Abstract
Innovative and low-energy solutions for the removal of nitrogen from domestic wastewater are needed to achieve regulatory ambitions. However, there is a lack of appropriate technologies for use in non-centralised applications, where receiving waterbodies also are potentially sensitive. Denitrifying down-flow hanging sponge (DDHS) reactors are a promising solution but their performance has not been assessed under colder operating conditions pertinent to northern climates. Two DDHS reactor configurations (short and tall anoxic zones) were tested under “typical” UK winter, summer, and spring/autumn temperatures. At 22 °C, both reactors achieved >58% total nitrogen (TN) removal from domestic wastewater with no significant differences in removal rates between configurations. However, denitrification was lost at 13 °C in the reactor with the short anoxic zone, and was lost totally in both systems at 6 °C. Efficient nitrification was retained at 6 °C in both reactors (>90% removal NH4–N), suggesting that while elevated TN removal was not retained under colder conditions, the DDHS systems still effectively removed ammonia under UK winter conditions. DDHS reactors show promise for use under colder temperature conditions, although optimisation is needed, including the derivation of temperature correction factors for nitrogen removal.
1. Introduction
Ever-tightening wastewater discharge standards demand innovative solutions to deliver reliable and efficient nitrogen removal, especially at lower temperatures. Many solutions exist for large-scale wastewater treatment facilities, but achieving efficient nitrogen removal with smaller, decentralised systems is more difficult. The definition of “decentralised” varies across the world. Here, we consider a decentralised system to be one that serves a community of 250 population-equivalent or less. This size is relevant to rural UK and equates to approximately 50 m3 mean-flow per day, which is the low-end for the numerical regulation of continuous discharges [1]. Such small-scale systems are often overlooked because their influence is usually local, and they are typically subject to less stringent regulatory control [1]. However, smaller discharges also can be to ecologically sensitive receiving waters [2] and it may be necessary in the future to ensure more decentralised wastewater releases will achieve legislative compliance [3]. Decentralisation is also considered a potentially sustainable approach for wastewater treatment in the emerging world [4].
Nitrogen (N) removal is typically achieved using energy-intensive technologies, chemical resources, and/or large-footprint technologies. Therefore, few viable treatment options exist for N-removal in decentralised applications [5]. For example, the use of breakpoint chlorination or ion exchange for ammonia removal requires onsite chemical storage and access for heavy goods vehicles, which may not be feasible in decentralised or rural locations. Biotechnologies, such as denitrifying down-flow hanging sponge (DDHS) reactors, show promise. DDHS systems require little energy input and have been optimised for total nitrogen (TN) removal from domestic wastewater at an ambient temperature of 22–23 °C [6]. These systems also have the benefit of reducing antibiotic-resistant genes in their effluent [6,7], and sponge-based technologies have shown successful removal of viruses [8] and faecal coliforms [9]. However, the performance of such systems has not yet been assessed at colder temperatures so their viability for use in temperate climates, such as the UK, is not understood. Here, we sought to fill this knowledge gap and describe the semi-optimised performance of DDHS reactors under temperatures down to 6 °C, assessing their potential use in the UK under winter conditions.
As background, DDHS systems use sponges as the support media, which offer greater surface area-to-volume ratios compared with more traditional media, such as stone or plastic. Typically, specific surface areas of traditional media are between 50 and 100 m2/m3 [10]. Porous sponge media made of polyurethane, with 97% void fraction (density = 30 kg/m3), have a high specific surface area of 250–270 m2/m3 [11,12]. The reticulated structure of sponge also allows higher biomass retention [11] and potentially provides more diverse ecological niches for microbial community development within biofilms. As such, an ancillary goal was to assess whether sponge media provide insulation against the external environment and more stable performance [13], even under colder conditions.
2. Methods
2.1. Reactor Configuration
Two lab-scale (140 × 500 mm; diameter × height cylinders) DDHS reactors with varying sponge configurations were built to test the effect of aerobic–anoxic ratios on lower temperature operations. The aerobic–anoxic ratio is the ratio of sponge volumes in the aerobic versus the anoxic sections (see Figure 1). DDHS reactors use a bipartite design that consists of sequential redox compartments for aerobic then anoxic treatment. Anoxic conditions are created by submerging the lower sponges in the effluent from the preceding aerobic step. Here, the anoxic compartments were supplied with additional wastewater at a bypass point located at the top of the anoxic sponge section. The “bypass” shunts a portion of “raw” wastewater across the top aerobic sponge core, which encourages anoxia by adding additional carbon to the submerged layers to promote denitrification [14].
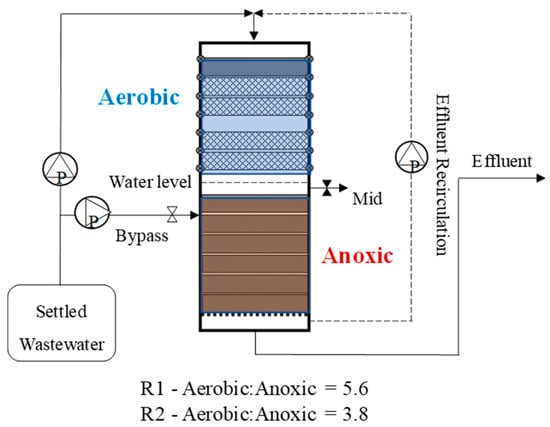
Figure 1.
Schematic of a denitrifying down-flow hanging sponge (DDHS) reactor showing the relationship between the aerobic and anoxic sponge layers. Additionally, the bypass and recirculation lines are shown. Reactors R1 and R2 differ based on the relative volumes of the aerobic versus and anoxic zones. Figure not to scale.
Reactor 1 (R1) was set up as described by Bundy et al. [6], with an aerobic–anoxic ratio of 5.6 by sponge volume. Reactor 2 (R2) was designed to potentially be more resilient to temperature changes by extending the anoxic zone to allow for greater DO solubility in colder water and reduced microbial activity at lower temperatures. The aerobic–anoxic ratio of R2 was 3.8. Eleven polyurethane sponge cylinders (140 × 30 mm each) were placed in each reactor to act as a support matrix for biofilm development. The total sponge volume in each reactor was 5.08 × 10−3 m3. Coarse (45 pores per inch) and fine (20 pores per inch) sponges were used in the aerobic and anoxic zones, respectively, with a single fine sponge disk positioned at the top of each aerobic zone to aid solids management. The aerobic sponge layers were supported by PVC-coated wire mesh and hung suspended from the top of the reactors to maximise passive aeration.
2.2. Influent
Primary settled wastewater (termed, “influent”) was collected weekly from a conventional activated sludge plant in North East England. It was stored at 4 °C in sealed containers until required. Every second day, influent was transferred to a 25 L carboy stored in a fridge adjacent to the DDHS units, and influent was continuously drawn in parallel into each reactor. Sponges (both aerobic and anoxic sponges) were seeded prior to reactor commissioning, by soaking in an inoculum mixture containing 50% nitrifying activated sludge and 50% primary settled wastewater for 24 h. Activated sludge and primary settled wastewater were collected from the treatment plant as above. Inoculation was performed at room temperature (21–23 °C) after three hours of inoculum collection.
2.3. Reactor Operation
The reactor columns were gravity-fed with an overall mean hydraulic retention time (HRT) of 34.8 h, a 30% influent bypass (% of influent flowrate) to the anoxic zone, and a recirculation ratio of 4.9 to promote uniform flow distribution (see [6]). The mean flow rate was 2.43 mL/min, which provided a hydraulic loading of 689 L/m3-sponge/day to each reactor. HRT was calculated using sponge volume. The experiment was carried out in a temperature-controlled incubator (Panasonic MIR 554-PE) for 6 months between April and October 2017. Reactor performance, in terms of % and load of pollutant removal, was compared at temperatures simulating meteorological summer (22 °C), autumn/spring (13 °C), and winter (6 °C) conditions in North East England. Values were guided by the maximum mean temperatures recorded at the weather station in Durham, UK, between 1981 and 2010 [15]. Air temperature ranges for this period were 1.0–6.9 °C, 5.1–12.7 °C, and 10.4–19.2 °C, for winter, autumn/spring, and summer, respectively.
2.4. Sample Analysis
Weekly samples were taken from the influent, post-aerobic effluent, and the final effluent from each reactor. Samples were collected during quasi-steady-state reactor operating conditions, allowing four weeks of reactor stabilisation before sampling at each temperature. Weekly replicate samples used for chemical analyses, which included soluble and total chemical oxygen demand (sCOD and tCOD), ammonium (NH4–N), total Kjeldahl nitrogen (TKN), nitrite (NO2–N) and nitrate (NO3–N), pH, and dissolved oxygen (DO). All of the analysis was performed in triplicate immediately after sample collection, in accordance with Standard Methods for Examinations of Water and Wastewater [16].
Colourimetric test kits (Merck, Darmstadt, Germany) were used for COD and NH4–N analysis. TN is defined as the sum of TKN and nitrogenous anions. Determination of anions was performed using ion chromatography (IC) using an ICS-1000 system (Sunnyvale, CA, USA) fitted with an AS40 auto sampler (Waltham, MA, USA). The IC was equipped with a conductivity detector and an anion column for separations (Ionpac AS14A, 4250 mm analytical, Sunnyvale, CA, USA). Samples for soluble analysis were filtered using 0.2 µm nylon syringe filters (Monroeville, PA, USA) and all analyses were carried out alongside pre-prepared standards (Monroeville, PA, U). pH and DO concentrations were quantified using 3310 (Cole-Palmer, Stone, UK) and HQ40d portable multi meters (Ames, IA, USA), respectively. Prior to use, instrument calibration was performed according to manufacturers’ instructions.
2.5. Data Analysis and Statistics
Percentage and load removal per day were calculated for each operating temperature. All of the statistical analysis was performed using R [17] and significance was defined by 95% confidence limits (p < 0.05), unless otherwise stated. Unpaired t-tests were carried out to compare nutrient concentrations and removal rates in equivalent samples from the two reactors, and concentrations and removal rates from the same reactor at different operating temperatures were compared using paired t-tests. Single-factor analysis of variance (ANOVA) was used to compare the difference in TN removal between the two reactors at each operating temperature.
Pearson correlation analysis was used to compare relationships among influent characteristics, such as concentrations of influent wastewater constituents, and operating conditions relative to the removal of TN and NH4–N. Ideally, statistical models would have been used to infer underlying causes of observed removal rates, however, the limited data can sometimes lead to model overfitting, which occurs when sample numbers are low relative to independent variables. Therefore, for the sake of clarity, correlation analysis was used and only example conclusions were drawn. Correlation analysis allows the comparison of relationships between variables that are likely to influence the removal and/or final effluent concentration of key pollutants. Throughout, the term “treatment performance” is used to refer to the removal of pollutants in terms of load or percentage and is made clear within each context.
3. Results and Discussion
3.1. Trends in TN Removal and Effluent Quality
The relative performance of two reactors under each temperature condition are summarised in Table 1. Significant differences in TN load removal were observed vs. temperature in both reactors (ANOVA, p = 3.1 × 10−5). Mean TN load removal at 22 °C was 0.11 g/L/day in R1 and R2 (Table 1), whereas TN removal was only −0.045 and 0.036 g/L/day at 13 °C in R1 and R2, respectively. This equates to a decrease in TN load removal of 140% in R1 and 34.3% in R2 between 22 °C and 13 °C, respectively. Comparably, a load removal of sCOD decreased by 48.8% and 49.2% in R1 and R2 between 22 °C and 13 °C, suggesting that TN removal cannot be exclusively explained by the decrease in temperature affecting oxygen solubility.

Table 1.
A summary of mean influent and effluent quality after different stages of treatment by both reactors, at each temperature increment. Standard deviations are shown in brackets. R% = percentage removal. Load removal (g/L/day) = load of constituent removed per litre, per day. Inf = Influent, Eff = Effluent, sCOD = soluble chemical oxygen demand, tCOD = total chemical oxygen demand, TN = total nitrogen, NH4-N = ammonium nitrogen, DO = dissolved oxygen. n = 24.
A dramatic reduction in TN load removal was seen at 13 °C, probably due to a slowing of metabolic rates (see Figure 2). However, while R2 retained some TN removal capacity, a net increase in TN was observed in R1. This is likely due to the extended anoxic zone in R2 providing a “buffering” effect to the temperature change. The greater volume of anoxic sponge in R2 may provide protection from the effects of the external temperature decrease: namely the increase in oxygen solubility in water and the slowing of metabolic rates. The final effluent DO concentration in R1 doubled between 22 °C and 13 °C, whereas the increase observed in R2 was only 23.5% (Table 1). This suggests a more rapid loss of denitrification in R1, which is consistent with the increase in NO3–N in the R1 effluent compared with R2 at 13 °C (Figure 3). NO3–N constitutes 68.5% of TN in the R1 effluent at 13 °C, compared with only 32.5% of TN in the R2 effluent at 13 °C.
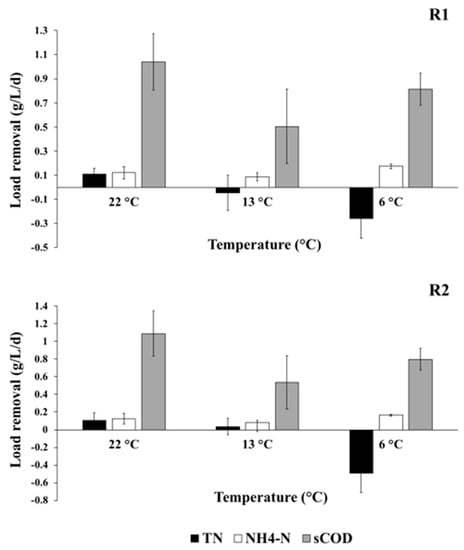
Figure 2.
Reactor performance shown as mean load removal per litre, per day for total nitrogen (TN), ammonium, and soluble COD. Error bars show the standard deviation range. For samples, n = 24.
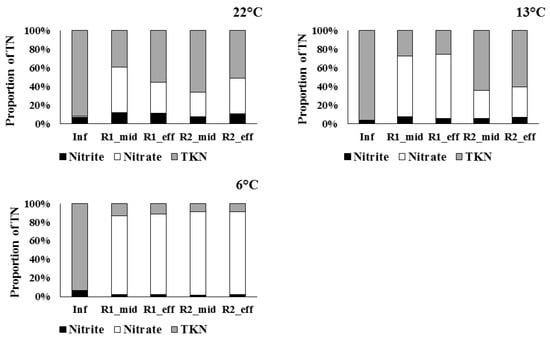
Figure 3.
Proportion of nitrite, nitrate, and TKN (as % of TN) measured in the influent (Inf), post-aerobic effluent (mid), and final effluent (eff) from R1 and R2, at 22 °C, 13 °C, and 6 °C. n = 24.
The importance of improved TN removal on receiving water quality is highlighted when one considers changes in removal at lower temperatures. TN load removal decreased dramatically in both reactors, from −0.045 at 13 °C to −0.260 g/L/day at 6 °C in R1 and from 0.036 at 13 °C to −0.494 g/L/day at 6 °C in R2. The mean final effluent TN concentration from R2 was 66.7 mg-N/L higher than R1 at 6 °C, which almost exclusively results from the higher NO3–N levels in the effluent (Figure 3). While phosphate is generally considered the limiting nutrient in freshwater ecosystems, NO3–N may be more common and influential than previously recognised [18], particularly in rural areas, which often are dominated by agricultural land use and where associated inputs to wastewater collection systems exist.
The elevated level of nitrate at 6 °C in the final effluents is probably attributable to the ammonification from organic matter, which mobilises N from accumulated biosolids within the reactors. This provides extra soluble organic N and, in turn, ammonia, which gets converted by AOB/NOB to nitrate, hence the reduced proportion of TKN within the post-aerobic and effluent samples (Figure 3). A reduction in biosolids removal, most likely due to lower endogenous activity, could, in theory, increase abundances of internal biosolids at lower temperatures, increasing ammonification and nitrification, and nitrate levels in the reactor effluents [19,20]. However, characterising resident microbial communities would be needed to confirm this speculation.
No significant difference in TN removal (un-paired t-test, p = 0.31) was observed between R1 and R2 when considering performance over the entire experiment. This is supported by the difference in TN removal between the two reactors (i.e., two difference reactor configurations) not being significant (unpaired t-test, p = 0.31), which may be an indication that the effects of temperature can only be mitigated against, to a certain point (in this case to 13 °C), through changes to the configuration of the DDHS bioreactors.
The amount of TN removed appears somewhat linked to the TN concentration of the influent, which is expected. However, a correlation of 0.32 between influent TN concentration and percentage TN removal suggests that both reactors were sized appropriately for treating the waste load (see influent data in Table 1). In other words, the relatively weak positive relationship between the concentration of influent TN and the removal of TN may suggest that other factors are important to TN removal in DDHS systems operating at different temperatures. Correlations between the percentage removal of TN and temperature (correlation = −0.70) and NO3–N concentration in the post-aerobic effluent (correlation = −0.94) suggest that net TN removal was dominated by the bio-removal of ammonia through nitrification. However, analysis of a larger dataset would be required to confirm this and the previous speculation.
3.2. Nitrification at Low Temperatures
Influent characteristics appear to affect observed rates of nitrification, which may, in part, explain relatively weak correlations between temperature and NH4–N percentage removal. At 22 °C, the average effluent concentration of NH4–N was 13.0 mg N/L in R1 and 12.6 mg N/L in R2 (Table 1), which is below the Urban Wastewater Treatment Directive [21] limit of 15 mg/L. Further, the percentage removal of NH4–N increased with each temperature drop, becoming >90% removal in both reactors at 6 °C. There was not a significant difference between percentage NH4–N removal at 22 °C and 13 °C in R1 or R2 (paired t-test, p = 0.22 and p = 1.8). However, there was a significant difference between the percentage NH4–N removal at 13 °C and 6 °C in both reactors (paired t-test, p = 0.004 and p = 0.003 for R1 and R2, respectively). The increase in NH4–N percentage removal at 13 °C compared with 22 °C is likely because of the decrease in influent TN and NH4–N concentration (Table 1), whereas the changes observed at 6 °C demand a more refined explanation.
NH4–N load removal decreased in both reactors at 13 °C (0.09 and 0.08 g/L/day, respectively), but increased to 0.17 g/L/day load removal in R1 and R2 when the temperature was dropped to 6 °C, an increase of 49.3% and 48.8% from 13 °C, respectively. The decrease in NH4–N load removal following the first temperature drop to 13 °C was probably due to lower metabolic rates. Elevated effluent DO levels at 13 °C (3.2 and 2.1 mg/L, respectively) also suggest denitrification was suppressed, especially in R1 with the shorter anoxic zone. However, NH4–N removals at 13 °C were sufficiently high to suggest there was enough oxygen in the lower layers to support some nitrification in both reactors. At 6 °C, all final effluent DO concentrations were between 5 and 6 mg/L (Table 1). Therefore, we suspect the “anoxic” zones became capable of supporting a nitrifying community [22], even though apparent metabolic rates were lower at 6 °C.
Finally, the relative abundance of NO2–N in the final effluent was at its lowest at 6 °C for both reactors (Figure 3), implying complete nitrification was evident at this low temperature, i.e., no accumulation of NO2–N as an intermediate N species during the two-step oxidation process. This is best explained by the fact that the reactors became entirely aerobic and oversized relative to the influent NH4–N loading rates. In other words, lower metabolic rates in conjunction with increased oxygen solubility at lower temperature increased net nitrification, functionally making the reactors wholly nitrification treatment systems.
3.3. Implications for Reactor Design
The greater resilience of R2 to the temperature drop to 13 °C suggests that modifiable “package-style” designs may be most appropriate for DDHS applications in places with a seasonal climate. For example, an “oversized” anoxic zone, which is placed directly below the aerobic layer, provides an extended biologically active volume that is essential to compensate for the increased oxygen solubility of the colder water. This is key because microbial substrate affinities and specific growth rates are typically lower under colder temperatures [23], i.e., an oversized lower zone provides extra volume to reduce oxygen levels to anoxia, which is critical for denitrification.
However, altered load removal of TN at 6 °C suggests that combined nitrification–denitrification only occurs at higher temperatures. Under UK winter temperatures, effluent DO concentrations were found to increase dramatically (Table 1), which is expected [24]. However, if one separates the aerobic and anoxic reaction compartments, one could have greater control over DO levels in the lower anoxic zone. Further, the inclusion of a buffer tank between the aerobic and anoxic units could provide greater volume for additional microbial activity, which might help reduce DO and increase the probability of denitrification in the lower layers.
Compared with traditional trickling filter systems, relatively high recirculation rates were used for both DDHS reactors in this experiment. Besides providing adequate distribution of the influent over the upper sponge layers, which is essential where flows are lower or the wastewater is stronger, a by-product of elevated recirculation may have been to “over-aerate” the influent by repeated passage through the aerobic zone. This may have overloaded the anoxic zone with oxygen and suppressed establishment of a denitrifying community. Optimising the recirculation ratio for denitrification at a range of temperatures was beyond the scope of this study. However, the possible influence of this consideration is evident and warrants further investigation.
It is well known that nitrification is among the most temperature-sensitive of all biological processes [25]. A correction factor, derived from Arrhenius’ equation, is often applied to the design of nitrifying wastewater treatment systems [26]. Several studies (e.g., [27,28]) have shown that appropriately designed systems can function within a moderate range when operated under a variety of temperatures, with separate correction factors existing for below 7 °C [25]. With this in mind, correction factors should be developed for DDHS reactors, not least because of the intrinsic link with denitrification being so prevalent at low temperatures in these systems. In order to determine suitable correction factors and more effectively utilise the technology, the aerobic and anoxic zones should be separated physically, and the anoxic zone should be a sealed unit.
The results presented here show that DDHS systems have the potential for effective nitrogen, solids, and organic matter removal across a range of temperatures. For example, we report removal rates of 58% for TN at 22 °C, which, encouragingly, is within the range seen in other sponge-based systems when being used for post-treatment with up-flow anaerobic sludge blanket systems [12,29].
4. Conclusions
DDHS reactors offer a low energy solution for carbon and nutrient removal in decentralised locations. The performance of the previous configuration (R1) shows promise at 22 °C with >58% removal of TN and >81% removal of sCOD from domestic wastewater. However, simple adjustments can be made to allow effective wastewater treatment at lower temperatures. For example, the loss of denitrification with decreasing temperature may be minimised by decreasing the aerobic–anoxic ratio. This was particularly effective at 13 °C. TN removal may be further improved by adjusting recirculation rates with different temperature conditions. A lower aerobic–anoxic ratio appears better for lower temperatures, although local conditions may drive optimal DDHS configurations.
Further work is necessary to fully optimise the system for seasonal climates, including the derivation of temperature correction factors for nitrification. In this experiment, the effect of increased oxygen solubility at colder temperatures was more evident at 6 °C than at 13 °C. These effects were strongly influenced by the reactor configuration and resulted in enhanced nitrification occurring at the lowest temperature due a more extended aerobic zone. Overall, results show DDHS systems can effectively operate at lower temperatures, but how they operate and what they remove differs as the temperature declines. Therefore, reactor designs that allow operational flexibility are preferred, especially for use in regions with wider variations in seasonal temperatures.
Author Contributions
Conceptualization, D.W.G., J.T.B., M.-C.J.; methodology, E.J.C., J.T.B., M.-C.J.; formal analysis, E.J.C.; investigation, E.J.C.; writing—original draft preparation, E.J.C., J.T.B.; writing—review and editing, D.W.G., J.T.B., M.-C.J., E.J.C.; supervision, D.W.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Engineering and Physical Sciences Research Council (EPSRC) grant number: EP/M50791X/1) and Northumbrian Water Ltd.
Conflicts of Interest
The authors declare no conflict of interest.
References
- DEFRA. Environmental Permitting (England and Wales) Regulations 2010; Statutory Instrument No. 675 Environmental Permitting (England and Wales) Regulations 2010; DEFRA: London, UK, 2010.
- Pujol, R.; Lienard, A. Qualitative and quantitative characterization of wastewater for small communities. In Proceedings of the International Specialized Conference on Design and Operation of Small Wastewater Treatment Plants, Trondheim, Norway, 26–28 June 1989; Odegaard, H., Ed.; Tapir: Bergen, Norway, 1989; pp. 267–274. [Google Scholar]
- Council of European Communities. Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off. J. Eur. Parliam. 2000, L327, 1–82. [Google Scholar]
- Graham, D.W.; Bunce, J.T.; Giesen, M.J. Strategic approach for prioritising local and regional sanitation interventions for reducing global antibiotic resistance. Water 2019, 11, 27. [Google Scholar] [CrossRef]
- Bunce, J.T.; Ndam, E.; Ofiteru, I.D.; Moore, A.; Graham, D.W. A review of phosphorus removal technologies and their applicability to small-scale domestic wastewater treatment systems. Front. Environ. Sci. 2018, 6, 8. [Google Scholar] [CrossRef]
- Bundy, C.A.; Wu, D.; Jong, M.C.; Edwards, S.R.; Ahammad, Z.S.; Graham, D.W. Enhanced denitrification in Downflow Hanging Sponge reactors for decentralised domestic wastewater treatment. Bioresour. Technol. 2017, 226, 1–8. [Google Scholar] [CrossRef]
- Jong, M.C.; Su, J.Q.; Bunce, J.T.; Harwood, C.R.; Snape, J.R.; Zhu, Y.G.; Graham, D.W. Co-optimization of sponge-core bioreactors for removing total nitrogen and antibiotic resistance genes from domestic wastewater. Sci. Total Environ. 2018, 634, 1417–1423. [Google Scholar] [CrossRef]
- Kobayashi, N.; Oshiki, M.; Ito, T.; Segawa, T.; Hatamoto, M.; Kato, T.; Yamaguchi, T.; Kubota, K.; Takahashi, M.; Iguchi, A.; et al. Removal of human pathogenic viruses in a down-flow hanging sponge (DHS) reactor treating municipal wastewater and health risks associated with utilization of the effluent for agricultural irrigation. Water Res. 2017, 110, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Beas, Y.R.E.; Kujawa-Roeleveld, K.; van Lier, J.B.; Zeeman, G.A. Downflow Hanging Sponge (DHS) reactor for faecal coliform removal from an Upflow Anaerobic Sludge Bed (UASB) effluent. Water Sci. Technol. 2015, 72, 2034–2044. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Harrison, J.R.; Daigger, G.T. Comparison of Trickling Filter Media. J. Water Pollut. Control Fed. 1987, 59, 679–685. [Google Scholar]
- Tawfik, A.; Ohashi, A.; Harada, H. Effect of sponge volume on the performance of down-flow hanging sponge system treating UASB reactor effluent. Bioprocess Biosyst. Eng. 2010, 33, 779–785. [Google Scholar] [CrossRef]
- Onodera, T.; Tandukar, M.; Sugiyana, D.; Uemura, S.; Ohashi, A.; Harada, H. Development of a sixth-generation down-flow hanging sponge (DHS) reactor using rigid sponge media for post-treatment of UASB treating municipal sewage. Bioresour. Technol. 2014, 152, 93–100. [Google Scholar] [CrossRef]
- Ikuma, K.; Decho, A.W.; Lau, B.L.T. The Extracellular Bastions of Bacteria—A Biofilm Way of Life. Nat. Educ. Knowl. 2013, 4, 2. [Google Scholar]
- Schipper, L.A.; Robertson, W.D.; Gold, A.J.; Jaynes, D.B.; Cameron, S.C. Denitrifying bioreactors—An approach for reducing nitrate loads to receiving waters. Ecol. Eng. 2010, 36, 1532–1543. [Google Scholar] [CrossRef]
- Met Office. Met Office: UK Climate—Historic Station Data 1981–2010. Available online: https://www.metoffice.gov.uk/public/weather/climate-historic/#?tab=climateHistoric (accessed on 26 May 2020).
- APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2009. [Google Scholar]
- R Foundation for Statistical Computing. R Core R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Paerl, H.W.; Xu, H.; Mccarthy, M.J.; Zhu, G.; Qin, B.; Li, Y.; Gardner, W.S. Controlling harmful cyanobacterial blooms in a hyper-eutrophic lake (Lake Taihu, China): The need for a dual nutrient (N & P) management strategy. Water Res. 2011, 45, 1973–1983. [Google Scholar] [PubMed]
- Lishman, L.A.; Legge, R.L.; Farquhar, G.J. Temperature effects on wastewater treatment under aerobic and anoxic conditions. Water Res. 2000, 34, 2263–2276. [Google Scholar] [CrossRef]
- Ducey, T.F.; Vanotti, M.B.; Shriner, A.D.; Szogi, A.A.; Ellison, A.Q. Characterization of a microbial community capable of nitrification at cold temperature. Bioresour. Technol. 2010, 101, 491–500. [Google Scholar] [CrossRef]
- Council of European Communities. Directive concerning urban waste water treatment. Off. J. Eur. Commun. 1991, 135, 40–52. [Google Scholar]
- Daigger, G.T. Oxygen and carbon requirements for biological nitrogen removal processes accomplishing nitrification, nitritation, and anammox. Water Environ. Res. 2014, 86, 204–209. [Google Scholar] [CrossRef]
- Nedwell, D.B. Effect of low temperature on microbial growth: Lowered affinity for substrates limits growth at low temperature. FEMS Microbiol. Ecol. 1999, 30, 101–111. [Google Scholar] [CrossRef]
- Weiss, R.F. The solubility of nitrogen, oxygen and argon in water and seawater. Deep Sea Res. Oceanogr. Abstr. 1970, 17, 721–735. [Google Scholar] [CrossRef]
- Hwang, J.H.; Oleszkiewicz, J.A. Effect of cold-temperature shock on nitrification. Water Environ. Res. 2007, 79, 964–968. [Google Scholar] [CrossRef]
- Oleszkiewicz, J.A.; Berquist, S.A. Low temperature nitrogen removal in sequencing batch reactors. Water Res. 1988, 22, 1163–1171. [Google Scholar] [CrossRef]
- Painter, H.A.; Loveless, J.E. Effect of temperature and pH value on the growth-rate constants of nitrifying bacteria in the activated sludge process. Water Res. 1983, 17, 237–248. [Google Scholar] [CrossRef]
- Head, M.A.; Oleszkiewicz, J.A. Bioaugmentation for nitrification at cold temperatures. Water Res. 2004, 38, 523–530. [Google Scholar] [CrossRef]
- Onodera, T.; Okubo, T.; Uemura, S.; Yamaguchi, T.; Ohashi, A.; Harada, H. Long-term performance evaluation of down-flow hanging sponge reactor regarding nitrification in a full-scale experiment in India. Bioresour. Technol. 2016, 204, 177–184. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).