Nutrient Removal in Sequential Batch Polishing Ponds
Abstract
:1. Introduction
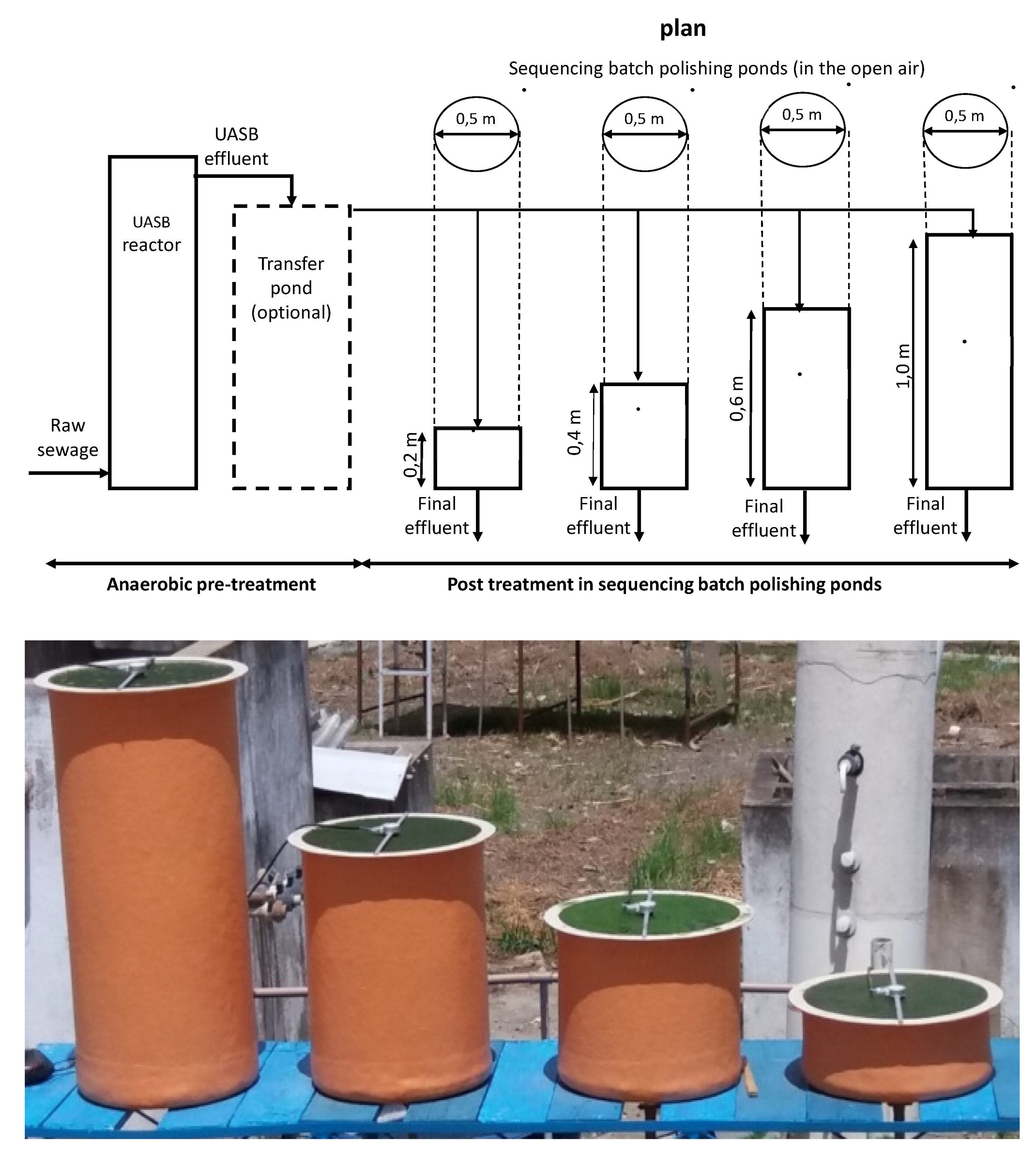
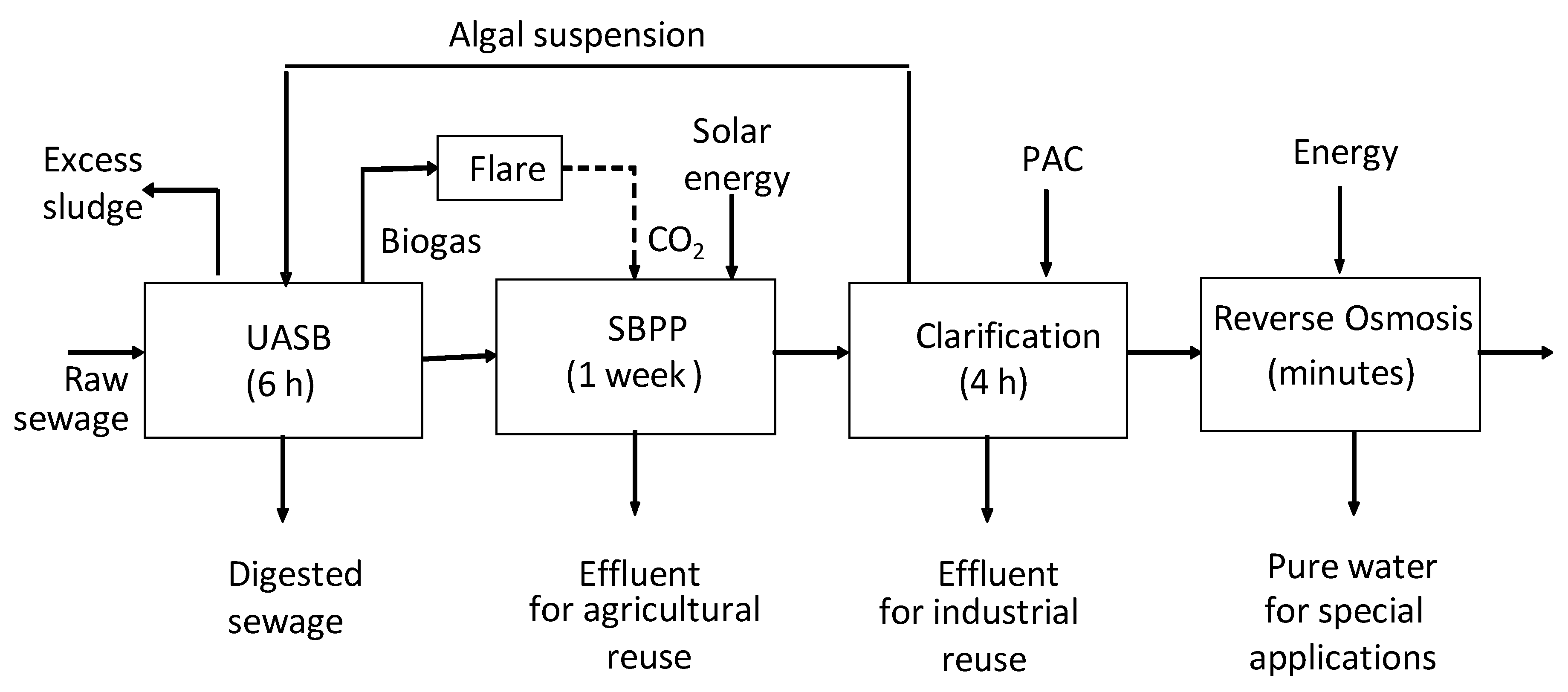
2. Materials and Methods
2.1. pH Value and Variation in Treatment Systems
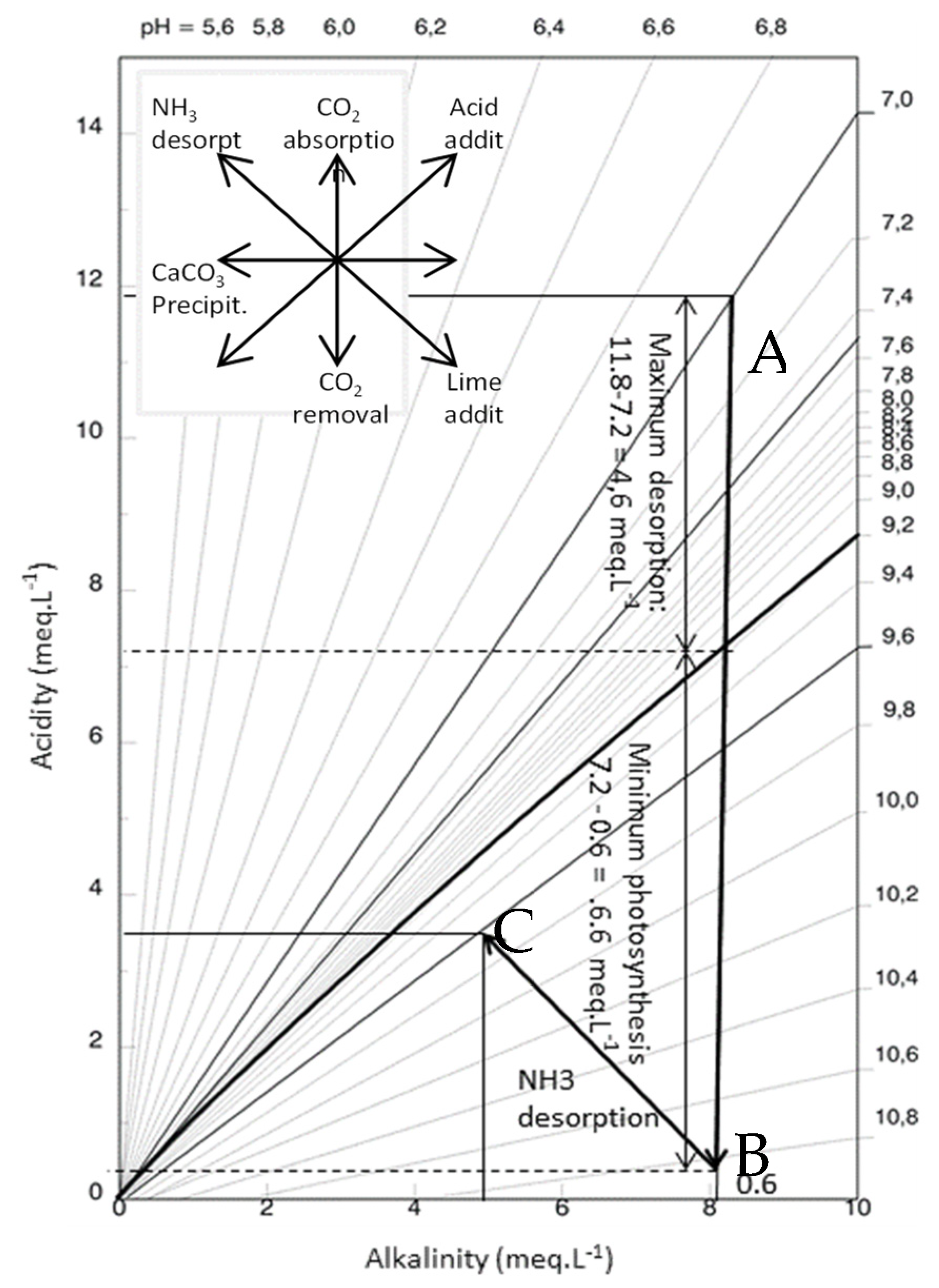
2.1.1. CO2 Removal
2.1.2. Physical CO2 Removal
2.1.3. Biological CO2 Removal
2.1.4. Ammonium Removal
3. Results and Discussion
3.1. Experimental Results
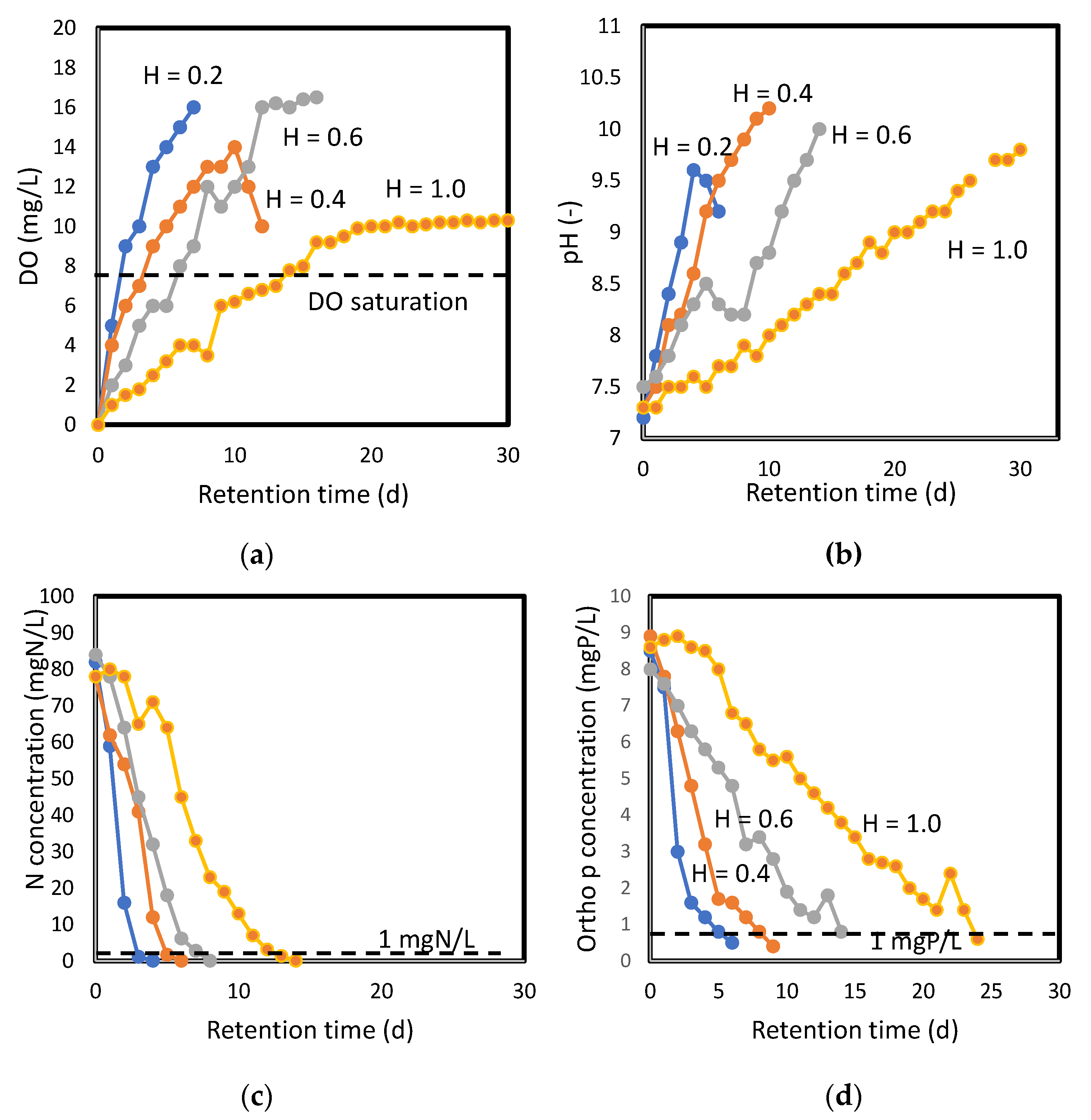
3.1.1. DO Profile
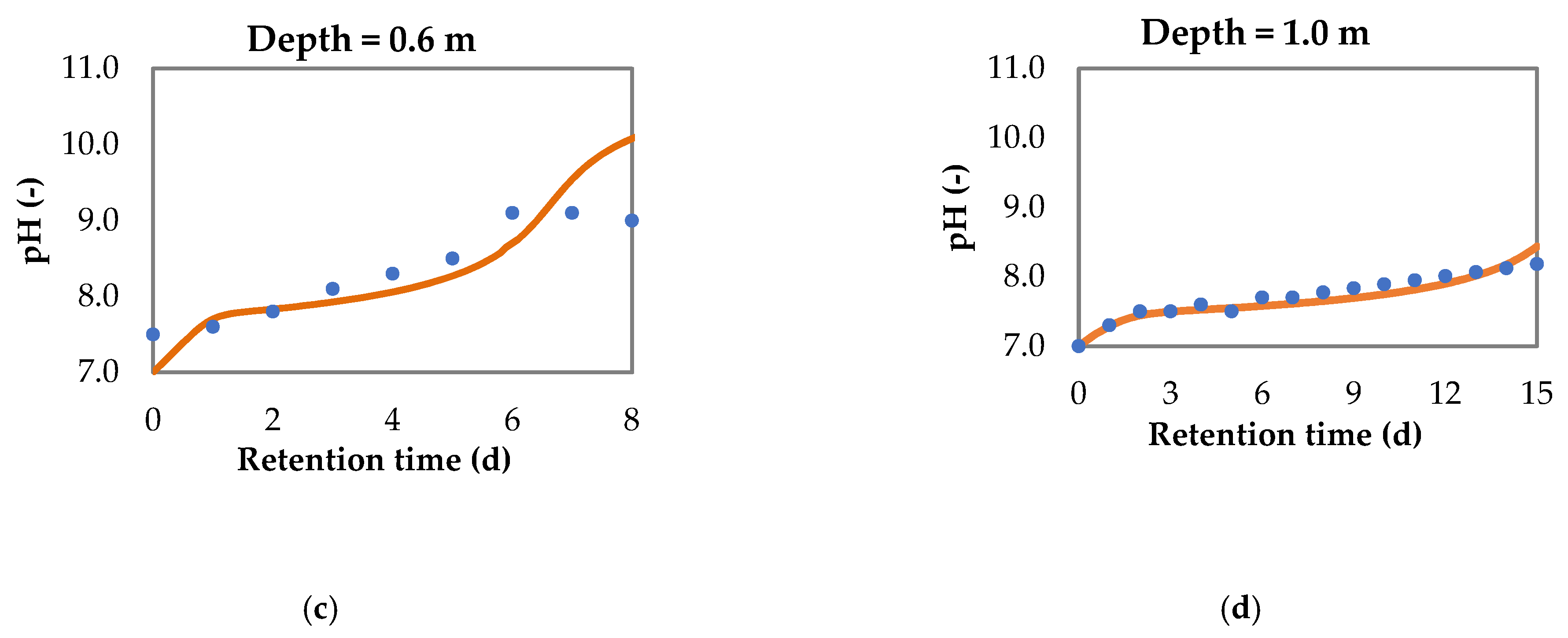
3.1.2. pH Increase
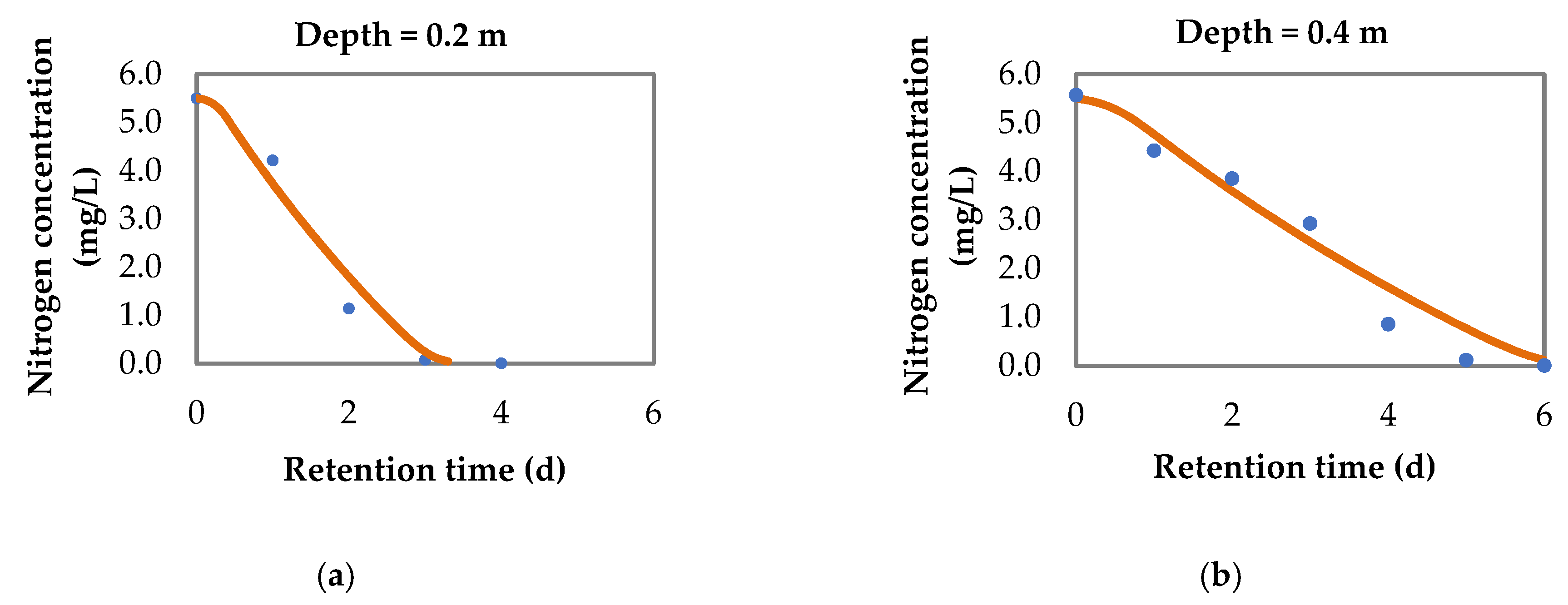
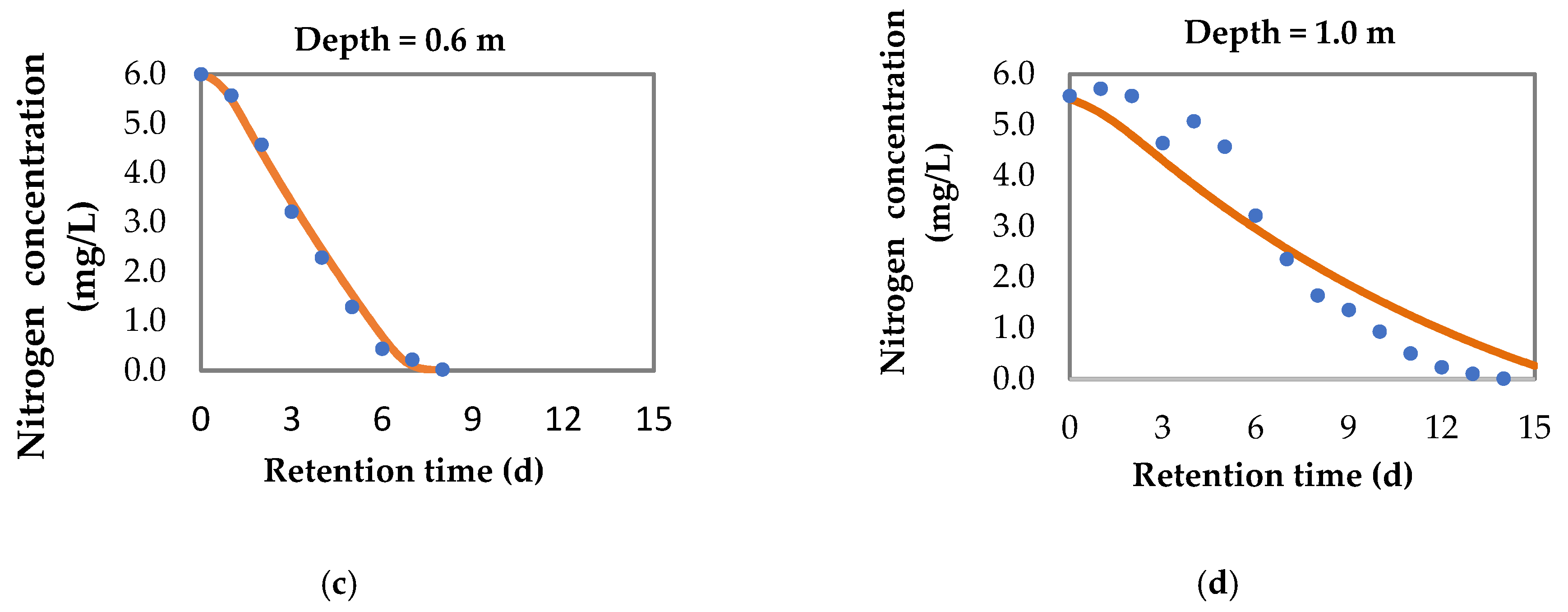
3.1.3. Nitrogen Removal
3.1.4. Phosphorus Removal
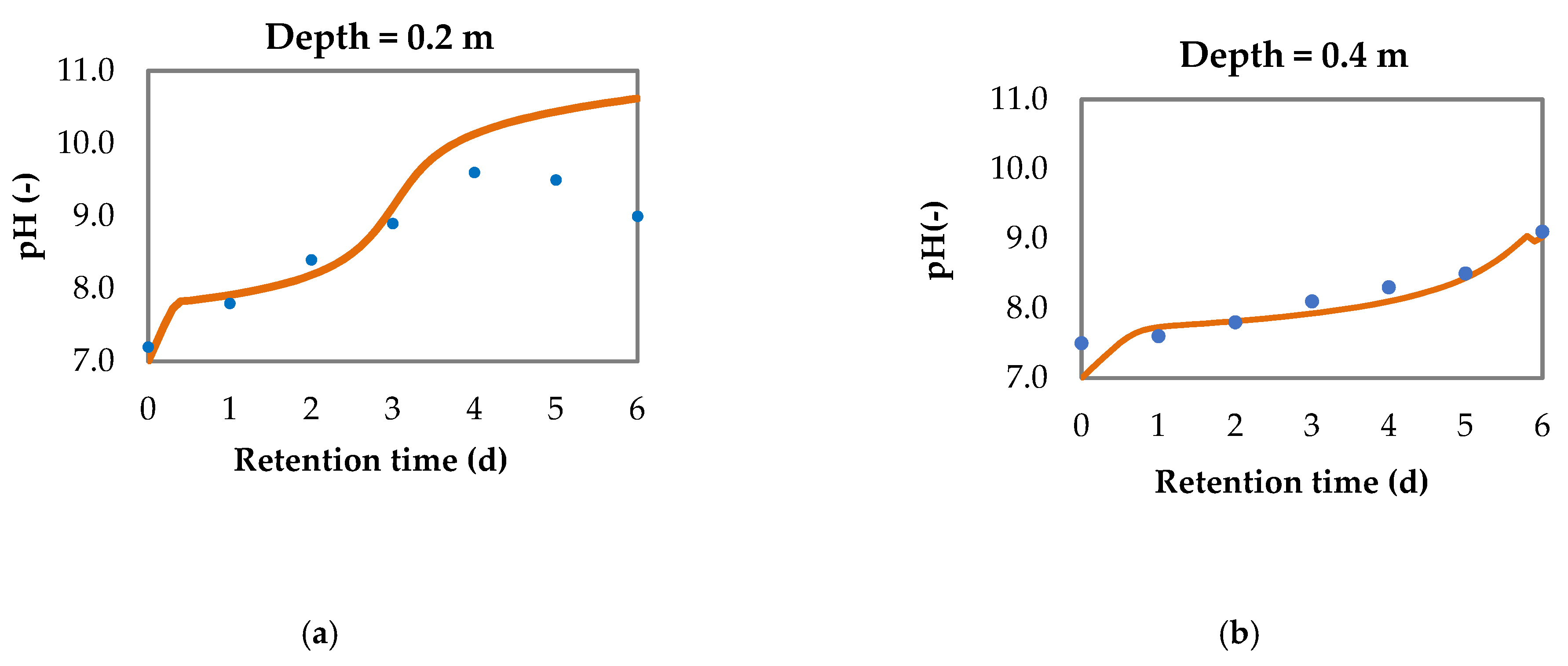
3.2. Model for pH Variation and Nitrogen Removal in Sequential Batch Polishing Ponds
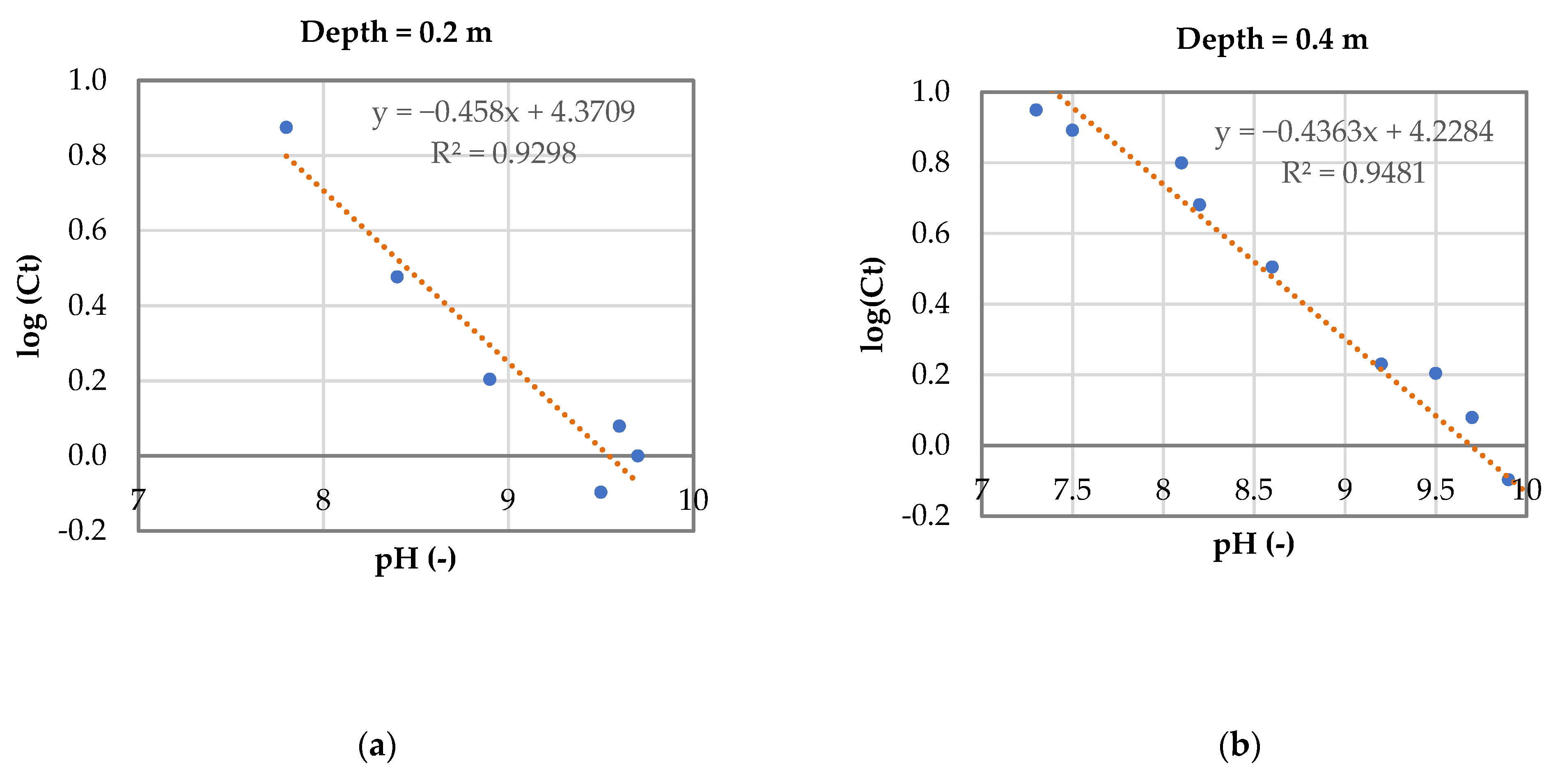
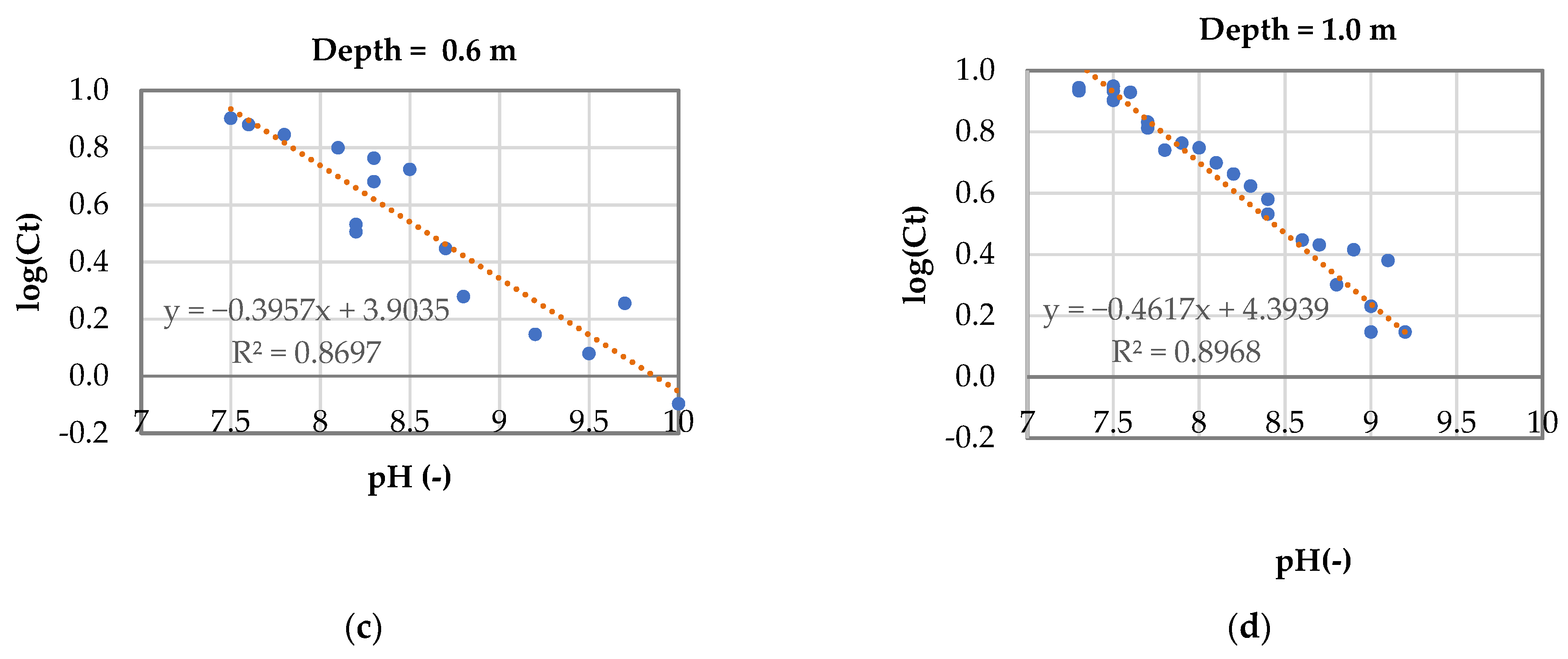
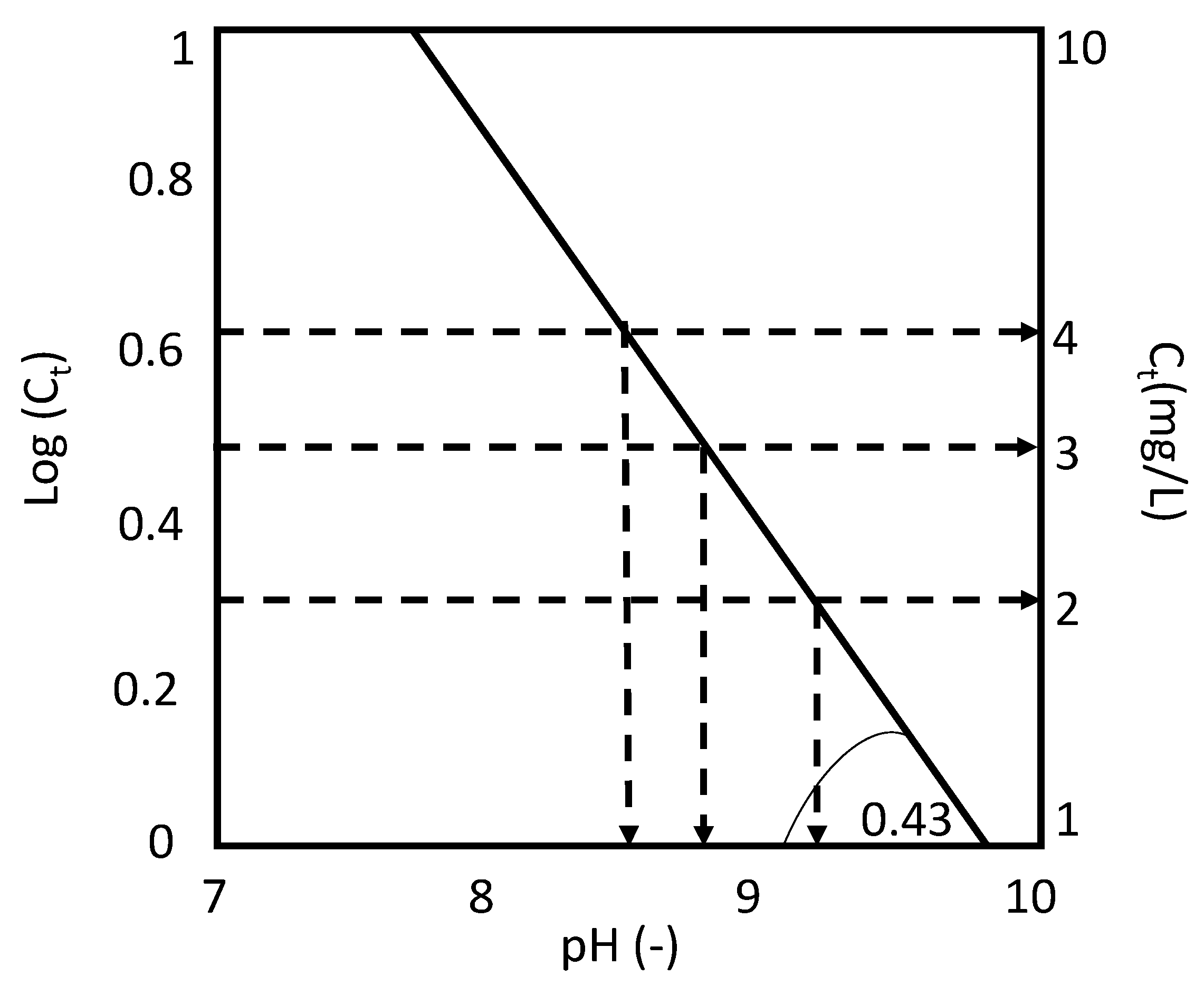
3.3. Model for Phosphorus Removal in Sequential Batch Polishing Ponds
- Log(Ct) = −0.43pH + 4.29
- 2.
- For an effluent P concentration of 2 mg·L−1: logCt = log 2 = 0.32;
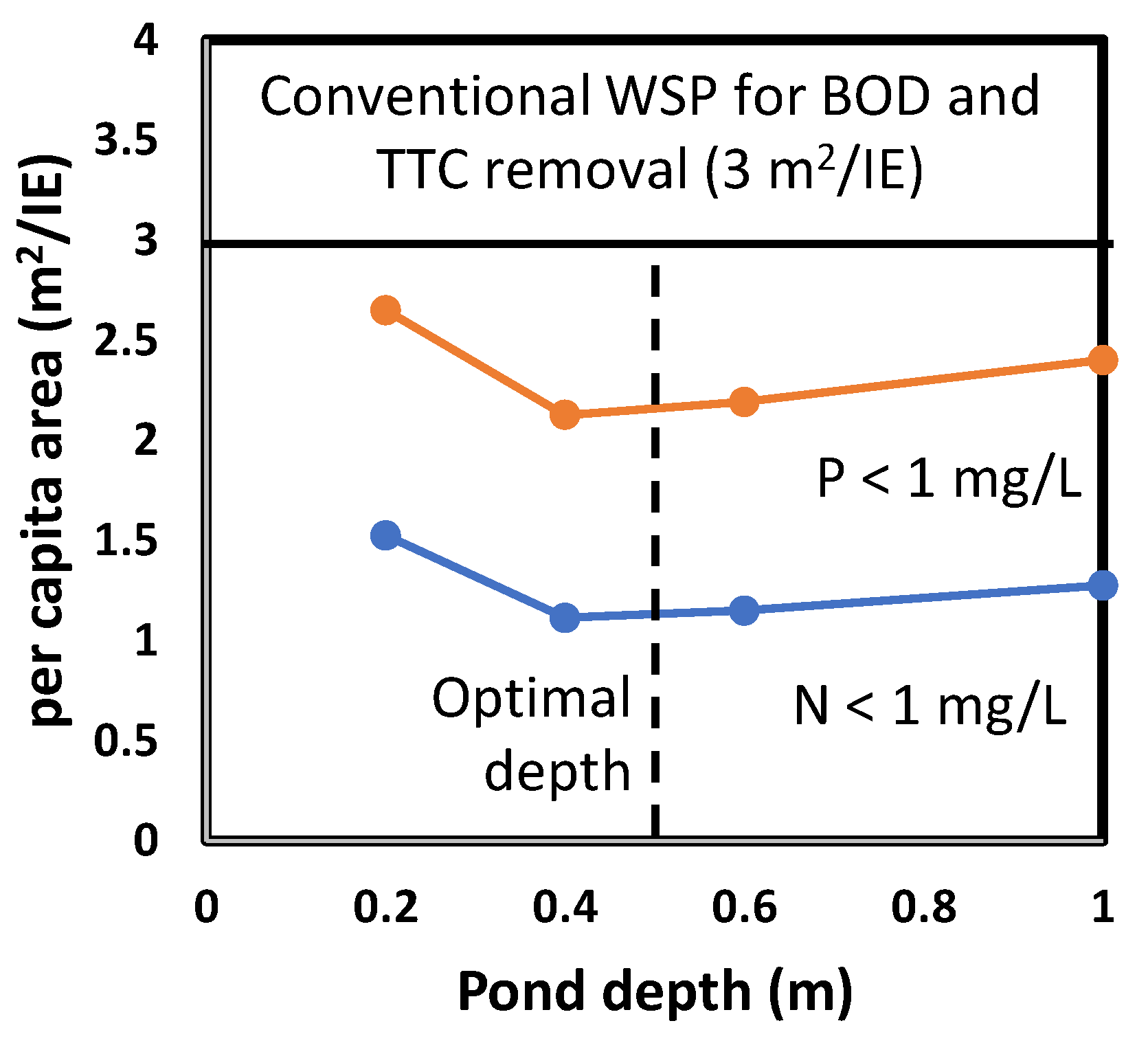
3.4. Per Capita Area
- The experimental data show that over the range of 0.2 to 1.0 m there is little influence of the pond depth on the pond area. The optimal pond depth is about 0.5 m, which is very much smaller than the depth of maturation ponds in WSP where the depth is usually in the range of 1.0 to 1.2 m. The shallow depth of the pond is an important factor in the reduction of construction costs.
- The area of shallow ponds is much smaller than 3 m2 per inhabitant equivalent, normally used for design of conventional WSP, where nutrient removal does not occur.
- The area required for P removal is about double the value for N removal.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Parker, C.D.; Jones, H.L.; Taylor, W.S. Purification of Sewage in Lagoons. Sew. Ind. Waste 1950, 22, 760–775. [Google Scholar]
- Mara, D.D. Proposed design for oxidation ponds in hot climates. J. Environ. Eng. Div. ASCE 1976, 101, 296–300. [Google Scholar] [CrossRef]
- Van Haandel, A.C.; Van der Lubbe, J. Anaerobic Sewage Digestion: Theory and Applications; International Water Association: London, UK, 2019. [Google Scholar]
- Pano, A.E.; Middlebrooks, J. Ammonia Nitrogen Removal in Facultative Wastewater Stabilization Ponds. J. Water Pollut. Control Fed. 1982, 54, 344–351. [Google Scholar] [CrossRef]
- Bastos, R.K.X.; Rios, E.N.E.; Sánchez, I.A. Further contributions to the understanding of nitrogen removal in waste stabilization ponds. Water Sci. Technol. 2018, 77, 2635–2641. [Google Scholar] [CrossRef] [PubMed]
- Zimmo, O.R.; Van Der Steen, N.P.; Gijzen, H.J. Comparison of ammonia volatilization rates in algae and duckweed-based waste stabilization ponds treating domestic wastewater. Water Res. 2003, 37, 4587–4594. [Google Scholar] [CrossRef] [PubMed]
- Camargo Valero, M.A.; Mara, D.D. Ammonia volatilisation in waste stabilisation ponds: A cascade of misinterpretations? Water Sci. Technol. 2010, 61, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Gomez, E.; Paing, J.; Casellas, C.; Picot, B. Characterisation of phosphorus in sediments from waste stabilization ponds. Water Sci. Technol. 2000, 24, 257–264. [Google Scholar] [CrossRef]
- Assunção, F.A.L.; Von Sperling, M. Importance of the ammonia volatilization rates in shallow maturation ponds treating UASB reactor effluent. Water Sci Technol. 2012, 66, 1239–1246. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti, P.F.F. Integrated Application of the UASB Reactor and Ponds for Domestic Sewage Treatment in Tropical Regions. Ph.D. Thesis, University of Wageningen, Wageningen, The Netherlands, 2003. [Google Scholar]
- Kube, M.; Jefferson, B.; Fan, L.; Rodicck, F. The impact of wastewater characteristics, algal species selection and immobilisation on simultaneous nitrogen and phosphorus removal. Algal Res. 2018, 31, 478–488. [Google Scholar] [CrossRef] [Green Version]
- Cai, T.; Park, S.Y.; Li, Y. Nutrient recovery from wastewater streams by microalgae: Status and prospects. Renew. Sustain. Energy Rev. 2013, 19, 360–369. [Google Scholar] [CrossRef]
- Capri, M.G.; Marais, G.V.R. Anaerobic Filter Treatment of Wine Distillery Waste; Research Report W1; Department of Civil Engineering, University of Cape Town: Cape Town, South Africa, 1974. [Google Scholar]
- Albuquerque, M.S.; Santos, S.L.; Van Haandel, A. Influência do regime hidrodinâmico sobre o desempenho de lagoas de polimento. Revista DAE 2021, 69, 25–38. [Google Scholar] [CrossRef]
- American Public Health Association—APHA; American Water Works Association—AWWA; Water Environment Association—WEF. Standard Methods for the Examination of Water and Wastewater, 23th ed.; APHA: Washington, DC, USA, 2017. [Google Scholar]
- Deffeyes, K.S. Carbonate Equilibria: A Graphic and Algebraic Approach. Limnol. Oceanogr. 1965, 66, 412–426. [Google Scholar] [CrossRef]
- Loewenthal, R.E.; Marais, G.v.R. Carbonate Chemistry of Aquatic Systems, Theory and Application; Ann Arbor Science Publishers EUA: Ann Arbor, MI, USA, 2006; ISBN 0-250-40141. [Google Scholar]
- Emerson, K.; Russo, R.C.; Lund, R.E.; Thurston, T.T. Aqueous ammonium calculations: Effect of pH and temperature. JFRBC 1975, 23, 2379–2383. [Google Scholar] [CrossRef]
- Morais, C.E.P.; Santos, S.L.; Van Haandel, A. Determinação de constantes de transferência de compostos voláteis em lagoas de tratamento de esgoto. Revista DAE 2021, 69, 39–51. [Google Scholar] [CrossRef]
- CONAMA (National Committee Board for the Environment). Resolution n° 430/2011; Diário Oficial da União; Imprensa Nacional: São Paulo, Brazil, 2011. [Google Scholar]
| Process | Reaction | ΔAlk (eq·mol−1) | ΔAc (eq·mol−1) | ΔpH |
|---|---|---|---|---|
| Physical CO2 removal | No reaction | 0 | −2 | increase |
| Biological CO2 removal | CO2 + H2O → MO + O2 | 0 | −2 | increase |
| NH3 removal | NH4+ → NH3 + H+ | −1 | +1 | decrease |
| DO production rate | OPR = ΔDO/Δt = −(∆CO2/∆t)f | (23) |
| CO2 desorption rate | rdc = (∆ [CO2]/∆t)d = kdc([CO2] − [CO2]s) | (17) |
| NH3 desorption rate | rdn = (∆ [NH3]/∆t)d = kdn[NH3] | (18) |
| Alkalinity change | ∆Alk/∆t = −kdn[NH3] | (12) |
| Acidity change | ∆Ac/∆t = –2{kdc([CO2] − [CO2]s) − 2OPR + kdn[NH3]} | (13) |
| pH change | pH = −log{(0.5(k1(Ac1/Alk1 − 1)/2 + ((k1(Ac1/Alk1/2)2 − 4k1K2)1/2)} | (16) |
| CO2 conc. change | ∆[CO2]/∆t = −kdc([CO2] − [CO2]s) – OPR | (24) |
| NH3 conc. change | ∆[NH3]/∆t = −kdn[NH3] | (25) |
| Total N conc. change | ∆Ntot/∆t = Ntot − kdn[NH3] | (28) |
| 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|
| Pond Depth | HRT (d) for 1 mgN·L−1 | HRT (d) for 1 mgP·L−1 | Per Capita Area for 1 mgN·L−1 | Per Capita Area for 1 mgP·L−1 |
| L1 = 0.2 m | 3 | 5 | 1.52 | 2.65 |
| L2 = 0.4 m | 4.5 | 8.5 | 1.11 | 2.12 |
| L3 = 0.6 m | 7 | 13 | 1.15 | 2.19 |
| L4 = 1.0 m | 13 | 24 | 1.28 | 2.40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
dos Santos, S.L.; van Haandel, A. Nutrient Removal in Sequential Batch Polishing Ponds. Water 2021, 13, 1584. https://doi.org/10.3390/w13111584
dos Santos SL, van Haandel A. Nutrient Removal in Sequential Batch Polishing Ponds. Water. 2021; 13(11):1584. https://doi.org/10.3390/w13111584
Chicago/Turabian Styledos Santos, Silvânia Lucas, and Adrianus van Haandel. 2021. "Nutrient Removal in Sequential Batch Polishing Ponds" Water 13, no. 11: 1584. https://doi.org/10.3390/w13111584
APA Styledos Santos, S. L., & van Haandel, A. (2021). Nutrient Removal in Sequential Batch Polishing Ponds. Water, 13(11), 1584. https://doi.org/10.3390/w13111584





