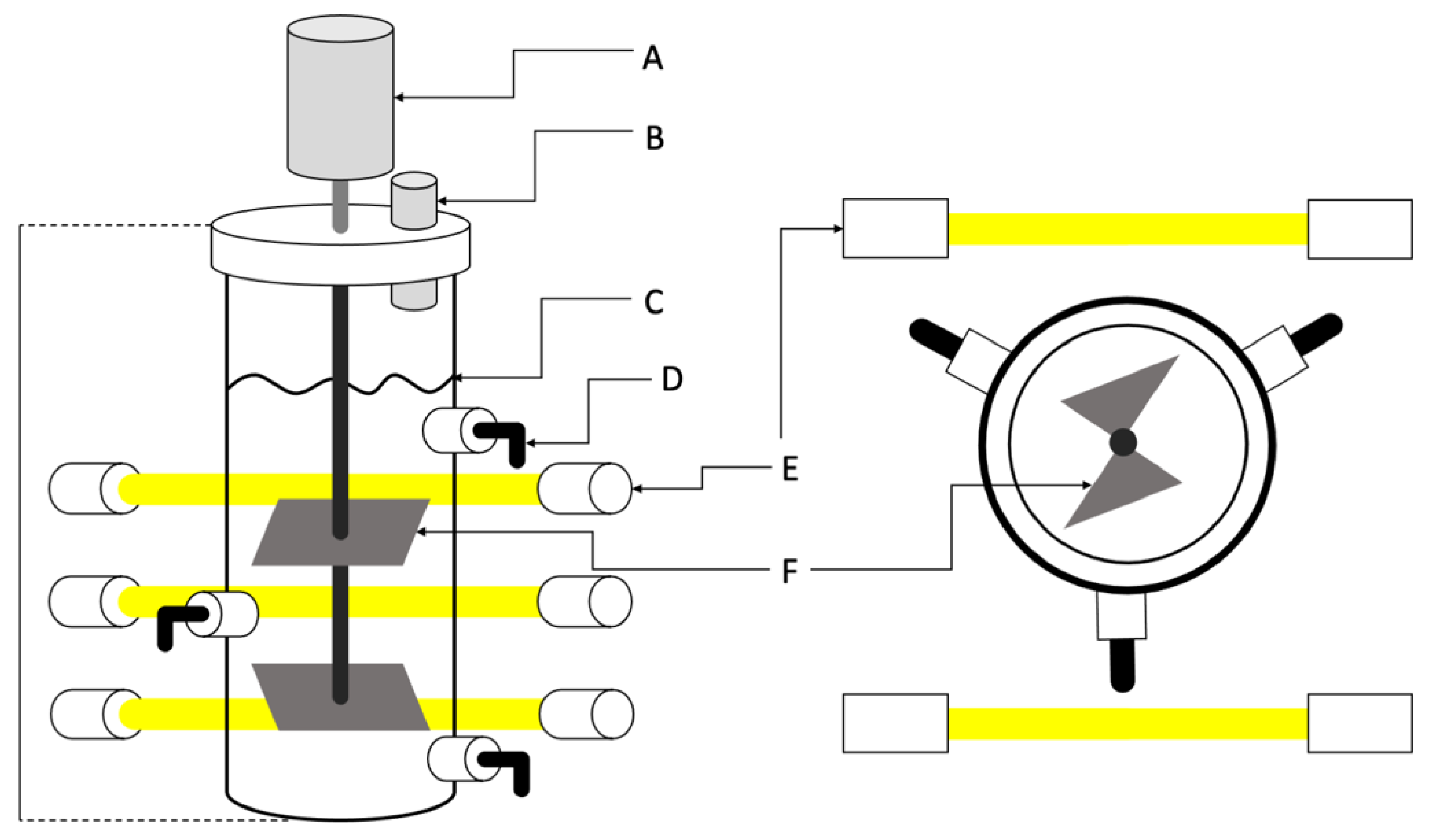
2.4. Cultivation Conditions and Biomass Generation
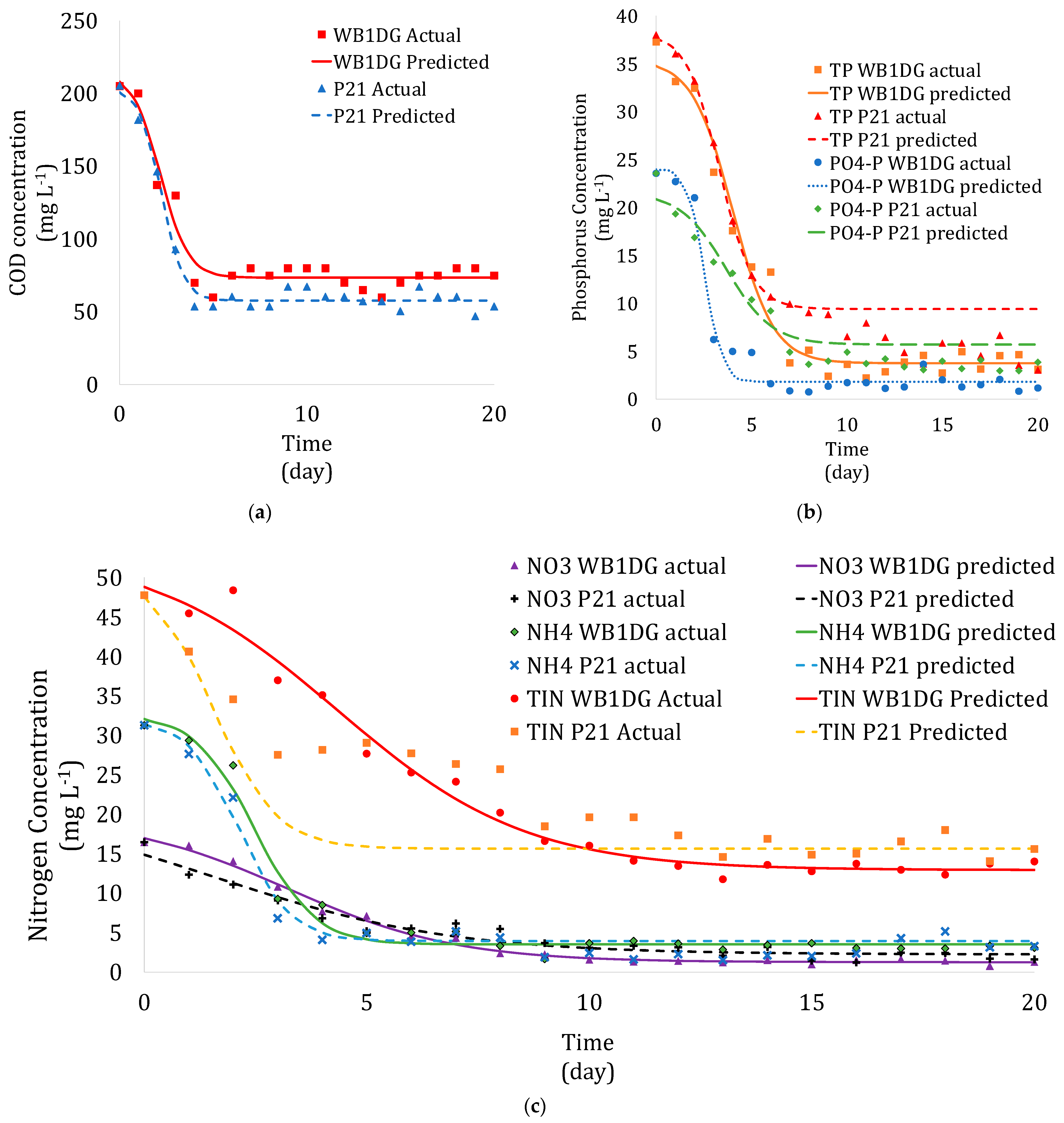
2.5. Kinetics Analysis
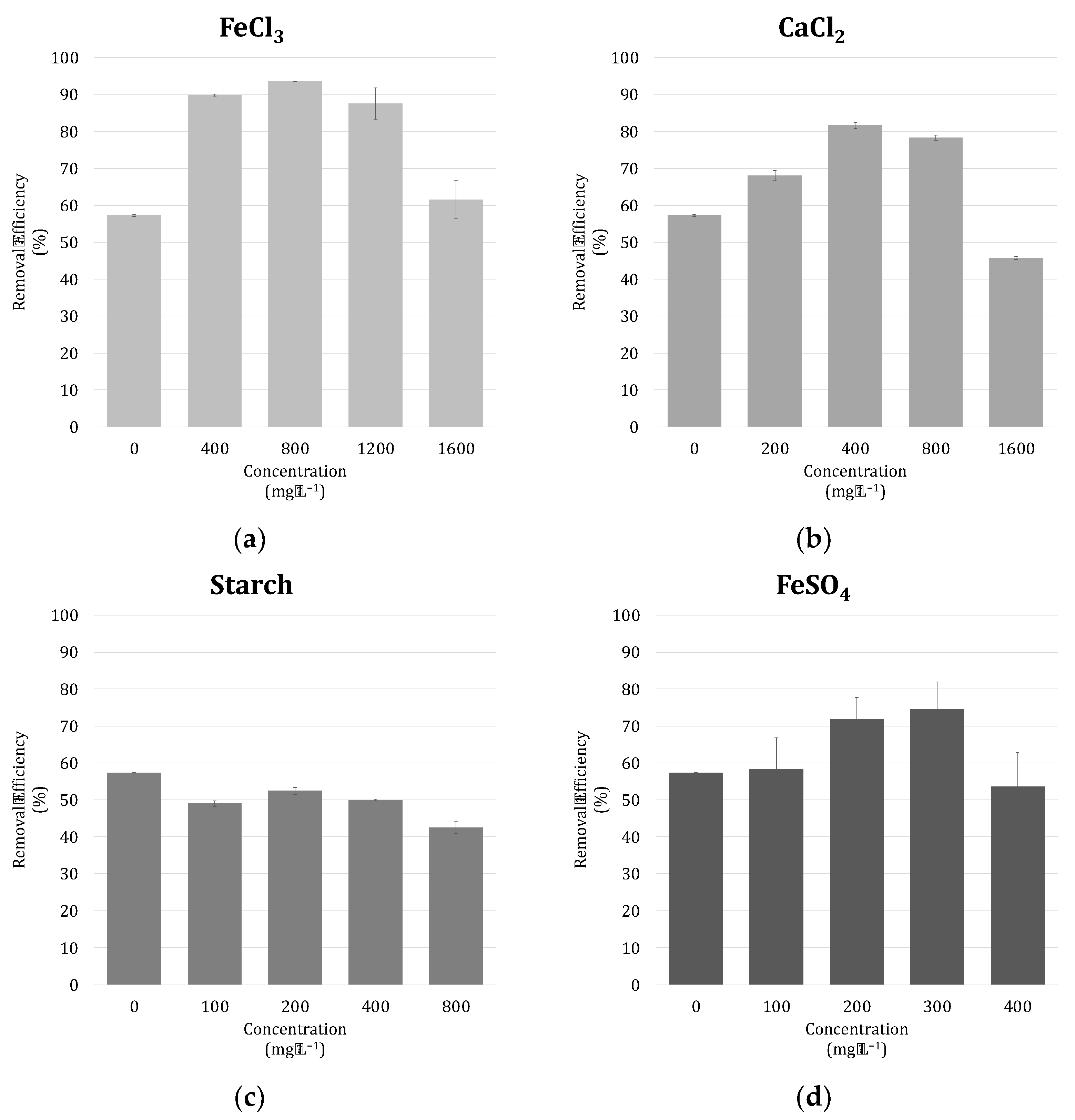
2.6. Harvestability Tests
2.7. Statistical Analyses
3. Results and Discussion
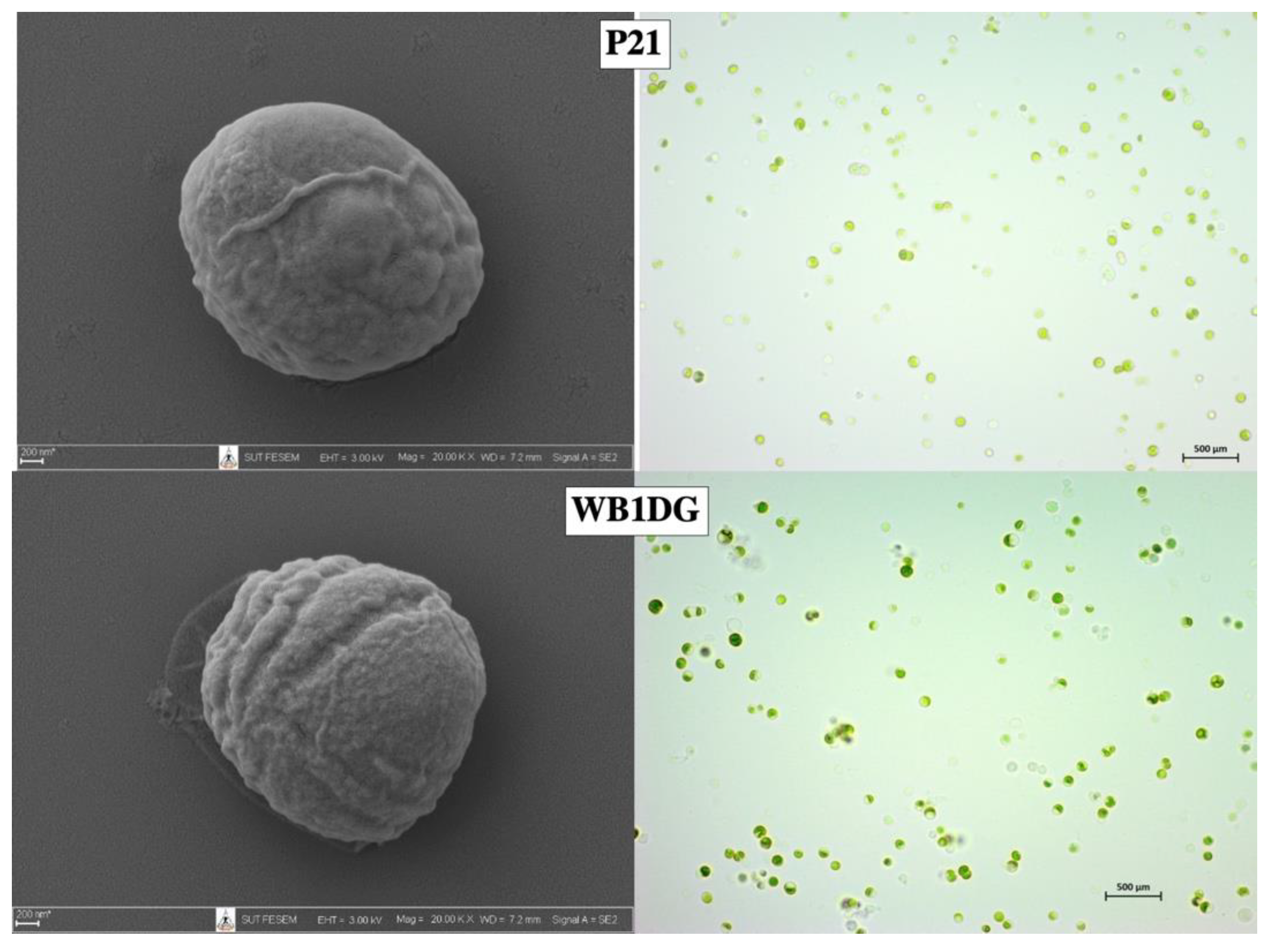
3.1. Isolation and Screening of Microalgae
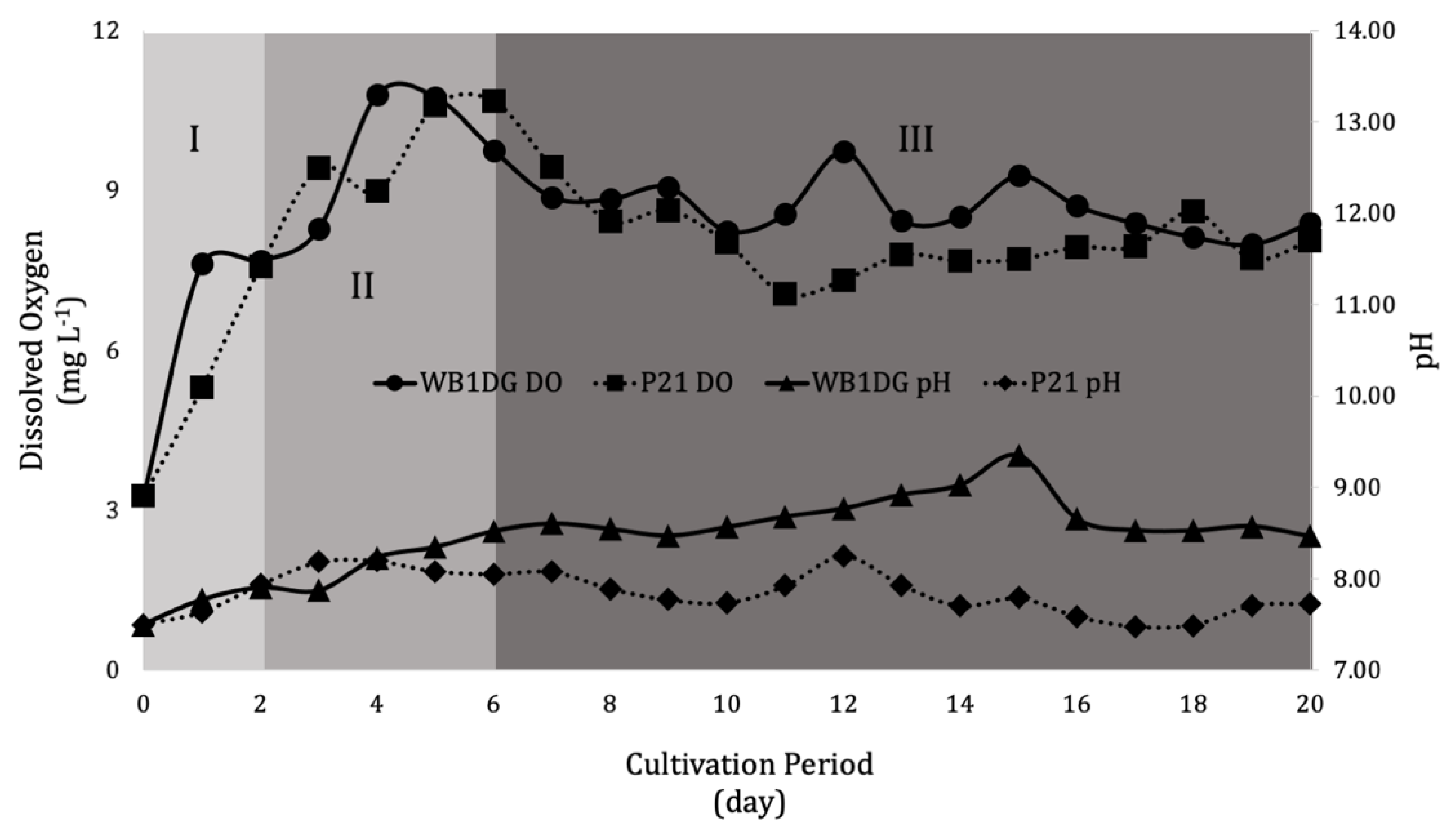
3.2. Culture Conditions during Cultivation
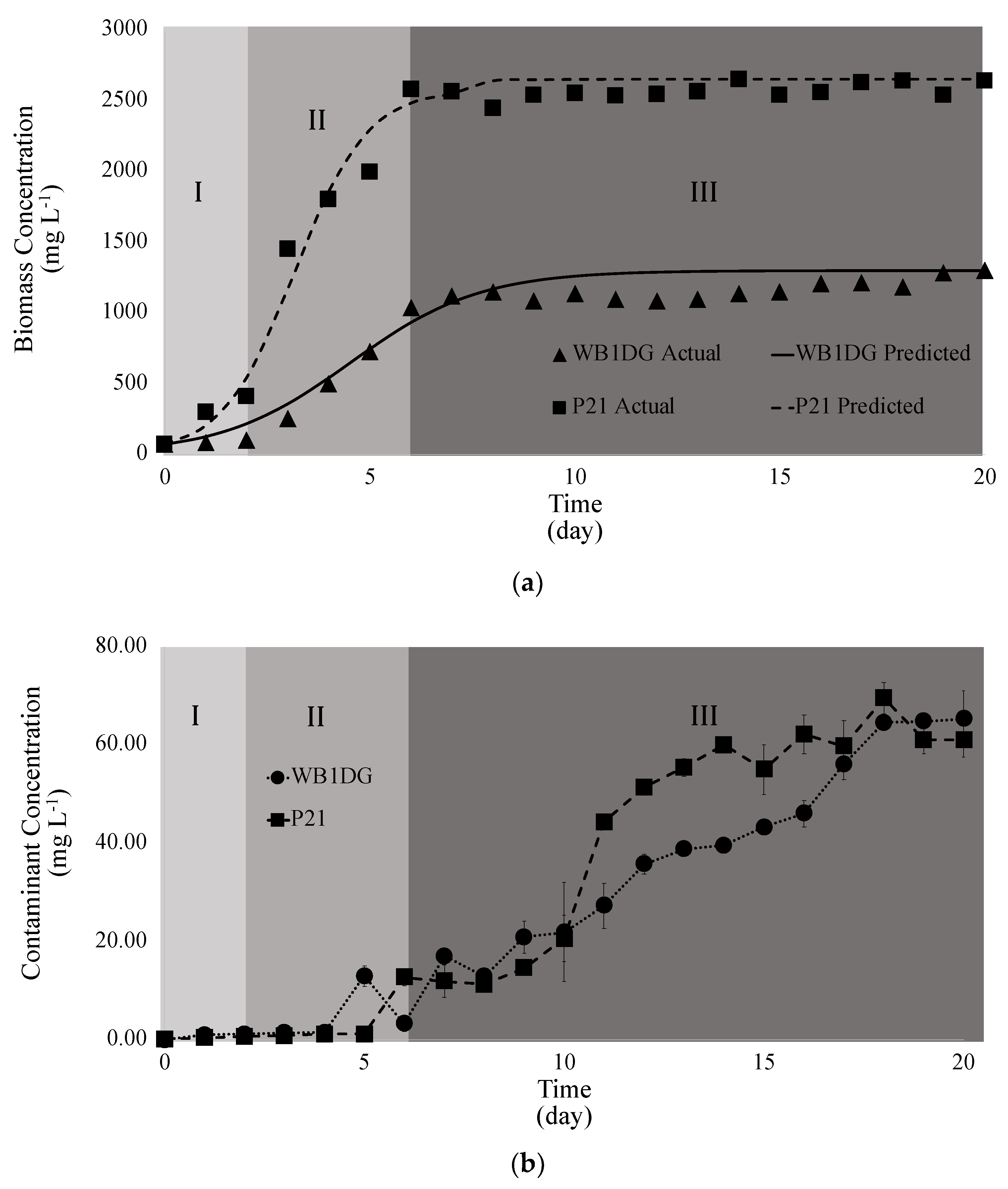
3.3. Kinetics of Microalgae Growth in Wastewater
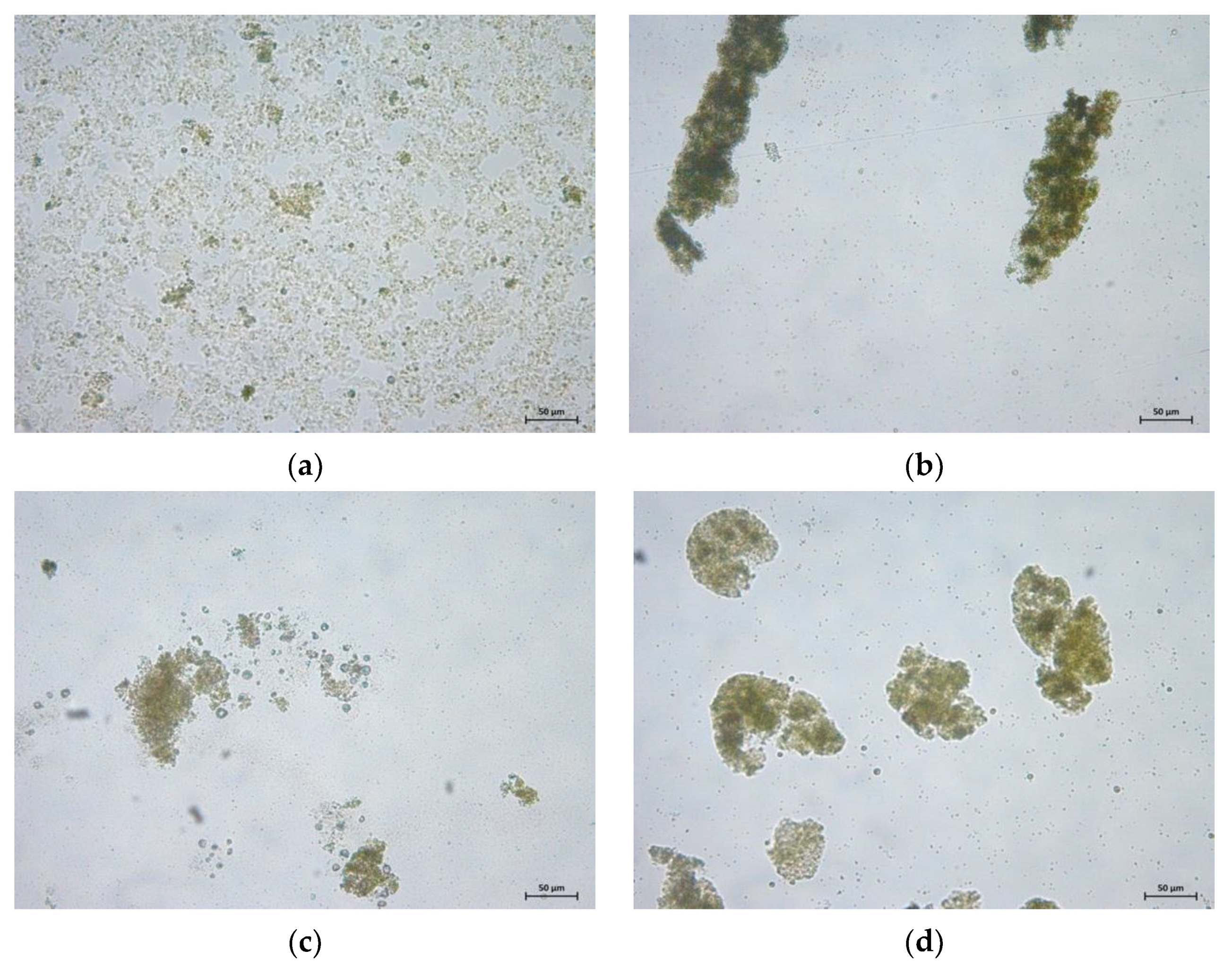
3.4. Harvestability of Microalgae
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jiraprasertwong, A.; Maitriwong, K.; Chavadej, S. Production of biogas from cassava wastewater using a three-stage upflow anaerobic sludge blanket (UASB) reactor. Renew. Energy 2019, 130, 191–205. [Google Scholar] [CrossRef]
- Sreethawong, T.; Chatsiriwatana, S.; Rangsunvigit, P.; Chavadej, S. Hydrogen production from cassava wastewater using an anaerobic sequencing batch reactor: Effects of operational parameters, COD:N ratio, and organic acid composition. Int. J. Hydrog. Energy 2010, 35, 4092–4102. [Google Scholar] [CrossRef]
- Pingmuanglek, P.; Jakrawatana, N.; Gheewala, S.H. Supply chain analysis for cassava starch production: Cleaner production opportunities and benefits. J. Clean. Prod. 2017, 162, 1075–1084. [Google Scholar] [CrossRef]
- Tuszynska, A.; Wilinska, A.; Czerwionka, K. Phosphorus and nitrogen forms in liquid fraction of digestates from agricultural biogas plants. Environ. Technol. 2020, 1–13. [Google Scholar] [CrossRef]
- Wandera, S.M.; Qiao, W.; Algapani, D.E.; Bi, S.; Yin, D.; Qi, X.; Liu, Y.; Dach, J.; Dong, R. Searching for possibilities to improve the performance of full scale agricultural biogas plants. Renew. Energy 2018, 116, 720–727. [Google Scholar] [CrossRef]
- Zeppilli, M.; Mattia, A.; Villano, M.; Majone, M. Three-chamber Bioelectrochemical System for Biogas Upgrading and Nutrient Recovery. Fuel Cells 2017, 17, 593–600. [Google Scholar] [CrossRef]
- Colin, X.; Farinet, J.L.; Rojas, O.; Alazard, D. Anaerobic treatment of cassava starch extraction wastewater using a horizontal flow filter with bamboo as support. Bioresour. Technol. 2007, 98, 1602–1607. [Google Scholar] [CrossRef]
- Lin, H.; Gan, J.; Rajendran, A.; Reis, C.E.R.; Hu, B. Phosphorus removal and recovery from digestate after biogas production. In Biofuels-Status and Perspective; IntechOpen: London, UK, 2015. [Google Scholar]
- Xie, B.; Gong, W.; Tian, Y.; Qu, F.; Luo, Y.; Du, X.; Tang, X.; Xu, D.; Lin, D.; Li, G.; et al. Biodiesel production with the simultaneous removal of nitrogen, phosphorus and COD in microalgal-bacterial communities for the treatment of anaerobic digestion effluent in photobioreactors. Chem. Eng. J. 2018, 350, 1092–1102. [Google Scholar] [CrossRef]
- Guldhe, A.; Ansari, F.A.; Singh, P.; Bux, F. Heterotrophic cultivation of microalgae using aquaculture wastewater: A biorefinery concept for biomass production and nutrient remediation. Ecol. Eng. 2017, 99, 47–53. [Google Scholar] [CrossRef]
- Benedetti, M.; Vecchi, V.; Barera, S.; Dall’Osto, L. Biomass from microalgae: The potential of domestication towards sustainable biofactories. Microb. Cell Factories 2018, 17, 173. [Google Scholar] [CrossRef] [Green Version]
- Wollmann, F.; Dietze, S.; Ackermann, J.-U.; Bley, T.; Walther, T.; Steingroewer, J.; Krujatz, F. Microalgae wastewater treatment: Biological and technological approaches. Eng. Life Sci. 2019, 19, 860–871. [Google Scholar] [CrossRef] [Green Version]
- Sadvakasova, A.K.; Akmukhanova, N.R.; Bolatkhan, K.; Zayadan, B.K.; Usserbayeva, A.A.; Bauenova, M.O.; Akhmetkaliyeva, A.E.; Allakhverdiev, S.I. Search for new strains of microalgae-producers of lipids from natural sources for biodiesel production. Int. J. Hydrog. Energy 2019, 44, 5844–5853. [Google Scholar] [CrossRef]
- Cheng, J.; Zhu, Y.; Zhang, Z.; Yang, W. Modification and improvement of microalgae strains for strengthening CO2 fixation from coal-fired flue gas in power plants. Bioresour. Technol. 2019, 291, 121850. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Ma, H.; Chen, S.; Yu, J.; Xu, W.; Zhu, X.; Gujar, A.; Ji, C.; Xue, J.; Zhang, C.; et al. Mitigating excessive ammonia nitrogen in chicken farm flushing wastewater by mixing strategy for nutrient removal and lipid accumulation in the green alga Chlorella sorokiniana. Bioresour. Technol. 2020, 303, 122940. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Srivastava, S.; Kumar, S. Isolation, screening and comprehensive characterization of candidate microalgae for biofuel feedstock production and dairy effluent treatment: A sustainable approach. Bioresour. Technol. 2019, 293, 121998. [Google Scholar] [CrossRef] [PubMed]
- Ferro, L.; Gentili, F.G.; Funk, C. Isolation and characterization of microalgal strains for biomass production and wastewater reclamation in Northern Sweden. Algal Res. 2018, 32, 44–53. [Google Scholar] [CrossRef]
- Choi, Y.-K.; Jang, H.M.; Kan, E. Microalgal Biomass and Lipid Production on Dairy Effluent Using a Novel Microalga, Chlorella sp. Isolated from Dairy Wastewater. Biotechnol. Bioprocess Eng. 2018, 23, 333–340. [Google Scholar] [CrossRef]
- Wen, Y.; He, Y.; Ji, X.; Li, S.; Chen, L.; Zhou, Y.; Wang, M.; Chen, B. Isolation of an indigenous Chlorella vulgaris from swine wastewater and characterization of its nutrient removal ability in undiluted sewage. Bioresour. Technol. 2017, 243, 247–253. [Google Scholar] [CrossRef]
- Tait, K.; White, D.A.; Kimmance, S.A.; Tarran, G.; Rooks, P.; Jones, M.; Llewellyn, C.A. Characterisation of bacteria from the cultures of a Chlorella strain isolated from textile wastewater and their growth enhancing effects on the axenic cultures of Chlorella vulgaris in low nutrient media. Algal Res. 2019, 44, 101666. [Google Scholar] [CrossRef]
- Eladel, H.; Abomohra, A.E.-F.; Battah, M.; Mohmmed, S.; Radwan, A.; Abdelrahim, H. Evaluation of Chlorella sorokiniana isolated from local municipal wastewater for dual application in nutrient removal and biodiesel production. Bioprocess Biosyst. Eng. 2019, 42, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.; Baird, E.; Eaton, R. Standard Methods for Examination of Water and Wastewater, 23rd ed.; American Public Health Association, American Water Works Association and Water Environment Federation: Washington, DC, USA, 2017. [Google Scholar]
- Abou-Shanab, R.A.I.; Matter, I.A.; Kim, S.-N.; Oh, Y.-K.; Choi, J.; Jeon, B.-H. Characterization and identification of lipid-producing microalgae species isolated from a freshwater lake. Biomass Bioenergy 2011, 35, 3079–3085. [Google Scholar] [CrossRef]
- Salgueiro, J.L.; Perez, L.; Maceiras, R.; Sanchez, A.; Cancela, A. Bioremediation of Wastewater using Chlorella Vulgaris Microalgae: Phosphorus and Organic Matter. Int. J. Environ. Res. 2016, 10, 465–470. [Google Scholar] [CrossRef]
- Fazal, T.; Rehman, M.S.U.; Javed, F.; Akhtar, M.; Mushtaq, A.; Hafeez, A.; Alaud Din, A.; Iqbal, J.; Rashid, N.; Rehman, F. Integrating bioremediation of textile wastewater with biodiesel production using microalgae (Chlorella vulgaris). Chemosphere 2021, 281, 130758. [Google Scholar] [CrossRef]
- Barsanti, L.; Gualtieri, P. Algae: Anatomy, Biochemistry, and Biotechnology; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Tale, M.; Ghosh, S.; Kapadnis, B.; Kale, S. Isolation and characterization of microalgae for biodiesel production from Nisargruna biogas plant effluent. Bioresour. Technol. 2014, 169, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Khaw, Y.S.; Khong, N.M.H.; Shaharuddin, N.A.; Yusoff, F.M. A simple 18S rDNA approach for the identification of cultured eukaryotic microalgae with an emphasis on primers. J. Microbiol. Methods 2020, 172, 105890. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Eisterhold, M.L.; Rindi, F.; Palanisami, S.; Nam, P.K. Isolation and screening of microalgae from natural habitats in the midwestern United States of America for biomass and biodiesel sources. J. Nat. Sci. Biol. Med. 2014, 5, 333–339. [Google Scholar] [CrossRef] [Green Version]
- Jagielski, T.; Gawor, J.; Bakuła, Z.; Zuchniewicz, K.; Żak, I.; Gromadka, R. An optimized method for high quality DNA extraction from microalga Prototheca wickerhamii for genome sequencing. Plant Methods 2017, 13, 77. [Google Scholar] [CrossRef] [Green Version]
- Fawley, M.W.; Fawley, K.P. A Simple and Rapid Technique for the Isolation of DNA from Microalgae1. J. Phycol. 2004, 40, 223–225. [Google Scholar] [CrossRef]
- Ding, G.T.; Mohd Yasin, N.H.; Takriff, M.S.; Kamarudin, K.F.; Salihon, J.; Yaakob, Z.; Mohd Hakimi, N.I.N. Phycoremediation of palm oil mill effluent (POME) and CO2 fixation by locally isolated microalgae: Chlorella sorokiniana UKM2, Coelastrella sp. UKM4 and Chlorella pyrenoidosa UKM7. J. Water Process Eng. 2020, 35, 101202. [Google Scholar] [CrossRef]
- Kumsiri, B.; Pekkoh, J.; Pathom-Aree, W.; Lumyong, S.; Pumas, C. Synergistic effect of co-culture of microalga and actinomycete in diluted chicken manure digestate for lipid production. Algal Res. 2018, 33, 239–247. [Google Scholar] [CrossRef]
- Nguyen, T.-T.-D.; Nguyen, T.-T.; An Binh, Q.; Bui, X.-T.; Ngo, H.H.; Vo, H.N.P.; Andrew Lin, K.-Y.; Vo, T.-D.-H.; Guo, W.; Lin, C.; et al. Co-culture of microalgae-activated sludge for wastewater treatment and biomass production: Exploring their role under different inoculation ratios. Bioresour. Technol. 2020, 314, 123754. [Google Scholar] [CrossRef] [PubMed]
- Krzemińska, I.; Pawlik-Skowrońska, B.; Trzcińska, M.; Tys, J. Influence of photoperiods on the growth rate and biomass productivity of green microalgae. Bioprocess Biosyst. Eng. 2014, 37, 735–741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Mattos, L.F.A.; Bastos, R.G. COD and nitrogen removal from sugarcane vinasse by heterotrophic green algae Desmodesmus sp. Desalin. Water Treat. 2016, 57, 9465–9473. [Google Scholar] [CrossRef]
- Mennaa, F.Z.; Arbib, Z.; Perales, J.A. Urban wastewater treatment by seven species of microalgae and an algal bloom: Biomass production, N and P removal kinetics and harvestability. Water Res. 2015, 83, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, J.; Álvarez-Díaz, P.D.; Arbib, Z.; Garrido-Pérez, C.; Barragán, J.; Perales, J.A. Performance of a flat panel reactor in the continuous culture of microalgae in urban wastewater: Prediction from a batch experiment. Bioresour. Technol. 2013, 127, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, J.; Zhao, Q.; Wei, W.; Sun, Y. Comparative study of wastewater treatment and nutrient recycle via activated sludge, microalgae and combination systems. Bioresour. Technol. 2016, 211, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Zou, S.; Fei, C.; Yang, W.; Huang, Z.; He, M.; Wang, C. High-efficiency 18S microalgae barcoding by coalescent, distance and character-based approaches: A test in Chlorella and Scenedesmus. J. Oceanol. Limnol. 2018, 36, 1771–1777. [Google Scholar] [CrossRef]
- Friedl, T. The evolution of the Green Algae. In Origins of Algae and Their Plastids; Bhattacharya, D., Ed.; Springer: Vienna, Austria, 1997; pp. 87–101. [Google Scholar]
- Asadi, P.; Rad, H.A.; Qaderi, F. Lipid and biodiesel production by cultivation isolated strain Chlorella sorokiniana pa.91 and Chlorella vulgaris in dairy wastewater treatment plant effluents. J. Environ. Health Sci. Eng. 2020, 18, 573–585. [Google Scholar] [CrossRef] [PubMed]
- Izadpanah, M.; Gheshlaghi, R.; Mahdavi, M.A.; Elkamel, A. Effect of light spectrum on isolation of microalgae from urban wastewater and growth characteristics of subsequent cultivation of the isolated species. Algal Res. 2018, 29, 154–158. [Google Scholar] [CrossRef]
- Cecchin, M.; Benfatto, S.; Griggio, F.; Mori, A.; Cazzaniga, S.; Vitulo, N.; Delledonne, M.; Ballottari, M. Molecular basis of autotrophic vs mixotrophic growth in Chlorella sorokiniana. Sci. Rep. 2018, 8, 6465. [Google Scholar] [CrossRef] [PubMed]
- León-Vaz, A.; León, R.; Díaz-Santos, E.; Vigara, J.; Raposo, S. Using agro-industrial wastes for mixotrophic growth and lipids production by the green microalga Chlorella sorokiniana. New Biotechnol. 2019, 51, 31–38. [Google Scholar] [CrossRef]
- Chai, S.; Shi, J.; Huang, T.; Guo, Y.; Wei, J.; Guo, M.; Li, L.; Dou, S.; Liu, L.; Liu, G. Characterization of Chlorella sorokiniana growth properties in monosaccharide-supplemented batch culture. PLoS ONE 2018, 13, e0199873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perez-Garcia, O.; Bashan, Y. Microalgal Heterotrophic and Mixotrophic Culturing for Bio-refining: From Metabolic Routes to Techno-economics. In Algal Biorefineries: Products and Refinery Design; Prokop, A., Bajpai, R.K., Zappi, M.E., Eds.; Springer International Publishing: Cham, Switzerland, 2015; Volume 2, pp. 61–131. [Google Scholar]
- Nagi, M.; He, M.; Li, D.; Gebreluel, T.; Cheng, B.; Wang, C. Utilization of tannery wastewater for biofuel production: New insights on microalgae growth and biomass production. Sci. Rep. 2020, 10, 1530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, K.Y.; Lim, B.-R.; Lee, K. Growth of microalgae in diluted process water of the animal wastewater treatment plant. Water Sci. Technol. 2009, 59, 2111–2116. [Google Scholar] [CrossRef] [PubMed]
- Pleissner, D.; Lindner, A.V.; Ambati, R.R. Techniques to Control Microbial Contaminants in Nonsterile Microalgae Cultivation. Appl. Biochem. Biotechnol. 2020, 192, 1376–1385. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Chinnasamy, S.; Singh, M.; Das, K.C. Renewable biomass production by mixotrophic algae in the presence of various carbon sources and wastewaters. Appl. Energy 2011, 88, 3425–3431. [Google Scholar] [CrossRef]
- Mujtaba, G.; Rizwan, M.; Lee, K. Removal of nutrients and COD from wastewater using symbiotic co-culture of bacterium Pseudomonas putida and immobilized microalga Chlorella vulgaris. J. Ind. Eng. Chem. 2017, 49, 145–151. [Google Scholar] [CrossRef]
- Gupta, S.K.; Ansari, F.A.; Shriwastav, A.; Sahoo, N.K.; Rawat, I.; Bux, F. Dual role of Chlorella sorokiniana and Scenedesmus obliquus for comprehensive wastewater treatment and biomass production for bio-fuels. J. Clean. Prod. 2016, 115, 255–264. [Google Scholar] [CrossRef] [Green Version]
- González, C.; Marciniak, J.; Villaverde, S.; León, C.; García, P.A.; Muñoz, R. Efficient nutrient removal from swine manure in a tubular biofilm photo-bioreactor using algae-bacteria consortia. Water Sci. Technol. 2008, 58, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Tsioptsias, C.; Lionta, G.; Deligiannis, A.; Samaras, P. Enhancement of the performance of a combined microalgae-activated sludge system for the treatment of high strength molasses wastewater. J. Environ. Manag. 2016, 183, 126–132. [Google Scholar] [CrossRef]
- Leong, W.H.; Lim, J.W.; Lam, M.K.; Lam, S.M.; Sin, J.C.; Samson, A. Novel sequential flow baffled microalgal-bacterial photobioreactor for enhancing nitrogen assimilation into microalgal biomass whilst bioremediating nutrient-rich wastewater simultaneously. J. Hazard. Mater. 2021, 409, 124455. [Google Scholar] [CrossRef] [PubMed]
- Yang, A. Modeling and Evaluation of CO2 Supply and Utilization in Algal Ponds. Ind. Eng. Chem. Res. 2011, 50, 11181–11192. [Google Scholar] [CrossRef] [Green Version]
- Salbitani, G.; Carfagna, S. Ammonium Utilization in Microalgae: A Sustainable Method for Wastewater Treatment. Sustainability 2021, 13, 956. [Google Scholar] [CrossRef]
- Delgadillo-Mirquez, L.; Lopes, F.; Taidi, B.; Pareau, D. Nitrogen and phosphate removal from wastewater with a mixed microalgae and bacteria culture. Biotechnol. Rep. 2016, 11, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Young, E.B.; Berges, J.A.; He, Z. Integrated Photo-Bioelectrochemical System for Contaminants Removal and Bioenergy Production. Environ. Sci. Technol. 2012, 46, 11459–11466. [Google Scholar] [CrossRef] [PubMed]
- Ayre, J.M.; Moheimani, N.R.; Borowitzka, M.A. Growth of microalgae on undiluted anaerobic digestate of piggery effluent with high ammonium concentrations. Algal Res. 2017, 24, 218–226. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Xiong, Z.; Yang, L.; Ren, Z.; Shao, P.; Shi, H.; Xiao, X.; Pavlostathis, S.G.; Fang, L.; Luo, X. Successful isolation of a tolerant co-flocculating microalgae towards highly efficient nitrogen removal in harsh rare earth element tailings (REEs) wastewater. Water Res. 2019, 166, 115076. [Google Scholar] [CrossRef]
- Czerwionka, K.; Makinia, J.; Pagilla, K.R.; Stensel, H.D. Characteristics and fate of organic nitrogen in municipal biological nutrient removal wastewater treatment plants. Water Res. 2012, 46, 2057–2066. [Google Scholar] [CrossRef]
- Patel, A.; Barrington, S.; Lefsrud, M. Microalgae for phosphorus removal and biomass production: A six species screen for dual-purpose organisms. GCB Bioenergy 2012, 4, 485–495. [Google Scholar] [CrossRef]
- Brown, N.; Shilton, A. Luxury uptake of phosphorus by microalgae in waste stabilisation ponds: Current understanding and future direction. Rev. Environ. Sci. Biotechnol. 2014, 13, 321–328. [Google Scholar] [CrossRef]
- Liao, Q.; Chang, H.-X.; Fu, Q.; Huang, Y.; Xia, A.; Zhu, X.; Zhong, N. Physiological-phased kinetic characteristics of microalgae Chlorella vulgaris growth and lipid synthesis considering synergistic effects of light, carbon and nutrients. Bioresour. Technol. 2018, 250, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Del Río, E.; Armendáriz, A.; García-Gómez, E.; García-González, M.; Guerrero, M.G. Continuous culture methodology for the screening of microalgae for oil. J. Biotechnol. 2015, 195, 103–107. [Google Scholar] [CrossRef] [Green Version]
- Shoener, B.D.; Schramm, S.M.; Béline, F.; Bernard, O.; Martínez, C.; Plósz, B.G.; Snowling, S.; Steyer, J.-P.; Valverde-Pérez, B.; Wágner, D.; et al. Microalgae and cyanobacteria modeling in water resource recovery facilities: A critical review. Water Res. 2019, 2, 100024. [Google Scholar] [CrossRef] [PubMed]
- Larsdotter, K.; Jansen, J.L.C.; Dalhammar, G. Biologicaly Mediated Phosporus Precipitation in Wastewater Treatment with Microalgae. Environ. Technol. 2007, 28, 953–960. [Google Scholar] [CrossRef]
- Ruiz, J.; Álvarez, P.; Arbib, Z.; Garrido, C.; Barragán, J.; Perales, J.A. Effect of Nitrogen and Phosphorus Concentration on Their Removal Kinetic in Treated Urban Wastewater by Chlorella Vulgaris. Int. J. Phytoremediation 2011, 13, 884–896. [Google Scholar] [CrossRef]
- Oliveira, C.Y.B.D.; Viegas, T.L.; Lopes, R.G.; Cella, H.; Menezes, R.S.; Soares, A.T.; Antoniosi Filho, N.R.; Derner, R.B. A comparison of harvesting and drying methodologies on fatty acids composition of the green microalga Scenedesmus obliquus. Biomass Bioenergy 2020, 132, 105437. [Google Scholar] [CrossRef]
- Caetano, N.S.; Martins, A.A.; Gorgich, M.; Gutiérrez, D.M.; Ribeiro, T.J.; Mata, T.M. Flocculation of Arthrospira maxima for improved harvesting. Energy Rep. 2020, 6, 423–428. [Google Scholar] [CrossRef]
- Li, Y.; Xu, Y.; Zheng, T.; Wang, H. Flocculation mechanism of the actinomycete Streptomyces sp. hsn06 on Chlorella vulgaris. Bioresour. Technol. 2017, 239, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Vandamme, D.; Foubert, I.; Meesschaert, B.; Muylaert, K. Flocculation of microalgae using cationic starch. J. Appl. Phycol. 2010, 22, 525–530. [Google Scholar] [CrossRef] [Green Version]
- El-Naggar, M.E.; Samhan, F.A.; Salama, A.A.A.; Hamdy, R.M.; Ali, G.H. Cationic starch: Safe and economic harvesting flocculant for microalgal biomass and inhibiting E. coli growth. Int. J. Biol. Macromol. 2018, 116, 1296–1303. [Google Scholar] [CrossRef]
- Gerde, J.A.; Yao, L.; Lio, J.; Wen, Z.; Wang, T. Microalgae flocculation: Impact of flocculant type, algae species and cell concentration. Algal Res. 2014, 3, 30–35. [Google Scholar] [CrossRef]
- Vergini, S.; Aravantinou, A.F.; Manariotis, I.D. Harvesting of freshwater and marine microalgae by common flocculants and magnetic microparticles. J. Appl. Phycol. 2016, 28, 1041–1049. [Google Scholar] [CrossRef]
- Chekli, L.; Eripret, C.; Park, S.H.; Tabatabai, S.A.A.; Vronska, O.; Tamburic, B.; Kim, J.H.; Shon, H.K. Coagulation performance and floc characteristics of polytitanium tetrachloride (PTC) compared with titanium tetrachloride (TiCl4) and ferric chloride (FeCl3) in algal turbid water. Sep. Purif. Technol. 2017, 175, 99–106. [Google Scholar] [CrossRef] [Green Version]
- Agbovi, H.K.; Wilson, L.D. Flocculation Optimization of Orthophosphate with FeCl3 and Alginate Using the Box–Behnken Response Surface Methodology. Ind. Eng. Chem. Res. 2017, 56, 3145–3155. [Google Scholar] [CrossRef]
- Dong, Q.; Parker, W.; Dagnew, M. Impact of FeCl3 dosing on AnMBR treatment of municipal wastewater. Water Res. 2015, 80, 281–293. [Google Scholar] [CrossRef]
- Zhu, L.; Hu, T.; Li, S.; Nugroho, Y.K.; Li, B.; Cao, J.; Show, P.-L.; Hiltunen, E. Effects of operating parameters on algae Chlorella vulgaris biomass harvesting and lipid extraction using metal sulfates as flocculants. Biomass Bioenergy 2020, 132, 105433. [Google Scholar] [CrossRef]
- Behera, B.; Nageshwari, K.; Darshini, M.; Balasubramanian, P. Evaluating the harvesting efficiency of inorganic coagulants on native microalgal consortium enriched with human urine. Water Sci. Technol. 2020, 82, 1217–1226. [Google Scholar] [CrossRef]
- Branyikova, I.; Prochazkova, G.; Potocar, T.; Jezkova, Z.; Branyik, T. Harvesting of Microalgae by Flocculation. Fermentation 2018, 4, 93. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Graham, N.; Liu, T.; Cheng, S.; Yu, W. A comparison of the coagulation performance of PAFC and FeSO4 for the treatment of leach liquor from Stevia processing. Sep. Purif. Technol. 2021, 255, 117680. [Google Scholar] [CrossRef]
- Pal, P. Chapter 2-Chemical Treatment Technology. In Industrial Water Treatment Process Technology; Pal, P., Ed.; Butterworth-Heinemann: Oxford, UK, 2017; pp. 21–63. [Google Scholar]
- McKinley, K.; McLellan, I.; Gagné, F.; Quinn, B. The toxicity of potentially toxic elements (Cu, Fe, Mn, Zn and Ni) to the cnidarian Hydra attenuata at environmentally relevant concentrations. Sci. Total Environ. 2019, 665, 848–854. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Yuan, R.; Liu, C.; Zhou, B. Effect of Fe2+ adding period on the biogas production and microbial community distribution during the dry anaerobic digestion process. Process Saf. Environ. Prot. 2020, 136, 234–241. [Google Scholar] [CrossRef]
| Parameters | Value | Units |
|---|---|---|
| COD | 205 ± 12.3 | mg L−1 |
| BOD | 75 ± 9.52 | mg L−1 |
| TP | 37.26 ± 2.05 | mg L−1 |
| PO4-P | 23.53 ± 1.70 | mg L−1 |
| TKN | 54.1 ± 3.21 | mg L−1 |
| NO2-N | 0.08 ± 0.02 | mg L−1 |
| NO3-N | 16.43 ± 0.69 | mg L−1 |
| NH4-N | 31.24 ± 1.67 | mg L−1 |
| pH | 7.6 ± 0.03 | - |
| DO | 3.21 ± 2.4 | mg L−1 |
| Conductivity | 2699 ± 43.60 | mS cm−1 |
| Alkalinity | 700 ± 32.57 | mg L−1 as |
| Salinity | 2.2 ± 1.3 | parts per thousand (PPT) |
| Kinetic Parameters | P21 | WB1DG |
|---|---|---|
| X0 (mg L−1) | 70 | 70 |
| Xm (mg L−1) | 2652.99 | 1301.85 |
| μ (day−1) | 1.11 | 0.61 |
| R2 | 0.99 | 0.96 |
| P (mg L−1 day−1) | 179.42 | 49.09 |
| Nutrient | Strain | Kinetic Parameters | |||||
|---|---|---|---|---|---|---|---|
| S0 (mg L−1) | Sna (mg L−1) | μ (Day−1) | R2 | 1/Y0 (% S) | CRs (mg L−1 Day−1) | ||
| COD | P21 | 200.72 | 57.65 | 1.69 | 0.98 | 4.6 | 53.57 |
| WB1DG | 207.92 | 73.36 | 1.35 | 0.94 | 10 | 62.85 | |
| NO3-N | P21 | 14.88 | 2.21 | 0.36 | 0.95 | 10 | 4.10 |
| WB1DG | 16.96 | 1.24 | 0.53 | 0.99 | 4 | 3.25 | |
| NH4-N | P21 | 31.35 | 3.03 | 1.70 | 0.98 | 1 | 15.29 |
| WB1DG | 32.07 | 3.50 | 1.50 | 0.98 | 1 | 16.92 | |
| TIN-N | P21 | 41.61 | 15.62 | 0.35 | 0.91 | 10 | 7.13 |
| WB1DG | 48.83 | 12.91 | 0.47 | 0.98 | 6 | 11.41 | |
| PO4-P | P21 | 19.85 | 4.37 | 1.26 | 0.94 | 1 | 4.33 |
| WB1DG | 23.97 | 1.79 | 2.24 | 0.98 | 1 | 14.81 | |
| TP | P21 | 37.05 | 6.08 | 1.01 | 0.98 | 1 | 8.17 |
| WB1DG | 34.75 | 3.71 | 0.94 | 0.98 | 1 | 9.47 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Padri, M.; Boontian, N.; Teaumroong, N.; Piromyou, P.; Piasai, C. Application of Two Indigenous Strains of Microalgal Chlorella sorokiniana in Cassava Biogas Effluent Focusing on Growth Rate, Removal Kinetics, and Harvestability. Water 2021, 13, 2314. https://doi.org/10.3390/w13172314
Padri M, Boontian N, Teaumroong N, Piromyou P, Piasai C. Application of Two Indigenous Strains of Microalgal Chlorella sorokiniana in Cassava Biogas Effluent Focusing on Growth Rate, Removal Kinetics, and Harvestability. Water. 2021; 13(17):2314. https://doi.org/10.3390/w13172314
Chicago/Turabian StylePadri, Mohamad, Nittaya Boontian, Neung Teaumroong, Pongdet Piromyou, and Chatlada Piasai. 2021. "Application of Two Indigenous Strains of Microalgal Chlorella sorokiniana in Cassava Biogas Effluent Focusing on Growth Rate, Removal Kinetics, and Harvestability" Water 13, no. 17: 2314. https://doi.org/10.3390/w13172314
APA StylePadri, M., Boontian, N., Teaumroong, N., Piromyou, P., & Piasai, C. (2021). Application of Two Indigenous Strains of Microalgal Chlorella sorokiniana in Cassava Biogas Effluent Focusing on Growth Rate, Removal Kinetics, and Harvestability. Water, 13(17), 2314. https://doi.org/10.3390/w13172314






