Effectiveness of Advanced Oxidation Processes in Wastewater Treatment: State of the Art
Abstract
1. Introduction
2. Advanced Oxidation Processes for Wastewater Treatment
3. Methodology
4. Compounds of Emerging Concern (CECs)
4.1. Pharmaceuticals Compounds (PH)
4.2. Personal Care Products (PCPs)
4.3. Illicit Drugs
4.4. Pesticides
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Monteoliva-García, A.; Martín-Pascual, J.; Muñío, M.M.; Poyatos, J.M. Removal of a pharmaceutical mix from urban wastewater coupling membrane bioreactor with advanced oxidation processes. J. Environ. Eng. 2019, 145, 04019055. [Google Scholar] [CrossRef]
- Lee, S.-H.; Kim, K.-H.; Lee, M.; Lee, B.-D. Detection status and removal characteristics of pharmaceuticals in wastewater treatment effluent. J. Water Process. Eng. 2019, 31, 100828. [Google Scholar] [CrossRef]
- Brillas, E. A review on the photoelectro-Fenton process as efficient electrochemical advanced oxidation for wastewater re-mediation. Treatment with UV light, sunlight, and coupling with conventional and other photo-assisted advanced technologies. Chemosphere 2020, 250, 126198. [Google Scholar] [CrossRef] [PubMed]
- Miniero, R.; Abate, V.; Brambilla, G.; Davoli, E.; De Felip, E.; De Filippis, S.P.; Dellatte, E.; De Luca, S.; Fanelli, R.; Fattore, E.; et al. Persistent toxic substances in Mediterranean aquatic species. Sci. Total Environ. 2014, 494–495, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Teodosiu, C.; Gilca, A.-F.; Barjoveanu, G.; Fiore, S. Emerging pollutants removal through advanced drinking water treatment: A review on processes and environmental performances assessment. J. Clean. Prod. 2018, 197, 1210–1221. [Google Scholar] [CrossRef]
- Bui, X.T.; Vo, T.P.T.; Ngo, H.H.; Guo, W.S.; Nguyen, T.T. Multicriteria assessment of advanced treatment technologies for mi-cropollutants removal at large-scale applications. Sci. Total Environ. 2016, 563–564, 1050–1067. [Google Scholar] [CrossRef] [PubMed]
- Rozas, O.; Vidal, C.; Baeza, C.; Jardim, W.F.; Rossner, A.; Mansilla, H.D. Organic micropollutants (OMPs) in natural waters: Oxidation by UV/H2O2 treatment and toxicity assessment. Water Res. 2016, 98, 109–118. [Google Scholar] [CrossRef]
- Watkinson, A.; Murby, E.; Kolpin, D.; Costanzo, S. The occurrence of antibiotics in an urban watershed: From wastewater to drinking water. Sci. Total Environ. 2009, 407, 2711–2723. [Google Scholar] [CrossRef]
- Moldovan, Z. Occurrences of pharmaceutical and personal care products as micropollutants in rivers from Romania. Chemosphere 2006, 64, 1808–1817. [Google Scholar] [CrossRef]
- Silva, B.F.D.; Jelic, A.; López-Serna, R.; Mozeto, A.A.; Petrovic, M.; Barceló, D. Occurrence and distribution of pharmaceuticals in surface water, suspended solids and sediments of the Ebro river basin, Spain. Chemosphere 2011, 85, 1331–1339. [Google Scholar] [CrossRef]
- Bartelt-Hunt, S.L.; Snow, D.D.; Damon, T.; Shockley, J.; Hoagland, K. The occurrence of illicit and therapeutic pharmaceuticals in wastewater effluent and surface waters in Nebraska. Environ. Pollut. 2009, 157, 786–791. [Google Scholar] [CrossRef]
- Xu, W.-H.; Zhang, G.; Zou, S.-C.; Li, X.; Liu, Y.-C. Determination of selected antibiotics in the Victoria Harbour and the Pearl River, South China using high-performance liquid chromatography-electrospray ionization tandem mass spectrometry. Environ. Pollut. 2007, 145, 672–679. [Google Scholar] [CrossRef]
- Li, W.; Shi, Y.; Gao, L.; Liu, J.; Cai, Y. Occurrence of antibiotics in water, sediments, aquatic plants, and animals from Baiyangdian Lake in North China. Chemosphere 2012, 89, 1307–1315. [Google Scholar] [CrossRef]
- Matamoros, V.; Arias, C.A.; Nguyen, L.X.; Salvado, V.; Brix, H. Occurrence and behavior of emerging contaminants in surface water and a restored wetland. Chemosphere 2012, 88, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- Vilimanovic, D.; Andaluri, G.; Hannah, R.; Suri, R.; MacGillivray, A.R. Occurrence and aquatic toxicity of contaminants of emerging concern (CECs) in tributaries of an urbanized section of the Delaware River Watershed. AIMS Environ. Sci. 2020, 7, 302–319. [Google Scholar] [CrossRef]
- Boczkaj, G.; Fernandes, A. Wastewater treatment by means of advanced oxidation processes at basic pH conditions: A review. Chem. Eng. J. 2017, 320, 608–633. [Google Scholar] [CrossRef]
- Poyatos, J.M.; Muñio, M.M.; Almecija, M.C.; Torres, J.C.; Hontoria, E.; Osorio, F. Advanced oxidation processes for wastewater treatment: State of the art. Water Air Soil Pollut. 2010, 205, 187–204. [Google Scholar] [CrossRef]
- Rosenfeldt, E.J.; Chen, P.-J.; Kullman, S.; Linden, K.G. Destruction of estrogenic activity in water using UV advanced oxidation. Sci. Total Environ. 2007, 377, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Baghirzade, B.S.; Yetis, U.; Dilek, F.B. Imidacloprid elimination by O3 and O3/UV: Kinetics study, matrix effect, and mechanism insight. Environ. Sci. Pollut. Res. 2021, 28, 24535–24551. [Google Scholar] [CrossRef] [PubMed]
- Maniakova, G.; Salmerón, I.; Polo-López, M.I.; Oller, I.; Rizzo, L.; Malato, S. Simultaneous removal of contaminants of emerging concern and pathogens from urban wastewater by homogeneous solar driven advanced oxidation processes. Sci. Total Environ. 2021, 766, 144320. [Google Scholar] [CrossRef]
- Ferro, G.; Guarino, F.; Castiglione, S.; Rizzo, L. Antibiotic resistance spread potential in urban wastewater effluents disinfected by UV/H2O2 process. Sci. Total Environ. 2016, 560-561, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Climent, L.F.; Olmos, R.G.; Anfruns, A.; Aymerich, I.; Corominas, L.; Barceló, D.; Rodriguez-Mozaz, S. Elimination study of the chemotherapy drug tamoxifen by different advanced oxidation processes: Transformation products and toxicity assessment. Chemosphere 2017, 168, 284–292. [Google Scholar] [CrossRef]
- Bianco, A.; Polo−López, M.; Fernandez-Ibañez, P.; Brigante, M.; Mailhot, G. Disinfection of water inoculated with Enterococcus faecalis using solar/Fe(III)EDDS-H2O2 or S2O82—process. Water Res. 2017, 118, 249–260. [Google Scholar] [CrossRef]
- He, Z.; Song, S.; Ying, H.; Xu, L.; Chen, J. p-Aminophenol degradation by ozonation combined with sonolysis: Operating conditions influence and mechanism. Ultrason. Sonochem. 2007, 14, 568–574. [Google Scholar] [CrossRef]
- Suri, R.P.S.; Andaluri, G. Oxidative sonication of estrogen hormones in water and municipal wastewater. Res. J. Environ. Sci. 2017, 11, 71–81. [Google Scholar] [CrossRef]
- Shemer, H.; Narkis, N. Trihalomethanes aqueous solutions sono-oxidation. Water Res. 2005, 39, 2704–2710. [Google Scholar] [CrossRef] [PubMed]
- Amadelli, R.; De Battisti, A.; Girenko, D.; Kovalyov, S.; Velichenko, A. Electrochemical oxidation of trans-3,4-dihydroxycinnamic acid at PbO2 electrodes: Direct electrolysis and ozone mediated reactions compared. Electrochim. Acta 2000, 46, 341–347. [Google Scholar] [CrossRef]
- Soares, I.C.D.C.; Da Silva, Á.R.L.; Santos, E.C.M.D.M.; dos Santos, E.V.; Da Silva, D.R.; Martínez-Huitle, C.A. Understanding the electrochemical oxidation of dyes on platinum and boron-doped diamond electrode surfaces: Experimental and computational study. J. Solid State Electrochem. 2020, 24, 3245–3256. [Google Scholar] [CrossRef]
- Damodhar, G.; Ghosh, P. Removal of organic compounds found in the wastewater through electrochemical advanced oxidation processes: A review. Russ. J. Electrochem. 2019, 55, 591–620. [Google Scholar] [CrossRef]
- Florenza, X.; Solano AM, S.; Centellas, F.; Martínez-Huitle, C.A.; Brillas, E.; Garcia-Segura, S. Degradation of the azo dye Acid Red 1 by anodic oxidation and indirect electrochemical processes based on Fenton’s reaction chemistry. Relationship between decolorization, mineralization and products. Electrochim. Acta. 2014, 142, 276–288. [Google Scholar] [CrossRef]
- Peralta-Hernández, J.; Meas-Vong, Y.; Rodríguez, F.J.; Chapman, T.W.; Maldonado, M.I.; Godínez, L.A. Comparison of hydrogen peroxide-based processes for treating dye-containing wastewater: Decolorization and destruction of Orange II azo dye in dilute solution. Dye Pigment. 2008, 76, 656–662. [Google Scholar] [CrossRef]
- Raschitor, A.; Llanos, J.; Rodrigo, M.A.; Cañizares, P. Combined electrochemical processes for the efficient degradation of non-polar organochlorine pesticides. J. Environ. Manag. 2019, 248, 109289. [Google Scholar] [CrossRef] [PubMed]
- Kourdali, S.; Badis, A.; Boucherit, A. Degradation of direct yellow 9 by electro-Fenton: Process study and optimization and, monitoring of treated water toxicity using catalase. Ecotoxicol. Environ. Saf. 2014, 110, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Alaton, I.A.; Balcioglu, I.A.; Bahnemann, D.W. Advanced oxidation of a reactive dyebath effluent: Comparison of O3, H2O2/UV-C and TiO2/UV-A processes. Water Res. 2002, 36, 1143–1154. [Google Scholar] [CrossRef]
- Dogruel, S.; Atesci, Z.C.; Aydin, E.; Pehlivanoglu-Mantas, E. Ozonation in advanced treatment of secondary municipal wastewater effluents for the removal of micropollutants. Environ. Sci. Pollut. Res. 2020, 27, 45460–45475. [Google Scholar] [CrossRef]
- Al Momani, F. Degradation of cyanobacteria anatoxin-a by advanced oxidation processes. Sep. Purif. Technol. 2007, 57, 85–93. [Google Scholar] [CrossRef]
- Lou, W.; Kane, A.; Wolbert, D.; Rtimi, S.; Assadi, A.A. Study of a photocatalytic process for removal of antibiotics from wastewater in a falling film photoreactor: Scavenger study and process intensification feasibility. Chem. Eng. Process. Process. Intensif. 2017, 122, 213–221. [Google Scholar] [CrossRef]
- Hong, P.; Li, Y.; He, J.; Saeed, A.; Zhang, K.; Wang, C.; Kong, L.; Liu, J. Rapid degradation of aqueous doxycycline by surface CoFe2O4/H2O2 system: Behaviors, mechanisms, pathways and DFT calculation. Appl. Surf. Sci. 2020, 526, 146557. [Google Scholar] [CrossRef]
- Aguilar-Melo, C.; Rodríguez, J.L.; Chairez, I.; Tiznado, H.; Poznyak, T. Naphthalene degradation by catalytic ozonation based on nickel oxide: Study of the ethanol as cosolvent. Environ. Sci. Pollut. Res. 2016, 24, 25550–25560. [Google Scholar] [CrossRef]
- Tanaka, K.; Abe, K.; Hisanaga, T. Photocatalytic water treatment on immobilized TiO2 combined with ozonation. J. Photochem. Photobiol. A Chem. 1996, 101, 85–87. [Google Scholar] [CrossRef]
- Aguinaco, A.; Beltran, F.J.; García-Araya, J.F.; Oropesa, A.L. Photocatalytic ozonation to remove the pharmaceutical diclofenac from water: Influence of variables. Chem. Eng. J. 2012, 189-190, 275–282. [Google Scholar] [CrossRef]
- García, J.; Oliveira, J.; Silva, A.; Oliveira, C.; Nozaki, J.; De Souza, N. Comparative study of the degradation of real textile effluents by photocatalytic reactions involving UV/TiO2/H2O2 and UV/Fe2+/H2O2 systems. J. Hazard. Mater. 2007, 147, 105–110. [Google Scholar] [CrossRef]
- Kümmerer, K. The presence of pharmaceuticals in the environment due to human use—Present knowledge and future challenges. J. Environ. Manag. 2009, 90, 2354–2366. [Google Scholar] [CrossRef] [PubMed]
- Naidu, R.; Jit, J.; Kennedy, B.; Arias, V. Emerging contaminant uncertainties and policy: The chicken or the egg conundrum. Chemosphere 2016, 154, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Bu, Q.; Wang, B.; Huang, J.; Deng, S.; Yu, G. Pharmaceuticals and personal care products in the aquatic environment in China: A review. J. Hazard. Mater. 2013, 262, 189–211. [Google Scholar] [CrossRef]
- Petrović, M.; Gonzalez, S.; Barceló, D. Analysis and removal of emerging contaminants in wastewater and drinking water. Trends Anal. Chemistry 2003, 22, 685–696. [Google Scholar] [CrossRef]
- Carballa, M.; Omil, F.; Lema, J.M.; Llompart, M.; García-Jares, C.; Rodríguez, I.; Gomez, M.; Ternes, T. Behavior of pharmaceuticals, cosmetics and hormones in a sewage treatment plant. Water Res. 2004, 38, 2918–2926. [Google Scholar] [CrossRef]
- Rivera-Utrilla, J.; Sánchez-Polo, M.; Ferro-García, M.Á.; Prados-Joya, G.; Ocampo-Pérez, R. Pharmaceuticals as emerging contaminants and their removal from water. A review. Chemosphere 2013, 93, 1268–1287. [Google Scholar] [CrossRef]
- Jiang, J.-Q.; Zhou, Z.; Sharma, V. Occurrence, transportation, monitoring and treatment of emerging micro-pollutants in waste water—A review from global views. Microchem. J. 2013, 110, 292–300. [Google Scholar] [CrossRef]
- Michael, S.G.; Michael-Kordatou, I.; Nahim-Granados, S.; Polo-López, M.I.; Rocha, J.; Martínez-Piernas, A.B.; Fernández-Ibáñez, P.; Agüera, A.; Manaia, C.M.; Fatta-Kassinos, D. Investigating the impact of UV-C/H2O2 and sunlight/H2O2 on the removal of antibiotics, antibiotic resistance determinants and toxicity present in urban wastewater. Chem. Eng. J. 2020, 388, 124383. [Google Scholar] [CrossRef]
- Gao, Y.-Q.; Zhang, J.; Li, C.; Tian, F.-X.; Gao, N.-Y. Comparative evaluation of metoprolol degradation by UV/chlorine and UV/H2O2 processes. Chemosphere 2020, 243, 125325. [Google Scholar] [CrossRef]
- Tayo, L.L.; Caparanga, A.R.; Doma, B.T.; Liao, C.-H. A Review on the removal of Pharmaceutical and Personal Care Products (PPCPs) using advanced oxidation processes. J. Adv. Oxid. Technol. 2018, 21, 196–214. [Google Scholar] [CrossRef]
- Yan, C.; Yang, Y.; Zhou, J.; Liu, M.; Nie, M.; Shi, H.; Gu, L. Antibiotics in the surface water of the Yangtze estuary: Occurrence, distribution and risk assessment. Environ. Pollut. 2013, 175, 22–29. [Google Scholar] [CrossRef]
- Oviedo Cordoba, H.; Cortina Navarro, C.E.; Osorio Coronel, J.A.; Romero Torres, S.M. Realidades de la práctica de la auto-medicación en estudiantes de la Universidad del Magdalena. Enfermería Global. 2021, 20, 531–556. [Google Scholar] [CrossRef]
- Jardim, W.F.; Montagner, C.C.; Pescara, I.C.; Umbuzeiro, G.; Bergamasco, A.M.D.D.; Eldridge, M.L.; Sodré, F.F. An integrated approach to evaluate emerging contaminants in drinking water. Sep. Purif. Technol. 2012, 84, 3–8. [Google Scholar] [CrossRef]
- Bletsou, A.A.; Jeon, J.; Hollender, J.; Archontaki, E.; Thomaidis, N.S. Targeted and non-targeted liquid chromatography-mass spectrometric workflows for identification of transformation products of emerging pollutants in the aquatic environment. TrAC Trends Anal. Chem. 2015, 66, 32–44. [Google Scholar] [CrossRef]
- Monteoliva-García, A.; Martín-Pascual, J.; Muñío, M.M.; Poyatos, J.M. Removal of carbamazepine, ciprofloxacin and ibuprofen in real urban wastewater by using light-driven advanced oxidation processes. Int. J. Environ. Sci. Technol. 2019, 16, 6005–6018. [Google Scholar] [CrossRef]
- Yuan, F.; Hu, C.; Hu, X.; Wei, D.; Chen, Y.; Qu, J. Photodegradation and toxicity changes of antibiotics in UV and UV/H2O2 process. J. Hazard. Mater. 2011, 185, 1256–1263. [Google Scholar] [CrossRef]
- Rosal, R.; Rodríguez, A.; Melon, J.A.P.; Mezcua, M.; Hernando, D.; Leton, P.; Calvo, E.G.; Agüera, A.; Fernández-Alba, A. Removal of pharmaceuticals and kinetics of mineralization by O3/H2O2 in a biotreated municipal wastewater. Water Res. 2008, 42, 3719–3728. [Google Scholar] [CrossRef]
- De Witte, B.; Dewulf, J.; Demeestere, K.; Van Langenhove, H. Ozonation and advanced oxidation by the peroxone process of ciprofloxacin in water. J. Hazard. Mater. 2009, 161, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Lekkerkerker-Teunissen, K.; Benotti, M.J.; Snyder, S.; van Dijk, H.C. Transformation of atrazine, carbamazepine, diclofenac and sulfamethoxazole by low and medium pressure UV and UV/H2O2 treatment. Sep. Purif. Technol. 2012, 96, 33–43. [Google Scholar] [CrossRef]
- Yang, Y.; Lu, X.; Jiang, J.; Ma, J.; Liu, G.; Cao, Y.; Liu, W.; Li, J.; Pang, S.; Kong, X.; et al. Degradation of sulfamethoxazole by UV, UV/H2O2 and UV/persulfate (PDS): Formation of oxidation products and effect of bicarbonate. Water Res. 2017, 118, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-H.; Kim, S.D.; Kim, H.Y.; Lim, S.J.; Lee, M.; Yu, S. Degradation and toxicity assessment of sulfamethoxazole and chlortetracycline using electron beam, ozone and UV. J. Hazard. Mater. 2012, 227-228, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.J.; Kim, W.G.; Yoon, Y.; Kang, J.-W.; Hong, Y.M.; Kim, H.W. Removal of amoxicillin by UV and UV/H2O2 processes. Sci. Total Environ. 2012, 420, 160–167. [Google Scholar] [CrossRef]
- Elmolla, E.S.; Chaudhuri, M. Photocatalytic degradation of amoxicillin, ampicillin and cloxacillin antibiotics in aqueous solution using UV/TiO2 and UV/H2O2/TiO2 photocatalysis. Desalination 2010, 252, 46–52. [Google Scholar] [CrossRef]
- Torre, J.A.C.; Lehmann Ah González, C.; Carmona Em Muñoz, A.H. Presencia de Fármacos en Aguas Residuales y Eficacia de los Procesos Convencionales en su Eliminación; Cátedra de Ingeriería Sanidad y Ambiente de la ETS Ingenieros de Caminos, Canales y Puertos, Universidad Politécnica de Madrid: Madrid, Spain, 2006. [Google Scholar]
- Shemer, H.; Kunukcu, Y.K.; Linden, K.G. Degradation of the pharmaceutical Metronidazole via UV, Fenton and photo-Fenton processes. Chemosphere 2006, 63, 269–276. [Google Scholar] [CrossRef]
- Andreozzi, R.; Canterino, M.; Giudice, R.L.; Marotta, R.; Pinto, G.; Pollio, A. Lincomycin solar photodegradation, algal toxicity and removal from wastewaters by means of ozonation. Water Res. 2006, 40, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, S.K.; Kang, J.; Nghiem, L.; van de Merwe, J.; Leusch, F.; Price, W.E. Photolysis and UV/H2O2 of diclofenac, sulfamethoxazole, carbamazepine, and trimethoprim: Identification of their major degradation products by ESI–LC–MS and assessment of the toxicity of reaction mixtures. Process. Saf. Environ. Prot. 2017, 112, 222–234. [Google Scholar] [CrossRef]
- Ternes, T.A.; Meisenheimer, M.; McDowell, D.; Sacher, F.; Brauch, H.-J.; Haist-Gulde, B.; Preuss, G.; Wilme, U.; Zulei-Seibert, N. Removal of pharmaceuticals during drinking water treatment. Environ. Sci. Technol. 2002, 36, 3855–3863. [Google Scholar] [CrossRef]
- Huber, M.M.; Canonica, S.; Park, G.-Y.; Von Gunten, U. Oxidation of pharmaceuticals during ozonation and advanced oxidation processes. Environ. Sci. Technol. 2003, 37, 1016–1024. [Google Scholar] [CrossRef]
- Vogna, D.; Marotta, R.; Napolitano, A.; Andreozzi, R.; D’Ischia, M. Advanced oxidation of the pharmaceutical drug diclofenac with UV/H2O2 and ozone. Water Res. 2004, 38, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Andreozzi, R.; Campanella, L.; Fraysse, B.; Garric, J.; Gonnella, A.; Giudice, R.L.; Marotta, R.; Pinto, G.; Pollio, A. Effects of advanced oxidation processes (AOPs) on the toxicity of a mixture of pharmaceuticals. Water Sci. Technol. 2004, 50, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Sein, M.M.; Zedda, M.; Tuerk, J.; Schmidt, T.C.; Golloch, A.; Von Sonntag, C. Oxidation of diclofenac with ozone in aqueous solution. Environ. Sci. Technol. 2008, 42, 6656–6662. [Google Scholar] [CrossRef] [PubMed]
- Zwiener, C.; Frimmel, F.H. Oxidative treatment of pharmaceuticals in water. Water Res. 2000, 34, 1881–1885. [Google Scholar] [CrossRef]
- Ravina, M.; Campanella, L.; Kiwi, J. Accelerated mineralization of the drug Diclofenac via Fenton reactions in a concentric photo-reactor. Water Res. 2002, 36, 3553–3560. [Google Scholar] [CrossRef]
- Ternes, T.A.; Stüber, J.; Herrmann, N.; McDowell, D.; Ried, A.; Kampmann, M.; Teiser, B. Ozonation: A tool for removal of pharmaceuticals, contrast media and musk fragrances from wastewater? Water Res. 2003, 37, 1976–1982. [Google Scholar] [CrossRef]
- Witte, B.D.; Langenhove, H.V.; Hemelsoet, K.; Demeestere, K.; Wispelaere, P.D.; Van Speybroeck, V.; Dewulf, J. Levofloxacin ozonation in water: Rate determining process parameters and reaction pathway elucidation. Chemosphere 2009, 76, 683–689. [Google Scholar] [CrossRef]
- Lima, V.B.; Goulart, L.A.; Rocha, R.S.; Steter, J.R.; Lanza, M.R. Degradation of antibiotic ciprofloxacin by different AOP systems using electrochemically generated hydrogen peroxide. Chemosphere 2020, 247, 125807. [Google Scholar] [CrossRef] [PubMed]
- Lange, F.; Cornelissen, S.; Kubac, D.; Sein, M.M.; von Sonntag, J.; Hannich, C.B.; Golloch, A.; Heipieper, H.J.; Möder, M.; von Sonntag, C. Degradation of macrolide antibiotics by ozone: A mechanistic case study with clarithromycin. Chemosphere 2006, 65, 17–23. [Google Scholar] [CrossRef]
- Garoma, T.; Umamaheshwar, S.K.; Mumper, A. Removal of sulfadiazine, sulfamethizole, sulfamethoxazole, and sulfathiazole from aqueous solution by ozonation. Chemosphere 2010, 79, 814–820. [Google Scholar] [CrossRef]
- Chen, X.; Richard, J.; Liu, Y.; Dopp, E.; Tuerk, J.; Bester, K. Ozonation products of triclosan in advanced wastewater treatment. Water Res. 2012, 46, 2247–2256. [Google Scholar] [CrossRef]
- Ellis, J. Pharmaceutical and personal care products (PPCPs) in urban receiving waters. Environ. Pollut. 2006, 144, 184–189. [Google Scholar] [CrossRef]
- Ebele, A.J.; Abdallah, M.A.-E.; Harrad, S. Pharmaceuticals and personal care products (PPCPs) in the freshwater aquatic environment. Emerg. Contam. 2017, 3, 1–16. [Google Scholar] [CrossRef]
- Xia, K.; Bhandari, A.; Das, K.; Pillar, G. Occurrence and fate of pharmaceuticals and personal care products (PPCPs) in biosolids. J. Environ. Qual. 2005, 34, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Hu, J. Effect of alpha-hydroxy acids on transformation products formation and degradation mechanisms of carbamaz-epine by UV/H2O2 process. Sci. Total. Environ. 2019, 689, 70–78. [Google Scholar] [CrossRef]
- Huang, Y.; Kong, M.; Coffin, S.; Cochran, K.H.; Westerman, D.C.; Schlenk, D.; Richardson, S.D.; Lei, L.; Dionysiou, D.D. Degradation of contaminants of emerging concern by UV/H2O2 for water reuse: Kinetics, mechanisms, and cytotoxicity analysis. Water Res. 2020, 174, 115587. [Google Scholar] [CrossRef]
- Tay, K.S.; Rahman, N.A.; Bin Abas, M.R. Kinetic studies of the degradation of parabens in aqueous solution by ozone oxidation. Environ. Chem. Lett. 2010, 8, 331–337. [Google Scholar] [CrossRef]
- De la Cruz, N.; Giménez, J.; Esplugas, S.; Grandjean, D.; de Alencastro, L.F.; Pulgarín, C. Degradation of 32 emergent contaminants by UV and neutral photo-Fenton in domestic wastewater effluent previously treated by activated sludge. Water Res. 2012, 46, 1947–1957. [Google Scholar] [CrossRef] [PubMed]
- Zuccato, E.; Castiglioni, S.; Bagnati, R.; Chiabrando, C.; Grassi, P.; Fanelli, R. Illicit drugs, a novel group of environmental con-taminants. Water Res. 2008, 42, 961–968. [Google Scholar] [CrossRef]
- Ahmed, F.; Tscharke, B.; O’Brien, J.W.; Cabot, P.J.; Hall, W.D.; Mueller, J.F.; Thomas, K.V. Can wastewater analysis be used as a tool to assess the burden of pain treatment within a population? Environ. Res. 2020, 188, 109769. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Zhou, Z.; Bai, Y.; Xu, Z.; Gao, T.; Fu, X.; Li, X. Estimating heroin abuse in major Chinese cities through wastewater-based epidemiology. Sci. Total. Environ. 2017, 605–606, 158–165. [Google Scholar] [CrossRef]
- Subedi, B.; Kannan, K. Mass loading and removal of select illicit drugs in two wastewater treatment plants in New York State and estimation of illicit drug usage in communities through wastewater analysis. Environ. Sci. Technol. 2014, 48, 6661–6670. [Google Scholar] [CrossRef]
- Been, F.; Benaglia, L.; Lucia, S.; Gervasoni, J.-P.; Esseiva, P.; Delémont, O. Data triangulation in the context of opioids monitoring via wastewater analyses. Drug Alcohol Depend. 2015, 151, 203–210. [Google Scholar] [CrossRef] [PubMed]
- McCall, A.-K.; Bade, R.; Kinyua, J.; Lai, F.Y.; Thai, P.; Covaci, A.; Bijlsma, L.; van Nuijs, A.; Ort, C. Critical review on the stability of illicit drugs in sewers and wastewater samples. Water Res. 2016, 88, 933–947. [Google Scholar] [CrossRef] [PubMed]
- Mercan, S.; Kuloglu, M.; Tekin, T.; Turkmen, Z.; Dogru, A.O.; Safran, A.N.; Acikkol, M.; Asicioglu, F. Wastewater-based monitoring of illicit drug consumption in Istanbul: Preliminary results from two districts. Sci Total Environ. 2019, 656, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Damien, D.A.; Thomas, N.; Hélène, P.; Sara, K.; Yves, L. First evaluation of illicit and licit drug consumption based on wastewater analysis in Fort de France urban area (Martinique, Caribbean), a transit area for drug smuggling. Sci. Total Environ. 2014, 490, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Kasprzyk-Hordern, B.; Dinsdale, R.; Guwy, A.J. The occurrence of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs in surface water in South Wales, UK. Water Res. 2008, 42, 3498–3518. [Google Scholar] [CrossRef]
- Jones-Lepp, T.L.; Alvarez, D.A.; Petty, J.D.; Huckins, J.N. Polar organic chemical integrative sampling and liquid chromatography? Electrospray/ion-trap mass spectrometry for assessing selected prescription and illicit drugs in treated sewage effluents. Arch. Environ. Contam. Toxicol. 2004, 47, 427–439. [Google Scholar] [CrossRef]
- Huerta-Fontela, M.; Galceran, M.T.; Martin-Alonso, J.; Ventura, F. Occurrence of psychoactive stimulatory drugs in wastewaters in north-eastern Spain. Sci. Total Environ. 2008, 397, 31–40. [Google Scholar] [CrossRef]
- Spasiano, D.; Russo, D.; Vaccaro, M.; Siciliano, A.; Marotta, R.; Guida, M.; Reis, N.; Puma, G.L.; Andreozzi, R. Removal of benzoylecgonine from water matrices through UV254/H2O2 process: Reaction kinetic modeling, ecotoxicity and genotoxicity assessment. J. Hazard. Mater. 2016, 318, 515–525. [Google Scholar] [CrossRef]
- Russo, D.; Spasiano, D.; Vaccaro, M.; Cochran, K.H.; Richardson, S.D.; Andreozzi, R.; Puma, G.L.; Reis, N.; Marotta, R. Investigation on the removal of the major cocaine metabolite (benzoylecgonine) in water matrices by UV254 /H2O2 process by using a flow microcapillary film array photoreactor as an efficient experimental tool. Water Res. 2016, 89, 375–383. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gooddy, D.C.; Bloomfield, J.P.; Chilton, P.J.; Johnson, A.C.; Williams, R.J. Assessing herbicide concentrations in the saturated and unsaturated zone of a Chalk aquifer in southern England. Ground Water 2001, 39, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Gooddy, D.C.; Chilton, P.J.; Harrison, I. A field study to assess the degradation and transport of diuron and its metabolites in a calcareous soil. Sci. Total Environ. 2002, 297, 67–83. [Google Scholar] [CrossRef]
- Kolpin, D.W.; Thurman, E.M.; Linhart, S.M. Occurrence of cyanazine compounds in groundwater: Degradates More Prevalent than the parent compound. Environ. Sci. Technol. 2001, 35, 1217–1222. [Google Scholar] [CrossRef]
- Panno, S.; Kelly, W. Nitrate and herbicide loading in two groundwater basins of Illinois’ sinkhole plain. J. Hydrol. 2004, 290, 229–242. [Google Scholar] [CrossRef]
- Lapworth, D.; Gooddy, D.; Stuart, M.; Chilton, P.; Cachandt, G.; Knapp, M.; Bishop, S. Pesticides in groundwater: Some observations on temporal and spatial trends. Water Environ. J. 2006, 20, 55–64. [Google Scholar] [CrossRef]
- Hans-Rudolf, B. Atrazine and others-triazine herbicides in lakes and in rain in Switzerland. Environ. Sci. Technol. 1990, 24, 1049–1058. [Google Scholar] [CrossRef]
- Chen, L.; Hu, X.; Yang, Y.; Jiang, C.; Bian, C.; Liu, C.; Zhang, M.; Cai, T. Degradation of atrazine and structurally related s-triazine herbicides in soils by ferrous-activated persulfate: Kinetics, mechanisms and soil-types effects. Chem. Eng. J. 2018, 351, 523–531. [Google Scholar] [CrossRef]
- Serna-Galvis, E.A.; Botero-Coy, A.M.; Martínez-Pachón, D.; Moncayo-Lasso, A.; Ibáñez, M.; Hernández, F.; Torres-Palma, R.A. Degradation of seventeen contaminants of emerging concern in municipal wastewater effluents by sonochemical advanced oxidation pro-cesses. Water Res. 2019, 154, 349–360. [Google Scholar] [CrossRef]
- Ma, D.; Yi, H.; Lai, C.; Liu, X.; Huo, X.; An, Z.; Li, L.; Fu, Y.; Li, B.; Zhang, M.; et al. Critical review of advanced oxidation processes in organic wastewater treatment. Chemosphere 2021, 275, 130104. [Google Scholar] [CrossRef]
- Berberidou, C.; Kitsiou, V.; Lambropoulou, D.; Antoniadis, A.; Ntonou, E.; Zalidis, G.C.; Poulios, I. Evaluation of an alternative method for wastewater treatment containing pesticides using solar photocatalytic oxidation and constructed wetlands. J. Environ. Manag. 2017, 195, 133–139. [Google Scholar] [CrossRef]
- Karaolia, P.; Michael-Kordatou, I.; Hapeshi, E.; Drosou, C.; Bertakis, Y.; Christofilos, D.; Armatas, G.S.; Sygellou, L.; Schwartz, T.; Xekoukoulotakis, N.P.; et al. Removal of antibiotics, antibi-otic-resistant bacteria and their associated genes by graphene-based TiO2 composite photocatalysts under solar radiation in urban wastewaters. Appl. Catal. B Environ. 2018, 224, 810–824. [Google Scholar] [CrossRef]
- Talwar, S.; Sangal, V.K.; Verma, A. Feasibility of using combined TiO2 photocatalysis and RBC process for the treatment of real pharmaceutical wastewater. J. Photochem. Photobiol. A Chem. 2018, 353, 263–270. [Google Scholar] [CrossRef]
- Rueda-Marquez, J.J.; Levchuk, I.; Fernández Ibañez, P.; Sillanpää, M. A critical review on application of photocatalysis for toxicity reduction of real wastewaters. J. Clean. Prod. 2020, 258, 120694. [Google Scholar] [CrossRef]
- Ibáñez, M.; Lor, E.G.; Bijlsma, L.; Morales, E.; Pastor, L.; Hernandez, F. Removal of emerging contaminants in sewage water subjected to advanced oxidation with ozone. J. Hazard. Mater. 2013, 260, 389–398. [Google Scholar] [CrossRef]
- Martínez, F.; Molina, R.; Rodríguez, I.; Pariente, M.I.; Segura, Y.; Melero, J.A. Techno-economical assessment of coupling Fen-ton/biological processes for the treatment of a pharmaceutical wastewater. J. Environ. Chem. Eng. 2018, 6, 485–494. [Google Scholar] [CrossRef]
- Olvera-Vargas, H.; Oturan, N.; Buisson, D.; Oturan, M.A. A coupled Bio-EF process for mineralization of the pharmaceuticals furosemide and ranitidine: Feasibility assessment. Chemosphere 2016, 155, 606–613. [Google Scholar] [CrossRef]
- Ganzenko, O.; Trellu, C.; Oturan, N.; Huguenot, D.; Péchaud, Y.; Van Hullebusch, E.D.; Oturan, M.A. Electro-Fenton treatment of a complex pharmaceutical mixture: Mineralization efficiency and biodegradability enhancement. Chemosphere 2020, 253, 126659. [Google Scholar] [CrossRef] [PubMed]
- Karaolia, P.; Michael-Kordatou, I.; Hapeshi, E.; Alexander, J.; Schwartz, T.; Fatta-Kassinos, D. Investigation of the potential of a membrane BioReactor followed by solar Fenton oxidation to remove antibiotic-related microcontaminants. Chem. Eng. J. 2017, 310, 491–502. [Google Scholar] [CrossRef]
- Lastre-Acosta, A.M.; Palharim, P.H.; Barbosa, I.M.; Mierzwa, J.C.; Teixeira, A.C.S.C. Removal of sulfadiazine from simulated industrial wastewater by a membrane bioreactor and ozonation. J. Environ. Manag. 2020, 271, 111040. [Google Scholar] [CrossRef]
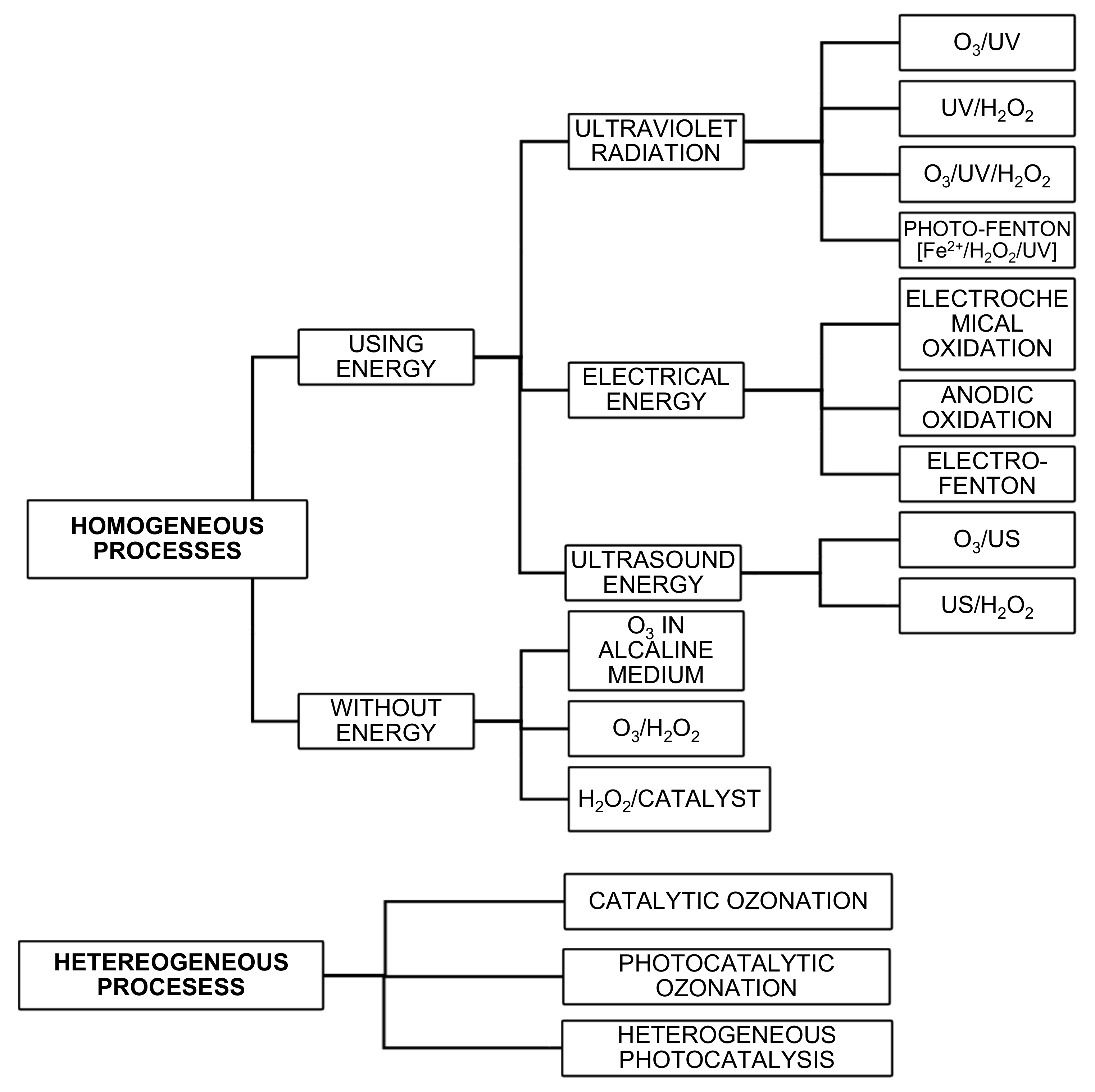
| Advanced Oxidation Processes | Reactions | Description | Examples of Pollutants That Can Be Removed |
|---|---|---|---|
| O3/UV | The photolysis of the ozone produces the formation of hydroxyl radicals [17]. | Imidacloprid [19] | |
| H2O2/UV | The formation of hydroxyl radicals is generated by photolysis of H2O2 [17]. | Bacterias (E. Coli, S. Enteritidis and E. Faecalis) [20] Inactivation of a multidrug resistant E. Coli [21] | |
| O3/H2O2/UV | Combination of systems O3/UV and H2O2/UV. The use of H2O2 it accelerates the decomposition of ozone and increases the generation of OH• [17]. | Tamoxifen [22] | |
| Fe2+/H2O2/UV | The Fenton reaction is the process most often applied when it is necessary to remove recalcitrant compounds [17]. | E. Faecalis [23] Inactivation of a multidrug resistant E. Coli [21] | |
| O3/US | Ultrasounds are represented as ‘(((‘ [24]. | 7α -estradiol, 17β-estradiol, estrone, 17α-dihydroequilin, 17α-ethinyl estradiol, estriol and equilin [25] | |
| H2O2/US | The combination of ultrasound and H2O2 achieves the formation of hydroxyl radicals [26]. | 7α -estradiol, 17β-estradiol, estrone, 17α-dihydroequilin, 17α-ethinyl estradiol, estriol and equilin [25] | |
| Electrochemical oxidation | [Anode] [Cathode] | In this type of oxidation, the compound to be degraded reacts to the oxidants, which have been electrochemically generated in situ [27]. | Reactive Orange 16, Reactive Violet 4, Reactive Red 228, and Reactive Black 5 [28] |
| Anodic oxidation | In this process, organic compounds directly react with heterogeneous hydroxyl radicals (OH•), formed by oxidation of water at the surface of anode with high oxygen overpotential [29]. | Acid Red 1 azo dye [30] | |
| Electro-Fenton | Hydrogen peroxide is produced electrochemically through the cathodic reduction of dissolved oxygen on a carbon electrode [31]. | Non-polar organochlorine pesticides [32] Direct yellow 9 azo dye [33] | |
| Ozonation in an alkaline medium | The degradation of the compound occurs through the action of the ozone itself as well as through the radicals generated in the alkaline medium [34]. | Bisphenol A, paraxantina [35] | |
| Ozonation with hydrogen peroxide | Hydrogen peroxide in an aqueous solution is partially dissociated to hydroperoxide anion which reacts with ozone [36]. | Fluoroquinolone antibiotics and Clarithromycin [37] | |
| Hydrogen peroxide and catalyst | This type of reaction is very similar to photo-Fenton processes but the formation rate of OH• radicals is lower [17]. | Doxycycline (catalyst CoFe2O4) [38] | |
| Catalytic ozonation | In catalytic ozonation the most widely used catalyst is Fe2+ [36]. | Naphthalene [39] | |
| Photocatalytic ozonation: O3/UV/TiO2 | Titanium dioxide (TiO2) is the most effective catalyst of those used in AOPs. The basic mechanism is described by [40]. | Diclofenac [41] | |
| Heterogeneous photocatalysis: UV/TiO2/H2O2 | In these processes, titanium dioxide is combined with hydrogen peroxide and UV radiation [42]. | Inactivation of A multidrug resistant E. Coli [21] |
| Pharmaceutical Compounds | Use | Examples [52] |
|---|---|---|
| Antibiotics | Antibacterial activity. Human and veterinary use | Amoxicillin, ampicillin, cefaclor, cefalexin, ciprofloxacin, chlortetracycline, clarithromycin, difloxacine, doxycycline, enoxacin, erythromycin, lincomycin, levofloxacin, metromidazole, mecillinam, ofloxacin, oxytetracycline, penicillin, sulfamethoxazole, sulfadiazine, sulfamethizole, sulfathiazole, sulphapyridine, tetracyclines, trimethoprim, tylosin… |
| Analgesics and anti-Inflammatory Pharmaceuticals | Pain relief and the reduction of inflammation | Ibuprofen, diclofenac, paracetamol, acetaminophen, acetylsalicylic acid, fenoprofen, indomethacin, naproxen, nimesulide, mefenamic acid, fluoxetine, ketoprofen, phenazone… |
| Antidepressants and Anticonvulsants | Relief from mental symptoms and the treatment of epileptic seizures | Diazepam, carbamazepine, doxepin, imipramine, amitriptyline, primidone, salbutamol, meprobamate, fluxetine, oxazepam, gabapentin, phenobarbital, thioridazine, dilantin… |
| Lipid Regulators | Regulation of cholesterol and blood triglycerides | Clofibric acid, clofibrate, benzafibrate, fenofibric acid, etofibrate, gemfibrozil, simvastatin, furosemide, Bendroflumethiazide… |
| β-blockers | Reduction of blood pressure | Atenolol, metoprolol, propranolol, sotalol, timolol… |
| X-ray contrasts | Diagnostic contrast (organ visibility) | Iopromide, iopamidol, diatrizoate… |
| Eestrogens, Progestogens, Androgens, Glucocorticoids, Phytoestrogens& Hormones | Regulation of female/male sexual development, maintenance of pregnancy, growth promotion in meat-producing animals, control of immune function, treatment of breast cancer, lymphomas and leukaemias | 17b-Oestradiol (E2), estrone (E1), estriol, diethylstilbestrol (DES), 17-α ethynylestradiol, mestranol, zerranol, trenbolone acetate, melengestrol acetate, tamoxifen, testosterone, phytosterols, sesquiterpenes, androstenedione, beclomethasone, progesterone, norethindrone… |
| Personal Care Products | Use | Examples [52] |
|---|---|---|
| Fragrances | Pleasant odours | Polycyclic and macrocyclic musks (musk xylol and musk ketone), phthalates, tonalide, celestolide, galaxolide…. |
| Sun–screen agents | Skin protection from solar ultraviolet radiation | Benzophenone, 4-methylbenzylidene camphor, homosalate, octocrylene, oxybenzone, octylmethoxycinnamate, octyldimethyl-PABA… |
| Insect repellents | Repellent | N,N-diethyltoluamide,… |
| Antiseptics | Disinfectants | Clorophene, triclosan, bromoprene, 2-phenylphenol, 4-chlorocresol, 4-chloroxylenol… |
| Preservatives | Prevention of undesirable chemical changes or microbial decomposition | Methylparaben, 2-phenoxyethanol, ethyl 4-hydroxybenzoate, butyl 4-hydroxybenzoate… |
| Advanced Oxidation Processes | Examples of Representative PCPs, Illicit Drugs and Pesticides That Can Be Removed |
|---|---|
| O3-based AOPs | PCPs: Triclosan [84]; Ethylparaben, propylparaben, and butylparaben [90] |
| O3/UV | PCPs: Triclosan [88]; Butylparaben [91]; Illicit drugs: Cocaine and its metabolite [103,104,110] |
| VUV/UV/chlorine | Pesticides: Dimethoate [DMT], atrazine [ATZ], prometon [PMT], propoxur [PPX], bromacil [BRM], and propachlor [PPC] [109] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bermúdez, L.A.; Pascual, J.M.; Martínez, M.d.M.M.; Poyatos Capilla, J.M. Effectiveness of Advanced Oxidation Processes in Wastewater Treatment: State of the Art. Water 2021, 13, 2094. https://doi.org/10.3390/w13152094
Bermúdez LA, Pascual JM, Martínez MdMM, Poyatos Capilla JM. Effectiveness of Advanced Oxidation Processes in Wastewater Treatment: State of the Art. Water. 2021; 13(15):2094. https://doi.org/10.3390/w13152094
Chicago/Turabian StyleBermúdez, Laura Antiñolo, Jaime Martín Pascual, María del Mar Muñio Martínez, and Jose Manuel Poyatos Capilla. 2021. "Effectiveness of Advanced Oxidation Processes in Wastewater Treatment: State of the Art" Water 13, no. 15: 2094. https://doi.org/10.3390/w13152094
APA StyleBermúdez, L. A., Pascual, J. M., Martínez, M. d. M. M., & Poyatos Capilla, J. M. (2021). Effectiveness of Advanced Oxidation Processes in Wastewater Treatment: State of the Art. Water, 13(15), 2094. https://doi.org/10.3390/w13152094







