Nitrification Process in a Nuclear Wastewater with High Load of Nitrogen, Uranium and Organic Matter under ORP Controlled
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation Synthetic Medium
2.2. Blended Real Nuclear Wastewater (BRNW)
2.3. Test Analysis
2.4. Monod Kinetics Model
2.5. Control of Environmental Conditions to Monitor ORP and in Relation to Monod Parameters
2.6. Inhibition Assays: Maximum Load of Blended Real Wastewater
2.7. Pre Scale-Up Operations in Moving Bed Bioreactor (MBBR) using BRNW
2.8. Mathematical Models
3. Results and Discussion
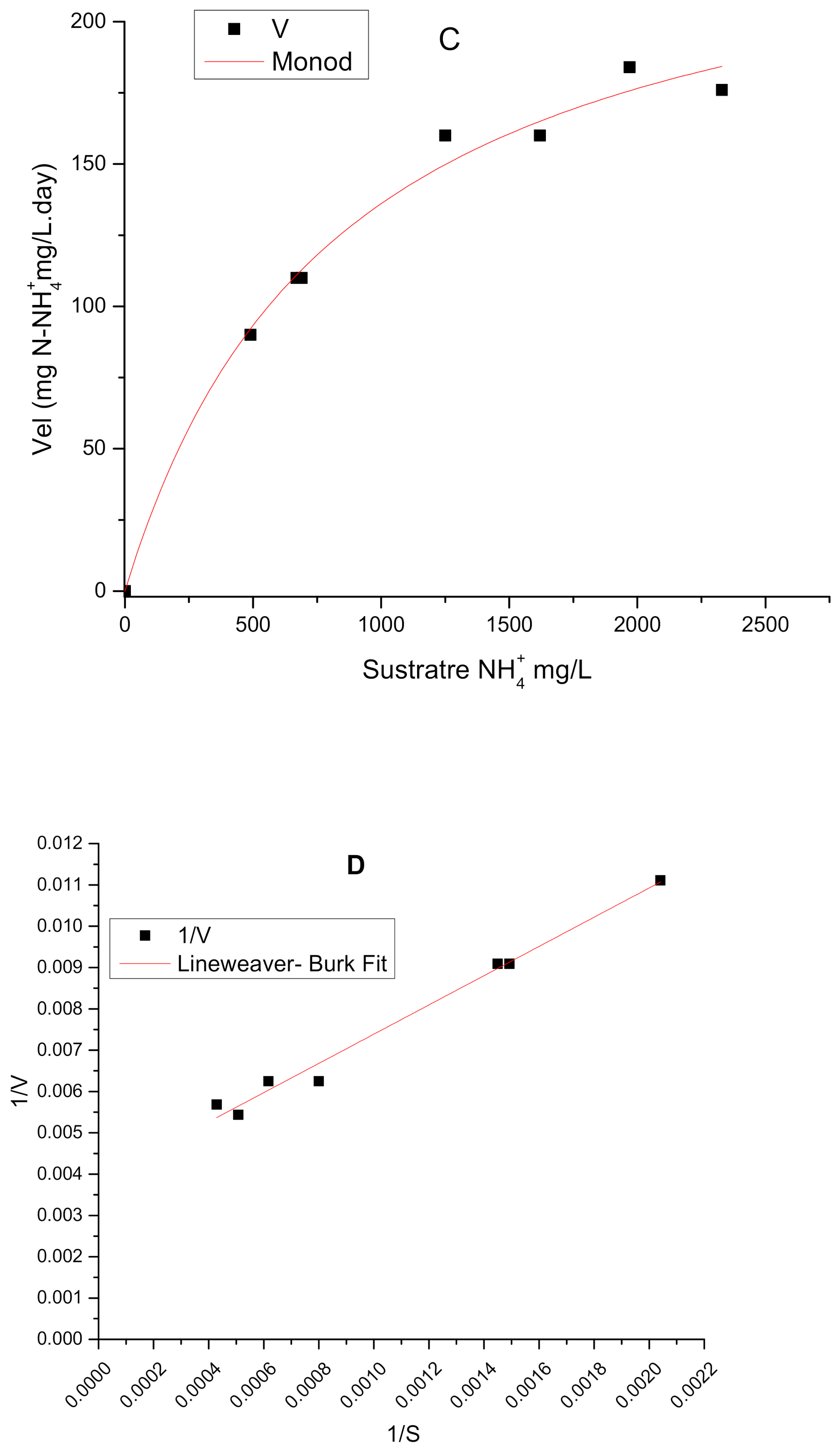
3.1. Monod Kinetic Parameters
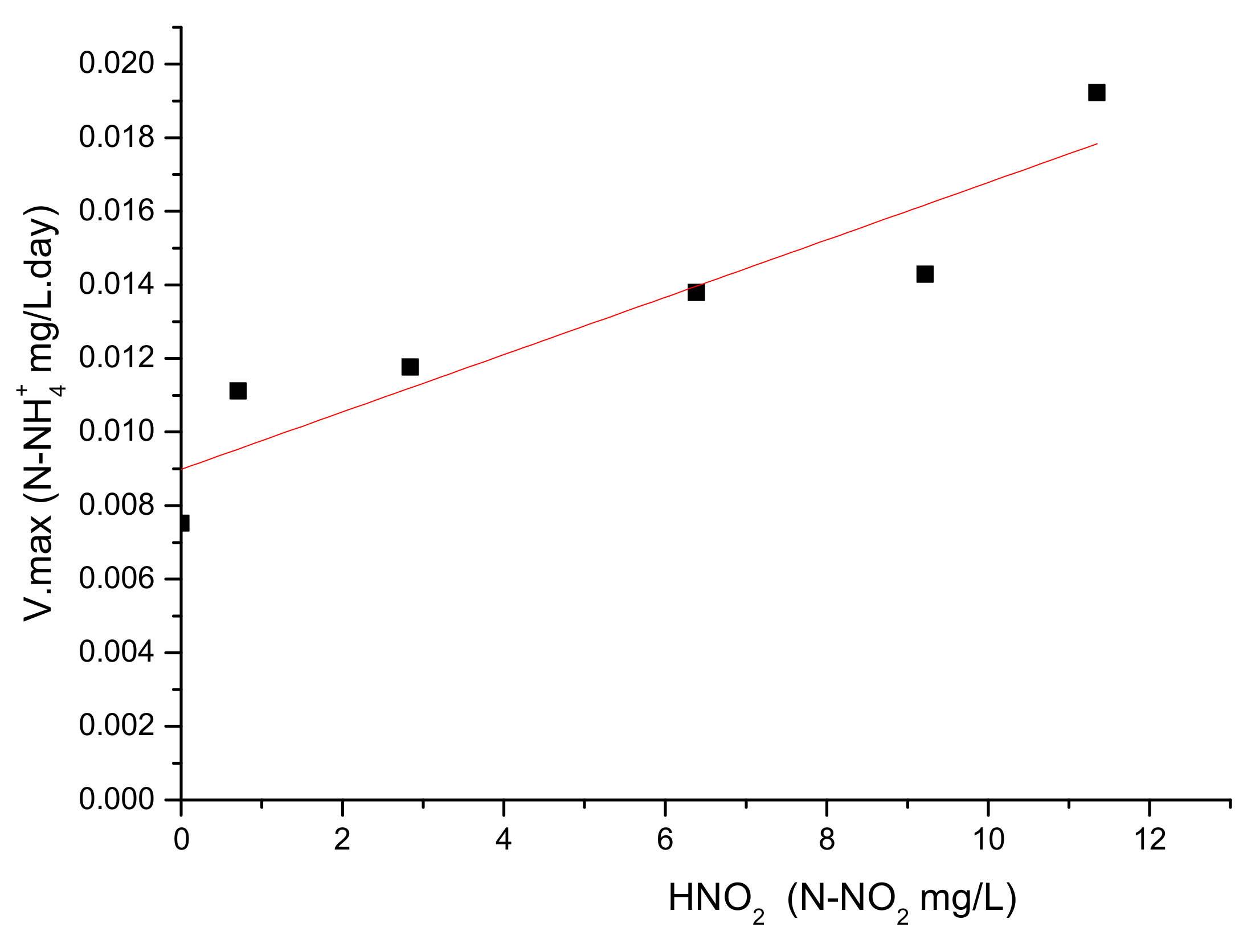
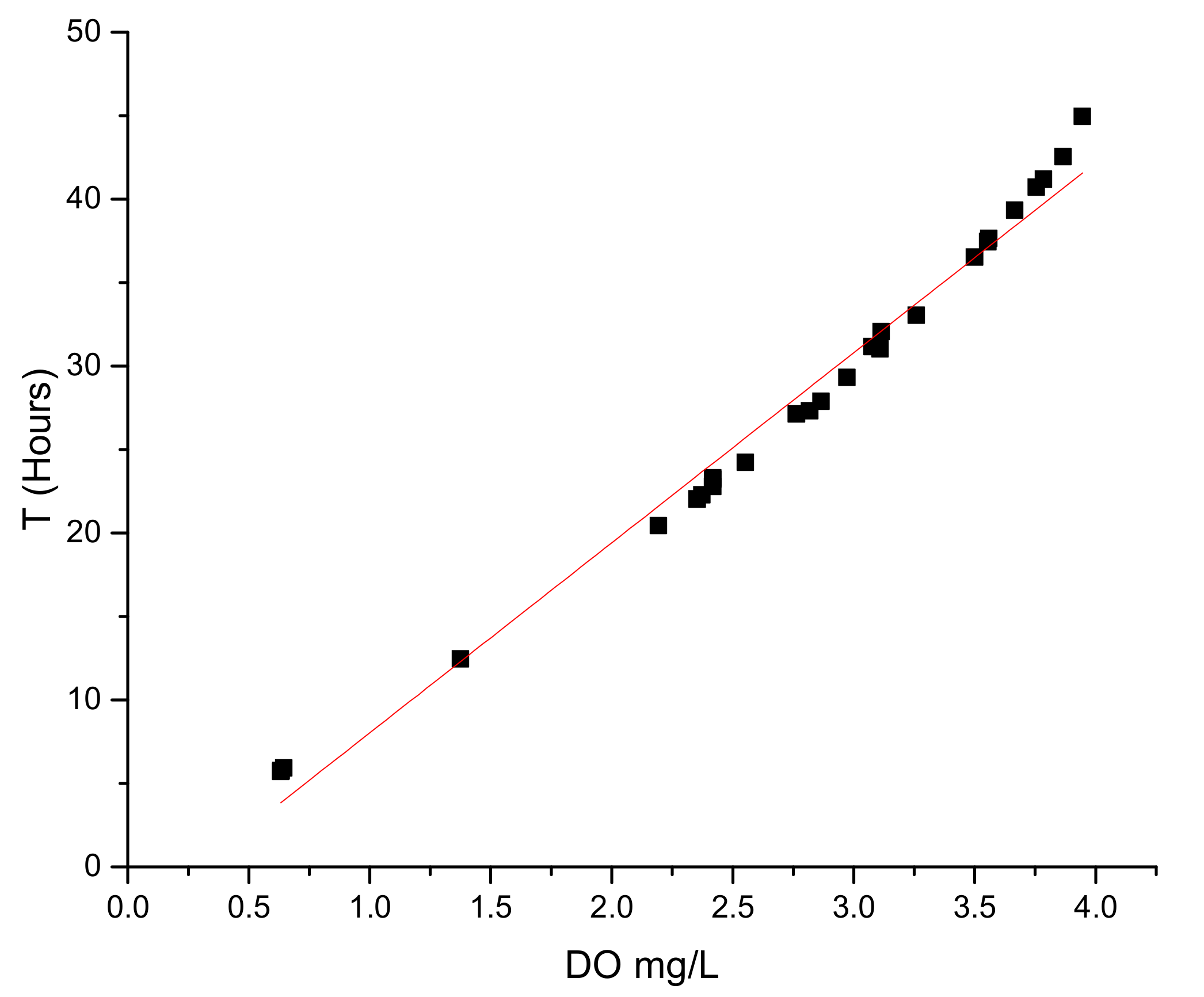
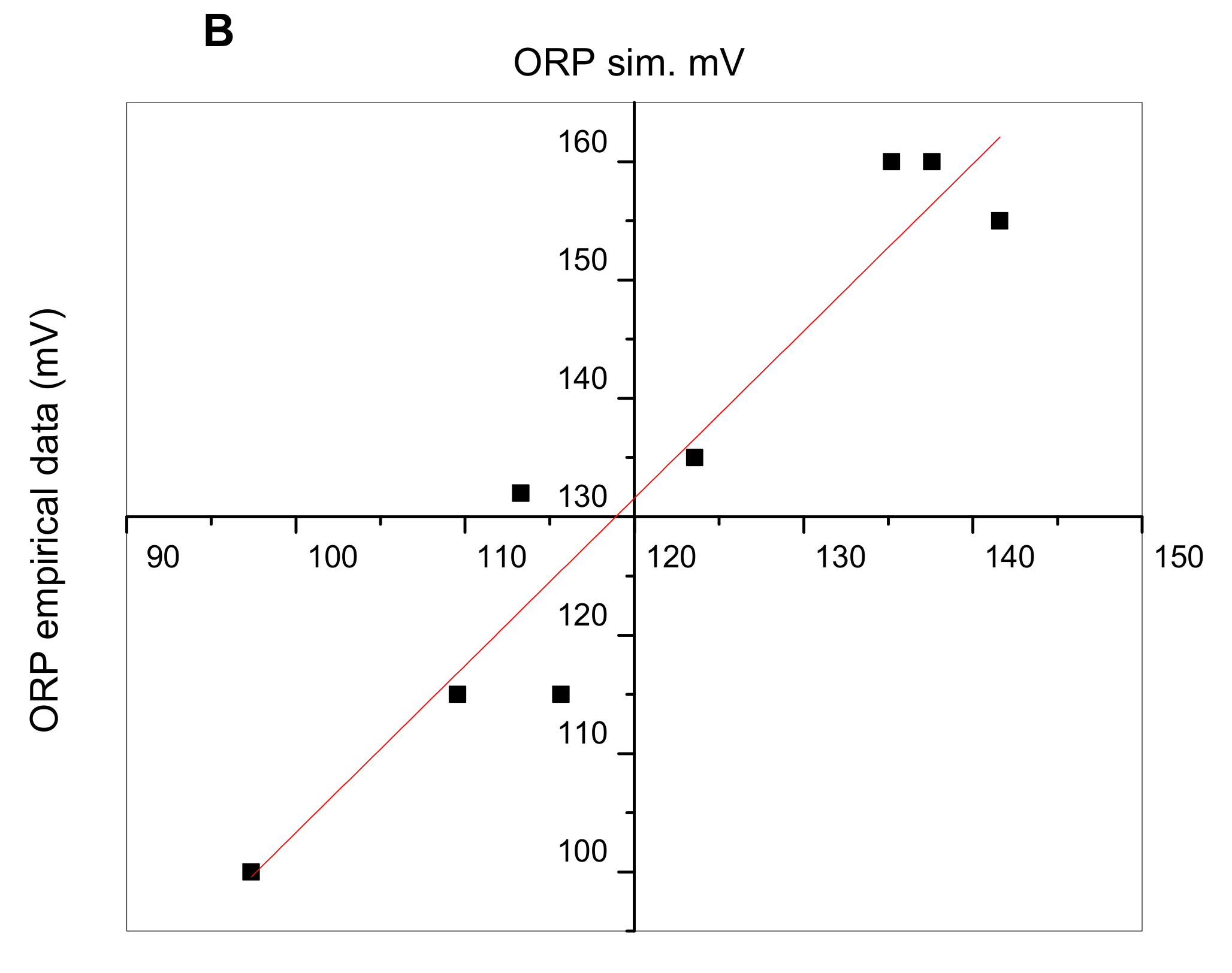
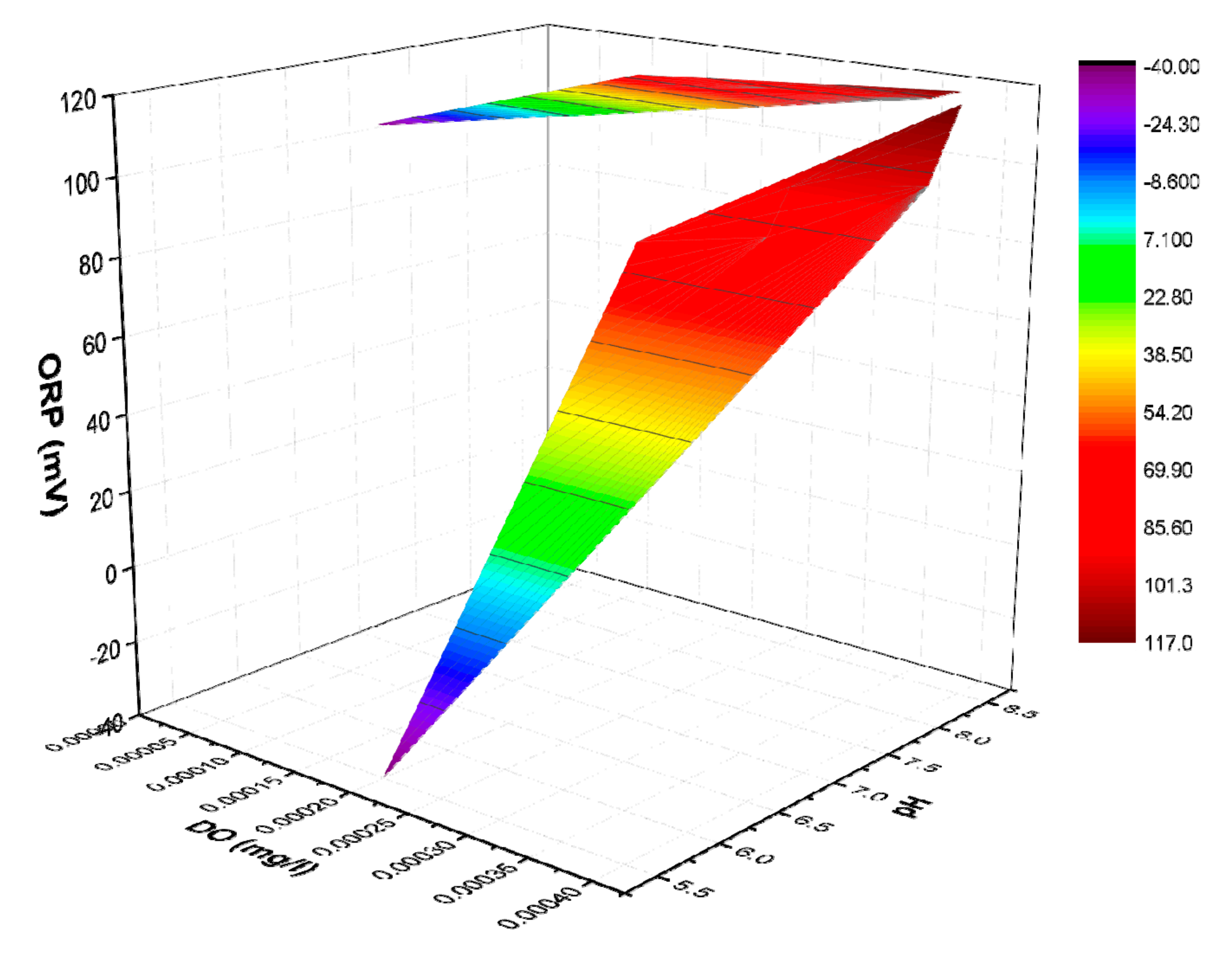
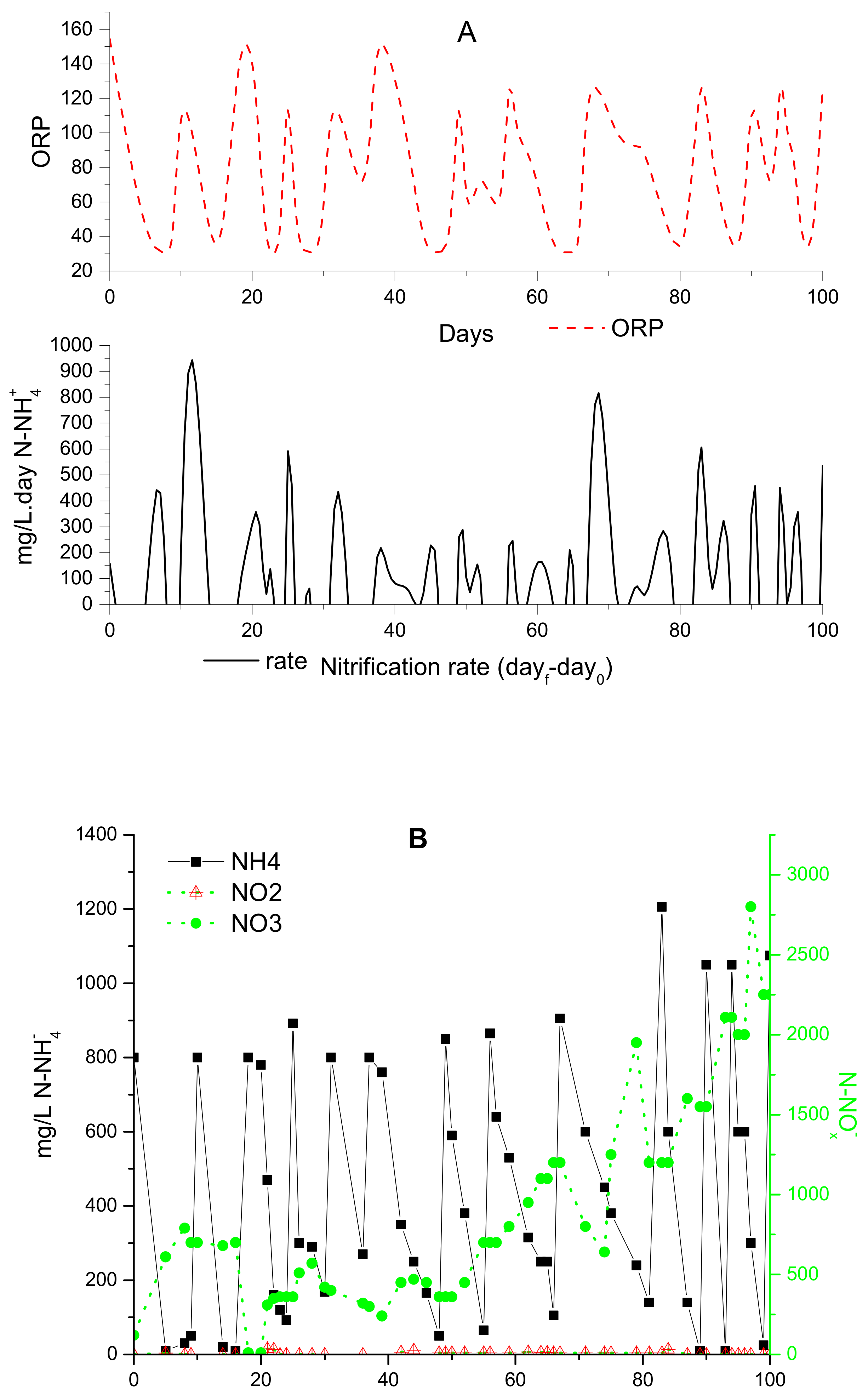
3.2. Control of ORP and Relation with Monod Parameters under Autotrophic Conditions
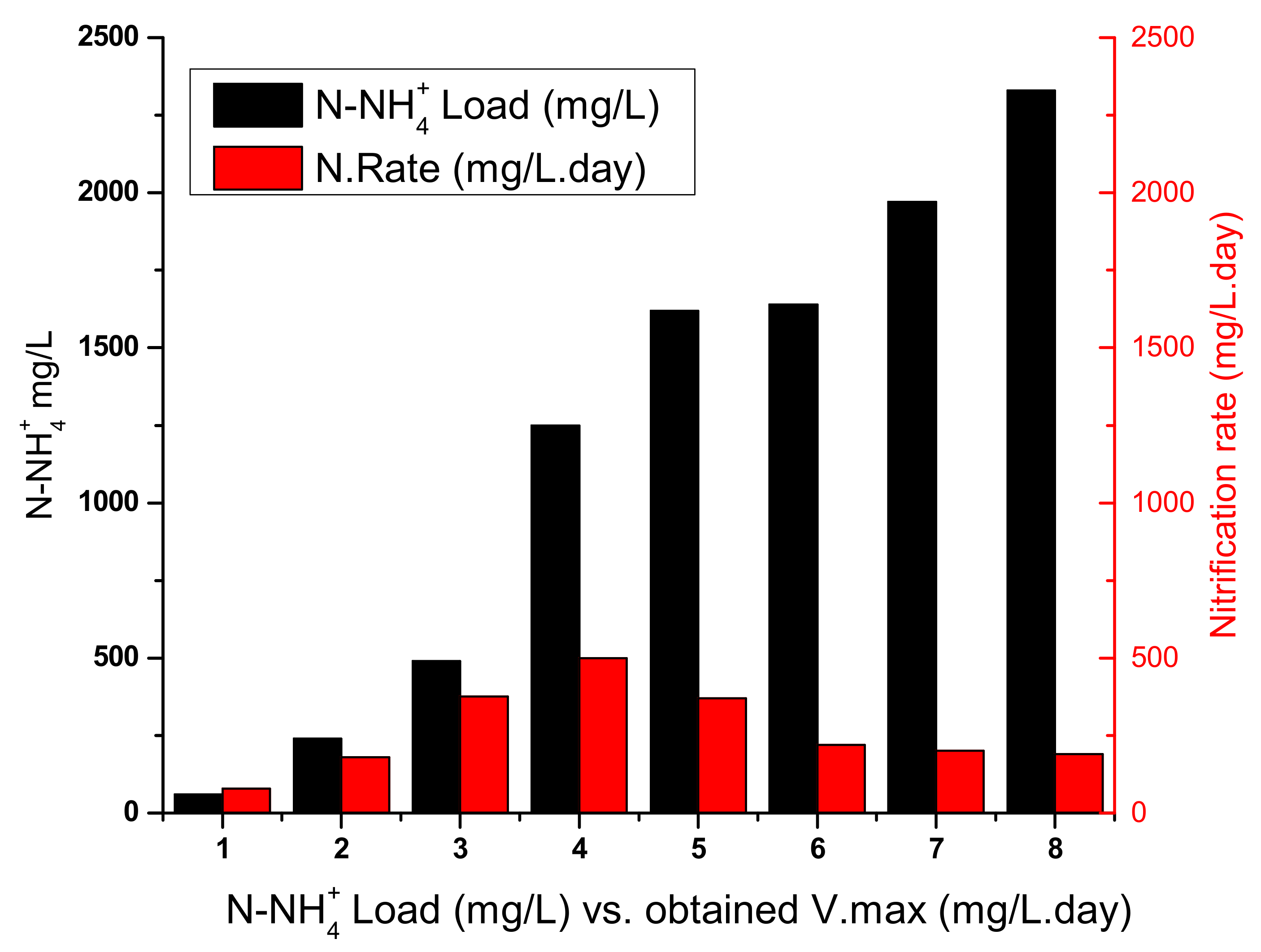
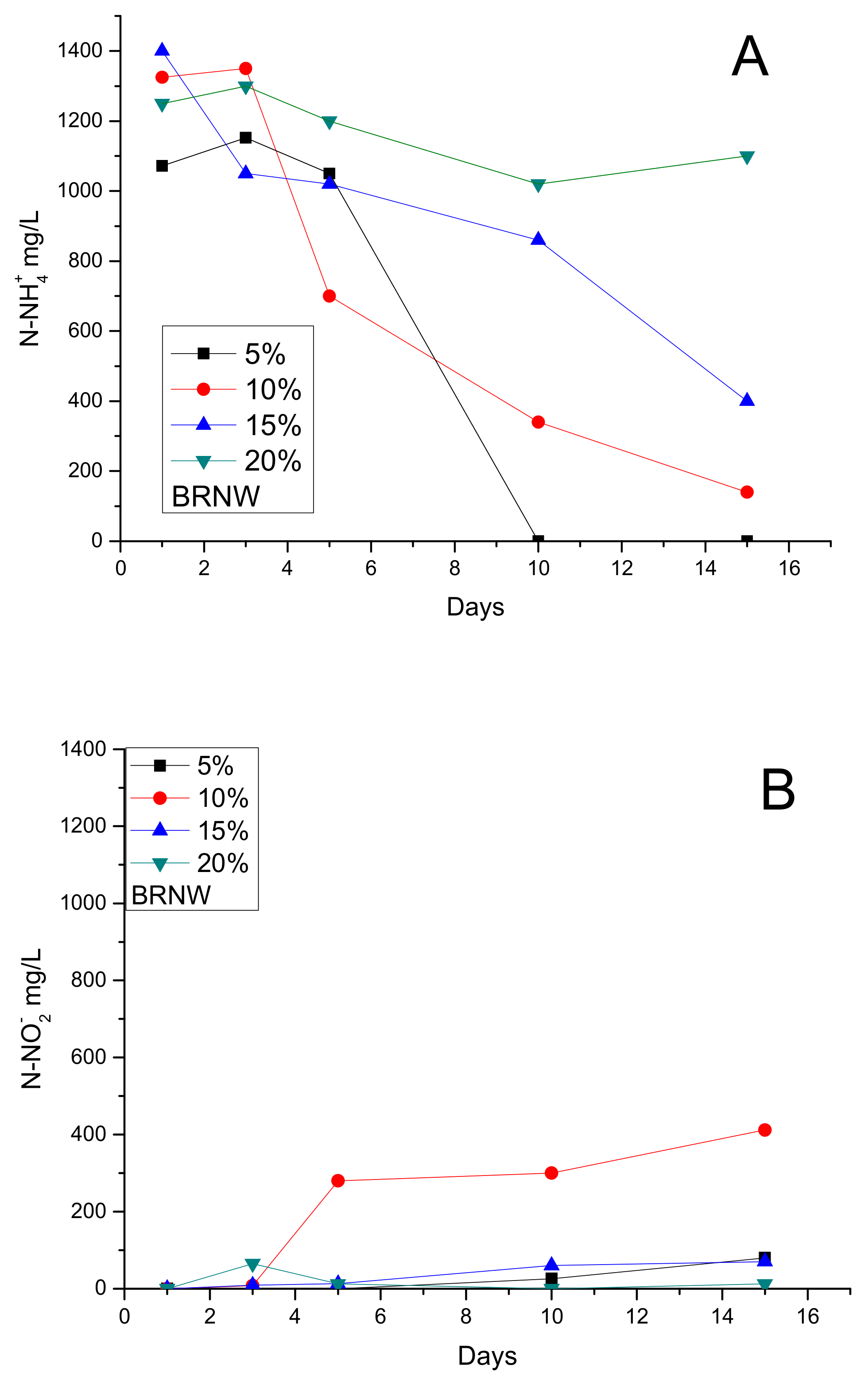
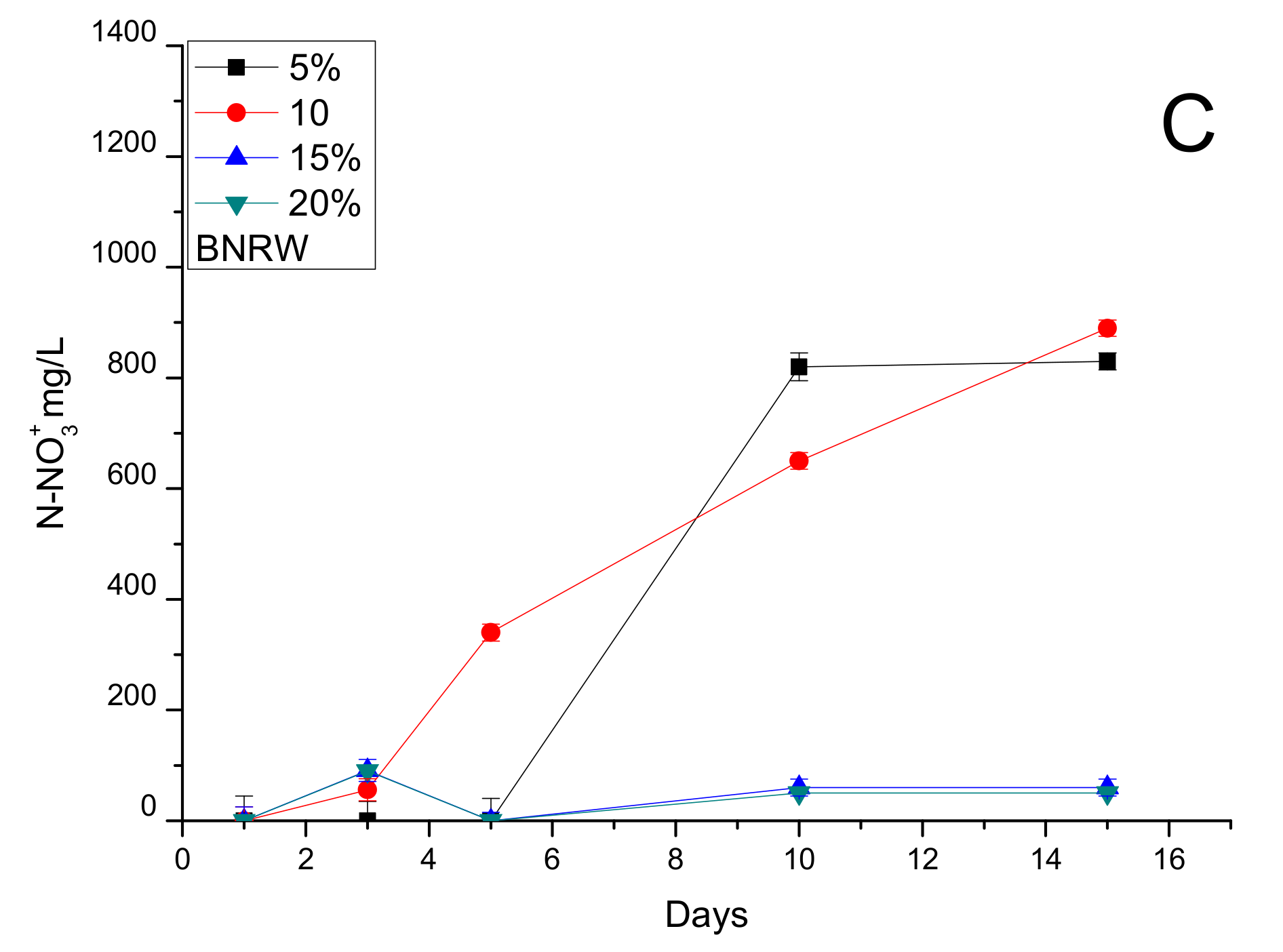
3.3. Inhibition Assays: Maximum Load of BRNW
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Cao, B.; Wu, J.; Yang, T.; Ma, X.; Chen, Y. Preliminary Study on Nuclear Fuel Cycle Scenarios of China before 2050. Energy Procedia 2013, 39, 294–299. [Google Scholar] [CrossRef]
- Setting Authorized Limits for Radioactive Discharges: Practical Issues to Consider. Available online: https://www.iaea.org/publications/8312/setting-authorized-limits-for-radioactive-discharges-practical-issues-to-consider (accessed on 27 March 2021).
- Edwards, C.R.; Oliver, A.J. Uranium Processing: A Review of Current Methods and Technology. JOM 2000, 52, 12–20. [Google Scholar] [CrossRef]
- Bhanot, P.; Celin, S.M.; Sreekrishnan, T.R.; Kalsi, A.; Sahai, S.K.; Sharma, P. Application of Integrated Treatment Strategies for Explosive Industry Wastewater—A Critical Review. J. Water Process Eng. 2020, 35, 101232. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A.; Zouboulis, A.I. Removal of Uranium from Contaminated Drinking Water: A Mini Review of Available Treatment Methods. Desalination Water Treat. 2013, 51, 2915–2925. [Google Scholar] [CrossRef]
- InfoLEG—Ministerio de Economía y Finanzas Públicas—Argentina. Available online: http://servicios.infoleg.gob.ar/infolegInternet/anexos/40000-44999/43795/norma.htm (accessed on 31 March 2021).
- National Research Council Uranium Mining in Virginia: Scientific, Technical, Environmental, Human Health and Safety, and Regulatory Aspects of Uranium Mining and Processing in Virginia; The National Academies Press: Washington, DC, USA, 2011; ISBN 978-0-309-22087-3.
- Smičiklas, I.; Šljivić-Ivanović, M. Radioactive Contamination of the Soil: Assessments of Pollutants Mobility with Implication to Remediation Strategies. Soil Contam.-Curr. Conseq. Furth. Solut. 2016. [Google Scholar] [CrossRef]
- Orrego, P.; Hernández, J.; Manríquez, J. Modeling Operational Parameters for Uranium Dioxide Production Reactor through Uranium Trioxide Reaction Using Hydrogen. World J. Nucl. Sci. Technol. 2016, 06, 131–139. [Google Scholar] [CrossRef]
- Jeong, D.; Cho, K.; Lee, C.-H.; Lee, S.; Bae, H. Integration of Forward Osmosis Process and a Continuous Airlift Nitrifying Bioreactor Containing PVA/Alginate-Immobilized Cells. Chem. Eng. J. 2016, 306, 1212–1222. [Google Scholar] [CrossRef]
- Quan, X.; Huang, K.; Li, M.; Lan, M.; Li, B. Nitrogen Removal Performance of Municipal Reverse Osmosis Concentrate with Low C/N Ratio by Membrane-Aerated Biofilm Reactor. Front. Environ. Sci. Eng. 2018, 12, 5. [Google Scholar] [CrossRef]
- Zero Liquid Discharge (ZLD). Available online: https://condorchem.com/en/zld/ (accessed on 6 October 2018).
- Roch Nicholas Analysis of Ammonia Removal from Wastewater Market: Feasibility of Saltworks Introducing New Technology. Master’s Thesis, Simon Fraser University, Burnaby, BC, Canada, 2015.
- Dhangar, K.; Kumar, M. Tricks and Tracks in Removal of Emerging Contaminants from the Wastewater through Hybrid Treatment Systems: A Review. Sci. Total Environ. 2020, 738, 140320. [Google Scholar] [CrossRef]
- Schoeman, J.; Steyn, A. Nitrate Removal with Reverse Osmosis in a Rural Area in South Africa. Desalination 2003, 155, 15–26. [Google Scholar] [CrossRef]
- Gadd, G.M.; Pan, X. Biomineralization, Bioremediation and Biorecovery of Toxic Metals and Radionuclides. Geomicrobiol. J. 2016, 33, 175–178. [Google Scholar] [CrossRef]
- Puyol, D.; Batstone, D.J.; Hülsen, T.; Astals, S.; Peces, M.; Krömer, J.O. Resource Recovery from Wastewater by Biological Technologies: Opportunities, Challenges, and Prospects. Front. Microbiol. 2017, 7. [Google Scholar] [CrossRef]
- Daphne, L. Stoner Biotechnological Treatment of Liquid and Solid Inorganic Wastes; Library of Congress Cataloging-in-Publication Data; LEWIS PUBLISHERS; CRC Press: Boca Raton, FL, USA, 1993; pp. 71–95. ISBN 978-1-315-13844-2. [Google Scholar]
- Rai, A.; Chauhan, P.S.; Bhattacharya, S. Remediation of Industrial Effluents. In Water Remediation; Energy, Environment, and Sustainability; Bhattacharya, S., Gupta, A.B., Gupta, A., Pandey, A., Eds.; Springer: Singapore, 2018; pp. 171–187. ISBN 978-981-10-7551-3. [Google Scholar]
- Waugh, W.J.; Glenn, E.P.; Benson, C.H.; Albright, W.H.; Brusseau, M.L.; Bush, R.P.; Dayvault, J. Applications of Ecological Engineering Remedies for Uranium Processing Sites; US Department of Energy/Office of Legacy Management: Washington, DC, USA, 2016; p. 2. [Google Scholar]
- Jaramillo, F.; Orchard, M.; Muñoz, C.; Zamorano, M.; Antileo, C. Advanced Strategies to Improve Nitrification Process in Sequencing Batch Reactors—A Review. J. Environ. Manag. 2018, 218, 154–164. [Google Scholar] [CrossRef]
- What Is the Best Biological Process for Nitrogen Removal: When and Why? | Environmental Science & Technology. Available online: https://pubs.acs.org/doi/10.1021/acs.est.7b05832 (accessed on 29 December 2020).
- Sri Shalini, S.; Joseph, K. Combined SHARON and ANAMMOX Processes for Ammoniacal Nitrogen Stabilisation in Landfill Bioreactors. Bioresour. Technol. 2018, 250, 723–732. [Google Scholar] [CrossRef]
- Stoichiometric Evaluation of Partial Nitritation, Anammox and Denitrification Processes in a Sequencing Batch Reactor and Interpretation of Online Monitoring Parameters. Available online: https://europepmc.org/article/med/27614985 (accessed on 27 March 2021).
- Wu, P.; Zhang, X.; Wang, X.; Wang, C.; Faustin, F.; Liu, W. Characterization of the Start-up of Single and Two-Stage Anammox Processes with Real Low-Strength Wastewater Treatment. Chemosphere 2020, 245, 125572. [Google Scholar] [CrossRef]
- Li, J.; Peng, Y.; Zhang, L.; Li, X.; Zhang, Q.; Yang, S.; Gao, Y.; Li, S. Improving Efficiency and Stability of Anammox through Sequentially Coupling Nitritation and Denitritation in a Single-Stage Bioreactor. Environ. Sci. Technol. 2020, 54, 10859–10867. [Google Scholar] [CrossRef]
- Huynh, T.V.; Nguyen, P.D.; Phan, T.N.; Luong, D.H.; Truong, T.T.V.; Huynh, K.A.; Furukawa, K. Application of CANON Process for Nitrogen Removal from Anaerobically Pretreated Husbandry Wastewater. Int. Biodeterior. Biodegrad. 2019, 136, 15–23. [Google Scholar] [CrossRef]
- Lv, Y.; Chen, X.; Wang, L.; Ju, K.; Chen, X.; Miao, R.; Wang, X. Microprofiles of Activated Sludge Aggregates Using Microelectrodes in Completely Autotrophic Nitrogen Removal over Nitrite (CANON) Reactor. Front. Environ. Sci. Eng. 2016. [Google Scholar] [CrossRef]
- Pochana, K.; Keller, J. Study of Factors Affecting Simultaneous Nitrification and Denitrification (SND). Water Sci. Technol. 1999, 39, 61–68. [Google Scholar] [CrossRef]
- Lozano Avilés, A.B.; Del Cerro Velázquez, F.; Lloréns Pascual del Riquelme, M. Methodology for Energy Optimization in Wastewater Treatment Plants. Phase III: Implementation of an Integral Control System for the Aeration Stage in the Biological Process of Activated Sludge and the Membrane Biological Reactor. Sensors 2020, 20, 4342. [Google Scholar] [CrossRef]
- Majtacz, J.; Grubba, D.; Czerwionka, K. Application of the Anammox Process for Treatment of Liquid Phase Digestate. Water 2020, 12, 2965. [Google Scholar] [CrossRef]
- Gao, D.-W.; Peng, Y.-Z.; Liang, H.; Wang, P. Using Oxidation-Reduction Potential (ORP) and PH Value for Process Control of Shortcut Nitrification-Denitrification. J. Environ. Sci. Health A Toxic Hazard. Subst. Environ. Eng. 2003, 38, 2933–2942. [Google Scholar] [CrossRef]
- Rodriguez-Sanchez, A.; Margareto, A.; Robledo-Mahon, T.; Aranda, E.; Diaz-Cruz, S.; Gonzalez-Lopez, J.; Barcelo, D.; Vahala, R.; Gonzalez-Martinez, A. Performance and Bacterial Community Structure of a Granular Autotrophic Nitrogen Removal Bioreactor Amended with High Antibiotic Concentrations. Chem. Eng. J. 2017, 325, 257–269. [Google Scholar] [CrossRef]
- Yu, R.-F.; Liaw, S.-L.; Chang, C.-N.; Lu, H.-J.; Cheng, W.-Y. Monitoring and Control Using On-Line Orp on the Continuous-Flow Activated Sludge Batch Reactor System. Water Sci. Technol. 1997, 35, 57–66. [Google Scholar] [CrossRef]
- Rodríguez, D.C.; Pino, N.; Peñuela, G. Monitoring the Removal of Nitrogen by Applying a Nitrification–Denitrification Process in a Sequencing Batch Reactor (SBR). Bioresour. Technol. 2011, 102, 2316–2321. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-N.; Cheng, H.-B.; Chao, A.C. Applying the Nernst Equation to Simulate Redox Potential Variations for Biological Nitrification and Denitrification Processes. Environ. Sci. Technol. 2004, 38, 1807–1812. [Google Scholar] [CrossRef] [PubMed]
- Ying, X.-B.; Tang, C.-Y.; Guo, W.; Sheng, D.-S.; Wang, M.-Z.; Feng, H.-J. Quantifying the Electron-Donating and -Accepting Capacities of Wastewater for Evaluating and Optimizing Biological Wastewater Treatment Processes. J. Environ. Sci. 2021, 102, 235–243. [Google Scholar] [CrossRef]
- Kassem Alef, P.N. Methods in Applied Soil Microbiology and Biochemistry, 1st ed.; 1era 1995; Academic Press: London, UK, 2002; ISBN 978-0-08-052748-2. [Google Scholar]
- Ying, Q.; Xiao-tong, C.; Bing, L.; Gen-na, F.; Yang, W.; You-lin, S.; Zhen-ming, L.; Ya-ping, T.; Chun-he, T. The Conceptual Flowsheet of Effluent Treatment during Preparing Spherical Fuel Elements of HTR. Nucl. Eng. Des. 2014, 271, 189–192. [Google Scholar] [CrossRef]
- How Uranium Ore Is Made into Nuclear Fuel—World Nuclear Association. Available online: https://www.world-nuclear.org/nuclear-basics/how-is-uranium-ore-made-into-nuclear-fuel.aspx (accessed on 27 July 2019).
- Chen, X.; He, L.; Liu, B.; Tang, Y.; Tang, C. The Uranium Recovery from UO2 Kernel Production Effluent. Nucl. Eng. Des. 2016, 310, 187–191. [Google Scholar] [CrossRef]
- Métodos Normalizados Para el Análisis de Aguas Potables y Residuales APHA-AWWA-WPCF 978-84-7978-031-9. Available online: http://www.diazdesantosargentina.com//libros/apha-awwa-wpcf-metodos-normalizados-para-el-analisis-de-aguas-potables-y-residuales-L03000310201.html (accessed on 30 May 2021).
- Bhattacharya, R.; Mazumder, D. Evaluation of Nitrification Kinetics for Treating Ammonium Nitrogen Enriched Wastewater in Moving Bed Hybrid Bioreactor. J. Environ. Chem. Eng. 2020, 104589. [Google Scholar] [CrossRef]
- Monod, J.; Wyman, J.; Changeux, J.-P. On the Nature of Allosteric Transitions: A Plausible Model. J. Mol. Biol. 1965, 12, 88–118. [Google Scholar] [CrossRef]
- Carrera, J.; Jubany, I.; Carvallo, L.; Chamy, R.; Lafuente, J. Kinetic Models for Nitrification Inhibition by Ammonium and Nitrite in a Suspended and an Immobilised Biomass Systems. Process Biochem. 2004, 39, 1159–1165. [Google Scholar] [CrossRef]
- Wiesmann, U. Biological Nitrogen Removal from Wastewater. Adv. Biochem. Eng. Biotechnol. 1994, 51, 113–154. [Google Scholar] [CrossRef] [PubMed]
- Ciudad, G.; Werner, A.; Bornhardt, C.; Muñoz, C.; Antileo, C. Differential Kinetics of Ammonia- and Nitrite-Oxidizing Bacteria: A Simple Kinetic Study Based on Oxygen Affinity and Proton Release during Nitrification. Process Biochem. 2006, 41, 1764–1772. [Google Scholar] [CrossRef]
- Ciudad, G.; González, R.; Bornhardt, C.; Antileo, C. Modes of Operation and PH Control as Enhancement Factors for Partial Nitrification with Oxygen Transport Limitation. Water Res. 2007, 41, 4621–4629. [Google Scholar] [CrossRef] [PubMed]
- Venturini, M.; Perez, M.; Curutchet, G.; Silva Paulo, P.; Pizarro, R. Biological Nitrogen Removal of Wastewater from the Nuclear Industry with High Load. In Environmental Biotechnology and Engineering Advances; 2016; CINVESTAV: Mexico City, Mexico, 2018; pp. 716–726. ISBN 978-607-9023-51-5. [Google Scholar]
- Liu, Y.; Ngo, H.H.; Guo, W.; Peng, L.; Wang, D.; Ni, B. The Roles of Free Ammonia (FA) in Biological Wastewater Treatment Processes: A Review. Environ. Int. 2019, 123, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Anthonisen, A.C.; Loehr, R.C.; Prakasam, T.B.S.; Srinath, E.G. Inhibition of Nitrification by Ammonia and Nitrous Acid. Journal (Water Pollut. Control Fed.) 1976, 48, 835–852. [Google Scholar] [PubMed]
- Chandran, K.; Hu, Z.; Smets, B.F. A Critical Comparison of Extant Batch Respirometric and Substrate Depletion Assays for Estimation of Nitrification Biokinetics. Biotechnol. Bioeng. 2008, 101, 62–72. [Google Scholar] [CrossRef]
- Ginestet, P.; Audic, J.M.; Urbain, V.; Block, J.C. Estimation of Nitrifying Bacterial Activities by Measuring Oxygen Uptake in the Presence of the Metabolic Inhibitors Allylthiourea and Azide. Appl. Environ. Microbiol. 1998, 64, 2266–2268. [Google Scholar] [CrossRef]
- Cheng, H.-B.; Kumar, M.; Lin, J.-G. Interpretation of Redox Potential Variation during Biological Denitrification Using Linear Non-Equilibrium Thermodynamic Model. Int. Biodeterior. Biodegrad. 2012, 67, 28–39. [Google Scholar] [CrossRef]
- Muloiwa, M.; Nyende-Byakika, S.; Dinka, M. Comparison of Unstructured Kinetic Bacterial Growth Models. S. Afr. J. Chem. Eng. 2020, 33, 141–150. [Google Scholar] [CrossRef]
- Ianus GmbH Simulation of (De-)Nitrification Tanks. Ianus-Simulation GmbH. Available online: https://ianus-simulation.de/en/de-nitrification-tanks/ (accessed on 27 May 2021).
- dos Santos, A.L.S.; Galdino, A.C.M.; de Mello, T.P.; Ramos, L.d.S.; Branquinha, M.H.; Bolognese, A.M.; Columbano Neto, J.; Roudbary, M.; dos Santos, A.L.S.; Galdino, A.C.M.; et al. What Are the Advantages of Living in a Community? A Microbial Biofilm Perspective! Memórias do Instituto Oswaldo Cruz 2018, 113. [Google Scholar] [CrossRef]
- Wang, X.; Wang, W.; Li, Y.; Zhang, J.; Zhang, Y.; Li, J. Biofilm Activity, Ammonia Removal and Cell Growth of the Heterotrophic Nitrifier, Acinetobacter sp., Facilitated by Exogenous N-Acyl-Homoserine Lactones. RSC Adv. 2018, 8, 30783–30793. [Google Scholar] [CrossRef]
- Roy, D.; Benkaraache, S.; Lemay, J.-F.; Landry, D.; Drogui, P.; Tyagi, R.D. High-Strength Ammonium Wastewater Treatment by MBR: Steady-State Nitrification Kinetic Parameters. J. Water Process Eng. 2019, 32, 100945. [Google Scholar] [CrossRef]

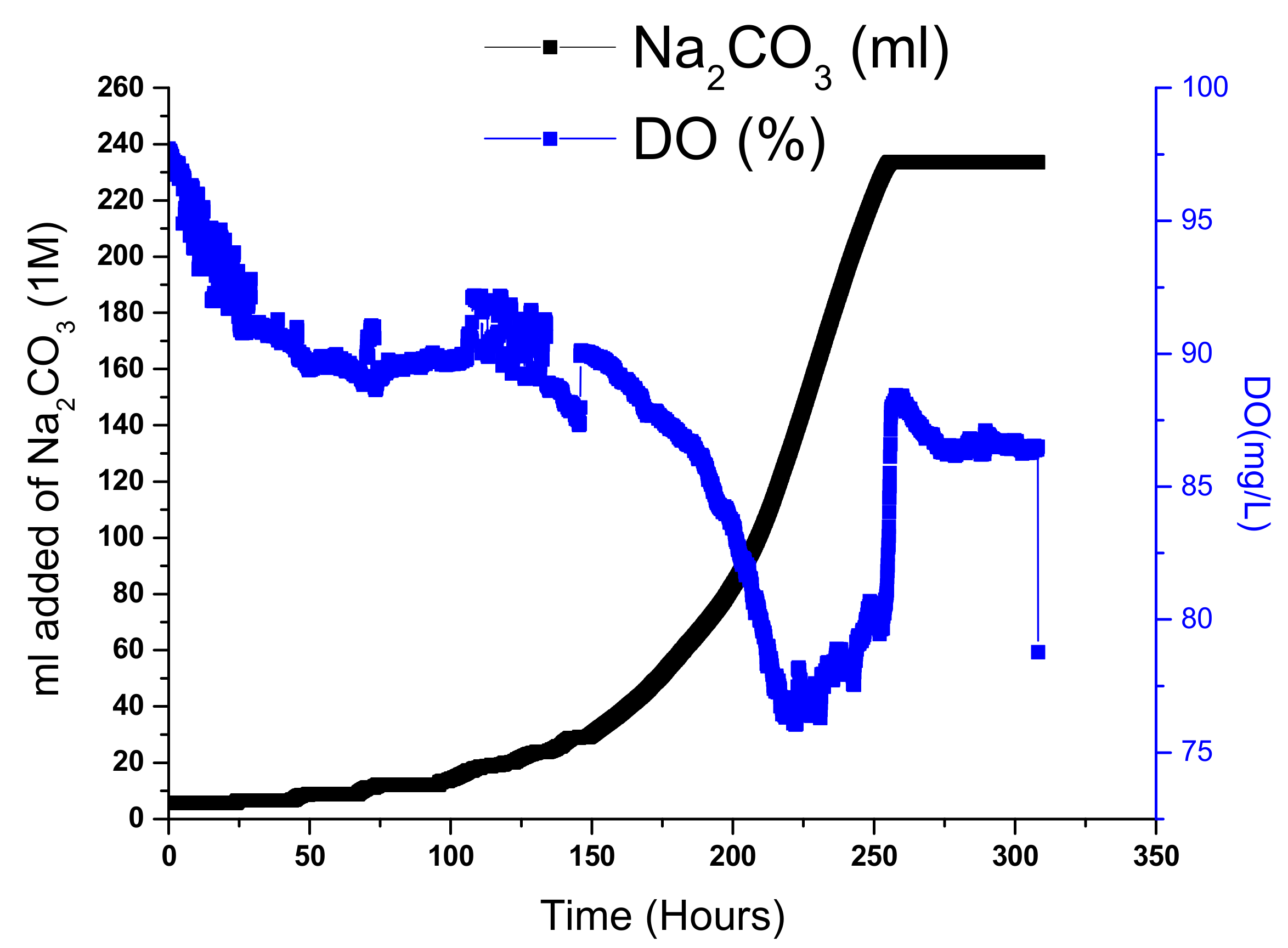
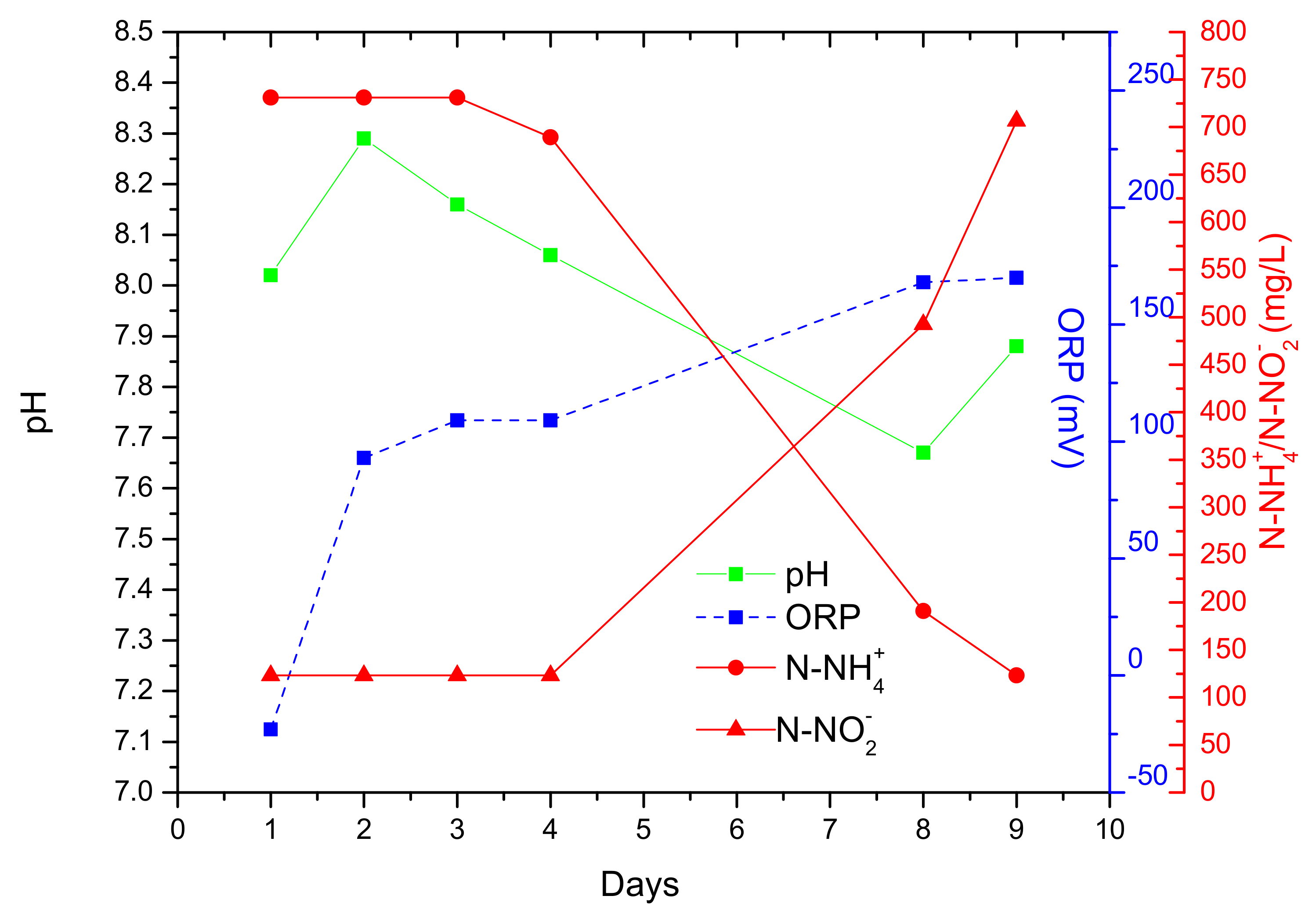
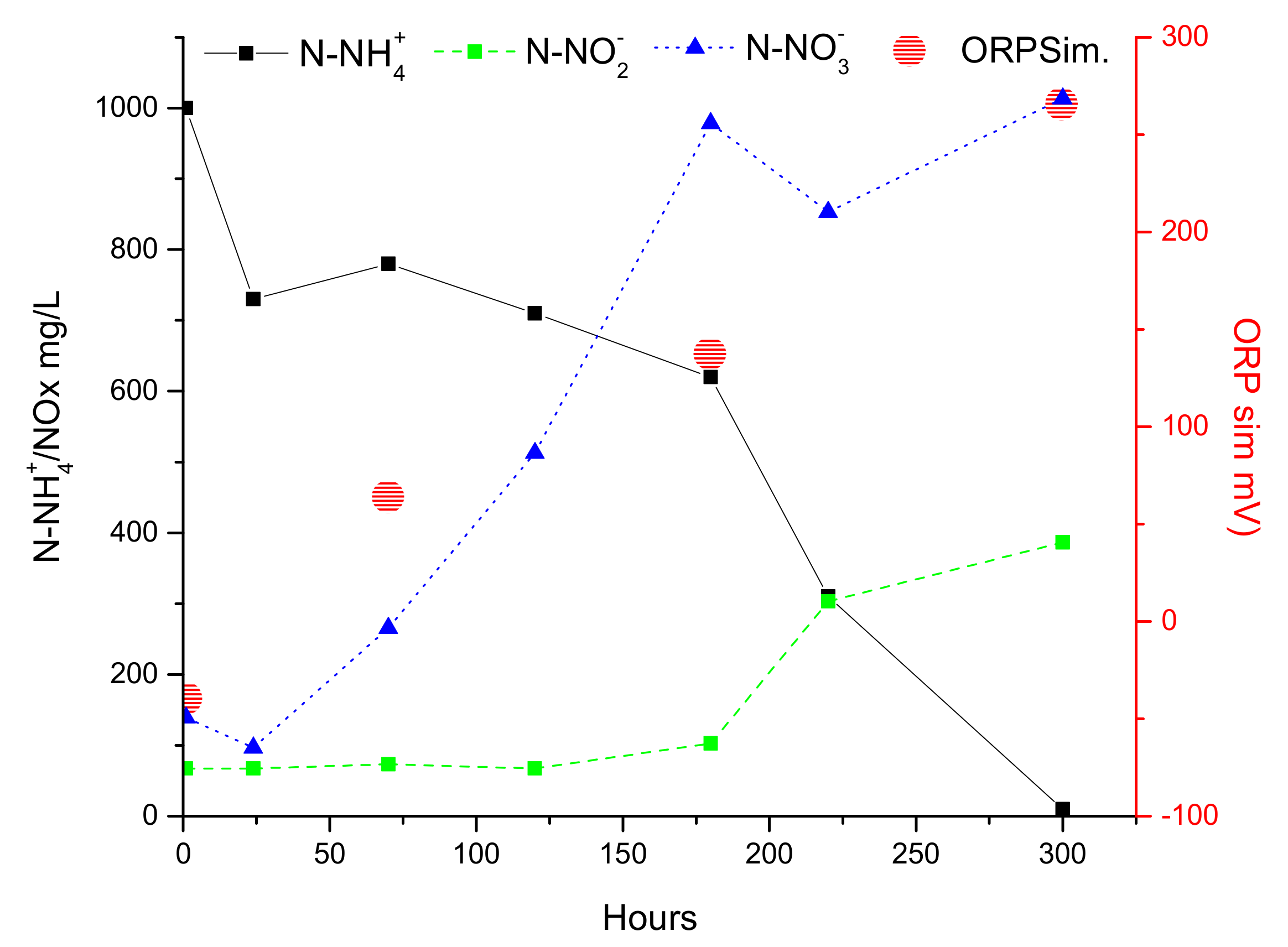
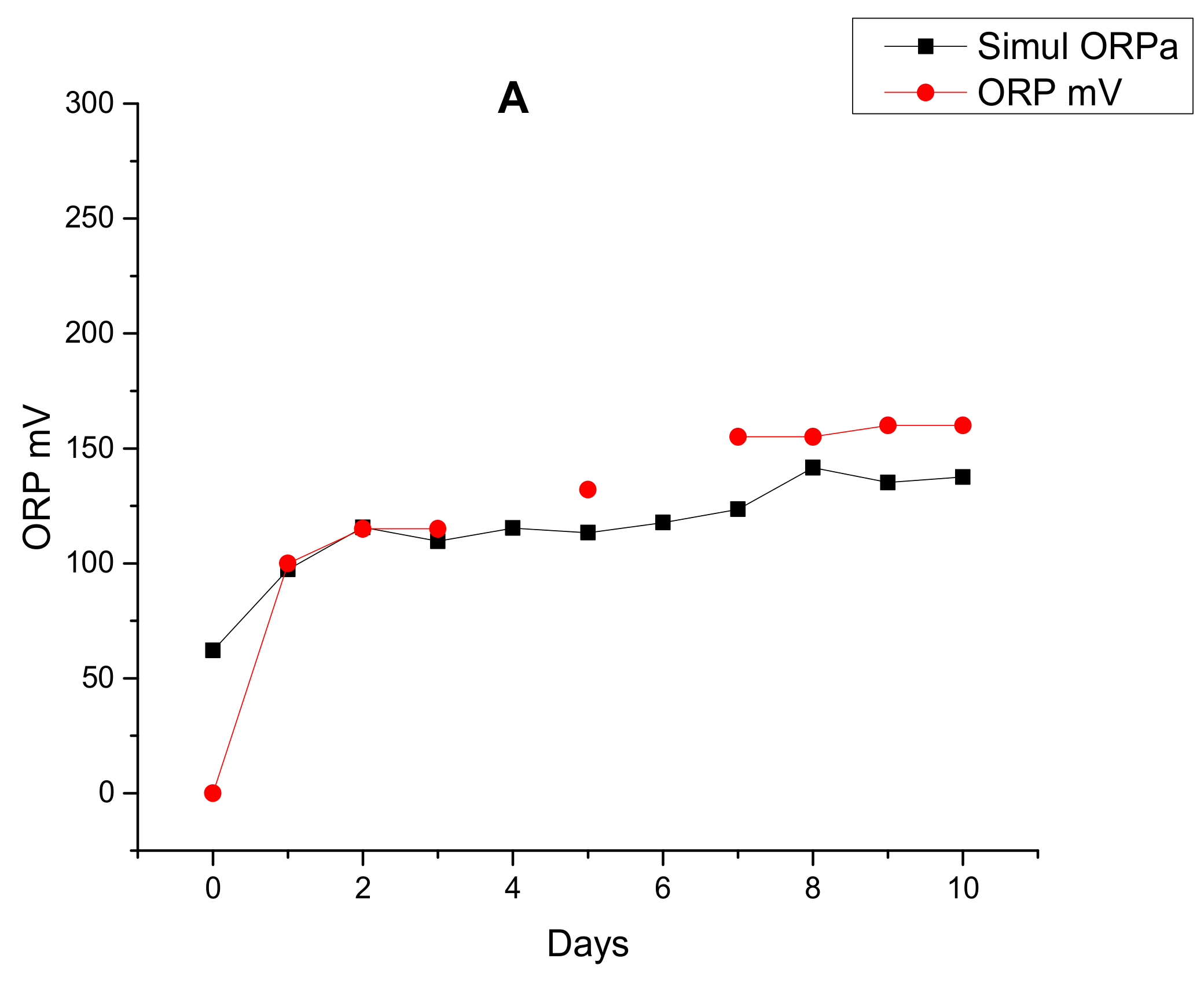
| Parameters | Acclimation-Autothropic Medium-Nuclear Effluent (Stage 1) | Acclimation-Heterothropic Medium (Stage 2) | Blended Real Nuclear Wastewater (BRNW) | |
|---|---|---|---|---|
| Unit | Concentration Minimum-Maximum | Concentration Minimum-Maximum | Concentration Minimum-Maximum | |
| Ammonium | mg/L | 500–700 | 600–900 | 600–1400 |
| Nitrate | mg/L | 0–400 | 100–800 | 1000–1300 |
| COD | mg/L | 0 | 9000 | 1500 |
| pH | 6.0–8.0 | 6.0–8.0 | 7.0–7.8 | |
| Uranium | mg/L | 300–600 | 0 | 300–600 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venturini, M.; Rossen, A.; Paulo, P.S. Nitrification Process in a Nuclear Wastewater with High Load of Nitrogen, Uranium and Organic Matter under ORP Controlled. Water 2021, 13, 1607. https://doi.org/10.3390/w13111607
Venturini M, Rossen A, Paulo PS. Nitrification Process in a Nuclear Wastewater with High Load of Nitrogen, Uranium and Organic Matter under ORP Controlled. Water. 2021; 13(11):1607. https://doi.org/10.3390/w13111607
Chicago/Turabian StyleVenturini, Mariano, Ariana Rossen, and Patricia Silva Paulo. 2021. "Nitrification Process in a Nuclear Wastewater with High Load of Nitrogen, Uranium and Organic Matter under ORP Controlled" Water 13, no. 11: 1607. https://doi.org/10.3390/w13111607
APA StyleVenturini, M., Rossen, A., & Paulo, P. S. (2021). Nitrification Process in a Nuclear Wastewater with High Load of Nitrogen, Uranium and Organic Matter under ORP Controlled. Water, 13(11), 1607. https://doi.org/10.3390/w13111607






