Observations of Tidal Flat Sedimentation within a Native and an Exotic Spartina Species
Abstract
1. Introduction
2. Materials and Methods
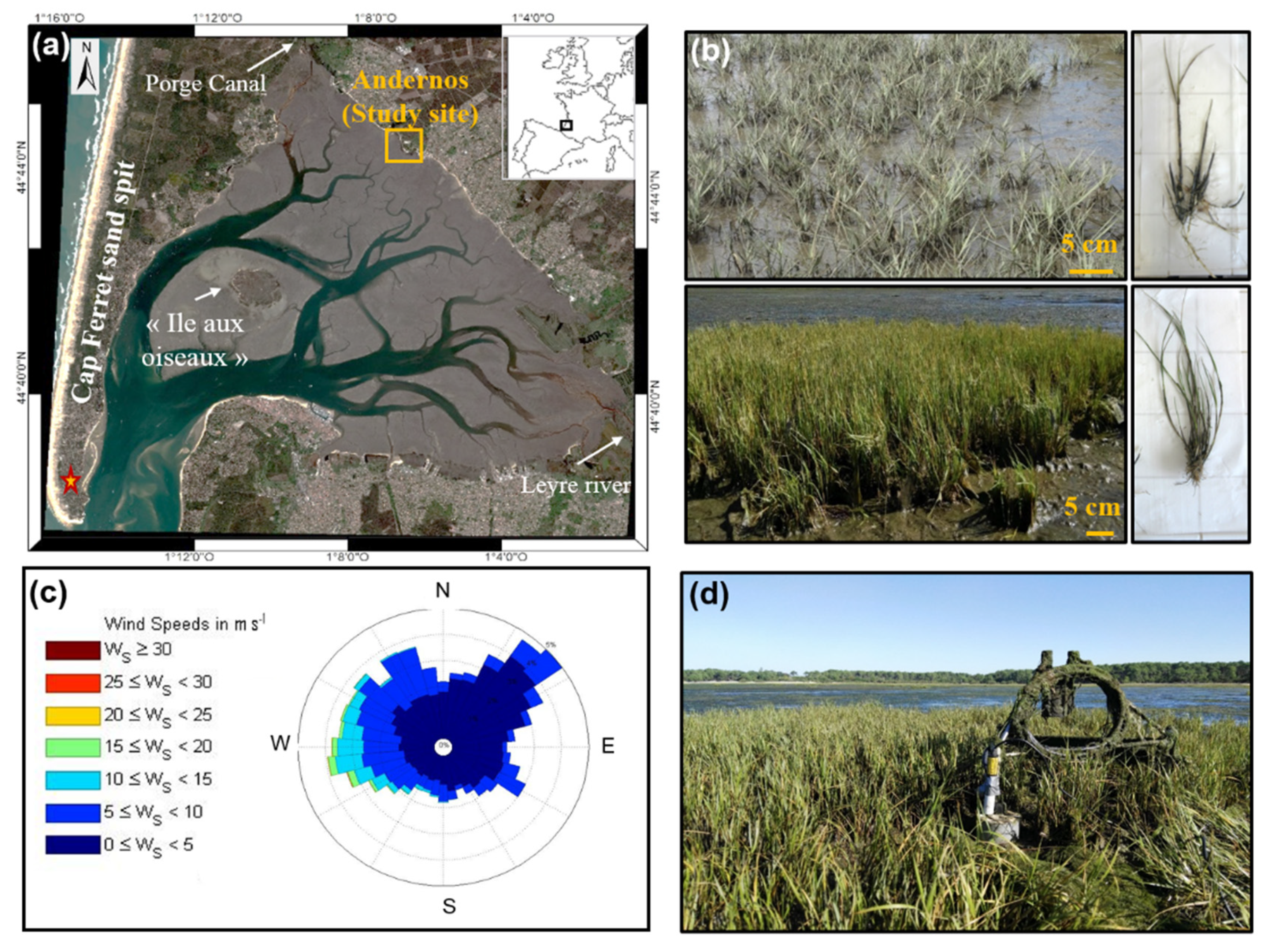
2.1. Study Area
2.2. Vegetation in the Bay of Arcachon
2.3. Sampling and Analysis
3. Results
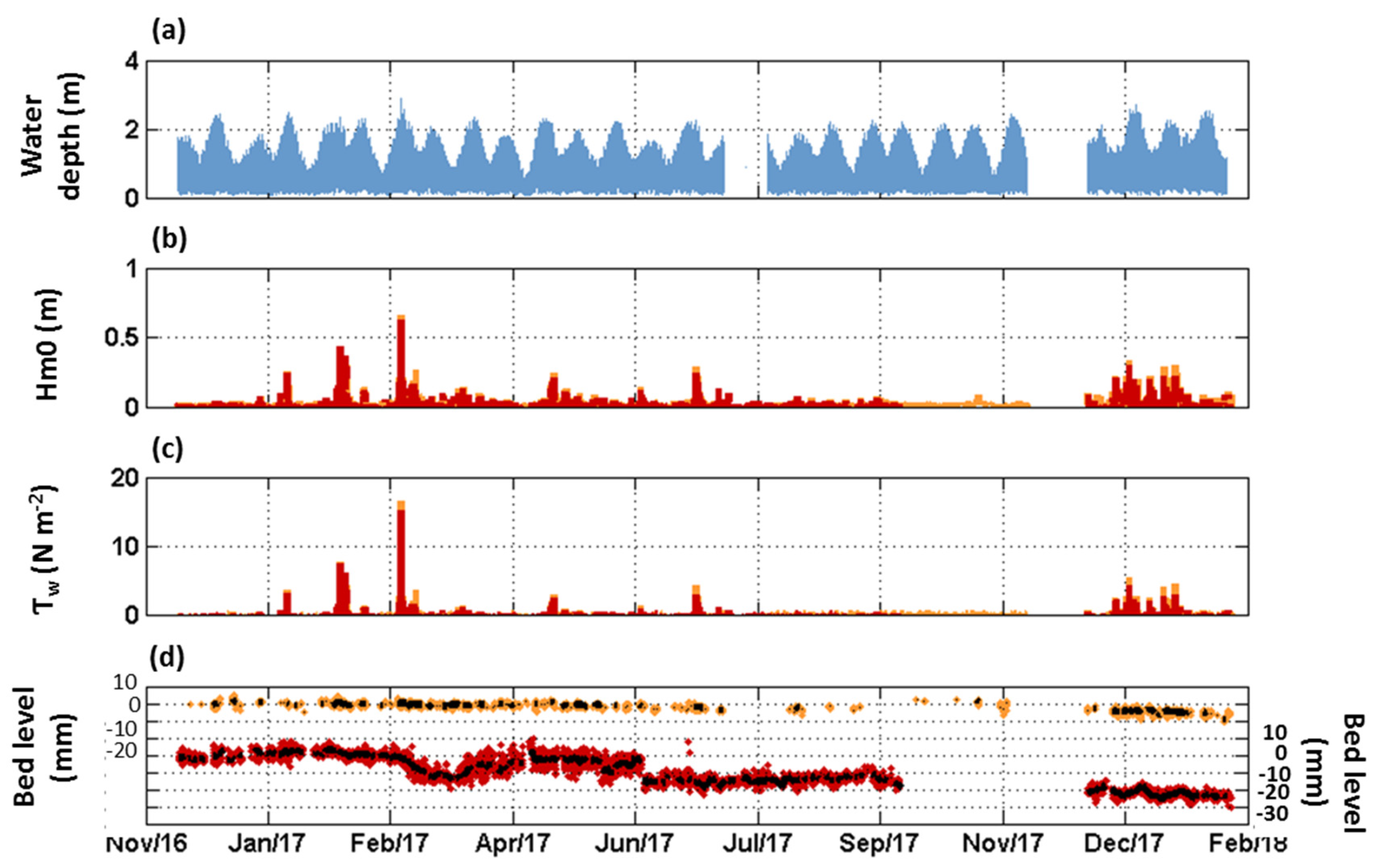
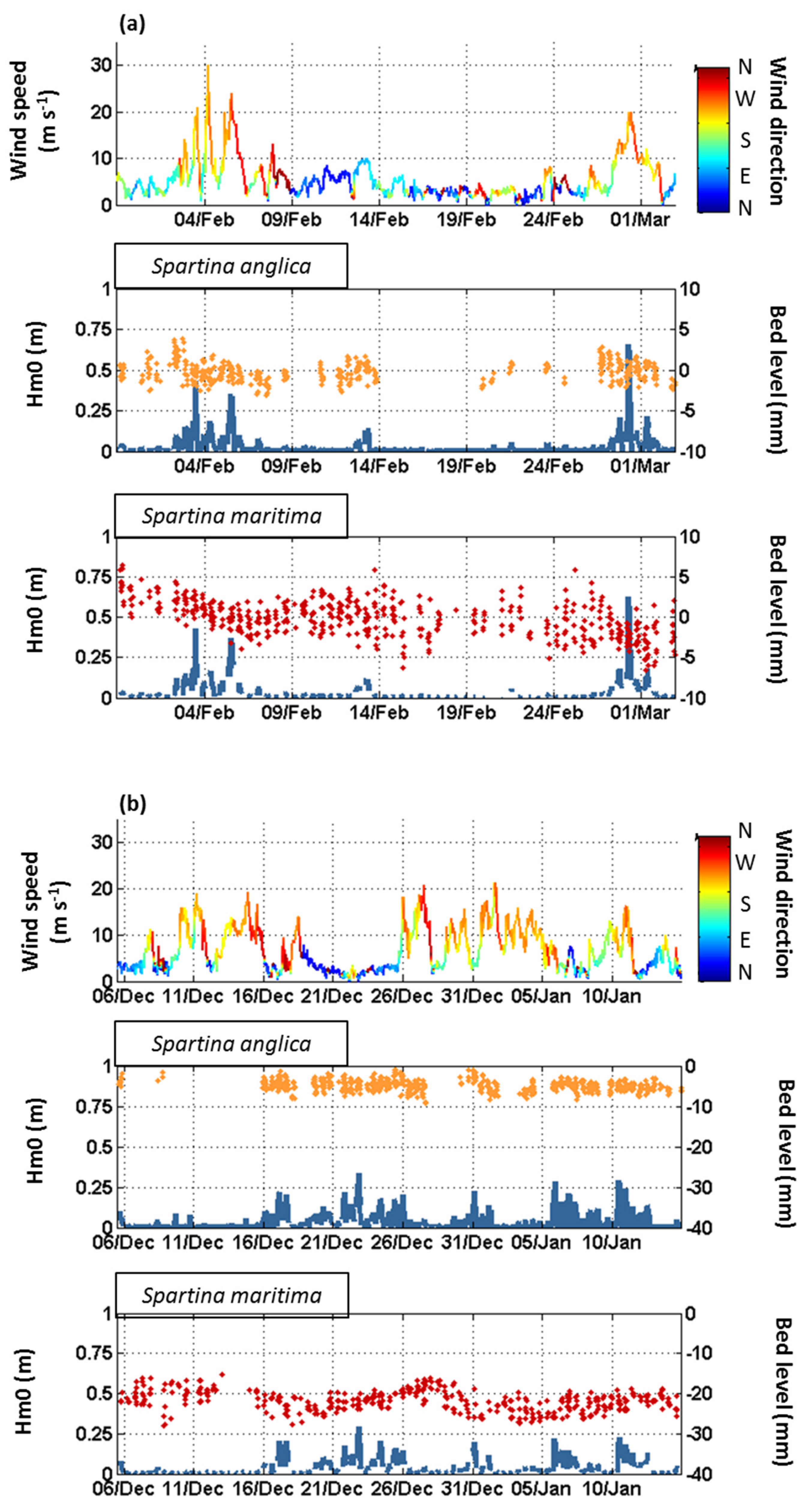
3.1. Seasonal to Event Patterns of Sediment Dynamics within S. anglica and S. maritima
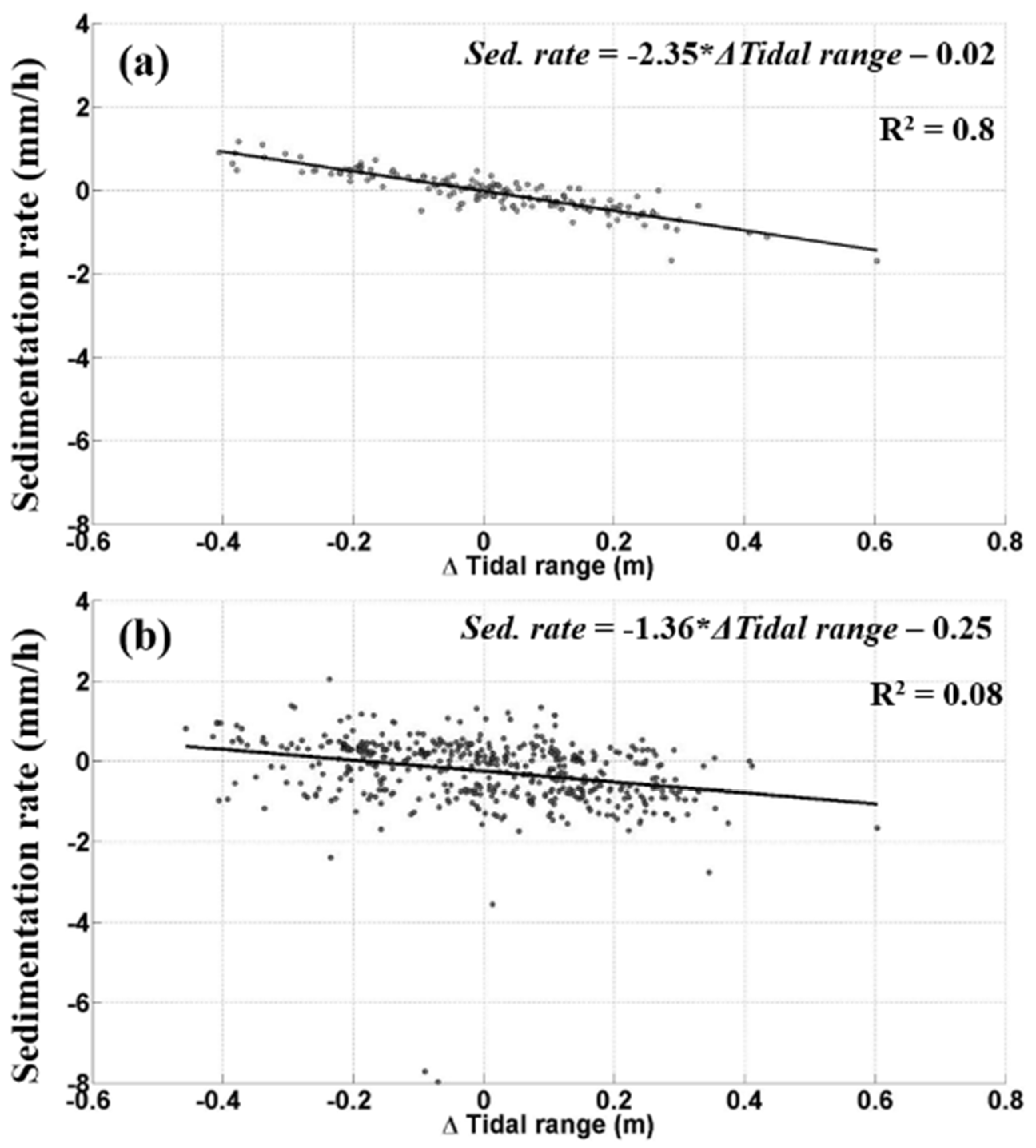
3.2. Tide-Induced Variations in Sediment Dynamics
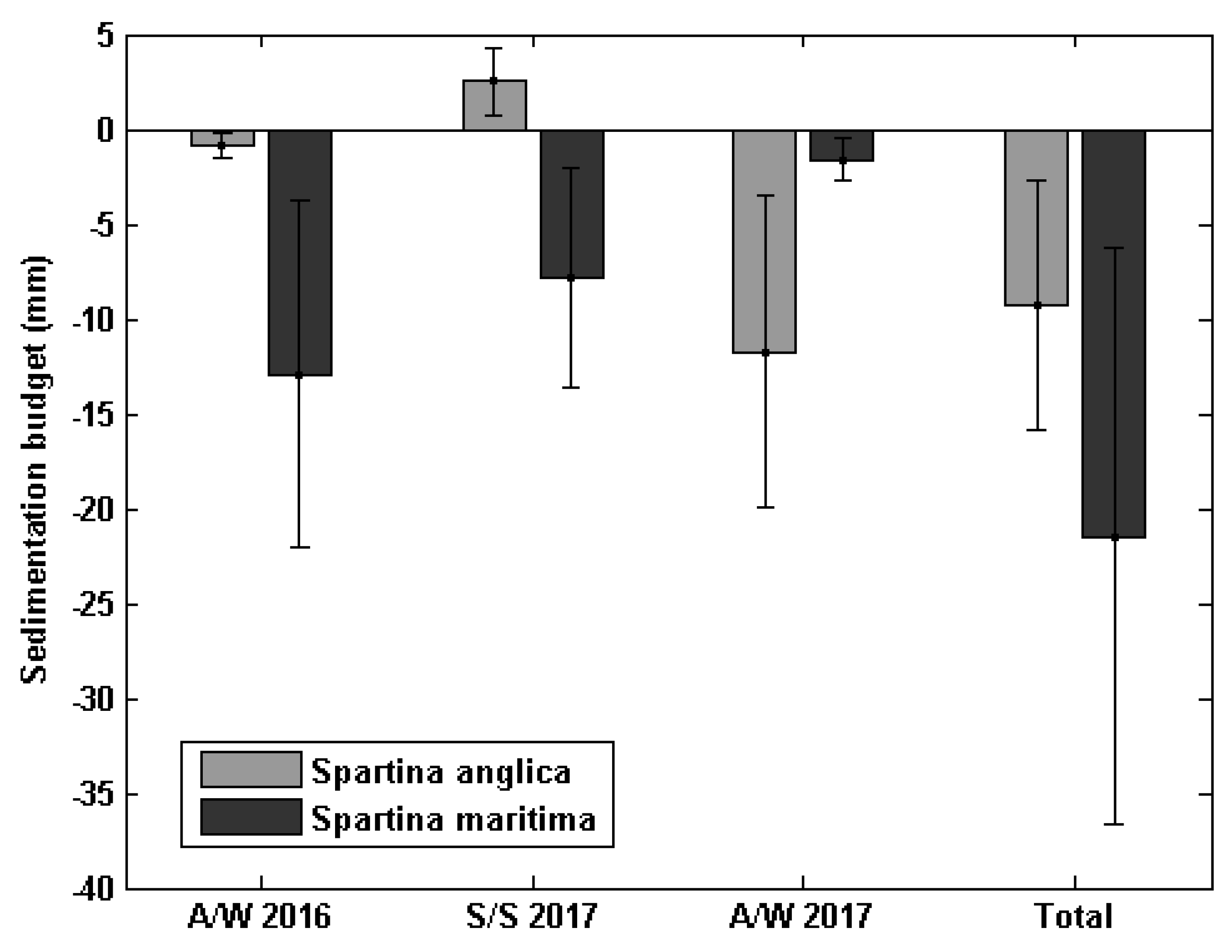
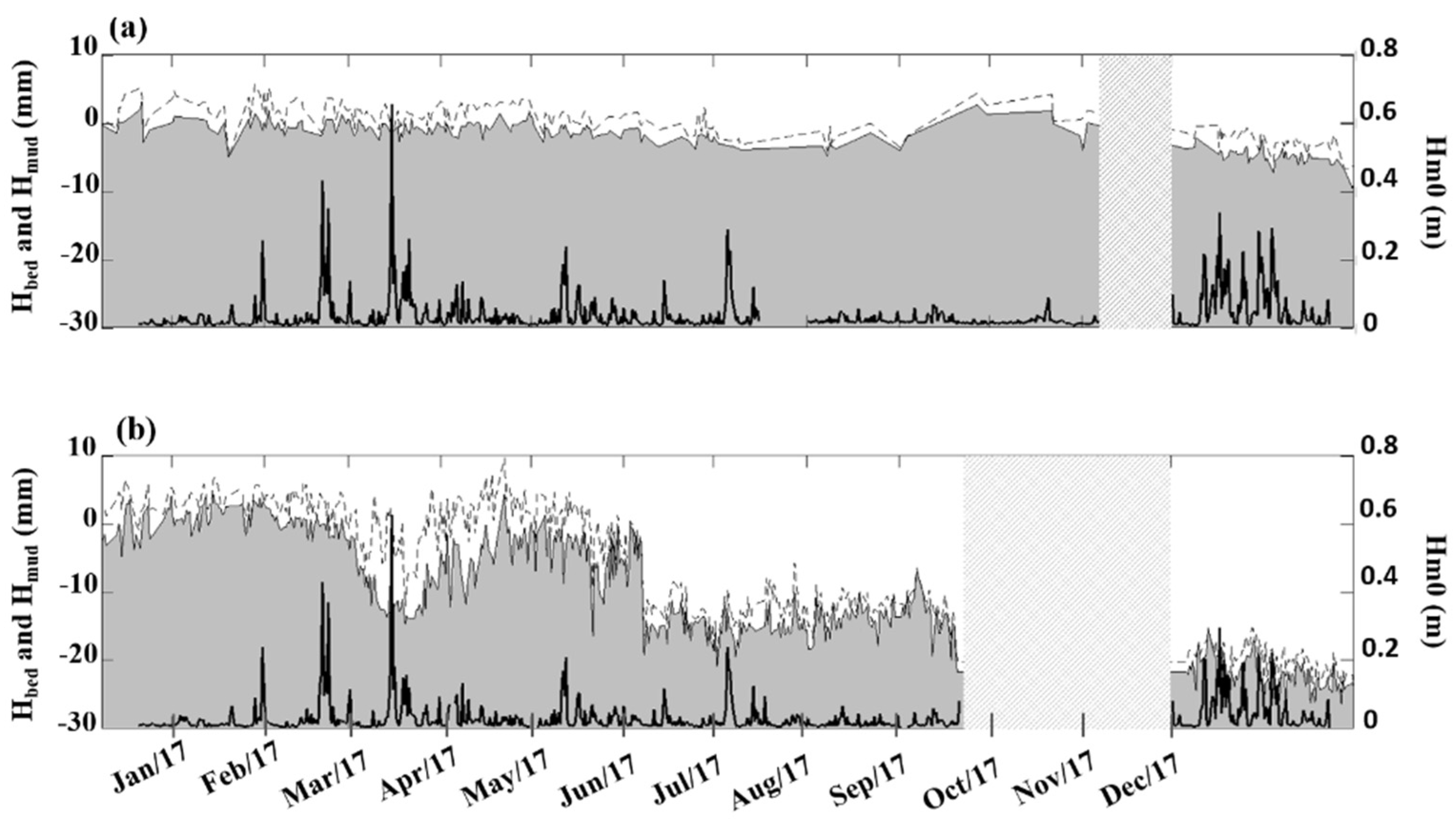
3.3. Annual Bed Level Evolution under Spartina Influence
4. Discussion
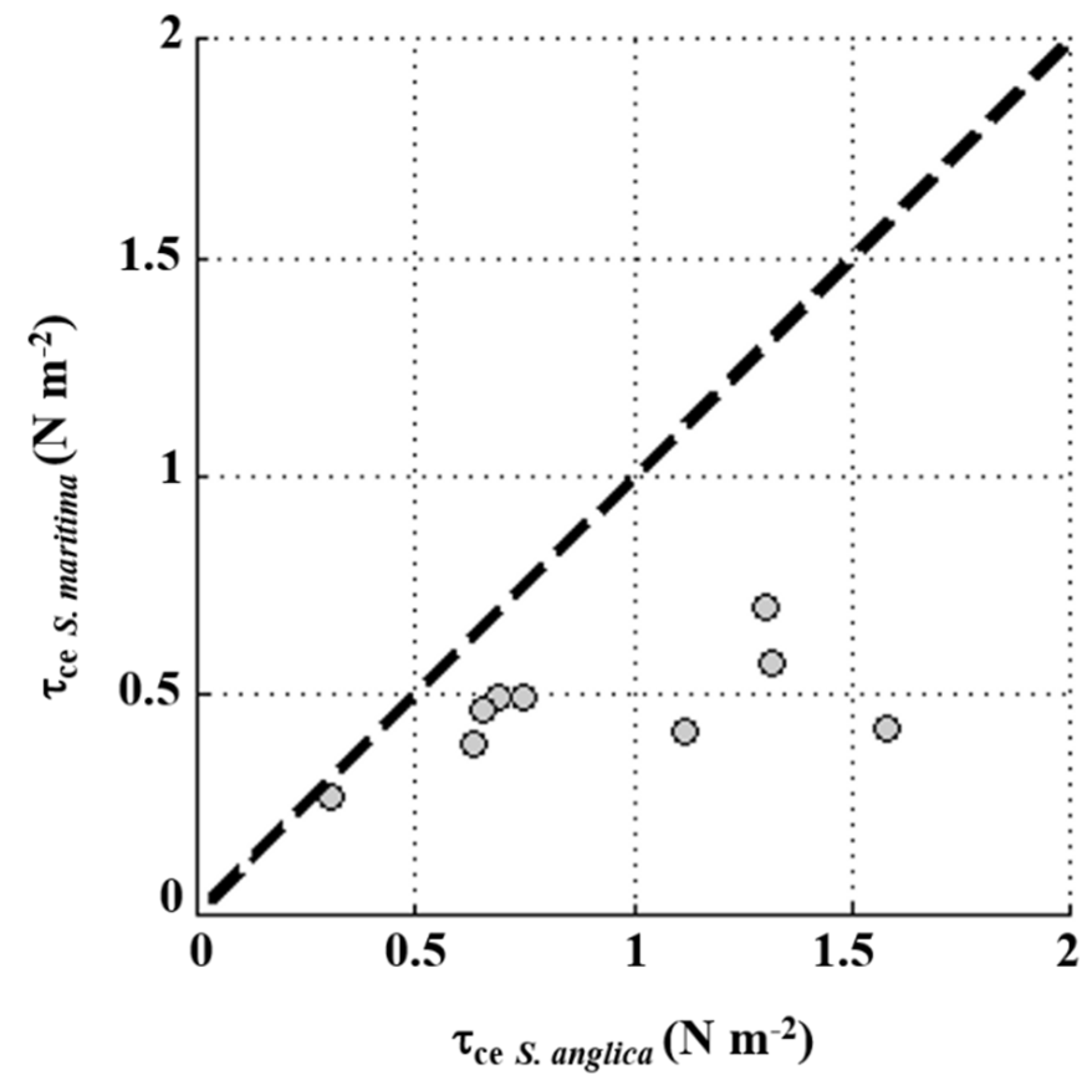
4.1. Sediment Dynamics under the Influence of Waves
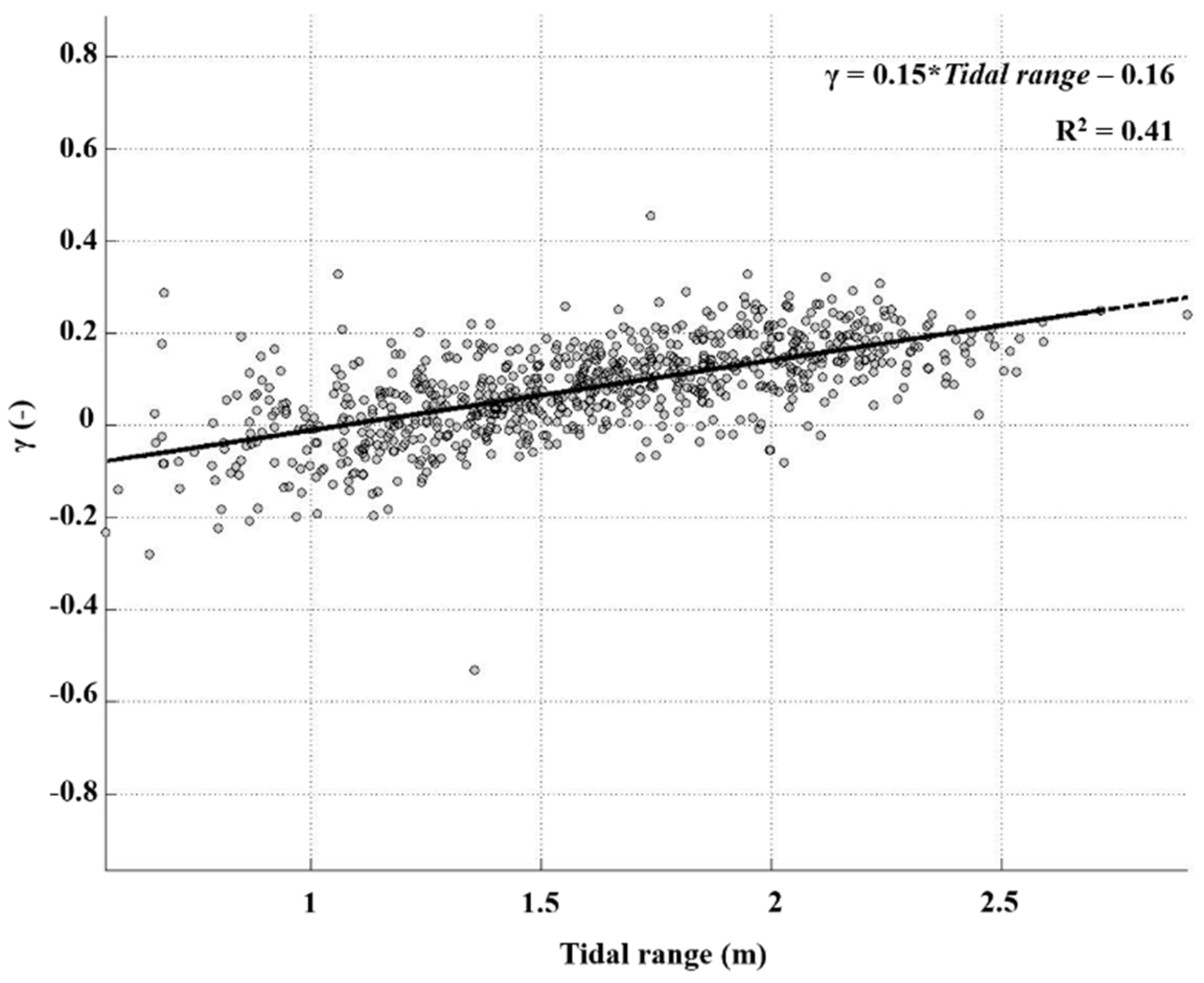
4.2. Tidally Driven Sediment Dynamics
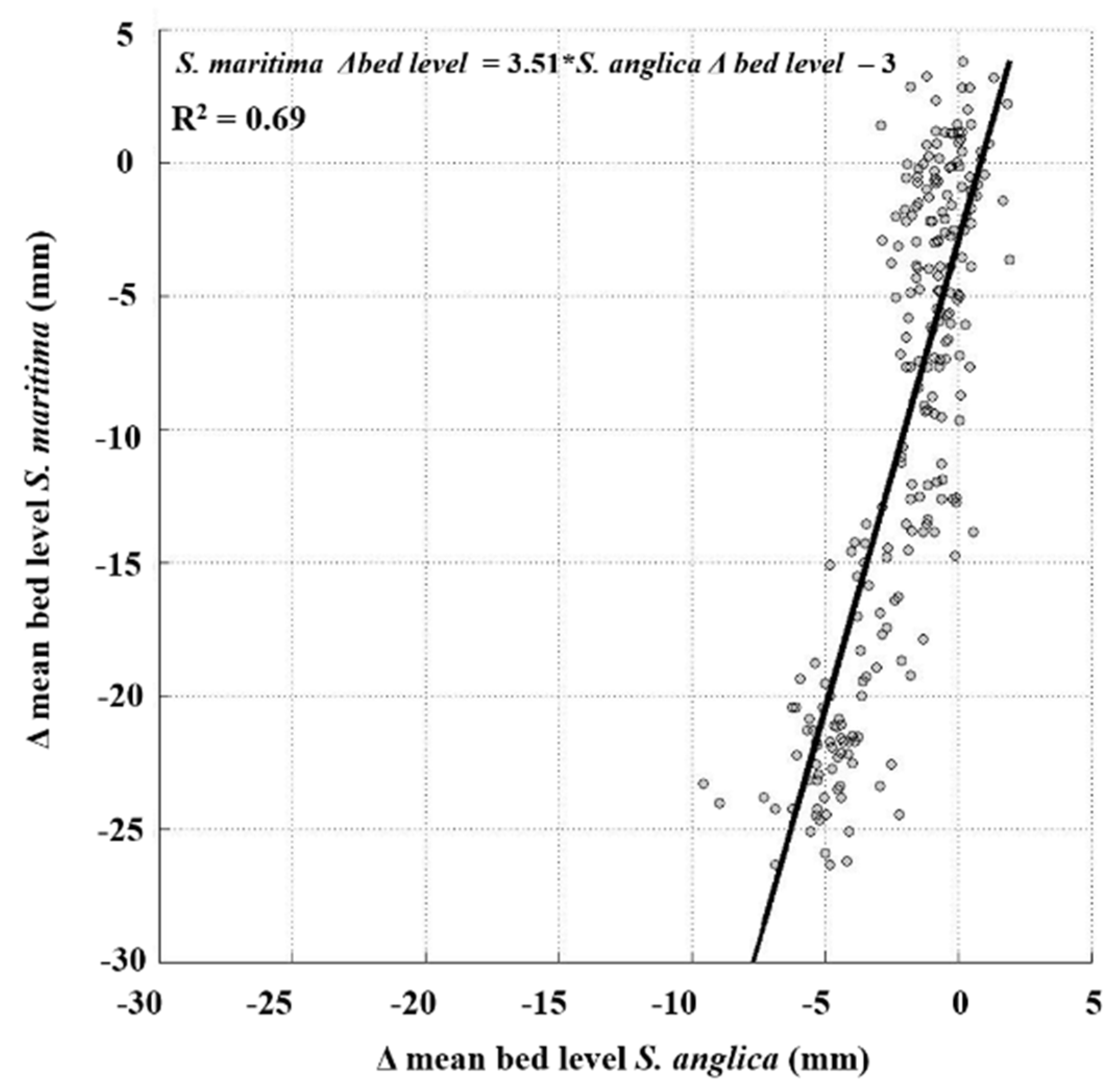
4.3. Long-Term Differences in Bed Level Variation between S. anglica and S. maritima
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- D’Alpaos, A.; Lanzoni, S.; Marani, M.; Rinaldo, A. Landscape evolution in tidal embayments: Modeling the interplay of erosion, sedimentation, and vegetation dynamics. J. Geophys. Res. Space Phys. 2007, 112. [Google Scholar] [CrossRef]
- Kirwan, M.L.; Murray, A.B. A coupled geomorphic and ecological model of tidal marsh evolution. Proc. Natl. Acad. Sci. USA 2007, 104, 6118–6122. [Google Scholar] [CrossRef]
- Temmerman, S.; Bouma, T.; Van De Koppel, J.; Van Der Wal, D.; De Vries, M.; Herman, P. Vegetation causes channel erosion in a tidal landscape. Geology 2007, 35, 631–634. [Google Scholar] [CrossRef]
- Murray, A.B.; Knaapen, M.A.F.; Tal, M.; Kirwan, M.L. Biomorphodynamics: Physical-biological feedbacks that shape landscapes. Water Resour. Res. 2008, 44. [Google Scholar] [CrossRef]
- Bouma, T.; Temmerman, S.; van Duren, L.; Martini, E.; Vandenbruwaene, W.; Callaghan, D.; Balke, T.; Biermans, G.; Klaassen, P.; van Steeg, P.; et al. Organism traits determine the strength of scale-dependent bio-geomorphic feedbacks: A flume study on three intertidal plant species. Geomorphology 2013, 180–181, 57–65. [Google Scholar] [CrossRef]
- Fagherazzi, S.; Mariotti, G.; Wiberg, P.; McGlathery, K. Marsh Collapse Does Not Require Sea Level Rise. Oceanography 2013, 26, 70–77. [Google Scholar] [CrossRef]
- Tinoco, R.O.; Coco, G. Turbulence as the Main Driver of Resuspension in Oscillatory Flow Through Vegetation. J. Geophys. Res. Earth Surf. 2018, 123, 891–904. [Google Scholar] [CrossRef]
- Coco, G.; Zhou, Z.; van Maanen, B.; Olabarrieta, M.; Tinoco, R.; Townend, I. Morphodynamics of tidal networks: Advances and challenges. Mar. Geol. 2013, 346, 1–16. [Google Scholar] [CrossRef]
- Hu, Z.; Willemsen, P.W.J.M.; Borsje, B.W.; Wang, C.; Wang, H.; van der Wal, D.; Zhu, Z.; Oteman, B.; Vuik, V.; Evans, B.; et al. Synchronized high-resolution bed-level change and biophysical data from 10 marsh–mudflat sites in northwestern Europe. Earth Syst. Sci. Data 2021, 13, 405–416. [Google Scholar] [CrossRef]
- Borsje, B.W.; van Wesenbeeck, B.K.; Dekker, F.; Paalvast, P.; Bouma, T.J.; van Katwijk, M.M.; de Vries, M.B. How ecological engineering can serve in coastal protection. Ecol. Eng. 2011, 37, 113–122. [Google Scholar] [CrossRef]
- Temmerman, S.; Meire, P.; Bouma, T.J.; Herman, P.M.J.; Ysebaert, T.; De Vriend, H.J. Ecosystem-based coastal defence in the face of global change. Nature 2013, 504, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Ondiviela, B.; Losada, I.J.; Lara, J.L.; Maza, M.; Galván, C.; Bouma, T.J.; Van Belzen, J. The role of seagrasses in coastal protection in a changing climate. Coast. Eng. 2014, 87, 158–168. [Google Scholar] [CrossRef]
- Barbier, E.B.; Hacker, S.D.; Kennedy, C.; Koch, E.W.; Stier, A.C.; Silliman, B.R. The value of estuarine and coastal ecosystem services. Ecol. Monogr. 2011, 81, 169–193. [Google Scholar] [CrossRef]
- Bouma, T.J.; De Vries, M.B.; Low, E.; Peralta, G.; Tánczos, I.C.; Van De Koppel, J.; Herman, P.M.J. Trade-offs related to ecosystem engineering: A case study on stiffness of emerging macrophytes. Ecology 2005, 86, 2187–2199. [Google Scholar] [CrossRef]
- Bouma, T.J.; van Duren, L.A.; Temmerman, S.; Claverie, T.; Blanco-Garcia, A.; Ysebaert, T.; Herman, P.M.J. Spatial flow and sedimentation patterns within patches of epibenthic structures: Combining field, flume and modelling experiments. Cont. Shelf Res. 2007, 27, 1020–1045. [Google Scholar] [CrossRef]
- Neumeier, U.; Amos, C.L. The influence of vegetation on turbulence and flow velocities in European salt-marshes. Sedimentology 2006, 53, 259–277. [Google Scholar] [CrossRef]
- Nepf, H.M. Flow and Transport in Regions with Aquatic Vegetation. Annu. Rev. Fluid Mech. 2012, 44, 123–142. [Google Scholar] [CrossRef]
- Ganthy, F.; Soissons, L.; Sauriau, P.-G.; Verney, R.; Sottolichio, A. Effects of short flexible seagrass Zostera noltei on flow, erosion and deposition processes determined using flume experiments. Sedimentology 2015, 62, 997–1023. [Google Scholar] [CrossRef]
- Maza, M.; Lara, J.; Losada, I.; Ondiviela, B.; Trinogga, J.; Bouma, T. Large-scale 3-D experiments of wave and current interaction with real vegetation. Part 2: Experimental analysis. Coast. Eng. 2015, 106, 73–86. [Google Scholar] [CrossRef]
- Weitzman, J.S.; Zeller, R.B.; Thomas, F.I.M.; Koseff, J.R. The attenuation of current- and wave-driven flow within submerged multispecific vegetative canopies. Limnol. Oceanogr. 2015, 60, 1855–1874. [Google Scholar] [CrossRef]
- Losada, I.J.; Maza, M.; Lara, J.L. A new formulation for vegetation-induced damping under combined waves and currents. Coast. Eng. 2016, 107, 1–13. [Google Scholar] [CrossRef]
- Nowacki, D.J.; Beudin, A.; Ganju, N.K. Spectral wave dissipation by submerged aquatic vegetation in a back-barrier estuary. Limnol. Oceanogr. 2017, 62, 736–753. [Google Scholar] [CrossRef]
- Horstman, E.M.; Dohmen-Janssen, C.M.; Hulscher, S.J. Flow routing in mangrove forests: A field study in Trang province, Thailand. Cont. Shelf Res. 2013, 71, 52–67. [Google Scholar] [CrossRef]
- Neumeier, U. Velocity and turbulence variations at the edge of saltmarshes. Cont. Shelf Res. 2007, 27, 1046–1059. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Y.; Cai, T.; Thompson, C.; Li, Y. A comparison of biohydrodynamic interaction within mangrove and saltmarsh boundaries. Earth Surf. Process. Landf. 2016, 41, 1967–1979. [Google Scholar] [CrossRef]
- Temmerman, S.; Bouma, T.J.; Govers, G.; Wang, Z.B.; De Vries, M.; Herman, P.M.J. Impact of vegetation on flow routing and sedimentation patterns: Three-dimensional modeling for a tidal marsh. J. Geophys. Res. Earth Surf. 2005, 110. [Google Scholar] [CrossRef]
- Chen, Y.; Cai, T.; Chang, Y.; Huang, S.; Xia, T. Comparison of Flow and Energy Reduction by Representative Intertidal Plants, Southeast China. In Proceedings of the Twenty-Eighth (2018) International Ocean and Polar Engineering Conference, Sapporo, Japan, 15 June 2018. [Google Scholar]
- Bernik, B.M.; Eppinga, M.B.; Kolker, A.S.; Blum, M.J. Clonal Vegetation Patterns Mediate Shoreline Erosion. Geophys. Res. Lett. 2018, 45, 6476–6484. [Google Scholar] [CrossRef]
- Peralta, G.; Van Duren, L.; Morris, E.; Bouma, T. Consequences of shoot density and stiffness for ecosystem engineering by benthic macrophytes in flow dominated areas: A hydrodynamic flume study. Mar. Ecol. Prog. Ser. 2008, 368, 103–115. [Google Scholar] [CrossRef]
- Bouma, T.J.; Ortells, V.; Ysebaert, T. Comparing biodiversity effects among ecosystem engineers of contrasting strength: Macrofauna diversity in Zostera noltii and Spartina anglica vegetations. Helgol. Mar. Res. 2009, 63, 3–18. [Google Scholar] [CrossRef]
- Green, M.O.; Coco, G. Review of wave-driven sediment resuspension and transport in estuaries-. Rev. Geophys. 2014, 52, 77–117. [Google Scholar] [CrossRef]
- Balke, T.; Klaassen, P.C.; Garbutt, A.; van der Wal, D.; Herman, P.M.; Bouma, T.J. Conditional outcome of ecosystem engineering: A case study on tussocks of the salt marsh pioneer Spartina anglica. Geomorphology 2012, 153–154, 232–238. [Google Scholar] [CrossRef]
- Fagherazzi, S.; Kirwan, M.L.; Mudd, S.M.; Guntenspergen, G.R.; Temmerman, S.; D’Alpaos, A.; Van De Koppel, J.; Rybczyk, J.M.; Reyes, E.; Craft, C.B.; et al. Numerical models of salt marsh evolution: Ecological, geomorphic, and climatic factors. Rev. Geophys. 2012, 50. [Google Scholar] [CrossRef]
- Kirwan, M.L.; Megonigal, J.P. Tidal wetland stability in the face of human impacts and sea-level rise. Nature 2013, 504, 53–60. [Google Scholar] [CrossRef]
- Friedrichs, C.T.; Perry, J.E. Tidal Salt Marsh Morphodynamics: A Synthesis. J. Coast. Res. 2001, SI 27, 7–37. [Google Scholar]
- Coops, H.; Geilen, N.; Verheij, H.J.; Boeters, R.; Van Der Velde, G. Interactions between waves, bank erosion and emergent vegetation: An experimental study in a wave tank. Aquat. Bot. 1996, 53, 187–198. [Google Scholar] [CrossRef]
- Schwarz, M.; Lehmann, P.; Or, D. Quantifying lateral root reinforcement in steep slopes—From a bundle of roots to tree stands. Earth Surf. Process. Landf. 2010, 35, 354–367. [Google Scholar] [CrossRef]
- Twomey, A.J.; Saunders, M.I.; Callaghan, D.P.; Bouma, T.J.; Han, Q.; O’Brien, K.R. Lateral sediment erosion with and without the non-dense root-mat forming seagrass Enhalus acoroides. Estuarine Coast. Shelf Sci. 2021, 253, 107316. [Google Scholar] [CrossRef]
- Vannoppen, W.; Poesen, J.; Peeters, P.; De Baets, S.; Vandevoorde, B. Root properties of vegetation communities and their impact on the erosion resistance of river dikes. Earth Surf. Process. Landf. 2016, 41, 2038–2046. [Google Scholar] [CrossRef]
- Ganthy, F.; Sottolichio, A.; Verney, R. The Stability of Vegetated Tidal Flats in a Coastal Lagoon Through Quasi In-Situ Measurements of Sediment Erodibility. J. Coast. Res. 2011, SI 64, 1500–1504. [Google Scholar]
- Willemsen, P.W.J.M.; Borsje, B.W.; Hulscher, S.J.M.H.; Van Der Wal, D.; Zhu, Z.; Oteman, B.; Evans, B.; Möller, I.; Bouma, T.J. Quantifying Bed Level Change at the Transition of Tidal Flat and Salt Marsh: Can We Understand the Lateral Location of the Marsh Edge? J. Geophys. Res. Earth Surf. 2018, 123, 2509–2524. [Google Scholar] [CrossRef]
- Baumel, A.; Ainouche, M.L.; Levasseur, J.E. Molecular investigations in populations of Spartina anglica C.E. Hubbard (Poaceae) invading coastal Brittany (France). Mol. Ecol. 2001, 10, 1689–1701. [Google Scholar] [CrossRef]
- Proença, B.; Nez, T.; Poli, A.; Ciutat, A.; Devaux, L.; Sottolichio, A.; Montaudouin, X.; Michalet, R. Intraspecific facilitation explains the spread of the invasive engineer Spartina anglica in Atlantic salt marshes. J. Veg. Sci. 2019, 30, 212–223. [Google Scholar] [CrossRef]
- Bouma, T.J.; Van Belzen, J.; Balke, T.; Van Dalen, J.; Klaassen, P.; Hartog, A.M.; Callaghan, D.P.; Hu, Z.; Stive, M.J.F.; Temmerman, S.; et al. Short-term mudflat dynamics drive long-term cyclic salt marsh dynamics: Lateral Salt Marsh Dynamics. Limnol. Oceanogr. 2016, 61, 2261–2275. [Google Scholar] [CrossRef]
- Plus, M.; Dumas, F.; Stanisière, J.-Y.; Maurer, D. Hydrodynamic characterization of the Arcachon Bay, using model-derived descriptors. Cont. Shelf Res. 2009, 29, 1008–1013. [Google Scholar] [CrossRef]
- Allard, J.; Chaumillon, E.; Fenies, H. A synthesis of morphological evolutions and Holocene stratigraphy of a wave-dominated estuary: The Arcachon lagoon, SW France. Cont. Shelf Res. 2009, 29, 957–969. [Google Scholar] [CrossRef]
- Parisot, J.-P.; Diet-Davancens, J.; Sottolichio, A.; Crosland, E.; Drillon, C.; Verney, R. Modélisation des agitations dans le bassin d’Arcachon. Xèmes Journées Nationales Génie Côtier—Génie Civil JNGCGC 2008, 435–444. [Google Scholar] [CrossRef]
- Cayocca, F. Long-term morphological modeling of a tidal inlet: The Arcachon Basin, France. Coast. Eng. 2001, 42, 115–142. [Google Scholar] [CrossRef]
- Salles, P.; Sottolichio, A.; Sénéchal, N.; Valle-Levinson, A. Wind-driven modifications to the residual circulation in an ebb-tidal delta: Arcachon Lagoon, Southwestern France-Salles-2015. J. Geophys. Res. Oceans 2015, 120, 728–740. [Google Scholar] [CrossRef]
- Ganthy, F.; Sottolichio, A.; Verney, R. Seasonal modification of tidal flat sediment dynamics by seagrass meadows of Zostera noltii (Bassin d’Arcachon, France). J. Mar. Syst. 2013, 109–110, S233–S240. [Google Scholar] [CrossRef]
- Martin, P.; Sébastien, D.; Gilles, T.; Auby, I.; de Montaudouin, X.; Emery, É.; Claire, N.; Christophe, V. Long-term evolution (1988–2008) of Zostera spp. meadows in Arcachon Bay (Bay of Biscay). Estuar. Coast. Shelf Sci. 2010, 87, 357–366. [Google Scholar] [CrossRef]
- Blanchet, H.; De Montaudouin, X.; Chardy, P.; Bachelet, G. Structuring factors and recent changes in subtidal macrozoobenthic communities of a coastal lagoon, Arcachon Bay (France). Estuar. Coast. Shelf Sci. 2005, 64, 561–576. [Google Scholar] [CrossRef]
- Kombiadou, K.; Ganthy, F.; Verney, R.; Plus, M.; Sottolichio, A. Modelling the effects of Zostera noltei meadows on sediment dynamics: Application to the Arcachon lagoon. Ocean Dyn. 2014, 64, 1499–1516. [Google Scholar] [CrossRef]
- Costanza, R.; Pérez-Maqueo, O.; Martinez, M.L.; Sutton, P.; Anderson, S.J.; Mulder, K. The value of coastal wetlands for hurricane protection. AMBIO 2008, 37, 241–248. [Google Scholar] [CrossRef]
- Cognat, M.; Ganthy, F.; Auby, I.; Barraquand, F.; Rigouin, L.; Sottolichio, A. Environmental factors controlling biomass development of seagrass meadows of Zostera noltei after a drastic decline (Arcachon Bay, France). J. Sea Res. 2018, 140, 87–104. [Google Scholar] [CrossRef]
- Strong, D.R.; Ayres, D.R. Ecological and Evolutionary Misadventures of Spartina. Annu. Rev. Ecol. Evol. Syst. 2013, 44, 389–410. [Google Scholar] [CrossRef]
- Proença, B.; Romuald, M.; Auby, I.; Ganthy, F.; Sottolichio, A.; Michalet, R. Disentangling ecosystem engineering from short-term biotic effects of a strong invader on a native foundation species. Mar. Ecol. Prog. Ser. 2019, 621, 69–81. [Google Scholar] [CrossRef]
- Bassoullet, P.; Le Hir, P.; Gouleau, D.; Robert, S. Sediment transport over an intertidal mudflat: Field investigations and estimation of fluxes within the “Baie de Marenngres-Oleron” (France). Cont. Shelf Res. 2000, 20, 1635–1653. [Google Scholar] [CrossRef]
- Jestin, H.; Bassoulet, P.; Le Hir, P.; L’Yavanc, J.; Degres, Y. Development of ALTUS, A High Frequency Acoustic Submersible Recording Altimeter to Accurately Monitor Bed Elevation and Quantify Deposition or Erosion of Sediments. In Proceedings of the IEEE Oceanic Engineering Society. OCEANS’98. Conference Proceedings (Cat. No.98CH36259), Nice, France, 28 September–1 October 1998; IEEE: Nice, France, 1998. [Google Scholar]
- Cea, L.; Puertas, J.; Pena, L. Velocity measurements on highly turbulent free surface flow using ADV. Exp. Fluids 2007, 42, 333–348. [Google Scholar] [CrossRef]
- Nidzieko, N.J.; Ralston, D.K. Tidal asymmetry and velocity skew over tidal flats and shallow channels within a macrotidal river delta. J. Geophys. Res. Ocean. 2012, 117. [Google Scholar] [CrossRef]
- Soulsby, R.L. Dynamics of Marine Sands; Thomas Telford: London, UK, 1997. [Google Scholar]
- Soulsby, R.L. Modelling of Coastal Processes; Thomas Telford: London, UK, 1993; pp. 111–118. [Google Scholar]
- Ariathurai, R.; Arulanandan, K. Erosion Rates of Cohesive Soils. J. Hydraul. Div. 1978, 104, 279–283. [Google Scholar] [CrossRef]
- Mengual, B.; Le Hir, P.; Cayocca, F.; Garlan, T. Modelling Fine Sediment Dynamics: Towards a Common Erosion Law for Fine Sand, Mud and Mixtures. Water 2017, 9, 564. [Google Scholar] [CrossRef]
- Partheniades, E. Erosion and Deposition of Cohesive Soils. J. Hydraul. Div. (ASCE) 1965, 91, 105–139. [Google Scholar] [CrossRef]
- Hu, Z.; Wang, Z.B.; Zitman, T.J.; Stive, M.J.F.; Bouma, T.J. Predicting long-term and short-term tidal flat morphodynamics using a dynamic equilibrium theory: Modeling Tidal Flat Dynamic Equilibrium. J. Geophys. Res. Earth Surf. 2015, 120, 1803–1823. [Google Scholar] [CrossRef]
- Belliard, J.-P.; Silinski, A.; Meire, D.; Kolokythas, G.; Levy, Y.; Van Braeckel, A.; Bouma, T.J.; Temmerman, S. High-resolution bed level changes in relation to tidal and wave forcing on a narrow fringing macrotidal flat: Bridging intra-tidal, daily and seasonal sediment dynamics. Mar. Geol. 2019, 412, 123–138. [Google Scholar] [CrossRef]
- Forsberg, P.; Ernstsen, V.; Andersen, T.; Winter, C.; Becker, M.; Kroon, A. The effect of successive storm events and seagrass coverage on sediment suspension in a coastal lagoon. Estuar. Coast. Shelf Sci. 2018, 212, 329–340. [Google Scholar] [CrossRef]
- Kirwan, M.L.; Guntenspergen, G.R.; D’Alpaos, A.; Morris, J.T.; Mudd, S.M.; Temmerman, S. Limits on the adaptability of coastal marshes to rising sea level. Geophys. Res. Lett. 2010, 37. [Google Scholar] [CrossRef]
- Escapa, M.; Perillo, G.M.; Iribarne, O. Sediment dynamics modulated by burrowing crab activities in contrasting SW Atlantic intertidal habitats. Estuar. Coast. Shelf Sci. 2008, 80, 365–373. [Google Scholar] [CrossRef]
- Nyman, J.A.; Walters, R.J.; Delaune, R.D.; Patrick, W.H. Marsh vertical accretion via vegetative growth. Estuar. Coast. Shelf Sci. 2006, 69, 370–380. [Google Scholar] [CrossRef]
- Thompson, J.D. The Biology of an Invasive Plant. What Makes Spartina Anglica so Successful? BioScience 1991, 41, 393–401. [Google Scholar] [CrossRef]
- Castellanos, E.M.; Figueroa, M.E.; Davy, A.J. Nucleation and Facilitation in Saltmarsh Succession: Interactions between Spartina Maritima and Arthrocnemum Perenne. J. Ecol. 1994, 82, 239–248. [Google Scholar] [CrossRef]
- Sanchez, J.; SanLeon, D.; Izco, J. Primary colonisation of mudflat estuaries by Spartina maritima (Curtis) Fernald in Northwest Spain: Vegetation structure and sediment accretion. Aquat. Bot. 2001, 69, 15–25. [Google Scholar] [CrossRef]
- Lauzon, R.; Murray, A.B.; Moore, L.J.; Walters, D.C.; Kirwan, M.L.; Fagherazzi, S. Effects of Marsh Edge Erosion in Coupled Barrier Island-Marsh Systems and Geometric Constraints on Marsh Evolution. J. Geophys. Res. Earth Surf. 2018, 123, 1218–1234. [Google Scholar] [CrossRef]
- Nahon, A.; Idier, D.; Sénéchal, N.; Féniès, H.; Mallet, C.; Mugica, J. Imprints of wave climate and mean sea level variations in the dynamics of a coastal spit over the last 250 years: Cap Ferret, SW France. Earth Surf. Process. Landf. 2019, 44, 2112–2125. [Google Scholar] [CrossRef]
- Schoutens, K.; Heuner, M.; Minden, V.; Ostermann, T.S.; Silinski, A.; Belliard, J.-P.; Temmerman, S. How effective are tidal marshes as nature-based shoreline protection throughout seasons? Limnol. Oceanogr. 2019, 64, 1750–1762. [Google Scholar] [CrossRef]
- Reguero, B.G.; Losada, I.J.; Méndez, F.J. A recent increase in global wave power as a consequence of oceanic warming. Nat. Commun. 2019, 10, 205. [Google Scholar] [CrossRef]
- Doody, J.P. ‘Coastal Squeeze’—An Historical Perspective. J. Coast. Conserv. 2004, 10, 129–138. [Google Scholar] [CrossRef]
| Spartina anglica | Spartina maritima | ||||
|---|---|---|---|---|---|
| Parameters | Autumn/Winter | Spring/Summer | Autumn/Winter | Spring/Summer | |
| Plant properties | Patch density (n° individuals m−2 ±SE) | 39 (±8) | 63 (±13) | 124 (±16) | 130 (±24) |
| Plant height (cm) | 15 | 15 | 20 | 20 | |
| Sediment properties | Water content (%) | 188 (±60) | 216 (±23) | 224 (±20) | 218 (±15) |
| ρ dry (Kg m−3) | 505 (±152) | 391 (±34) | 380 (±29) | 384 (±9) | |
| ρ bulk (Kg m−3) | 1363 (±90) | 1229 (±19) | 1226 (±25) | 1220 (±14) | |
| Porosity (−) | 0.86 (±0.06) | 0.84 (±0.02) | 0.85 (±0.02) | 0.84 (±0.01) | |
| D50 (μm) | 23.1 (±7.5) | 28.5 (±3.1) | 18.3 (±1.9) | 28.7 (±3.18) | |
| Fine sed. fraction (mud and silt %) | 81.3 (±11) | 73.6 (±4.7) | 87.5 (±1.9) | 74.7 (±4.4) | |
| Sand fraction (%) | 18.7 (±11) | 26.4 (±4.7) | 12.5 (±1.9) | 25.3 (±4.4) | |
| Hydrodynamics | Hm0 (min, max) | <0.05, 0.65 | <0.05, 0.34 | <0.05, 0.63 | <0.05, 0.29 |
| Hmax (min, max) | 0.06, 0.85 | 0.06, 0.38 | 0.05, 0.86 | 0.06, 0.33 | |
| Tp (min, max) | 1, 9 | 1, 9 | 1, 9 | 1, 9 | |
| τw (min, max) | 0.09, 16.42 | 0.1, 5.29 | 0.11, 15.12 | 0.16, 0.5 | |
| Hm0/h | 0.02, 0.27 | 0.02, 0.36 | 0.03, 0.47 | 0.08, 0.27 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Proença, B.; Ganthy, F.; Michalet, R.; Sottolichio, A. Observations of Tidal Flat Sedimentation within a Native and an Exotic Spartina Species. Water 2021, 13, 1566. https://doi.org/10.3390/w13111566
Proença B, Ganthy F, Michalet R, Sottolichio A. Observations of Tidal Flat Sedimentation within a Native and an Exotic Spartina Species. Water. 2021; 13(11):1566. https://doi.org/10.3390/w13111566
Chicago/Turabian StyleProença, Barbara, Florian Ganthy, Richard Michalet, and Aldo Sottolichio. 2021. "Observations of Tidal Flat Sedimentation within a Native and an Exotic Spartina Species" Water 13, no. 11: 1566. https://doi.org/10.3390/w13111566
APA StyleProença, B., Ganthy, F., Michalet, R., & Sottolichio, A. (2021). Observations of Tidal Flat Sedimentation within a Native and an Exotic Spartina Species. Water, 13(11), 1566. https://doi.org/10.3390/w13111566







