Hydrogeochemical Characters of Karst Aquifers in Central Italy and Relationship with Neotectonics
Abstract
1. Introduction
2. Materials and Methods
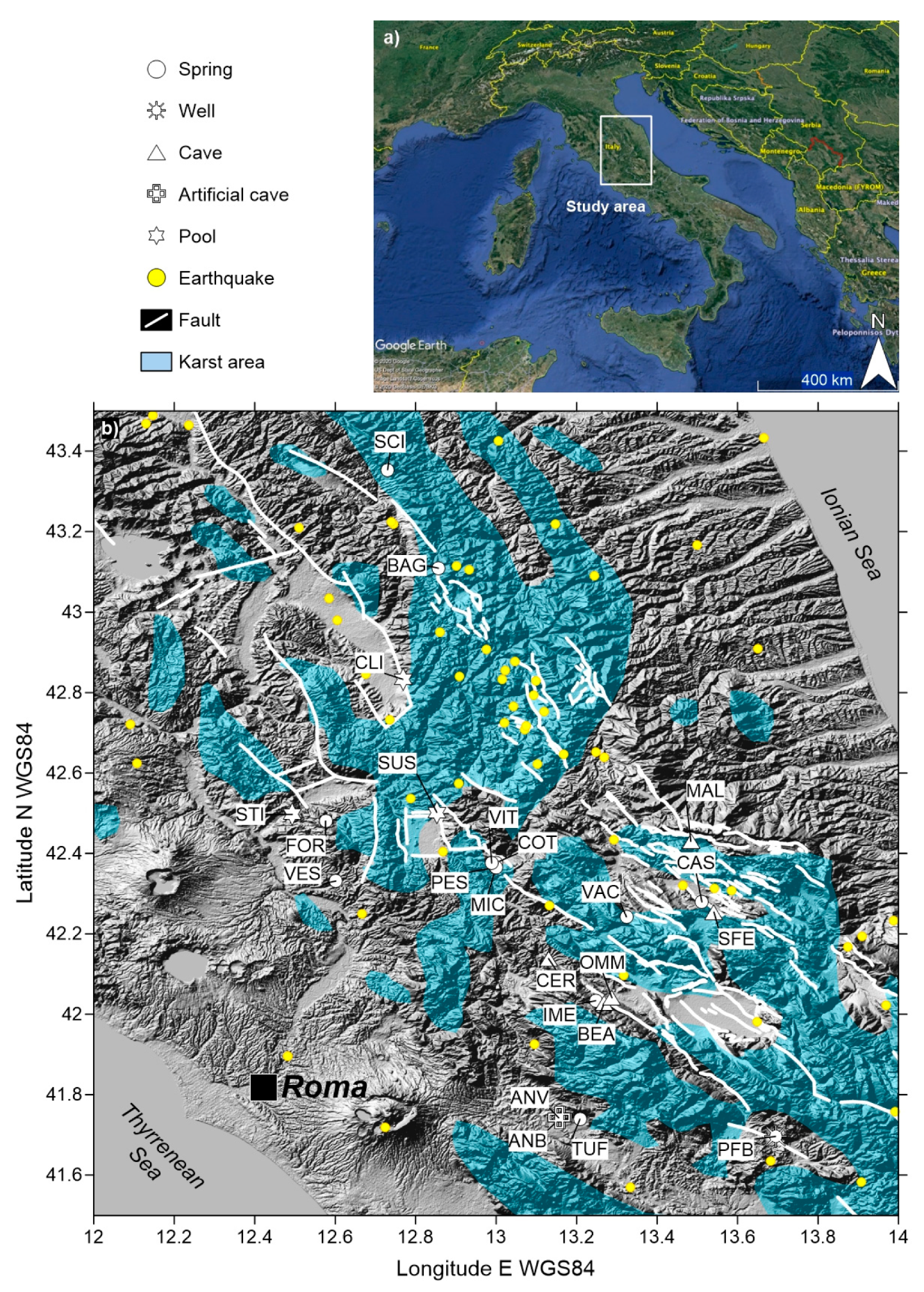
2.1. Study Area Settings
2.2. Anaytical Methodologies
3. Results
4. Discussion
4.1. Major Ion Chemistry
4.2. Dissolved Gases
4.3. O, D, C Isotopic Composition
4.4. Potential Suitability of the Studied sites for the Monitoring of Neotectonic Activity and Its Spatial Distribution
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aggarwal, Y.P.; Sykes, L.R.; Armbruster, J.; Sbar, M.L. Premonitory Changes in Seismic Velocities and Prediction of Earthquakes. Nature 1973, 241, 101–104. [Google Scholar] [CrossRef]
- Roeloffs, E.A. Hydrologic precursors to earthquakes: A review. Pure Appl. Geophys. 1988, 126, 177–209. [Google Scholar] [CrossRef]
- Reasenberg, P.A. Foreshock occurrence before large earthquakes. J. Geophys. Res. Solid Earth 1999, 104, 4755–4768. [Google Scholar] [CrossRef]
- King, C.Y.; Azuma, S.; Ohno, M.; Asai, Y.; He, P.; Kitagawa, Y.; Igarashi, G.; Wakita, H. In search of earthquake precursors in the water-level data of 16 closely clustered wells at Tono, Japan. Geophys. J. Int. 2000, 143, 469–477. [Google Scholar] [CrossRef]
- Favara, R.; Grassa, F.; Inguaggiato, S.; Valenza, M. Hydrogeochemisty and stable isotopes of the thermal springs: Earthquake-related chemical changes along Belice Fault (Western Sicily). Appl. Geochem. 2001, 16, 1–17. [Google Scholar] [CrossRef]
- Lucente, F.P.; De Gori, P.; Margheriti, L.; Piccinini, D.; Di Bona, M.; Chiarabba, C.; Piana Agostinetti, N. Temporal variation of seismic velocity and anisotropy before the 2009 M W 6.3 L’Aquila earthquake, Italy. Geology 2010, 38, 1015–1018. [Google Scholar] [CrossRef]
- Amoruso, A.; Crescentini, L.; Petitta, M.; Rusi, S.; Tallini, M. Impact of the 6 April 2009 L’Aquila earthquake on groundwater flow in the Gran Sasso carbonate aquifer, Central Italy. Hydrol. Process 2010, 25, 1754–1764. [Google Scholar] [CrossRef]
- Riguzzi, F.; Crespi, M.; Devoti, R.; Doglioni, C.; Pietrantonio, G.; Pisani, A.R. Geodetic strain rate and earthquake size: New clues for seismic hazard studies. Phys. Earth Planet. Inter. 2012, 206–207, 67–75. [Google Scholar] [CrossRef]
- Barberio, M.D.; Barbieri, M.; Billi, M.; Doglioni, C.; Petitta, M. Hydrogeochemical changes before and during the 2016 Amatrice-Norcia seismic sequence (central Italy). Entif. Rep. 2017, 15, 11735. [Google Scholar] [CrossRef]
- Rikitake, T. Earthquake prediction. Earth Sci. Rev. 1968, 4, 245–282. [Google Scholar] [CrossRef]
- Wakita, H. Geochemistry as a tool for earthquake prediction. J. Phys. Earth. 1977, 25, 175–183. [Google Scholar] [CrossRef]
- Wakita, H. Changes in groundwater level and chemical composition. In Earth-quake Prediction Techniques; University of Tokyo: Tokyo, Japan, 1982; pp. 171–216. [Google Scholar]
- Carapezza, M.; Nuccio, P.M.; Valenza, M. Geochemical precursor of earthquake. In High Pressure Science & Technology; Pergamon Press: New York, NY, USA, 1980; pp. 90–103. [Google Scholar]
- Cai, Z.; Shi, H.; Zhang, W.; Luo, G.E.X.; Shi, X.; Yang, H. Some applications of fluid-geochemical methods to earthquake prediction in China: Proceeding of International Symposium on Continental Seismology and Earthquake Prediction; Seismological Press: Beijing, China, 1984; pp. 384–395. [Google Scholar]
- Barsukov, V.L.; Varshal, G.M.; Zamokina, N.S. Recent results of hydro- geochemical studies for earthquake prediction in USSR. Pure Appl. Geophys. 1985, 122, 143–156. [Google Scholar] [CrossRef]
- Thomas, D. Geochemical precursor to seismic activity. Pure Appl. Geophys. 1988, 126, 241–266. [Google Scholar] [CrossRef]
- Valenza, M.; Nuccio, P.M. Geochemical precursors of earthquakes. Some experiences in Italy. In Isotopic and Geochemical Precursors of Earthquakes and Volcanic Eruptions; IAEA: Vienna, Austria, 1993; pp. 44–47. [Google Scholar]
- Favara, R.; Italiano, F.; Martinelli, G. Earthquake-induced chemical changes in the thermal waters of the Umbria region during the 1997-1998 seismic swarm. Terra Nova 2001, 13, 227–233. [Google Scholar] [CrossRef]
- Sibson, R.H. Implications of fault-valve behaviour for rupture nucleation and recurrence. Tectonophysics 1992, 211, 283–293. [Google Scholar] [CrossRef]
- Muir-Wood, R.; King, G.C.P. Hydrological signatures of earthquake strain. J. Geophys. Res. Solid Earth 1993, 982, 22035–22068. [Google Scholar] [CrossRef]
- Doglioni, C.; Barba, S.; Carminati, E.; Riguzzi, F. Role of the brittle-ductile transition on fault activation. Phys. Earth Planet. Inter. 2011, 184, 160–171. [Google Scholar] [CrossRef]
- Doglioni, C.; Barba, S.; Carminati, E.; Riguzzi, F. Fault on-off versus coseismic fluids reaction. Geosci. Front. 2011, 5, 767–780. [Google Scholar] [CrossRef]
- Casale, P.; De Martin, M.; Germani, C. Il Progetto Ipodata: Stazioni sismiche in siti ipogei. In Proceedings of the Atti VIII Convegno Nazionale di Speleologia in Cavità Artificiali, Ragusa, Italy, 7–9 September 2012. [Google Scholar]
- Guidoboni, E.; Ferrari, G.; Mariotti, D.; Comastri, A.; Tarabusi, G.; Sgattoni, G.; Valensise, G. (2018) - CFTI5Med, Catalogo dei Forti Terremoti in Italia (461 a.C.-1997) e nell’area Mediterranea (760 a.C.-1500). Istituto Nazionale di Geofisica e Vulcanologia (INGV). Available online: http://storing.ingv.it/cfti/cfti5/ (accessed on 12 February 2020).
- ISPRA. ITHACA—Catalogo Delle Faglie Capaci. Available online: http://www.isprambiente.gov.it/it/progetti/suolo-e-territorio-1/ithaca-catalogo-delle-faglie-capaci (accessed on 12 February 2020).
- Società Speleologica Italiana—Carta Delle Principali Aree E Sorgenti Carsiche D’italia; ERGA Edizioni: Firenze, Italy, 2002.
- Barbieri, M.; Nigro, A.; Petitta, M. Groundwater mixing in the discharge area of San Vittorino Plain (Central Italy): Geochemical characterization and implication for drinking uses. Environ. Earth Sci. 2017, 76, 393. [Google Scholar] [CrossRef]
- Petitta, M.; Primavera, P.; Tuccimei, P.; Aravena, R. Interaction between deep and shallow groundwater systems in areas affected by Quaternary tectonics (Central Italy): A geochemical and isotope approach. Environ. Earth Sci. 2001, 63, 11–30. [Google Scholar] [CrossRef]
- Bernard, P. From the search of ‘precursor’ to the research on ‘crustal transients’. Tectonophysics 2001, 338, 225–232. [Google Scholar] [CrossRef]
- Favara, R.; Grassa, F.; Madonia, P.; Valenza, M. Flow Changes and Geochemical Anomalies in Warm and Cold Springs Associated with the 1992–1994 Seismic Sequence at Pollina, Central Sicily, Italy. Pure Appl. Geophys. 2007, 164, 1–20. [Google Scholar] [CrossRef]
- Madonia, P.; Cusano, P.; Diliberto, I.S.; Cangemi, M. Thermal anomalies in fumaroles at Vulcano island (Italy) and their relationship with seismic activity. Phys. Chem. Earth 2013, 63, 160–169. [Google Scholar] [CrossRef]
- Capasso, G.; Inguaggiato, S. A simple method for the determination of dissolved gases in natural waters. An application to thermal waters from Vulcano Island. Appl. Geochem. 1998, 13, 631–642. [Google Scholar]
- Capasso, G.; Favara, R.; Grassa, F.; Inguaggiato, S.; Longo, M. On-line technique for preparing and measuring stable carbon isotope of total dissolved inorganic carbon in water samples (δ13C). Ann. Geophys. 2005, 48, 159–166. [Google Scholar]
- Zhang, J.; Quay, P.D.; Wilbur, D.O. Carbon isotope fractionation during gas-water exchange and dissolution of CO2. Geoc. Cosm. Acta. 1995, 59, 107–114. [Google Scholar] [CrossRef]
- Langelier, W.F.; Ludwig, F. Graphical methods for indicating the mineral character of natural waters. J. Am. Water Works Assoc. 1942, 34, 335–352. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Appelo, C.A.J. Description of Input and Examples for PHREEQC Version 3—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations: U.S. Geological Survey Techniques and Methods. Available online: http://pubs.usgs.gov/tm/06/a43 (accessed on 12 February 2020).
- Célico, P.; Gonfiantini, R.; Koizumi, M.; Mangano, F. Environmental isotope studies of limestone aquifers in central Italy. In Isotope Hydrology; IAEA: Vienna, Austria, 1984; pp. 173–192. [Google Scholar]
- Longinelli, A.; Selmo, E. Isotopic composition of precipitation in Italy: A first overlap map. J. Hydrol. 2003, 270, 75–88. [Google Scholar]
- ISPRA. Carta Geologica d’Italia 1:50000, Foglio 398 Anagni; Litografia Artistica Cartografica: Firenze, Italy, 1975. [Google Scholar]
- Postpischl, D.; Agostini, S.; Forti, P.; Quinif, Y. Palaeoseismicity from karst sediments: The “Grotta de1 Cervo” cave case study (Central Italy). Tectonophysics 2011, 193, 33–44. [Google Scholar] [CrossRef]
- Chiodini, G.; Frondini, F.; Kerrick, D.M.; Rogie, J.; Parello, F.; Peruzzi, L.; Zanzari, A.R. Quantification of deep CO2 fluxes from Central Italy. Examples of carbon balance for regional aquifers and of soil diffuse degassing. Chem. Geol. 1999, 159, 205–222. [Google Scholar] [CrossRef]
- Madonia, P.; D’Aleo, R.; Di Maggio, C.; Favara, R.; Hartwig, A. The use of shallow dripwater as an isotopic marker of seepage in karst areas: A comparison between Western Sicily (Italy) and the Harz Mountains (Germany). Appl. Geoch. 2013, 34, 231–239. [Google Scholar] [CrossRef]
- Gat, J.R. Groundwater. In Stable Isotope Hydrology; Gat, J.R., Gonfiantini, R., Eds.; Technical Reports Series n. 210; IAEA: Vienna, Austria, 1981; pp. 223–240. [Google Scholar]
- Di Domenica, A.; Turtù, A.; Satolli, S.; Calamita, F. Relationships between thrusts and normal faults in curved belts: New insight in the inversion tectonics of the Central-Northern Apennines (Italy). J. Struct. Geol. 2012, 42, 104–117. [Google Scholar] [CrossRef]
- Kazakis, N.; Chalikakis, K.; Mazzilli, N.; Ollivier, C.; Manakos, A.; Voudouris, K. Management and research strategies of karst aquifers in Greece: Literature overview and exemplification based on hydrodynamic modelling and vulnerability assessment of a strategic karst aquifer. Sci. Total Environ. 2018, 643, 592–609. [Google Scholar] [CrossRef] [PubMed]









| ID | Lon | Lat | Ele | TP | T | pH | EC | Eh | Na | K | Mg | Ca | Cl | SO4 | Alk | δ18O | δD | δ13C | N2 | CH4 | CO2 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ANB | 13.2 | 41.7 | 445 | AC | 15.7 | 7.15 | 1036 | −16 | 1.53 | 1.89 | 2.01 | 5.06 | 1.87 | 1.71 | 6.13 | −6.7 | −43 | −12.3 | 12.69 | bdl | 9.25 |
| ANV | 13.2 | 41.7 | 415 | AC | 13.6 | 7.27 | 1230 | −22 | 2.25 | 1.82 | 2.74 | 6.74 | 1.61 | 1.22 | 8.29 | −6.7 | −42 | −6.0 | 11.52 | 421 | 9.59 |
| BAG | 12.9 | 43.1 | 630 | S | 12.2 | 7.34 | 390 | −30 | 0.15 | 0.02 | 0.15 | 4.08 | 0.17 | 0.22 | 3.95 | −9.1 | −57 | −12.4 | 20.46 | bdl | 9.90 |
| BAT | 12.9 | 43.1 | 630 | S | nd | 7.32 | 403 | 0.14 | 0.02 | 0.17 | 4.10 | 0.17 | 0.27 | 3.95 | −9.1 | −57 | nd | bdl | nd | ||
| CAS | 13.5 | 42.3 | 598 | S | 10.6 | 7.24 | 514 | −21 | 0.27 | 0.08 | 0.21 | 5.09 | 0.33 | 0.11 | 5.10 | −8.9 | −62 | −14.5 | 13.40 | bdl | 19.5 |
| COT | 13 | 42.4 | 409 | S | 17.7 | 6.19 | 2582 | 37 | 1.21 | 0.16 | 7.80 | 30.3 | 0.96 | 4.94 | 33.0 | −8.3 | −58 | 2.0 | 18.91 | bdl | 128 |
| FOR | 12.6 | 42.5 | 374 | S | 15 | 6.97 | 608 | −8 | 0.45 | 0.11 | 0.20 | 5.84 | 0.45 | 0.44 | 5.65 | −7.1 | −42 | −15.2 | 14.62 | bdl | 25.9 |
| IME | 13.2 | 42 | 1025 | S | 9.4 | 7.79 | 339 | −53 | 0.08 | 0.01 | 1.07 | 2.58 | 0.09 | 0.07 | 3.55 | −9.0 | −56 | −11.3 | 15.28 | 785 | 2.68 |
| MIC | 13 | 42.4 | 410 | S | 15.2 | 6.16 | 1497 | 40 | 0.38 | 0.10 | 4.93 | 20.0 | 0.30 | 2.75 | 22.2 | −8.9 | −55 | 0.1 | 7.67 | 7.7 | 550 |
| PES | 13 | 42.4 | 408 | S | 12.4 | 6.88 | 684 | −3 | 0.17 | 0.03 | 1.95 | 6.22 | 0.14 | 0.33 | 7.96 | −9.0 | −57 | −2.7 | 16.44 | 799 | 44.7 |
| SCI | 12.7 | 43.4 | 594 | S | 11.4 | 7.59 | 336 | −43 | 0.19 | 0.01 | 0.44 | 2.99 | 0.21 | 0.60 | 2.70 | −8.8 | −53 | −10.4 | 18.44 | 368 | 2.90 |
| TUF | 13.2 | 41.7 | 298 | S | 16.1 | 6.93 | 838 | −3 | 0.22 | 0.04 | 2.63 | 7.72 | 0.23 | 0.47 | 10.1 | −7.9 | −47 | −2.5 | 14.90 | 576 | 58.0 |
| VAC | 13.3 | 42.2 | 1235 | S | 10.9 | 8.18 | 318 | −70 | 0.06 | 0.01 | 0.09 | 3.50 | 0.07 | 0.03 | 3.42 | −8.9 | −62 | nd | 13.84 | 828 | 3.06 |
| VES | 12.6 | 42.3 | 129 | S | 16.3 | 7.54 | 919 | −40 | 1.56 | 0.15 | 1.07 | 7.42 | 1.15 | 0.49 | 7.34 | −6.3 | −38 | −14.4 | 15.36 | 785 | 9.02 |
| VIT | 13 | 42.4 | 408 | S | 12.3 | 6.04 | 1148 | 23 | 0.31 | 0.05 | 2.63 | 11.1 | 0.28 | 1.66 | 12.2 | −9.0 | −57 | −1.4 | 17.29 | 409 | 190 |
| BEA | 13.3 | 42 | 1058 | C | 9.6 | 7.72 | 354 | −50 | 0.09 | 0.01 | 0.19 | 3.68 | 0.11 | 0.04 | 3.57 | −8.5 | −52 | −11.0 | 14.58 | bdl | 3.66 |
| CER | 13.1 | 42.1 | 883 | C | nd | 7.22 | 374 | −20 | 0.15 | 0.01 | 0.13 | 3.79 | 0.17 | 0.05 | 3.87 | −7.7 | −47 | −14.8 | 14.85 | bdl | 9.40 |
| MAL | 13.5 | 42.4 | 939 | C | nd | 7.64 | 280 | −44 | 0.06 | 0.01 | 0.40 | 2.79 | 0.08 | 0.06 | 3.06 | −10.3 | −69 | −9.9 | 15.00 | bdl | 3.98 |
| OMM | 13.3 | 42 | 727 | C | 14.6 | 7.56 | 333 | −40 | 0.08 | 0.01 | 0.93 | 2.83 | 0.09 | 0.04 | 3.61 | −9.5 | −57 | −11.1 | 15.16 | 42 | bdl |
| SFE | 13.5 | 42.3 | 744 | C | 11.5 | 7.2 | 460 | −21 | 0.27 | 0.03 | 0.58 | 4.40 | 0.26 | 0.12 | 4.92 | −9.3 | −62 | −14.3 | 14.26 | bdl | 12.9 |
| PFB | 13.7 | 41.7 | 300 | W | 11.3 | 7.14 | 573 | −13 | 0.14 | 0.02 | 1.13 | 5.82 | 0.11 | 0.09 | 6.82 | −8.5 | −50 | −3.2 | 16.16 | 611 | 26.8 |
| CLI | 12.8 | 42.8 | 227 | P | 12.3 | 7.2 | 664 | −20 | 0.20 | 0.03 | 2.14 | 6.56 | 0.17 | 5.13 | 3.69 | −8.4 | −54 | −7.5 | 19.58 | bdl | 10.8 |
| STI | 12.5 | 42.5 | 77 | P | 16.8 | 6.54 | 3600 | 17 | 20.4 | 0.11 | 4.67 | 16.59 | 20.7 | 9.89 | 11.8 | −7.9 | −47 | −2.6 | 18.91 | bdl | 128 |
| SUS | 12.9 | 42.5 | 384 | P | 10.6 | 7.32 | 825 | −27 | 0.13 | 0.02 | 2.74 | 6.79 | 0.11 | 6.11 | 3.57 | −8.6 | −54 | −6.5 | 21.86 | bdl | 7.77 |
| Ranking | |||||
|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 |
| BEA | CER | BAG | ANB | CLI | COT |
| CAS | FOR | ANV | SUS | MIC | |
| IME | SCI | PFB | PES | ||
| MAL | VES | STI | |||
| OMM | TUF | ||||
| SFE | VIT | ||||
| VAC | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madonia, P.; Cangemi, M.; Oliveri, Y.; Germani, C. Hydrogeochemical Characters of Karst Aquifers in Central Italy and Relationship with Neotectonics. Water 2020, 12, 1926. https://doi.org/10.3390/w12071926
Madonia P, Cangemi M, Oliveri Y, Germani C. Hydrogeochemical Characters of Karst Aquifers in Central Italy and Relationship with Neotectonics. Water. 2020; 12(7):1926. https://doi.org/10.3390/w12071926
Chicago/Turabian StyleMadonia, Paolo, Marianna Cangemi, Ygor Oliveri, and Carlo Germani. 2020. "Hydrogeochemical Characters of Karst Aquifers in Central Italy and Relationship with Neotectonics" Water 12, no. 7: 1926. https://doi.org/10.3390/w12071926
APA StyleMadonia, P., Cangemi, M., Oliveri, Y., & Germani, C. (2020). Hydrogeochemical Characters of Karst Aquifers in Central Italy and Relationship with Neotectonics. Water, 12(7), 1926. https://doi.org/10.3390/w12071926






