Performance of Closed Loop Venturi Aspirated Aeration System: Experimental Study and Numerical Analysis with Discrete Bubble Model
Abstract
1. Introduction
2. Clean Water Tests
3. Experimental Procedure
4. Theory
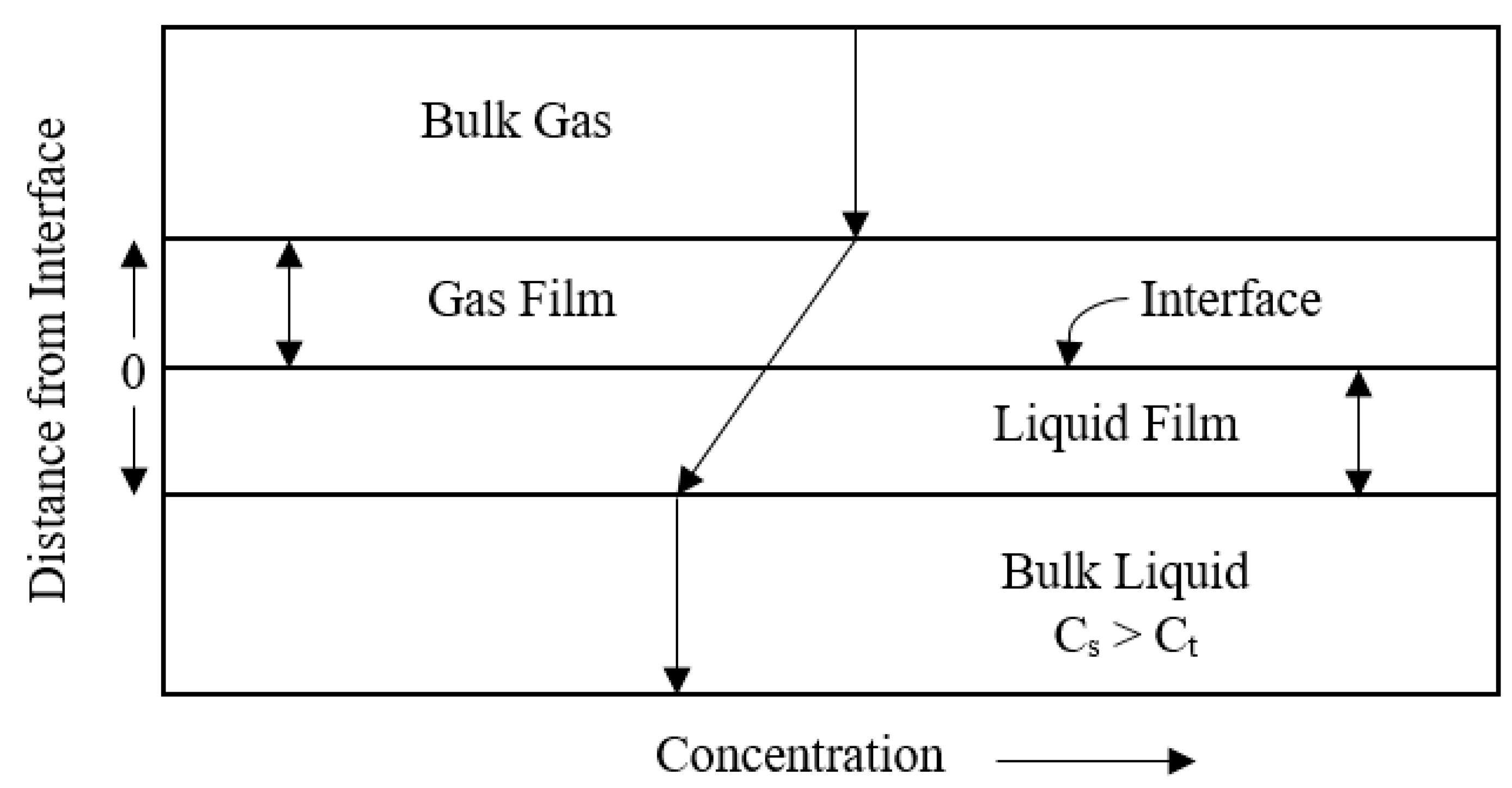
4.1. Two Film Theory
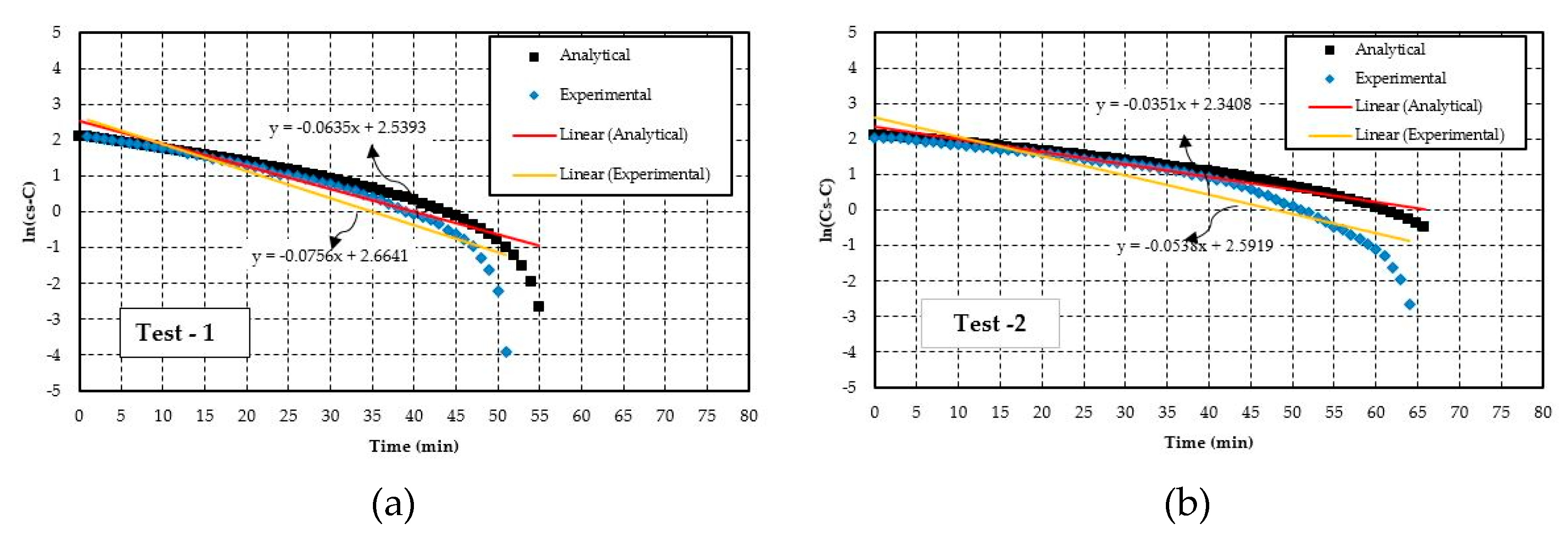
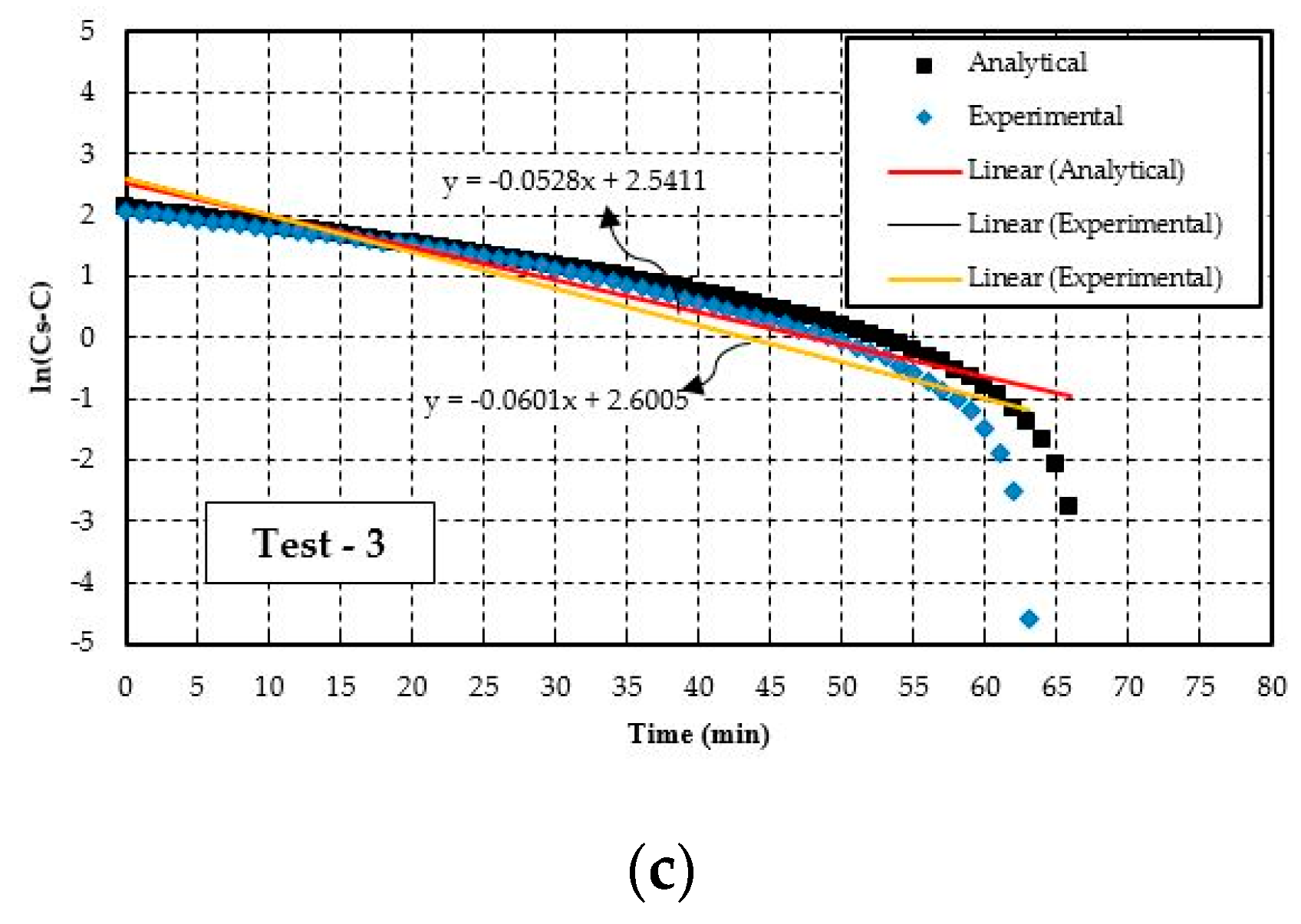
4.2. Experimental Results Evaluation Technique
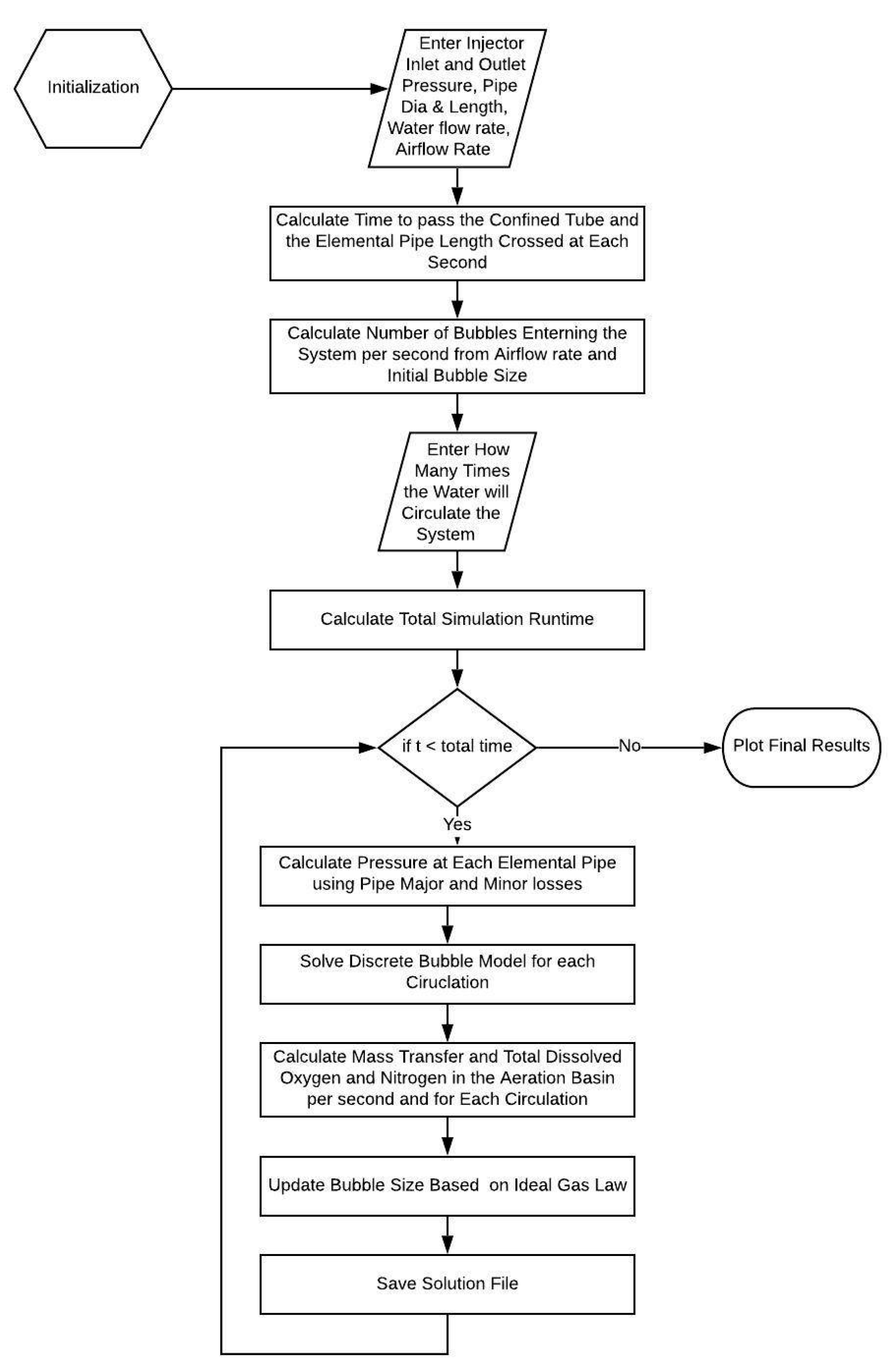
4.3. Discrete Bubble Model (Analytical Technique)
- The model is based on the Euler-Euler (E-E) approach.
- This model is capable of solving homogeneous multiphase flow.
- This E-E model employs the volume averaged mass and momentum conservation equation to describe time dependent motion of both phases.
- The bubble number contained in a computational cell is represented by a volume fraction.
- The bubble size information is obtained by incorporating population balance equations with break-up and coalesce of bubbles as well as growth or shrinkage of bubbles due to mass transfer across the bubble surface.
5. Discussion
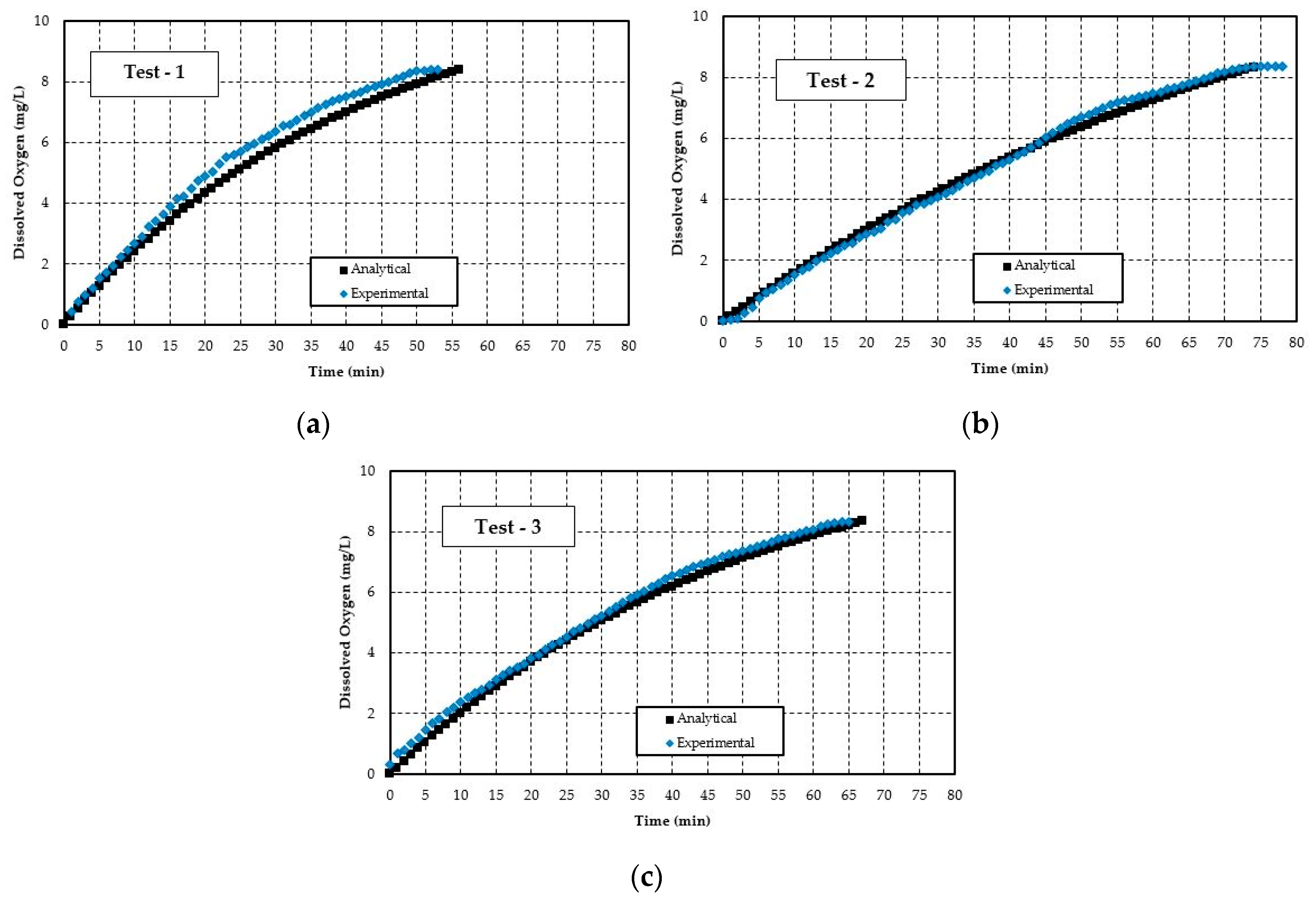
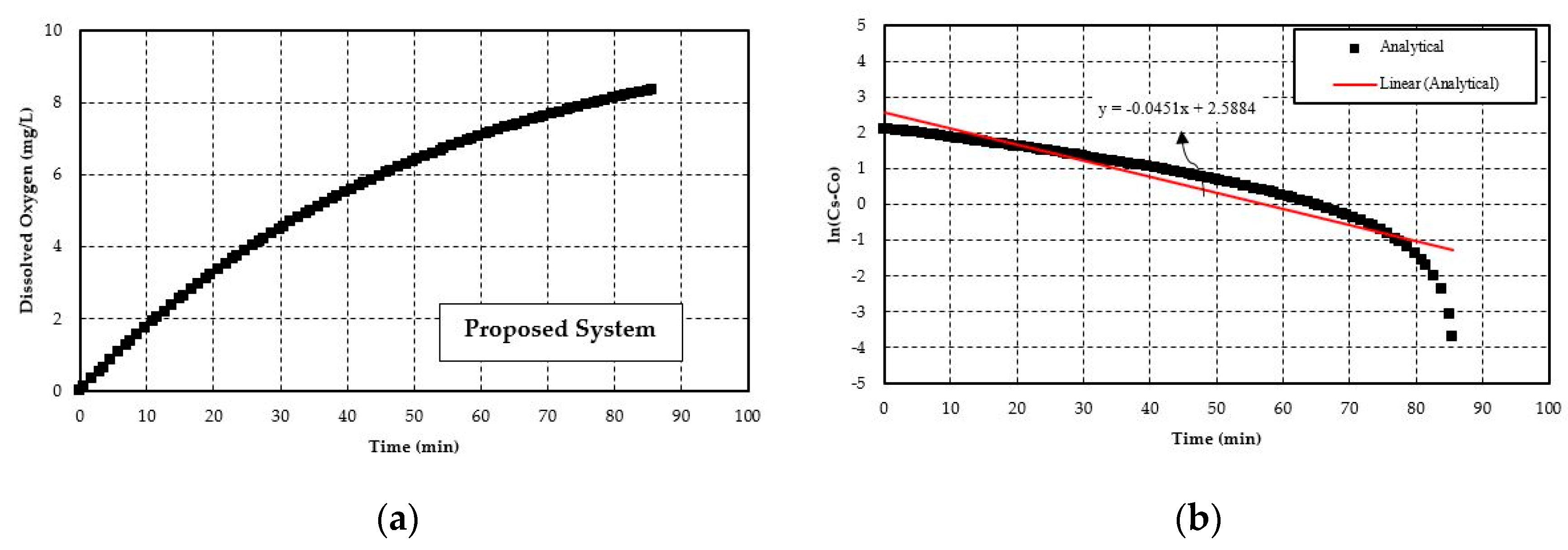
5.1. Validation of the Numerical Model
5.2. Parametric Study
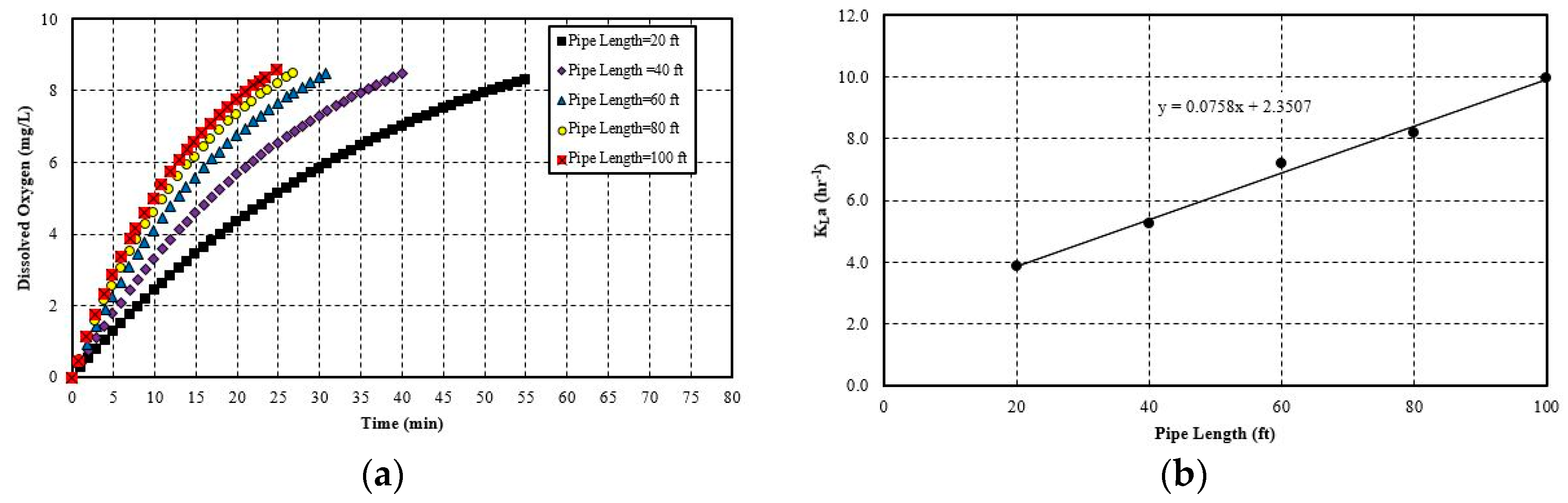
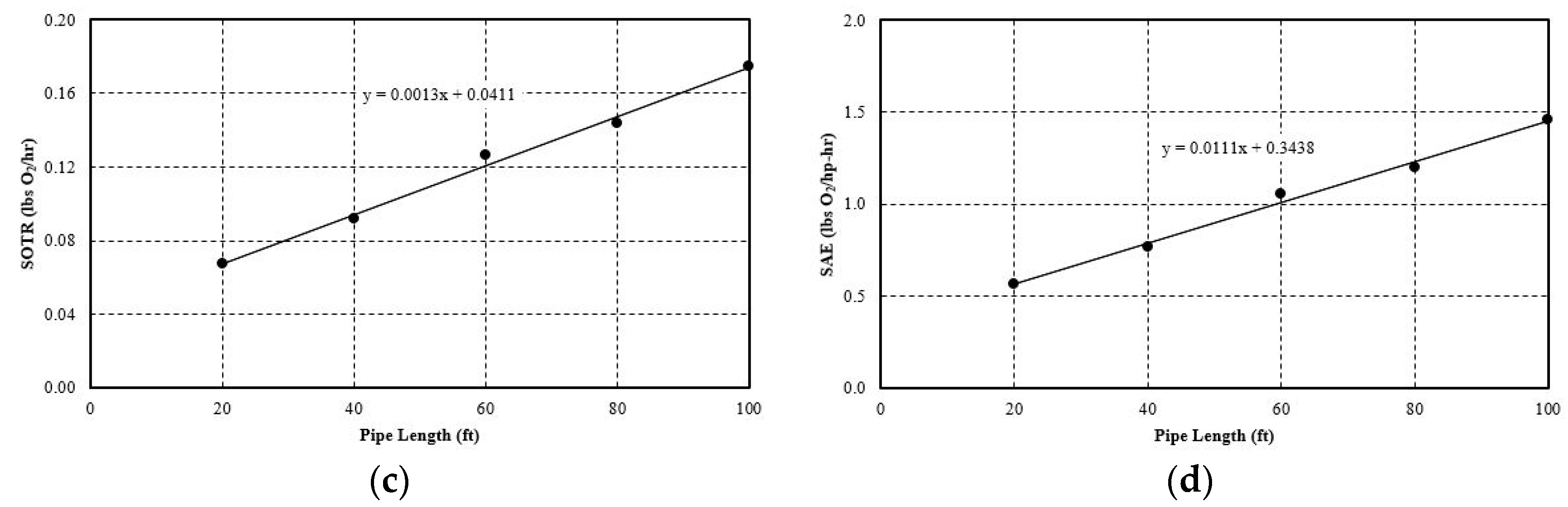
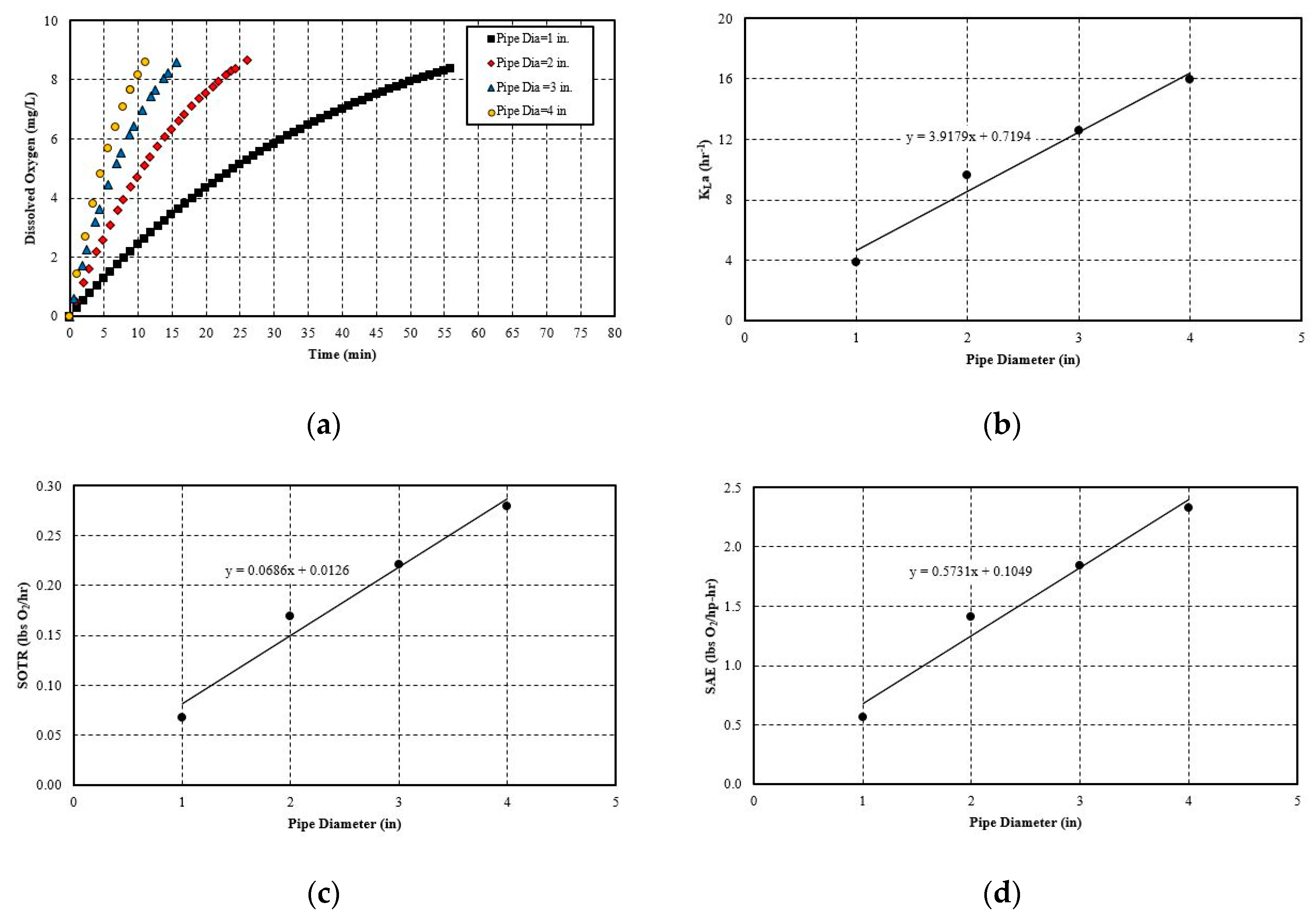
5.2.1. Effect of Pipe Length
5.2.2. Effect of Pipe Diameter
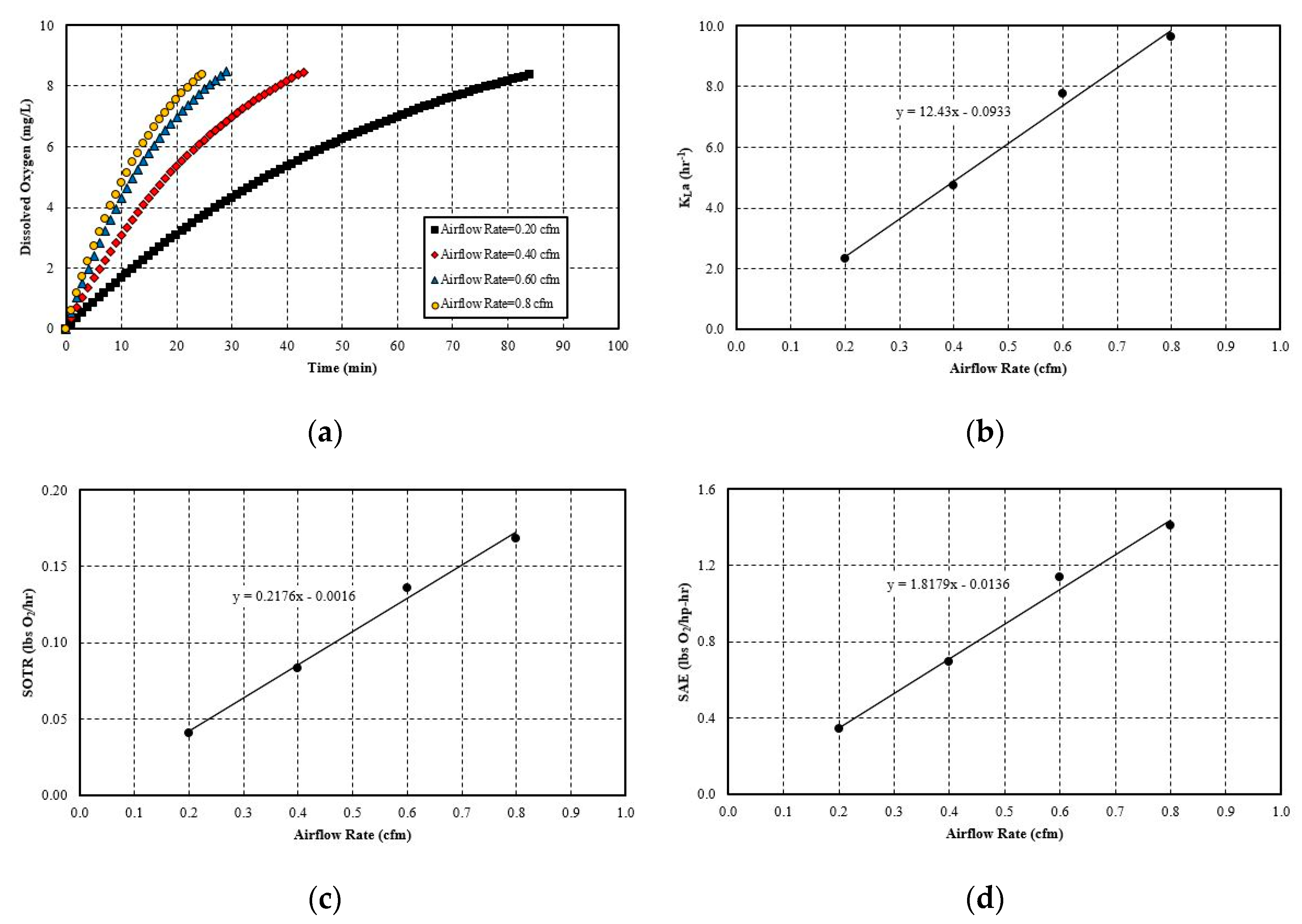
5.2.3. Effect of Gas Flow Rate
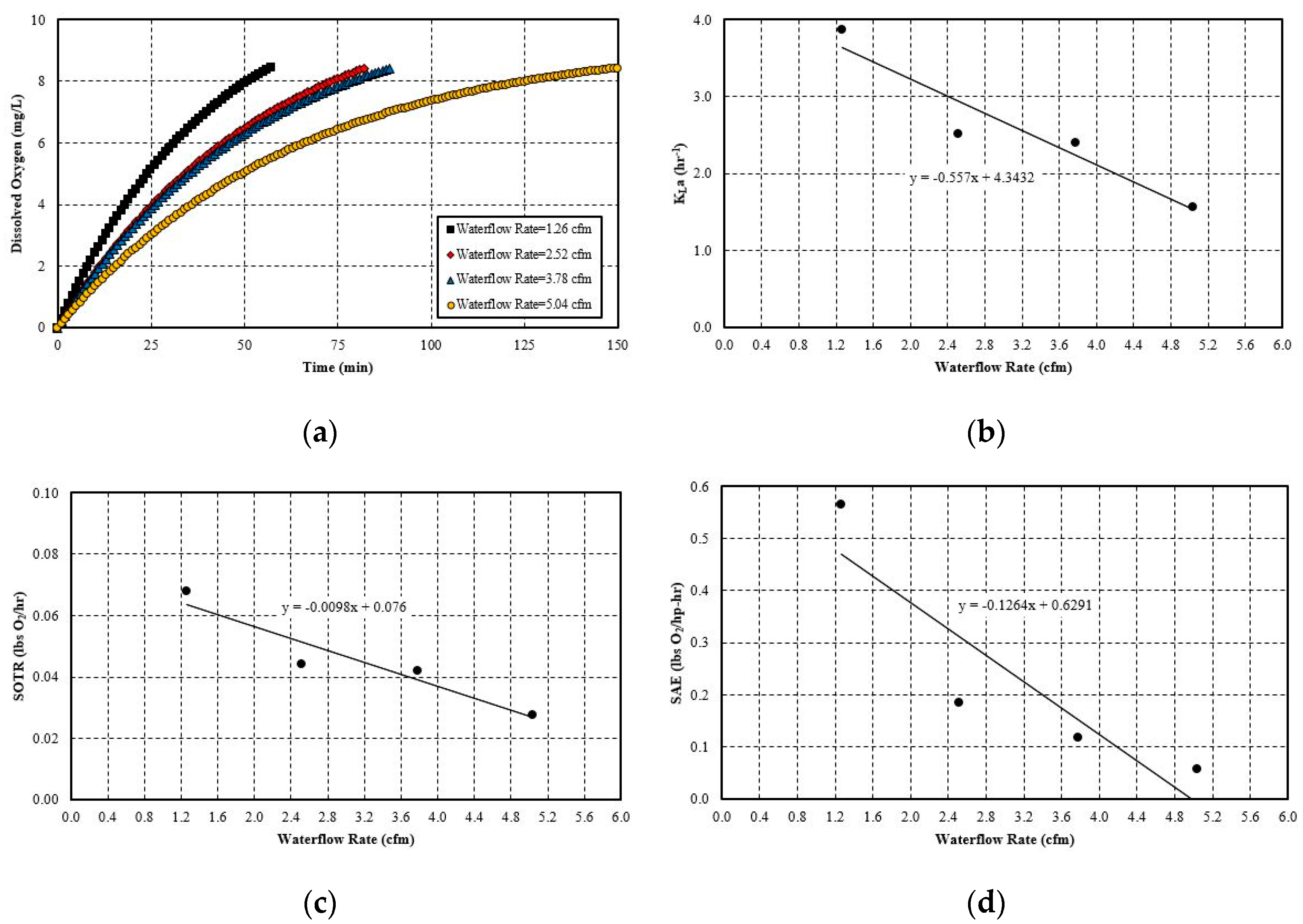
5.2.4. Effect of Liquid Flow Rate
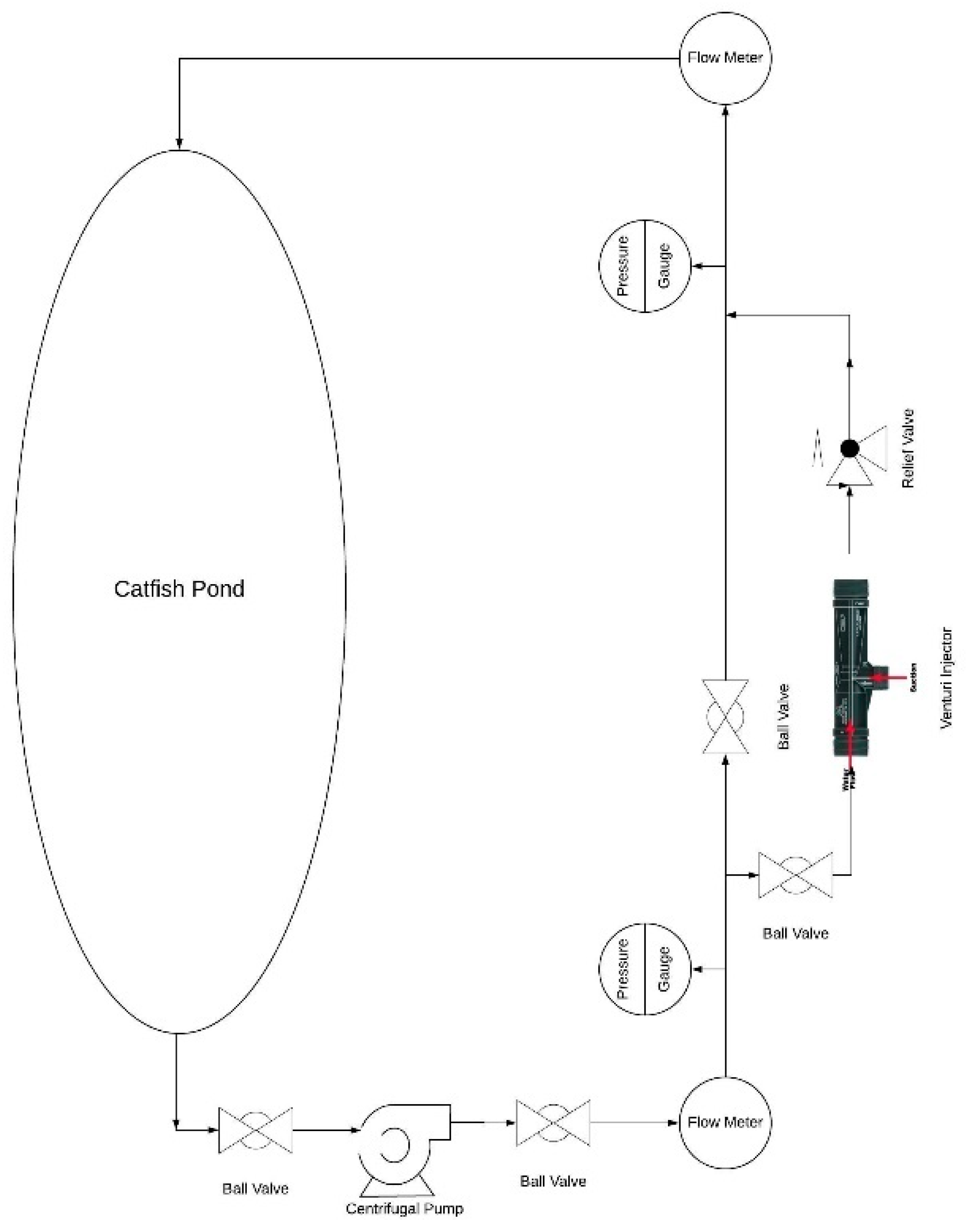
5.3. Proposed System
- The proposed system will be a closed loop system where a pump will be set up on the ground. The water intake of the pump will be at the bottom of the catfish pond.
- The water will reach the main discharge line located on the ground. There will be a pressure gauge to measure the pressure of the upcoming fluid before it enters into the injector.
- A pressure reducing valve will be attached to the proposed system in the discharge side of the injector to maintain the desired pressure differential. Therefore, maximum air suction can be achieved.
- The coiled pipe will start on the discharge side of injector and will hang in from the ground. The pressurized water will be evacuated from the pond on top surface.
- Finally, the water from the discharge side will be ejected into the catfish pond with high velocity. A schematic of the proposed system is shown in Figure 12.
6. Conclusions
- The experimental study of the current closed loop venturi aspirated aeration system achieved an SOTR in the range of 0.050–0.068 lbs O2/h and an SAE in the range of 0.42–0.63 lbs O2/hp-h. With the analytical study, SOTR is found to be similar for this system and is 0.047–0.059 lbs O2/h and SAE in the range of 0.37–0.55 lbs O2/hp-h. In all cases, the difference between experimental and analytical results are within 15%. The analytical model underestimates the experimental values, likely because it cannot account for turbulent mixing, bubble breakup, and bubble coalescing.
- The hydraulic parameters considered herein are the pipe diameter and pipe length. Both significantly impact the system’s aeration efficiency. Increasing the pipe length by a factor of five increases the SAE by 2.5 times. For pipe diameter increase, the effect is more evident, since increasing the diameter by 4 times increases the SAE by 4.1 times. The linear trend line is obtained with a simple mathematical relationship between SAE against the pipe length and diameter. The probable cause for this positive influence is that pipe length increases the bubble contact time with water. Moreover, the bubbles do not migrate towards the pipe wall and try to follow the centerline by avoiding coalesce and breakup. The diameter increase has a similar impact since it reduces the water flow rate and also reduces the frictional head loss. Thereby, it increases the system’s efficiency. The flow parameters that are considered here are: gas flow rate and water flow rate. Gas flow rate seems to play an important role in the system’s efficiency. It is observed that increasing the gas flow rate by 75% increases the SAE by almost 76%. The trend line of SAE versus gas flow rate also shows a linear relationship. The gas flow rate increases the amount of bubbles or gas volume fractions in the continuous medium of water flow at any instant. This results in a higher interfacial area for mass transfer. However, liquid flow rate increase impacts the system in a negative way. It does not give enough time for bubble contact and also reduces the gas volume fraction in the continuous media. There is no observable trend line found between SAE versus liquid flow rate.
- A system is proposed to optimize SAE based on the limited experimental information in this study. This proposed system is intended for aquaculture (e.g., catfish farm). The proposed system requires a 5-hp pump with 250 feet of 4-inch diameter coiled pipe. A 4-inch diameter venturi injector is chosen which can aspirate 7 scfm of air at 10 psi pressure differential with a water flow rate of 230 gpm. The system has an SOTR of 4.38 lbs O2/h and an SAE of 1.8.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alp, E.; Melching, C.S. Allocation of supplementary aeration stations in the Chicago waterway system for dissolved oxygen improvement. J. Environ. Manag. 2011, 92, 1577–1583. [Google Scholar] [CrossRef] [PubMed]
- Buscaglia, G.C.; Bombardelli, F.A.; García, M.H. Numerical modeling of large-scale bubble plumes accounting for mass transfer effects. Int. J. Multiph. Flow. 2003, 28, 1763–1785. [Google Scholar] [CrossRef]
- Zhang, Z.; Zeng, Y.; Kusiak, A. Minimizing pump energy in a wastewater processing plant. Energy 2012, 47, 505–514. [Google Scholar] [CrossRef]
- Brandt, M.; Middleton, R.; Wheale, G.; Schulting, F. Energy efficiency in the water industry, a Global Research Project. Water Pract. Technol. 2011, 6. [Google Scholar] [CrossRef]
- Oliver, J.D.; Somerset Environmental Solutions Inc. Apparatus and Method for Aerating Wastewater. U.S. Patent Application 16/090,676.9, 2019. [Google Scholar]
- Rosso, D.; Larson, L.E.; Stenstrom, M.K. Aeration of large-scale municipal wastewater treatment plants: State of the art. Water Sci. Technol. 2008, 57, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.X. Analysis on the effect of venturi tube structural parameters on fluid flow. AIP Adv. 2017, 7. [Google Scholar] [CrossRef]
- Simpson, A.; Ranade, V.V. Modeling hydrodynamic cavitation in venturi: Influence of venturi configuration on inception and extent of cavitation. AIChE J. 2019, 65, 421–433. [Google Scholar] [CrossRef]
- Zahradník, J.; Fialová, M.; Linek, V.; Sinkule, J.; Řezníčková, J.; Kaštánek, F. Dispersion efficiency of ejector-type gas distributors in different operating modes. Chem. Eng. Sci. 1997, 52, 4499–4510. [Google Scholar] [CrossRef]
- Terasaka, K.; Hirabayashi, A.; Nishino, T.; Fujioka, S.; Kobayashi, D. Development of microbubble aerator for waste water treatment using aerobic activated sludge. Chem. Eng. Sci. 2011, 66, 3172–3179. [Google Scholar] [CrossRef]
- Basso, A.; Hamad, F.A.; Ganesan, P. Effects of the geometrical configuration of air–water mixer on the size and distribution of microbubbles in aeration systems. Asia-Pacific J. Chem. Eng. 2018, 13, e2259. [Google Scholar] [CrossRef]
- Akhtar, M.S.; Rajesh, M.; Ciji, A.; Sharma, P.; Kamalam, B.S.; Patiyal, R.S.; Singh, A.K.; Sarma, D. Photo-thermal manipulations induce captive maturation and spawning in endangered golden mahseer (Tor putitora): A silver-lining in the strangled conservation efforts of decades. Aquaculture 2018, 497, 336–347. [Google Scholar] [CrossRef]
- Bagatur, T. Evaluation of Plant Growth with Aerated Irrigation Water Using Venturi Pipe Part. Arab. J. Sci. Eng. 2014, 39, 2525–2533. [Google Scholar] [CrossRef]
- Dahrazma, B.; Naghedinia, A.; Ghasemian Gorji, H.; Saghravani, S.F. Morphological and physiological responses of Cucumis sativus L. to water with micro-nanobubbles. J. Agric. Sci. Technol. 2019, 21, 181–192. [Google Scholar]
- Mitra, S.; Daltrophe, N.C.; Gilron, J. A novel eductor-based MBR for the treatment of domestic wastewater. Water Res. 2016, 100, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Kaya, Y.; Bacaksiz, A.M.; Bayrak, H.; Gönder, Z.B.; Vergili, I.; Hasar, H.; Yilmaz, G. Treatment of chemical synthesis-based pharmaceutical wastewater in an ozonation-anaerobic membrane bioreactor (AnMBR) system. Chem. Eng. J. 2017, 322, 293–301. [Google Scholar] [CrossRef]
- Unsal, M.; Baylar, A.; Tugal, M.; Ozkan, F. Increased aeration efficiency of high-head conduit flow systems. J. Hydraul. Res. 2008, 46, 711–714. [Google Scholar] [CrossRef]
- Bunea, F.; Ciocan, G.D. Experimental Study of Standard Aeration Efficiency in a Bubble Column. In Desalination and Water Treatment; IntechOpen: London, UK, 2018. [Google Scholar]
- Baylar, A.; Ozkan, F. Applications of venturi principle to water aeration systems. Environ. Fluid Mech. 2006, 6, 341–357. [Google Scholar] [CrossRef]
- Baylar, A.; Ozkan, F.; Unsal, M. On the Use of Venturi Tubes in Aeration. CLEAN Soil Air Water 2007, 35, 183–185. [Google Scholar] [CrossRef]
- Baylar, A.; Aydin, M.; Unsal, M.; Ozkan, F. Numerical Modeling of Venturi Flows for Determining Air Injection Rates Using Fluent V6.2. Math. Comput. Appl. 2009, 14, 97–108. [Google Scholar] [CrossRef]
- Water Resources Institute (US). Measurement of Oxygen Transfer in Clean Water: ASCE Standard, ASCE/EWRI 2-06; American Society of Civil Engineers: Reston, WV, USA, 2007. [Google Scholar]
- Mantha, R.; Taylor, K.E.; Biswas, N.; Bewtra, J.K. A continuous system for Fe0 reduction of nitrobenzene in synthetic wastewater. Environ. Sci. Technol. 2001, 35, 3231–3236. [Google Scholar] [CrossRef]
- Yin, Z.G.; David, Z.Z.; Liang, B.C.; Wang, L. Theoretical analysis and experimental study of oxygen transfer under regular and non-breaking waves. J. Hydrodyn. 2013, 25, 718–724. [Google Scholar] [CrossRef]
- Mazzei Injector Company, LLC. Available online: https://mazzei.net/ (accessed on 14 April 2020).
- Eckenfelder, W.W.; Ford, D.L. New concepts in oxygen transfer and aeration. In Advances in Water Quality Improvement; Texas Press: Austin, TX, USA, 1968; pp. 215–236. [Google Scholar]
- Barrut, B.; Blancheton, J.P.; Champagne, J.Y.; Grasmick, A. Mass transfer efficiency of a vacuum airlift-Application to water recycling in aquaculture systems. Aquac. Eng. 2012, 46, 18–26. [Google Scholar] [CrossRef]
- Kumar, A.; Moulick, S.; Singh, B.K.; Mal, B.C. Design characteristics of pooled circular stepped cascade aeration system. Aquac. Eng. 2013, 56, 51–58. [Google Scholar] [CrossRef]
- Kumar, A.; Moulick, S.; Mal, B.C. Selection of aerators for intensive aquacultural pond. Aquac. Eng. 2013, 56, 71–78. [Google Scholar] [CrossRef]
- Mueller, J.A.; Boyle, W.C.; Pöpel, H.J. Aeration: Principles and Practice. Popel-Google Books. Available online: https://books.google.com/books?hl=en&lr=&id=t4W9BwAAQBAJ&oi=fnd&pg=PP1&dq=Aeration:+principles+and+practice+%5BM%5D,+vol+9.+CRC+Press&ots=Gh0HKOBfmm&sig=jZ8nJquSAEHwxavK5IKqGR793-U#v=onepage&q=Aeration%3Aprinciplesandpractice%5BM%5D%2Cvol9.CRCPress&f=false (accessed on 12 April 2020).
- Wüest, A.; Brooks, N.H.; Imboden, D.M. Bubble plume modeling for lake restoration. Water Resour. Res. 1992, 28, 3235–3250. [Google Scholar] [CrossRef]
- Delnoij, E.; Lammers, F.A.; Kuipers, J.A.M.; Van Swaaij, W.P.M. Dynamic simulation of dispersed gas-liquid two-phase flow using a discrete bubble model. Chem. Eng. Sci. 1997, 52, 1429–1458. [Google Scholar] [CrossRef]
- Sokolichin, A.; Eigenberger, G.; Lapin, A.; Lübbert, A. Dynamic numerical simulation of gas-liquid two-phase flows: Euler/Euler versus Euler/Lagrange. Chem. Eng. Sci. 1997, 52, 611–626. [Google Scholar] [CrossRef]
- Darmana, D.; Deen, N.G.; Kuipers, J.A.M. Detailed modeling of hydrodynamics, mass transfer and chemical reactions in a bubble column using a discrete bubble model. Chem. Eng. Sci. 2005, 60, 3383–3404. [Google Scholar] [CrossRef]
- Van Den Hengel, E.I.V.; Deen, N.G.; Kuipers, J.A.M. Application of coalescence and breakup models in a discrete bubble model for bubble columns. Ind. Eng. Chem. Res. 2005, 44, 5233–5245. [Google Scholar] [CrossRef]
- Ellis, C.; Karn, A.; Hong, J.; Lee, S.J.; Kawakami, E.; Scott, D.; Gulliver, J.; Arndt, R.E.A. Measurements in the wake of a ventilated hydrofoil: A step towards improved turbine aeration techniques. In Proceedings of the IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2014; Volume 22, p. 62009. [Google Scholar]
- Zhao, L.; Sun, L.; Mo, Z.; Tang, J.; Hu, L.; Bao, J. An investigation on bubble motion in liquid flowing through a rectangular Venturi channel. Exp. Therm. Fluid Sci. 2018, 97, 48–58. [Google Scholar] [CrossRef]
- Burris, V.L.; Little, J.C. Bubble dynamics and oxygen transfer in a hypolimnetic aerator. In Water Science and Technology; Elsevier Sci Ltd.: Amsterdam, The Netherlands, 1998; Volume 37, pp. 293–300. [Google Scholar]
- Burris, V.L.; McGinnis, D.F.; Little, J.C. Predicting oxygen transfer and water flow rate in airlift aerators. Water Res. 2002, 36, 4605–4615. [Google Scholar] [CrossRef]
- Little, J.C.; McGinnis, D.F. Hypolimnetic oxygenation: Predicting performance using a discrete-bubble model. In Water Science and Technology: Water Supply; IWA Publishing: London, UK, 2001; Volume 1, pp. 185–191. [Google Scholar]
- Popel, H.J.; Wagner, M. Prediction of oxygen transfer rates from simple measurements of bubble characteristics. In Water Science and Technology; IWA Publishing: London, UK, 1991; Volume 23, pp. 1941–1950. [Google Scholar]
- Biń, A.K. Application of A Single-Bubble Model in Estimation of Ozone Transfer Efficiency in Water. Ozone Sci. Eng. 1995, 17, 469–484. [Google Scholar] [CrossRef]
- Orsat, V.; Vigneault, C.; Raghavan, G.S.V. Air Diffusers Characterization using a Digitized Image Analysis System. Appl. Eng. Agric. 1993, 9, 115–121. [Google Scholar] [CrossRef]
- McGinnis, D.F.; Little, J.C. Predicting diffused-bubble oxygen transfer rate using the discrete-bubble model. Water Res. 2002, 36, 4627–4635. [Google Scholar] [CrossRef]
- Heat Transfer Handbook-Google Books. Available online: https://books.google.com/books?hl=en&lr=&id=d4cgNG_IUq8C&oi=fnd&pg=PR9&dq=Bejan,+Adrian%3B+Kraus,+Allan+D.+(2003).+Heat+Transfer+Handbook.+John+Wiley+%26+Son&ots=27DYj60qWH&sig=P02nCTSSS5a-3b8hlgPtqcQPLqM#v=onepage&q=Bejan%2CAdrian%3BKraus%2CAllan,D.(2003).HeatTransferHandbook.JohnWiley%26Son&f=false (accessed on 12 April 2020).
- Gordiychuk, A.; Svanera, M.; Benini, S.; Poesio, P. Size distribution and Sauter mean diameter of micro bubbles for a Venturi type bubble generator. Exp. Therm. Fluid Sci. 2016, 70, 51–60. [Google Scholar] [CrossRef]
- Saffari, H.; Moosavi, R.; Gholami, E.; Nouri, N.M. The effect of bubble on pressure drop reduction in helical coil. Exp. Therm. Fluid Sci. 2013, 51, 251–256. [Google Scholar] [CrossRef]
- Ekambara, K.; Sanders, R.S.; Nandakumar, K.; Masliyah, J.H. CFD Modeling of Gas-Liquid Bubbly Flow in Horizontal Pipes: Influence of Bubble Coalescence and Breakup. Int. J. Chem. Eng. 2012, 2012, 20. [Google Scholar] [CrossRef]
- Dobbins, W.E. Mechanism of Gas Absorption by Turbulent Liquids. In Advances in Water Pollution Research; Elsevier: Amsterdam, The Netherlands, 1964; pp. 61–96. [Google Scholar]
- Bewtra, J.; Federation, W.N.-J.; Water, P.C. Oxygenation from diffused air in aeration tanks. Journal 1964, 36, 1195–1224. [Google Scholar]
- Bewtra, J.K.; Nicholas, W.R.; Polkowski, L.B. Effect of temperature on oxygen transfer in water. Water Res. 1970, 4, 115–123. [Google Scholar] [CrossRef]
- Schmit, F.; Wren, J.; Control, D.R.-J.; Water, P. The effect of tank dimensions and diffuser placement on oxygen transfer. JSTOR 1978, 50, 1750–1767. [Google Scholar]
- Tchobanoglous, G.; Burton, F.L.; David Stensel, H. Wastewater Engineering. Management 1991, 7, 1–4. [Google Scholar]
- Zhu, J.; Miller, C.F.; Dong, C.; Wu, X.; Wang, L.; Mukhtar, S. Aerator Module Development Using Venturi Air Injectors to Improve Aeration Efficiency. Appl. Eng. Agric. 2007, 23, 661–667. [Google Scholar] [CrossRef]
- Dong, C.; Zhu, J.; Wu, X.; Miller, C.F. Aeration efficiency influenced by venturi aerator arrangement, liquid flow rate and depth of diffusing pipes. Environ. Technol. (UK) 2012, 33, 1289–1298. [Google Scholar] [CrossRef] [PubMed]
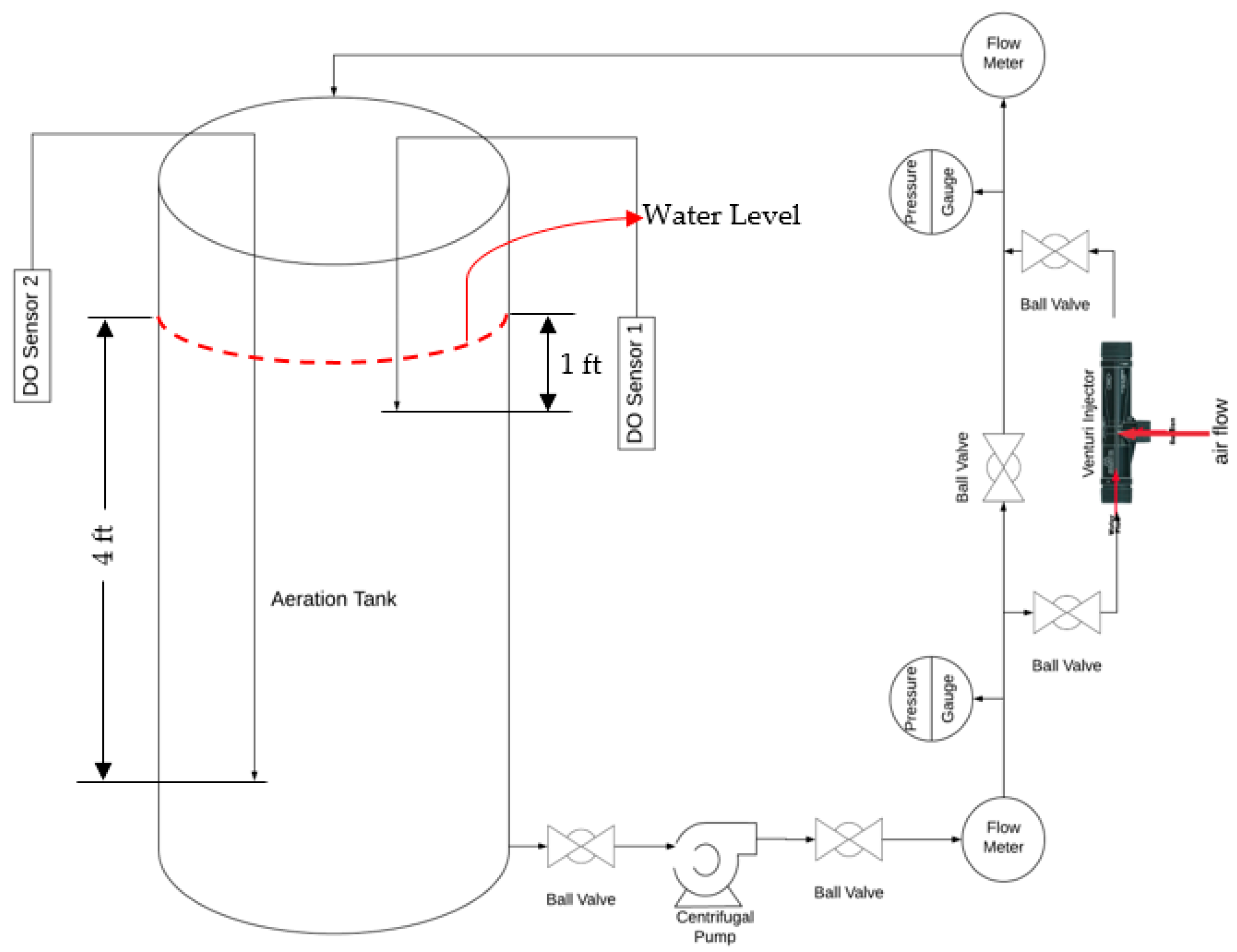

| Equipment | Quantity |
|---|---|
| ½ hp Centrifugal Pump | 1 |
| PEX Pipe (1 in. Dia) | 20 ft. |
| Mazzei [25] Venturi Injectors (1 in. Dia) | 1 |
| Digital Pressure Gauge | 2 |
| Digital Flow Meter | 2 |
| Aeration Tank Size (300 Gallons) | 1 |
| Ball Valve | 6 |
| Optical DO Probe | 2 |
| Temperature Sensor | 1 |
| Parameter | Dimensions | Experimental Value | ||
|---|---|---|---|---|
| Test-1 | Test-2 | Test-3 | ||
| Pipe length | ft | 20 | 20 | 20 |
| Pipe diameter | inch | 1 | 1 | 1 |
| Venturi injectors | – | 1 | 1 | 1 |
| Injector inlet pressure | psi | 17.8 | 17.8 | 14.05 |
| Injector outlet pressure | psi | 5.4 | 11.2 | 4.1 |
| Air flow rate | SCFM | 0.302 | 0.142 | 0.2183 |
| Water flow rate | CFM | 1.26 | 1.26 | 1.13 |
| Aerated water volume | Gallons | 250 | 250 | 250 |
| Temperature | °C | 25 | 25 | 25 |
| Reynolds Number (Water) | 33,493 | 33,493 | 30,037 | |
| Reynolds Number (Mixture) | 1,234,100 | 1,434,400 | 1,172,600 | |
| Tests | Experimental Value (EV) | Analytical Value (AV) | Percentage Difference | ||||||
|---|---|---|---|---|---|---|---|---|---|
| KLa (h−1) | SOTR (lbsO2/h) | SAE (lbsO2/hp-h) | KLa (h−1) | SOTR (lbsO2/h) | SAE (lbsO2/hp-h) | KLa (%) | SOTR (%) | SAE (%) | |
| Test-1 | 4.03 | 0.068 | 0.56 | 3.38 | 0.059 | 0.49 | 16 | 13 | 13 |
| Test-2 | 2.87 | 0.050 | 0.42 | 2.55 | 0.044 | 0.37 | 11 | 12 | 12 |
| Test-3 | 3.20 | 0.054 | 0.63 | 2.81 | 0.047 | 0.55 | 12 | 13 | 12 |
| Parameter | Values | Analytical Value | ||
|---|---|---|---|---|
| KLa (h−1) | SOTR (lbsO2/h) | SAE (lbsO2/hp-h) | ||
| Pipe Length | 20 ft. | 3.38 | 0.06 | 0.49 |
| 40 ft. | 5.24 | 0.09 | 0.77 | |
| 60 ft. | 7.21 | 0.13 | 1.05 | |
| 80 ft. | 8.20 | 0.14 | 1.20 | |
| 100 ft. | 9.97 | 0.17 | 1.46 | |
| Pipe Diameter | 1 in. | 3.38 | 0.06 | 0.49 |
| 2 in | 9.65 | 0.17 | 1.41 | |
| 3 in. | 12.60 | 0.22 | 1.84 | |
| 4 in. | 15.94 | 0.28 | 2.33 | |
| Airflow Rate | 0.2 cfm | 2.34 | 0.04 | 0.34 |
| 0.4 cfm | 4.75 | 0.08 | 0.69 | |
| 0.6 cfm | 7.77 | 0.14 | 1.14 | |
| 0.8 cfm | 9.62 | 0.17 | 1.41 | |
| Water flow Rate | 1.26 cfm | 3.87 | 0.07 | 0.57 |
| 2.52 cfm | 2.52 | 0.04 | 0.18 | |
| 3.78 cfm | 2.40 | 0.04 | 0.12 | |
| 5.04 cfm | 1.57 | 0.03 | 0.06 | |
| Parameter | Dimensions | Input Value |
|---|---|---|
| Pipe length | ft. | 250 |
| Pipe diameter | inch | 4 |
| Venturi injectors | – | 1 |
| Injector inlet pressure | psi | 15 |
| Injector outlet pressure | psi | 5 |
| Air flow rate | SCFM | 7 |
| Water flow rate | CFM | 31 |
| Aerated water volume | Gallons | 26,041 |
| Temperature | °C | 25 |
| Type | SAE (lbs O2/hp-h) |
|---|---|
| Closed Loop Venturi Aspirated Aeration | 1.8 |
| Low-Speed Surface Aerators | 2.5–3.4 |
| High-Speed Surface Aerators | 1.8–2.3 |
| Submersed Jet Aerators | 1.6–2.3 |
| Fine Bubble Diffusers, Disks | 3–10 |
| High Density Low Flux (HDLF) Fine Bubble Diffuser | 5–13 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahmud, R.; Erguvan, M.; MacPhee, D.W. Performance of Closed Loop Venturi Aspirated Aeration System: Experimental Study and Numerical Analysis with Discrete Bubble Model. Water 2020, 12, 1637. https://doi.org/10.3390/w12061637
Mahmud R, Erguvan M, MacPhee DW. Performance of Closed Loop Venturi Aspirated Aeration System: Experimental Study and Numerical Analysis with Discrete Bubble Model. Water. 2020; 12(6):1637. https://doi.org/10.3390/w12061637
Chicago/Turabian StyleMahmud, Roohany, Mustafa Erguvan, and David W. MacPhee. 2020. "Performance of Closed Loop Venturi Aspirated Aeration System: Experimental Study and Numerical Analysis with Discrete Bubble Model" Water 12, no. 6: 1637. https://doi.org/10.3390/w12061637
APA StyleMahmud, R., Erguvan, M., & MacPhee, D. W. (2020). Performance of Closed Loop Venturi Aspirated Aeration System: Experimental Study and Numerical Analysis with Discrete Bubble Model. Water, 12(6), 1637. https://doi.org/10.3390/w12061637





