Diffuse Water Pollution from Agriculture: A Review of Nature-Based Solutions for Nitrogen Removal and Recovery
Abstract
1. Introduction
2. Nitrogen Removal Mechanisms
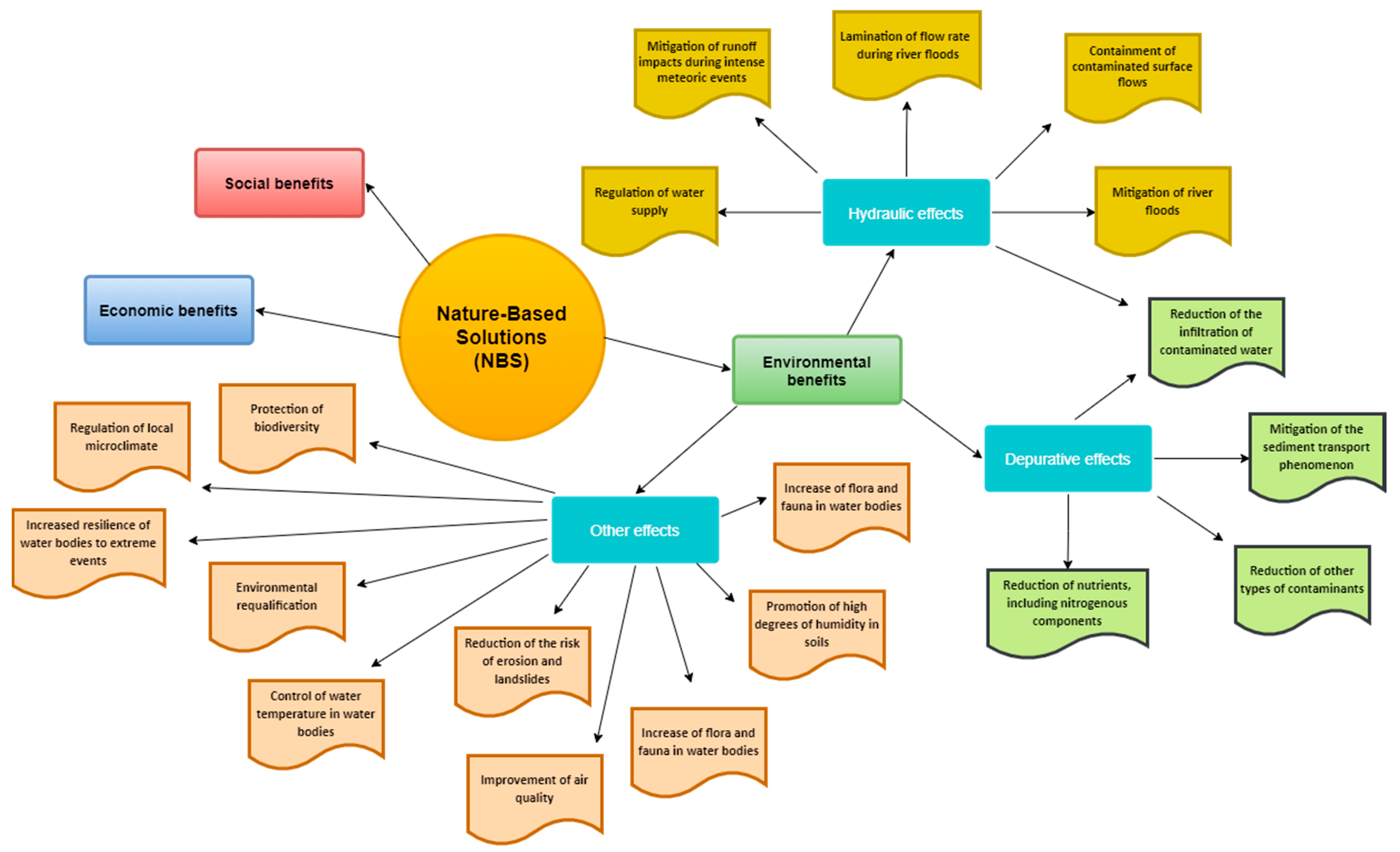
3. Nature-Based Solutions
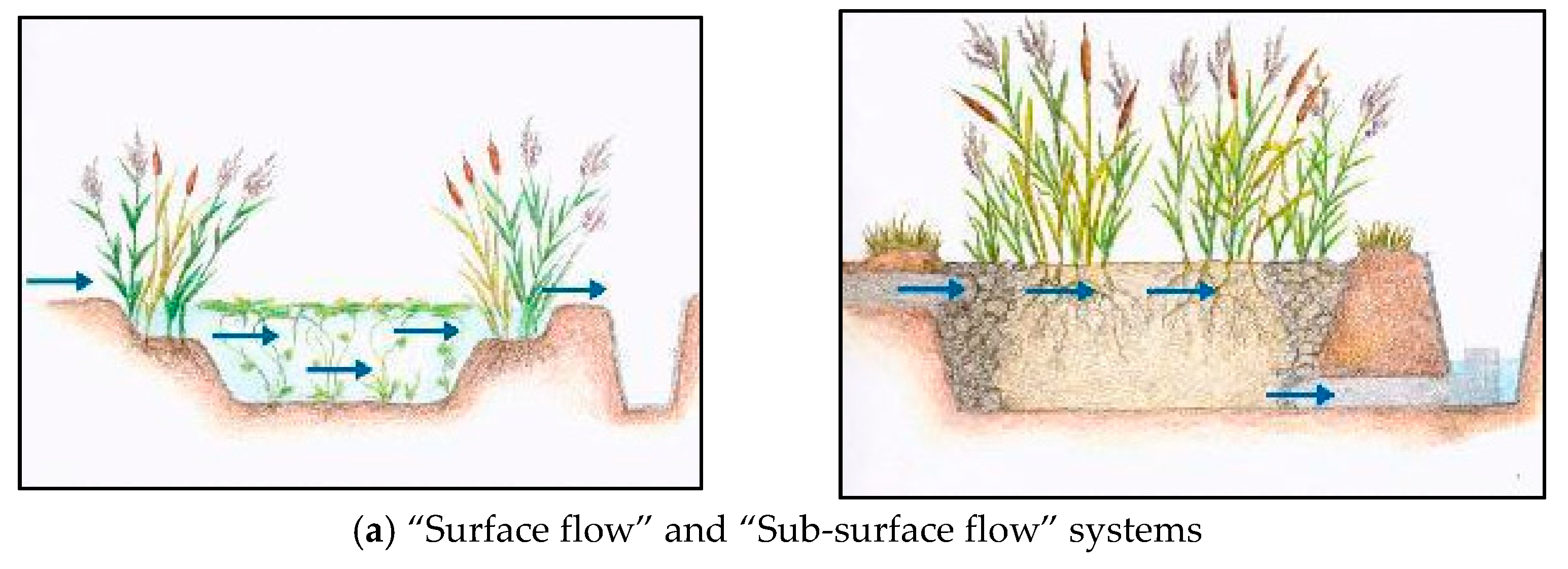
3.1. Constructed Wetlands
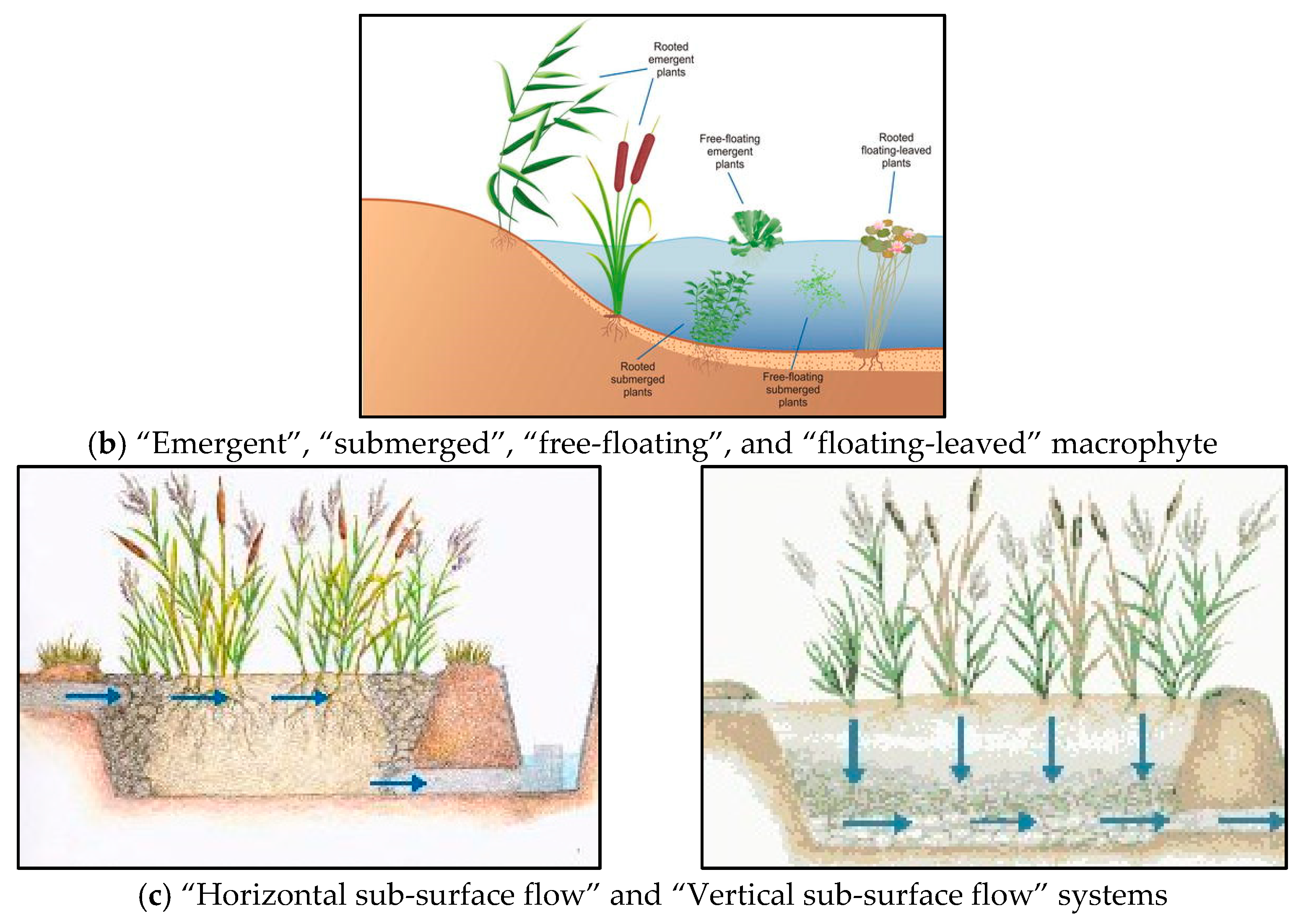
3.1.1. Vegetation Typology
3.1.2. Substrate Materials
3.1.3. Microbial Communities
3.1.4. Improved Systems and the Influence of Different Operating Conditions
3.2. Buffer Strips
3.2.1. Vegetation Typology
3.2.2. Influence of Dimensional Parameters and Operating Conditions
3.3. Other Types of NBSs
3.3.1. Vegetated Channels
3.3.2. Water Sediment Control Basins
4. Nitrogen Recovery Strategies
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FAO (Food and Agriculture Organization of the United Nations). Fertilizer and plant nutrition bullettin. In Plant Nutrition for Food Security: A Guide for Integrated Nutrient Management; FAO: Rome, Italy, 2006. [Google Scholar]
- Smith, L.E.D.; Siciliano, G. A comprehensive review of constraints to improved management of fertilizers in China and mitigation of diffuse water pollution from agriculture. Agric. Ecosyst. Environ. 2005, 209, 15–25. [Google Scholar] [CrossRef]
- Mas-Pla, J.; Menció, A. Groundwater nitrate pollution and climate change: Learnings from a water balance-based analysis of several aquifers in a western Mediterranean region (Catalonia). Environ. Sci. Pollut. Res. 2019, 26, 2184–2202. [Google Scholar] [CrossRef] [PubMed]
- Scanlon, B.R.; Jolly, I.; Sophocleous, M.; Zhang, L. Global impacts of conversions from natural to agricultural ecosystems on water resources: Quantity versus quality. Water Resour. Res. 2007, 43, 1–18. [Google Scholar] [CrossRef]
- Huang, J.; Xu, C.C.; Ridoutt, B.G.; Wang, X.C.; Ren, P. Nitrogen and phosphorus losses and eutrophication potential associated with fertilizer application to cropland in China. J. Clean. Prod. 2017, 159, 171–179. [Google Scholar] [CrossRef]
- Liang, L.Z.; Zhao, X.Q.; Yi, X.Y.; Chen, Z.C.; Dong, X.Y.; Chen, R.F.; Shen, R.F. Excessive application of nitrogen and phosphorus fertilizers induces soil acidification and phosphorus enrichment during vegetable production in Yangtze River Delta, China. Soil Use Manag. 2013, 29, 161–168. [Google Scholar] [CrossRef]
- Van Miegroet, H. The relative importance of sulfur and nitrogen compounds in the acidification of freshwater. In Proceedings of the Dahlem Workshop on Acidification of Freshwater Ecosystems, Berlin, Germany, 27 September–2 October 1992. [Google Scholar]
- Sutton, M.A.; Mason, K.E.; Sheppard, L.J.; Sverdrup, H.; Haeuber, R.; Hicks, W.K. Nitrogen Deposition, Critical Loads and Biodiversity; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- FAOSTAT Environment Statistics of Food and Agriculture Organization of the United Nations (FAO), Rome. 2019. Available online: http://www.fao.org/faostat/en/#data/RFN/visualize (accessed on 14 July 2020).
- Smith, K.A. Nitrous Oxide and Climate Change; Earthscan: London, UK, 2010. [Google Scholar]
- FAOSTAT Environment Statistics of Food and Agriculture Organization of the United Nations (FAO), Rome—Ferilizers by Nutrient. 2019. Available online: http://www.fao.org/faostat/en/ (accessed on 27 July 2020).
- EEA European Environment Agency. Agriculture and Environment in EU-15: The IRENA Indicator Report; EEA Report No. 6/2005; EEA: Copenhagen, Denmark, 2005. [Google Scholar]
- EEA European Environment Agency. Environmental Indicator Report 2018—In Support to the Monitoring of the Seventh Environment Action Programme; EEA Report|N. 19/2018; Publications Office of the European Union: Luxembourg, 2018. [Google Scholar]
- Council Directive 91/676/EEC of 12 December 1991 concerning the Protection of Waters against Pollution Caused by Nitrates from Agricultural Sources; Council Directive: Brussels, Belgium, 1991.
- Baker, J.L. Limitations of improved nitrogen management to reduce nitrate leaching and increase use efficiency. In Optimizing Nitrogen Management in Food and Energy Production and Environmental Protection, Proceedings of the 2nd International Nitrogen Conference on Science and Policy, Potomac, MD, USA, 14–18 October 2001; TheScientificWorld, Hindawi: London, UK, 2001; pp. 10–16. [Google Scholar]
- IUCN. International Union for Conservation of Nature; IUCN: Marseille, France, 2020. [Google Scholar]
- Marcatili, M.; Giordano, S.; Toscano, A.; Mancuso, G.; Perez-Blanco, D. Valutazione dei Servizi Ecosistemici Della Gestione Integrata Delle Acque dei Canali; Report Tecnico; Autorità del bacino Po: Parma, Italy, 2020. [Google Scholar]
- Vymazal, J. The use of constructed wetlands for nitrogen removal from agricultural drainage: A review. Sci. Agric. Bohem. 2017, 48, 82–91. [Google Scholar] [CrossRef]
- Dollinger, J.; Dagès, C.; Bailly, J.S.; Lagacherie, P.; Voltz, M. Managing ditches for agroecological engineering of landscape. A review. Agron. Sustain. Dev. 2015, 35, 999–1020. [Google Scholar] [CrossRef]
- Hickey, M.B.C.; Doran, B. A review of the efficiency of buffer strips for the maintenance and enhancement of riparian ecosystems. Water Qual. Res. J. Can. 2004, 39, 311–317. [Google Scholar] [CrossRef]
- Khatiwada, N.R.; Polprasert, C. Assessment of effective specific surface area for free water surface constructed wetlands. Water Sci. Technol. 1999, 40, 83–89. [Google Scholar] [CrossRef]
- Hamlett, J.M.; Epp, D.J. Water quality impacts of conservation and nutrient management practices in Pennsylvania. J. Soil Water Conserv. 1994, 49, 59–66. [Google Scholar]
- Vymazal, J. Removal of nutrients in various types of constructed wetlands. Sci. Total Environ. 2007, 380, 48–65. [Google Scholar] [CrossRef]
- Zhu, G.; Jetten, M.S.M.; Kuschk, P.; Ettwig, K.F.; Yin, C. Potential roles of anaerobic ammonium and methane oxidation in the nitrogen cycle of wetland ecosystems. Appl. Microbiol. Biotechnol. 2010, 86, 1043–1055. [Google Scholar] [CrossRef]
- Kartal, B.; Van Niftrik, L.; Rattray, J.; Van De Vossenberg, J.L.C.M.; Schmid, M.C.; Sinninghe Damsté, J.; Jetten, M.S.M.; Strous, M. Candidatus “Brocadia fulgida”: An autofluorescent anaerobic ammonium oxidizing bacterium. FEMS Microbiol. Ecol. 2008, 63, 46–55. [Google Scholar] [CrossRef]
- Van De Graaf, A.A.; De Bruijn, P.; Robertson, L.A.; Jetten, M.S.M.; Kuenen, J.G. Metabolic pathway of anaerobic ammonium oxidation on the basis of 15N studies in a fluidized bed reactor. Microbiology 1997, 143, 2415–2421. [Google Scholar] [CrossRef] [PubMed]
- Vymazal, J. Algae and Element Cycling in Wetlands; Lewis Publishers: Chelsea, UK, 1995. [Google Scholar]
- Taylor, G.D.; Fletcher, T.D.; Wong, T.H.F.; Breen, P.F. Nitrogen composition in urban runoff—Implication for stormwater management. Water Res. 2005, 39, 1982–1989. [Google Scholar] [CrossRef]
- Vymazal, J. Constructed wetlands for treatment of industrial wastewaters: A review. Ecol. Eng. 2014, 73, 724–751. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Wallace, S. Treatment Wetlands; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2008. [Google Scholar]
- Van de Moortel, A.M.K.; Rousseau, D.P.L.; Tack, F.M.G.; De Pauw, N. A comparative study of surface and subsurface flow constructed wetlands for treatment of combined sewer overflows: A greenhouse experiment. Ecol. Eng. 2009, 35, 175–183. [Google Scholar] [CrossRef]
- Lee, C.G.; Fletcher, T.D.; Sun, G. Nitrogen removal in constructed wetland systems. Eng. Life Sci. 2009, 9, 11–22. [Google Scholar] [CrossRef]
- Cooper, P.F.; Boon, A.G. The use of Phragmites for wastewater treatment by the root zone method: The UK approach. In Aquatic Plants for Water Treatment and Resource Recovery; Reddy, K.R., Smith, W.H., Eds.; Magnolia Publishing Inc.: Orlando, FL, USA, 1987. [Google Scholar]
- Bachand, P.A.M.; Horne, A.J. Denitrification in constructed free-water surface wetlands: II. Effects of vegetation and temperature. Ecol. Eng. 2000, 14, 17–32. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, Y.; Liu, R.; Morgan, D. Global development of various emerged substrates utilized in constructed wetlands. Bioresour. Technol. 2018, 261, 441–452. [Google Scholar] [CrossRef]
- Rajan, R.J.; Sudarsan, J.S.; Nithiyanantham, S. Microbial population dynamics in constructed wetlands: Review of recent advancements for wastewater treatment. Environ. Eng. Res. 2019, 24, 181–190. [Google Scholar] [CrossRef]
- Tanner, C.C. Plants for constructed wetland treatment systems—A comparison of the growth and nutrient uptake of eight emergent species. Ecol. Eng. 1996, 7, 59–83. [Google Scholar] [CrossRef]
- Ge, Y.; Han, W.; Huang, C.; Wang, H.; Liu, D.; Chang, S.X.; Gu, B.; Zhang, C.; Gu, B.; Fan, X.; et al. Positive effects of plant diversity on nitrogen removal in microcosms of constructed wetlands with high ammonium loading. Ecol. Eng. 2015, 82, 614–623. [Google Scholar] [CrossRef]
- EPA. Design Manual: Constructed Wetlands and Aquatic Plant Systems for Municipal Wastewater Treatment; EPA/625/1-88/022; EPA: Cincinnati, OH, USA, 1988. [Google Scholar]
- Brix, H. Do macrophytes play a role in constructed treatment wetlands? Water Sci. Technol. 1997, 35, 11–17. [Google Scholar] [CrossRef]
- Braskerud, B.C. Factors affecting nitrogen retention in small constructed wetlands treating agricultural non-point source pollution. Ecol. Eng. 2002, 18, 351–370. [Google Scholar] [CrossRef]
- Wood, A. Constructed wetlands in water pollution control: Fundamentals to their understanding. Water Sci. Technol. 1995, 32, 21–29. [Google Scholar] [CrossRef]
- Daneshvar, F.; Nejadhashemi, A.P.; Adhikari, U.; Elahi, B.; Abouali, M.; Herman, M.R.; Martinez-Martinez, E.; Calappi, T.J.; Rohn, B.G. Evaluating the significance of wetland restoration scenarios on phosphorus removal. J. Environ. Manag. 2017, 192, 184–196. [Google Scholar] [CrossRef]
- Kröpfelová, L.; Vymazal, J.; Švehla, J.; Štíchová, J. Removal of trace elements in three horizontal sub-surface flow constructed wetlands in the Czech Republic. Environ. Pollut. 2009, 157, 1186–1194. [Google Scholar] [CrossRef]
- Matagi, S.; Swai, D.; Mugabe, R. A review of heavy metal removal mechanisms in wetlands. Afr. J. Trop. Hydrobiol. Fish. 1998, 8, 23–35. [Google Scholar] [CrossRef]
- Stottmeister, U.; Wießner, A.; Kuschk, P.; Kappelmeyer, U.; Kästner, M.; Bederski, O.; Müller, R.A.; Moormann, H. Effects of plants and microorganisms in constructed wetlands for wastewater treatment. Biotechnol. Adv. 2003, 22, 93–117. [Google Scholar] [CrossRef]
- Wang, Y.; Cai, Z.; Sheng, S.; Pan, F.; Chen, F.; Fu, J. Comprehensive evaluation of substrate materials for contaminants removal in constructed wetlands. Sci. Total Environ. 2020, 701, 134736. [Google Scholar] [CrossRef]
- Zhu, W.L.; Cui, L.H.; Ouyang, Y.; Long, C.F.; Tang, X.D. Kinetic adsorption of ammonium nitrogen by substrate materials for constructed wetlands. Pedosphere 2011, 21, 454–463. [Google Scholar] [CrossRef]
- Daothaisong, A.; Yimrattanabovorn, J. Nitrogen adsorption of shale for use as media in constructed wetland. Environ. Eng. Manag. J. 2009, 8, 1073–1079. [Google Scholar]
- Shannon, R.D.; Kirk, B.A.; Flite, O.P.; Hunter, M.S. Constructed wetland nitrogen and phosphorus removal at a rural campground and conference center. In Proceedings of the Ninth National Symosium on Individual and Small Community Sewage Systems—The Radisson Plaza, Fort Worth, TX, USA, 11–14 March 2001. [Google Scholar]
- Cyrus, J.S.; Reddy, G.B. Sorption and desorption of ammonium by zeolite: Batch and column studies. J. Environ. Sci. Health 2011, 46, 408–414. [Google Scholar] [CrossRef]
- Zou, J.; Guo, X.; Han, Y.; Liu, J.; Liang, H. Study of a novel vertical flow constructed wetland system with drop aeration for rural wastewater treatment. Water Air Soil Pollut. 2012, 223, 889–900. [Google Scholar] [CrossRef]
- Srinivasan, R.; Hoffman, D.W.; Wolf, D.W. Evaluation of removal of orthophosphate and ammonia from rainfall runoff using aboveground permeable reactive barrier composed of limestone and zeolite. J. Environ. Sci. Health 2008, 43, 1441–1450. [Google Scholar] [CrossRef]
- Zhao, J.; Zhao, Y.; Xu, Z.; Doherty, L.; Liu, R. Highway runoff treatment by hybrid adsorptive media-baffled subsurface flow constructed wetland. Ecol. Eng. 2016, 91, 231–239. [Google Scholar] [CrossRef]
- Zhao, Y.Q.; Babatunde, A.O.; Zhao, X.H.; Li, W.C. Development of alum sludge-based constructed wetland: An innovative and cost effective system for wastewater treatment. J. Environ. Sci. Health 2009, 44, 827–832. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wendling, L.A.; Douglas, G.B.; Coleman, S.; Yuan, Z. Nutrient and dissolved organic carbon removal from natural waters using industrial by-products. Sci. Total Environ. 2013, 442, 63–72. [Google Scholar] [CrossRef]
- Zhou, I.; Huang, Z.J.; Li, T. Application of Wasted Architecture Walling Materials Used as a Constructed Wetland Media; Iwa Publishing: London, UK, 2010. [Google Scholar]
- Cheng, G.; Li, Q.; Su, Z.; Sheng, S.; Fu, J. Preparation, optimization, and application of sustainable ceramsite substrate from coal fly ash/waterworks sludge/oyster shell for phosphorus immobilization in constructed wetlands. J. Clean. Prod. 2018, 175, 572–581. [Google Scholar] [CrossRef]
- Fu, J.; Lee, W.N.; Coleman, C.; Meyer, M.; Carter, J.; Nowack, K.; Huang, C.H. Pilot investigation of two-stage biofiltration for removal of natural organic matter in drinking water treatment. Chemosphere 2017, 166, 311–322. [Google Scholar] [CrossRef]
- Cheng, Y.; Cheng, J.; Niu, S.; Kim, Y. Evaluation of the different filter media in vertical flow stormwater wetland. Desalin. Water Treat. 2013, 51, 4097–4106. [Google Scholar] [CrossRef]
- Cheng, Y.; Guerra, H.B.; Min, K.S.; Kim, Y. Operation of the vertical subsurface flow and partly submersed stormwater wetland with an intermittent recycle. Desalin. Water Treat. 2012, 38, 349–359. [Google Scholar] [CrossRef]
- Calder, N.; Anderson, B.C.; Martin, D.G. Field investigation of advanced filtration for phosphorus removal from constructed treatment wetland effluents. Environ. Technol. 2006, 27, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Molle, P.; Liénard, A.; Grasmick, A.; Iwema, A. Phosphorus retention in subsurface constructed wetlands: Investigations focused on calcareous materials and their chemical reactions. Water Sci. Technol. 2003, 48, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Jesus, J.M.; Calheiros, C.S.C.; Castro, P.M.L.; Borges, M.T. Feasibility of Typha Latifolia for high salinity effluent treatment in constructed wetlands for integration in resource management systems. Int. J. Phytoremed. 2014, 16, 334–346. [Google Scholar] [CrossRef]
- Kozub, D.D.; Liehr, S.K. Assessing denitrification rate limiting factors in a constructed wetland receiving landfill leachate. Water Sci. Technol. 1999, 40, 75–82. [Google Scholar] [CrossRef]
- Tao, W.; Hall, K.J.; Duff, S.J.B. Microbial biomass and heterotrophic production of surface flow mesocosm wetlands treating woodwaste leachate: Responses to hydraulic and organic loading and relations with mass reduction. Ecol. Eng. 2007, 31, 132–139. [Google Scholar] [CrossRef]
- Xu, G.; Lv, Y.; Sun, J.; Shao, H.; Wei, L. Recent advances in biochar applications in agricultural soils: Benefits and environmental implications. Clean Soil Air Water 2012, 40, 1093–1098. [Google Scholar] [CrossRef]
- Kasak, K.; Truu, J.; Ostonen, I.; Sarjas, J.; Oopkaup, K.; Paiste, P.; Kõiv-Vainik, M.; Mander, Ü.; Truu, M. Biochar enhances plant growth and nutrient removal in horizontal subsurface flow constructed wetlands. Sci. Total Environ. 2018, 639, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chi, Z.; Yan, B.; Cheng, L.; Li, J. An innovative wood-chip-framework substrate used as slow-release carbon source to treat high-strength nitrogen wastewater. J. Environ. Sci. (China) 2017, 51, 275–283. [Google Scholar] [CrossRef]
- Karunarathna, A.K.; Tanaka, N.; Jinadasa, K.B.S.N. Effect of external organic matter on nutrient removal and growth of Phragmites australis in a laboratory-scale subsurface-flow treatment wetland. Water Sci. Technol. 2007, 55, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Groh, T.A.; Gentry, L.E.; David, M.B. Nitrogen Removal and Greenhouse Gas Emissions from Constructed Wetlands Receiving Tile Drainage Water. J. Environ. Qual. 2015, 44, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.R.; Patrick, W.H.; Lindau, C.W. Nitrification-denitrification at the plant root-sediment interface in wetlands. Limnol. Oceanogr. 1989, 34, 1004–1013. [Google Scholar] [CrossRef]
- Mander, Ü.; Tournebize, J.; Espenberg, M.; Chaumont, C.; Torga, R.; Garnier, J.; Muhel, M.; Maddison, M.; Lebrun, J.D.; Uher, E.; et al. High denitrification potential but low nitrous oxide emission in a constructed wetland treating nitrate-polluted agricultural run-off. Sci. Total Environ. 2021, 779, 146614. [Google Scholar] [CrossRef]
- Haandel, A.; Lubbe, J. Handbook of Biological Waste Water Treatment. Design and Optimization of Activated Sludge Systems; Quist Publishing: Leidschendam, The Netherlands, 2007. [Google Scholar]
- Meng, P.; Pei, H.; Hu, W.; Shao, Y.; Li, Z. How to increase microbial degradation in constructed wetlands: Influencing factors and improvement measures. Bioresour. Technol. 2014, 157, 316–326. [Google Scholar] [CrossRef]
- Jia, W.; Zhang, J.; Wu, J.; Xie, H.; Zhang, B. Effect of intermittent operation on contaminant removal and plant growth in vertical flow constructed wetlands: A microcosm experiment. Desalination 2010, 262, 202–208. [Google Scholar] [CrossRef]
- Sànchez Constructed wetlands revisited: Microbial diversity in the –omics Era. Microb. Ecol. 2017, 73, 722–733. [CrossRef]
- Riva, V.; Riva, F.; Vergani, L.; Crotti, E.; Borin, S.; Mapelli, F. Microbial assisted phytodepuration for water reclamation: Environmental benefits and threats. Chemosphere 2020, 241, 124843. [Google Scholar] [CrossRef]
- Kadlec, R.H. Comparison of free water and horizontal subsurface treatment wetlands. Ecol. Eng. 2009, 35, 159–174. [Google Scholar] [CrossRef]
- Pucher, B.; Langergraber, G. The state of the art of clogging in vertical flow wetlands. Water 2019, 11, 2400. [Google Scholar] [CrossRef]
- Sengorur, B.; Ozdemir, S. Performance of a constructed wetland system for the treatment of domestic wastewater. Fresenius Environ. Bull. 2006, 15, 242–244. [Google Scholar]
- Nivala, J.; Wallace, S.; Headley, T.; Kassa, K.; Brix, H.; van Afferden, M.; Müller, R. Oxygen transfer and consumption in subsurface flow treatment wetlands. Ecol. Eng. 2013, 61, 544–554. [Google Scholar] [CrossRef]
- Kantawanichkul, S.; Somprasert, S. Using a compact combined constructed wetland system to treat agricultural wastewater with high nitrogen. Water Sci. Technol. 2005, 51, 47–53. [Google Scholar] [CrossRef]
- Saeed, T.; Sun, G. Enhanced denitrification and organics removal in hybrid wetland columns: Comparative experiments. Bioresour. Technol. 2011, 102, 967–974. [Google Scholar] [CrossRef]
- Wu, H.; Fan, J.; Zhang, J.; Ngo, H.H.; Guo, W.; Hu, Z.; Lv, J. Optimization of organics and nitrogen removal in intermittently aerated vertical flow constructed wetlands: Effects of aeration time and aeration rate. Int. Biodeterior. Biodegrad. 2016, 113, 139–145. [Google Scholar] [CrossRef]
- Platzer, C.; Mauch, K. Soil clogging in vertical flow reed beds—Mechanisms, parameters, consequences and.......solutions? Water Sci. Technol. 1997, 35, 175–181. [Google Scholar] [CrossRef]
- Langergraber, G.; Leroch, K.; Pressl, A.; Sleytr, K.; Rohrhofer, R.; Haberl, R. High-rate nitrogen removal in a two-stage subsurface vertical flow constructed wetland. Desalination 2009, 246, 55–68. [Google Scholar] [CrossRef]
- Lavrnić, S.; Nan, X.; Blasioli, S.; Braschi, I.; Anconelli, S.; Toscano, A. Performance of a full scale constructed wetland as ecological practice for agricultural drainage water treatment in Northern Italy. Ecol. Eng. 2020, 154, 105927. [Google Scholar] [CrossRef]
- Al-Isawi, R.; Scholz, M.; Wang, Y.; Sani, A. Clogging of vertical-flow constructed wetlands treating urban wastewater contaminated with a diesel spill. Environ. Sci. Pollut. Res. 2015, 22, 12779–12803. [Google Scholar] [CrossRef] [PubMed]
- Truu, M.; Juhanson, J.; Truu, J. Microbial biomass, activity and community composition in constructed wetlands. Sci. Total Environ. 2009, 407, 3958–3971. [Google Scholar] [CrossRef]
- Yin, X.; Zhang, J.; Hu, Z.; Xie, H.; Guo, W.; Wang, Q.; Ngo, H.H.; Liang, S.; Lu, S.; Wu, W. Effect of photosynthetically elevated pH on performance of surface flow-constructed wetland planted with Phragmites australis. Environ. Sci. Pollut. Res. 2016, 23, 15524–15531. [Google Scholar] [CrossRef] [PubMed]
- Sirivedhin, T.; Gray, K.A. Factors affecting denitrification rates in experimental wetlands: Field and laboratory studies. Ecol. Eng. 2006, 26, 167–181. [Google Scholar] [CrossRef]
- Lavrnić, S.; Cristino, S.; Zapater-Pereyra, M.; Vymazal, J.; Cupido, D.; Lucchese, G.; Mancini, B.; Mancini, M.L. Effect of earthworms and plants on the efficiency of vertical flow systems treating university wastewater. Environ. Sci. Pollut. Res. 2019, 26, 10354–10362. [Google Scholar] [CrossRef] [PubMed]
- Barling, R.D.; Moore, I.D. Role of buffer strips in management of waterway pollution: A review. Environ. Manag. 1994, 18, 543–558. [Google Scholar] [CrossRef]
- Gumiero, B.; Boz, B.; Cornelio, P.; Casella, S. Shallow groundwater nitrogen and denitrification in a newly afforested, subirrigated riparian buffer. J. Appl. Ecol. 2011, 48, 1135–1144. [Google Scholar] [CrossRef]
- Li, Y.; Shao, X.; Sheng, Z. Field experiments on reducing pollutants in agricultural-drained water using soil-vegetation buffer strips. Pol. J. Environ. Stud. 2016, 25, 195–204. [Google Scholar] [CrossRef]
- Dong, Y.; Xiong, D.; Su, Z.; Yang, D.; Zheng, X.; Shi, L.; Poesen, J. Effects of vegetation buffer strips on concentrated flow hydraulics and gully bed erosion based on in situ scouring experiments. Land Degrad. Dev. 2018, 29, 1672–1682. [Google Scholar] [CrossRef]
- Fortier, J.; Gagnon, D.; Truax, B.; Lambert, F. Nutrient accumulation and carbon sequestration in 6-year-old hybrid poplars in multiclonal agricultural riparian buffer strips. Agric. Ecosyst. Environ. 2010, 137, 276–287. [Google Scholar] [CrossRef]
- Uri, N.D. Conservation practices in U.S. agriculture and their impact on carbon sequestration. Environ. Monit. Assess. 2001, 70, 323–344. [Google Scholar] [CrossRef]
- Hefting, M.M.; Clement, J.C.; Bienkowski, P.; Dowrick, D.; Guenat, C.; Butturini, A.; Topa, S.; Pinay, G.; Verhoeven, J.T.A. The role of vegetation and litter in the nitrogen dynamics of riparian buffer zones in Europe. Ecol. Eng. 2005, 24, 465–482. [Google Scholar] [CrossRef]
- Parkyn, S. Review of Riparian Buffer Zone Effectiveness; MAF: Wellington, New Zealand, 2004; ISBN 0478078234. [Google Scholar]
- Helmers, M.J.; Isenhart, T.; Dosskey, M.; Dabney, S.; Strock, J. Buffers and vegetative filter strips. Bus. Dict. 2011, 298, 1–17. [Google Scholar]
- Jabłońska, E.; Winkowska, M.; Wiśniewska, M.; Geurts, J.; Zak, D.; Kotowski, W. Impact of vegetation harvesting on nutrient removal and plant biomass quality in wetland buffer zones. Hydrobiologia 2020, 848, 3273–3289. [Google Scholar] [CrossRef]
- Zak, D.; Kronvang, B.; Carstensen, M.V.; Hoffmann, C.C.; Kjeldgaard, A.; Larsen, S.E.; Audet, J.; Egemose, S.; Jorgensen, C.A.; Feuerbach, P.; et al. Nitrogen and phosphorus removal from agricultural runoff in integrated buffer zones. Environ. Sci. Technol. 2018, 52, 6508–6517. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.A.; Fischenich, J.C. Design Recommendations for Riparian Corridors and Vegetated Buffer Strips; EMRRP Technical Notes Collection (ERDC TN-EMRRP-SR-24); U.S. Army Engineer Research and Development Center: Vicksburg, MS, USA, 2000. [Google Scholar]
- Schultz, R.C.; Wray, P.H.; Colletti, J.P.; Isenhart, T.M.; Rodriguez, C.A. Buffer Strip Design, Establishment, and Maintenance; Agriculture and Environment Extension Publications Agriculture and Natural Resources: Ames, IA, USA, 1997. [Google Scholar]
- Ferrero, F.D.P.; Giorgio Borreani, E.T. Le Fasce Tampone Vegetate Riparie Erbacee Realizzazione e Gestione. Available online: https://www.regione.piemonte.it/web/sites/default/files/media/documenti/2019-01/le_fasce_tampone_vegetate_riparie_erbacee_0.pdf (accessed on 7 July 2021).
- Osborne, L.L. Riparian vegetated buffer strips in water-quality restoration and stream management. Biol. Conserv. 1995, 71, 215. [Google Scholar] [CrossRef]
- Deaver, E.; Moor, M.T.; Cooper, C.M.; Knight, S.S. Efficiency of three aquatic macrophytes in mitigating nutrient run-off. Int. J. Ecol. Environ. Sci. 2005, 31, 1–7. [Google Scholar]
- Hodaj, A.; Bowling, L.C.; Frankenberger, J.R.; Chaubey, I. Impact of a two-stage ditch on channel water quality. Agric. Water Manag. 2017, 192, 126–137. [Google Scholar] [CrossRef]
- Christopher, S.F.; Tank, J.L.; Mahl, U.H.; Yen, H.; Arnold, J.G.; Trentman, M.T.; Sowa, S.P.; Herbert, M.E.; Ross, J.A.; White, M.J.; et al. Modeling nutrient removal using watershed-scale implementation of the two-stage ditch. Ecol. Eng. 2017, 108, 358–369. [Google Scholar] [CrossRef]
- Castaldelli, G.; Soana, E.; Racchetti, E.; Vincenzi, F.; Anna, E.; Bartoli, M. Vegetated canals mitigate nitrogen surplus in agricultural watersheds. Agric. Ecosyst. Environ. 2015, 212, 253–262. [Google Scholar] [CrossRef]
- Soana, E.; Vincenzi, F.; Bartoli, M.; Castaldelli, G. Mitigation of nitrogen pollution in vegetated ditches fed by nitrate-rich spring waters. Agric. Ecosyst. Environ. 2017, 243, 74–82. [Google Scholar] [CrossRef]
- Kröger, R.; Holland, M.M.; Moore, M.T.; Cooper, C.M. Plant senescence: A mechanism for nutrient release in temperate agricultural wetlands. Environ. Pollut. 2007, 146, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Mielke, L.N. Performance of water and sediment control basins in northeastern Nebraska. J. Soil Water Conserv. 1985, 40, 524–528. [Google Scholar]
- United States Department of Agriculture. Natural Resources Conservation Service Conservation Practice Standard—Water and Sediment Control Basin; USDA: Washington, DC, USA, 1991; pp. 1–5. [Google Scholar]
- United States Department of Agriculture. Natural Resources Conservation Service Conservation Practice Standard—Sediment Basin; USDA: Washington, DC, USA, 2017; pp. 1–5. [Google Scholar]
- Sengupta, S.; Nawaz, T.; Beaudry, J. Nitrogen and Phosphorus Recovery from Wastewater. Curr. Pollut. Rep. 2015, 1, 155–166. [Google Scholar] [CrossRef]
- Shilton, A.N.; Powell, N.; Guieysse, B. Plant based phosphorus recovery from wastewater via algae and macrophytes. Curr. Opin. Biotechnol. 2012, 23, 884–889. [Google Scholar] [CrossRef]
- Oron, G. Duckweed culture for wastewater renovation and biomass production. Agric. Water Manag. 1994, 26, 27–40. [Google Scholar] [CrossRef]
- Acién Fernández, F.G.; Gómez-Serrano, C.; Fernández-Sevilla, J.M. Recovery of Nutrients From Wastewaters Using Microalgae. Front. Sustain. Food Syst. 2018, 2, 1–13. [Google Scholar] [CrossRef]
- Mehta, C.M.; Khunjar, W.O.; Nguyen, V.; Tait, S.; Batstone, D.J. Technologies to recover nutrients from waste streams: A critical review. Crit. Rev. Environ. Sci. Technol. 2015, 45, 385–427. [Google Scholar] [CrossRef]
- Emami Moghaddam, S.A.; Harun, R.; Mokhtar, M.N.; Zakaria, R. Potential of Zeolite and Algae in Biomass Immobilization. Biomed. Res. Int. 2018, 2018, 6563196. [Google Scholar] [CrossRef]
- Cai, T.; Park, S.Y.; Li, Y. Nutrient recovery from wastewater streams by microalgae: Status and prospects. Renew. Sustain. Energy Rev. 2013, 19, 360–369. [Google Scholar] [CrossRef]
- Manios, T. The composting potential of different organic solid wastes: Experience from the island of Crete. Environ. Int. 2004, 29, 1079–1089. [Google Scholar] [CrossRef]
- Sultana, M.Y.; Chowdhury, A.K.M.M.B.; Michailides, M.K.; Akratos, C.S.; Tekerlekopoulou, A.G.; Vayenas, D.V. Integrated Cr(VI) removal using constructed wetlands and composting. J. Hazard. Mater. 2015, 281, 106–113. [Google Scholar] [CrossRef]
- Kouki, S.; Saidi, N.; M’hiri, F.; Hafiane, A.; Hassen, A. Co-Composting of Macrophyte Biomass and Sludge as an Alternative for Sustainable Management of Constructed Wetland By-Products. Clean Soil Air Water 2016, 44, 694–702. [Google Scholar] [CrossRef]
- Song, U. Post-remediation use of macrophytes as composting materials for sustainable management of a sanitary landfill. Int. J. Phytoremed. 2017, 19, 395–401. [Google Scholar] [CrossRef]
- Tripathi, M.; Sahu, J.N.; Ganesan, P. Effect of process parameters on production of biochar from biomass waste through pyrolysis: A review. Renew. Sustain. Energy Rev. 2016, 55, 467–481. [Google Scholar] [CrossRef]
- Ghezzehei, T.A.; Sarkhot, D.V.; Berhe, A.A. Biochar can be used to capture essential nutrients from dairy wastewater and improve soil physico-chemical properties. Solid Earth 2014, 5, 953–962. [Google Scholar] [CrossRef]
- Mancuso, G.; Langone, M.; Di Maggio, R.; Toscano, A.; Andreottola, G. Effect of hydrodynamic cavitation on flocs structure in sewage sludge to increase stabilization for efficient and safe reuse in agriculture. Bioremediat. J. 2021, 1–12. [Google Scholar] [CrossRef]
- Mancuso, G.; Langone, M.; Andreottola, G.; Bruni, L. Effects of hydrodynamic cavitation, low-level thermal and low-level alkaline pre-treatments on sludge solubilisation. Ultrason. Sonochem. 2019, 59, 104750. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, G.; Langone, M.; Andreottola, G. A critical review of the current technologies in wastewater treatment plants by using hydrodynamic cavitation process: Principles and applications. J. Environ. Health Sci. Eng. 2020, 18, 311–333. [Google Scholar] [CrossRef]
- Roj-Rojewski, S.; Wysocka-Czubaszek, A.; Czubaszek, R.; Kamocki, A.; Banaszuk, P. Anaerobic digestion of wetland biomass from conservation management for biogas production. Biomass Bioenergy 2019, 122, 126–132. [Google Scholar] [CrossRef]
- Makadi, M.; Tomcsik, A.; Orosz, V. Digestate: A New Nutrient Source—Review. Biogas 2012, 14, 295–312. [Google Scholar]
- Monlau, F.; Sambusiti, C.; Ficara, E.; Aboulkas, A.; Barakat, A.; Carrère, H. New opportunities for agricultural digestate valorization: Current situation and perspectives. Energy Environ. Sci. 2015, 8, 2600–2621. [Google Scholar] [CrossRef]
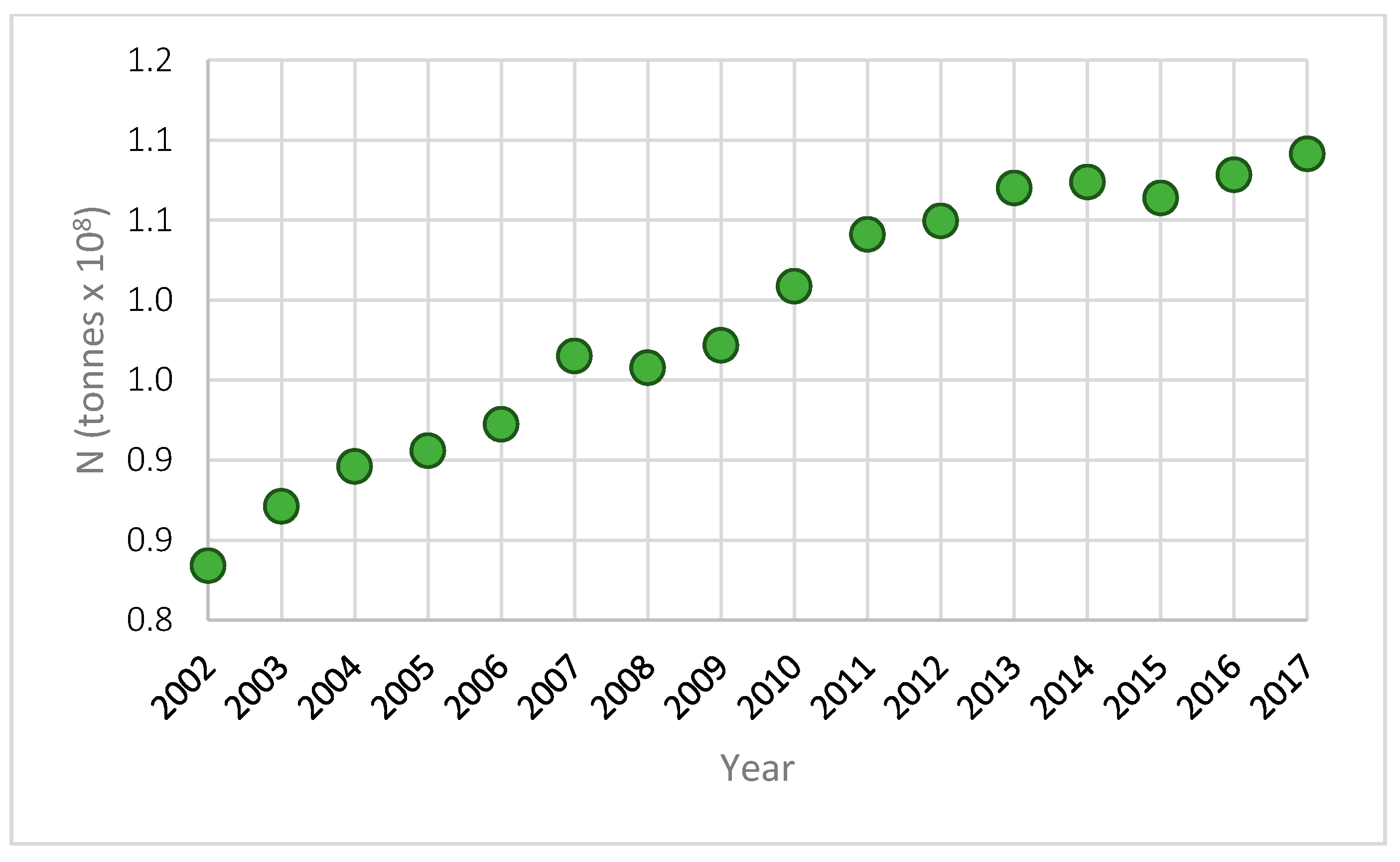

| Influencing Factors | NBS Type | |||
|---|---|---|---|---|
| CWs | Buffer Strips | Vegetated Channels | Water Sediment Control Basins | |
| Vegetation typology | ✓ | ✓ | ✓ | ✓ |
| Substrate materials | ✓ | X | X | X |
| Microbial communities | ✓ | X | ✓ | X |
| Operating conditions | ✓ | ✓ | ✓ | ✓ |
| Total studies (n.) | 87 | 71 | 7 | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mancuso, G.; Bencresciuto, G.F.; Lavrnić, S.; Toscano, A. Diffuse Water Pollution from Agriculture: A Review of Nature-Based Solutions for Nitrogen Removal and Recovery. Water 2021, 13, 1893. https://doi.org/10.3390/w13141893
Mancuso G, Bencresciuto GF, Lavrnić S, Toscano A. Diffuse Water Pollution from Agriculture: A Review of Nature-Based Solutions for Nitrogen Removal and Recovery. Water. 2021; 13(14):1893. https://doi.org/10.3390/w13141893
Chicago/Turabian StyleMancuso, Giuseppe, Grazia Federica Bencresciuto, Stevo Lavrnić, and Attilio Toscano. 2021. "Diffuse Water Pollution from Agriculture: A Review of Nature-Based Solutions for Nitrogen Removal and Recovery" Water 13, no. 14: 1893. https://doi.org/10.3390/w13141893
APA StyleMancuso, G., Bencresciuto, G. F., Lavrnić, S., & Toscano, A. (2021). Diffuse Water Pollution from Agriculture: A Review of Nature-Based Solutions for Nitrogen Removal and Recovery. Water, 13(14), 1893. https://doi.org/10.3390/w13141893









