Nonlinear Isotherm and Kinetic Modeling of Cu(II) and Pb(II) Uptake from Water by MnFe2O4/Chitosan Nanoadsorbents
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Adsorbent Preparation
2.3. Instrumentation and Analytical Methods
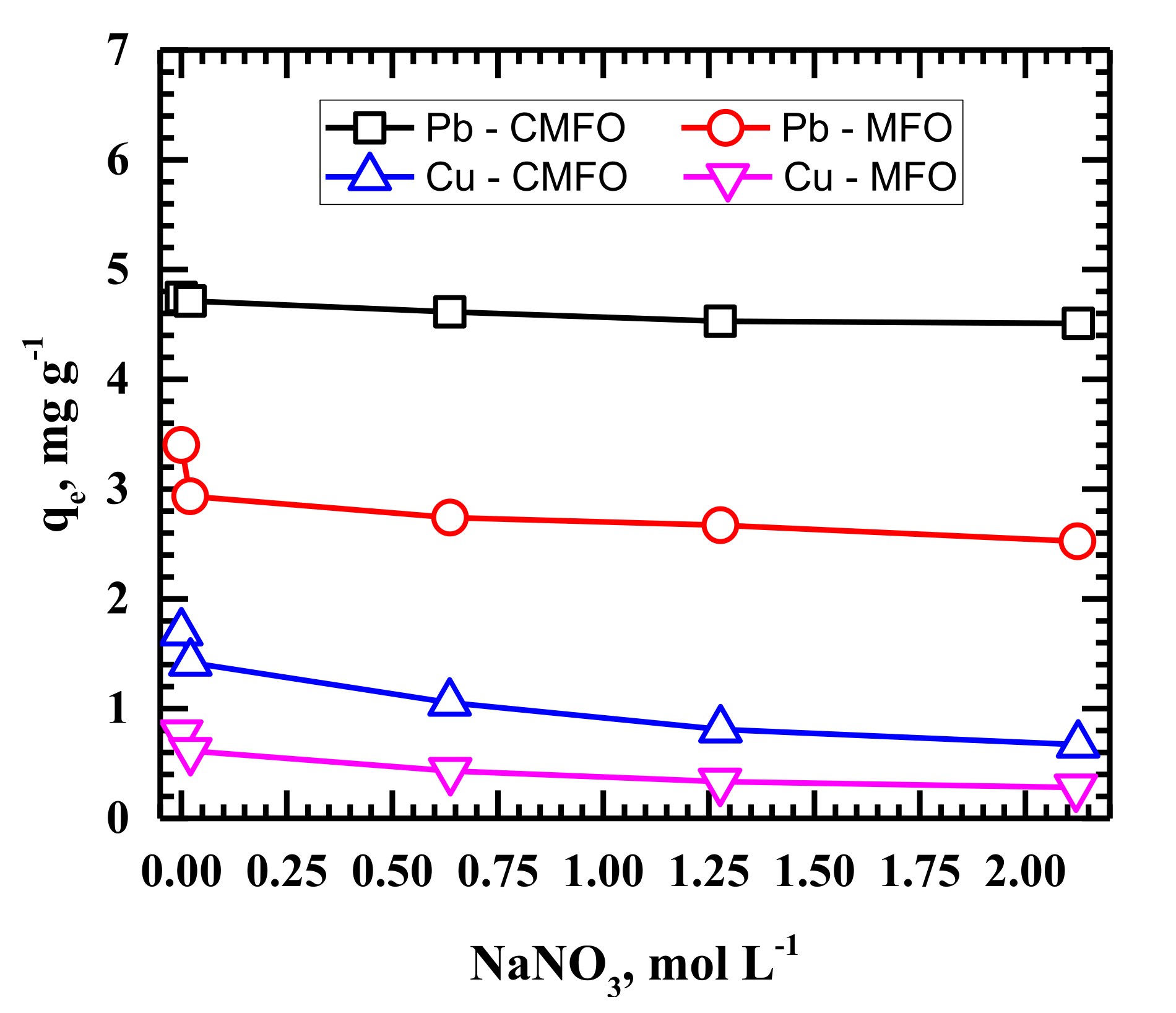
2.4. Adsorption Studies
3. Results and Discussion
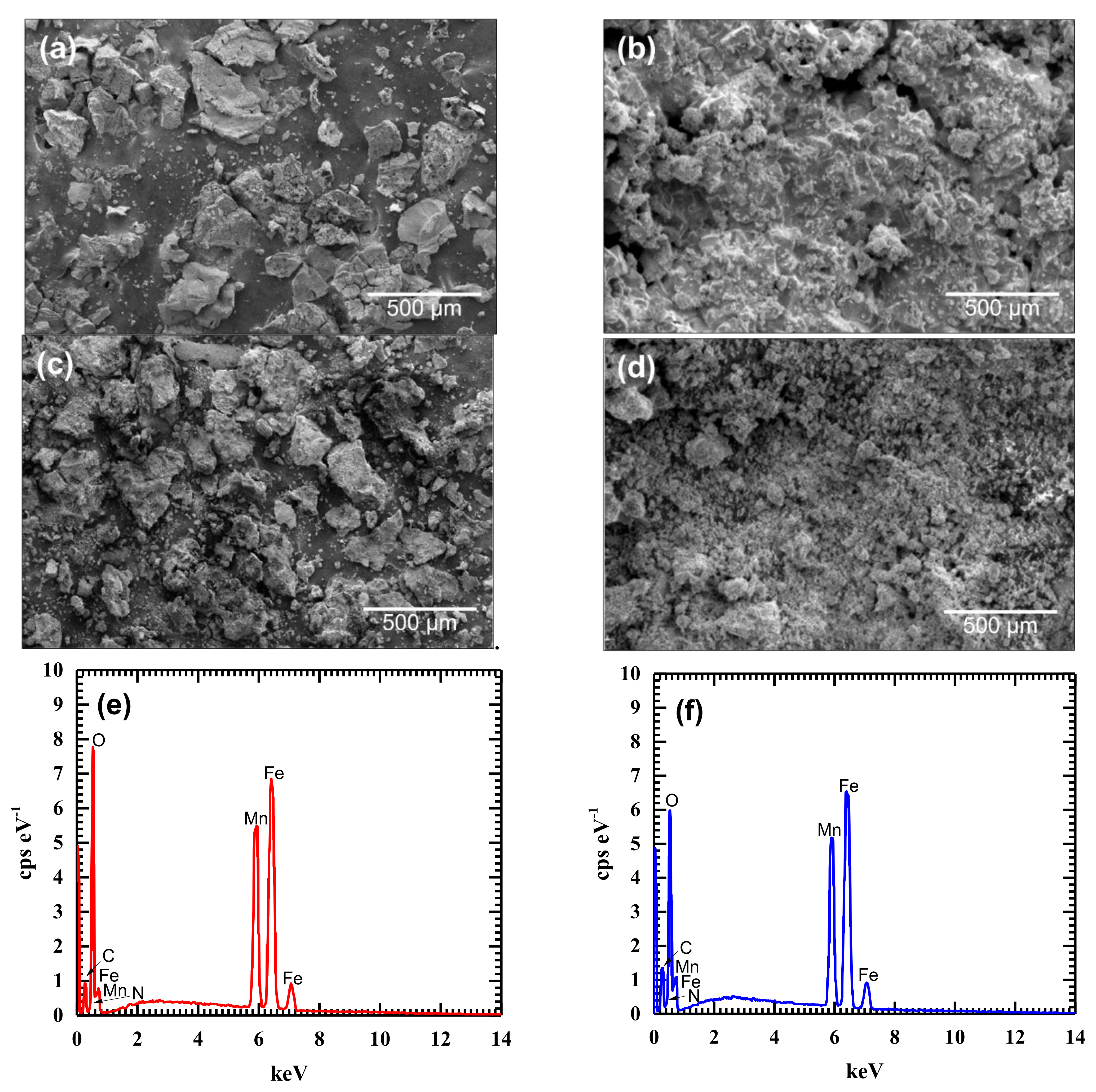
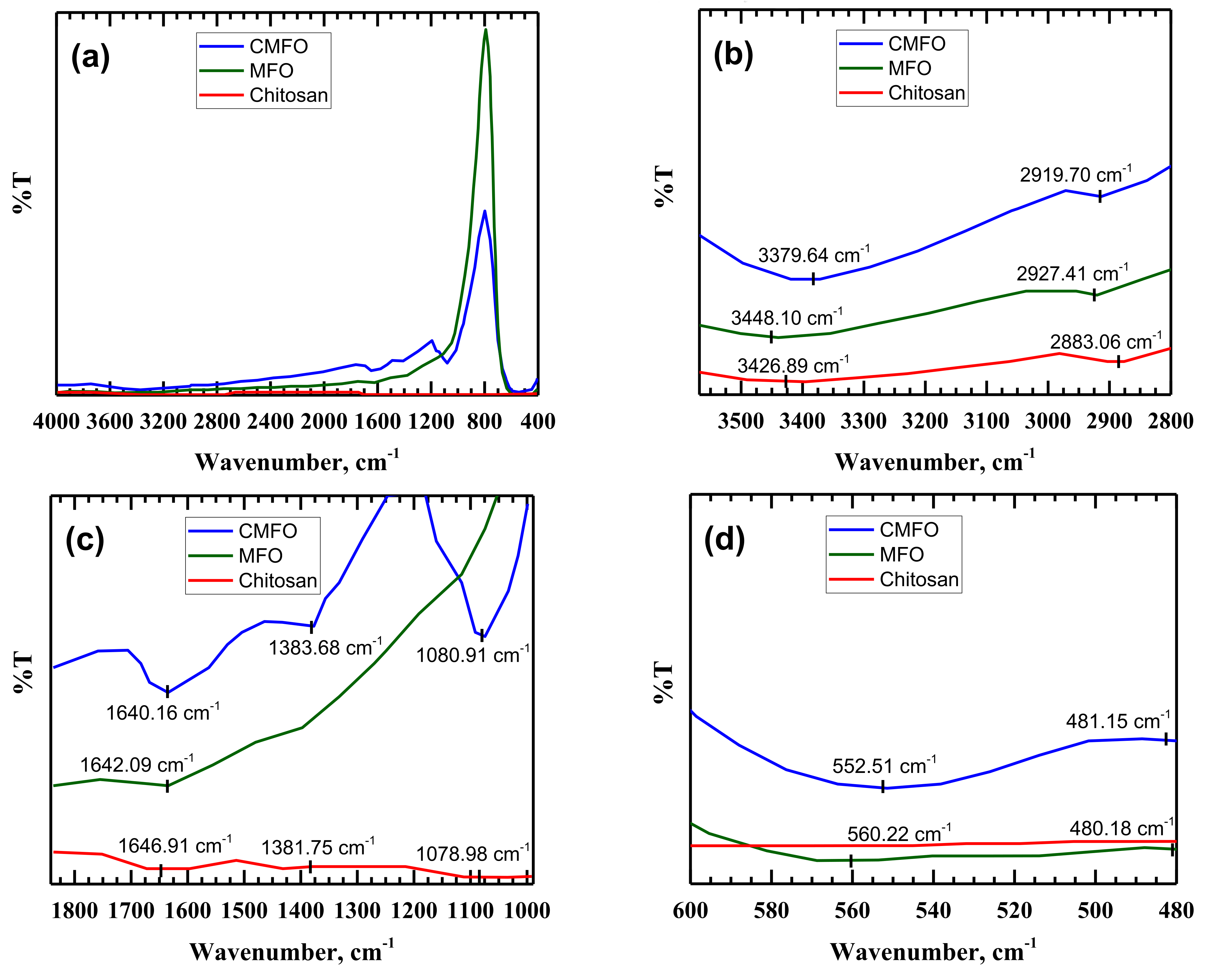
3.1. Adsorbent Characterization
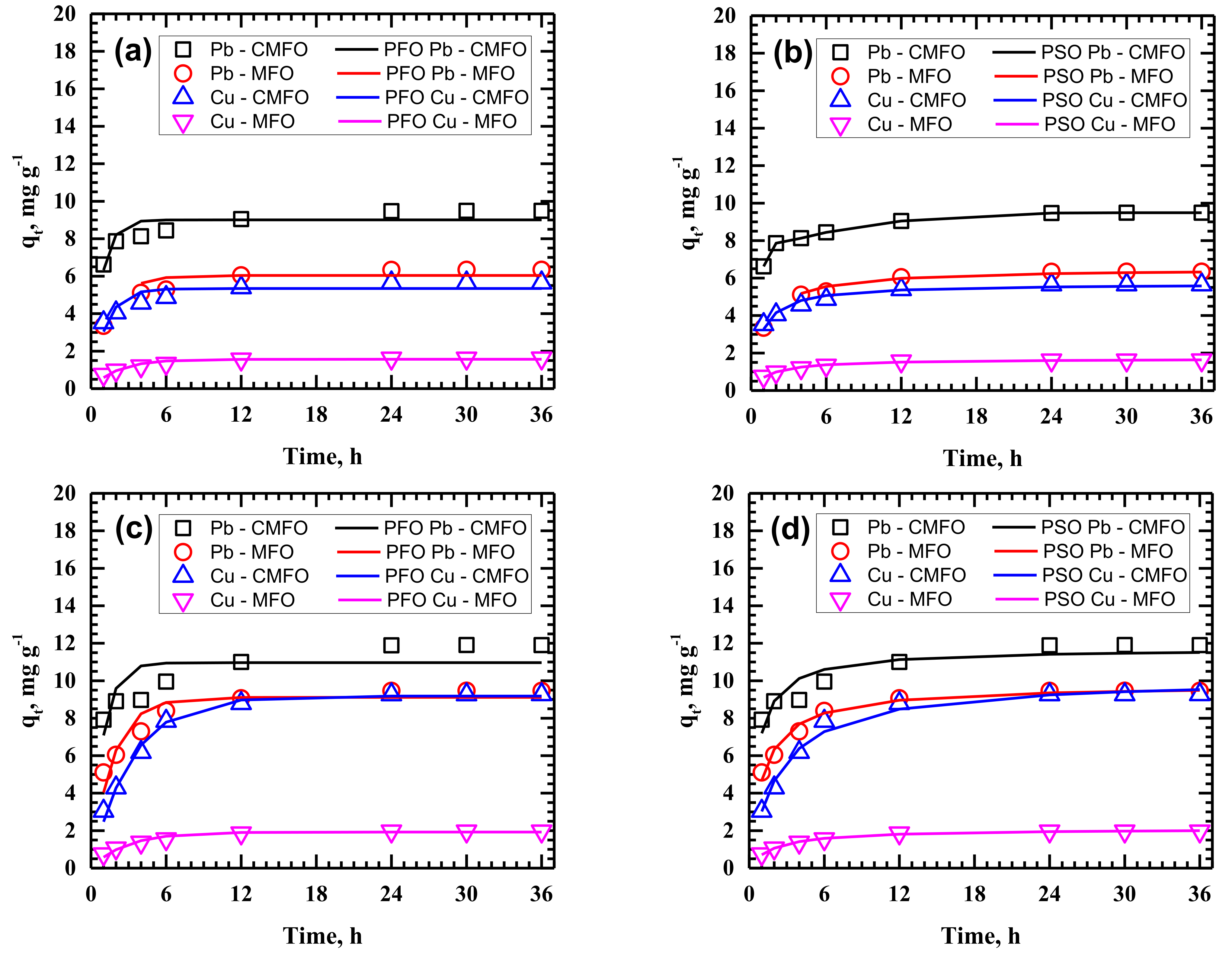
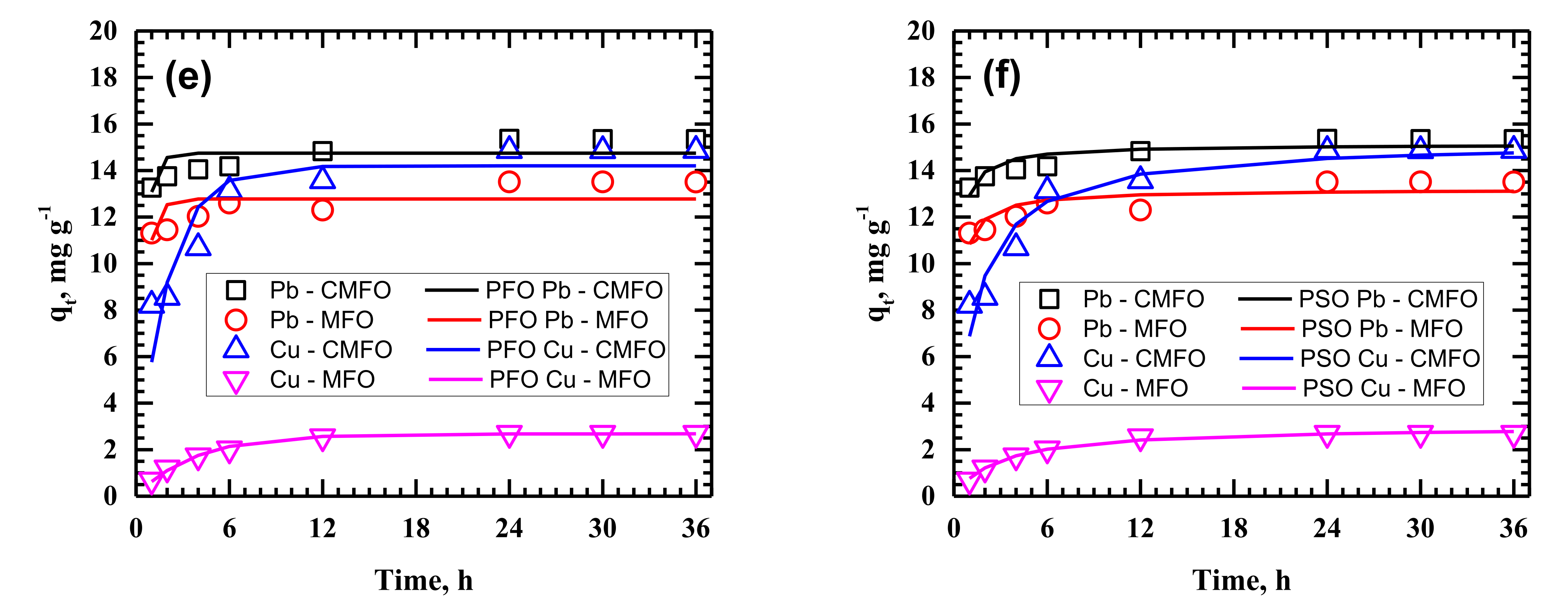
3.2. Adsorption Kinetics
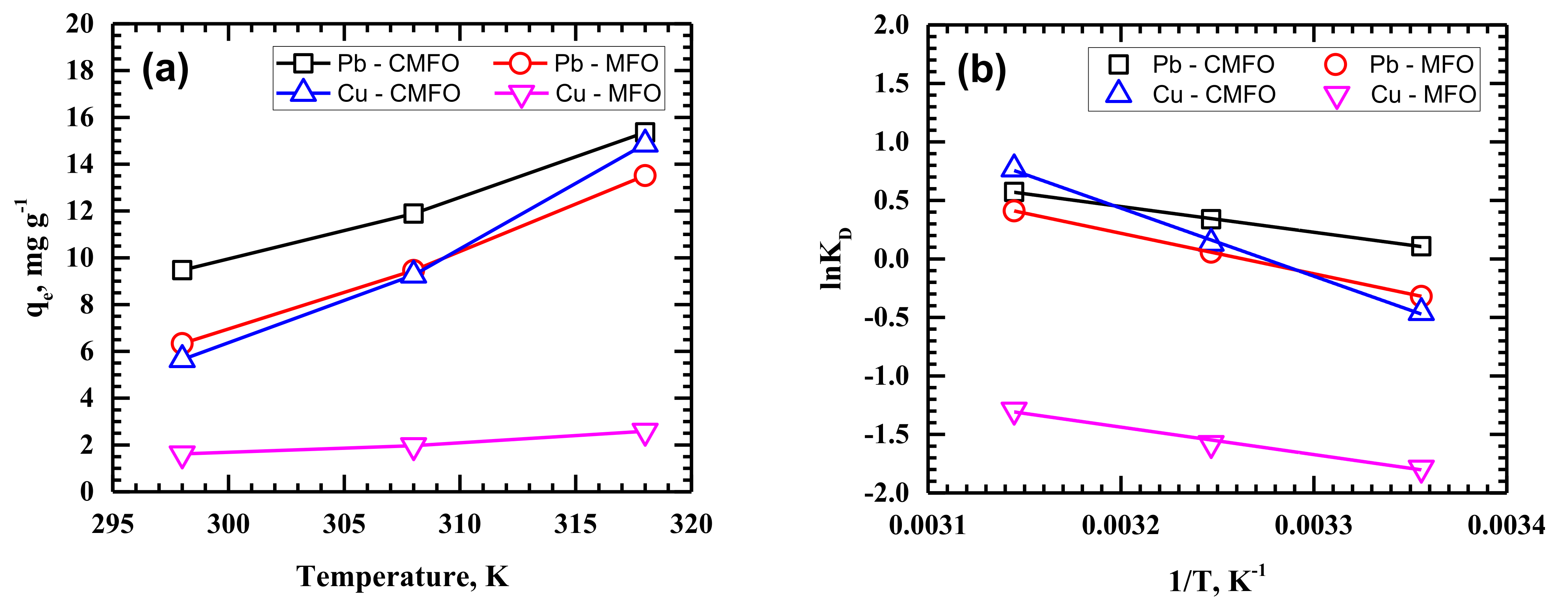
3.3. Adsorption Thermodynamics
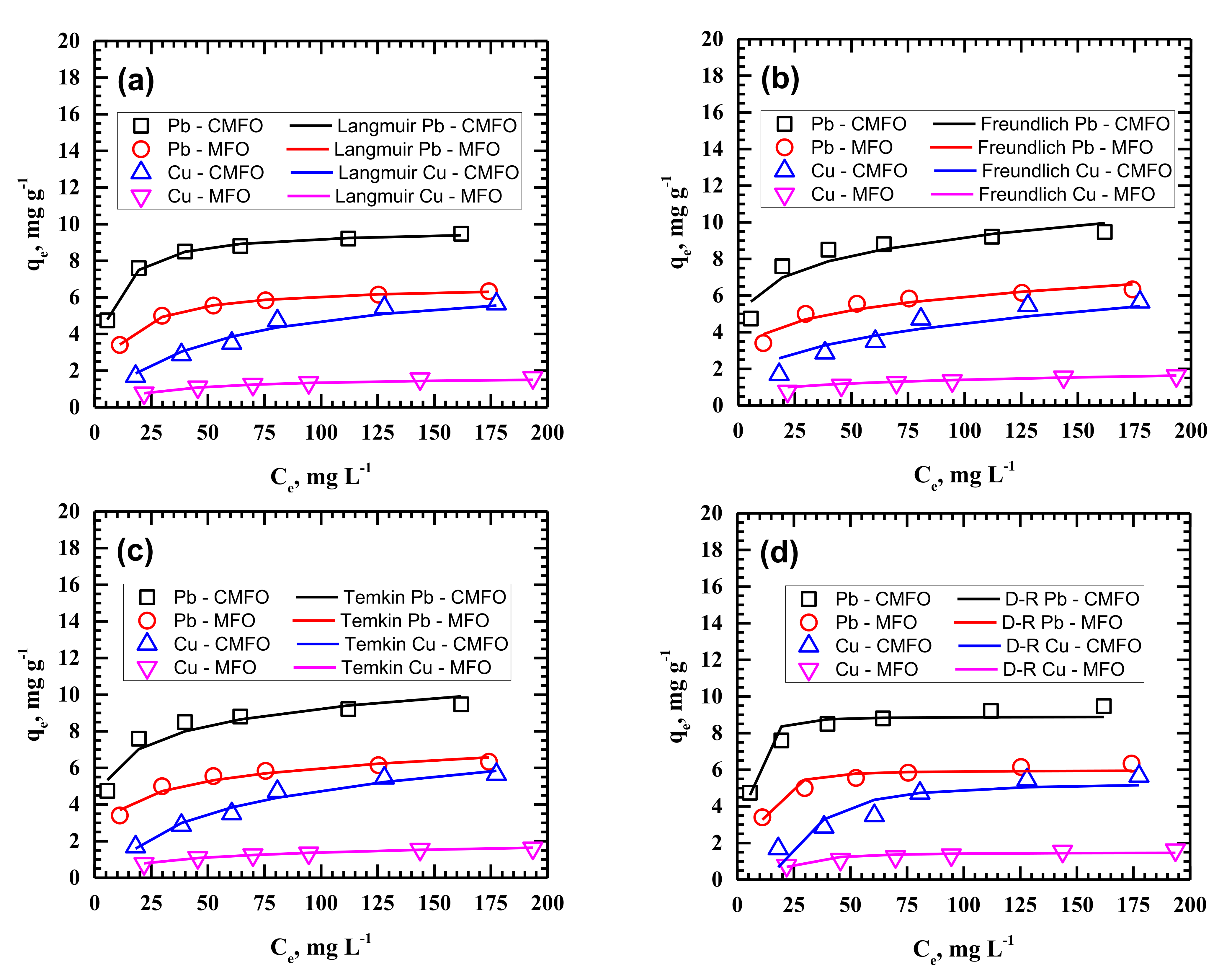
3.4. Adsorption Isotherms
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Viriyatum, R.; Boyd, C.E. Slow-Release Coated Copper Sulfate as an Algicide for Aquaculture. J. World Aquac. Soc. 2016, 47, 667–675. [Google Scholar] [CrossRef]
- Kholghi Eshkalak, S.; Khatibzadeh, M.; Kowsari, E.; Chinnappan, A.; Ramakrishna, S. A Novel Surface Modification of Copper (II) Phthalocyanine with Ionic Liquids as Electronic Ink. Dye. Pigment. 2018, 154, 296–302. [Google Scholar] [CrossRef]
- Isahak, W.N.R.W.; Hasan, S.Z.; Ramli, Z.A.C.; Ba-Abbad, M.M.; Yarmo, M.A. Enhanced Physical and Chemical Adsorption of Carbon Dioxide Using Bimetallic Copper–Magnesium Oxide/Carbon Nanocomposite. Res. Chem. Intermed. 2018, 44, 829–841. [Google Scholar] [CrossRef]
- Rihel, J. Copper on the Brain. Nat. Chem. Biol. 2018, 14, 638–639. [Google Scholar] [CrossRef]
- Kumar, J.; Sathua, K.B.; Flora, S.J.S. Chronic Copper Exposure Elicit Neurotoxic Responses in Rat Brain: Assessment of 8-Hydroxy-2-Deoxyguanosine Activity, Oxidative Stress and Neurobehavioral Parameters. Cell. Mol. Biol. 2019, 65, 27–35. [Google Scholar] [CrossRef]
- Kumar, A.; Cabral-Pinto, M.; Kumar, A.; Kumar, M.; Dinis, P.A. Estimation of Risk to the Eco-Environment and Human Health of Using Heavy Metals in the Uttarakhand Himalaya, India. Appl. Sci. 2020, 10, 7078. [Google Scholar] [CrossRef]
- Marques, D.M.; Veroneze Júnior, V.; da Silva, A.B.; Mantovani, J.R.; Magalhães, P.C.; de Souza, T.C. Copper Toxicity on Photosynthetic Responses and Root Morphology of Hymenaea Courbaril L. (Caesalpinioideae). WaterAirSoil Pollut. 2018, 229, 1–14. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, A.M.M.S.C.-P.; Chaturvedi, A.K.; Shabnam, A.A.; Subrahmanyam, G.; Mondal, R.; Gupta, D.K.; Malyan, S.K.; Kumar, S.S.; Khan, S.A.; et al. Lead Toxicity: Health Hazards, Influence on Food Chain, and Sustainable Remediation Approaches. Int. J. Environ. Res. Public Health 2020, 17, 2179. [Google Scholar] [CrossRef]
- De Luna, M.D.G.; Bellotindos, L.M.; Asiao, R.N.; Lu, M.C. Removal and Recovery of Lead in a Fluidized-Bed Reactor by Crystallization Process. Hydrometallurgy 2015, 155, 6–12. [Google Scholar] [CrossRef]
- García-Lestón Julia, J.; Méndez, J.; Pásaro, E.; Laffon, B. Genotoxic Effects of Lead: An Updated Review. Environ. Int. 2010, 36, 623–636. [Google Scholar] [CrossRef]
- Liao, L.M.; Friesen, M.C.; Xiang, Y.-B.; Cai, H.; Koh, D.-H.; Ji, B.-T.; Yang, G.; Li, H.-L.; Locke, S.J.; Rothman, N.; et al. Occupational Lead Exposure and Associations with Selected Cancers: The Shanghai Men’s and Women’s Health Study Cohorts. Environ. Health Perspect. 2016, 124, 97–103. [Google Scholar] [CrossRef]
- Mitra, P.; Sharma, S.; Purohit, P.; Sharma, P. Clinical and Molecular Aspects of Lead Toxicity: An Update. Crit. Rev. Clin. Lab. Sci. 2017, 54, 506–528. [Google Scholar] [CrossRef]
- Harvey, P.J.; Handley, H.K.; Taylor, M.P. Widespread Copper and Lead Contamination of Household Drinking Water, New South Wales, Australia. Environ. Res. 2016, 151, 275–285. [Google Scholar] [CrossRef]
- Abedi Sarvestani, R.; Aghasi, M. Health Risk Assessment of Heavy Metals Exposure (Lead, Cadmium, and Copper) through Drinking Water Consumption in Kerman City, Iran. Environ. Earth Sci. 2019, 78, 1–11. [Google Scholar] [CrossRef]
- Ong, D.C.; de Luna, M.D.G.; Pingul-Ong, S.M.B.; Kan, C.C. Manganese and Iron Recovery from Groundwater Treatment Sludge by Reductive Acid Leaching and Hydroxide Precipitation. J. Environ. Manag. 2018, 223, 723–730. [Google Scholar] [CrossRef]
- De Luna, M.D.G.; Flores, E.D.; Cenia, M.C.B.; Lu, M.-C. Removal of Copper Ions from Aqueous Solution by Adlai Shell (Coix Lacryma-Jobi L.) Adsorbents. Bioresour. Technol. 2015, 192, 841–844. [Google Scholar] [CrossRef]
- Tsai, W.C.; De Luna, M.D.G.; Bermillo-Arriesgado, H.L.P.; Futalan, C.M.; Colades, J.I.; Wan, M.W. Competitive Fixed-Bed Adsorption of Pb(II), Cu(II), and Ni(II) from Aqueous Solution Using Chitosan-Coated Bentonite. Int. J. Polym. Sci. 2016, 2016. [Google Scholar] [CrossRef]
- De Castro, M.L.F.A.; Abad, M.L.B.; Sumalinog, D.A.G.; Abarca, R.R.M.; Paoprasert, P.; de Luna, M.D.G. Adsorption of Methylene Blue Dye and Cu(II) Ions on EDTA-Modified Bentonite: Isotherm, Kinetic and Thermodynamic Studies. Sustain. Environ. Res. 2018, 28, 197–205. [Google Scholar] [CrossRef]
- Tumampos, S.B.; Ensano, B.M.B.; Pingul-Ong, S.M.B.; Ong, D.C.; Kan, C.-C.; Yee, J.-J.; de Luna, M.D.G. Isotherm, Kinetics and Thermodynamics of Cu(II) and Pb(II) Adsorption on Groundwater Treatment Sludge-Derived Manganese Dioxide for Wastewater Treatment Applications. Int. J. Environ. Res. Public Health 2021, 18, 3050. [Google Scholar] [CrossRef]
- Yang, L.X.; Wang, F.; Meng, Y.F.; Tang, Q.H.; Liu, Z.Q. Fabrication and Characterization of Manganese Ferrite Nanospheres as a Magnetic Adsorbent of Chromium. J. Nanomater. 2013, 2013. [Google Scholar] [CrossRef]
- Aryee, A.A.; Mpatani, F.M.; Du, Y.; Kani, A.N.; Dovi, E.; Han, R.; Li, Z.; Qu, L. Fe3O4 and Iminodiacetic Acid Modified Peanut Husk as a Novel Adsorbent for the Uptake of Cu (II) and Pb (II) in Aqueous Solution: Characterization, Equilibrium and Kinetic Study. Environ. Pollut. 2021, 268, 115729. [Google Scholar] [CrossRef]
- Behbahani, E.S.; Dashtian, K.; Ghaedi, M. Fe3O4-FeMoS4: Promise Magnetite LDH-Based Adsorbent for Simultaneous Removal of Pb (II), Cd (II), and Cu (II) Heavy Metal Ions. J. Hazard. Mater. 2021, 410, 124560. [Google Scholar] [CrossRef] [PubMed]
- Tran, C.V.; Quang, D.V.; Nguyen Thi, H.P.; Truong, T.N.; La, D.D. Effective Removal of Pb(II) from Aqueous Media by a New Design of Cu–Mg Binary Ferrite. Acs Omega 2020, 5, 7298–7306. [Google Scholar] [CrossRef]
- Hou, X.; Feng, J.; Xu, X.; Zhang, M. Synthesis and Characterizations of Spinel MnFe2O4 Nanorod by Seed-Hydrothermal Route. J. Alloys Compd. 2010, 491, 258–263. [Google Scholar] [CrossRef]
- Kafshgari, L.A.; Ghorbani, M.; Azizi, A. Synthesis and Characterization of Manganese Ferrite Nanostructure by Co-Precipitation, Sol-Gel, and Hydrothermal Methods. Part. Sci. Technol. 2019, 37, 900–906. [Google Scholar] [CrossRef]
- Kan, C.C.; Ibe, A.H.; Rivera, K.K.P.; Arazo, R.O.; de Luna, M.D.G. Hexavalent Chromium Removal from Aqueous Solution by Adsorbents Synthesized from Groundwater Treatment Residuals. Sustain. Environ. Res. 2017, 27, 163–171. [Google Scholar] [CrossRef]
- Ong, D.C.; Pingul-Ong, S.M.B.; Kan, C.C.; de Luna, M.D.G. Removal of Nickel Ions from Aqueous Solutions by Manganese Dioxide Derived from Groundwater Treatment Sludge. J. Clean. Prod. 2018, 190, 443–451. [Google Scholar] [CrossRef]
- Reddy, D.H.K.; Yun, Y.S. Spinel Ferrite Magnetic Adsorbents: Alternative Future Materials for Water Purification? Coord. Chem. Rev. 2016, 315, 90–111. [Google Scholar] [CrossRef]
- Xiao, Y.; Liang, H.; Wang, Z. MnFe2O4/Chitosan Nanocomposites as a Recyclable Adsorbent for the Removal of Hexavalent Chromium. Mater. Res. Bull. 2013, 48, 3910–3915. [Google Scholar] [CrossRef]
- Habiba, U.; Joo, T.C.; Siddique, T.A.; Salleh, A.; Ang, B.C.; Afifi, A.M. Effect of Degree of Deacetylation of Chitosan on Adsorption Capacity and Reusability of Chitosan/Polyvinyl Alcohol/TiO2 Nano Composite. Int. J. Biol. Macromol. 2017, 104, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Liang, H.; Chen, W.; Wang, Z. Synthesis and Adsorption Behavior of Chitosan-Coated MnFe2O 4 Nanoparticles for Trace Heavy Metal Ions Removal. Appl. Surf. Sci. 2013, 285, 498–504. [Google Scholar] [CrossRef]
- Zeiger, E.; Gollapudi, B.; Spencer, P. Genetic Toxicity and Carcinogenicity Studies of Glutaraldehyde—A Review. Mutat. Res. Rev. Mutat. Res. 2005, 589, 136–151. [Google Scholar] [CrossRef]
- Takigawa, T.; Endo, Y. Effects of Glutaraldehyde Exposure on Human Health. J. Occup. Health 2006, 48, 75–87. [Google Scholar] [CrossRef]
- Sam, S.; Nesaraj, S. Preparation of MnFe 2 O 4 Nanoceramic Particles by Soft Chemical Routes. Int. J. 2011, 9, 223–239. [Google Scholar]
- Sessarego, S.; Rodrigues, S.C.G.; Xiao, Y.; Lu, Q.; Hill, J.M. Phosphonium-Enhanced Chitosan for Cr(VI) Adsorption in Wastewater Treatment. Carbohydr. Polym. 2019, 211, 249–256. [Google Scholar] [CrossRef]
- Mende, M.; Schwarz, D.; Steinbach, C.; Boldt, R.; Schwarz, S. Simultaneous Adsorption of Heavy Metal Ions and Anions from Aqueous Solutions on Chitosan—Investigated by Spectrophotometry and SEM-EDX Analysis. Colloids Surf. A Physicochem. Eng. Asp. 2016, 510, 275–282. [Google Scholar] [CrossRef]
- Ahamad, T.; Naushad, M.; Al-Shahrani, T.; Al-hokbany, N.; Alshehri, S.M. Preparation of Chitosan Based Magnetic Nanocomposite for Tetracycline Adsorption: Kinetic and Thermodynamic Studies. Int. J. Biol. Macromol. 2020, 147, 258–267. [Google Scholar] [CrossRef]
- Wang, G.; Zhao, D.; Ma, Y.; Zhang, Z.; Che, H.; Mu, J.; Zhang, X.; Zhang, Z. Synthesis and Characterization of Polymer-Coated Manganese Ferrite Nanoparticles as Controlled Drug Delivery. Appl. Surf. Sci. 2018, 428, 258–263. [Google Scholar] [CrossRef]
- Futalan, C.M.; Kan, C.C.; Dalida, M.L.; Hsien, K.J.; Pascua, C.; Wan, M.W. Comparative and Competitive Adsorption of Copper, Lead, and Nickel Using Chitosan Immobilized on Bentonite. Carbohydr. Polym. 2011, 83, 528–536. [Google Scholar] [CrossRef]
- Giraldo, L.; Erto, A.; Moreno-Piraján, J.C. Magnetite Nanoparticles for Removal of Heavy Metals from Aqueous Solutions: Synthesis and Characterization. Adsorption 2013, 19, 465–474. [Google Scholar] [CrossRef]
- Lagergren, S.Y. Zur Theorie Der Sogenannten Adsorption Gelöster Stoffe. K. Sven. Vetensk. Handl. 1898, 24, 1–39. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. A Comparison of Chemisorption Kinetic Models Applied to Pollutant Removal on Various Sorbents. Process Saf. Environ. Prot. 1998, 76, 332–340. [Google Scholar] [CrossRef]
- Blanchard, G.; Maunaye, M.; Martin, G. Removal of Heavy Metals from Waters by Means of Natural Zeolites. Water Res. 1984, 18, 1501–1507. [Google Scholar] [CrossRef]
- Weber, W.J.; Morris, J.C. Kinetics of Adsorption on Carbon from Solution. J. Sanit. Eng. Div. 1963, 89, 31–59. [Google Scholar] [CrossRef]
- Ren, Y.; Li, N.; Feng, J.; Luan, T.; Wen, Q.; Li, Z.; Zhang, M. Adsorption of Pb(II) and Cu(II) from Aqueous Solution on Magnetic Porous Ferrospinel MnFe2O4. J. Colloid Interface Sci. 2012, 367, 415–421. [Google Scholar] [CrossRef]
- Kumar, A.; Yadav, K.L. Synthesis and Characterization of MnFe2O4–BiFeO3 Multiferroic Composites. Phys. B Condens. Matter 2011, 406, 1763–1766. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Chao, H.-P. Thermodynamic Parameters of Cadmium Adsorption onto Orange Peel Calculated from Various Methods: A Comparison Study. J. Environ. Chem. Eng. 2016, 4, 2671–2682. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Hosseini-Bandegharaei, A.; Chao, H.-P. Mistakes and Inconsistencies Regarding Adsorption of Contaminants from Aqueous Solutions: A Critical Review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef] [PubMed]
- Chella, S.; Kollu, P.; Komarala, E.V.P.R.; Doshi, S.; Saranya, M.; Felix, S.; Ramachandran, R.; Saravanan, P.; Koneru, V.L.; Venugopal, V.; et al. Solvothermal Synthesis of MnFe2O4-Graphene Composite-Investigation of Its Adsorption and Antimicrobial Properties. Appl. Surf. Sci. 2015, 327, 27–36. [Google Scholar] [CrossRef]
- Langmuir, I. The Adsorption of Gases on Plane Surfaces of Glass, Mica and Platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Über Die Adsorption in Lösungen. Z. Phys. Chem. 1906, 57, 385–470. [Google Scholar] [CrossRef]
- Temkin, M.J.; Pyzhev, V. Recent Modifications to Langmuir Isotherms. Acta Physicochim. URSS 1940, 12, 217–222. [Google Scholar]
- Dubinin, M.; Radushkevich, L. The Equation of the Characteristic Curve Ofactivated Charcoal. Proc. Acad. Sci. USSR 1947, 55, 331–333. [Google Scholar]
- Meng, Y.; Chen, D.; Sun, Y.; Jiao, D.; Zeng, D.; Liu, Z. Adsorption of Cu2+ Ions Using Chitosan-Modified Magnetic Mn Ferrite Nanoparticles Synthesized by Microwave-Assisted Hydrothermal Method. Appl. Surf. Sci. 2015, 324, 745–750. [Google Scholar] [CrossRef]
- Sezgin, N. Removal of Copper From Industrial Wastewater Using Manganese Ferrite Nanoparticles: Evaluation Of Equilibrium And Kinetic Model. Env. Eng. Manag. J. 2018, 17, 2147–2156. [Google Scholar] [CrossRef]
- Wang, Y.; Cheng, R.; Wen, Z.; Zhao, L. Synthesis and Characterization of Single-Crystalline MnFe2O 4 Ferrite Nanocrystals and Their Possible Application in Water Treatment. Eur. J. Inorg. Chem. 2011, 2942–2947. [Google Scholar] [CrossRef]
- Bateer, B.; Tian, C.; Qu, Y.; Du, S.; Yang, Y.; Ren, Z.; Pan, K.; Fu, H. Synthesis, Size and Magnetic Properties of Controllable MnFe2O4 Nanoparticles with Versatile Surface Functionalities. Dalton Trans. 2014, 43, 9885–9891. [Google Scholar] [CrossRef]
- Jung, K.-W.; Lee, S.Y.; Lee, Y.J. Facile One-Pot Hydrothermal Synthesis of Cubic Spinel-Type Manganese Ferrite/Biochar Composites for Environmental Remediation of Heavy Metals from Aqueous Solutions. Bioresour. Technol. 2018, 261, 1–9. [Google Scholar] [CrossRef]
- Israelachvili, J.N. Interactions Involving Polar Molecules. In Intermolecular and Surface Forces; Elsevier: Amsterdam, The Netherlands, 2011; pp. 71–90. [Google Scholar]
- Kara, İ.; Yilmazer, D.; Akar, S.T. Metakaolin Based Geopolymer as an Effective Adsorbent for Adsorption of Zinc(II) and Nickel(II) Ions from Aqueous Solutions. Appl. Clay Sci. 2017, 139, 54–63. [Google Scholar] [CrossRef]
- Kumar, S.; Nair, R.R.; Pillai, P.B.; Gupta, S.N.; Iyengar, M.A.R.; Sood, A.K. Graphene Oxide–MnFe2O4 Magnetic Nanohybrids for Efficient Removal of Lead and Arsenic from Water. Acs Appl. Mater. Interfaces 2014, 6, 17426–17436. [Google Scholar] [CrossRef]
- Shirkhodaie, M.; Beyki, M.H.; Shemirani, F. Simple Route Synthesis of MnFe2O4 @ Alunite Composite for Preconcentration of Trace Level of Copper and Lead from Food and Water Samples. Desalination Water Treat. 2016, 57, 22480–22492. [Google Scholar] [CrossRef]
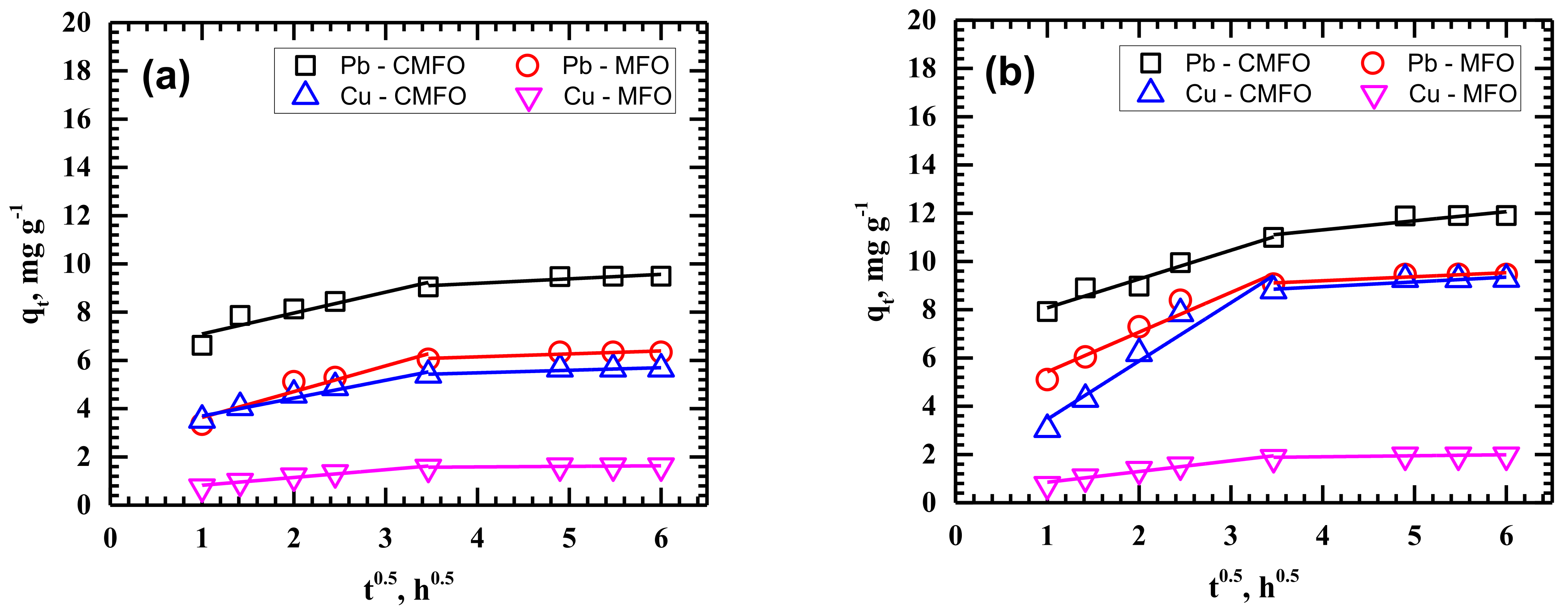
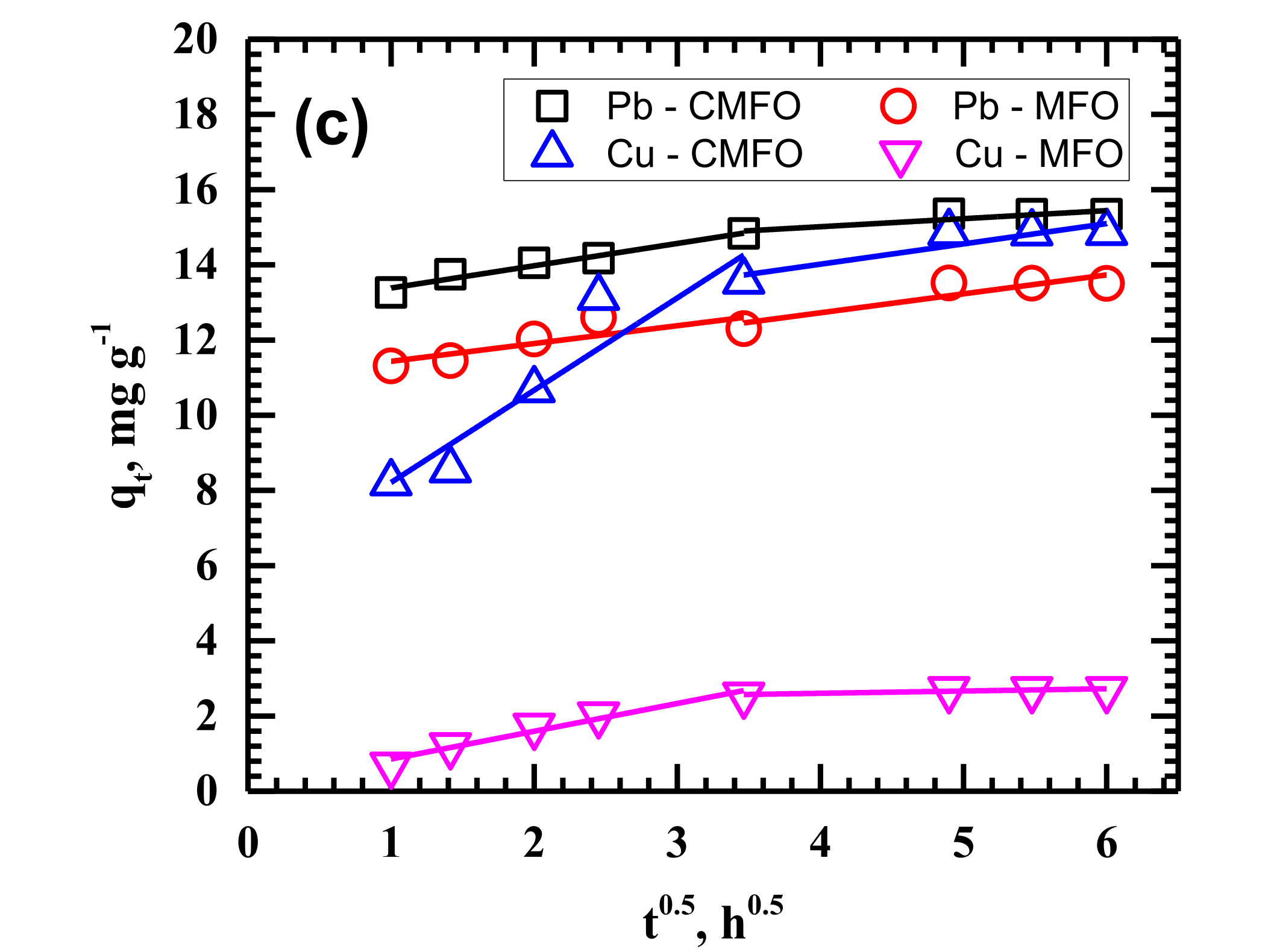
| Temperature | 25 °C | 35 °C | 45 °C | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adsorbent | CMFO | MFO | CMFO | MFO | CMFO | MFO | |||||||
| Metal | Cu | Pb | Cu | Pb | Cu | Pb | Cu | Pb | Cu | Pb | Cu | Pb | |
| qe exp | 5.649 | 9.472 | 1.622 | 6.337 | 9.263 | 11.887 | 1.971 | 9.46 | 14.861 | 15.358 | 2.587 | 13.515 | |
| PFO | k1 | 0.014 | 0.0202 | 0.0078 | 0.0111 | 0.0052 | 0.0173 | 0.006 | 0.0097 | 0.0087 | 0.0363 | 0.0044 | 0.0329 |
| R2 | 0.7529 | 0.7439 | 0.9126 | 0.8465 | 0.9877 | 0.5419 | 0.9537 | 0.8696 | 0.7959 | 0.4447 | 0.9931 | 0.3693 | |
| qe | 5.345 | 9.007 | 1.571 | 6.04 | 9.185 | 10.964 | 1.925 | 9.117 | 14.2 | 14.747 | 2.681 | 12.779 | |
| PSO | k2 | 0.004 | 0.0039 | 0.0069 | 0.0025 | 0.0007 | 0.0023 | 0.0041 | 0.0016 | 0.0009 | 0.0065 | 0.0019 | 0.0058 |
| R2 | 0.9571 | 0.9465 | 0.9892 | 0.9847 | 0.9842 | 0.8382 | 0.9943 | 0.9779 | 0.9258 | 0.7982 | 0.9921 | 0.7212 | |
| qe | 5.697 | 9.459 | 1.698 | 6.501 | 10.155 | 11.71 | 2.107 | 9.774 | 15.256 | 15.126 | 3.006 | 13.192 | |
| ID | kip1 | 0.7416 | 0.8672 | 0.3234 | 1.0690 | 2.4165 | 1.1922 | 0.4447 | 1.6458 | 2.4546 | 0.5926 | 0.7394 | 0.4738 |
| Ci1 | 2.9530 | 6.2287 | 0.5009 | 2.5678 | 1.0413 | 6.8871 | 0.4048 | 3.7799 | 5.7577 | 12.7914 | 0.1162 | 10.9599 | |
| R2 | 0.9639 | 0.8562 | 0.9627 | 0.9208 | 0.9358 | 0.9555 | 0.9642 | 0.9372 | 0.8872 | 0.9712 | 0.9644 | 0.6839 | |
| kip2 | 0.1064 | 0.1852 | 0.0232 | 0.1222 | 0.1970 | 0.3770 | 0.0418 | 0.1673 | 0.5397 | 0.2140 | 0.0608 | 0.5037 | |
| Ci2 | 5.0592 | 8.4550 | 1.4921 | 5.6587 | 8.1680 | 9.8019 | 1.7383 | 8.5318 | 11.8592 | 14.1589 | 2.3615 | 10.7103 | |
| R2 | 0.8340 | 0.8566 | 0.8325 | 0.8338 | 0.8342 | 0.8401 | 0.8274 | 0.8345 | 0.8327 | 0.8159 | 0.8284 | 0.8273 | |
| Adsorbate | Temperature (°C) | MFO | CMFO | ||||||
|---|---|---|---|---|---|---|---|---|---|
| ΔG° (kJ mol−1) | ΔH° (kJ mol−1) | ΔS° (kJ mol−1 K−1) | R2 | ΔG° (kJ mol−1) | ΔH° (kJ mol−1) | ΔS° (kJ mol−1 K−1) | R2 | ||
| Pb | 25 | −16.32 | 28.75 | 0.151 | 0.9999 | −17.38 | 18.28 | 0.120 | 0.9995 |
| 35 | −17.84 | −18.57 | |||||||
| 45 | −19.35 | −19.77 | |||||||
| Cu | 25 | −12.65 | 19.49 | 0.108 | 0.9888 | −15.95 | 48.33 | 0.216 | 0.9985 |
| 35 | −13.73 | −18.10 | |||||||
| 45 | −14.81 | −20.26 | |||||||
| Adsorption Isotherm | Parameters | CMFO | MFO | ||
|---|---|---|---|---|---|
| Cu | Pb | Cu | Pb | ||
| Langmuir | qmax | 8.0970 | 9.7250 | 1.8720 | 6.6911 |
| KL | 0.0146 | 0.1726 | 0.0307 | 0.0945 | |
| RL | 0.25–0.73 | 0.03–0.19 | 0.14–0.57 | 0.05–0.30 | |
| R2 | 0.9774 | 0.9977 | 0.9912 | 0.9988 | |
| Freundlich | n | 2.0965 | 5.9625 | 3.1984 | 5.1734 |
| KF | 0.5120 | 4.2435 | 0.3222 | 2.4413 | |
| R2 | 0.9436 | 0.8718 | 0.9773 | 0.9066 | |
| Temkin | ΒT | 1.8575 | 1.3615 | 0.3897 | 1.0450 |
| AT | 0.1307 | 8.9403 | 0.3496 | 3.1112 | |
| R2 | 0.9717 | 0.9231 | 0.9958 | 0.9488 | |
| D-R | qD | 5.1952 | 8.8887 | 1.4775 | 5.9577 |
| β | 1.02 × 10−4 | 3.98 × 10−6 | 6.10 × 10−5 | 1.31 × 10−5 | |
| R2 | 0.8081 | 0.9258 | 0.8370 | 0.9161 | |
| Metal Ions | Magnetic Nanosorbents | Experimental Conditions * | qe (mg g−1) | Reference |
|---|---|---|---|---|
| Cu(II) | MnFe2O4 | m = 0.1 g L−1; pH = 4.5; T = 45 °C; t = 24 h | 2.59 | This study |
| Chitosan-coated MnFe2O4 | m = 0.1 g L−1; pH = 4.5; T = 45 °C; t = 24 h | 14.86 | This study | |
| Chitosan-coated MnFe2O4 | m = 0.03 g L−1; pH = 6; T = 20 °C; t = 6 h | 22.6 | [31] | |
| MnFe2O4 | m = 1 g L−1; pH = 6; T = 25 ± 2 °C; t = 3 h | 60.5 | [45] | |
| Chitosan-coated MnFe2O4 | m = 3.33 g L−1; pH = 6.5; T = 20 °C; t = 8.33 h | 65.1 | [54] | |
| MnFe2O4 | m = 2 g L−1; pH = 5; t = 2 h | 43.02 | [55] | |
| Pb(II) | MnFe2O4 | m = 0.1 g L−1; pH = 4.5; T = 45 °C; t = 24 h | 13.52 | This study |
| Chitosan-coated MnFe2O4 | m = 0.1 g L−1; pH = 4.5; T = 45 °C; t = 24 h | 15.36 | This study | |
| MnFe2O4 | m = 0.8 g L−1; pH = N.S.; T = 20 °C; t = 1 h | 8.3 | [56] | |
| MnFe2O4 | m = 1 g L−1; pH = 6.8 ± 0.4; T = N.S.; t = 24 h | 5.2 | [57] | |
| MnFe2O4 | m = 1 g L−1; pH = 6; T = 25 ± 2 °C; t = 3 h | 69.1 | [45] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taguba, M.A.M.; Ong, D.C.; Ensano, B.M.B.; Kan, C.-C.; Grisdanurak, N.; Yee, J.-J.; de Luna, M.D.G. Nonlinear Isotherm and Kinetic Modeling of Cu(II) and Pb(II) Uptake from Water by MnFe2O4/Chitosan Nanoadsorbents. Water 2021, 13, 1662. https://doi.org/10.3390/w13121662
Taguba MAM, Ong DC, Ensano BMB, Kan C-C, Grisdanurak N, Yee J-J, de Luna MDG. Nonlinear Isotherm and Kinetic Modeling of Cu(II) and Pb(II) Uptake from Water by MnFe2O4/Chitosan Nanoadsorbents. Water. 2021; 13(12):1662. https://doi.org/10.3390/w13121662
Chicago/Turabian StyleTaguba, Manny Anthony M., Dennis C. Ong, Benny Marie B. Ensano, Chi-Chuan Kan, Nurak Grisdanurak, Jurng-Jae Yee, and Mark Daniel G. de Luna. 2021. "Nonlinear Isotherm and Kinetic Modeling of Cu(II) and Pb(II) Uptake from Water by MnFe2O4/Chitosan Nanoadsorbents" Water 13, no. 12: 1662. https://doi.org/10.3390/w13121662
APA StyleTaguba, M. A. M., Ong, D. C., Ensano, B. M. B., Kan, C.-C., Grisdanurak, N., Yee, J.-J., & de Luna, M. D. G. (2021). Nonlinear Isotherm and Kinetic Modeling of Cu(II) and Pb(II) Uptake from Water by MnFe2O4/Chitosan Nanoadsorbents. Water, 13(12), 1662. https://doi.org/10.3390/w13121662







