Spatial and Temporal Vertical Distribution of Chlorophyll in Relation to Submarine Wastewater Effluent Discharges
Abstract
1. Introduction
2. Materials and Methods
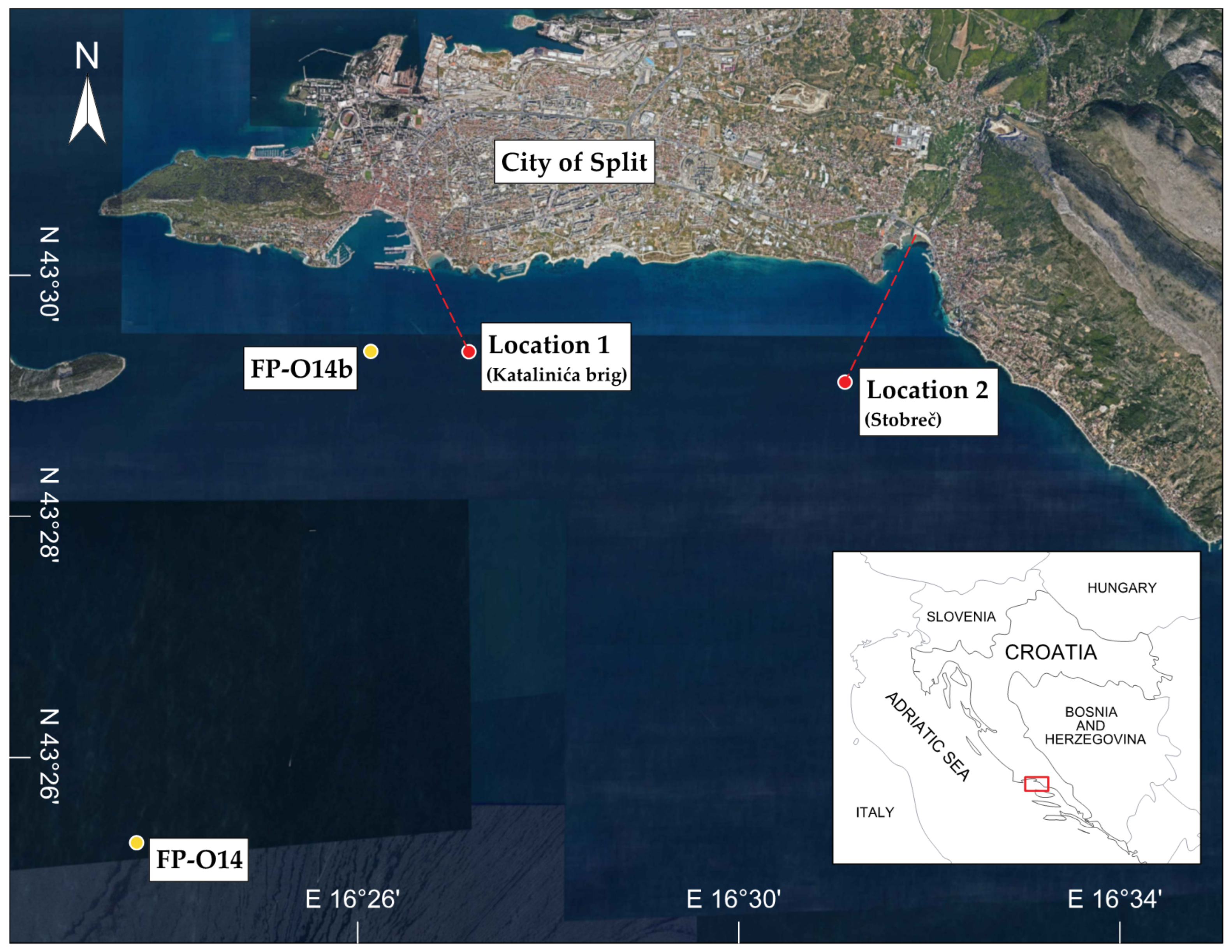
2.1. Study Site
2.2. Sampling Methodology
2.3. Data Analysis
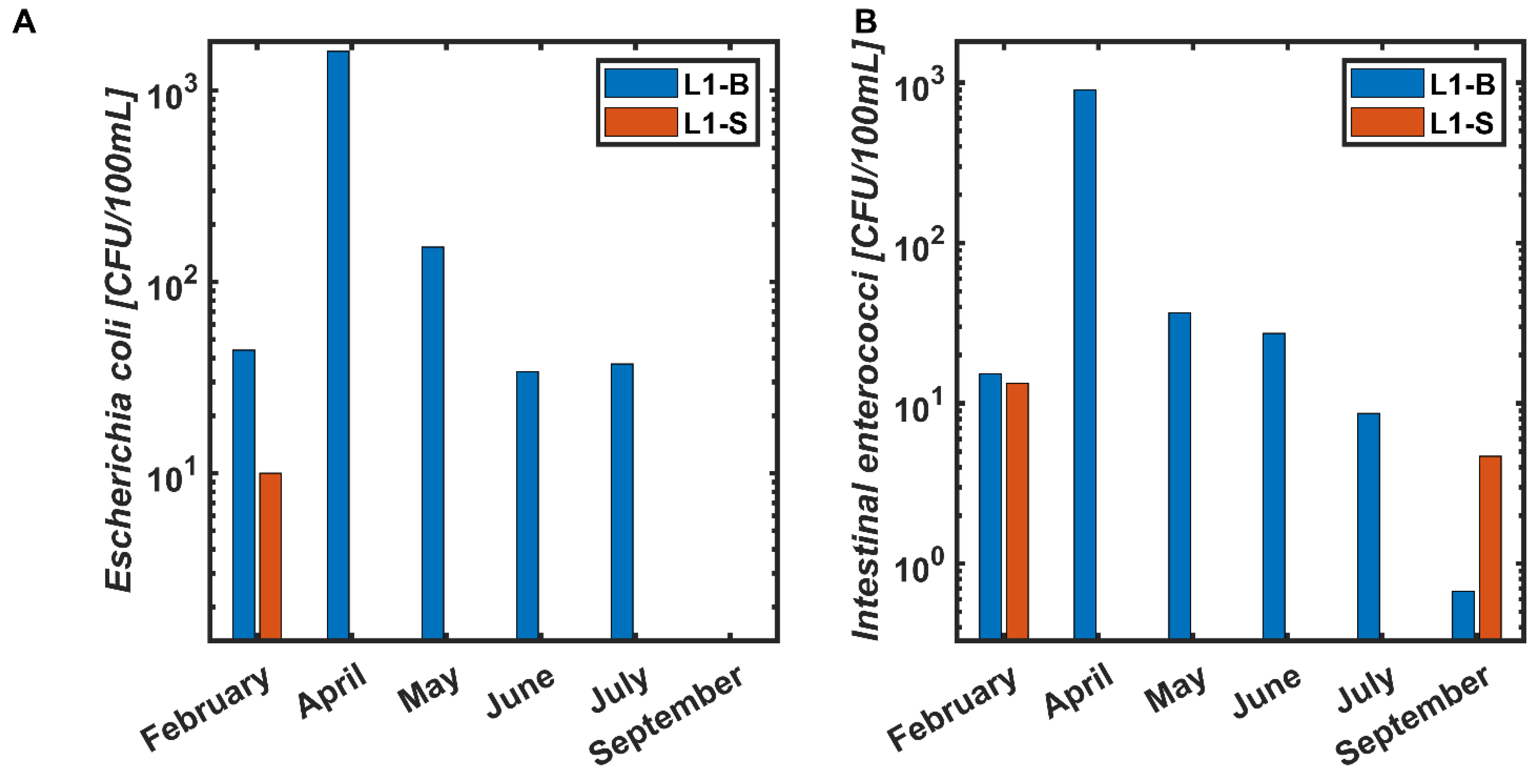
3. Results
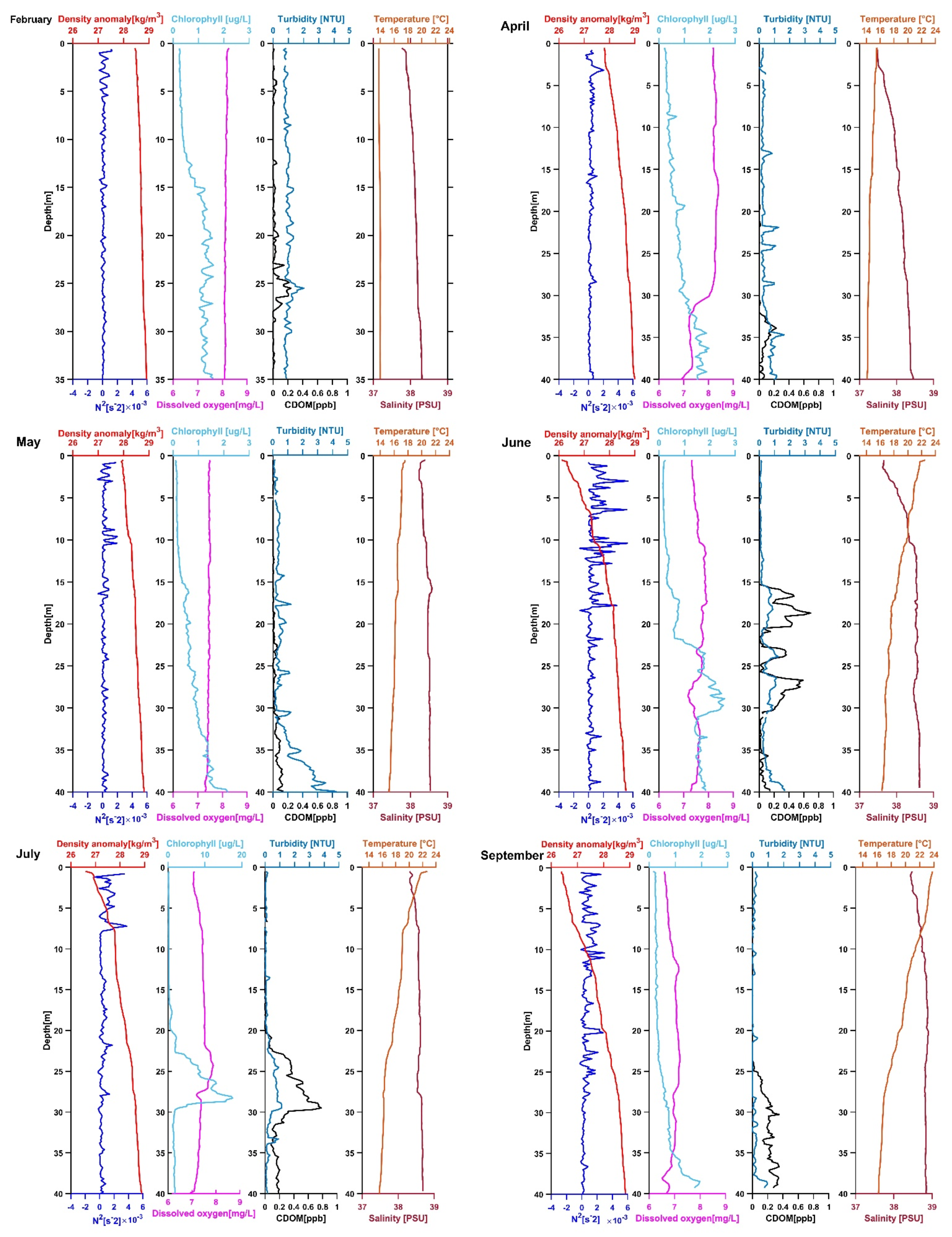
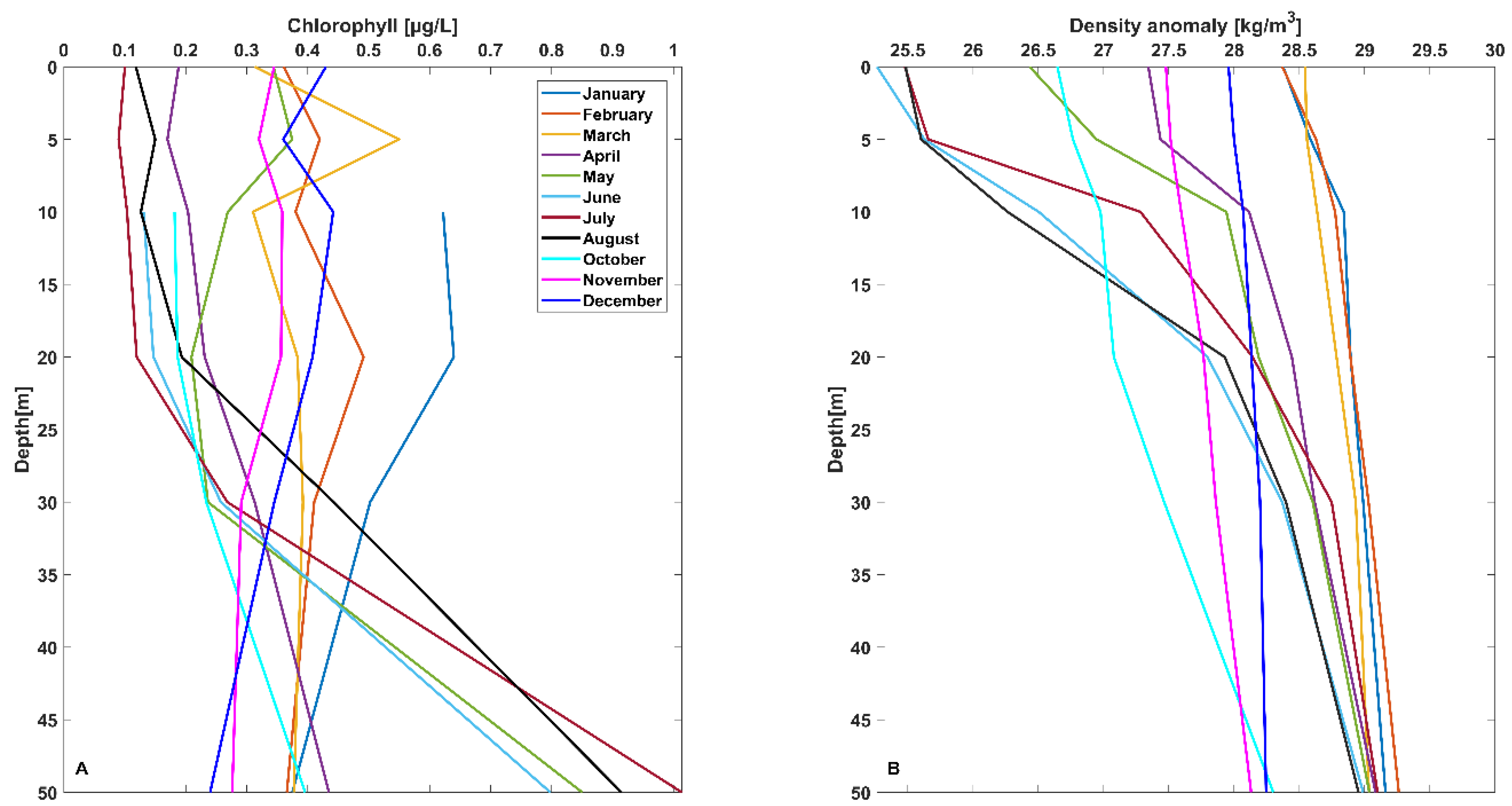
3.1. Location 1
3.1.1. February 2020
3.1.2. April 2020
3.1.3. May 2020
3.1.4. June 2020
3.1.5. July 2020
3.1.6. September 2020
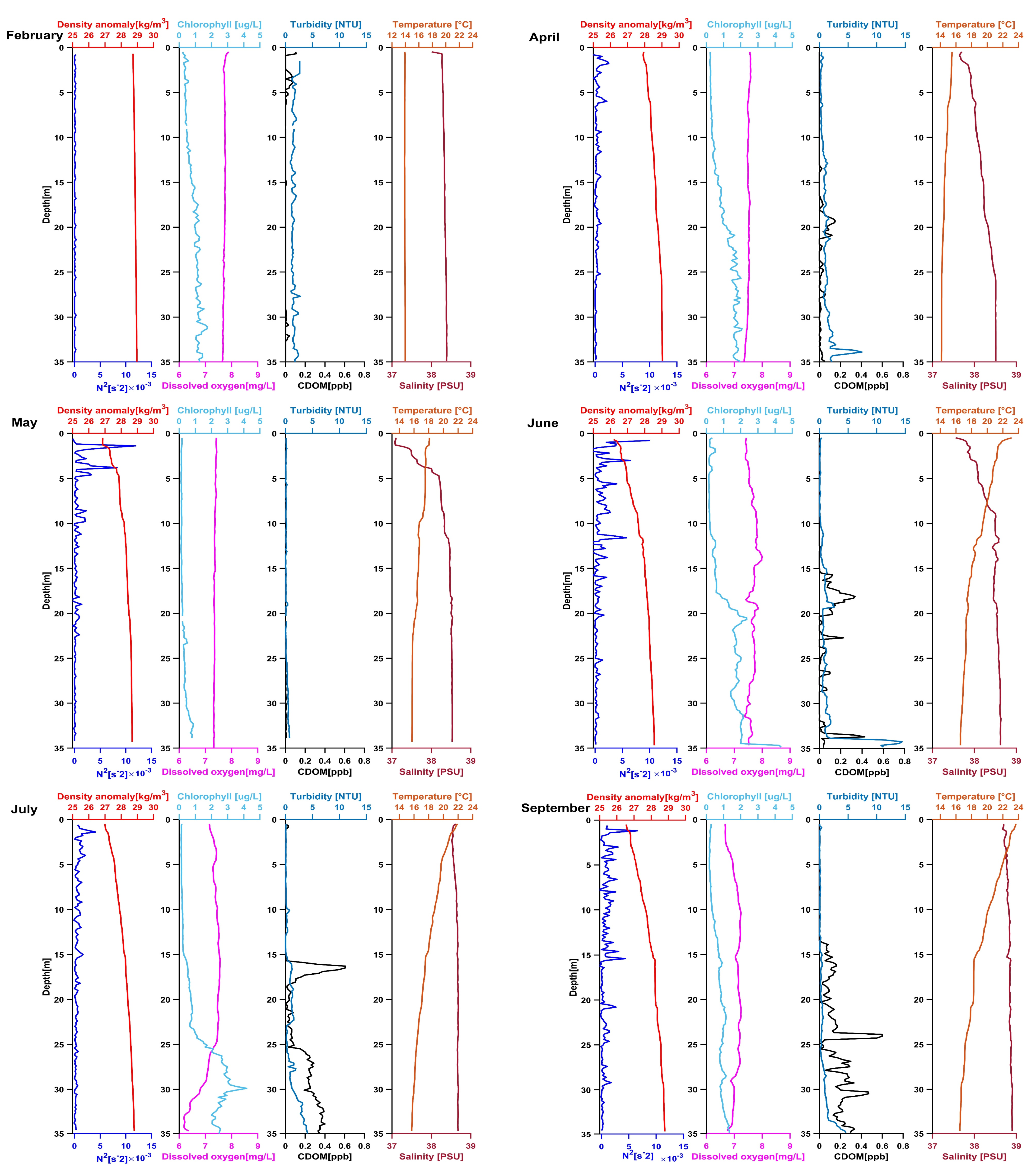
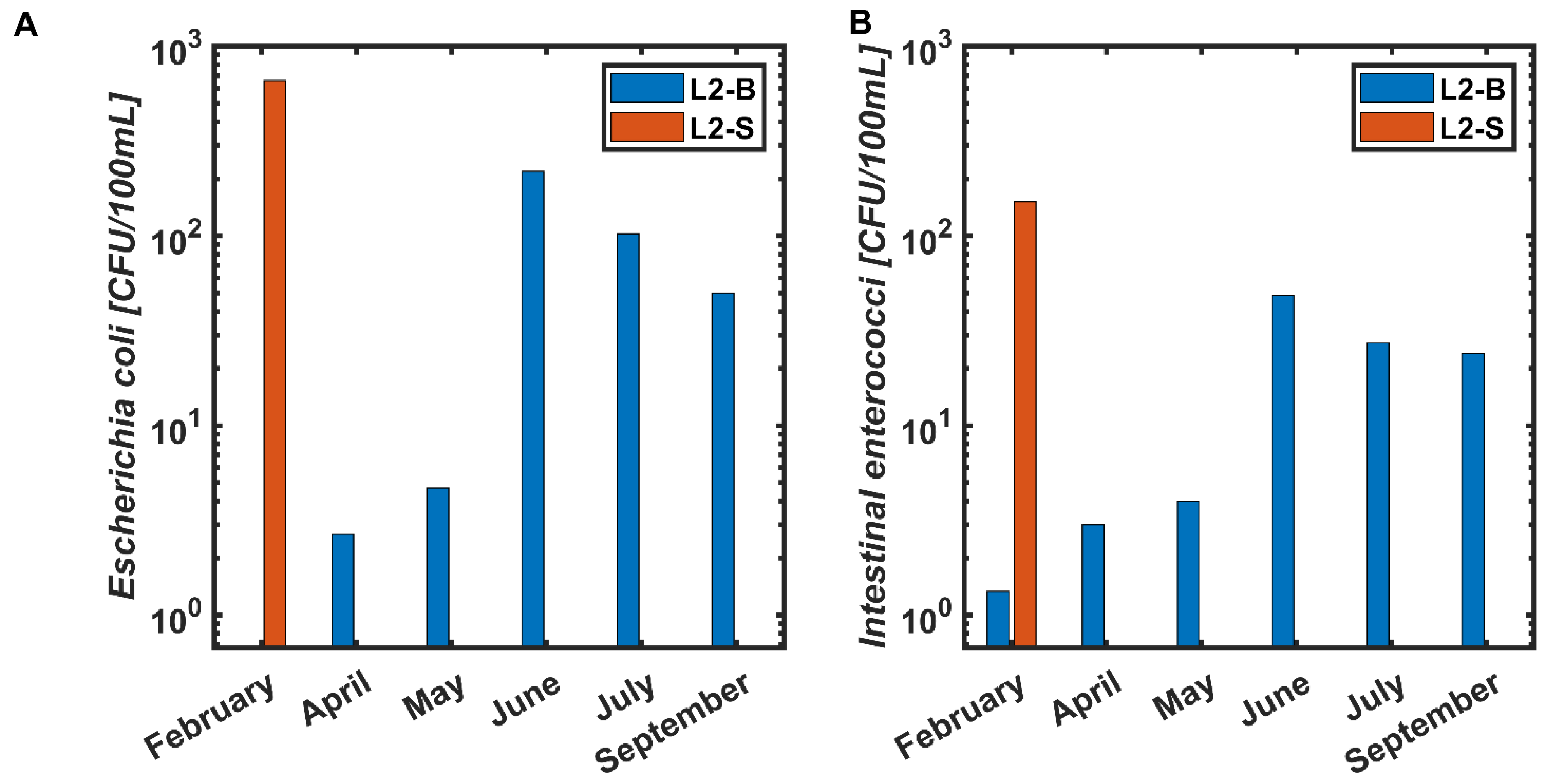
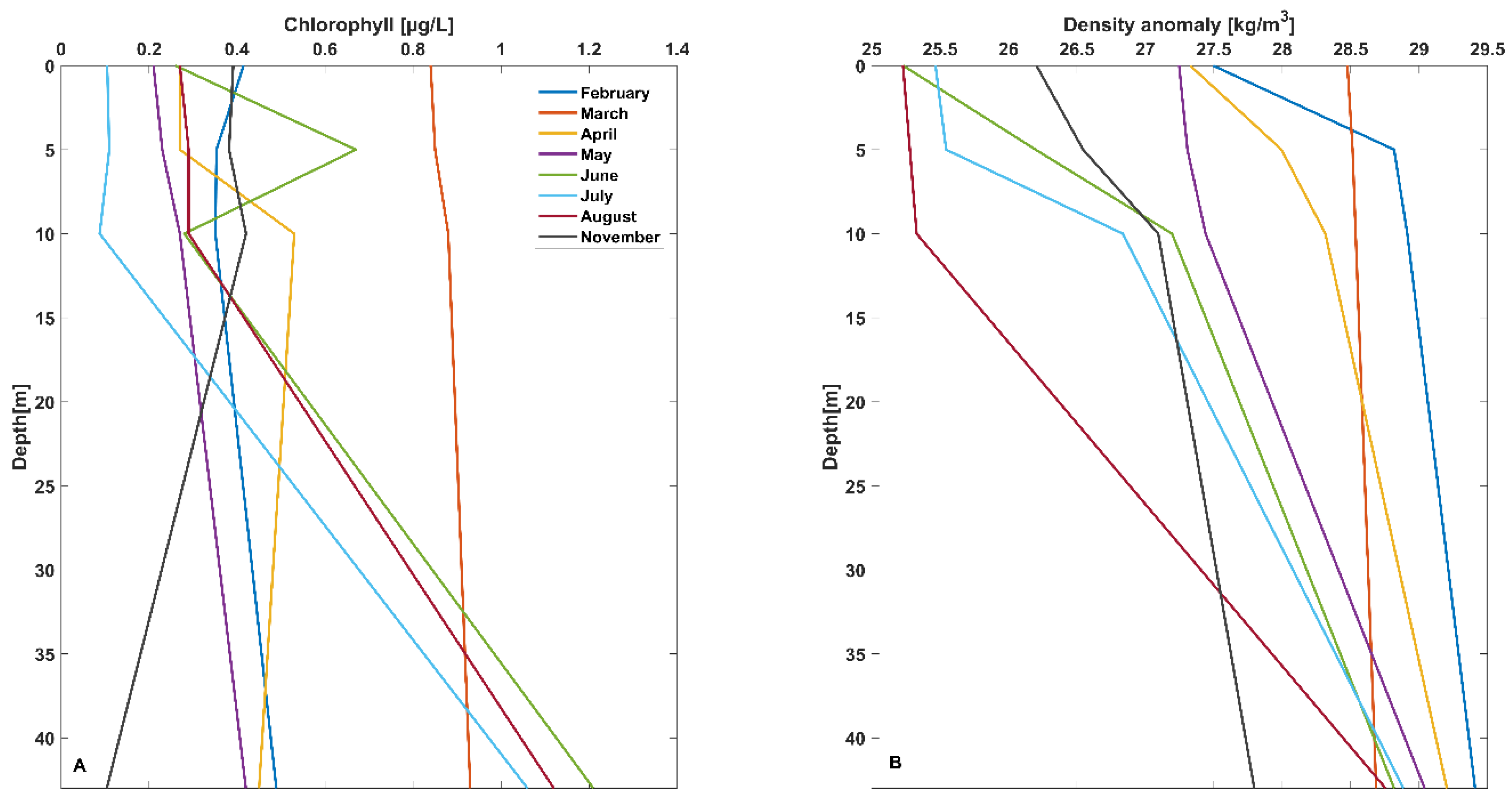
3.2. Location 2
3.2.1. February 2020
3.2.2. April 2020
3.2.3. May 2020
3.2.4. June 2020
3.2.5. July 2020
3.2.6. September 2020
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lorenzen, C.J. A method for the continuous measurement of in vivo chlorophyll concentration. Deep Sea Res. Oceanogr. Abstr. 1966, 13, 223–227. [Google Scholar] [CrossRef]
- Cullen, J.J. The Deep Chlorophyll Maximum: Comparing Vertical Profiles of Chlorophyll a. Can. J. Fish. Aquat. Sci. 1982, 39, 791–803. [Google Scholar] [CrossRef]
- Palter, J.; Coto, S.L.; Ballestero, D. The distribution of nutrients, dissolved oxygen and chlorophyll a in the upper Gulf of Nicoya, Costa Rica, a tropical estuary. Rev. Biol. Trop. 2007, 55, 427–436. [Google Scholar] [CrossRef][Green Version]
- Lavigne, H.; D’Ortenzio, F.; Migon, C.; Claustre, H.; Testor, P.; D’Alcalà, M.R.; Lavezza, R.; Houpert, L.; Prieur, L. Enhancing the comprehension of mixed layer depth control on the Mediterranean phytoplankton phenology. J. Geophys. Res. Ocean. 2013, 118, 3416–3430. [Google Scholar] [CrossRef]
- Lavigne, H.; D’Ortenzio, F.; Ribera D’Alcalà, M.; Claustre, H.; Sauzède, R.; Gacic, M. On the vertical distribution of the chlorophyll a concentration in the Mediterranean Sea: A basin-scale and seasonal approach. Biogeosciences 2015, 12, 5021–5039. [Google Scholar] [CrossRef]
- Dugdale, R.C.; Wilkerson, F.P. Nutrient sources and primary production in the Eastern Mediterranean. Oceanologica Acta. In Proceedings of the Oceanographie Pelagique Mediterraneene, Villefranche-sur-Mer, France, 16–20 September 1988; pp. 179–184. [Google Scholar]
- Bosc, E.; Bricaud, A.; Antoine, D. Seasonal and interannual variability in algal biomass and primary production in the Mediterranean Sea, as derived from 4 years of SeaWiFS observations. Glob. Biogeochem. Cycles 2004, 18. [Google Scholar] [CrossRef]
- Barale, V.; Jaquet, J.M.; Ndiaye, M. Algal blooming patterns and anomalies in the Mediterranean Sea as derived from the SeaWiFS data set (1998–2003). Remote Sens. Environ. 2008, 112, 3300–3313. [Google Scholar] [CrossRef]
- D’Ortenzio, F.; Ribera d’Alcalà, M. On the trophic regimes of the Mediterranean Sea: A satellite analysis. Biogeosciences 2009, 6, 139–148. [Google Scholar] [CrossRef]
- Volpe, G.; Nardelli, B.B.; Cipollini, P.; Santoleri, R.; Robinson, I.S. Seasonal to interannual phytoplankton response to physical processes in the Mediterranean Sea from satellite observations. Remote Sens. Environ. 2012, 117, 223–235. [Google Scholar] [CrossRef]
- Falkowski, P.; Woodhead, A. Primary Productivity and Biogeochemical Cycles in the Sea; Springer: Heidelberg, Germany, 1992; Volume 43, ISBN 0306441926. [Google Scholar]
- Macías, D.; Stips, A.; Garcia-Gorriz, E. The relevance of deep chlorophyll maximum in the open Mediterranean Sea evaluated through 3D hydrodynamic-biogeochemical coupled simulations. Ecol. Modell. 2014, 281, 26–37. [Google Scholar] [CrossRef]
- Crise, A.; Allen, J.I.; Baretta, J.; Crispi, G.; Mosetti, R.; Solidoro, C. The Mediterranean pelagic ecosystem response to physical forcing. Prog. Oceanogr. 1999, 44, 219–243. [Google Scholar] [CrossRef]
- Moutin, T.; Raimbault, P. Primary production, carbon export and nutrients availability in western and eastern Mediterranean Sea in early summer 1996 (MINOS cruise). J. Mar. Syst. 2002, 33–34, 273–288. [Google Scholar] [CrossRef]
- Crombet, Y.; Leblanc, K.; Quéuiner, B.; Moutin, T.; Rimmelin, P.; Ras, J.; Claustre, H.; Leblond, N.; Oriol, L.; Pujo-Pay, M. Deep silicon maxima in the stratified oligotrophic Mediterranean Sea. Biogeosciences 2011, 8, 459–475. [Google Scholar] [CrossRef]
- Dolan, J.R.; Claustre, H.; Carlotti, F.; Plounevez, S.; Moutin, T. Microzooplankton diversity: Relationships of tintinnid ciliates with resources, competitors and predators from the Atlantic Coast of Morocco to the Eastern Mediterranean. Deep. Res. Part I Oceanogr. Res. Pap. 2002, 49, 1217–1232. [Google Scholar] [CrossRef]
- Christaki, U.; Giannakourou, A.; Van Wambeke, F.; Grégori, G. Nanoflagellate predation on auto- and heterotrophic picoplankton in the oligotrophic Mediterranean Sea. J. Plankton Res. 2001, 23, 1297–1310. [Google Scholar] [CrossRef]
- Estrada, M.; Marrase, C.; Latasa, M.; Berdalet, E.; Delgado, M.; Riera, T. Variability of deep chlorophyll maximum characteristics in the northwestern Mediterranean. Mar. Ecol. Prog. Ser. 1993, 92, 289–300. [Google Scholar] [CrossRef]
- Casotti, R.; Landolfi, A.; Brunet, C.; D’Ortenzio, F.; Mangoni, O.; Ribera d’Alcalà, M.; Denis, M. Composition and dynamics of the phytoplankton of the Ionian Sea (eastern Mediterranean). J. Geophys. Res. Ocean. 2003, 108. [Google Scholar] [CrossRef]
- Marty, J.C.; Chiavérini, J.; Pizay, M.D.; Avril, B. Seasonal and interannual dynamics of nutrients and phytoplankton pigments in the western Mediterranean Sea at the DYFAMED time-series station (1991–1999). Deep. Res. Part II Top. Stud. Oceanogr. 2002, 49, 1965–1985. [Google Scholar] [CrossRef]
- Psarra, S.; Tselepides, A.; Ignatiades, L. Primary productivity in the oligotrophic Cretan Sea (NE Mediterranean): Seasonal and interannual variability. Prog. Oceanogr. 2000, 46, 187–204. [Google Scholar] [CrossRef]
- Krom, M.D.; Brenner, S.; Kress, N.; Neori, A.; Gordon, L.I. Nutrient dynamics and new production in a warm-core eddy from the Eastern Mediterranean Sea. Deep Sea Res. Part A Oceanogr. Res. Pap. 1992, 39, 467–480. [Google Scholar] [CrossRef]
- Siokou-Frangou, I.; Christaki, U.; Mazzocchi, M.G.; Montresor, M.; Ribera D’Alcala, M.; Vaque, D.; Zingone, A. Plankton in the open mediterranean Sea: A review. Biogeosciences 2010, 7, 1543–1586. [Google Scholar] [CrossRef]
- Barale, V.; Rizzoli, P.M.; Hendershott, M.C. Remotely sensing the surface dynamics of the Adriatic Sea. Deep Sea Res. Part A Oceanogr. Res. Pap. 1984, 31, 1433–1459. [Google Scholar] [CrossRef]
- Barale, V.; McClain, C.R.; Malanotte-Rizzoli, P. Space and time variability of the surface color field in the northern Adriatic Sea. J. Geophys. Res. 1986, 91, 12957. [Google Scholar] [CrossRef]
- Böhm, E.; Banzon, V.; D’Acunzo, E.; D’Ortenzio, F.; Santoleri, R. Adriatic Sea surface temperature and ocean colour variability during the MFSPP. Ann. Geophys. 2003, 21, 137–149. [Google Scholar] [CrossRef]
- Krivokapić, S.; Stanković, Ž.; Vuksanović, N. Seasonal variations of phytoplankton biomass and environmental conditions in the inner Boka Kotorska Bay (Eastern Adriatic sea). Acta Bot. Croat. 2009, 68, 45–55. [Google Scholar]
- Ninčević, Ž.; Marasović, I.; Kušpilić, G. Deep chlorophyll-a maximum at one station in the middle Adriatic Sea. J. Mar. Biol. Assoc. UK 2002, 82, 9–19. [Google Scholar] [CrossRef]
- Mauri, E.; Poulain, P.M.; Južnič-Zonta, Ž. MODIS chlorophyll variability in the northern Adriatic Sea and relationship with forcing parameters. J. Geophys. Res. Ocean. 2007, 112. [Google Scholar] [CrossRef]
- Kišević, M.; Morović, M.; Smailbegović, A.; Andričević, R. Above and in-water measurements of reflectance and chlorophyll algorithms in the Kaštela Bay in the Adriatic Sea. In Proceedings of the 2nd Workshop on Hyperspectral Image and Signal Processing: Evolution in Remote Sensing, WHISPERS 2010—Workshop Program, Reykjavik, Iceland, 14–16 June 2010; pp. 1–4. [Google Scholar] [CrossRef]
- Mozetič, P.; Solidoro, C.; Cossarini, G.; Socal, G.; Precali, R.; Francé, J.; Bianchi, F.; De Vittor, C.; Smodlaka, N.; Fonda Umani, S. Recent trends towards oligotrophication of the northern adriatic: Evidence from chlorophyll a time series. Estuaries Coasts 2010, 33, 362–375. [Google Scholar] [CrossRef]
- Cozzi, S.; Cabrini, M.; Kralj, M.; De Vittor, C.; Celio, M.; Giani, M. Climatic and anthropogenic impacts on environmental conditions and phytoplankton community in the gulf of trieste (Northern adriatic sea). Water 2020, 12, 2652. [Google Scholar] [CrossRef]
- Viličić, D.; Orlić, M.; Jasprica, N. The deep chlorophyll maximum in the coastal north eastern Adriatic Sea, July 2007. Acta Bot. Croat. 2008, 67, 33–43. [Google Scholar]
- Mozetič, P.; Malačič, V.; Turk, V. A case study of sewage discharge in the shallow coastal area of the Northern Adriatic Sea (Gulf of Trieste). Mar. Ecol. 2008, 29, 483–494. [Google Scholar] [CrossRef]
- Ninčević-Gladan, Ž.; Bužančić, M.; Kušpilić, G.; Grbec, B.; Matijević, S.; Skejić, S.; Marasović, I.; Morović, M. The response of phytoplankton community to anthropogenic pressure gradient in the coastal waters of the eastern Adriatic Sea. Ecol. Indic. 2015, 56, 106–115. [Google Scholar] [CrossRef]
- Bužančić, M.; Ninčević Gladan, Ž.; Marasović, I.; Kušpilić, G.; Grbec, B. Eutrophication influence on phytoplankton community composition in three bays on the eastern Adriatic coast. Oceanologia 2016, 58, 302–316. [Google Scholar] [CrossRef]
- Teodoro, A.C.; Duleba, W.; Gubitoso, S.; Prada, S.M.; Lamparelli, C.C.; Bevilacqua, J.E. Analysis of foraminifera assemblages and sediment geochemical properties to characterise the environment near Araçá and Saco da Capela domestic sewage submarine outfalls of São Sebastião Channel, São Paulo State, Brazil. Mar. Pollut. Bull. 2010, 60, 536–553. [Google Scholar] [CrossRef] [PubMed]
- Despland, L.M.; Vancov, T.; Aragno, M.; Clark, M.W. Diversity of microbial communities in an attached-growth system using BauxsolTM pellets for wastewater treatment. Sci. Total Environ. 2012, 433, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Seon, J.; Park, Y.; Cho, S.; Lee, T. Electricity generation and microbial community in a submerged-exchangeable microbial fuel cell system for low-strength domestic wastewater treatment. Bioresour. Technol. 2012, 117, 172–179. [Google Scholar] [CrossRef]
- Decision, C. Directive 2006/7/EC of the European Parliament and of the Council of 15 February 2006 concerning the management of bathing water quality and repealing Directive 76/160/EEC. Off. J. Eur. Union 2006, L064, 37–51. [Google Scholar]
- Di Dato, M.; Galešić, M.; Šimundić, P.; Andričević, R. A novel screening tool for the health risk in recreational waters near estuary: The Carrying Capacity indicator. Sci. Total Environ. 2019, 694. [Google Scholar] [CrossRef] [PubMed]
- Brettum, P.; Andersen, T. The use of phytoplankton as indicators of water quality. In Norwegian Institute for Water Research SNO Report; NIVA: Oslo, Norway, 2004; p. 33. [Google Scholar]
- Howarth, R.W.; Sharpley, A.; Walker, D. Sources of nutrient pollution to coastal waters in the United States: Implications for achieving coastal water quality goals. Estuaries 2002, 25, 656–676. [Google Scholar] [CrossRef]
- Halpern, B.S.; Walbridge, S.; Selkoe, K.A.; Kappel, C.V.; Micheli, F.; D’Agrosa, C.; Bruno, J.F.; Casey, K.S.; Ebert, C.; Fox, H.E.; et al. A global map of human impact on marine ecosystems. Science 2008, 319, 948–952. [Google Scholar] [CrossRef]
- Windom, H.L. Contamination of the marine environment from land-based sources. Mar. Pollut. Bull. 1992, 25, 32–36. [Google Scholar] [CrossRef]
- Lapointe, B.E.; Thacker, K.; Hanson, C.; Getten, L. Sewage pollution in Negril, Jamaica: Effects on nutrition and ecology of coral reef macroalgae. Chin. J. Oceanol. Limnol. 2011, 29, 775–789. [Google Scholar] [CrossRef]
- Kiefer, D.A. Fluorescence properties of natural phytoplankton populations. Mar. Biol. 1973, 22, 263–269. [Google Scholar] [CrossRef]
- Röttgers, R.; Koch, B.P. Spectroscopic detection of a ubiquitous dissolved pigment degradation product in subsurface waters of the global ocean. Biogeosciences 2012, 9, 2585–2596. [Google Scholar] [CrossRef]
- Sosik, H.M.; Chisholm, S.W.; Olson, R.J. Chlorophyll fluorescence from single cells: Interpretation of flow cytometric signals. Limnol. Oceanogr. 1989, 34, 1749–1761. [Google Scholar] [CrossRef]
- Kolber, Z.; Falkowski, P.G. Use of active fluorescence to estimate phytoplankton photosynthesis in situ. Limnol. Oceanogr. 1993, 38, 1646–1665. [Google Scholar] [CrossRef]
- Müller, P.; Li, X.P.; Niyogi, K.K. Update on Photosynthesis Non-Photochemical Quenching. A Response to Excess Light Energy. Plant Physiol. 2001, 125, 1558–1566. [Google Scholar] [CrossRef]
- Intergovernmental Oceanographic Commission; Scientific Committee on Oceanic Research; International Association for the Physical Sciences of the Ocean. The international thermodynamic equation of seawater—2010: Calculation and use of thermodynamic properties. In Intergovernmental Oceanographic Commission, Manuals and Guides No. 56; UNESCO: Paris, France, 2010; p. 196. [Google Scholar]
- Petersen, W.; Schroeder, F.; Bockelmann, F.-D. FerryBox—Application of continuous water quality observations along transects in the North Sea. Ocean Dyn. 2011, 61, 1541–1554. [Google Scholar] [CrossRef]
- Cunningham, A. Variability of In-Vivo Chlorophyll Fluorescence and Its Implication for Instrument Development in Bio-Optical Oceanography. Sci. Mar. 1996, 60, 309–315. [Google Scholar]
- Falkowski, P.; Kiefer, D.A. Chlorophyll a fluorescence in phytoplankton: Relationship to photosynthesis and biomass. J. Plankton Res. 1985, 7, 715–731. [Google Scholar] [CrossRef]
- Croatian Standards Institute (HRN EN ISO 9308-1:2014/A1:2017). Water Quality—Enumeration of Escherichia Coli and Coliform Bacteria—Part 1: Membrane Filtration Method for Waters with Low Bacterial Background Flora; Croatian Standards Institute: Zagreb, Croatia, 2017. [Google Scholar]
- Jozić, S.; Vukić Lušić, D.; Ordulj, M.; Frlan, E.; Cenov, A.; Diković, S.; Kauzlarić, V.; Fiorido Đurković, L.; Stilinović Totić, J.; Ivšinović, D.; et al. Performance characteristics of the temperature-modified ISO 9308-1 method for the enumeration of Escherichia coli in marine and inland bathing waters. Mar. Pollut. Bull. 2018, 135, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Croatian Standards Institute (HR EN ISO 7899-2:2000). Water Quality—Detection and Enumeration of Intestinal Enterococci—Part 2: Membrane Filtration Method; Croatian Standards Institute: Zagreb, Croatia, 2000. [Google Scholar]
- Zhao, C.; Maerz, J.; Hofmeister, R.; Röttgers, R.; Wirtz, K.; Riethmüller, R.; Schrum, C. Characterizing the vertical distribution of chlorophyll a in the German Bight. Cont. Shelf Res. 2019, 175, 127–146. [Google Scholar] [CrossRef]
- Goosen, N.K.; Kromkamp, J.; Peene, J.; Van Rijswijk, P.; Van Breugel, P. Bacterial and phytoplankton production in the maximum turbidity zone of three European estuaries: The Elbe, Westerschelde and Gironde. J. Mar. Syst. 1999, 22, 151–171. [Google Scholar] [CrossRef]
- Ni, X.; Huang, D.; Zeng, D.; Zhang, T.; Li, H.; Chen, J. The impact of wind mixing on the variation of bottom dissolved oxygen off the Changjiang Estuary during summer. J. Mar. Syst. 2016, 154, 122–130. [Google Scholar] [CrossRef]
- Epifanio, C.E.; Maurer, D.; Dittel, A.I. Seasonal changes in nutrients and dissolved oxygen in the Gulf of Nicoya, a tropical estuary on the Pacific coast of Central America. Hydrobiologia 1983, 101, 231–238. [Google Scholar] [CrossRef]
- Frankignoulle, M.; Abril, G.; Borges, A.; Bourge, I.; Canon, C.; Delille, B.; Libert, E.; Théate, J.M. Carbon dioxide emission from European estuaries. Science 1998, 282, 434–436. [Google Scholar] [CrossRef] [PubMed]
- Kress, N.; Coto, S.L.; Brenes, C.L.; Brenner, S.; Arroyo, G. Horizontal transport and seasonal distribution of nutrients, dissolved oxygen and chlorophyll-a in the Gulf of Nicoya, Costa Rica: A tropical estuary. Cont. Shelf Res. 2002, 22, 51–66. [Google Scholar] [CrossRef]
- Gazeau, F.; Smith, S.V.; Gentili, B.; Frankignoulle, M.; Gattuso, J.P. The European coastal zone: Characterization and first assessment of ecosystem metabolism. Estuar. Coast. Shelf Sci. 2004, 60, 673–694. [Google Scholar] [CrossRef]
- Gazeau, F.; Gattuso, J.P.; Middelburg, J.J.; Brion, N.; Schiettecatte, L.S.; Frankignoulle, M.; Borges, A.V. Planktonic and whole system metabolism in a nutrient-rich estuary (the Scheldt estuary). Estuaries 2005, 28, 868–883. [Google Scholar] [CrossRef]
- Yuan, X.; Yin, K.; Harrison, P.J.; Cai, W.J.; He, L.; Xu, J. Bacterial production and respiration in subtropical Hong Kong waters: Influence of the Pearl River discharge and sewage effluent. Aquat. Microb. Ecol. 2010, 58, 167–179. [Google Scholar] [CrossRef]
- Stanley, D.W.; Nixon, S.W. Stratification and bottom-water hypoxia in the Pamlico River estuary. Estuaries 1992, 15, 270–281. [Google Scholar] [CrossRef]
- Breitburg, D.L.; Adamack, A.; Rose, K.A.; Kolesar, S.E.; Decker, M.B.; Purcell, J.E.; Keister, J.E.; Cowan, J.H. The pattern and influence of low dissolved oxygen in the Patuxent River, a seasonally hypoxic estuary. Estuaries 2003, 26, 280–297. [Google Scholar] [CrossRef]
- Bergondo, D.L.; Kester, D.R.; Stoffel, H.E.; Woods, W.L. Time-series observations during the low sub-surface oxygen events in Narragansett Bay during summer 2001. Mar. Chem. 2005, 97, 90–103. [Google Scholar] [CrossRef]
- Roth, F.; Lessa, G.C.; Wild, C.; Kikuchi, R.K.P.; Naumann, M.S. Impacts of a high-discharge submarine sewage outfall on water quality in the coastal zone of Salvador (Bahia, Brazil). Mar. Pollut. Bull. 2016, 106, 43–48. [Google Scholar] [CrossRef]
- Ramos, P.A.; Neves, M.V.; Pereira, F.L. Mapping and initial dilution estimation of an ocean outfall plume using an autonomous underwater vehicle. Cont. Shelf Res. 2007, 27, 583–593. [Google Scholar] [CrossRef]
- Petrenko, A.A. Effects of a sewage plume on the biology, optical characteristics, and particle size distributions of coastal waters. J. Geophys. Res. C Ocean. 1997, 102, 25061–25071. [Google Scholar] [CrossRef]
- Washburn, B.L.; Jones, B.H.; Bratkovich, A.; Dickey, D.; Chen, M. Mixing, Dispersion, and resuspension in vicinity of ocean wastewater plume. J. Hydraul. Eng. 1992, 118, 38–58. [Google Scholar] [CrossRef]
| Location/Parameter | QSUMMER (m3/Day) | QWINTER (m3/Day) | COD (mg/L) | BOD (mg/L) | TSS (mg/L) | TN (mg/L) | TP (mg/L) |
|---|---|---|---|---|---|---|---|
| Stupe | 30.000 | 26.000 | 372 ± 235 | 199 ± 113 | 89 ± 59 | 52 ± 15 | 6 ± 1.5 |
| Katalinića brig | 38.000 | 34.000 | 246 ± 107 | 135 ± 58 | 69 ± 38 | 32 ± 5 | 4 ± 2 |
| Location 1 | Location 2 | |||||
|---|---|---|---|---|---|---|
| Month | Stratification | Effluent | Type | Stratification | Effluent | Type |
| February | X | X | UCP | X | X | UCP |
| April | X | √ | UCP-ECP | X | X | UCP |
| May | √ | X | BCP | √ | X | BCP |
| June | √ | √ | ECP | √ | √ | ECP |
| July | √ | √ | ECP | √ | √ | ECP |
| September | √ | √ | ECP | √ | X | BCP |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kvesić, M.; Vojković, M.; Kekez, T.; Maravić, A.; Andričević, R. Spatial and Temporal Vertical Distribution of Chlorophyll in Relation to Submarine Wastewater Effluent Discharges. Water 2021, 13, 2016. https://doi.org/10.3390/w13152016
Kvesić M, Vojković M, Kekez T, Maravić A, Andričević R. Spatial and Temporal Vertical Distribution of Chlorophyll in Relation to Submarine Wastewater Effluent Discharges. Water. 2021; 13(15):2016. https://doi.org/10.3390/w13152016
Chicago/Turabian StyleKvesić, Marija, Marin Vojković, Toni Kekez, Ana Maravić, and Roko Andričević. 2021. "Spatial and Temporal Vertical Distribution of Chlorophyll in Relation to Submarine Wastewater Effluent Discharges" Water 13, no. 15: 2016. https://doi.org/10.3390/w13152016
APA StyleKvesić, M., Vojković, M., Kekez, T., Maravić, A., & Andričević, R. (2021). Spatial and Temporal Vertical Distribution of Chlorophyll in Relation to Submarine Wastewater Effluent Discharges. Water, 13(15), 2016. https://doi.org/10.3390/w13152016







